Patents
Literature
83931results about How to "High purity" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
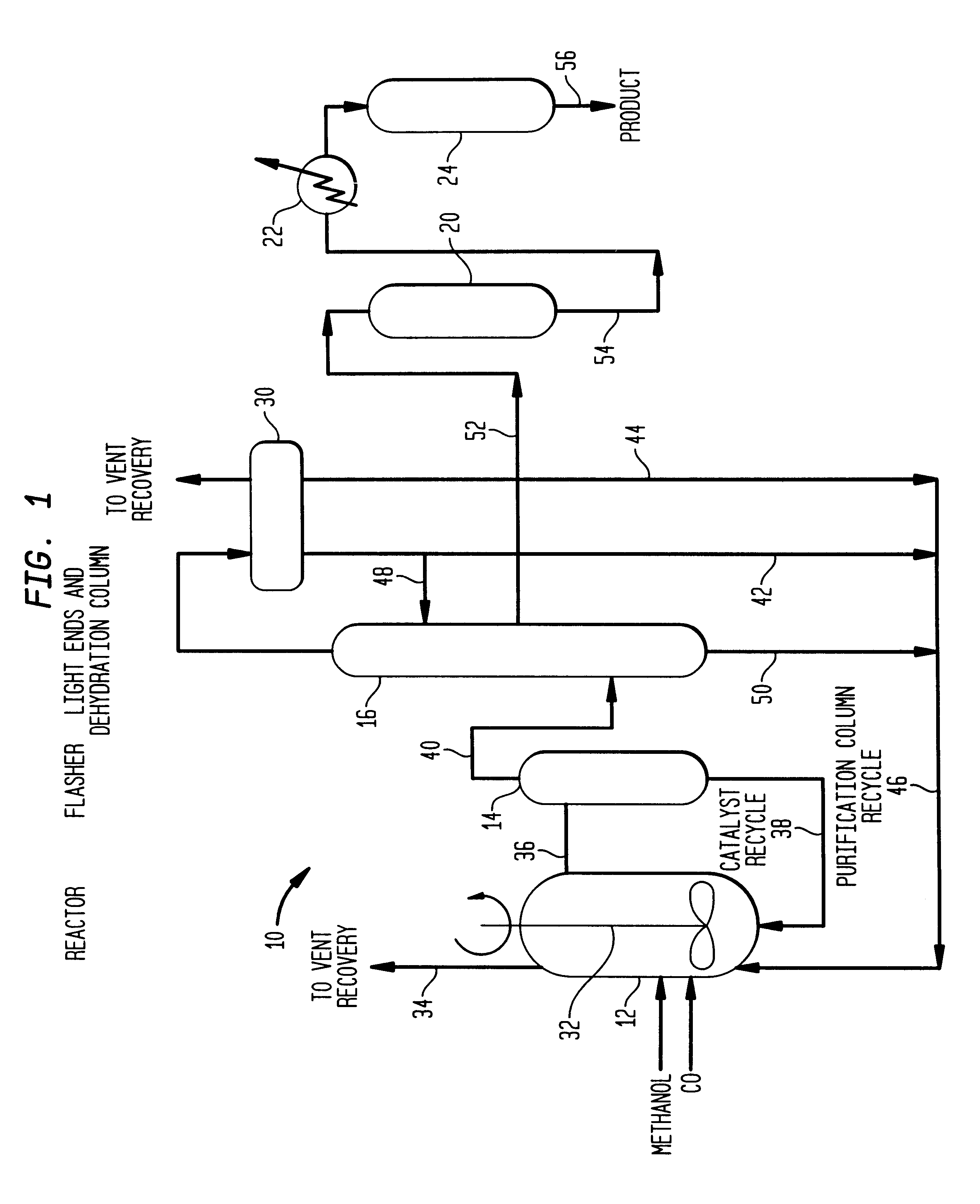
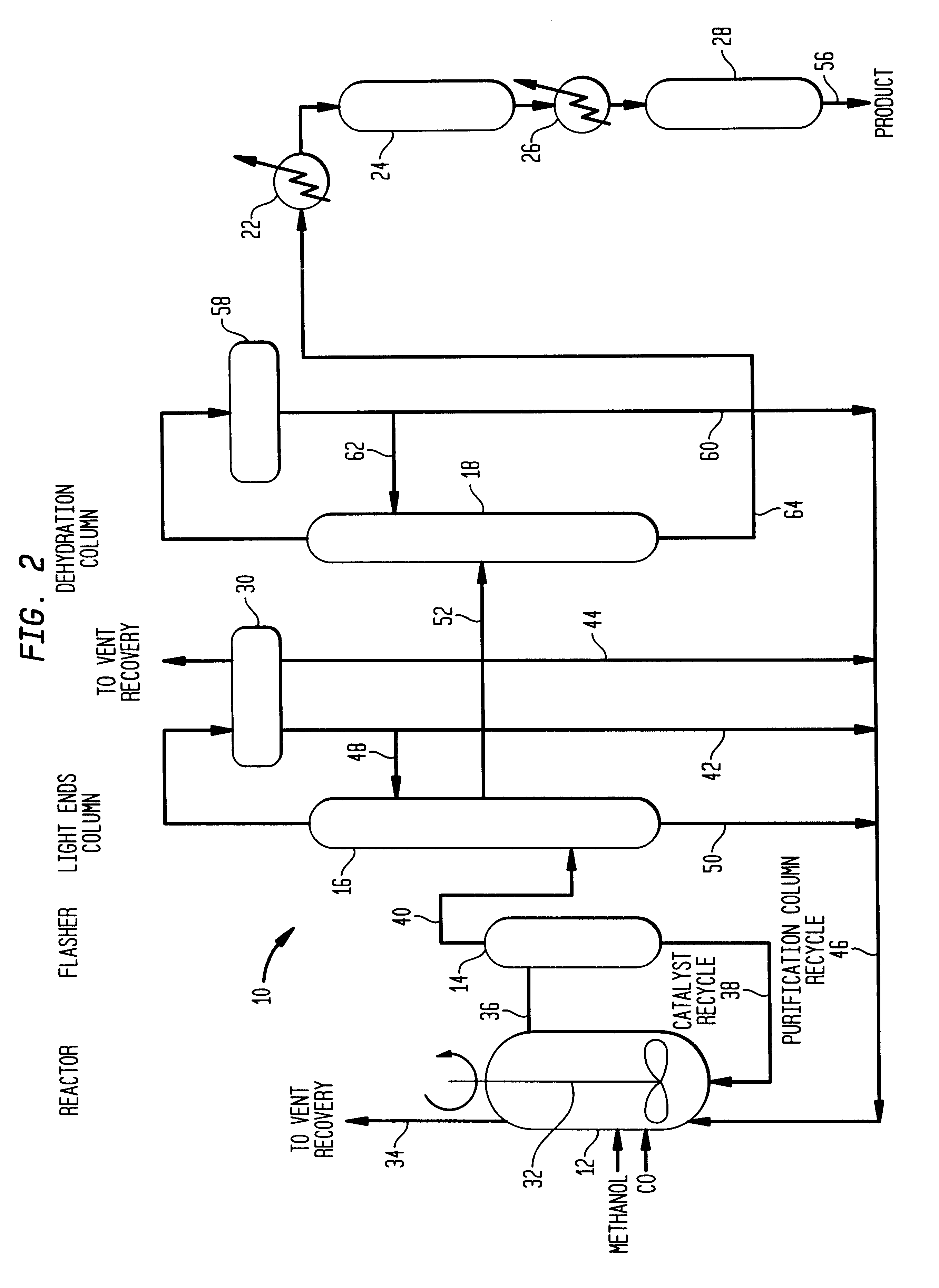
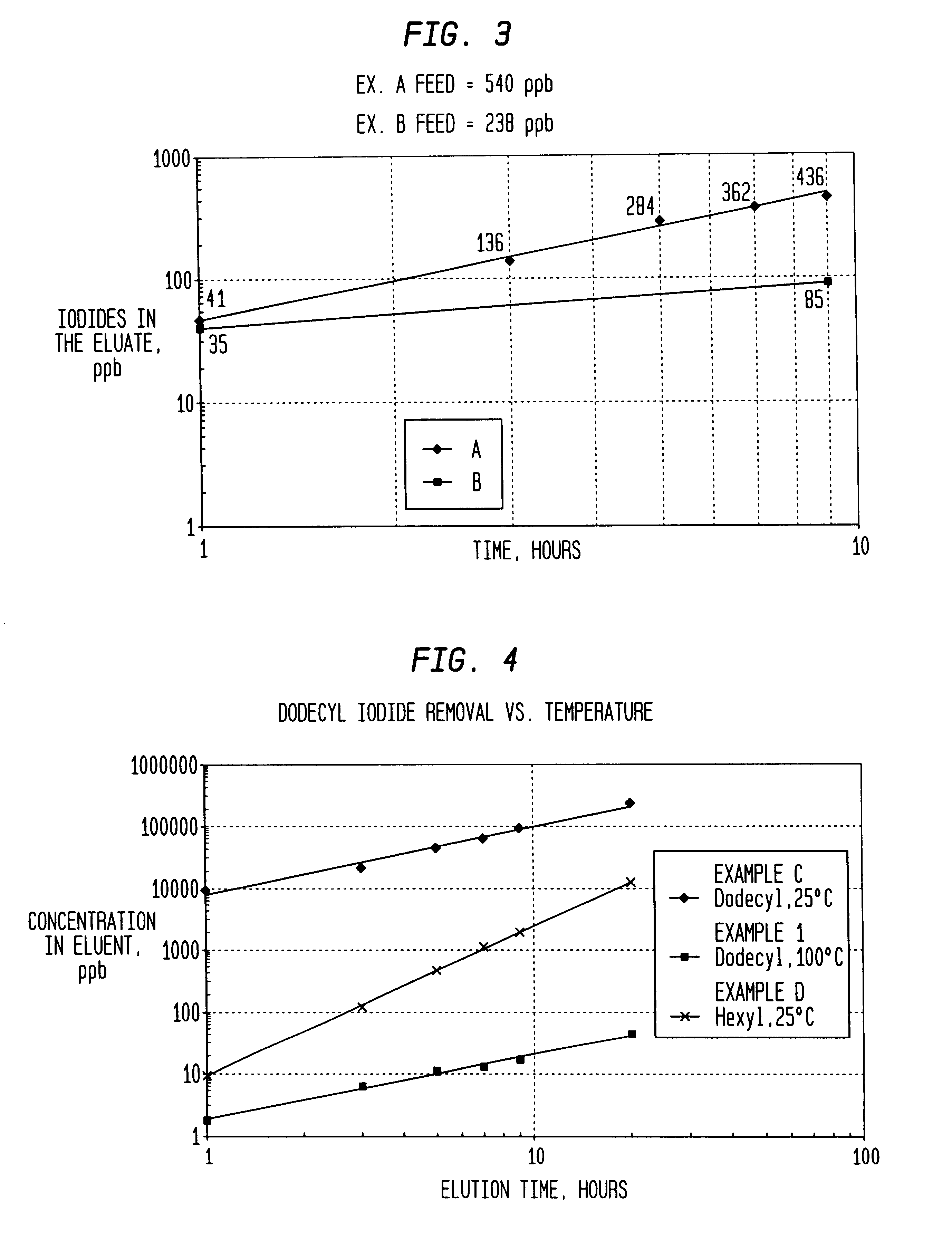
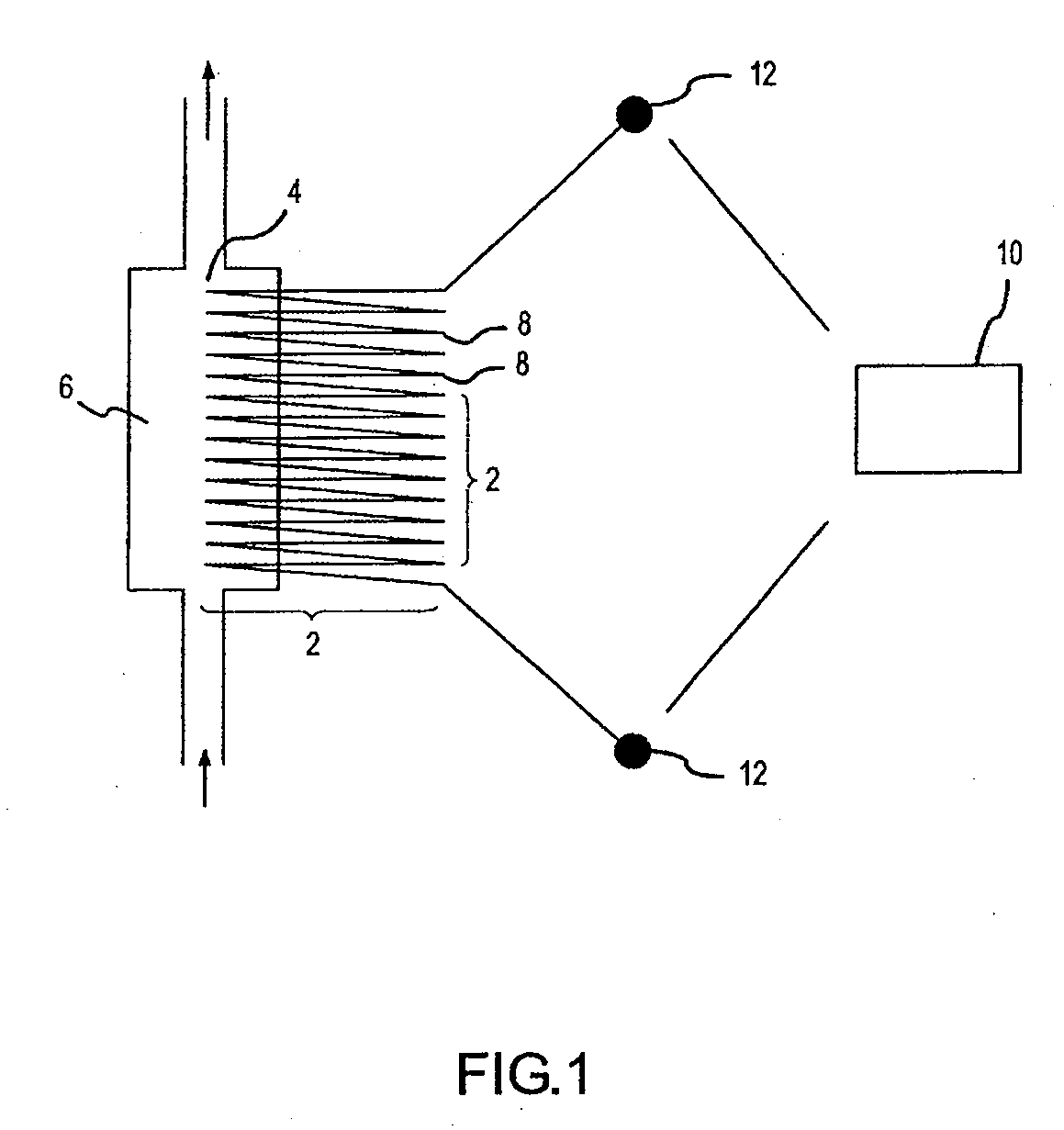
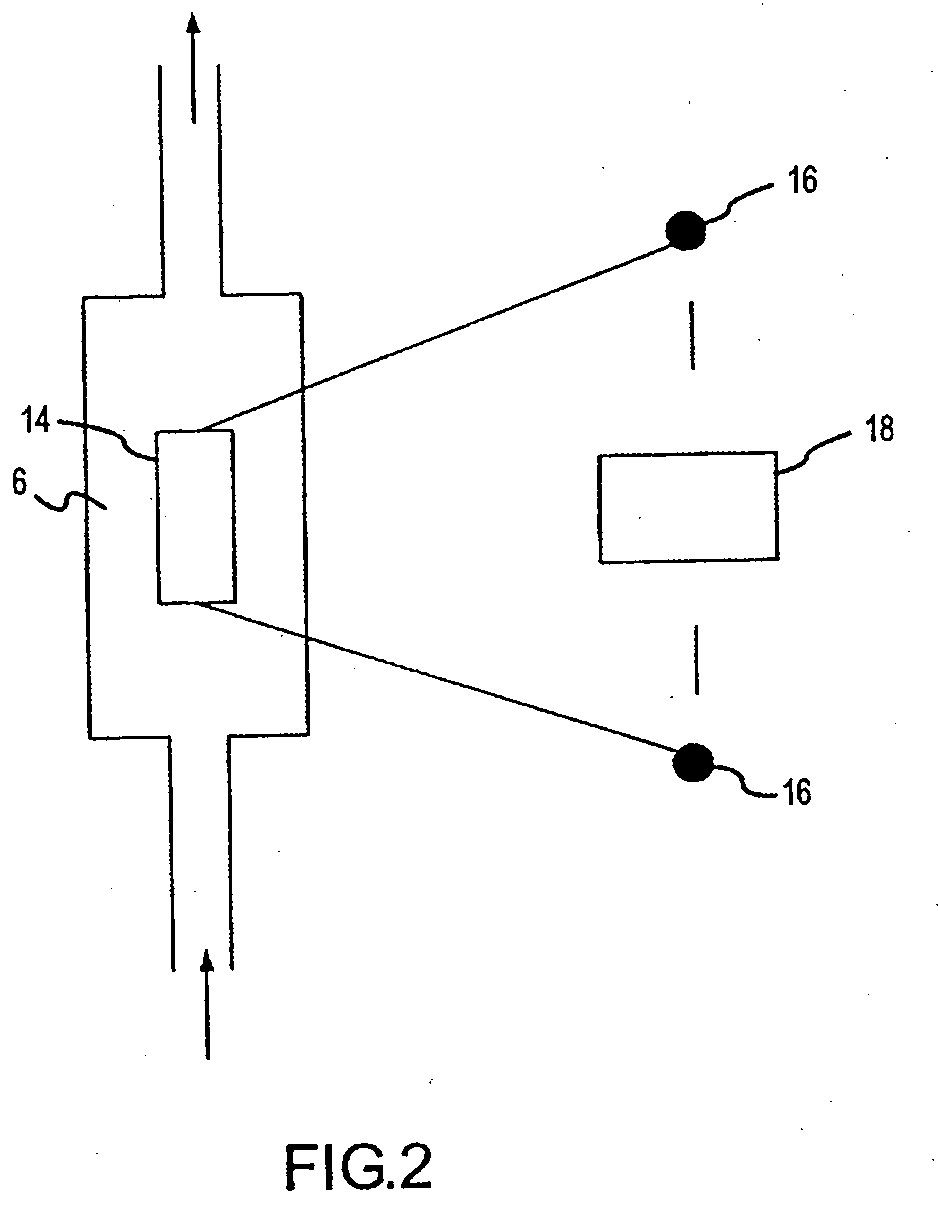
Low energy carbonylation process
InactiveUS6657078B2Weaken energyHigh purityOrganic compound preparationOrganic chemistry methodsPropanoic acidIodide
A low energy process for producing acetic acid by the carbonylation of methanol is disclosed. The process involves a rhodium-catalyzed system operated at less than about 14% water utilizing up to 2 distillation columns. The process is preferably controlled such that the product stream has a low level of propionic acid impurity and the level of aldehyde impurities is minimized by way of aldehyde removal or minimizing aldehyde generation. The level of iodides is controlled by contacting the product, at elevated temperatures, with ion exchange resins. In preferred embodiments, at least one silver or mercury exchanged macroreticular strong acid ion exchange resin is used to purify the product. The high temperature treatment provides the added benefit of controlling the Color Value (Pt-Co units) of the product stream.
