Patents
Literature
865 results about "BALB/c" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
BALB/c is an albino, laboratory-bred strain of the house mouse from which a number of common substrains are derived. Now over 200 generations from New York in 1920, BALB/c mice are distributed globally, and are among the most widely used inbred strains used in animal experimentation.
Human SARS-CoV-2 monoclonal antibody and preparation method and application thereof
PendingCN111153991AHigh affinityStrong specificitySsRNA viruses positive-senseVirus peptidesBALB/cMonoclonal antibody agent
The invention discloses a human SARS-CoV-2 monoclonal antibody. The preparation method of the human SARS-CoV-2 monoclonal antibody comprises the steps: adopting SARS-CoV Nucleocapsid recombinant protein as immunogen, immunizing BALB / c mice, performing fusion and subcloning on spleen cells and myeloma cells of mice, then performing a large amount of repeated screening and domestication of cell lines through commercialized products SARS-CoV-2 Nucleocapsid and MERS Nucleocapsid so as to obtain a hybridoma cell line capable of secreting the SARS-CoV-2-resistant N monoclonal antibody with high affinity and high specificity finally and successfully, and finally performing ascites preparation and purification so as to obtain the monoclonal antibody, wherein the amino acid sequence of the SARS-CoVNucleocapsid recombinant protein is shown in SEQ ID No. 1. The invention also discloses application of the monoclonal antibody in preparation of SARS-CoV-2 virus detection products and preparation ofdrugs for inhibiting the SARS-CoV-2 viruses. The monoclonal antibody can be used for detecting the SARS-CoV-2 in human throat swabs / pulmonary secretions and other samples by using a double-antibody sandwich method, and can be applied to diagnosis and prevention and control of SARS-CoV-2 virus infection and scientific researches of viruses and other study.
Owner:BEIJING BIOSYNTHESIS BIOTECH
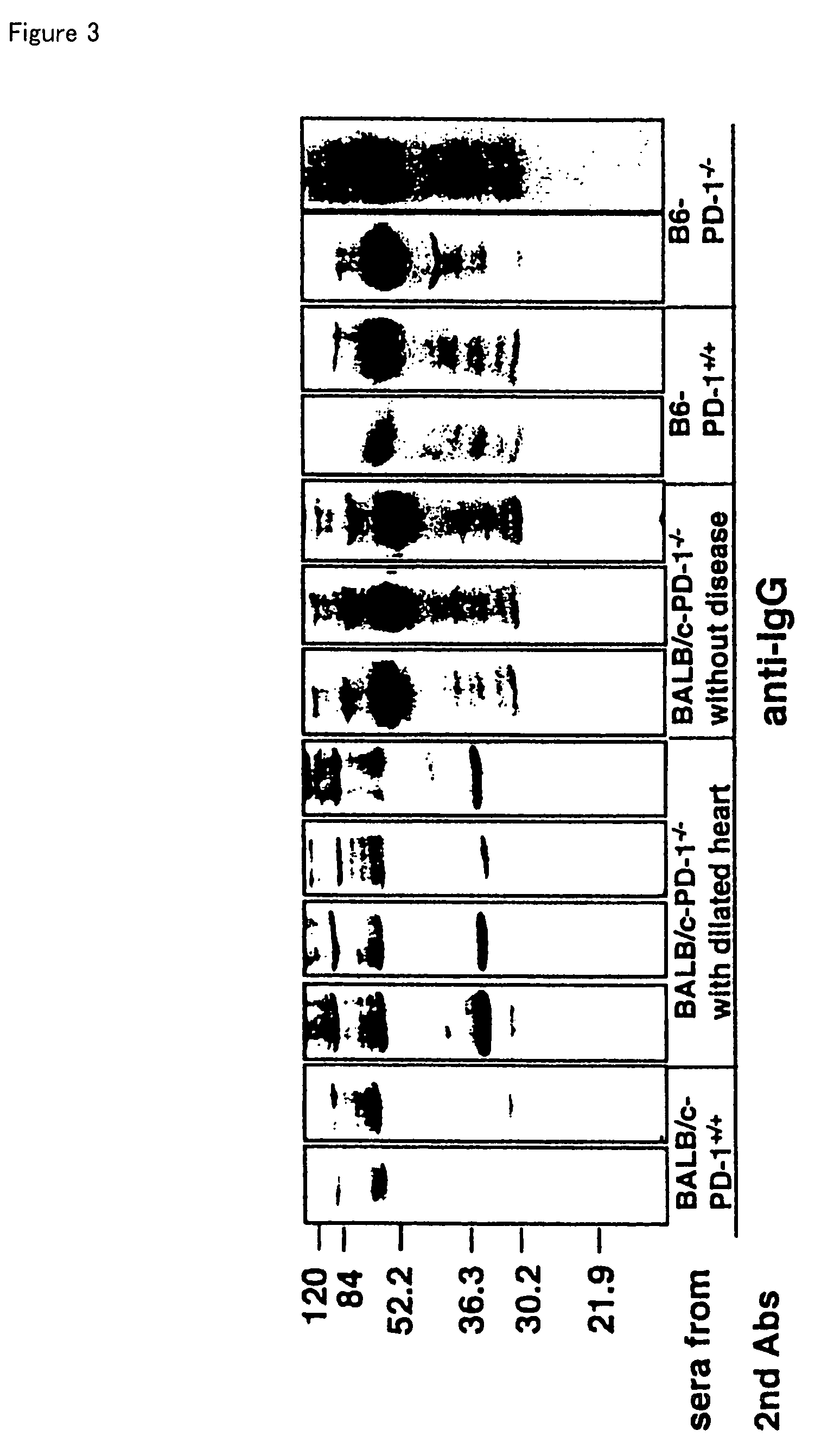
PD-1-lacking mouse and use thereof
InactiveUS7414171B2Immunoglobulin superfamilyMicrobiological testing/measurementBALB/cAutoimmune disease
The invention relates to BALB / c mice that are deficient in the programmed cell death-1 receptor (PD-1), a screening method for autoimmune disease medicine using these mice, an IgG self-reactive antibody that the mice produce, a protein that specifically reacts to the antibody and is produced in heart, and a diagnostic method for dilated cardiomyopathy using the protein. Because PD-1 deficient BALB / c mice spontaneously develop autoimmune disease, and specifically dilated cardiomyopathy, they are useful for screening for medicines against these diseases.
Owner:ONO PHARMA CO LTD +1
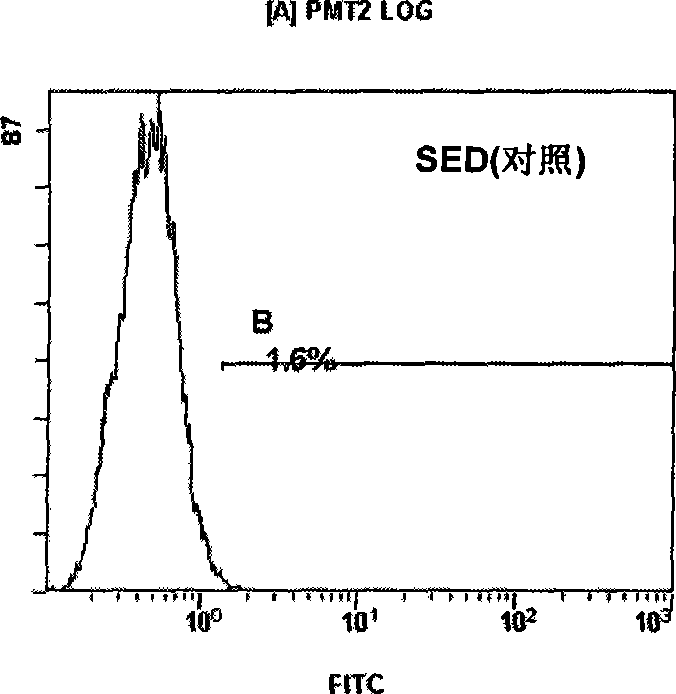
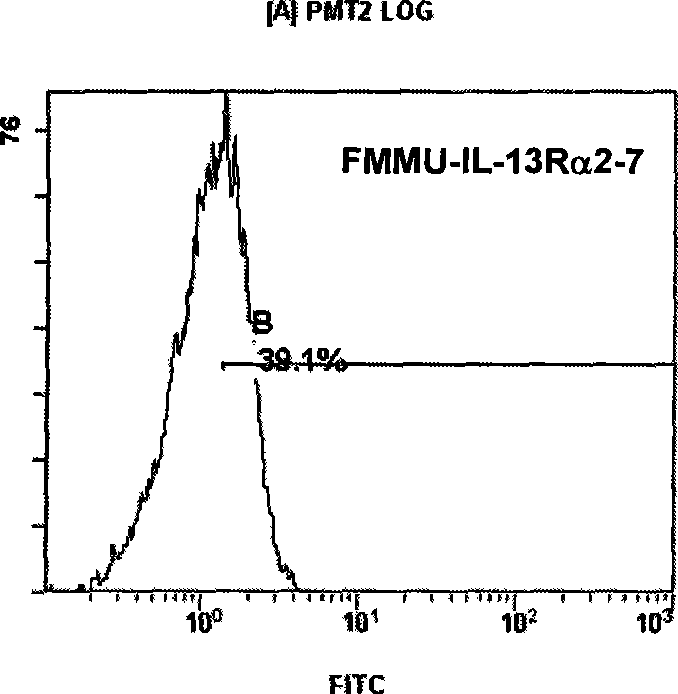
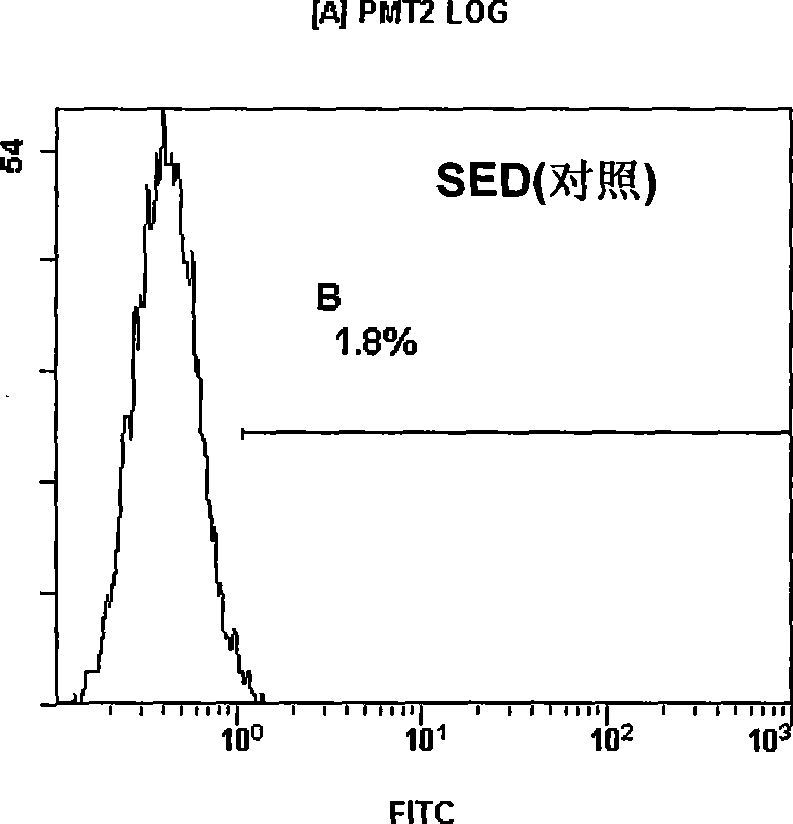
Variable regions of heavy chain and light chain of antihuman IL-13R alpha 2 monoclonal antibody
InactiveCN101440130AHigh medicinal valueImmunoglobulins against animals/humansAntibody ingredientsBALB/cSingle-Chain Antibodies
The invention discloses a variable region of heavy and light chains of an anti-human IL-13R alpha 2 single clone antibody. The invention uses recombinant human IL-13R alpha 2 to immunize a BALB / c rat to prepare a group of rate anti-human IL-13R alpha 2 single clone antibody and screen anti-human IL-13R alpha 2 single clone antibody FMMU-IL-13R alpha 2-7 with high affinity. The variable region genes of heavy and light chains of the single clone antibody are cloned to obtain the sequences of genes and amino acid in the variable region of the heavy and light chains of the single clone antibody, and confirm the uniqueness of the gene and protein sequences. The amino acid sequence of the variable region and the gene sequences encoding the variable zones have great potential application value in single chain antibody, chimeric antibody, humanized antibody or vaccine for treating malignancy with human IL-13R alpha 2 as the target.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
Heavy chain and light chain variable regions of anti-human BAFF monoclonal antibody
The invention discloses heavy chain and light chain variable regions of an anti-human BAFF monoclonal antibody. The anti-human BAFF monoclonal antibody is FMMU-BAFF-4, wherein the gene sequences of the monoclonal antibody variable regions are shown as SEQ ID NO.3 and SEQ ID NO.4; and the amino acid sequences of the monoclonal antibody variable regions are shown as SEQ ID NO.1 and SEQ ID NO.2. In the variable regions, a mouse anti-human BAFF monoclonal antibody is prepared by using a recombined human BAFF immunological BALB / c mouse; the mouse anti-human BAFF monoclonal antibody is cloned and screened to obtain a hybridoma cell line which can secrete specific human BAFF monoclonal antibody FMMU-BAFF-NO.4; the genes of the monoclonal antibody light chain and heavy chain variable regions are cloned to obtain the gene sequences and the amino acid sequences of the monoclonal antibody light and heavy chain variable regions, and CDR sequences of the variable regions; and the uniqueness of the amino acid sequences and the gene sequences is determined.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
