Patents
Literature
Hiro is an intelligent assistant for R&D personnel, combined with Patent DNA, to facilitate innovative research.
5754results about "SsRNA viruses positive-sense" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Microparticles with adsorbent surfaces, methods of making same, and uses thereof
InactiveUS6884435B1Stimulate immune responseEasy to usePowder deliverySsRNA viruses positive-senseAntigenDisease
The present invention is directed to microparticles, to microparticle compositions containing the same, to methods of forming the same, and to uses for the same, including use for a vaccine, for raising an immune response, for treatment of a disease and for diagnosis of a disease. The microparticles comprise a biodegradable polymer, such as a poly(α-hydroxy acid), a polyhydroxy butyric acid, a polycaprolactone, a polyorthoester, a polyanhydride, or a polycyanoacrylate, and a detergent selected from a cationic detergent and an anionic detergent. The microparticles further comprise an antigen adsorbed on the surface of the microparticle.
Owner:NOVARTIS VACCINES & DIAGNOSTICS INC
Antigen-binding molecule capable of binding to two or more antigen molecules repeatedly
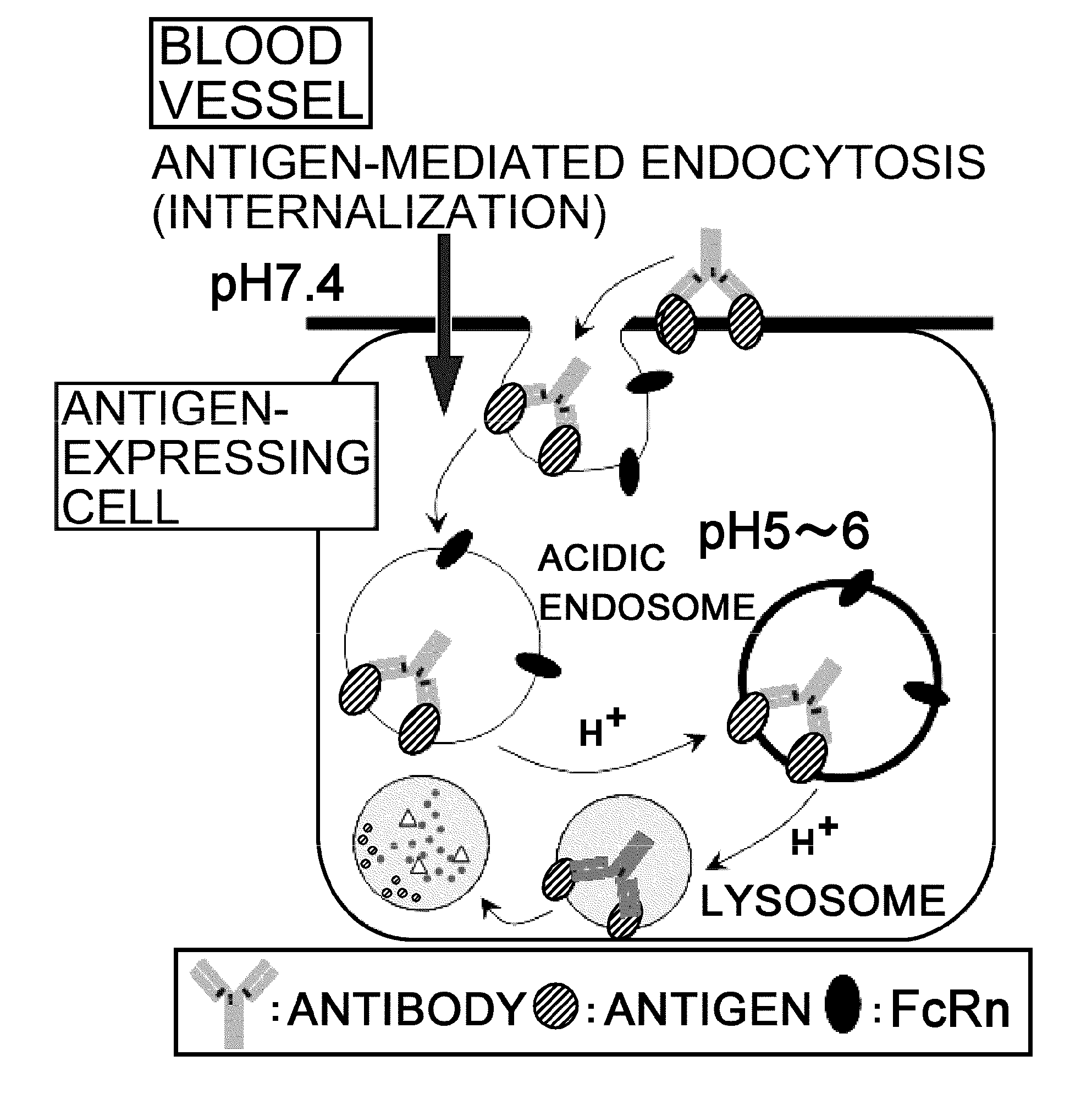
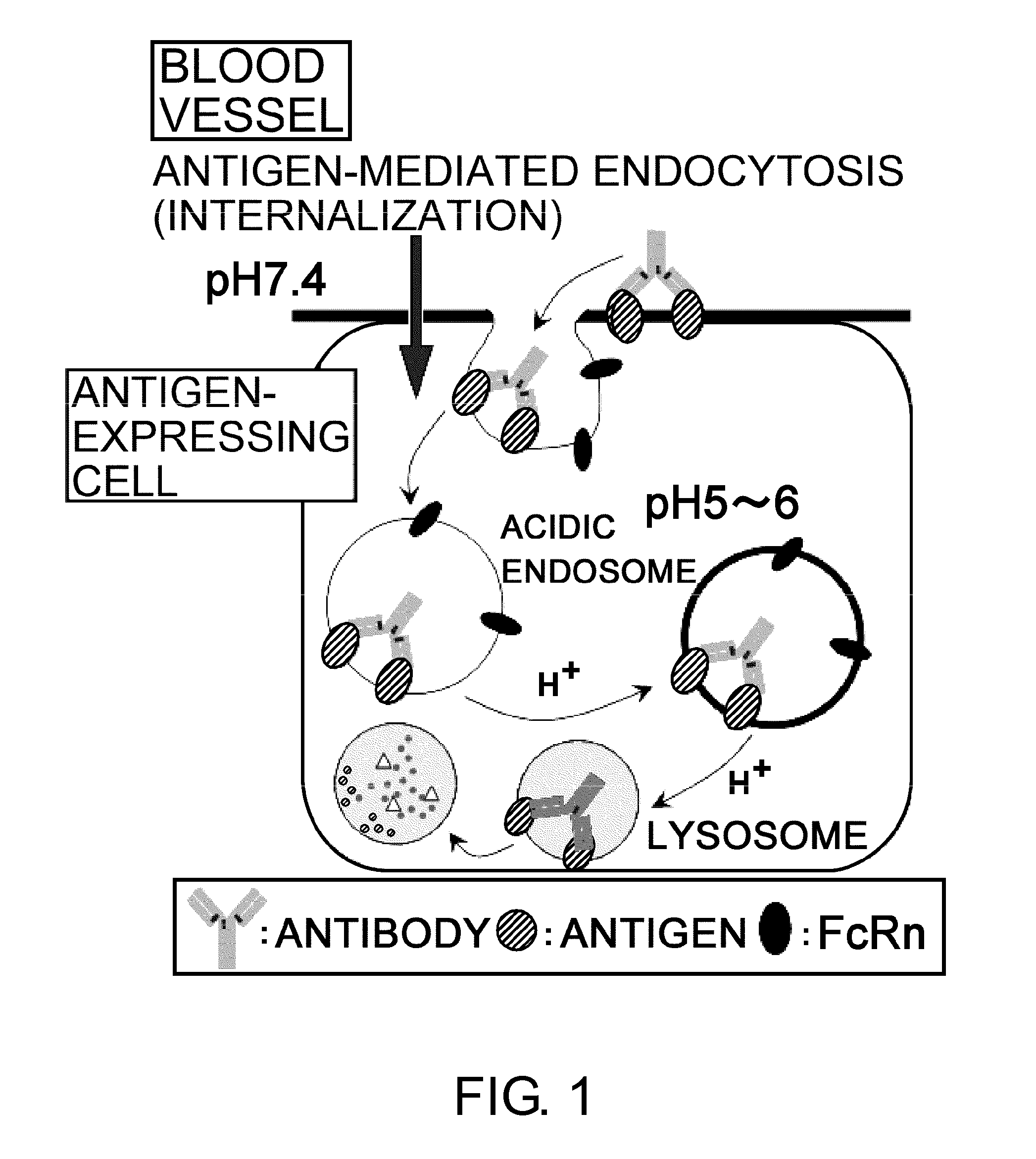
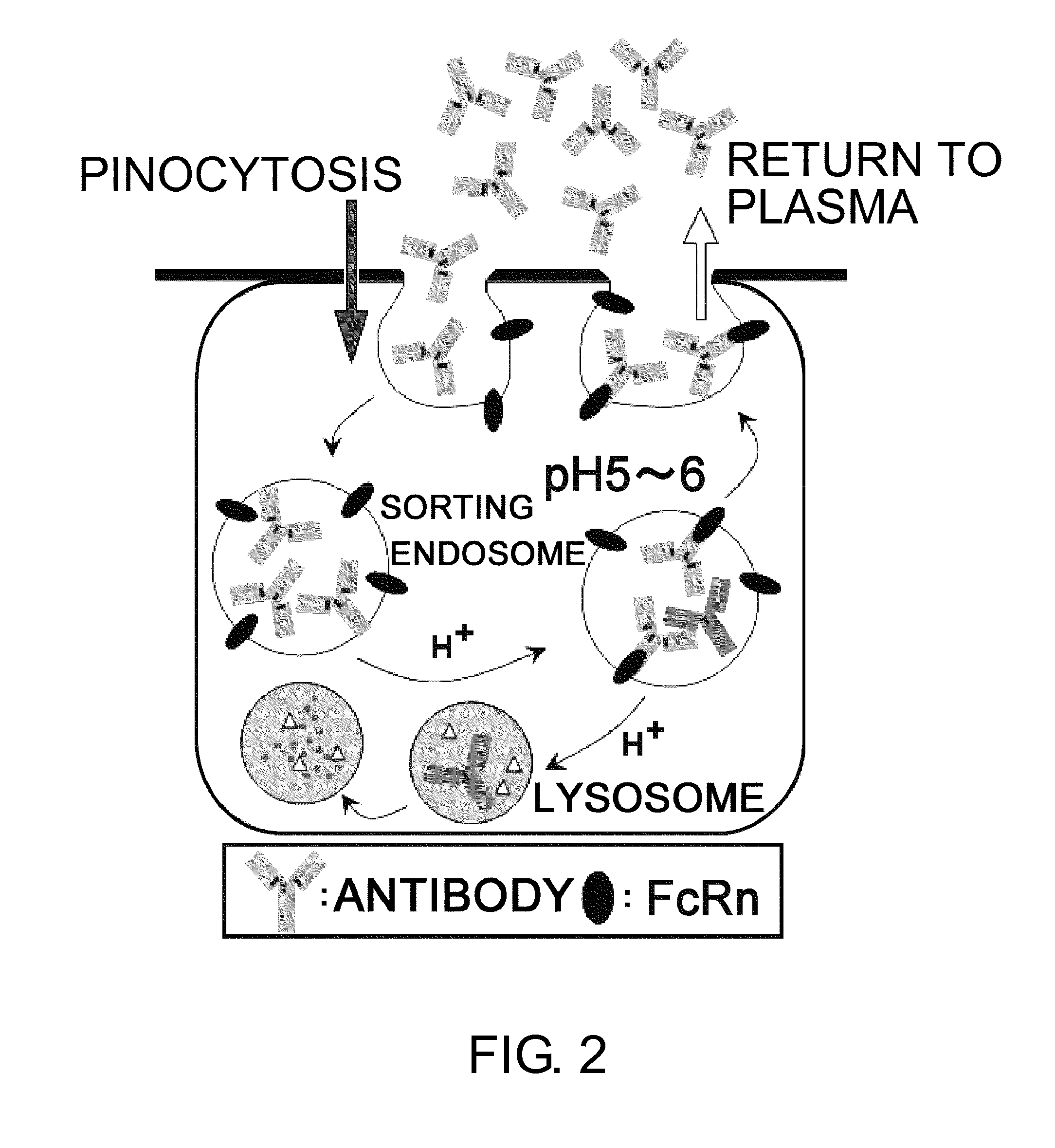
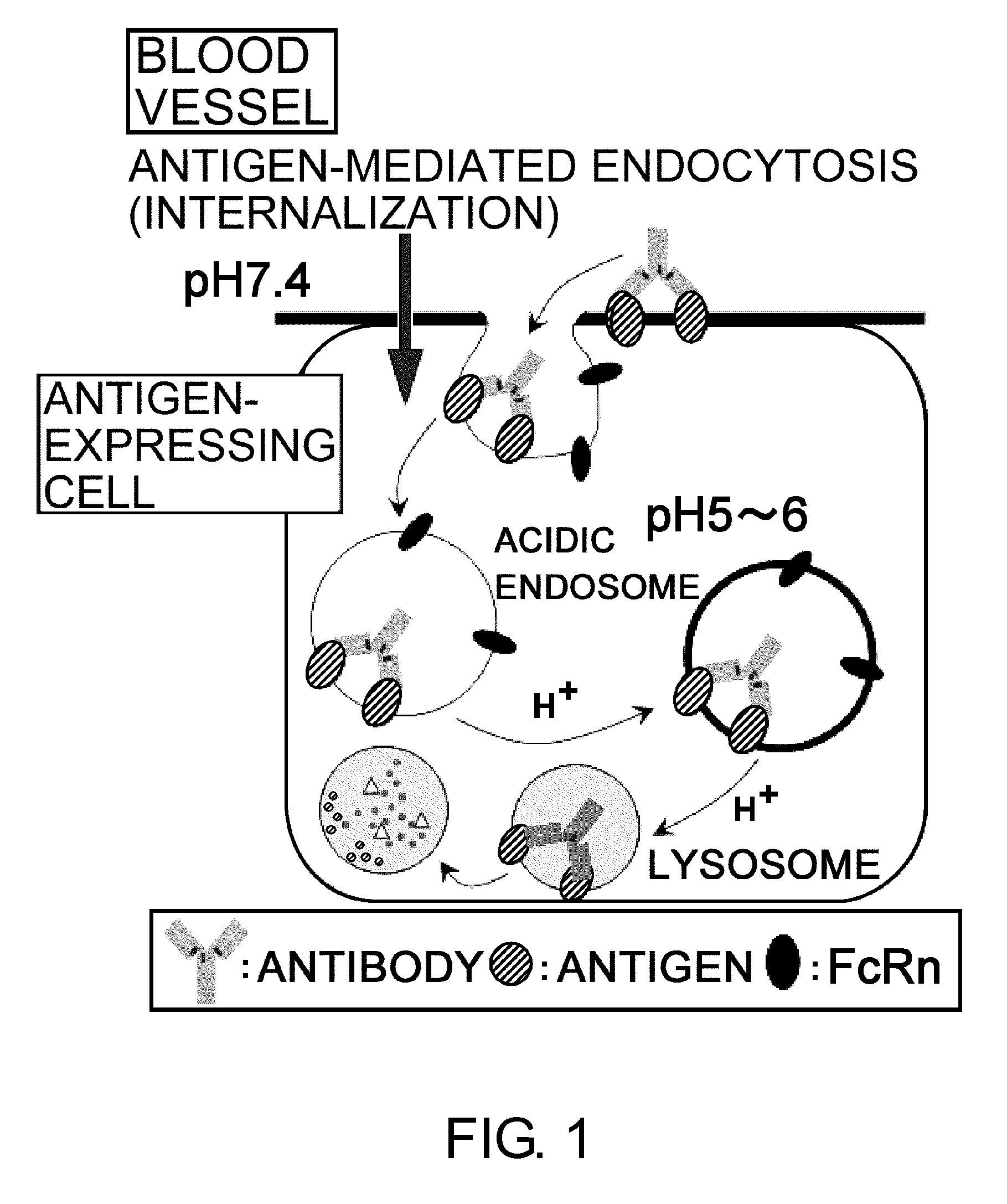
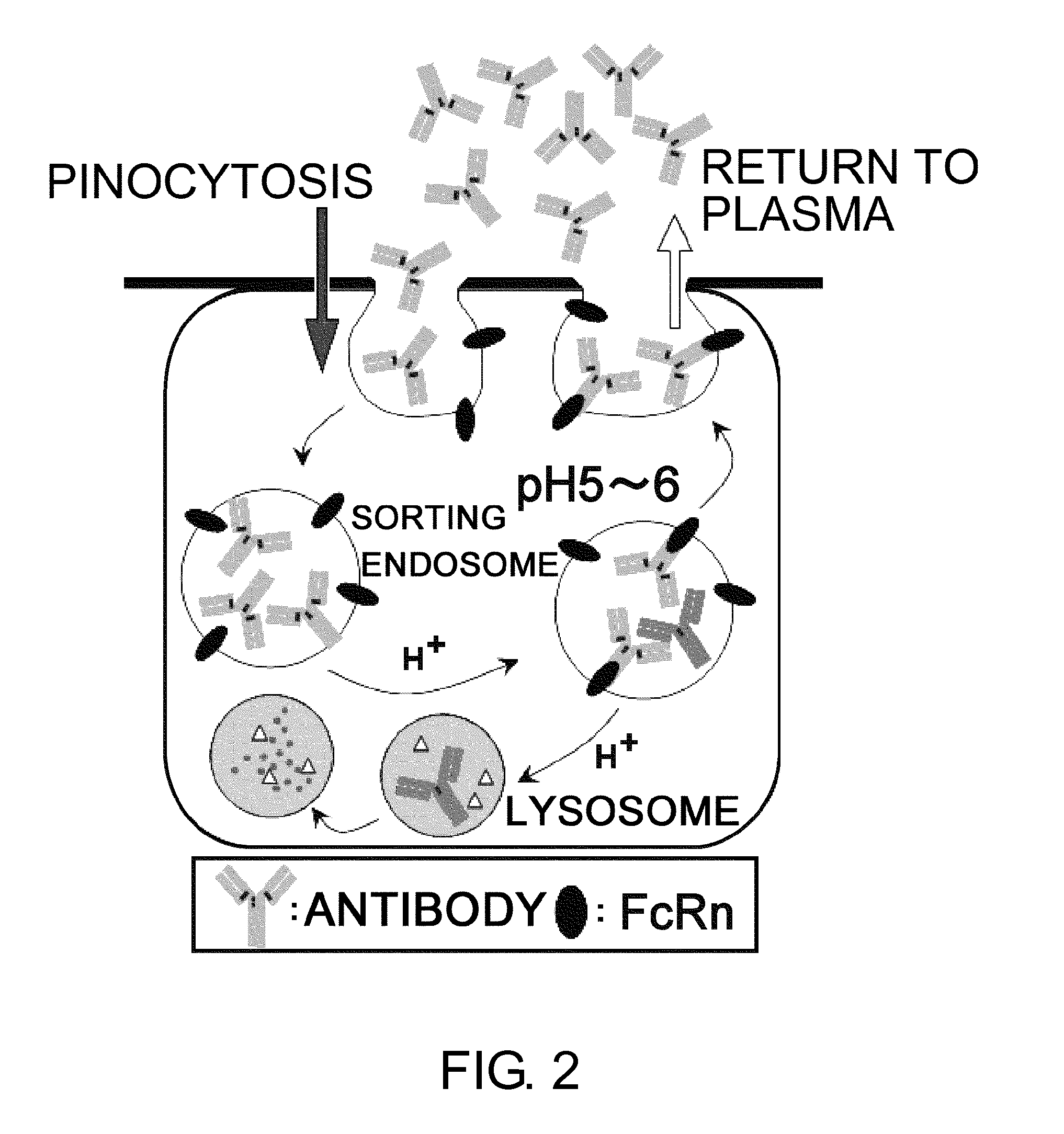
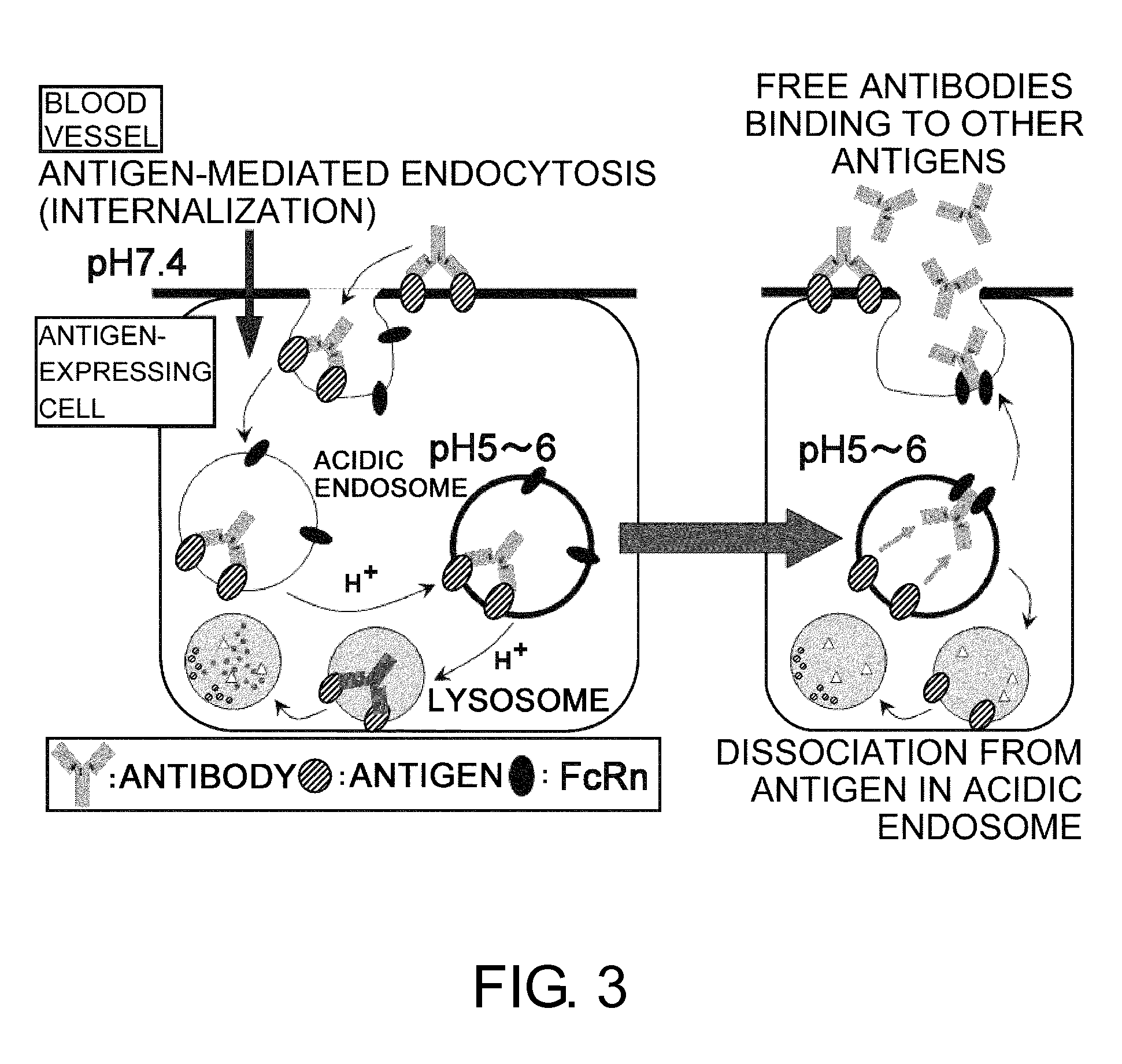