Owner:CELANESE INT CORP
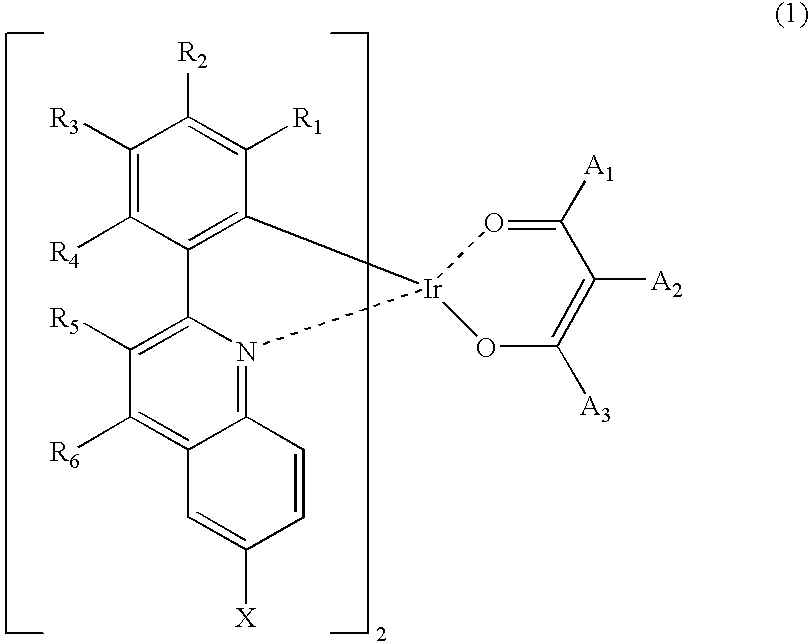
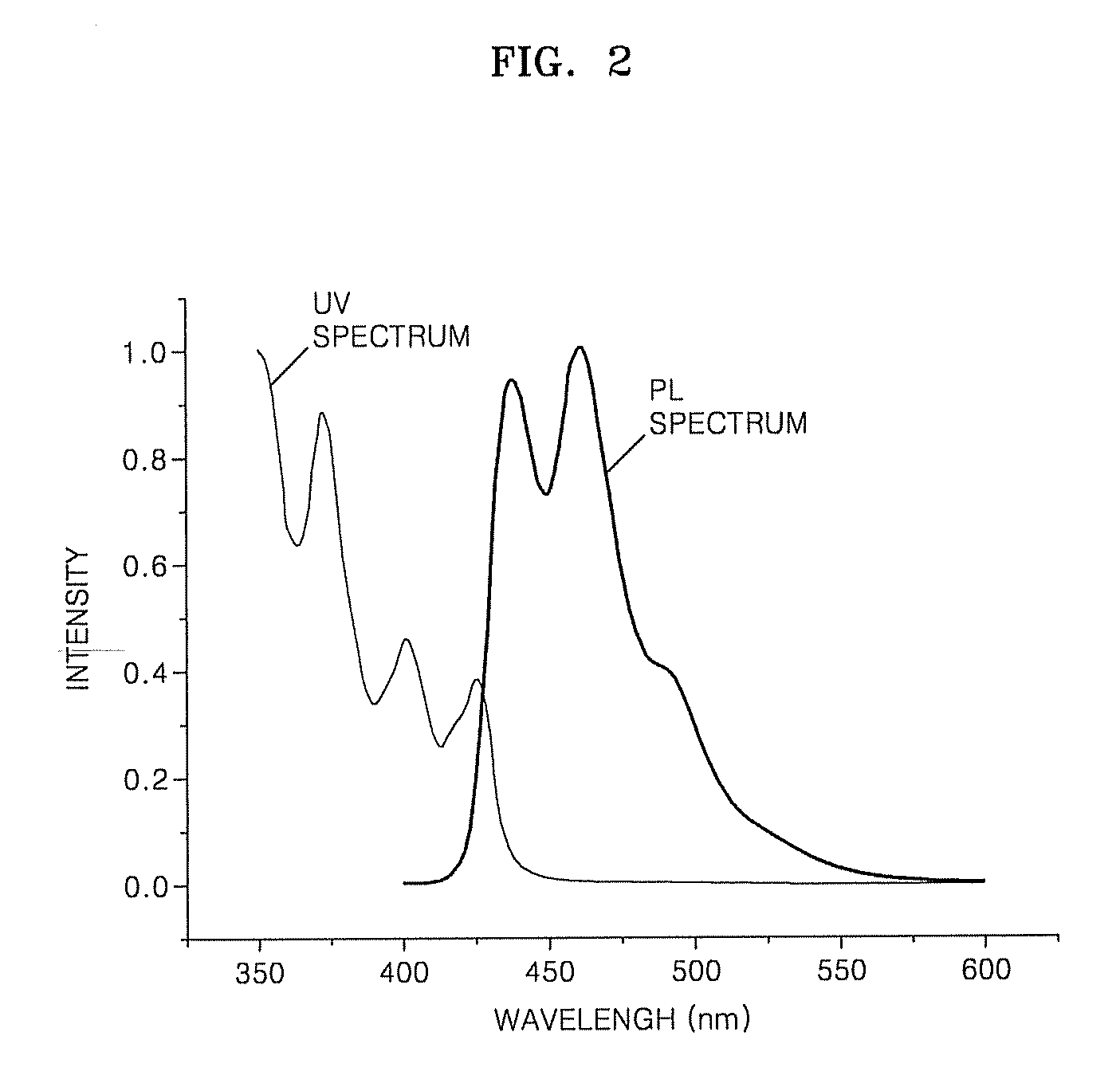
Red phosphorescent compounds and organic electroluminescent devices using the same
ActiveUS20090104472A1Good red color puritySolve low luminous efficiencyIndium organic compoundsElectroluminescent light sourcesQuantum efficiencyOrganic electroluminescence
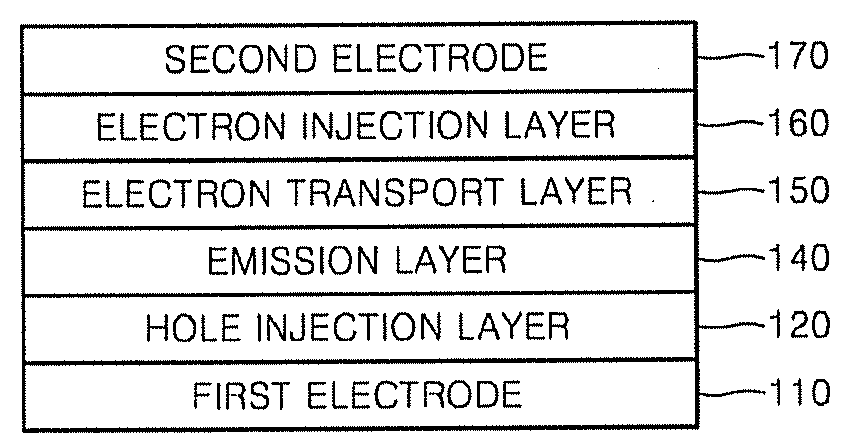
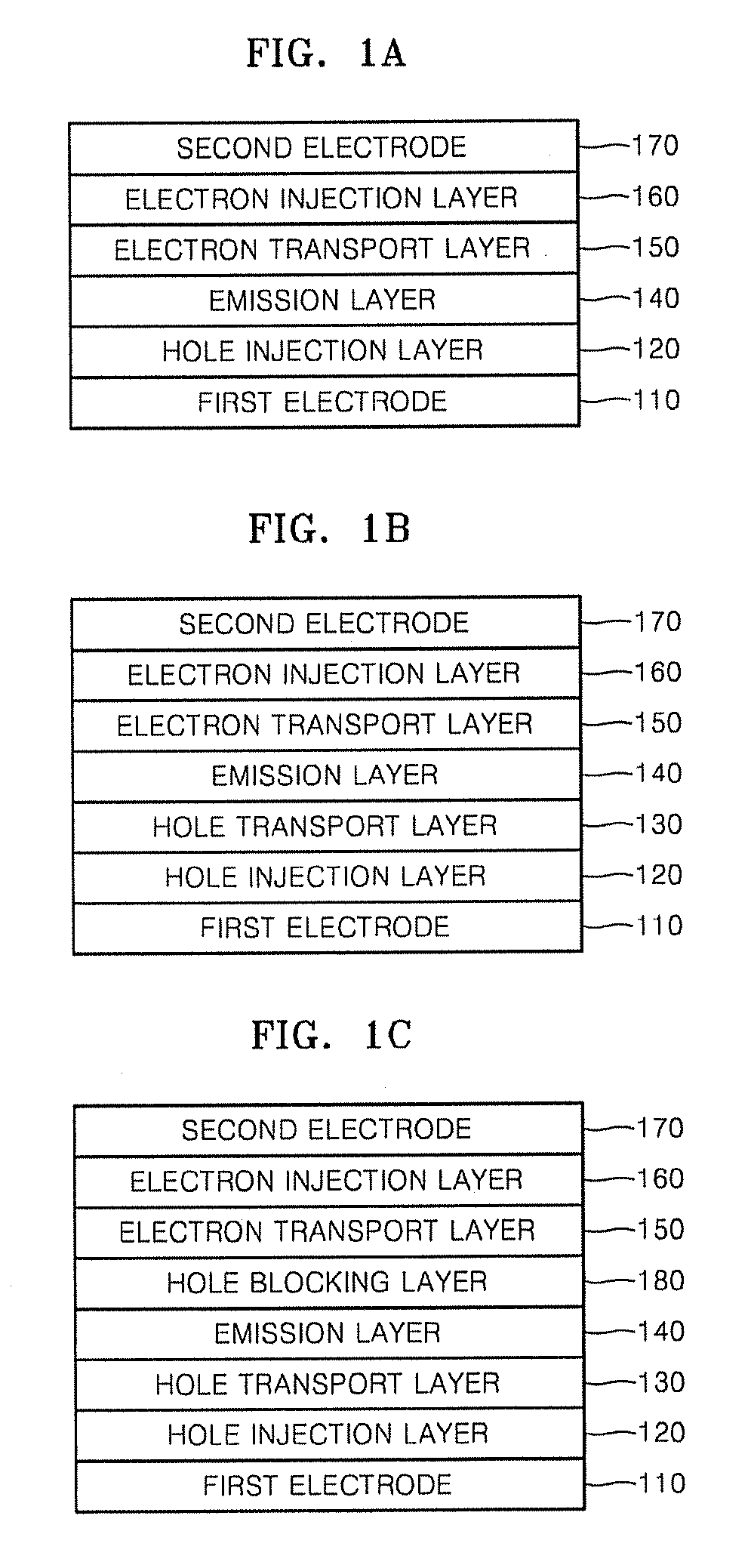
Provided are red phosphorescent compounds represented by Formula 1:Organic electroluminescent devices using the red phosphorescent compounds are further provided. The organic electroluminescent devices exhibit good red color purity, high quantum efficiency, high power efficiency, high luminance, and long lifetime.
