Patents
Literature
31992 results about "Nickel" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Nickel is a chemical element with the symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel belongs to the transition metals and is hard and ductile. Pure nickel, powdered to maximize the reactive surface area, shows a significant chemical activity, but larger pieces are slow to react with air under standard conditions because an oxide layer forms on the surface and prevents further corrosion (passivation). Even so, pure native nickel is found in Earth's crust only in tiny amounts, usually in ultramafic rocks, and in the interiors of larger nickel–iron meteorites that were not exposed to oxygen when outside Earth's atmosphere.
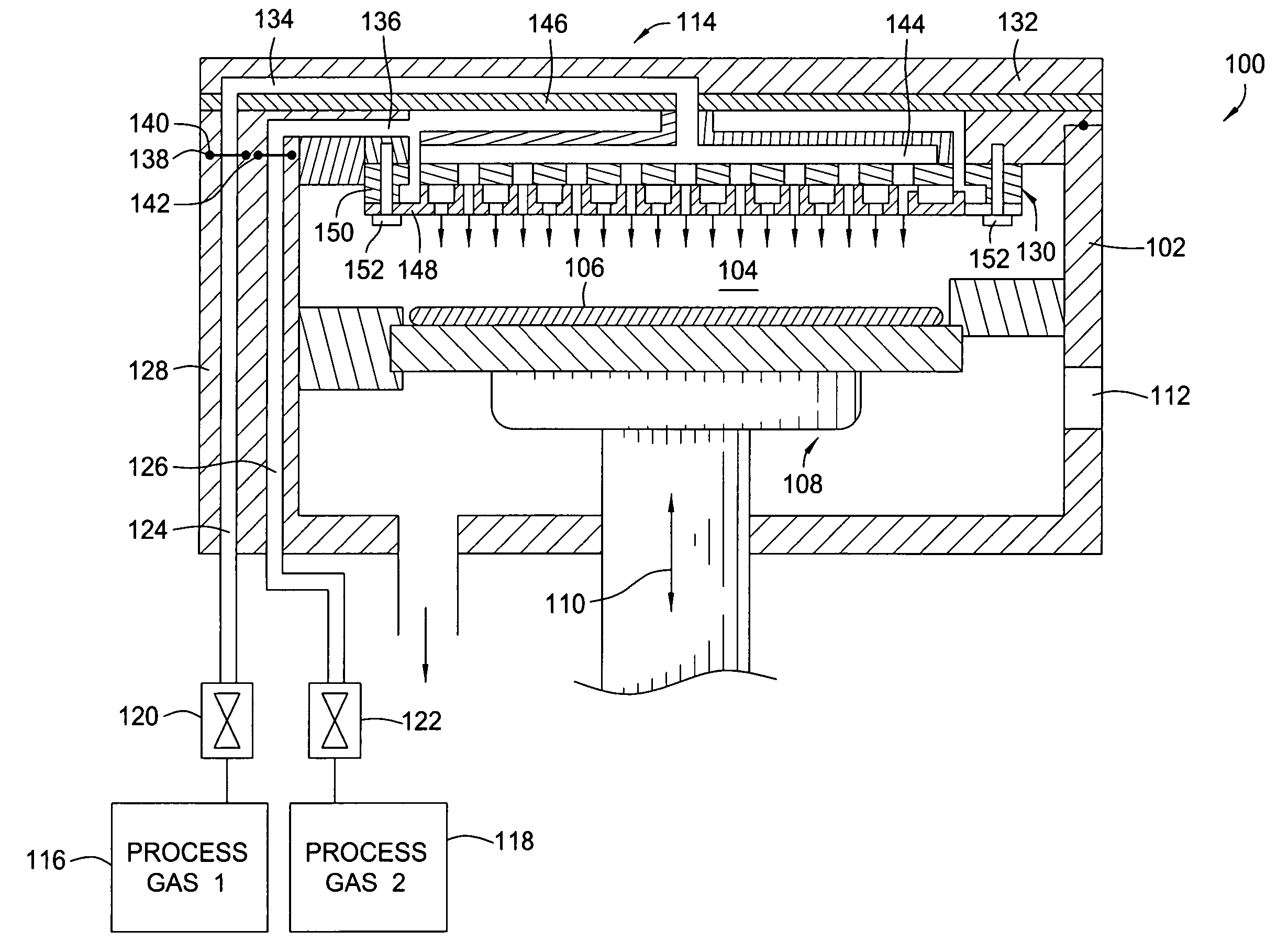
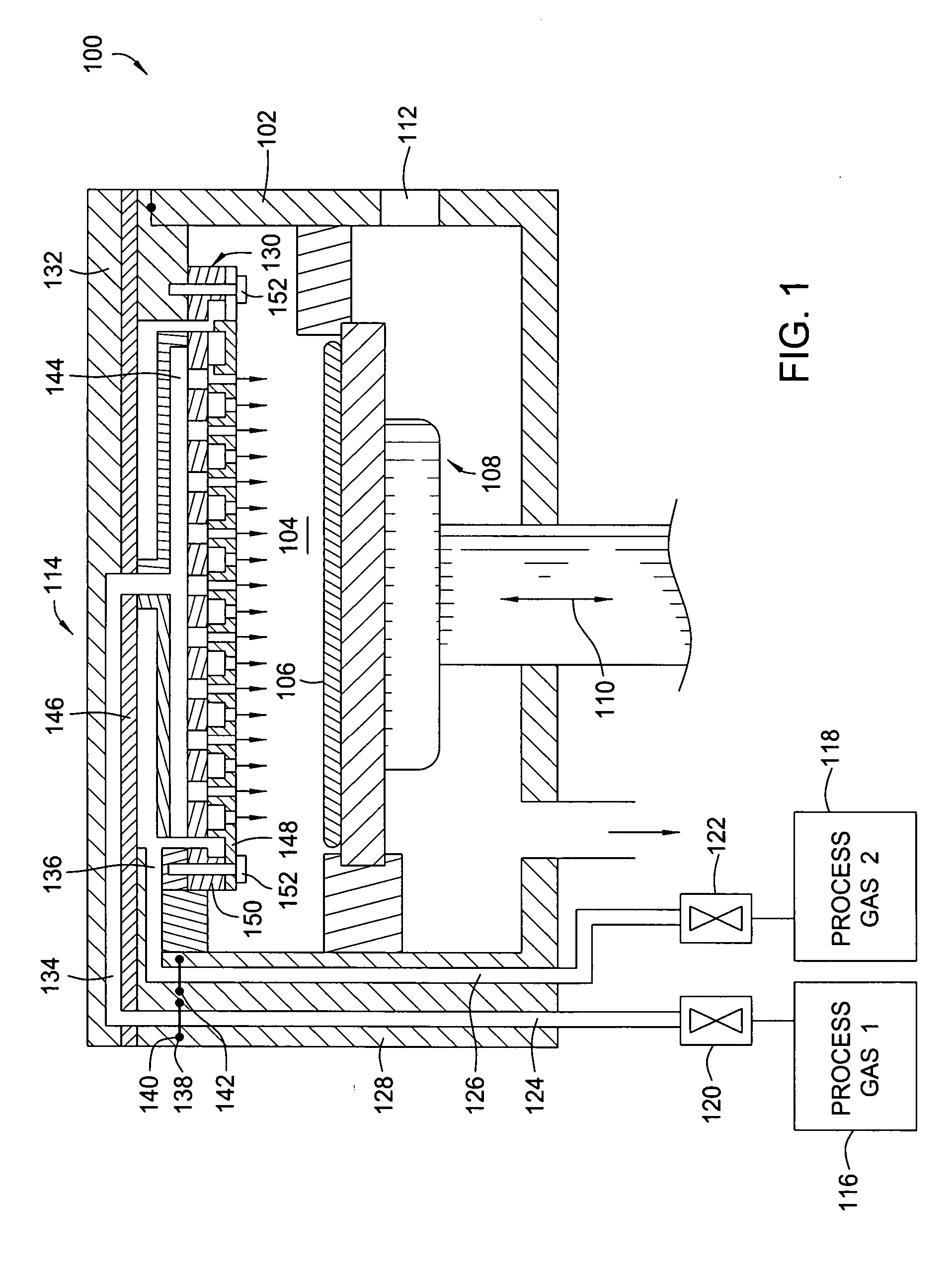
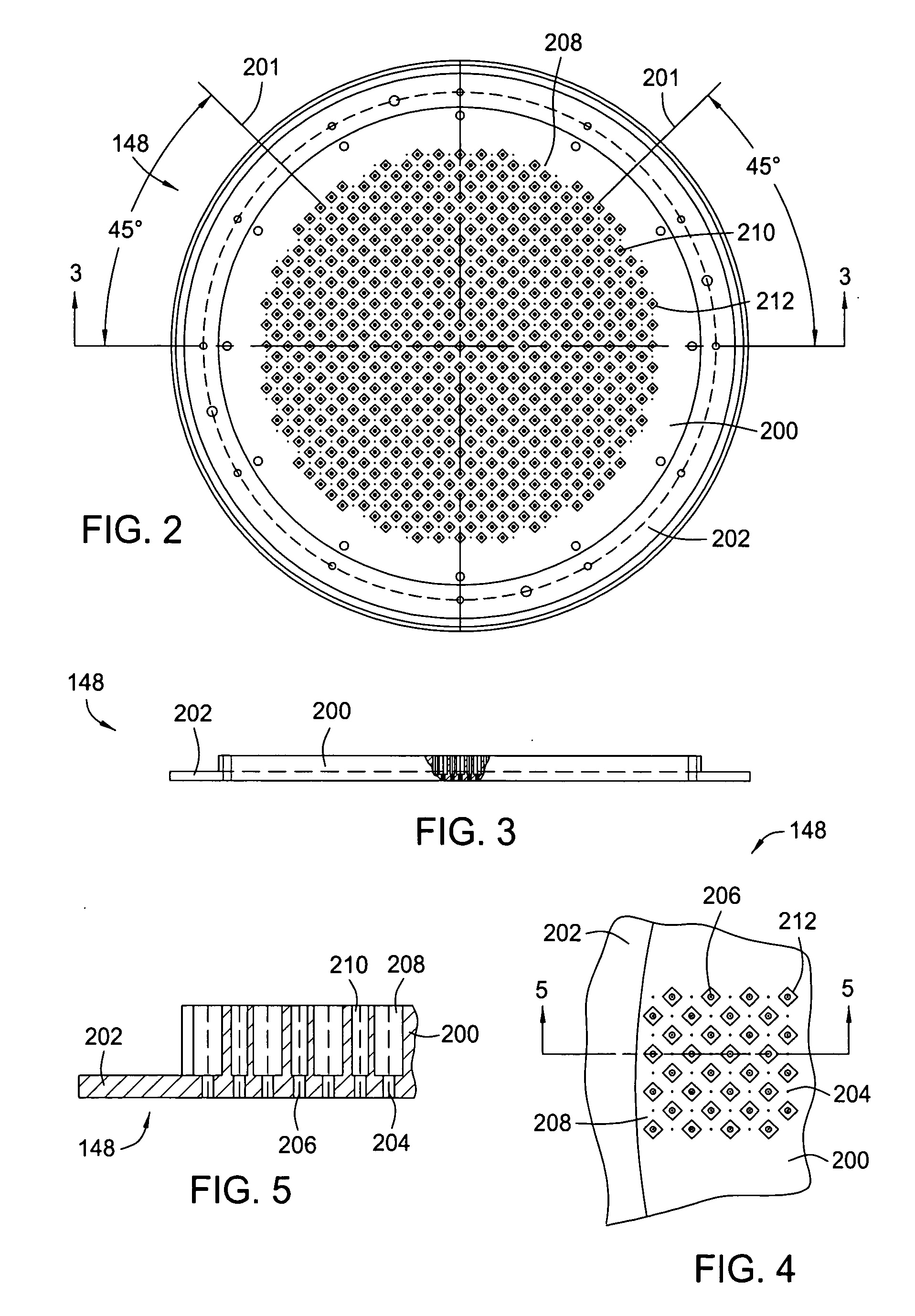
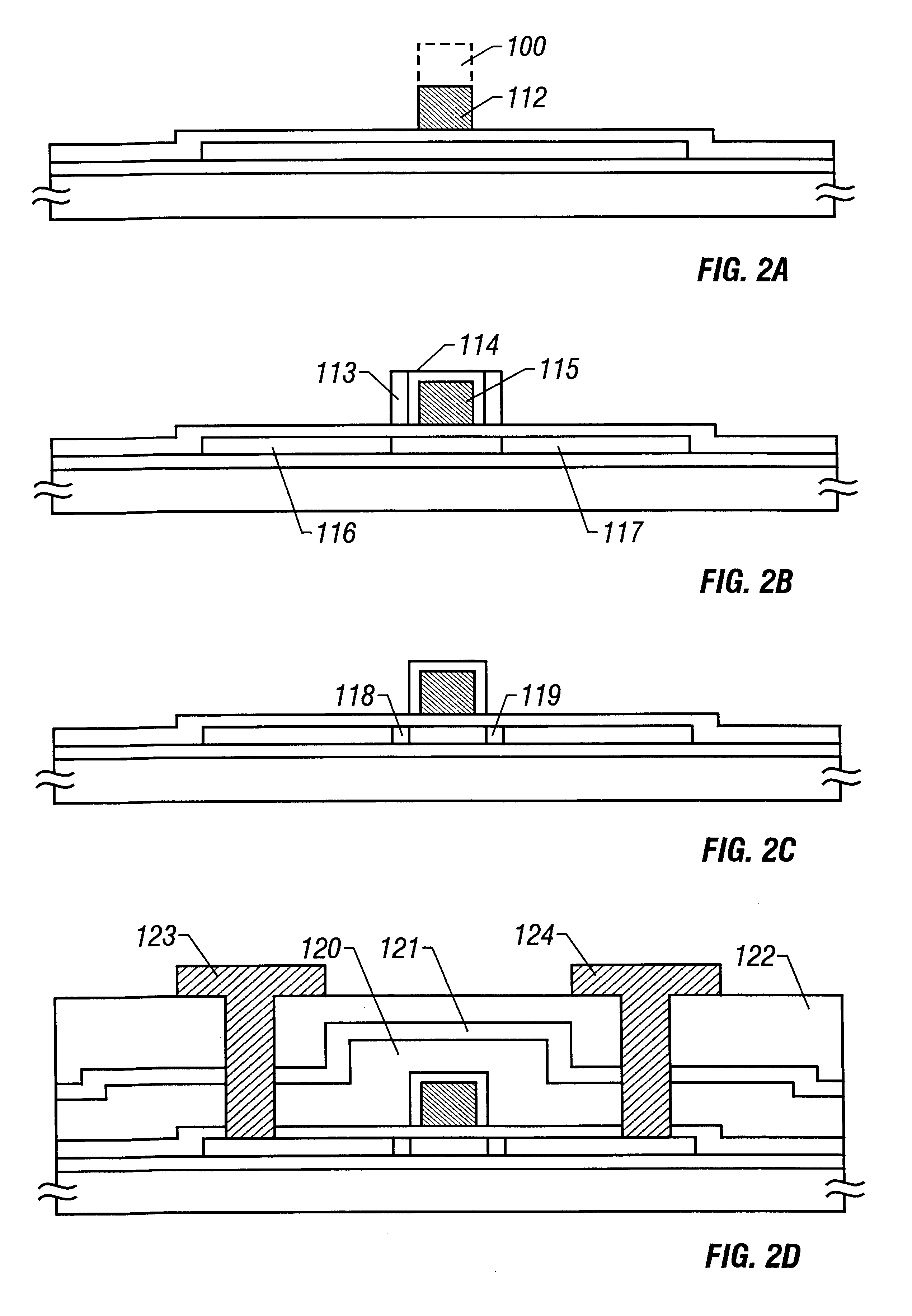
Dual gas faceplate for a showerhead in a semiconductor wafer processing system
InactiveUS20060021703A1Even gas distributionElectric discharge tubesSemiconductor/solid-state device manufacturingProcess regionProcess engineering
Owner:APPLIED MATERIALS INC
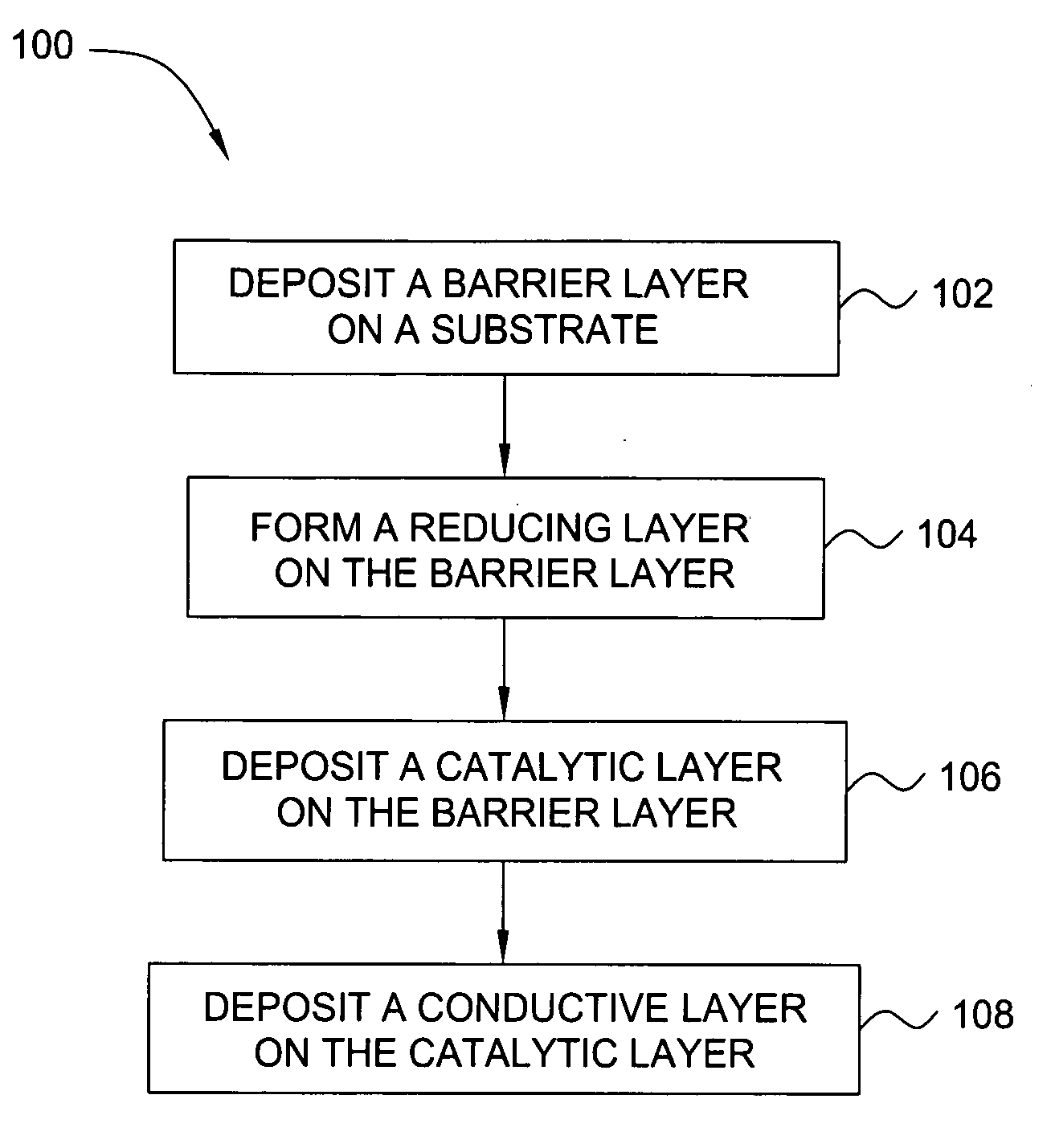
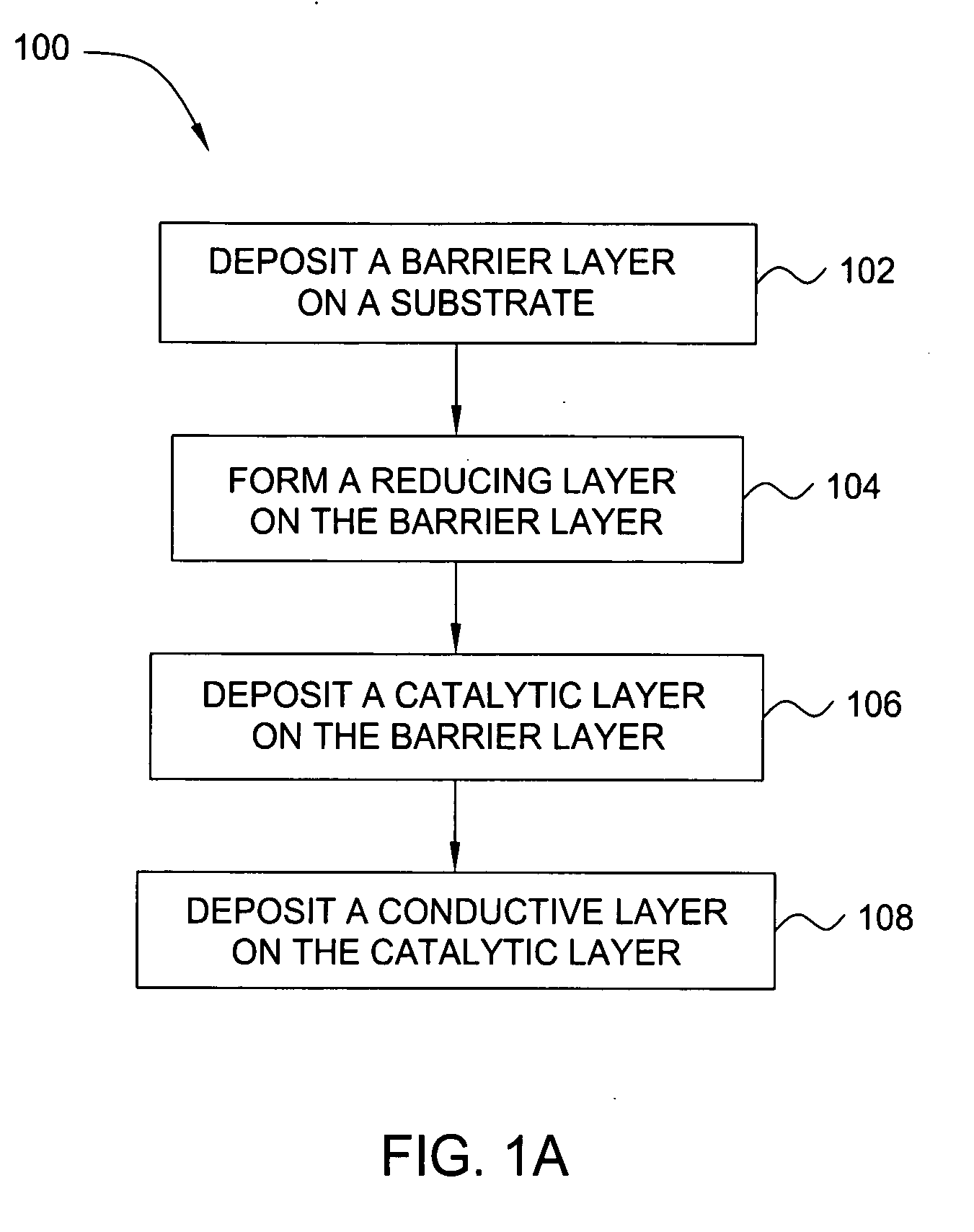
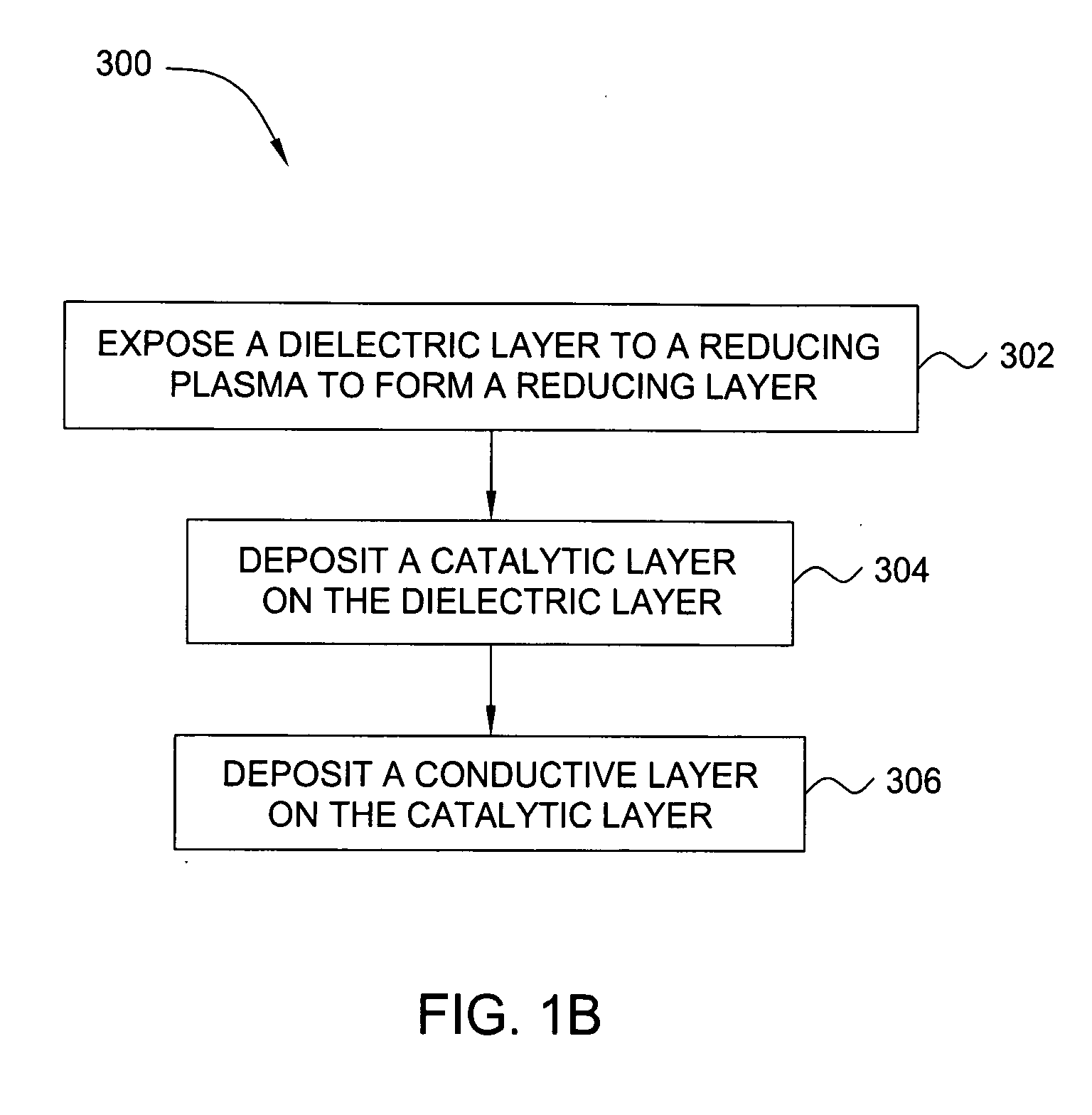
