Patents
Literature
1248 results about "Mass" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
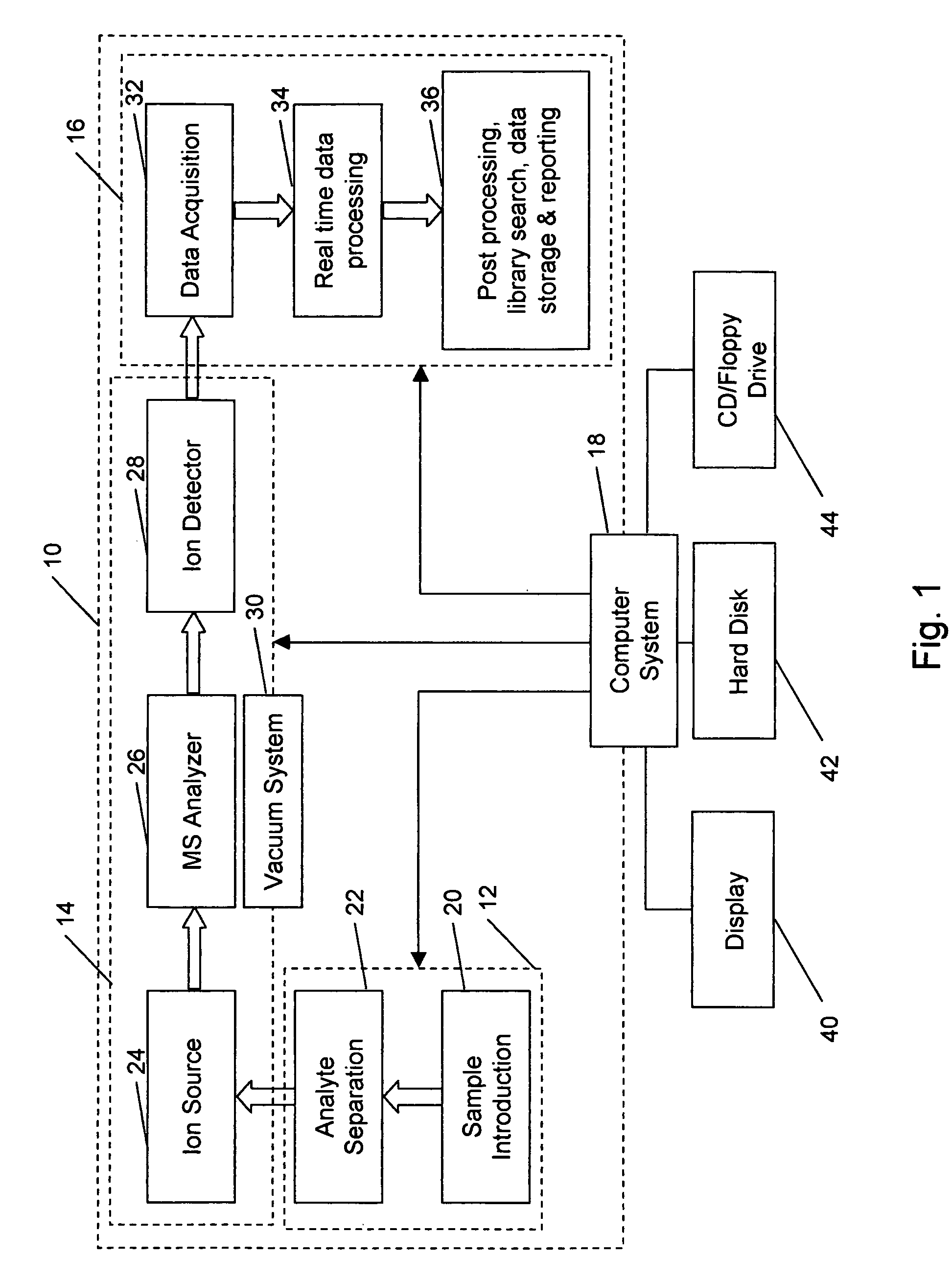
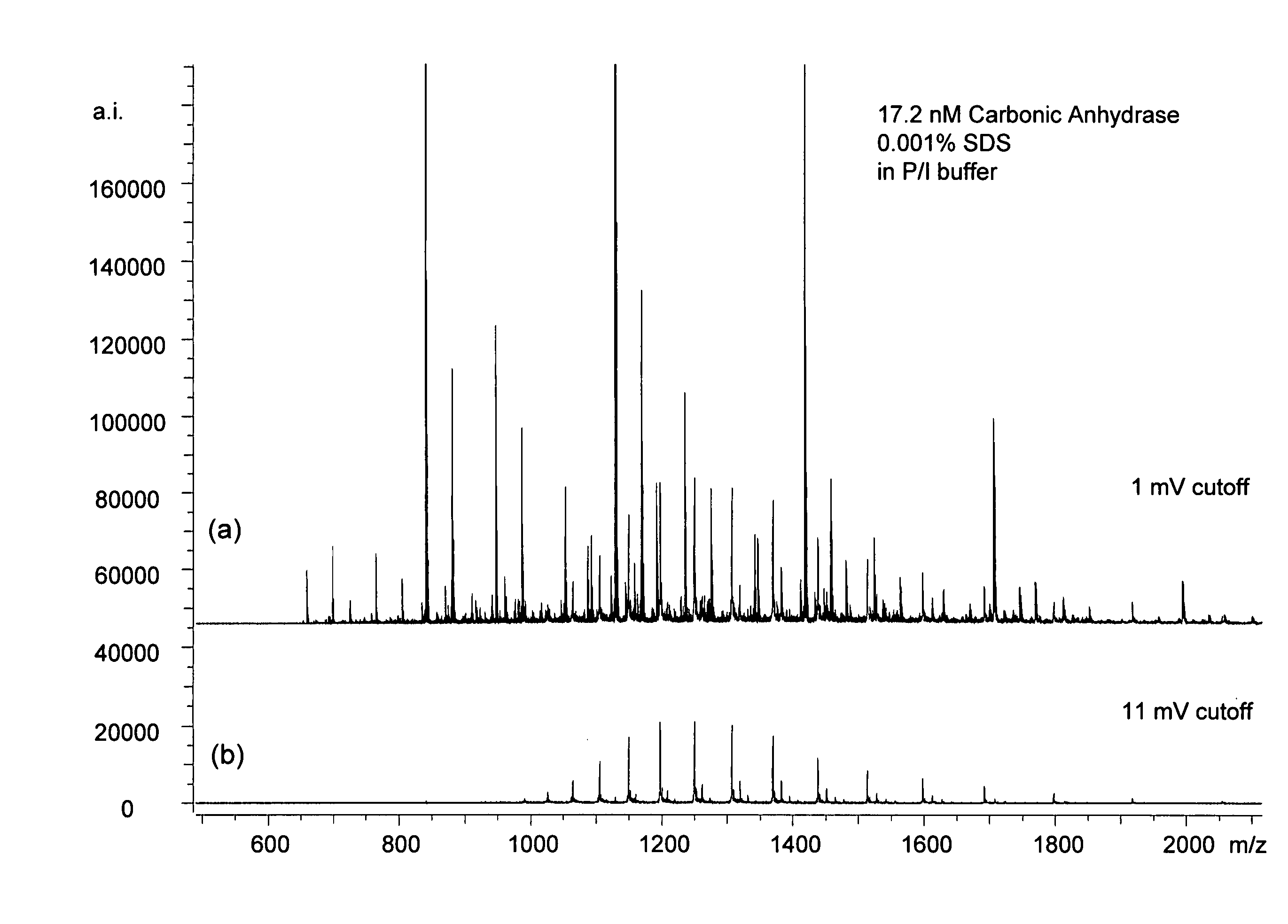
The mass recorded by a mass spectrometer can refer to different physical quantities depending on the characteristics of the instrument and the manner in which the mass spectrum is displayed.
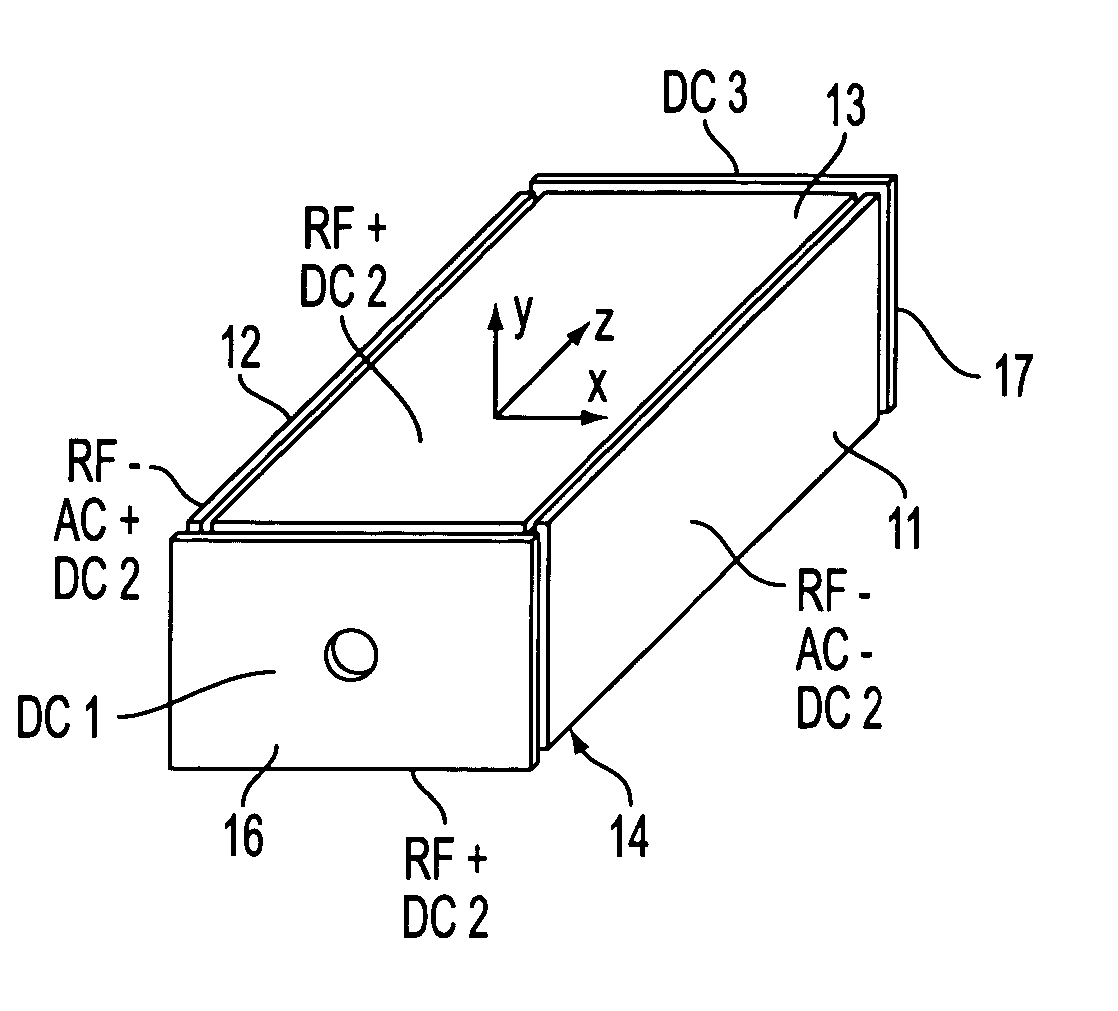
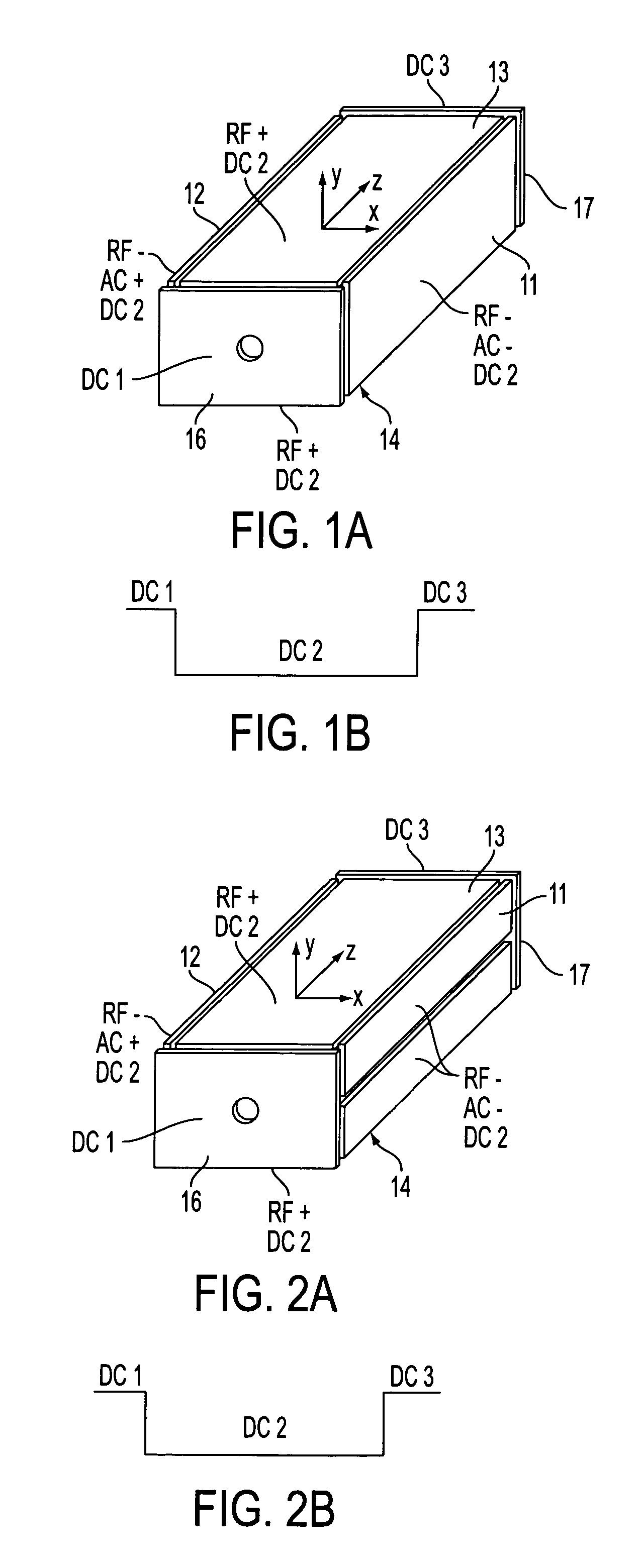
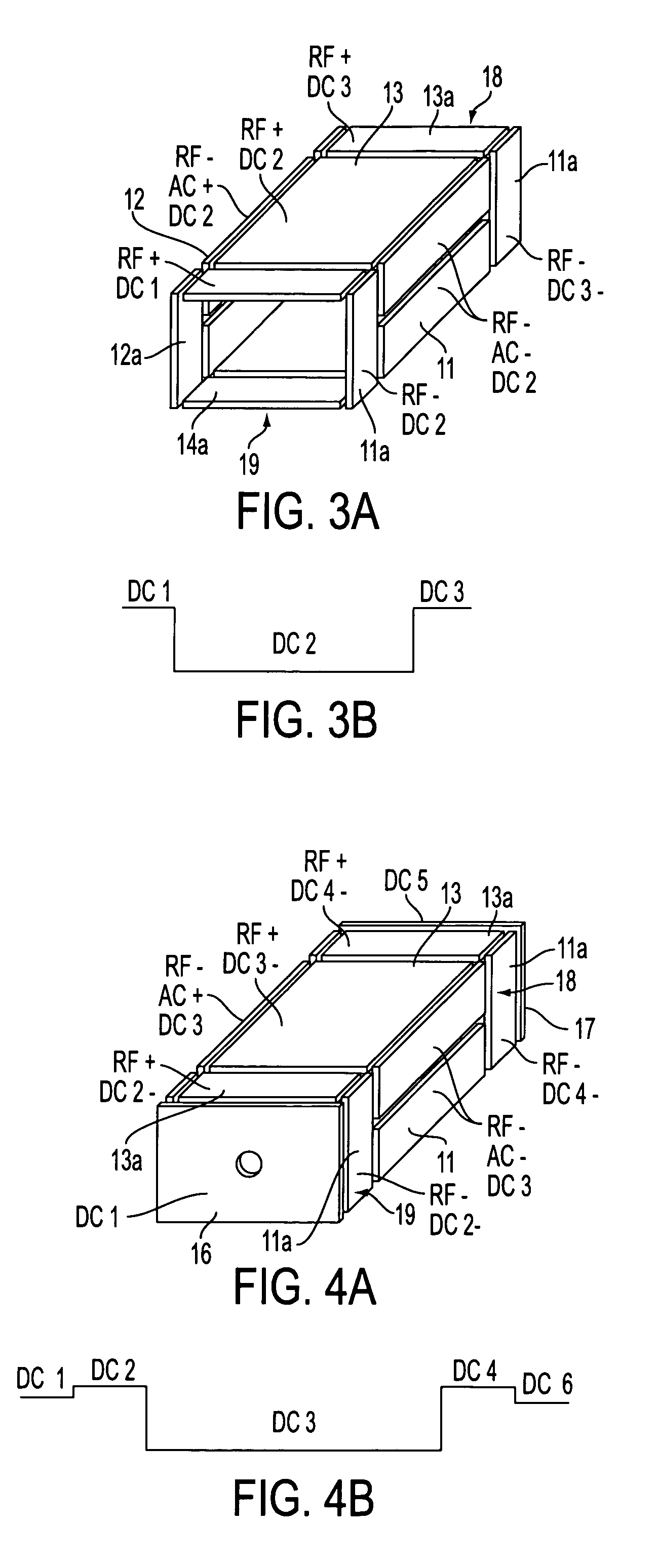
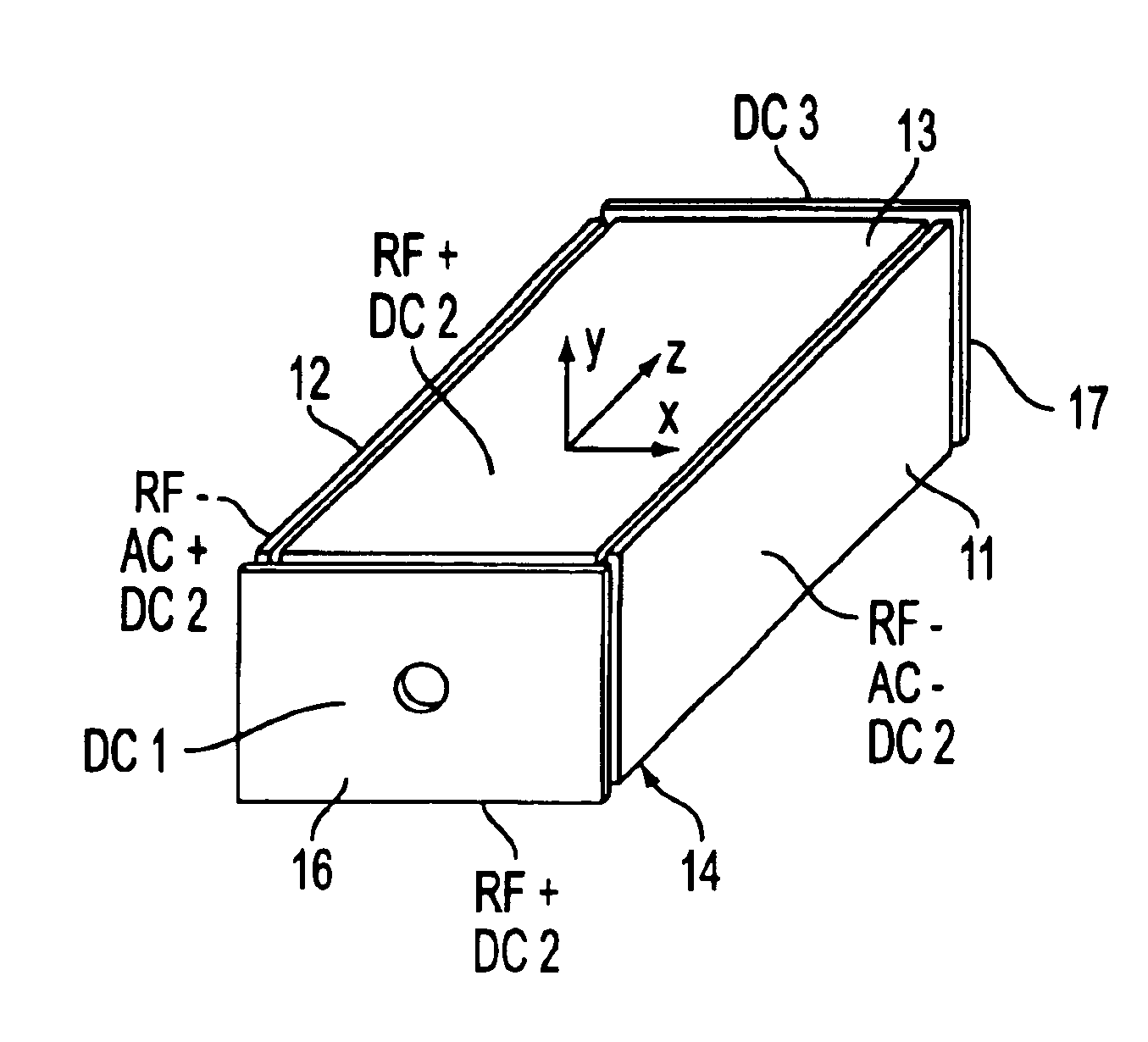
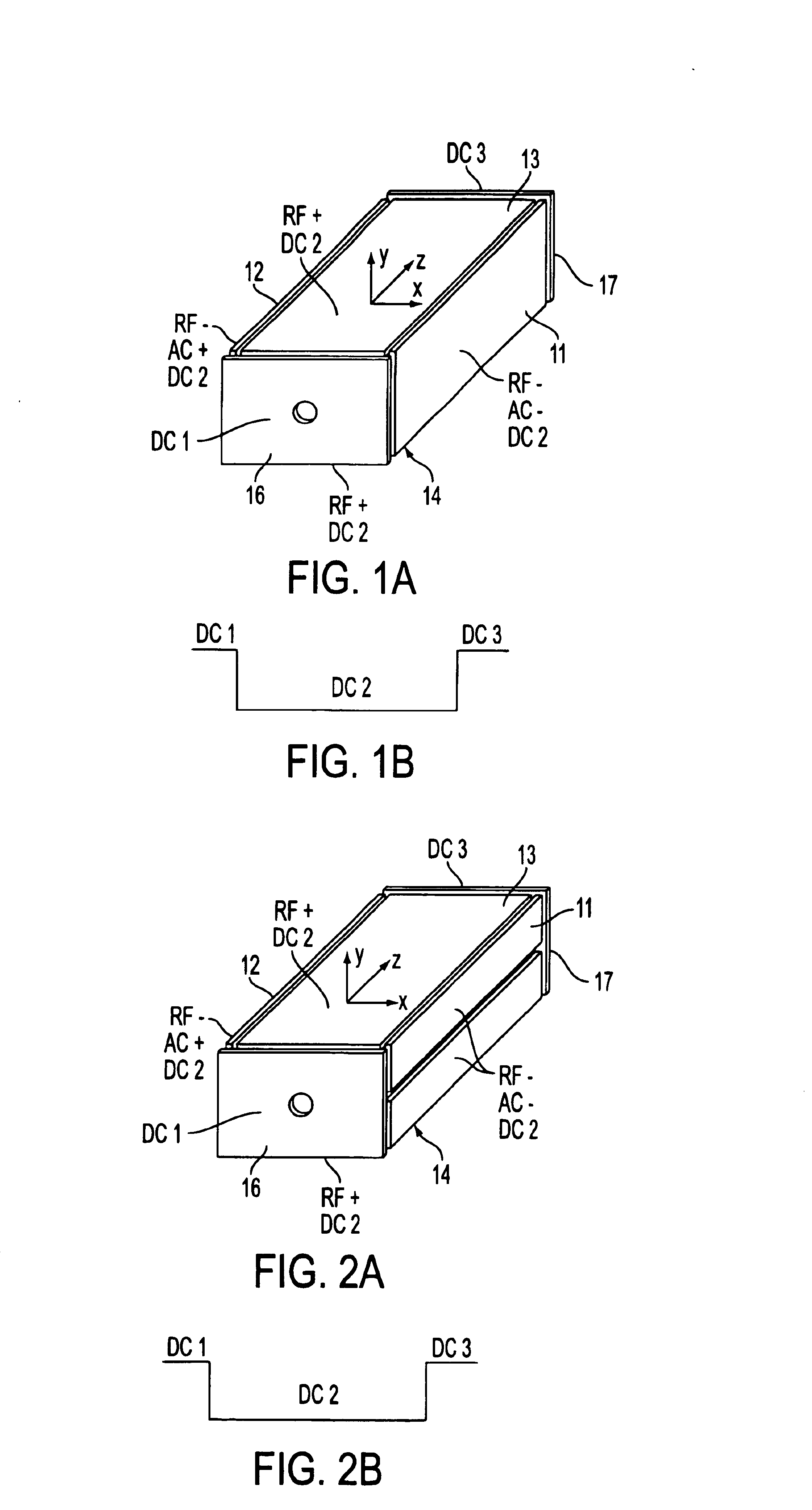
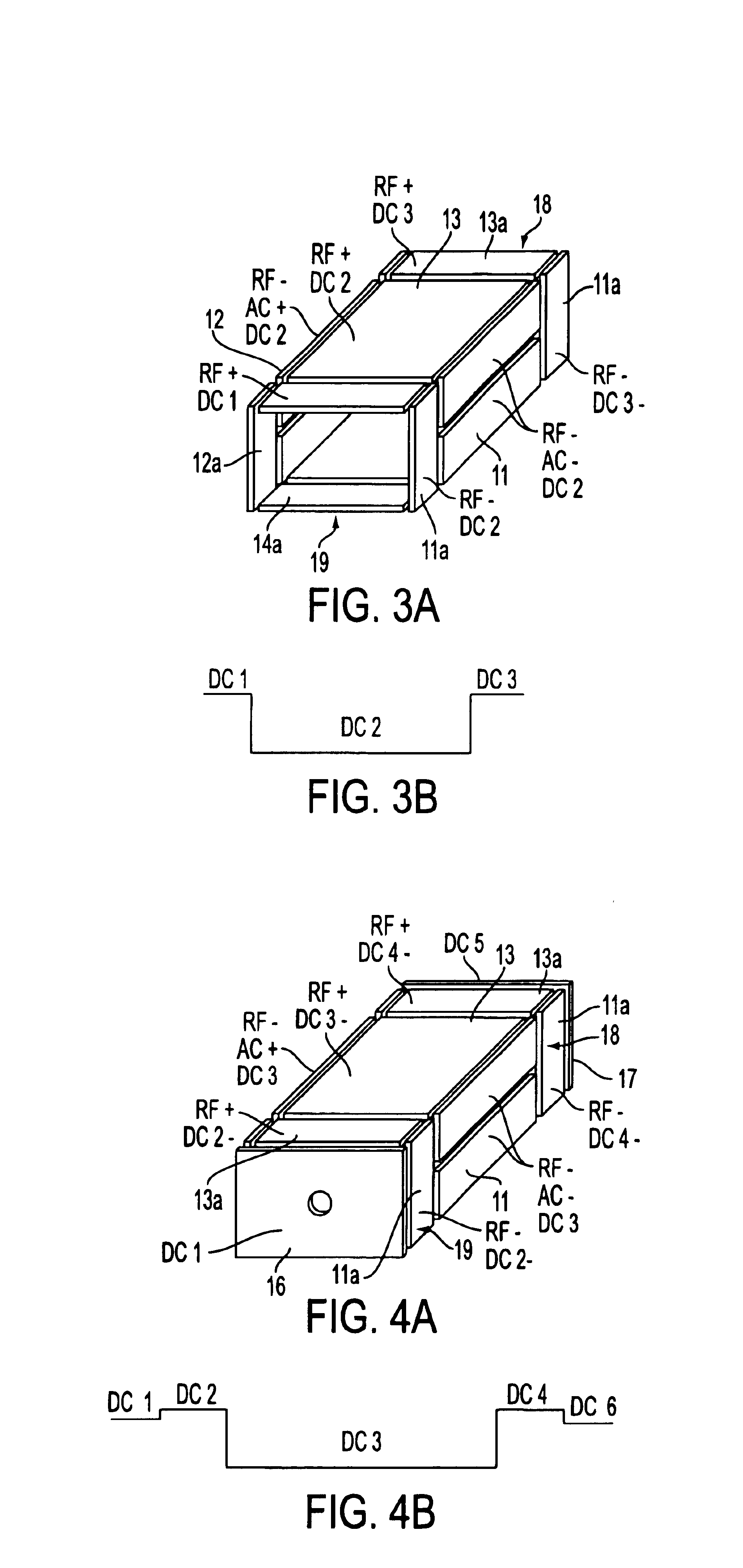
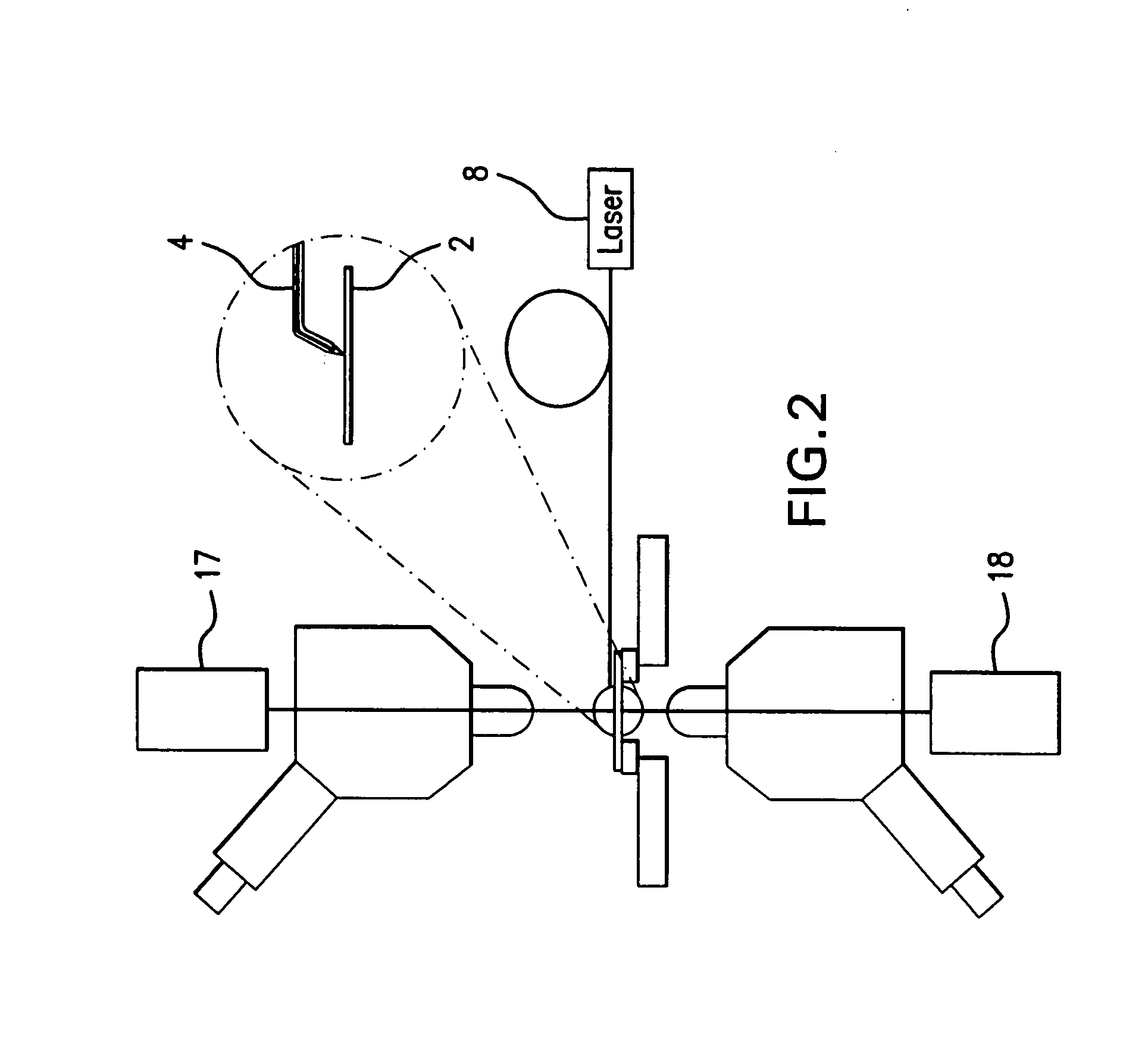
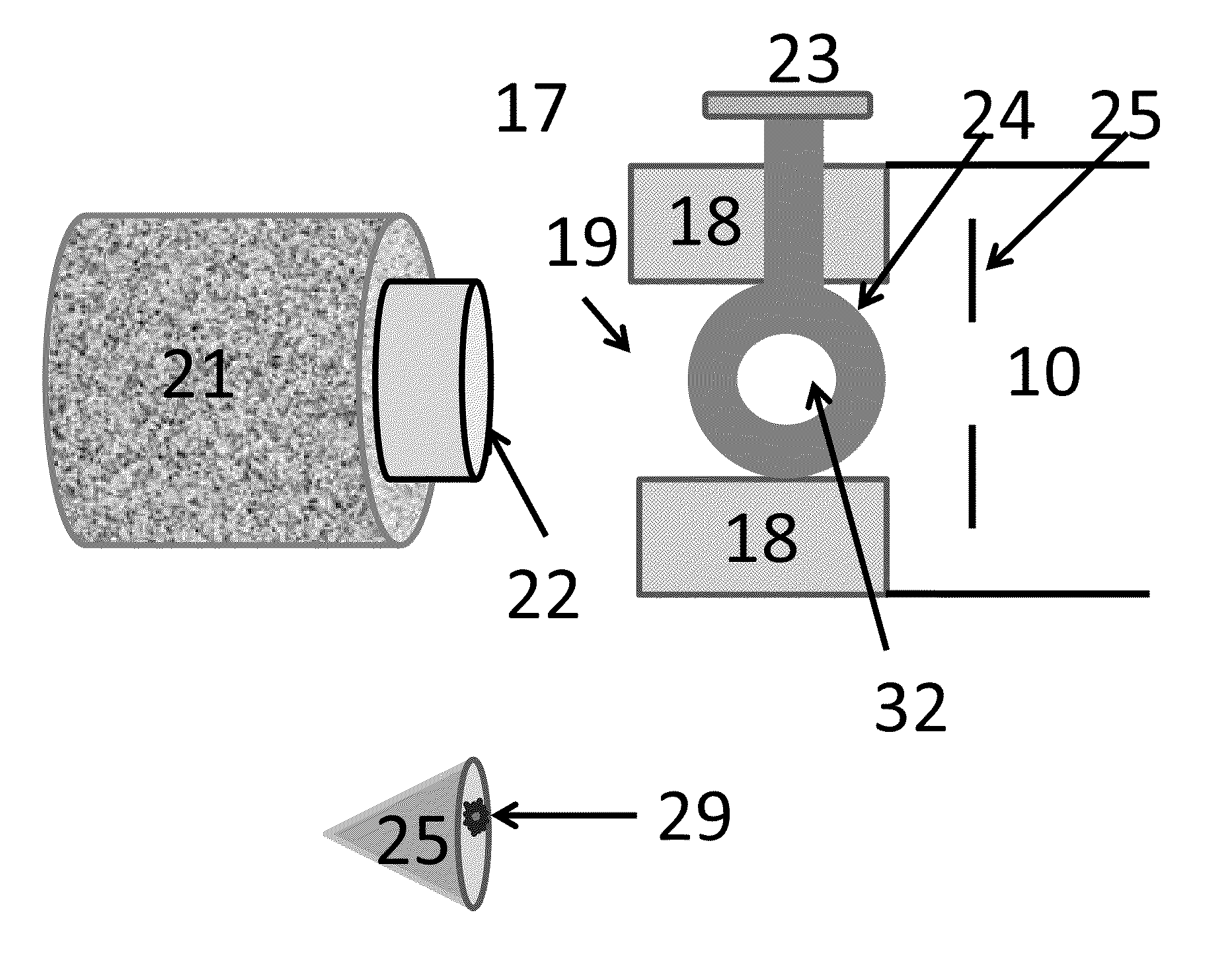
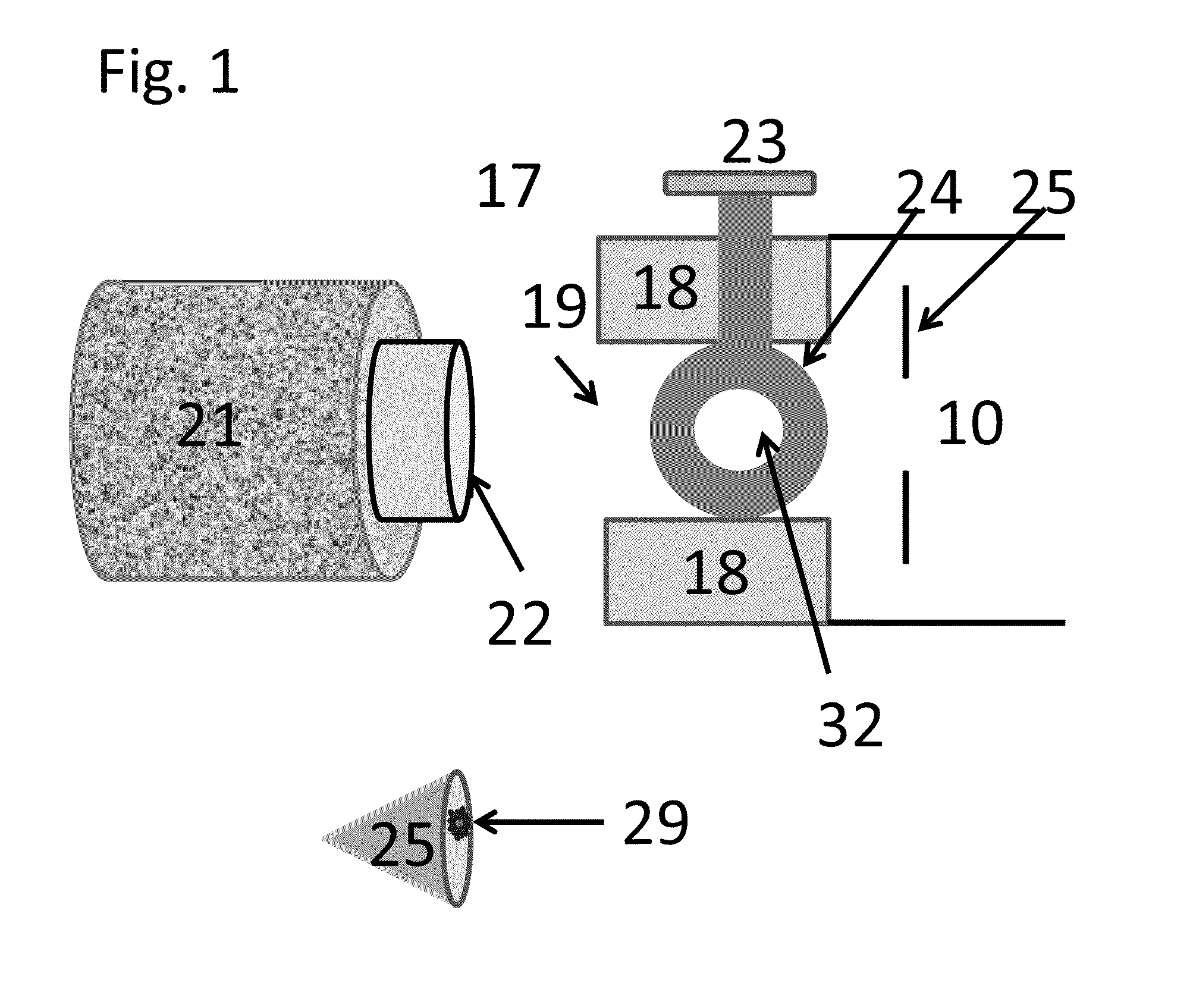
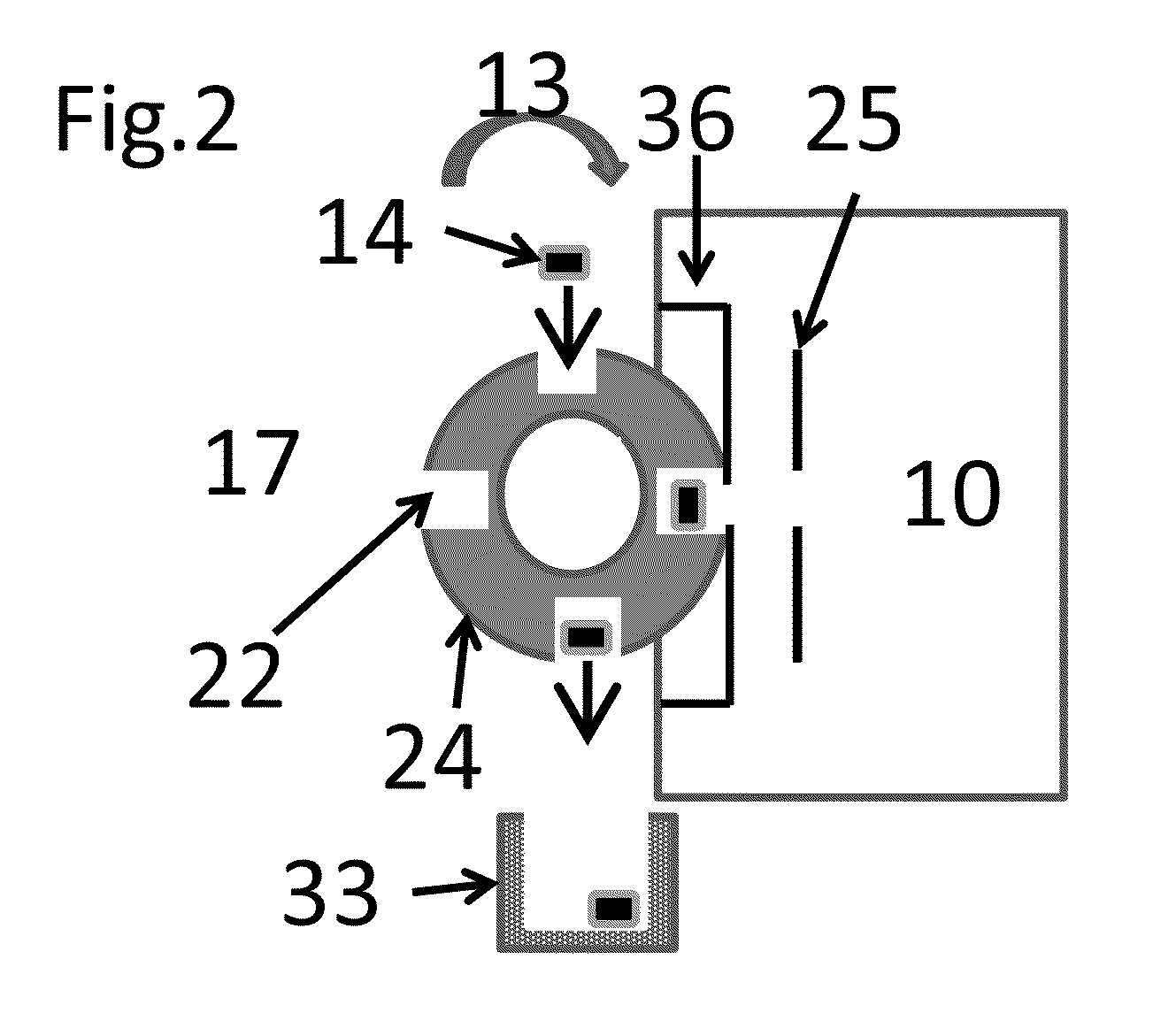
Rectilinear ion trap and mass analyzer system and method
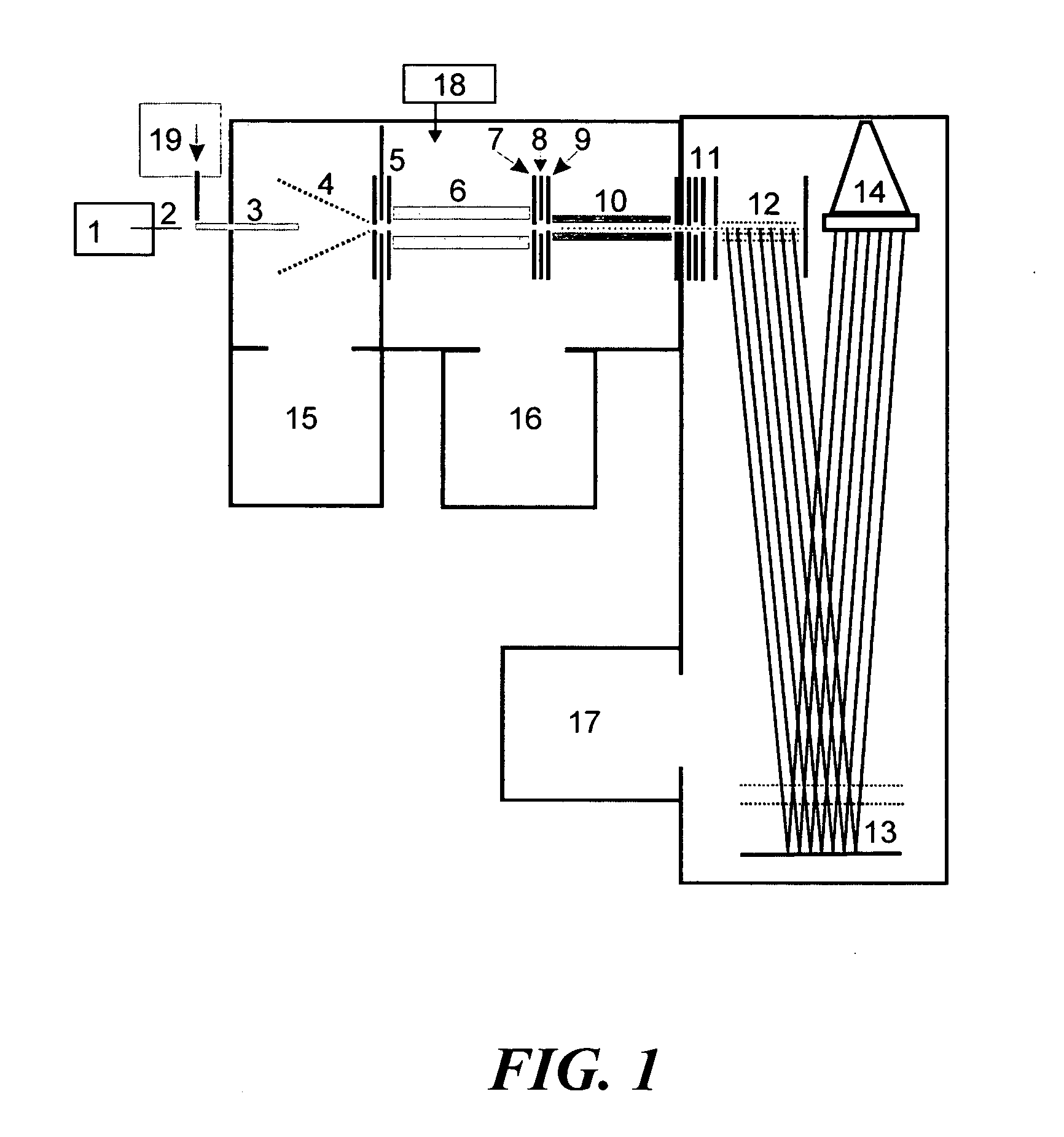
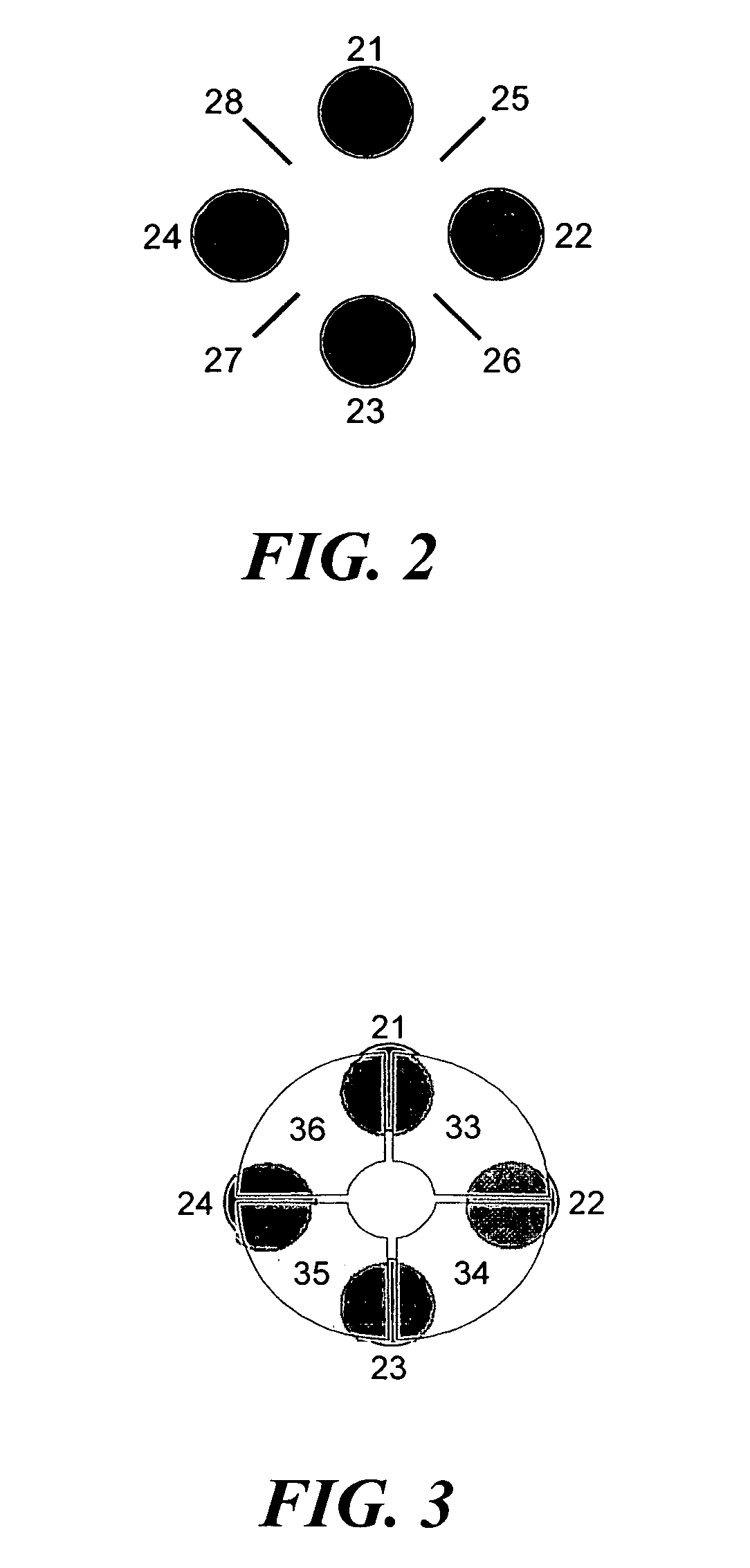
ActiveUS20040135080A1New and simple geometryImprove capture abilityStability-of-path spectrometersIsotope separationMass storageIon trap mass spectrometry
A new geometry ion trap and its use as a mass spectrometer is described. The ion traps can be combined linearly and in parallel to form systems for mass storage, analysis, fragmentation, separation, etc. of ions. The ion trap has a simple rectilinear geometry with a high trapping capacity. It can be operated to provide mass analysis in the mass-selective instability mode as well as the mass-selective stability mode. Arrays of multiple ion traps allow combinations of multiple gas-phase processes to be applied to the trapped ions to achieve high sensitivity, high selectivity and / or higher throughput in the analysis of ions.
Owner:PURDUE RES FOUND INC
Rectilinear ion trap and mass analyzer system and method
InactiveUS6838666B2New and simple geometryImprove capture abilityStability-of-path spectrometersIsotope separationMass storageIon trap mass spectrometry
A new geometry ion trap and its use as a mass spectrometer is described. The ion traps can be combined linearly and in parallel to form systems for mass storage, analysis, fragmentation, separation, etc. of ions. The ion trap has a simple rectilinear geometry with a high trapping capacity. It can be operated to provide mass analysis in the mass-selective instability mode as well as the mass-selective stability mode. Arrays of multiple ion traps allow combinations of multiple gas-phase processes to be applied to the trapped ions to achieve high sensitivity, high selectivity and / or higher throughput in the analysis of ions.