Owner:SAMSUNG ELECTRONICS CO LTD
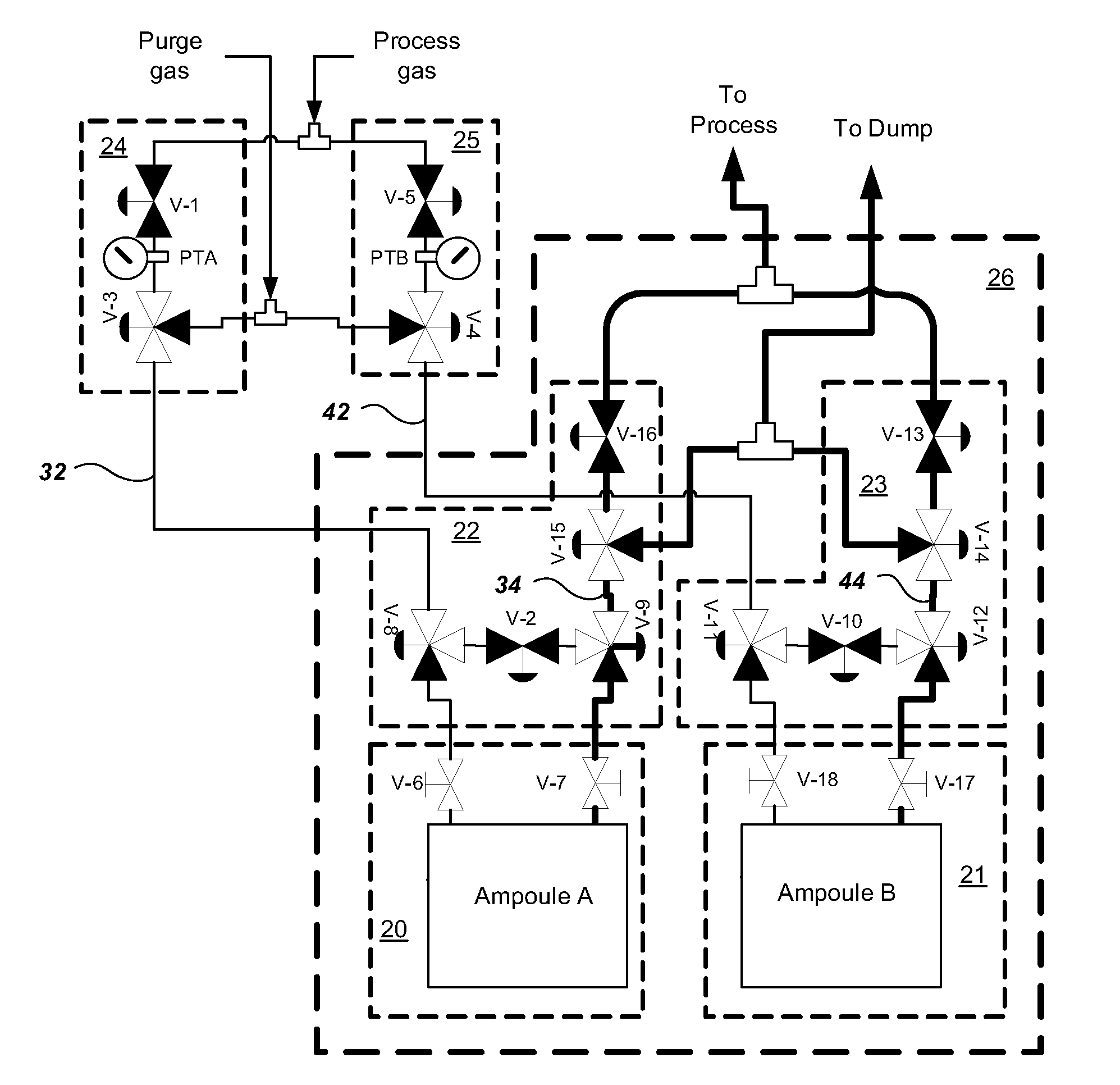
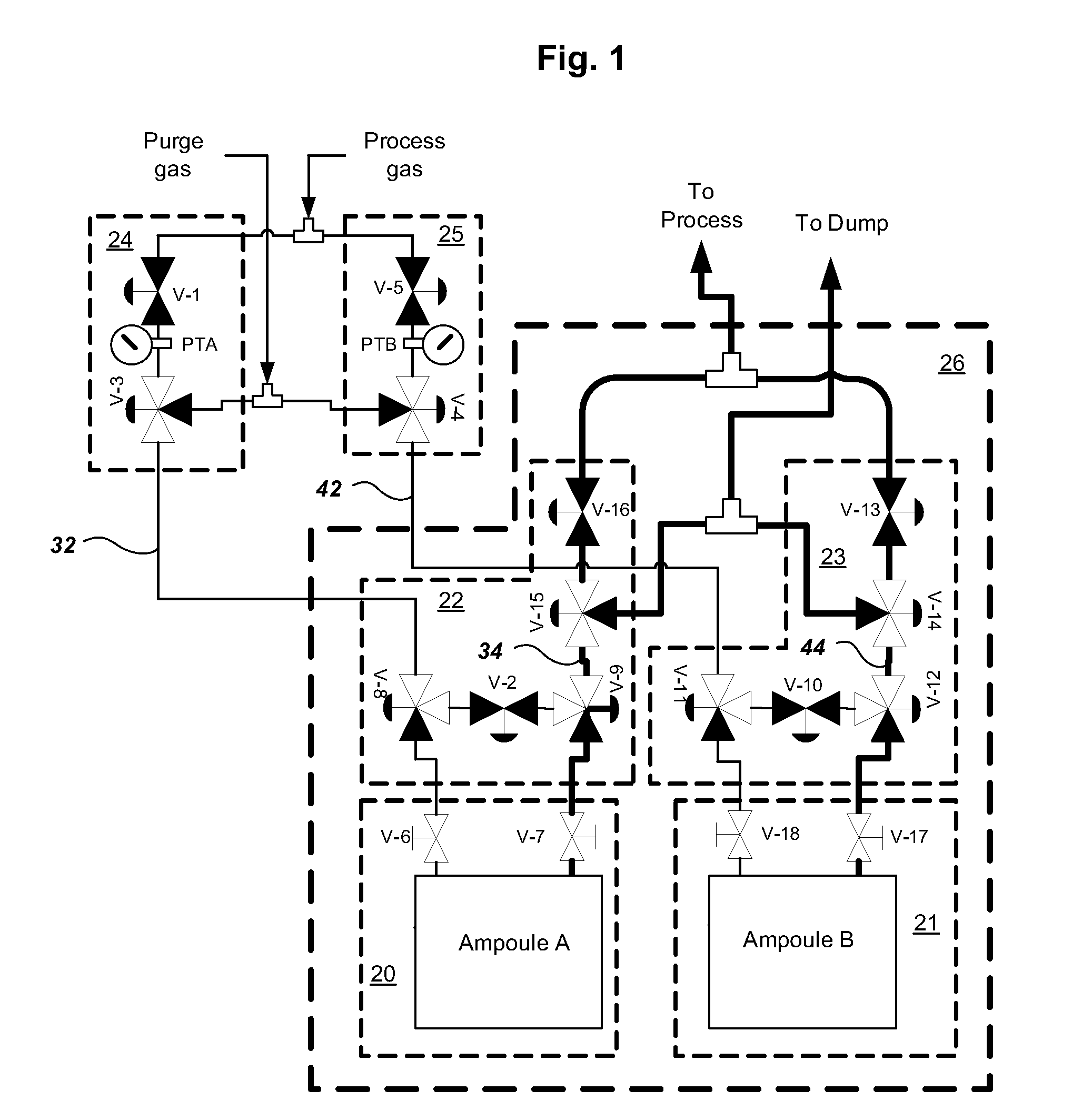
Multiple ampoule delivery systems
InactiveUS20090211525A1Easy to useReduce wasteLiquid surface applicatorsPipeline systemsSemiconductor materialsDelivery system
This invention relates to an integrated vapor or liquid phase reagent dispensing apparatus having a plurality of vessels and a plurality of carrier or inert gas feed / vapor or liquid phase reagent delivery manifolds, that may be used for continuously dispensing vapor or liquid phase reagents such as precursors for deposition of materials in the manufacture of semiconductor materials and devices.
Owner:PRAXAIR TECH INC
Diamond coatings on reactor wall and method of manufacturing thereof
InactiveUS20020086501A1High purityIncrease resistanceFrom solid stateVacuum evaporation coatingMetallurgySemiconductor
A corrosion resistant component of semiconductor processing equipment such as a plasma chamber includes a diamond containing surface and process for manufacture thereof.
Owner:LAM RES CORP
Carbonitride coated component of semiconductor processing equipment and method of manufacturing thereof
InactiveUS20020094378A1High purityIncrease resistanceCellsElectric discharge tubesCorrosion resistantMetallurgy
A corrosion resistant component of semiconductor processing equipment such as a plasma chamber includes a carbonitride containing surface and process for manufacture thereof.
Owner:LAM RES CORP
Rigid porous carbon structures, methods of making, methods of using and products containing same
InactiveUS6099965AHigh accessible surface areaHigh activityPigmenting treatmentMaterial nanotechnologyFiberPorous carbon
This invention relates to rigid porous carbon structures and to methods of making same. The rigid porous structures have a high surface area which are substantially free of micropores. Methods for improving the rigidity of the carbon structures include causing the nanofibers to form bonds or become glued with other nanofibers at the fiber intersections. The bonding can be induced by chemical modification of the surface of the nanofibers to promote bonding, by adding "gluing" agents and / or by pyrolyzing the nanofibers to cause fusion or bonding at the interconnect points.
Owner:HYPERION CATALYSIS INT
Method of determining the nucleotide sequence of oligonucleotides and DNA molecules
InactiveUS6780591B2Eliminate needHigh purityMicrobiological testing/measurementRecombinant DNA-technologyFluorescenceOligonucleotide primers
The present invention relates to a novel method for analyzing nucleic acid sequences based on real-time detection of DNA polymerase-catalyzed incorporation of each of the four nucleotide bases, supplied individually and serially in a microfluidic system, to a reaction cell containing a template system comprising a DNA fragment of unknown sequence and an oligonucleotide primer. Incorporation of a nucleotide base into the template system can be detected by any of a variety of methods including but not limited to fluorescence and chemiluminescence detection. Alternatively, microcalorimetic detection of the heat generated by the incorporation of a nucleotide into the extending template system using thermopile, thermistor and refractive index measurements can be used to detect extension reactions.
Owner:LIFE TECH CORP
High efficiency solid-state light source and methods of use and manufacture
ActiveUS20050152146A1Eliminate needImprove light outputOptical radiation measurementPoint-like light sourceDevice materialFluorescence
A high-intensity light source is formed by a micro array of a semiconductor light source such as a LEDs, laser diodes, or VCSEL placed densely on a liquid or gas cooled thermally conductive substrate. The semiconductor devices are typically attached by a joining process to electrically conductive patterns on the substrate, and driven by a microprocessor controlled power supply. An optic element is placed over the micro array to achieve improved directionality, intensity, and / or spectral purity of the output beam. The light module may be used for such processes as, for example, fluorescence, inspection and measurement, photopolymerzation, ionization, sterilization, debris removal, and other photochemical processes.
Owner:SILICON VALLEY BANK
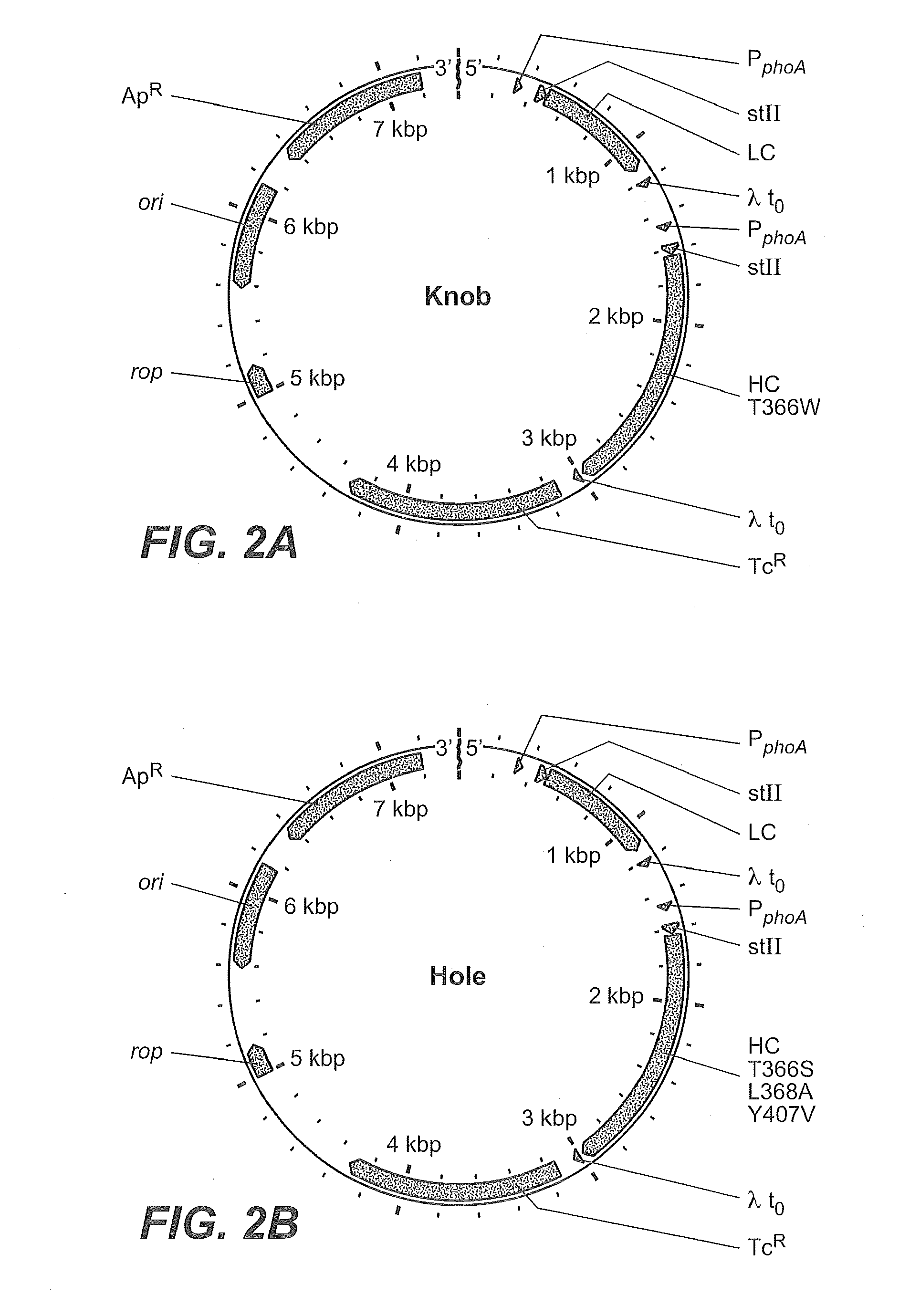
Production of Heteromultimeric Proteins
ActiveUS20110287009A1Reduction in yieldDecreased/elimination of effector functionAntipyreticAnalgesicsEpitopeBiochemistry
Described herein are methods for the efficient production of antibodies and other multimeric protein complexes (collectively referred to herein as heteromultimeric proteins) capable of specifically binding to more than one target. The targets may be, for example, different epitopes on a single molecule or located on different molecules. The methods combine efficient, high gene expression level, appropriate assembly, and ease of purification for the heteromultimeric proteins. The invention also provides methods of using these heteromultimeric proteins, and compositions, kits and articles of manufacture comprising these antibodies.
Owner:F HOFFMANN LA ROCHE & CO AG
Multi-formed collagenous biomaterial medical device
InactiveUS6666892B2Great benefitFavorable immune responseStentsSurgical adhesivesBiomedical engineeringMedical device
Owner:COOK BIOTECH
Method of reforming insulating film deposited on substrate with recess pattern
ActiveUS10283353B2Improve propertiesHigh puritySemiconductor/solid-state device manufacturingAtmosphereMetal
A method of reforming an insulating film deposited on a substrate having a recess pattern constituted by a bottom and sidewalls, includes: providing the film deposited on the substrate having the recess pattern in an evacuatable reaction chamber, wherein a property of a portion of the film deposited on the sidewalls is inferior to that of a portion of the film deposited on a top surface of the substrate; adjusting a pressure of an atmosphere of the reaction chamber to 10 Pa or less, which atmosphere is constituted by H2 and / or He without a precursor and without a reactant; and applying RF power to the atmosphere of the pressure-adjusted reaction chamber to generate a plasma to which the film is exposed, thereby reforming the portion of the film deposited on the sidewalls to improve the property of the sidewall portion of the film.
