Patents
Literature
2925results about "Iron compounds" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Ternary oxide nanostructures and methods of making same
InactiveUS20070138459A1Suitable for preparationFrom gel stateAlkaline earth titanatesNanostructureDislocation
A single crystalline ternary nanostructure having the formula AxByOz, wherein x ranges from 0.25 to 24, and y ranges from 1.5 to 40, and wherein A and B are independently selected from the group consisting of Ag, Al, As, Au, B, Ba, Br, Ca, Cd, Ce, Cl, Cm, Co, Cr, Cs, Cu, Dy, Er, Eu, F, Fe, Ga, Gd, Ge, Hf, Ho, I, In, Ir, K, La, Li, Lu, Mg, Mn, Mo, Na, Nb, Nd, Ni, Os, P, Pb, Pd, Pr, Pt, Rb, Re, Rh, Ru, S, Sb, Sc, Se, Si, Sm, Sn, Sr, Ta, Tb, Tc, Te, Ti, Ti, Tm, U, V, W, Y, Yb, and Zn, wherein the nanostructure is at least 95% free of defects and / or dislocations.
Owner:THE RES FOUND OF STATE UNIV OF NEW YORK
Catalyst for manufacturing hydrogen or synthesis gas and manufacturing method of hydrogen or synthesis gas
InactiveUS6361757B1Produce hydrogenEfficient productionIron compoundsCobalt compoundsIridiumForming gas
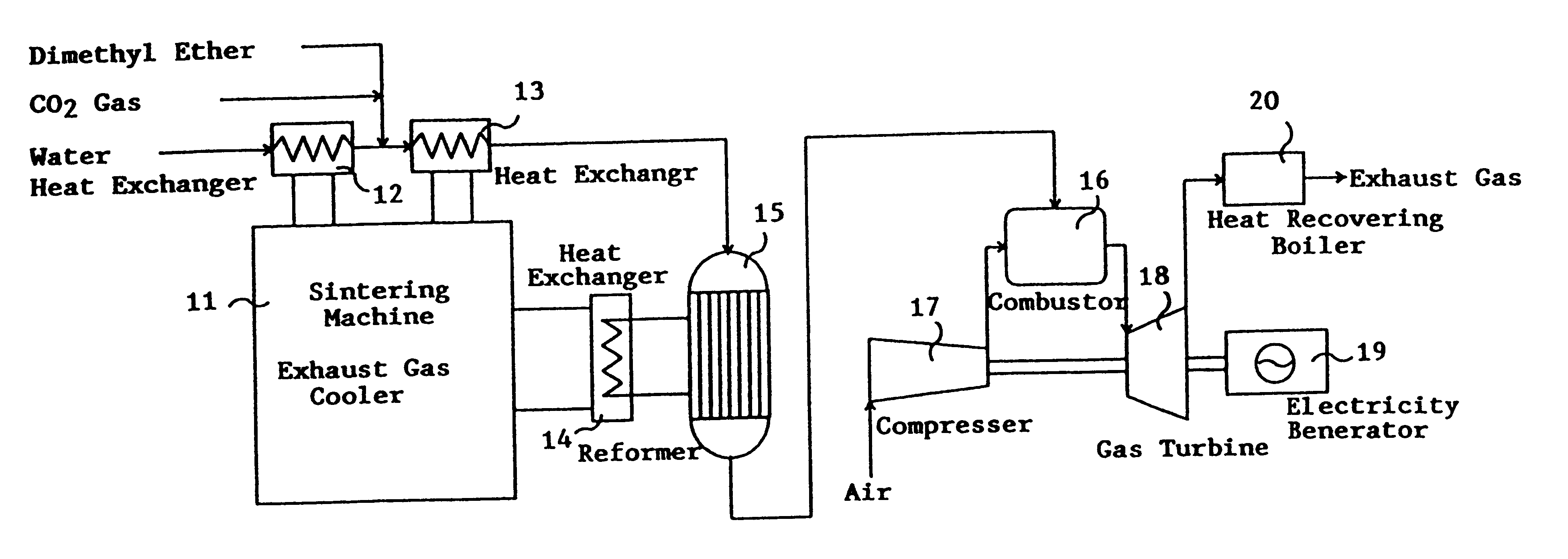
This invention provides a catalyst for producing hydrogen gas from a mixed gas comprising dimethyl ether and water vapor or carbon dioxide gas, which comprises copper, iron, cobalt, palladium, iridium, platinum, rhodium, or nickel as an active component, and a method of producing synthesis gas or hydrogen gas in a high yield at a low temperature. By using the catalyst, a fuel cell, electricity generation, reduction of iron ore and the like can be carried out.