InactiveUS20110111406A1Pharmacokinetics of the antigen-binding molecule can be improvedGood effectSsRNA viruses negative-senseCompound screeningHalf-lifeAntigen binding
The present inventors discovered that antibodies having weaker antigen-binding activity at the early endosomal pH in comparison with that at the pH of plasma are capable of binding to multiple antigen molecules with a single antibody molecule, have long half-lives in plasma, and have improved durations of time in which they can bind to antigen.
Owner:CHUGAI PHARMA CO LTD
Expression of heterologous proteins
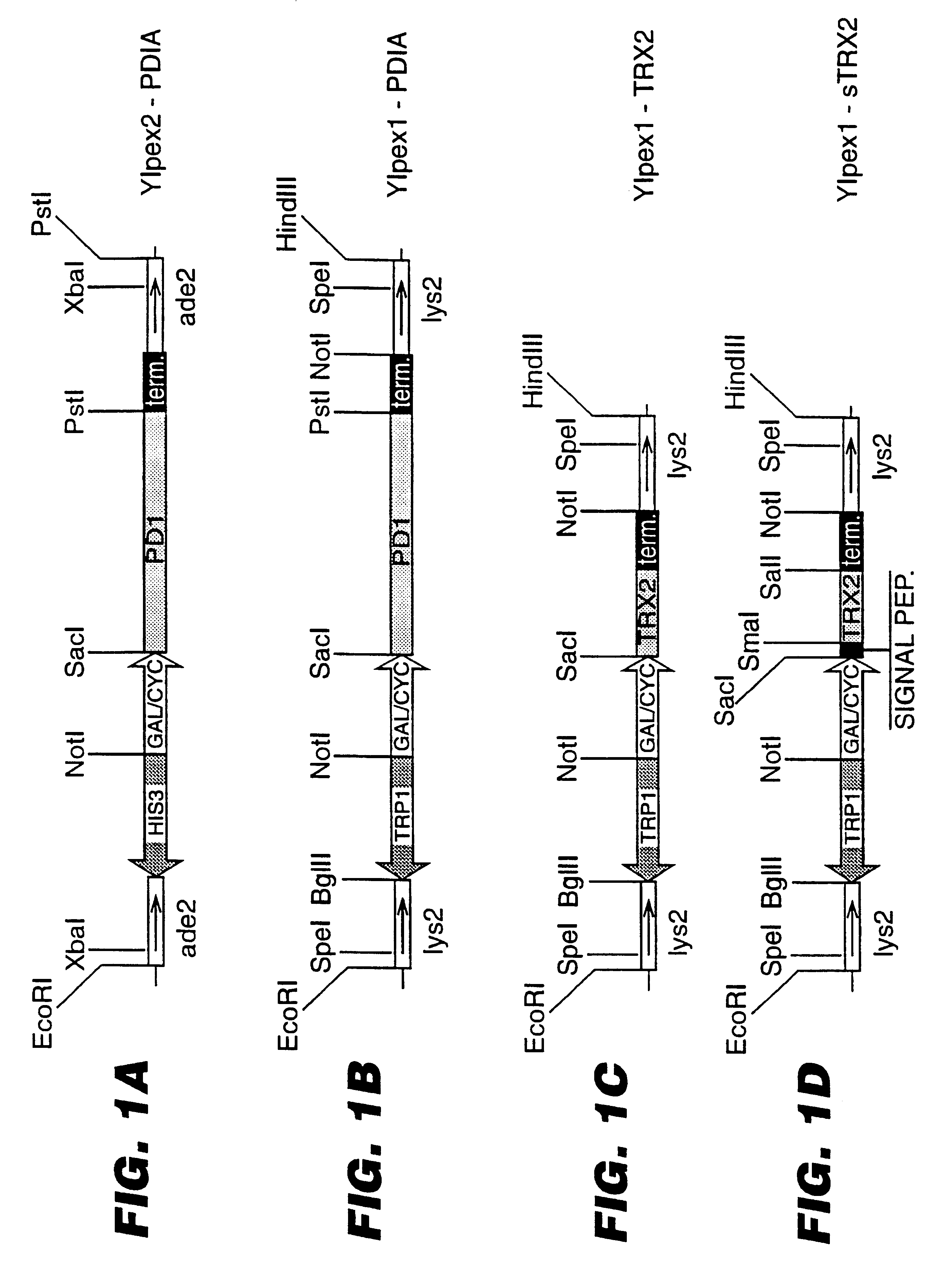
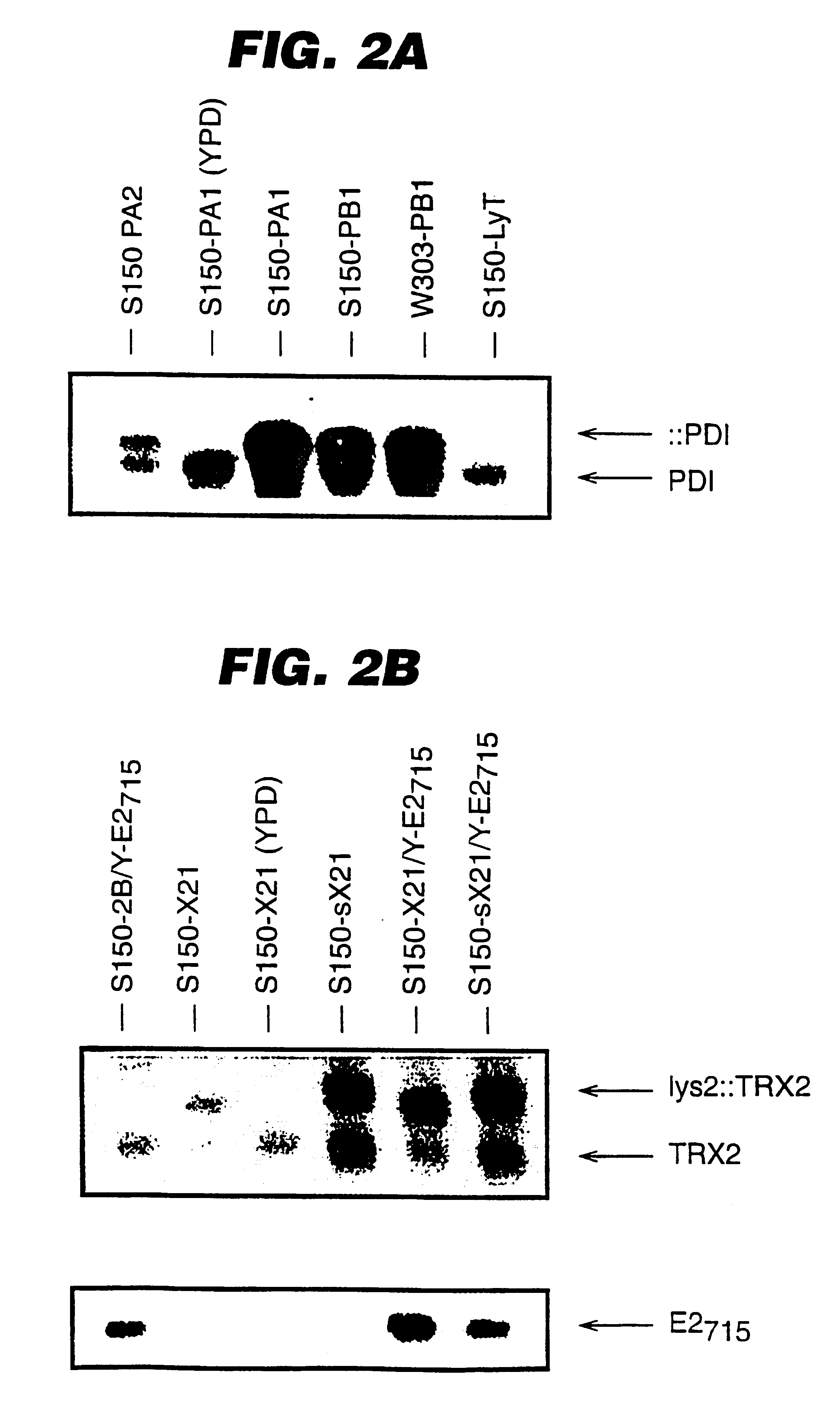
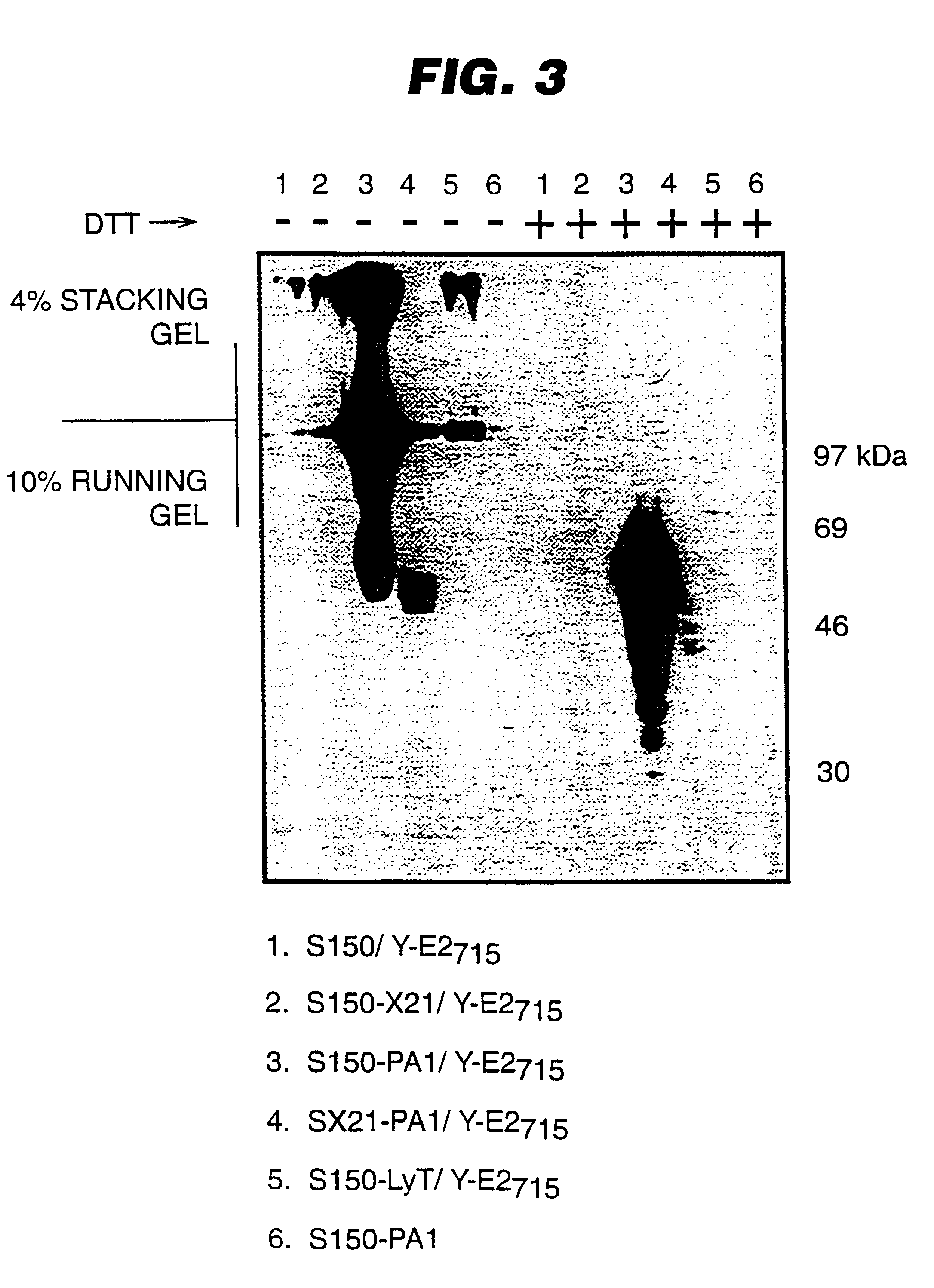
An expression system which provides heterologous proteins expressed by a non-native host organism but which have native-protein-like biological activity and / or structure. Disclosed are vectors, expression hosts and methods for expressing the heterologous proteins. The expression system involves co-expression of protein factor(s) which is / are capable of catalyzing disulphide bond formation and desired heterologous protein(s). The expression system is presented using yeast cells as the preferred host, protein disulphide isomerase (PDI) and thioredoxin (TRX) as the preferred examples of the protein factors and HCV-E2715 envelope glycoprotein and human FIGF as the preferred examples of the heterologous proteins.