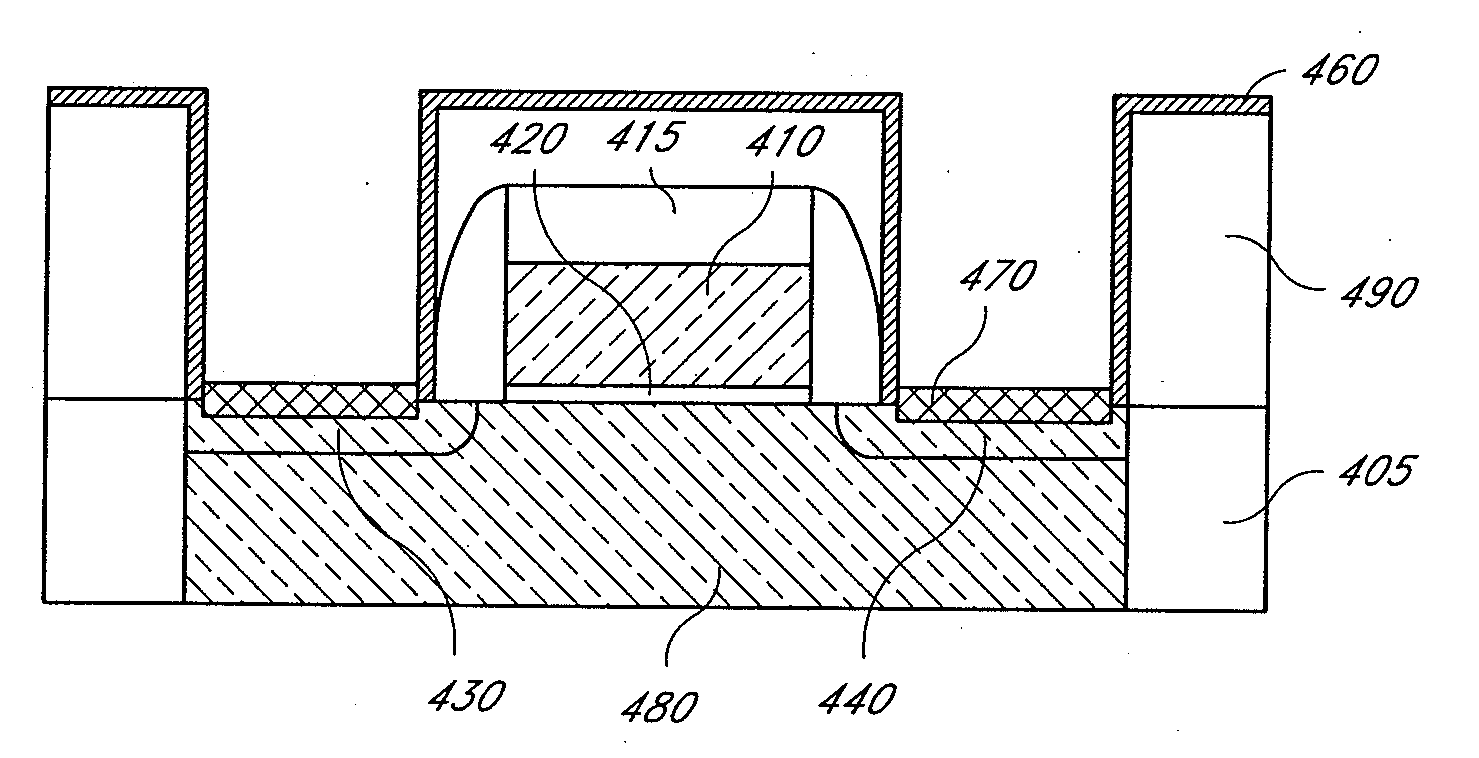
Deposition of an intermediate catalytic layer on a barrier layer for copper metallization
InactiveUS20060240187A1Solid-state devicesSemiconductor/solid-state device manufacturingIridiumSilanes
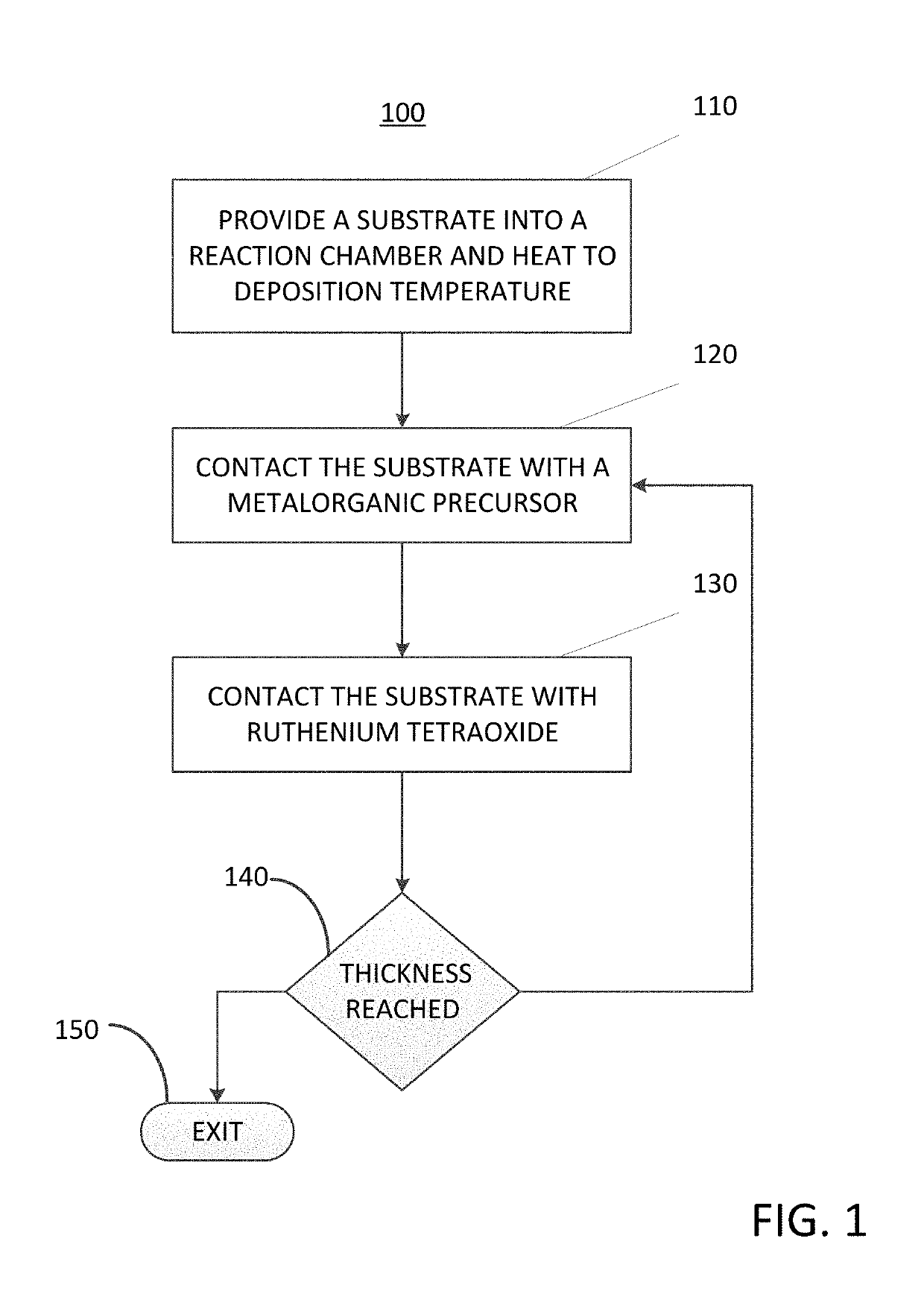
In one embodiment, a method for depositing a conductive material on a substrate is provided which includes exposing a substrate containing a barrier layer to a volatile reducing precursor to form a reducing layer during a soak process, exposing the reducing layer to a catalytic-metal precursor to deposit a catalytic metal-containing layer on the barrier layer, and depositing a conductive layer (e.g., copper) on the catalytic metal-containing layer. The volatile reducing precursor may include phosphine, diborane, silane, a plasma thereof, or a combination thereof and be exposed to the substrate for a time period within a range from about 1 second to about 30 seconds during the soak process. The catalytic metal-containing layer may contain ruthenium, cobalt, rhodium, iridium, nickel, palladium, platinum, silver, or copper. In one example, the catalytic metal-containing layer is deposited by a vapor deposition process utilizing ruthenium tetroxide formed by an in situ process.