Owner:NIPPON KOKAN KK
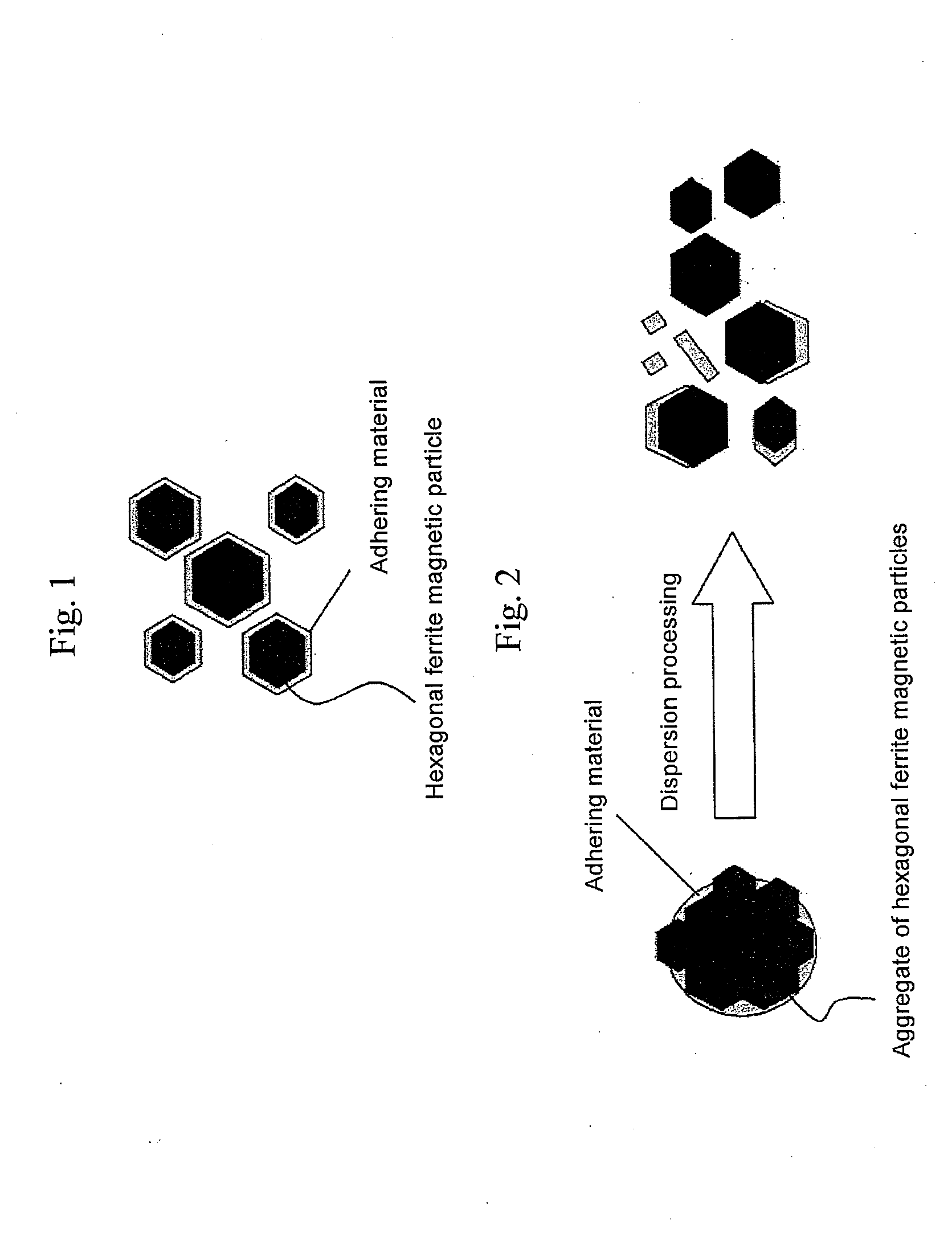
Hexagonal ferrite magnetic particle and method of manufacturing the same, and magnetic recording medium
ActiveUS20110244272A1Improve coercive forceReduce particlesMaterial nanotechnologyMagnetic materials for record carriersHeat treatedMaterials science
An aspect of the present invention relates to a method of manufacturing a hexagonal ferrite magnetic particle comprising melting an Al-containing starting material mixture to prepare a melt and quenching the melt to obtain an amorphous material; subjecting the amorphous material to heat treatment to cause a hexagonal ferrite magnetic particle to precipitate in a product obtained by the heat treatment; collecting a hexagonal ferrite magnetic particle by subjecting the product to treatment with an acid and washing, wherein the hexagonal ferrite magnetic particle collected has a particle size ranging from 15 to 30 nm, comprises 0.6 to 8.0 weight percent of Al, based on Al2O3 conversion, relative to a total weight of the particle, and Al adheres to a surface of the hexagonal ferrite magnetic particle.
Owner:FUJIFILM CORP
Anode active material, manufacturing method thereof, and non-aqueous electrolyte secondary battery
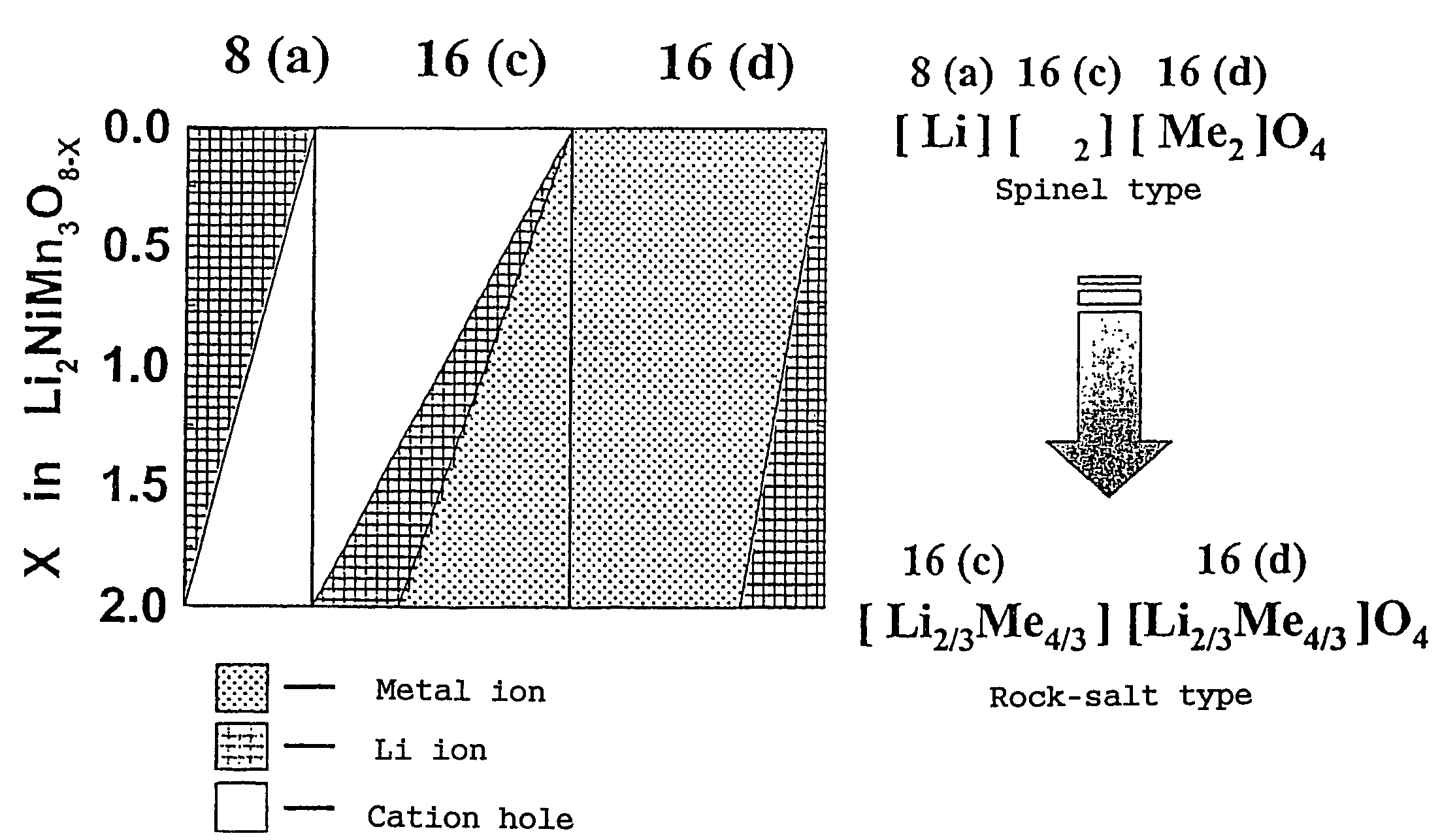
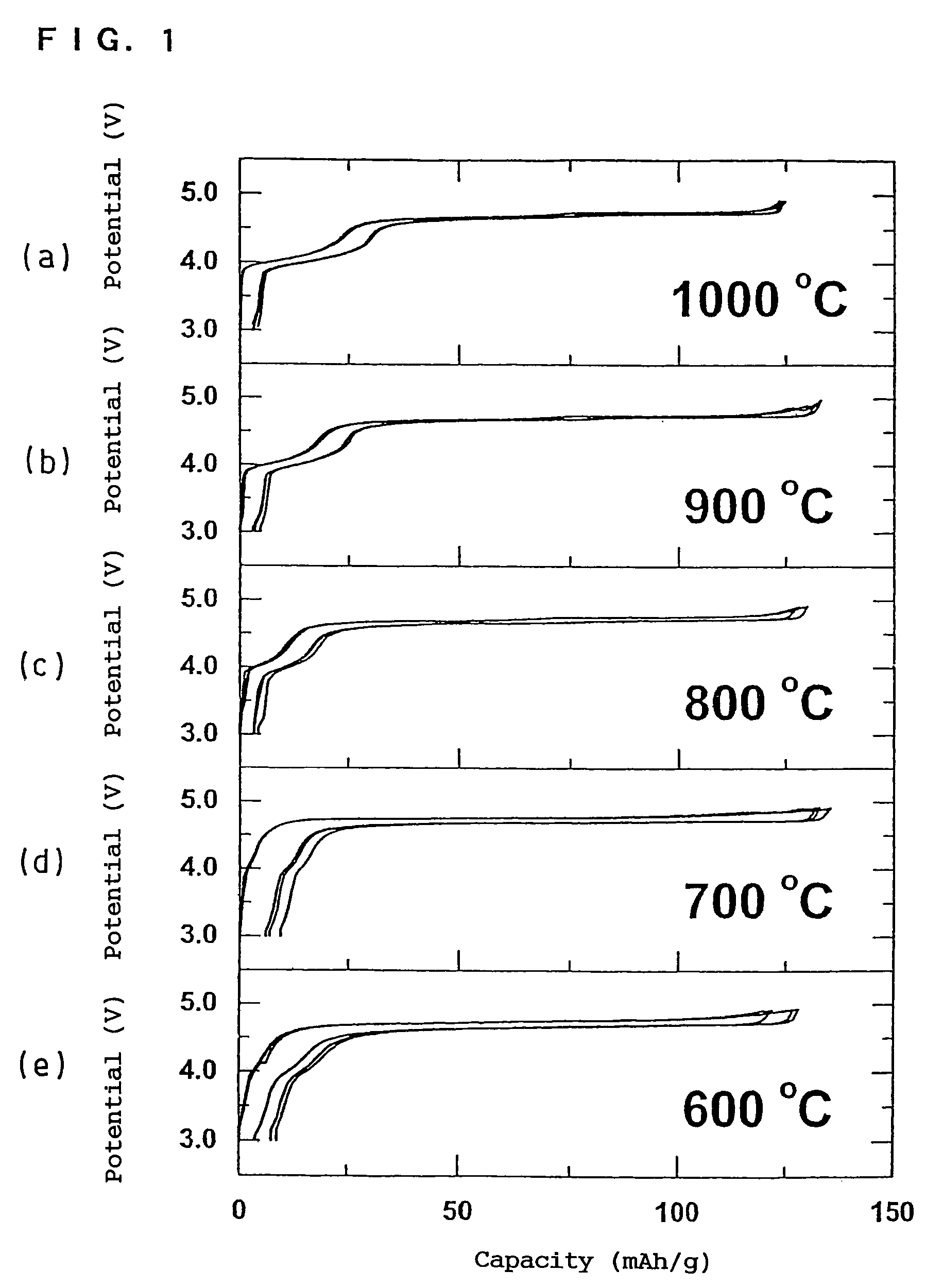
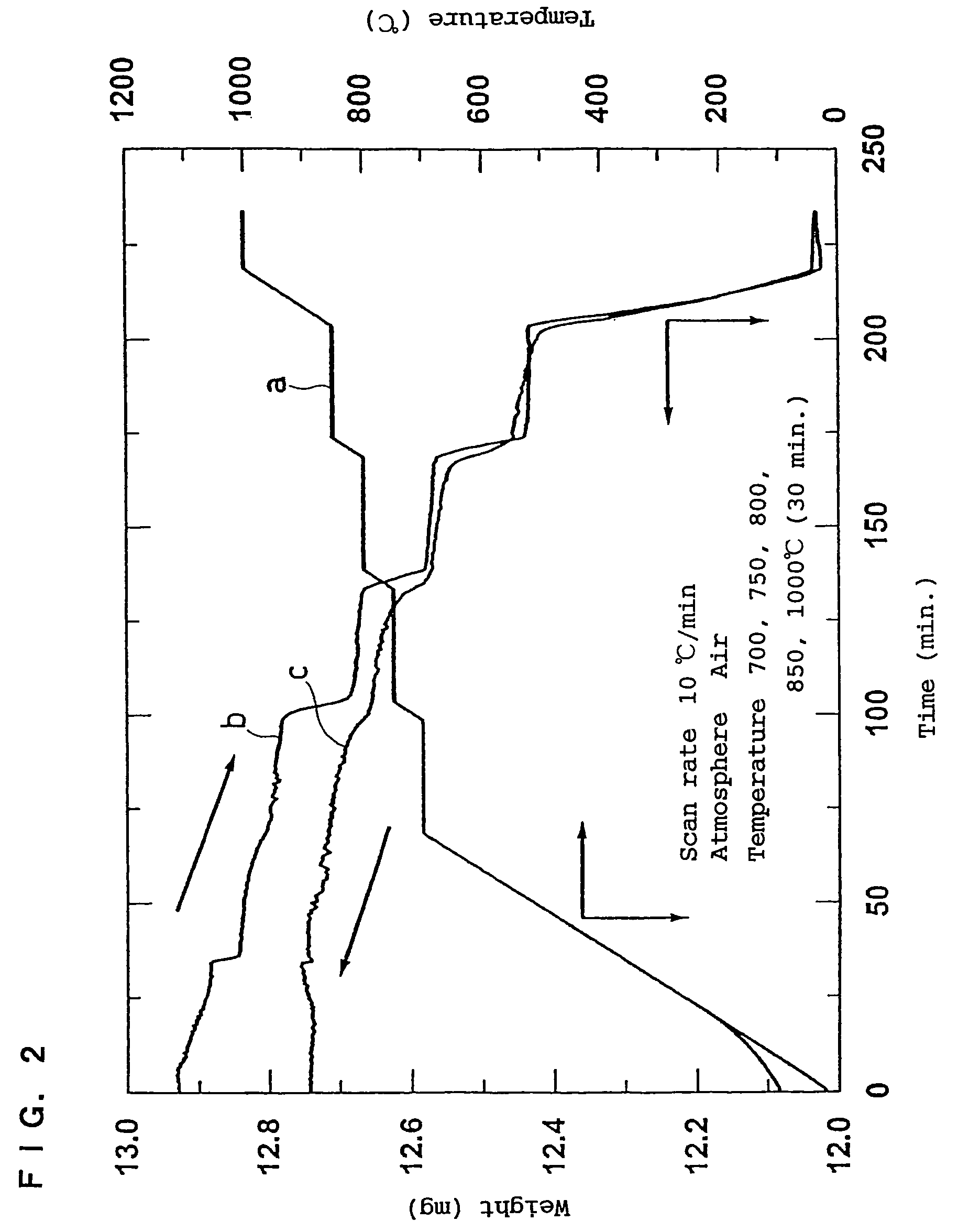
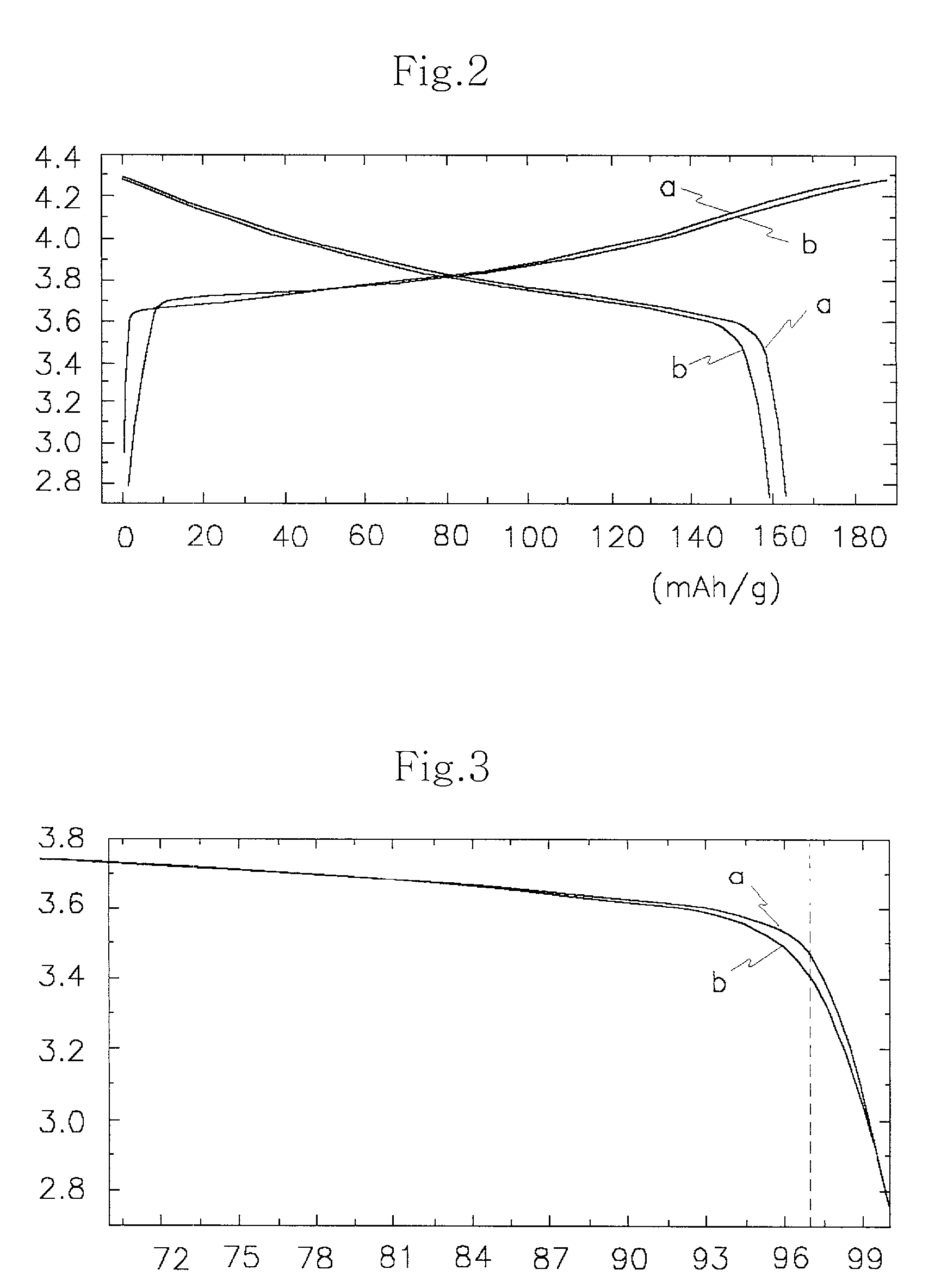
