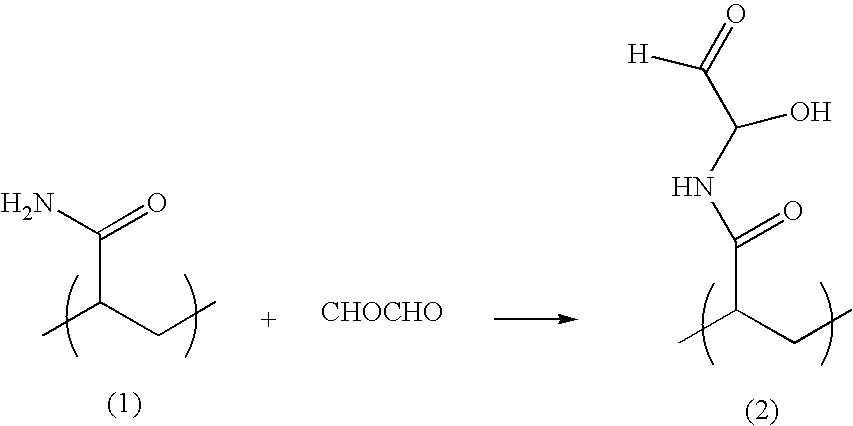
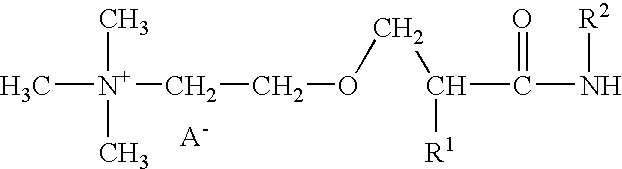
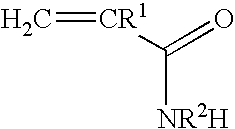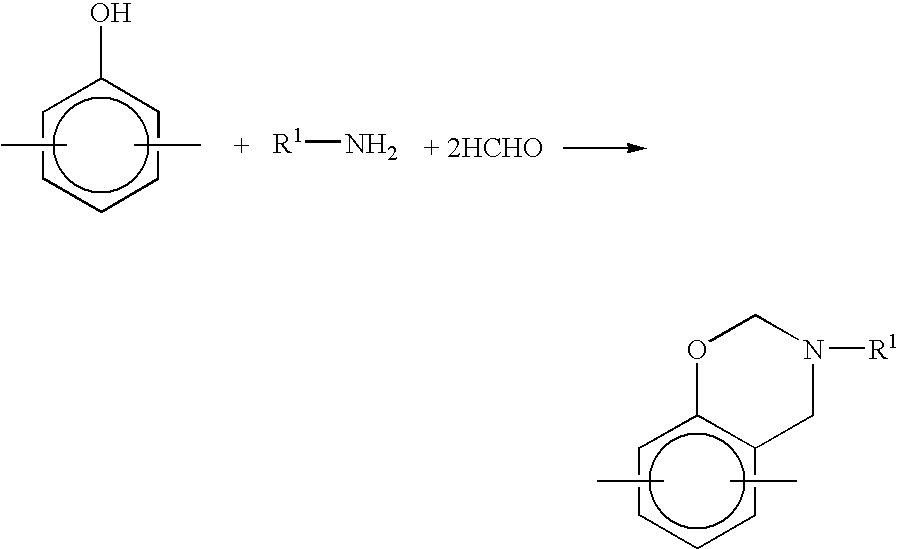Patents
Literature
17619 results about "Aldehyde" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
An aldehyde /ˈældɪhaɪd/ is a compound containing a functional group with the structure −CHO, consisting of a carbonyl center (a carbon double-bonded to oxygen) with the carbon atom also bonded to hydrogen and to an R group, which is any generic alkyl or side chain. The group—without R—is the aldehyde group, also known as the formyl group. Aldehydes are common in organic chemistry, and many fragrances are aldehydes.
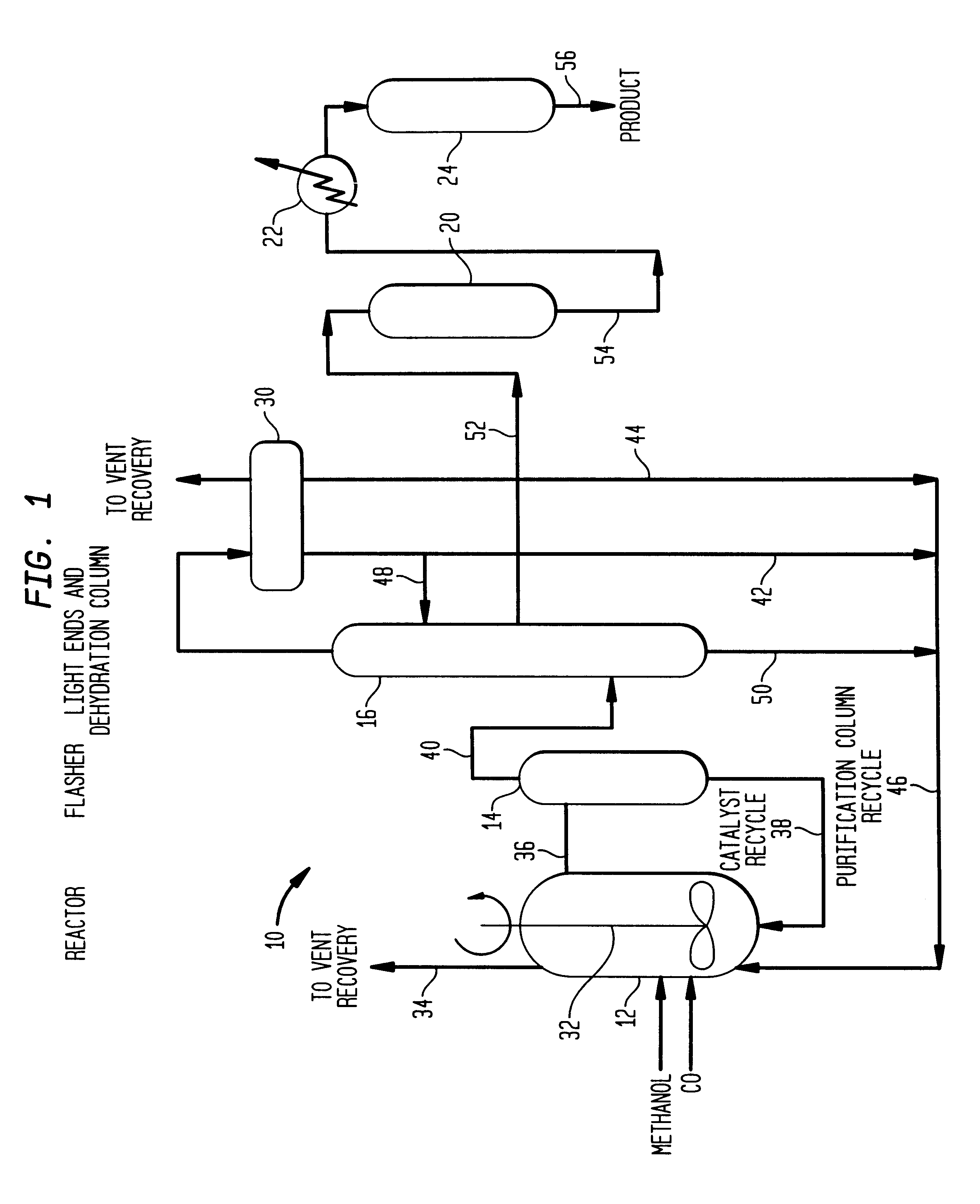
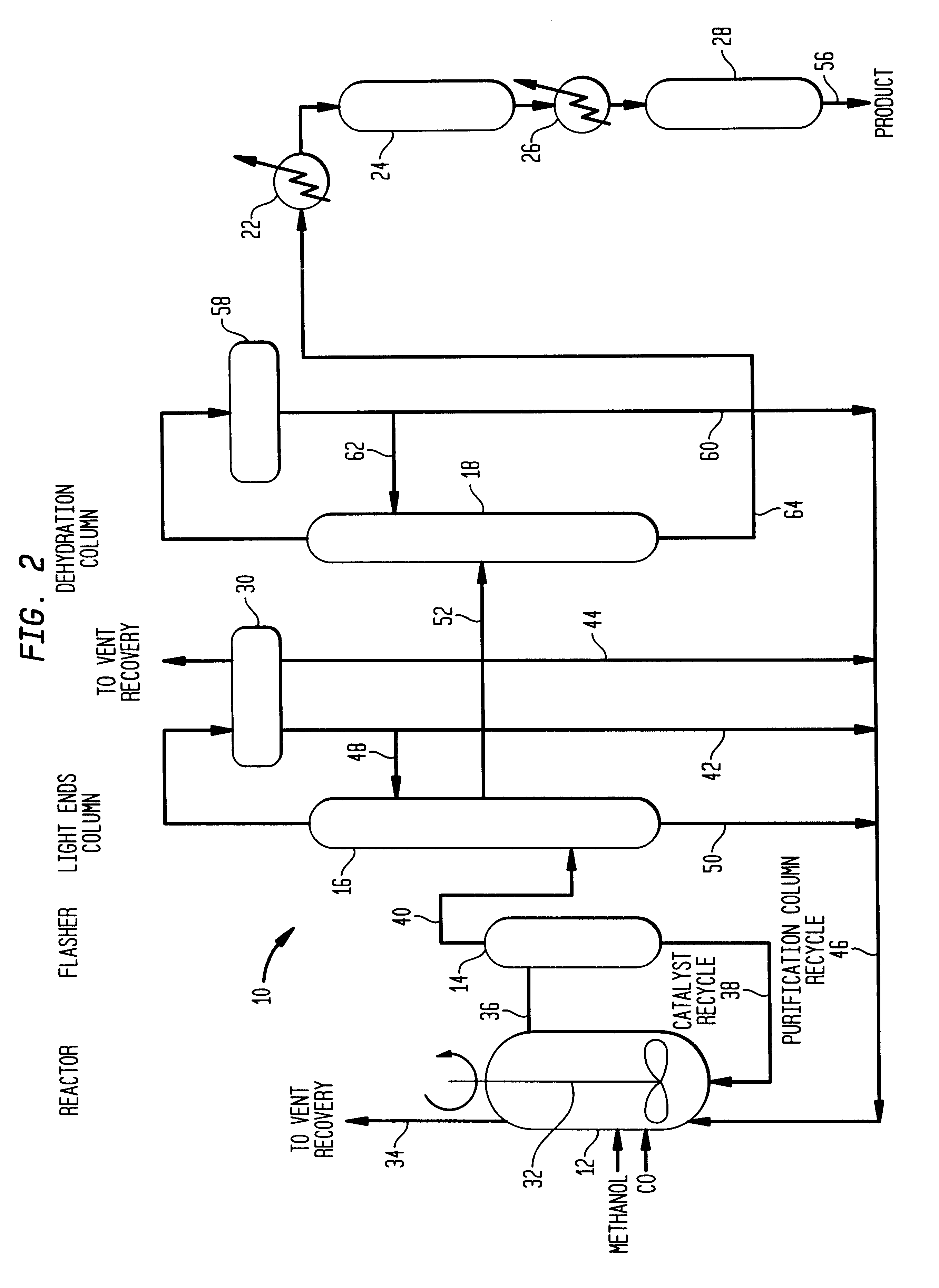
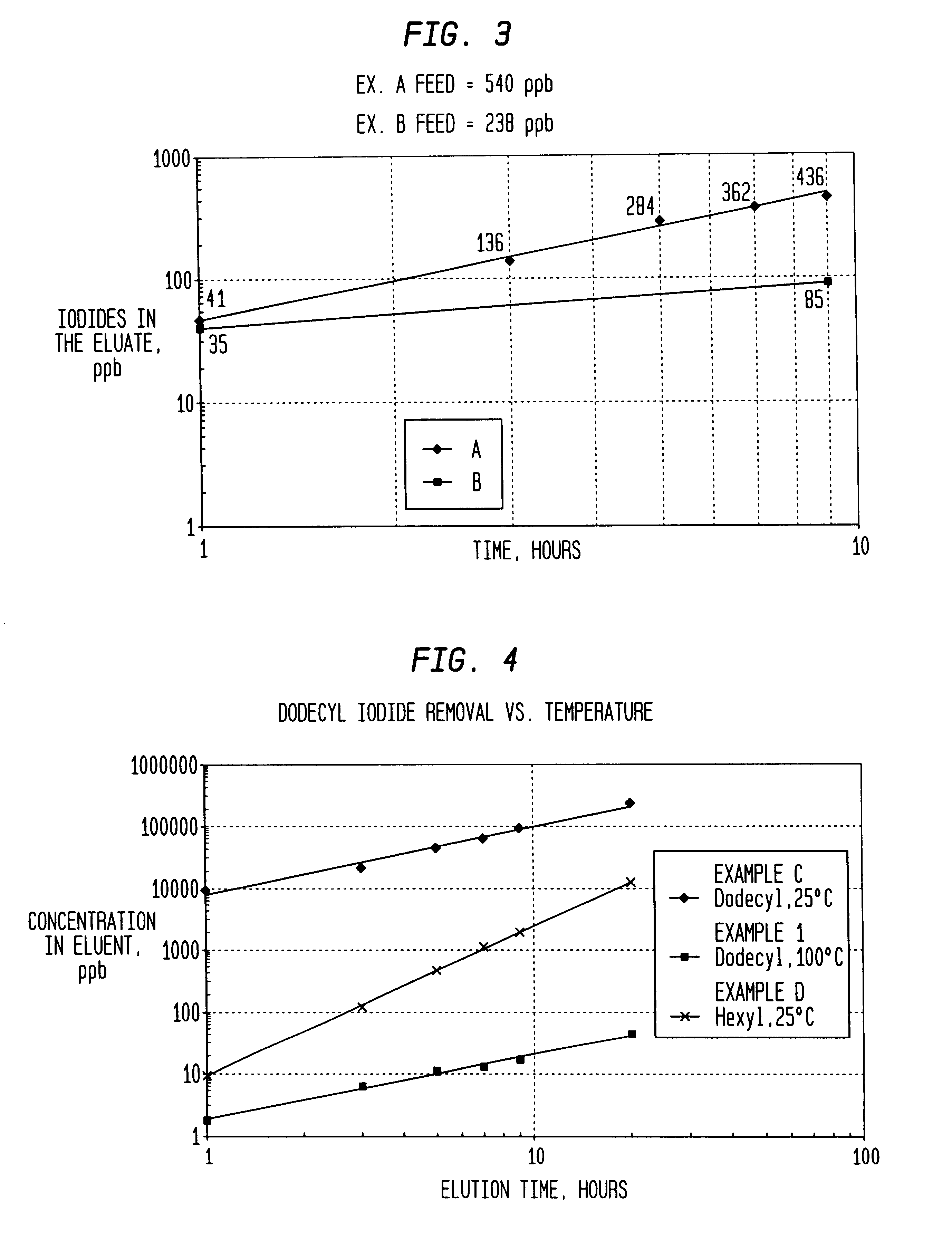
Low energy carbonylation process
InactiveUS6657078B2Weaken energyHigh purityOrganic compound preparationOrganic chemistry methodsPropanoic acidIodide
A low energy process for producing acetic acid by the carbonylation of methanol is disclosed. The process involves a rhodium-catalyzed system operated at less than about 14% water utilizing up to 2 distillation columns. The process is preferably controlled such that the product stream has a low level of propionic acid impurity and the level of aldehyde impurities is minimized by way of aldehyde removal or minimizing aldehyde generation. The level of iodides is controlled by contacting the product, at elevated temperatures, with ion exchange resins. In preferred embodiments, at least one silver or mercury exchanged macroreticular strong acid ion exchange resin is used to purify the product. The high temperature treatment provides the added benefit of controlling the Color Value (Pt-Co units) of the product stream.
Owner:CELANESE INT CORP
Precursor source mixtures
A precursor source mixture useful for CVD or ALD of a film comprising: at least one precursor composed of an element selected from the group consisting of Li, Na, K, Rb, Cs, Fr, Be, Mg, Ti, Zr, Hf, Sc, Y, La, V, Nb, Ta, Cr, Mo, W, Mn, Re, Fe, Ru, Os, Co, Rh, Ir, Ni, Pd, Pt, Cu, Ag, Au, Zn, Cd, Hg, B, Al, Ga, In, Tl, Si, Ge, Sn, Pb, As, P, Sb and Bi, to which is bound at least one ligand selected from the group consisting of hydride, alkyl, alkenyl, cycloalkenyl, aryl, alkyne, carbonyl, amido, imido, hydrazido, phosphido, nitrosyl, nitryl, nitrate, nitrile, halide, azide, alkoxy, siloxy, silyl, and halogenated, sulfonated or silyated derivatives thereof, which is dissolved, emulsified or suspended in an inert liquid selected from the group consisting of aliphatic hydrocarbons, aromatic hydrocarbons, alcohols, ethers, aldehydes, ketones, acids, phenols, esters, amines, alkylnitrile, halogenated hydrocarbons, silyated hydrocarbons, thioethers, amines, cyanates, isocyanates, thiocyanates, silicone oils, nitroalkyl, alkylnitrate, and mixtures thereof. The precursor source mixture may be a solution, emulsion or suspension and may consist of a mixture of solid, liquid and gas phases which are distributed throughout the mixture.