Owner:APPLIED MATERIALS INC
Methods for depositing nickel films and for making nickel silicide and nickel germanide
ActiveUS20130115768A1Semiconductor/solid-state device manufacturingSemiconductor devicesGas phaseMetal silicide
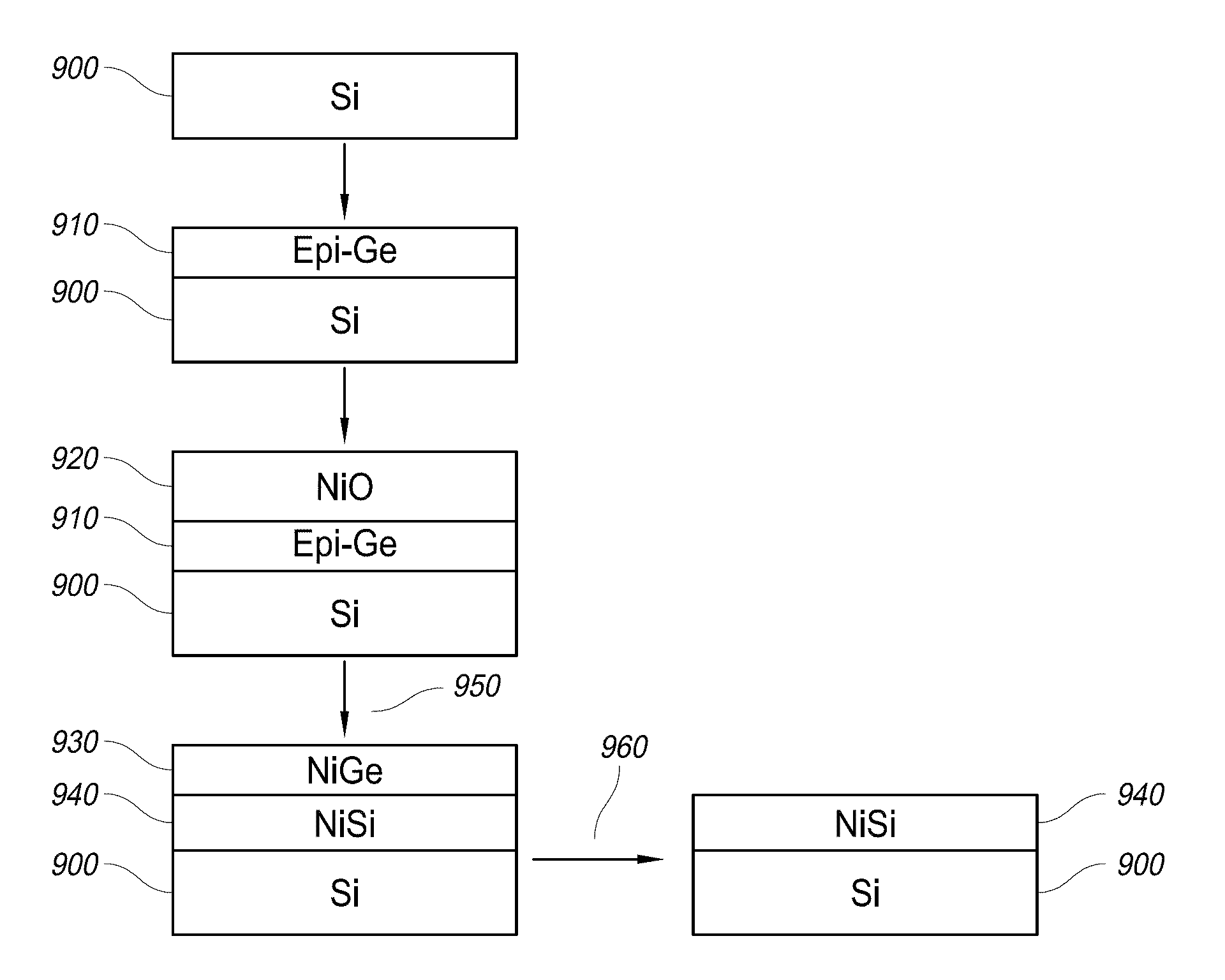
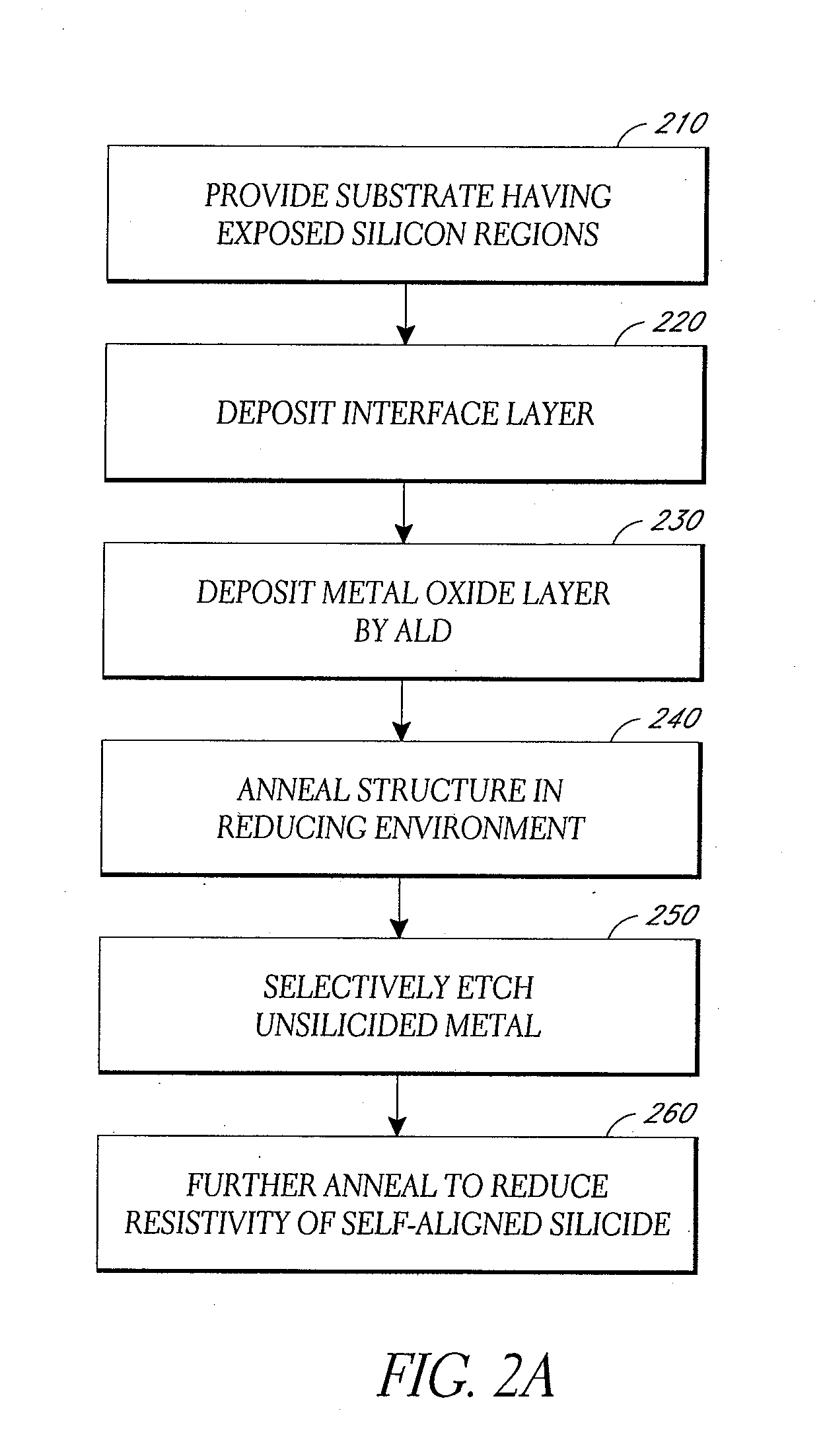
In one aspect, methods of silicidation and germanidation are provided. In some embodiments, methods for forming metal silicide can include forming a non-oxide interface, such as germanium or solid antimony, over exposed silicon regions of a substrate. Metal oxide is formed over the interface layer. Annealing and reducing causes metal from the metal oxide to react with the underlying silicon and form metal silicide. Additionally, metal germanide can be formed by reduction of metal oxide over germanium, whether or not any underlying silicon is also silicided. In other embodiments, nickel is deposited directly and an interface layer is not used. In another aspect, methods of depositing nickel thin films by vapor phase deposition processes are provided. In some embodiments, nickel thin films are deposited by ALD. Nickel thin films can be used directly in silicidation and germanidation processes.
Owner:ASM INTERNATIONAL
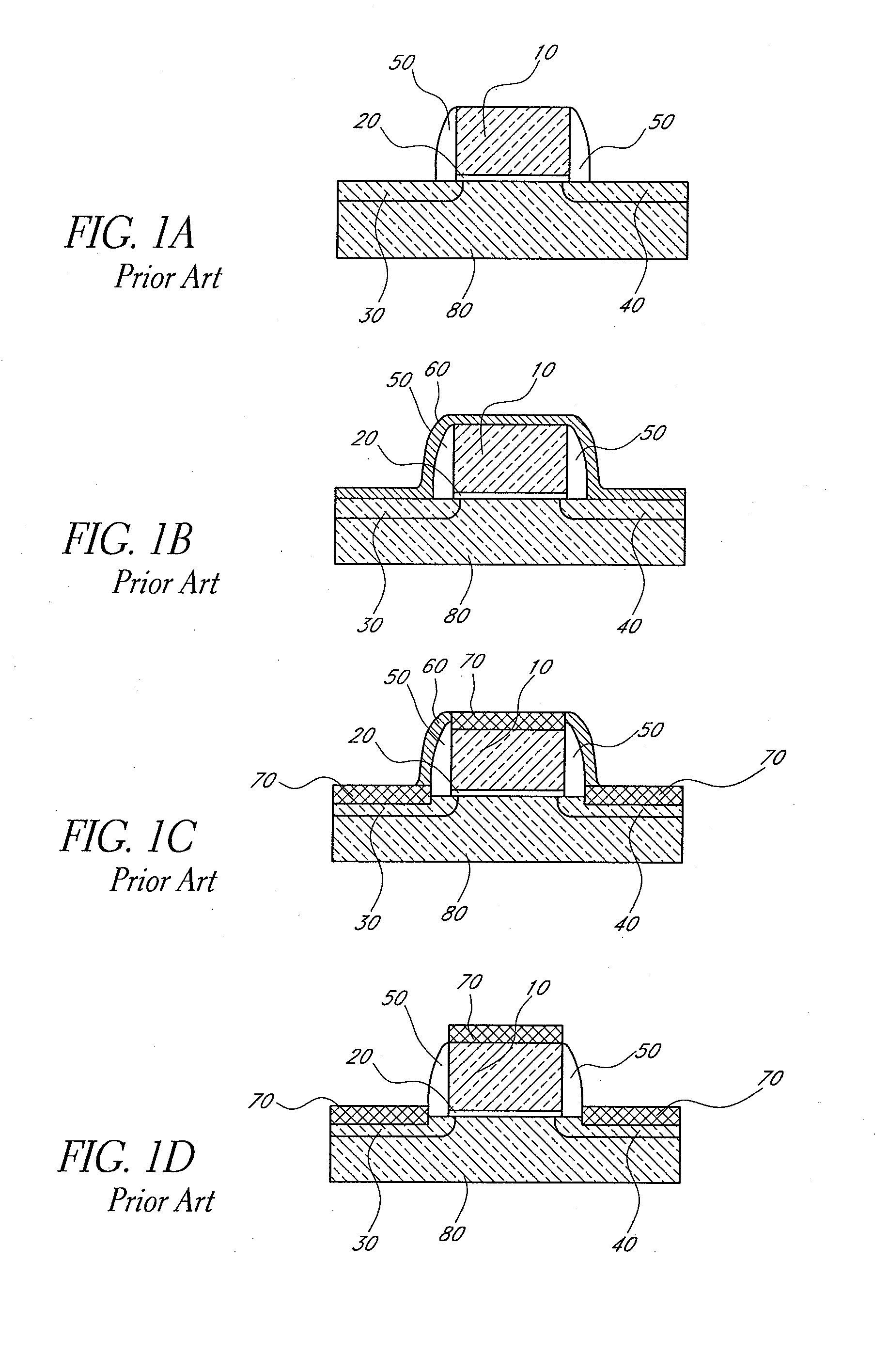
Method of manufacturing a semiconductor device
InactiveUS6165824ATransistorSemiconductor/solid-state device manufacturingCrystallinityCrystal growth
A crystal growth 301 is carried out by diffusing a metal element, and a nickel element is moved into regions 108 and 109 which has been doped with phosphorus. An axis coincident with the moving directions 302 and 303 of the nickel element at this time is made to coincide with an axis coincident with the direction of the crystal growth, and a TFT having the regions as channel forming regions is manufactured. In the path of the region where nickel moved, since high crystallinity is obtained in the moving direction, the TFT having high characteristics can be obtained by this way.