In order to provide a 3V level non-aqueous electrolyte secondary battery with a flat voltage and excellent cycle life at a high rate with low cost, the present invention provides a positive electrode represented by the formula: Li2±α[Me]4O8−x, wherein 0≦α<0.4, 0≦x<2, and Me is a transition metal containing Mn and at least one selected from the group consisting of Ni, Cr, Fe, Co and Cu, said active material exhibiting topotactic two-phase reactions during charge and discharge.
Owner:OSAKA CITY UNIV +1
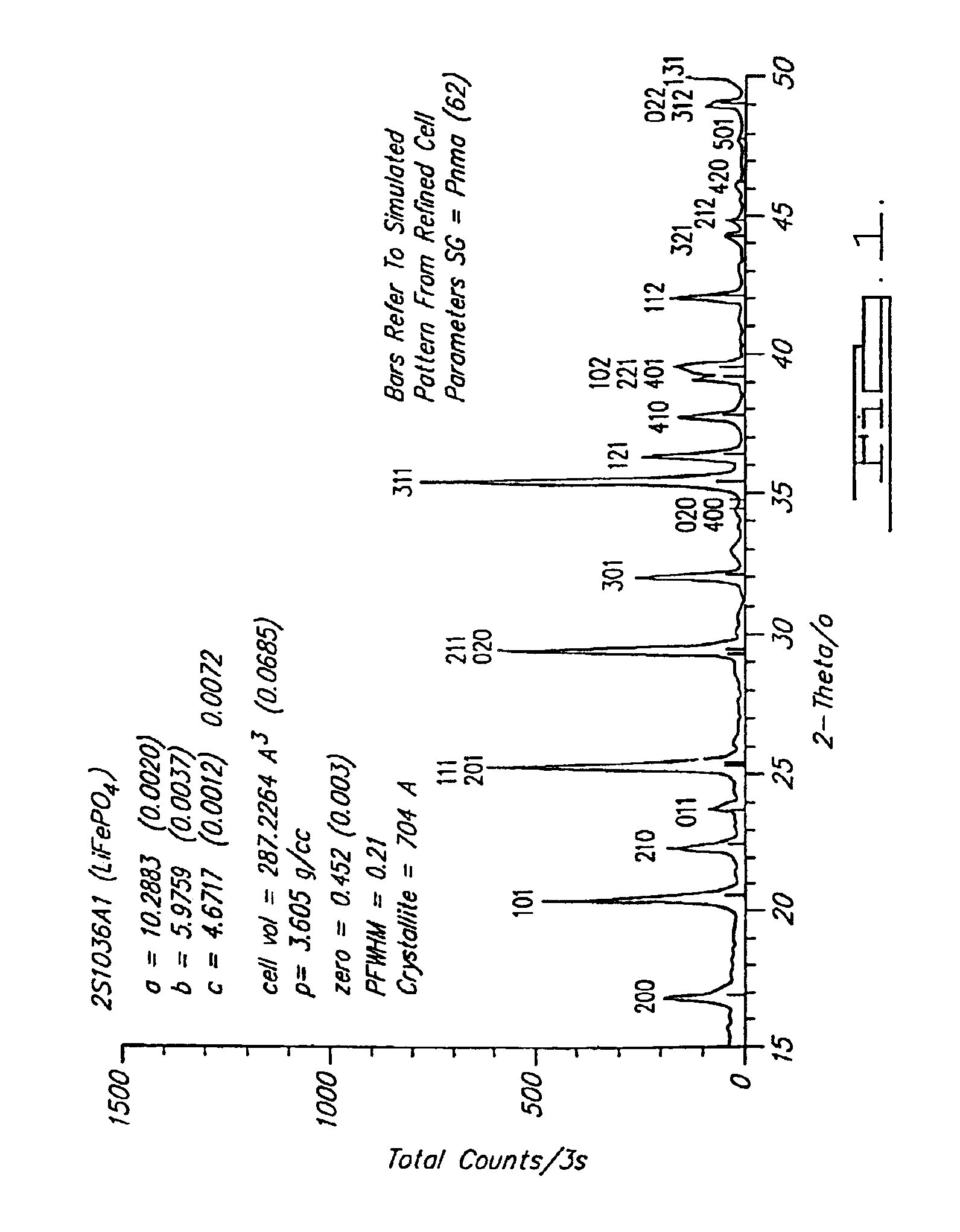
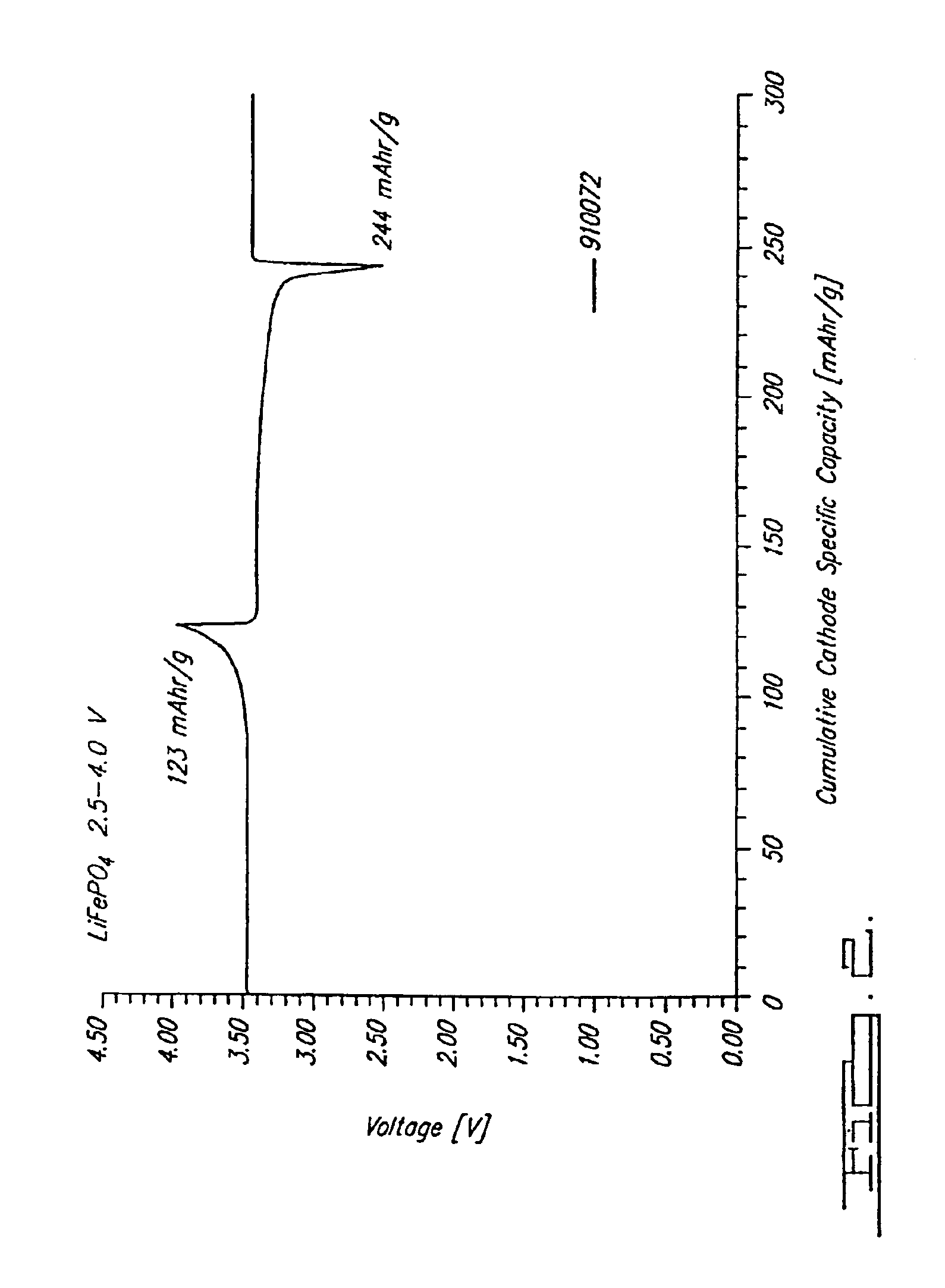
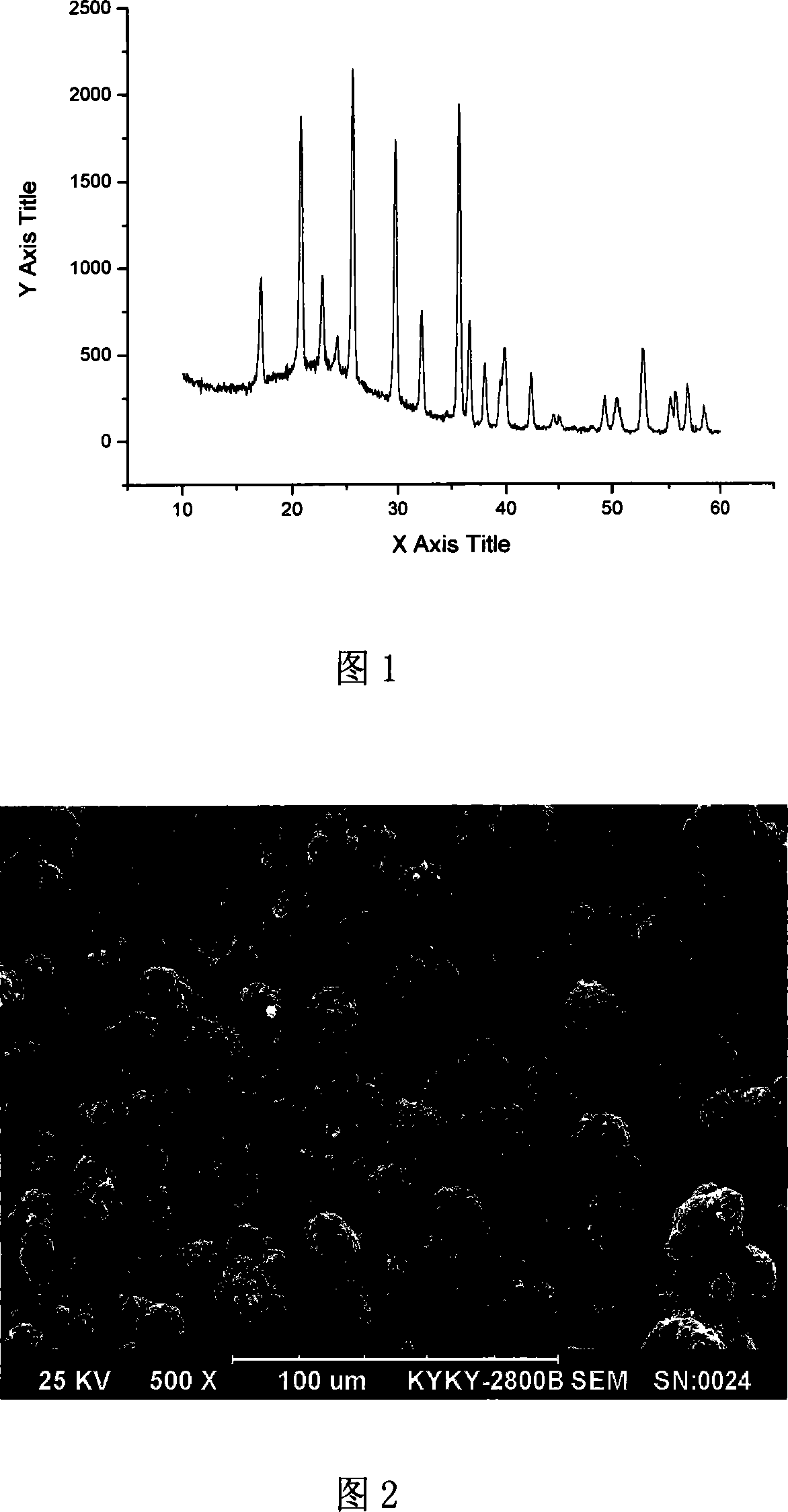
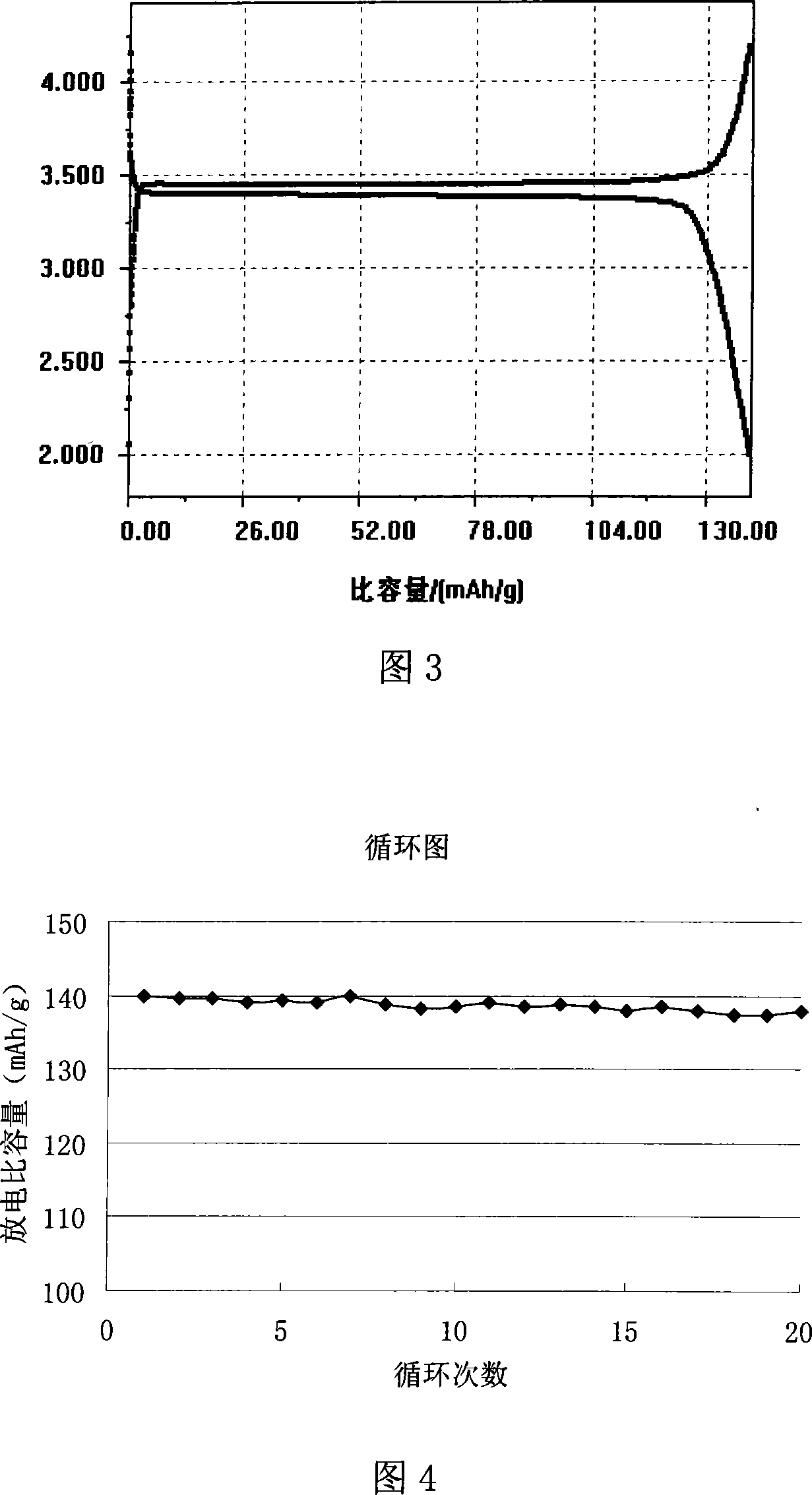
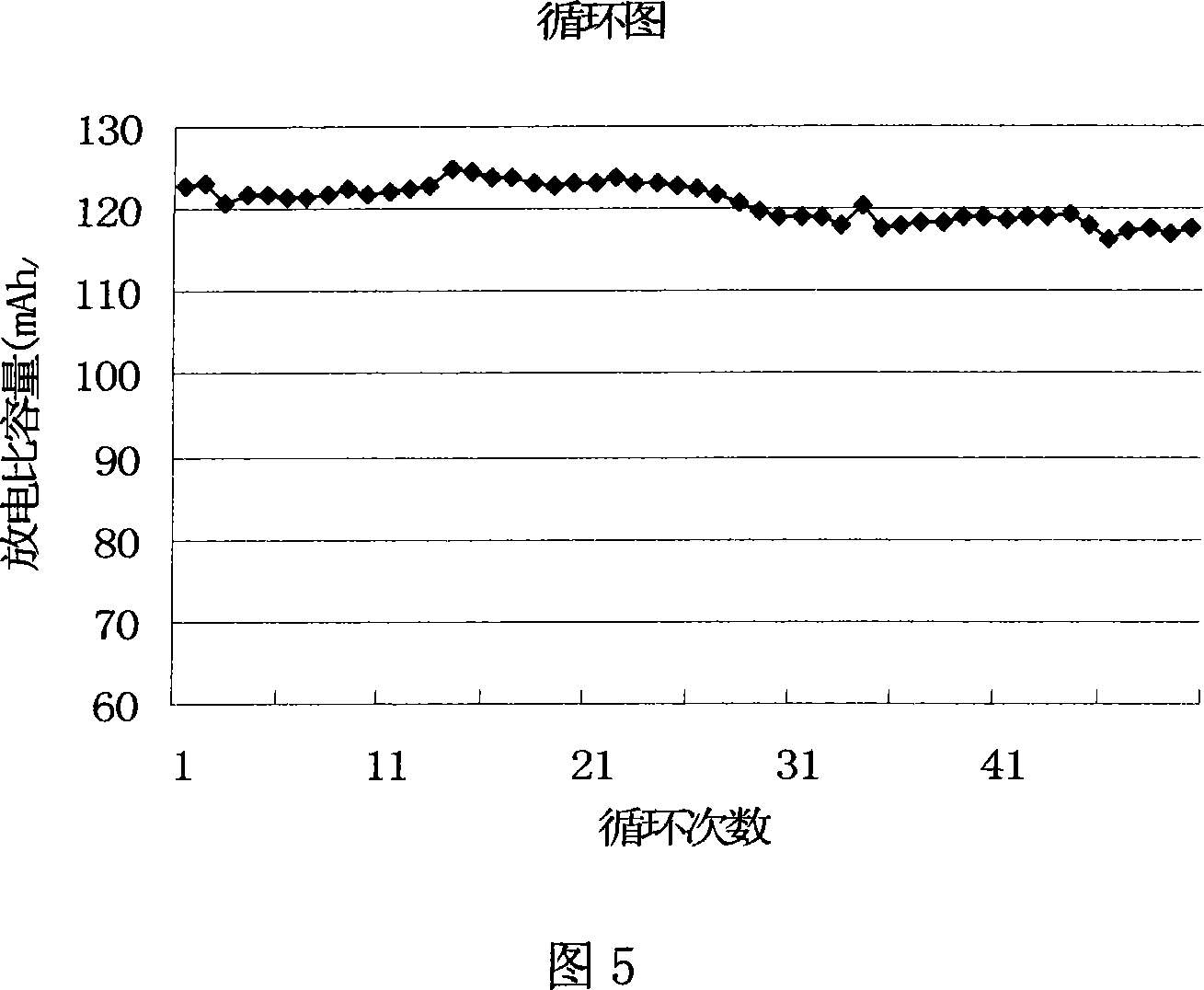
Method for preparing high-density spherical ferric lithium phosphate as anode material of lithium-ion battery
InactiveCN1635648AHigh bulk densityIncrease volume capacityElectrode manufacturing processesLithium compoundsPhosphateLithium-ion battery
This invention discloses the method for preparing high-density spherical lithium ferric phosphate used as the positive material of lithium ion cell, which contains synthesizing the ferric iron salt aqueous solution, phosphorus source aqueous solution and alkali aqueous solution to form spherical or spheroid ferric phosphate precursor, uniformly mixed with lithium source, carbon source and doped metal compound after being washed and dried, high temperature heat treating at 600-900 degree centigrade for 8-48 hr under inertia or reducing atmosphere protection to obtain lithium ferric phosphate with mean grain size of 7-12 micrometer,2.0-2.2g / cm3 of tap density, high buck density of 140-155mAh / g first discharge ratio capacity at normal temperature, and high volume ratio capacity.
