Patents
Literature
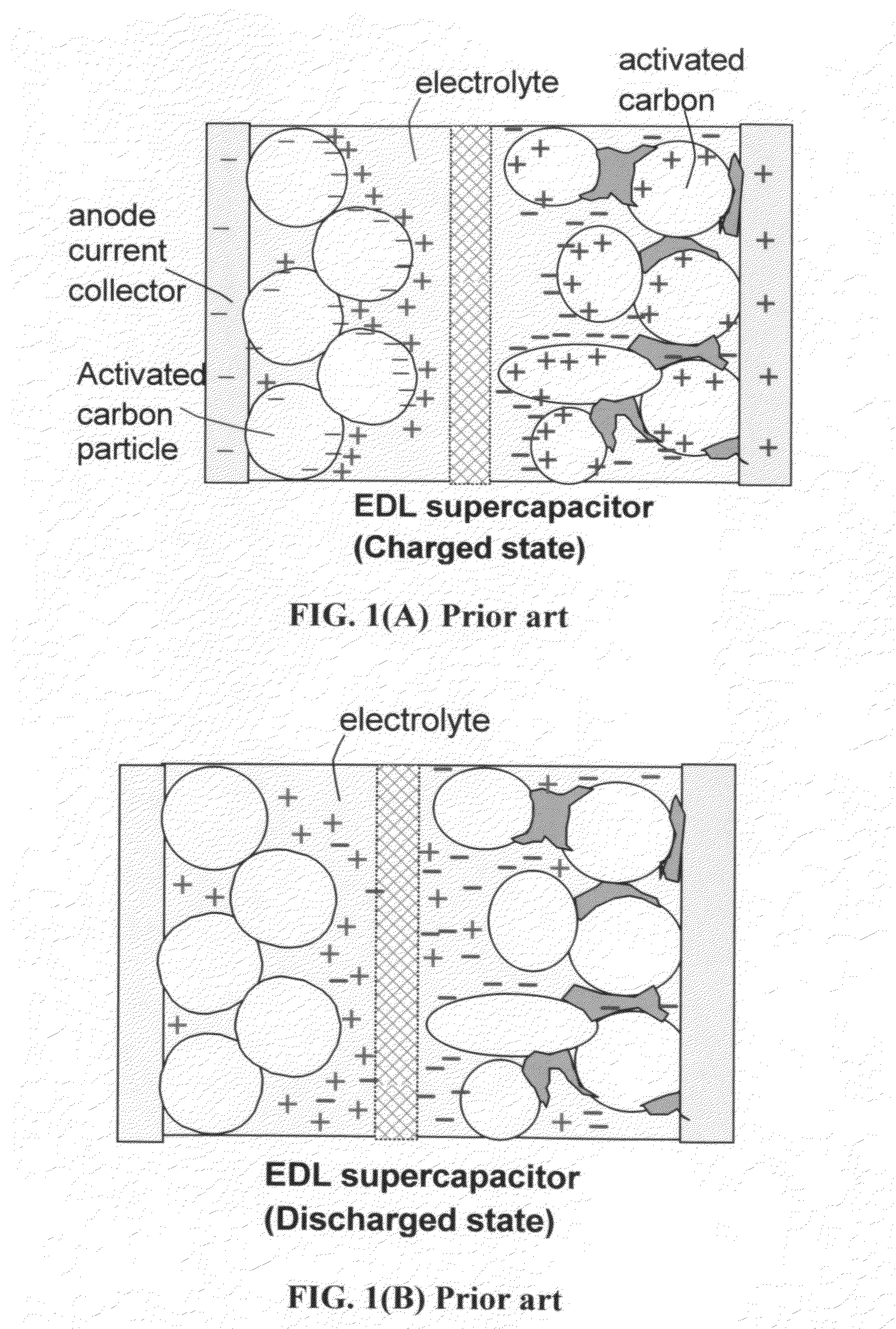
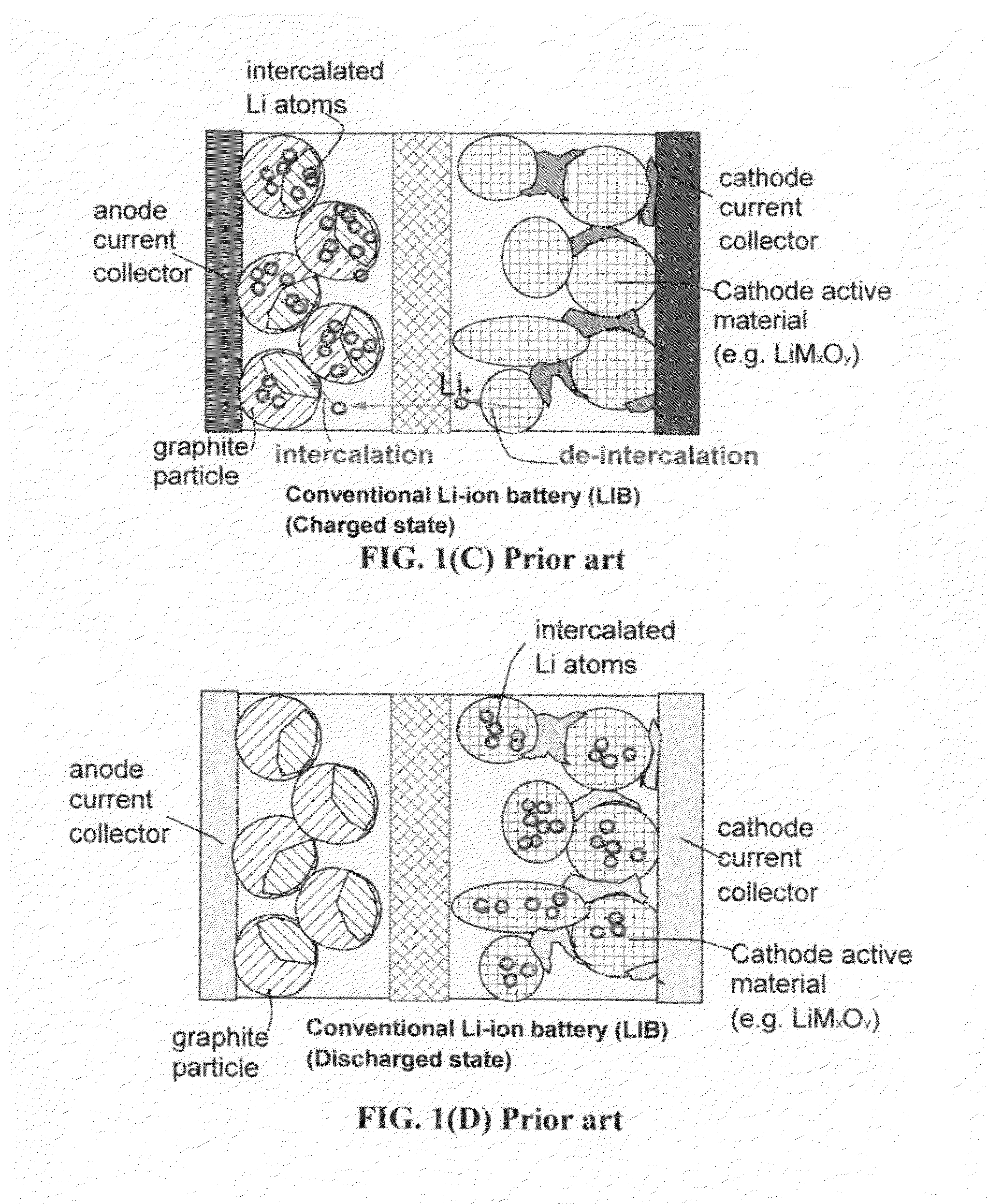
41240 results about "Lithium-ion battery" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
A lithium-ion battery or Li-ion battery (abbreviated as LIB) is a type of rechargeable battery. Lithium-ion batteries are commonly used for portable electronics and electric vehicles and are growing in popularity for military and aerospace applications. The technology was largely developed by John Goodenough, Stanley Whittingham, Rachid Yazami and Akira Yoshino during the 1970s–1980s, and then commercialized by a Sony and Asahi Kasei team led by Yoshio Nishi in 1991.
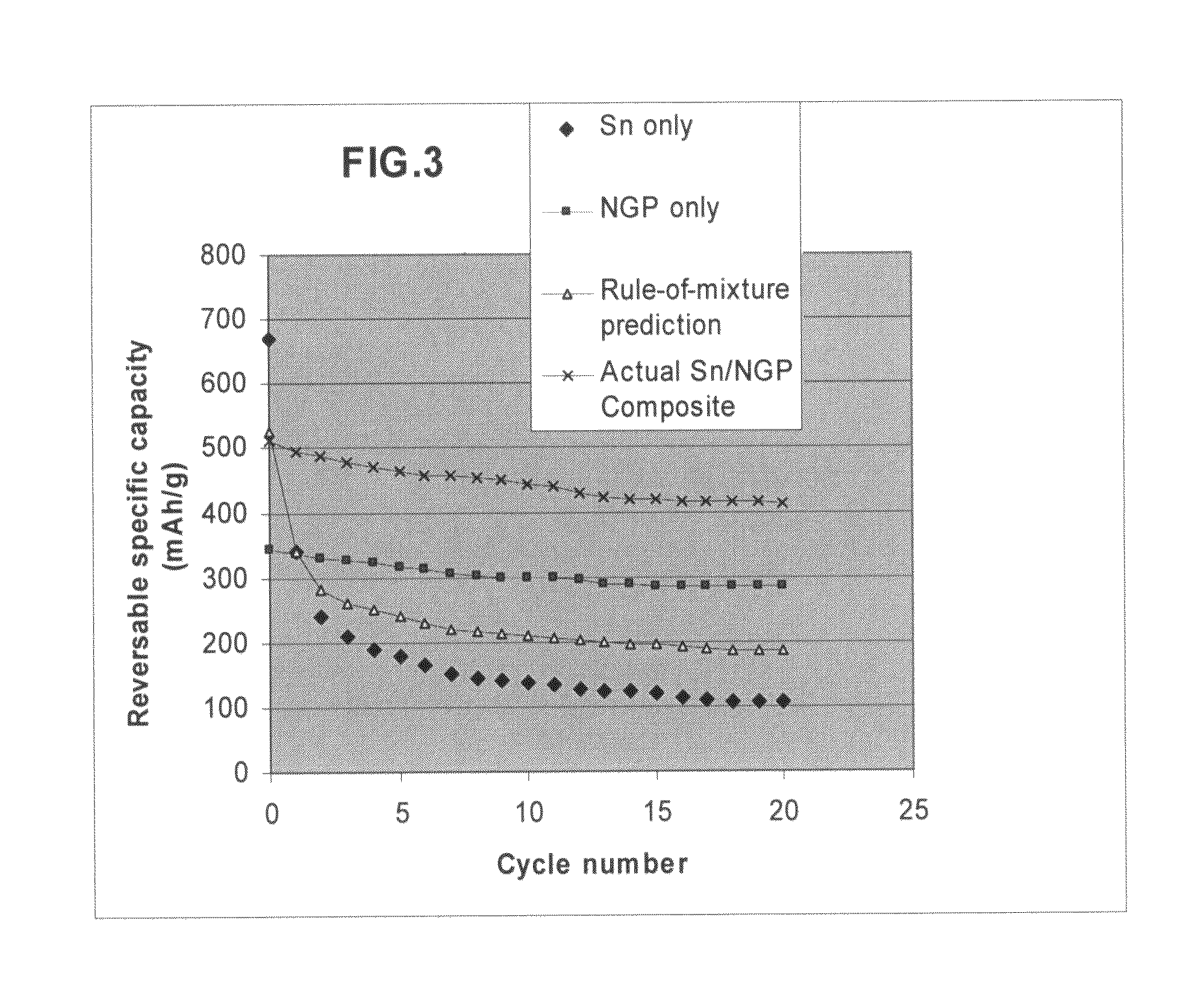
Nano graphene platelet-based composite anode compositions for lithium ion batteries
ActiveUS20090117467A1Improve conductivityLower internal resistanceElectrolytic capacitorsSecondary cellsGraphene flakeGraphite
The present invention provides a nano-scaled graphene platelet-based composite material composition for use as an electrode, particularly as an anode of a lithium ion battery. The composition comprises: (a) micron- or nanometer-scaled particles or coating which are capable of absorbing and desorbing lithium ions; and (b) a plurality of nano-scaled graphene platelets (NGPs), wherein a platelet comprises a graphene sheet or a stack of graphene sheets having a platelet thickness less than 100 nm; wherein at least one of the particles or coating is physically attached or chemically bonded to at least one of the graphene platelets and the amount of platelets is in the range of 2% to 90% by weight and the amount of particles or coating in the range of 98% to 10% by weight. Also provided is a lithium secondary battery comprising such a negative electrode (anode). The battery exhibits an exceptional specific capacity, an excellent reversible capacity, and a long cycle life.