Owner:ASM IP HLDG BV
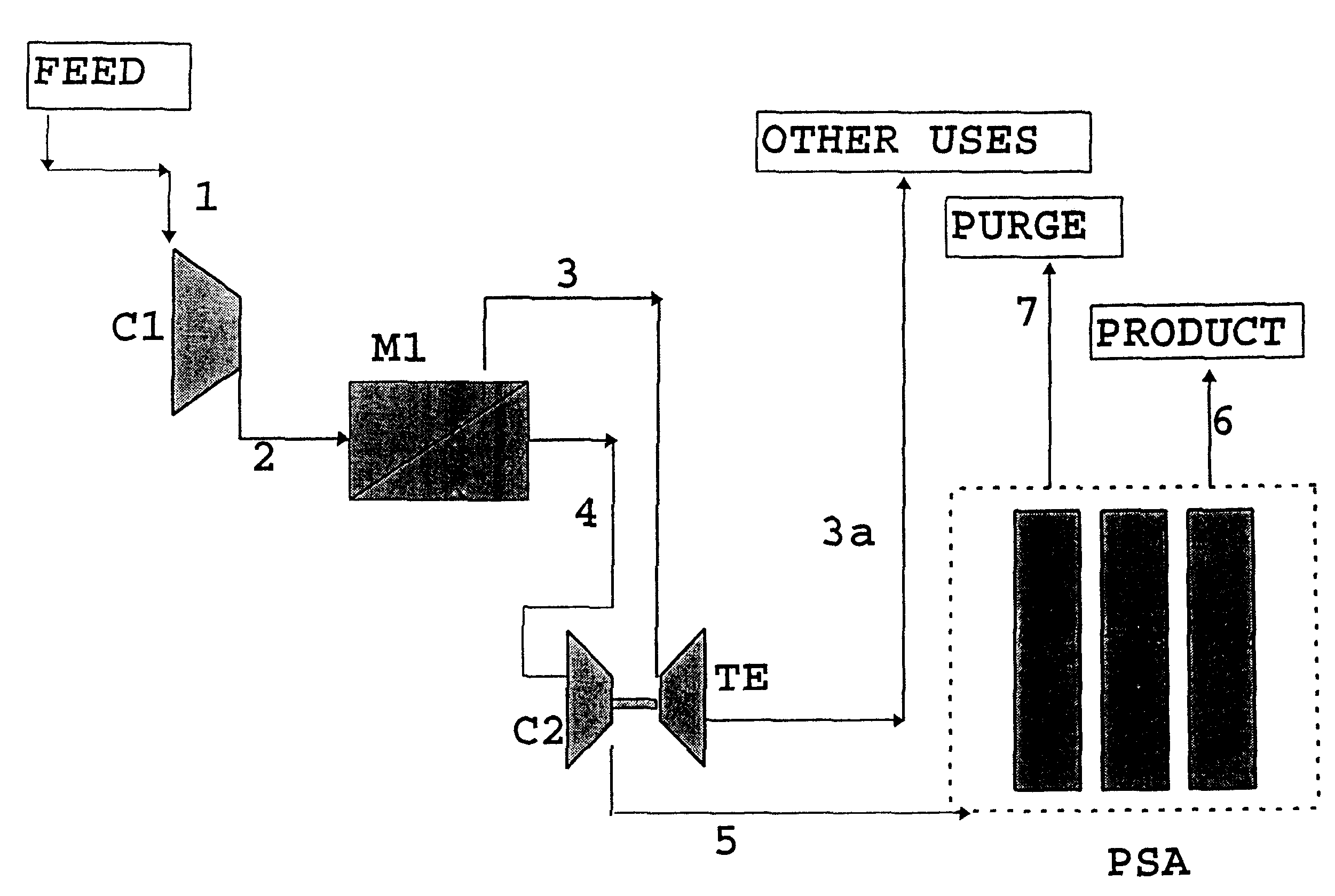
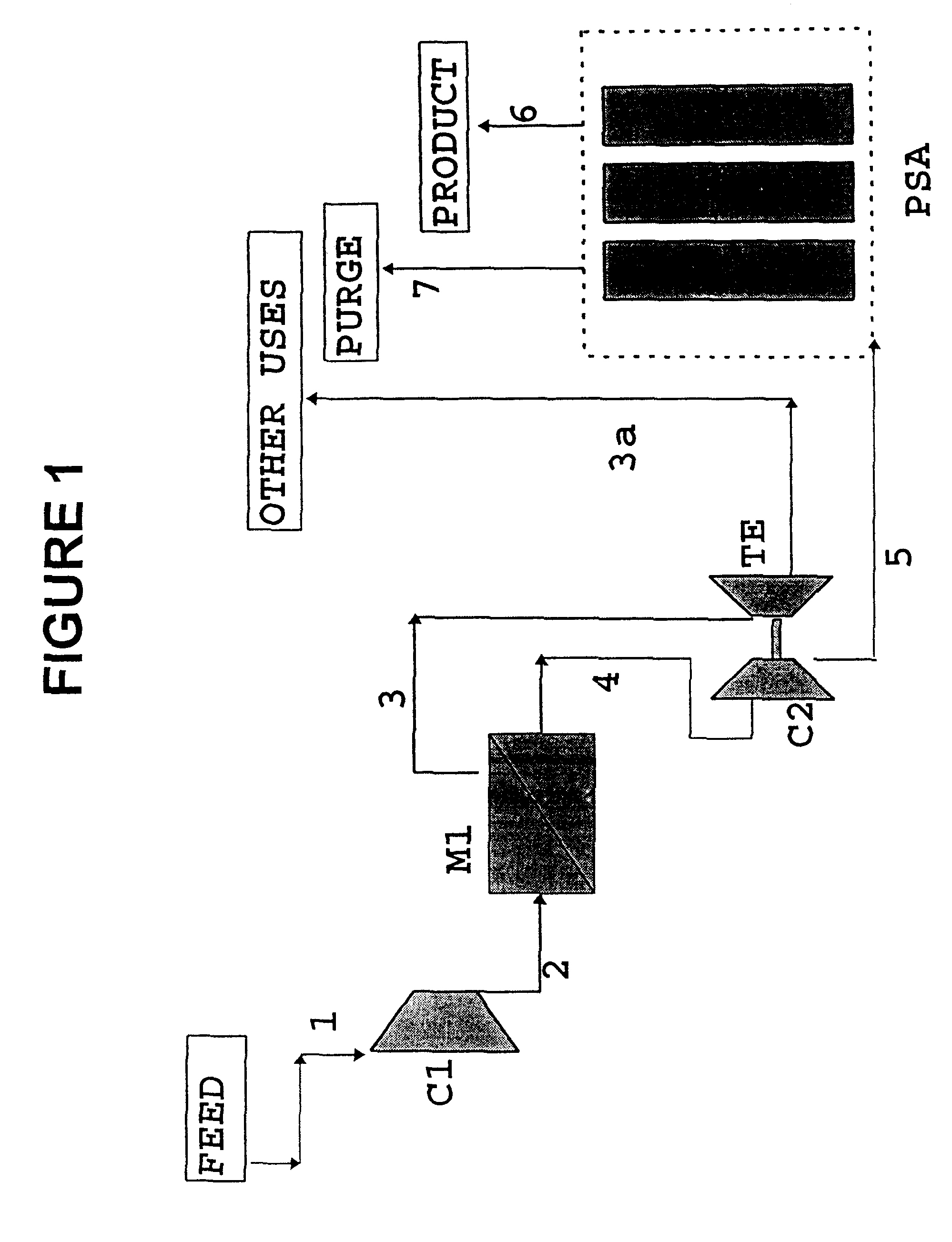
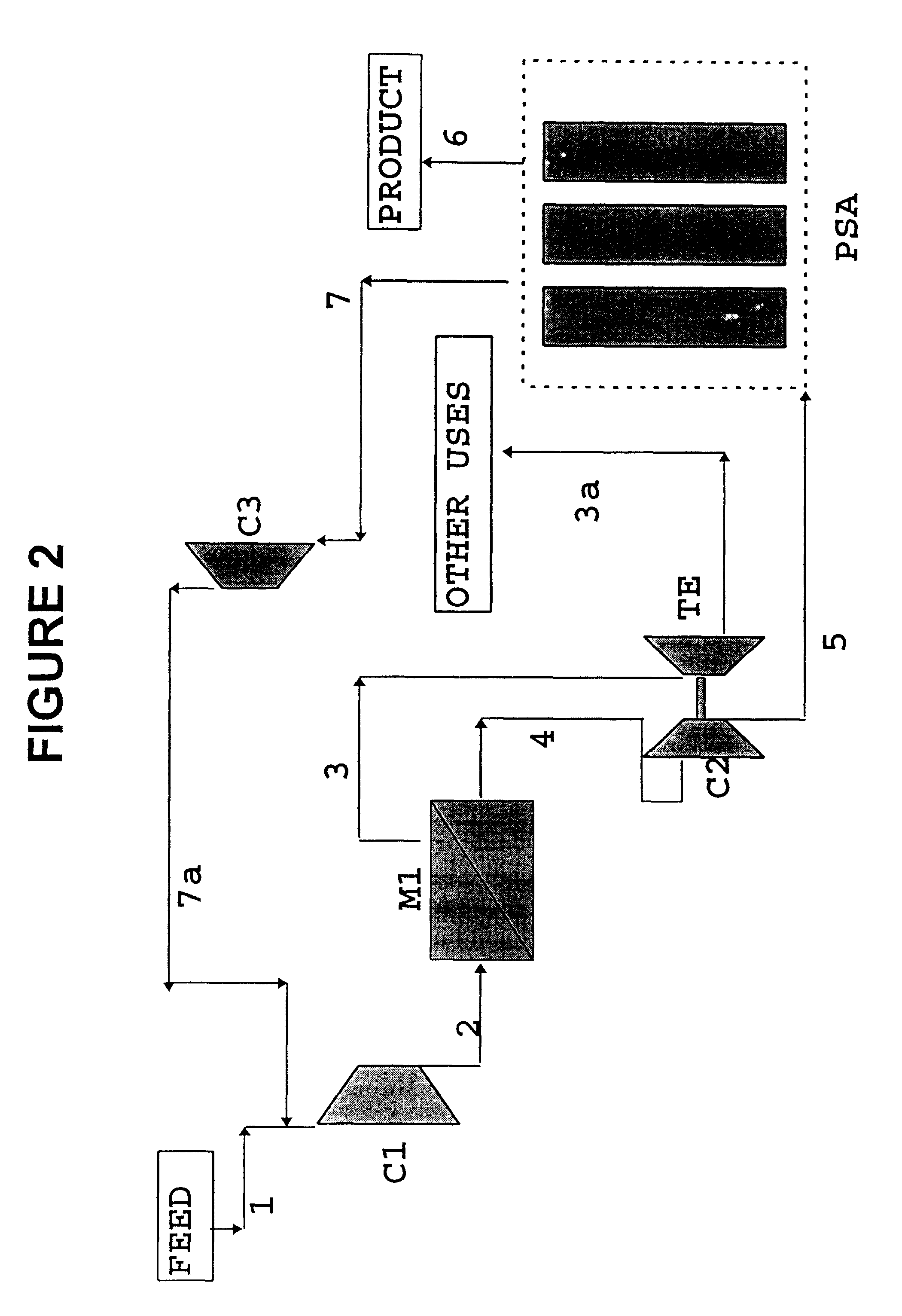
High frequency pressure swing adsorption
InactiveUS6176897B1High purityRecoverable expansion workNitrogen purification/separationGas treatmentSorbentEngineering
Pressure swing adsorption separation of a feed gas mixture, to obtain a purified product gas of the less strongly adsorbed fraction of the feed gas mixture, is performed in a plurality of preferably an even number of adsorbent beds, with each adsorbent bed communicating at its product end directly to a variable volume expansion chamber, and at its feed end by directional valves to a feed compressor and an exhaust vacuum pump. For high frequency operation of the pressure swing adsorption cycle, a high surface area layered support is used for the adsorbent. The compressor and vacuum pump pistons may be integrated with the cycle, reciprocating at twice the cycle frequency. Alternative configurations of the layered adsorbent beds are disclosed.
Owner:AIR PROD & CHEM INC
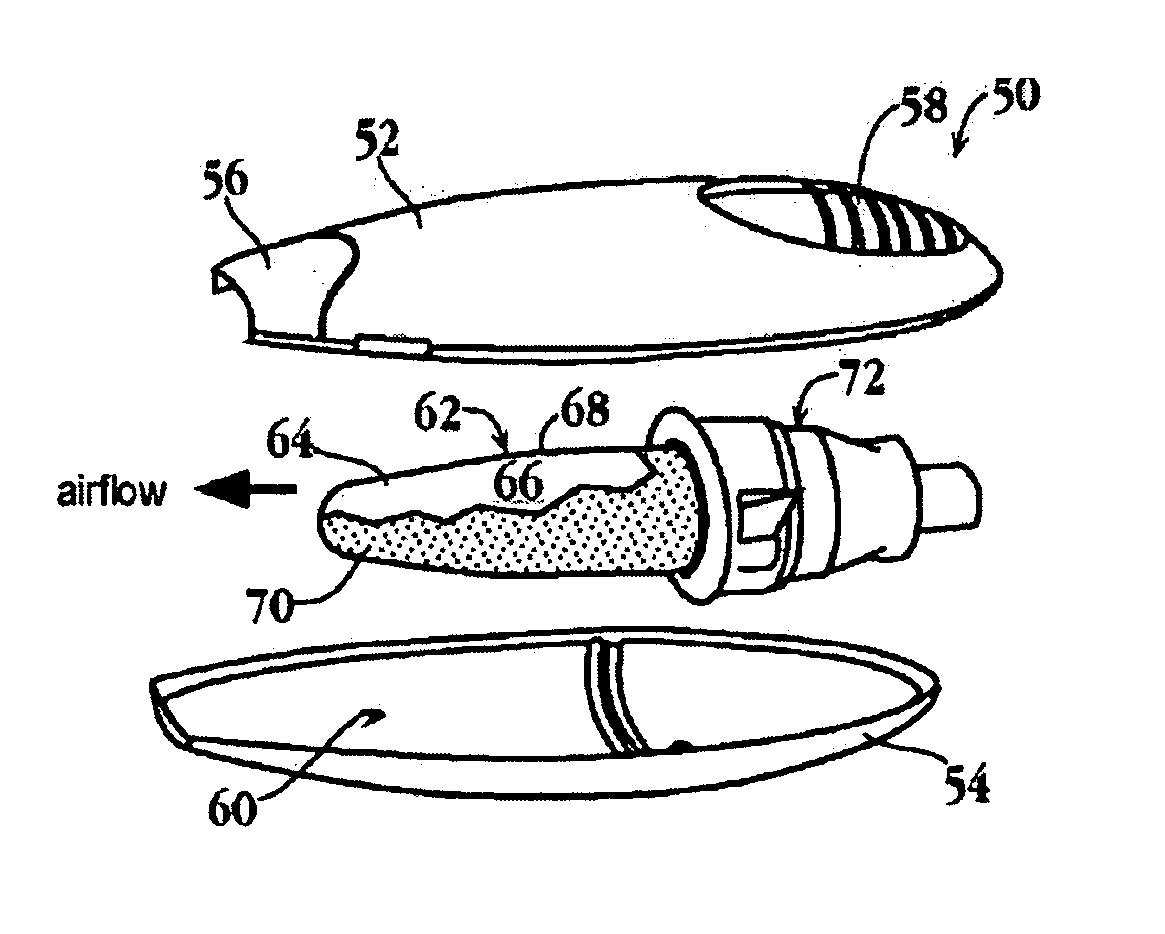
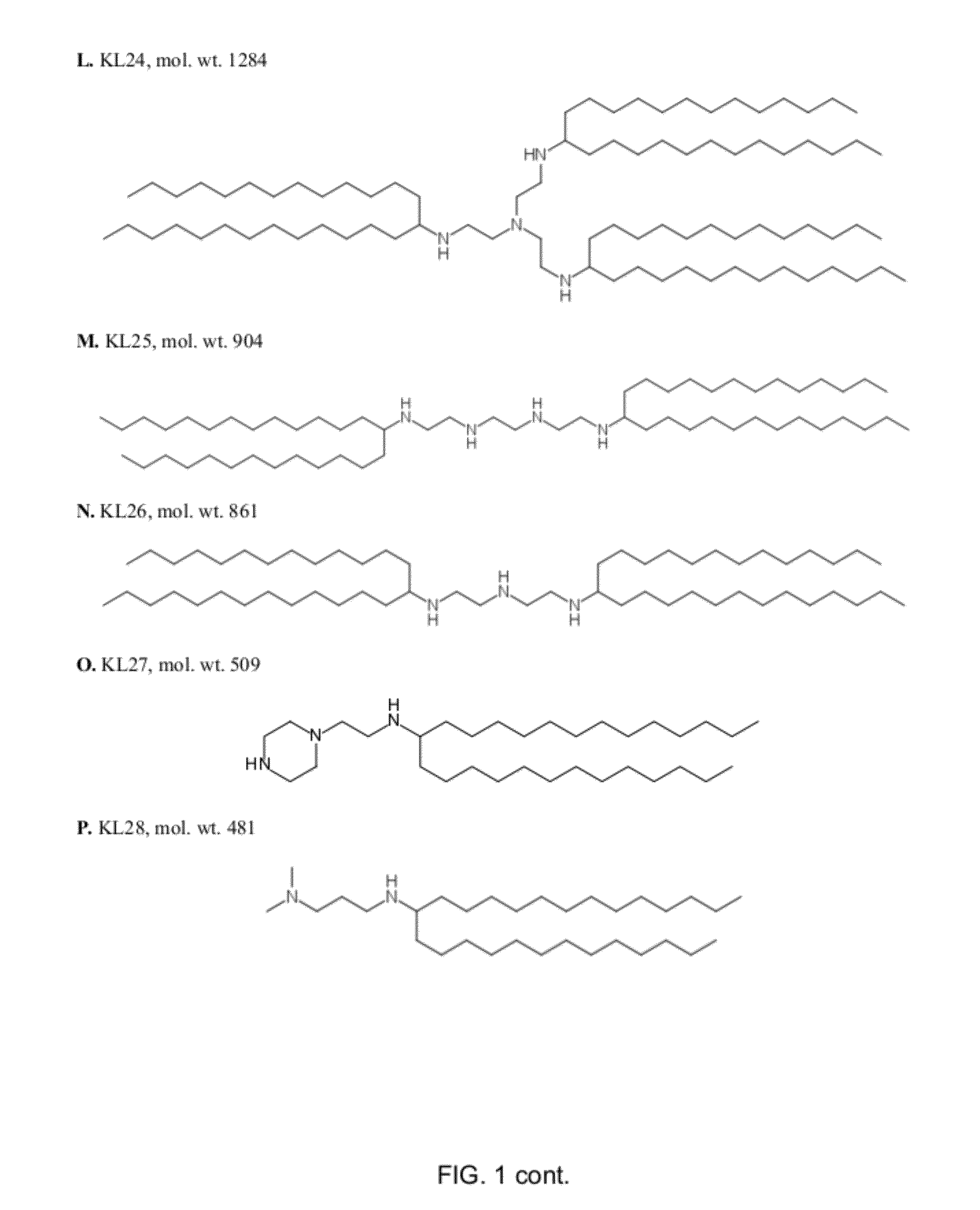
Lipids and compositions for intracellular delivery of biologically active compounds
ActiveUS8691750B2High yieldHigh purityPowder deliveryMaterial nanotechnologyLipid formationIntracellular drug delivery
The present invention provides novel amino-lipids, compositions comprising such amino-lipids and methods of producing them. In addition, lipid nanoparticles comprising the novel amino-lipids and a biologically active compound are provided, as well as methods of production and their use for intracellular drug delivery.