Owner:PURDUE RES FOUND INC
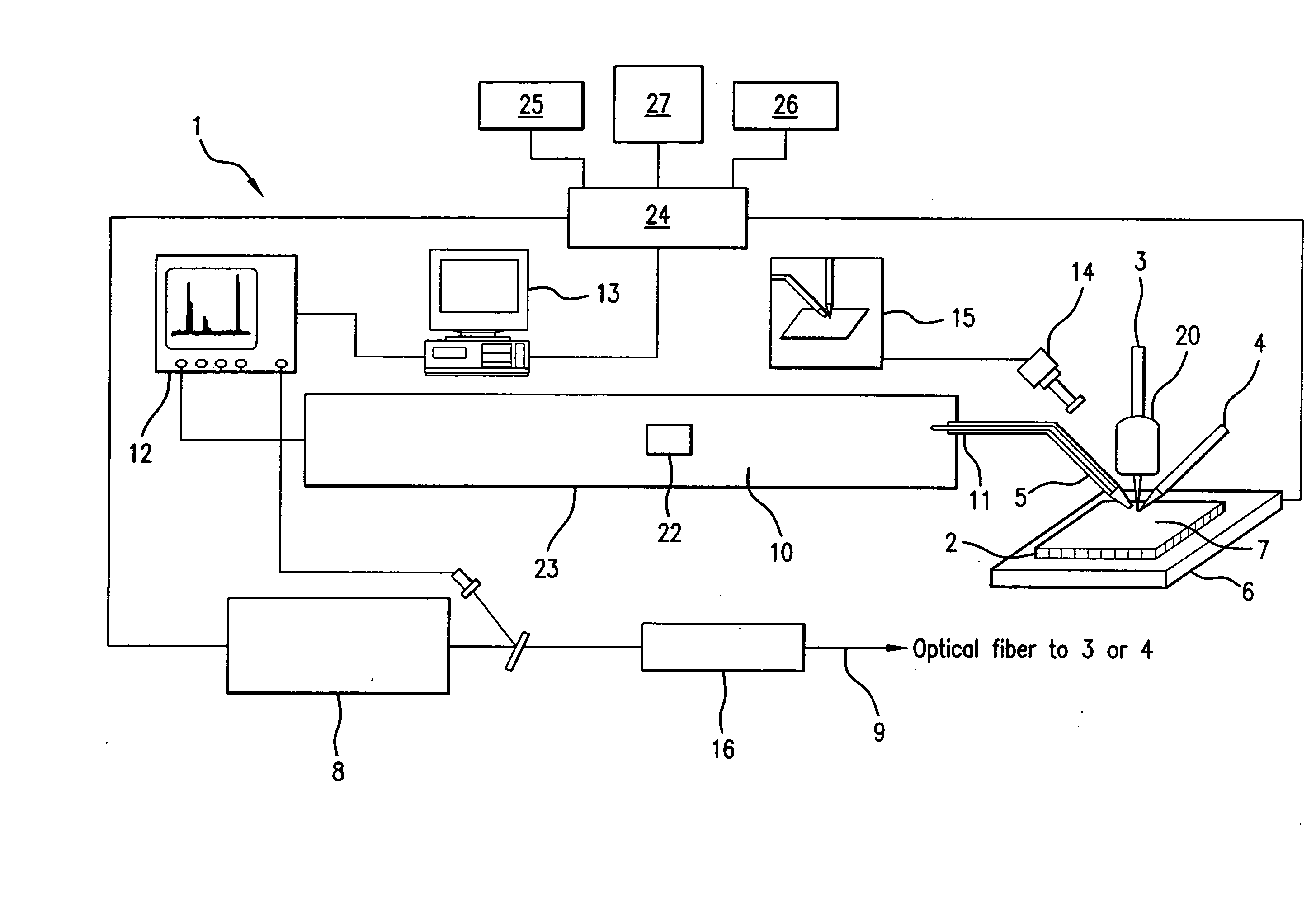
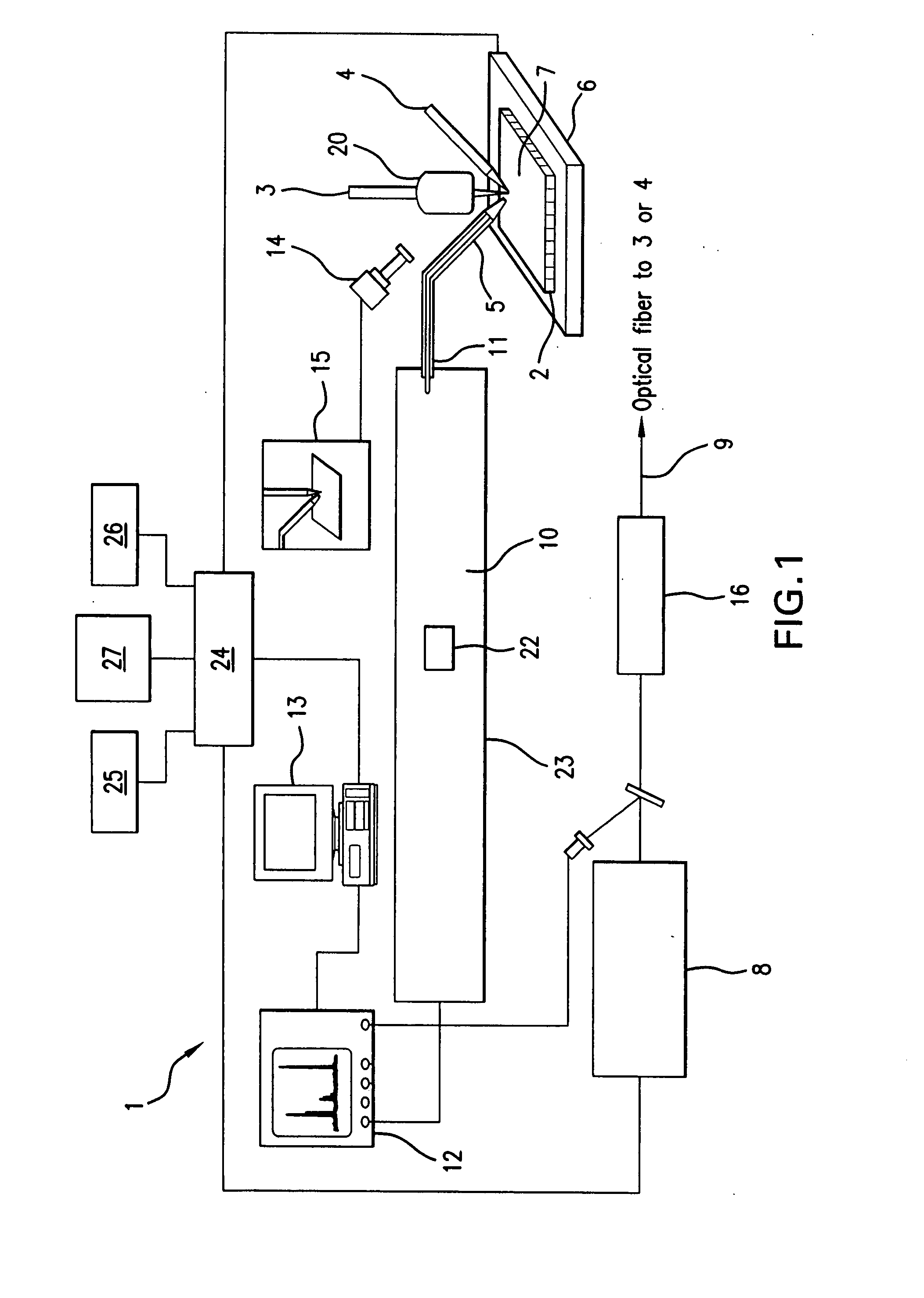
Protein Microscope
A system and method for analyzing and imaging a sample containing molecules of interest combines modified MALDI mass spectrometer and SNOM devices and techniques, and includes: (A) an atmospheric-pressure or near-atmospheric-pressure ionization region; (B) a sample holder for holding the sample; (C) a laser for illuminating said sample; (D) a mass spectrometer having at least one evacuated vacuum chamber; (E) an atmospheric pressure interface connecting said ionization region and said mass spectrometer; (F) a scanning near-field optical microscopy instrument comprising a near-field probe for scanning the sample; a vacuum capillary nozzle for sucking in particles which are desorbed by said laser, the nozzle being connected to an inlet orifice of said atmospheric pressure interface; a scanner platform connected to the sample holder, the platform being movable to a distance within a near-field distance of the probe; and a controller for maintaining distance information about a current distance between said probe and said sample; (G) a recording device for recording topography and mass spectrum measurements made during scanning of the sample with the near-field probe; (H) a plotting device for plotting said topography and mass spectrum measurements as separate x-y mappings; and (I) an imaging device for providing images of the x-y mappings.
Owner:GEORGE WASHINGTON UNIVERSITY
Mass spectrometer
ActiveUS6992283B2Increase speedLow ion mobilityTime-of-flight spectrometersMaterial analysis by electric/magnetic meansMass analyzerSpectrometer
A mass spectrometer is disclosed wherein ions are passed through an ion mobility separator and are then mass analysed by a Time of Flight mass analyzer. Multiple sets of mass spectral data are obtained which are then post-processed so that mass spectral data relating to ions having undesired charge state(s) is filtered out. The resultant mass spectrum comprises ions having a desired charge state.
Owner:MICROMASS UK LTD
Methods for accurate component intensity extraction from separations-mass spectrometry data
InactiveUS20050255606A1Accurate representationBiological testingRecognisation of pattern in signalsMass Spectrometry-Mass SpectrometryUltimate tensile strength
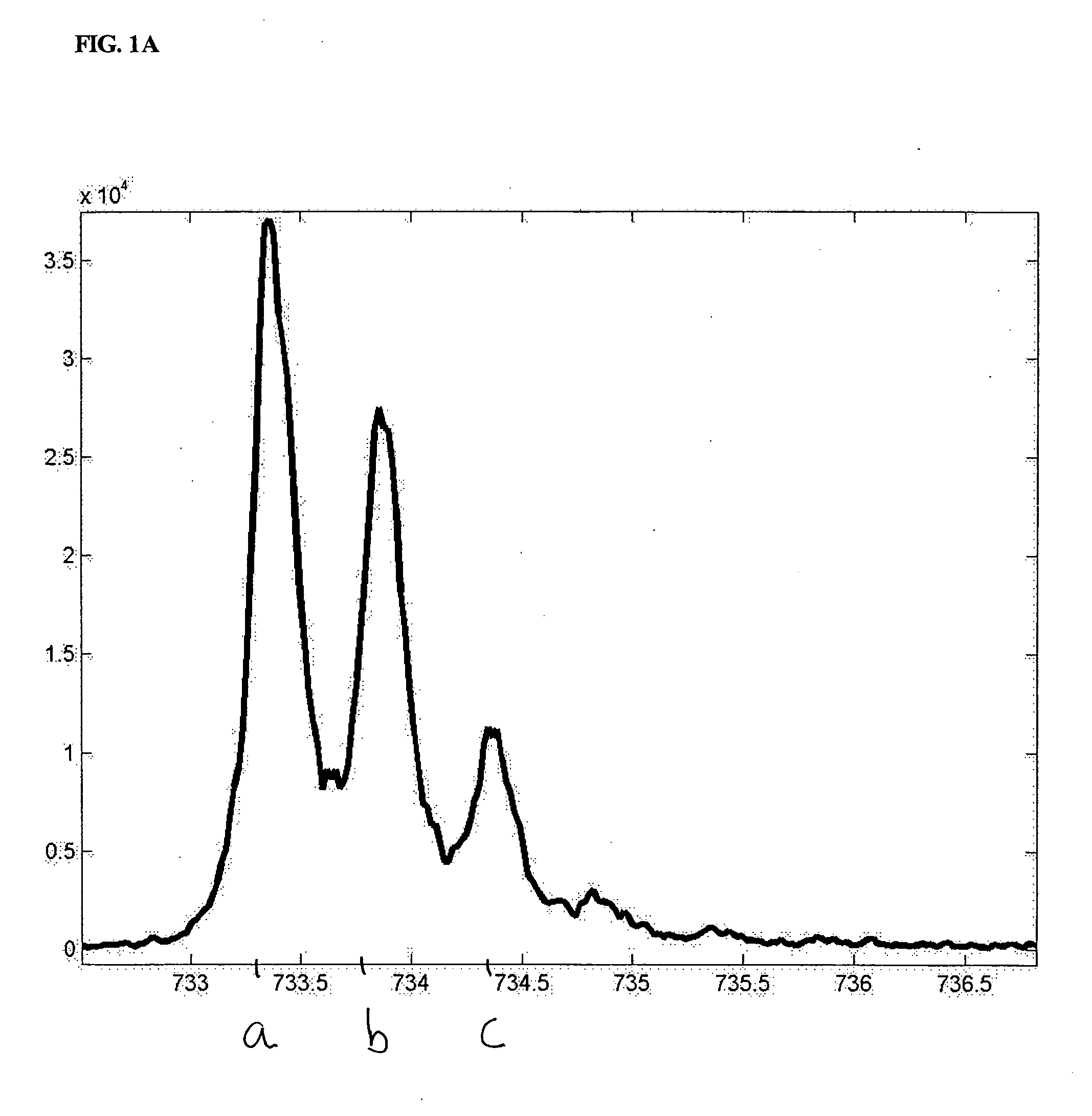
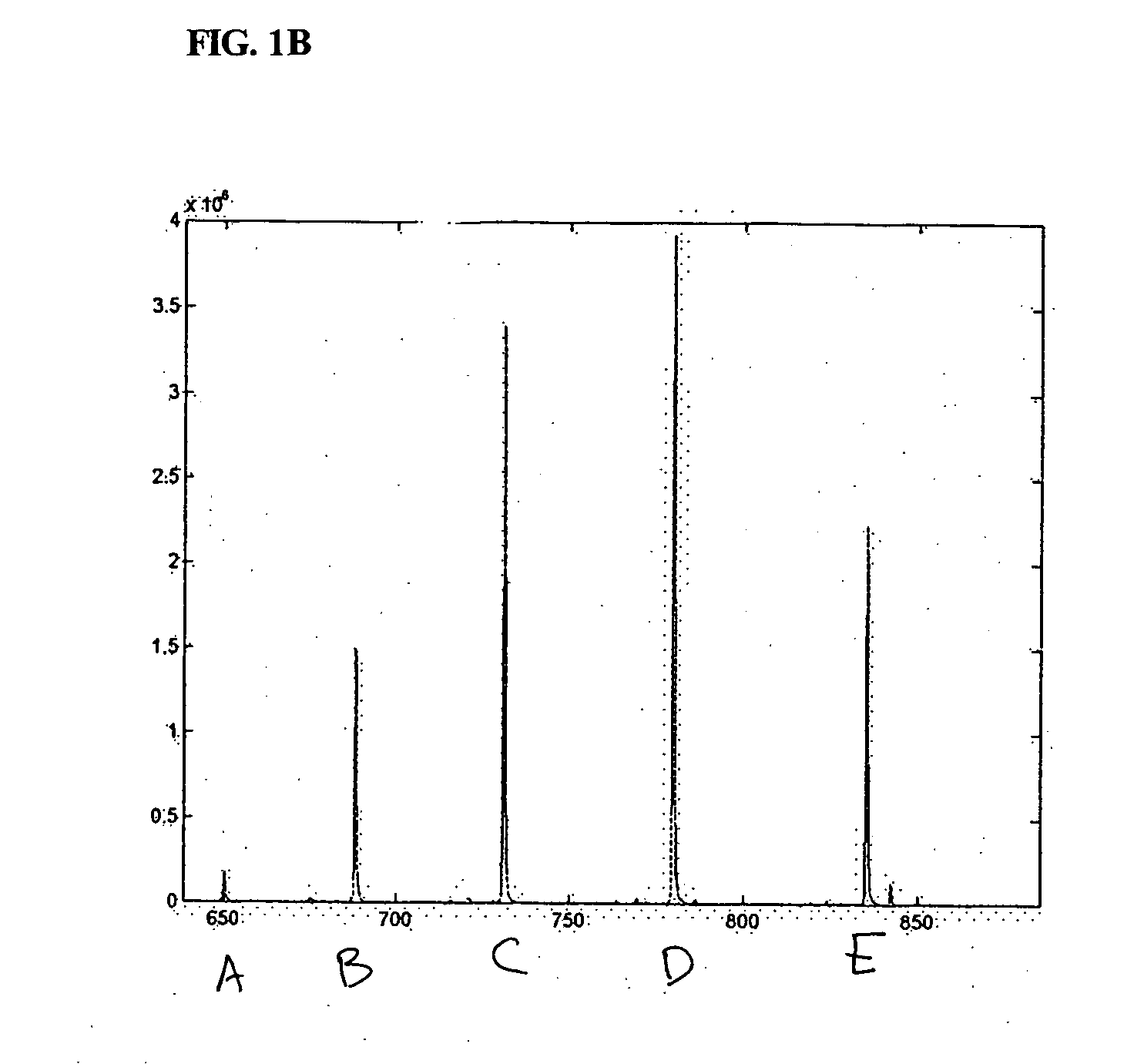
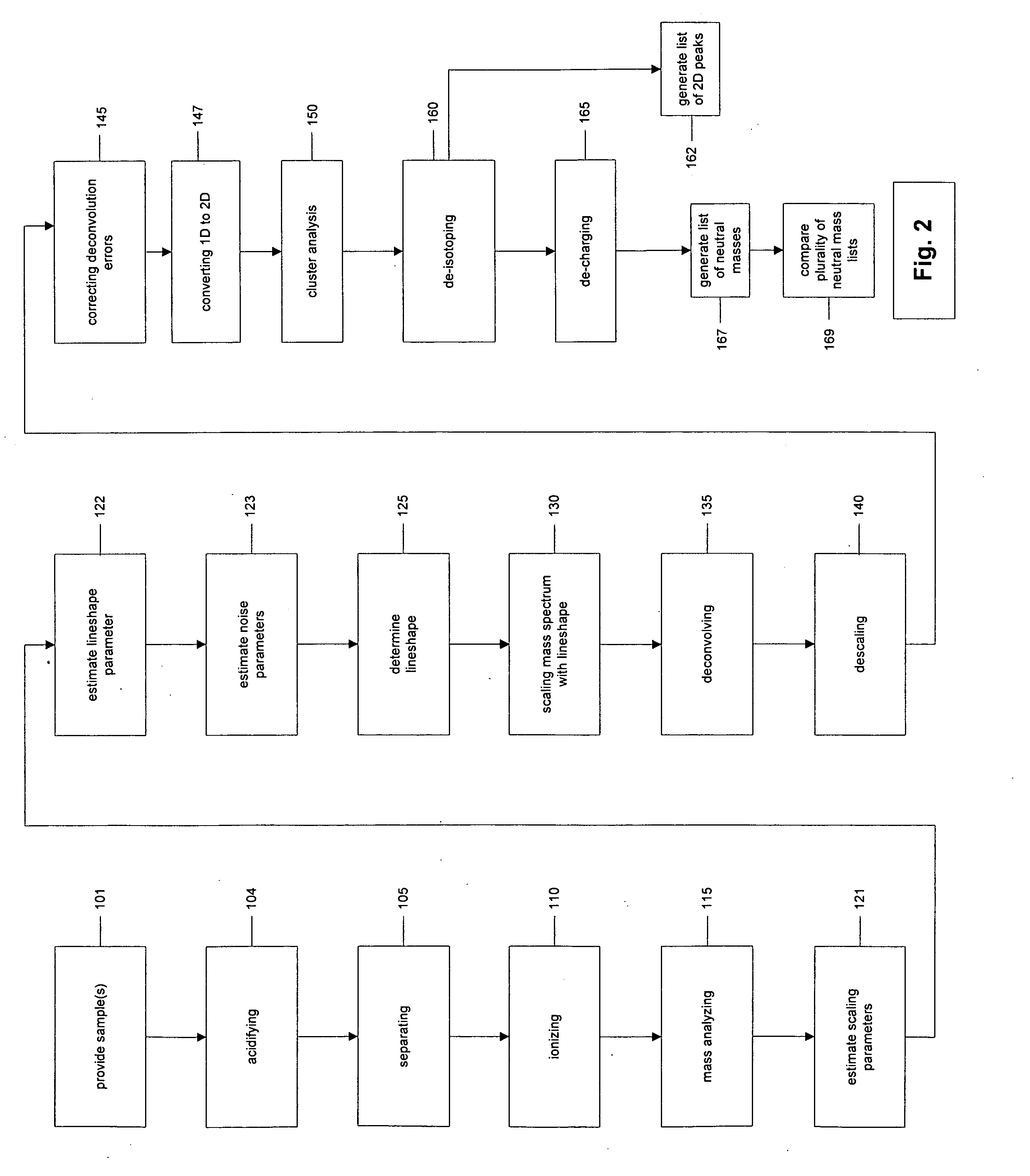
The present invention discloses methods for deconvolving and converting 1D mass spectra to 2D mass spectrum in order to obtain migration time centers and total intensities of the neutral mass envelopes of 2D spectra. The present invention also discloses devices that include a preparation / separation unit coupled to a mass spectrometer unit, and a computer unit capable of deconvolving mass spectra and calculating neutral mass envelopes.
Owner:V&M TCA LP +1
Particle analysis system and method
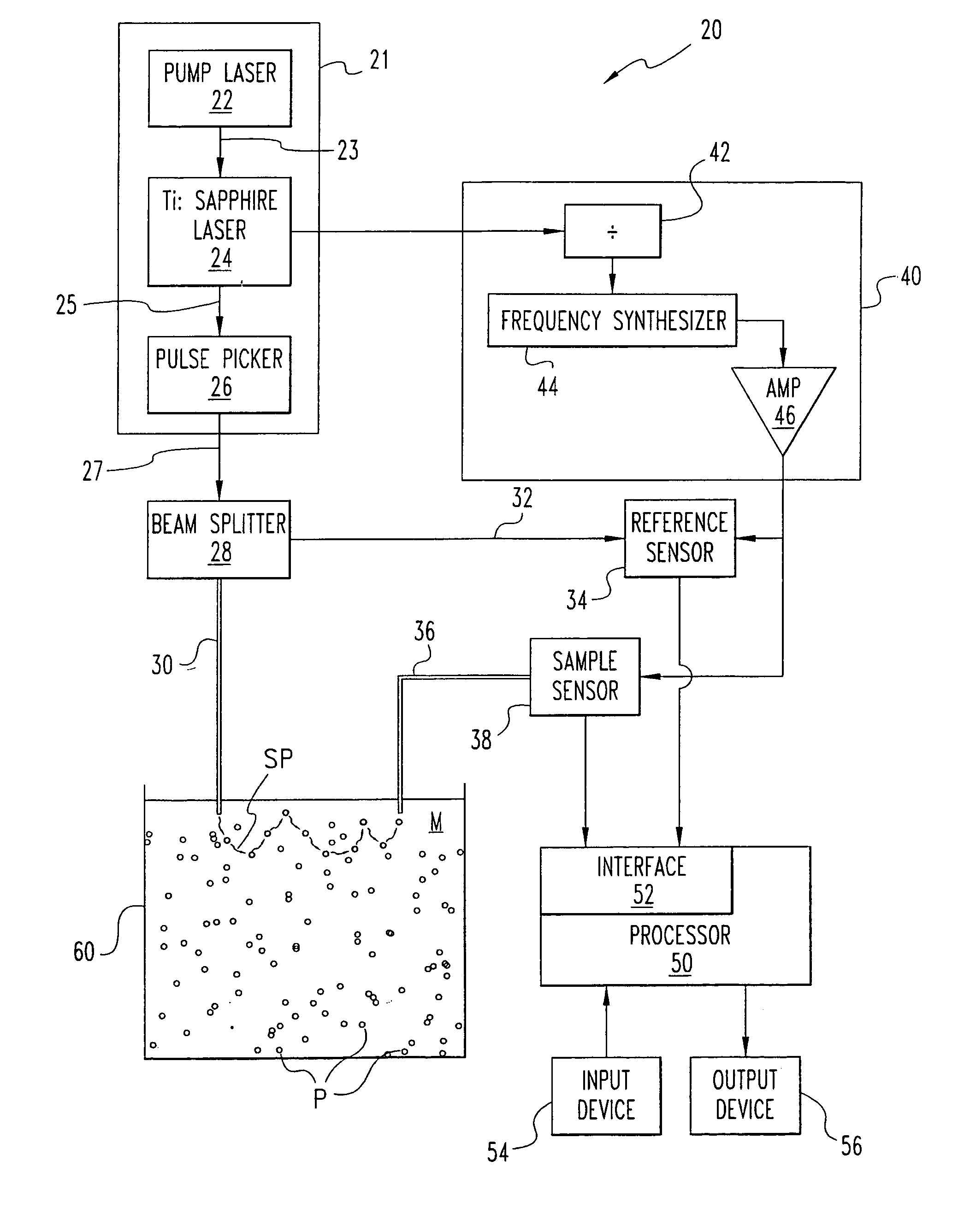
InactiveUS7187441B1Scattering properties measurementsParticle size analysisScattered lightLight source
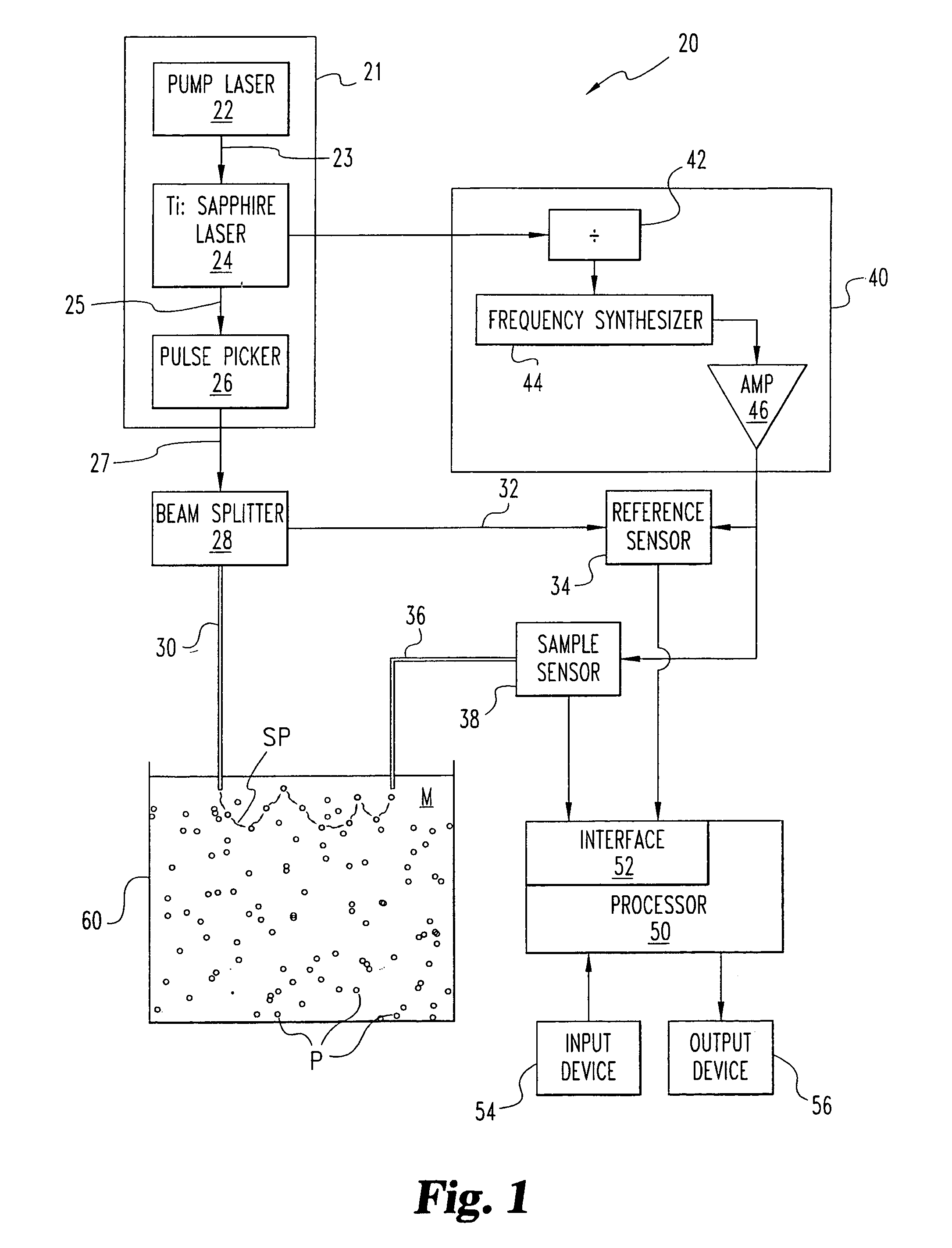
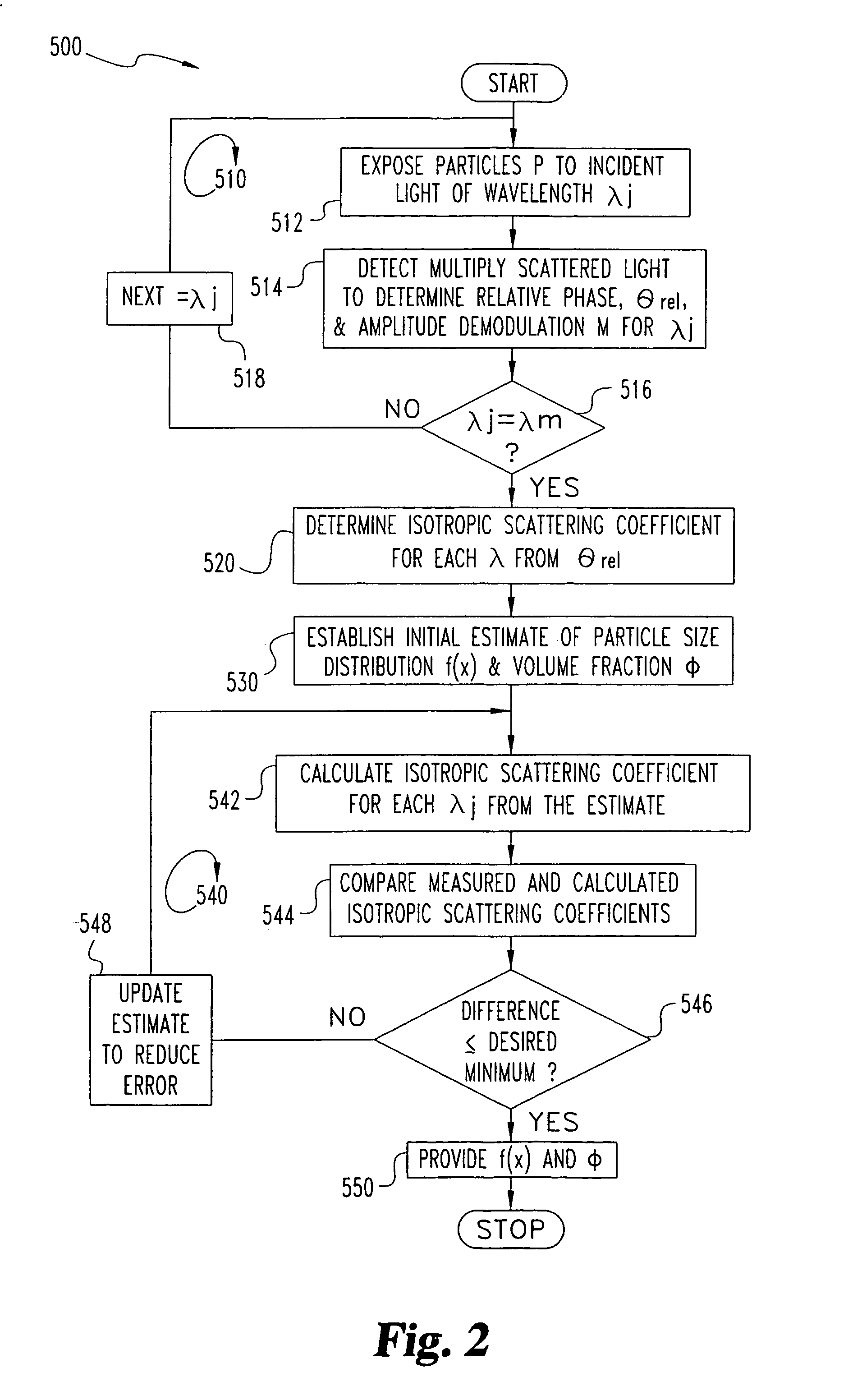
A system (20) and method are disclosed for the self-calibrating, on-line determination of size distribution f(x) and volume fraction φ of a number of particles (P) dispersed in a medium (M) by detecting one or more propagation characteristics of multiply scattered light from the particles (P). The multiply scattered light is re-emitted in response to exposure to a light source (21) configured to provide light at selected wavelengths. The determination includes calculating the isotropic scattering and absorption coefficients for the particles (P) by comparing the incident and detected light to determine a measurement corresponding to the propagation time through the scattering medium (M), and iteratively estimating the size distribution f(x) and volume fraction φ as a function of the coefficients for each of the wavelengths. An estimation approach based on an expected form of the distribution and the mass of the particles is also disclosed. Furthermore, techniques to determine a particle structure factor indicative of particle-to-particle interactions which vary with particle concentration and influence light scattering at high concentrations is disclosed.