Owner:SAMSUNG ELECTRONICS CO LTD
Methods and reagents for enhancing the cycling efficiency of lithium polymer batteries
InactiveUS6017651AImprove efficiencyElectrode rolling/calenderingElectrochemical processing of electrodesLithium metalSulfur electrode
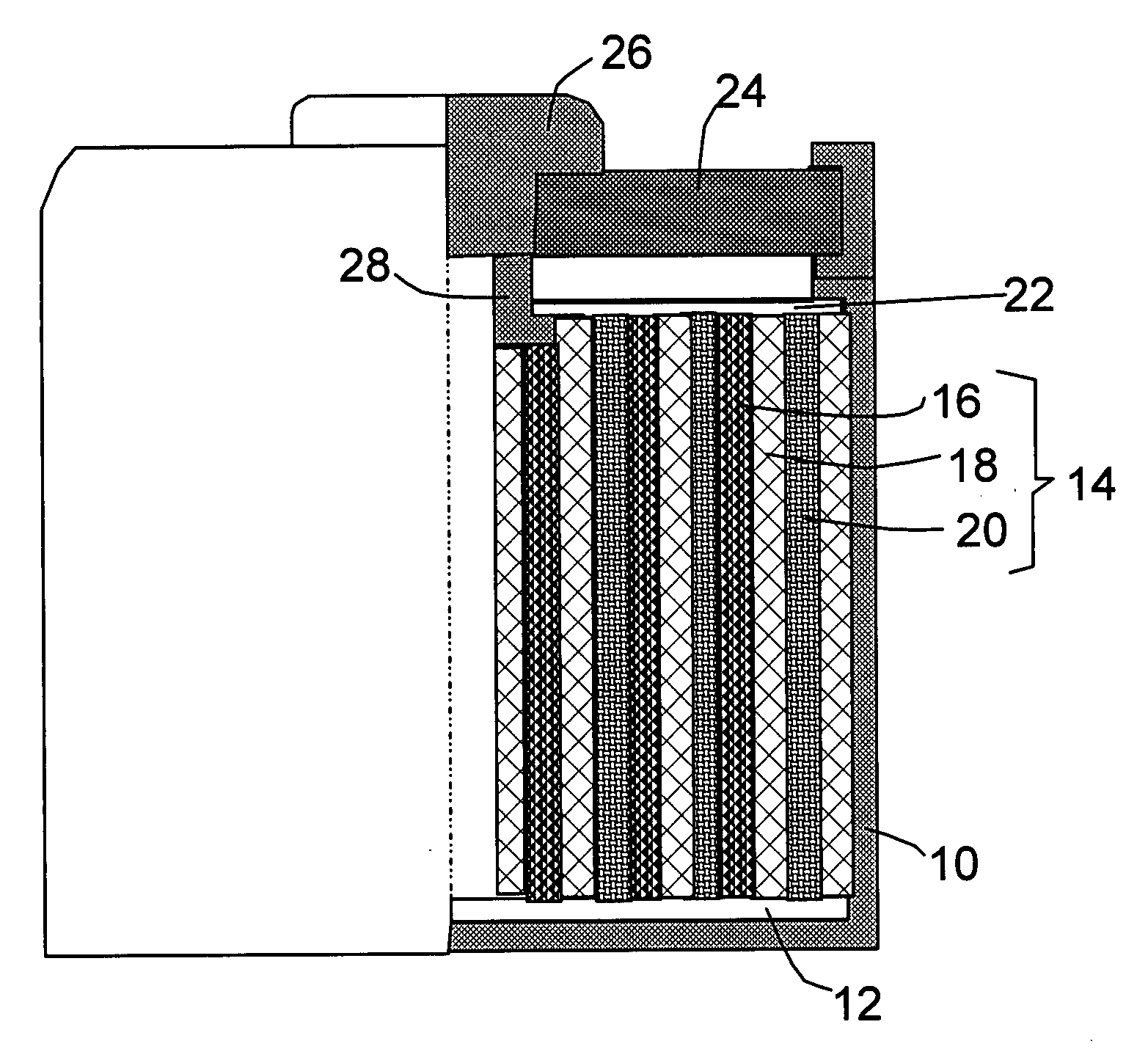
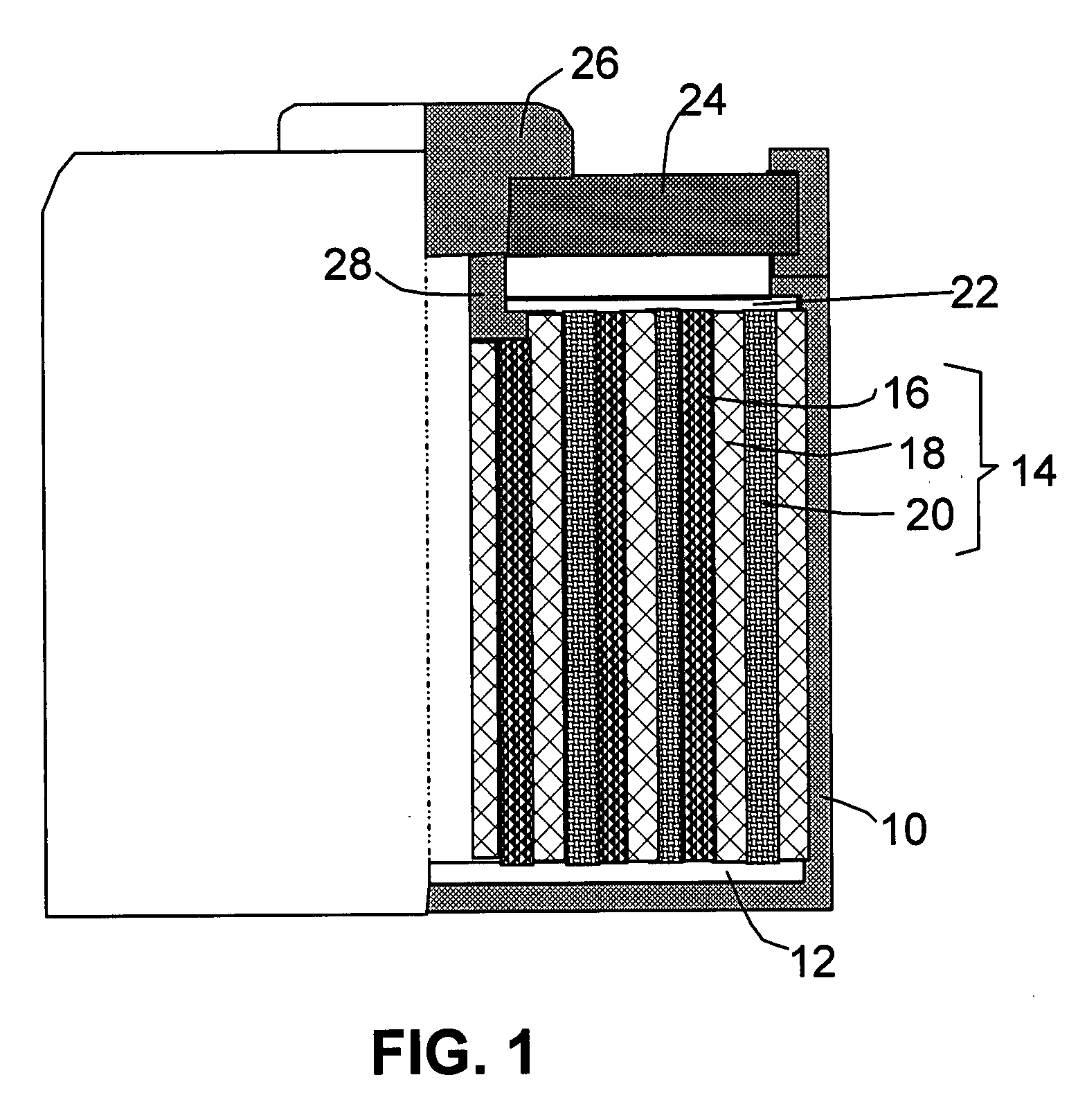
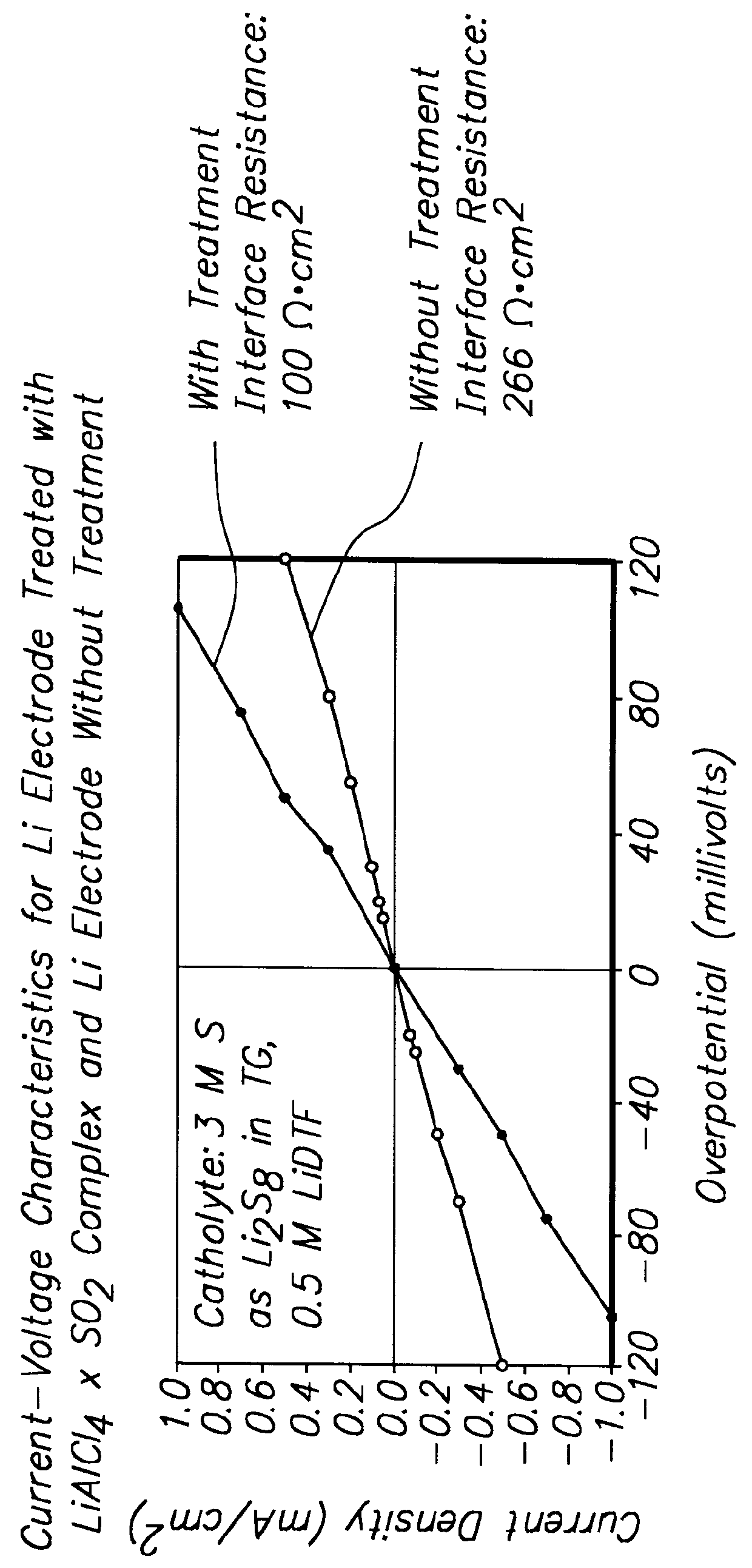
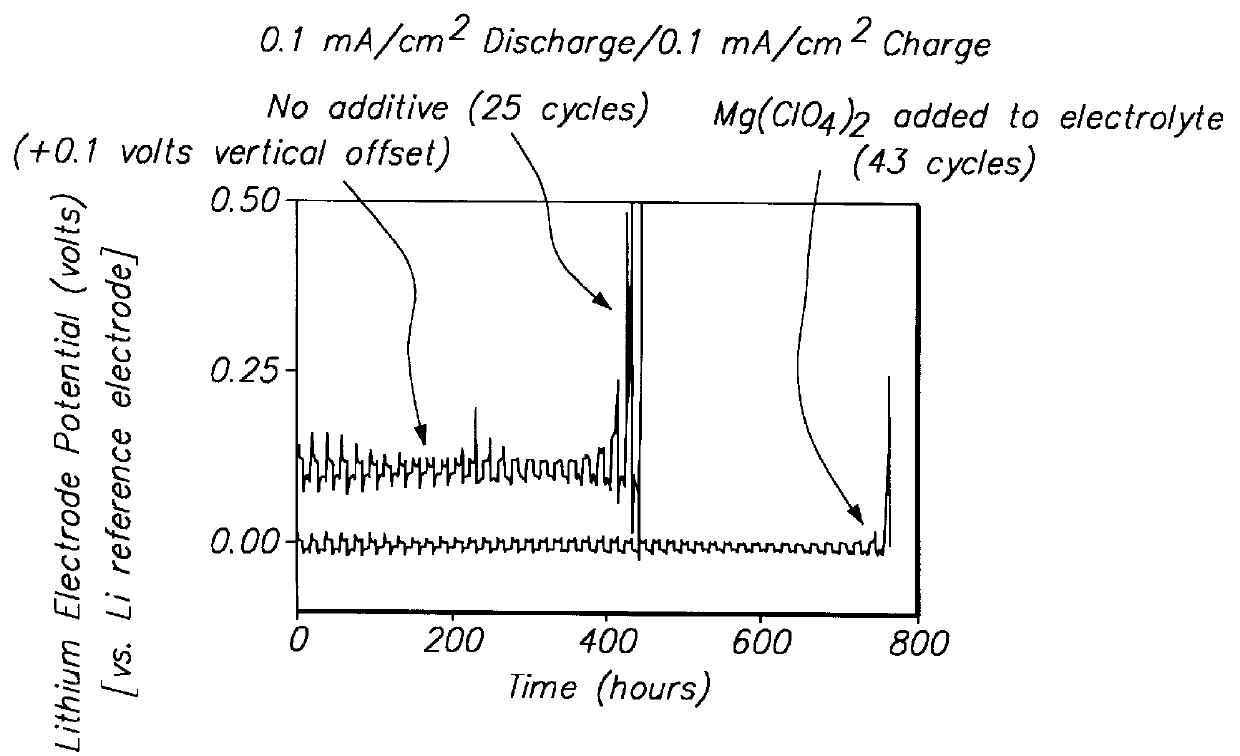
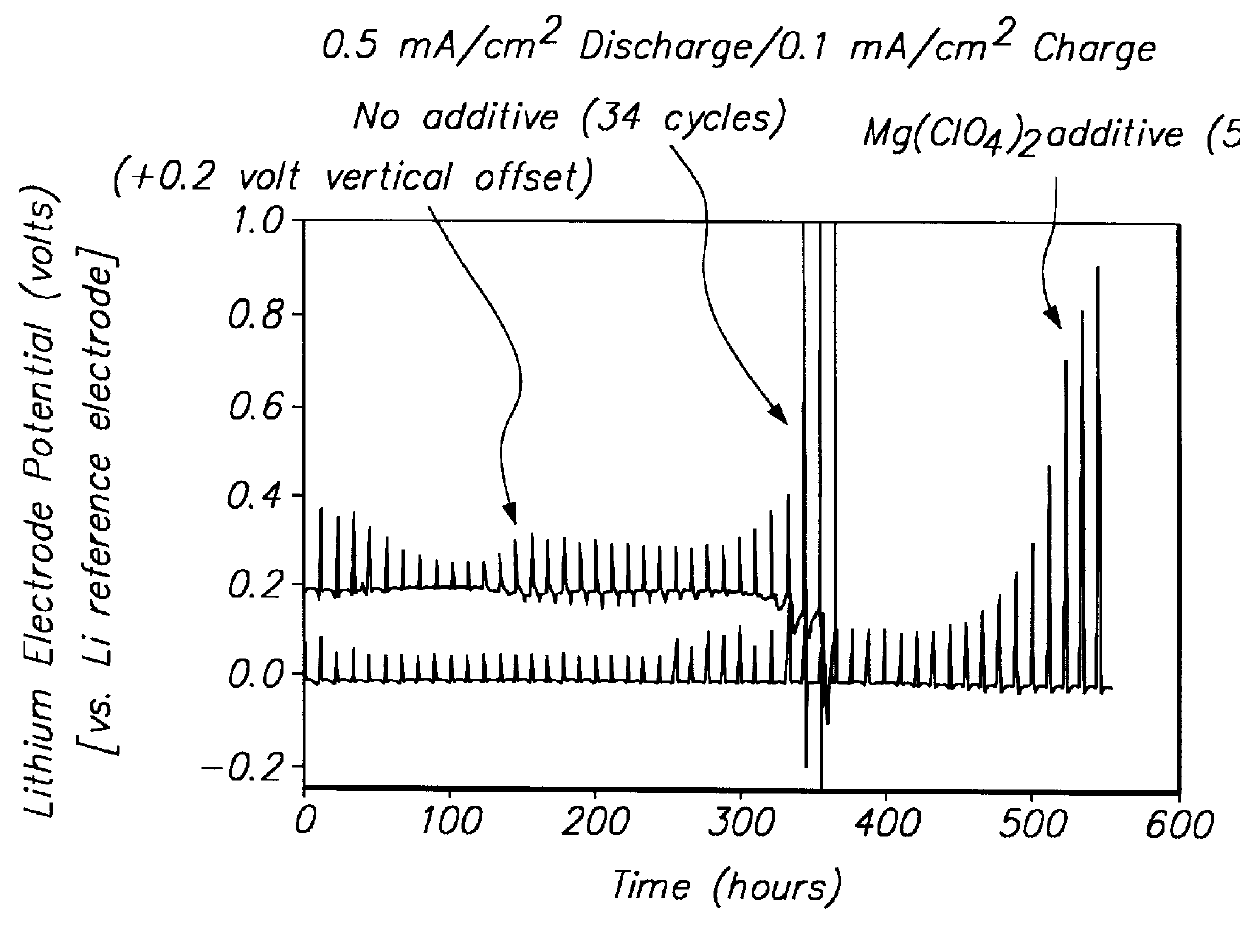
Batteries including a lithium electrode and a sulfur counter electrode that demonstrate improved cycling efficiencies are described. In one embodiment, an electrochemical cell having a lithium electrode and a sulfur electrode including at least one of elemental sulfur, lithium sulfide, and a lithium polysulfide is provided. The lithium electrode includes a surface coating that is effective to increase the cycling efficiency of said electrochemical cell. In a more particular embodiment, the lithium electrode is in an electrolyte solution, and, more particularly, an electrolyte solution including either elemental sulfur, a sulfide, or a polysulfide. In another embodiment, the coating is formed after the lithium electrode is contacted with the electrolyte. In a more particular embodiment, the coating is formed by a reaction between the lithium metal of the lithium electrode and a chemical species present in the electrolyte.
Owner:POLYPLUS BATTERY CO INC
Electrode Including Nanostructures for Rechargeable Cells
InactiveUS20100285358A1Lower resistanceNanostructure manufactureMicroscopic fiber electrodesRechargeable cellSilicon nanowires
A lithium ion battery electrode includes silicon nanowires used for insertion of lithium ions and including a conductivity enhancement, the nanowires growth-rooted to the conductive substrate.
Owner:AMPRIUS INC
High Capacity Anode Materials for Lithium Ion Batteries
High capacity silicon based anode active materials are described for lithium ion batteries. These materials are shown to be effective in combination with high capacity lithium rich cathode active materials. Supplemental lithium is shown to improve the cycling performance and reduce irreversible capacity loss for at least certain silicon based active materials. In particular silicon based active materials can be formed in composites with electrically conductive coatings, such as pyrolytic carbon coatings or metal coatings, and composites can also be formed with other electrically conductive carbon components, such as carbon nanofibers and carbon nanoparticles. Additional alloys with silicon are explored.