Owner:NOVARTIS AG
Method for the preparation of purified HCV RNA by exosome separation
InactiveUS7198923B1Simple preparation processPromote progressSsRNA viruses positive-senseMicrobiological testing/measurementExosomeBlood plasma
Owner:GRIFOLS WORLDWIDE OPERATIONS +1
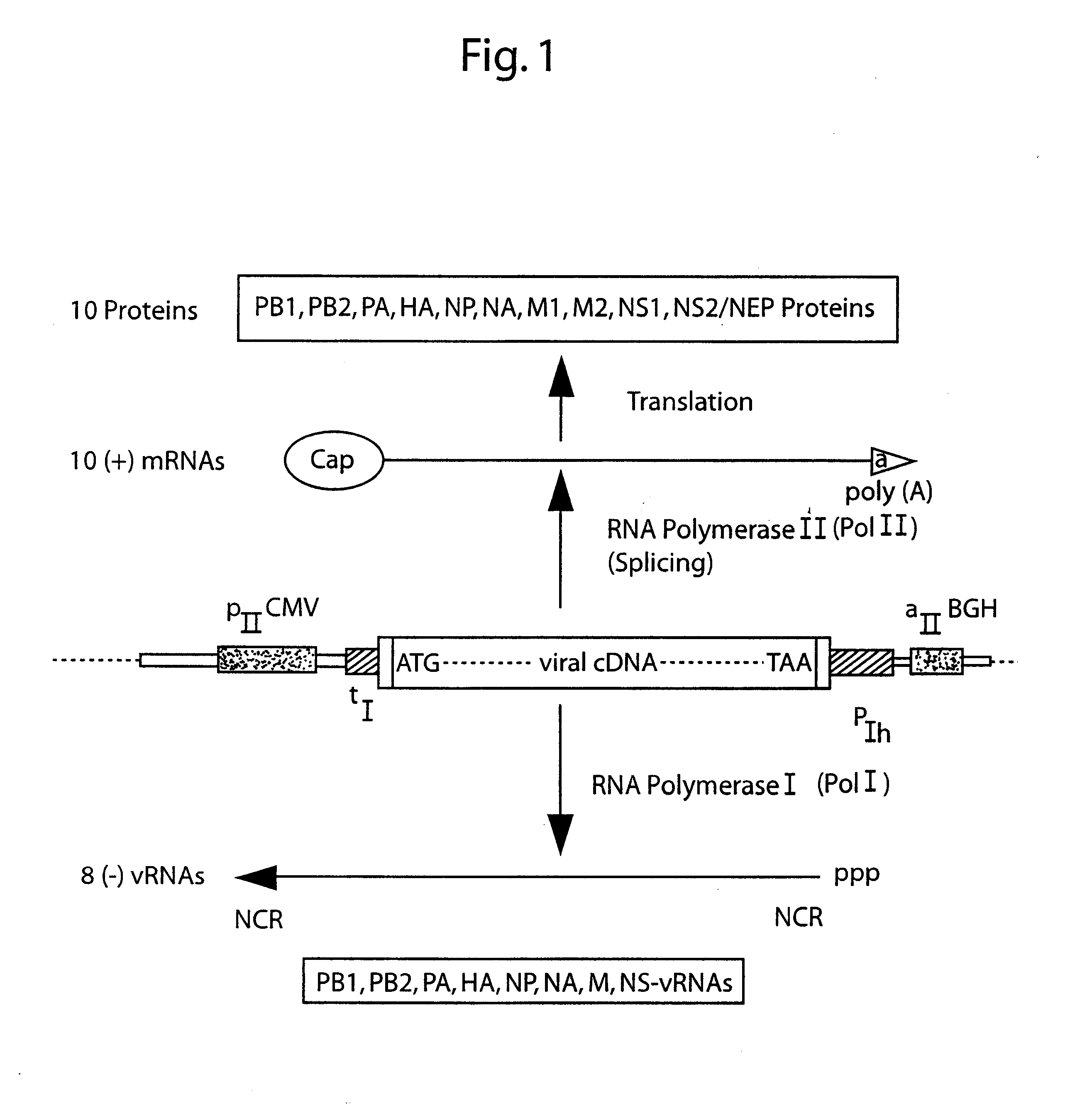
DNA transfection system for the generation of infectious influenza virus
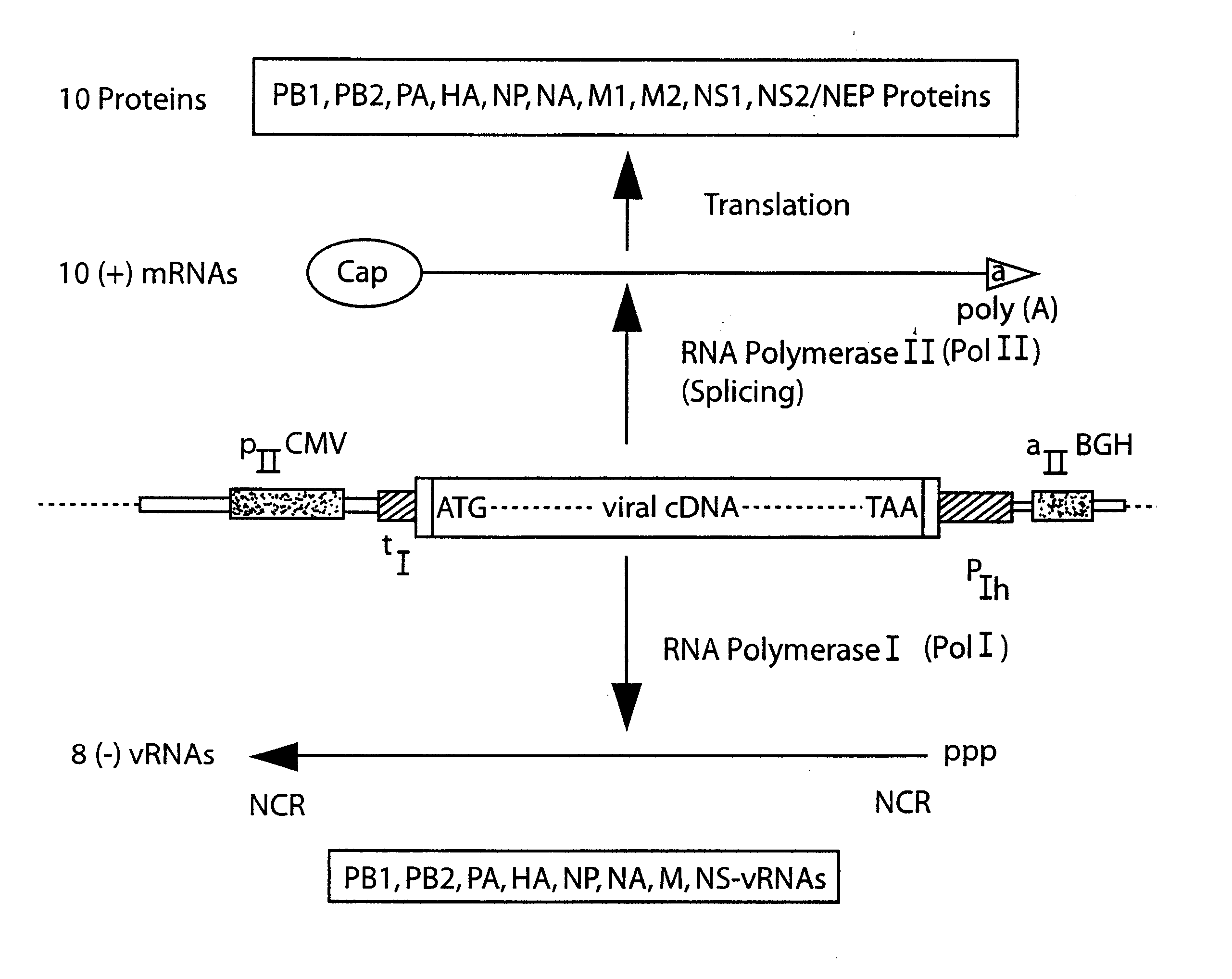
The present invention is based on the development of a dual promoter system (preferably a RNA pol I-pol II system) for the efficient intracellular synthesis of viral RNA. The resultant minimal plasmid-based system may be used to synthesize any RNA virus, preferably viruses with a negative single stranded RNA genome. The viral product of the system is produced when the plasmids of the system are introduced into a suitable host cell. One application of the system is production of attenuated, reassortant influenza viruses for use as antigens in vaccines. The reassortant viruses generated by cotransfection of plasmids may comprise genes encoding the surface glycoproteins hemagglutinin and neuramimidase from an influenza virus currently infecting the population and the internal genes from an attenuated influenza virus. An advantageous property of the present invention is its versatility; the system may be quickly and easily adapted to synthesize an attenuated version of any RNA virus. Attenuated or inactivated RNA viruses produced by the present invention may be administered to a patient in need of vaccination by any of several routes including intranasally or intramuscularly.
Owner:ST JUDE CHILDRENS RES HOSPITAL INC
Chimeric and/or growth-restricted flaviviruses
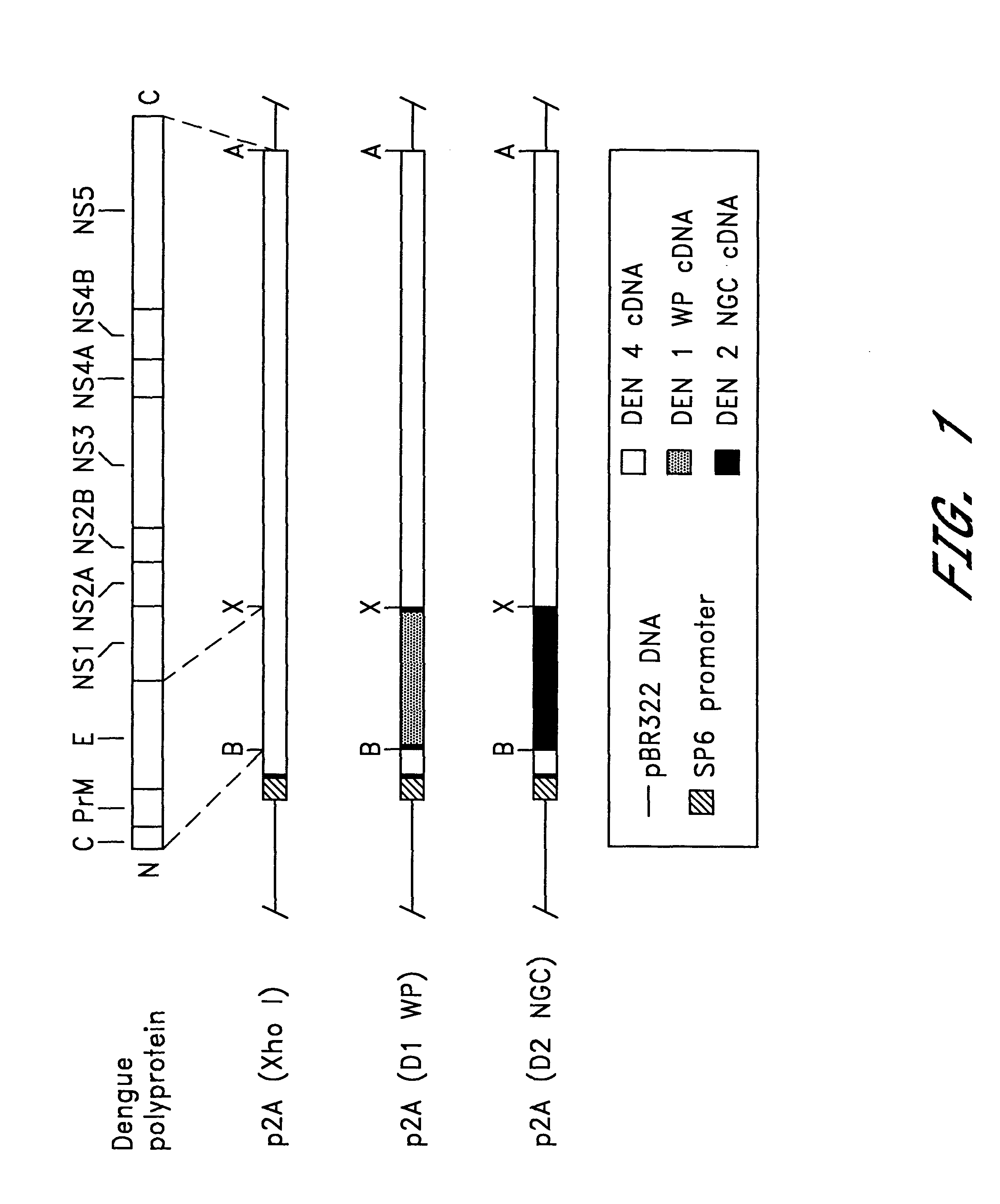
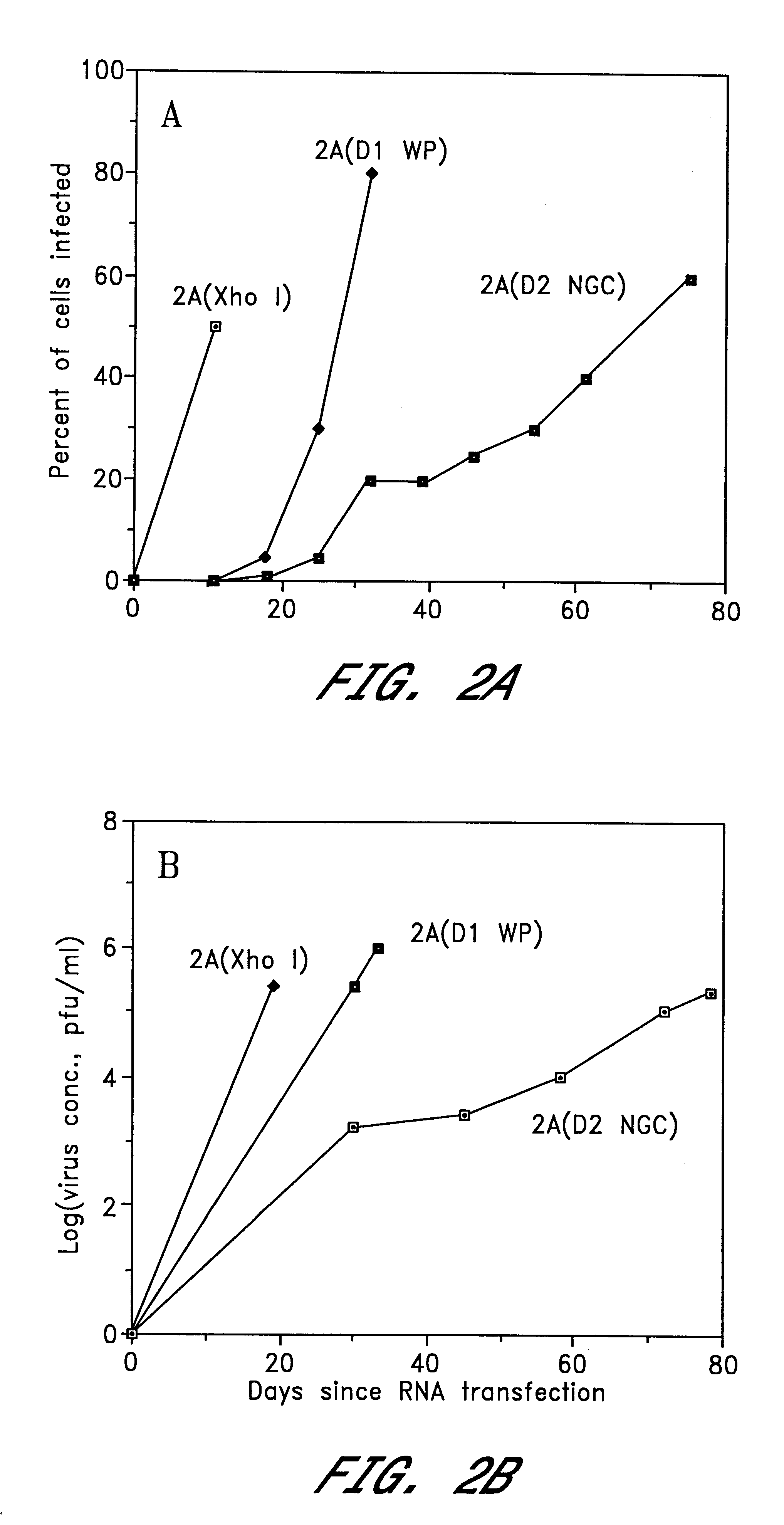
InactiveUS6184024B2BiocideSsRNA viruses positive-senseViral matrix proteinJapanese encephalitis viruses
The invention includes a chimeric virus for use in a vaccine preparation having a genome comprising nucleic acid sequences encoding at least one structural protein from one flavivirus and nucleic acid sequences encoding nonstructural protein from another flavivirus. The genome preferably includes mutations within the viral genome that reduce virus virulence and in a particularly preferred embodiment these vaccines are directed to flaviviruses such as dengue virus, tick-borne encephalitis virus and Japanese encephalitis virus. The invention also includes a baculovirus having a recombinant dengue cDNA sequence which encodes: (1) dengue virus capsid protein, pre-matrix protein, envelope glycoprotein and NS1 and NS2a nonstructural proteins or (2) dengue envelope glycoprotein or (3) dengue non-structural proteins NS1 and NS2a. The invention further includes a baculovirus having a recombinant Japanese B encephalitis virus cDNA sequence which encodes the Japanese B encephalitis virus capsid protein, pre-matrix protein, envelope glycoprotein and non-structural proteins NS1 and NS2a. The invention further includes a vaccine and a method to produce that vaccine.