Owner:TEXAS A&M UNIVERSITY
Nanostructure-initiator mass spectrometry
InactiveUS20080128608A1Increase horizontal resolutionHigh sensitivityParticle separator tubesLayered productsMass Spectrometry-Mass SpectrometrySpatial mapping
A substrate for use in providing an ionized target comprising a structured substrate has a plurality of recesses, at least a portion of the plurality of recesses containing an initiator, the substrate being capable of having a target loaded on it. In one methods, irradiation of the substrate can cause the initiator to restructure, releasing it from the recesses and thereby desorbing and ionizing the target. The target so desorbed and ionized can be detected by mass analyzers. The mass of the targets at a given point on the surface can be recorded to provide a spatial mapping of the targets on the surface.
Owner:THE SCRIPPS RES INST
Methods and apparatus for reducing artifacts in mass spectrometers
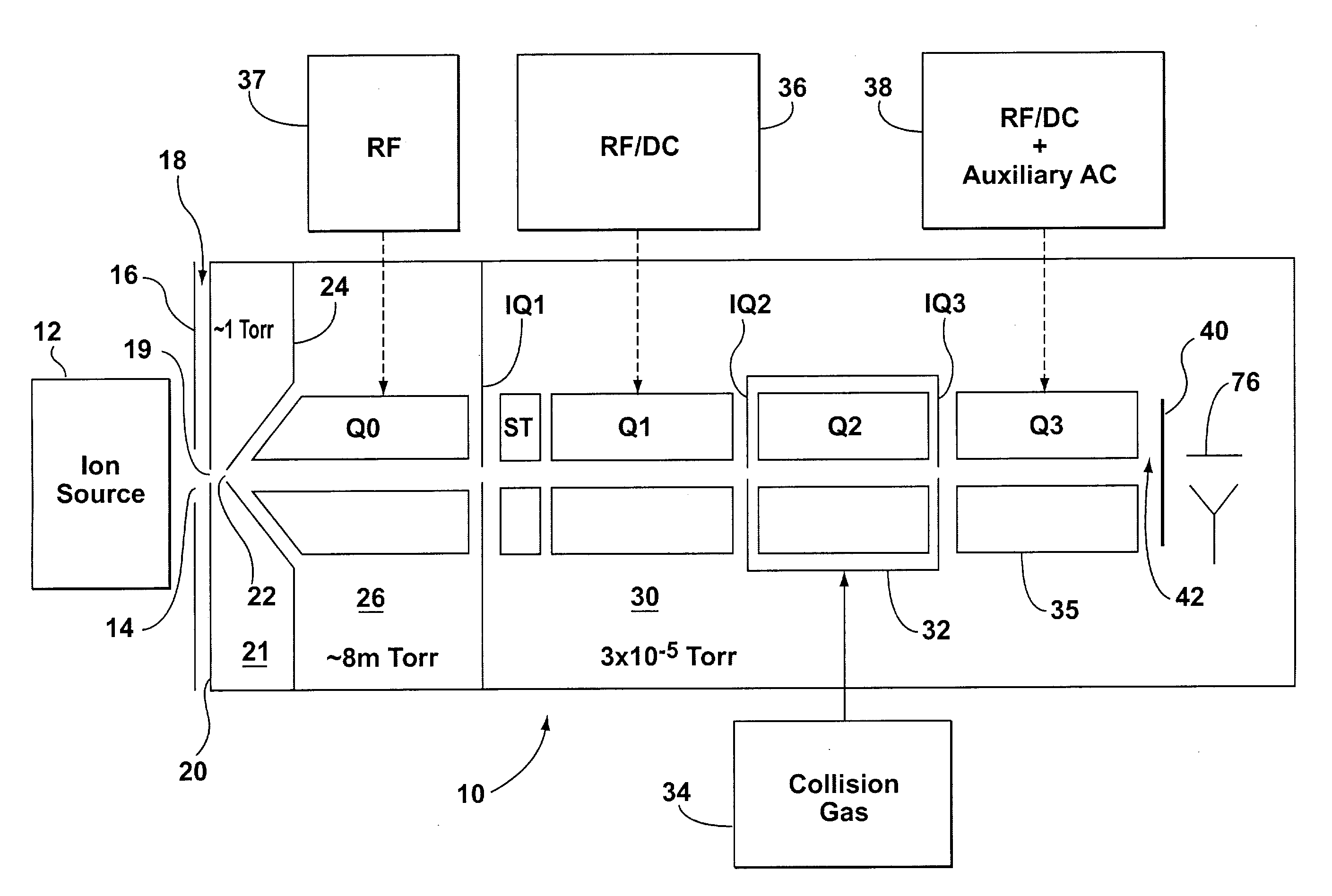
InactiveUS20040011956A1Excellent characteristicsImprove propertiesStability-of-path spectrometersMaterial analysis by electric/magnetic meansVoltage gradientMass analysis
The invention solves the problem of artifact ghost peaks which can sometimes arise in mass spectrometers that employ a quadrupole rod set for both trapping and mass analyzing the trapped ions. The problem arises as a result of randomly distributed voltage gradients along the length of the rods. Three solutions are presented. The first approach involves improving the conduction characteristics of the rod sets. The second approach involves the application of at least one continuous axial DC field to the trapping quadrupole rod set in order to urge ions towards a pre-determined region of the trap, thereby avoiding voltage gradients. The third approach involves the application of one or more discrete axial fields to create one or more potential barriers along the axial dimension of the trap (in addition to the barriers used to initially trap the ions). These barriers prevent ions of differing voltage gradients from equilibrating with one another.
Owner:MDS CO LTD +2
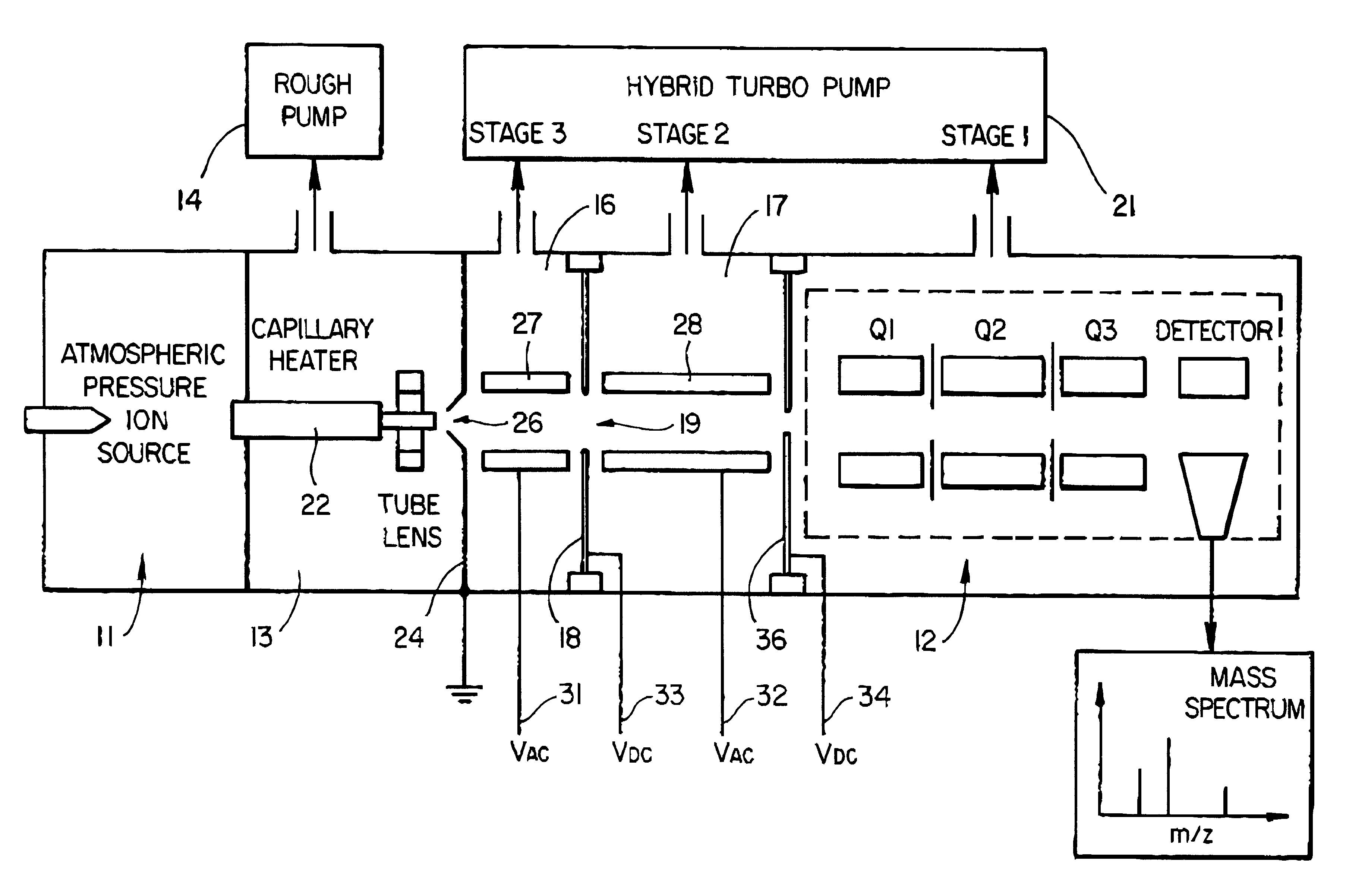
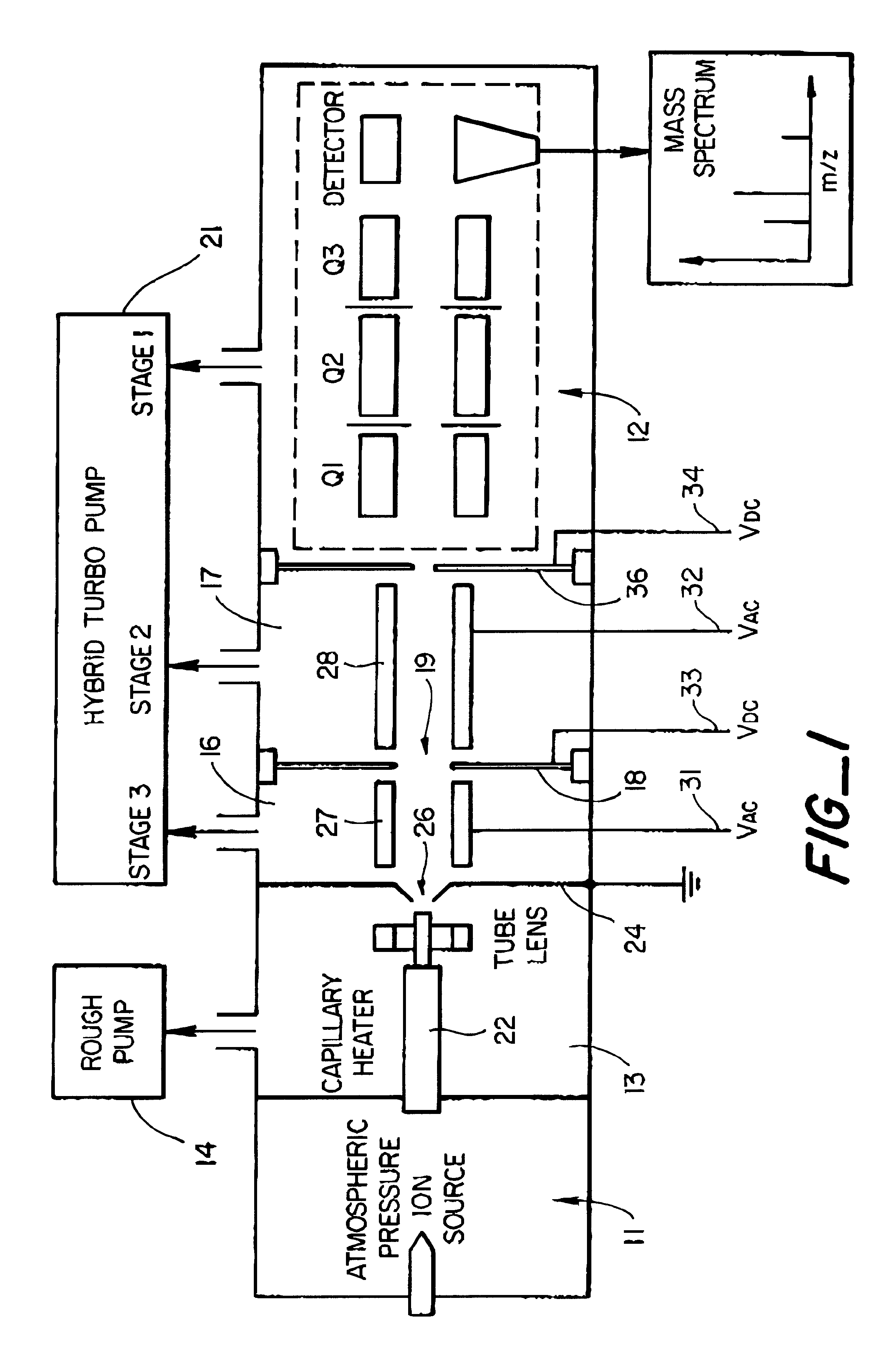
Mass spectrometer system including a double ion guide interface and method of operation
InactiveUS6528784B1Increase currentHigh sensitivityStability-of-path spectrometersSamples introduction/extractionVacuum pressureHigh pressure
There is described an interface for delivering ions generated in an ion source into a mass analyzer in a chamber under vacuum pressure. In particular, the interface employs two consecutive ion guides operated to dissociate adduct ions formed in the ion source or high pressure regions of the interface between the ion source and the mass analyzer, thus improving the limit of detection or limit of quantitation of the mass analyzer by increasing the analyte ion current.
Owner:THERMO FINNIGAN
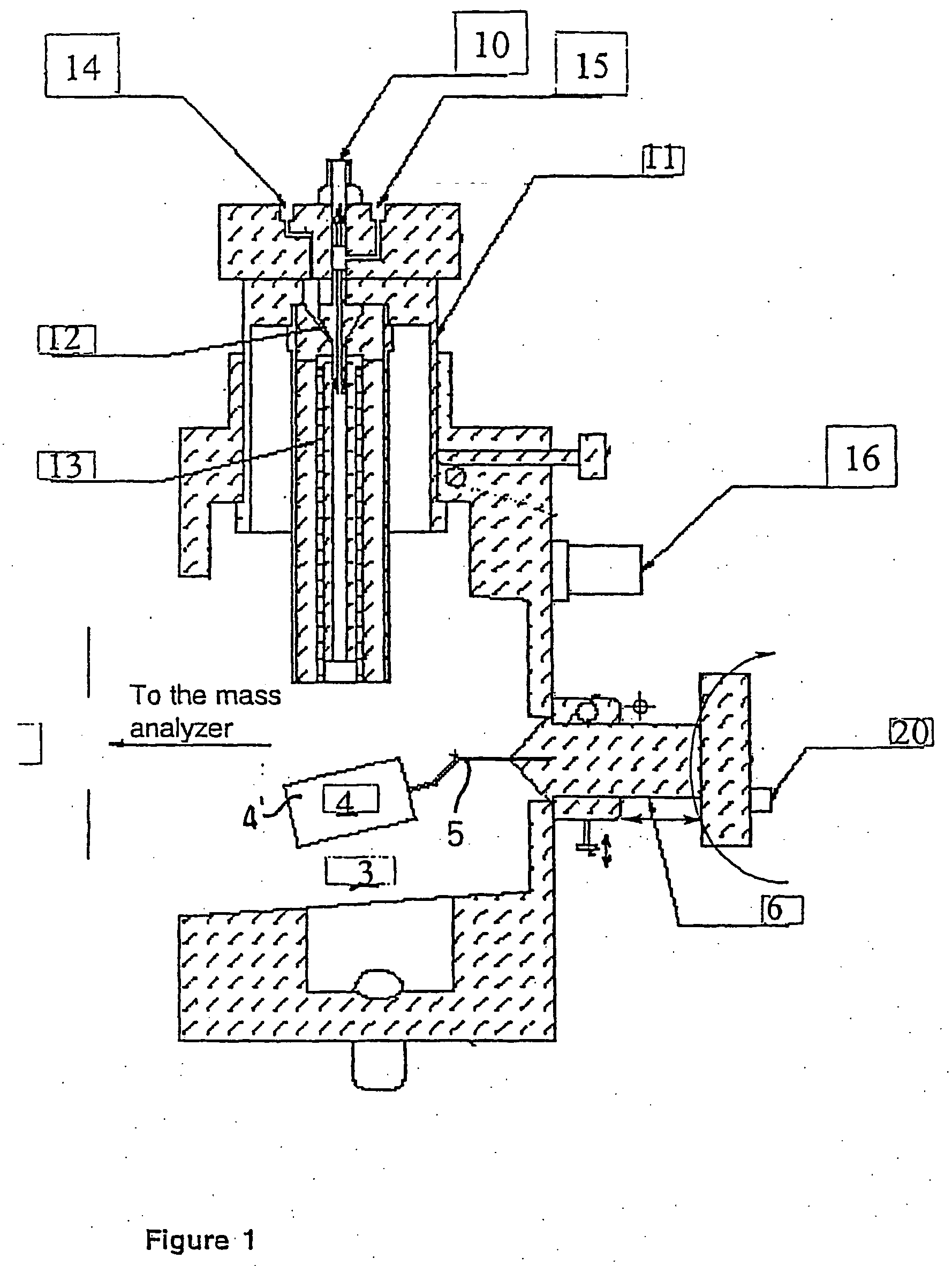
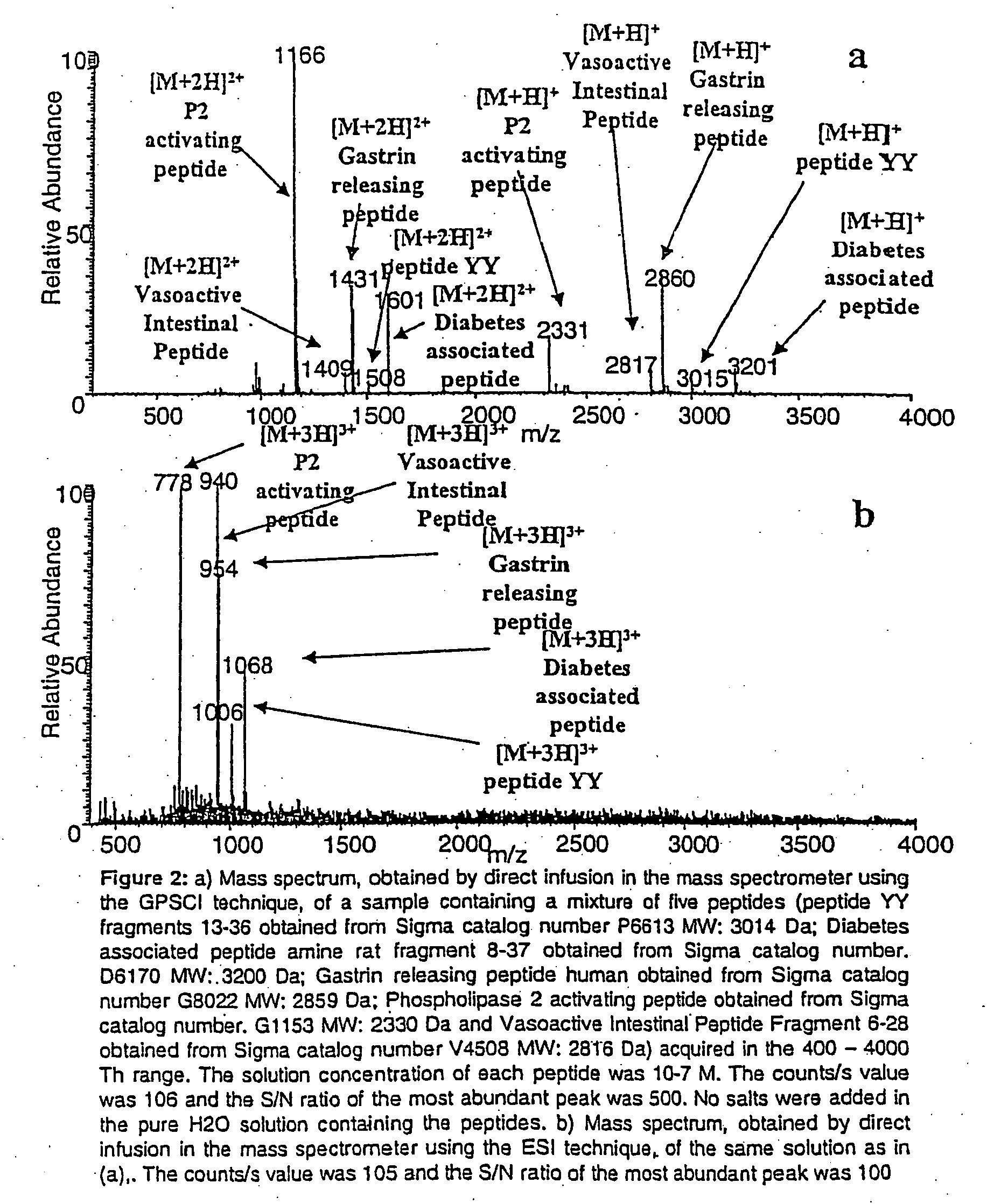
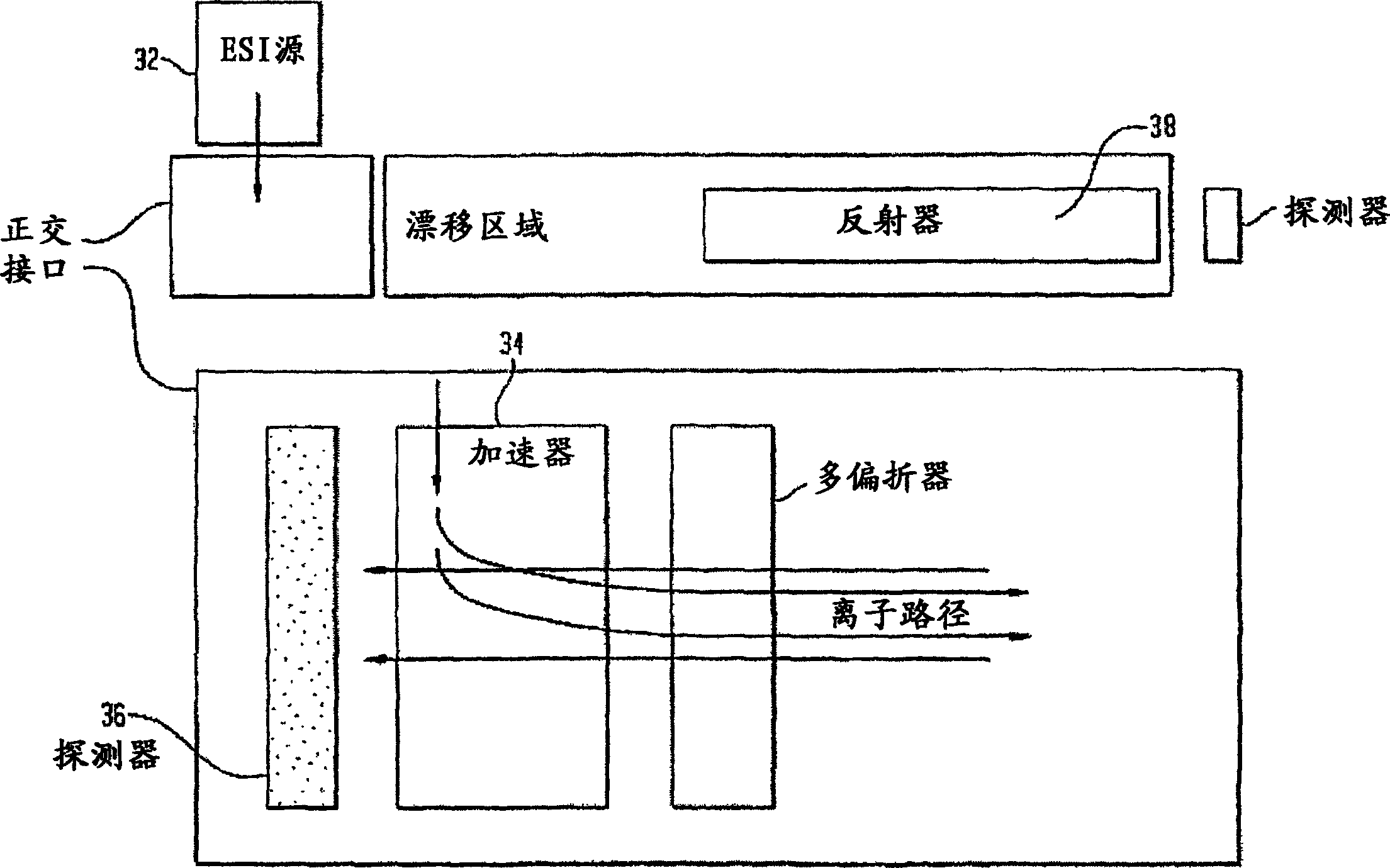
Ionization source for mass spectrometry analysis
ActiveUS20060145089A1Samples introduction/extractionMaterial analysis by optical meansGas phaseMass analyzer
A new ionization source named Surface Activated Chemical Ionization (SACI) has been discovered and used to improve the sensitivity of the mass spectrometer. According to this invention the ionization chamber of a mass spectrometer is heated and contains a physical new surface to improve the ionization process. The analyte neutral molecules that are present in gas phase are ionized on this surface. The surface can be made of various materials and may also chemically modified so to bind different molecules. This new ionization source is able to generate ions with high molecular weight and low charge, an essential new key feature of the invention so to improve sensitivity and reduce noise. The new device can be especially used for the analysis of proteins, peptides and other macromolecules. The new invention overcomes some of the well known and critical limitations of the Electrospray (ESI) and Matrix Assisted Laser Desorption Ionization (MALDI) mass spectrometric techniques.