Owner:GLOBALFOUNDRIES INC
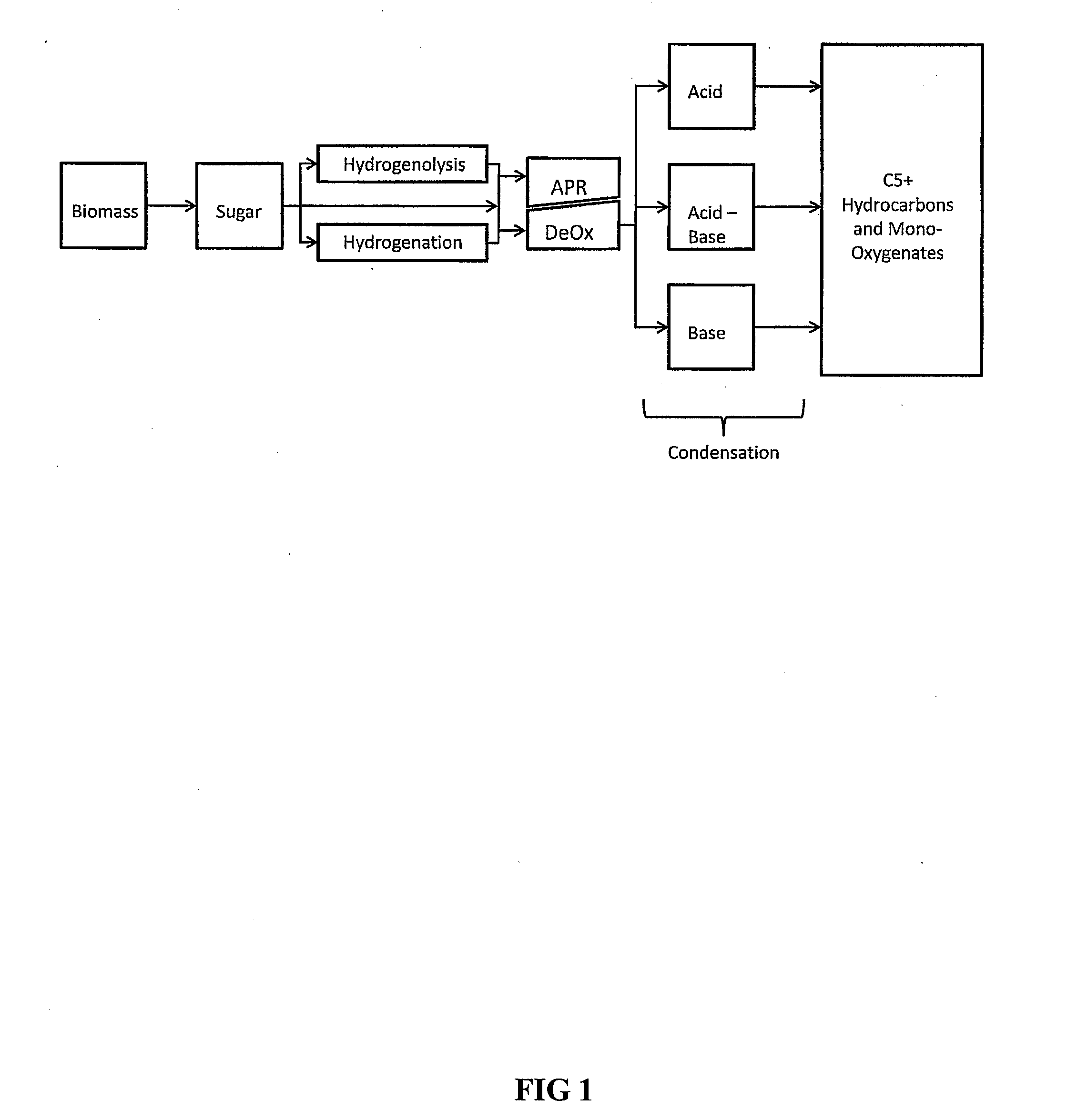
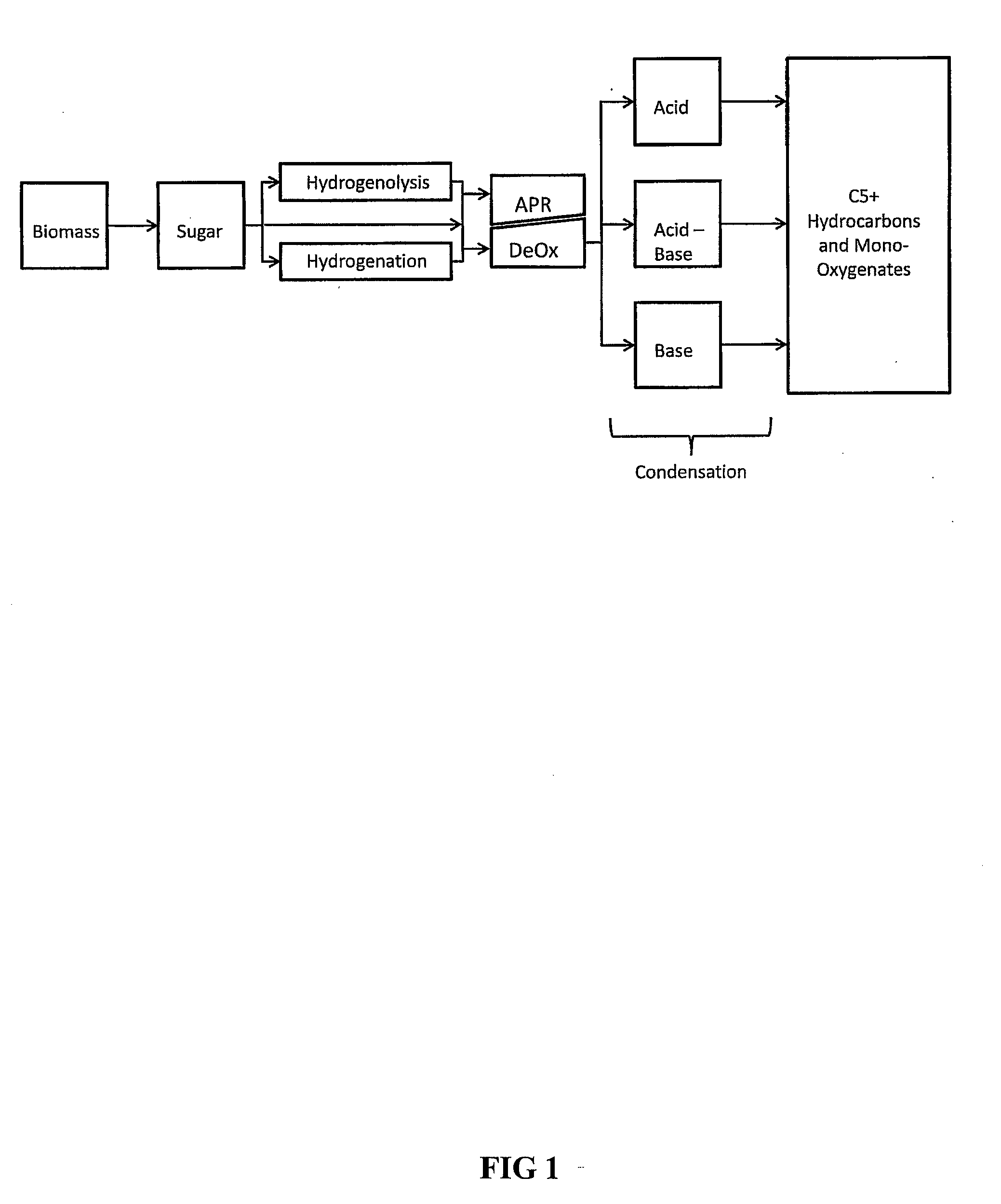
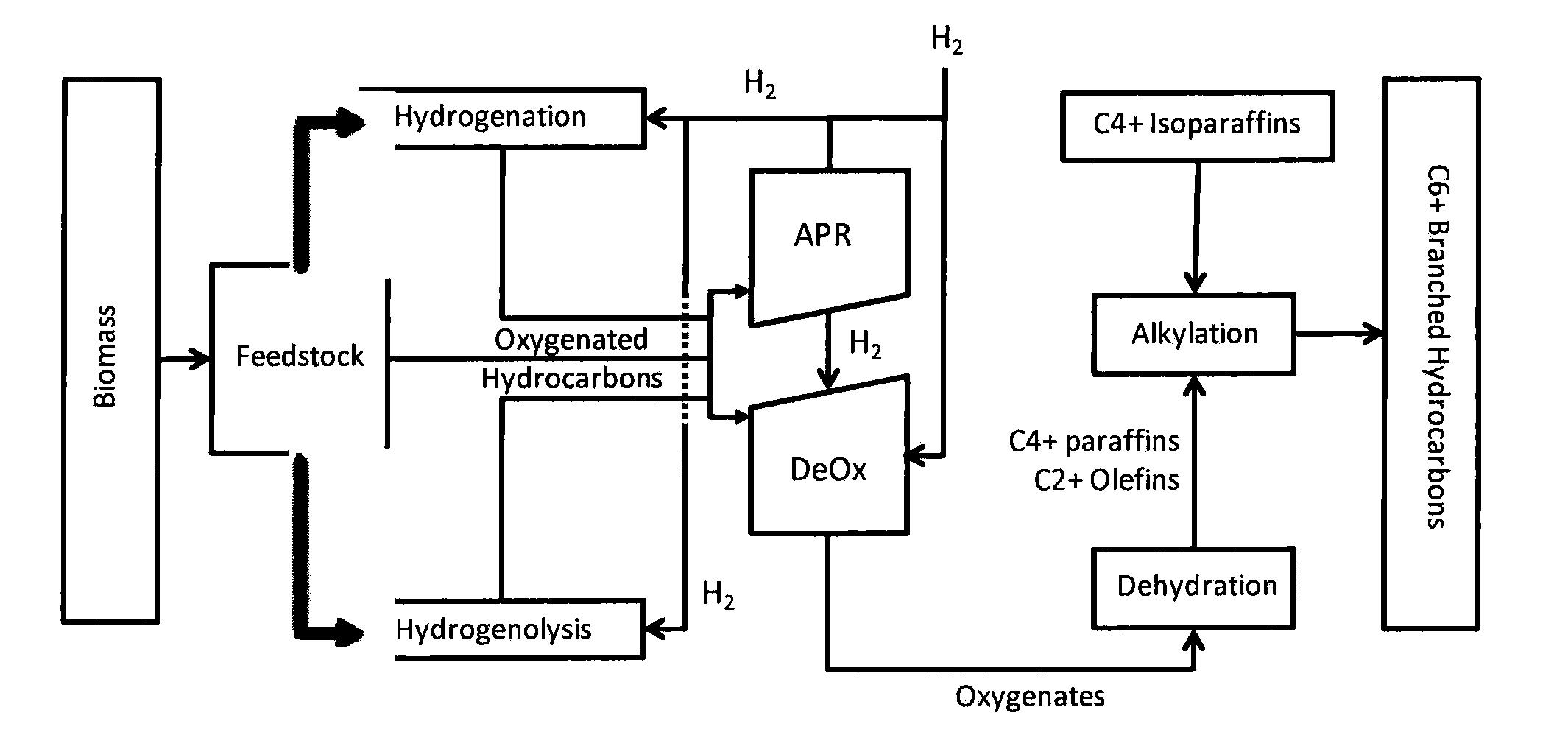
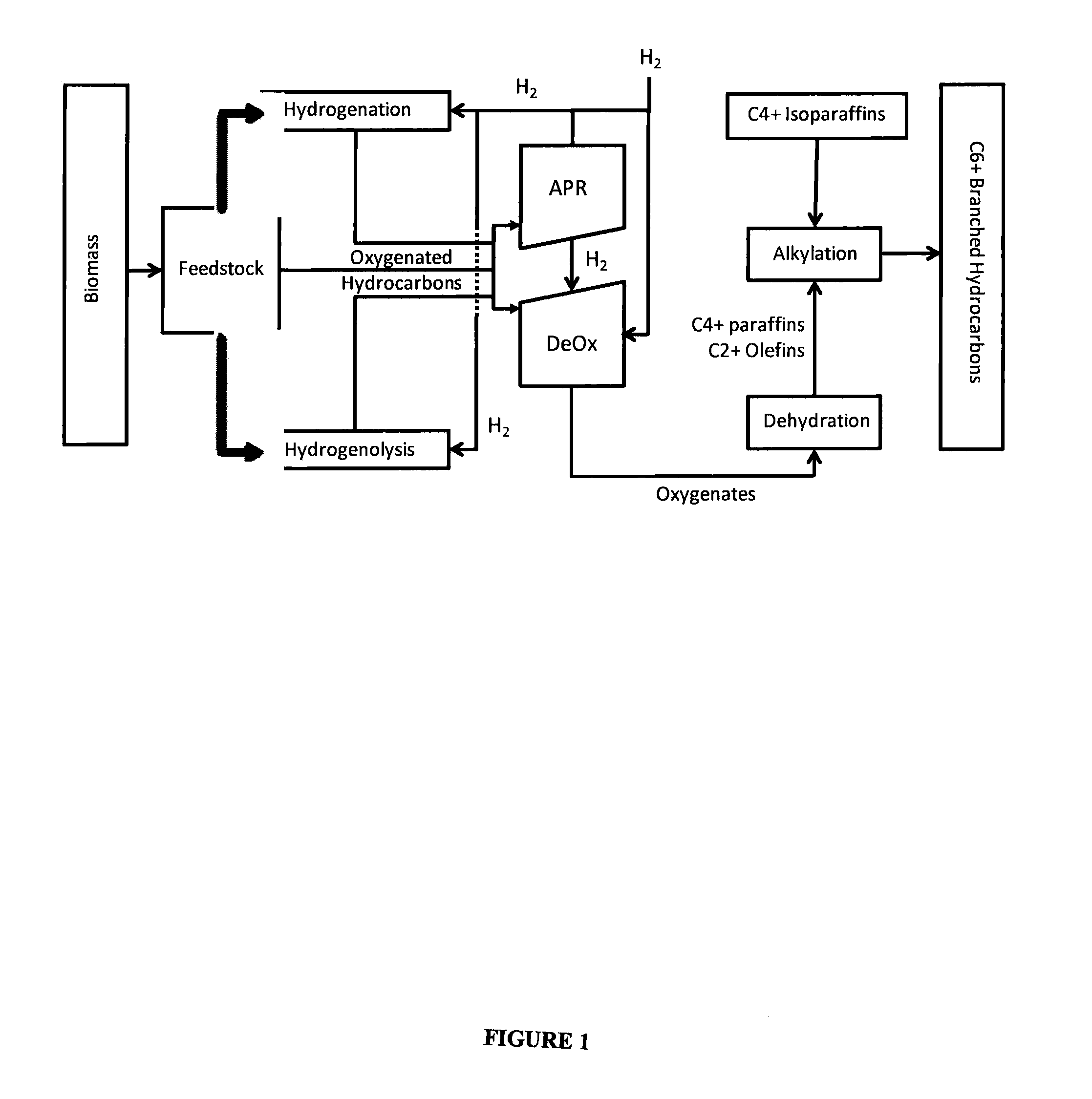
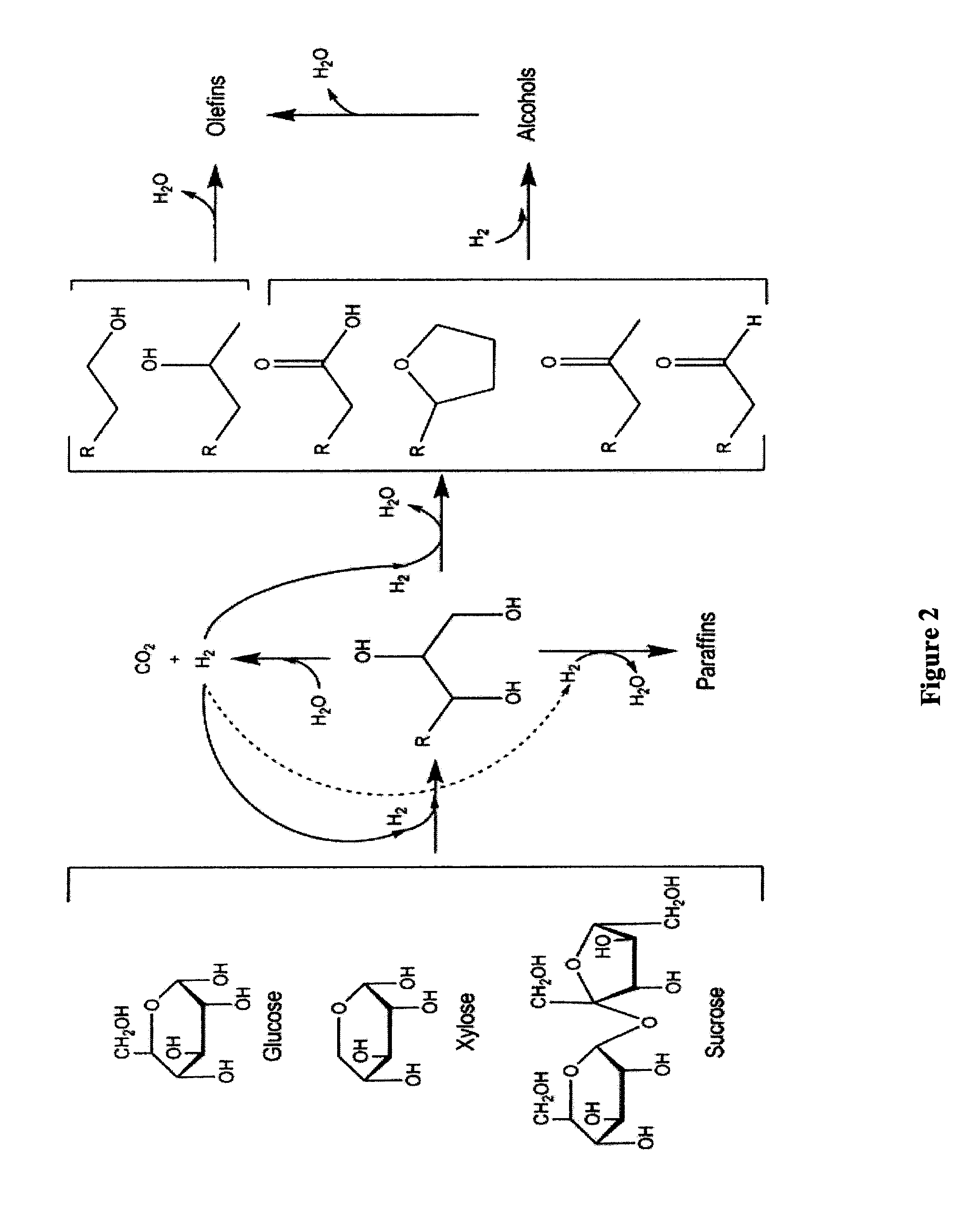
Synthesis of liquid fuels and chemicals from oxygenated hydrocarbons
ActiveUS20080216391A1Organic compound preparationHydrocarbon from oxygen organic compoundsFuranLiquid fuel
Processes and reactor systems are provided for the conversion of oxygenated hydrocarbons to hydrocarbons, ketones and alcohols useful as liquid fuels, such as gasoline, jet fuel or diesel fuel, and industrial chemicals. The process involves the conversion of mono-oxygenated hydrocarbons, such as alcohols, ketones, aldehydes, furans, carboxylic acids, diols, triols, and / or other polyols, to C4+ hydrocarbons, alcohols and / or ketones, by condensation. The oxygenated hydrocarbons may originate from any source, but are preferably derived from biomass.