Owner:US DEPT OF HEALTH & HUMAN SERVICES
Expression of HIV polypeptides and production of virus-like particles
InactiveUS6602705B1Sufficient amountImprove efficiencyOrganic active ingredientsSsRNA viruses positive-senseProteinDna immunization
The present invention relates to the efficient expression of HIV polypeptides in a variety of cell types, including, but not limited to, mammalian, insect, and plant cells. Synthetic expression cassettes encoding the HIV Gag-containing polypeptides are described, as are uses of the expression cassettes in applications including DNA immunization, generation of packaging cell lines, and production of Env-, tat- or Gag-containing proteins. The invention provides methods of producing Virus-Like Particles (VLPs), as well as, uses of the VLPs including, but not limited to, vehicles for the presentation of antigens and stimulation of immune response in subjects to whom the VLPs are administered.
Owner:CHIRON CORP
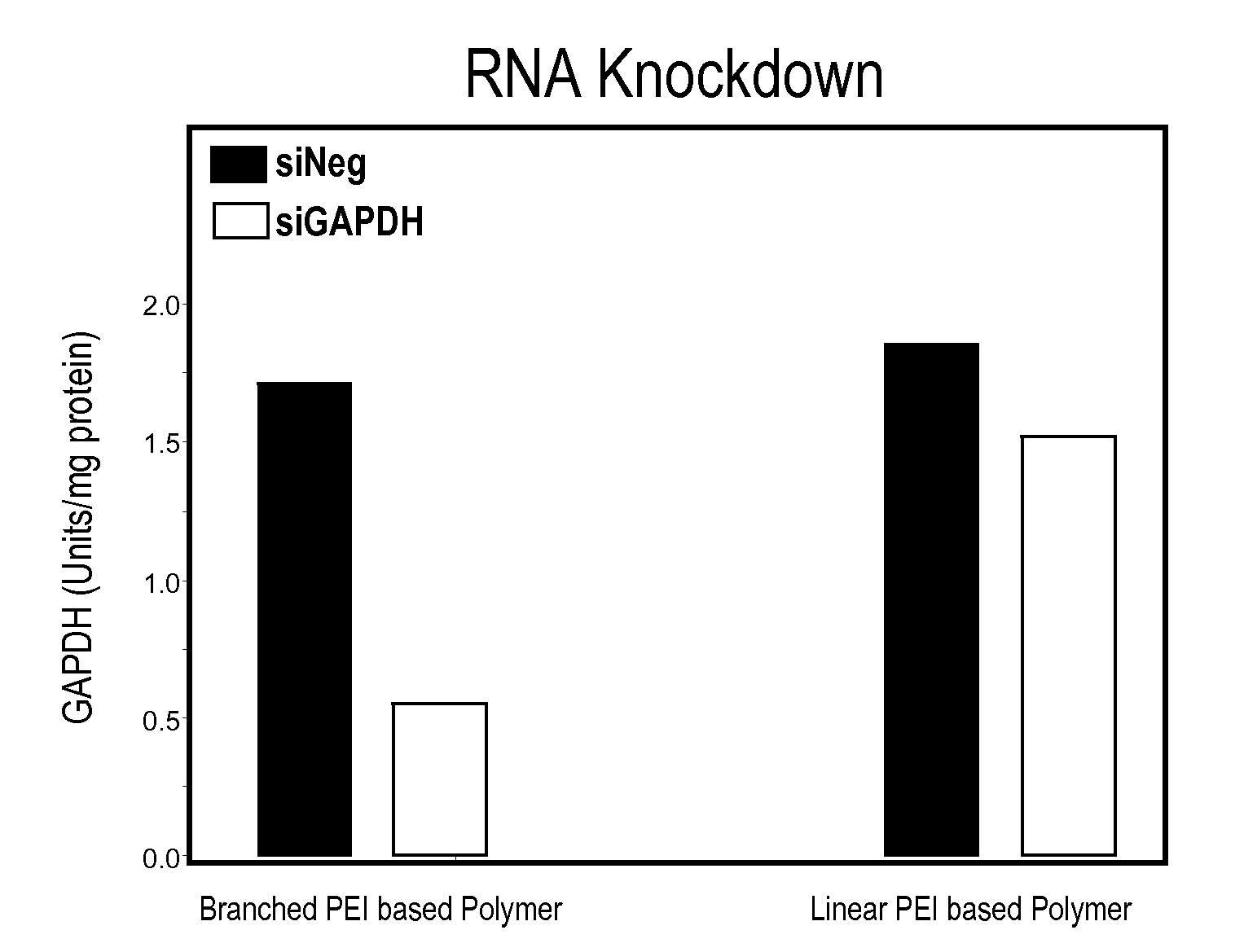
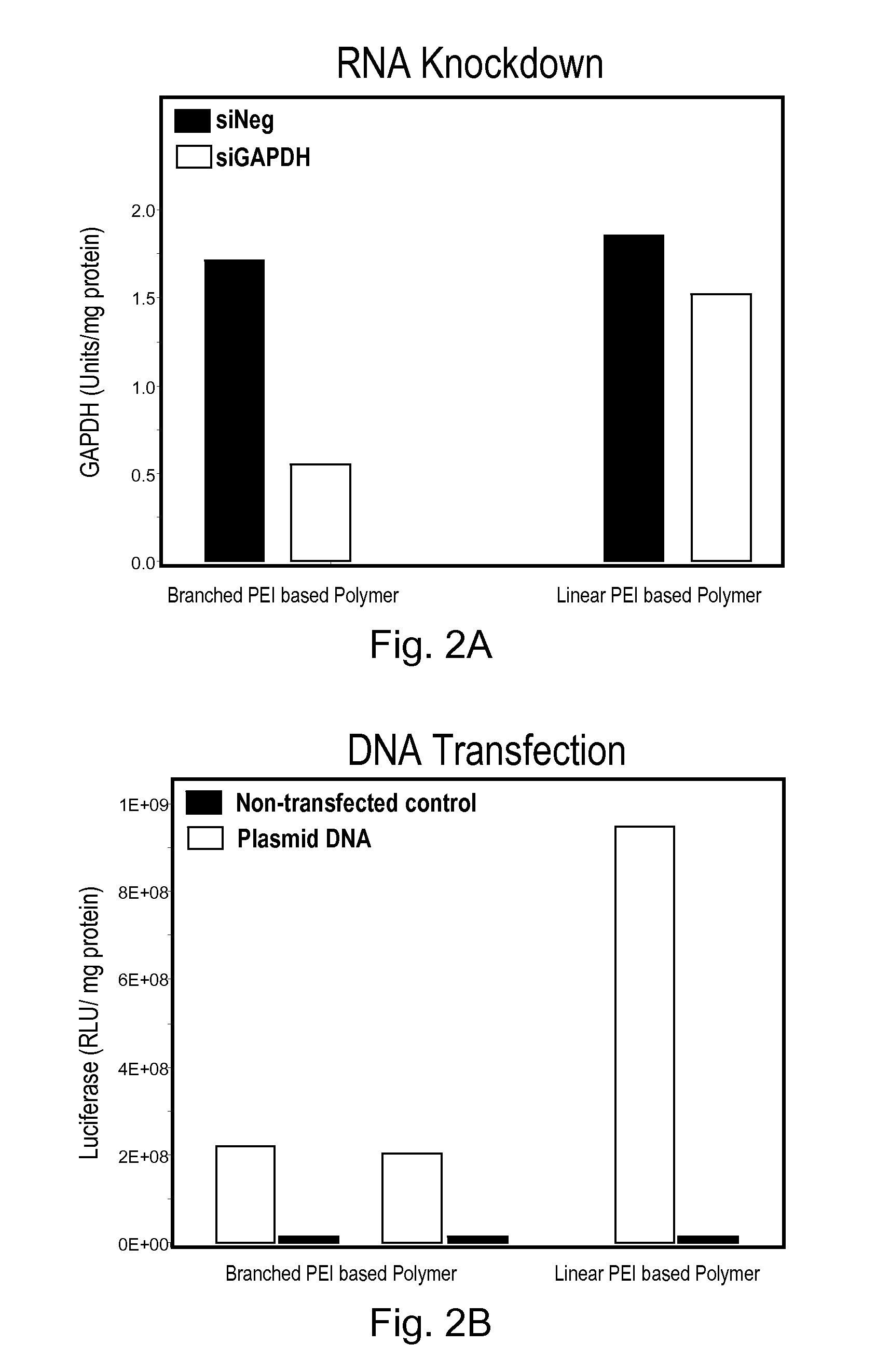
Biodegradable Cross-Linked Branched Poly(Alkylene Imines)
InactiveUS20100004315A1Easy to controlGuaranteed effective sizeBiocideSenses disorderCross-linkNucleotide
Disclosed is a cross-linked branched poly(alkylenimine) and compositions thereof and nucleotide molecules. Also disclosed are methods for preparing the cross-linked branched poly(alkylenimine).
Owner:CLSN LAB
Treatment of neoplasms with viruses
InactiveUS20030044384A1Convenient treatmentReduce productionSsRNA viruses negative-senseBiocideDiseaseAnti viral response
The subject invention relates to viruses that are able to replicate and thereby kill neoplastic cells with a deficiency in the IFN-mediated antiviral response, and their use in treating neoplastic disease including cancer and large tumors. RNA and DNA viruses are useful in this regard. The invention also relates to methods for the selection, design, purification and use of such viruses for cancer therapy.
Owner:PRO VIRUS
Prevention and treatment of HCV infection employing antibodies that inhibit the interaction of HCV virions with their receptor
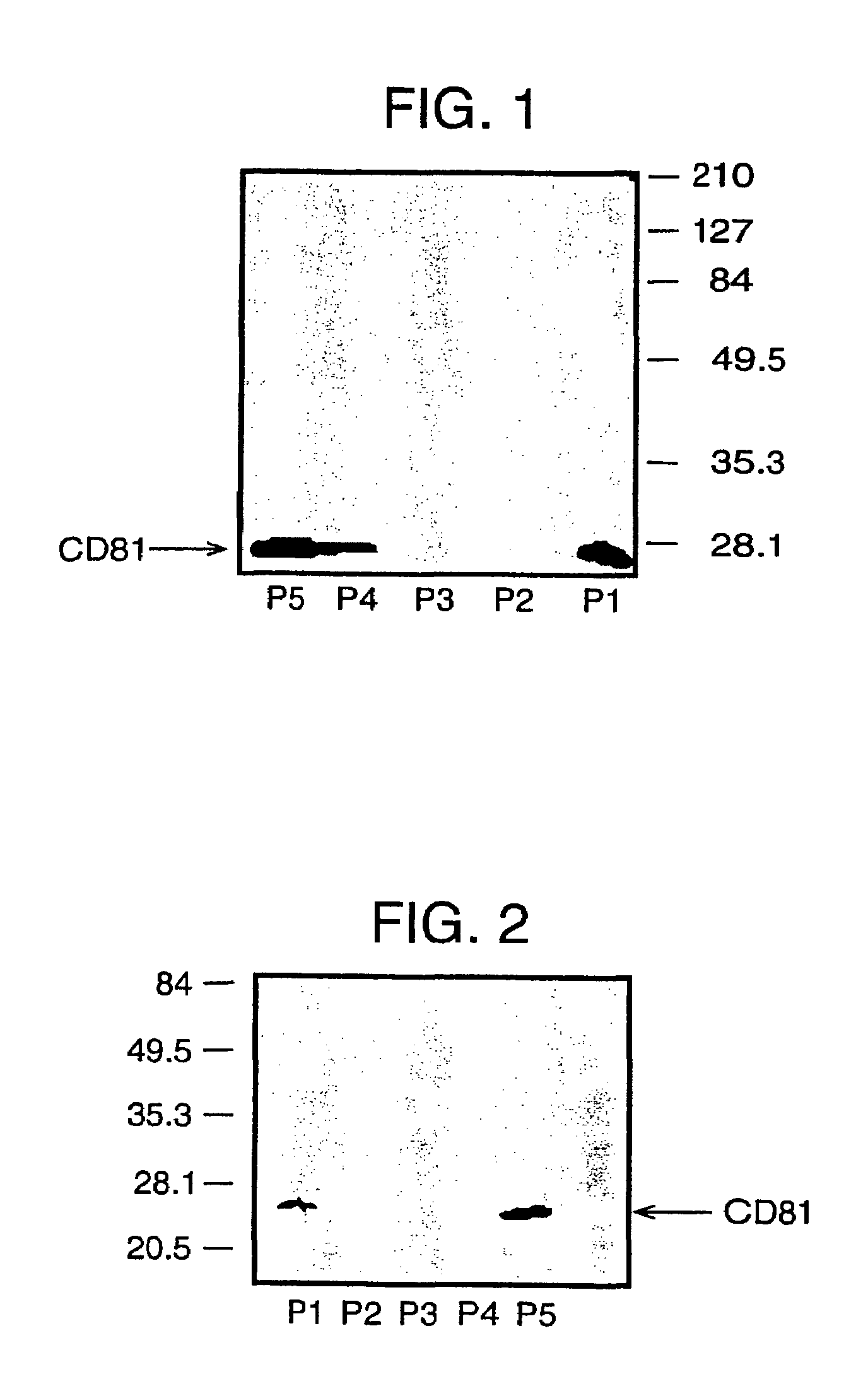
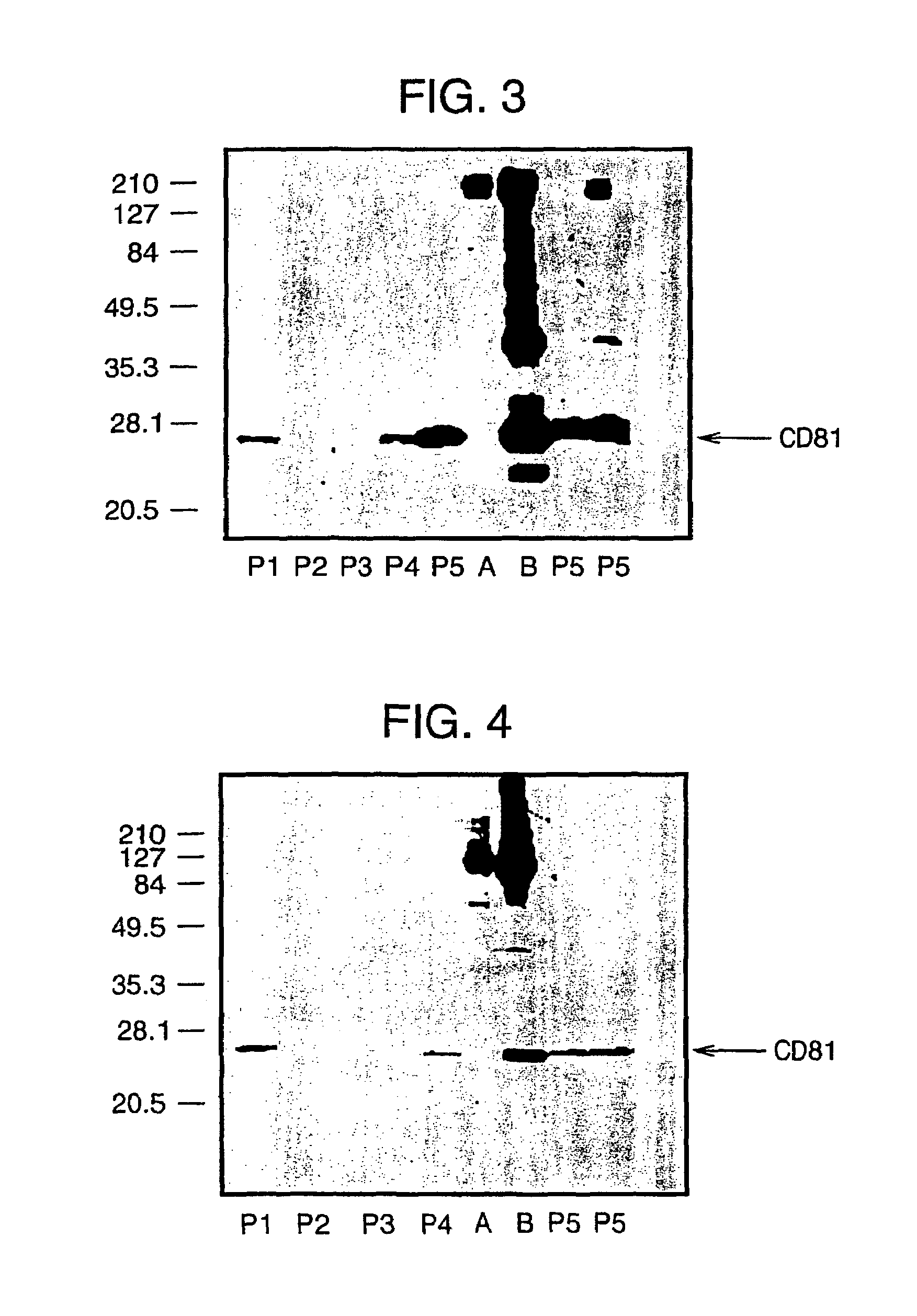
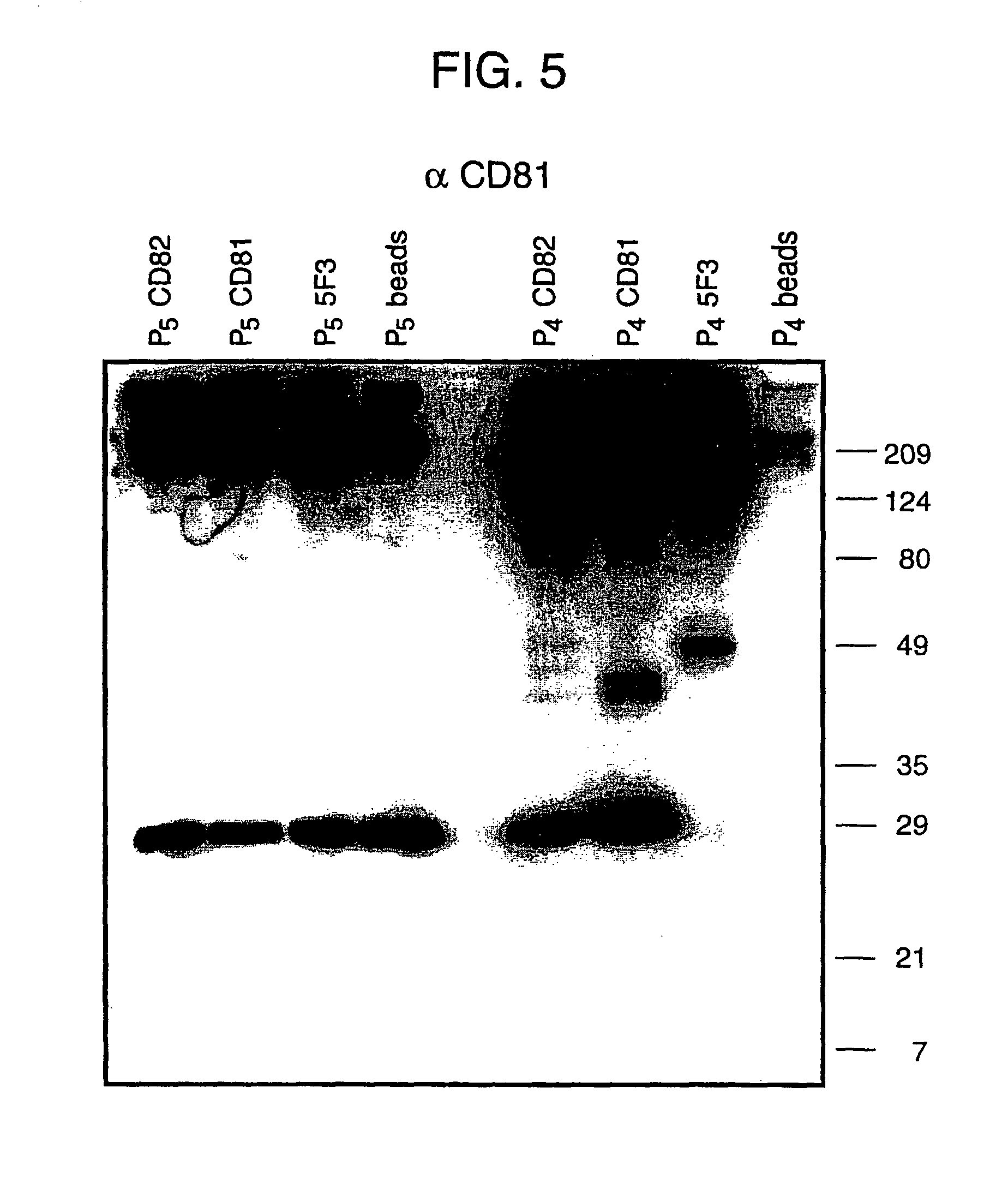
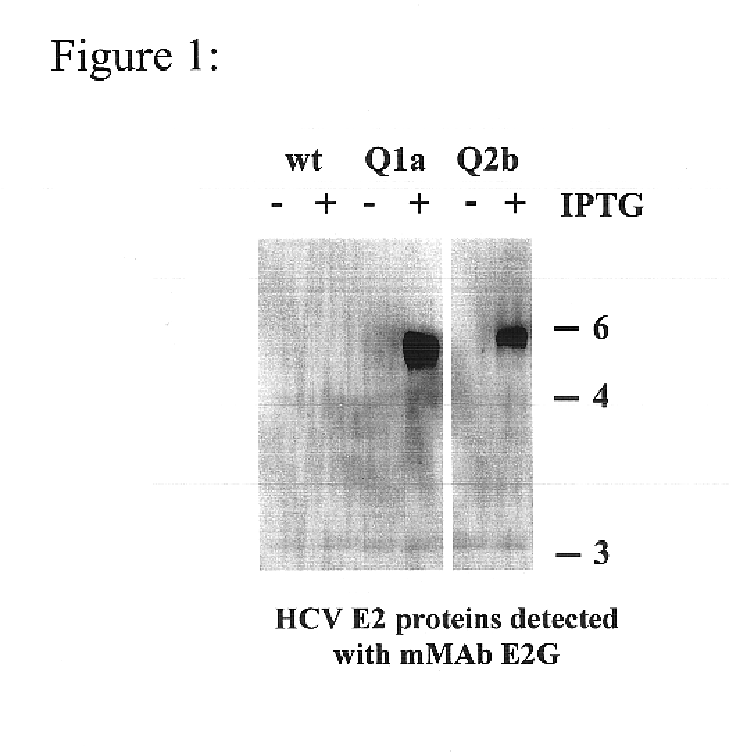