Variable region gene of high affinity monoclonal antibody of tumor necrosis factor and its preparation
The invention discloses a process for preparing variable zone genes of high affinity tumor necrosis factor (TNF) resistant monoclonal antibody (F6 mAb) comprising, using recombinated human TNF immune BALB / c mouse to prepare a plurality of mouse Anti-TNF monoclonal antibody, screening high affinity F6 mAb using indirect ELISA method. By cloning the monoclonal antibody light chain and heavy chain variable zone genes, the monoclonal antibody light chain and heavy chain variable zone gene sequence and amino acid sequence can be obtained, and the unicity of the gene sequence and protein sequence can be confirmed.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
Hybridoma cell line of monoclonal antibody against African swine fever virus and secreted monoclonal antibody thereof
InactiveCN101831407AHigh utility valueMicroorganism based processesImmunoglobulins against virusesBALB/cPurification methods
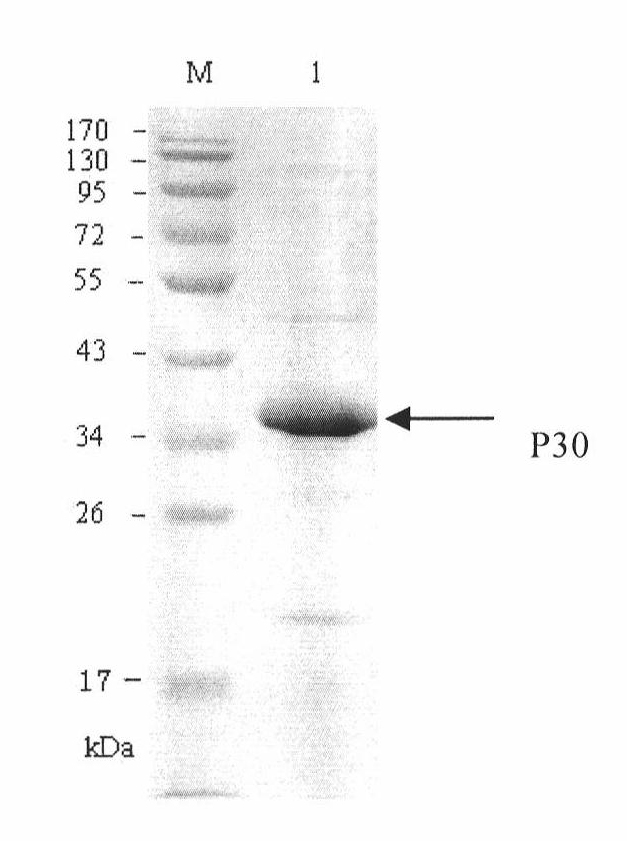
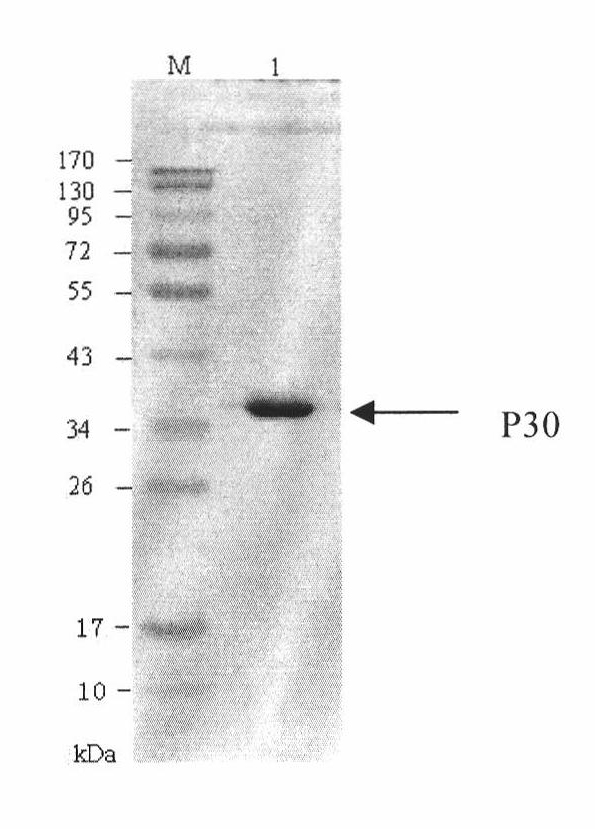
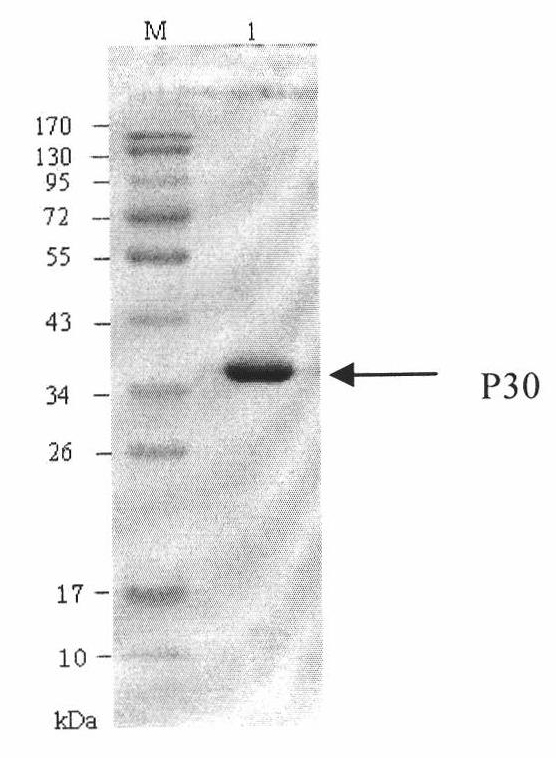
The invention discloses a hybridoma cell line of a monoclonal antibody against African swine fever virus and the secreted monoclonal antibody thereof. The preparation method of the invention comprises the following steps: preparing a recombined P30 soluble antigen by prokaryotic expression; immunizing a BALB / c mouse; and finally fusing, screening and cloning by a hybridoma technology to obtain the hybridoma cell line which can stably secrete the monoclonal antibody against African swine fever virus P30 protein. The invention further discloses a method for preparing the monoclonal antibody with the cell line, an antibody purification method and a labeling method for horseradish peroxidase of the antibody. The monoclonal antibody can be used in detecting the African swine fever viral antibody in pig serum.
Owner:ANIMAL & PLANT & FOOD INSPECTION CENT OF TIANJIN ENTRY EXIT INSPECTION & QUARANTINE BUREAU
Preparation method for PCV-II Cap protein monoclonal antibody, antibody and application
InactiveCN101768218AAvoid distortionThe ability to secrete antibodies is strong and stableImmunoglobulins against virusesFluorescence/phosphorescenceBALB/cIndirect elisa
The invention discloses a preparation method for a PCV-II Cap protein monoclonal antibody, an antibody and application. The invention adopts ultracentrifuged and purified PCV-II as an immunogen to immunize a BALB / c mouse by the conventional method, takes spleen cells of the immunized BALB / c mouse to fuse with SP2 / 0 cells, obtains two strains of hybridoma cells secreting the PCV2-Cap protein monoclonal antibodies by indirect ELISA screening, respectively names the two strains of hybridoma cells as 8-60 and 10-48, identifies biological characteristics of the two strains 8-60 and 10-48, and usesthe two strains 8-60 and 10-48 as the first antibodies to establish an indirect immunofluorescence diagnostic method. The result of the indirect immunofluorescence diagnostic method is basically consistent with that of the PCR diagnostic method, and the positive and negative coincidence rates are respectively 93.75 percent and 100 percent so as to provide reference for preventing and treating theporcine circovirus disease.
Owner:INST OF ANIMAL HUSBANDRY & VETERINARY FUJIAN ACADEMY OF AGRI SCI +7
Kit for rapid detection of staphylococcus aureus in sample and detection method thereof
InactiveCN102323416ASuitable for mass inspectionAccurate detectionColor/spectral properties measurementsBALB/cStaphylococcus cohnii
The invention discloses a kit for rapid detection of staphylococcus aureus in a sample and a detection method thereof, belonging to the technical field of immunological detection. In the invention, a staphylococcus aureus immunogen inactivated with formaldehyde is used for immunizing a healthy New Zealand rabbit to obtain a polyclonal antibody to serve as a coated antibody, and used for immunizing a BALB / C mouse and performing cell fusion to obtain a monoclonal antibody to serve as a secondary antibody, thus, the kit for performing a double antibody sandwich enzyme-linked immunosorbent assay on the staphylococcus aureus in food (milk) is established, and a rapid and efficient detection means is provided for residual detection of the staphylococcus aureus in the food, and the advantages of lower cost and better stability and repeatability are achieved. A detection limit of the kit is 105cfu / mL and is suitable for detecting mass samples.