Owner:IONBLOX INC
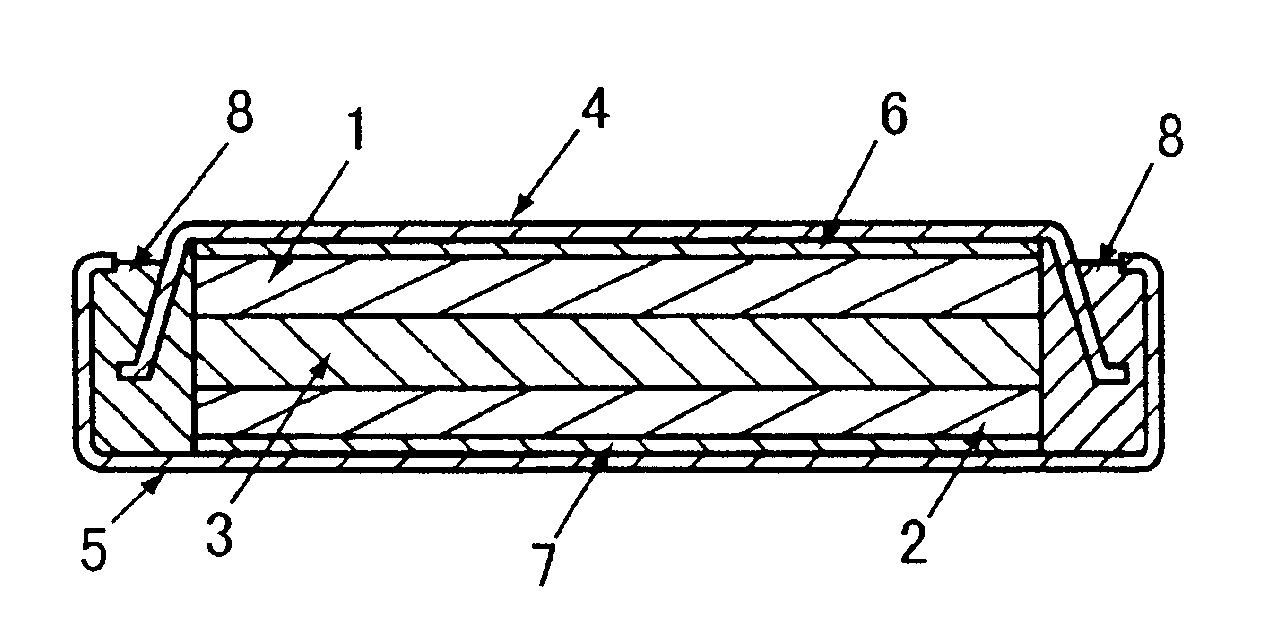
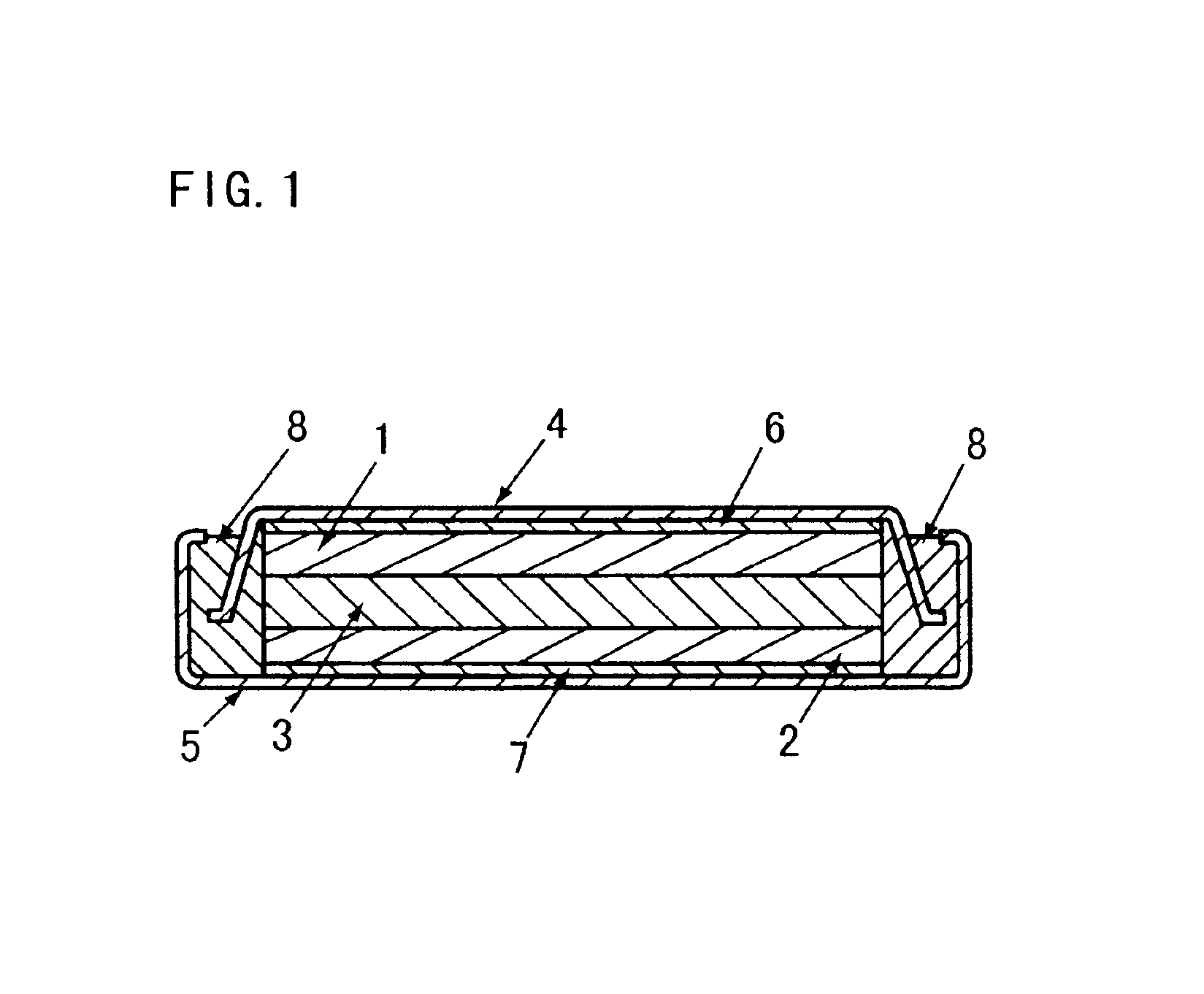
Laminated lithium ion battery, battery pack comprising same and pole piece of laminated lithium ion battery
InactiveCN104882635AImprove cooling effectReduce welding processFinal product manufactureElectrode carriers/collectorsInternal resistanceElectrical battery
Disclosed are a laminated lithium ion battery, a battery pack comprising the same and a pole piece of the laminated lithium ion battery. The pole piece comprises a current collector and an active material layer, wherein the current collector is coated with the active material layer. A section of continuous uncoated area is arranged at the tail end of a first width end portion of the pole piece, the top face and the bottom face of the uncoated area are not coated with the active material layer, the current collector is exposed in the uncoated area, and the exposed current collector serves as a pole lug of the pole piece. By the pole piece, internal resistance of the lithium ion battery is reduced and heat dissipation performance of the battery is improved.
Owner:SHENZHEN GREPOW BATTERY CO LTD
Porous silicon particulates for lithium batteries
An anode structure for lithium batteries includes nanofeatured silicon particulates dispersed in a conductive network. The particulates are preferably made from metallurgical grade silicon powder via HF / HNO3 acid treatment, yielding crystallite sizes from about 1 to 20 nm and pore sizes from about 1 to 100 nm. Surfaces of the particles may be terminated with selected chemical species to further modify the anode performance characteristics. The conductive network is preferably a carbonaceous material or composite, but it may alternatively contain conductive ceramics such as TiN or B4C. The anode structure may further contain a current collector of copper or nickel mesh or foil.
Owner:TIEGS TERRY N
Method of depositing silicon on carbon materials and forming an anode for use in lithium ion batteries
ActiveUS20080261116A1Increase rangeMaterial nanotechnologyNanoinformaticsPhysical chemistryCarbon nanofiber
A method of modifying the surface of carbon materials such as vapor grown carbon nanofibers is provided in which silicon is deposited on vapor grown carbon nanofibers using a chemical vapor deposition process. The resulting silicon-carbon alloy may be used as an anode in a rechargeable lithium ion battery.
Owner:APPLIED SCI
Nano graphene platelet-base composite anode compositions for lithium ion batteries
ActiveUS7745047B2Improve conductivityLower internal resistanceAlkaline accumulatorsElectrolytic capacitorsGraphiteGraphene
The present invention provides a nano-scaled graphene platelet-based composite material composition for use as an electrode, particularly as an anode of a lithium ion battery. The composition comprises: (a) micron- or nanometer-scaled particles or coating which are capable of absorbing and desorbing lithium ions; and (b) a plurality of nano-scaled graphene platelets (NGPs), wherein a platelet comprises a graphene sheet or a stack of graphene sheets having a platelet thickness less than 100 nm; wherein at least one of the particles or coating is physically attached or chemically bonded to at least one of the graphene platelets and the amount of platelets is in the range of 2% to 90% by weight and the amount of particles or coating in the range of 98% to 10% by weight. Also provided is a lithium secondary battery comprising such a negative electrode (anode). The battery exhibits an exceptional specific capacity, an excellent reversible capacity, and a long cycle life.