Owner:VIRENT
Synthesis of liquid fuels and chemicals from oxygenated hydrocarbons
ActiveUS20080300435A1Oxygen-containing compound preparationLiquid hydrocarbon mixture productionFuranLiquid fuel
Processes and reactor systems are provided for the conversion of oxygenated hydrocarbons to hydrocarbons, ketones and alcohols useful as liquid fuels, such as gasoline, jet fuel or diesel fuel, and industrial chemicals. The process involves the conversion of mono-oxygenated hydrocarbons, such as alcohols, ketones, aldehydes, furans, carboxylic acids, diols, triols, and / or other polyols, to C4+ hydrocarbons, alcohols and / or ketones, by condensation. The oxygenated hydrocarbons may originate from any source, but are preferably derived from biomass.
Owner:VIRENT
Pro-fragrances
InactiveUS6861402B1Enhanced perfume longevityCosmetic preparationsOrganic chemistryFlavorAdditive ingredient
The present invention relates to fragrance delivery systems which comprise: A) from about 0.01% by weight of a pro-fragrance component which comprises pro-fragrances or pro-accords selected from at least two of the following: i) aldehyde and ketone releasing pro-fragrances, preferably an oxazolidine pro-fragrance; ii) β-amino pro-fragrances; and iii) orthoester pro-accords; and B) the balance carries and others adjunct ingredients.
Owner:THE PROCTER & GAMBLE COMPANY
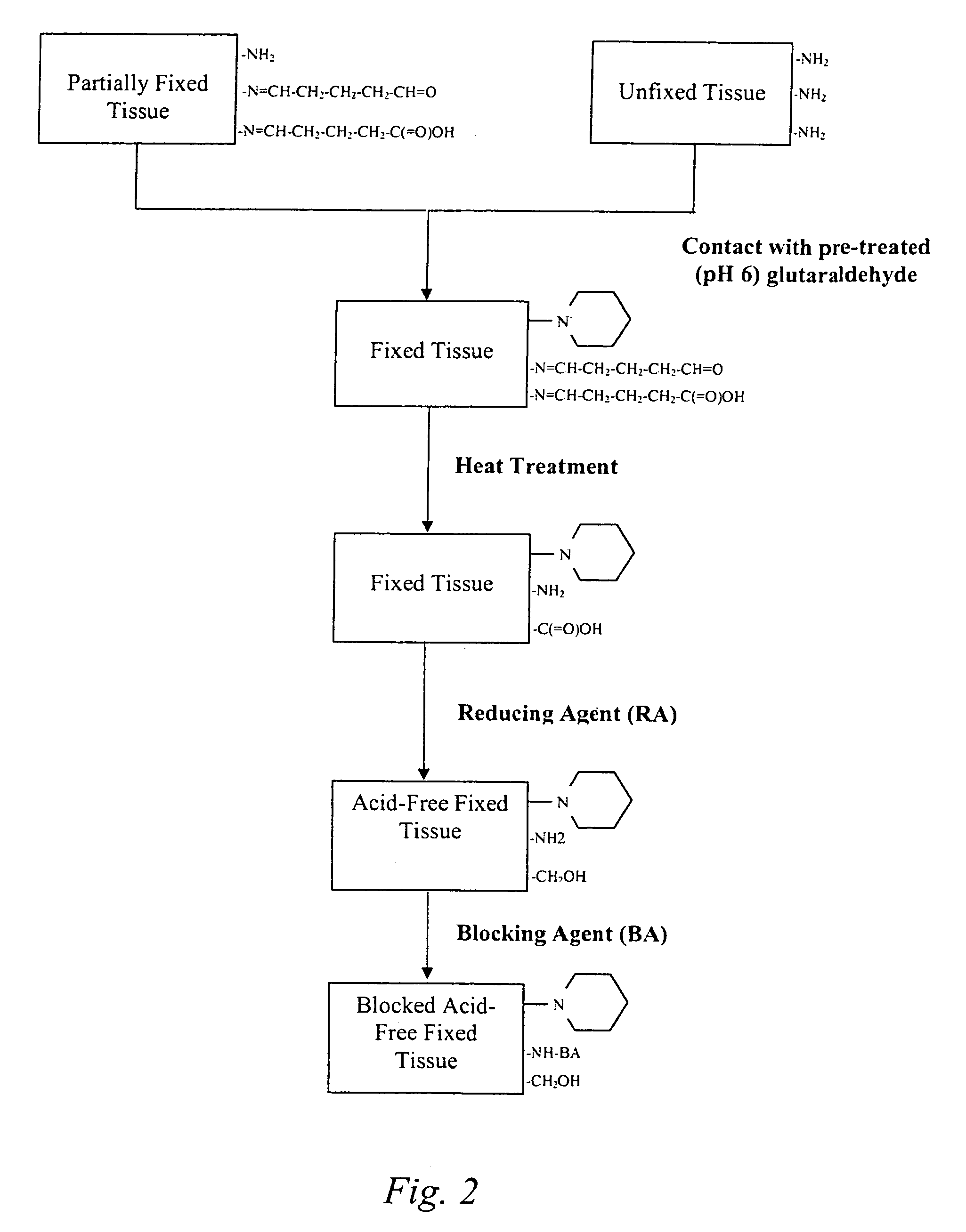
Treatment of bioprosthetic tissues to mitigate post implantation calcification
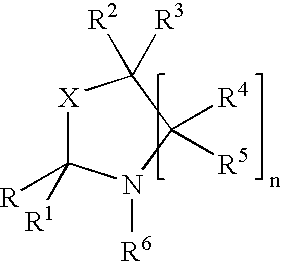
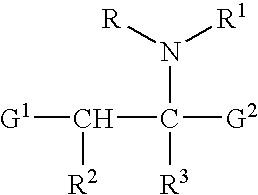
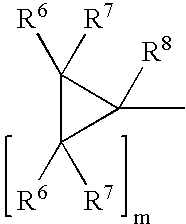
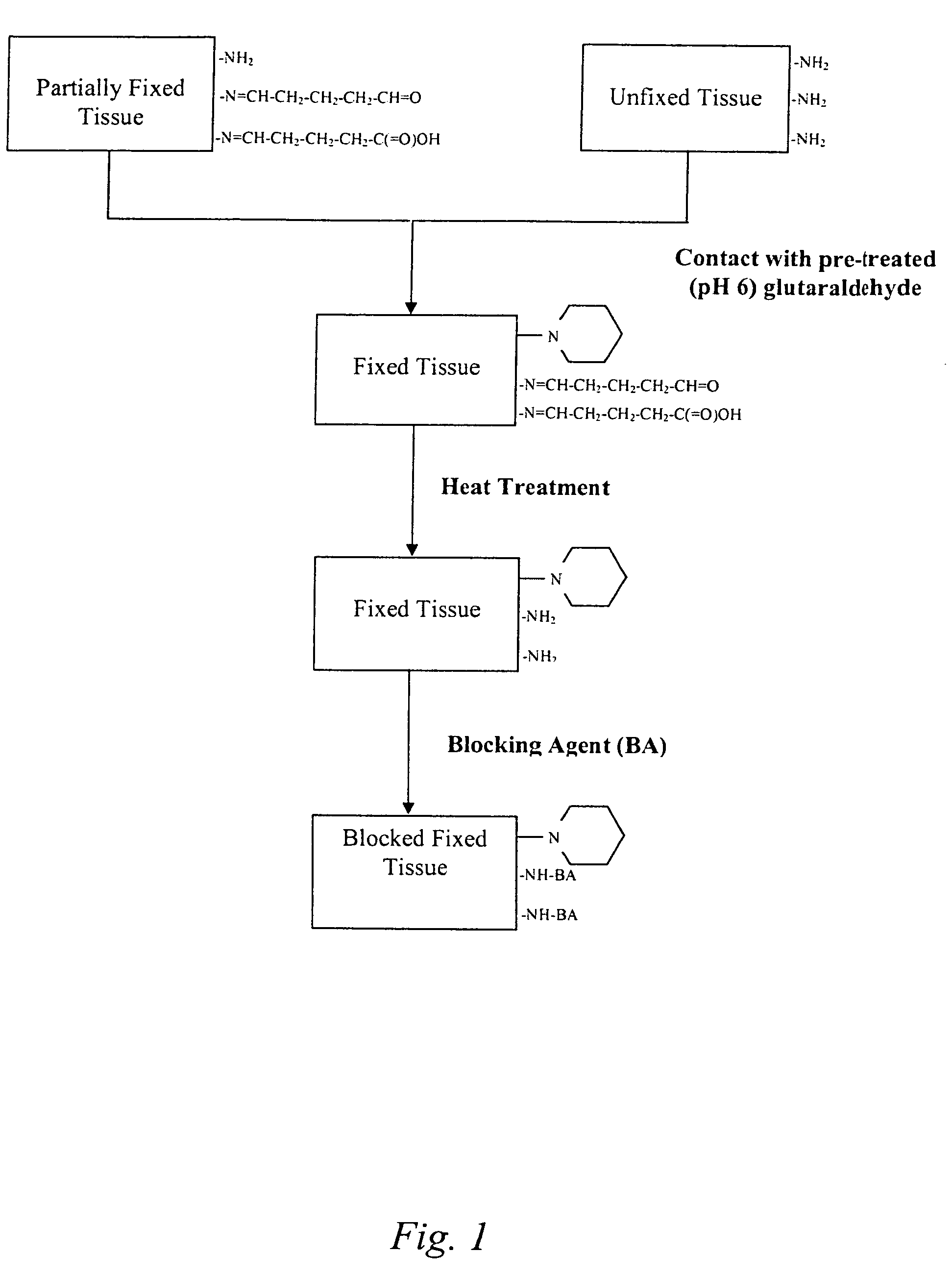
The present invention provides methods for treating tissue to inhibit post-implant calcification of a biological tissue. In one method of this invention, a tissue is immersed in or otherwise contacted with a pretreated glutaraldehyde solution, i.e., a heat-treated or pH-adjusted glutaraldehyde solution. The tissue may be partially fixed with glutaraldehyde prior to, after, or concurrently with the step of contacting the tissue with the pretreated gluteraldehyde. Contact with the pretreated gluteraldehyde produces free amine groups on the tissue, which are subsequently blocked by contacting the crosslinked tissue with a blocking agent. In another embodiment, a tissue is contacted with either a non-pretreated glutaraldehyde or a pH-adjusted glutaraldehyde solution for a period of time sufficient to crosslink the tissue. The crosslinked tissue is then treated with a reducing agent that reduces aldehyde and carboxylic acid groups on the fixed tissue.
Owner:EDWARDS LIFESCIENCES CORP
Synthesis of liquid fuels from biomass
ActiveUS20100076233A1Hydrocarbon by metathesis reactionLiquid hydrocarbon mixture productionFuranAlkane
Processes and reactor systems are provided for the conversion of oxygenated hydrocarbons to paraffins useful as liquid fuels. The process involves the conversion of water soluble oxygenated hydrocarbons to oxygenates, such as alcohols, furans, ketones, aldehydes, carboxylic acids, diols, triols, and / or other polyols, followed by the subsequent conversion of the oxygenates to paraffins by dehydration and alkylation. The oxygenated hydrocarbons may originate from any source, but are preferably derived from biomass.
Owner:VIRENT
Fragrance pro-accords and aldehyde and ketone fragrance libraries
InactiveUS7018978B2Extend your lifeEasy to prepareCosmetic preparationsOrganic chemistryFlavorOxazolidine E
The present invention relates to novel heterocyclic pro-fragrances, preferably oxazolidines, tertahydro-1,3-oxazines, thiazolidines, or tetrahydro-1,3-thiazines, more preferably oxazolidines, or tertahydro-1,3-oxazines, most preferably oxazolidines, which are capable of sustained release of fragrance raw material ketones and aldehydes and to fragrance delivery systems which comprise said pro-fragrances.
Owner:THE PROCTER & GAMBLE COMPANY
Pharmaceutical co-crystal compositions
A pharmaceutical composition comprising a co-crystal of an API and a co-crystal former; wherein the API has at least one functional group selected from ether, thioether, alcohol, thiol, aldehyde, ketone, thioketone, nitrate ester, phosphate ester, thiophosphate ester, ester, thioester, sulfate ester, carboxylic acid, phosphonic acid, phosphinic acid, sulfonic acid, amide, primary amine, secondary amine, ammonia, tertiary amine, sp2 amine, thiocyanate, cyanamide, oxime, nitrile diazo, organohalide, nitro, s-heterocyclic ring, thiophene, n-heterocyclic ring, pyrrole, o-heterocyclic ring, furan, epoxide, peroxide, hydroxamic acid, imidazole, pyridine and the co-crystal former has at least one functional group selected from amine, amide, pyridine, imidazole, indole, pyrrolidine, carbonyl, carboxyl, hydroxyl, phenol, sulfone, sulfonyl, mercapto and methyl thio, such that the API and co-crystal former are capable of co-crystallizing from a solution phase under crystallization conditions.