Owner:JIANGNAN UNIV
Schistosoma japonicum recombinant multi-epitope antigens, method for expressing and purifying same and application thereof
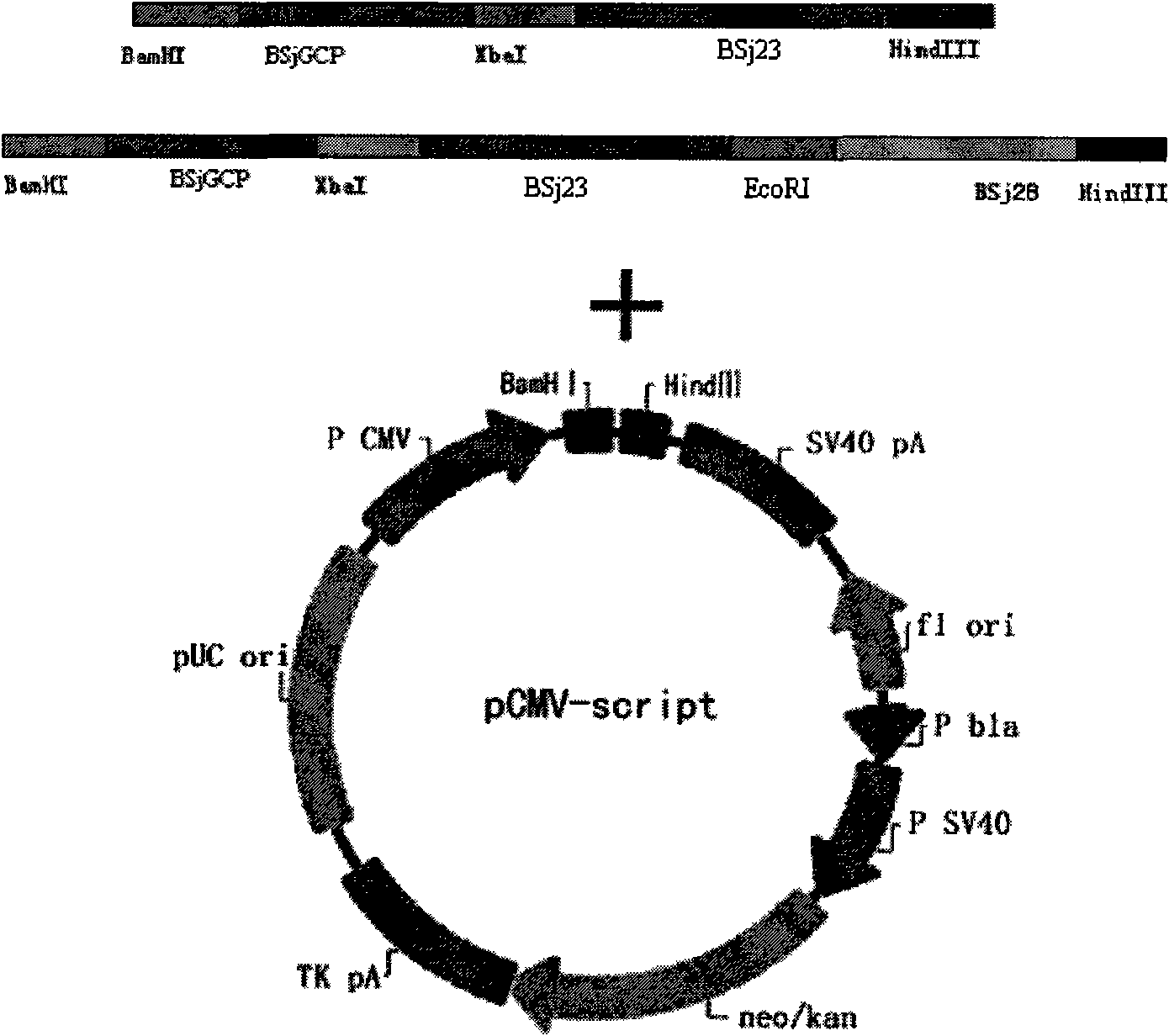
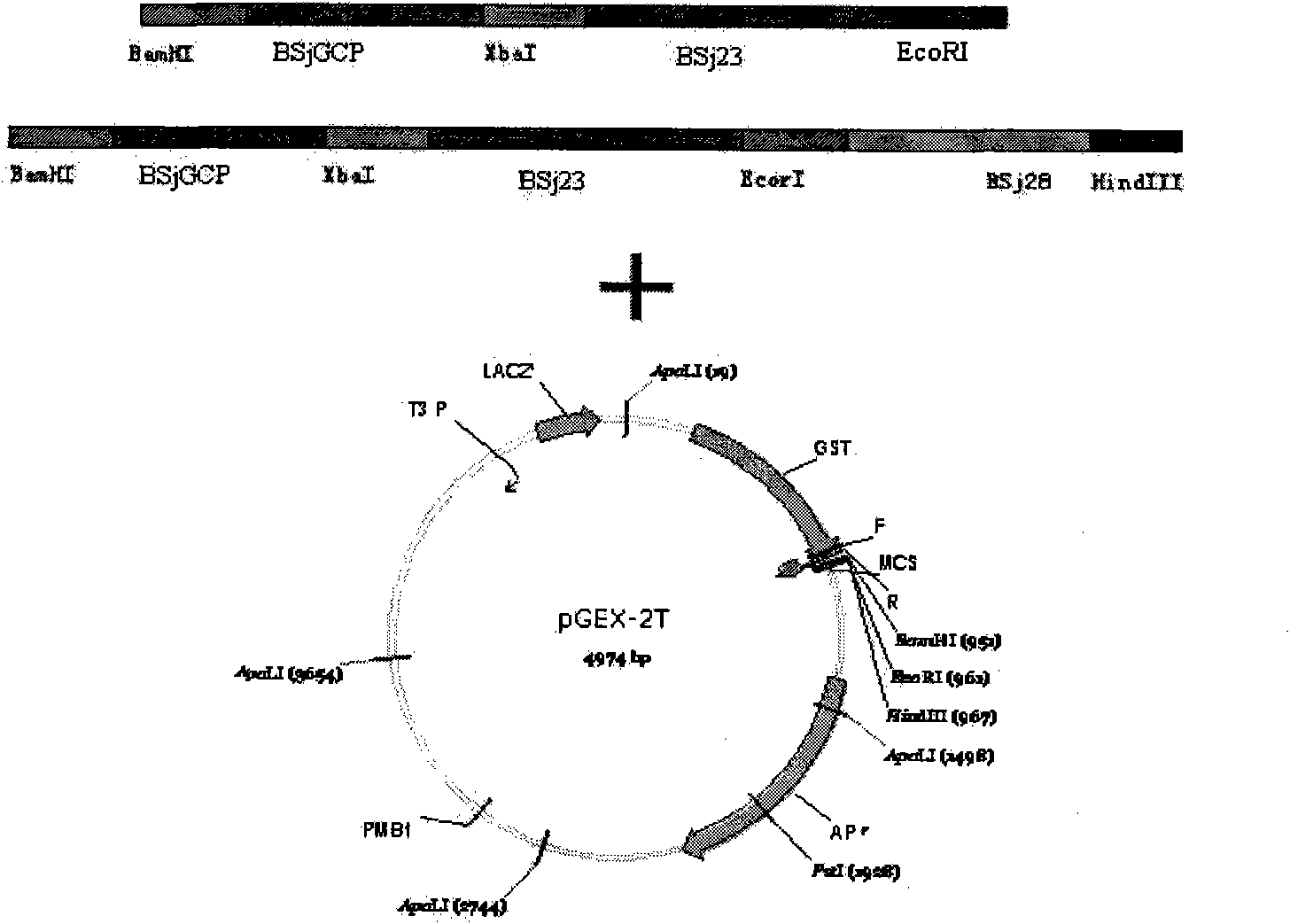
The invention discloses gene orders of schistosoma japonicum recombinant multi-epitope antigens BSjGCP-BSj23 and BSjGCP-BSj23-BSj28, a method for expressing and purifying the same, and application thereof in preparing schistosomiasis japonica immunity prevention vaccines and diagnostic reagents. Recombinant multi-epitope nucleic acid vaccines pCMV-BSjGCP-BSj23 and pCMV-BSjGCP-BSj23-BSj28 obtain 14.76 percent and 64.95 percent of worm reduction rates respectively in Kunming mice. The recombinant multi-epitope antigens pGEX-BSjGCP-BSj23 and pGEX-BSjGCP-BSj23-BSj28 obtain 15.7 percent and 57.99 percent of worm reduction rates in immunizing BalB / c mice, and obtain 91.0 percent and 89.9 percent of sensitivities as well as 97.8 percent and 93.4 percent of specificities respectively as diagnostic antigens.
Owner:SHANGHAI VETERINARY RES INST CHINESE ACAD OF AGRI SCI
Hybridoma cell strain capable of secreting monoclonal antibodies to quinolones and application of monoclonal antibodies thereof
InactiveCN102618502AStrong specificityHigh sensitivityTissue cultureImmunoglobulinsBALB/cAntibody types
The invention discloses a hybridoma cell strain capable of secreting monoclonal antibodies to quinolones and application of the monoclonal antibodies thereof. Ciprofloxacin (CIP) coupled with bovine serum albumin is used as an antigen to immunize BALB / c mice and cell fusion, screening and cloning are carried out so as to obtain one hybridoma cell strain 1F1 capable of stable passage and secretion of monoclonal antibodies (MAb) to quinolones, wherein, the accession number of the hybridoma cell strain 1F1 is CGMCC No. 5608. The titres of ascitic fluids of the 1F1 monoclonal antibodies are up to 10<-7>, and the type and the subclass of the monoclonal antibodies are IgG1 and kappa chain. According to indirect competitive ELISA analysis, the 1F1 monoclonal antibodies perform specific reactions to quinolones like ciprofloxacin, enrofloxacin, ofloxacin, danofloxacin, norfloxacin, enoxacin, marbofloxacin, sarafloxacin and difloxacin. An ELISA method, a kit and test paper for detecting residual of quinolones in food are developed by using the 1F1 monoclonal antibodies.
Owner:ZHEJIANG UNIV
Compound probiotics and drug for treating ulcerative colitis and preparation method thereof
The invention discloses compound probiotics and drug for treating ulcerative colitis and a preparation method thereof. The compound probiotics comprises Lactobacillus acidophilus CGMCC10436 and Lactobacillus plantarum CGMCC9961, and the drug for treating the ulcerative colitis adopts the two strains of the probiotics in a combined mode. According to the compound probiotics and drug for treating the ulcerative colitis and the preparation method thereof, Balb / c mice serve as test objects to establish an ulcerative colitis model, and the effect of the drug for resisting the ulcerative colitis is researched by starting from the angles such as apparent states and pathological conditions of the tested mice. It is shown through the test results that the drug has a good effect on resisting the ulcerative colitis, relapse of symptoms does not occur in a short period, the intestinal mucosa damage caused by the ulcerative colitis can be well repaired, and the drug for treating the ulcerative colitis can be widely applied to the field of preclinical medicine.
Owner:NORTHEAST AGRICULTURAL UNIVERSITY
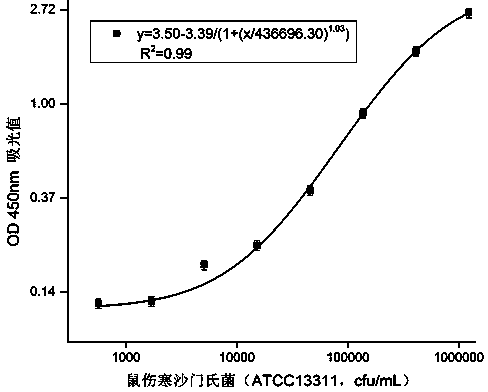
Double-antibody sandwich method for detecting salmonella typhimurium in food based on monoclonal antibodies
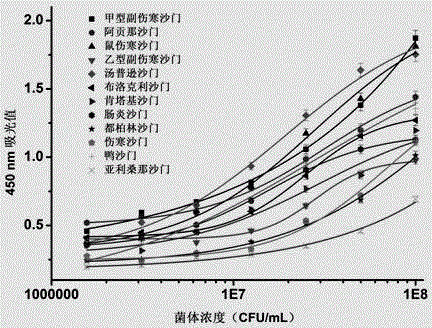
The invention discloses a double-antibody sandwich method for detecting salmonella typhimurium in food based on monoclonal antibodies, belonging to the technical field of immunoassay. Salmonella typhimurium ATCC13311 and smooth salmonella typhimurium LPS are adopted for mixed immunity of a 7-week BALB / c mouse, 10 LPS monoclonal antibodies are obtained by immunity, fusion and screening, horse radish peroxidases (HRP) are labeled respectively, and the salmonella typhimurium is paired two by two. A sandwich enzyme-linked immuno sorbent assay (ELISA) method is established by taking 6E2 CGMCC No.7206 monoclonal antibodies as coated antibodies and enzyme-labeled antibodies and by taking the salmonella typhimurium as standards, and the LOD is 500cfu / mL. The sandwich method, established by using the monoclonal antibodies which are highly uniform in physicochemical property and high in specificity and can be prepared on a large scale, is high in sensitivity and low in cost; the salmonella typhimurium is not in cross reaction with salmonella enteritidis, salmonella arizonae, E.coli, E.coliO157:H7, enterobacter sakazakii, staphylococcus aureus and listeria monocytogenes; a quick and efficient analysis way is provided for detection of the salmonella typhimurium in the food.