Owner:SEMICON ENERGY LAB CO LTD
Metal silicide, metal germanide, methods for making the same
ActiveUS20120270393A1Prevent oxidationSelective silicidationSemiconductor/solid-state device manufacturingChemical vapor deposition coatingMetal silicideGermanide
In one aspect, methods of silicidation and germanidation are provided. In some embodiments, methods for forming metal silicide can include forming a non-oxide interface, such as germanium or solid antimony, over exposed silicon regions of a substrate. Metal oxide is formed over the interface layer. Annealing and reducing causes metal from the metal oxide to react with the underlying silicon and form metal silicide. Additionally, metal germanide can be formed by reduction of metal oxide over germanium, whether or not any underlying silicon is also silicided. In other embodiments, nickel is deposited directly and an interface layer is not used. In another aspect, methods of depositing nickel thin films by vapor phase deposition processes are provided. In some embodiments, nickel thin films are deposited by ALD.
Owner:ASM INTERNATIONAL
Selective deposition
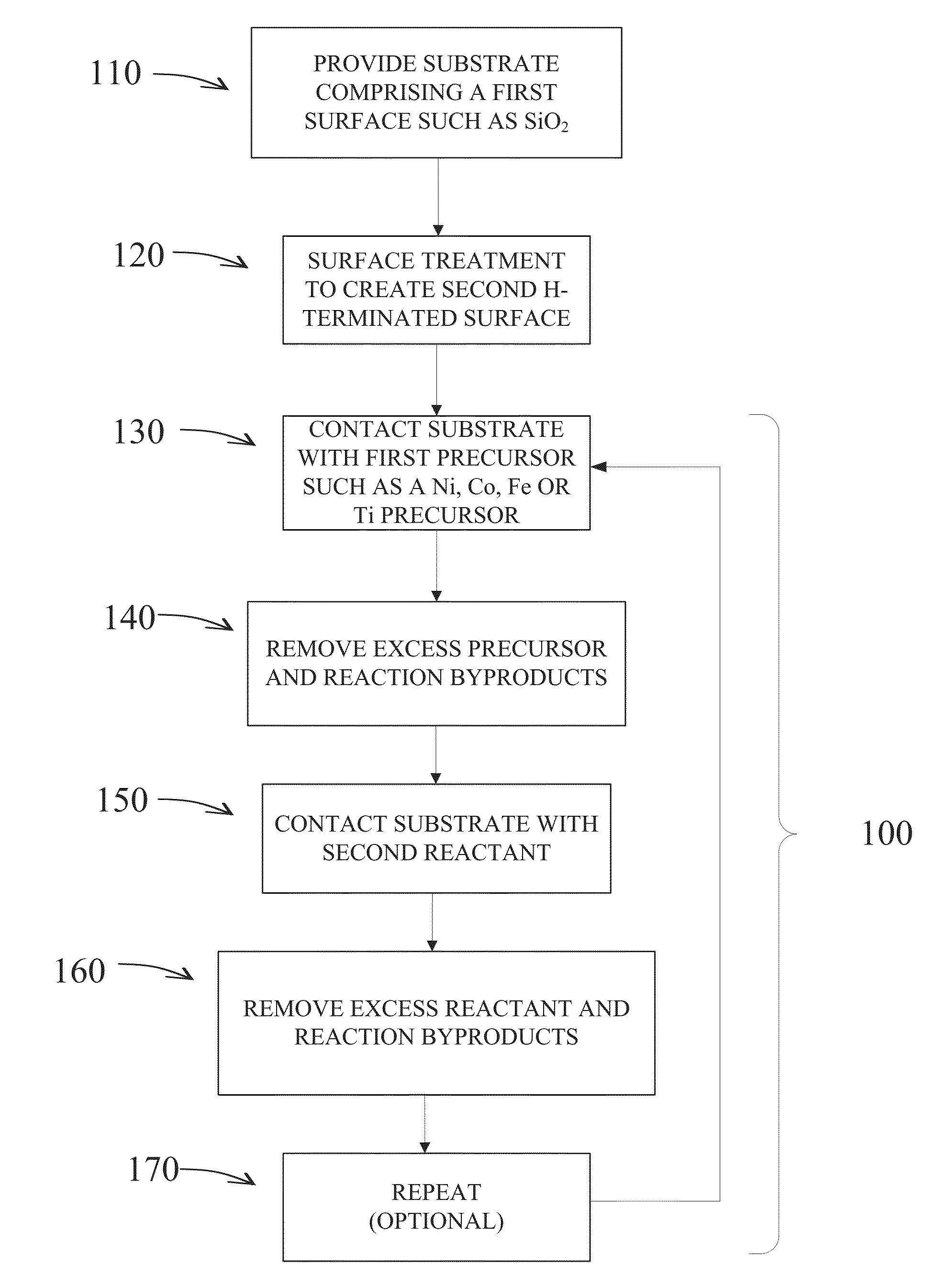
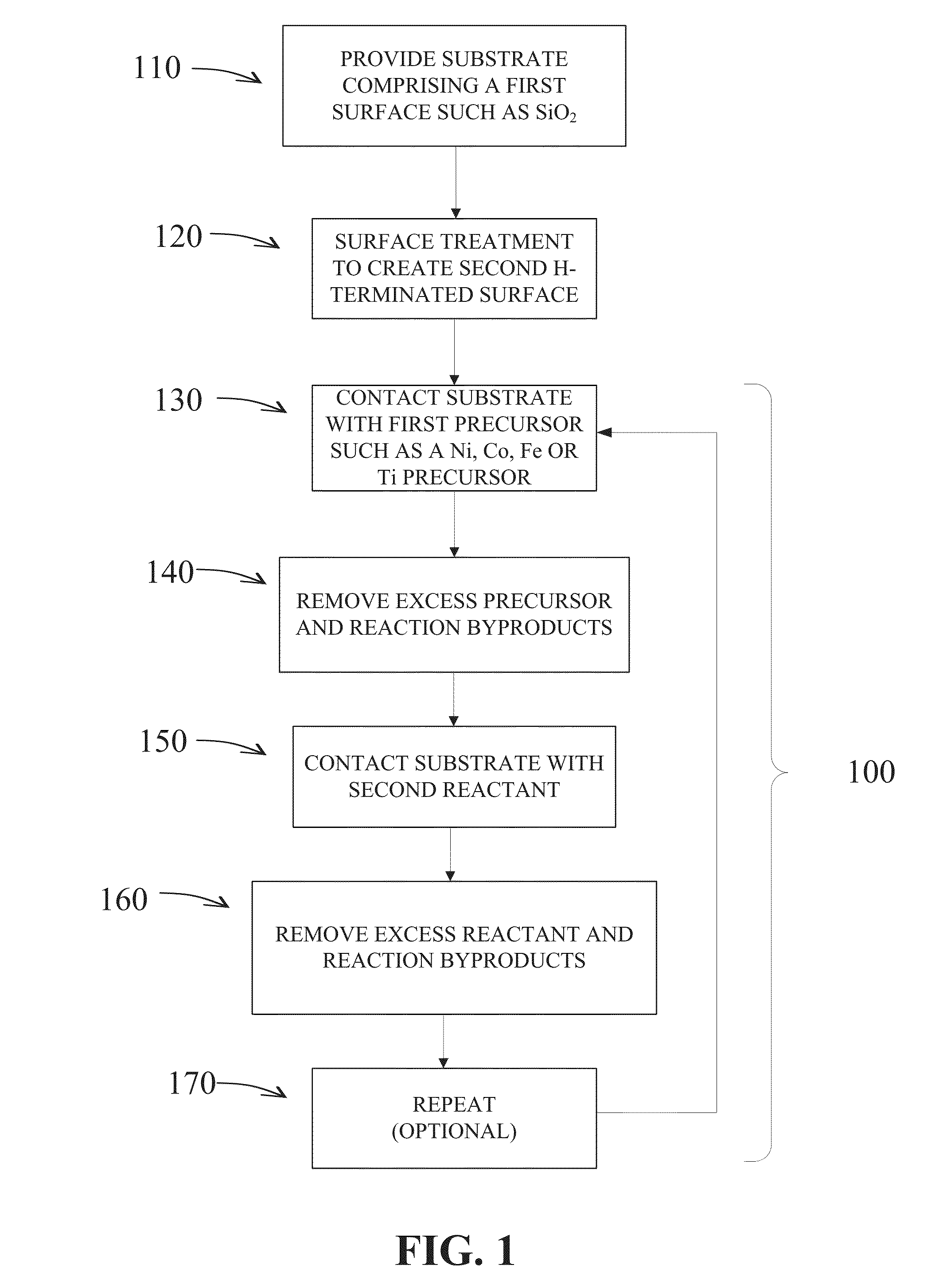
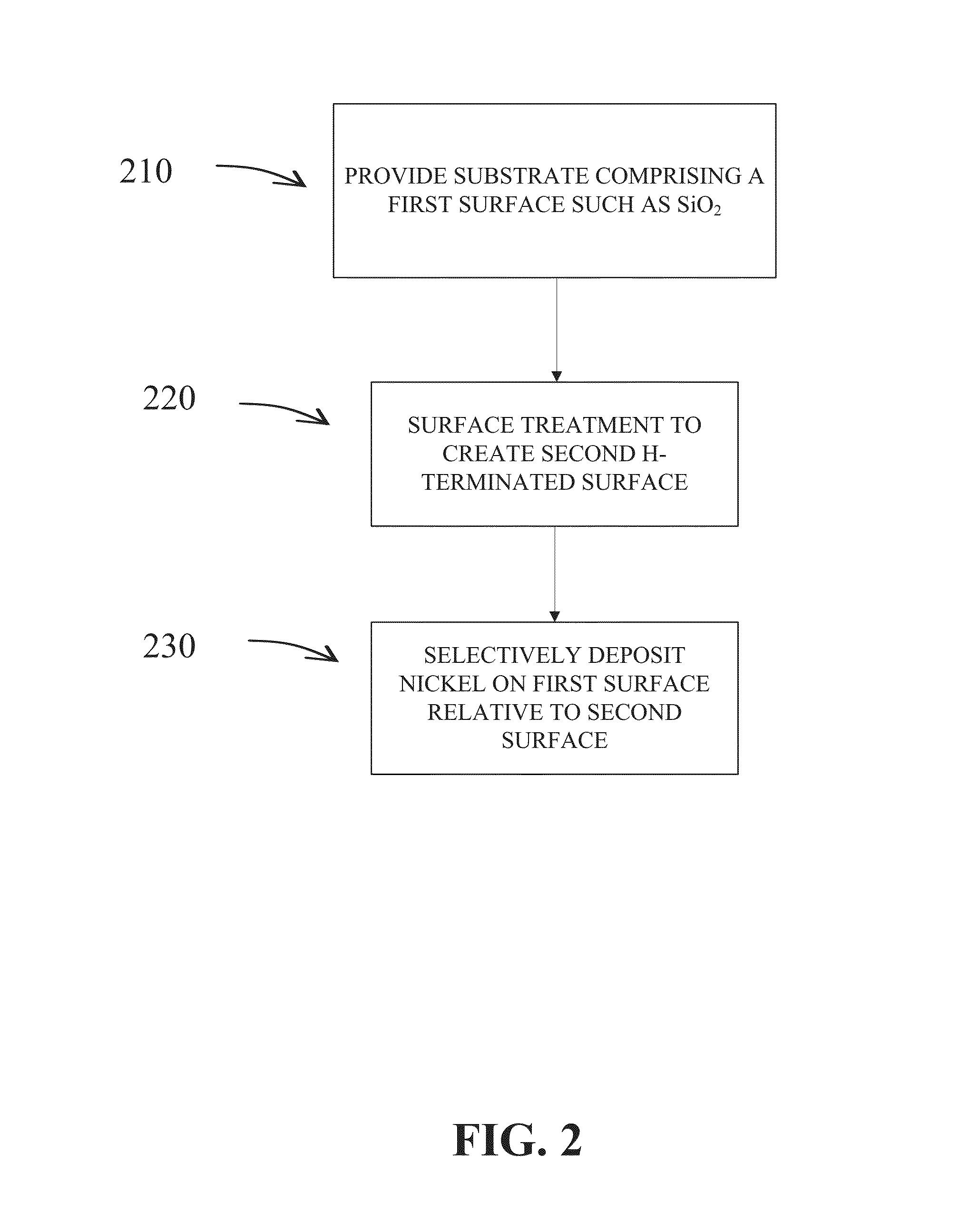
Methods are provided for selectively depositing a surface of a substrate relative to a second, different surface. An exemplary deposition method can include selectively depositing a material, such as a material comprising nickel, nickel nitride, cobalt, iron, and / or titanium oxide on a first surface, such as a SiO2 surface, relative to a second, different surface, such as a H-terminated surface, of the same substrate. Methods can include treating a surface of the substrate to provide H-terminations prior to deposition.
Owner:ASM IP HLDG BV
Alignable diffractive pigment flakes
Diffractive pigment flakes are selectively aligned to form an image. In one embodiment, flakes having a magnetic layer are shaped to facilitate alignment in a magnetic field. In another embodiment, the flakes include a magnetically discontinuous layer. In a particular embodiment, deposition of nickel on a diffraction grating pattern produces magnetic needles along the grating pattern that allow magnetic alignment of the resulting diffractive pigment flakes. Color scans of test samples of magnetically aligned flakes show high differentiation between illumination parallel and perpendicular to the direction of alignment of the magnetic diffractive pigment flakes.
Owner:VIAVI SOLUTIONS INC
Electroless deposition process on a silicon contact
ActiveUS20060264043A1Material nanotechnologySemiconductor/solid-state device manufacturingSalicideAlloy
Embodiments as described herein provide methods for depositing a material on a substrate during electroless deposition processes, as well as compositions of the electroless deposition solutions. In one embodiment, the substrate contains a contact aperture having an exposed silicon contact surface. In another embodiment, the substrate contains a contact aperture having an exposed silicide contact surface. The apertures are filled with a metal contact material by exposing the substrate to an electroless deposition process. The metal contact material may contain a cobalt material, a nickel material, or alloys thereof. Prior to filling the apertures, the substrate may be exposed to a variety of pretreatment processes, such as preclean processes and activations processes. A preclean process may remove organic residues, native oxides, and other contaminants during a wet clean process or a plasma etch process. Embodiments of the process also provide the deposition of additional layers, such as a capping layer.
Owner:APPLIED MATERIALS INC
Electroless deposition processes and compositions for forming interconnects
InactiveUS20060252252A1Material nanotechnologySemiconductor/solid-state device detailsHydrogen fluorideTungstate
In one embodiment, a method for depositing a material on a substrate is provided which includes positioning a substrate containing a contact within a process chamber, exposing the substrate to at least one pretreatment step and depositing a fill the contact vias by an electroless deposition process. The pretreatment step contains multiple processes for exposing the substrate to a wet-clean solution, a hydrogen fluoride solution, a tungstate solution, a palladium activation solution, an acidic rinse solution, a complexing agent solution or combinations thereof. Generally, the HARC via contains a tungsten oxide surface and the shallow contact via may contain a tungsten silicide surface. In some example, the substrate is pretreated such that both vias are filled at substantially the same time by a nickel-containing material through an electroless deposition process.