Owner:AXOLABS
Substrates for drug delivery device and methods of preparing and use
An assembly and method for producing a condensation aerosol are disclosed. The assembly includes a heat-conductive metal substrate with an oxidation resistant exterior surface and a drug composition film on the exterior surface and is for use in an aerosol device. The thickness of the film and the surface of the substrate is such that the aerosol formed by vaporizing and condensing the drug composition the aerosol contain 10% by weight or less drug-degradation products and at least 50% of the total amount of the drug composition in the film. The methods for treating the exterior surface include heat and chemical treatment and formation of a protective overcoat.
Owner:ALEXZA PHARMA INC
Novel Lipids and Compositions for Intracellular Delivery of Biologically Active Compounds
ActiveUS20120295832A1High yieldHigh purityPowder deliveryBiocideLipid formationIntracellular drug delivery
The present invention provides novel amino-lipids, compositions comprising such amino-lipids and methods of producing them. In addition, lipid nanoparticles comprising the novel amino-lipids and a biologically active compound are provided, as well as methods of production and their use for intracellular drug delivery.
Owner:AXOLABS
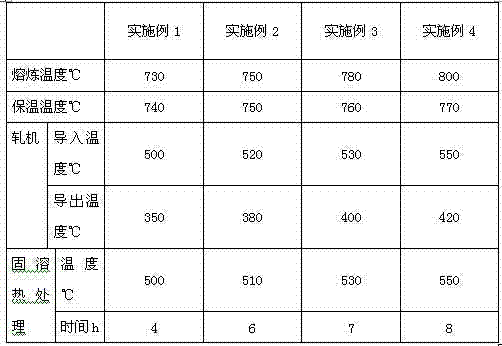
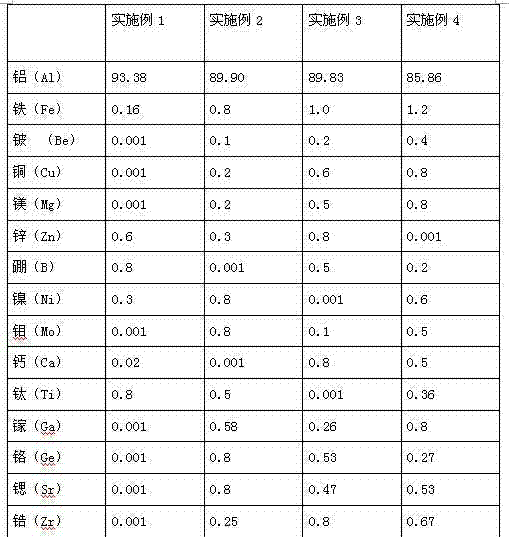
A kind of aluminum alloy material and preparation method thereof
The invention relates to an aluminum alloy material which is characterized in that the aluminum alloy material comprises the following components by weight percent: 0.16-1.2% of Fe, 0.001-0.8% of Cu, 0.001-0.8% of Mg, 0.001-0.8% of Zn, 0.001-0.8% of Ca, 0.001-1.0% of rare-earth elements, a trace amount of strontium, titanium, boron, nickel, chromium, zirconium, vanadium, beryllium, cobalt, lead, tin, bismuth, molybdenum, silver, indium, niobium and barium and the balance of aluminum. The alloy has excellent mechanical strength, processing performance and corrosion resistance and is suitable for the cable armored sheath.
Owner:GUANGDONG XINYI ALUMINUM ALLOY CABLE
Thermoplastic copolymer of tetrafluoroethylene and perfluoromethyl vinyl ether and medical devices employing the copolymer
An improved elastomeric material is described comprising an essentially noncross-linkable amorphous copolymer of tetrafluoroethylene (TFE) and perfluoromethyl vinyl ether (PMVE) which is both a thermoplastic and exhibits exceptional mechanical properties. This material is particularly suitable for use in ultra-clean environments, and particularly for use in an implantable device, since it does not contain contaminants that previous thermoset TFE / PMVE copolymers have required. Among the improved properties of the present invention are excellent biocompatibility, high matrix tensile strength, high clarity, high abrasion resistance, high purity, adequate elasticity, and ease of processing due to the thermoplastic, and noncross-linkable structure of the copolymer. The material of the present invention is also a high strength bonding agent particularly suited for bonding porous PTFE to itself or to other porous substances at room or elevated temperatures.
Owner:WL GORE & ASSOC INC
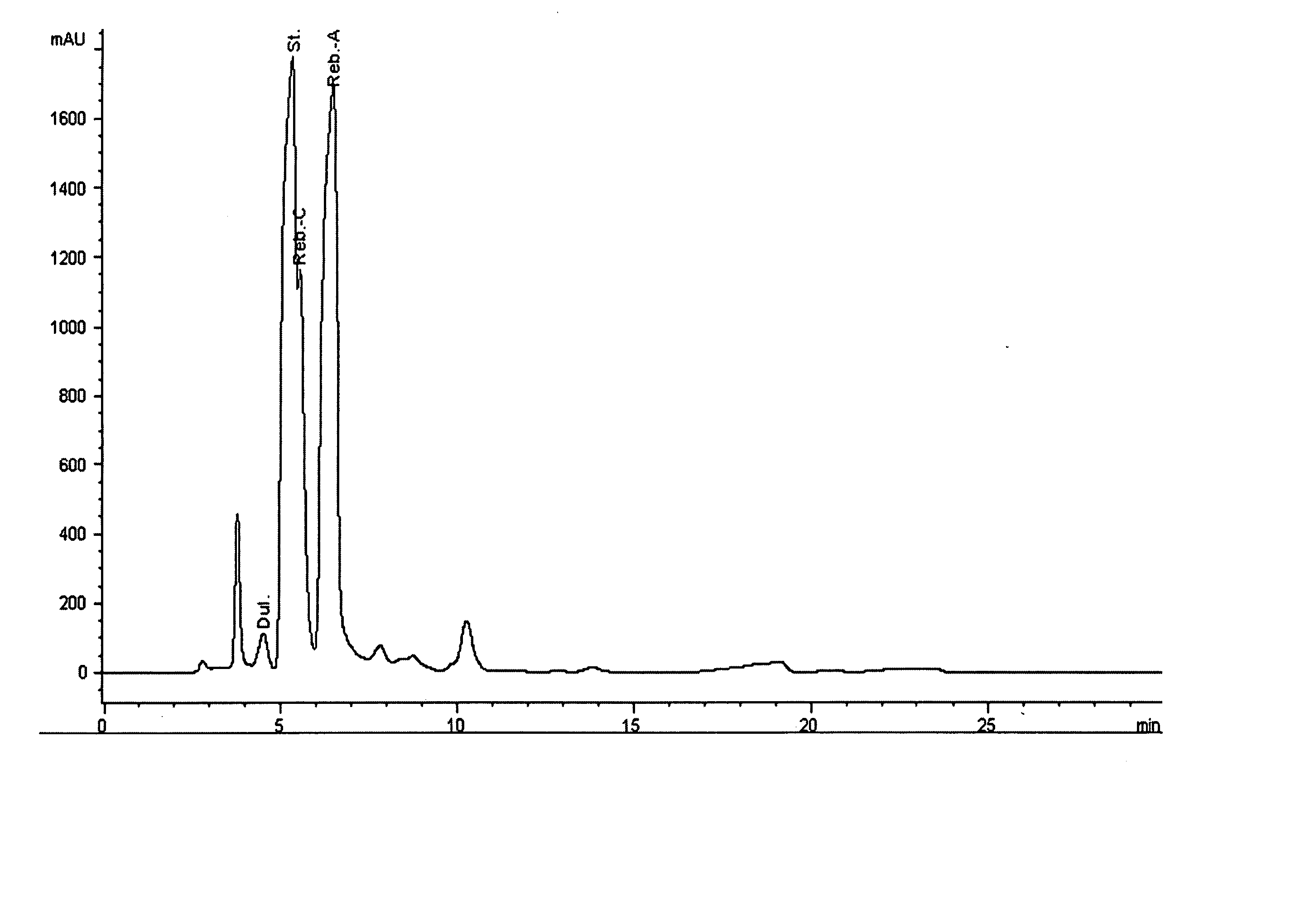
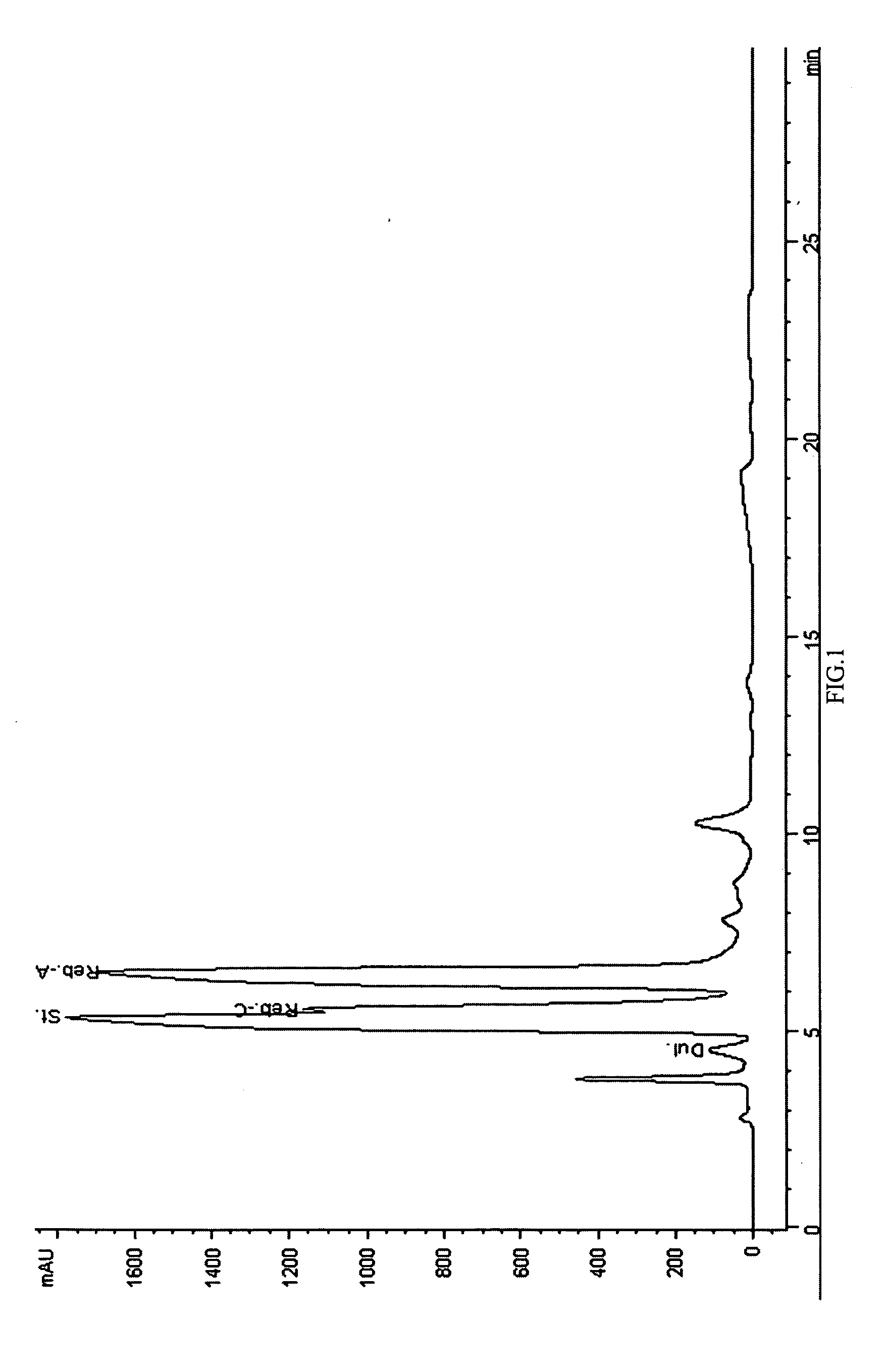
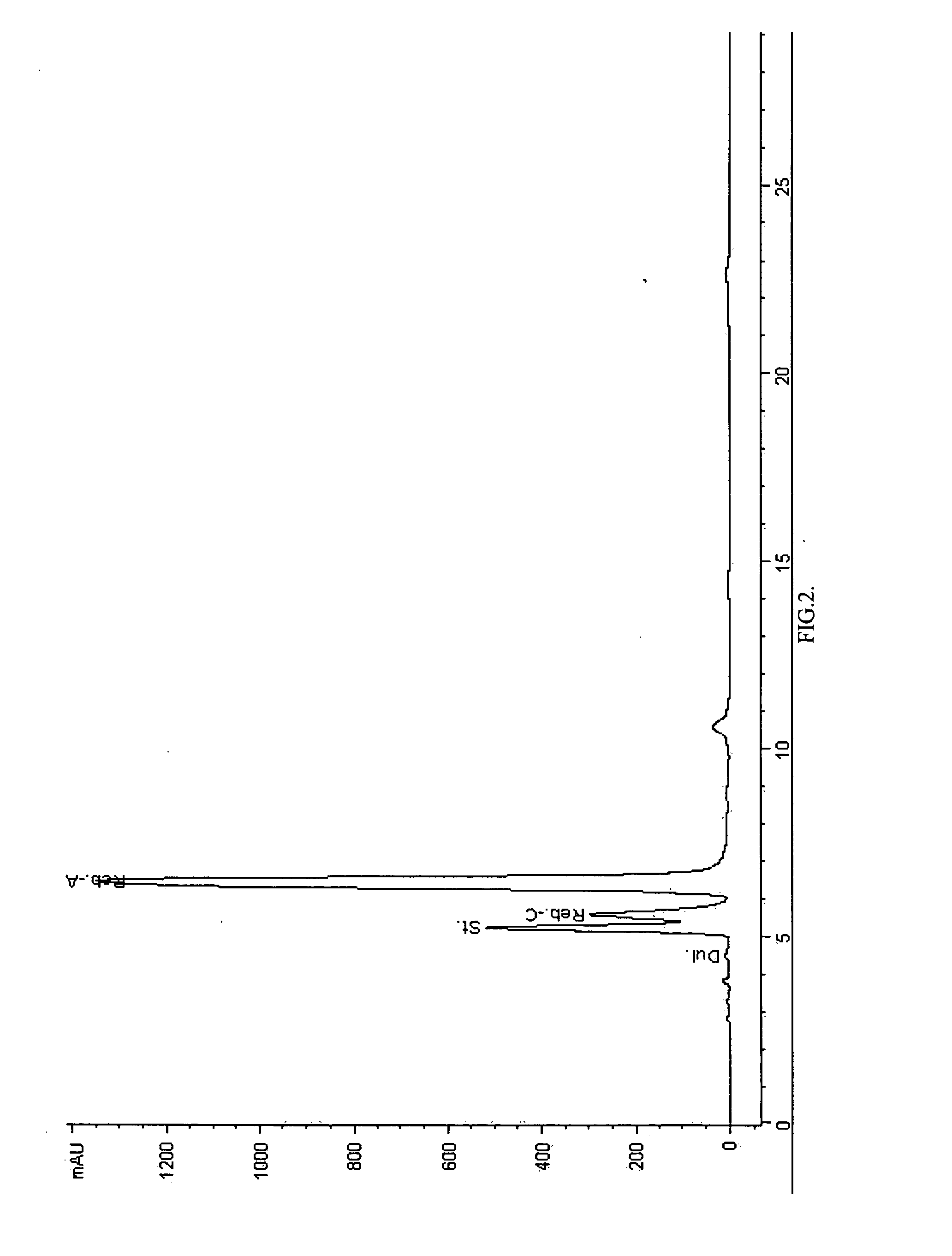
Extraction, separation and modification of sweet glycosides from the stevia rebaudiana plant
ActiveUS20060134292A1Increasing of degree of transglycosylationDelayed reaction timeBiocideAnimal repellantsPectinaseSodium Bentonite
The invention disclosed relates to a method for the extraction of sweet glycosides from the Stevia rebaudiana Bertoni plant and recovery of individual rebaudioside A and stevioside. The extraction is developed in the presence of pectinase, and the extract is purified using cyclodextrin and bentonite. High purity rebaudioside A is obtained by crystallization and recrystallization from ethanol. High purity stevioside is prepared from the filtrate by purification with cyclodextrin, bentonit, and ion exchange resins. The enzymatic modification of the rebaudioside A, stevioside and the purified extract is carried out using the transferring enzymes derived from Thermoactinomyces vilgaris and Bacillus halophilus.