Owner:JIANGNAN UNIV
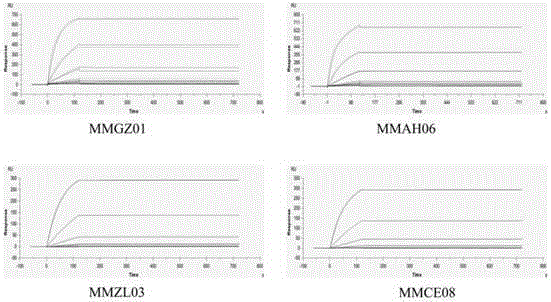
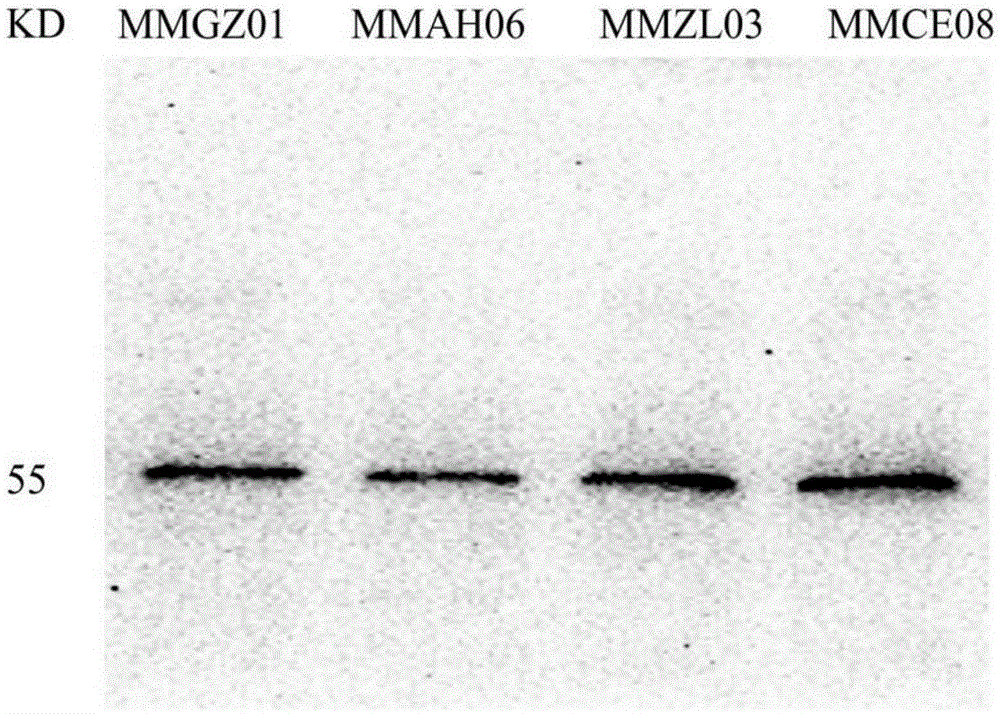
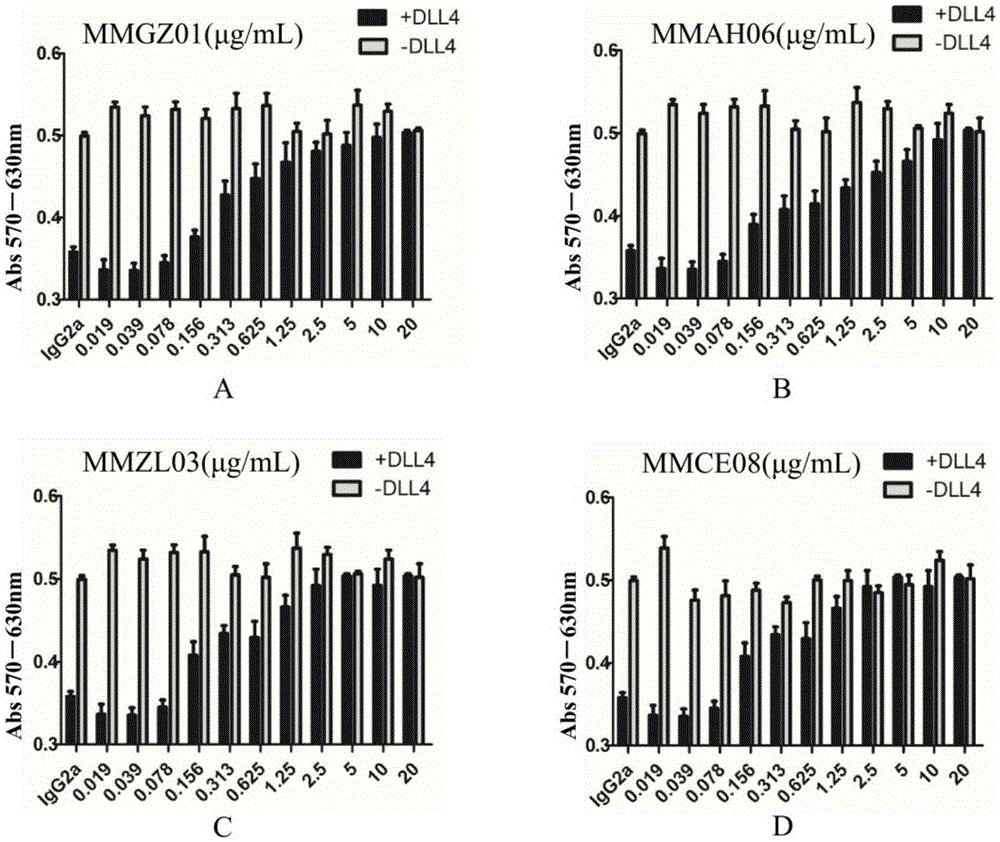
Anti-human Delta like 4 monoclonal antibody and application thereof
ActiveCN105384818AImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsBALB/cAntigen
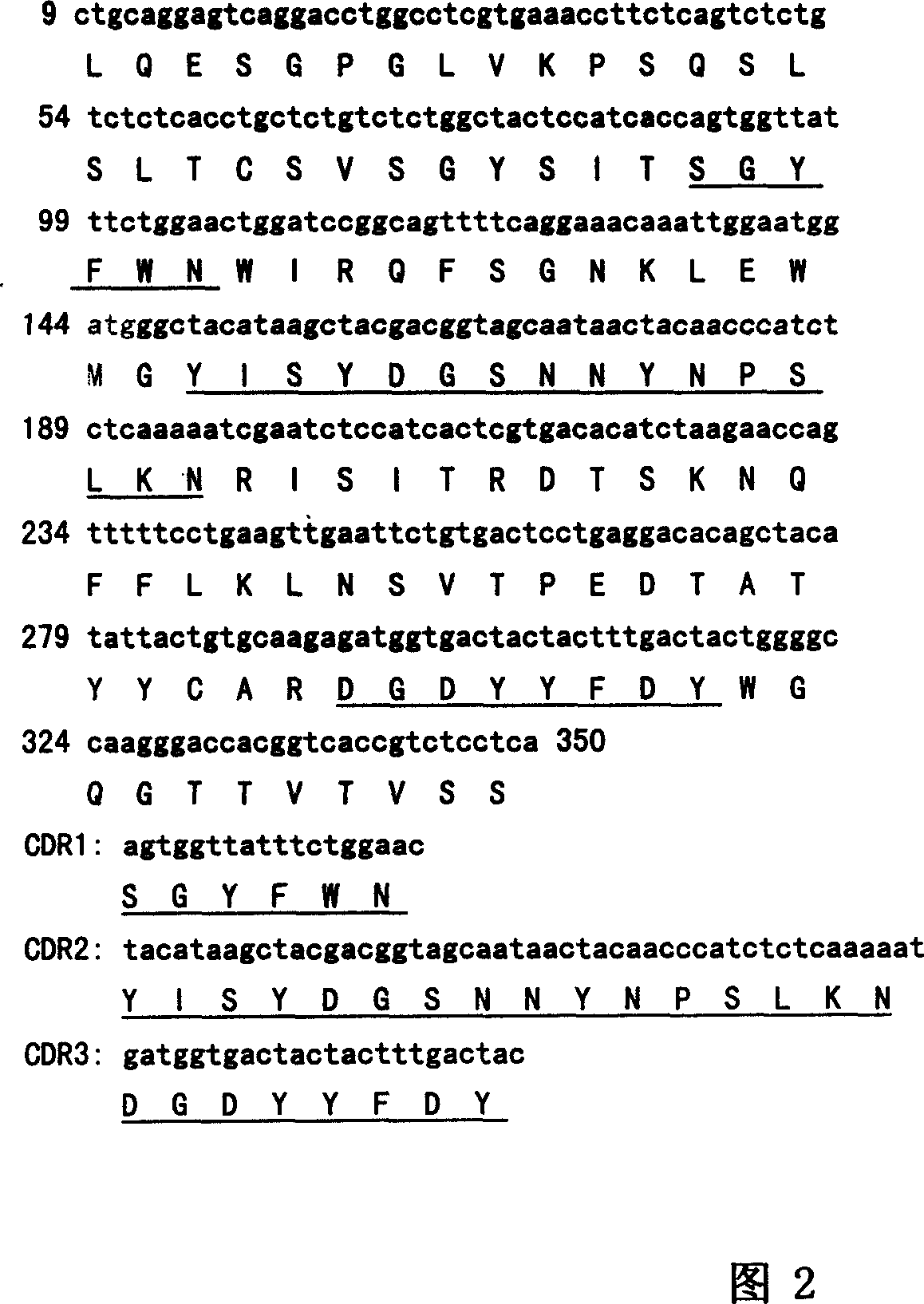
By use of hybridoma technology, recombinant human Delta like 4 (rhDll4) is used as an antigen for immunizing a BALB / c mice to obtain a high affinity and biological activity anti-human Delta like 4 monoclonal antibody. The monoclonal antibody is characterized in that: the monoclonal antibody can be combined with rhDll4 specifically, and can block human umbilical vein endothelial cells (HUVEC) proliferation suppression of the rhDll4. Specifically, screening, a preparation method, and nucleotide and amino acid sequences of the heavy chain variable region and the light chain variable region of the anti-human Delta like 4 monoclonal antibody are disclosed, and the nucleotide and amino acid sequences comprise nucleotide and amino acid sequences corresponding to complementarity determining regions CDR1, CDR2 and CDR3.
Owner:CHINA PHARM UNIV
Preparing method for paclobutrazol monoclonal antibodies
The invention discloses a preparing method for paclobutrazol monoclonal antibodies. The preparing method comprises the main steps as follows: (1) synthesizing artificial hapten paclobutrazol hemisuccinates by a microwave solvent free method; (2) performing coupling to obtain artificial antigens of the paclobutrazol by using the paclobutrazol hemisuccinates as raw materials; (3) performing the immune treatment on Balb / c mice by using the synthesized artificial antigens; (4) selecting the mice with optimal serum valence and optimal specificity, and mixing spleen cells and myeloma cells of the mice together in an external manner; (5) culturing and screening the fused cells by using the selective culture medium, and further cloning, propagating and storing by freezing; (7) injecting the expanded cell strains into the abdominal cavities of the mice to generate a lot of abdominal dropsy; (8) purifying the paclobutrazol-resistant monoclonal antibodies in the abdominal dropsy by a caprylic acid-saturated ammonium sulfate method. The prepared paclobutrazol-resistant monoclonal antibodies have high sensitivity and strong specificity, and can be further applied to the construction of technologies of immune sensors, colloidal gold immune chromatographic methods and the like.