Owner:APPLIED MATERIALS INC
Ni film forming method
InactiveUS20120183689A1Semiconductor/solid-state device manufacturingChemical vapor deposition coatingNickelNitrogen
Owner:TOKYO ELECTRON LTD
Methods for forming a metallic film on a substrate by a cyclical deposition and related semiconductor device structures
ActiveUS20190252196A1Semiconductor/solid-state device manufacturingChemical vapor deposition coatingHydrazine compoundOptoelectronics
Methods for forming a metallic film on a substrate by cyclical deposition are provided. In some embodiments methods may include contacting the substrate with a first reactant comprising a non-halogen containing metal precursor comprising at least one of copper, nickel or cobalt and contacting the substrate with a second reactant comprising a hydrocarbon substituted hydrazine. In some embodiments related semiconductor device structures may include at least a portion of a metallic interconnect formed by cyclical deposition processes.
Owner:ASM IP HLDG BV
Electro-polishing fixture and electrolyte solution for polishing stents and method
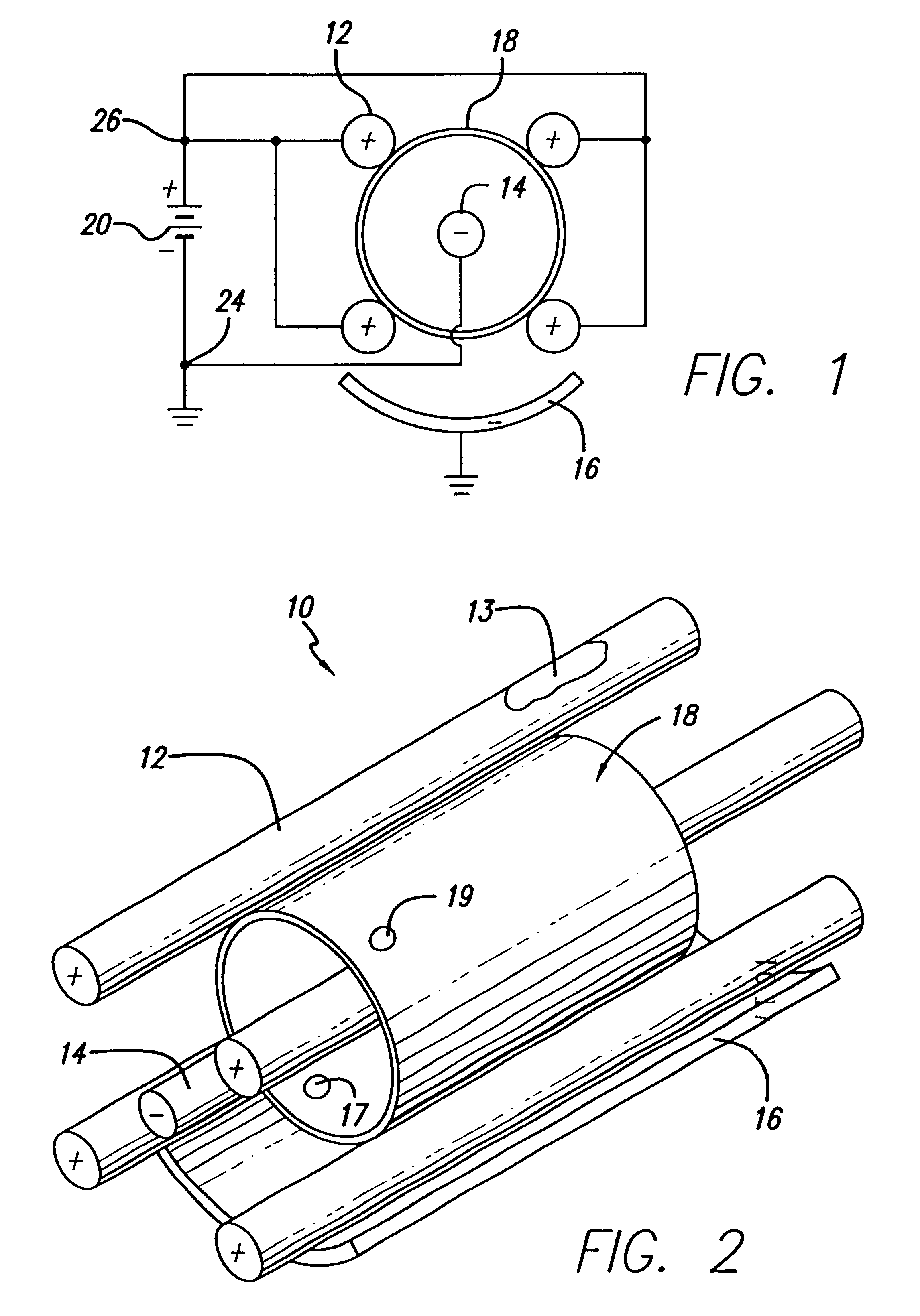
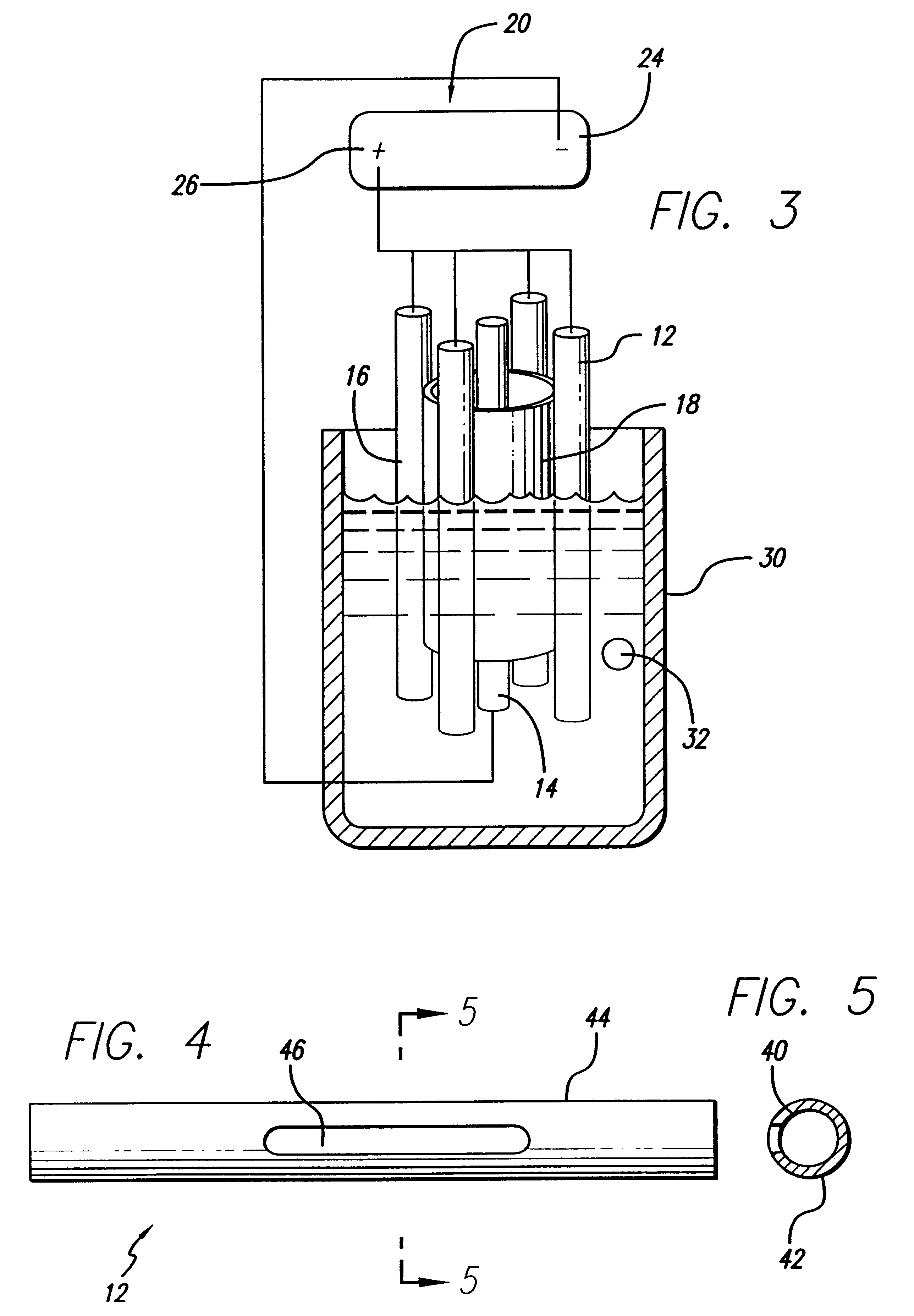
An electro-polishing fixture for polishing stents which incorporates multiple anodes in contact with the stent and a center cathode disposed coaxially within the interior of the stent and a curved exterior cathode disposed about the perimeter of the stent. The invention further includes an electrolyte solution adapted for polishing stents composed of nickel-titanium alloy and a method of using the electrolyte in combination with the electro-polishing fixture.
Owner:ABBOTT CARDIOVASCULAR
Method for depositing a ruthenium-containing film on a substrate by a cyclical deposition process
A method for depositing a ruthenium-containing film on a substrate by a cyclical deposition process is disclosed. The method may include: contacting the substrate with a first vapor phase reactant comprising a metalorganic precursor, the metalorganic precursor comprising a metal selected from the group consisting of a cobalt, nickel, tungsten, molybdenum, manganese, iron, and combinations thereof. The method may also include; contacting the substrate with a second vapor phase reactant comprising ruthenium tetroxide (RuO4); wherein the ruthenium-containing film comprises a ruthenium-metal alloy. Semiconductor device structures including ruthenium-metal alloys deposited by the methods of the disclosure are also disclosed.
Owner:ASM IP HLDG BV
Method for producing alcohols by hydrogenation of carbonyl compounds
InactiveUS6486366B1Less amountHigh strengthSugar derivativesOrganic compound preparationCobaltPt element
A method for preparation of alcohols by catalytic hydrogenation of carbonyl compounds with hydrogen or hydrogen-containing gases in the presence of a hydrogenation catalyst of Raney type, where the catalyst is used in the form of hollow bodies, Preferred as catalytically active components are nickel, cobalt, copper, iron, platinum, palladium or ruthenium.
Owner:DEGUSSA AG
Radiopaque nitinol alloys for medical devices
Owner:ABBOTT CARDIOVASCULAR
Heat and corrosion resistant cast CF8C stainless steel with improved high temperature strength and ductility
A CF8C type stainless steel alloy and articles formed therefrom containing about 18.0 weight percent to about 22.0 weight percent chromium and 11.0 weight percent to about 14.0 weight percent nickel; from about 0.05 weight percent to about 0.15 weight percent carbon; from about 2.0 weight percent to about 10.0 weight percent manganese; and from about 0.3 weight percent to about 1.5 weight percent niobium. The present alloys further include less than 0.15 weight percent sulfur which provides high temperature strength both in the matrix and at the grain boundaries without reducing ductility due to cracking along boundaries with continuous or nearly-continuous carbides. The disclosed alloys also have increased nitrogen solubility thereby enhancing strength at all temperatures because nitride precipitates or nitrogen porosity during casting are not observed. The solubility of nitrogen is dramatically enhanced by the presence of manganese, which also retains or improves the solubility of carbon thereby providing additional solid solution strengthening due to the presence of manganese and nitrogen, and combined carbon.
Owner:UT BATTELLE LLC
Slag oil hydro-demetallization catalyst and its preparing method
ActiveCN1782031AImprove performanceEasy to operateRefining to eliminate hetero atomsAluminum CarbonateAlumina hydrate
The slag oil hydrodemetallizing catalyst contains a kind of double-peak hole alumina carrier and supported Mo and / or W and Ni and / or Co metal components. The double-peak hole alumina carrier has pore volume of 0.8-1.6 ml / g, specific surface area 150-350 sq m / g, pore volume fraction of 10-30 nm size holes in 40-90 % and pore volume fraction of 100-2000 nm size holes in 10-60 %, and is prepared through mixing alumina hydrate and ammonium aluminum carbonate, forming and roasting. The slag oil hydrodemetallizing catalyst has simple preparation process and high hydrodemetallizing performance.
Owner:CHINA PETROLEUM & CHEM CORP +1
Methods for forming through-wafer interconnects, intermediate structures so formed, and devices and systems having at least one solder dam structure
ActiveUS20070045779A1Conveniently attachedEliminate cross-contaminationSemiconductor/solid-state device detailsSolid-state devicesVitrificationDevice material
A method for forming through-wafer interconnects (TWI) in a substrate of a thickness in excess of that of a semiconductor die such as a semiconductor wafer. Blind holes are formed from the active surface, sidewalls thereof passivated and coated with a solder-wetting material. A vent hole is then formed from the opposite surface (e.g., wafer back side) to intersect the blind hole. The blind hole is solder filled, followed by back thinning of the vent hole portion of the wafer to a final substrate thickness to expose the solder and solder-wetting material at both the active surface and the thinned back side. A metal layer such as nickel, having a glass transition temperature greater than that of the solder, may be plated to form a dam structure covering one or both ends of the TWI including the solder and solder-wetting material to prevent leakage of molten solder from the TWI during high temperature excursions. Intermediate structures of semiconductor devices, semiconductor devices and systems are also disclosed.
Owner:MICRON TECH INC
Method for fabricating a semiconductor device
InactiveUS6180439B1Promote crystallizationReduce concentrationTransistorSolid-state devicesAtmospheric airLaser light
Concentration of metal element which promotes crystallization of silicon and which exists within a crystalline silicon film obtained by utilizing the metal element is reduced. A first heat treatment for crystallization is performed after introducing nickel to an amorphous silicon film 103. Then, laser light is irradiated to diffuse nickel element which is concentrated locally. After that, another heat treatment is performed within an oxidizing atmosphere at a temperature higher than that of the previous heat treatment. At this time, HCl or the like is added to the atmosphere. A thermal oxide film 106 is formed in this step. At this time, gettering of the nickel element into the thermal oxide film 106 takes place. Then, the thermal oxide film 106 is removed. Thereby, a crystalline silicon film 107 having low concentration of the metal element and a high crystallinity can be obtained.
Owner:SEMICON ENERGY LAB CO LTD
Radiopaque markers for medical devices
InactiveUS20050060025A1High level of radiopacitySufficient radiopacityStentsBlood vesselsIridiumRhenium
An implantable medical device includes a structural body made from a superelastic material and includes one or more marker holders integrally formed on the structural body. Each marker holder is designed to hold a radiopaque marker which has a level of radiopacity greater than the superelastic material. The radiopaque marker can be made from a nickel-titanium alloy which includes a ternary element. The ternary element can be selected from the group of elements consisting of iridium, platinum, gold, rhenium, tungsten, palladium, rhodium, tantalum, silver, ruthenium, and hafnium. In one form, the marker holder includes a pair of projecting fingers connected together at a notched region to cooperatively create a particular-shaped opening. This opening, in turn, is adapted to receive a similarly shaped portion formed on the radiopaque marker. In one form, the radiopaque marker includes an inner core which is partially, or completely, encased by an outer layer. This inner core can be made from a highly radiopaque material while the outer layer is formed from a material that is easier to weld to the marker.