Owner:UNIV DELGI STUDI DI MILANO
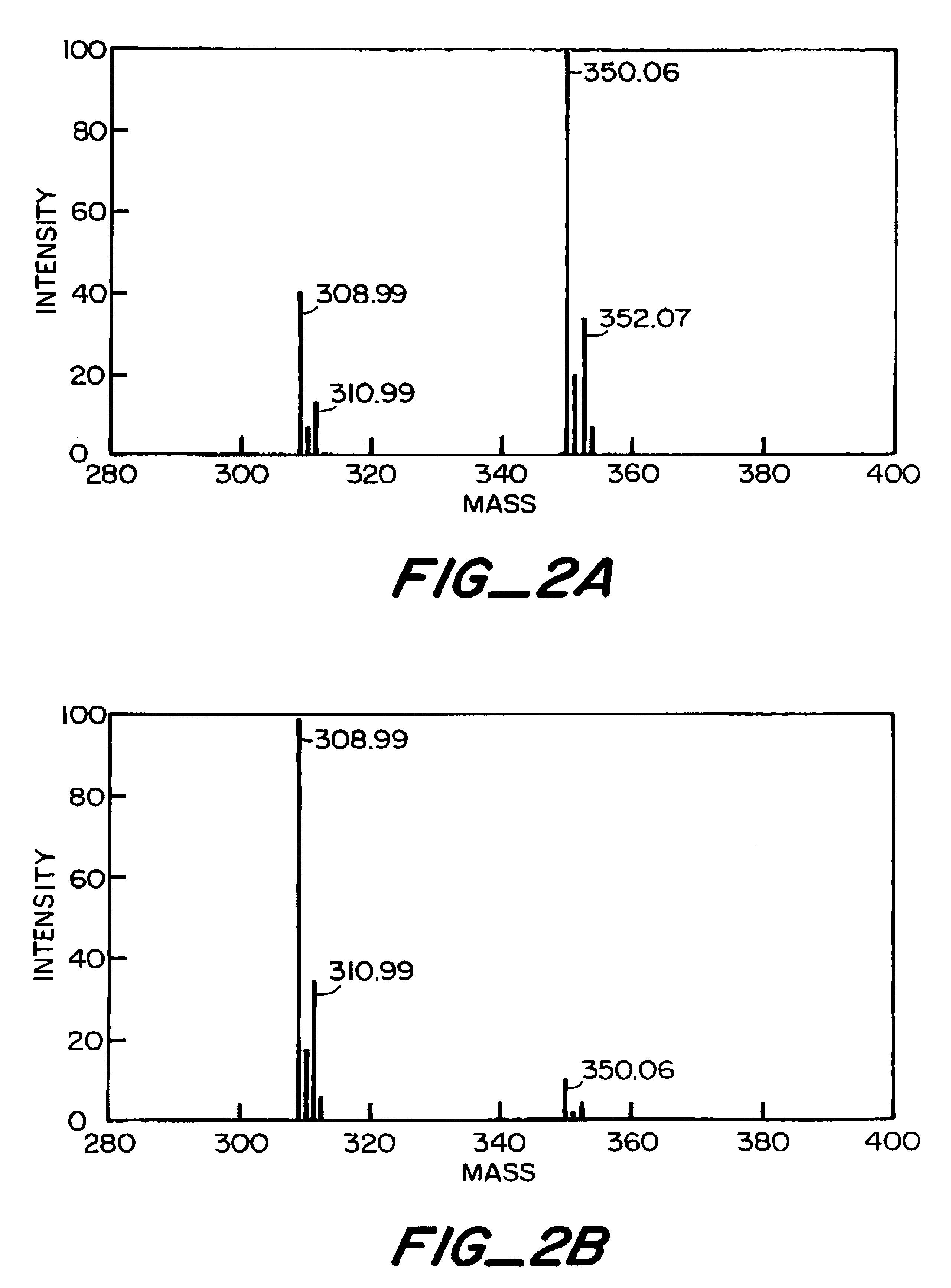
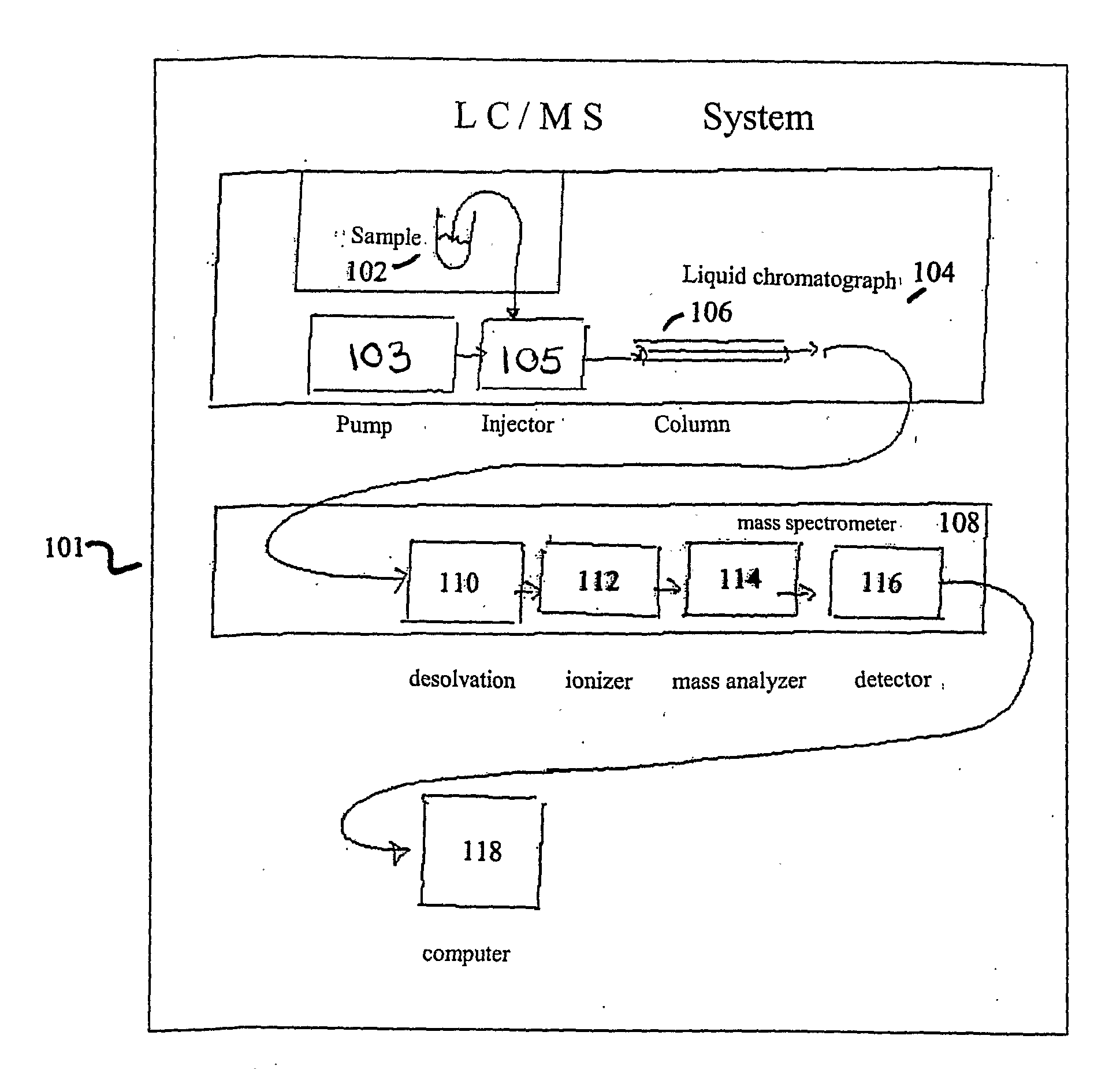
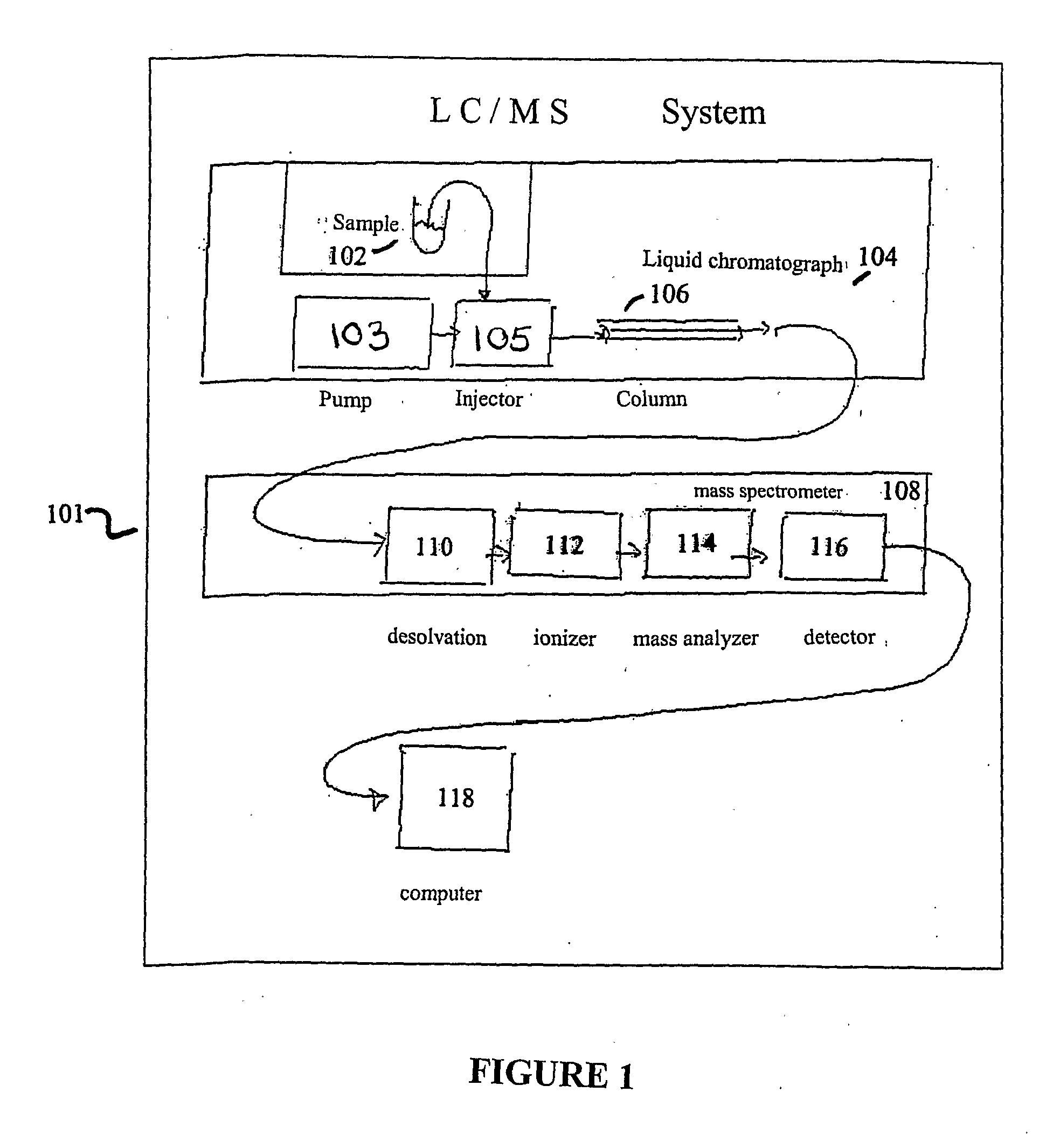
Apparatus and Method For Identifying Peaks In Liquid Chromatography/Mass Spectrometry And For Forming Spectra And Chromatograms
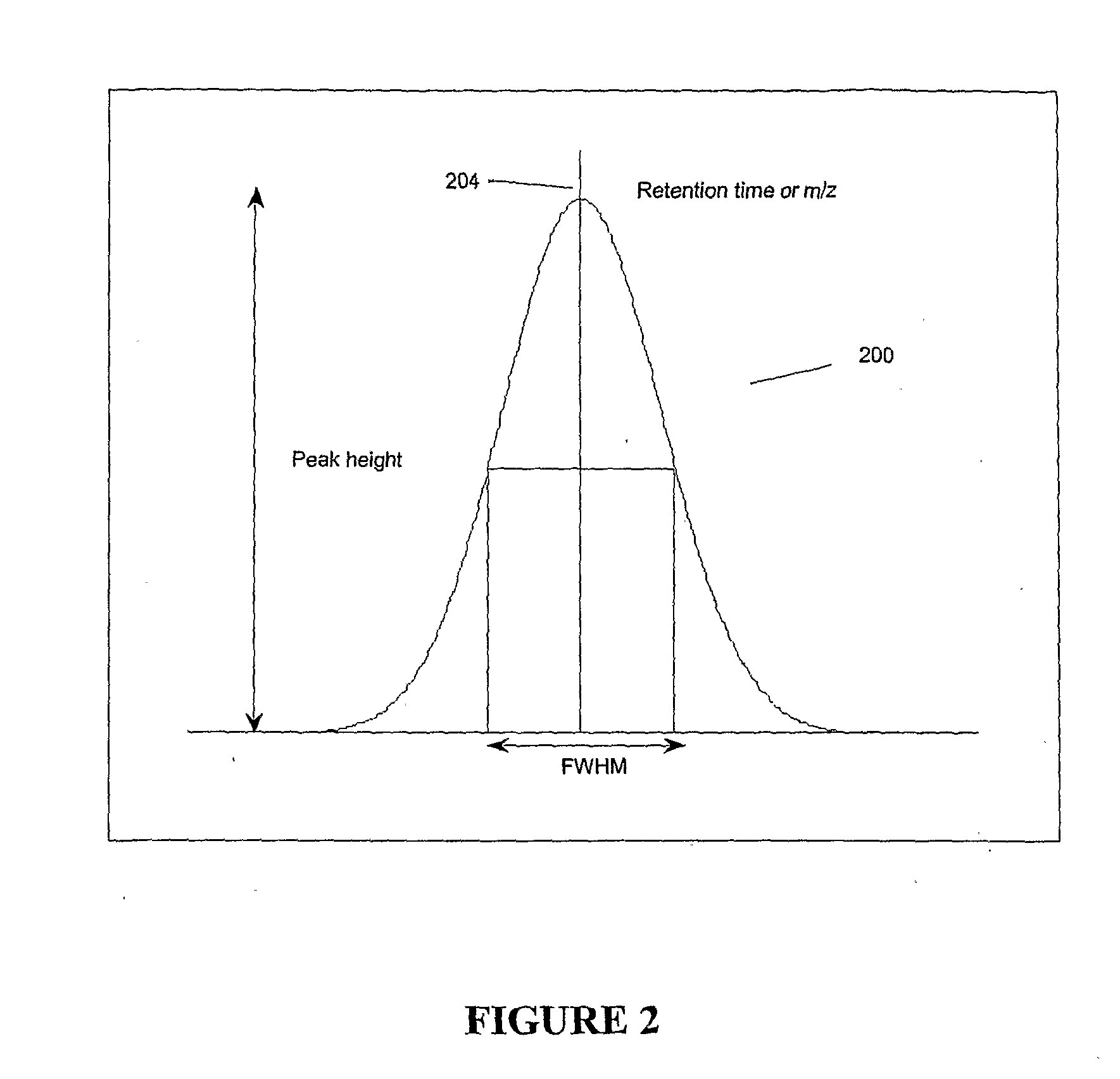
ActiveUS20070278395A1Enhanced completeness and accuracy and reproducibilityImproving completeness and accuracy and reproducibilitySpectral/fourier analysisThermometer detailsMass-to-charge ratioConvolution
Chromatograms and mass spectra produced by an LC / MS system are analyzed by creating a two-dimensional data matrix of the spectral and chromatographic data. The two-dimensional matrix can be created by placing the spectra generated by the mass spectrometer portion of the LC / MS system in successive columns of the data matrix. In this way, the rows of the data matrix correspond to chromatographic data and the columns of the data matrix correspond to the spectra. A two-dimensional filter is specified and applied to the data matrix to enhance the ability of the system to detect peaks associated with ions. The two-dimensional filter is specified according to desired criteria. Rank-1 and rank-2 filters can be specified to improve computational efficiency. One method of applying the two-dimensional filter is through convolution of the data matrix with the two-dimensional filter to produce an output data matrix. Peaks corresponding to detected ions are identified in the output data matrix. Parameters of the peaks are determined and stored for later processing including quantitation, or simplification of chromatograms or spectra by, for example, identifying peaks associating with ions having retention times falling within a specified retention time window or having mass-to charge ratios falling within a specified mass-to-charge ratio window.
Owner:WATERS TECH CORP
Mass spectrometer
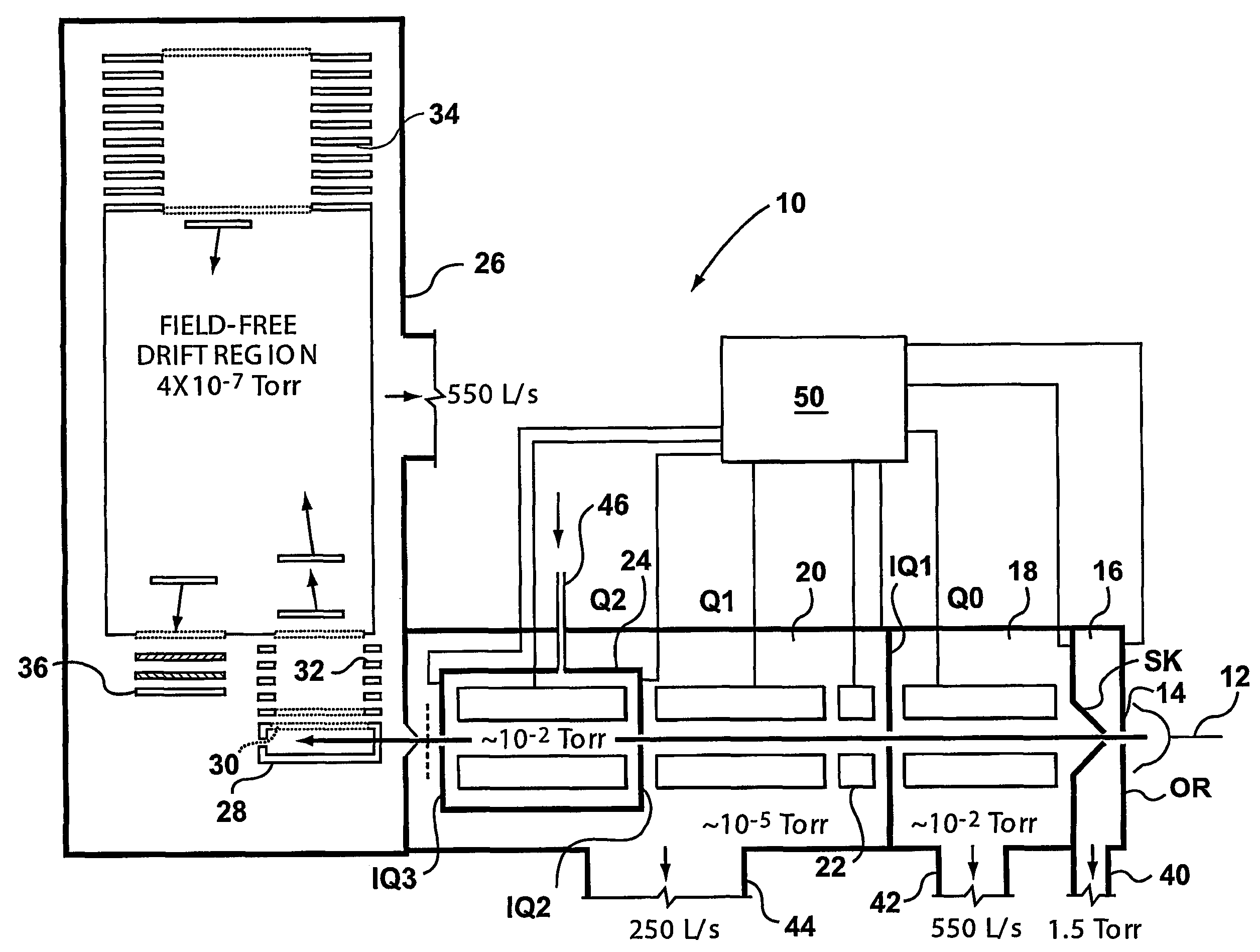
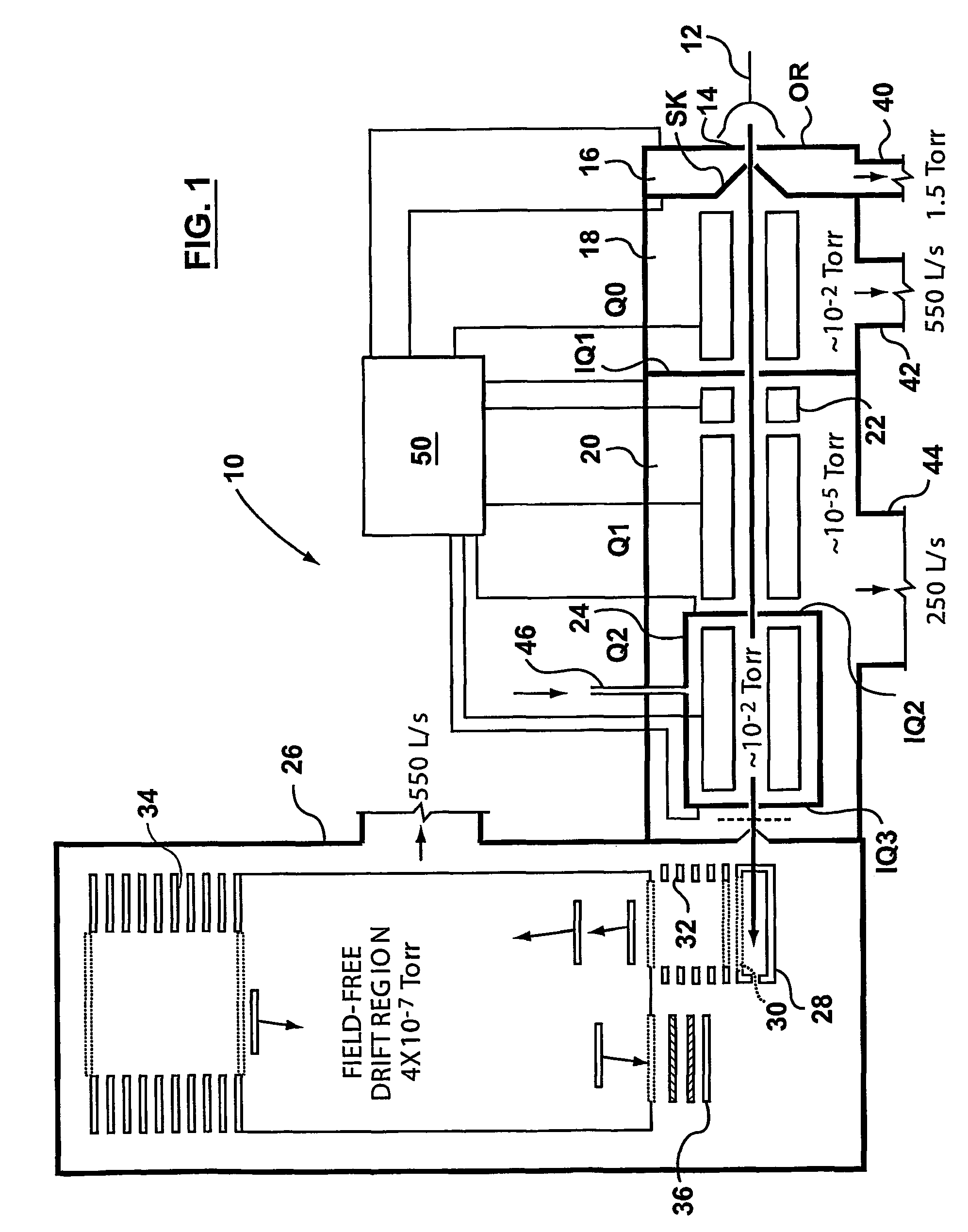
ActiveUS7071467B2Eliminate the effects ofHigh duty cycleStability-of-path spectrometersTime-of-flight spectrometersIon trap mass spectrometryTrapping region
A mass spectrometer is disclosed comprising an ion trap wherein ions which have been temporally separated according to their mass to charge ratio or ion mobility enter the ion trap. Once at least some of the ions have entered the ion trap, a plurality of ion trapping regions are created along the length of the ion trap in order to fractionate the ions. Alternatively, the ions may be received within one or more axial trapping regions which are translated along the ion trap with a velocity which is progressively reduced to zero.
Owner:MICROMASS UK LTD
Mass spectrometer
InactiveUS6875980B2Increases ion trapping volumeIncrease capacityStability-of-path spectrometersTime-of-flight spectrometersIon trap mass spectrometryMass analyzer
A mass spectrometer is disclosed wherein a relatively energetic pulse of ions having a relatively narrow spread of mass to charge ratios are ejected from a quadrupole ion trap and received in an ion trap upstream of a Time of Flight mass analyser. The ions are collisionally cooled within the ion trap and are pulsed out of the ion trap and into an extraction region of the Time of Flight mass analyser without substantially exciting the ions. This enables improved operation with the Time of Flight mass analyser. According to another embodiment, parent ions are fragmented and the resulting fragment ions are stored in two ion traps having different low mass cut-offs. The trapping system enables MS / MS experiments to be performed with a very high duty cycle.
Owner:MICROMASS UK LTD
Novel tandem mass spectrometer
InactiveUS20070084998A1Improve dynamic rangeImprove overall utilizationIsotope separationMass spectrometersPhysical chemistryMass analyzer
In a tandem mass spectrometer ions are created only once and stored in an ion reservoir. A particular ion species to be analyzed is then exported from the reservoir through a mass selective ion gate without damaging the other ion species remaining in the reservoir. All subsequent analyses are conducted on these stored ions, without adding further ions so that no changes in the concentrations of the stored ion species occur. The exported ions are fragmented, and a fragment ion mass spectrum is measured in a mass analyzer, preferably in a time-of-flight mass analyzer with orthogonal ion injection. The processes of exporting a selected ion species with subsequent fragmentation and the acquisition of the fragment ion spectrum can be repeated for any number of ion species stored in the reservoir.
Owner:BRUKER DALTONIK GMBH +1
Apparatus and method for MSnth in a tandem mass spectrometer system
InactiveUS7145133B2Simple capabilityImprove data accuracyStability-of-path spectrometersTime-of-flight spectrometersIon trap mass spectrometryPhysical chemistry
A method and apparatus are provided for effecting multiple mass selection or analysis steps. Fundamentally, the technique is based on moving ions in different directions through separate components of a mass spectrometer apparatus. To effect different steps, a precursor ion is selected in a first mass selector, and then passed into a collision cell, to effect fragmentation or reaction with a gas, to generate fragment or product ions. The generated product ions are then passed back into the first mass selector, and preferably back into an upstream ion trap. The product ions then pass through the first mass selector again, to select a desired product ion, for further fragmentation and analysis. These steps can be repeated a number of times. A final mass analysis step can be effected in either a time-of-flight section or other mass analyzer. The invention enables conventional triple quadrupole mass spectrometers and QqTOF mass spectrometers to effect multiple MS steps.
Owner:MDS CO LTD +2
Pattern recognition of whole cell mass spectra
ActiveUS20050061967A1Improve accuracyIncrease speedSamples introduction/extractionIsotope separationMass spectrometryAdduct
A method for reproducibly analyzing mass spectra from different sample sources is provided. The method deconvolutes the complex spectra by collapsing multiple peaks of different molecular mass that originate from the same molecular fragment into a single peak. The differences in molecular mass are apparent differences caused by different charge states of the fragment and / or different metal ion adducts and / or reactant products of one or more of the charge states. The deconvoluted spectrum is compared to a library of mass spectra acquired from samples of known identity to unambiguously determine the identity of one or more components of the sample undergoing analysis.