Owner:NANCHANG UNIV
Melamine and carrier protein couplet product, preparation method and uses of melamine antibody
InactiveCN101429243AEfficient detectionEasy to operateSerum immunoglobulinsOvalbuminBALB/cCarrier protein
The invention discloses a preparation method and application of a product obtained by coupling melamine with carrier protein, as well as a melamine antibody. The product obtained by coupling the carrier protein with the melamine is used as artificial antigen and applied to an immunological method for melamine detection. The preparation method comprises the following steps of immunizing animals with the coupled product so as to prepare an antibody used for melamine detection, fusing BALB / C mouse spleen cells immunized with the coupled product and SP2 / 0 mouse myeloma cells, obtaining hybridoma capable of stably transferring culture and secreting anti-melamine specific monoclonal antibodies by screening positive hybridoma and cloning cells, and preparing an ascites monoclonal antibody. The prepared monoclonal antibody is utilized to establish a direct competitive ELISA method having high specificity, sensitivity and accuracy to the melamine, as well as an immune colloidal gold test strip. The preparation method for the product obtained by coupling melamine with carrier protein, as well as the melamine antibody provides service for the rapid detection of melamine-type residue in foods.
Owner:ZHEJIANG UNIV
Anti-porcine reproductive and respiratory syndrome virus monoclonal antibody and application
ActiveCN101979512AImmunoglobulins against virusesMicroorganism based processesBALB/cBiological property
The invention discloses an anti-porcine reproductive and respiratory syndrome virus monoclonal antibody and application. The invention provides an anti-porcine reproductive and respiratory syndrome virus monoclonal antibody hybridoma cell strain 3C3 with collection number of CGMCC No. 4109. The invention also provides the anti-porcine reproductive and respiratory syndrome virus monoclonal antibody generated by the anti-porcine reproductive and respiratory syndrome virus monoclonal antibody hybridoma cell strain 3C3 with collection number of CGMCC No. 4109. An experiment of the invention proves that: a purified PRRSV JXA1 strain is used for immunizing BALB / c mice, one hybridoma cell strain which can stably secrete PRRSV monoclonal antibodies is screened by utilizing the hybridoma technology and the biological characteristic identification is performed on the hybridoma cell strain so as to lay the foundation for further establishing a specific, sensitive and rapid PRRSV detection method.
Owner:CHINA ANIMAL DISEASE CONTROL CENT
Preparation method of heavy metal mercury monoclonal antibody
InactiveCN101139398AFast pushStrong antigenicityImmunoglobulins against animals/humansBiological testingBALB/cSpleen cell
The present invention relates to a preparation method of heavy metal mercury monoclonal antibody, which belongs to the field of biotechnology. The present invention is particularly used in the preparation of specificity-recognition heavy metal mercury monoclonal antibody and in the fast mercury high-sensitivity measurement remained in the agricultural production and the agricultural production environment. The heavy metal mercury ion and the carrier albumen are coupled into full antigens through bifunctional metal chelate 1-(4-separate-cyano phenyl)-EDTA; the full antigens are used on Balb / C mouse and the spleen cells and the Sp2 / 0 myeloma cells are used to prepare hybridoma cells through the hybridoma technology. And monoclonal antibody which can stably excrete anti-Hg-EDTA is generated. The preparation technology in the present invention is easy and practical: the whole preparation process of the antigen requires no special instrument. The present invention is suitable for factory-scale production.
Owner:NANJING AGRICULTURAL UNIVERSITY
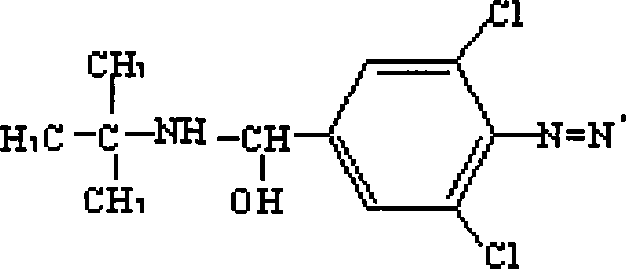
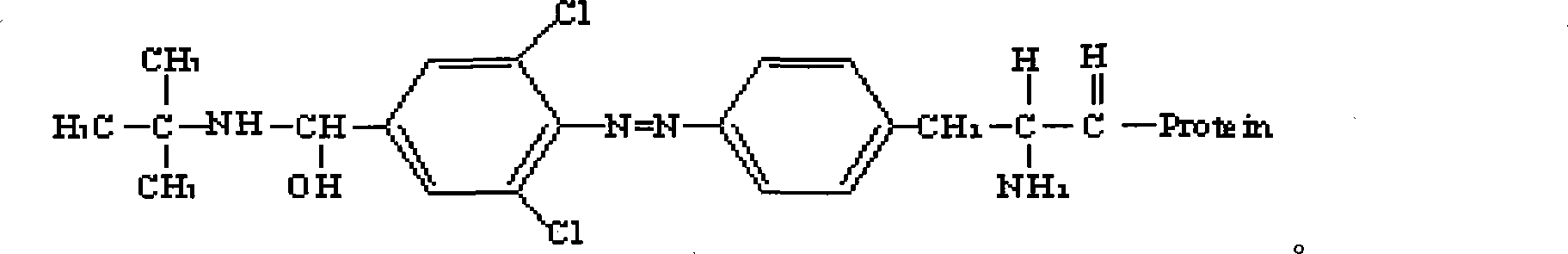
Clenbuterol complete antigen and method for preparing monoclonal antibody thereof
InactiveCN101182356AHigh sensitivityImprove featuresSerum albuminImmunoglobulins against animals/humansBALB/cAntigen
The invention discloses the preparation methods of a complete antigen of clenbuterol hydrochloride and a monoclonal antibody of the clenbuterol hydrochloride. Firstly, the clenbuterol hydrochloride reacts with sodium nitrite under the acid condition to obtain azo clenbuterol hydrochloride; then the azo clenbuterol hydrochloride coupled with bovine serum albumin under the alkaline condition to prepare for the complete antigen of the clenbuterol hydrochloride. A balb / c pure line rat is immunized, after IELISA test shows that the serum of the rat after immunity is eligible, the cell fusion is processed for preparing for the monoclonal antibody of the clenbuterol hydrochloride. The complete antigen of the clenbuterol hydrochloride prepared by the invention can be used for immunizing the animal. The prepared monoclonal antibody can be used for testing the residual quantity of the clenbuterol hydrochloride in meat, meat products, livestock feed and animal body before the animal is slaughtered. The coupling rate of the clenbuterol hydrochloride in the complete antigen of the clenbuterol hydrochloride obtained by the method of the invention with the bovine serum albumin is 17, and a molecular structure formula thereof is as above.
Owner:UNIV OF SHANGHAI FOR SCI & TECH
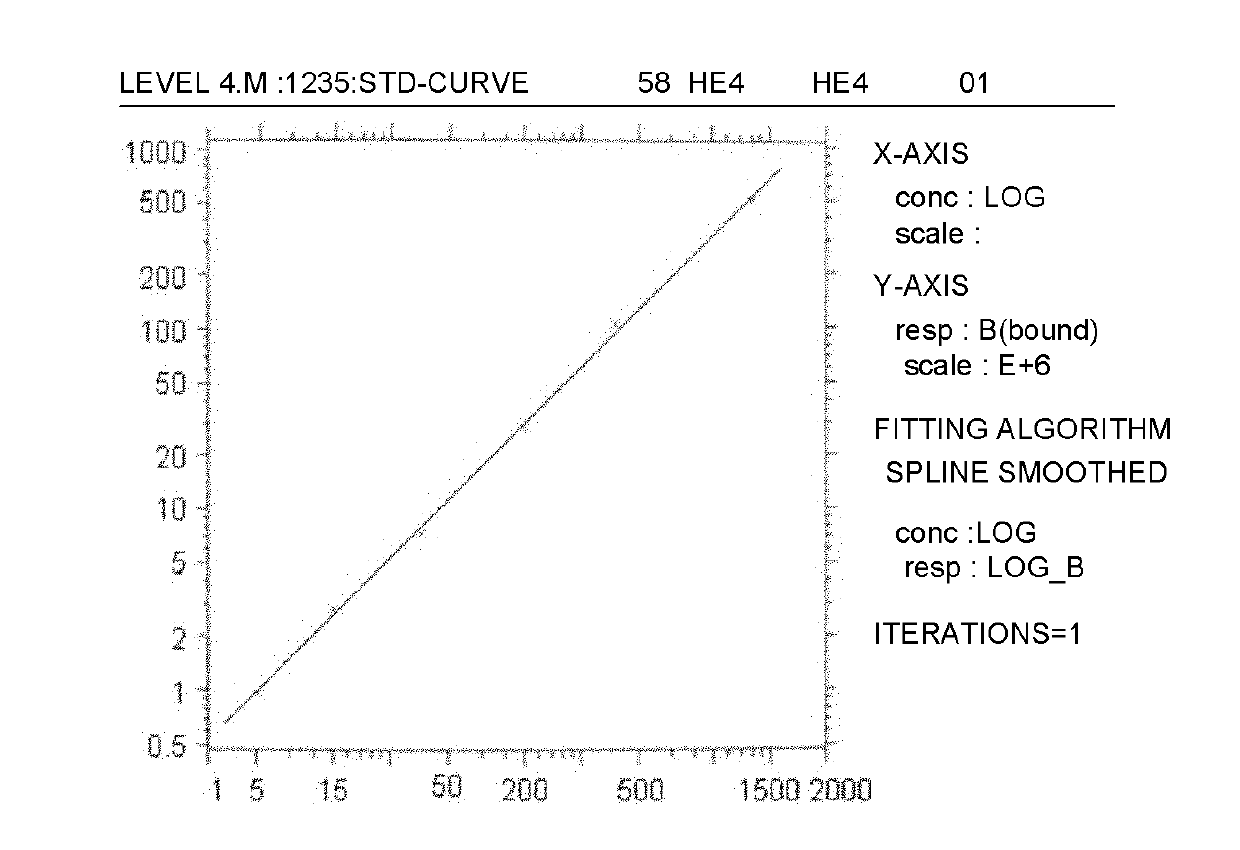
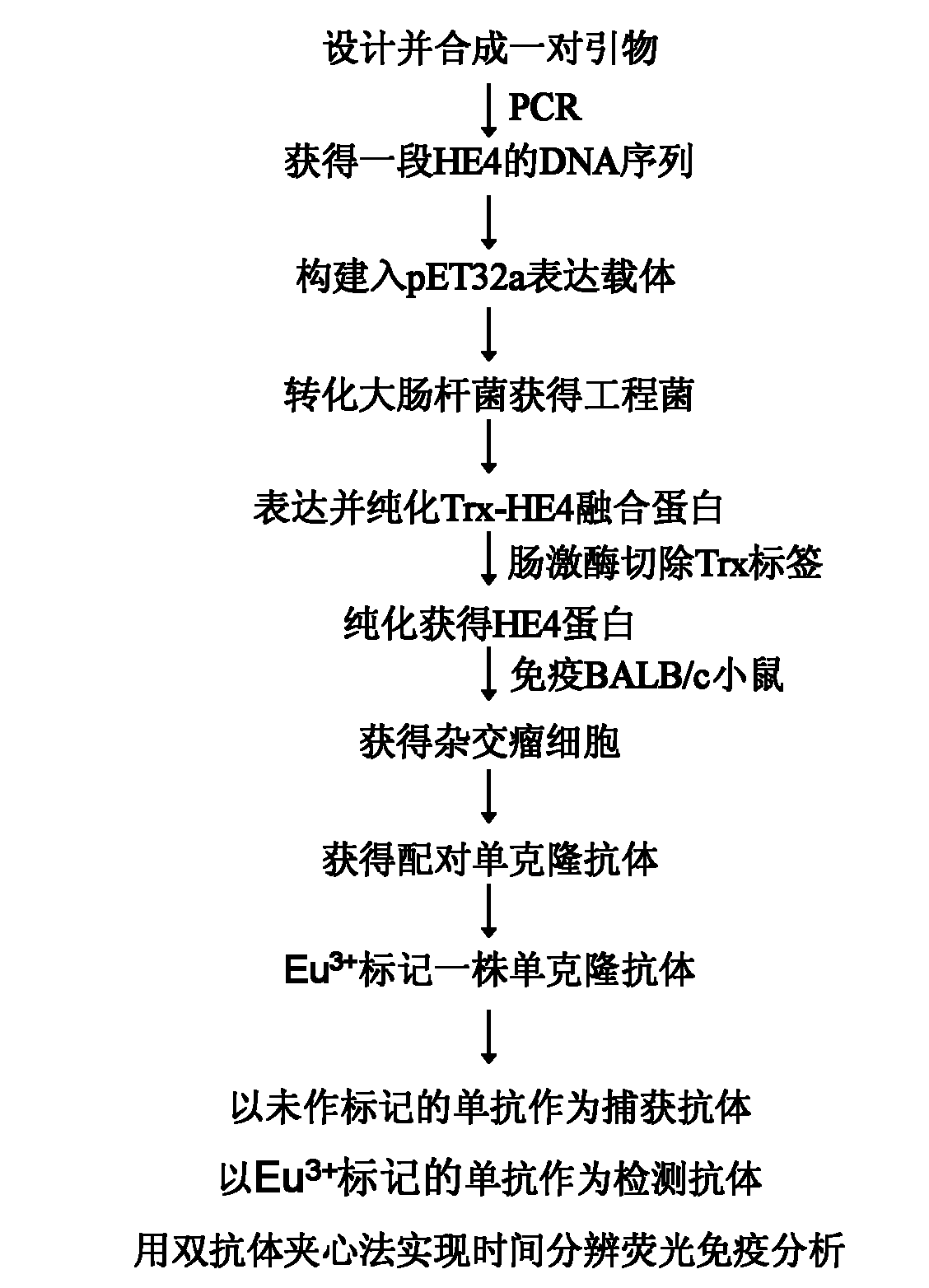
Time-resolved fluorescence (TRF) immunized detection kit of ovarian cancer tumor marker HE4
InactiveCN101949937AImmunoglobulins against animals/humansBiological testingRare-earth elementBALB/c
The invention relates to an analysis and detection technology of a human ovarian cancer tumor marker (human epididymis protein 4, HE4), in particular to a time-resolved fluorescence (TRF) immunized detection method and a kit of HE4, which is used for clinical auxiliary diagnosis, curative effect observation and prognosis judgment of the ovarian cancer. The invention comprises the following contents: constructing HE4 recombinant plasmids; expressing and purifying HE4 protein; immunizing BALB / c mice by the purified HE4 protein to prepare monoclonal antibodies; matching the obtained monoclonal antibodies to obtain two hybrid tumor cell strains (5A3 and 6C2) for secreting monoclonal antibodies of different epitopes of HE4 antigens, and marking the 5A3 monoclonal antibodies by the rare earth element Eu3+; and taking the unmarked 6C2 monoclonal antibodies as capture antibodies for coating a solid phase carrier, and taking the 5A3 monoclonal antibodies marked with the Eu3+ as detection antibodies to establish a double-antibody sandwich method for detecting HE4, thereby realizing the TRF immunized analysis of the invention.