Owner:ABBOTT VASCULAR SOLUTIONS
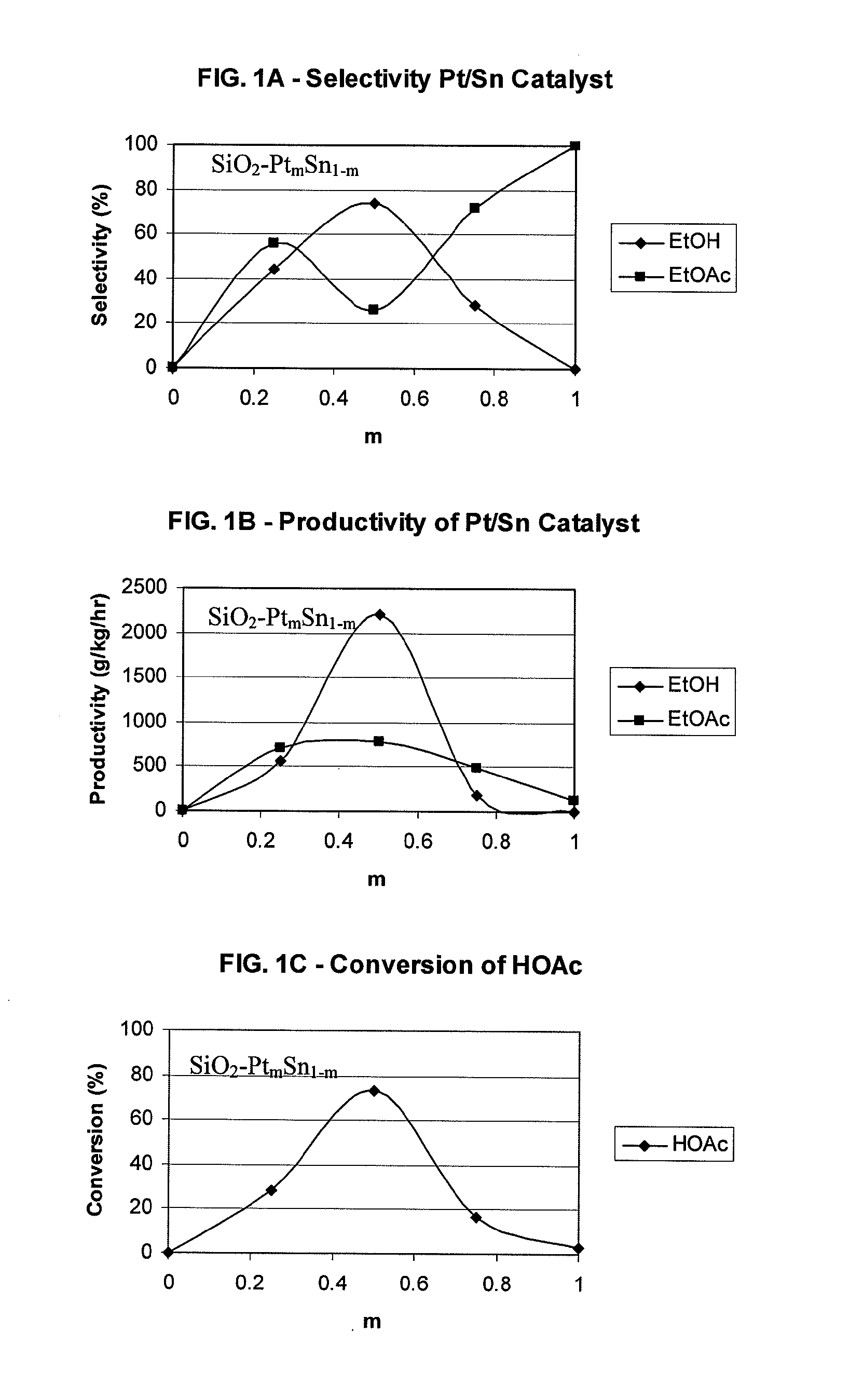
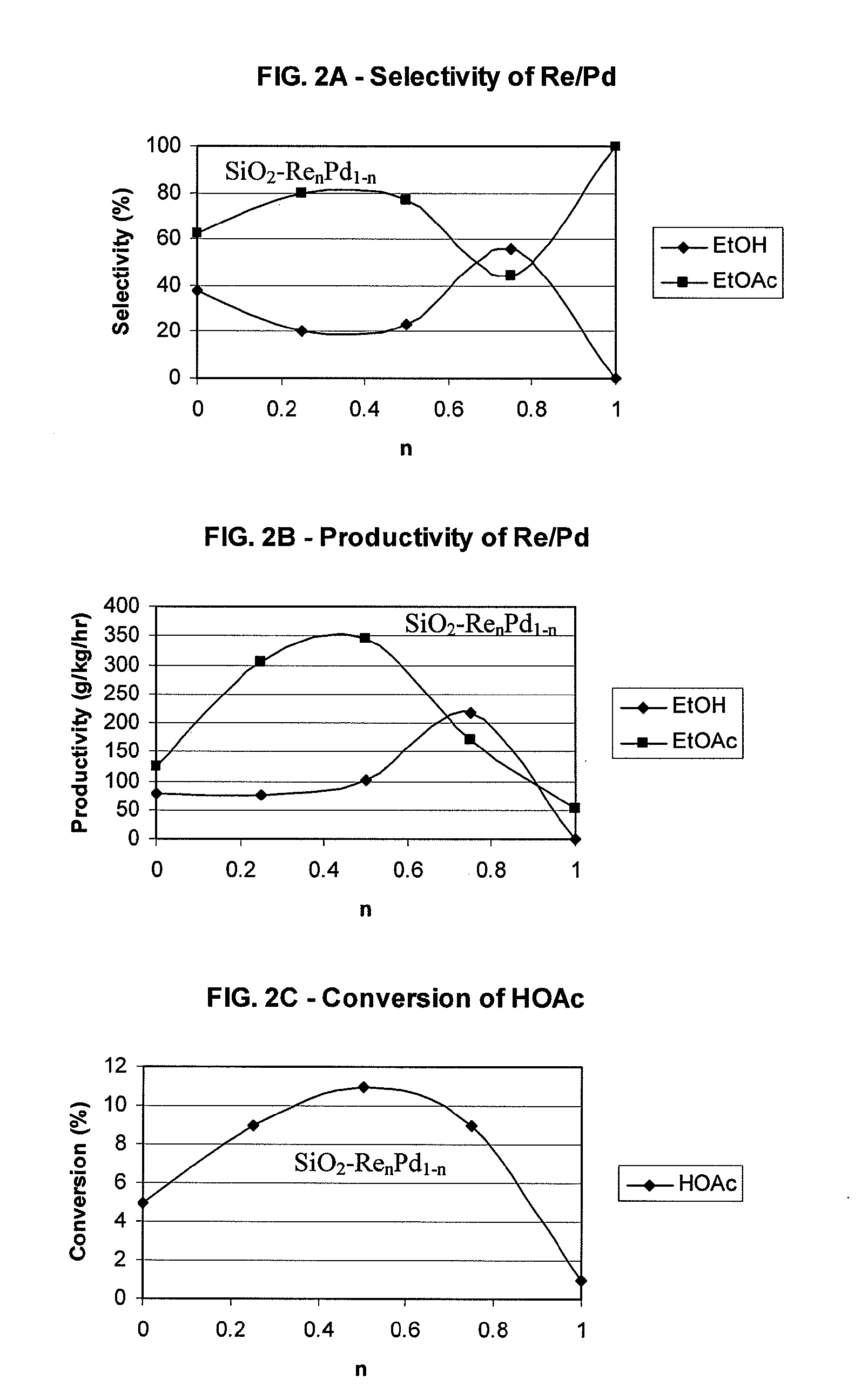
Processes for making ethanol from acetic acid
InactiveUS20100197985A1High selectivityPreparation by oxo-reaction and reductionEthylene productionCeriumCobalt
A process for selective formation of ethanol from acetic acid by hydrogenating acetic acid in the presence of first metal, a silicaceous support, and at least one support modifier. Preferably, the first metal is selected from the group consisting of copper, iron, cobalt, nickel, ruthenium, rhodium, palladium, osmium, iridium, platinum, titanium, zinc, chromium, rhenium, molybdenum, and tungsten. In addition the catalyst may comprise a second metal preferably selected from the group consisting of copper, molybdenum, tin, chromium, iron, cobalt, vanadium, tungsten, palladium, platinum, lanthanum, cerium, manganese, ruthenium, rhenium, gold, and nickel.
Owner:CELANESE INT CORP
Dimerized alcohol compositions and biodegradible surfactants made therefrom having cold water detergency
InactiveUS6222077B1Good cold water detergencyOrganic detergent compounding agentsOther chemical processesDouble bondCarboxylic acid
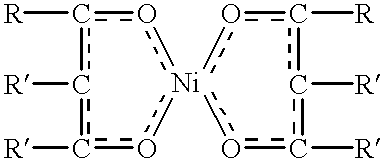
There is provided an alcohol composition obtained by dimerizing an olefin feed comprising C6-C10 linear olefins to obtain C12-C20 olefins, followed by conversion to alcohols, such as by hydroformylation. The composition has an average number of branches ranging from 0.9 to 2.0 per molecule. The linear olefin feed preferably comprises at least 85% of C6-C8-olefins. The primary alcohol compositions are then converted to anionic or nonionic surfactants, preferably sulfated or oxyalkylated or both. The sulfated compositions are biodegradable and possess good cold water detergency. The process for making the dimerized primary alcohol comprises dimerizing, in the presence of a homogeneous dimerization catalyst under dimerization conditions, an olefin feed comprising C6-C10 olefins and preferably at least 85 weight % of linear olefins based on the weight of the olefin feed, to obtain a C12-C20; optionally double bond isomerizing said C12-C20 olefins; and converting the C12-C20 olefins to alcohols, preferably through hydroformylation. The process is preferably a one-step dimerization. The homogenous catalyst comprises a mixture of a nickel carboxylate or a nickel chelate, with an alkyl aluminum halide or an alkyl aluminum alkoxide.
Owner:SHELL OIL CO
Electronic assembly having a wetting layer on a thermally conductive heat spreader
InactiveUS6504242B1Maintain temperatureSemiconductor/solid-state device detailsSolid-state devicesIndiumHeat cycling
An electronic assembly including a die, having an integrated circuit formed therein, a thermally conductive heat spreader, and indium located between the die and the heat spreader, is described. The heat spreader has a layer of nickel. A gold layer is formed on the nickel layer, and provides better wetting of indium than nickel. A better structural connection between the indium and the heat spreader is provided, especially during thermal cycling.