Human monoclonal antibodies binding to epitopes common to type 1 and 2 HCV are provided, as well as conformationally conserved HCV E2 2a and 2b proteins. Compositions comprising the antibodies find use in diagnosis and therapy. The antibodies recognize conformational epitopes that are conserved across multiple genotypes of HCV. Thus the antibodies have the potential to be useful in the prevention and treatment of the majority of HCV infections. A subset of the antibodies (CBH-2, CBH-5, CBH-7, CBH-8C, CBH-8E, and CBH-11) have the ability to prevent the binding of HCV E2 proteins of multiple genotypes to human CD81, a possible co-receptor for HCV infection. A subset of the antibodies (CBH-2 and CBH-5) have been shown to inhibit the binding of HCV virions (as opposed to purified E2 protein) to human CD81. A further subset of the antibodies (CBH-4D, CBH4B, CBH-8C, and CBH-9) have been shown to prevent HCV envelope mediated fusion using an HCV psuedotype system.
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV
Screening methods to identify agents that selectively inhibit hepatitis C virus replication
InactiveUS6030785APrevent dimerizationBlock viral inhibitionFungiSsRNA viruses positive-senseCellular defenseViral infection
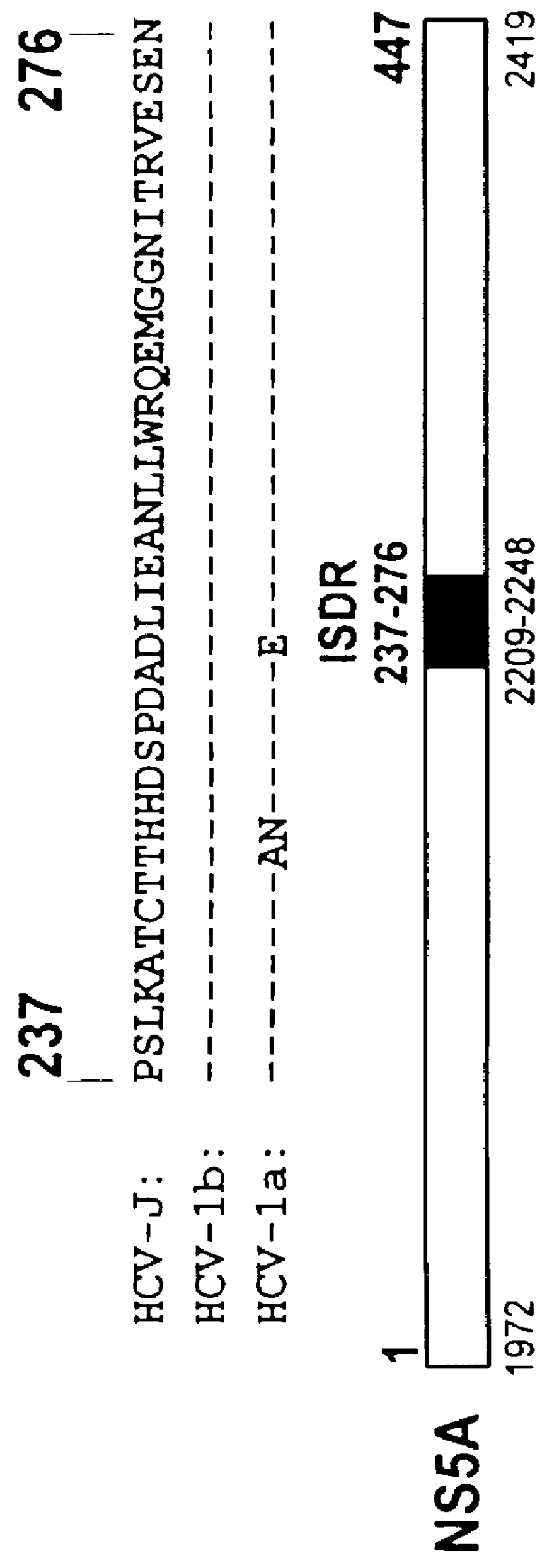
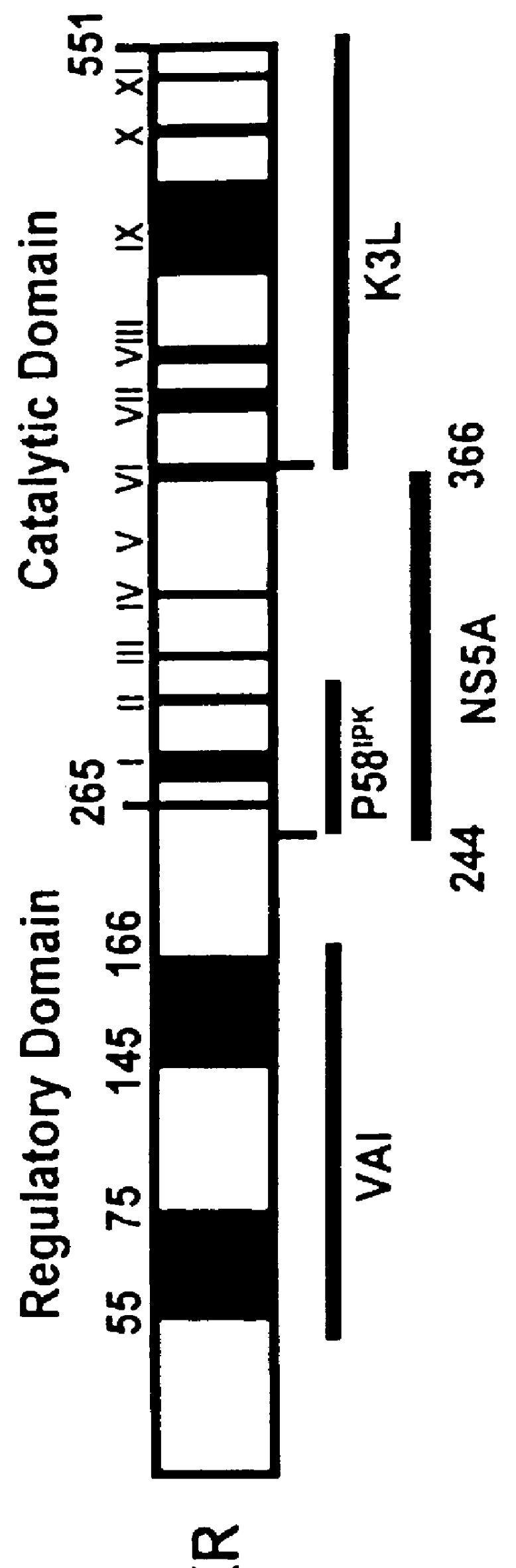
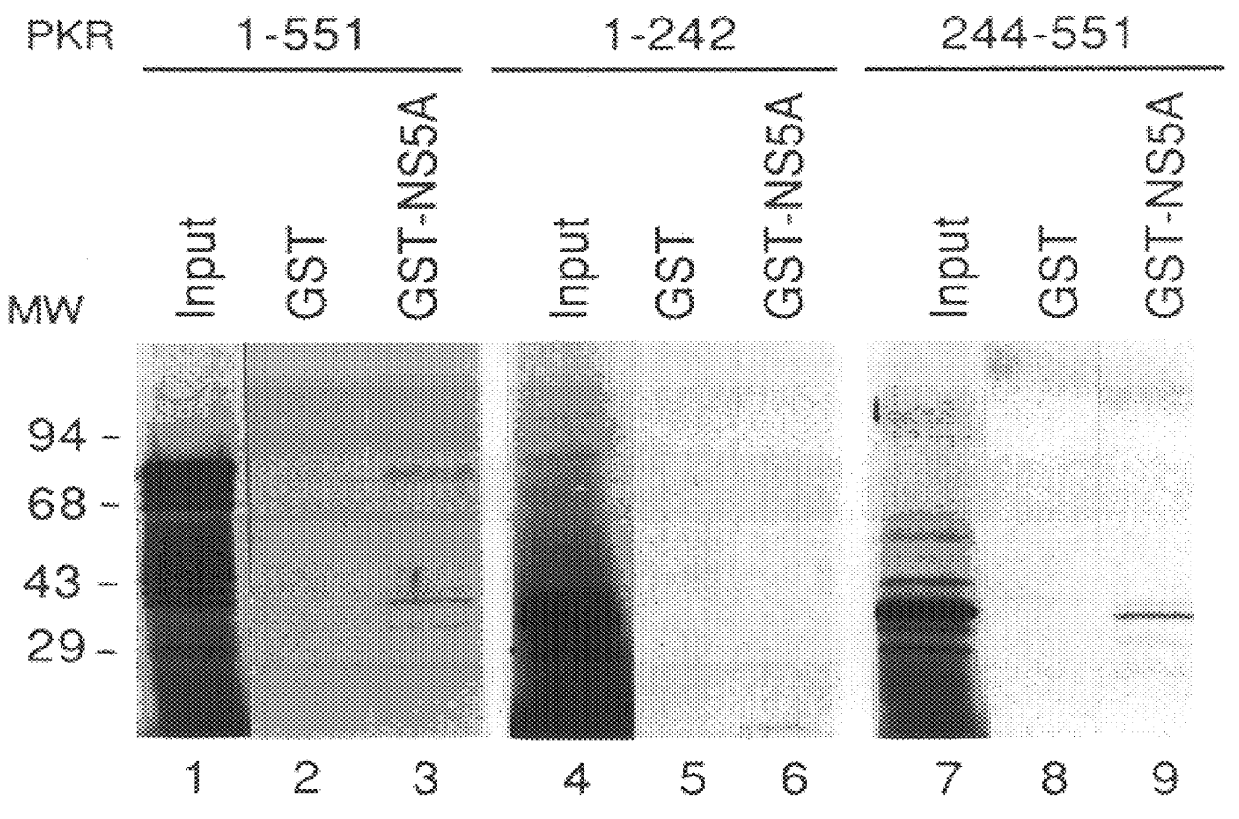
The present invention relates to novel methods for identifying antiviral agents which selectively interfere with viral proteins that override the interferon(IFN)-induced cellular defense mechanisms against viral infection. In particular, the present invention relates to screening assays that identify agents which selectively inhibit the interaction between viral proteins containing an interferon sensitivity determining region (ISDR) and IFN-induced PKR protein kinase. The present invention more particularly relates to screening assays that identify agents which selectively inhibit the interaction between hepatitis C virus (HCV) nonstructural 5A protein (NS5A), which contains an ISDR, and IFN-induced PKR protein kinase. The interaction between the viral ISDR and IFN-induced PKR protein kinase results in the override of IFN-induced cellular defense mechanisms to combat viral infection. Therefore the agents identified using the assays of the invention may have utility as antiviral agents.