Owner:TSINGHUA UNIV
Electromechanical actuators
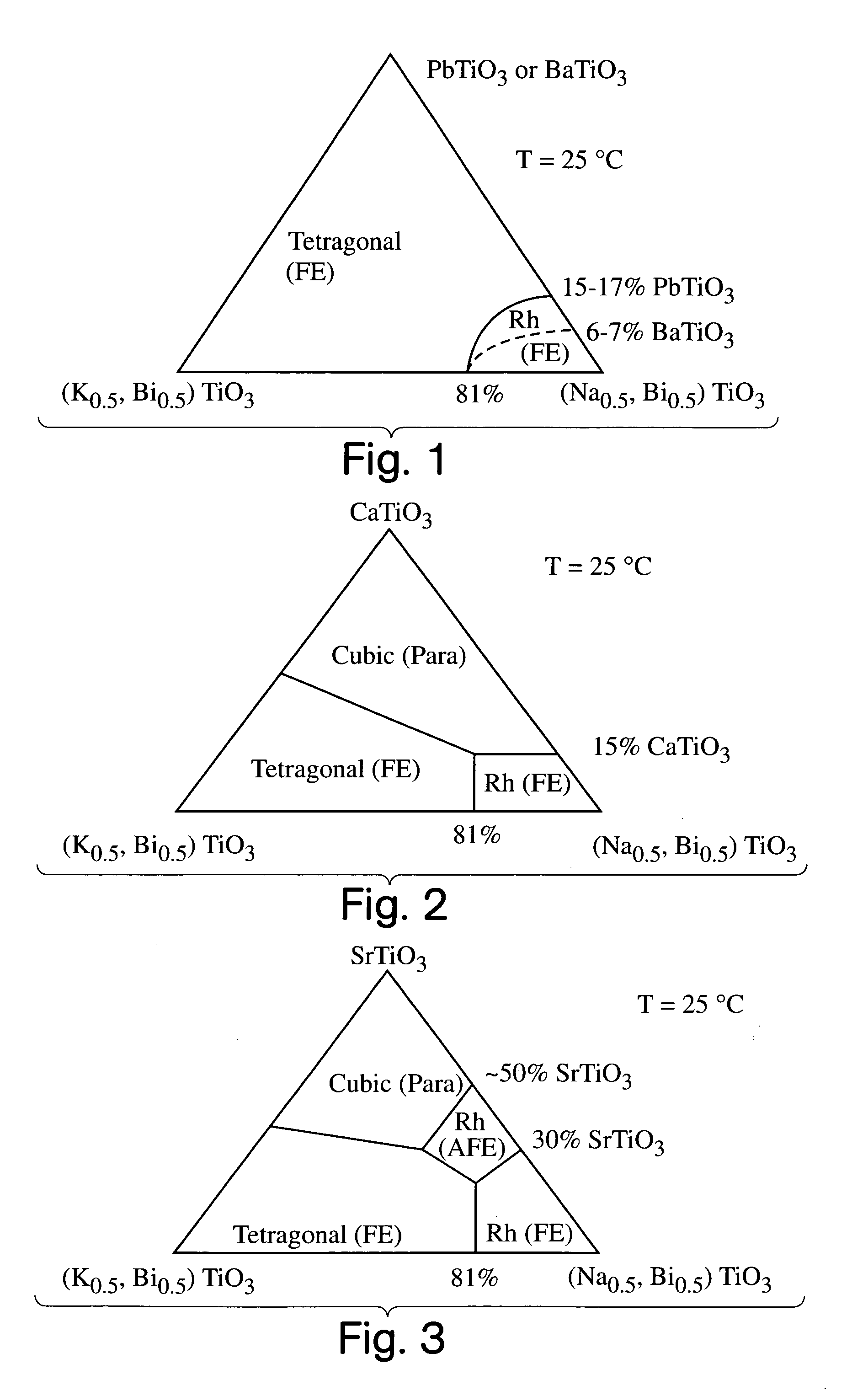
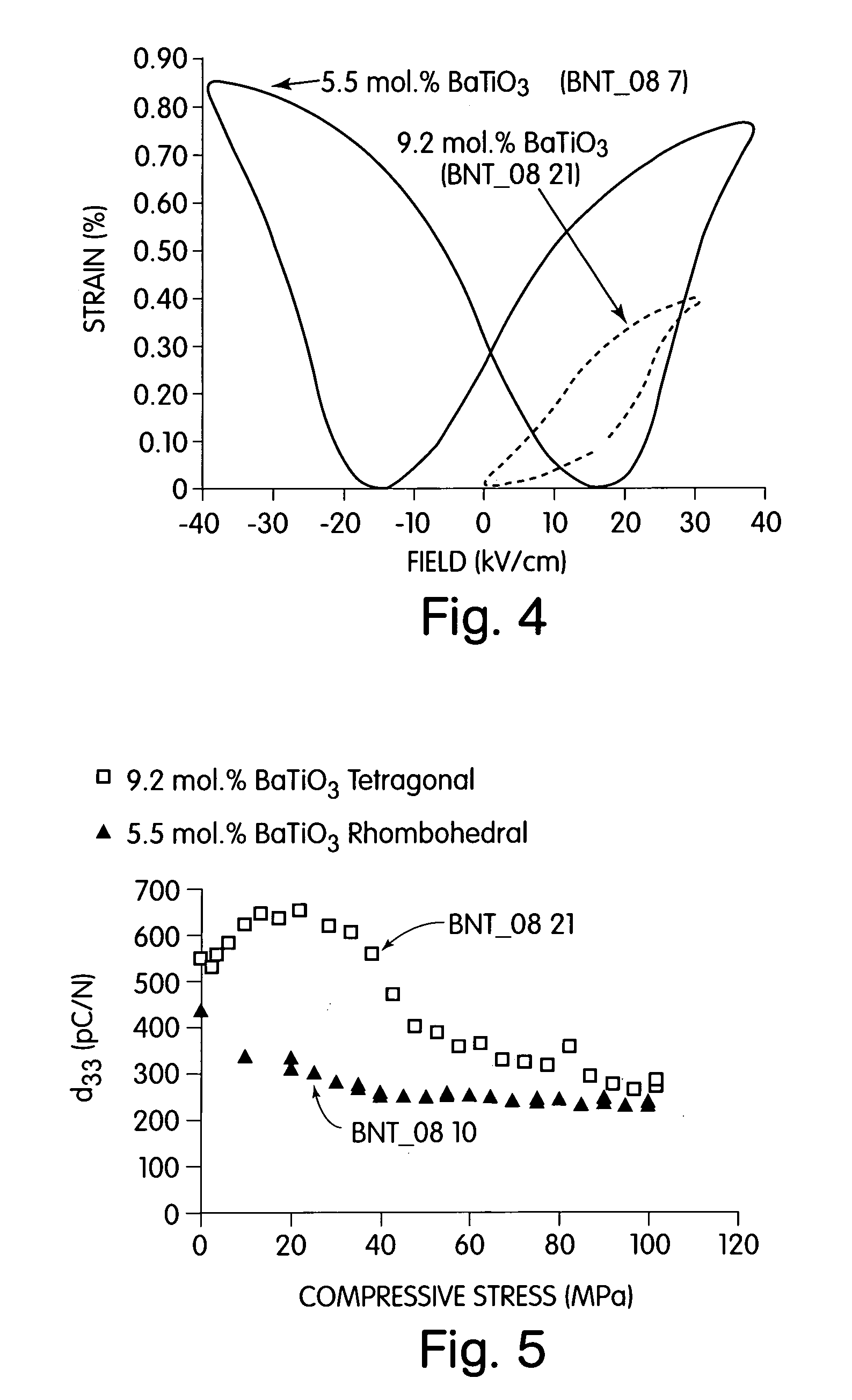
InactiveUS7090785B2Increased doping of BaTiOImprove propertiesAlkaline earth titanatesLighting support devicesElectroactive materialsMaterials science
A perovskite compound of the formula, (Na1 / 2Bi1 / 2)1-xMx(Ti1-yM′y)O3±z, where M is one or more of Ca, Sr, Ba, Pb, Y, La, Pr, Nd, Sm, Eu, Gd, Th, Dy, Ho, Er, Tm, Yb and Lu; and M′ is one or more of Zr, Hf, Sn, Ge, Mg, Zn, Al, Sc, Ga, Nb, Mo, Sb, Ta, W, Cr, Mn, Fe, Co and Ni, and 0.01<x<0.3, and 0.01<y<0.3, and z<0.1 functions as an electromechanically active material. The material may possess electrostrictive or piezoelectric characteristics.
Owner:MASSACHUSETTS INST OF TECH
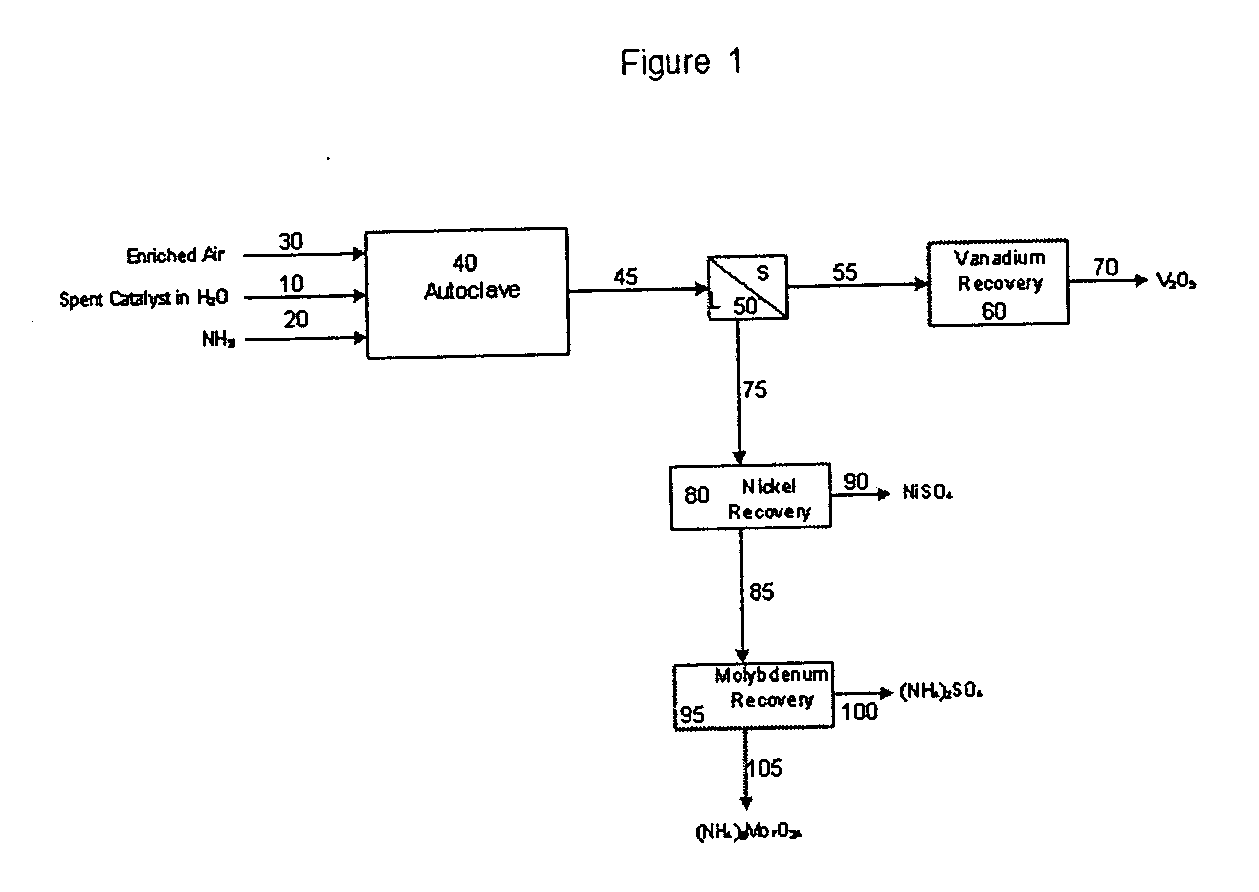
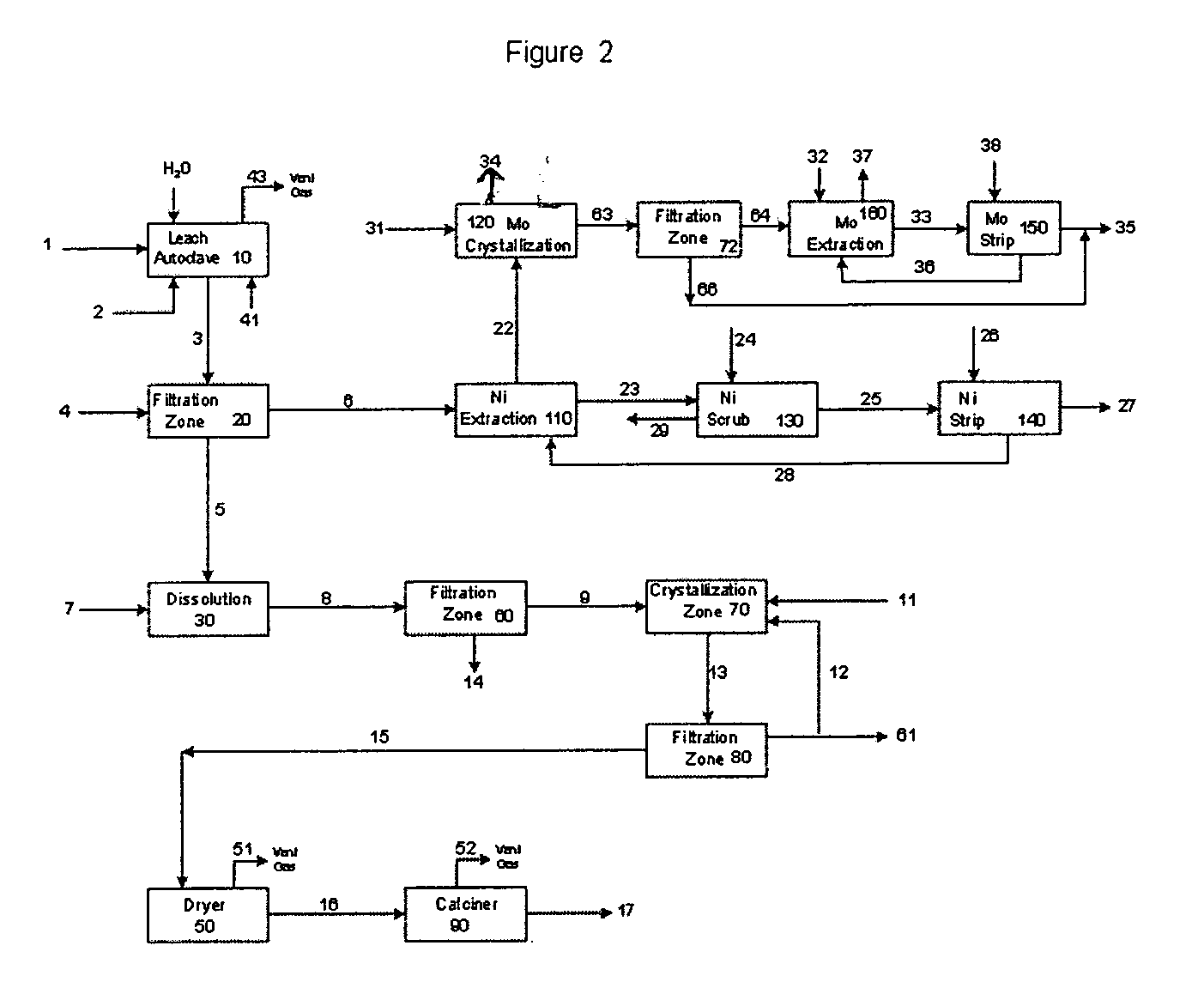
Process for metals recovery from spent catalyst
The process of this invention is directed to the removal of metals from an unsupported spent catalyst. The catalyst is subjected to leaching reactions. Vanadium is removed as a precipitate, while a solution comprising molybdenum and nickel is subjected to further extraction steps for the removal of these metals. Molybdenum may alternately be removed through precipitation.
Owner:CHEVROU USA INC
Method of hydrothermal liquid phase sintering of ceramic materials and products derived therefrom
Provided here is a method of producing a monolithic body from a porous matrix, comprising: (i) providing a porous matrix having interstitial spaces and comprising at least a first reactant; (ii) contacting the porous matrix with an infiltrating medium that carries at least a second reactant; (iii) allowing the infiltrating medium to infiltrate at least a portion of the interstitial spaces of the porous matrix under conditions that promote a reaction between the at least first reactant and the at least second reactant to provide at least a first product; and (iv) allowing the at least first product to form and fill at least a portion of the interstitial spaces of the porous matrix, thereby producing a monolithic body, wherein the monolithic body does not comprise barium titanate.
Owner:RUTGERS THE STATE UNIV
Lithium-based active materials and preparation thereof
InactiveUS6884544B2Easy to adaptHigh capacity retentionElectrode manufacturing processesPhosphatesElectrochemical cellChemistry
The invention provides novel active material represented by the general formula LiaMI1-yMIIyPO4, wherein MI is selected from the group consisting of Fe, Ni, Mn, V, Sn, Ti, Cr, and mixtures thereof; MII is selected from the group consisting of Zn, Cd, and a mixture thereof; a is about 1; and 0<y<1, which, upon electrochemical interaction, releases lithium ions, and is capable of reversibly cycling lithium ions. The invention provides a rechargeable lithium battery which comprises an electrode formed from the novel active material, wherein MI is selected from the group consisting of Fe, Co, Ni, Mn, Cu, V, Sn, Ti, Cr, and mixtures thereof. Methods for making the novel active material and methods for using such a material in electrochemical cells are also provided.