Owner:NAT INST OF HEALTH REPRESENTED BY THE SEC OF THE DEPT OF HEALTH & HUMAN SERVICES NAT INST OF HEALTH
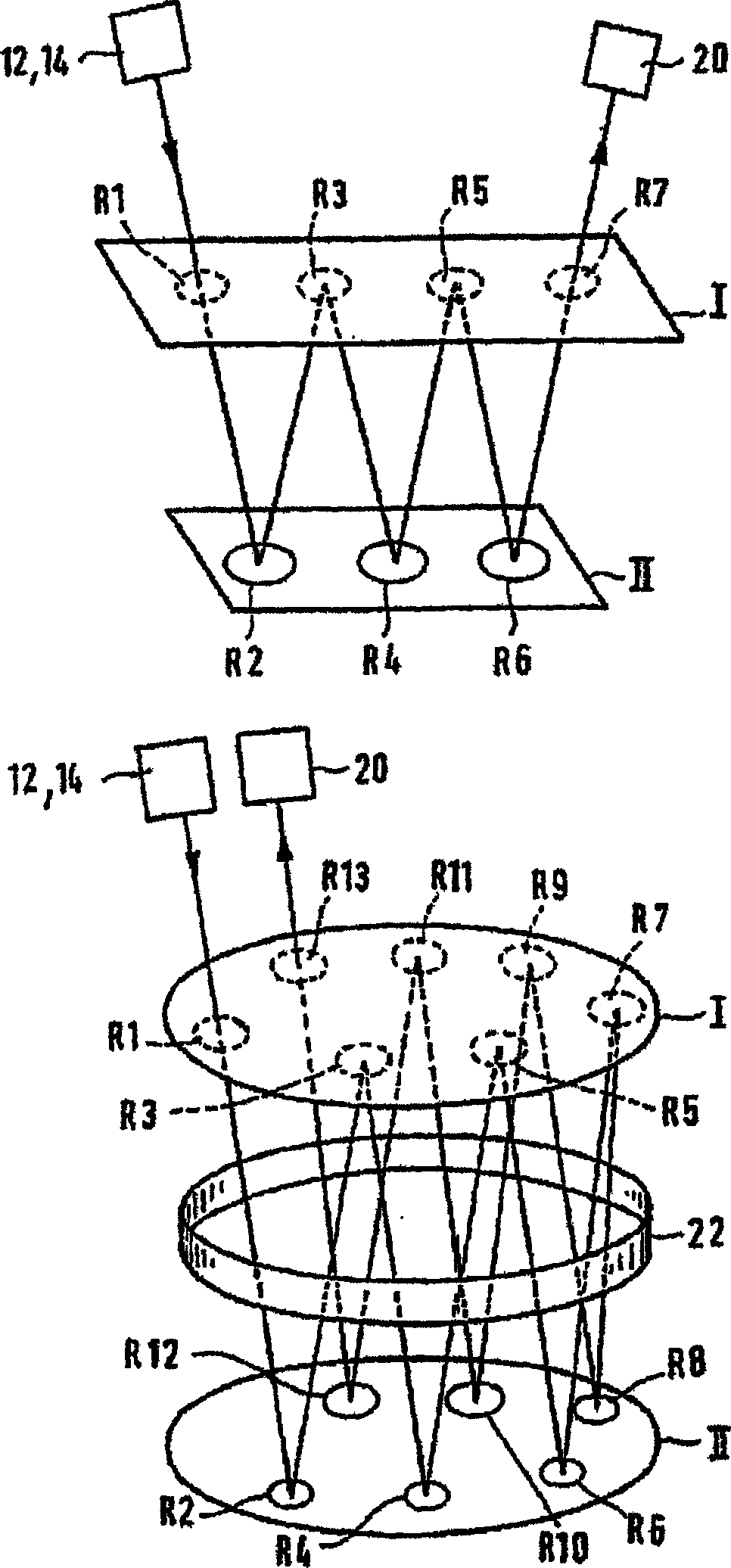
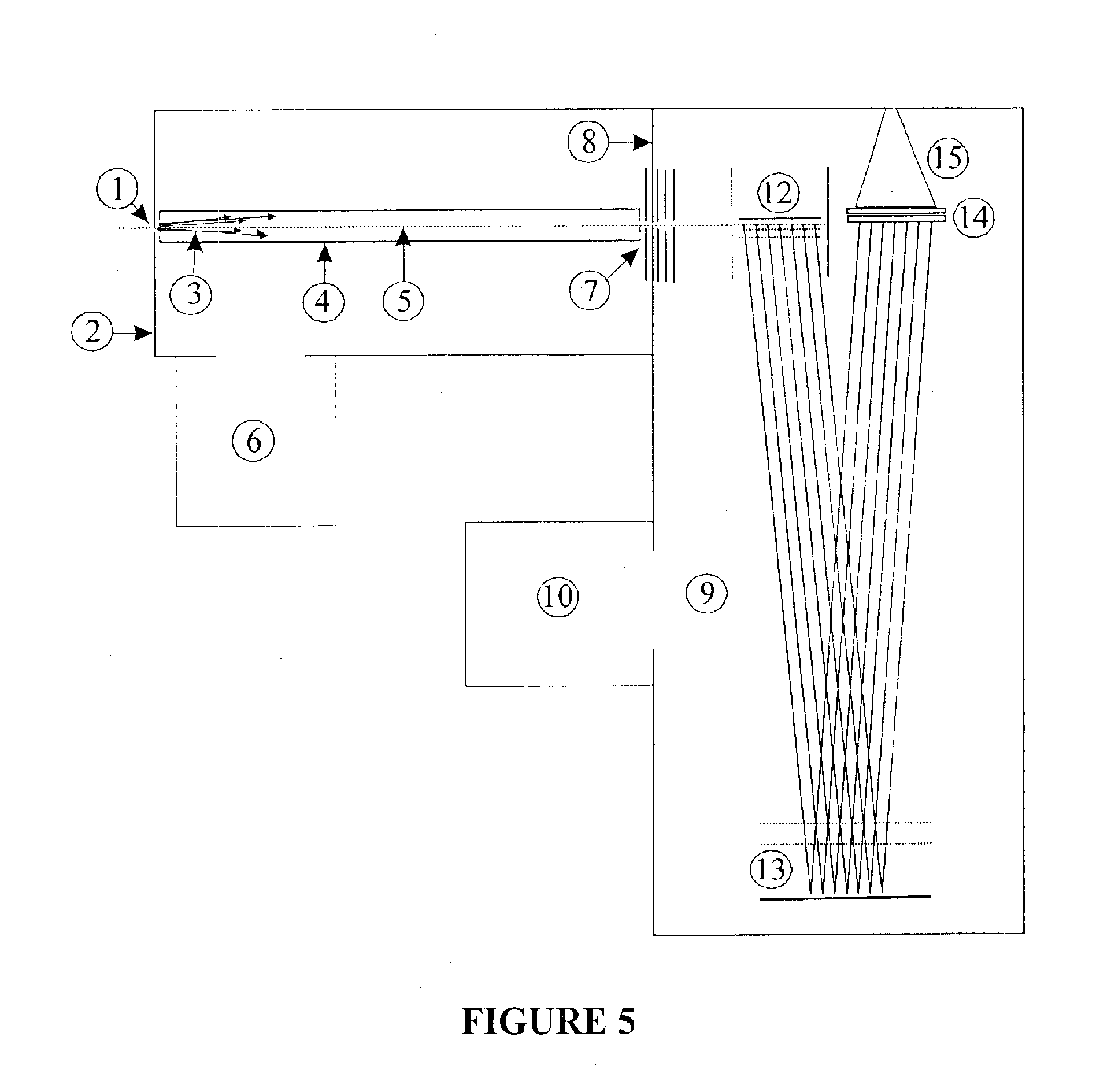
Multi-reflecting time-of-flight mass spectrometer and a method of use
ActiveCN1853255AFavorable combinationQuick jetTime-of-flight spectrometersMaterial analysis by electric/magnetic meansTime-of-flight mass spectrometryImage resolution
A multi reflecting time-of-flight mass spectrometer MR TOF MS 11. the flight path of ions from an ion source 12 to a receiver is folded along a trajectory by two parallel gridless electrostatic mirrors 15, elongated in the shift direction 7, orthogonal to the direction of reflection. A set of multiple lenses 17 is positioned in the drift space 14 between the mirrors to provide for spatial focusing of ions in the plane of the folded ion path. Each mirror consists of at least 4 electrodes 15C, 15E, 15L arranged and controlled so that to improve ion optics properties. Namely, in addition to time-of-flight focusing in energy and spatial focusing across the plane of the folded ion path, the mirrors also provide time-of-flight focusing with respect to the spatial spread of ions across the said plane. Because of improved spatial and time focusing, the MR TOF MS of the invention provides for a wider acceptance and confinement of ion beam along an extended folded ion path.
Owner:LECO CORPORATION
Aspects of mass spectral calibration
InactiveUS20060169883A1Improve accuracyHigh resolutionTime-of-flight spectrometersMaterial analysis by optical meansElemental compositionSpectral response
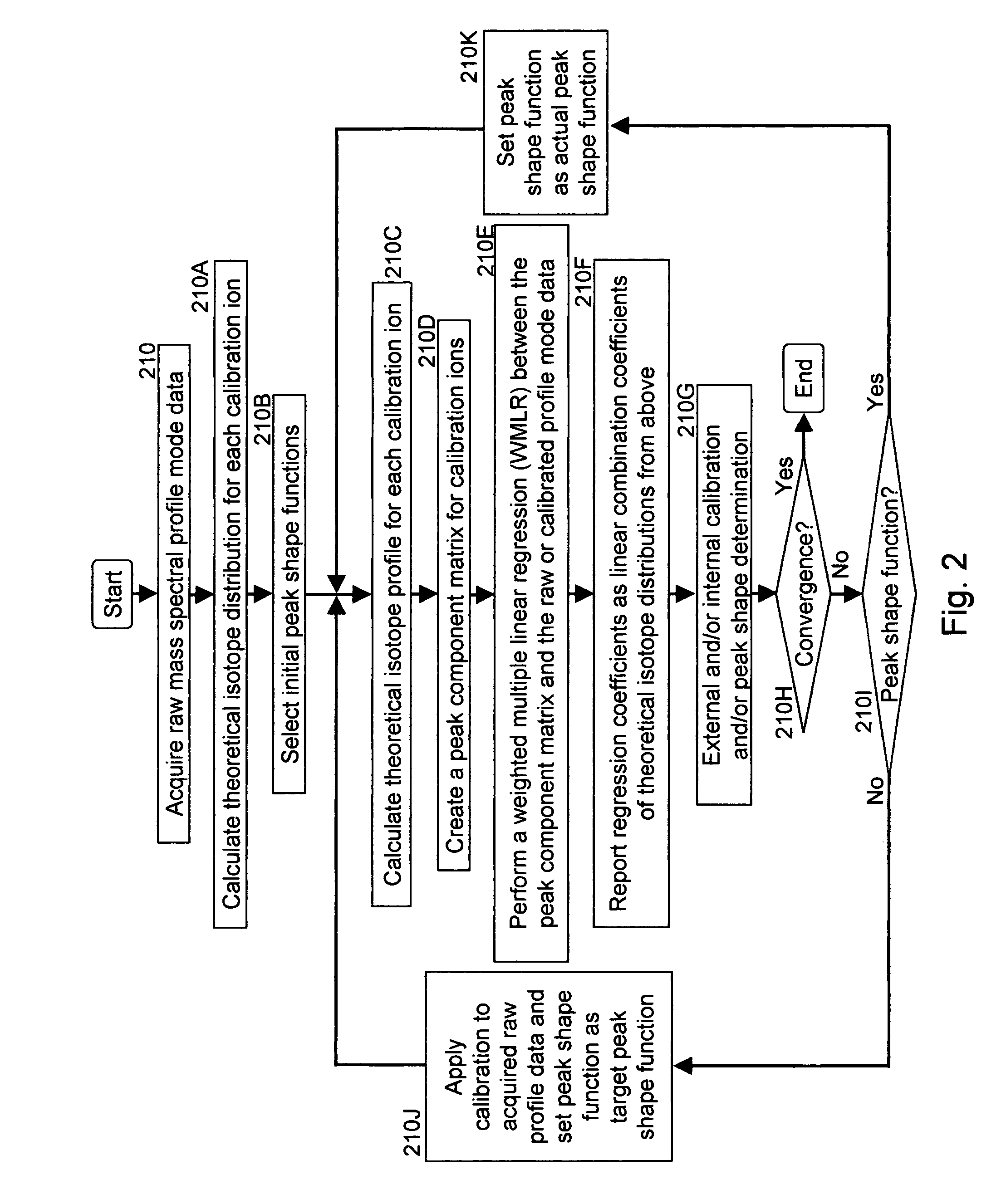
A method for calibrating and analyzing data from a mass spectrometer, comprising the steps of acquiring raw profile mode data containing mass spectral responses of ions with or without isotopes; calculating theoretical isotope distributions for each of at least one calibration ion based on elemental composition; convoluting the theoretical isotope distributions with an initial peak shape function to obtain theoretical isotope profiles for each ion; constructing a peak component matrix including the theoretical isotope profiles for calibration ions as peak components; performing a regression analysis between the raw profile mode mass spectral data and the peak component matrix; and reporting the regression coefficients as the relative concentrations for each of the components. A mass spectrometry system operated in accordance with the method and a computer readable medium having program code thereon for performing the method.
Owner:CERNO BIOSCI
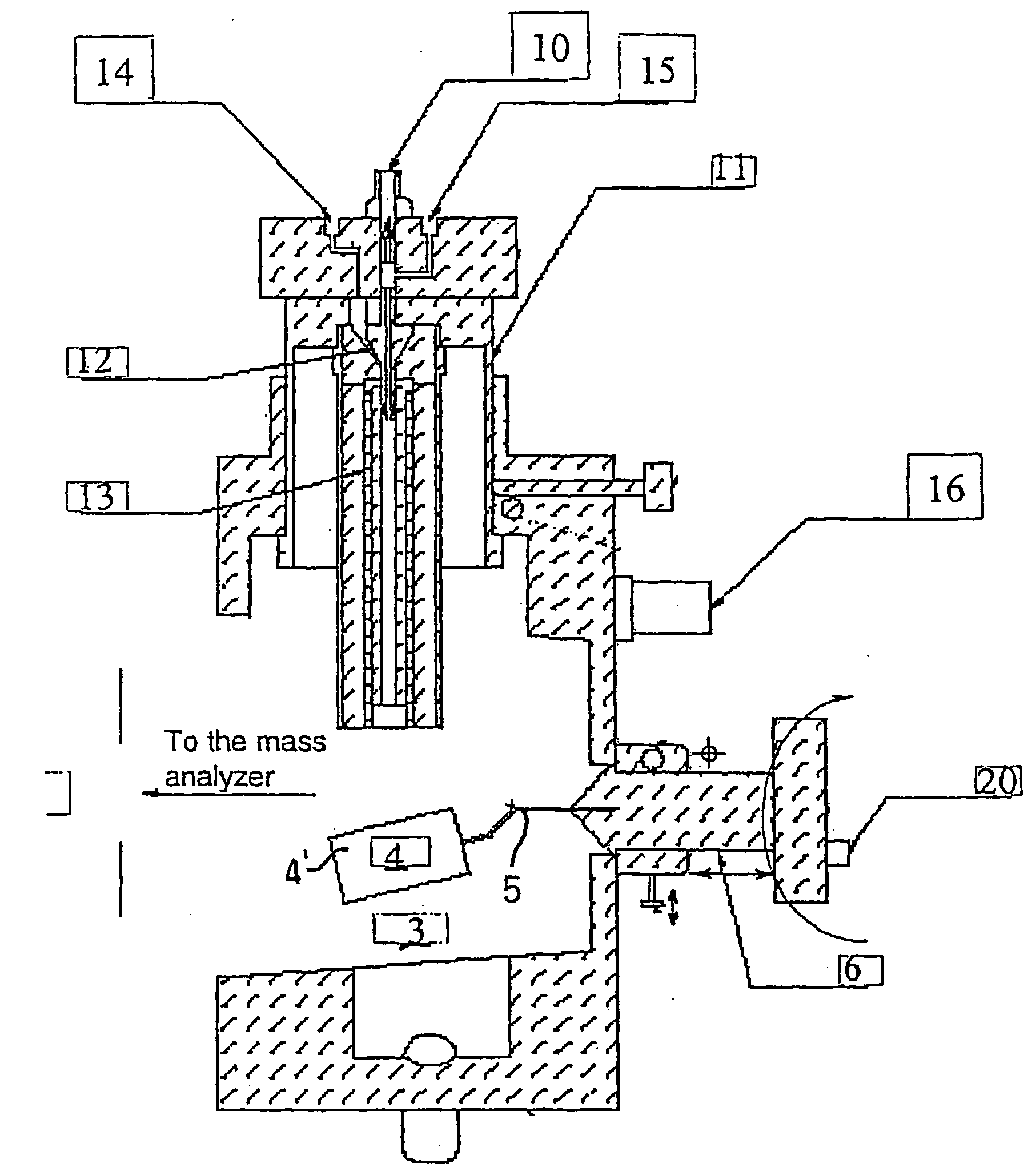
Method and apparatus for mass spectrometric analysis of samples
InactiveUS6861647B2Time-of-flight spectrometersSamples introduction/extractionPhysical chemistryIonization chamber
An MALDI mass spectrometer for the composition analysis of large batch sizes of samples includes a mass spectrometer having an ionization chamber and a sample chamber coupled to the ionization chamber. A transport cart is positioned in the sample chamber with a sample cassette removably coupled thereto. A method of operating a MALDI mass spectrometer is also disclosed.
Owner:INDIANA UNIV RES & TECH CORP
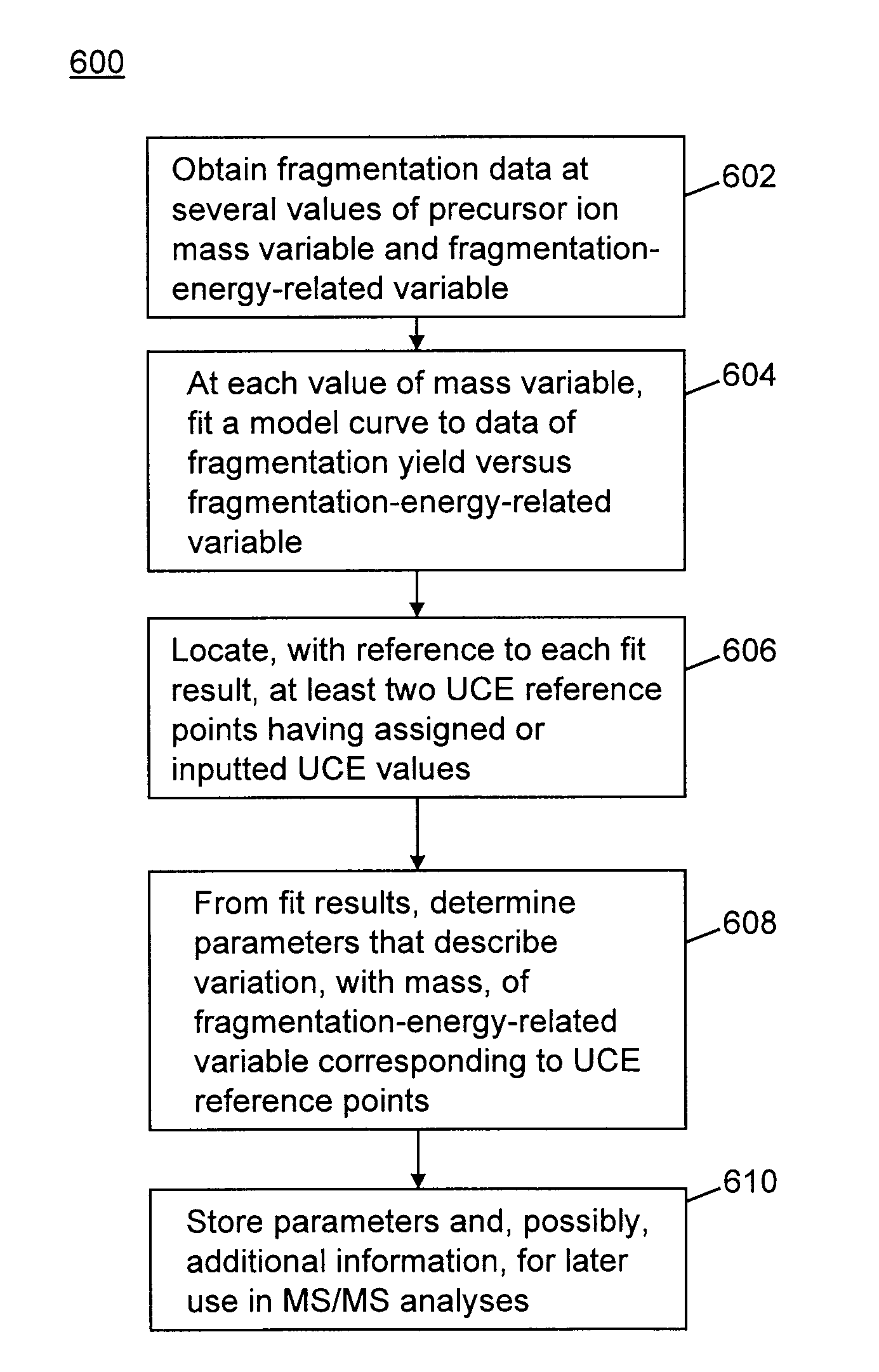
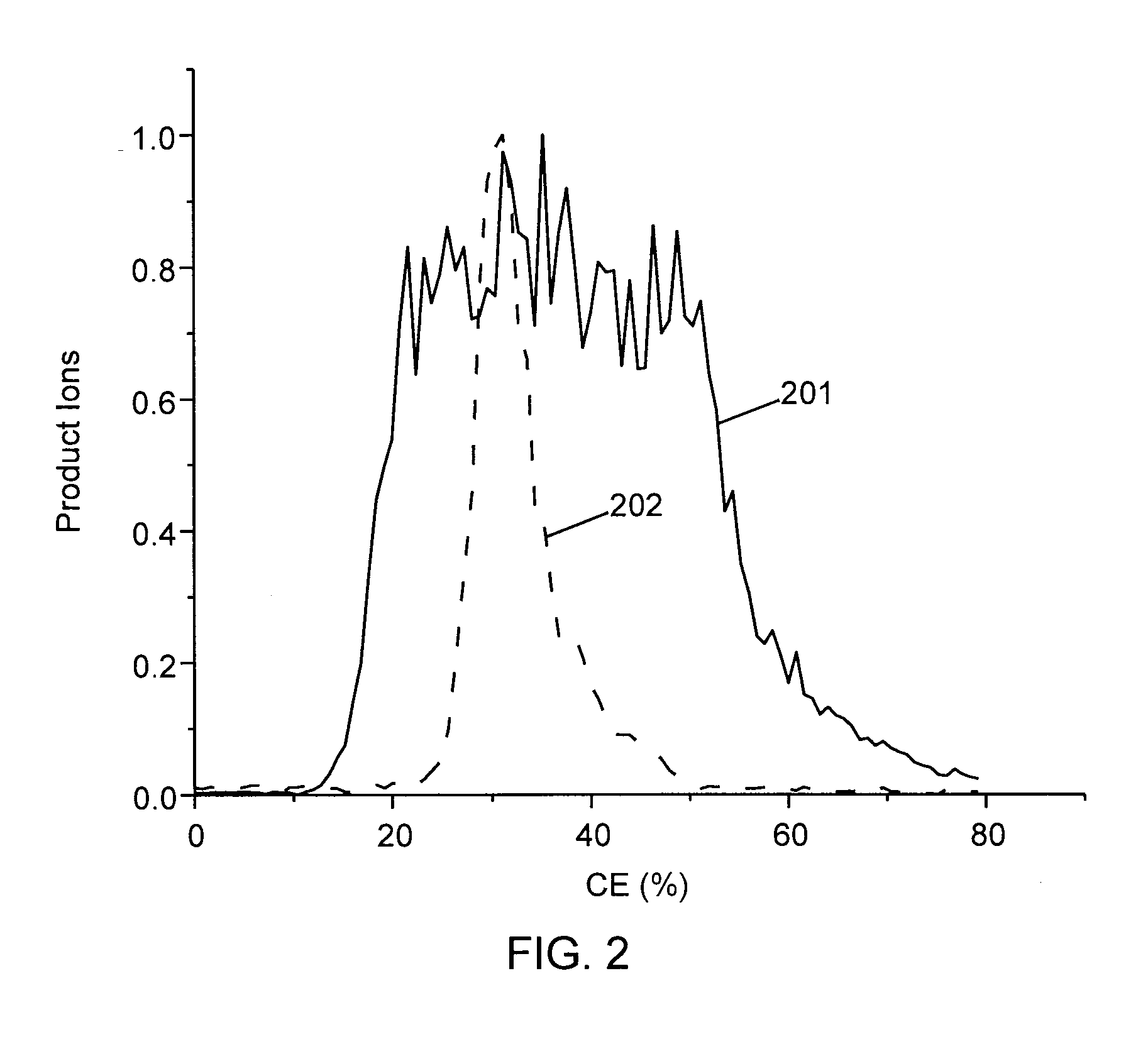
Methods for calibration of usable fragmentation energy in mass spectrometry
ActiveUS8278620B2Isotope separationCalibration apparatusMass Spectrometry-Mass SpectrometryMass analyzer
Owner:THERMO FINNIGAN
Precise and thorough background subtraction
ActiveUS20100213368A1Avoid enteringIsotope separationMass spectrometersTest sampleMass Spectrometry-Mass Spectrometry
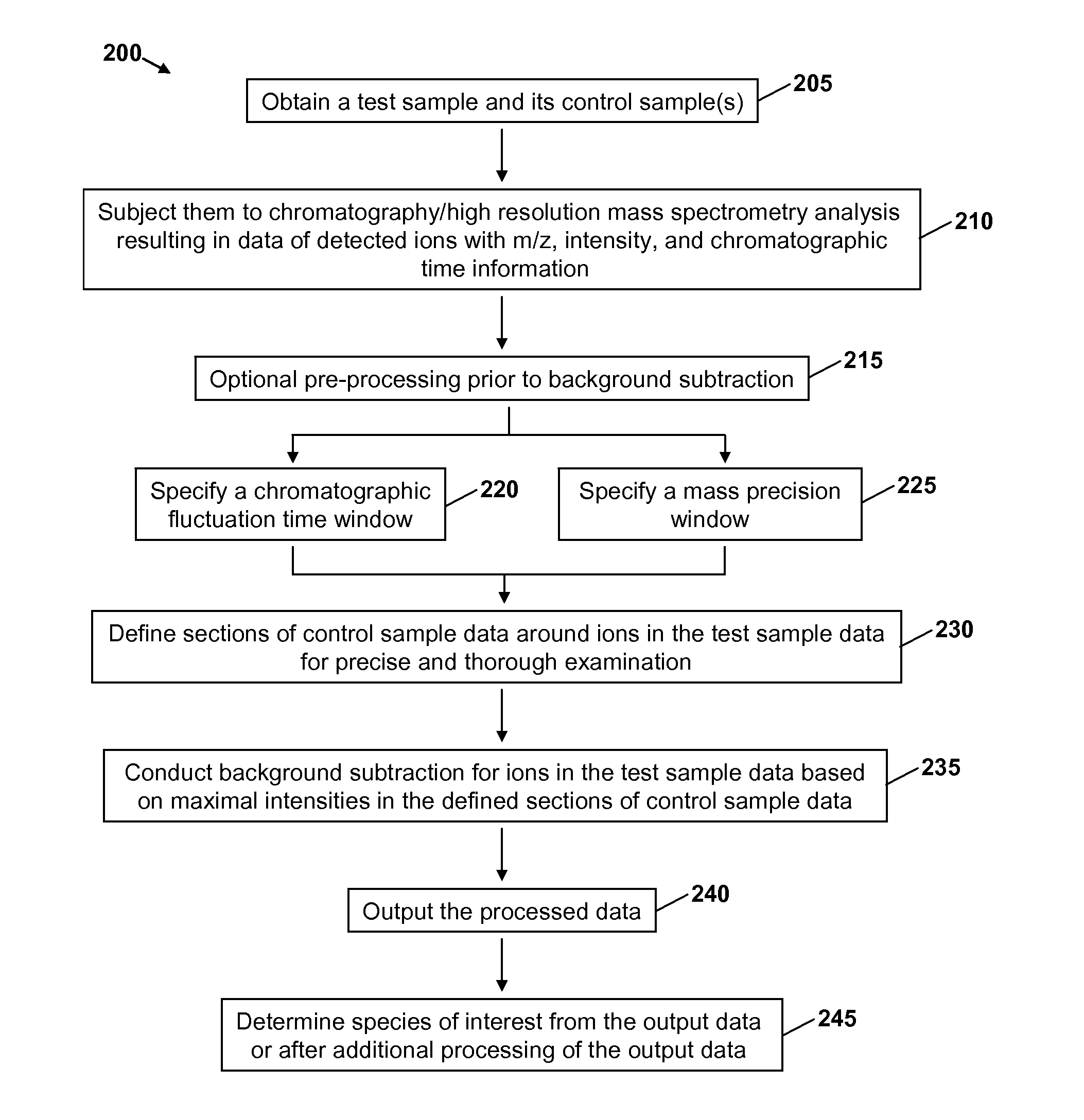
A method for identifying and characterizing components of interest in complex samples includes subjecting both a sample and its control samples to chromatography / high resolution mass spectrometry analysis to detect ions of the samples. The method includes defining sections of control sample data within specified chromatographic fluctuation time and mass precision windows around each ion or each group of the same ions of question in the test sample data. The defined sections of the control sample data are examined and the maximal intensities are subtracted from respective ions in the test sample. Components of interest are determined from the resultant data of the test sample. The method can be used for identifying molecular ions and / or their fragment ions for components of interest in complex samples.