Owner:UNIV OF WASHINGTON
Immunisation of large mammals with low doses of RNA
ActiveUS20130149375A1Conveniently preparedImprove stabilityAntibacterial agentsSsRNA viruses negative-senseMammalImmunity response
RNA encoding an immunogen is delivered to a large mammal at a dose of between 2 μg and 100 μg. Thus the invention provides a method of raising an immune response in a large mammal, comprising administering to the mammal a dose of between 2 μg and 100 μg of immunogen-encoding RNA. Similarly, RNA encoding an immunogen can be delivered to a large mammal at a dose of 3 ng / kg to 150 ng / kg. The delivered RNA can elicit an immune response in the large mammal
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
Human SARS-CoV-2 monoclonal antibody and preparation method and application thereof
PendingCN111153991AHigh affinityStrong specificitySsRNA viruses positive-senseVirus peptidesBALB/cMonoclonal antibody agent
The invention discloses a human SARS-CoV-2 monoclonal antibody. The preparation method of the human SARS-CoV-2 monoclonal antibody comprises the steps: adopting SARS-CoV Nucleocapsid recombinant protein as immunogen, immunizing BALB / c mice, performing fusion and subcloning on spleen cells and myeloma cells of mice, then performing a large amount of repeated screening and domestication of cell lines through commercialized products SARS-CoV-2 Nucleocapsid and MERS Nucleocapsid so as to obtain a hybridoma cell line capable of secreting the SARS-CoV-2-resistant N monoclonal antibody with high affinity and high specificity finally and successfully, and finally performing ascites preparation and purification so as to obtain the monoclonal antibody, wherein the amino acid sequence of the SARS-CoVNucleocapsid recombinant protein is shown in SEQ ID No. 1. The invention also discloses application of the monoclonal antibody in preparation of SARS-CoV-2 virus detection products and preparation ofdrugs for inhibiting the SARS-CoV-2 viruses. The monoclonal antibody can be used for detecting the SARS-CoV-2 in human throat swabs / pulmonary secretions and other samples by using a double-antibody sandwich method, and can be applied to diagnosis and prevention and control of SARS-CoV-2 virus infection and scientific researches of viruses and other study.
Owner:BEIJING BIOSYNTHESIS BIOTECH
Method for processing and preserving collagen-based tissues for transplantation
InactiveUS20060210960A1Minimize surface tension damageAugment selective preservationSsRNA viruses positive-senseViral antigen ingredientsOrganismBiology
A method for processing and preserving an acellular collagen-based tissue matrix for transplantation is disclosed. The method includes the steps of processing biological tissues with a stabilizing solution to reduce procurement damage, treatment with a processing solution to remove cells, treatment with a cryoprotectant solution followed by freezing, drying, storage and rehydration under conditions that preclude functionally significant damage and reconstitution with viable cells.
Owner:LIFECELL
Screening for west nile virus antiviral therapy
InactiveUS20050058987A1Improve efficiencySsRNA viruses positive-senseVectorsHigh-Throughput Screening MethodsImmunogenicity
The instant invention provides stable and novel lineage I WNV reverse genetics systems, and methods for making the reverse genetics systems, specifically, a fully-infectious lineage I WNV cDNA or replicon system engineered with one or more nucleotide sequences each encoding a reporter gene to be used in high throughput cell-based screening assays for the identification of novel antiflaviviral chemotherapeutics and / or vaccines effective to treat and / or immunize against infections by WNV and other emerging flaviviruses, such as, for example, JEV, SLEV, AV, KV, JV, CV, YV, TBEV, DENV-1, DENV-2, DENV-3, DENV-4, YFV and MVEV. The present invention further provides methods of high throughput screening of antiflaviviral compounds or improved derivatives thereof using novel lineage I WNV reverse genetics systems and / or cell lines stably containing the reverse genetics systems. Also, the invention provides novel pharmaceutical compositions comprising an attenuated lineage I WNV that is less virulent but similarly immunogenic as the parent WNV and is capable of providing a protective immune response in a host.
Owner:HEALTH RES INC
Methods for identification of coronaviruses
InactiveUS20050266397A1SsRNA viruses positive-senseSugar derivativesMass Spectrometry-Mass SpectrometryRapid identification
The present invention provides a method for rapid identification and quantitation of bacteria by amplification of a segment of bacterial nucleic acid followed by analysis by mass spectrometry. The compositions provide for characterization of the molecular masses and base compositions of bacterial nucleic acids which are used to rapidly identify bacteria.
Owner:IBIS BIOSCI
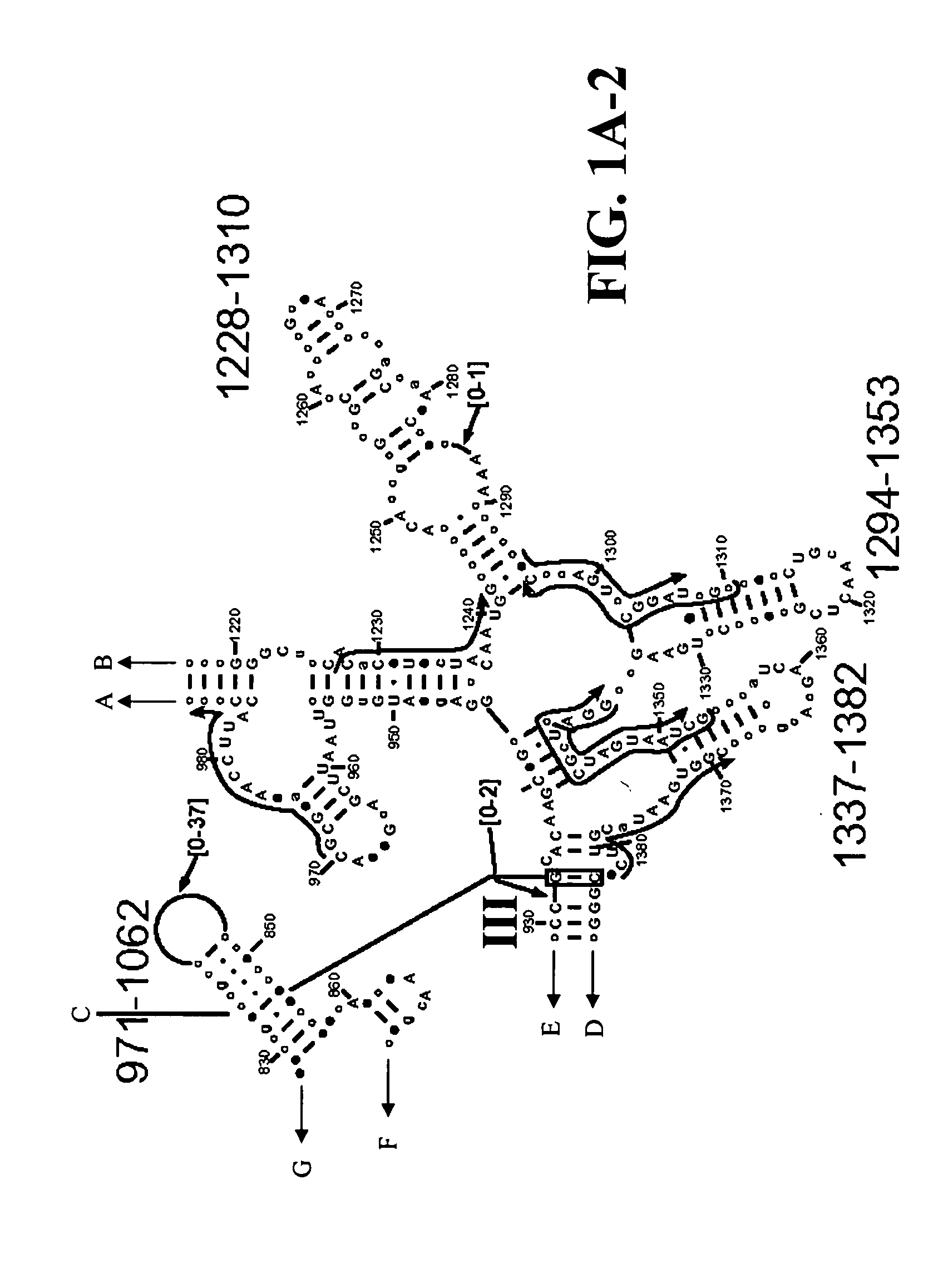
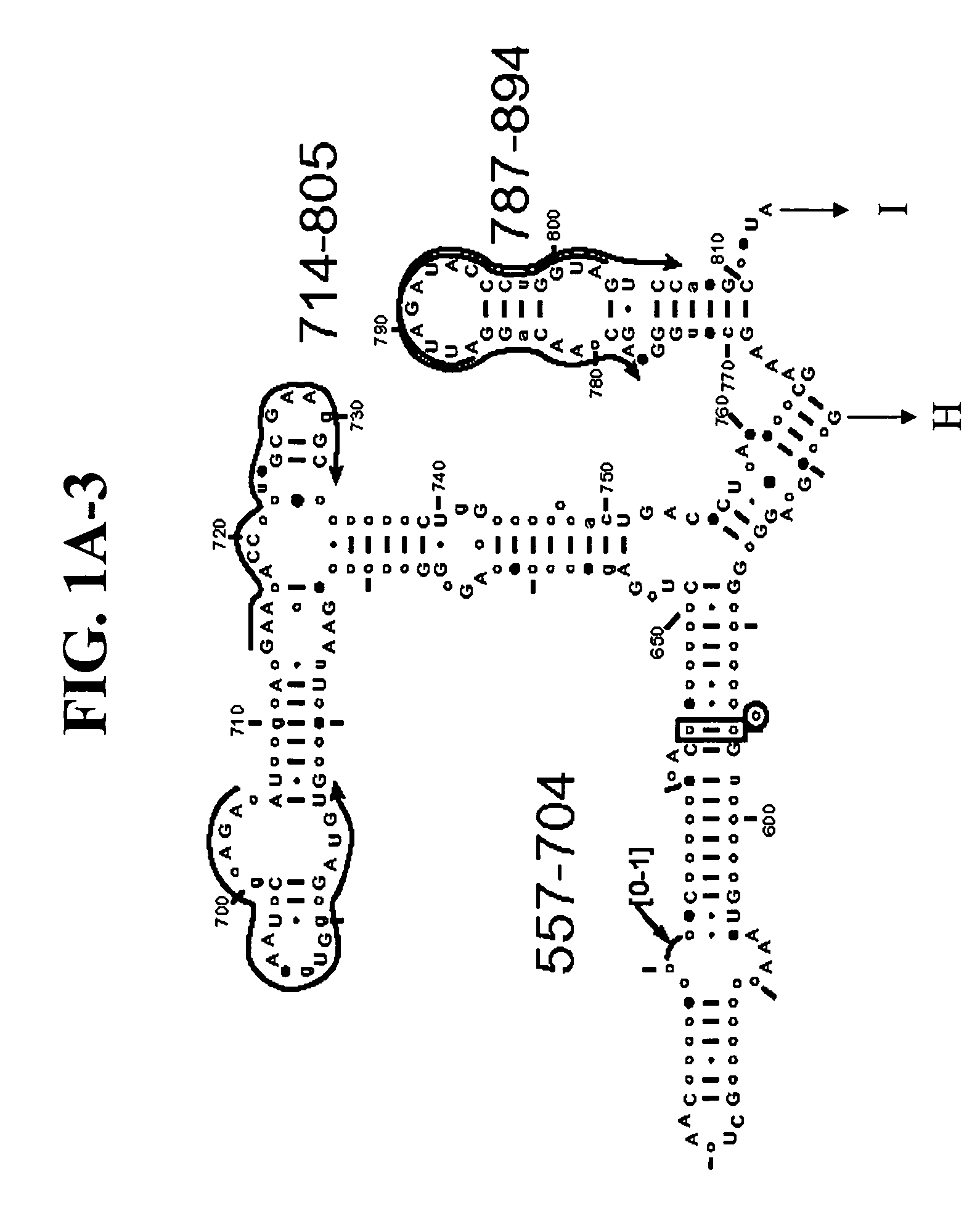
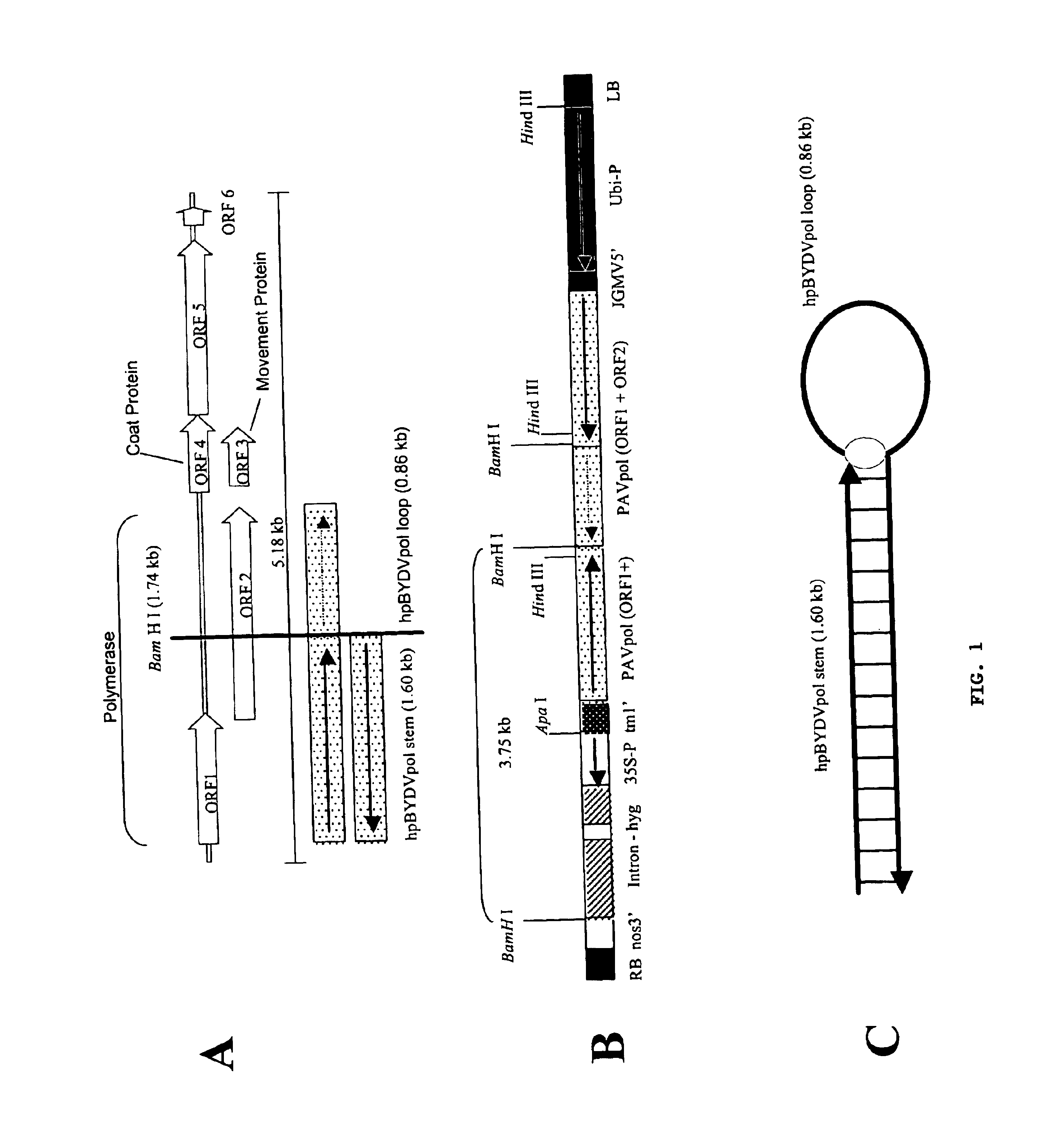
Methods and means for producing barley yellow dwarf virus resistant cereal plants
The invention relates to methods for producing transgenic cereal plants resistant to Barley Yellow Dwarf Virus, particularly in the presence of co-infecting Cereal Yellow Dwarf Virus, by stably integrating into the cells of the transgenic plant a chimeric gene comprising a DNA region operably linked to plant expressible promoter in such a way that the RNA molecule may be transcribed from the DNA region, the RNA molecule comprising both sense and antisense RNA capable of pairing and forming a double stranded RNA molecule or hairpin RNA.
Owner:COMMONWEALTH SCI & IND RES ORG
Method of producing a vaccine which raises an immunological response against a virus causing a porcine respiratory and reproductive disease
InactiveUS6251404B1Efficient responseAntibacterial agentsSsRNA viruses positive-senseInfectious agentRespiratory disease
The present invention provides a vaccine which protects pigs from a virus and / or an infectious agent causing a porcine respiratory and reproductive disease, a method of protecting a pig from a disease caused by a virus and / or an infectious agent which causes a respiratory and reproductive disease, a method of producing a vaccine against a virus and / or an infectious agent causing a porcine reproductive and respiratory disease, and a biologically pure sample of a virus and / or infectious agent associated with a porcine respiratory and reproductive disease, particularly the Iowa strain of porcine reproductive and respiratory syndrome virus (PRRSV), and an isolated polynucleotide which is at least 90% homologous with a polynucleotide obtained from the genome of a virus and / or infectious agent which causes a porcine respiratory and reproductive disease.