Owner:LITHIUM WERKS TECH BV
Positive active material for rechargeable lithium battery and method of preparing same
InactiveUS20020055042A1Improve thermal stabilityElectrode manufacturing processesZirconium compoundsPhysical chemistryLithium battery
Disclosed is a positive active material for a rechargeable lithium battery. The positive active material includes at least one compound represented by formulas 1 to 4 andl a metal oxide or composite metal oxide layer formed on the compound. <table-cwu id="TABLE-US-00001"> <number>1< / number> <tgroup align="left" colsep="0" rowsep="0" cols="3"> <colspec colname="OFFSET" colwidth="42PT" align="left" / > <colspec colname="1" colwidth="77PT" align="left" / > <colspec colname="2" colwidth="98PT" align="center" / > <row> <entry>< / entry> <entry>< / entry> < / row> <row> <entry>< / entry> <entry namest="OFFSET" nameend="2" align="center" rowsep="1">< / entry> < / row> <row> <entry>< / entry> <entry>LixNi1-yMnyF2< / entry> <entry>(1)< / entry> < / row> <row> <entry>< / entry> <entry>LixNi1-yMnyS2< / entry> <entry>(2)< / entry> < / row> <row> <entry>< / entry> <entry>LixNi1-y-zMnyMzO2-aFa< / entry> <entry>(3)< / entry> < / row> <row> <entry>< / entry> <entry>LixNi1-y-zMnyMzO2-aSa< / entry> <entry>(4)< / entry> < / row> <row> <entry>< / entry> <entry namest="OFFSET" nameend="2" align="center" rowsep="1">< / entry> < / row> < / tgroup> < / table-cwu> (where M is selected from the group consisting of Co, Mg, Fe, Sr, Ti, B, Si, Ga, Al, Sc, Y, La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu, Ac, Th, Pa, U, Np, IPu, Am, Cm, Bk, Cf, Es, Fm, Md, No and Lr, 0.95<=x<=1.1, 0<=y<=0.99, 0<=,z<=0.5, and 0<=a<=0.5)
Owner:SAMSUNG SDI CO LTD
Anode active material, manufacturing method thereof, and non-aqueous electrolyte secondary battery
In order to provide a 3V level non-aqueous electrolyte secondary battery with a flat voltage and excellent cycle life at a high rate with low cost, the present invention provides a positive electrode represented by the formula: Li2±α[Me]4O8−x, wherein 0≦α<0.4, 0≦x<2, and Me is a transition metal containing Mn and at least one selected from the group consisting of Ni, Cr, Fe, Co and Cu, said active material exhibiting topotactic two-phase reactions during charge and discharge.
Owner:OSAKA CITY UNIVERSITY +1
Ferric phosphate lithium material for lithium ion powder cell and preparation method thereof
ActiveCN101081696APromote crystallizationSingle structureElectrode manufacturing processesLithium compoundsCarbon layerLithium iron phosphate
The present invention discloses lithium iron phosphate material for lithium ion power cell and its preparation process, and dissolves the technological problem of raising the discharge power and safety performance. The lithium iron phosphate material is spherical or spheroid composite particle with one base body of lithium iron phosphate and doping and modifying agent and one coating carbon layer. Its preparation process includes wet ball milling the mixture comprising lithium salt, ferric salt, phosphoric acid, doping and modifying agent and dispersant, spraying to pelletize, heating for pre-treatment, maintaining temperature, cooling, pulverizing and shaping. Compared with available technology, the present invention has the features of high crystallization, single structure, average composite particle size of 5-60 microns, specific surface area of 8.0-15.0 sq m / g, bulk density of 1.4-1.7 g / cu cm, high specific capacity, high safety, etc.
Owner:BTR (TIANJIN) NANO MATERIAL MFG CO LTD
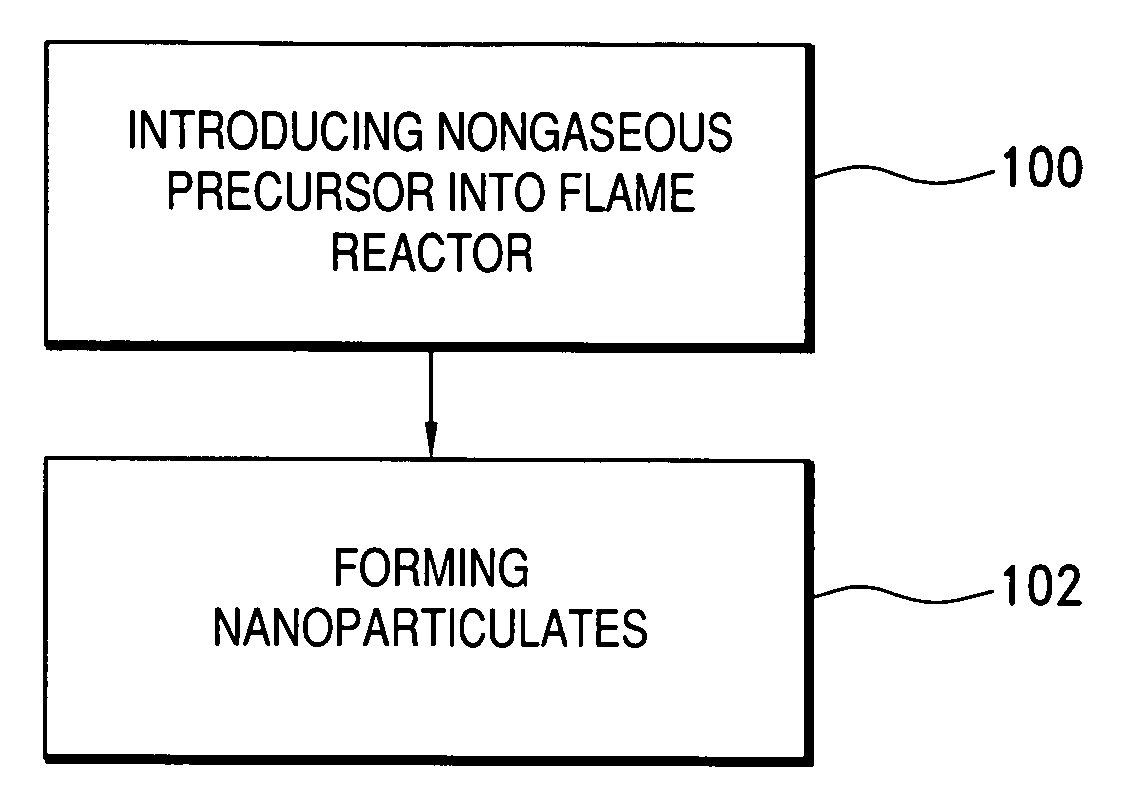
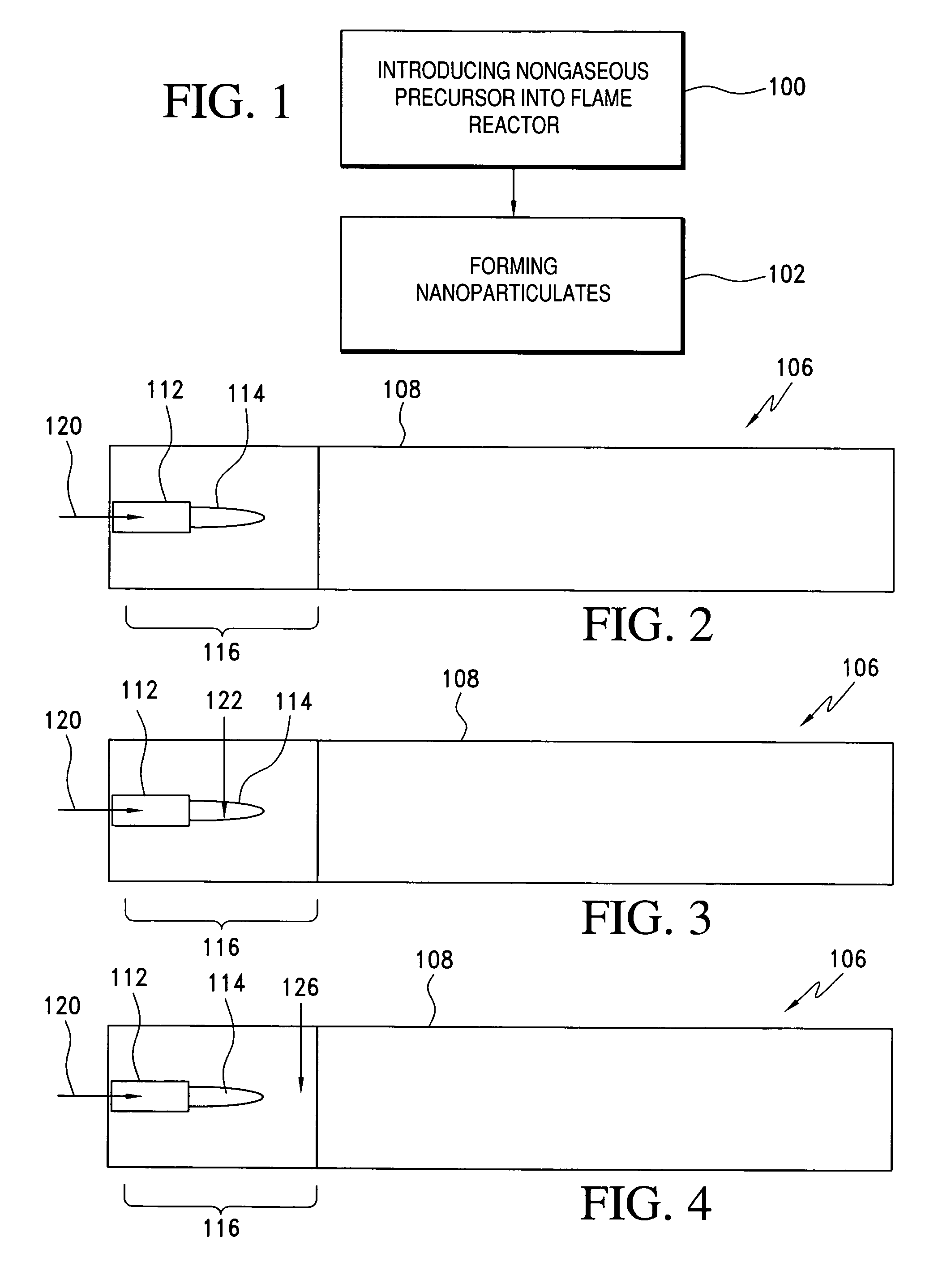
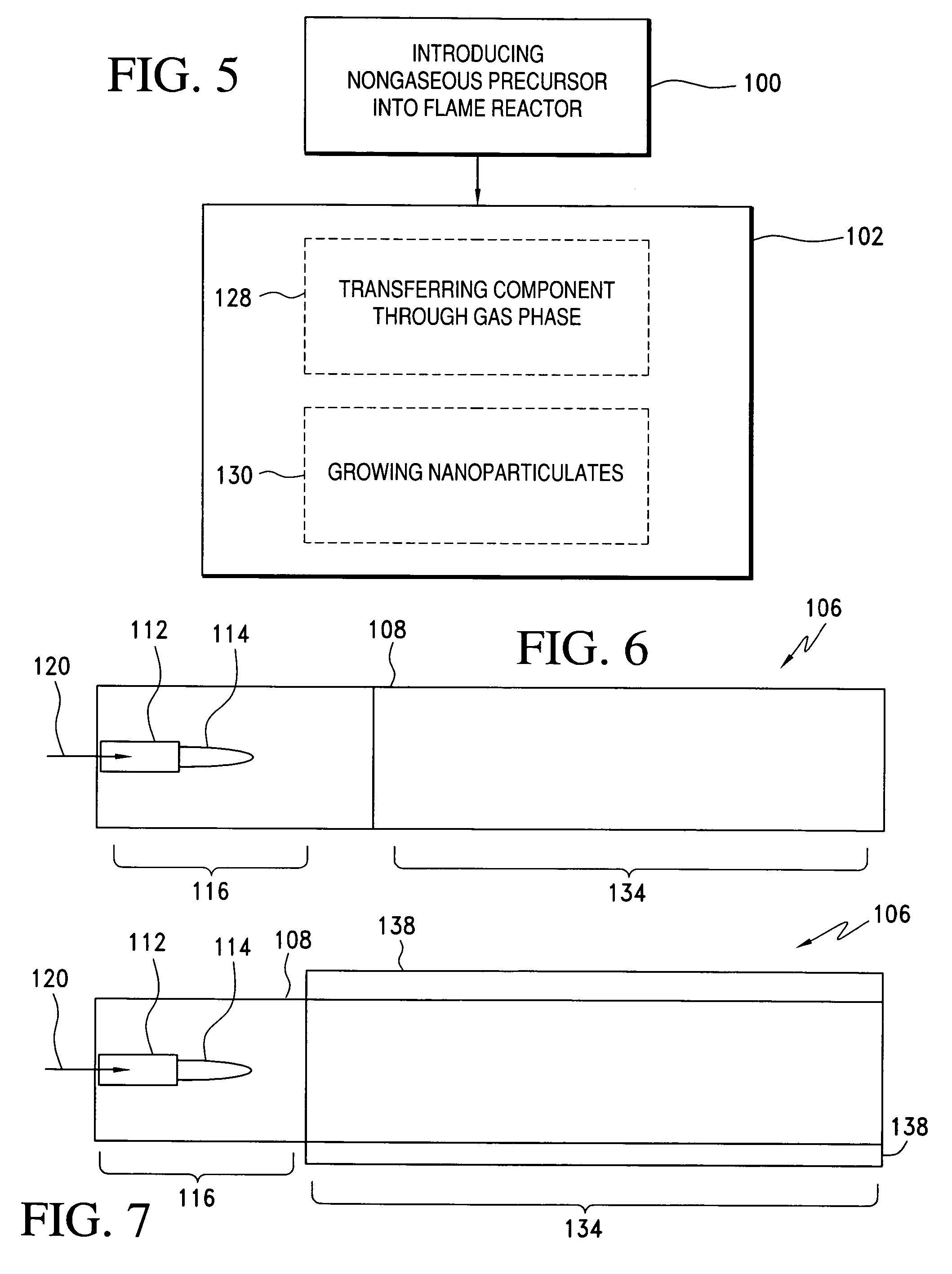
Method of making nanoparticulates and use of the nanoparticulates to make products using a flame reactor
InactiveUS20060166057A1Easy to joinLower melting temperatureMaterial nanotechnologyBurnersMulti materialNanoparticle
The present invention relates to a method of making nanoparticulates in a flame reactor, the nanoparticulates having controlled properties such as weight average particle size, composition and morphology. The nanoparticulates made with the method of present invention may be tailored to a specific weight average particle size range, such as from about 1 nm to about 500 nm. In addition to weight average particle size, the nanoparticulates made with the method of the present invention may include a variety of materials including metals, ceramics, organic materials, and combinations thereof. Moreover, the method of the present invention allows control over the morphology of the nanoparticulates, which allows the production of nanoparticulates with any desired morphology including spheroidal and unagglomerated; and agglomerated (aggregated) into larger units of hard aggregates.
Owner:CABOT CORP
Core-shell composite anode material for lithium ion battery and preparation method thereof
ActiveCN101740752AImprove conductivityHigh specific capacitySecondary cellsIron compoundsLithium iron phosphateElectrical battery
The invention discloses a core-shell composite anode material for a lithium ion battery. The composite anode material has a core-shell structure; the core-shell structure consists of a core-layer active material and a shell-layer active material; the core-layer active material is LiFePO4 or lithium manganate; the shell-layer active material is carbon-containing LiFePO4; the LiFePO4 has an Li1-XMXFePO4 or LiFe1-yMyPO4 structure; the lithium manganate has a LiMnO2 or LiMn2O4 structure; the carbon is selected from one or more of carbon nano tubes, superfine conductive black and an agraphitic carbon material; and the composite anode material comprises 65 to 99 mass percent of core-layer active material and 1 to 35 mass percent of shell-layer active material. The composite anode material of the invention has stable performance and excellent electrochemical properties; and the lithium ion battery manufactured by using the material has relatively high charge-discharge capacity and excellent cycle performance, can perform quick charging and large multiplying-factor discharging, can adapt to an ultra-low temperature working environment and is safe and stable.