Owner:SAMSUNG ELECTRONICS CO LTD
Hybrid anode compositions for lithium ion batteries
ActiveUS20090117466A1Superior multiple-cycle behaviorSmall capacity fadeAlkaline accumulatorsConductive materialHybrid materialSodium-ion battery
The present invention provides an exfoliated graphite-based hybrid material composition for use as an electrode, particularly as an anode of a lithium ion battery. The composition comprises: (a) micron- or nanometer-scaled particles or coating which are capable of absorbing and desorbing alkali or alkaline metal ions (particularly, lithium ions); and (b) exfoliated graphite flakes that are substantially interconnected to form a porous, conductive graphite network comprising pores, wherein at least one of the particles or coating resides in a pore of the network or attached to a flake of the network and the exfoliated graphite amount is in the range of 5% to 90% by weight and the amount of particles or coating is in the range of 95% to 10% by weight. Also provided is a lithium secondary battery comprising such a negative electrode (anode). The battery exhibits an exceptional specific capacity, excellent reversible capacity, and long cycle life.
Owner:SAMSUNG ELECTRONICS CO LTD
Si/c composite, anode active materials, and lithium battery including the same
ActiveUS20090029256A1Improve initial Coulombic efficiencyGood capacity retentionLiquid surface applicatorsElectrode manufacturing processesLithium-ion batteryPorous silicon
An Si / C composite includes carbon (C) dispersed in porous silicon (Si) particles. The Si / C composite may be used to form an anode active material to provide a lithium battery having a high capacity and excellent capacity retention.
Owner:SAMSUNG SDI CO LTD
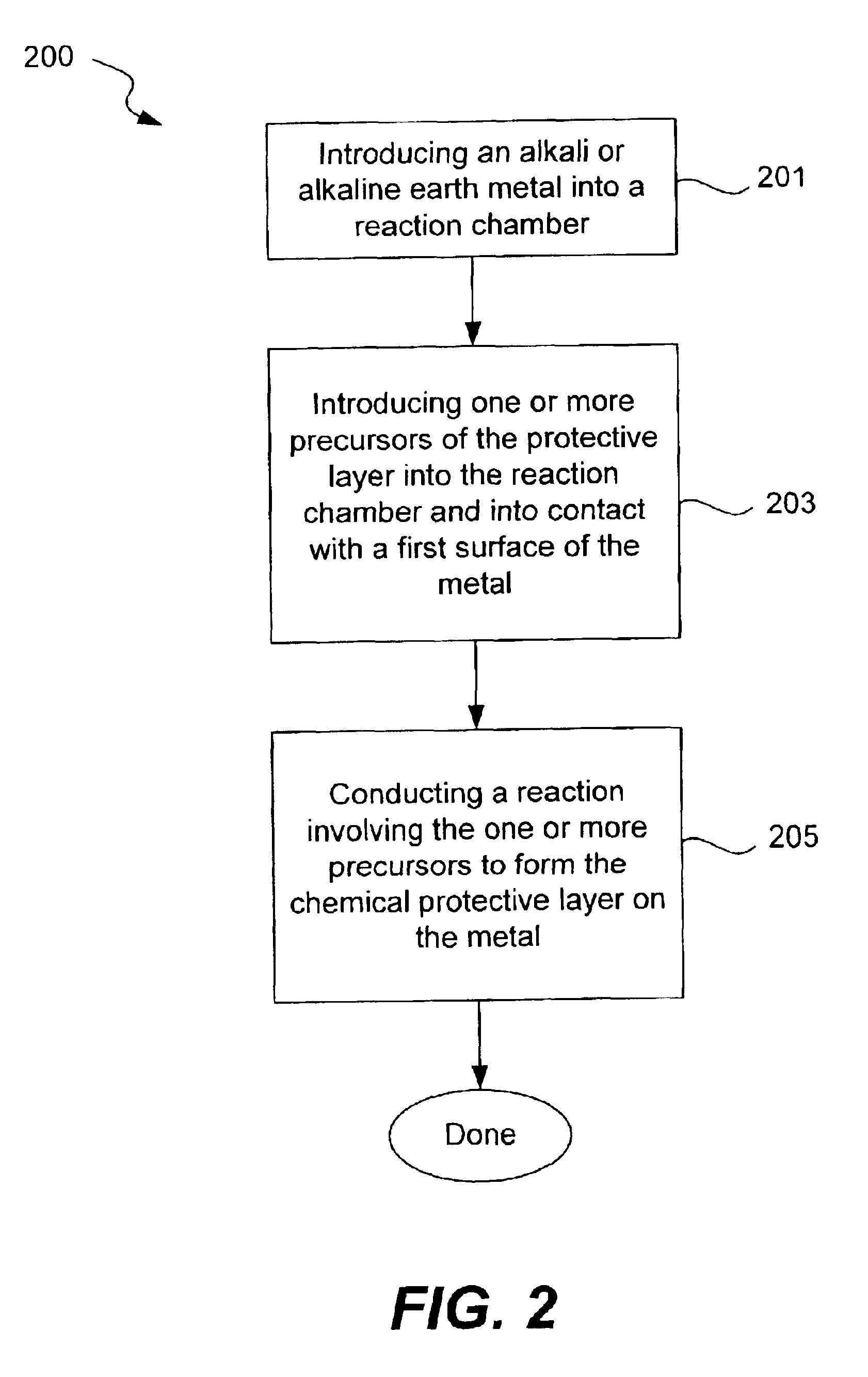
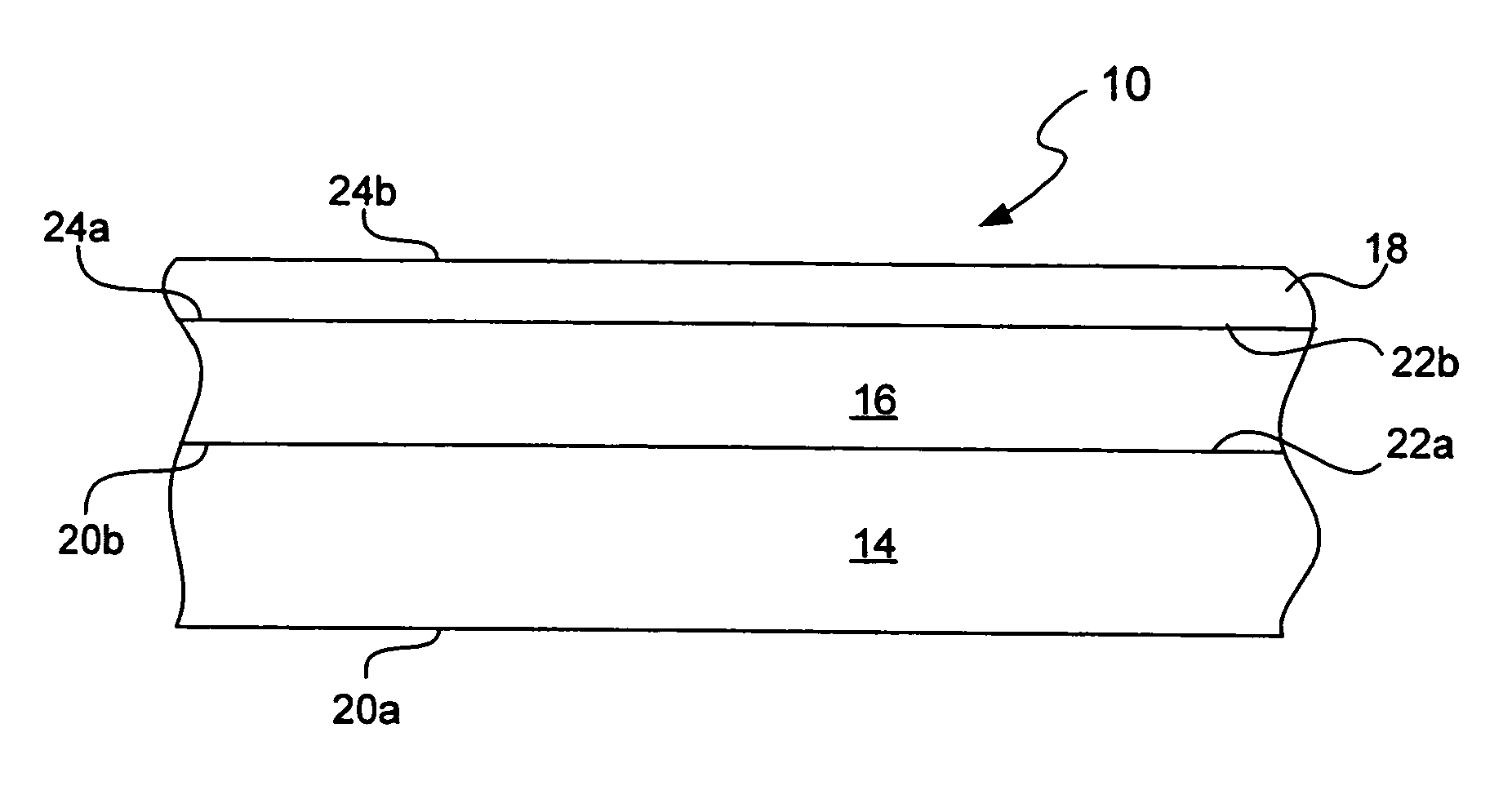
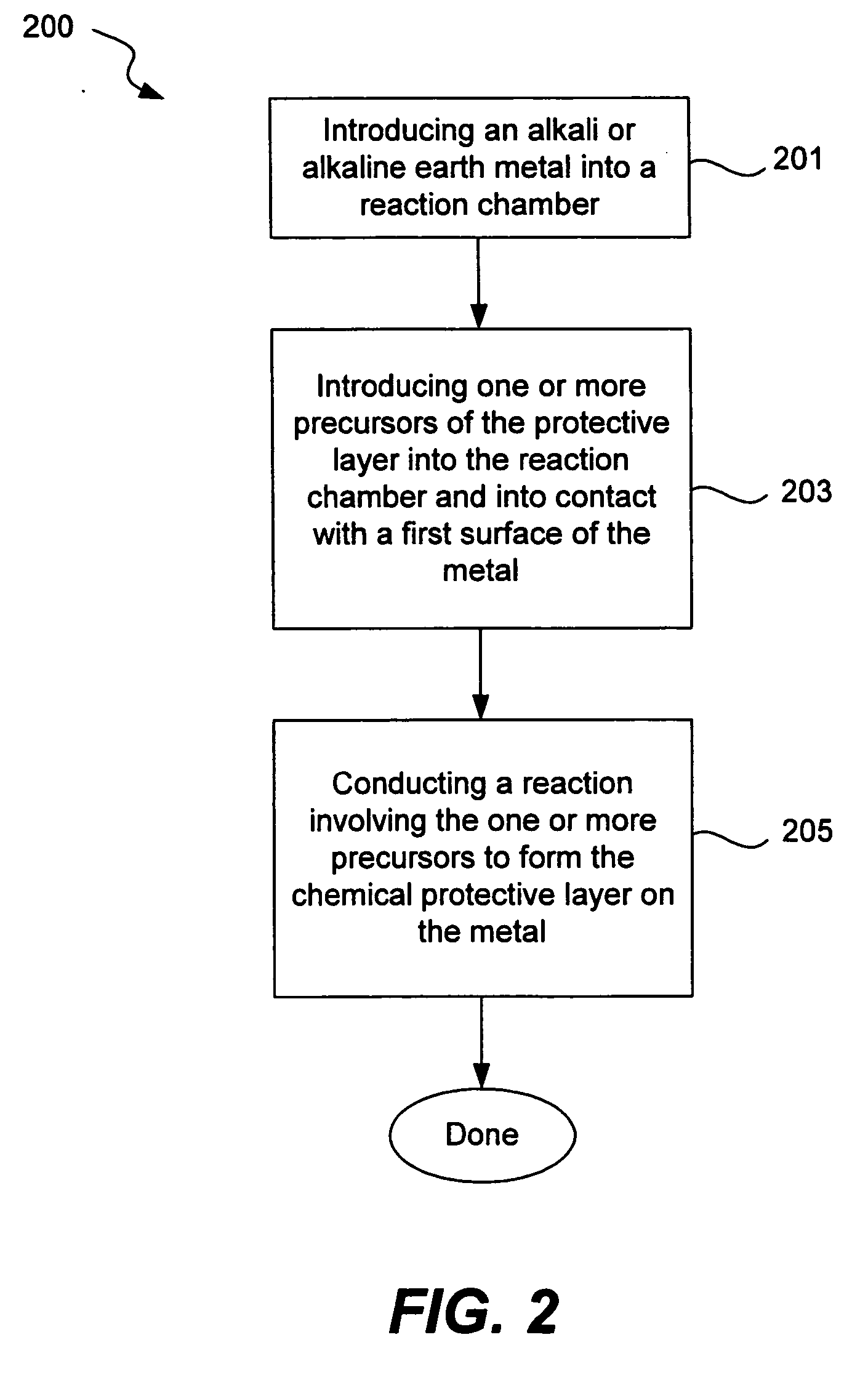
Chemical protection of a lithium surface
InactiveUS6911280B1Easy to produceSimple processElectrochemical processing of electrodesFinal product manufactureAlkaline earth metalLithium metal
Disclosed are compositions and methods for alleviating the problem of reaction of lithium or other alkali or alkaline earth metals with incompatible processing and operating environments by creating a ionically conductive chemical protective layer on the lithium or other reactive metal surface. Such a chemically produced surface layer can protect lithium metal from reacting with oxygen, nitrogen or moisture in ambient atmosphere thereby allowing the lithium material to be handled outside of a controlled atmosphere, such as a dry room. Production processes involving lithium are thereby very considerably simplified. One example of such a process in the processing of lithium to form negative electrodes for lithium metal batteries.
Owner:POLYPLUS BATTERY CO INC
Hybrid electrode and surface-mediated cell-based super-hybrid energy storage device containing same
PendingUS20130171502A1Primary cell to battery groupingMaterial nanotechnologyHigh energyLithium metal
The present invention provides a multi-component hybrid electrode for use in an electrochemical super-hybrid energy storage device. The hybrid electrode contains at least a current collector, at least an intercalation electrode active material storing lithium inside interior or bulk thereof, and at least an intercalation-free electrode active material having a specific surface area no less than 100 m2 / g and storing lithium on a surface thereof, wherein the intercalation electrode active material and the intercalation-free electrode active material are in electronic contact with the current collector. The resulting super-hybrid cell exhibits exceptional high power and high energy density, and long-term cycling stability that cannot be achieved with conventional supercapacitors, lithium-ion capacitors, lithium-ion batteries, and lithium metal secondary batteries.