Owner:ZOETIS WHC 2
Polypeptides fused with alfalfa mosaic virus or ilarvirus capsid proteins
InactiveUS6042832AEfficient presentation of antigenImprove protectionSsRNA viruses negative-senseSsRNA viruses positive-senseAntigenIlarvirus
Owner:THOMAS JEFFERSON UNIV
Hepatitis c virus inhibitors
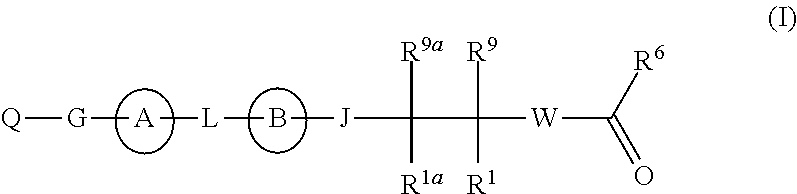
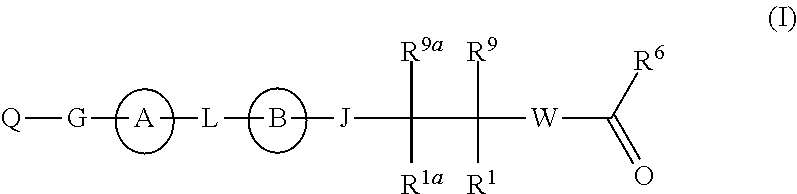
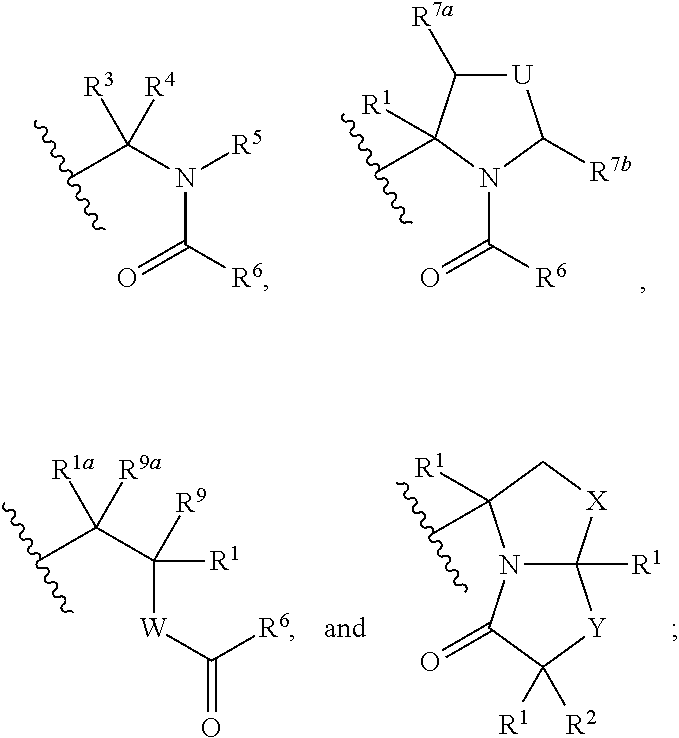
The present invention discloses compounds of Formula (I), or pharmaceutically acceptable salts, esters, or prodrugs thereof:which inhibit RNA-containing virus, particularly the hepatitis C virus (HCV). Consequently, the compounds of the present invention interfere with the life cycle of the hepatitis C virus and are also useful as antiviral agents. The present invention further relates to pharmaceutical compositions comprising the aforementioned compounds for administration to a subject suffering from HCV infection. The invention also relates to methods of treating an HCV infection in a subject by administering a pharmaceutical composition comprising the compounds of the present invention. The present invention relates to novel antiviral compounds represented herein above, pharmaceutical compositions comprising such compounds, and methods for the treatment or prophylaxis of viral (particularly HCV) infection in a subject in need of such therapy with said compounds.
Owner:ENANTA PHARM INC
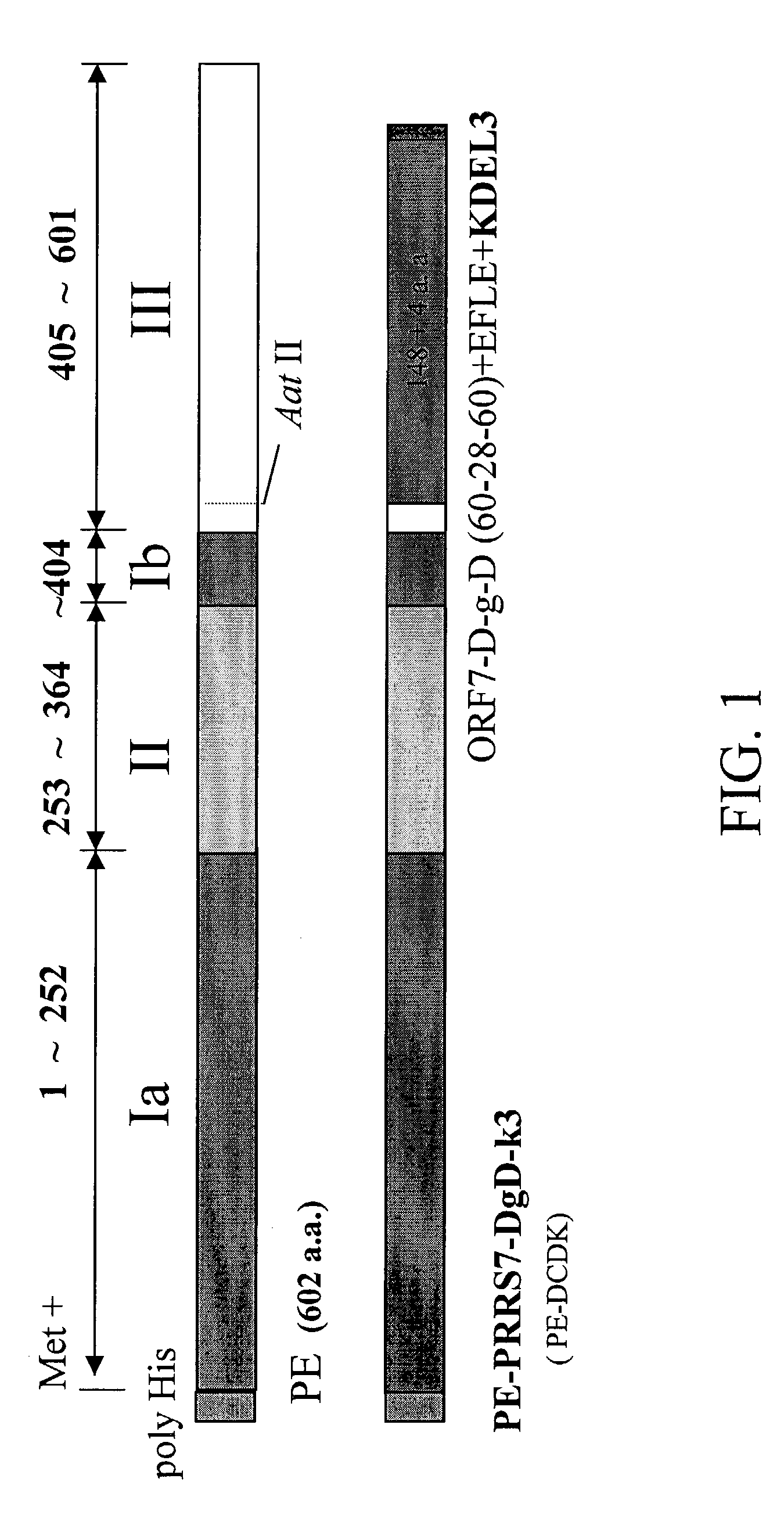
Fusion antigen used as vaccine
ActiveUS7335361B2SsRNA viruses positive-senseAntibody mimetics/scaffoldsPseudomonas tolaasiiReticulum cell
The present invention mainly provides a fusion antigen specific for a target cell comprising a ligand moiety which is capable of reacting, recognizing or binding to the receptors on the target cell, a Pseudomonas exotoxin A translocation domain II, an antigenic moiety, and a carboxyl terminal moiety which permits combination of the fusion antigen to the endoplasmic reticulum (ER) membrane of the target cell. A method of immunizing an animal using the fusion antigen is also provided.
Owner:AGRI TECH RES INST
Generation of replication competent viruses for therapeutic use
InactiveUS7550296B2Reduced viral genomeImprove effectivenessSsRNA viruses positive-sensePeptide/protein ingredientsDiseaseReplication competent virus
The present invention relates to the generation of replication-competent viruses having therapeutic utility. The replication-competent viruses of the invention can express proteins useful in the treatment of disease.
Owner:PSIOXUS THERAPEUTICS LTD
Modified plant virus particles and uses therefor
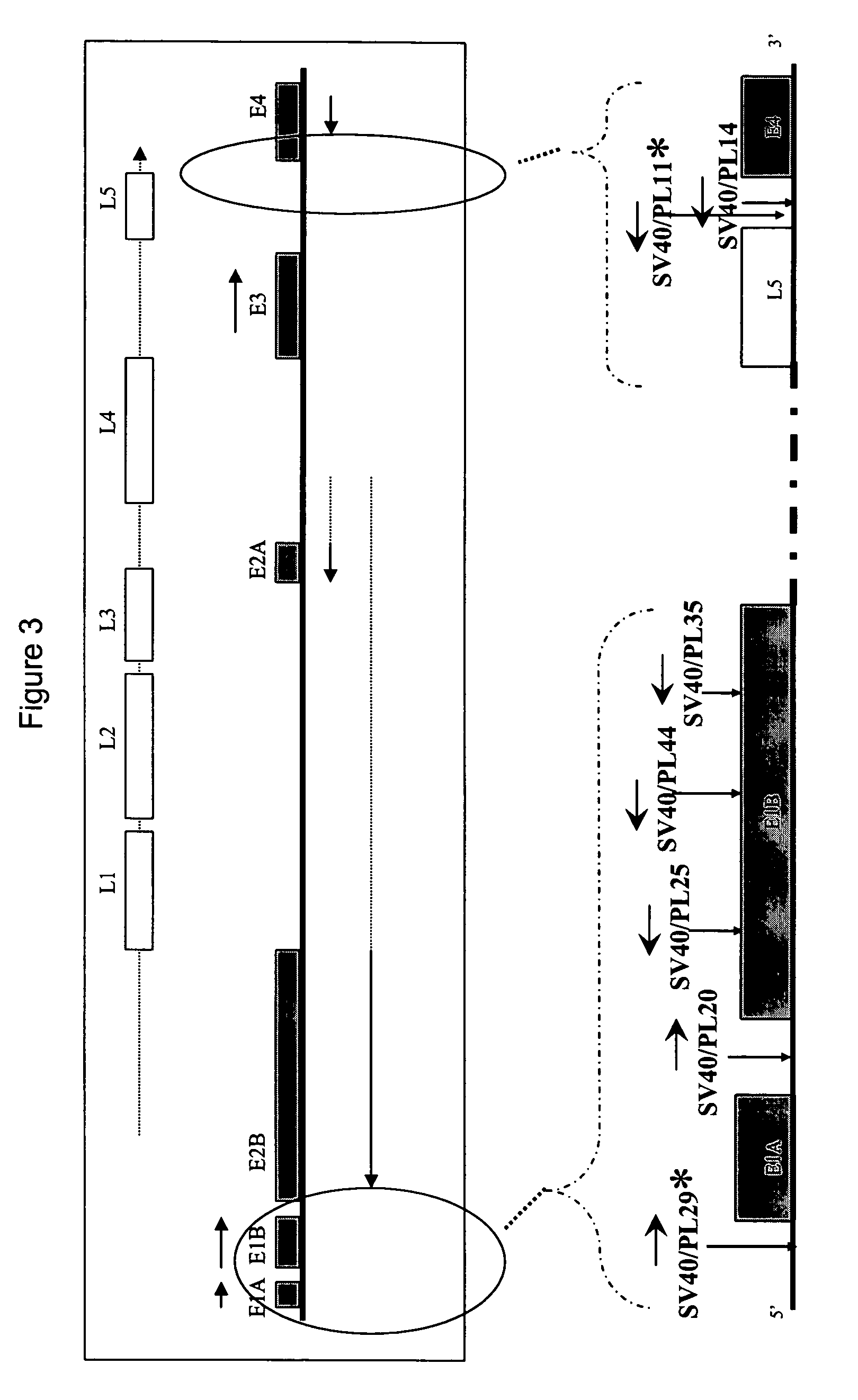
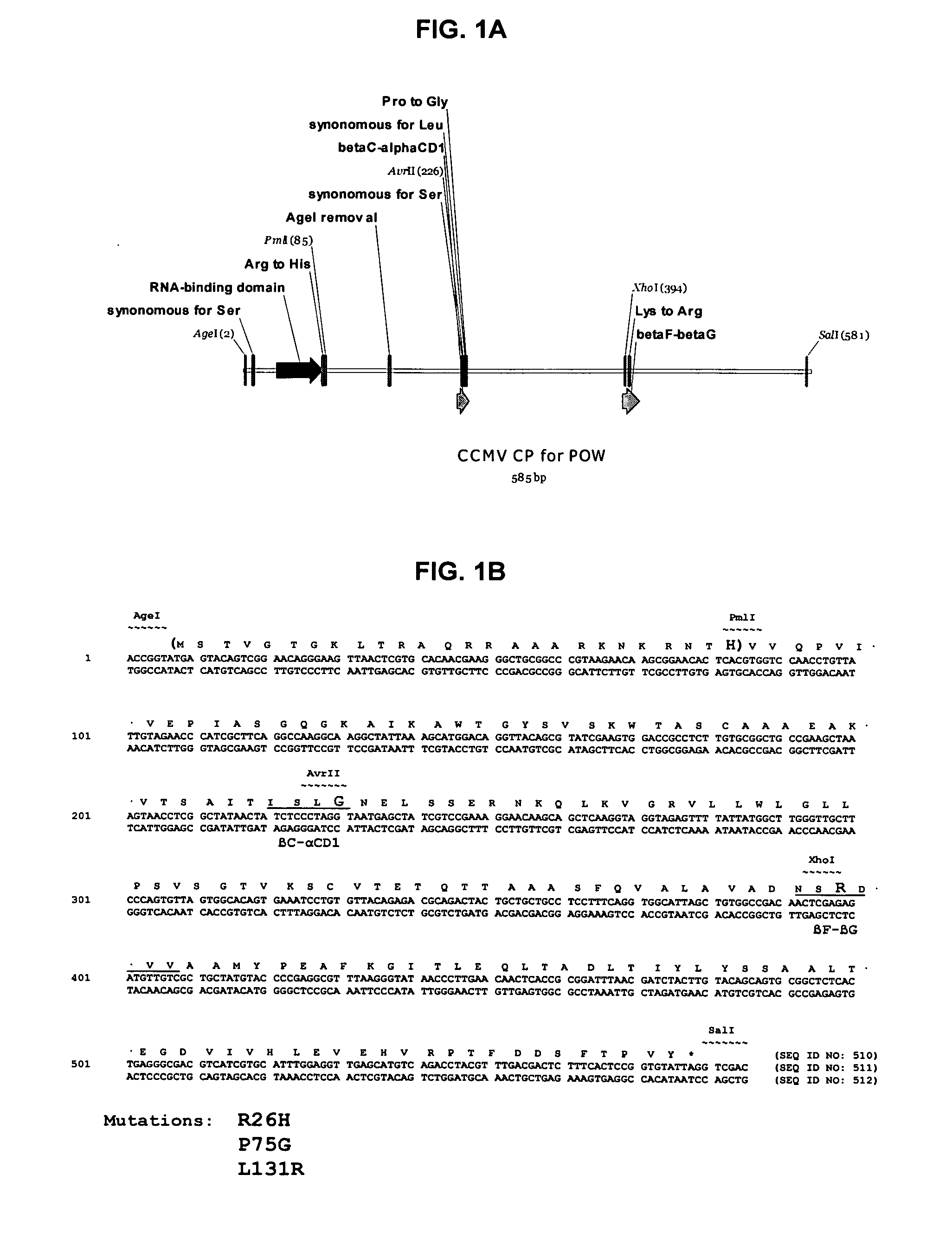
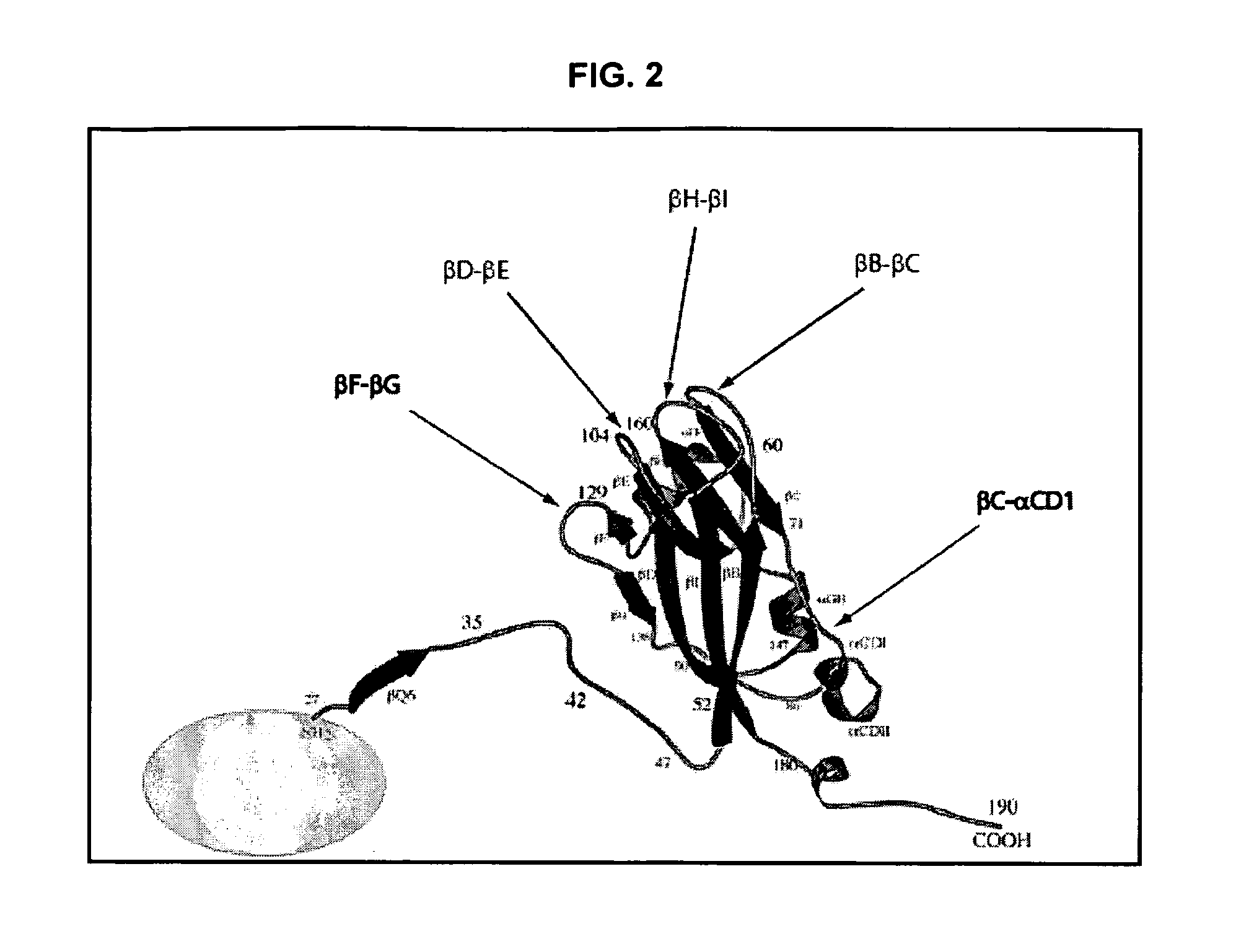
InactiveUS20120015899A1Broadening arrayEasy to assembleFusion with RNA-binding domainBiocideVirus-like particleCoat Proteins
Aspects of the invention provide modified virus-like particles that are designed for therapeutic applications. In particular, aspects of the invention provide CCMV coat proteins that are modified to generate virus-like particles, including mosaic virus-like particles, that can package and / or deliver one or more diagnostic and / or therapeutic agents. The invention also provides methods for treating subjects with one or more modified virus-like particles.