Owner:MASSDEFECT TECH
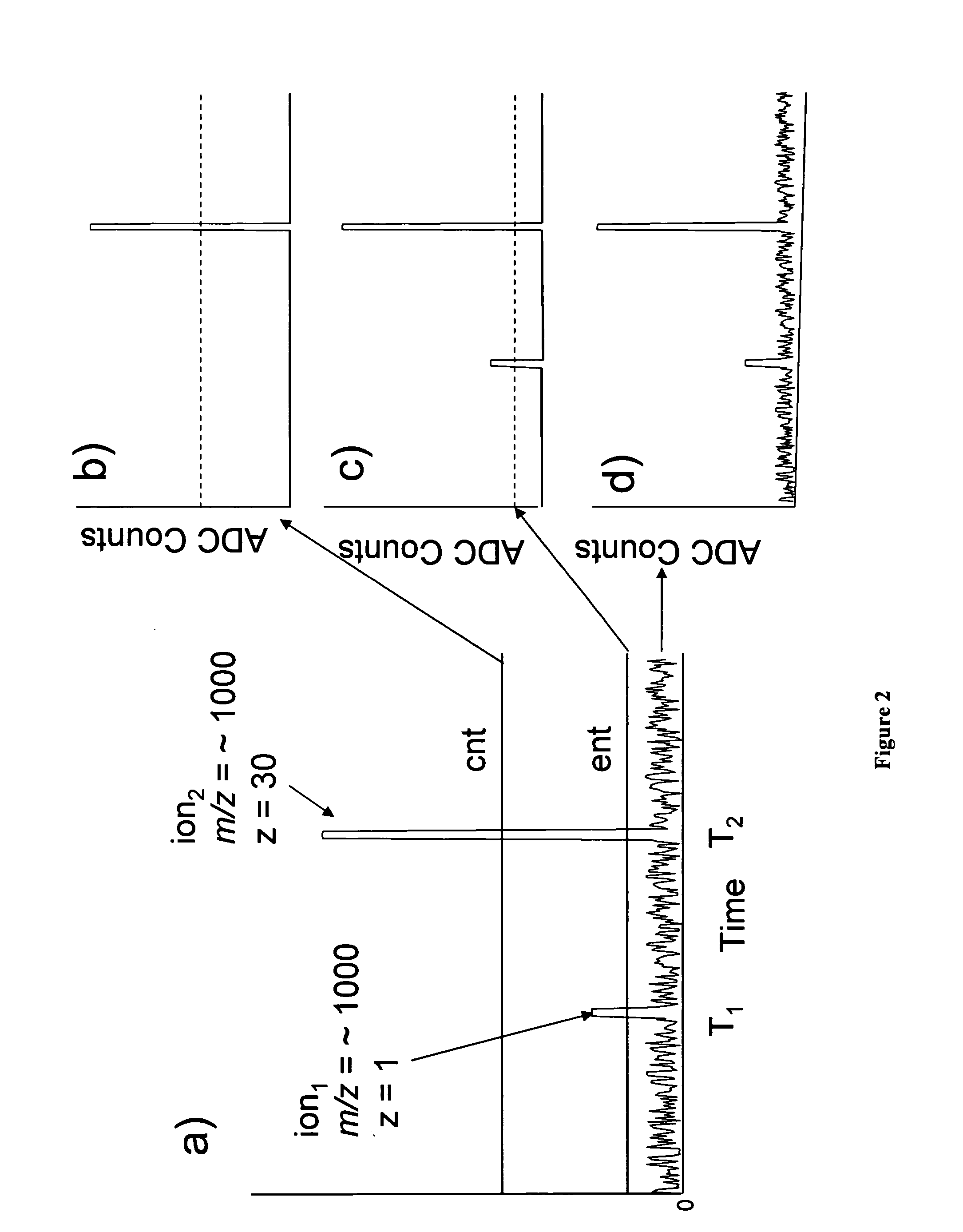
High resolution detection for time-of-flight mass spectrometers
InactiveUS6870156B2High resolutionInhibit transferSpectrometer detectorsTime-of-flight spectrometersEngineeringMass analyzer
The invention covers a method for detecting ions in high resolution time-of-flight mass spectrometers which operate with secondary electron multiplier multichannel plates and in which many single spectra are acquired and added to produce a sum spectrum. The invention involves (a) using an analog digital converter (ADC) for converting electron currents from secondary electron multipliers, instead of a time-to-digital converter (TDC) which was previously used for highest possible signal resolution, (b) performing a separate rapid peak recognition procedure for the ion signals of each spectrum by a fast calculation method, thereby collecting flight time and intensity value pairs for the ion peaks, and (c) constructing a time-of-flight / intensity histogram, which is further processed as a composite time-of-flight spectrum. The invention retains the significantly higher measurement dynamics of an ADC and achieves the improved resolution capability of a TDC, but without showing the latter's known signal distortion due to dead times.
Owner:BRUKER DALTONIK GMBH & CO KG
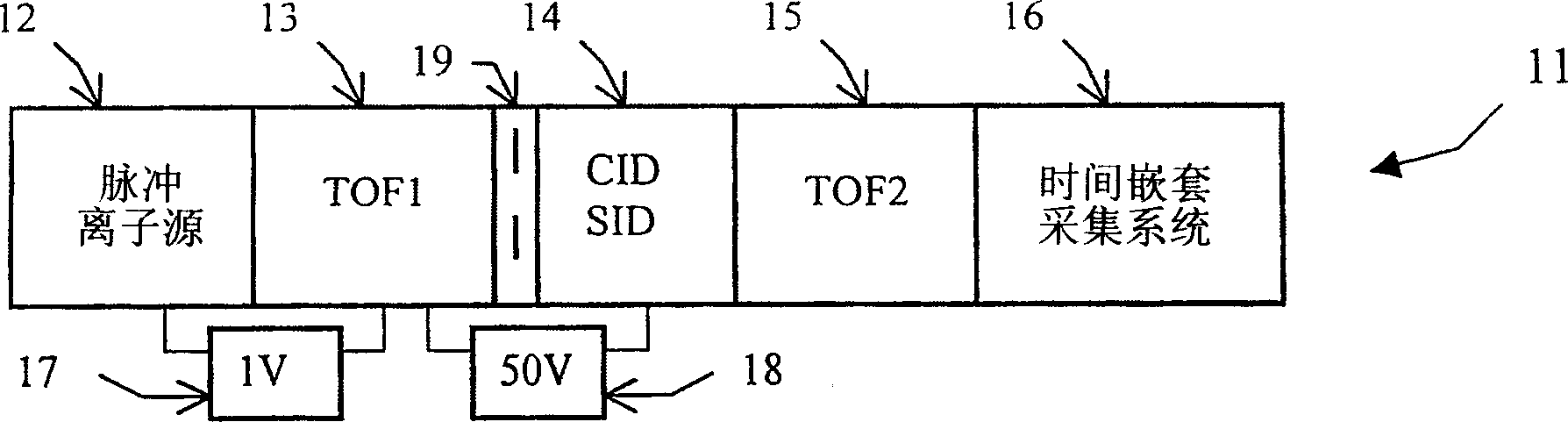
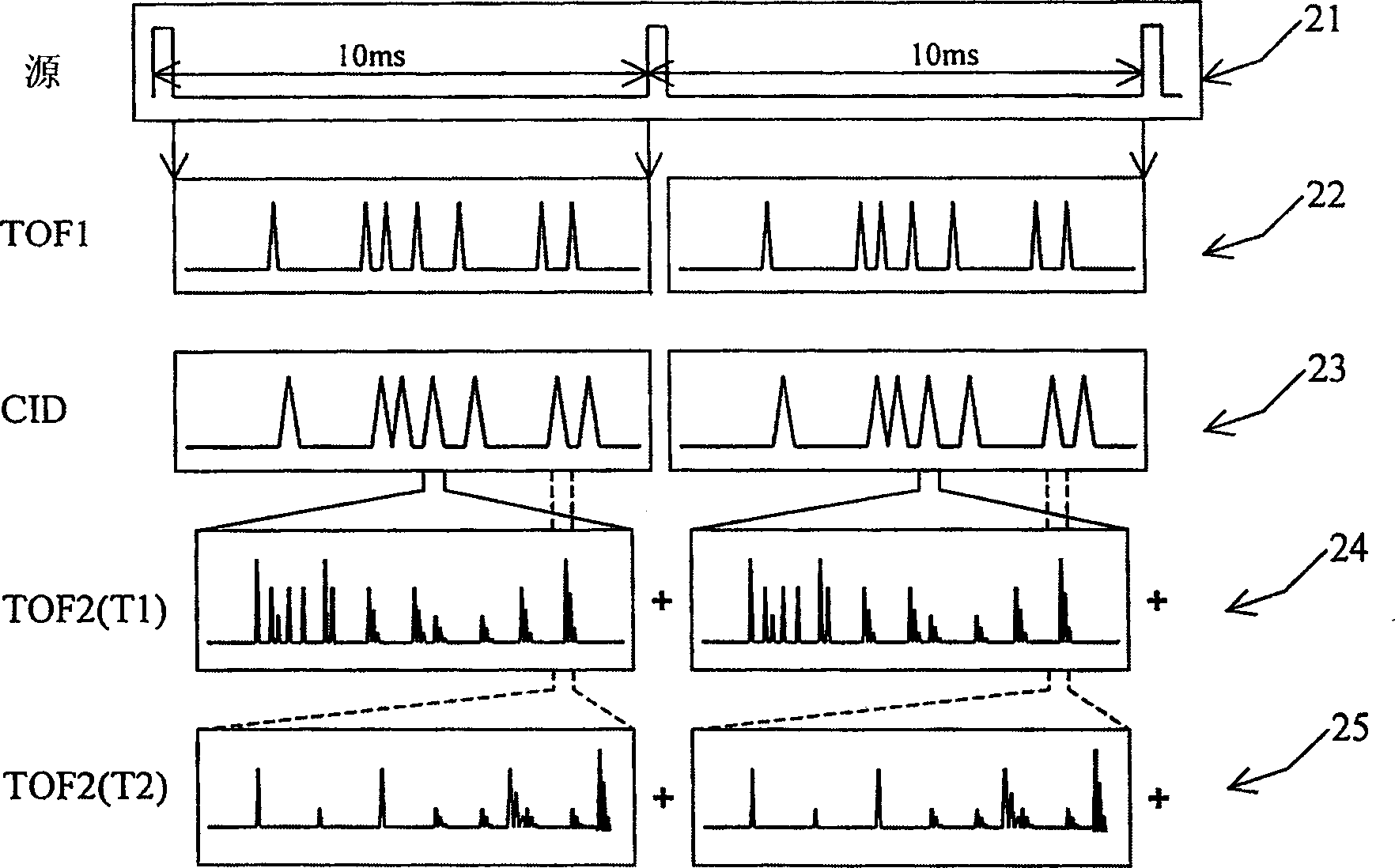
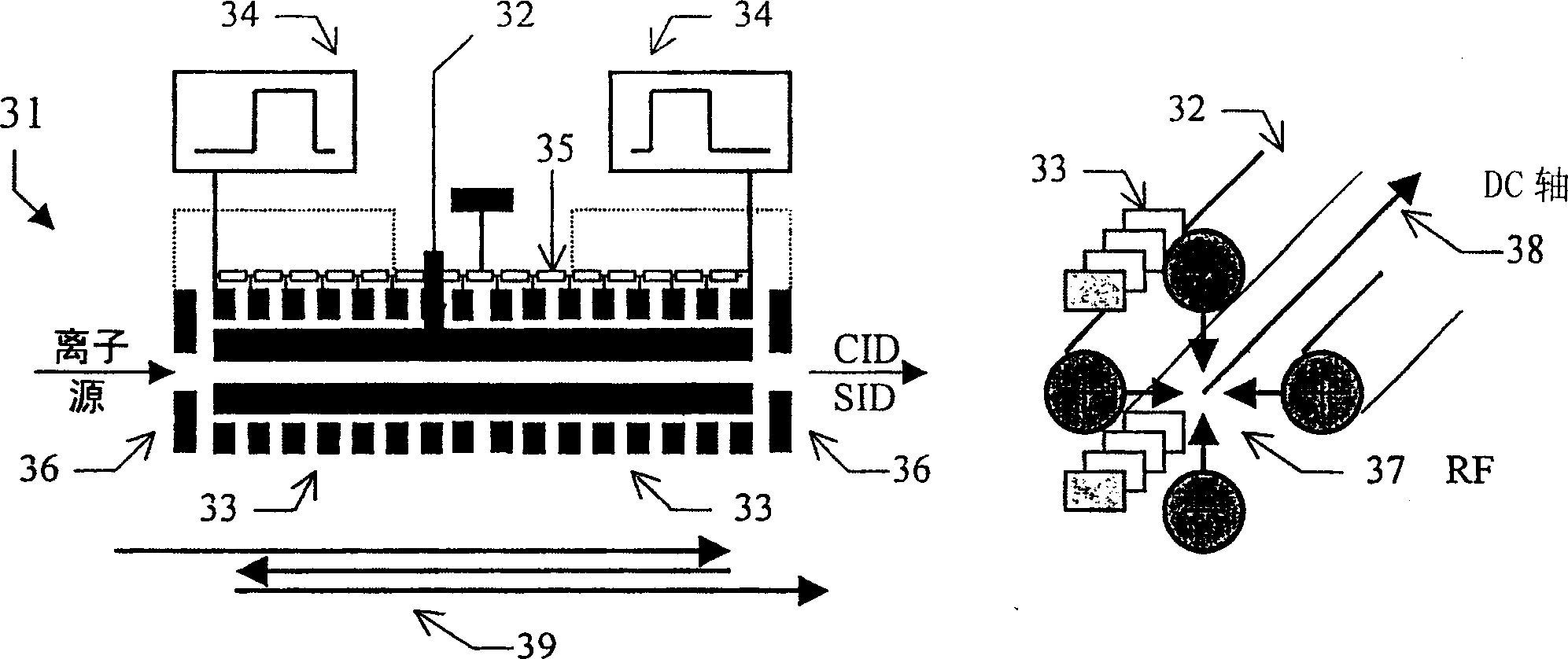
Tandem time of flight mass spectrometer and method of use
InactiveCN1689134AAcquisition speed is fastHigh sensitivityTime-of-flight spectrometersElectron/ion optical arrangementsRelative energyMass analyzer
To provide comprehensive (i.e. rapid and sensitive) MS-MS analysis, the inventor employs a time-nested separation, using two time-of-flight (TOF) mass spectrometers. Parent ions are separated in a slow and long TOF1, operating at low ion energy (1 to l00eV), and fragment ions are mass analyzed in a fast and short TOF2, operating at much higher keV energy. Low energy fragmentation cell between TOF1 and TOF2 is tailored to accelerate fragmentation and dampening steps, mostly by shortening the cell and employing higher gas pressure. Since separation in TOF1 takes milliseconds and mass analysis in TOF2- microseconds, the invention provides comprehensive MS-MS analysis of multiple precursor ions per single ion pulse. Slow separation in TOF1 becomes possible with an introduction of novel TOF1 analyzers. The TOF-TOF could be implemented using a static TOF1, here described on the examples of spiratron, planar and cylindrical multi-pass separators with griddles spatial focusing ion mirrors. Higher performance is expected with the use of novel hybrid TOF 1 analyzers, combining radio frequency (RF) and quadratic DC fields. RF field retains low-energy ions within TOF 1 analyzer, while quadratic DC field improves resolution by compensate for large relative energy spread.
Owner:力可公司
Mass spectrometer
ActiveUS20050230611A1Eliminate the effects ofReduce removalParticle separator tubesIsotope separationMass Spectrometry-Mass SpectrometryMass analyzer
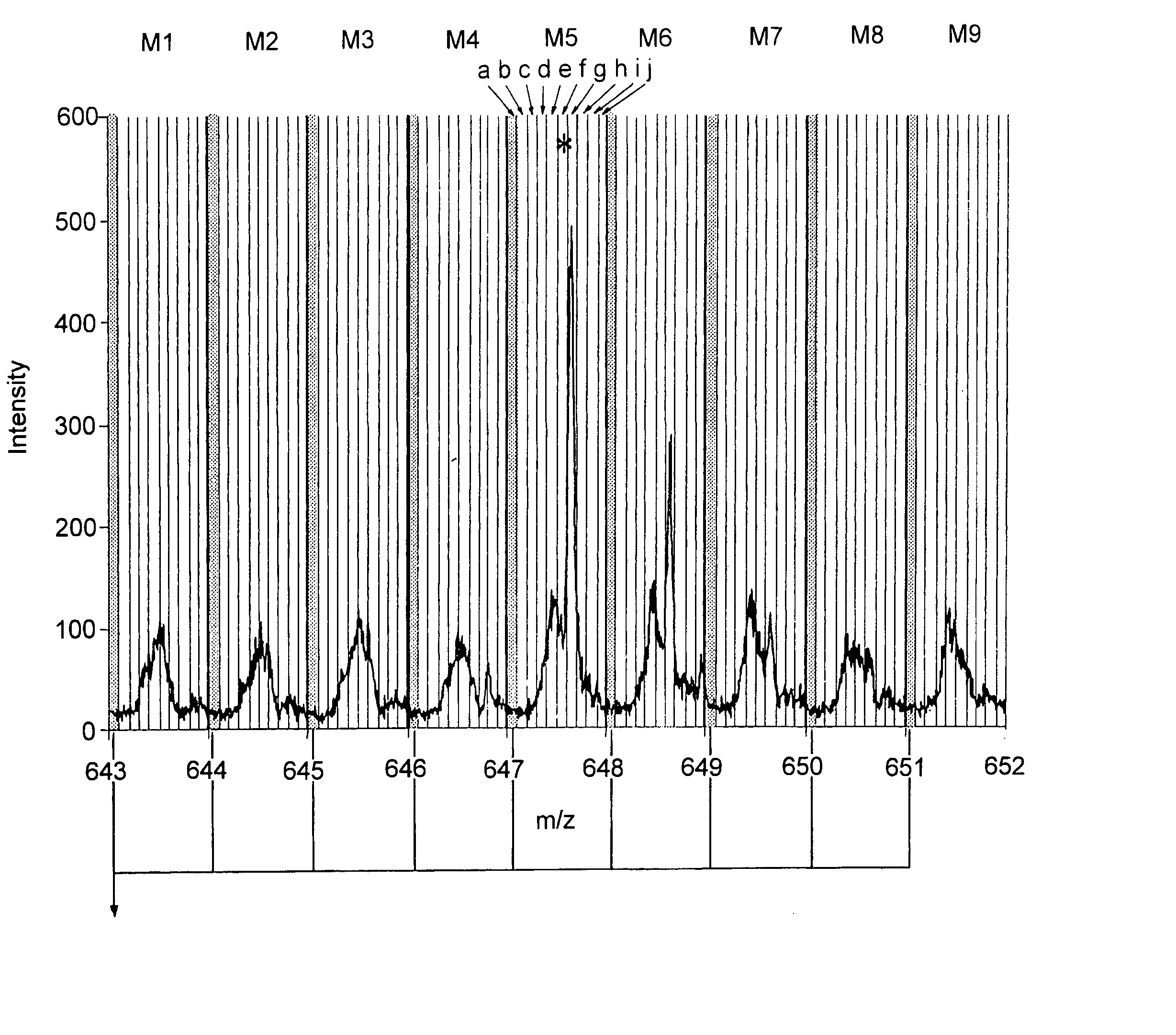
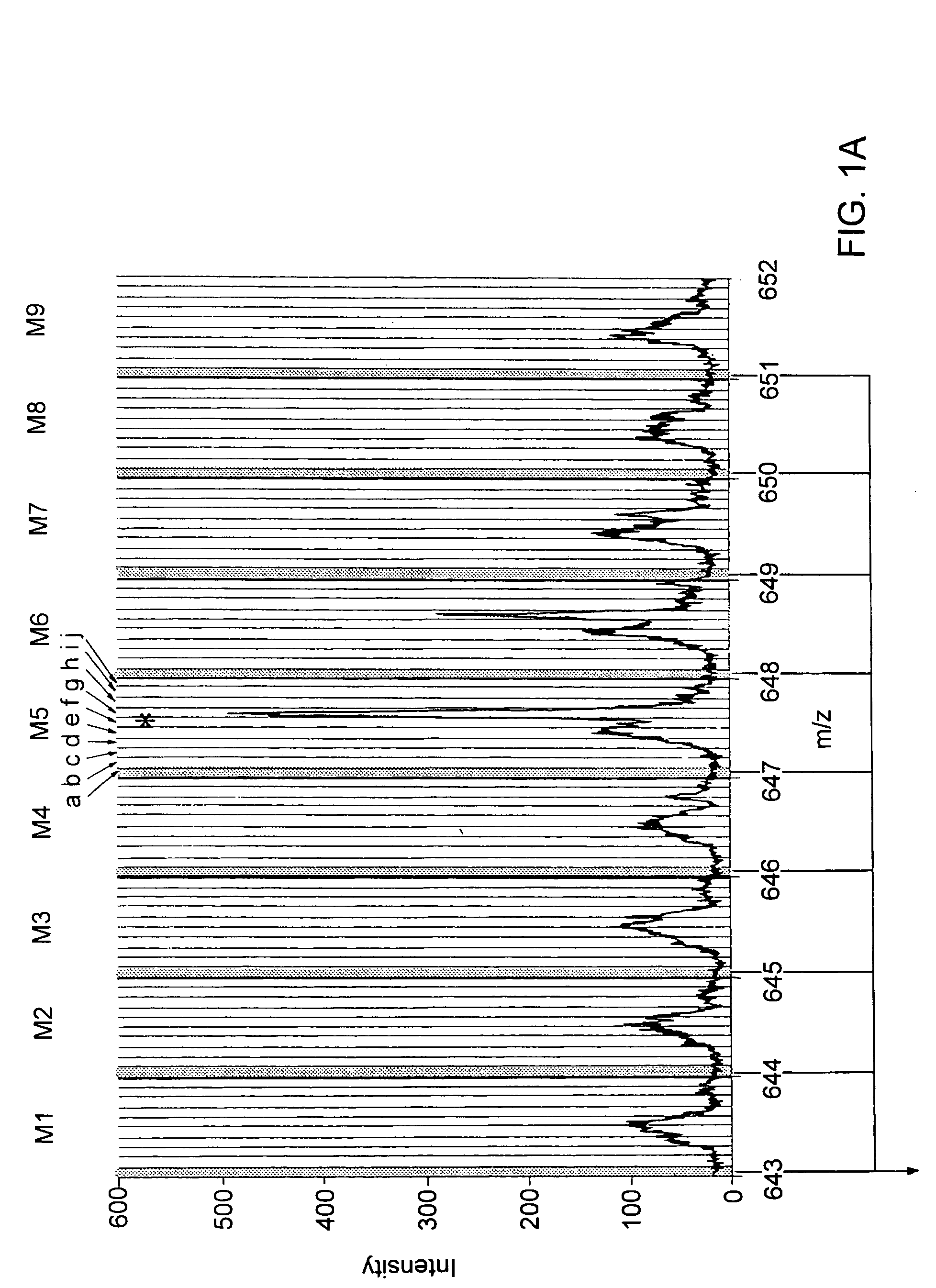
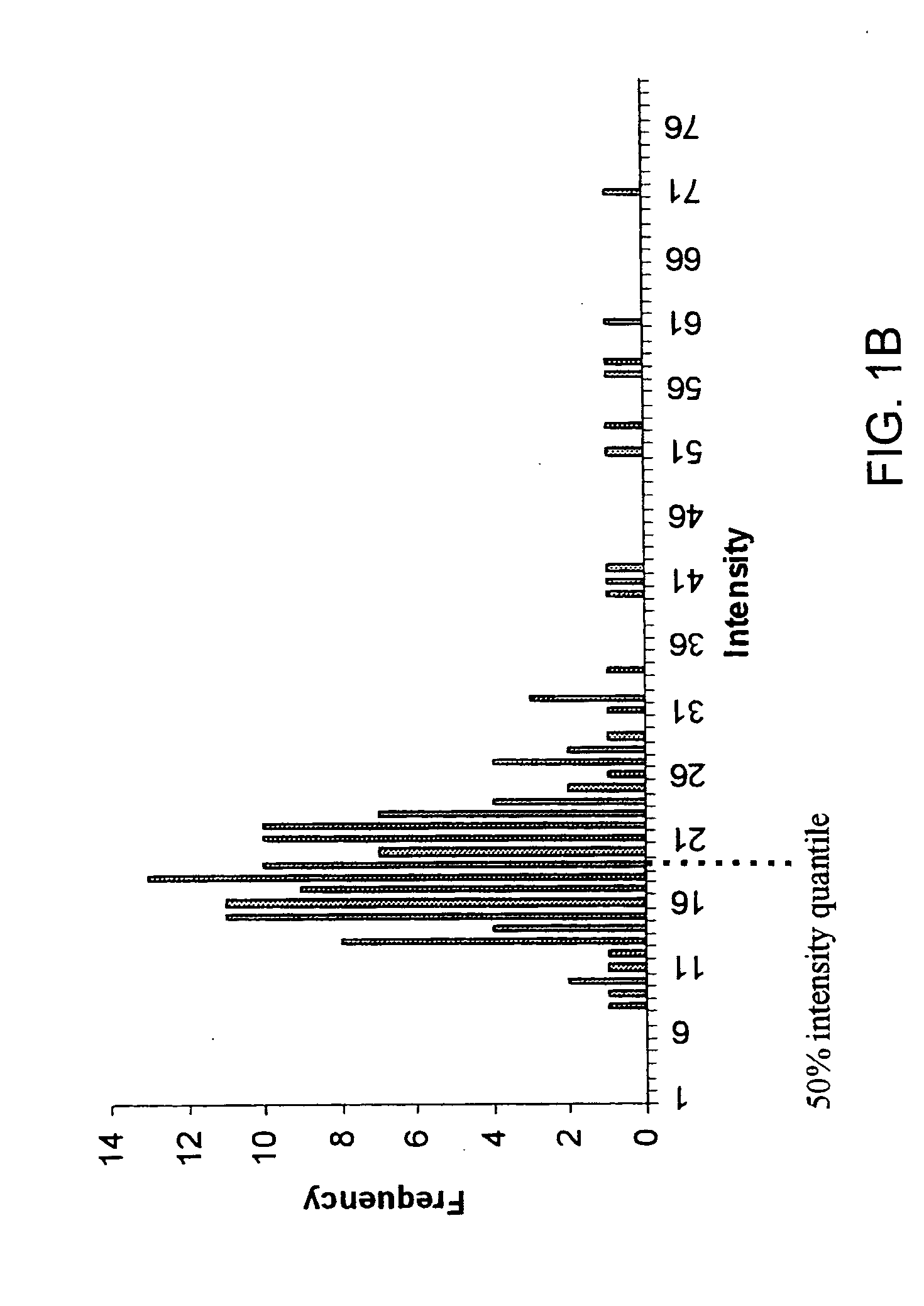
A mass spectrometer and a method of mass spectrometry are disclosed wherein periodic background noise is effectively filtered out from the mass spectral data. An overall mass window is superimposed upon the mass spectral data. The overall mass window preferably comprises 21 nominal mass windows each preferably having a width of 1.0005 amu. Each nominal mass window preferably comprises 20 channels. An intensity distribution relating to all the first channels of the 21 nominal mass windows is determined. An intensity quantile is determined from the intensity distribution. The intensity quantile is taken to represent the background intensity in the first channel of the central nominal mass window. This process is repeated for the other channels so that the background intensity across the whole of the central nominal mass window is estimated and then subtracted from the raw mass spectral data comprising the central nominal mass window. The overall mass window is then preferably advanced approximately 1 amu and the process is repeated multiple times.