Owner:大连美亿德生物科技有限公司
Method for quantitatively detecting allergen alpha-lactalbumin based on quantum dot fluorescence
InactiveCN102680705AIncreased sensitivitySimple and fast operationBiological testingFluorescence/phosphorescenceBALB/cFluorescence
The invention discloses a method for quantitatively detecting allergen alpha-lactalbumin based on quantum dot fluorescence and application of the allergen alpha-lactalbumin. The method comprises the following steps of: preparing a monoclonal antibody from alpha-lactalbumin as active immunization BALB / c mice; carrying out conjugation labeling on the alpha-lactalbumin monoclonal antibody by using a fluorescent quantum dot; forming an immunofluorescence complex by adopting a competitive immunosorbent assay; then detecting a fluorescent signal under a full-wavelength multifunctional ELIASA (Enzyme-Linked Immunosorbent Assay Apparatus); and quantitatively detecting the allergen alpha-lactalbumin in the food by establishing a standard curve. The method constructed by the invention can be widely applied to detection of relevant allergens in various powders and liquid milk products, has the characteristics of quickness, accuracy, high sensitivity, favorable repeatability, excellent specificity and the like, provides an effective means for high-throughput detection of relevant allergens in various foods as well as has a favorable popularization and application prospect.
Owner:NANCHANG UNIV
Penicillin and carrier protein couplet product, method for producing beta-lactam penicillin antibody, and uses thereof
The invention provides a preparation method and application of a product obtained by coupling penicillin with carrier protein, as well as a beta-lactam type penicillin antibody. Animals are immunized with penicillin artificial antigen coupled in the invention so as to prepare the antibody which can be used for detecting beta-lactam type penicillin in foods. The preparation method comprises the following steps: immune BALB / C mouse spleen cells and SP2 / 0 mouse myeloma cells are fused; beta-lactam type antibiotics coupled with the carrier protein are used as coating antigen to screen positive hybridoma; hybridoma capable of stably transferring culture and secreting anti-beta-lactam type antibiotic antibodies through cell clones is obtained; and an ascites monoclonal antibody is prepared. The prepared monoclonal antibody is utilized to establish a direct competitive ELISA method having high specificity, sensitivity and accuracy to the beta-lactam type antibiotics, as well as an immune colloidal gold test strip. The preparation method for the product obtained by coupling penicillin with carrier protein, as well as the beta-lactam type antibiotic antibodies can serve the rapid detection of beta-lactam type antibiotic residue in foods.
Owner:ZHEJIANG UNIV
HCMVPP65 antigenemia indirect immunofluorescence method detection reagent kit
InactiveCN101261272AImprove featuresIncreased sensitivityFluorescence/phosphorescenceBALB/cFluorescence
The invention discloses a HCMVPP65 antigenemia indirect immune fluorescence method detection kit, which uses cytomegalovirus-AD169 virus strain pp65 protein as the immunogen to immune a Balb / c mouse. The spleen cells of the immunized mouse and the myeloma cells of the mouse which belongs to the same type with the immune mouse are conventionally integrated, by indirect ELISA screening and finite dilution cloning, the hybridoma cell lines of the mouse cytomegalovirus pp65 protein cloning antibody are obtained, and the characteristics of the hybridoma cell lines are identified by ELISA, immune fluorescence experiment and other methods; two monoclonal antibodies that stably secrete the pp65 protein are established successfully and named respectively as 1A6 and 4A8. A monoclonal antibody which differs from the former report and aims at the pp65 protein of the cytomegalovirus (CMV) is prepared and a method used for preparing erythrocyte fast pyrolysis is established; compared with other detection kits which belongs to the same kind, the detection kit of the invention is faster, simpler and more convenient and has higher specificity and sensitivity.
Owner:天津市秀鹏生物技术开发有限公司
Chloramphenicol universal monoclonal antibody hybridoma cell strain and application thereof
ActiveCN104263701AHigh affinityHigh detection sensitivityMicroorganism based processesTissue cultureBALB/c1,3-Propanediol
Owner:JIANGNAN UNIV
Kit used for rapidly detecting Escherichia coli O157:H7 in sample, and detection method thereof
InactiveCN102435745ASuitable for mass inspectionAccurate detectionMaterial analysisBALB/cEscherichia coli
The invention relates to a kit used for rapidly detecting Escherichia coli O157:H7 in a sample, and a detection method thereof. The invention belongs to the technical field of immunological detection. According to the invention, a heated and deactivated Escherichia coli O157:H7 immunogen is used for immunizing a healthy New Zealand rabbit, such that a polyclonal antibody is obtained, and the polyclonal antibody is adopted as a coating antibody; a BALB / C mouse is immunized, and cell fusion is carried out, such that a monoclonal antibody is obtained, and the monoclonal antibody is adopted as a secondary antibody; and a double-antibody sandwich ELISA kit of Escherichia coli O157:H7 in foodstuffs (meat) is established. With the kit, a rapid and highly efficient detection means is provided for the detection of the residue of Escherichia coli O157:H7 in foodstuffs. The kit is advantaged in relatively low cost, relatively good stability, and relatively good repeatability. According to the invention, a detection limit is 105cfu / mL. The kit and the method are suitable for large-batch detections of samples.
Owner:王利兵 +2
Preparation method of HRPII protein monoclonal antibody of plasmodium falciparum
ActiveCN101659975AGood repeatabilityAchieve serial expressionMicroorganism based processesFermentationChemical synthesisEscherichia coli
The invention relates to a preparation method of HRPII protein monoclonal antibody of plasmodium falciparum. The preparation method comprises the following steps of: adopting HRPII protein of plasmodium falciparum as target antigen and respectively analyzing and selecting two dominant antigen epitopes of A and B; respectively repeating the two dominant antigen epitopes of A and B, then continuously connecting four glycine and forming recombinant protein C; adopting most securest code of escherichia coli and converting the amino acid sequence of the recombinant protein C into corresponding nucleotide sequence; carrying out chemical synthesis to the former step to obtain the nucleotide sequence, and respectively adding enzyme cutting sites BamHI and EcoRI at the upstream and downstream thereof; inserting nucleotide fragment obtained by the former step into expression carrier PET-28a(+), constructing recombinant protein C expression carrier and inducing to express the recombinant proteinC in the escherichia coli BL21 (DE3); carrying out ultrasonic bacteria breaking and low-temperature centrifugation, then taking supernatant of the solution, affining a chromatographic column by nickel-agarose, eluting and obtaining purified recombinant protein C; after immunizing Balb / c mouse with the recombinant protein C for a plurality of times, taking and fusing spleen cells with sp2 / 0 myelomacells, and obtaining six hybridoma cell lines by multiple rounds of screening; and purifying monoclonal antibody, respectively marking horse radish peroxidase and prorating matching and combination of optimum monoclonal antibody by ELISA orthogonal experiment.
Owner:杭州新脉生物科技有限公司
Anti-rabbit hemorrhagic disease virus VP60 albumen monoclonal antibody
InactiveCN101519447AThe preparation method is simple and feasibleStrong specificityImmunoglobulins against virusesTissue cultureBALB/cCell culture supernatant
The invention relates to an anti-rabbit hemorrhagic disease virus (RHDV)VP60 albumen monoclonal antibody, and belongs to the technical field of biology. An SP2 / 0 myeloma cell and a BALB / c mouse splenic cell immunized by utilizing RHDV to recombine VP60 albumen undergo cell fusion, are selectively cultured by an HAT culture medium and undergo double ELISA screening by utilizing the recombined VP60 albumen and RHDV; the obtained cell culture supernatant is checked up and screened respectively to obtain a hybrid tumor cell strain A3C which can stably excrete the anti-RHDV VP60 albumen monoclonal antibody; the ascitic fluid ELISA titer of the A3C is detected to be 1:327,600; and according to identification, the monoclonal antibody can specifically combine the expressed recombined VP60 albumen as well as the RHDV, and one single reaction strip appears in both specific combinations, thereby proving that the anti-RHDV VP60 albumen monoclonal antibody is a VP60 specific antibody of the RHDV capsid albumen.