Owner:PURECIRCLE SDN BHD
Carbon/silicon/carbon nano composite structure cathode material and preparation method thereof
The invention discloses a carbon / silicon / carbon nano composite structure cathode material and a preparation method thereof, belonging to the technical field of electrochemical power supply technologies. The cathode material consists of a carbon-based conductive substrate, nano silicon and a nano carbon coating layer, wherein the nano silicon is uniformly distributed on the carbon-based conductive substrate; the nano carbon coating layer is arranged on the surface of the nano silicon; the carbon-based conductive substrate is porous carbon, a carbon nanotube or graphene; the nano silicon exists in the state of nanoparticles or nano films; the weight percentage of the nano silicon in the cathode material is 10-90 percent; and the thickness of the nano carbon coating layer is 0.1-10 nanometers. The preparation method comprises the following steps of: depositing nano silicon on the carbon substrate in a reaction space in oxygen-free atmosphere by adopting a chemical vapor deposition process; and coating nano carbon on the surface of the nano silicon by adopting the chemical vapor deposition process. In the obtained carbon / silicon / carbon composite cathode material, the volume change of a silicon electrode material is controlled effectively in the charging and discharging processes, the electrode structure is kept complete, the circulation volume is large, the circulation service life is long, and the electrochemical performance is high.
Owner:TSINGHUA UNIV
Multi-channel mode converter and rotary joint operating with a series of TE or TM mode electromagnetic wave
A multi-channel mode converter operating with a series of TE or TM mode electromagnetic wave includes a plurality of coaxial waveguides arranged in overlay configuration. By controlling radius ratio and the number of coupling aperture of each coaxial waveguide, high power and high purity of operating mode of electromagnetic wave can be obtained and the major parasitic mode of electromagnetic wave can be suppressed, so as to avoid crosstalk between coaxial waveguides. A rotary joint including the above-mentioned mode converter with multi-channel is also disclosed.
Owner:NATIONAL TSING HUA UNIVERSITY
Method of Determining The Nucleotide Sequence of Oligonucleotides and DNA Molecules
InactiveUS20080213770A1Eliminate needHigh puritySugar derivativesMicrobiological testing/measurementFluorescenceOligonucleotide primers
The present invention relates to a novel method for analyzing nucleic acid sequences based on real-time detection of DNA polymerase-catalyzed incorporation of each of the four nucleotide bases, supplied individually and serially in a microfluidic system, to a reaction cell containing a template system comprising a DNA fragment of unknown sequence and an oligonucleotide primer. Incorporation of a nucleotide base into the template system can be detected by any of a variety of methods including but not limited to fluorescence and chemiluminescence detection. Alternatively, microcalorimetic detection of the heat generated by the incorporation of a nucleotide into the extending template system using thermopile, thermistor and refractive index measurements can be used to detect extension reactions.
Owner:LIFE TECH CORP
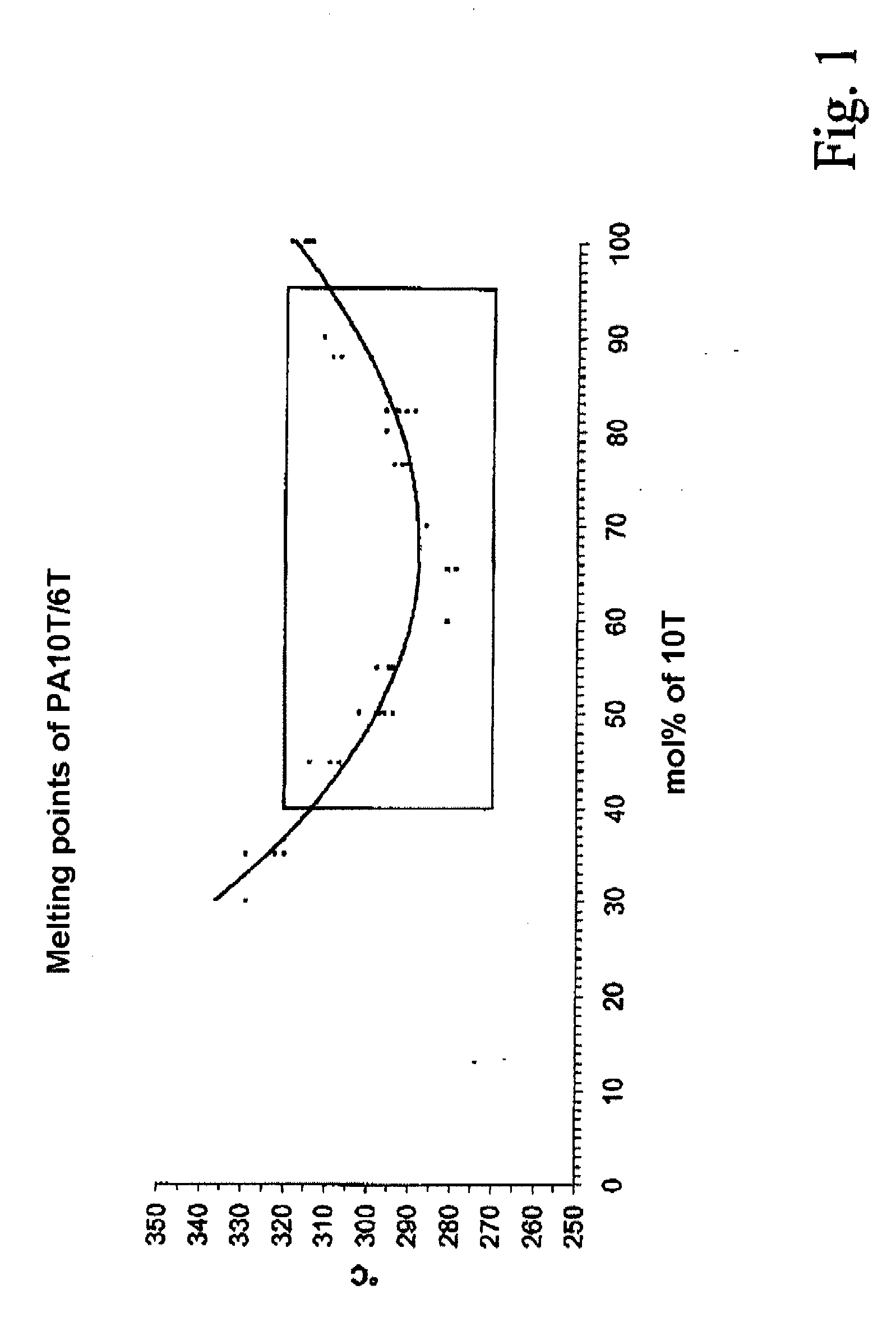
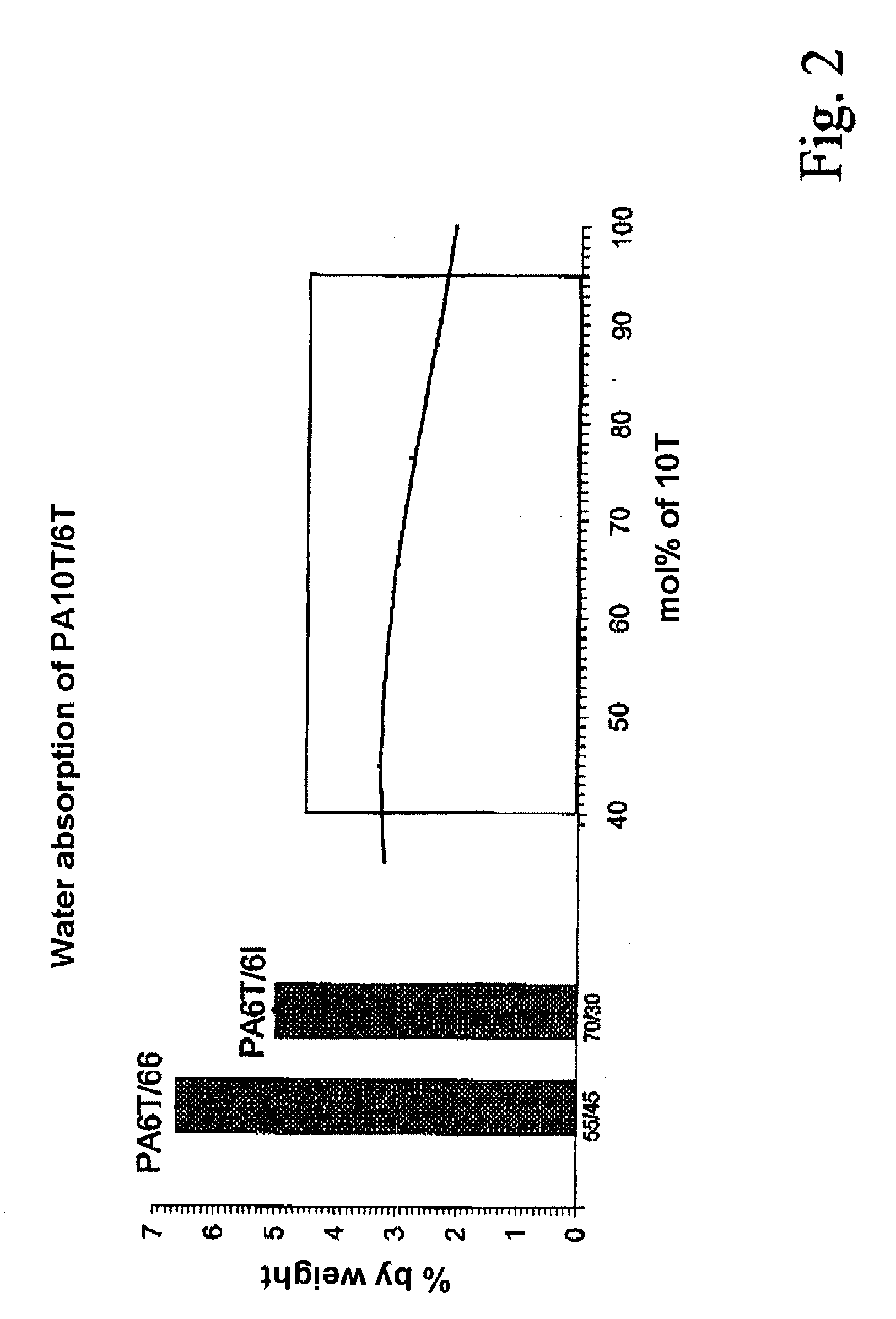
Semiaromatic polyamide molding compositions and their use
ActiveUS20080274355A1High strengthLow water absorptionPigmenting treatmentNon-fibrous pulp additionHexamethylenediamineAramides