Owner:INTEL CORP
Method of manufacturing a semiconductor device
InactiveUS6479333B1TransistorSemiconductor/solid-state device manufacturingCrystallinityCrystal growth
A crystal growth 301 is carried out by diffusing a metal element, and a nickel element is moved into regions 108 and 109 which has been doped with phosphorus. An axis coincident with the moving directions 302 and 303 of the nickel element at this time is made to coincide with an axis coincident with the direction of the crystal growth, and a TFT having the regions as channel forming regions is manufactured. In the path of the region where nickel moved, since high crystallinity is obtained in the moving direction, the TFT having high characteristics can be obtained by this way.
Owner:SEMICON ENERGY LAB CO LTD
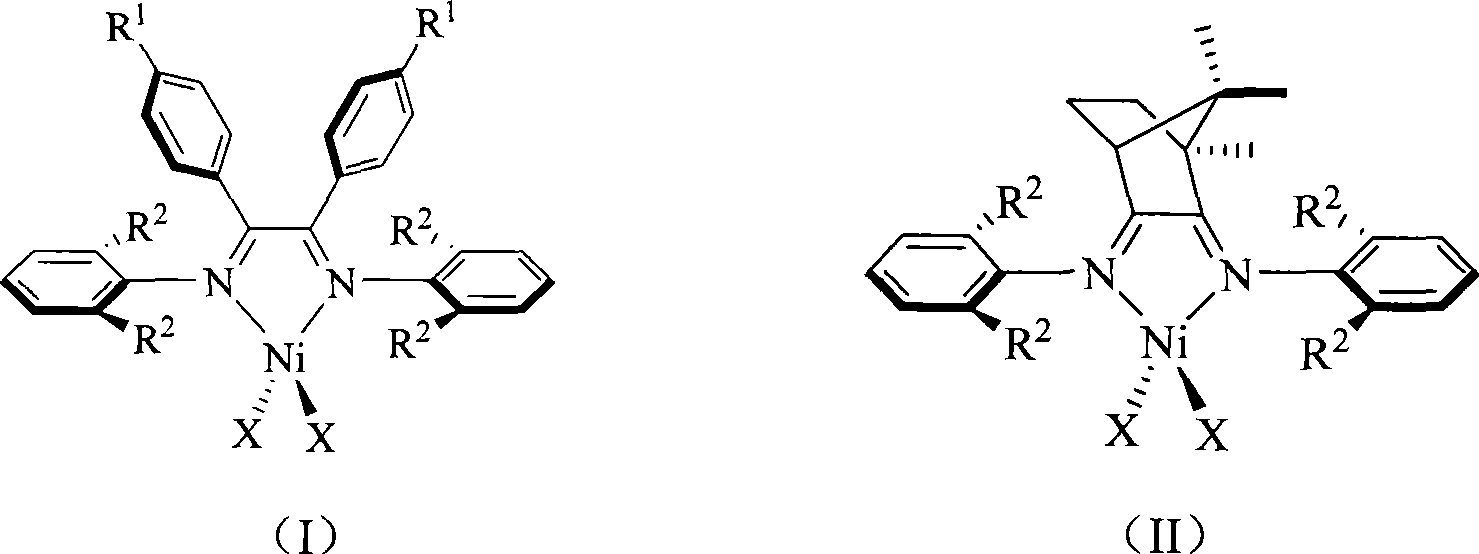
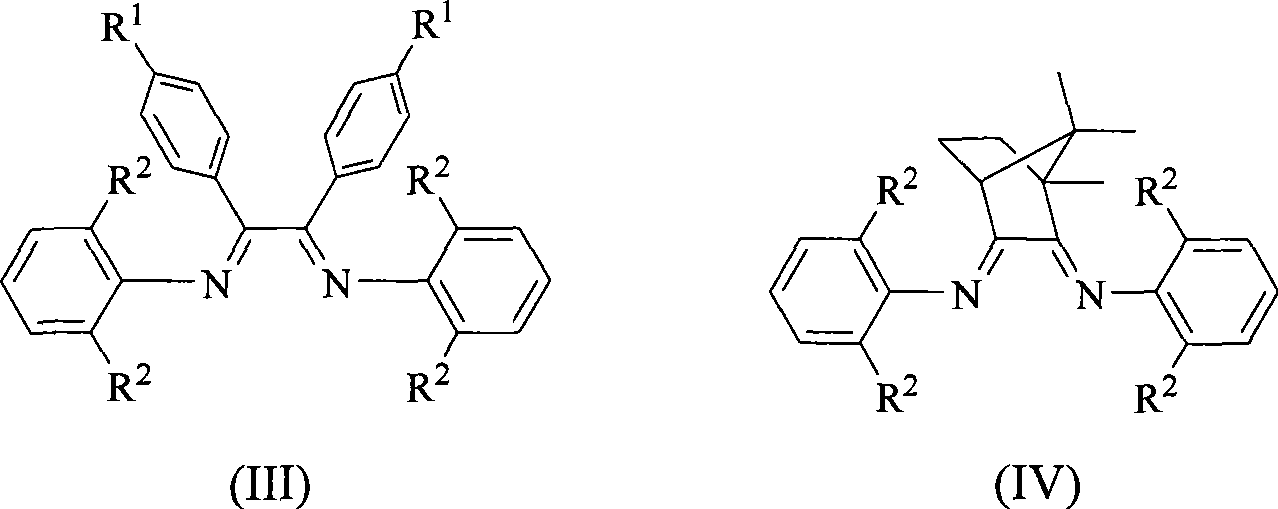
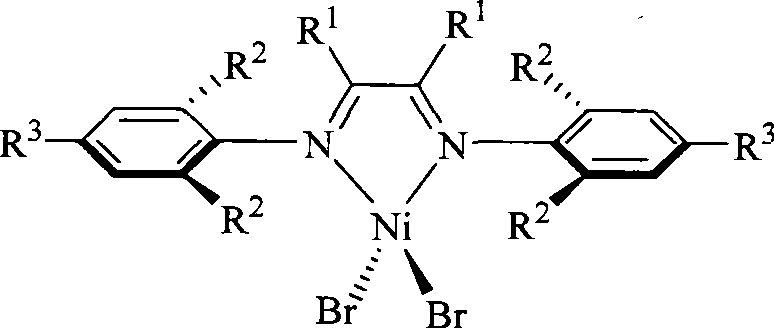
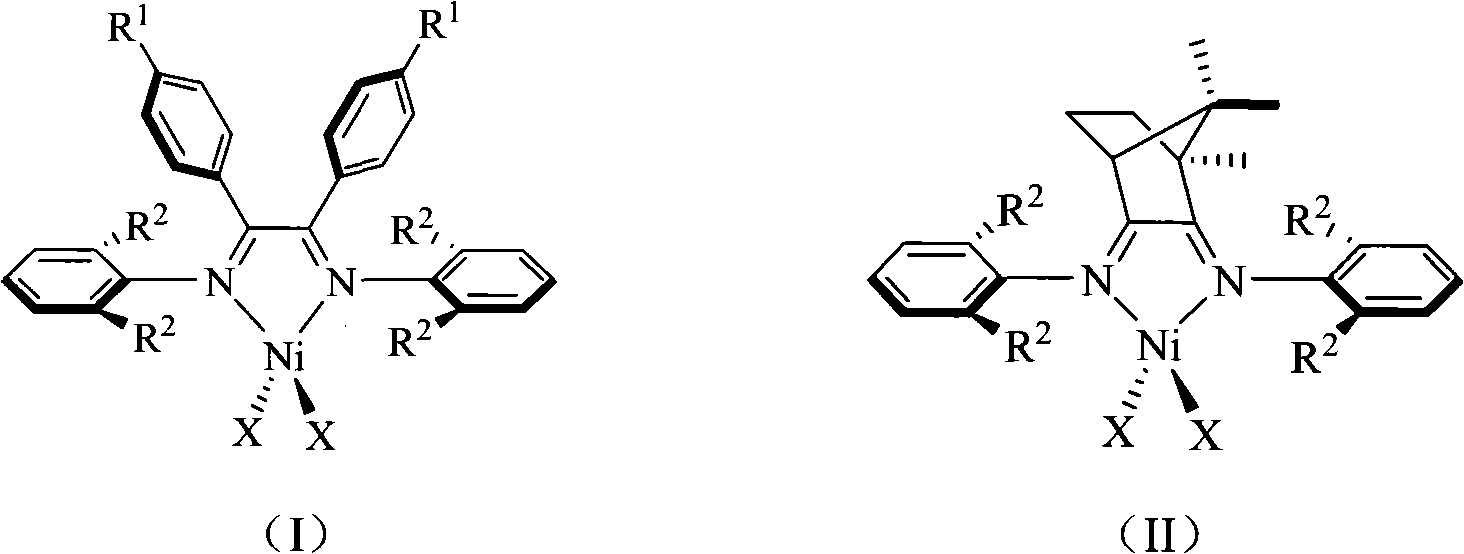
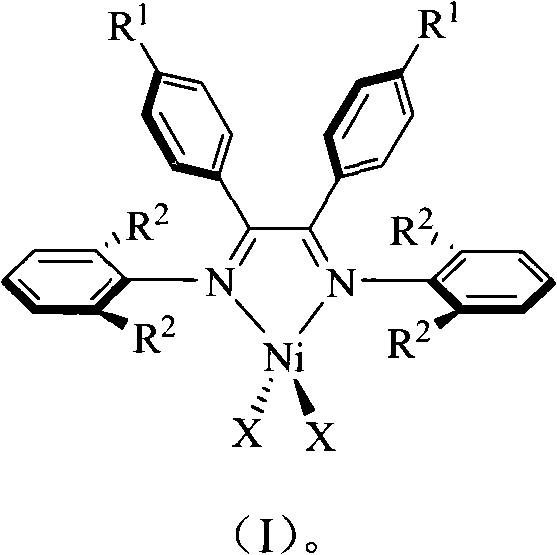
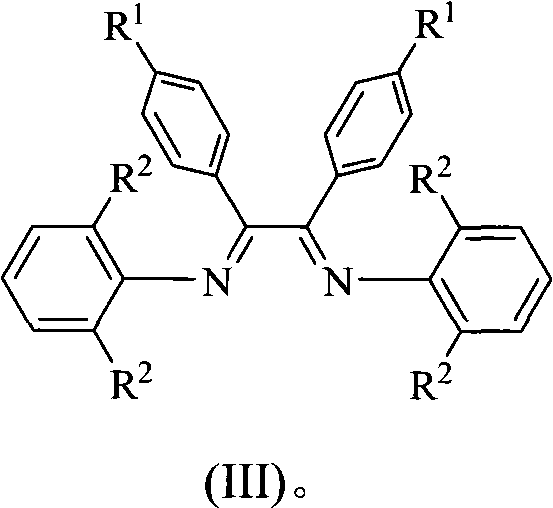
Alpha-nickel diimine compound olefin polymerization catalyst and preparation method thereof, and method for preparing branched polyethylene
The invention discloses an alpha-nickel diimine compound olefin polymerization catalyst and a preparation method thereof, and a method for preparing branched polyethylene. Structural formulas of the alpha-nickel diimine compound olefin polymerization catalyst are shown as a formula (I) and a formula (II), and the preparation method of the alpha-nickel diimine compound olefin polymerization catalyst is simple and low in cost, and can catalyze ethylene polymerization with high activity at a temperature of more than or equal to 80 DEG C to obtain high molecular weight polyethylene. The polyethylene prepared by catalyzing of the alpha-nickel diimine compound olefin polymerization catalyst has high molecular weight, and can prepare molecular weight which reaches more than 10<2>*kg / mol in the temperature range of between 0 and 80 DEG C.
Owner:SUN YAT SEN UNIV
Catalyst for manufacturing hydrogen or synthesis gas and manufacturing method of hydrogen or synthesis gas
InactiveUS6361757B1Produce hydrogenEfficient productionIron compoundsCobalt compoundsIridiumForming gas
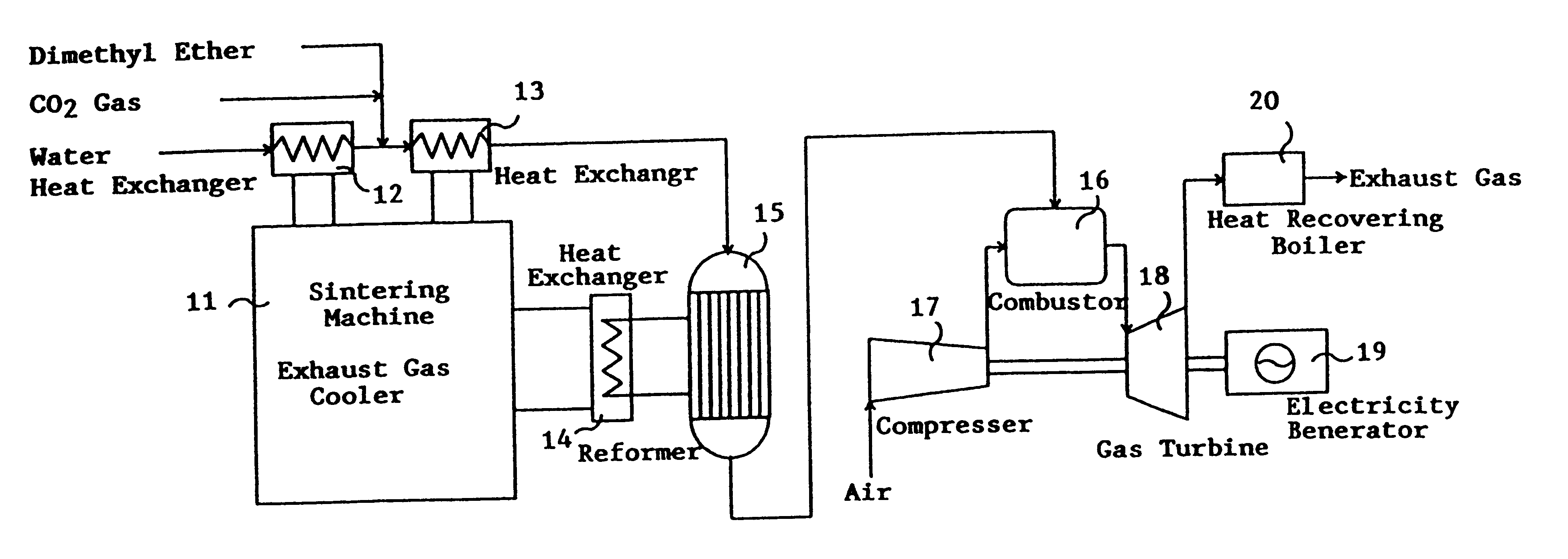
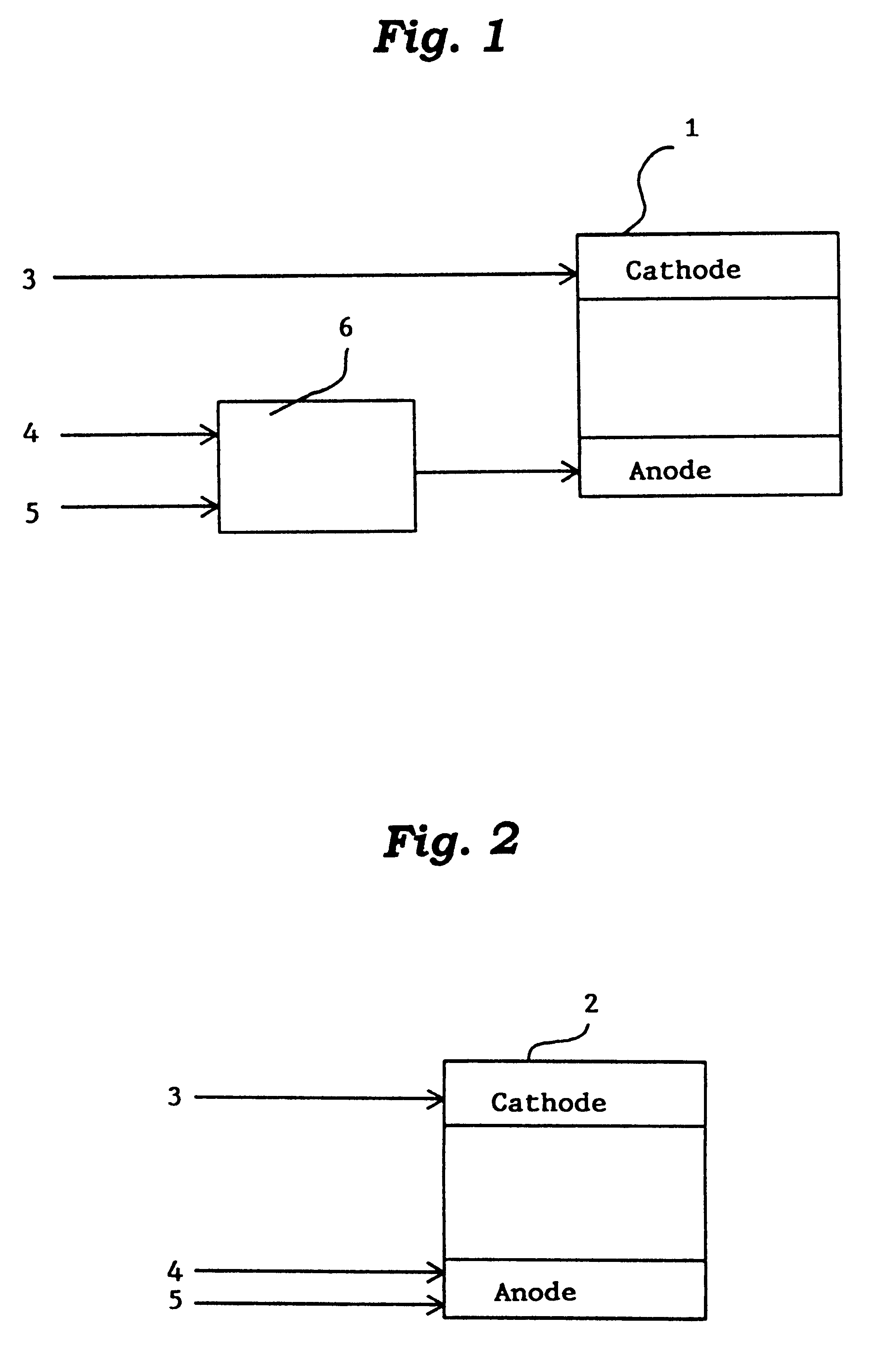
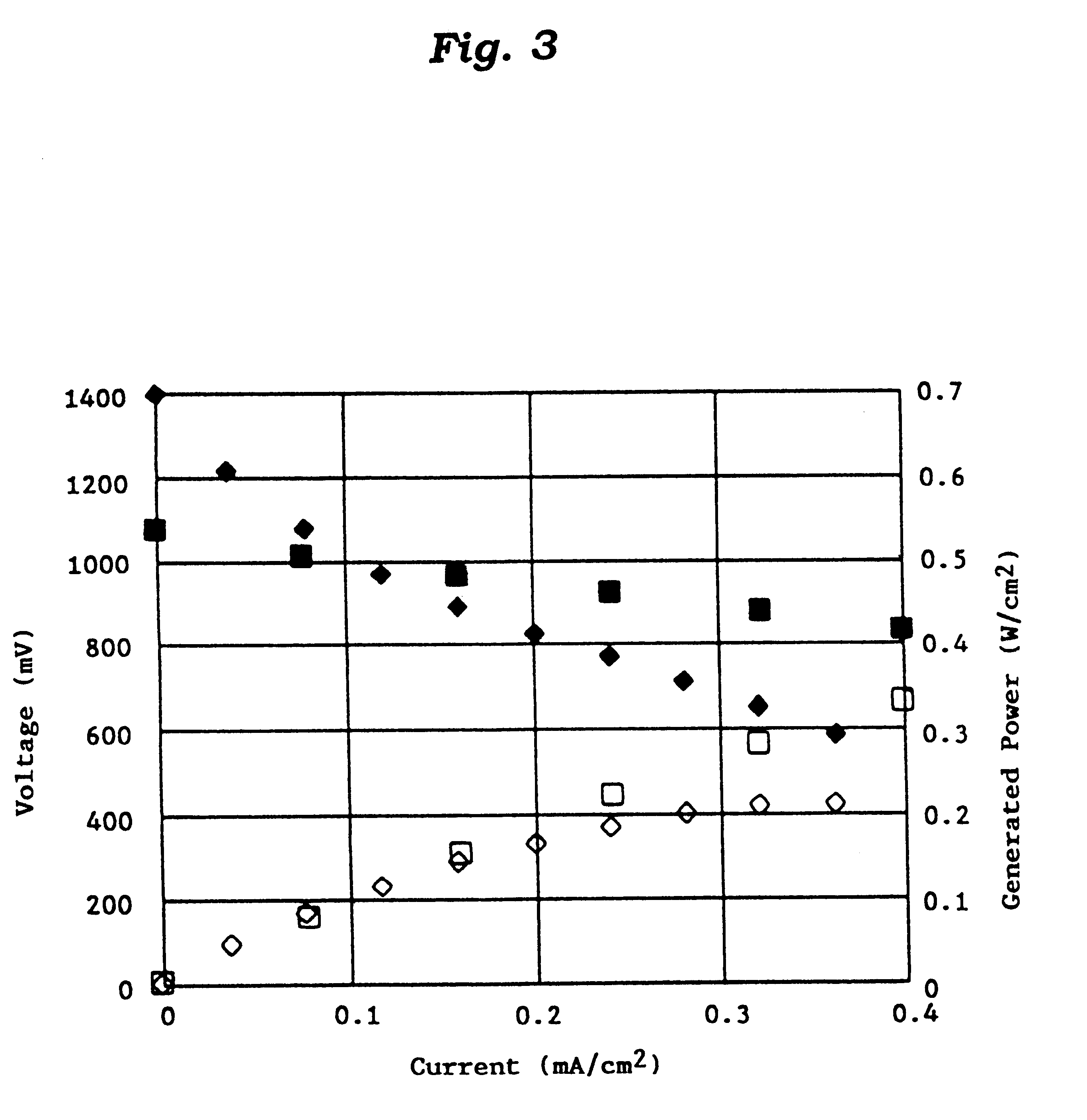
This invention provides a catalyst for producing hydrogen gas from a mixed gas comprising dimethyl ether and water vapor or carbon dioxide gas, which comprises copper, iron, cobalt, palladium, iridium, platinum, rhodium, or nickel as an active component, and a method of producing synthesis gas or hydrogen gas in a high yield at a low temperature. By using the catalyst, a fuel cell, electricity generation, reduction of iron ore and the like can be carried out.