Owner:GLOBAL GRAPHENE GRP INC +1
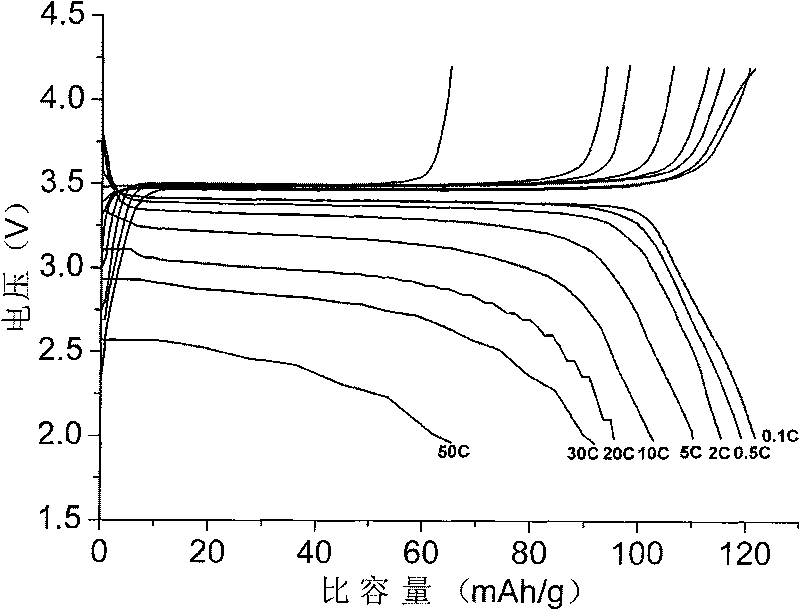
Positive electrode materials for lithium ion batteries having a high specific discharge capacity and processes for the synthesis of these materials
ActiveUS20100086853A1Electrode manufacturing processesAlkali metal oxidesDischarge rateLithium-ion battery
Owner:IONBLOX INC
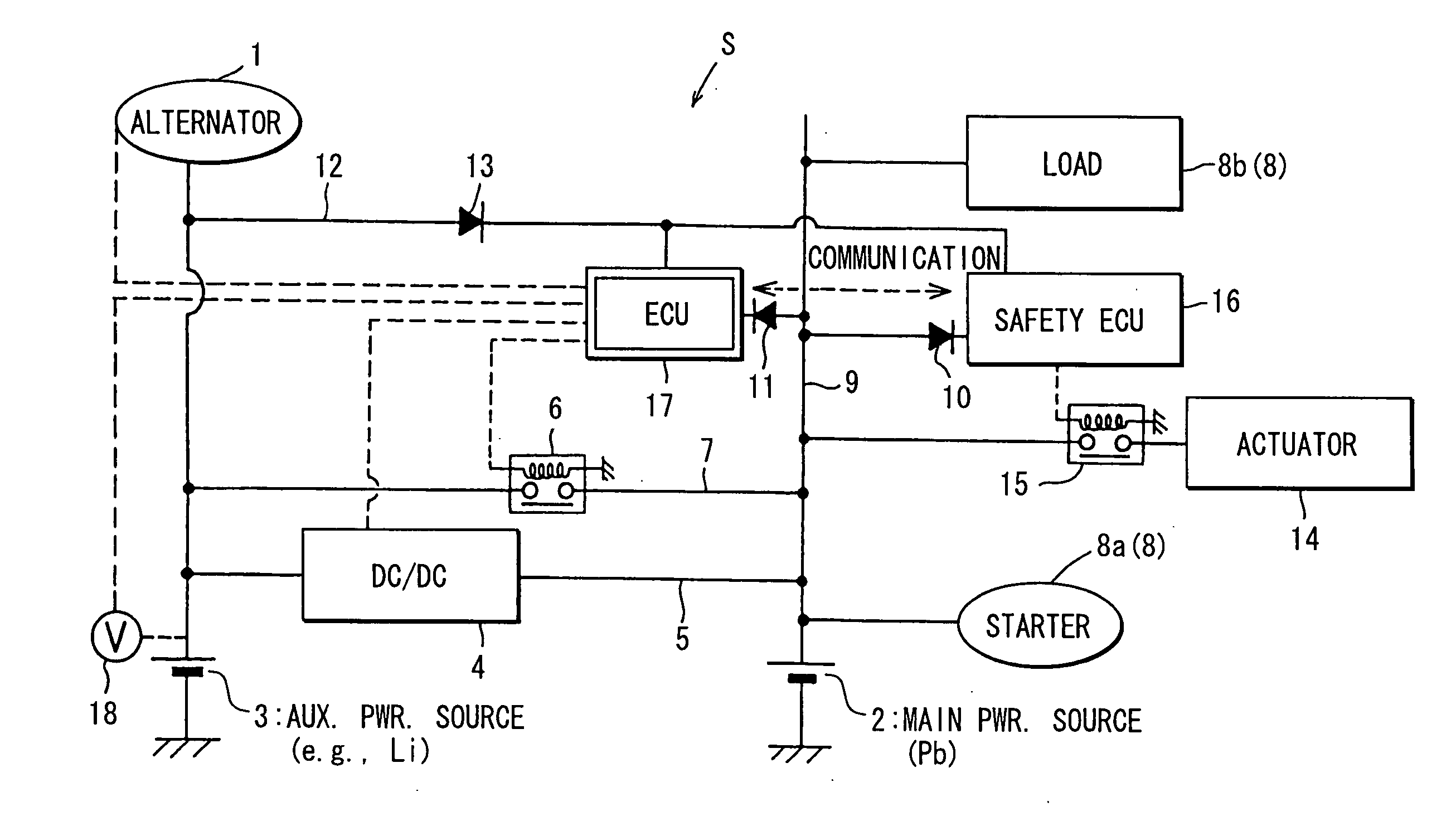
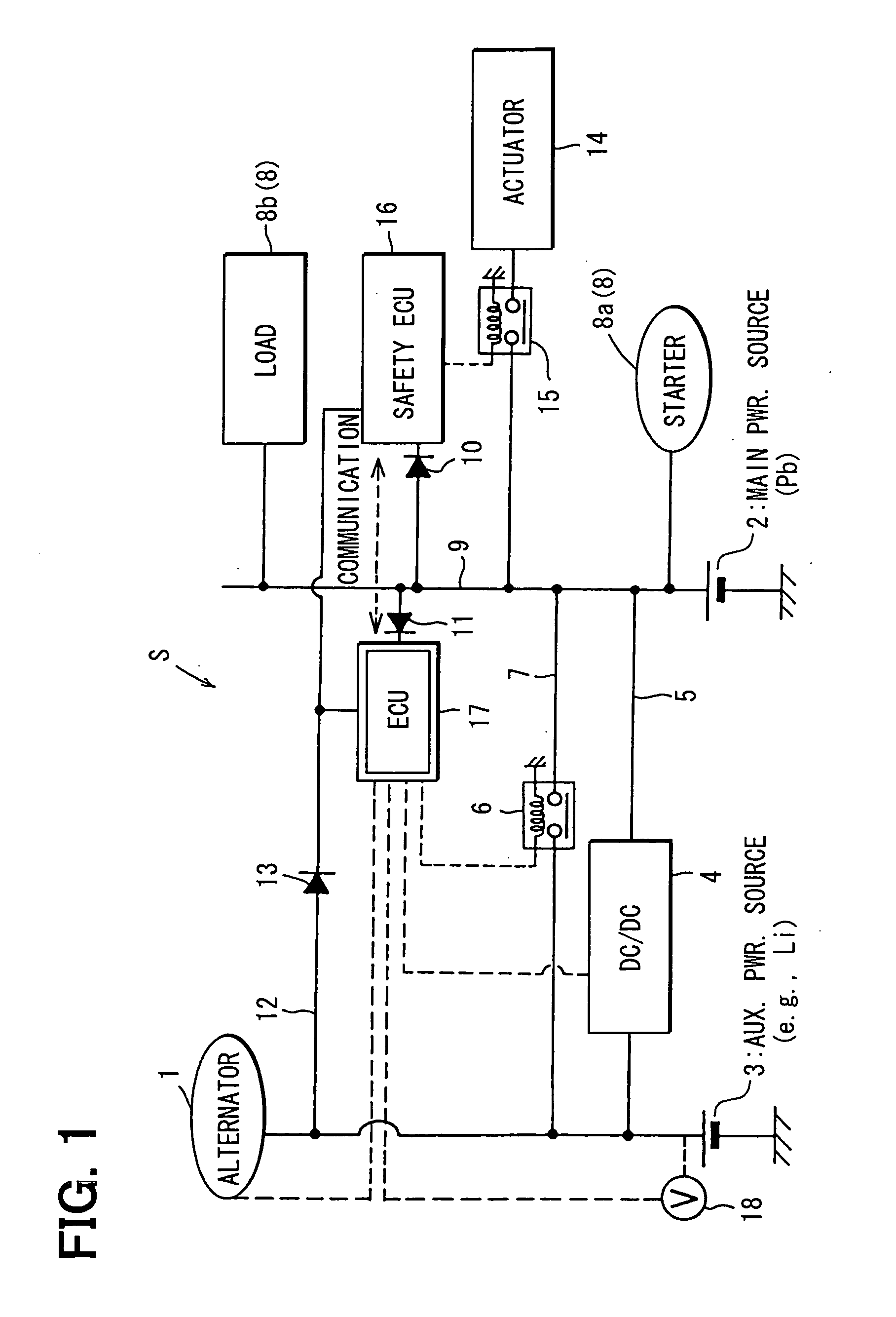
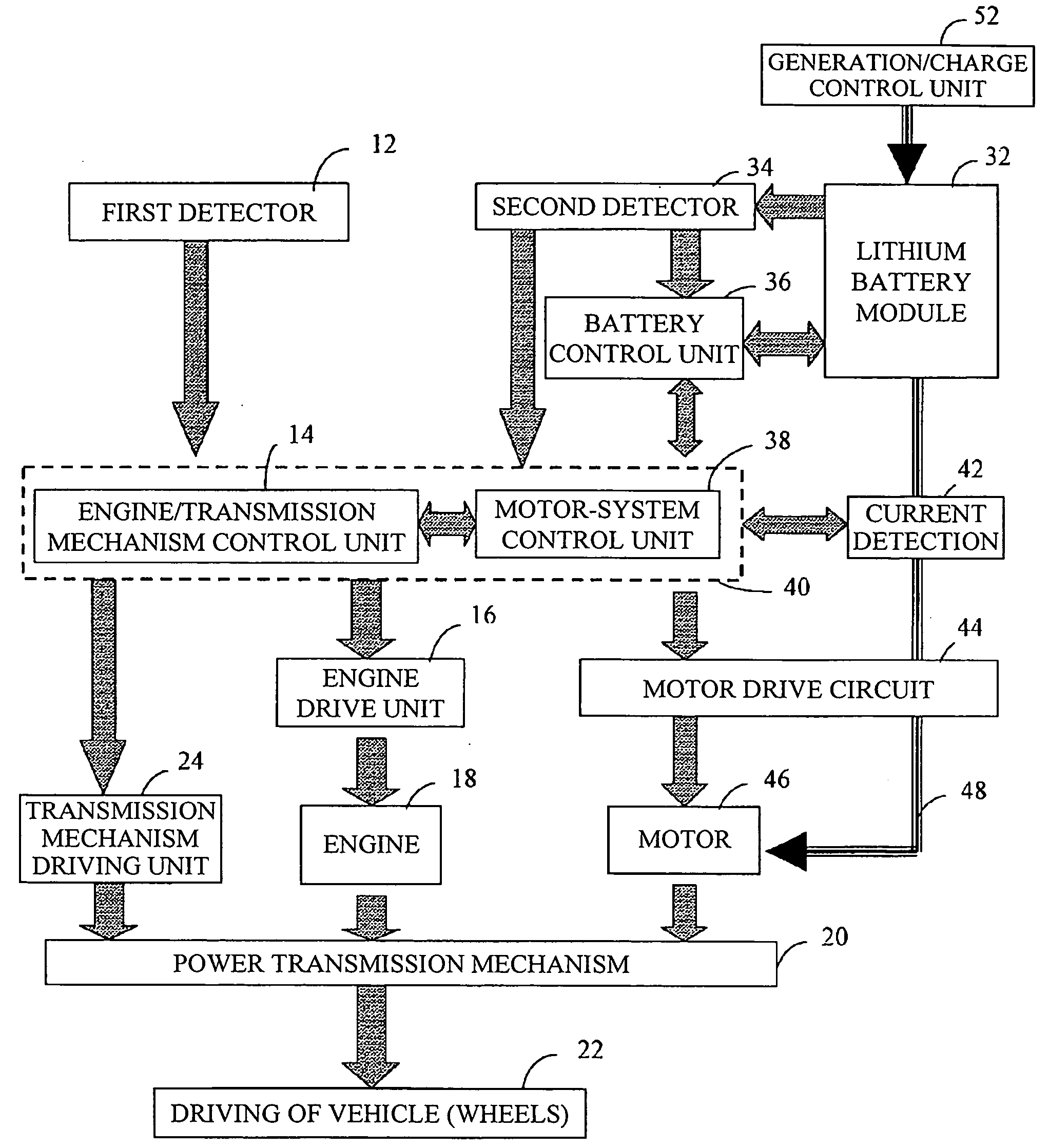
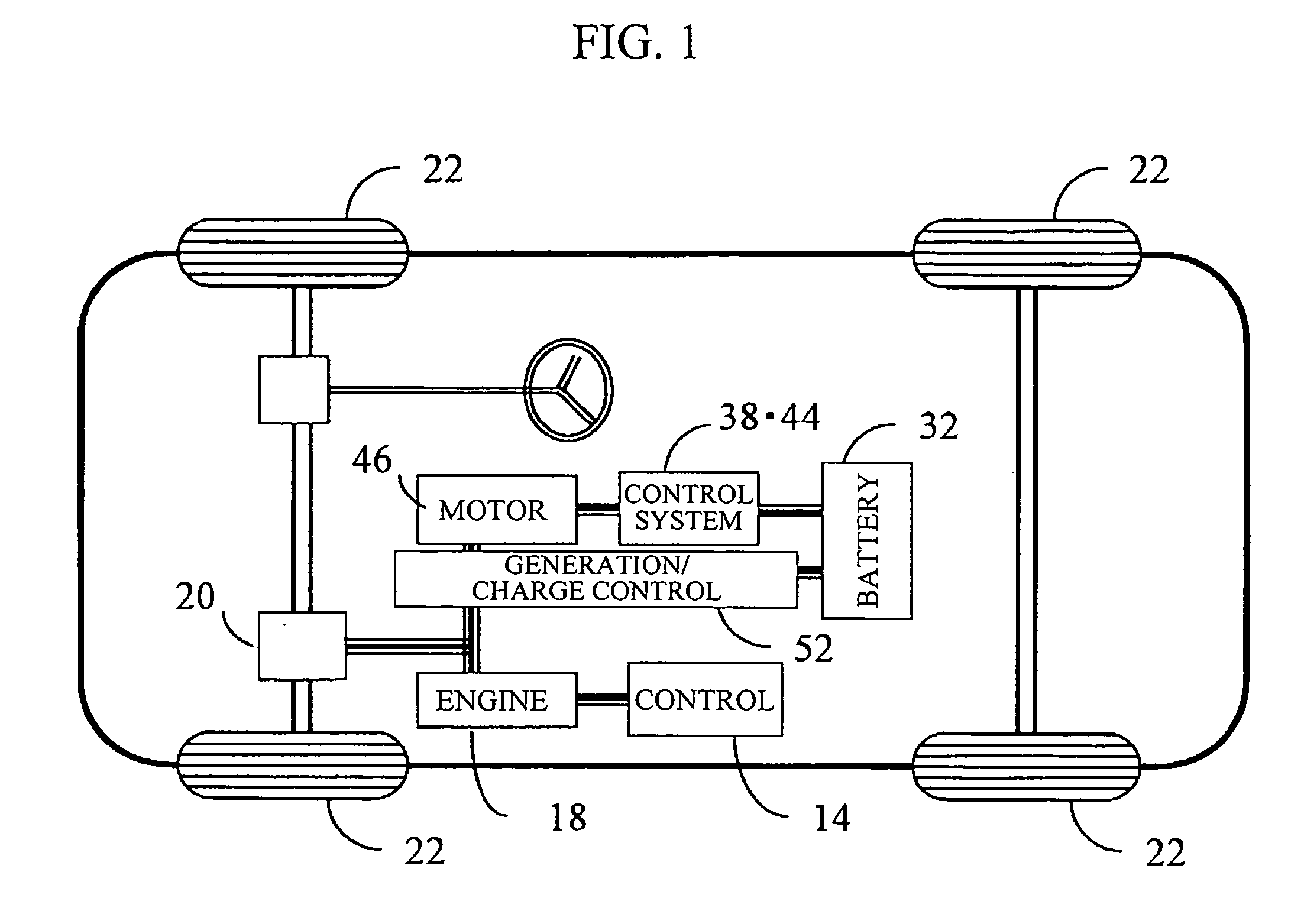
Vehicle power supply system
ActiveUS20060097577A1Efficient executionPower supplyBatteries circuit arrangementsDigital data processing detailsInternal resistanceEngineering
A main power source (2) is, for example, an ordinary Pb battery and generates a voltage of 12-13 V. At the time of starting an engine, the main power source (2) supplies power to a starter (8a). The main power source (2) is given a higher priority than an auxiliary power source (3) to supply power to ordinary loads (8b). The auxiliary power source (3) is a high performance battery (e.g., Li ion battery), which has superior charge acceptance capability and better state detectability over the main power source (2). Furthermore, the auxiliary power source (3) has an internal resistance per unit capacity, which is smaller than that of the main power source (2), and generates a voltage of 9-12 V. A generator (1) is directly connected to the auxiliary power source (3). The auxiliary power source (3) stores regenerative power, which is generated by the generator (1) at the time of deceleration of a vehicle, and is used as a redundant power source for the main power source (2). The main power source (2) and the auxiliary power source (3) are connected to each other through a supply circuit (5), which has a DC / DC converter (4), and a second supply circuit 7, which has a switch (6).
Owner:DENSO CORP
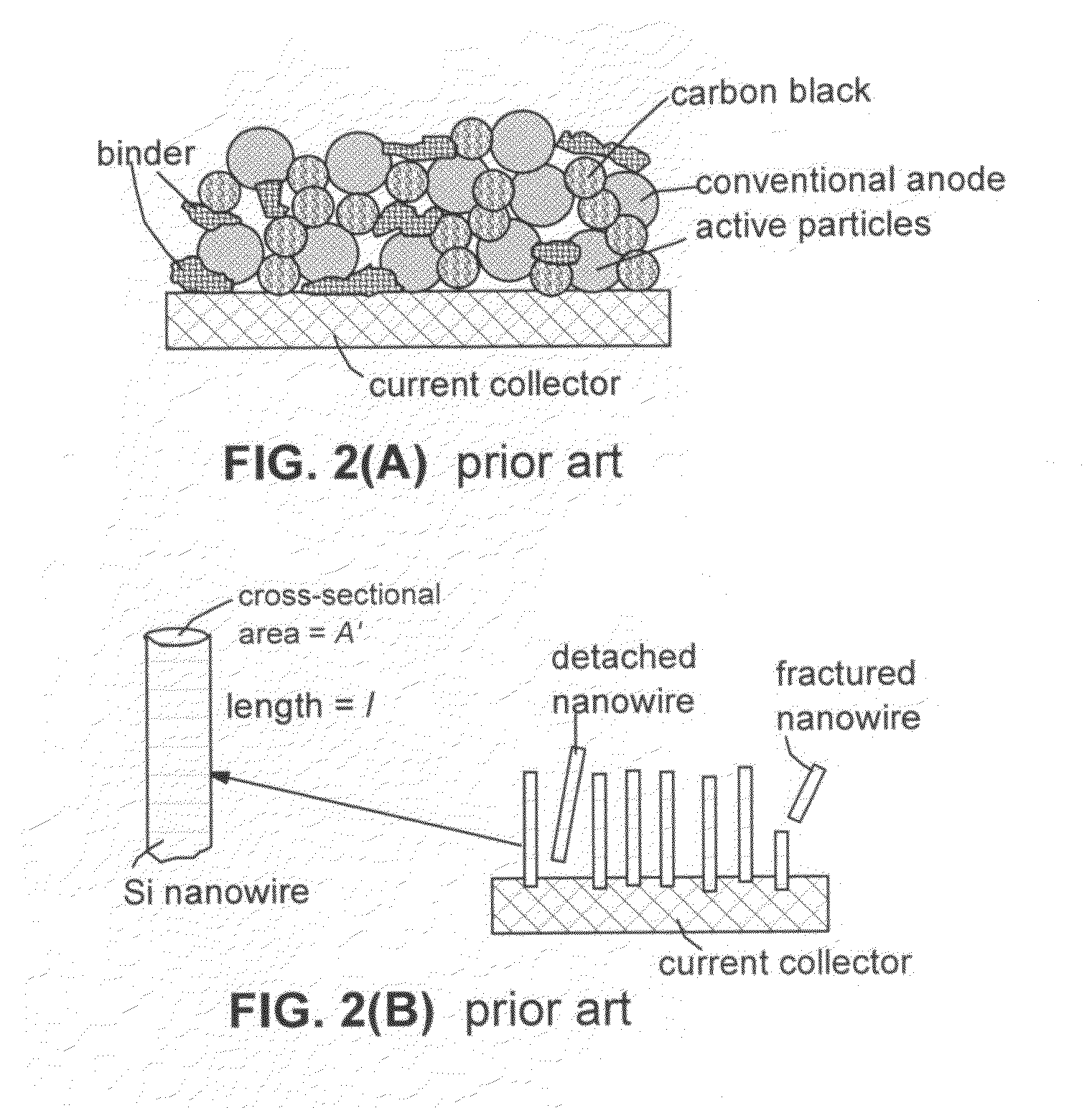
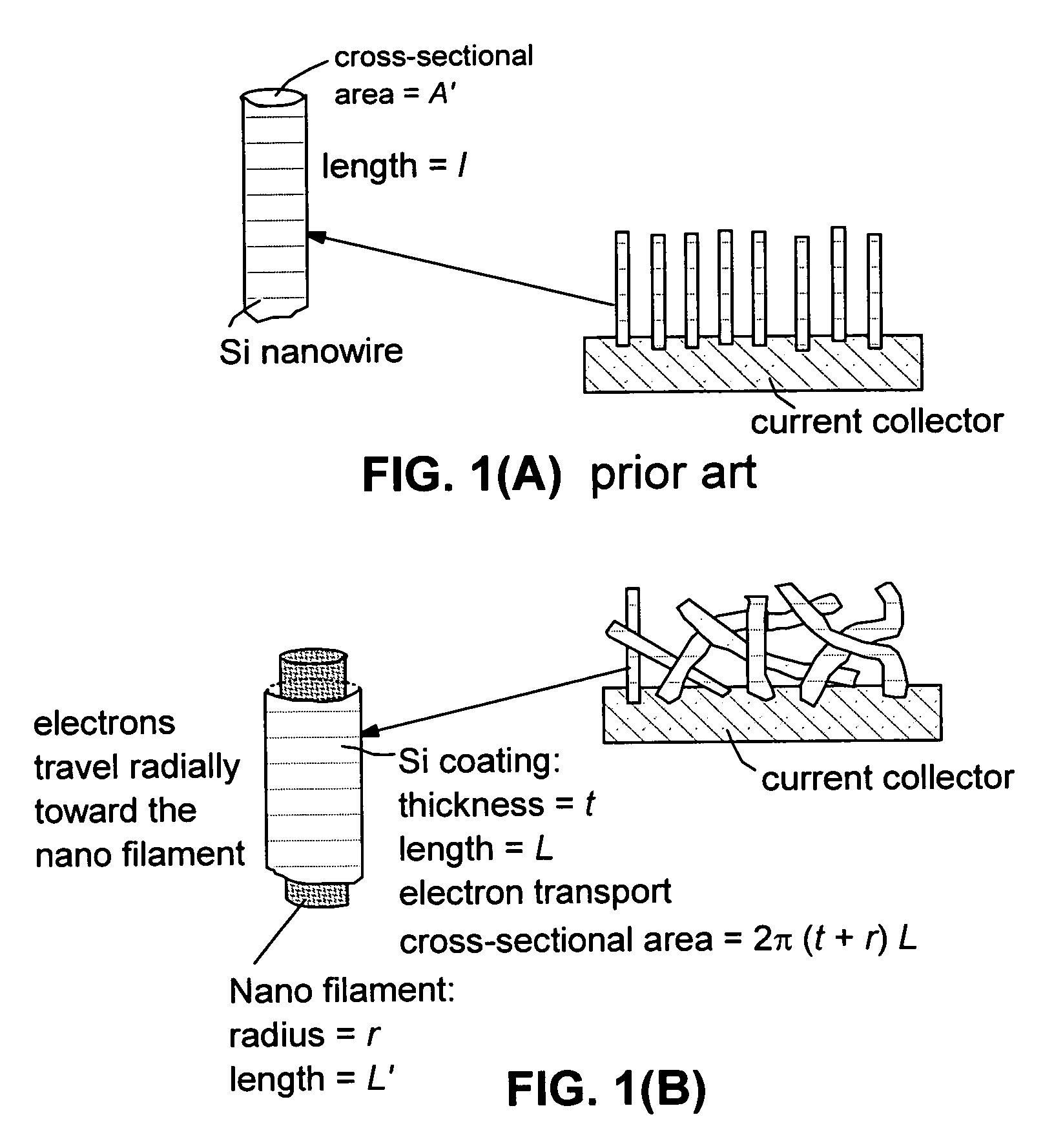
Core-shell high capacity nanowires for battery electrodes
InactiveUS20100330421A1Large capacityInhibition formationMaterial nanotechnologyNanostructure manufactureNanowireElectrochemistry
Provided are nanostructures containing electrochemically active materials, battery electrodes containing these nanostructures for use in electrochemical batteries, such as lithium ion batteries, and methods of forming the nanostructures and battery electrodes. The nanostructures include conductive cores, inner shells containing active materials, and outer shells partially coating the inner shells. The high capacity active materials having a stable capacity of at least about 1000 mAh / g can be used. Some examples include silicon, tin, and / or germanium. The outer shells may be configured to substantially prevent formation of Solid Electrolyte lnterphase (SEI) layers directly on the inner shells. The conductive cores and / or outer shells may include carbon containing materials. The nanostructures are used to form battery electrodes, in which the nanostructures that are in electronic communication with conductive substrates of the electrodes.