Owner:JOHNSON & JOHNSON CONSUMER COPANIES +2
Synthesis of liqiud fuels and chemicals from oxygenated hydrocarbons
ActiveUS20080300434A1Oxygen-containing compound preparationHydrocarbon purification/separationFuranCarboxylic acid
Processes and reactor systems are provided for the conversion of oxygenated hydrocarbons to hydrocarbons, ketones and alcohols useful as liquid fuels, such as gasoline, jet fuel or diesel fuel, and industrial chemicals. The process involves the conversion of mono-oxygenated hydrocarbons, such as alcohols, ketones, aldehydes, furans, carboxylic acids, diols, triols, and / or other polyols, to C4+ hydrocarbons, alcohols and / or ketones, by condensation. The oxygenated hydrocarbons may originate from any source, but are preferably derived from biomass.
Owner:VIRENT
Perfumed liquid laundry detergent compositions with functionalized silicone fabric care agents
InactiveUS20060003913A1Organic detergent compounding agentsNon-surface-active detergent compositionsLiquid laundry detergentNitrogen
The invention is directed to aqueous liquid laundry detergent compositions for cleaning and imparting fabric care benefits to fabrics laundered therewith and to methods for preparing such compositions. Such compositions comprise (A) at least one textile-cleaning surfactant; (B) droplets of miscible silicones comprising both a polarly-functionalized, preferably nitrogen-containing amino or ammonium functionalized, polysiloxane component and a nitrogen-free non-functionalized or non-polarly-functionalized polysiloxane component; and (C) a perfume component comprising fragrant aldehydes and / or ketones or a pro-perfume capable of providing such aldheyde and / or ketone perfume materials in situ. Incorporation of a polarly-functionalized polysiloxane fabric care agent into liquid laundry detergent compositions by miscibly combining it with a non-functionalized or non-polarly functionalized polysiloxane minimizes the undesirable interaction such polarly-functionalized silicone material might otherwise have with aldehyde and / or ketone perfume compounds.
Owner:THE PROCTER & GAMBLE COMPANY
Polypeptide compositions with improved stability
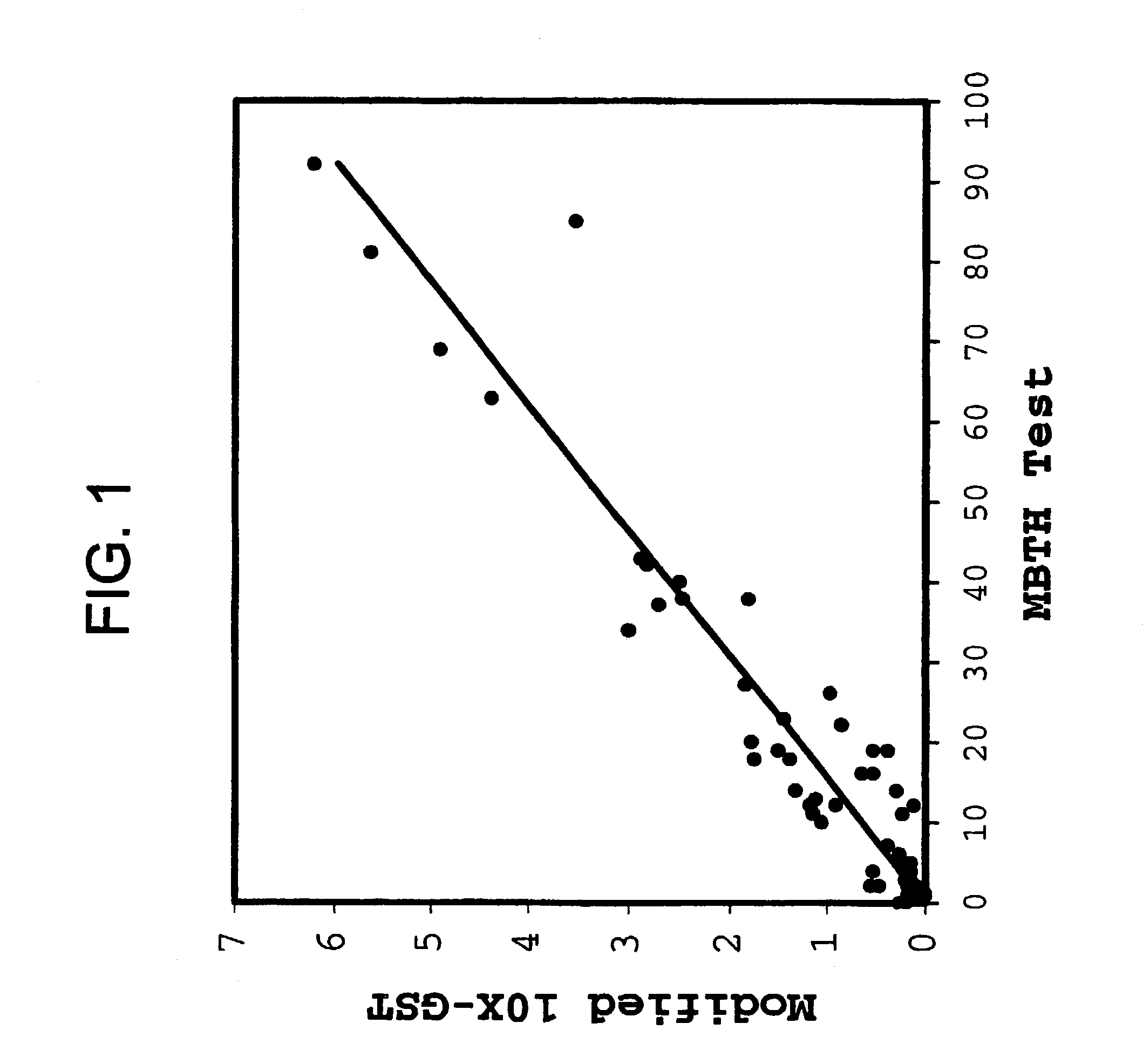
InactiveUS7022674B2Good chemical stabilityLower Level RequirementsPeptide/protein ingredientsMetabolism disorderLinear correlationGlycerol
The present invention provides means to improve the chemical stability of aqueous, parenteral pharmaceutical compositions comprising a polypeptide and glycerin. Reactive aldehydes are identified in commercial glycerins, and means for reducing such are provided. Convenient means are provided to assay for reactive aldehydes in glycerin, and a strong linear correlation between the level of reactive aldehydes in glycerin and chemical stability of compositions comprising a polypeptide and glycerin is demonstrated. The invention includes aqueous compositions comprising a polypeptide and glycerin having improved chemical stability compared to compositions previously known.
Owner:ELI LILLY & CO
Tissue adhesive sealant
Owner:COVALENT MEDICAL
Luminescent element material and luminescent element comprising the same
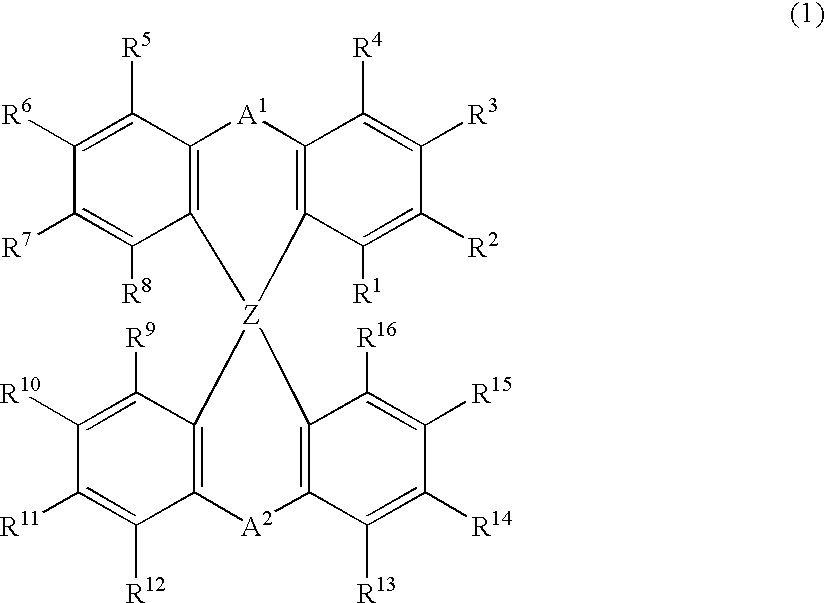
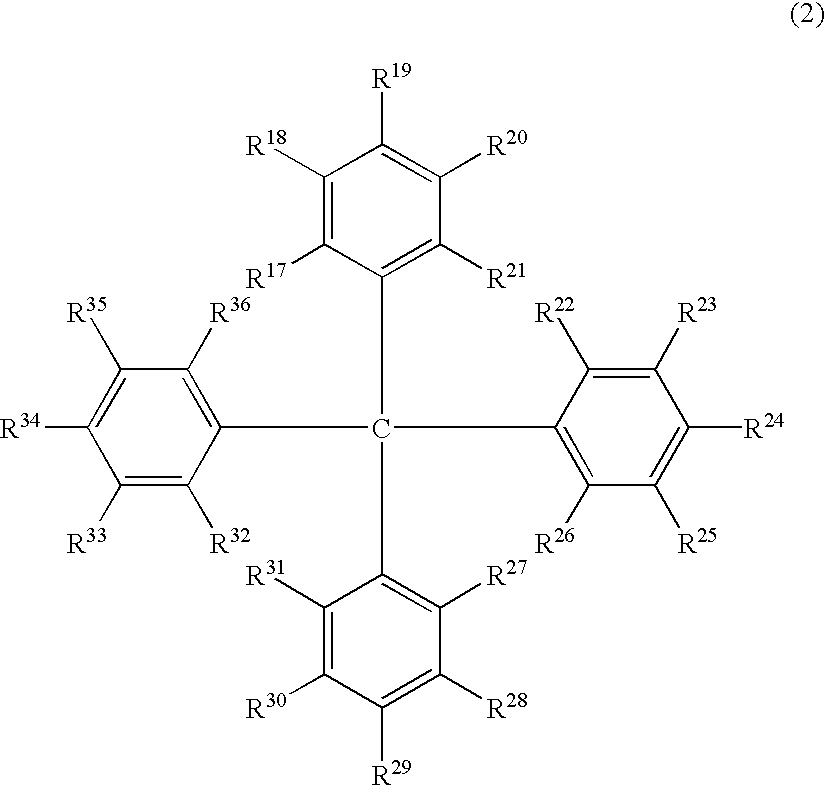
The light emitting device of the present invention relates to a light emitting device which is characterized in that it is a device with an emissive substance present between an anode and cathode, and which emits light by means of electrical energy, and said device has a least one type of compound denoted by (a) to (d) below. (a) A compound having a plurality of 1,7-phenanthroline skeletal structures (b) A benzoquinoline derivative (c) A spiro compound represented by general formula (1) A1 and A2 are each selected from single bonds, substituted or unsubstituted alkyl chains, ether chains, thioether chains, ketone chains and substituted or unsubstituted amino chains. However, A1<> A2. Z represents carbon or silicon. R1 to R16 are each selected from hydrogen, alkyl group, cycloalkyl group, aralkyl group, alkenyl group, cycloalkenyl group, alkynyl group, hydroxyl group, mercapto group, alkoxy group, alkylthio group, aryl ether group, aryl thioether group, aryl group, heterocyclic group, halogen, haloalkane, haloalkene, haloalkyne, cyano group, aldehyde group, carbonyl group, carboxyl group, ester group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group and a cyclic structure formed with an adjacent substituent. (d) A tetraphenylmethane derivative represented by general formula (2) R17 to R36 are each selected from hydrogen, alkyl group, cycloalkyl group, aralkyl group, alkenyl group, cycloalkenyl group, alkynyl group, hydroxyl group, mercapto group, alkoxy group, alkylthio group, aryl ether group, aryl thioether group, aryl group, heterocyclic group, halogen, haloalkane, haloalkene, haloalkyne, cyano group, aldehyde group, carbonyl group, carboxyl group, ester group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group and a cyclic structure formed with an adjacent substituent. However, at least one of R17 to R36 is selected from substituents represented by general formula (3). -X-Ar (3) X is a single bond or is selected from the following, and Ar denotes a condensed aromatic ring or heteroaromatic ring. In the case where X is phosphorus oxide, then Ar represents an aromatic hydrocarbon or heteroaromatic ring. n is an natural number.
Owner:TORAY IND INC
Functionalized derivatives of hyaluronic acid, formation of hydrogels in situ using same, and methods for making and using same
InactiveUS7196180B2Good physical propertiesSimple structureOrganic active ingredientsSugar derivativesCross-linkIn situ polymerization
Methods for chemical modification of hyaluronic acid, formation of amine or aldehyde functionalized hyaluronic acid, and the cross-linking thereof to form hydrogels are provided. Functionalized hyaluronic acid hydrogels of this invention can be polymerized in situ, are biodegradable, and can serve as a tissue adhesive, a tissue separator, a drug delivery system, a matrix for cell cultures, and a temporary scaffold for tissue regeneration.
Owner:ORTHOGENE
Process for producing integrated circuits including reduction using gaseous organic compounds
InactiveUS6921712B2Eliminate the problemNovel methodSolid-state devicesSemiconductor/solid-state device manufacturingAlcoholCarboxylic acid
This invention concerns a process for producing integrated circuits containing at least one layer of elemental metal which during the processing of the integrated circuit is at least partly in the form of metal oxide, and the use of an organic compound containing certain functional groups for the reduction of a metal oxide layer formed during the production of an integrated circuit. According to the present process the metal oxide layer is at least partly reduced to elemental metal with a reducing agent selected from organic compounds containing one or more of the following functional groups: alcohol (—OH), aldehyde (—CHO), and carboxylic acid (—COOH).