Owner:JIANGSU ACADEMY OF AGRICULTURAL SCIENCES
Hybridoma cell strain secreting thiamethoxam monoclonal antibody and application thereof
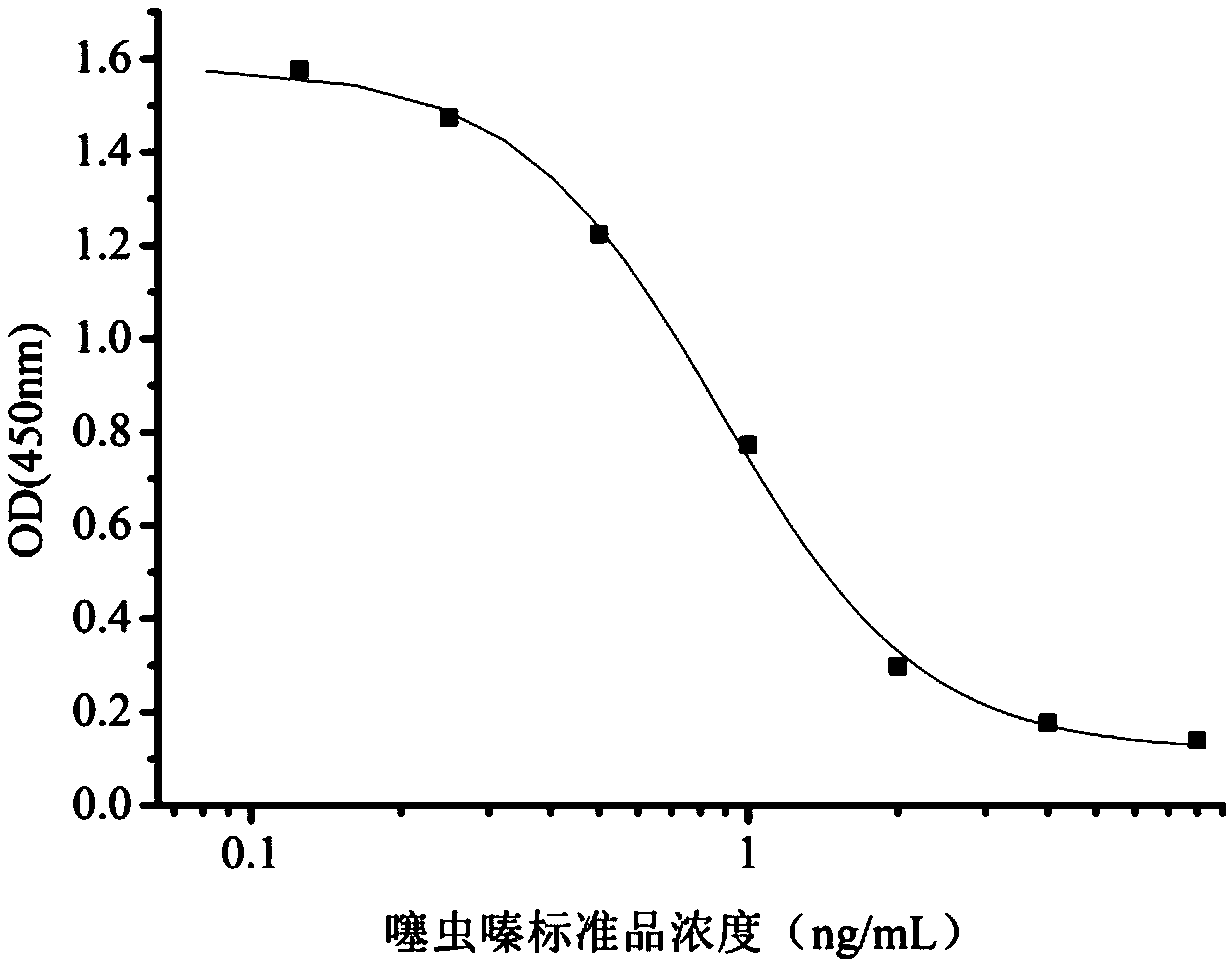
InactiveCN108998422AHigh detection sensitivityImprove featuresMicroorganism based processesDepsipeptidesBALB/cIc50 values
The invention relates to a hybridoma cell strain secreting thiamethoxacin monoclonal antibody and application thereof, belonging to the field of food safety immunodetection. The accession number of the hybridoma cell strain is CGMCC No. 14699. According to the invention, a complete Freund's adjuvant is used for primary immunization of a BALB / c mouse, then an incomplete Freund's adjuvant is used for booster immunization three times, and a thiamethoxam complete antigen containing no adjuvant is used for impact immunization once, so the BALB / c mouse is immunized; and then the high-titer low-IC50spleen cells of the immunized mouse are fused with mouse myeloma cells by using a PEG method, and then the cell strain is obtained through indirect competitive ELISA screening and subcloning three times. The monoclonal antibody secreted by the cell strain has good specificity and detection sensitivity (with an IC50 value of 0.81 ng / mL) to thiamethoxam and can be used for detection of thiamethoxamresidues in food.
Owner:JIANGNAN UNIV +1
Enrofloxacin monoclonal antibody and application
InactiveCN101565690AStrong specificityRapid and Sensitive DetectionTissue cultureImmunoglobulinsBALB/cIon exchange
The invention relates to an enrofloxacin monoclonal antibody and application, relates to hybridoma strains thereof, and belongs to the technical field of immunochemistry. The enrofloxacin monoclonal antibody is generated by mouse hybridoma strains 6A4 and 8E6. The preparation method comprises the following steps that: enrofloxacin and carrier proteins BSA, HAS and OVA are coupled by a carbodiimide method to synthesize artificial immunogens EnR-BSA, EnR-HSA and coatingen EnR-OVA; a Balb / c mouse is immunized by the synthesized artificial immunogens EnR-BSA and EnR-HSA; a spleen cell of the immunized mouse is extracted to be fused with a SP2 / O myeloma cell and coated by the coatingen EnR-OVA; indirect ELISA method and indirect competition ELISA method are established to screen the hybridoma strains which can stably secrete specific antibody; the obtained cell strain immunized Balb / c mouse is used to prepare ascites; the ascites is purified by a caprylic acid-ammonium method and an ion exchange method; and valences of antibodies of two purified cell strains reach over 1.024*10 and 1.28*10. The monoclonal antibody has strong specificity, can be applied to preparation of enrofloxacin residue inspection kit and aerosol test strip, and can sensibly and quickly inspect the enrofloxacin residue.
Owner:泰州市蛋白质工程研究院
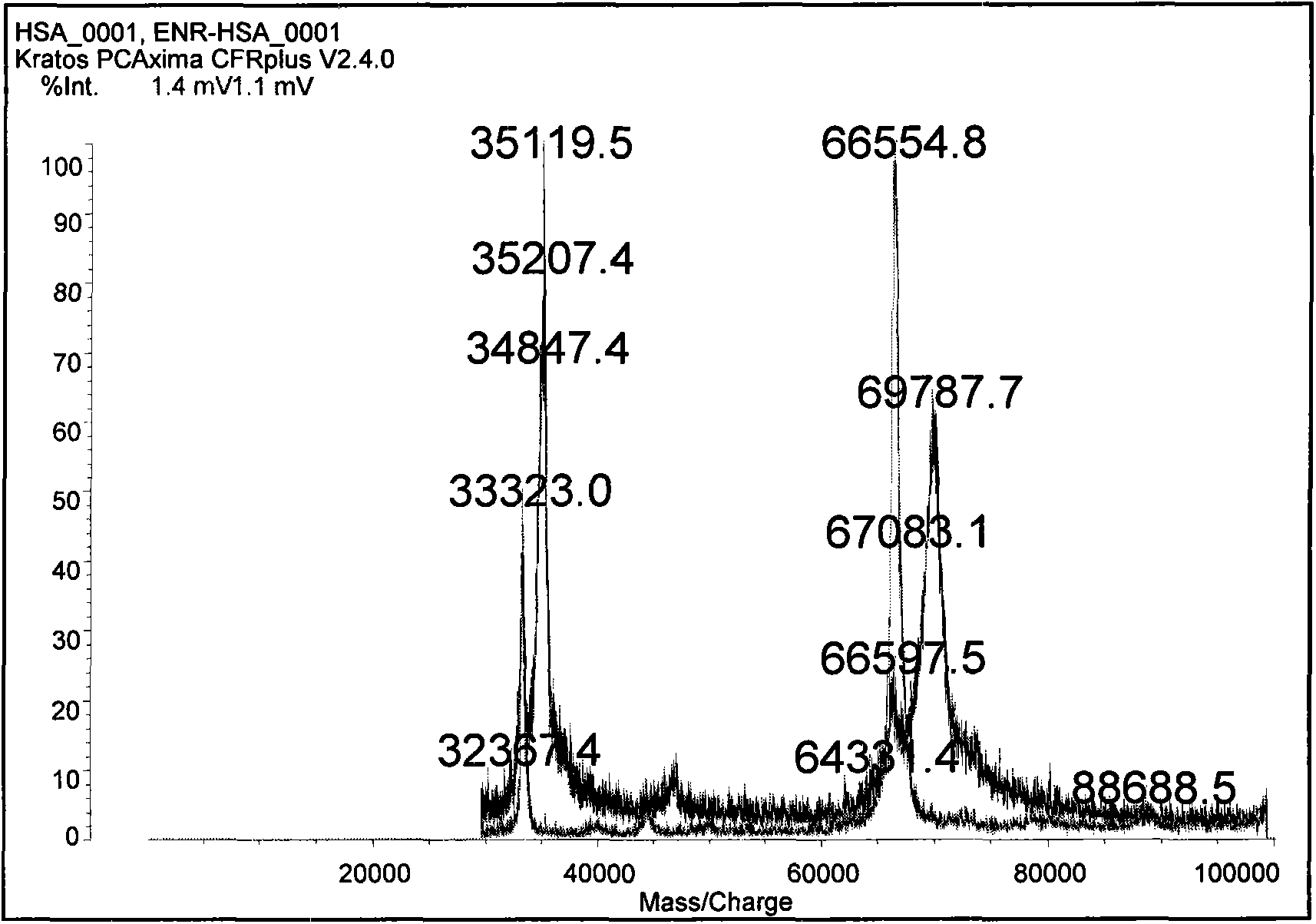
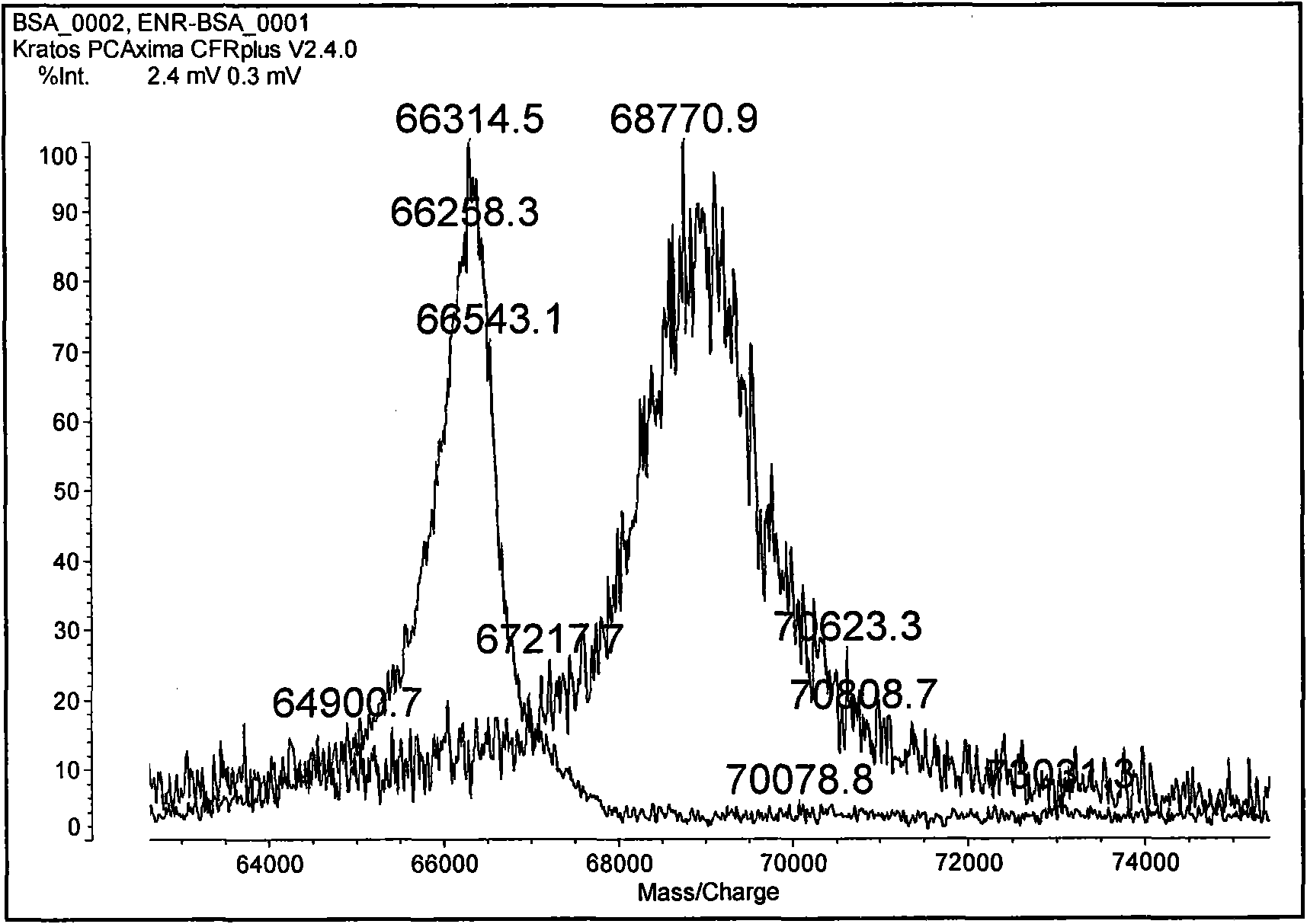
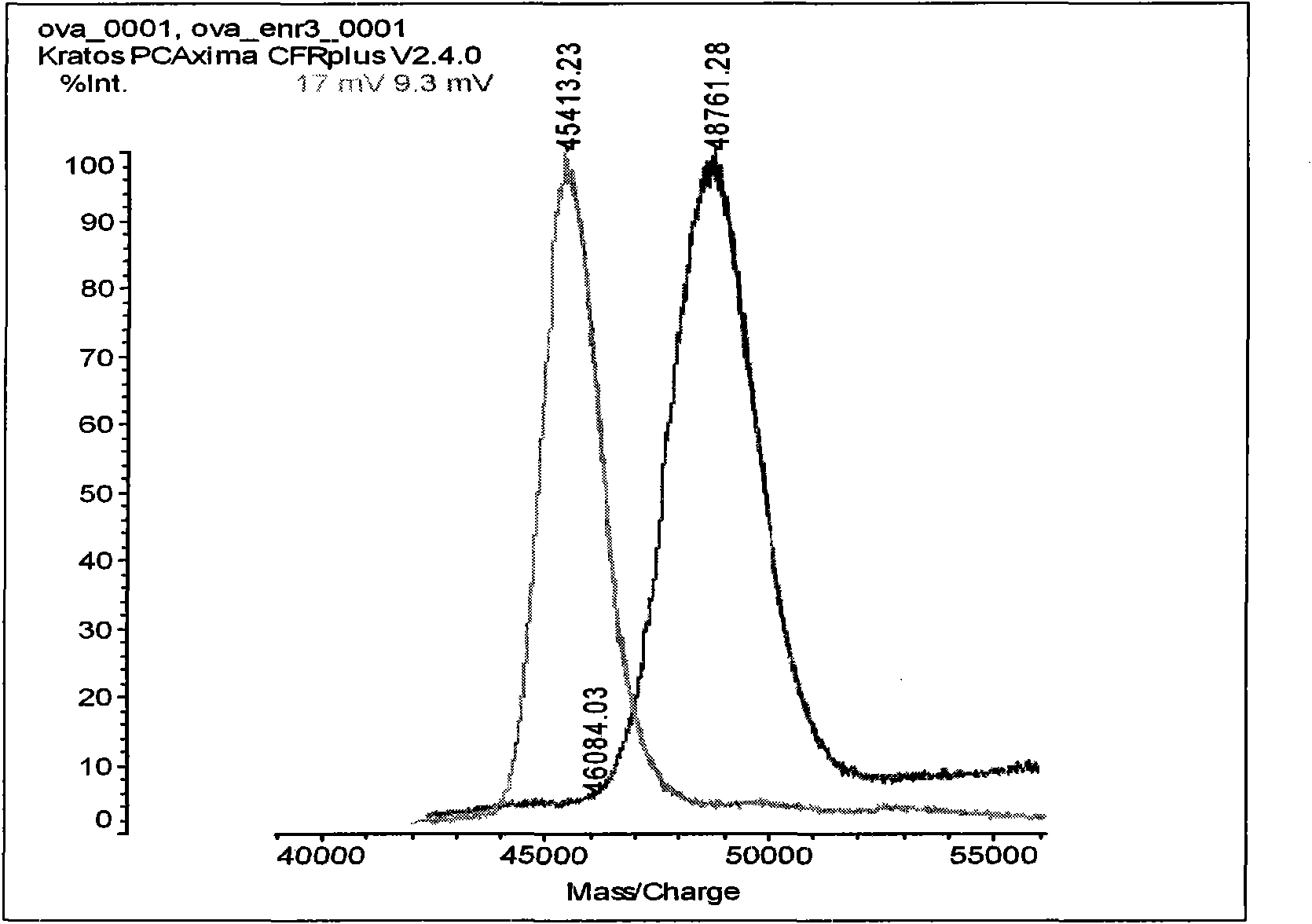