Owner:AMPRIUS INC
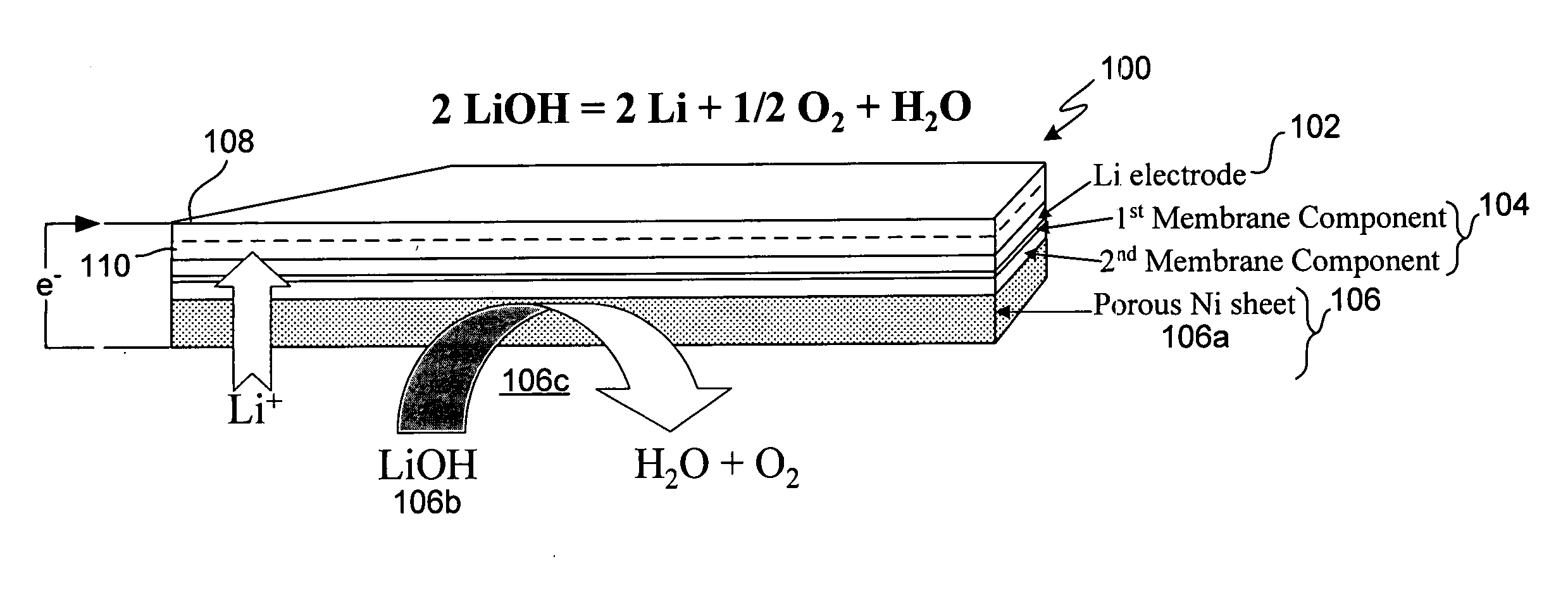
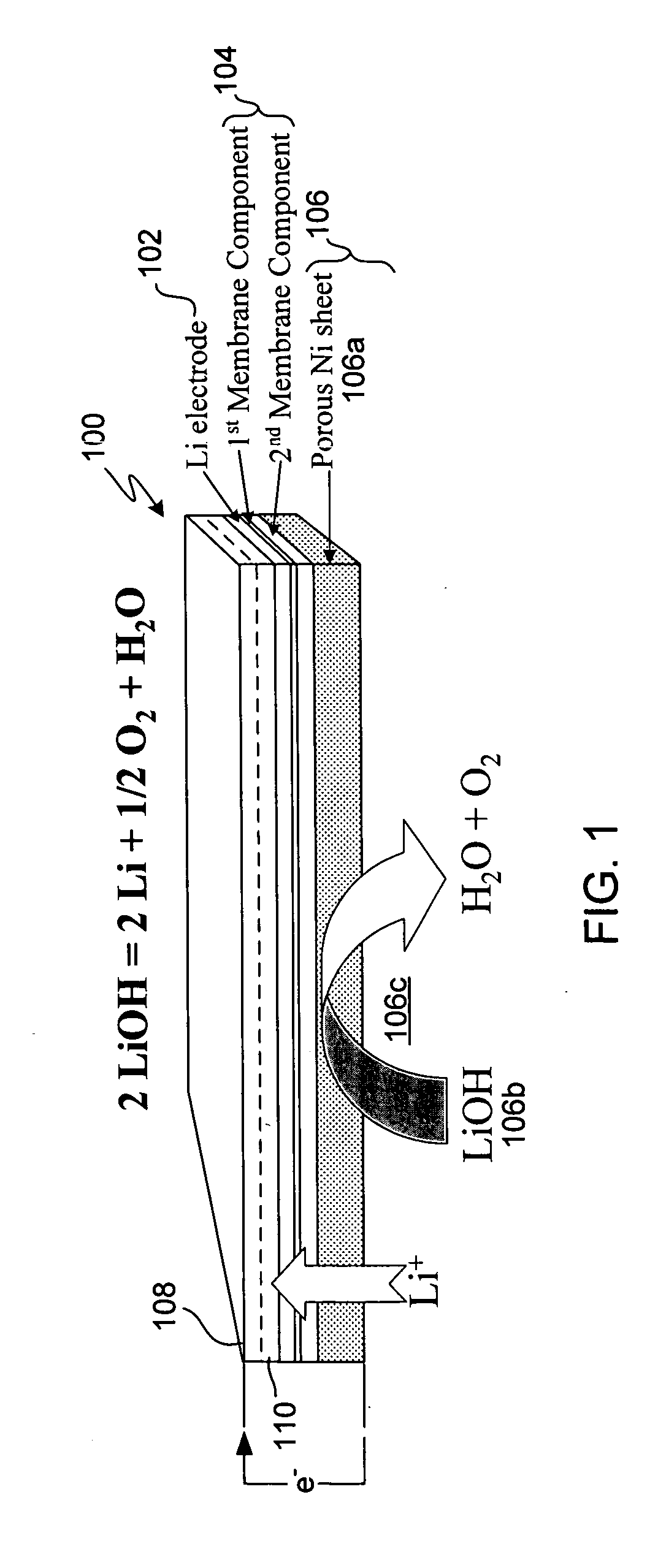
Active metal electrolyzer
InactiveUS20050100793A1Effective isolationReduce environmental pollutionPhotography auxillary processesElectrode manufacturing processesElectrolysisAqueous electrolyte
Electro-winning of active metal (e.g., lithium) ions from a variety of sources including industrial waste, and recycled lithium and lithium-ion batteries is accomplished with an electrolyzer having a protected cathode that is stable against aggressive solvents, including water, aqueous electrolytes, acid, base, and a broad range of protic and aprotic solvents. The electrolyzer has a highly ionically conductive protective membrane adjacent to the alkali metal cathode that effectively isolates (de-couples) the alkali metal electrode from solvent, electrolyte processing and / or cathode environments, and at the same time allows ion transport in and out of these environments. Isolation of the cathode from other components of a battery cell or other electrochemical cell in this way allows the use of virtually any solvent, electrolyte and / or anode material in conjunction with the cathode. The electrolyzer can be configured and operated to claim or reclaim lithium or other active metals from such sources.
Owner:POLYPLUS BATTERY CO INC
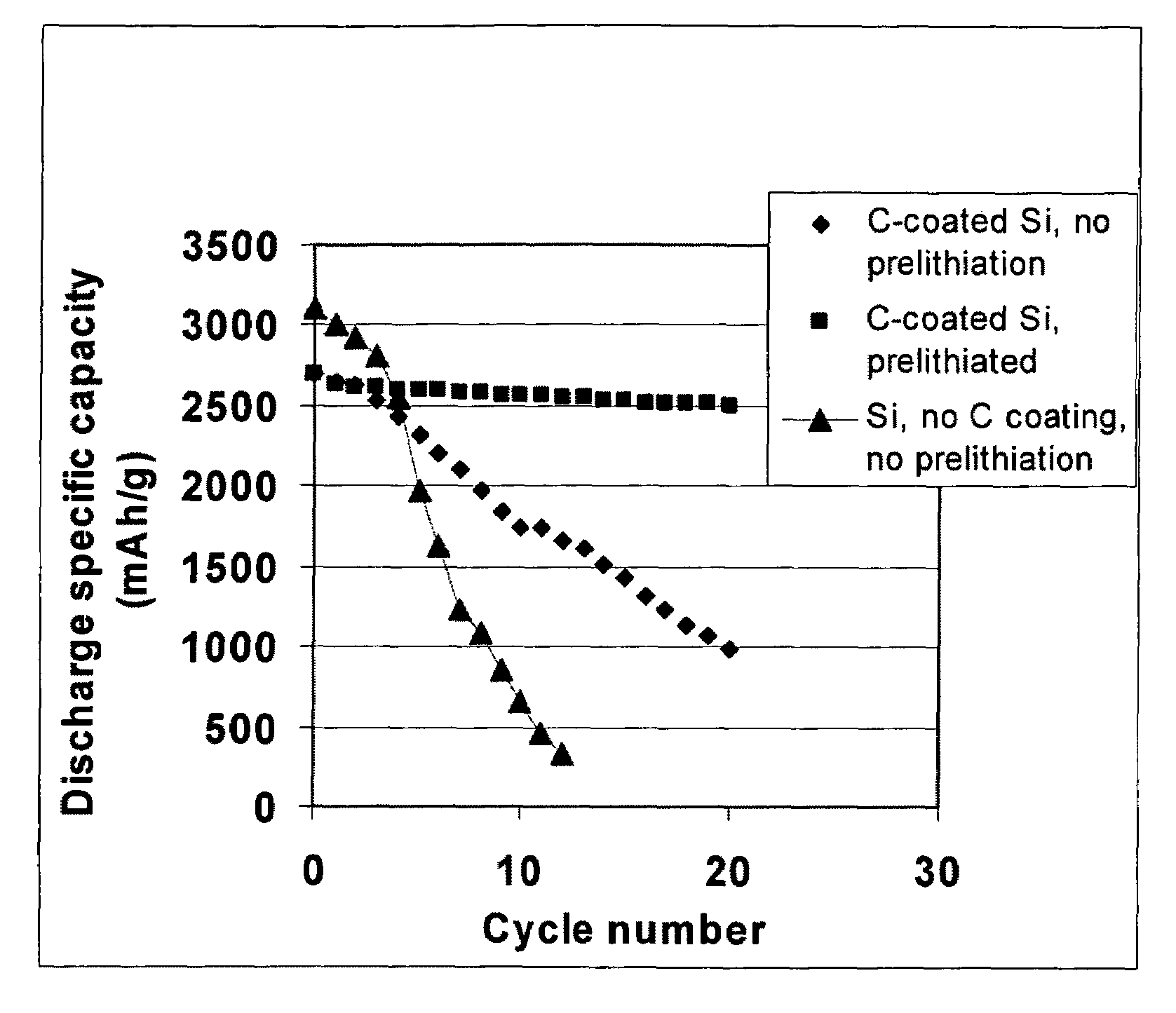
Secondary lithium ion battery containing a prelithiated anode
ActiveUS20100173198A1High specific capacityLong charge-discharge cycle lifeSecondary cellsActive material electrodesCharge dischargeGraphene
The present invention provides a lithium ion battery that exhibits a significantly improved specific capacity and much longer charge-discharge cycle life. In one preferred embodiment, the battery comprises an anode active material that has been prelithiated and pre-pulverized. This anode may be prepared with a method that comprises (a) providing an anode active material (preferably in the form of fine powder or thin film); (b) intercalating or absorbing a desired amount of lithium into the anode active material to produce a prelithiated anode active material; (c) comminuting the prelithiated anode active material into fine particles with an average size less than 10 μm (preferably <1 μm and most preferably <200 nm); and (d) combining multiple fine particles of the prelithiated anode active material with a conductive additive and / or a binder material to form the anode. Preferably, the prelithiated particles are protected by a lithium ion-conducting matrix or coating material. Further preferably, the matrix material is reinforced with nano graphene platelets.
Owner:GLOBAL GRAPHENE GRP INC
Lithium ion conductive solid electrolyte and production process thereof
InactiveUS20070231704A1Increase battery capacitySimple and convenient manufactureSecondary cellsSolid electrolyte cellsPorosityLithium metal
A lithium ion conductive solid electrolyte formed by sintering a molding product containing an inorganic powder and having a porosity of 10 vol % or less, which is obtained by preparing a molding product comprising an inorganic powder as a main ingredient and sintering the molding product after pressing and / or sintering the same while pressing, the lithium ion conductive solid electrolyte providing a solid electrolyte having high battery capacity without using a liquid electrolyte, usable stably for a long time and simple and convenient in manufacture and handling also in industrial manufacture in the application use of secondary lithium ion battery or primary lithium battery, a solid electrolyte having good charge / discharge cyclic characteristic in the application use of the secondary lithium ion battery a solid electrolyte with less water permeation and being safe when used for lithium metal-air battery in the application use of primary lithium battery, a manufacturing method of the solid electrolyte, and a secondary lithium ion battery and a primary lithium battery using the solid electrolyte.
Owner:OHARA
Graphite alkene iron lithium phosphate positive active material, preparing method thereof, and lithium ion twice battery based on the graphite alkene modified iron lithium phosphate positive active material
InactiveCN101752561AImprove conductivityImprove cycle stabilityLi-accumulatorsNon-aqueous electrolyte accumulator electrodesLithium-ion batteryCarbon coated
The present invention relates to graphite alkene iron lithium phosphate positive active material, a preparing method thereof, and a lithium ion twice battery based on the graphite alkene modified iron lithium phosphate positive active material. Graphite alkene and iron lithium phosphate are dispersed into water solution to be mixed evenly by stirring and ultra audible sound, then, are dried to obtain iron lithium phosphate material compounded by the graphite alkene and the iron lithium phosphate to be annealed by high temperature, and finally, the graphite alkene modified iron lithium phosphate positive active material is obtained. Compared with traditional carbon coated and conductive polymeric adulteration modified lithium batteries, the lithium ion twice battery based on the graphite alkene modified iron lithium phosphate positive active material has the advantages of high battery capacity, good charging-discharging circulating performance, long life and high circulating stability, and has great utility value.
Owner:宁波艾能锂电材料科技股份有限公司
Coated positive electrode materials for lithium ion batteries
ActiveUS20110111298A1Secondary cellsNon-aqueous electrolyte accumulator electrodesLithium metalInorganic composition
High specific capacity lithium rich lithium metal oxide materials are coated with inorganic compositions, such as metal fluorides, to improve the performance of the materials as a positive electrode active material. The resulting coated material can exhibit an increased specific capacity, and the material can also exhibit improved cycling. The materials can be formed while maintaining a desired relatively high average voltage such that the materials are suitable for the formation of commercial batteries. Suitable processes are described for the synthesis of the desired coated compositions that can be adapted for commercial production.