Owner:SHENZHEN DYNANONIC
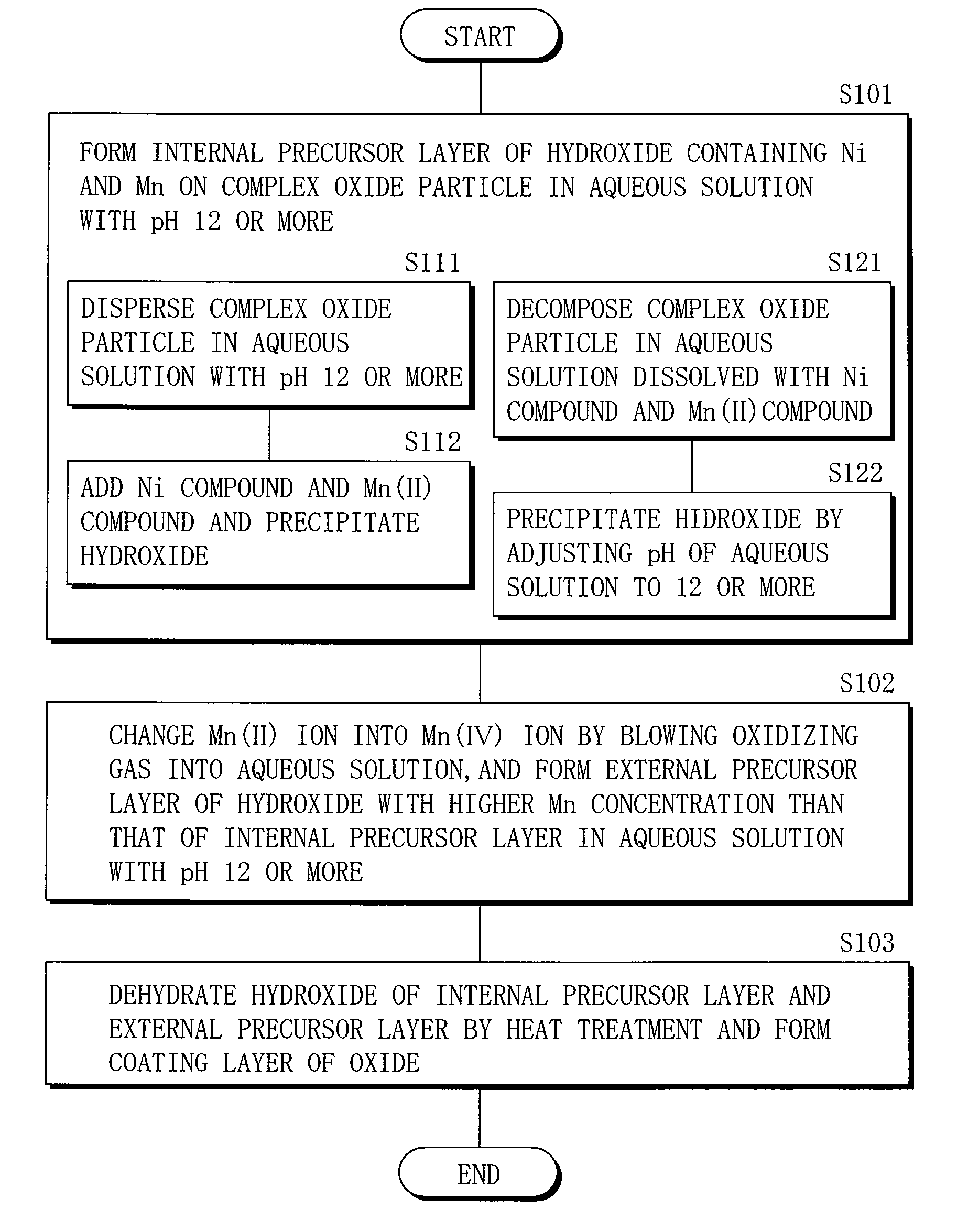
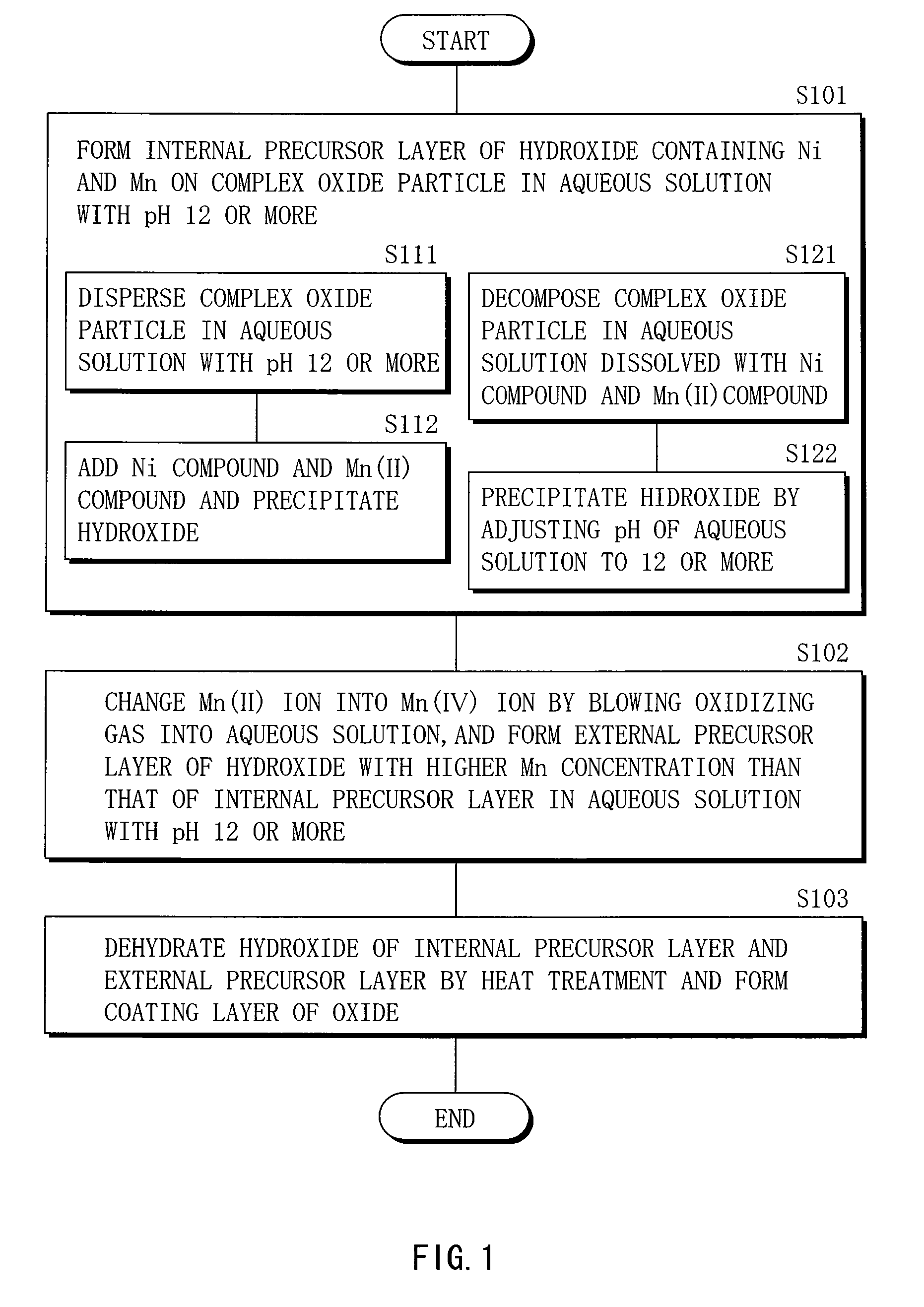
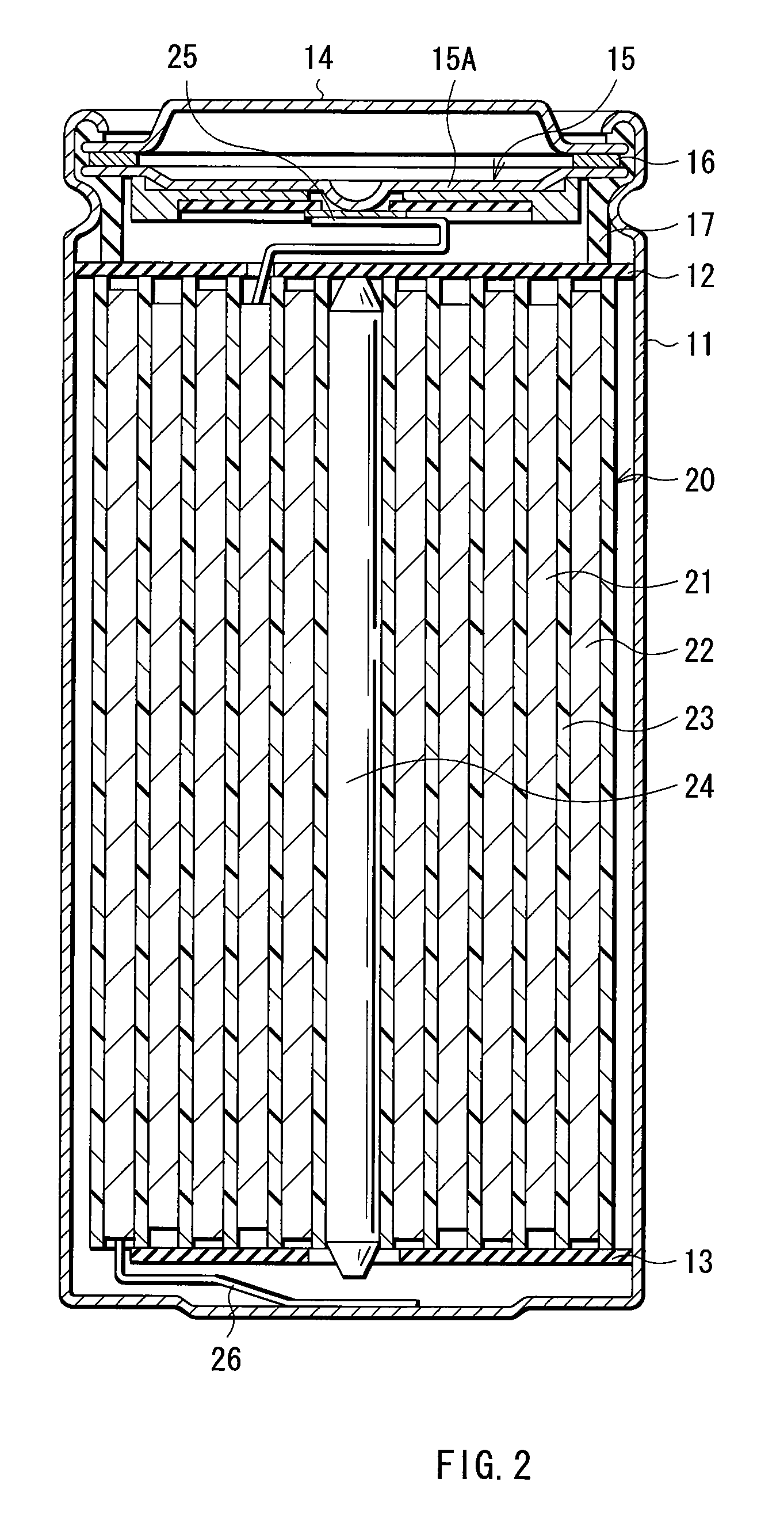
Cathode active material, method of manufacturing it, cathode, and battery
ActiveUS20060275667A1Large capacityGood chemical stabilityElectrode manufacturing processesNon-aqueous electrolyte accumulatorsLithiumManganese
A cathode active material capable of increasing a capacity and improving high temperature characteristics or cycle characteristics, a method of manufacturing it, a cathode using the cathode active material, and a battery using the cathode active material are provided. In a cathode active material contained in a cathode, a coating layer is provided on at least part of a complex oxide particle containing at least lithium (Li) and cobalt (Co). The coating layer is an oxide which contains lithium (Li) and at least one of nickel (Ni) and manganese (Mn).