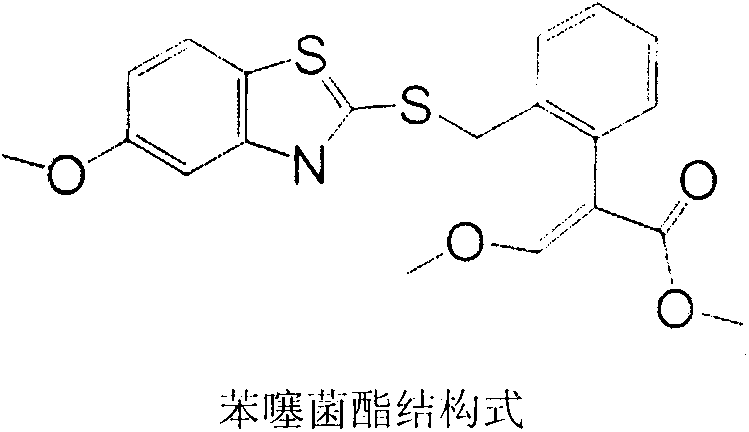
Preparation method of benzothiostrobin hapten, artificial antigen and specific antibody and application thereof
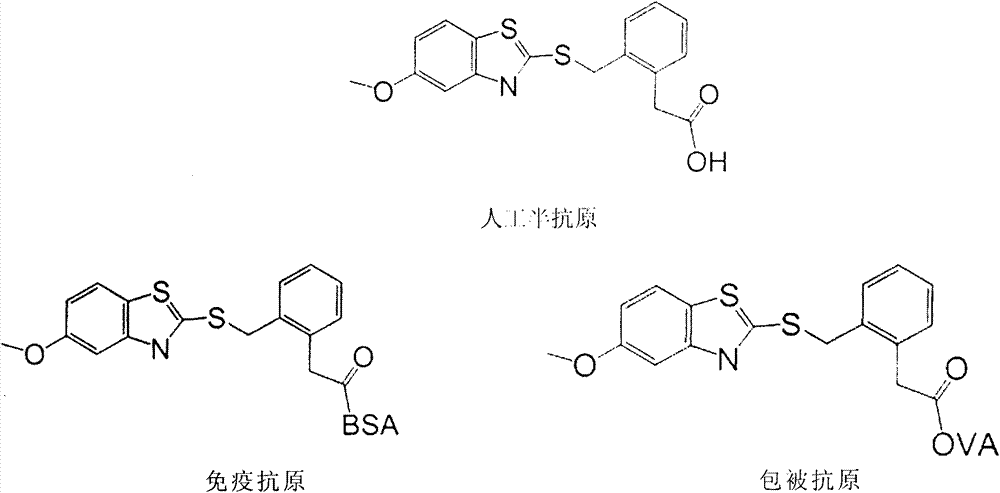
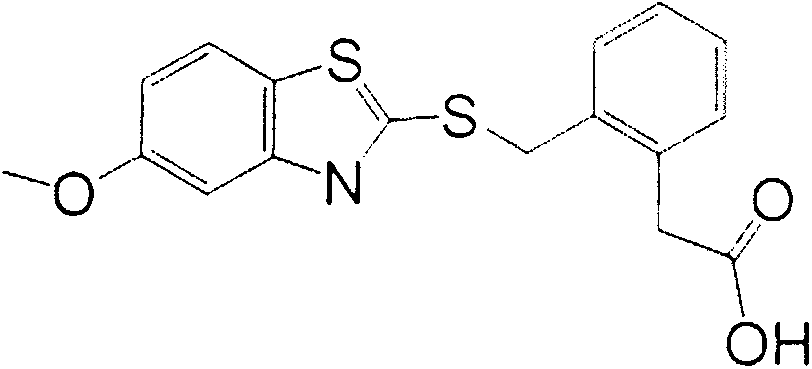
The invention relates to a preparation method of a benzothiostrobin antigen and antibody and an application thereof, belonging to the technical field of immunochemical analysis. The chemical name of benzothiostrobin is 2-[[(5-methoxy-2-benzothiazole)-thiomethyl]-alpha-(E)-methoxymethylene]methyl phenylacetate. Under an alkaline condition, carboxylic ester in the benzothiostrobin structure is hydrolyzed to synthesize an artificial hapten of which the chemical name is 2-[[(5-methoxy-2-benzothiazole)-thiomethyl]-alpha-(E)-methoxymethylene]phenylacetic acid; the artificial hapten is coupled with bovine serum albumin and ovalbumin respectively to prepared an artificial antigen. The BALB / c female mouse is immunized by the artificial antigen to obtain a specific monoclonal antibody of benzothiostrobin. The antibody does not experience a cross reaction with other compounds. An enzyme linked immunosorbent assay method established by use of the antibody can be applied to quick, sensitive, convenient and cheap detection of benzothiostrobin residues in environment and agricultural products.
Owner:NANJING AGRICULTURAL UNIVERSITY
Specific double antibody sandwich method for detecting salmonella in food based on monoclonal antibody
ActiveCN104792991ASuitable for testing requirementsImprove throughputBiological material analysisBALB/cImmune profiling
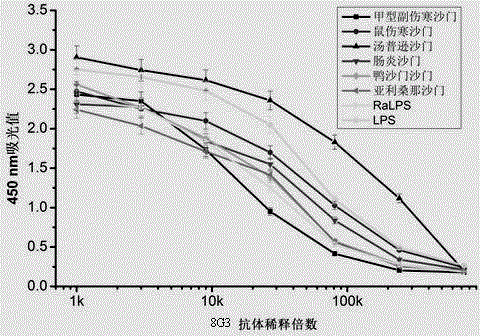
The invention discloses a specific double antibody sandwich method for detecting salmonella in food based on a monoclonal antibody, and belongs to the field of immunoassay. The specific double antibody sandwich method comprises the following steps: immunizing a 7-week-old BALB / c mouse by using smooth salmonella typhimurium LPS-coupled bovine serum albumin according to a sodium periodate method, and carrying out immunization, fusion and screening to obtain 12 monoclonal antibodies for specifically identifying salmonella LPS; marking the 12 monoclonal antibodies with horseradish peroxidase HRP respectively, and pairing the 12 monoclonal antibodies with salmonella paratyphi bacteria; using 8G3 as a coated antibody and 8G3-HRP as a detection antibody to establish the salmonella detecting specific double antibody sandwich method, so that salmonella paratyphi with the LOD of 1000000 CFU / mL can be detected. The specific double antibody sandwich method has cross reactions with all salmonella bacteria, but does not have cross sections with E.coli, E.coli O157:H7, enterobacter sakazakii, staphylococcus aureus and listeria monocytogenes, so that an overall, reliable, fast and efficient analysis method is provided for detection of the salmonella in the food.
Owner:JIANGNAN UNIV