Owner:IONBLOX INC
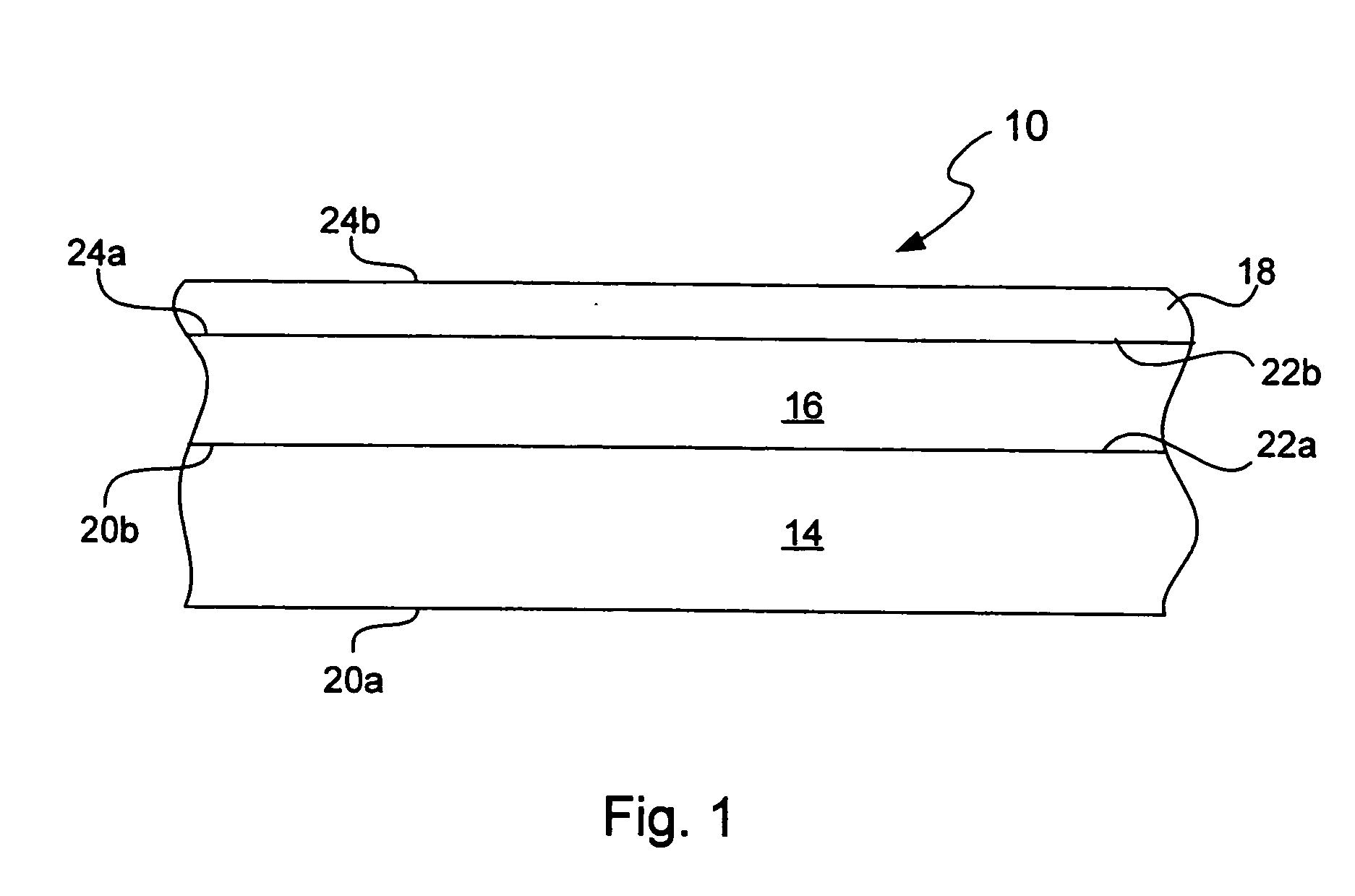
Chemical protection of a lithium surface
InactiveUS20050186469A1Easy to produceEasy to processElectrochemical processing of electrodesFinal product manufactureAlkaline earth metalLithium metal
Disclosed are compositions and methods for alleviating the problem of reaction of lithium or other alkali or alkaline earth metals with incompatible processing and operating environments by creating a ionically conductive chemical protective layer on the lithium or other reactive metal surface. Such a chemically produced surface layer can protect lithium metal from reacting with oxygen, nitrogen or moisture in ambient atmosphere thereby allowing the lithium material to be handled outside of a controlled atmosphere, such as a dry room. Production processes involving lithium are thereby very considerably simplified. One example of such a process is the processing of lithium to form negative electrodes for lithium metal batteries.
Owner:POLYPLUS BATTERY CO INC
Method for preparing electrode material for lithium battery
InactiveUS6887511B1Improve adhesionReduce expansionElectrode carriers/collectorsVacuum evaporation coatingAmorphous siliconOptoelectronics
Owner:SANYO ELECTRIC CO LTD
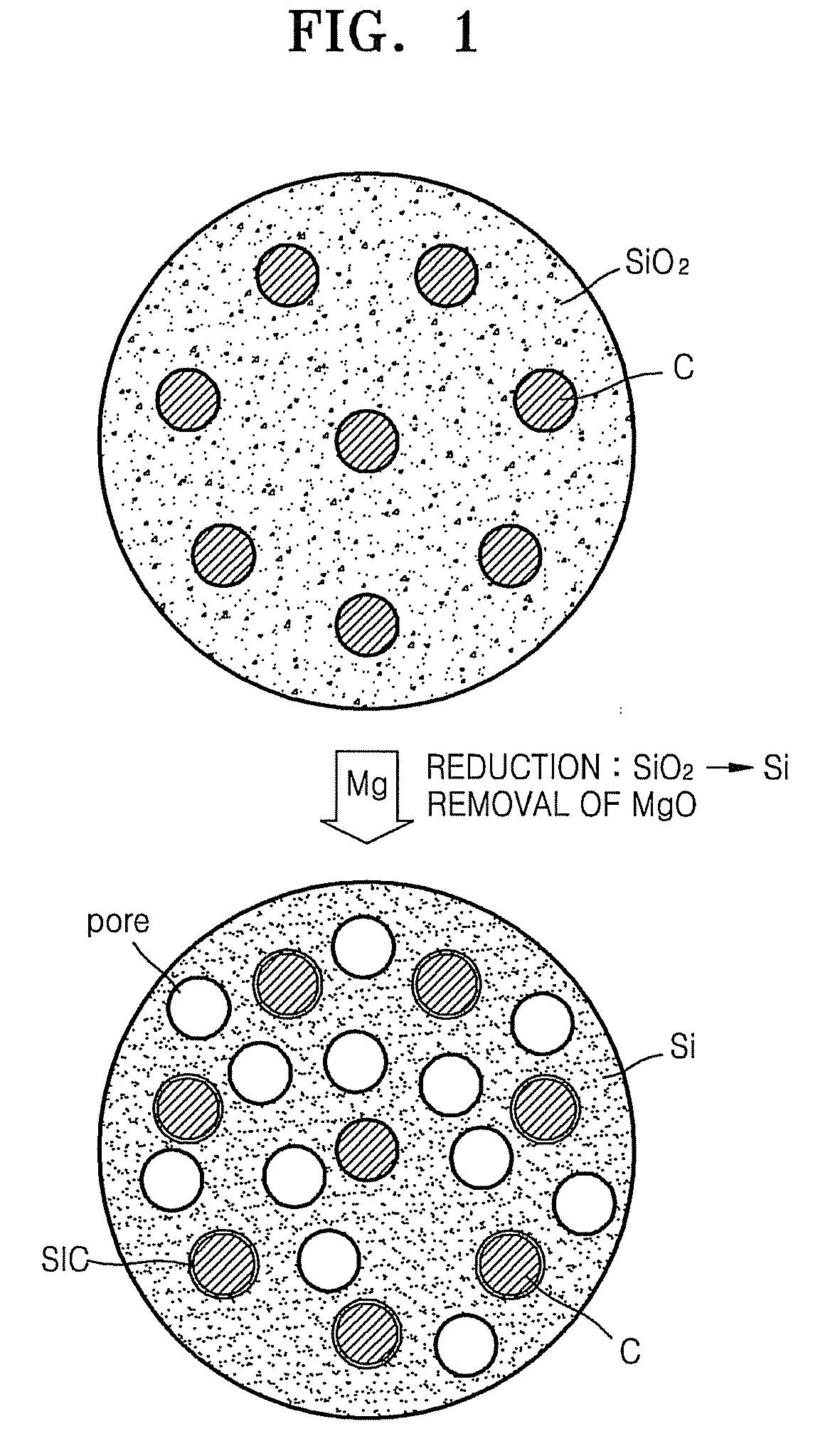
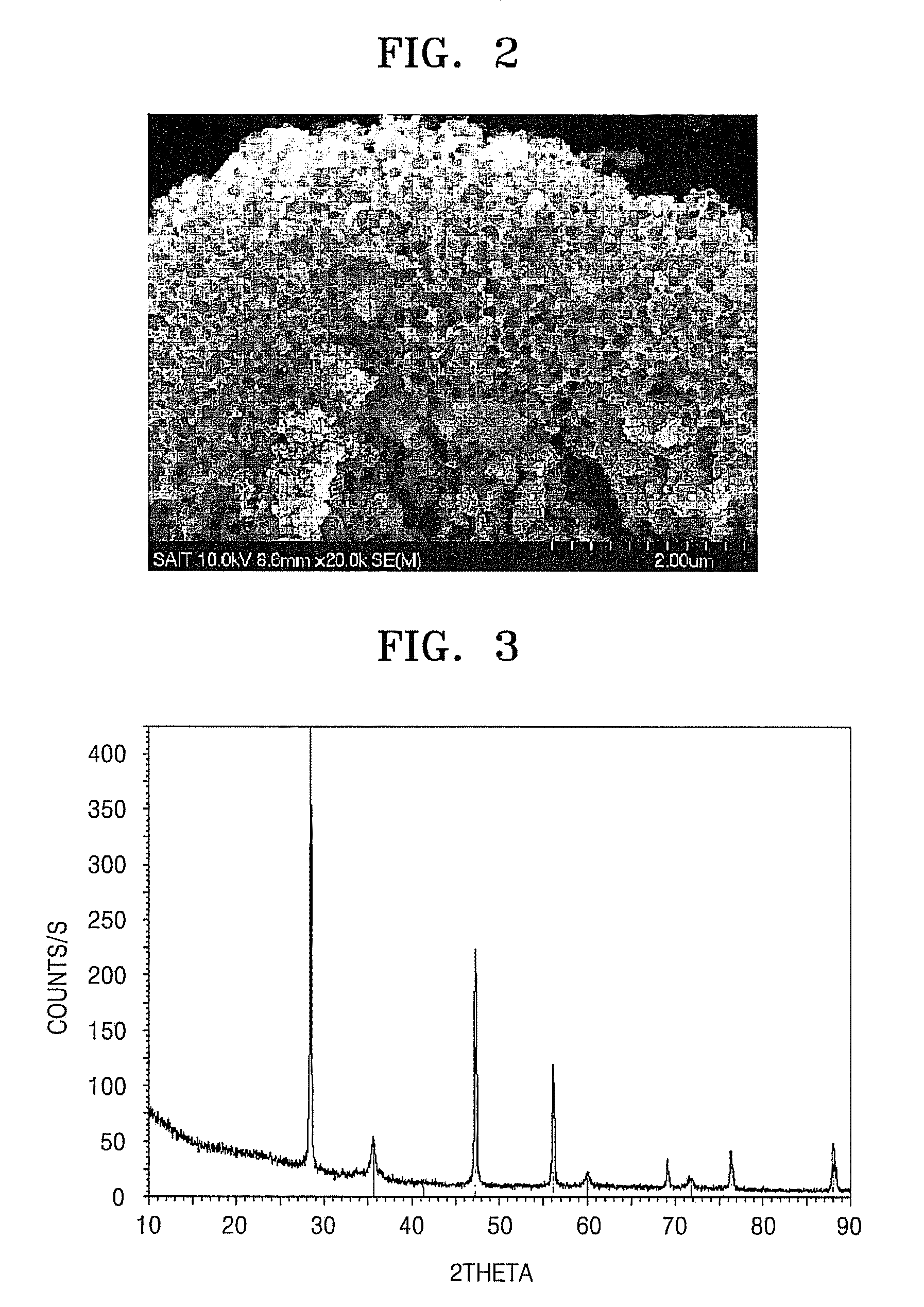
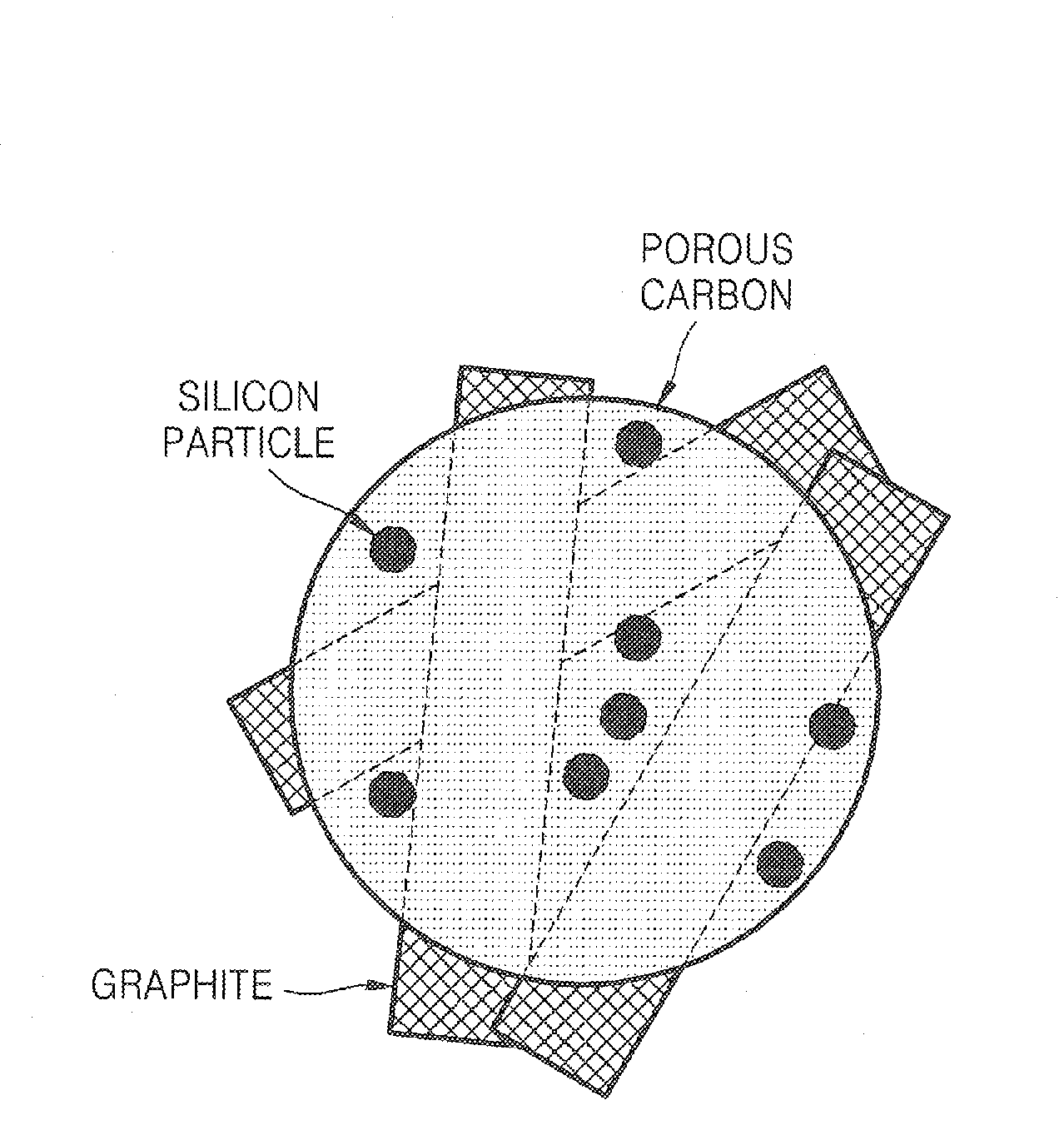
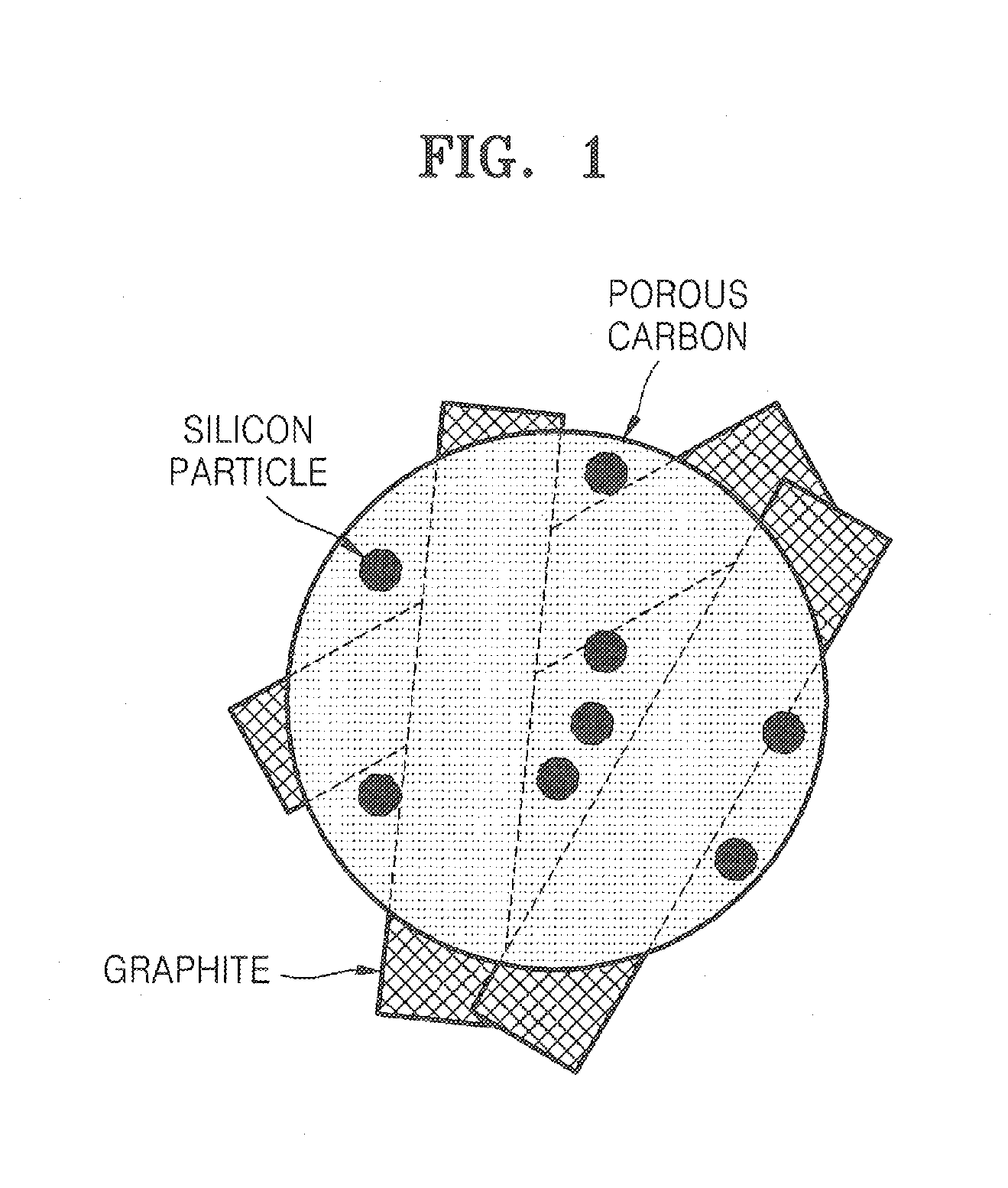
Porous anode active material, method of preparing the same, and anode and lithium battery employing the same
ActiveUS20080145757A1Efficiently remove stressExcellent charge and discharge characteristicsSilver accumulatorsMaterial nanotechnologyAlloyPore diameter
Provided are a porous anode active material, a method of preparing the same, and an anode and a lithium battery employing the same. The porous anode active material includes fine particles of metallic substance capable of forming a lithium alloy; a crystalline carboneous substance; and a porous carboneous material coating and attaching to the fine particles of metallic substance and the crystalline carboneous substance, the porous anode active material having pores exhibiting a bimodal size distribution with two pore diameter peaks as measured by a Barrett-Joyner-Halenda (BJH) pore size distribution from a nitrogen adsorption. The porous anode active material has the pores having a bimodal size distribution, and thus may efficiently remove a stress occurring due to a difference of expansion between a carboneous material and a metallic active material during charging and discharging. Further, the anode electrode and the lithium battery comprising the anode active material have excellent charge / discharge characteristics.