Owner:MICROMASS UK LTD
Mass chromatograph
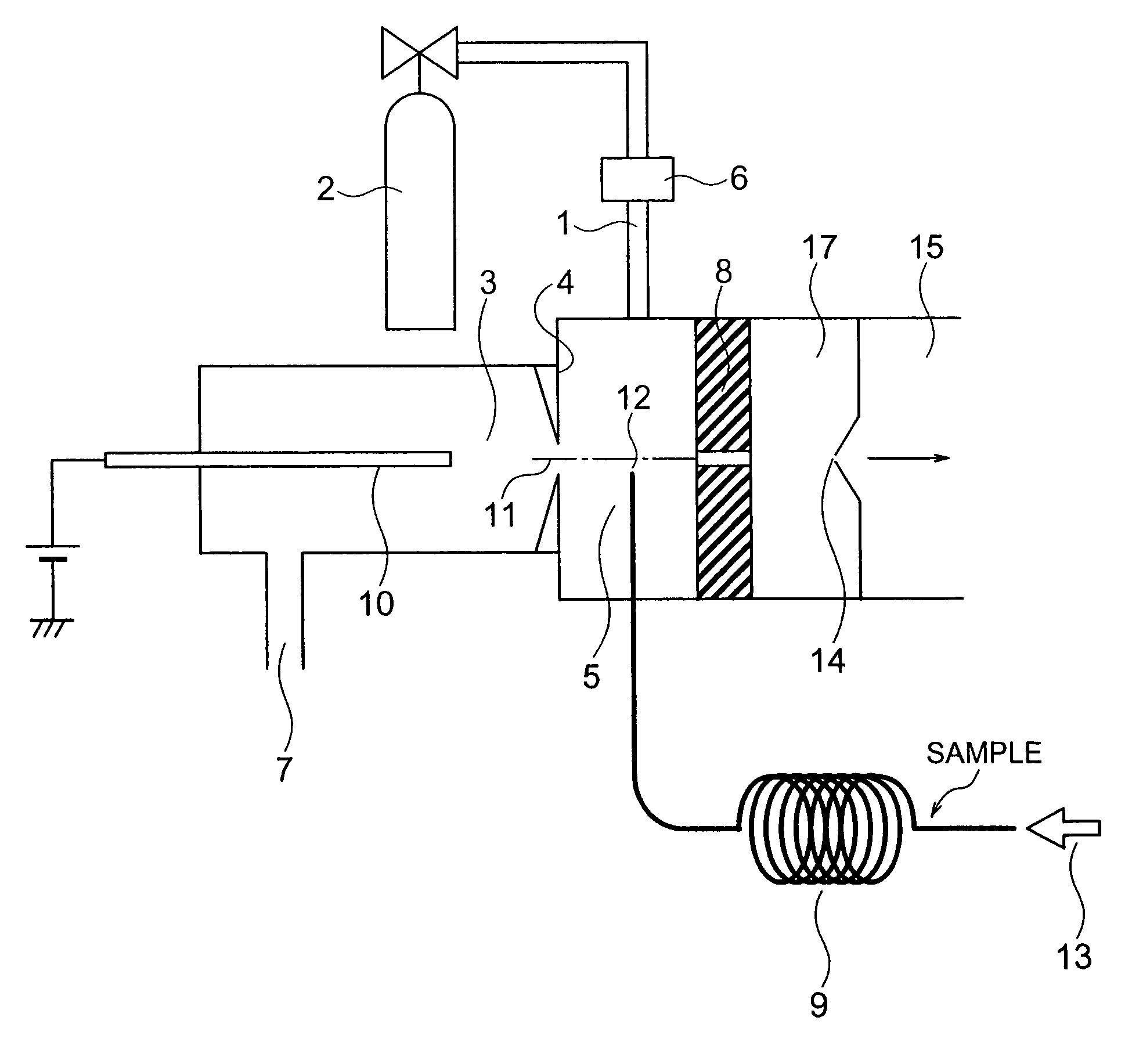
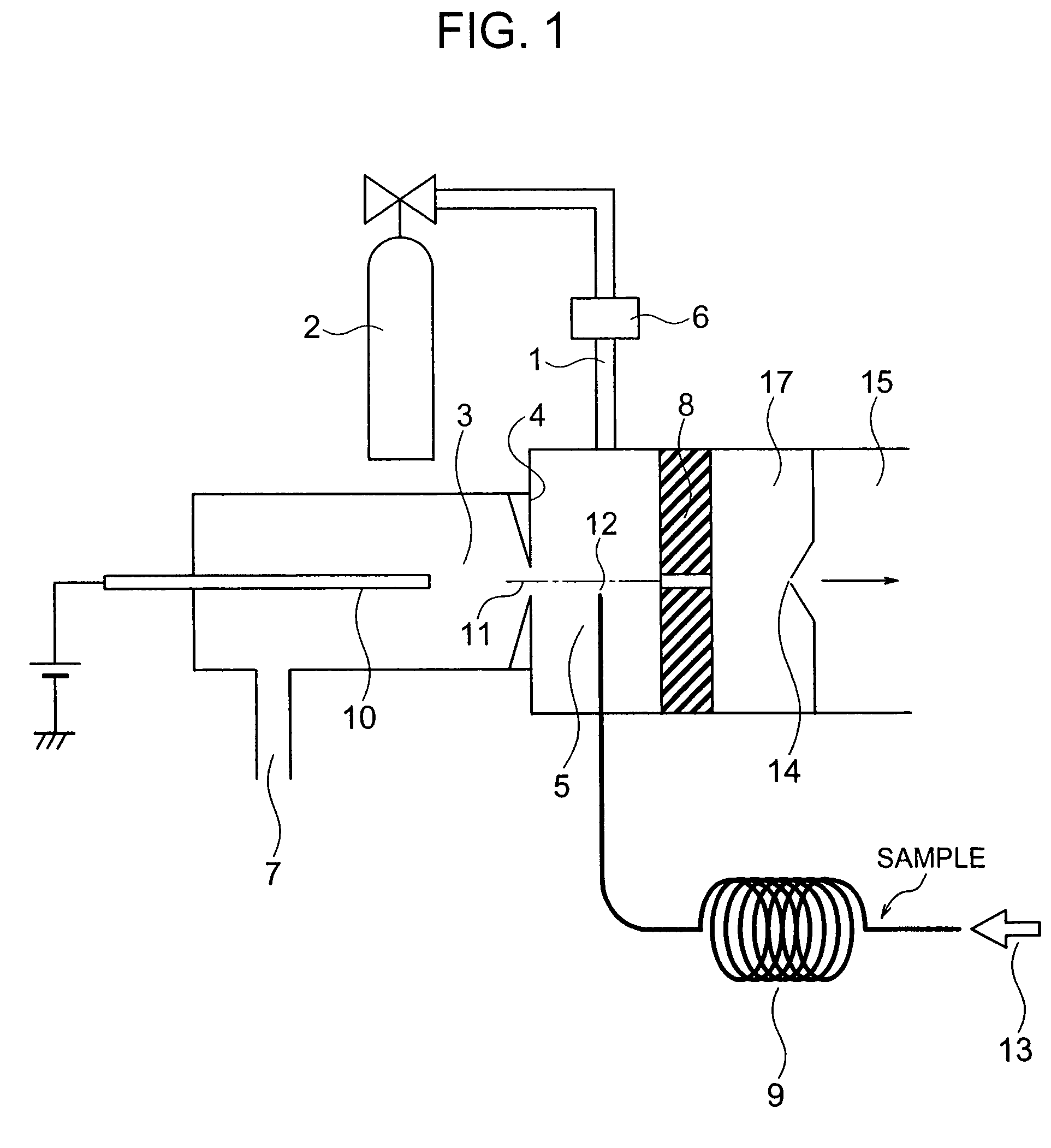
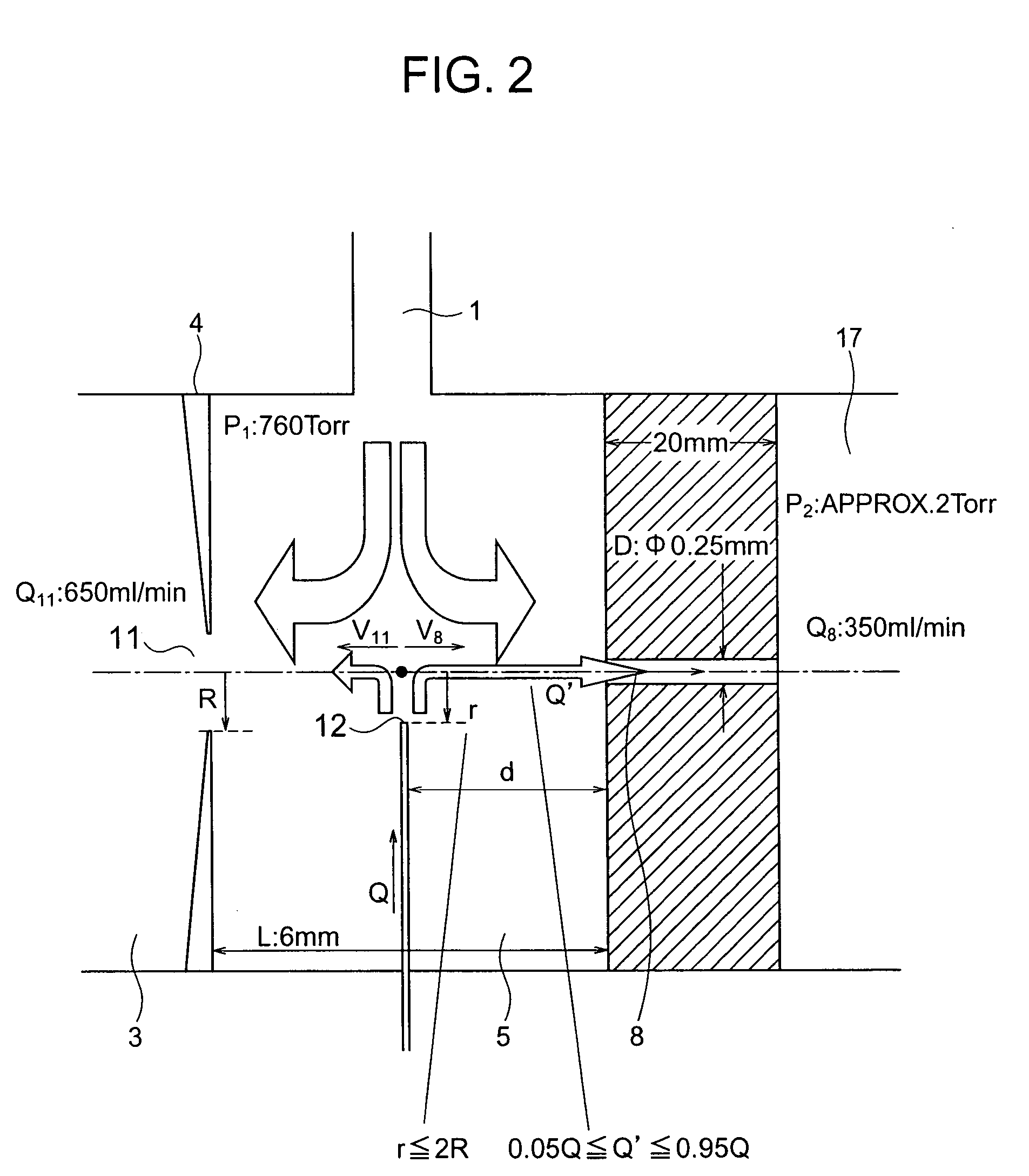
ActiveUS7064320B2Easily contaminatedHigh sensitivity analysisComponent separationMaterial analysis by electric/magnetic meansMass analyzerIonization
A mass spectrometer has an atmospheric pressure chemical ionization source that includes a first ionization portion including a needle electrode, for generating a primary ion by discharge of the needle electrode a primary ion introduction aperture, and a second ionization portion including an introduction aperture, for generating a sample ion by the reaction between the primary ion and the sample gas introduced from an end of a column of gas chromatography, a sample ion movement aperture; and a mass analysis portion; wherein the end is arranged at a position satisfying the relation r≦2R where R is a radius of an inner diameter of the aperture and r is a distance between an axis connecting the center of the aperture and the center of the aperture, and the end.
Owner:HITACHI LTD
Method for cascade mass spectrometry by using multiple ion traps
InactiveCN101320016AHigh sensitivityImprove analysis efficiencyMaterial analysis by electric/magnetic meansMass spectrometersQuadrupole fieldIon trap mass spectrometry
Owner:FUDAN UNIV +1
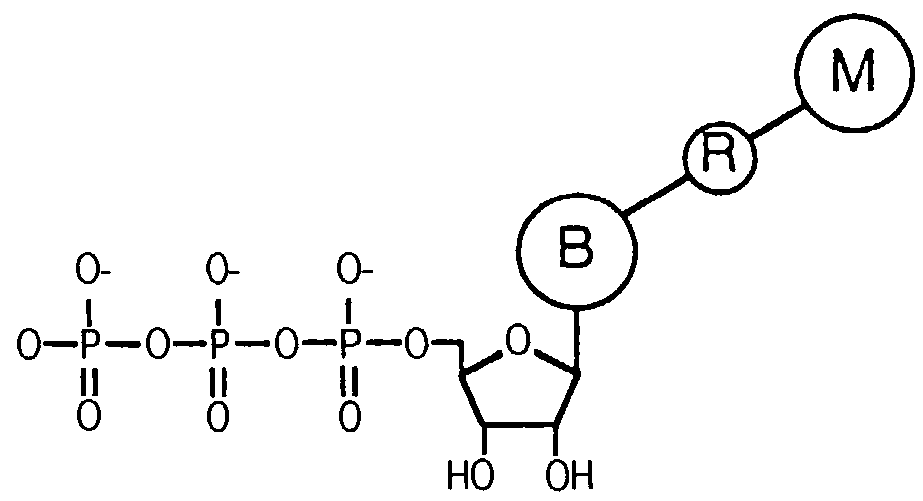
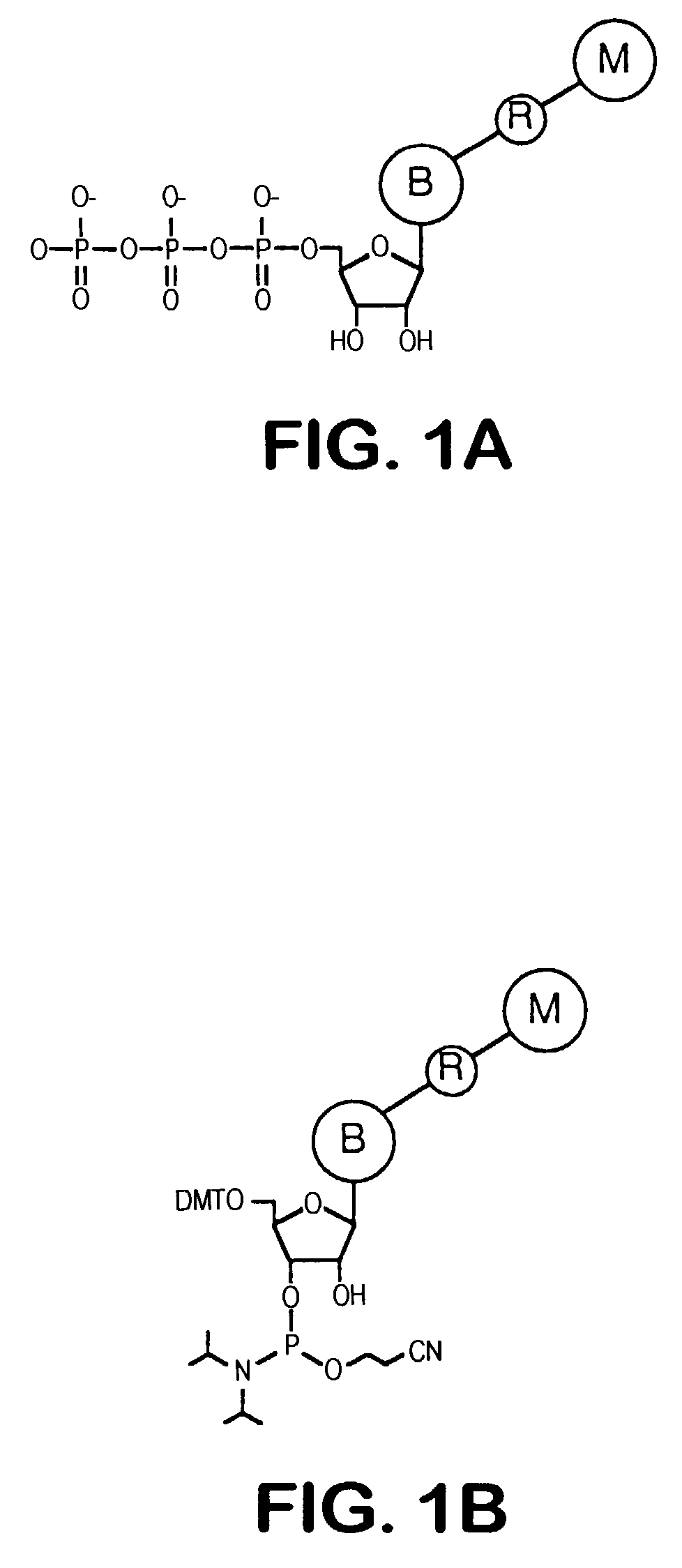
Releasable nonvolatile mass-label molecules
InactiveUS7132519B2Enhanced detection signalLow backgroundNanotechSugar derivativesMass spectrometricReagent
Releasable tag reagents for use in the detection and analysis of target molecules, particular in mass spectrometric analyses are provided. Also provided are methods of detection that employ releasable tag reagents.
Owner:AGENA BIOSCI
System and Methods for Ionizing Compounds using Matrix-assistance for Mass Spectometry and Ion Mobility Spectometry
ActiveUS20130306856A1Facilitated ionizationLow costSamples introduction/extractionMaterial analysis by electric/magnetic meansVacuum assistedGas phase
An ionization method for use with mass spectrometry or ion mobility spectrometry is a small molecule compound(s) as a matrix into which is incorporated analyte. The matrix has attributes of sublimation or evaporation when placed in vacuum at or near room temperature and produces both positive and negative charges. Placing the sample into a region of sub-atmospheric pressure, the region being in fluid communication with the vacuum of the mass spectrometer or ion mobility spectrometer, produces gas-phase ions of the analyte for mass-to-charge or drift-time analysis without use of a laser, high voltage, particle bombardment, or a heated ion transfer region. This matrix and vacuum assisted ionization process can operate from atmosphere or vacuum and produces ions from large (e.g. proteins) and small molecules (e.g. drugs) with charge states similar to those observed in electrospray ionization.
Owner:MSTM LLC
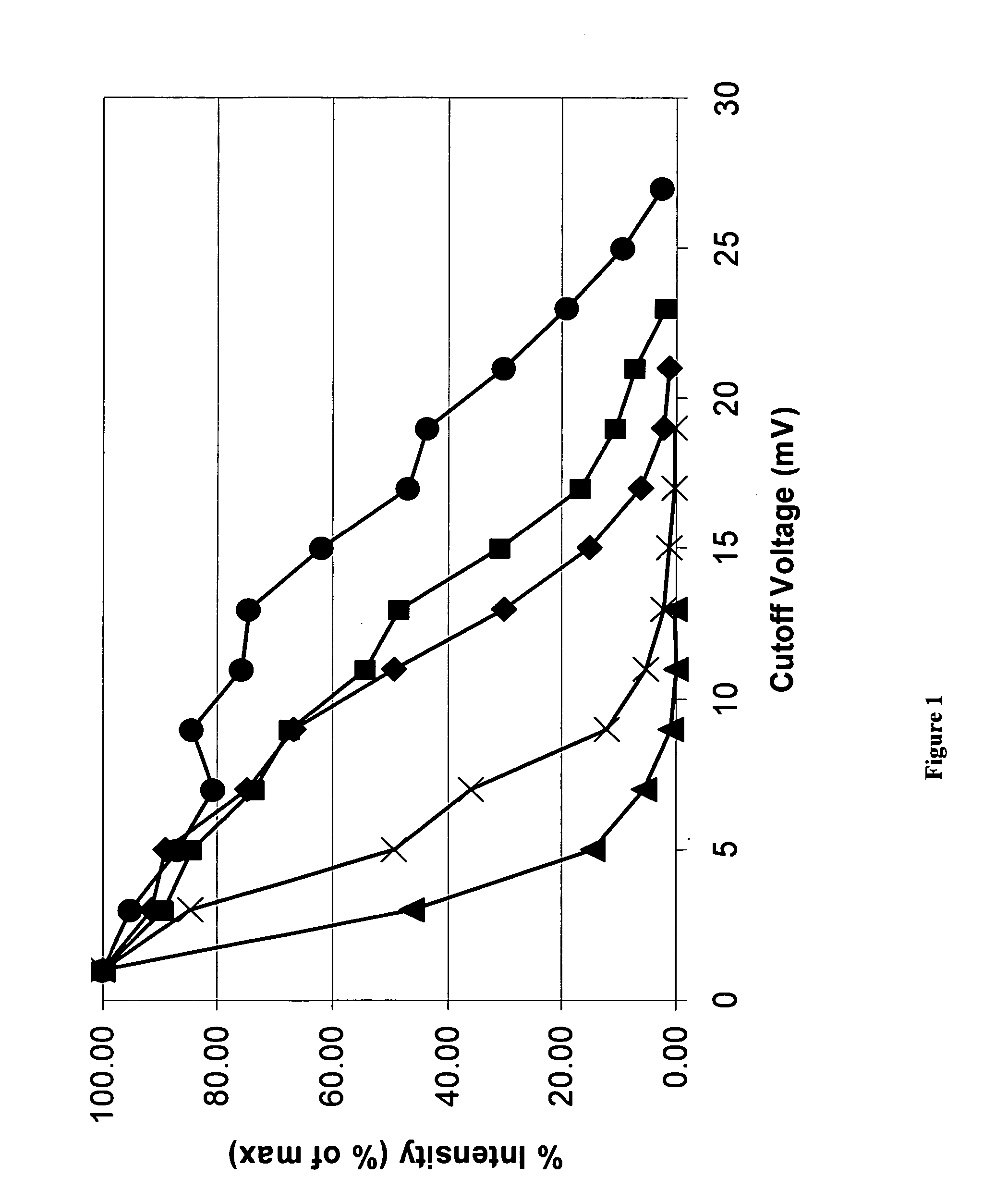
Mass spectrometry with selective ion filtration by digital thresholding
InactiveUS20050270191A1High levelStability-of-path spectrometersTime-of-flight spectrometersMass spectrum analysisMass analysis
The methods described herein generally relate to characterization of large analytes, such as biomolecules, by molecular mass analysis. Specifically, the methods are directed to molecular mass analysis of singly- or multiply-charged ions by selective ion filtering carried out by a digital thresholding process.
Owner:IBIS BIOSCI
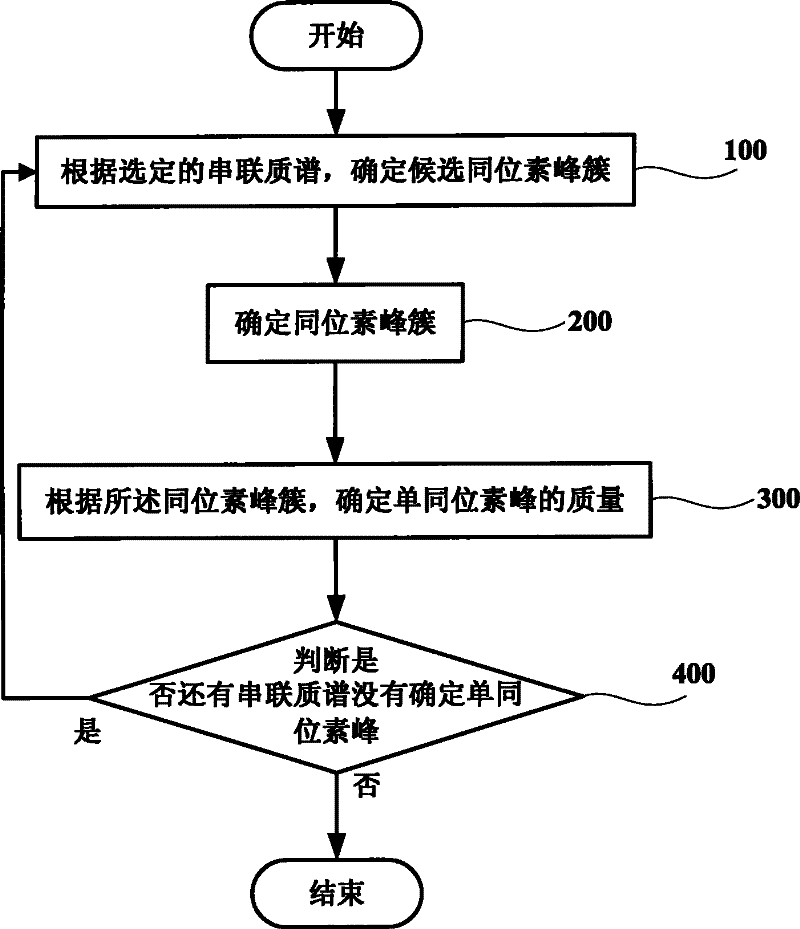
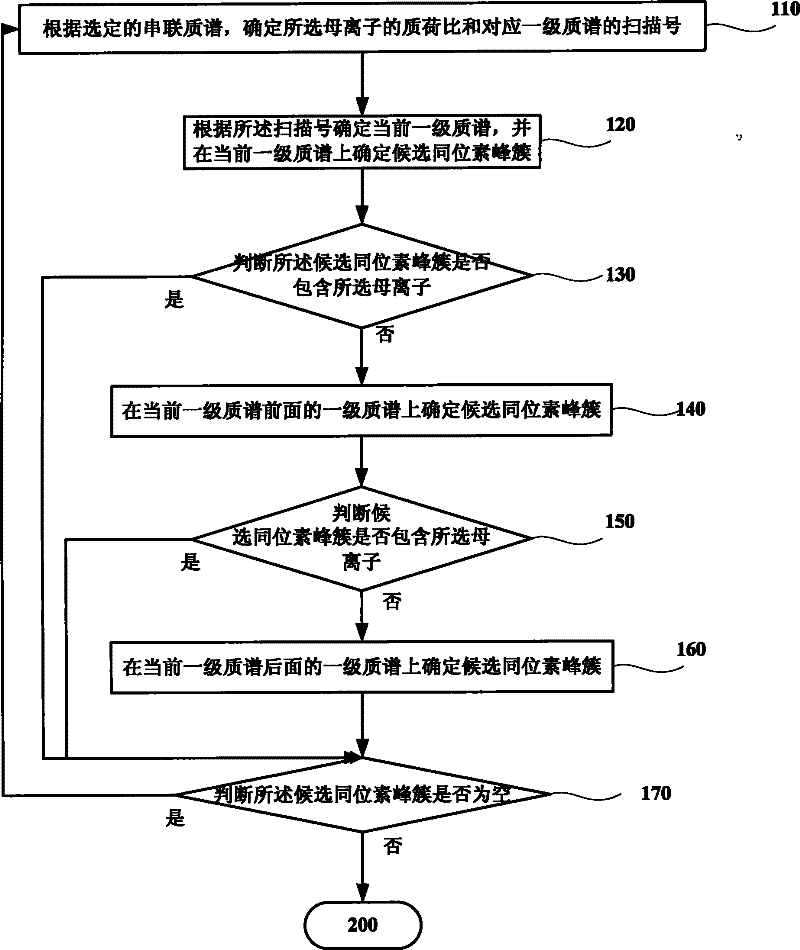
Method and system for increasing judgment accuracy of monoisotopic peaks
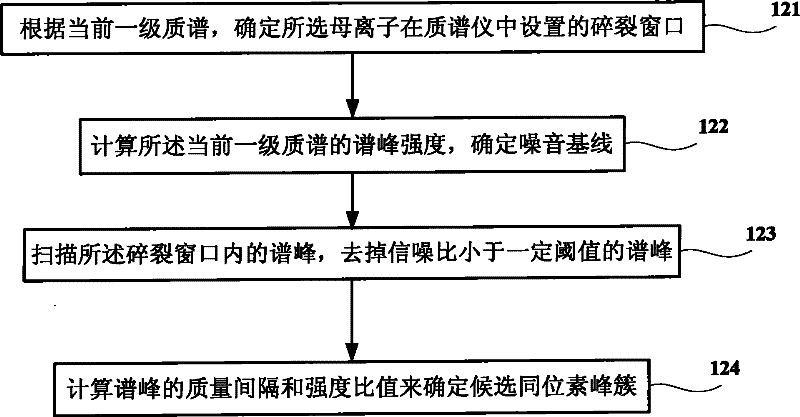
ActiveCN102445544AReduce mismatchSolve the problem of inaccurate intensity distributionMaterial analysis by electric/magnetic meansBiological testingComputer scienceTandem mass spectrometry
The invention discloses a method and system for increasing judgment accuracy of monoisotopic peaks. The method comprises the following steps: according to selected tandem mass spectrums, determining candidate isotopic peak clusters; according to the similarity and the strength ratio of the chromatographic outflow curves of the candidate isotopic peak clusters, determining the start and stop of the isotopic peak clusters; according to the start and stop of the isotopic peak clusters, determining the quality of the monoisotopic peaks; and judging whether the monoisotopic peaks of the tandem mass spectrums are not determined or not, if yes, returning and selecting a new tandem mass spectrum to determine a candidate isotopic peak cluster, and if not, ending.
Owner:INST OF COMPUTING TECH CHINESE ACAD OF SCI