Owner:ASM INTERNATIONAL
Bioabsorbable and biocompatible polyurethanes and polyamides for medical devices
ActiveUS20060188547A1Low toxicitySuture equipmentsOrganic active ingredientsAbsorbable polymersPolyester
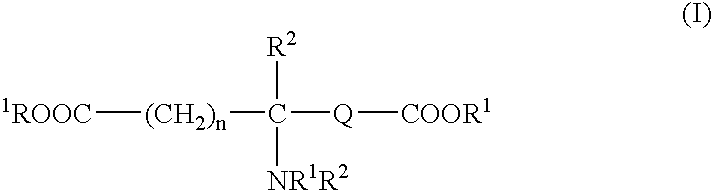
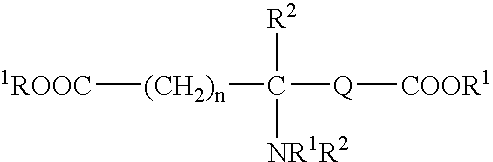
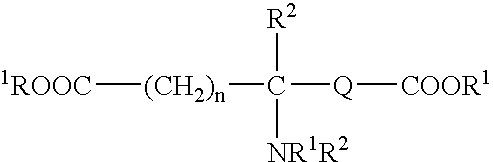
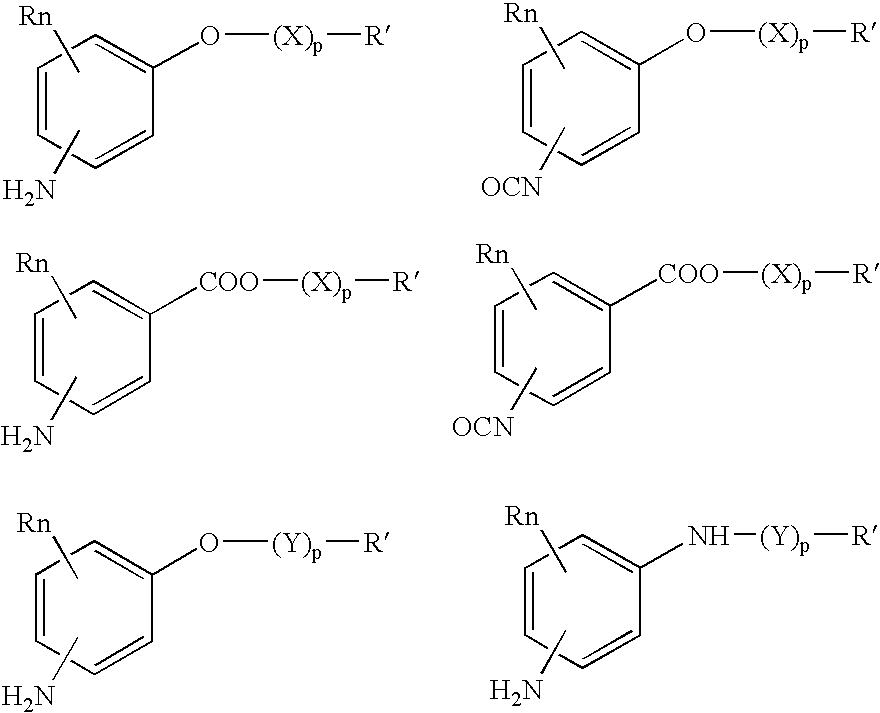
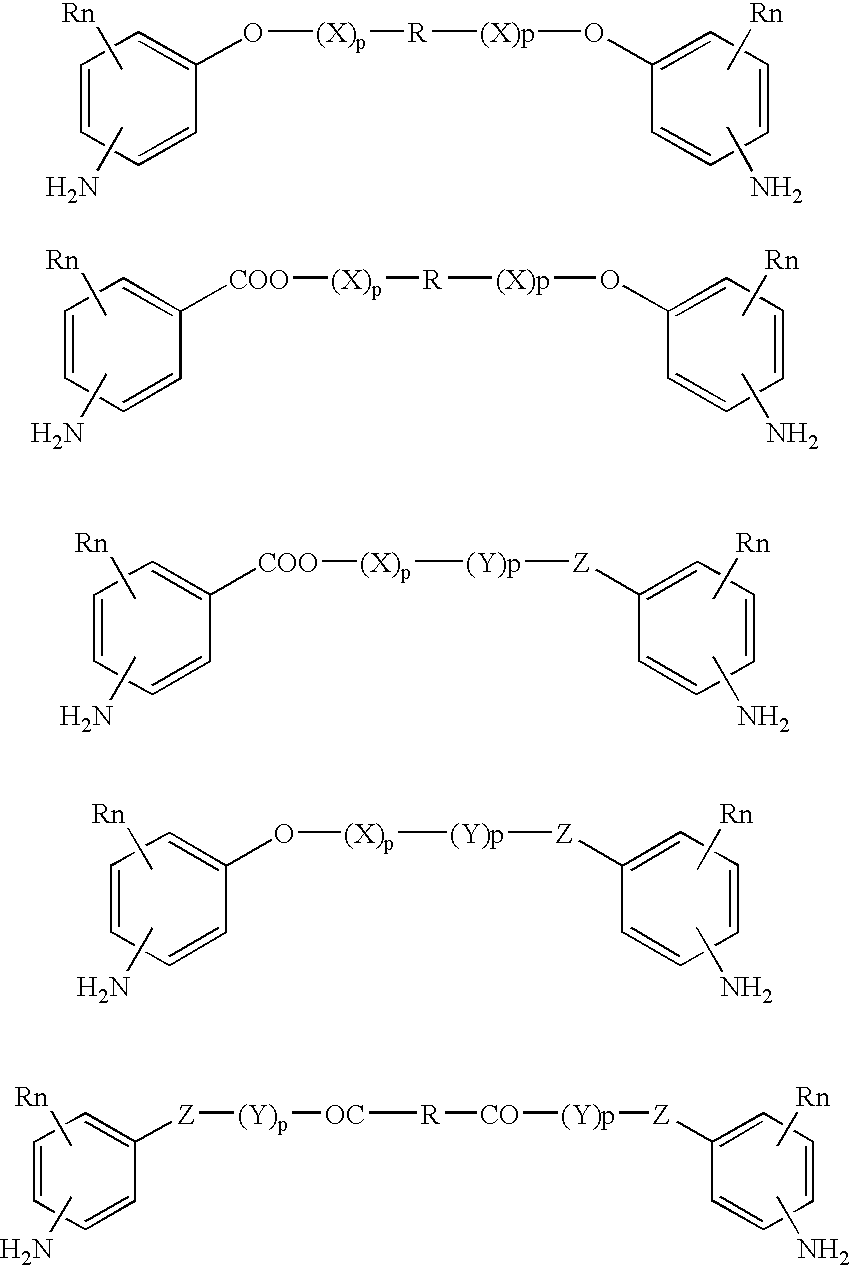
Absorbable polyurethanes, polyamides and polyester urethanes prepared from at least one compound selected from: or the diamines and diisocyanates thereof, wherein each X represents a member independently selected from —CH2COO— (glycolic acid moiety), —CH(CH3)COO— (lactic acid moiety), —CH2CH2OCH2COO— (dioxanone), —CH2CH2CH2CH2CH2COO— (caprolactone moiety), —(CH2)yCOO— where y is one of the numbers 2, 3, 4 or 6-24 inclusive, and —(CH2CH2O)z′CH2COO— where z′ is an integer between 2 and 24, inclusive; each Y represents a member independently selected from —COCH2O— (glycolic ester moiety), —COCH(CH3)O— (lactic ester moiety), —COCH2OCH2CH2O— (dioxanone ester), —COCH2CH2CH2CH2CH2O— (caprolactone ester), —CO(CH2)mO— where m is an integer between 2, 3, 4 or 6-24 inclusive, —COCH2O(CH2CH2O)n— where n is an integer between 2 and 24, inclusive; R′ is hydrogen, benzyl or an alkyl group, the alkyl group being either straight-chained or branched; p is an integer between 1 and 4, inclusive; and Rn represents one or more members selected from H, alkoxy, benzyloxy, aldehyde, halogen, carboxylic acid and —NO2, which is attached directly to an aromatic ring or attached through an aliphatic chain. Absorbable polymers prepared from these compounds are useful for drug delivery, tissue engineering, tissue adhesives, adhesion prevention and other implantable medical devices.
Owner:BEZWADA BIOMEDICAL LLC
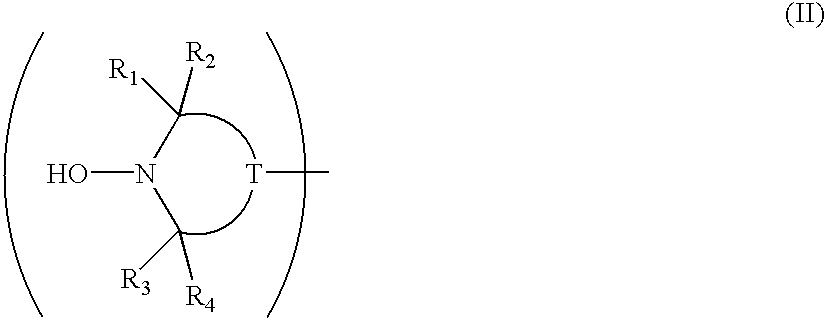
Polyester and polyamide compositions of low residual aldehyde content
A mixture of a polyester, such as poly(ethylene terephthalate) PET, or a polyamide, and a suitable stabilizer selected from the group consisting of hydroxylamine stabilizers, substituted hydroxylamine stabilizers, nitrone stabilizers and amine oxide stabilizers, when extrusion compounded exhibits a lower residual acetaldehyde content than does polyester or polyamide alone when similarly treated. The invention pertains to any polyester or polyamide used in the manufacture of molded articles, fibers or films, for instance bottles or containers which are used to store consumer materials, for example food, beverages and water.
Owner:CIBA SPECIALTY CHEM CORP
Methods of treating chronic inflammatory diseases using carbonyl trapping agents
InactiveUS6444221B1Improved therapeutic propertyImprove propertiesBiocidePeptide/protein ingredientsEtiologyBenzoic acid
Owner:SECANT PHARMA
Blends of glyoxalated polyacrylamides and paper strengthening agents
Compositions comprising a blend of two or more paper strengthening agents have improved stability compared with previously known paper strengthening agents. One component is a polymeric paper strengthening agent, preferably a wet strengthening agent. The other component is a stabilized polyacrylamide prepared by (1) reacting a first portion of glyoxal with a polyacrylamide having pendant amide groups to form a first reaction mixture comprising polyacrylamide; (2) adding an acid to the first reaction mixture to form a second reaction mixture having a reduced pH and comprising the polyacrylamide; and (3) adding a second portion of glyoxal to the second reaction mixture to form the stabilized polyacrylamide. If desired, an aldehyde scavenger can be included in one or more of step (1), step (2), step (3), or the stabilized polyacrylamide. Such compositions can be used to enhance the strength of paper, particularly the wet strength of paper.
Owner:ECOLAB USA INC
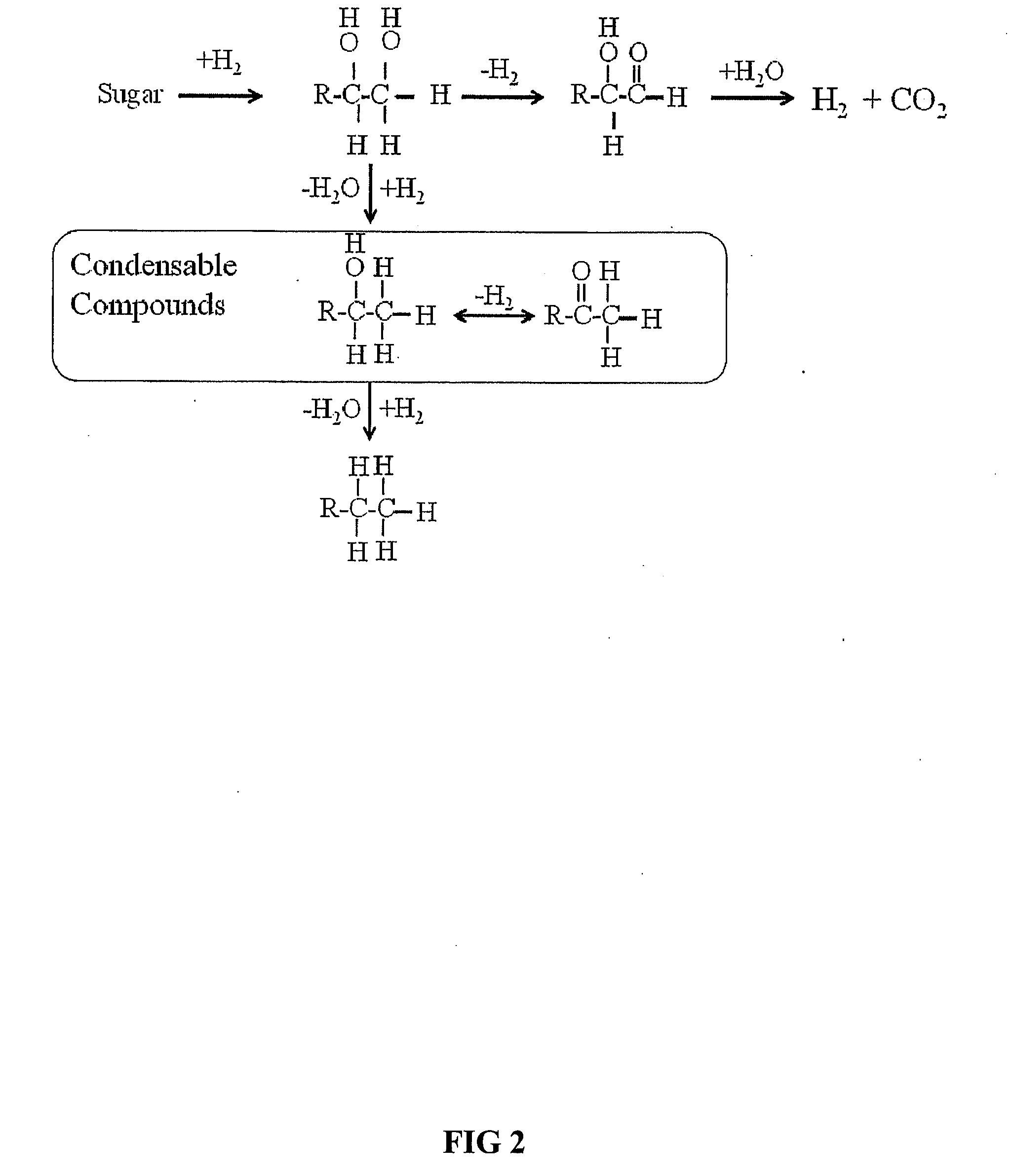
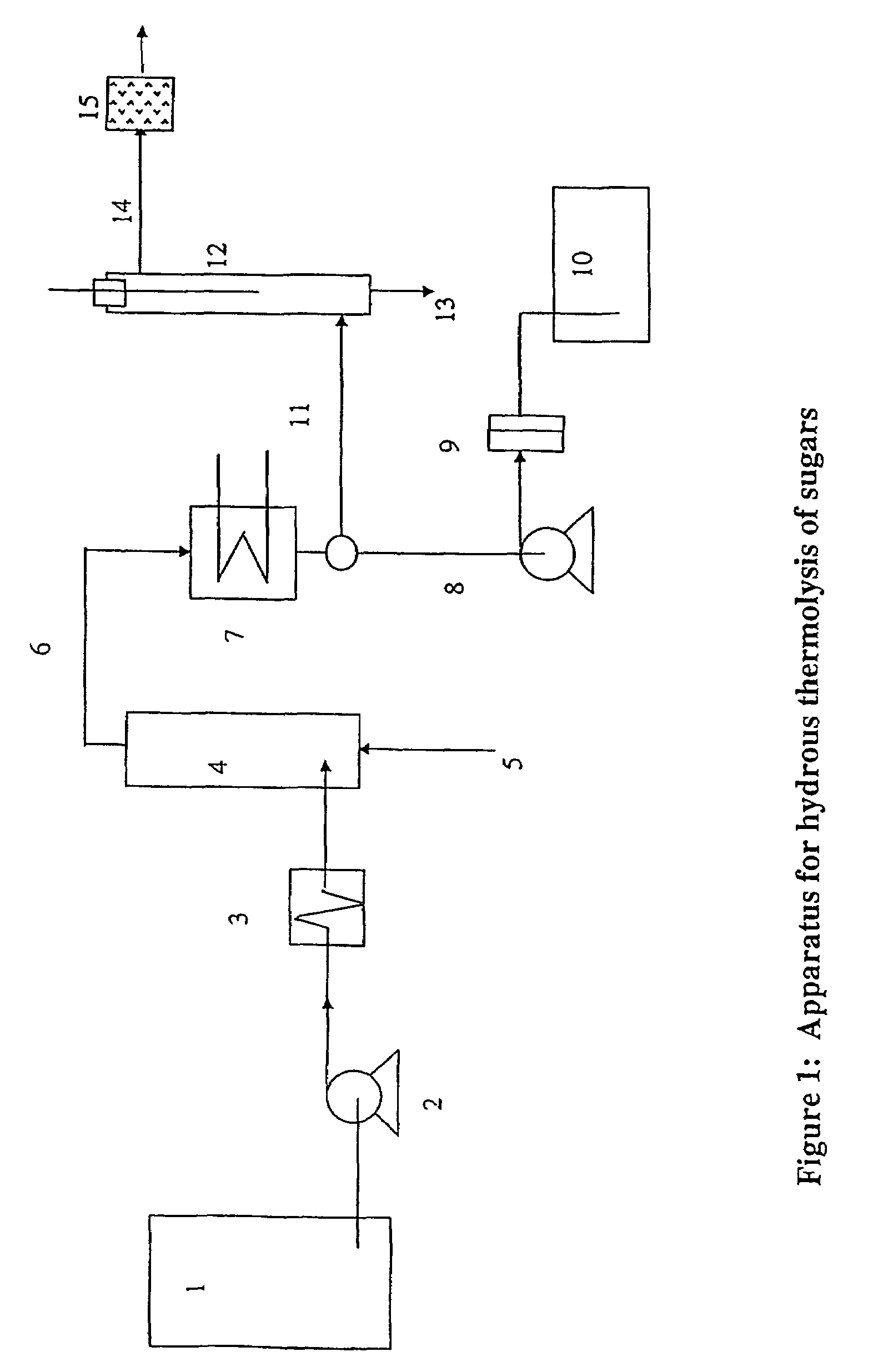
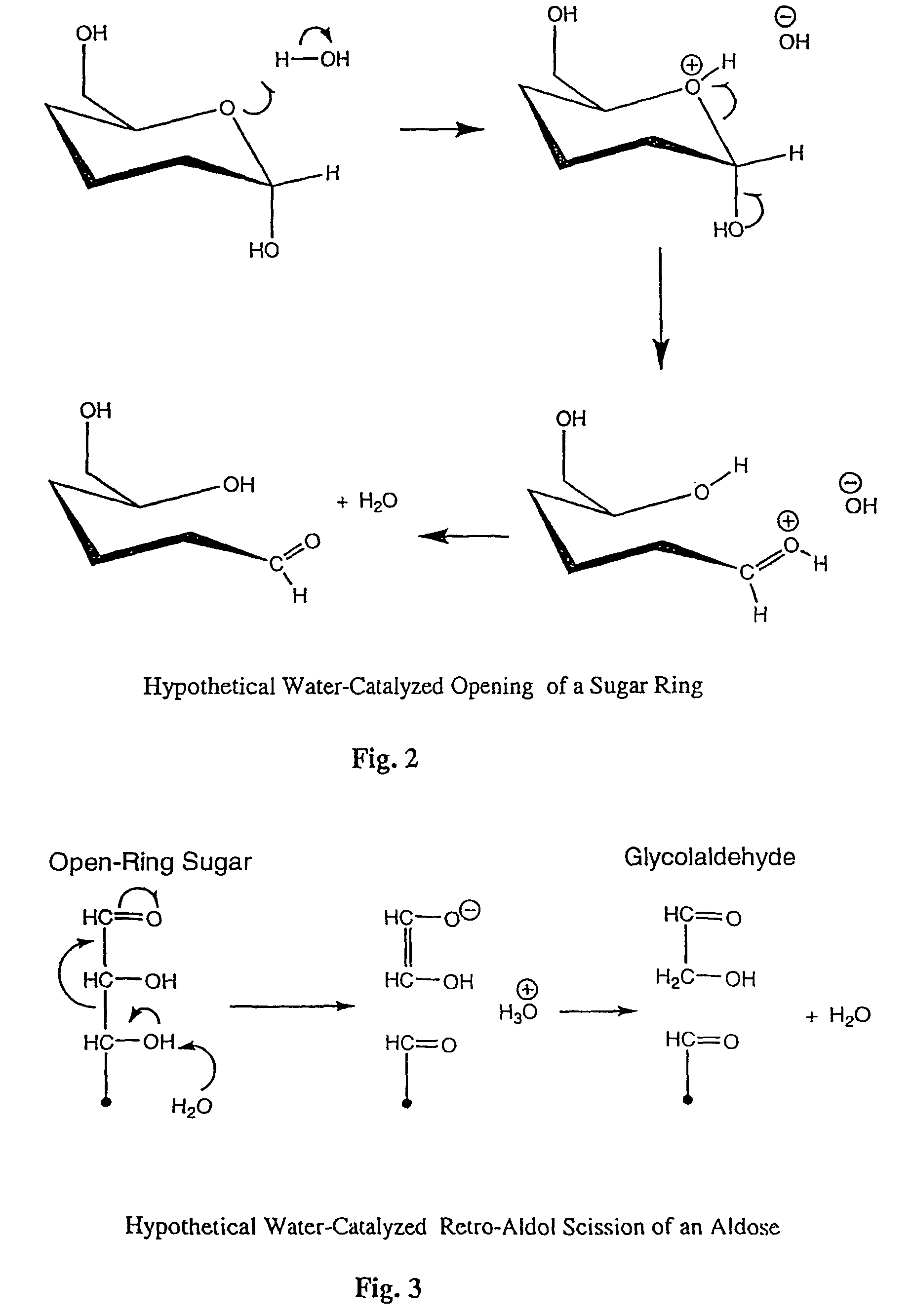
Production of glycolaldehyde by hydrous thermolysis of sugars
ActiveUS7094932B2Increase productionMinimize formationOrganic compound preparationConfectioneryGlucose polymersD-Glucose
The present invention provides a method for the production of glycolaldehyde with high specificity. The hydrous thermolysis consists of the spraying of aqueous sugar solutions containing from 25 to 80% of water but preferably 30 to 60% water, as a fine mist into a reactor held at the between 500 and 600° C., but preferably between 520 and 560° C. and the condensation of the resulting vaporous product in a surface condenser with optional heat recovery. The residence time of the vaporous product in the reactor should be in the range 0.1–5 seconds, but preferably in the range 0.5 to 2 seconds. Aldose monomeric sugars, preferably glucose (also known as dextrose), are preferred for use in the aqueous solution. The yield of glycolaldehyde in the condensed liquid is minimum 50% by weight of the sugar fed for glucose solutions.