A polyamide molding composition with the following constitution is described:(A) from 30 to 100% by weight of at least one 10T / 6T copolyamide, where this is composed of(A1) from 40 to 95 mol % of 10T units, formed from the monomers 1,10-decanediamine and terephthalic acid(A2) from 5 to 60 mol % of 6T units, formed from the monomers 1,6-hexanediamine and terephthalic acid(B) from 0 to 70% by weight of reinforcing materials and / or fillers(C) from 0 to 50% by weight of additives and / or further polymerswhere the entirety of components A to C is 100%, with the proviso that in component (A) up to 30 mol %, based on the entirety of the dicarboxylic acids, of the terephthalic acid can have been replaced by other aromatic, aliphatic, or cycloaliphatic dicarboxylic acids, and with the proviso that in component (A) up to 30 mol % of 1,10-decanediamine and respectively 1,6-hexanediamine, based on the entirety of the diamines, can have been replaced by other diamines, and with the proviso that not more than 30 mol % in component (A), based on the entirety of the monomers, can have been formed via lactams or amino acids. Uses of this polyamide molding composition are moreover described, as also are processes for the preparation of these polyamide molding compositions.
Owner:EMS PATENT AG
High purity tantalum, products containing the same, and methods of making the same
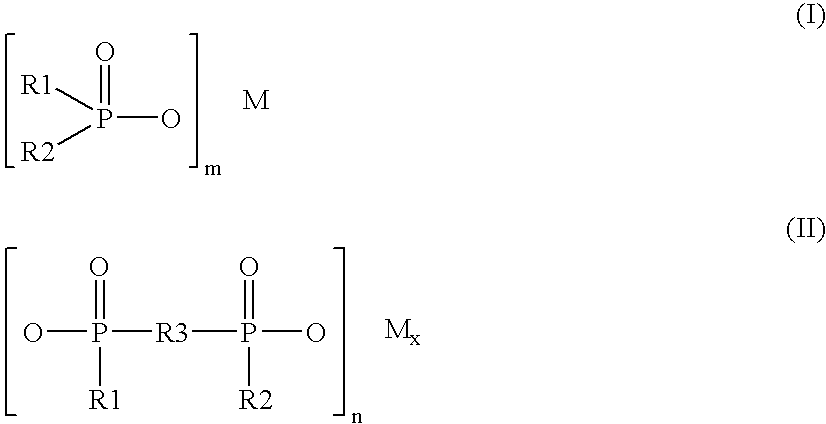
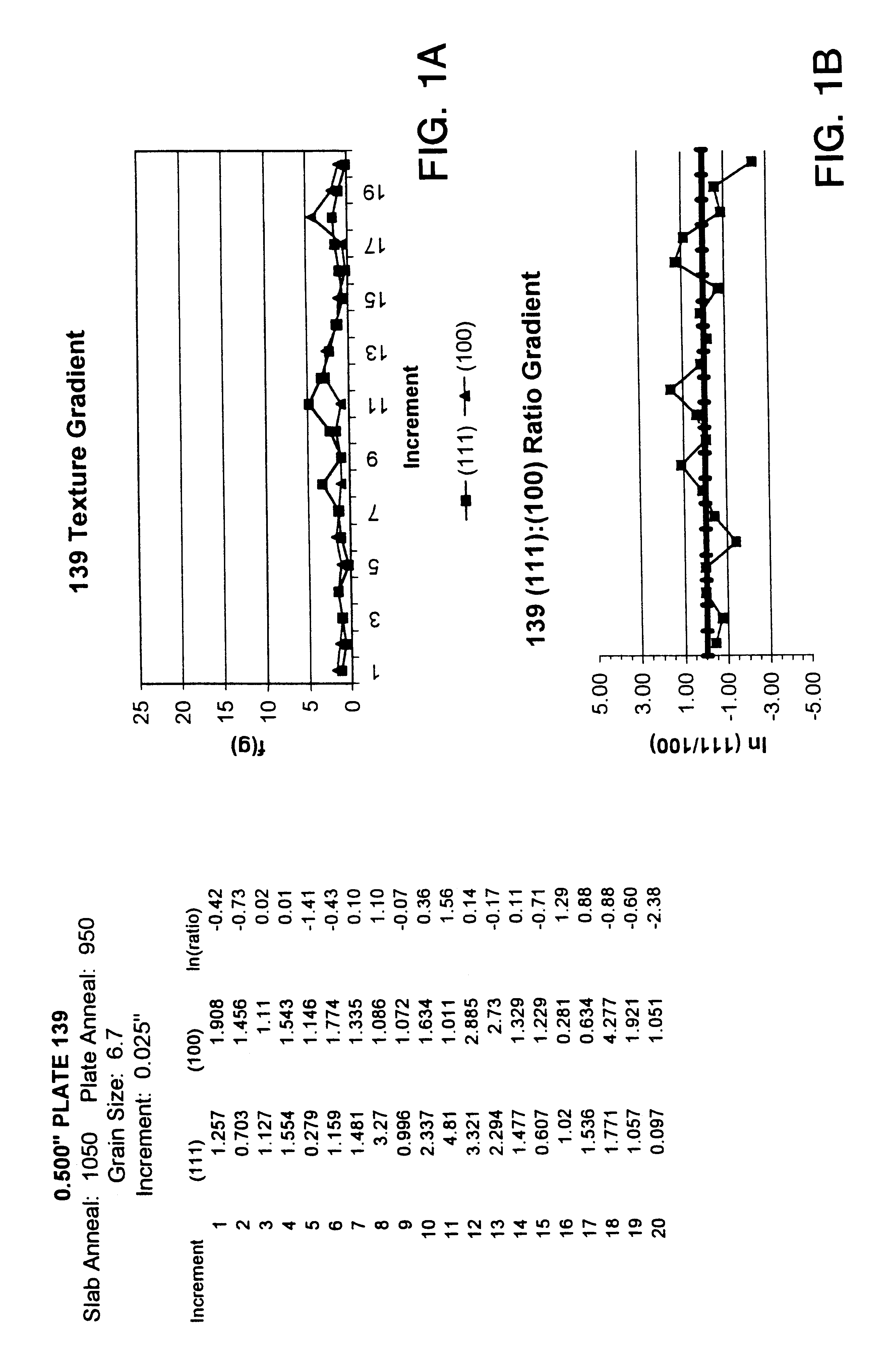
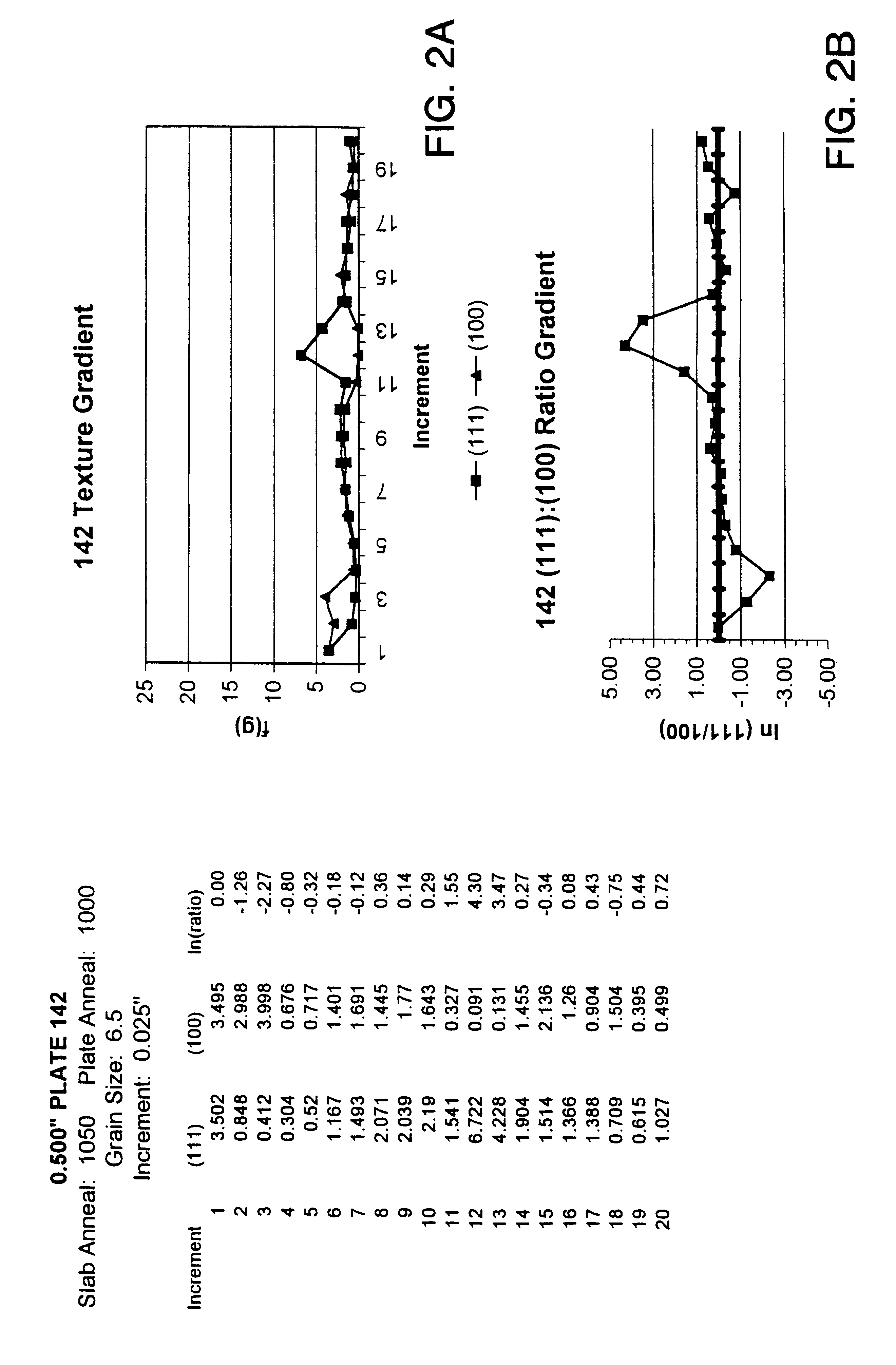
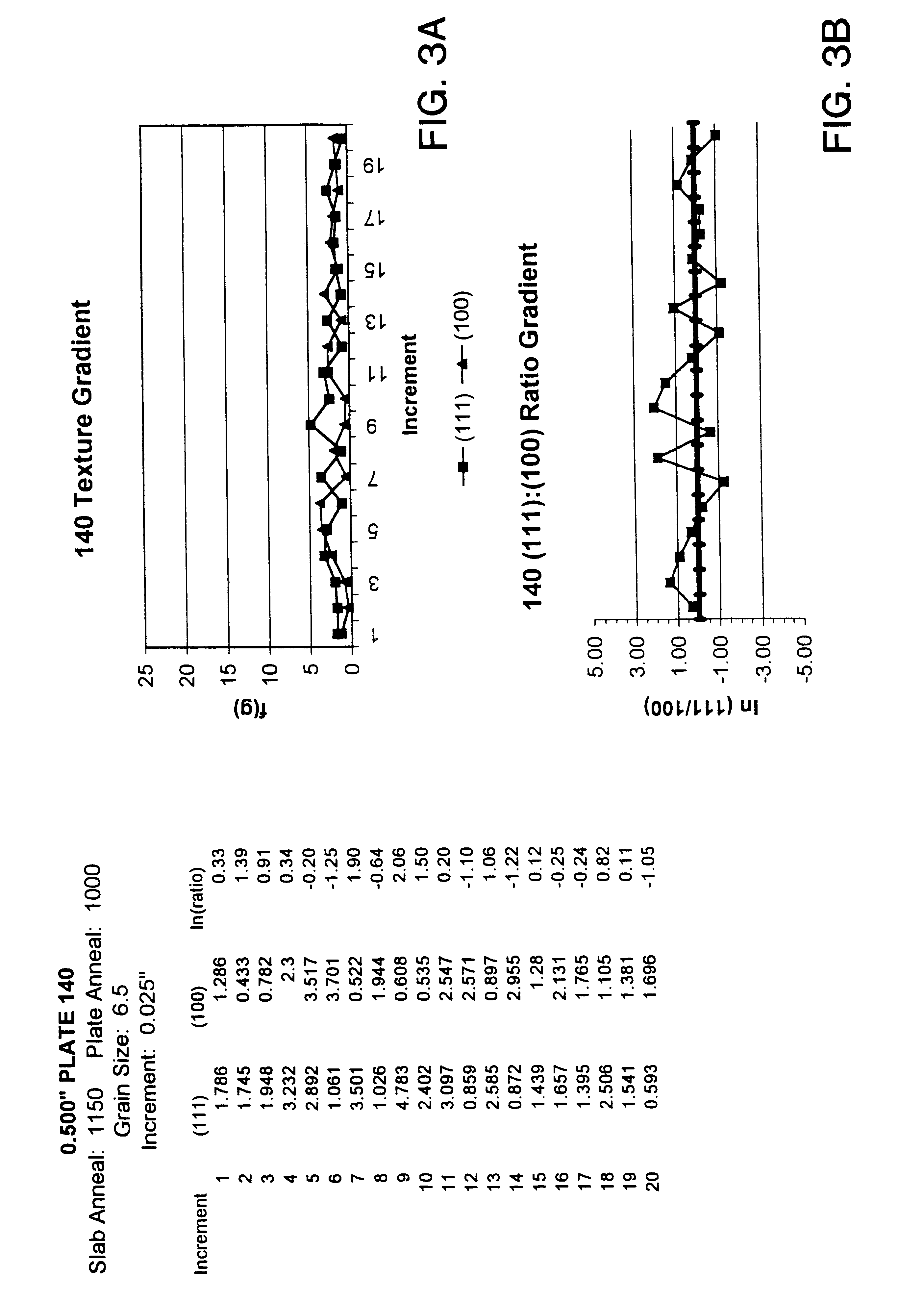
InactiveUS6348113B1Fine grain structureUniform textureVacuum evaporation coatingSputtering coatingMetallic materialsUltimate tensile strength
High purity tantalum metals and alloys containing the same are described. The tantalum metal preferably has a purity of at least 99.995% and more preferably at least 99.999%. In addition, tantalum metal and alloys thereof are described, which either have a grain size of about 50 microns or less, or a texture in which a (100) intensity within any 5% increment of thickness is less than about 15 random, or an incremental log ratio of (111):(100) intensity of greater than about -4.0, or any combination of these properties. Also described are articles and components made from the tantalum metal which include, but are not limited to, sputtering targets, capacitor cans, resistive film layers, wire, and the like. Also disclosed is a process for making the high purity metal which includes the step of reacting a salt-containing tantalum with at least one compound capable of reducing this salt to tantalum powder and a second salt in a reaction container. The reaction container or liner in the reaction container and the agitator or liner on the agitator are made from a metal material having the same or higher vapor pressure of melted tantalum. The high purity tantalum preferably has a fine and uniform microstructure.
Owner:GLOBAL ADVANCED METALS USA
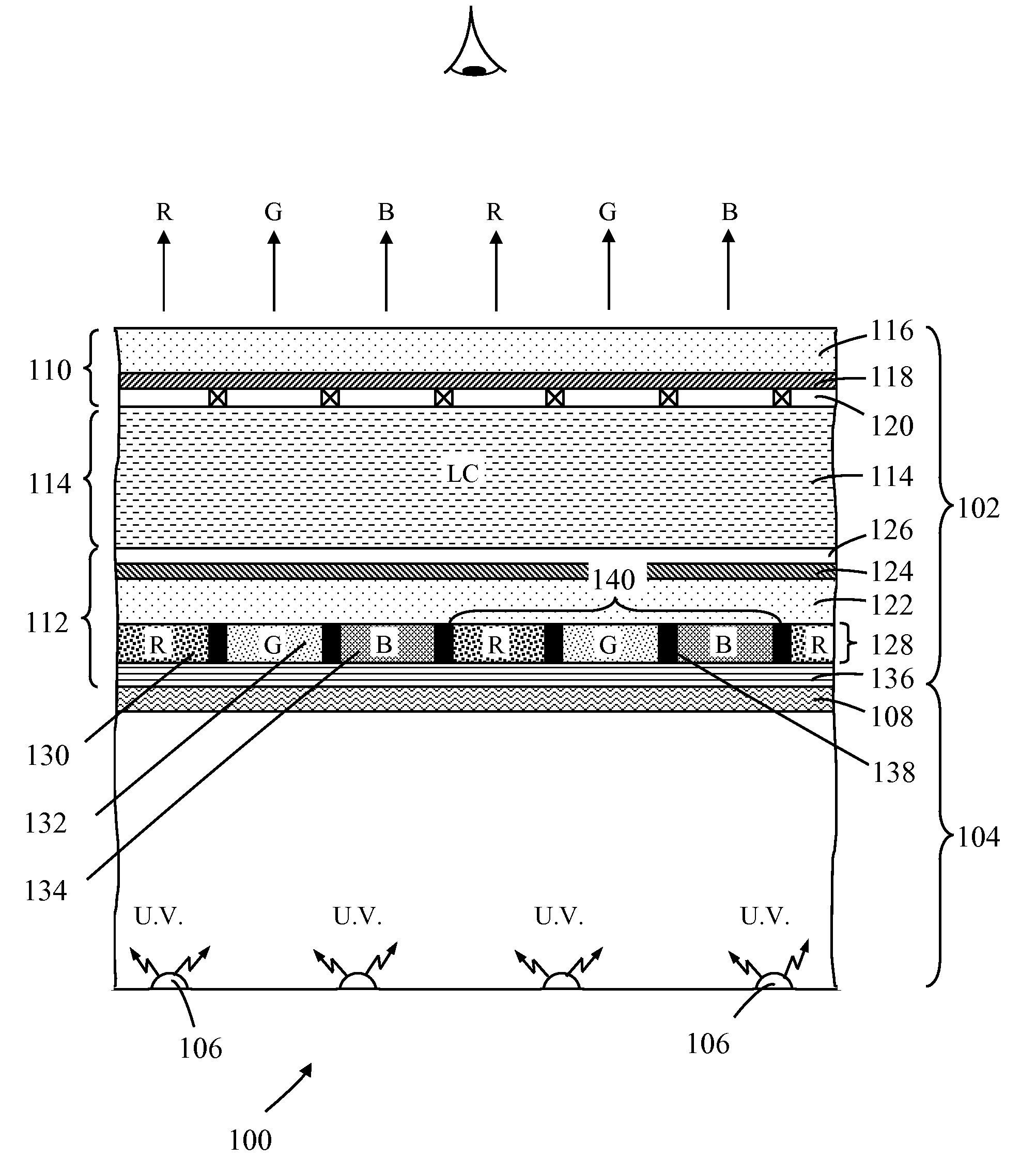
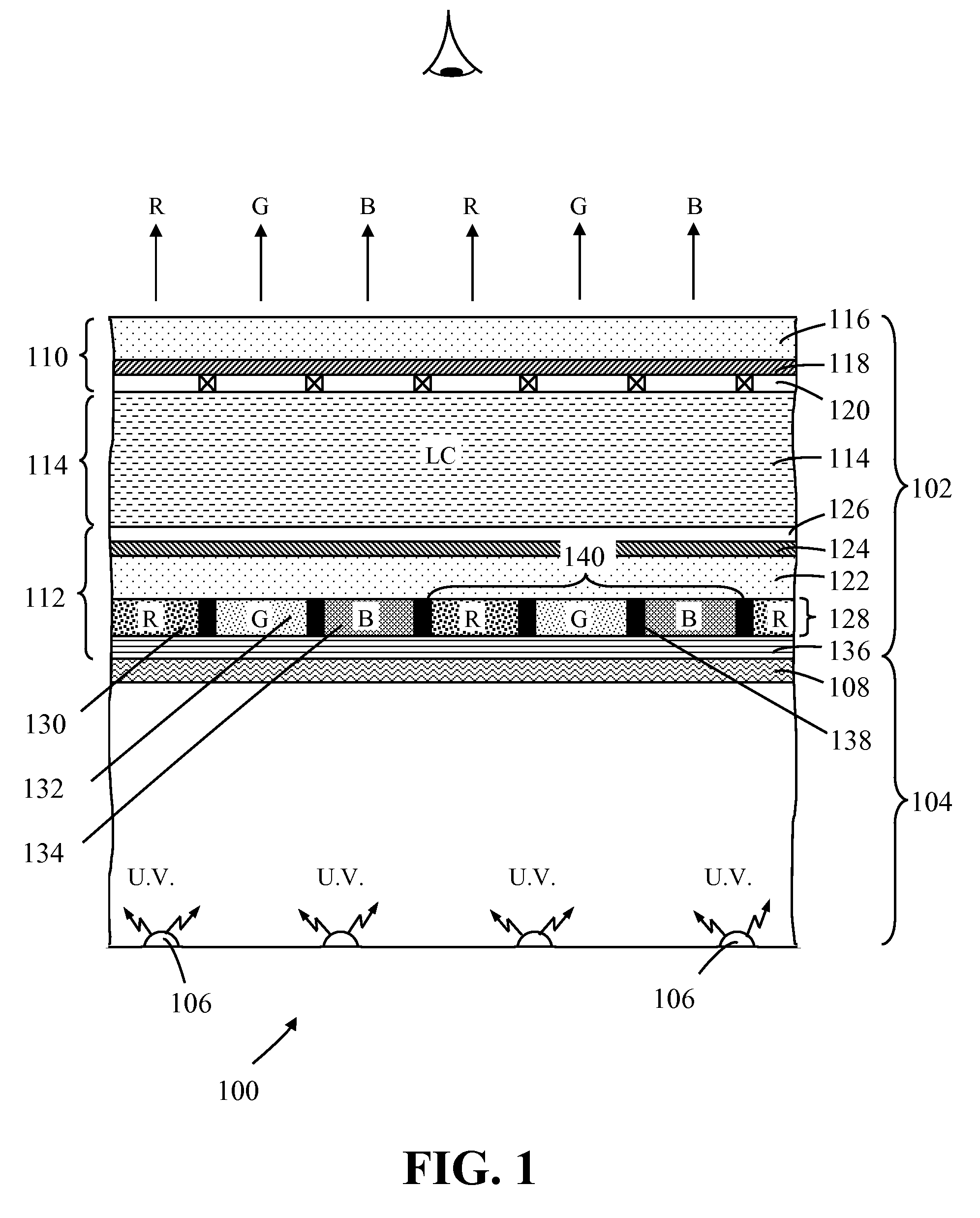
Photoluminescence color display
A photoluminescence color display comprises a display panel that displays red, green and blue pixel areas, an excitation source operable to generate excitation radiation for operating the display, and a photoluminescence color-element plate. The color-element plate comprises at least one photoluminescence material, such as a phosphor material or quantum dots, that is operable to emit light corresponding to red, green and blue pixel areas of the display in response to said excitation radiation. Additionally, the photo-luminescence color display comprises a wavelength selective filter that is provided between the color-element plate and the excitation source. The filter has a transmission characteristic that allows the passage of excitation radiation from the excitation source to excite the at least one photoluminescence material whilst preventing the passage of photoluminescence light back to the excitation source thereby prevent cross contamination of light among the different pixel areas of the display.