Owner:NIPPON KOKAN KK
Inorganic dopants, inks and related nanotechnology
InactiveUS6849109B2Facilitated DiffusionLower transition temperatureSelenium/tellurium compundsCell electrodesIndiumCerium
Ink compositions with modified properties result from using a powder size below 100 nanometers. Colored inks are illustrated. Nanoscale coated, uncoated, whisker inorganic fillers are included. The pigment nanopowders taught comprise one or more elements from the group actinium, aluminum, antimony, arsenic, barium, beryllium, bismuth, cadmuim, calcium, cerium, cesium, chalcogenide, cobalt, copper, dysprosium, erbium, europium, gadolinium, gallium, gold, hafnium, hydrogen, indium, iridium, iron, lanthanum, lithium, magnesium, manganese, mendelevium, mercury, molybdenum, neodymium, neptunium, nickel, niobium, nitrogen, oxygen, osmium, palladium, platinum, potassium, praseodymium, promethium, protactinium, rhenium, rubidium, scandium, silver, sodium, strontium, tantalum, terbium, thallium, thorium, tin, titanium, tungsten, vanadium, ytterbium, yttrium, zinc, and zirconium.
Owner:PPG IND OHIO INC
Corrosion resistant component of semiconductor processing equipment and method of manufacturing thereof
A corrosion resistant component of semiconductor processing equipment such as a plasma chamber includes a metal surface such as aluminum or aluminum alloy, stainless steel, or refractory metal coated with a phosphorus nickel plating and an outer ceramic coating such as alumina, silicon carbide, silicon nitride, boron carbide or aluminum nitride. The phosphorus nickel plating can be deposited by electroless plating and the ceramic coating can be deposited by thermal spraying. To promote adhesion of the ceramic coating, the phosphorus nickel plating can be subjected to a surface roughening treatment prior to depositing the ceramic coating.
Owner:LAM RES CORP
Zinc Ion-Exchanging Energy Storage Device
ActiveUS20160301096A1Quick releaseRapid depositionHybrid capacitor electrolytesAlkaline accumulatorsChemical treatmentZinc metal
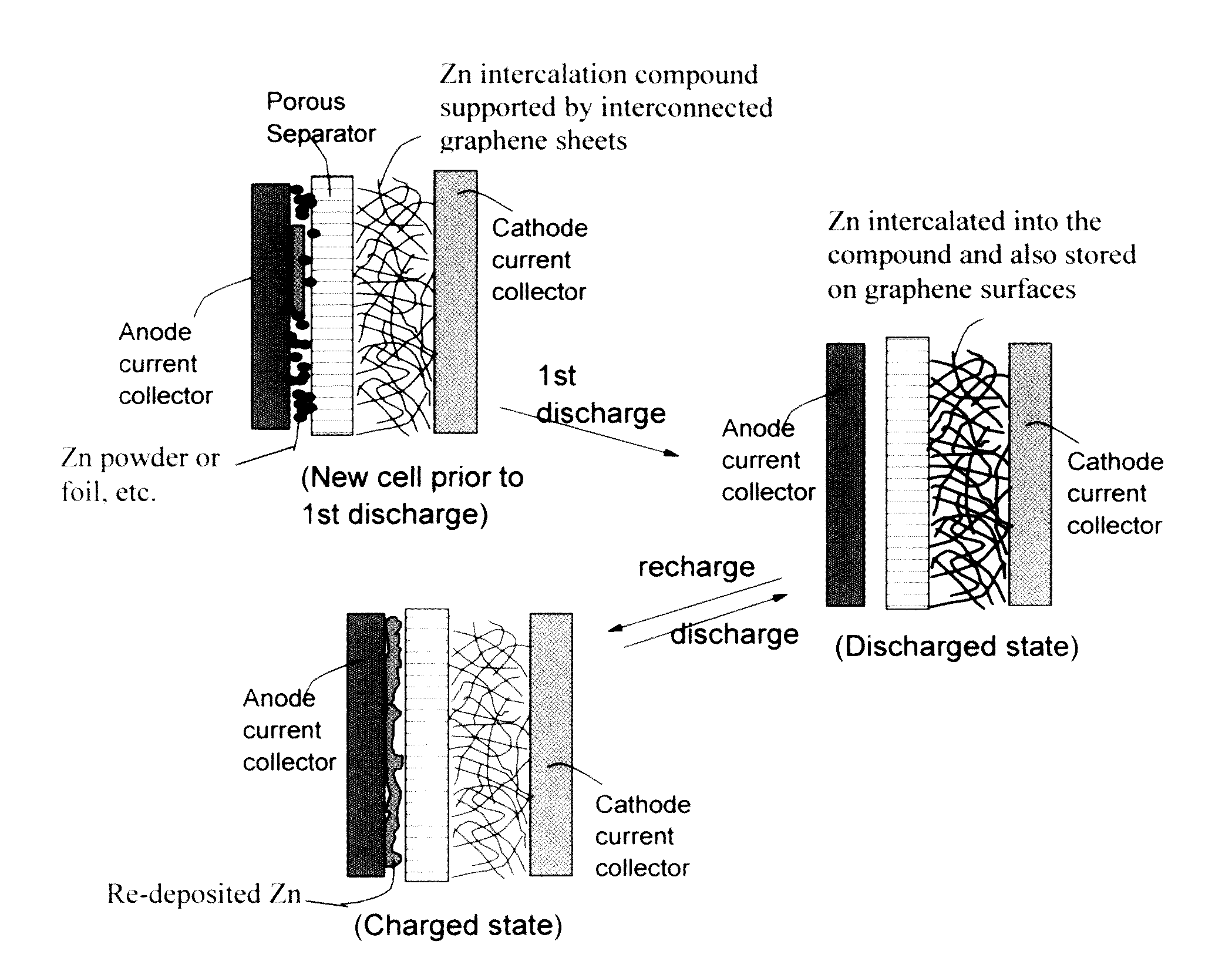
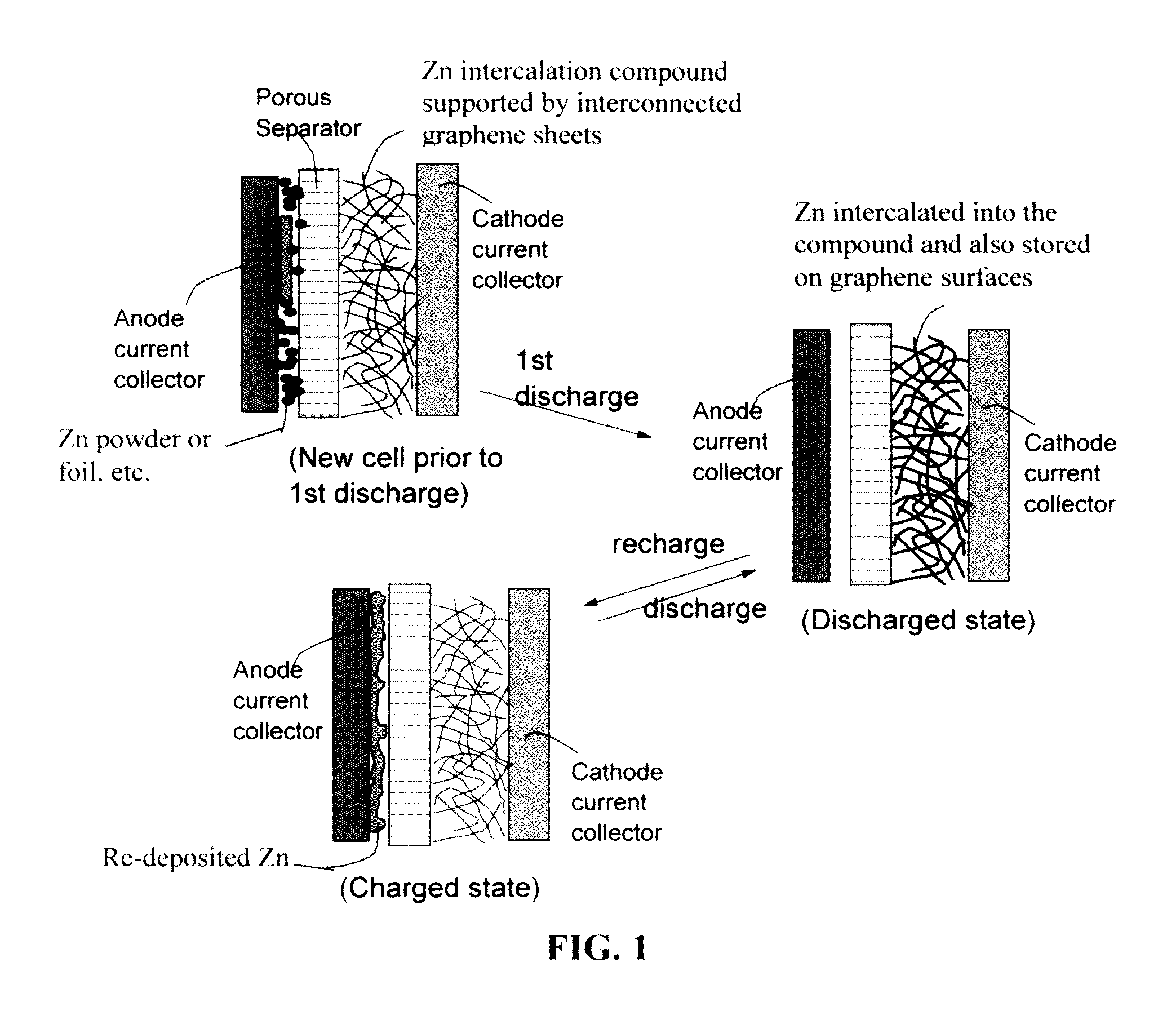
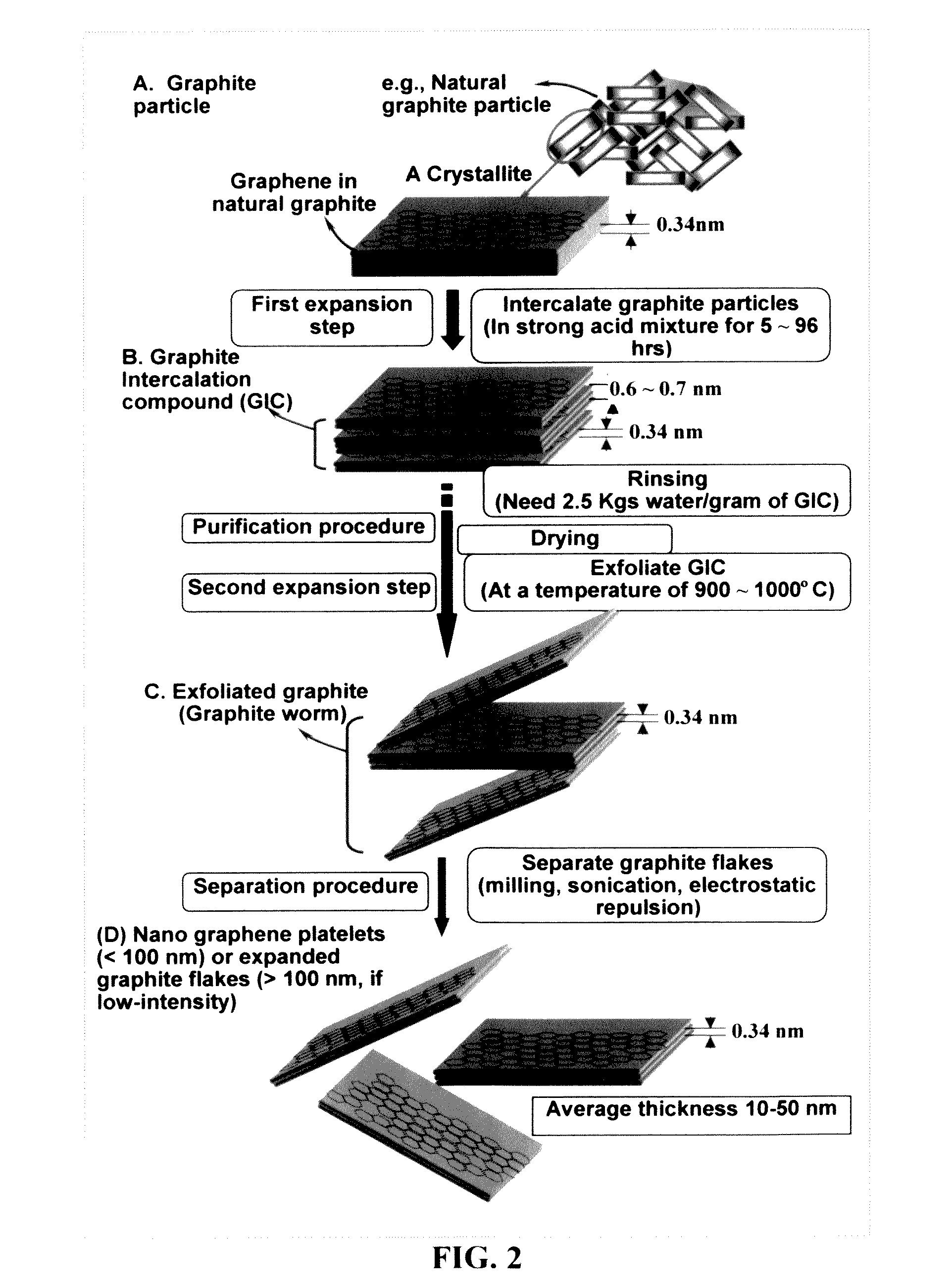
A zinc ion-exchanging battery device comprising: (A) a cathode comprising two cathode active materials (a zinc ion intercalation compound and a surface-mediating material); (B) an anode containing zinc metal or zinc alloy; (C) a porous separator disposed between the cathode and the anode; and (D) an electrolyte containing zinc ions that are exchanged between the cathode and the anode during battery charge / discharge. The zinc ion intercalation compound is selected from chemically treated carbon or graphite material having an expanded inter-graphene spacing d002 of at least 0.5 nm, or an oxide, carbide, dichalcogenide, trichalcogenide, sulfide, selenide, or telluride of niobium, zirconium, molybdenum, hafnium, tantalum, tungsten, titanium, vanadium, chromium, cobalt, manganese, iron, nickel, or a combination thereof. The surface-mediating material contains exfoliated graphite or multiple single-layer sheets or multi-layer platelets of a graphene material.
Owner:GLOBAL GRAPHENE GRP INC
Alpha-nickel diimine compound olefin polymerization catalyst, preparation method and method for preparing branched polyethylene
InactiveCN101812145AEasy to prepareLow costNickel organic compoundsBulk chemical productionPolymer scienceDiimine
The invention discloses an alpha-nickel diimine compound olefin polymerization catalyst, a preparation method and a method for preparing branched polyethylene. The structural formula of the alpha-nickel diimine compound olefin polymerization catalyst is shown as the formula (I) and the formula (II). The alpha-nickel diimine compound olefin polymerization catalyst has simple preparation method and low cost, and ethylene can be catalyzed to polymerize with high activity at a temperature not lower than 80 DEG C to obtain high molecular weight polyethylene. The polyethylene prepared from the alpha-nickel diimine compound olefin polymerization catalyst by catalyzing has high molecular weight, and the molecular weight can reach higher than 102x kg / mol within the range of 0-80 DEG C.
Owner:SUN YAT SEN UNIV