Owner:PLANT BIOSCI LTD +1
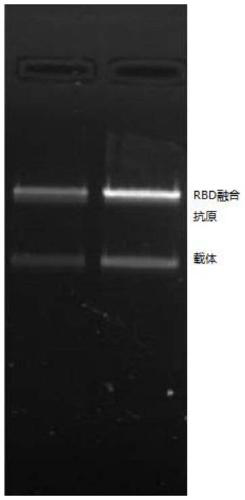
SARS-CoV-2 vaccine and preparation method thereof
ActiveCN111217917AEnter to helpImproving immunogenicityPolypeptide with localisation/targeting motifSsRNA viruses positive-senseAntigenDisease
The invention relates to a preparation method for a vaccine capable of treating and / or preventing SARS-CoV-2 infection or COVID-19 diseases. The core antigen of the vaccine comprises the RBD (receptor binding zone) fusion protein of the SARS-CoV-2, and a vaccine form comprises an RBD fusion protein subunit vaccine, an RBD fusion protein mRNA vaccine or an RBD fusion protein adenovirus vector vaccine. The above vaccine immunizes an organism, and immune reaction for treating and / or preventing the SARS-CoV-2 infection can be generated so as to be used for treating and / or preventing COVID-19. The invention also relates to an RBD fusion gene, the RBD fusion protein, a carrier, a cell, a preparation method, a treatment method or a pharmacy purpose of the SARS-CoV-2.
Owner:CANSINO BIOLOGICS INC
Immunodeficiency recombinant poxvirus
InactiveUS6596279B1SsRNA viruses negative-senseSsRNA viruses positive-senseFeline immunodeficiency virusCtl epitope
Attenuated recombinant viruses containing DNA encoding an immunodeficiency virus and / or CTL antigen, as well as methods and compositions employing the viruses, expression products therefrom, and antibodies generated from the viruses or expression products, are disclosed and claimed. The recombinant viruses can be NYVAC or ALVAC recombinant viruses. The DNA can code for at least one of: HIV1gag(+pro)(IIIB), gp120(MN)(+transmembrane), nef(BRU)CTL, pol(IIIB)CTL, ELDKWA or LDKW epitopes, preferably HIV1gag(+pro)(IIIB), gp120(MN) (+transmembrane), two (2) nef(BRU)CTL and three (3) pol(IIIB)CTL etpitopes; or two ELDKWA in gp120 V3 or another region or in gp160. The two (2) nef(BRU)CTL and three (3) pol(IIIB)CTL epitopes are preferably CTL1, CTL2, pol1, pol2 and pol3. The recombinant viruses and gene products therefrom and antibodies generated by the viruses and gene products have several preventive, therapeutic and diagnostic uses. DNA from the recombinant viruses are useful as probes or, for generating PCR primers or for immunization. Also disclosed and claimed are HIV immunogens and modified gp160 and gp120.
Owner:VIROGENETICS
Poxvirus-canine distemper virus (CDV) or measles virus recombinants and compositions and methods employing the recombinants
InactiveUS6309647B1Improve securityImprove security levelSsRNA viruses negative-senseSsRNA viruses positive-senseHemagglutininCanine distemper virus Antigen
Attenuated recombinant viruses containing DNA coding for a canine distemper virus antigen or measles M or N antigen, as well as methods and compositions employing the viruses, are disclosed and claimed. The recombinant viruses can be NYVAC or ALVAC recombinant viruses. The DNA can code for at least one of: canine distemper virus fusion protein, canine distemper virus hemagglutinin glycoprotein, canine distemper nucleocaspid protein, canine distemper matrix protein, measles virus nucleocaspid protein, and measles virus matrix protein. The recombinant viruses and gene products therefrom are useful for eliciting protection against canine distemper virus and / or measles virus, and, the gene products and antibodies elicited thereby are useful in assays. Additionally, DNA from the recombinants is used for probes or for generating PCR primers.
Owner:AVENTIS PASTEUR LTD
mVSV virus vector and virus vector vaccine, and COVID-19 vaccine based on mVSV mediation
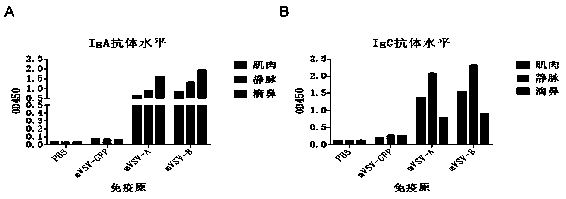
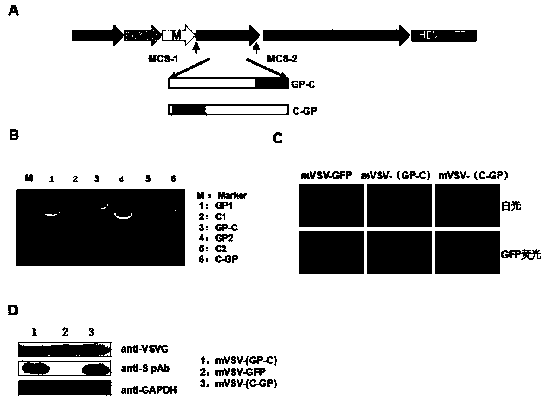
ActiveCN111088283AEnhance immune responseStrong immune responseSsRNA viruses negative-senseSsRNA viruses positive-senseHeterologousReceptor
The invention provides an mVSV virus vector, i.e., attenuated mVSV obtained after multiple modification mutations occur to an M protein amino acid site of a wild Indiana strain VSV, and an optimized heterologous antigen gene is preferentially integrated to a double cloning site area of an mVSV packaging core plasmid pmVSV-Core at the same time. The mVSV virus vector vaccine comprises a heterologous antigen gene which fuses or embeds a target virus between G and L genes of an mVSV vector envelope, wherein the antigen gene comprises an enveloped and embedded antigen gene encoding the target virus, an embedded combination antigen gene or a fused antigen gene; the mVSV virus vector is embedded or fused with a dominant antigen of spike protein S of an SARS-CoV-2 pathogen; the dominant antigen is preferably selected from a receptor binding domain of spike protein S, namely RBD; and a COVID-19 vaccine based on mVSV mediation is formed. The vaccine has good prevention or treatment effect on COVID-19 infected people.
Owner:FANTASIA BIOPHARMA ZHEJIANG CO LTD
Antigen-binding molecule capable of binding to two or more antigen molecules repeatedly
InactiveUS20130011866A1Pharmacokinetics of the antigen-binding molecule can be improvedGood effectSsRNA viruses negative-senseCompound screeningHalf-lifeAntigen binding
The present inventors discovered that antibodies having weaker antigen-binding activity at the early endosomal pH in comparison with that at the pH of plasma are capable of binding to multiple antigen molecules with a single antibody molecule, have long half-lives in plasma, and have improved durations of time in which they can bind to antigen.
Owner:CHUGAI PHARMA CO LTD
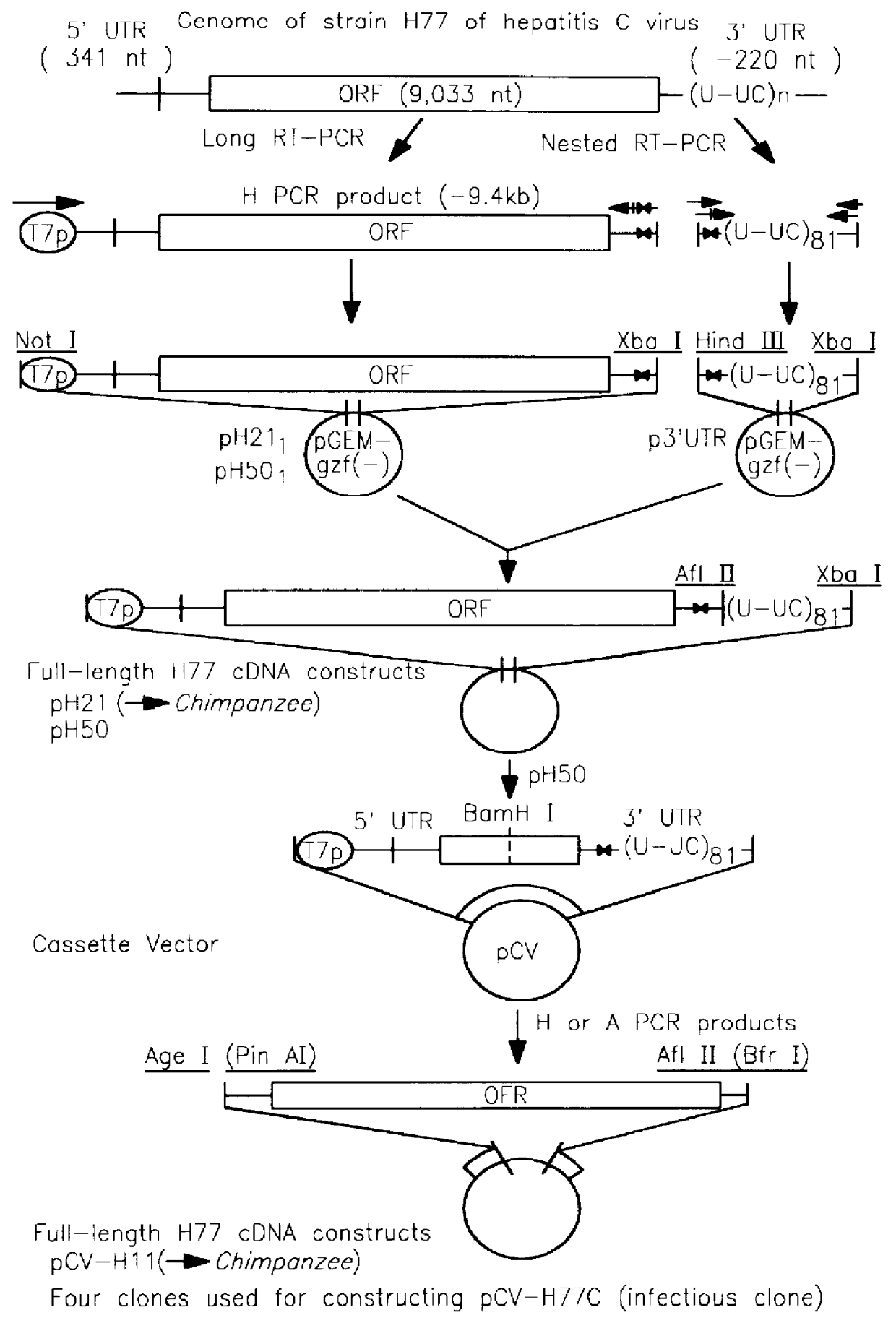
Cloned genomes of infectious hepatitis C viruses and uses thereof
InactiveUS6153421ADetermining effectSsRNA viruses positive-senseSugar derivativesNucleic acid sequencingGenome
The present invention discloses nucleic acid sequences which encode infectious hepatitis C viruses and the use of these sequences, and polypeptides encoded by all or part of these sequences, in the development of vaccines and diagnostics for HCV and in the development of screening assays for the identification of antiviral agents for HCV.
Owner:HEALTH & HUMAN SERVICES DEPT OF
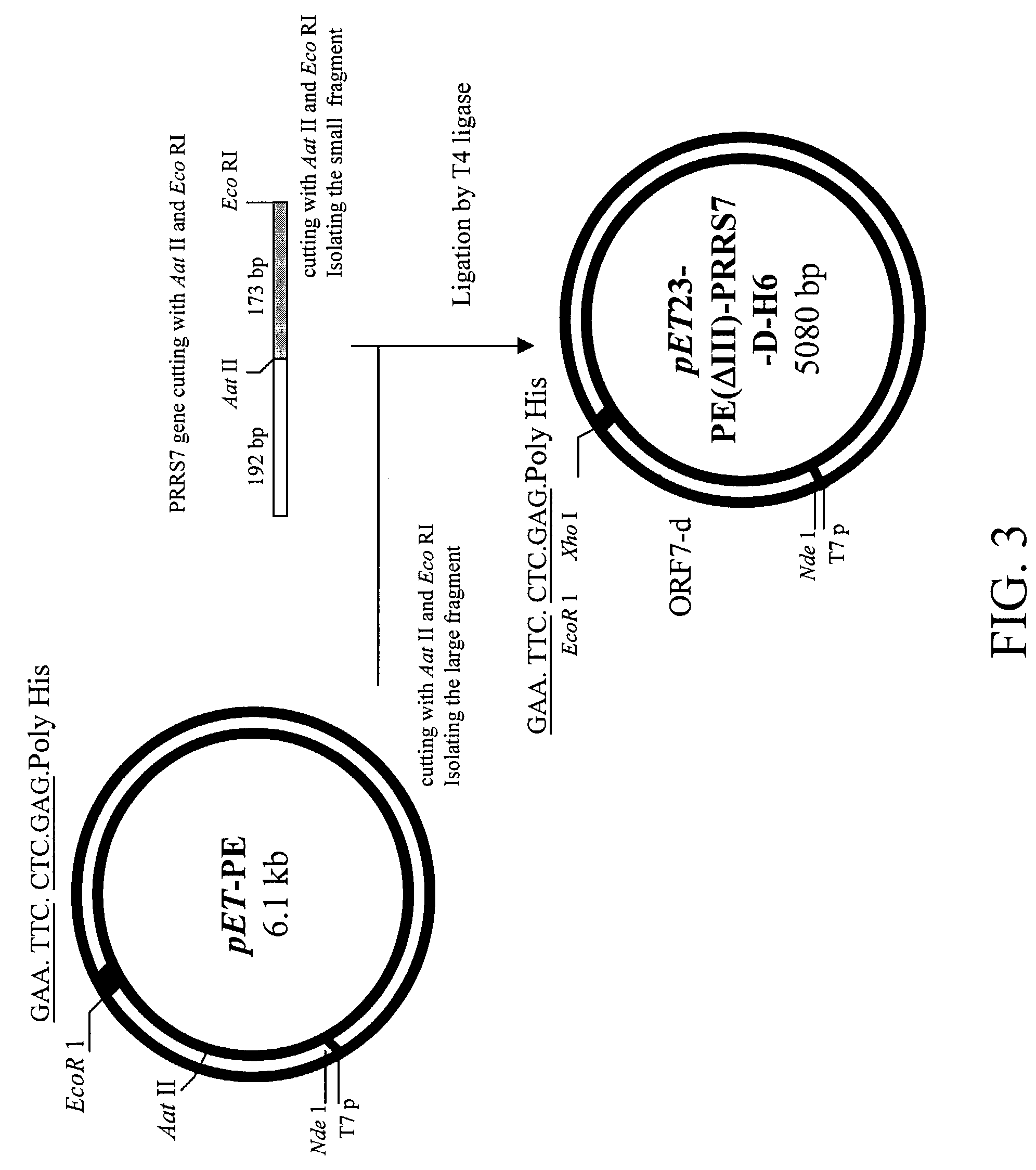
Infectious cDNA clones of porcine reproductive and respiratory syndrome virus and expression vectors thereof
InactiveUS6841364B2SsRNA viruses positive-senseSugar derivativesRespiratory syndrome virusGene mutation
Infectious cDNA clones of North American Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) are provided. Further provided are cDNA clones comprising genetic mutations. Also provided are vaccines comprising cDNAs, including genetically and immunologically marked vaccines for North American PRRSV.
Owner:PROTATEK INT
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com