Owner:INTEMATIX
Method of determining the nucleotide sequence of oligonucleotides and DNA molecules
InactiveUS7875440B2Eliminate needHigh puritySugar derivativesMicrobiological testing/measurementOligonucleotide primersFluorescence
The present invention relates to a novel method for analyzing nucleic acid sequences based on real-time detection of DNA polymerase-catalyzed incorporation of each of the four nucleotide bases, supplied individually and serially in a microfluidic system, to a reaction cell containing a template system comprising a DNA fragment of unknown sequence and an oligonucleotide primer. Incorporation of a nucleotide base into the template system can be detected by any of a variety of methods including but not limited to fluorescence and chemiluminescence detection. Alternatively, microcalorimetic detection of the heat generated by the incorporation of a nucleotide into the extending template system using thermopile, thermistor and refractive index measurements can be used to detect extension reactions.
Owner:LIFE TECH CORP
Organic light emitting compound, organic light emitting device comprising the same, and method of manufacturing the organic light emitting device
InactiveUS20080124455A1Improve solubilityImprove thermal stabilityOrganic chemistryDischarge tube luminescnet screensOrganic light emitting deviceHigh luminance
Provided are an organic light emitting compound represented by Formula 1 below, an organic light emitting device comprising the same, and a method of manufacturing the organic light emitting device:where CY1 and CY2 are each independently a fused C6-C50 aromatic ring, Ar1 is a substituted or unsubstituted C6-C50 arylene group, Ar2, Ar3, Ar4, and Ar5 are each independently a substituted or unsubstituted C6-C50 aryl group, m and n are independently 0-3, and R1 and R2 are substituent groups. An organic light emitting device comprising the organic light emitting compound has low turn-on voltage, high efficiency, high color purity and high luminance.
Owner:SAMSUNG ELECTRONICS CO LTD
Process for the separation/recovery of gases
InactiveUS6179900B1Low costHigh purityNitrogen purification/separationGas treatmentHydrogenHigh pressure
A process for the separation / recovery of gases is described where the desired component to be separated from the gaseous mixture is present in low / very low molar amount and / or low / moderate pressure, the process comprising contacting the gaseous mixture with a separation membrane unit containing high permeability / medium selectivity membranes which separate the feed stream into a permeate stream, enriched in the desired component and at a lower pressure and a retentate stream lean in the desired component and at a higher pressure. The retentate stream is made to convey its pressure to the permeate stream which then is directed to an adsorption PSA unit for further purification of the permeate stream so that the effluent stream from the PSA unit which contains the purified component has a purity of up to 99.90%. The process is useful for separating hydrogen from refinery off gases such as FCC gases and can also be applied to other gases such as helium and any other valuable gases present in gaseous streams in low to very low molar contents and low to moderate pressures.
Owner:GKSS FORSCHUNGSZENTRUM GEESTHACHT GMBH +1
Method of determining the nucleotide sequence of oligonucleotides and DNA molecules
InactiveUS20050032076A1Eliminate needHigh puritySugar derivativesMicrobiological testing/measurementThermopileOligonucleotide primers
The present invention relates to a novel method for analyzing nucleic acid sequences based on real-time detection of DNA poly-merase-catalyzed incorporation of each of the four nucleotide bases, supplied individually and serially in a microfluidic system, to a reaction cell containing a template system comprising a DNA fragment of unknown sequence and an oligonucleotide primer. Incorporation of a nucleotide base into the template system can be detected by any of a variety of methods including but not limited to fluorescence and chemiluminescence detection. Alternatively, microcalorimetic detection of the heat generated by the incorporation of a nucleotide into the extending template system using thermopile, thermistor and refractive index measurements can be used to detect extension reactions.
Owner:LIFE TECH CORP
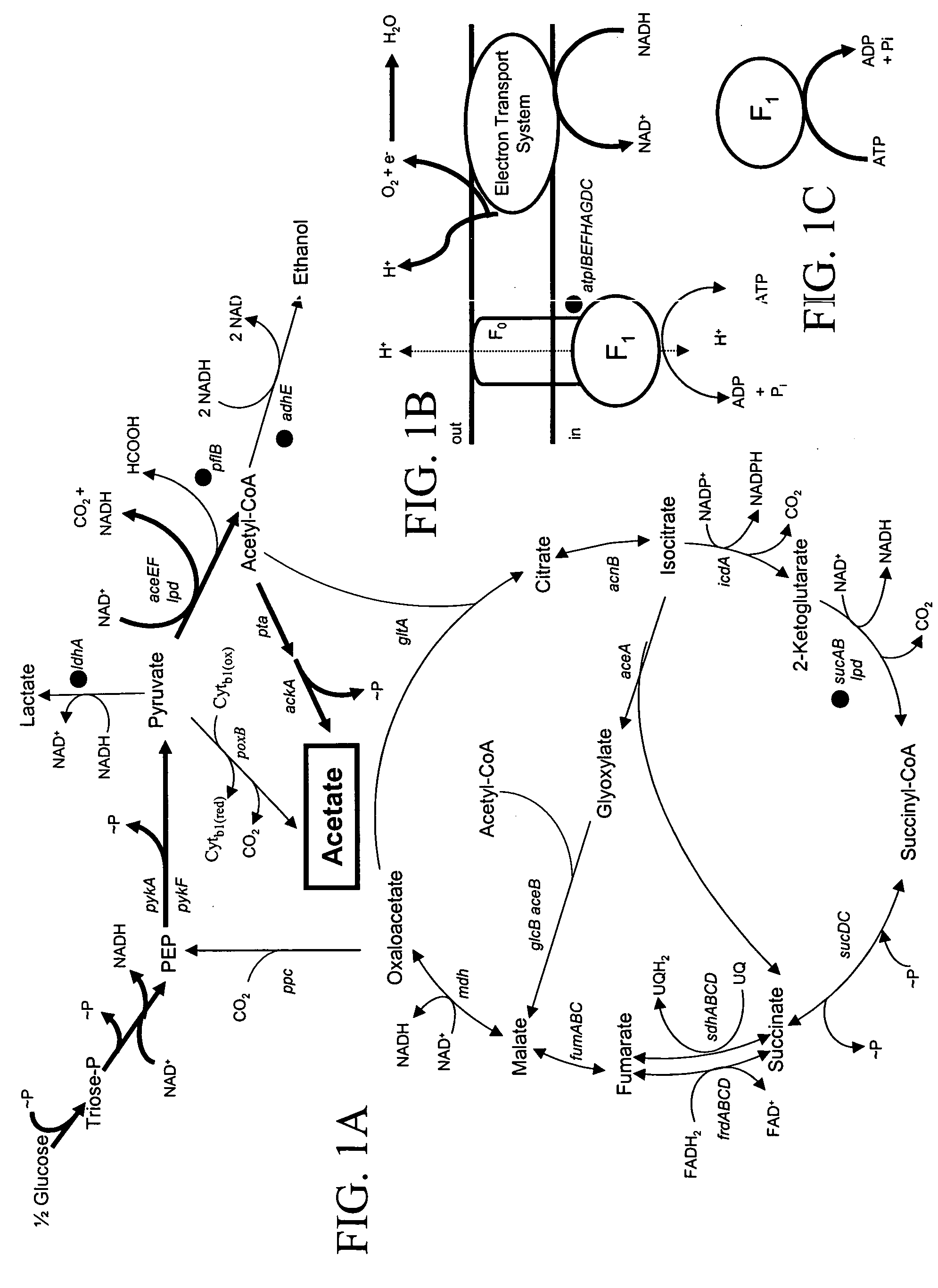
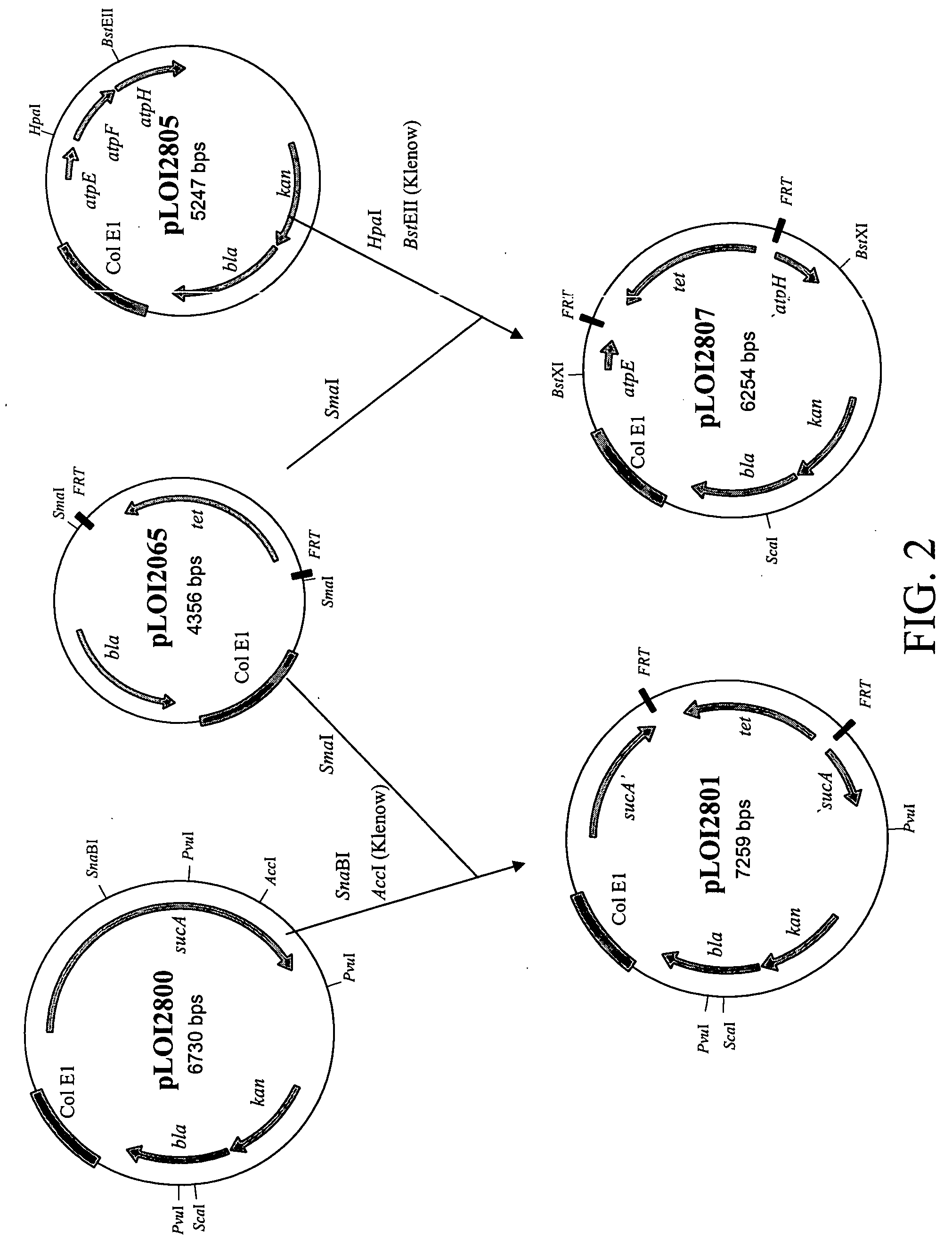
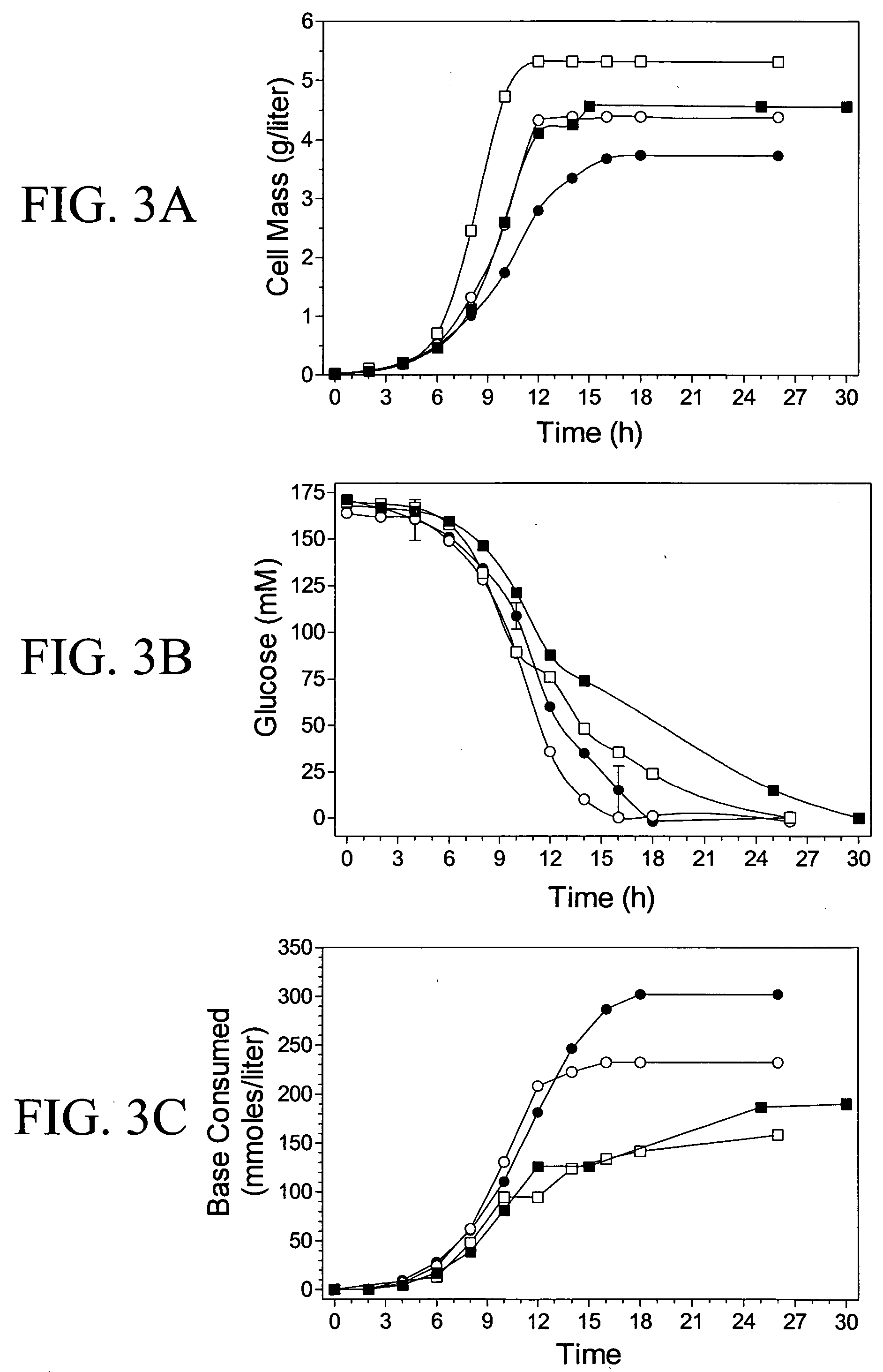
Materials and methods for the efficient production of acetate and other products
InactiveUS20040152159A1Increase productionIncrease acetate productionBacteriaHydrolasesGenes mutationAcetic acid
The subject invention provides materials and methods wherein unique and advantageous combinations of gene mutations are used to direct carbon flow from sugars to a single product. The techniques of the subject invention can be used to obtain products from native pathways as well as from recombinant pathways. In preferred embodiments, the subject invention provides new materials and methods for the efficient production of acetate and pyruvic acid.
Owner:UNIV OF FLORIDA RES FOUNDATION INC
Method for the chemical synthesis of oligonucleotides
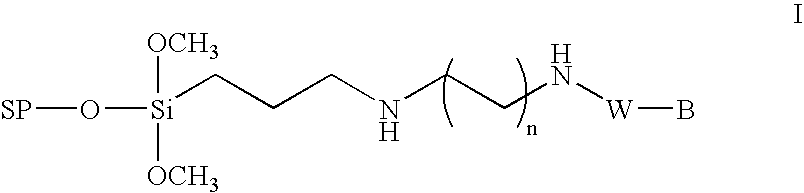
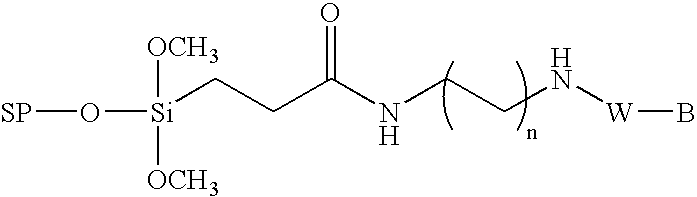
InactiveUS6995259B1High yieldHigh purityGroup 4/14 element organic compoundsSugar derivativesChemical synthesisCombinatorial chemistry
Owner:SIRNA THERAPEUTICS INC