Owner:KERRY GRP SERVICES INT
Process, composition and kit for providing a stable whole blood calibrator/control
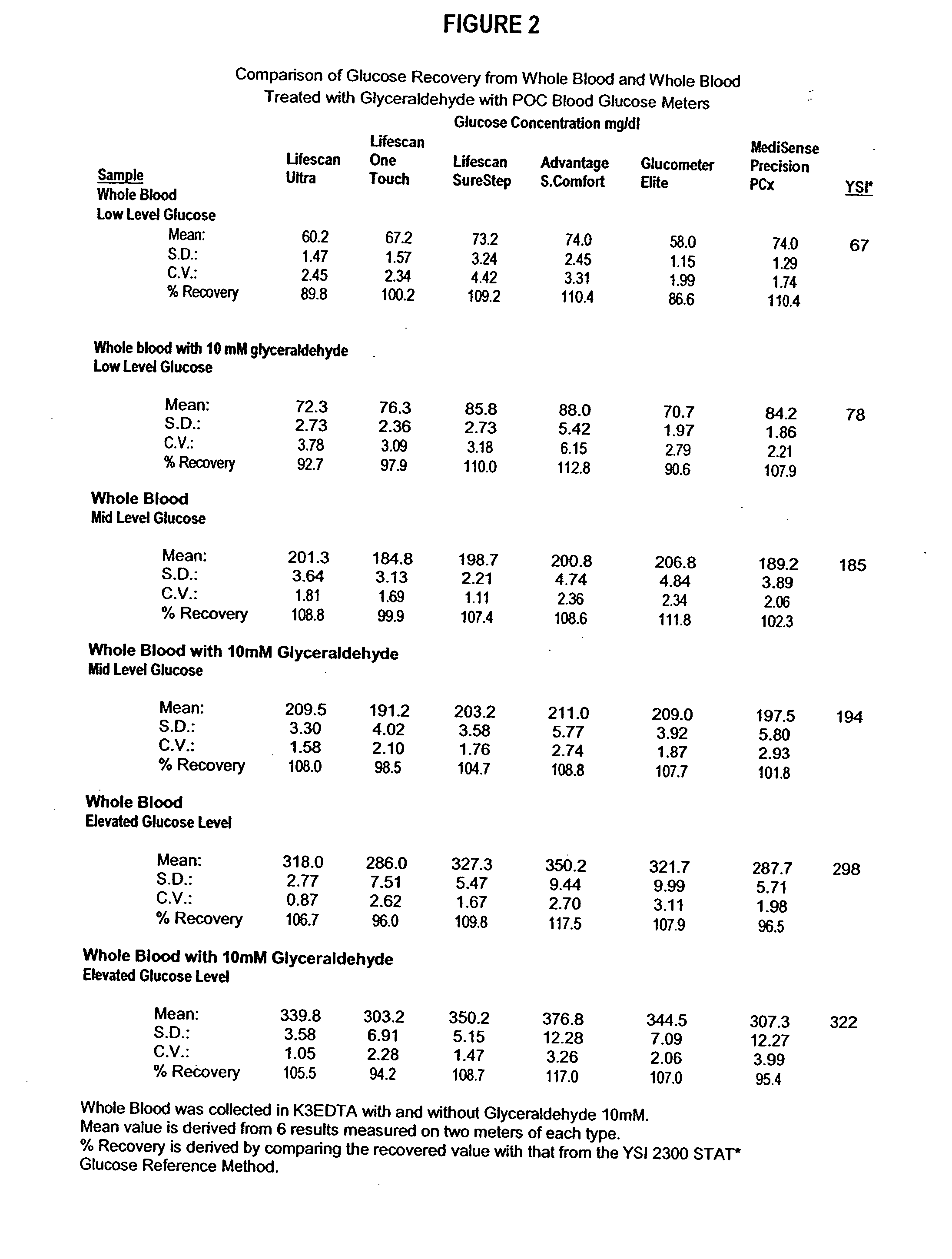
InactiveUS20060211072A1Quality improvementSimple and elegantSamplingMicrobiological testing/measurementMedicineConcentrations glucose
The present invention is directed toward a stable calibrator and / or control, kit and process for using in a glucose monitoring instrumentation. Principally, the instant invention teaches a glycolyzed red blood cell component which has been treated with a glycolysis stabilizing effective amount of at least one non-crosslinking aldehyde compound which may be added to fresh plasma along with an amount of glucose to form a simulated whole blood glucose control product, effective for maintaining a particular and essentially stable glucose concentration over a period of time sufficient for accurate measurement and calibration of a glucose measuring instrument.
Owner:STRECK INC
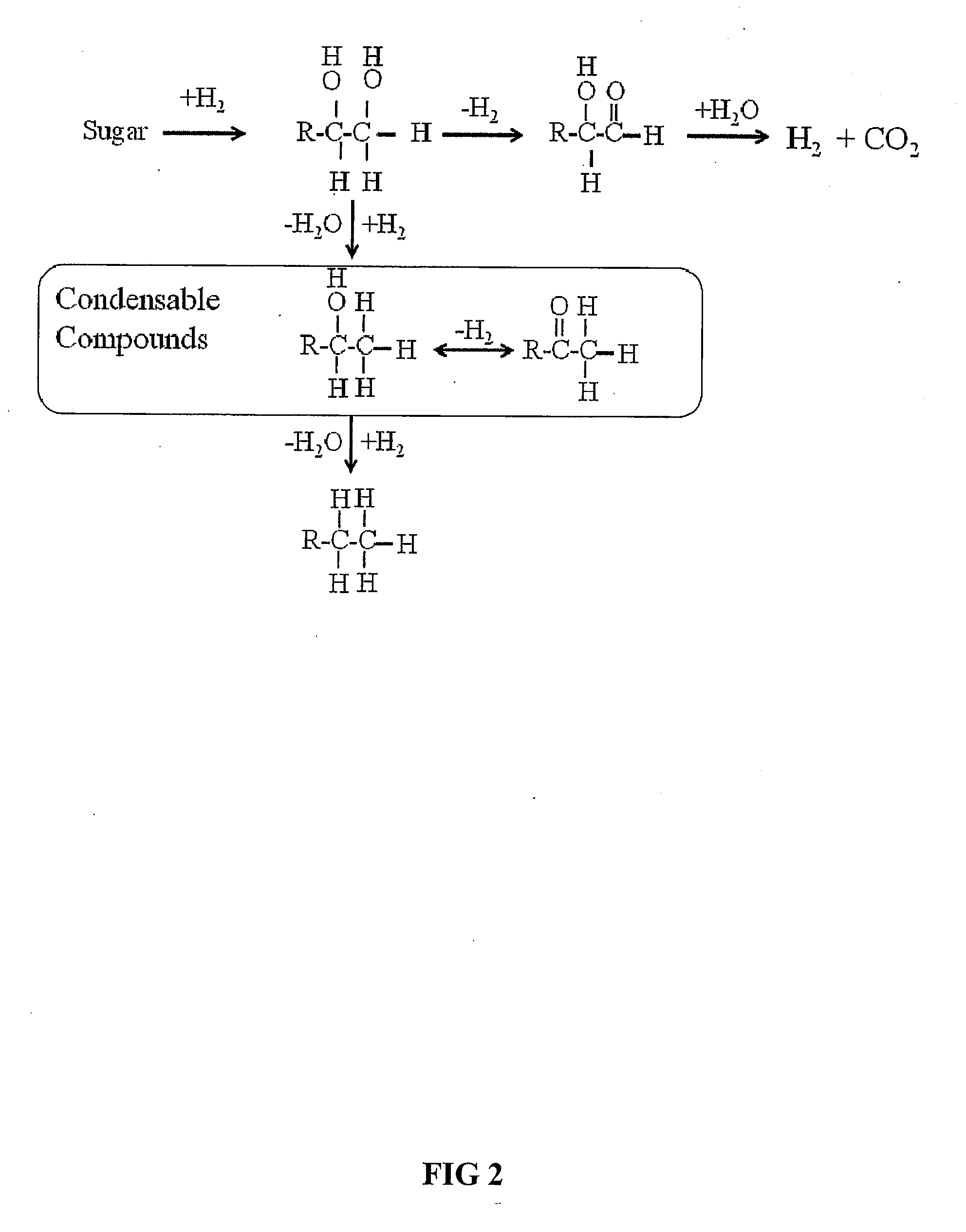
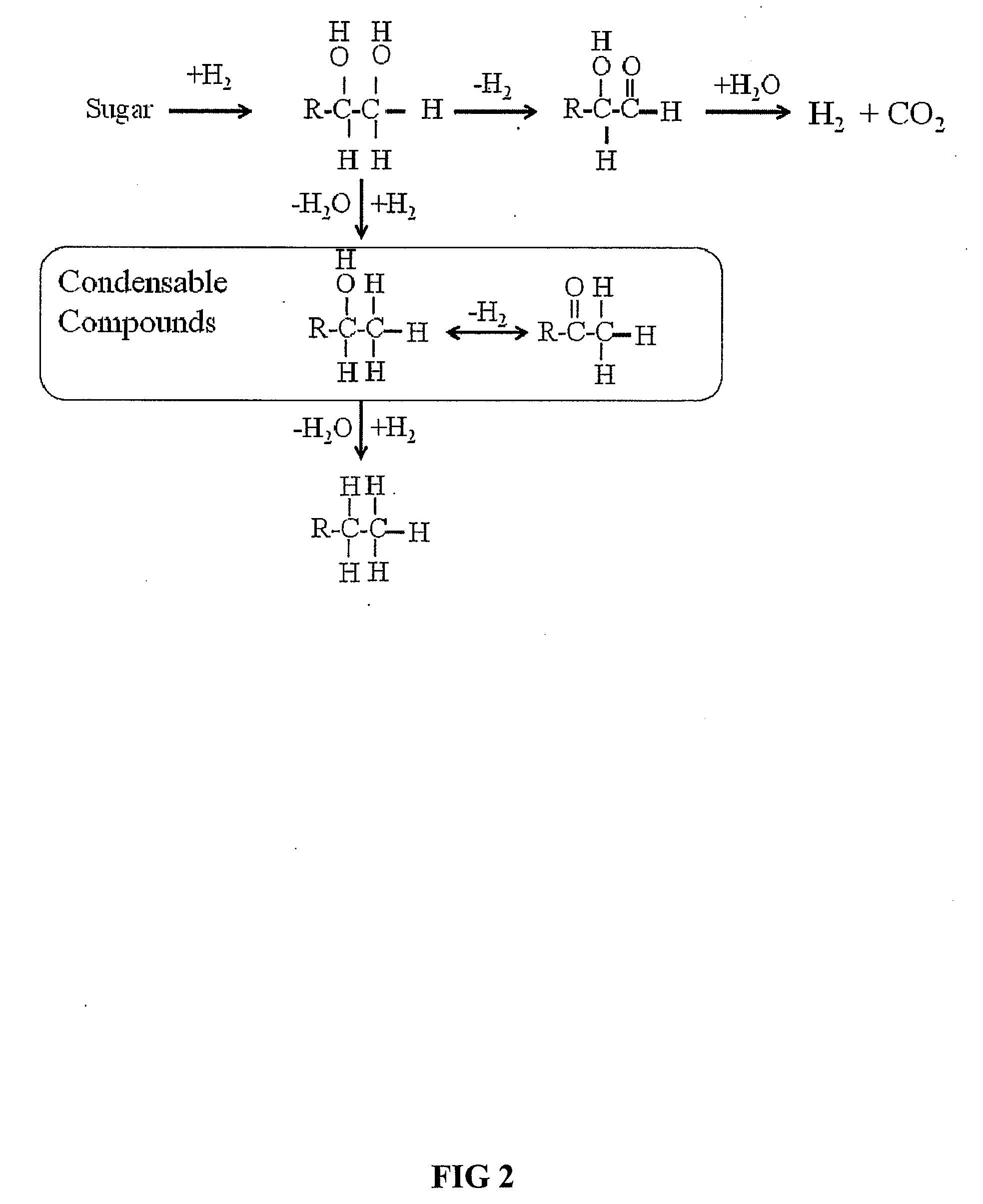
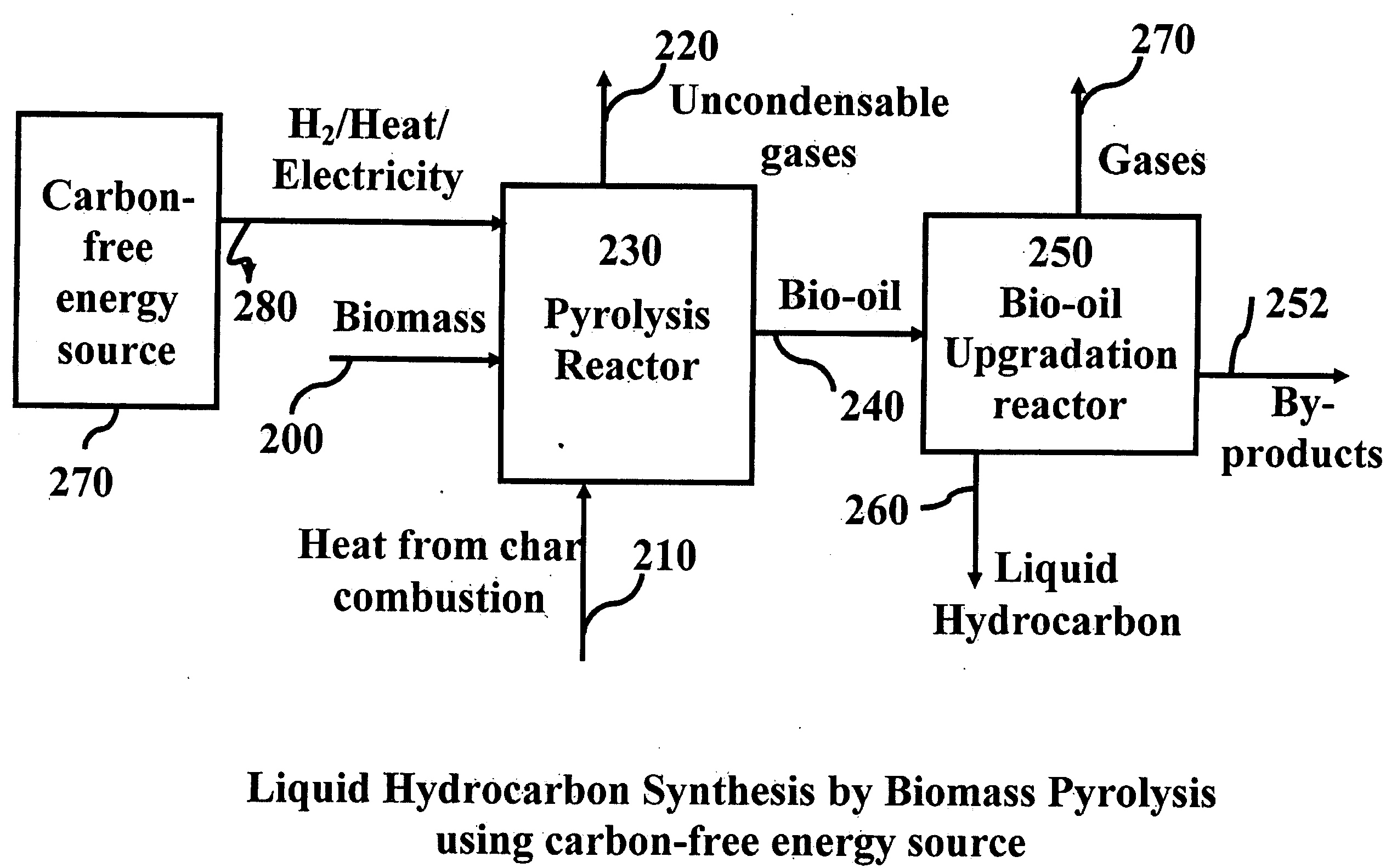
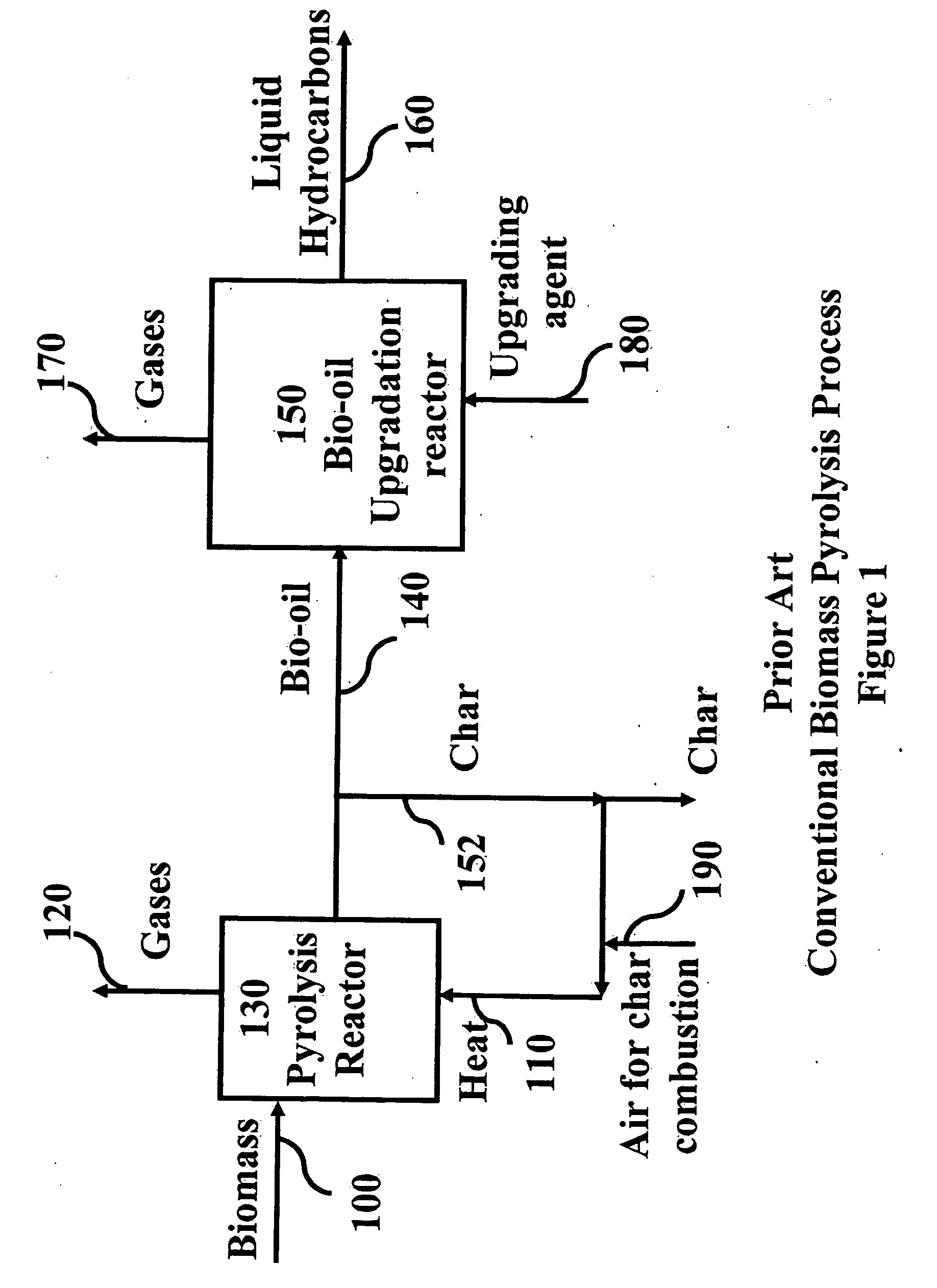
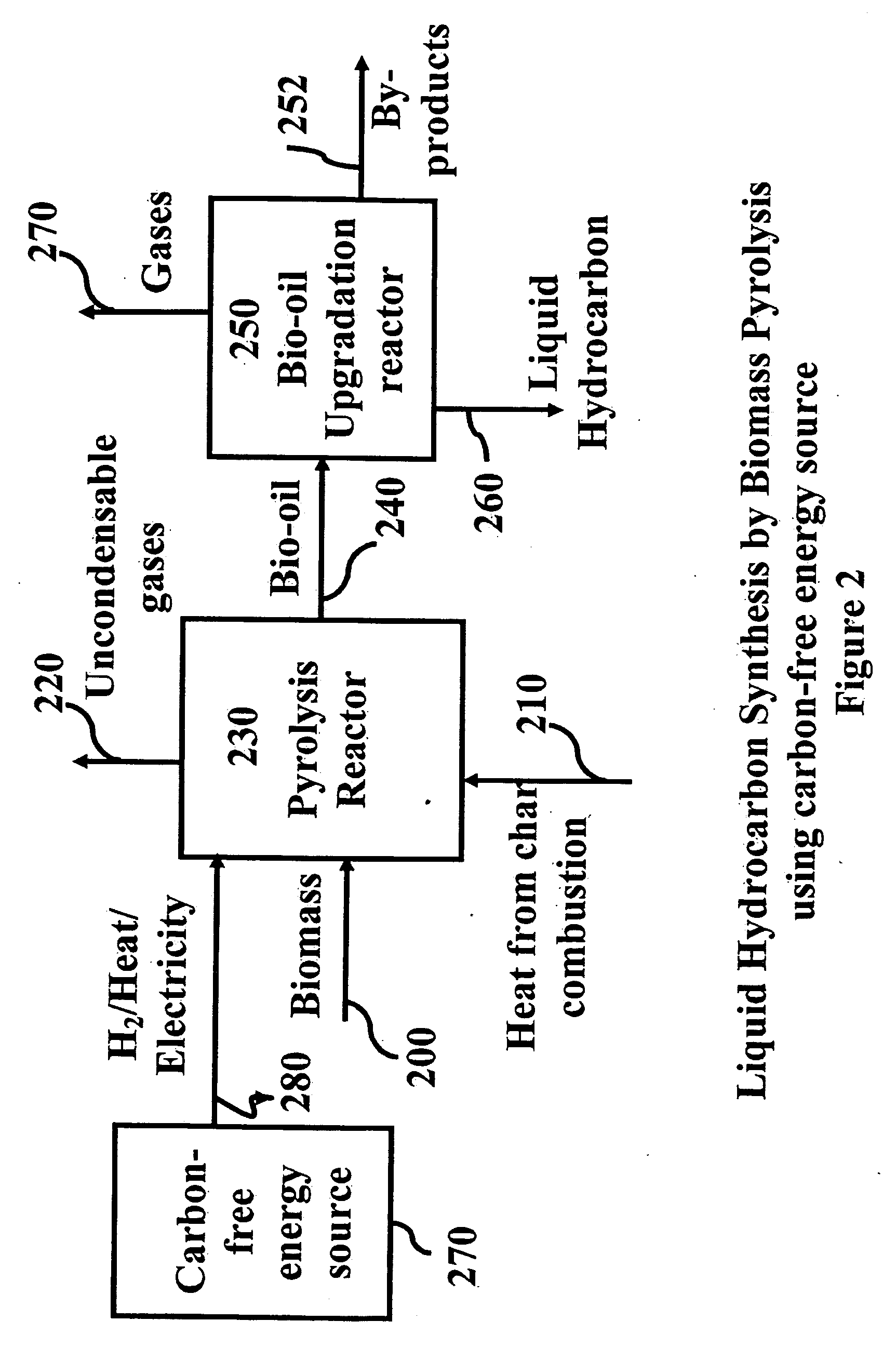
Novel process for producing liquid hydrocarbon by pyrolysis of biomass in presence of hydrogen from a carbon-free energy source
ActiveUS20090082604A1Process stabilityIncrease energy contentBioreactor/fermenter combinationsBiological substance pretreatmentsAlkaneHydrogen
In at least one embodiment of the present invention, a method for producing liquid hydrocarbons from biomass is provided. The method comprises pyrolizing the biomass with hydrogen (H2) to form bio-oil. The bio-oil comprises alkanes, alkenes, alcohols, aldehydes, ketones, aromatics, hydrocarbons or mixtures thereof. The H2 is formed from a carbon-free energy source.
Owner:PURDUE RES FOUND INC
Quaternary Ammonium Salt of a Mannich Compound
ActiveUS20080052985A1Organic chemistryTransportation and packagingCompound aQuaternary ammonium cation
A quaternary ammonium salt detergent made from the reaction product of the reaction of: (a) Mannich reaction product having a tertiary amino group, said Mannich reaction product being prepared from the reaction of a hydrocarbyl-substituted phenol, an aldehyde, and amine; and (b) a quaternizing agent suitable for converting the tertiary amino group to a quaternary nitrogen and the use of such quaternary ammonium salt detergents in a fuel composition to reduce intake valve deposits.