Owner:MURATA MFG CO LTD
Production Of Barium Titanate Compounds
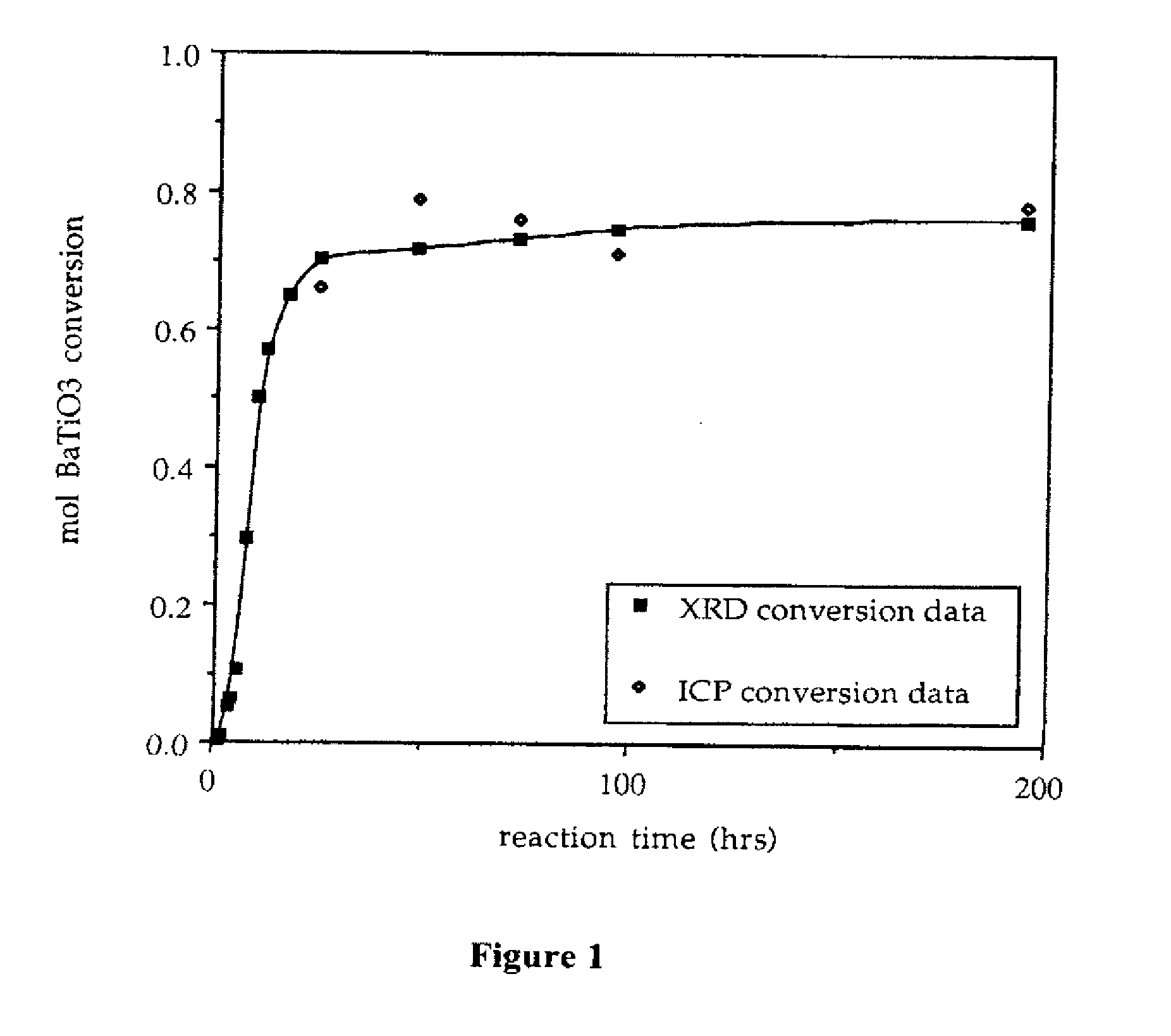
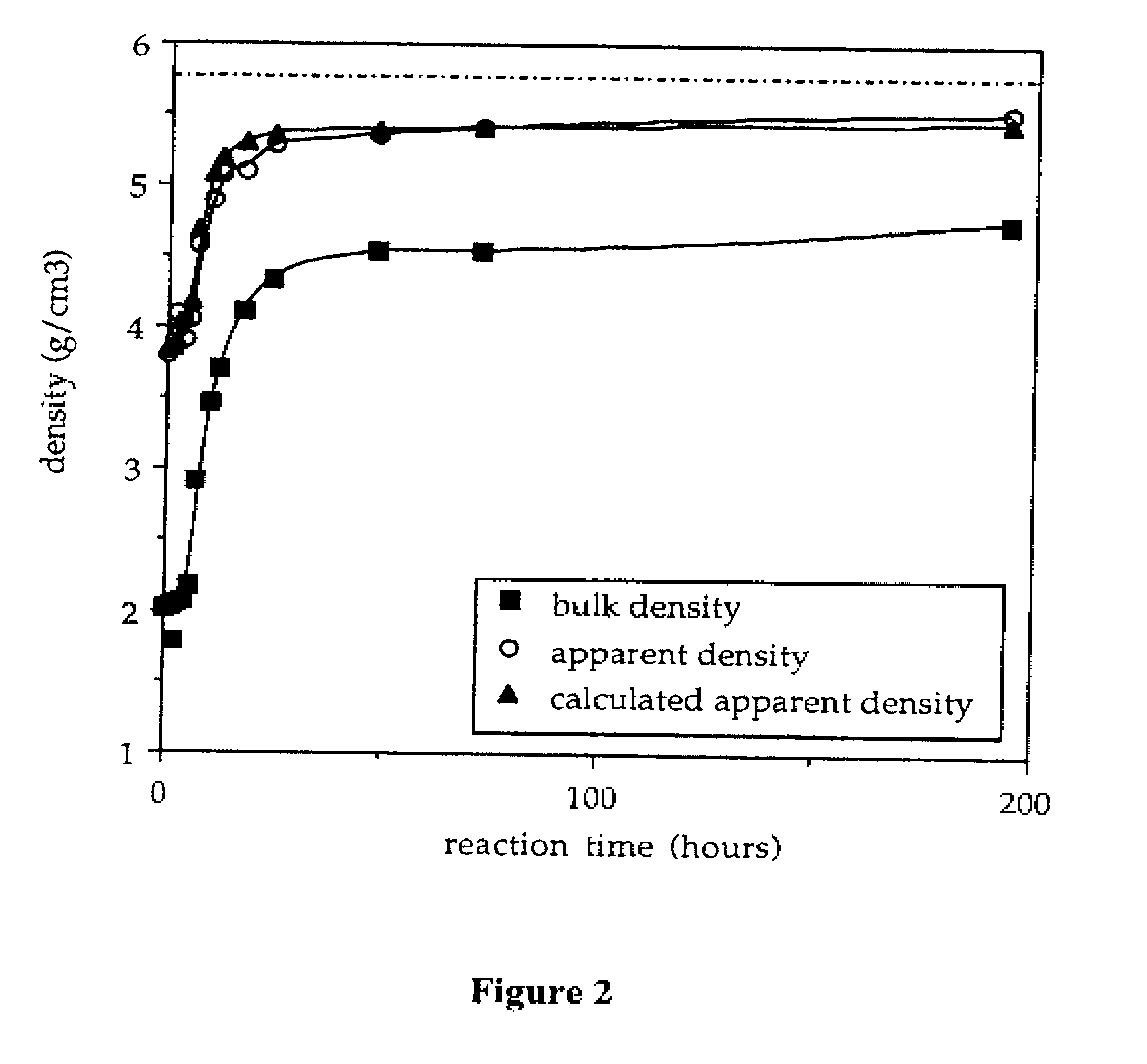
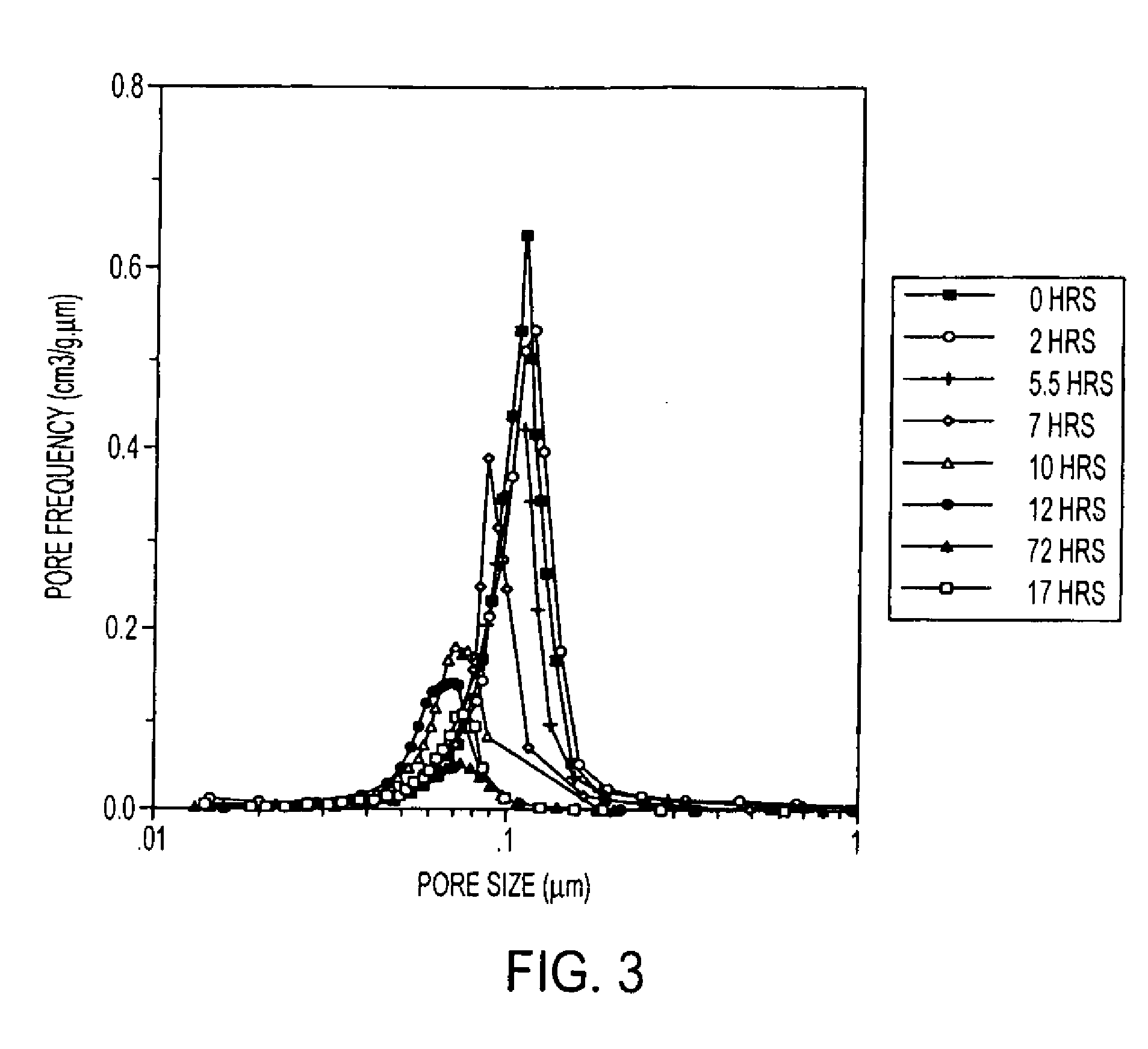
InactiveUS20070202036A1High purityLow costMaterial nanotechnologyAlkaline earth titanatesBarium titanateSpherical shaped
An ultrafine powder of barium titanate including solid solutions and doped compounds that meets up to specific characteristics is produced by method comprising two main steps. The first step is a reaction, typically in a Segmented Flow Tubular Reactor, between reactants to produce cubic-structure barium titanate composed of non-agglomerated ultrafine particles having a shape of given aspect ratio, usually a generally spherical shape, of low density corresponding at most to 90% of the intrinsic density, all particles being smaller than 1 micron and having a narrow particle size distribution and wherein the ratio of Ba:Ti including substitutents and dopants is very close to the ideal stoichiometry. This is followed by subjecting the powder produced in the first step to a second stage solvothermal post treatment typically in an autoclave at temperature less than 400° C. to convert the cubic-structure particles of low density to ultrafine tetragonal particles of increased density corresponding to at least 90% of the intrinsic density while maintaining the same aspect ratio, and maintaining the size of all particles below 1 micron, the narrow particle size distribution span, and the given ideal stoichiometry. The produced particles can have a non-spherical facetted shape such as cube-like.
Owner:JONGEN NATHALIE +1
Processes for forming nanoparticles in a flame spray system
In one aspect, the process includes providing a precursor medium comprising a liquid vehicle and a precursor to a component, and flame spraying the precursor medium under conditions effective to form a population of nanoparticles, wherein the nanoparticles include the component. The population of nanoparticles, as formed, comprises less than about 5 percent by volume particles having a particle size greater than 1.0 μm. A size distribution of the population of nanoparticles may have a d50 value less than about 500 nm, and it may be unimodal. The size distribution may have a geometric standard deviation of less than about 2. The process may occur continuously for at least four hours or more. Greater than about 90 percent by weight of the precursor to the component in the precursor medium may be converted to the component in the nanoparticles. The process typically occurs in an enclosed flame spray reactor.
Owner:CABOT CORP
Supported polyoxometalates and process for their preparation
InactiveUS7417008B2Group 4/14 element organic compoundsOxygen/ozone/oxide/hydroxideCatalytic oxidationMesoporous silica
Owner:EXXONMOBIL CHEM PAT INC
Controlling flame temperature in a flame spray reaction process
The invention relates to a process for decreasing flame temperature in a flame spray reaction system, the process comprising the steps of providing a precursor medium comprising a precursor to a component; flame spraying the precursor medium under conditions effective to form a population of product particles; and decreasing the flame temperature by contacting the flame with a cooling medium. The process of the present invention allows for the control of the size, composition and morphology of the nanoparticles made using the process. The invention also relates to a nozzle assembly that comprises a substantially longitudinally extending atomizing feed nozzle that comprises an atomizing medium conduit and one or more substantially longitudinally extending precursor medium feed conduits. The nozzle assembly of the present invention is used in a flame spray system to produce nanoparticles using the processes described herein.
Owner:CABOT CORP
Cathode active material and nonaqueous secondary battery containing the same
InactiveUS6103422ASuppress deterioration of electrode performanceReduce charge and dischargeAluminium compoundsActive material electrodesManganeseCobalt
PCT No. PCT / JP96 / 03738 Sec. 371 Date Jun. 24, 1998 Sec. 102(e) Date Jun. 24, 1998 PCT Filed Dec. 20, 1996 PCT Pub. No. WO97 / 23918 PCT Pub. Date Jul. 3, 1997The powdery cathode active material of the present invention for use in a nonaqueous electrolyte secondary battery comprises as the active ingredient a lithium manganese composite oxide and at least one nonmanganese metal element selected from the group consisting of aluminum, magnesium, vanadium, chromium, iron, cobalt, nickel, and zinc, provided that the nonmanganese metal element exists in the surface portions of fine particles constituting the powdery cathode active material. A nonaqueous electrolyte secondary battery comprising such a cathode active material, an anode active material and a nonaqueous electrolytic solution containing a lithium salt has a large charge-and-discharge capacity and a stable charge-and-discharge cycle durability, and can be prepared inexpensively.
Owner:KAO CORP
Methods of making lithium metal cathode active materials
The invention provides a novel method for making lithium mixed metal materials in electrochemical cells. The lithium mixed metal materials comprise lithium and at least one other metal besides lithium. The invention involves the reaction of a metal compound, a phosphate compound, with a reducing agent to reduce the metal and form a metal phosphate. The invention also includes methods of making lithium metal oxides involving reaction of a lithium compound, a metal oxide with a reducing agent.
Owner:LITHIUM WERKS TECH BV
Single-crystal-like materials
InactiveUS7022303B2Improve fracture toughnessReadily apparentMagnesium fluoridesCalcium/strontium/barium fluoridesMetallurgySingle crystal
Polycrystalline materials of macroscopic size exhibiting Single-Crystal-Like properties are formed from a plurality of Single-Crystal Particles, having Self-Aligning morphologies and optionally ling morphology, bonded together and aligned along at least one, and up to three, crystallographic directions.
Owner:RUTGERS THE STATE UNIV
Shrouded-plasma process and apparatus for the production of metastable nanostructured materials
InactiveUS20070044513A1Improve compactnessImprove sintering performanceMaterial nanotechnologyNitrogen compoundsLiquid jetShort range order
A method and apparatus for producing metastable nanostructured materials employing a ceramic shroud surrounding a plasma flame having a steady state reaction zone into which an aerosol or liquid jet of solution precursor or powder material is fed, causing the material to be pyrolyzed, melted, or vaporized, followed by quenching to form a metastable nanosized powder that has an amorphous (short-range ordered), or metastable microsized powder that has a crystalline (long-range ordered) structure, respectively.
Owner:RUTGERS THE STATE UNIV
Methods of synthesizing a ferrate oxidant and its use in ballast water
Novel devices for synthesizing ferrate and uses thereof are described. One aspect of the invention relates to devices and systems for synthesizing ferrate at a site proximal to the site of use.
Owner:FERRATE TREATMENT TECH
Lithium transition metal complex oxide for lithium ion secondary battery cathode active material and method for producing the same, lithium ion secondary battery cathode active material, and lithium ion secondary battery
InactiveUS20080241693A1Improve featuresAvoid security issuesIron compoundsCobalt compoundsLithium compoundSilicon
A lithium transition metal complex oxide for a lithium ion secondary battery cathode active material contains 100 to 1000 ppm of silicon and 300 to 900 ppm of fluorine. A method for producing the lithium transition metal complex oxide includes the step of mixing a lithium compound, a transition metal compound, a fluorine compound, and a silicon compound to prepare a raw material mixture, and the step of firing the raw material mixture to produce the lithium transition metal complex oxide.