Owner:THE LUBRIZOL CORP
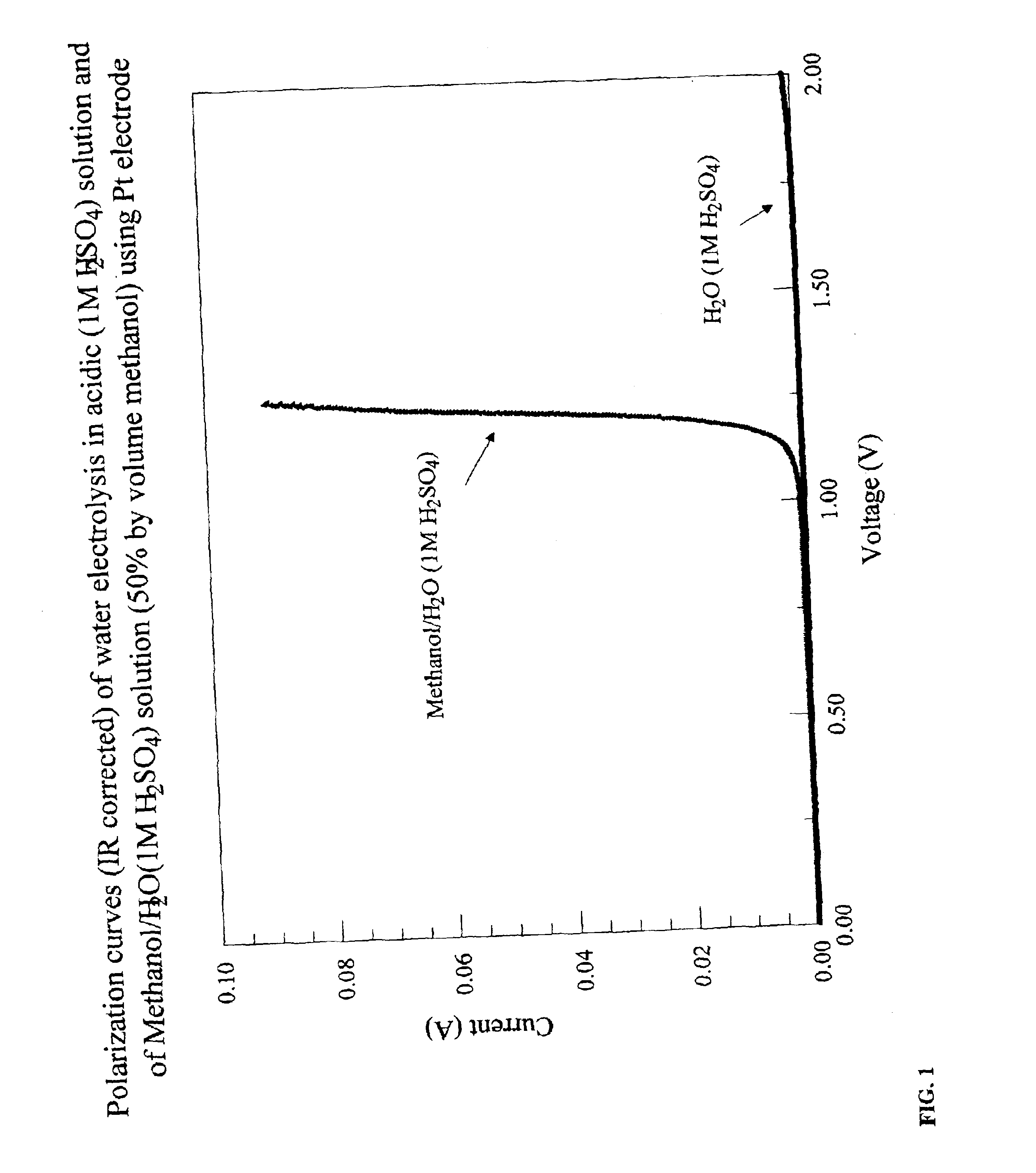
Electrolytic production of hydrogen
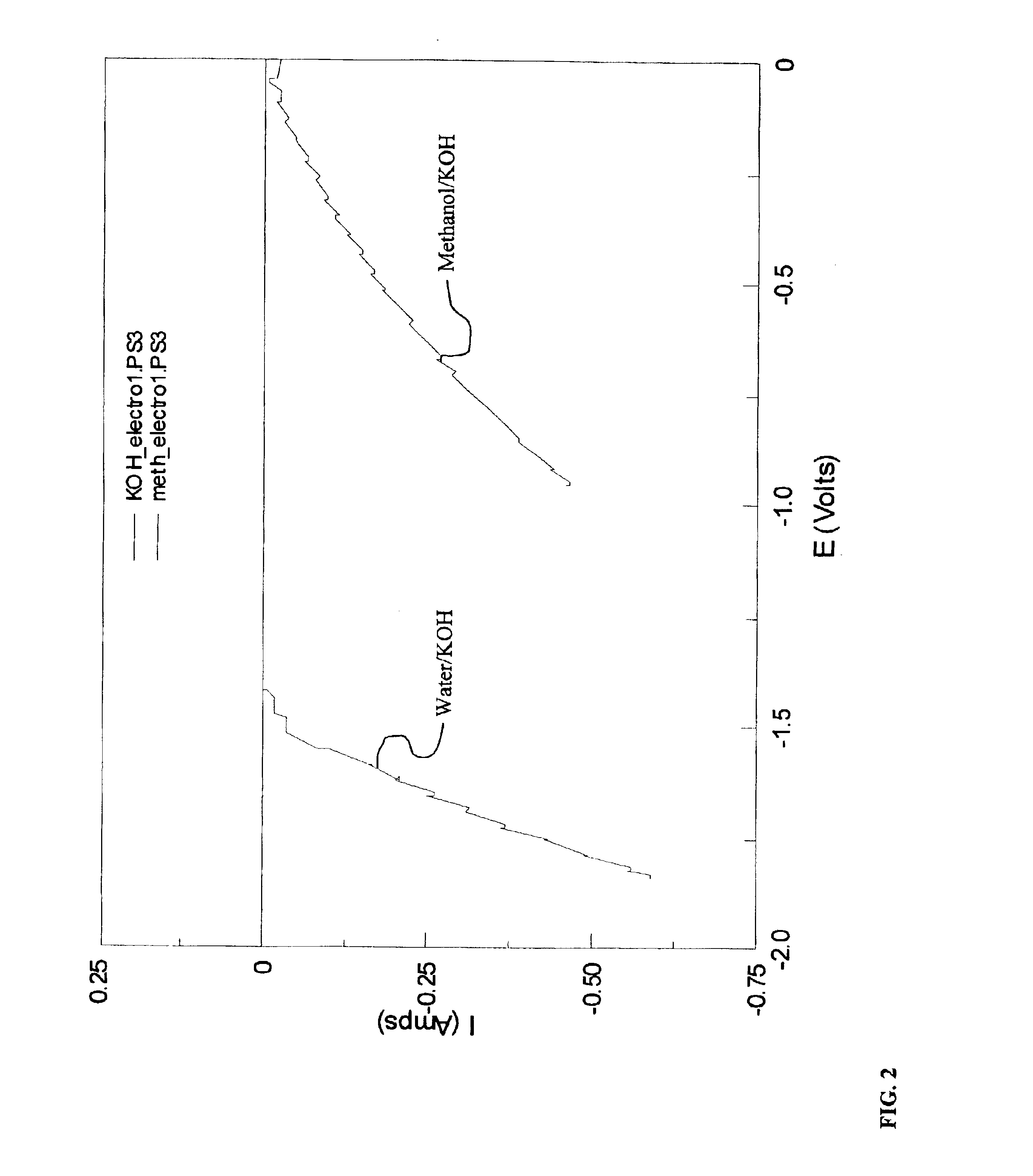
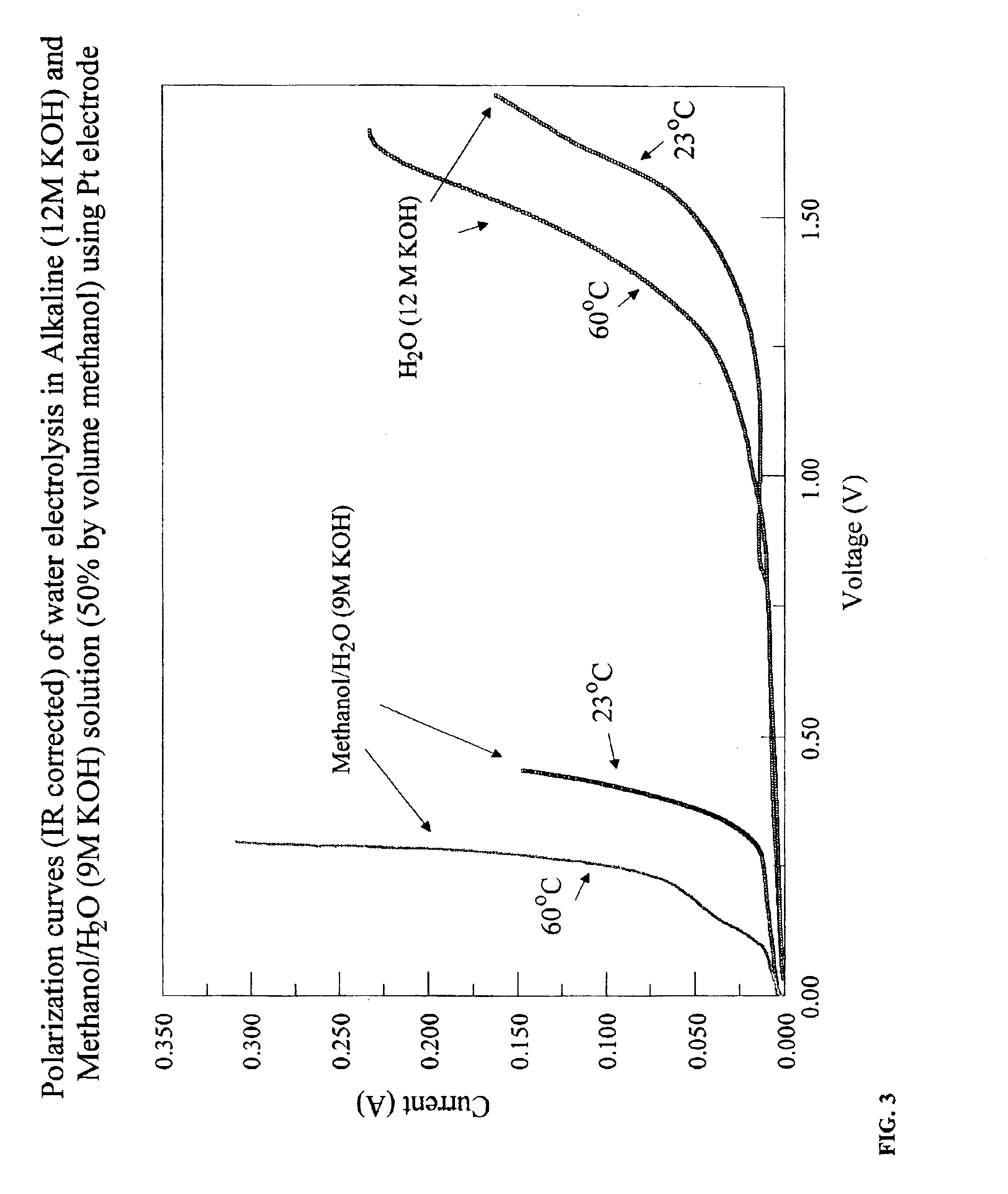
InactiveUS6890419B2Oxide/hydroxide preparationOrganic-compounds/hydrides/coordination-complexes catalystsElectrochemical responseElectrolysis
Methods for producing hydrogen gas from organic substances. According to the methods, hydrogen is produced from an electrochemical reaction of an organic substance with water or a base. The instant methods permit the production of hydrogen at lower operating voltages or lower operating temperatures relative to water electrolysis. Operable organic substances include alcohols, ethers, carboxylic acids, aldehydes, and ketones. In a preferred embodiment, hydrogen gas is produced from an electrochemical reaction of methanol in the presence of a base such as NaOH or KOH.
Owner:TACTICAL FUEL CELLS
Polyester and polyamide compositions of low residual aldehyde content
A mixture of a polyester, such as poly(ethylene terephthalate) PET, or a polyamide, and a suitable stabilizer selected from the group consisting of hydroxylamine stabilizers, substituted hydroxylamine stabilizers, nitrone stabilizers and amine oxide stabilizers, when extrusion compounded exhibits a lower residual acetaldehyde content than does polyester or polyamide alone when similarly treated. The invention pertains to any polyester or polyamide used in the manufacture of molded articles, fibers or films, for instance bottles or containers which are used to store consumer materials, for example food, beverages and water.
Owner:CIBA SPECIALTY CHEM CORP
Bioabsorbable and biocompatible polyurethanes and polyamides for medical devices
Absorbable polyurethanes, polyamides and polyester urethanes prepared from at least one compound selected from:or the diamines and diisocyanates thereof, wherein each X represents a member independently selected from —CH2COO— (glycolic acid moiety), —CH(CH3)COO— (lactic acid moiety), —CH2CH2OCH2COO— (dioxanone), —CH2CH2CH2CH2CH2COO— (caprolactone moiety), —(CH2)yCOO— where y is one of the numbers 2, 3, 4 or 6-24 inclusive, and —(CH2CH2O)z′CH2COO— where z′ is an integer between 2 and 24, inclusive; each Y represents a member independently selected from —COCH2O— (glycolic ester moiety), —COCH(CH3)O— (lactic ester moiety), —COCH2OCH2CH2O— (dioxanone ester), —COCH2CH2CH2CH2CH2O— (caprolactone ester), —CO(CH2)mO— where m is an integer between 2, 3, 4 or 6-24 inclusive, —COCH2O(CH2CH2O)n— where n is an integer between 2 and 24, inclusive; R′ is hydrogen, benzyl or an alkyl group, the alkyl group being either straight-chained or branched; p is an integer between 1 and 4, inclusive; and Rn represents one or more members selected from H, alkoxy, benzyloxy, aldehyde, halogen, carboxylic acid and —NO2, which is attached directly to an aromatic ring or attached through an aliphatic chain. Absorbable polymers prepared from these compounds are useful for drug delivery, tissue engineering, tissue adhesives, adhesion prevention and other implantable medical devices.
Owner:BEZWADA BIOMEDICAL LLC
Process for producing a branched hydrocarbon component
InactiveUS20070135316A1Reduce carbon dioxide emissionsImprove stabilityOrganic compound preparationCatalytic naphtha reformingIsomerizationHydrodeoxygenation
The invention relates to a process for producing high-quality hydrocarbon base oil particularly of biological origin. The process of the invention comprises aldol condensation, hydrodeoxygenation, and isomerization steps. Aldehydes and / or ketones, preferably of biological origin are used as the feedstock.
Owner:NESTE OIL OY
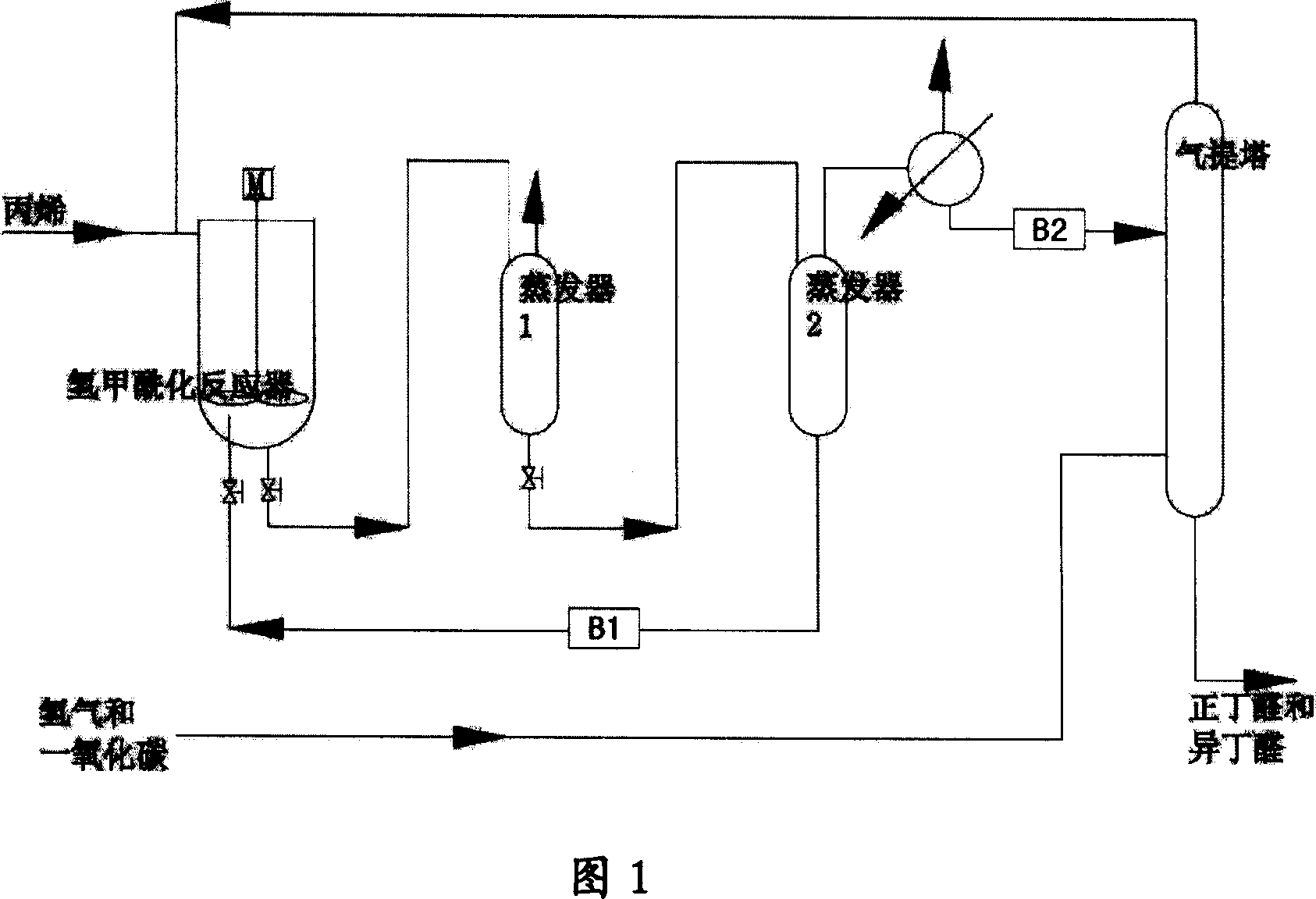
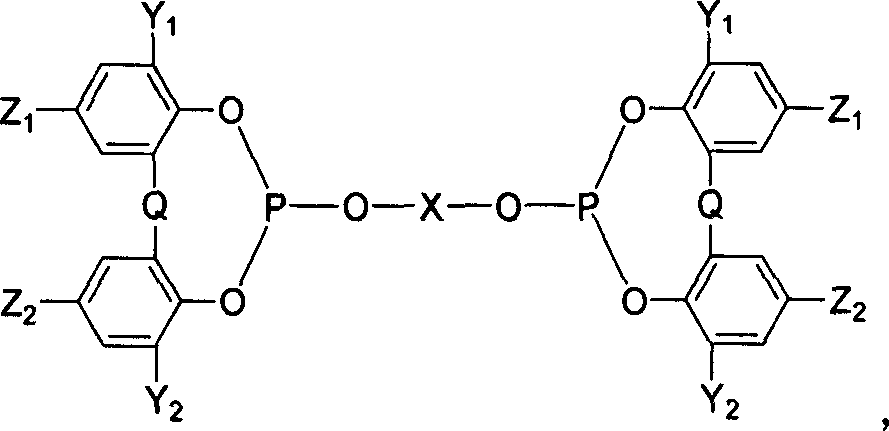
Catalyst system and catalyzing method of propylene hydrogenation and formylation
ActiveCN1986055AReduce dosageImprove stabilityOrganic-compounds/hydrides/coordination-complexes catalystsPreparation by carbon monoxide reactionFormylation reactionTriphenylphosphine
The present invention relates to catalyst system for propylene hydrogenation and formylation and process of catalytically synthesizing butyl aldehyde. The catalyst system is triaryl phosphine-Rh(I) catalyst system with proper additive, such as bisphosphite ester, in proper amount. Compared with similar available catalyst, the catalyst system has obviously higher catalytic acitivity, higher selectivity, higher stability and raised n-butyl aldehyde / isobutyl aldehyde ratio in the catalytically synthesized product.
Owner:SHANGHAI INST OF ORGANIC CHEM CHINESE ACAD OF SCI +1
Method for producing benzoxazine resin
InactiveUS7041772B2Improve efficiencyPromote safe productionNatural resin processChemical/physical/physico-chemical stationary reactorsOrganic solventReaction system
The present invention discloses a process for producing a benzoxazine resin which comprises the steps of reacting a phenol compound, an aldehyde compound and a primary amine in the presence of an organic solvent to synthesize a benzoxazine resin and removing generated condensation water and the organic solvent from a system under heating and a reduced pressure, wherein a pressure in the reaction system at the time of removal is set to 260 mmHg or higher.
Owner:HITACHI CHEM CO LTD