Owner:NIPPON CHECMICAL IND CO LTD
Non-volatile resistance-switching oxide thin film devices
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA
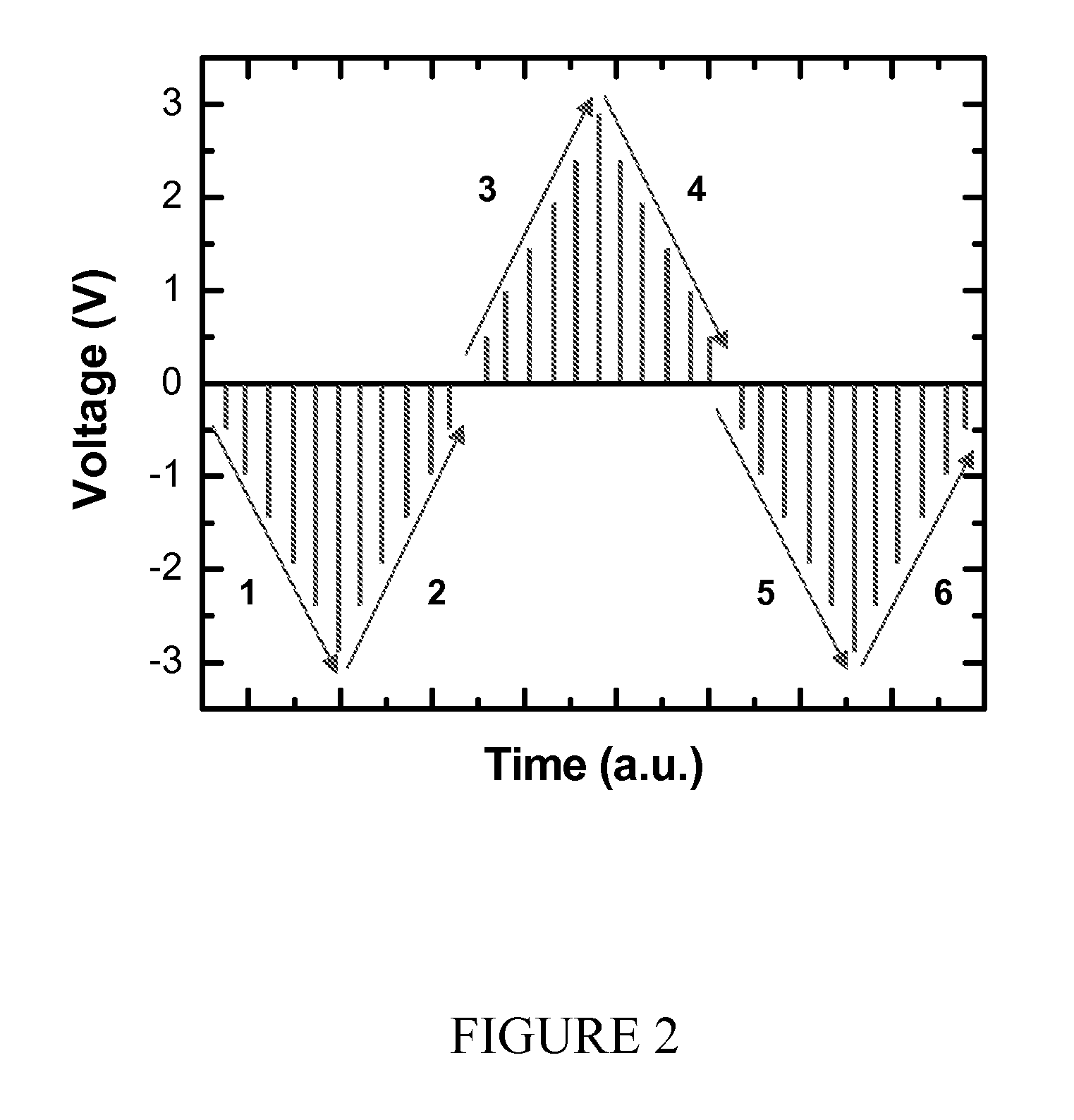
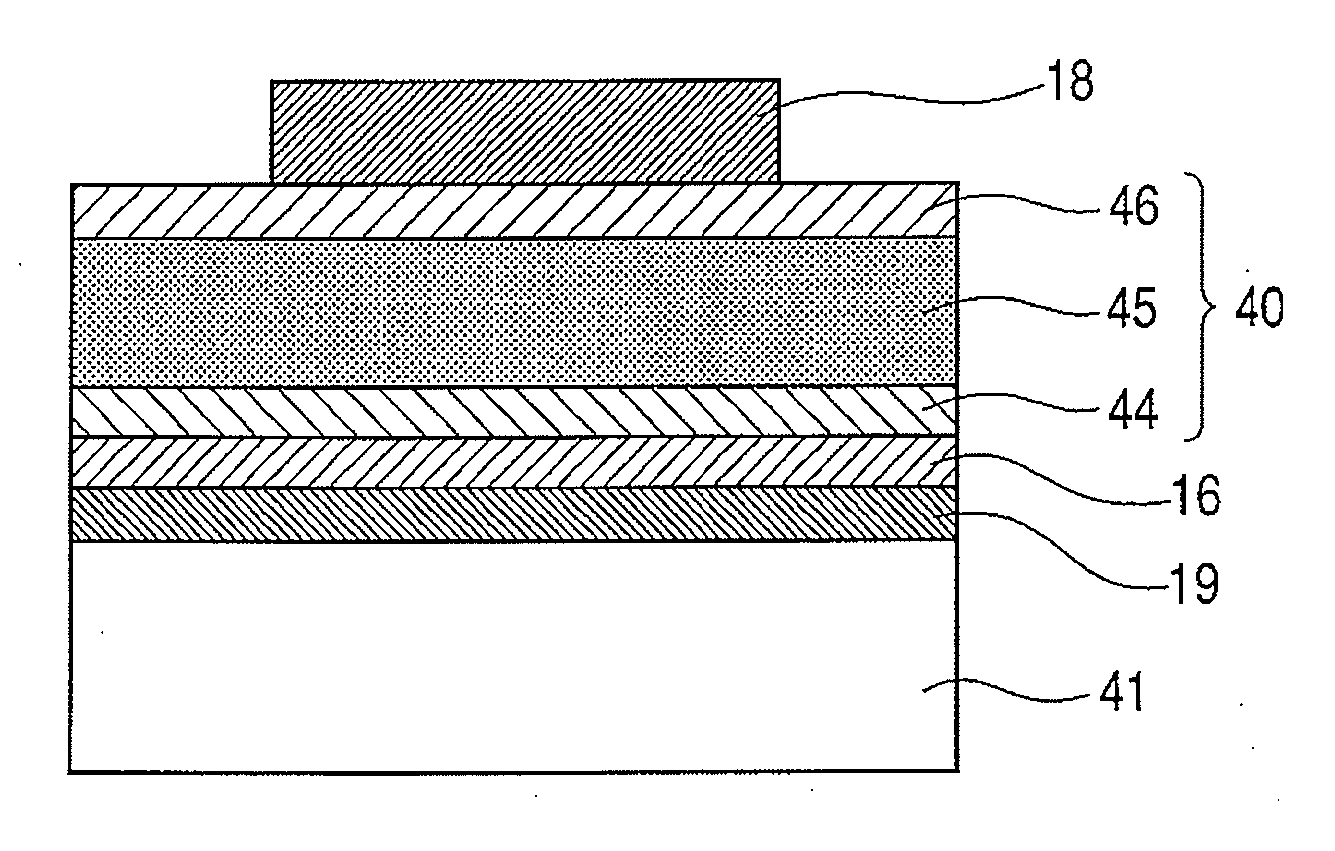
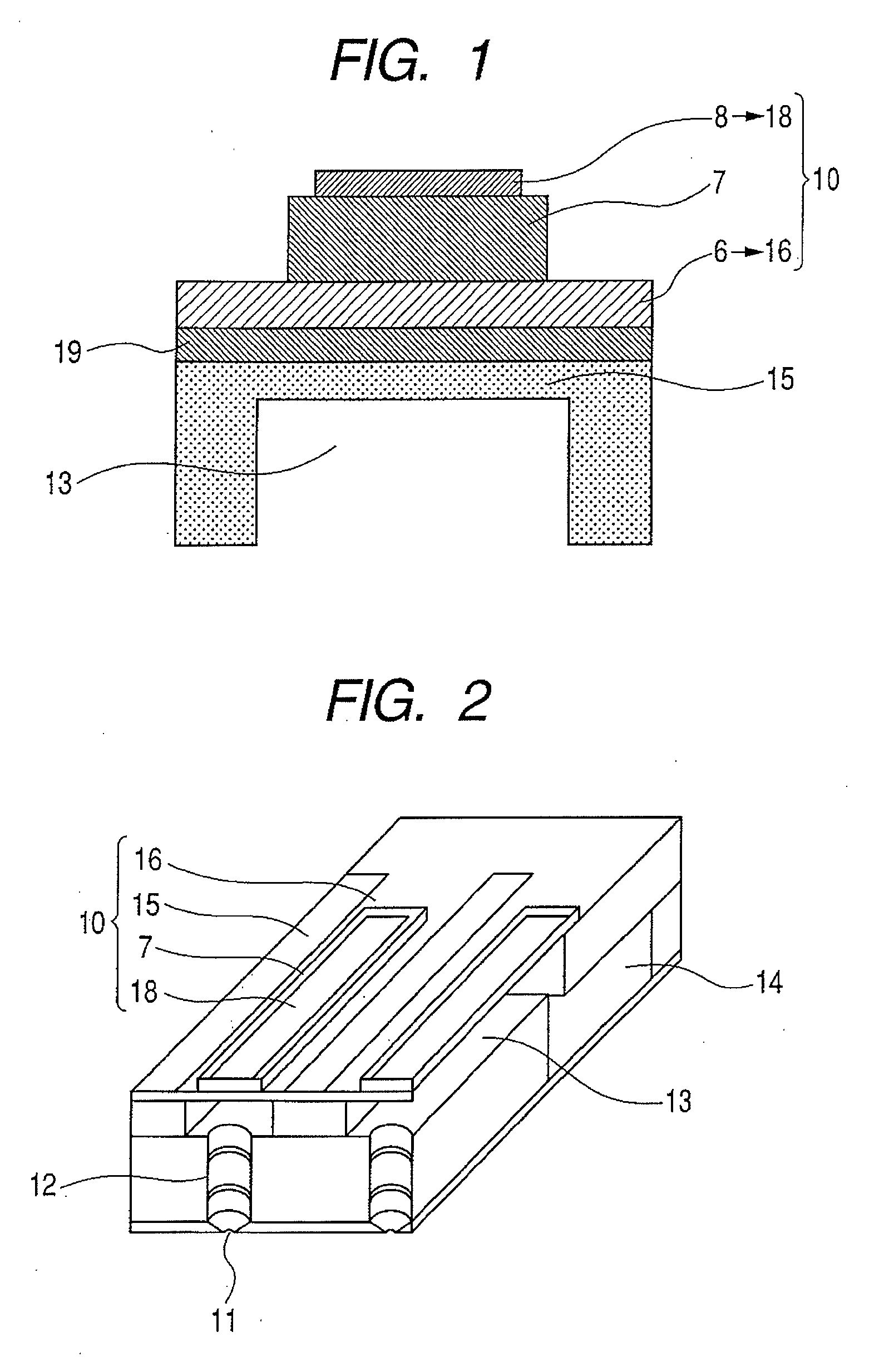
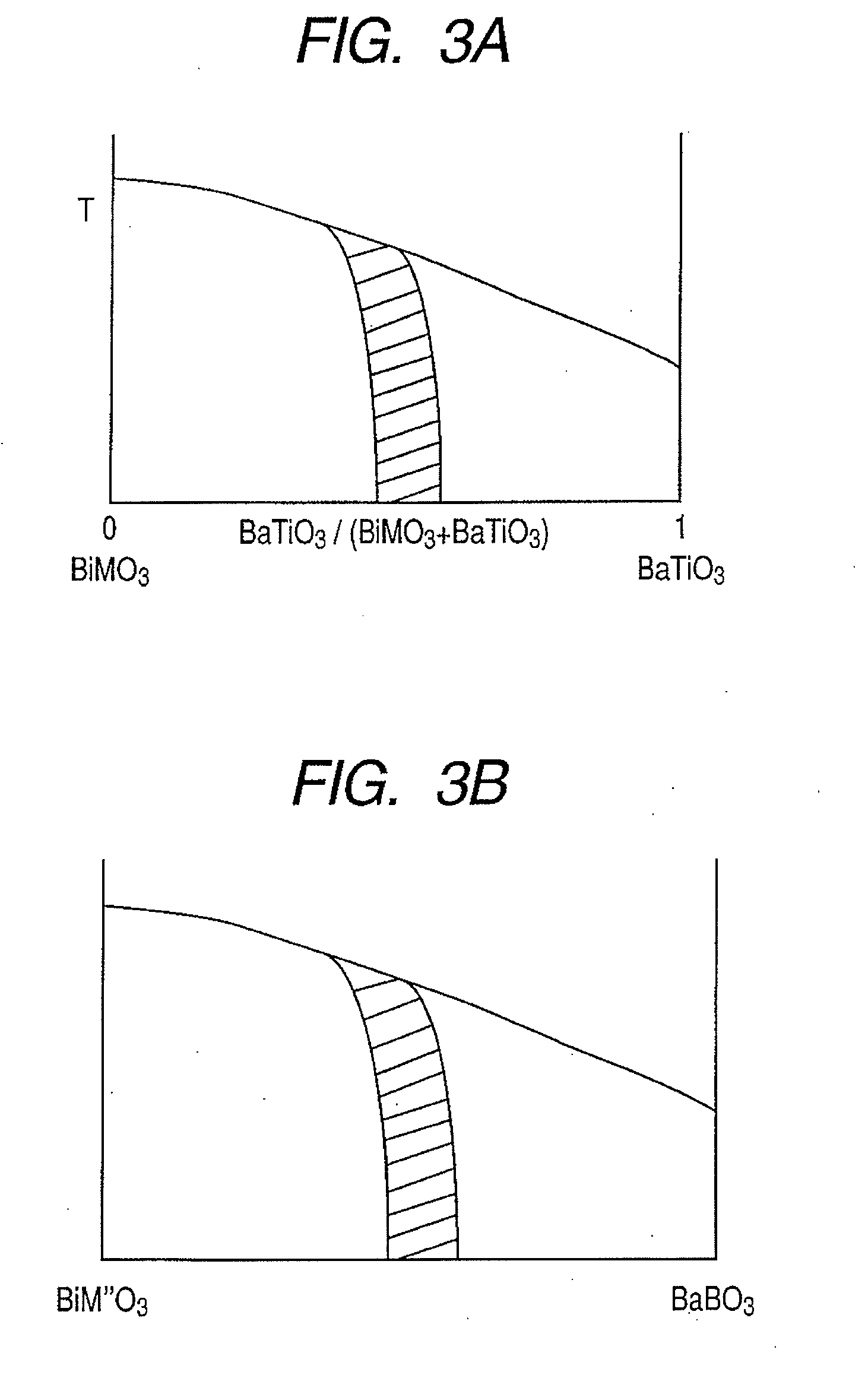
Piezoelectric element, and liquid jet head and ultrasonic motor using the piezoelectric element
InactiveUS20080067898A1Improve featuresInking apparatusPiezoelectric/electrostriction/magnetostriction machinesLiquid jetUltrasonic motor
There is disclosed a piezoelectric element having, on a substrate, a piezoelectric body and a pair of electrodes which come in contact with the piezoelectric body, wherein the piezoelectric body consists of a perovskite type oxide represented by the following formula (1):(Bi,Ba)(M,Ti)O3 (1)in which M is an atom of one element selected from the group consisting of Mn, Cr, Cu, Sc, In, Ga, Yb, Al, Mg, Zn, Co, Zr, Sn, Nb, Ta, and W, or a combination of the atoms of the plurality of elements.
Owner:CANON KK
LiFePO4 FLAKES FOR Li-ION BATTERY AND METHOD FOR MANUFACTURING THE SAME
ActiveUS20120328947A1Charge-discharge efficiency can be improvedShort diffusion pathMaterial nanotechnologyPhosphatesCharge dischargeEngineering
LiFePO4 flakes for a Li-ion battery and a method for manufacturing the same are disclosed. The LiFePO4 flakes of the present invention have a thickness of 5 nm-200 nm, and the angle between the flat surface normal of the flake and the Li-ion diffusion channel is 0°-80°. In addition, according to the present invention, the LiFePO4 flakes with short Li ion diffusion path can be prepared through a simple process. Hence, not only the charge-discharge efficiency of the Li-ion battery can be improved by use of the LiFePO4 flakes of the present invention, but also the cost of the Li-ion battery can be further reduced.
Owner:NATIONAL TSING HUA UNIVERSITY
Constrast media
InactiveUS6423296B1Increase productionPowder deliveryBiocideOxidative cleavageComposite nanoparticles
The invention relates to MR contrast media containing composite nanoparticles, preferably comprising a superparamagnetic iron oxide core provided with a coating comprising an oxidatively cleaved starch coating optionally together with a functionalized polyalkyleneoxide which serves to prolong blood residence.
Owner:GE HEALTHCARE AS