Owner:SAMSUNG SDI CO LTD
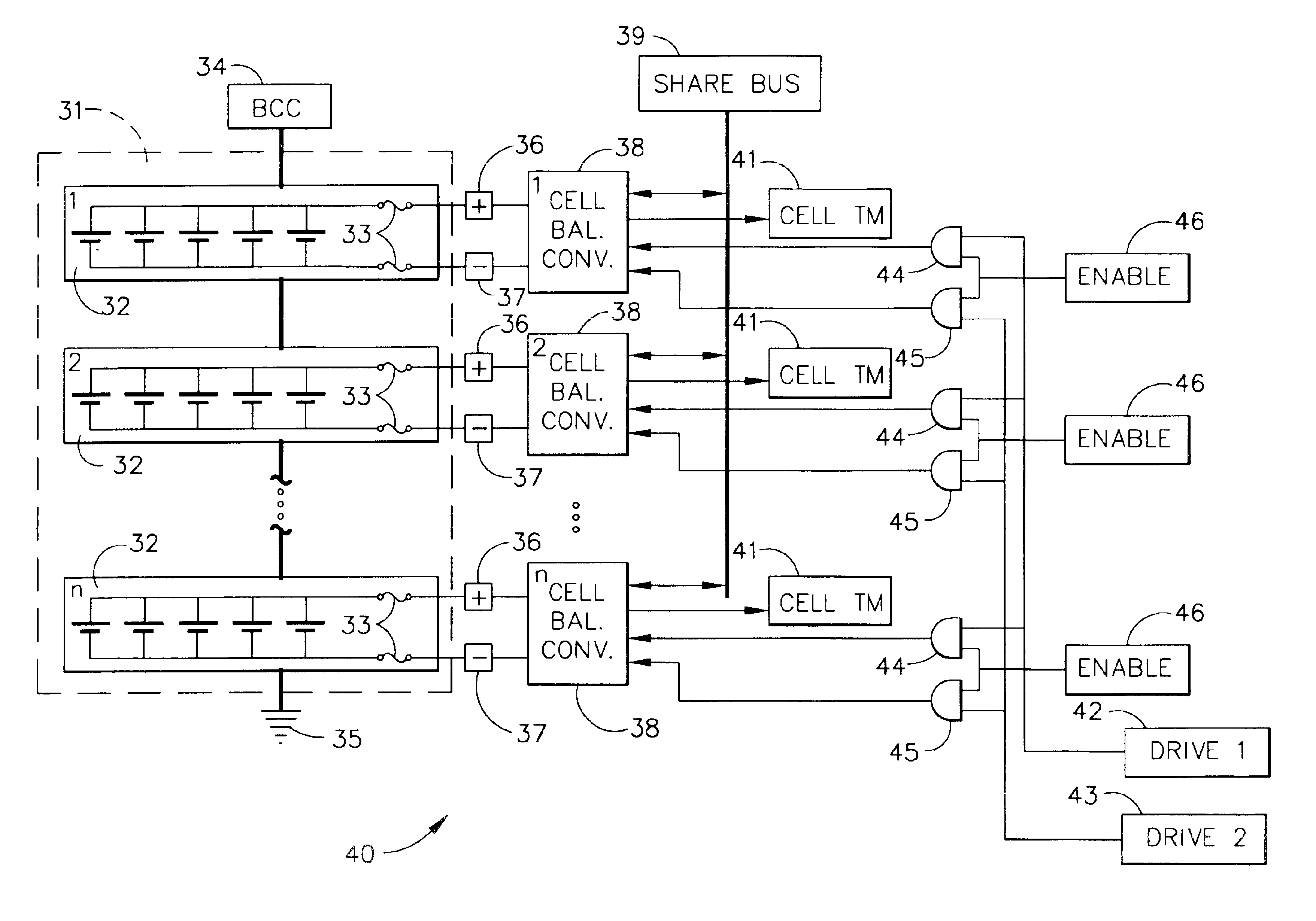
Autonomous battery cell balancing system with integrated voltage monitoring
InactiveUS6873134B2Circuit monitoring/indicationCharge equalisation circuitLithium-ion batteryControl circuit
An autonomous lithium-ion cell balancing system with integrated voltage monitoring includes a lithium-ion battery having a plurality of cells, a cell balancing circuit and a cell balancing control circuit. The heart of the cell balancing circuit is a plurality of gated cell balancing converters that are connected with the battery cells and a common share bus. Each gated lithium-ion cell balancing converter receives and input drive voltage and an input sample voltage and transfers energy from highly charged lithium-ion cells to the less charged lithium-ion cells, forcing all cells to have the same energy level. Further, the gated cell balancing converter provides a telemetry output voltage analog to an input cell voltage, eliminating the need for a separate cell voltage monitor.
Owner:THE BOEING CO
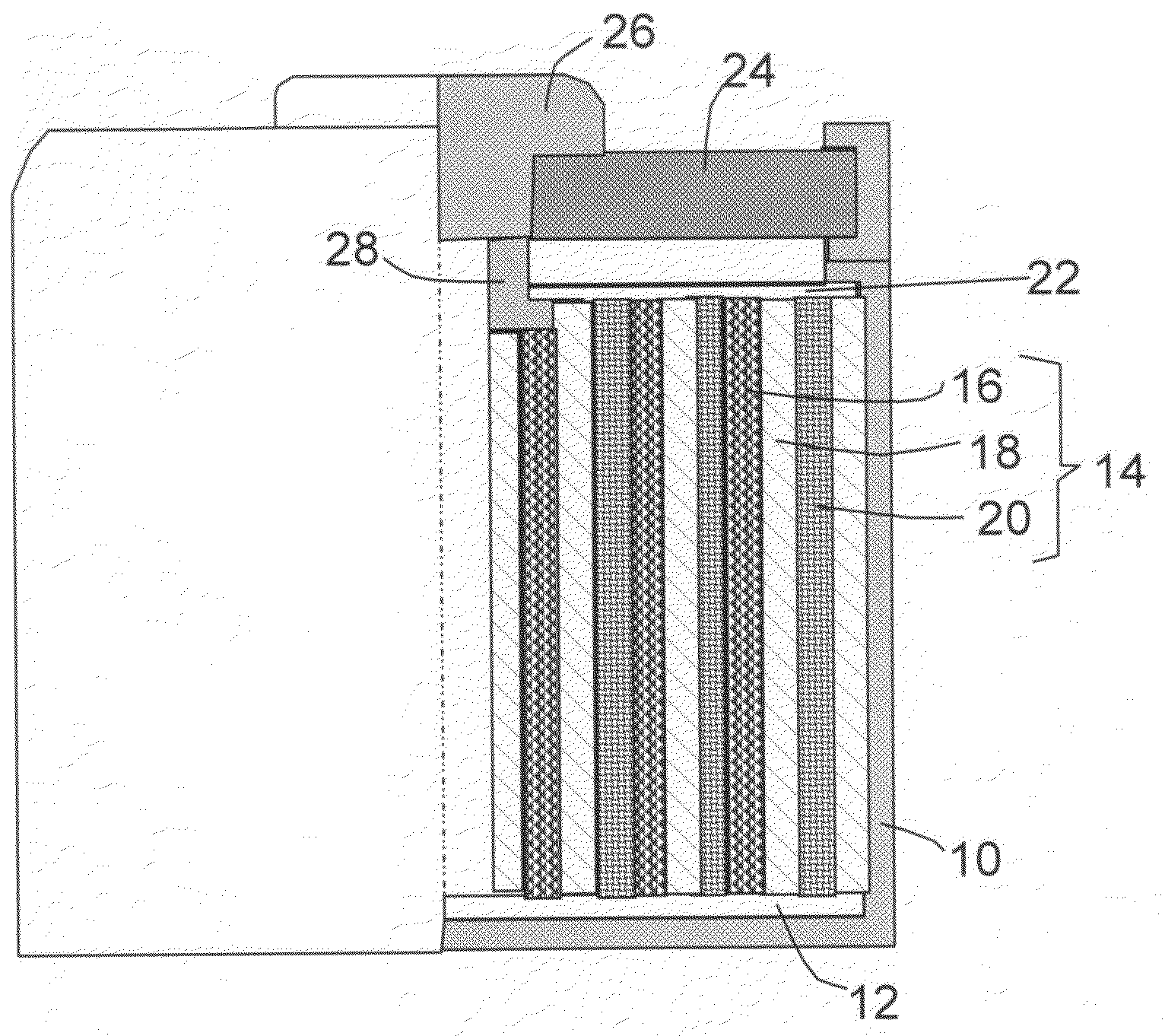
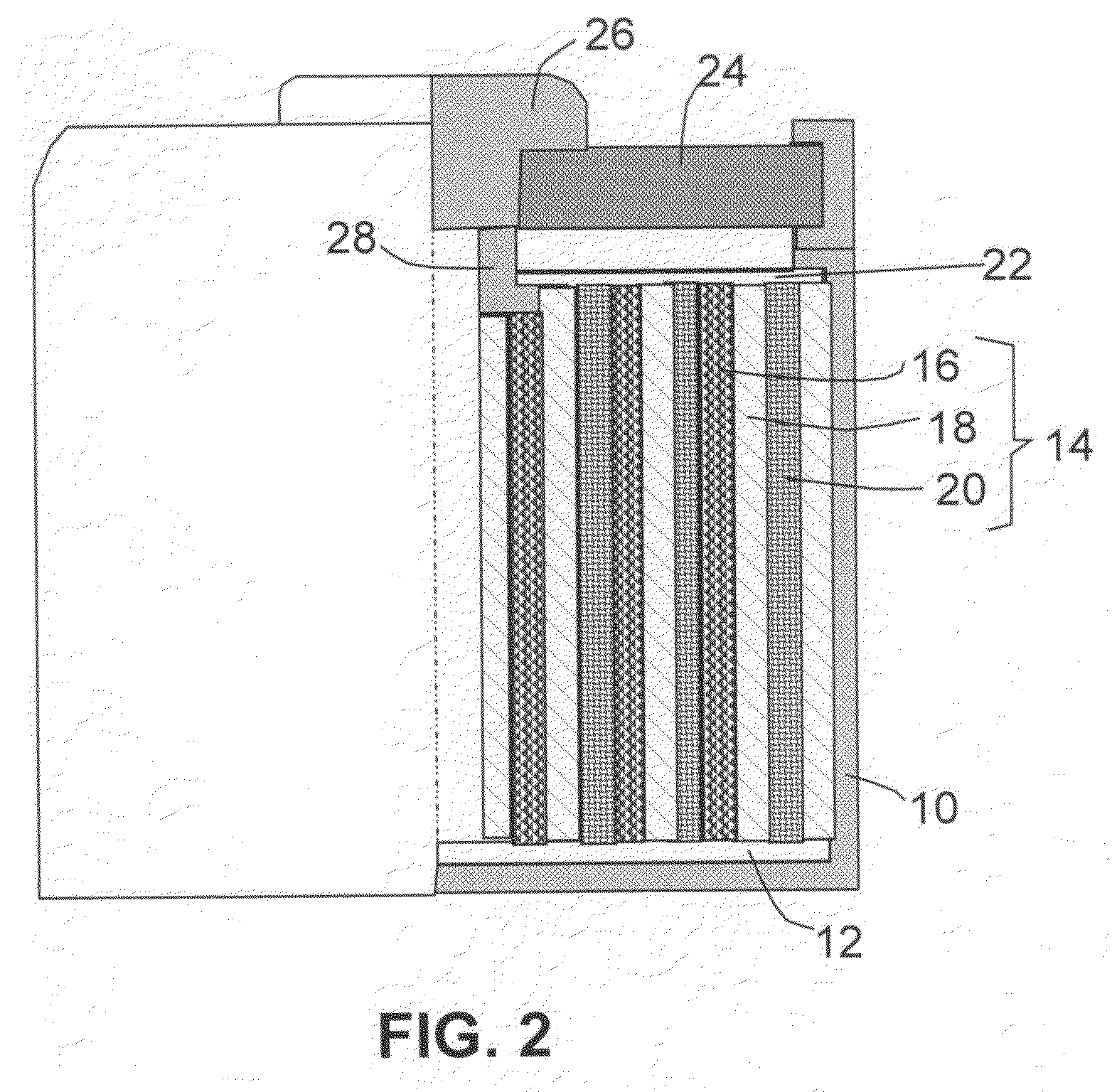
Hybrid nano-filament anode compositions for lithium ion batteries
ActiveUS20090169996A1Superior multiple-cycle behaviorImprove cycle lifeElectrochemical processing of electrodesElectrode thermal treatmentLithium-ion batteryNanometre
This invention provides a hybrid nano-filament composition for use as an electrochemical cell electrode. The composition comprises: (a) an aggregate of nanometer-scaled, electrically conductive filaments that are substantially interconnected, intersected, or percolated to form a porous, electrically conductive filament network comprising substantially interconnected pores, wherein the filaments have an elongate dimension and a first transverse dimension with the first transverse dimension being less than 500 nm (preferably less than 100 nm) and an aspect ratio of the elongate dimension to the first transverse dimension greater than 10; and (b) micron- or nanometer-scaled coating that is deposited on a surface of the filaments, wherein the coating comprises an anode active material capable of absorbing and desorbing lithium ions and the coating has a thickness less than 20 μm (preferably less than 1 μm). Also provided is a lithium ion battery comprising such an electrode as an anode. The battery exhibits an exceptionally high specific capacity, an excellent reversible capacity, and a long cycle life.
Owner:GLOBAL GRAPHENE GRP INC
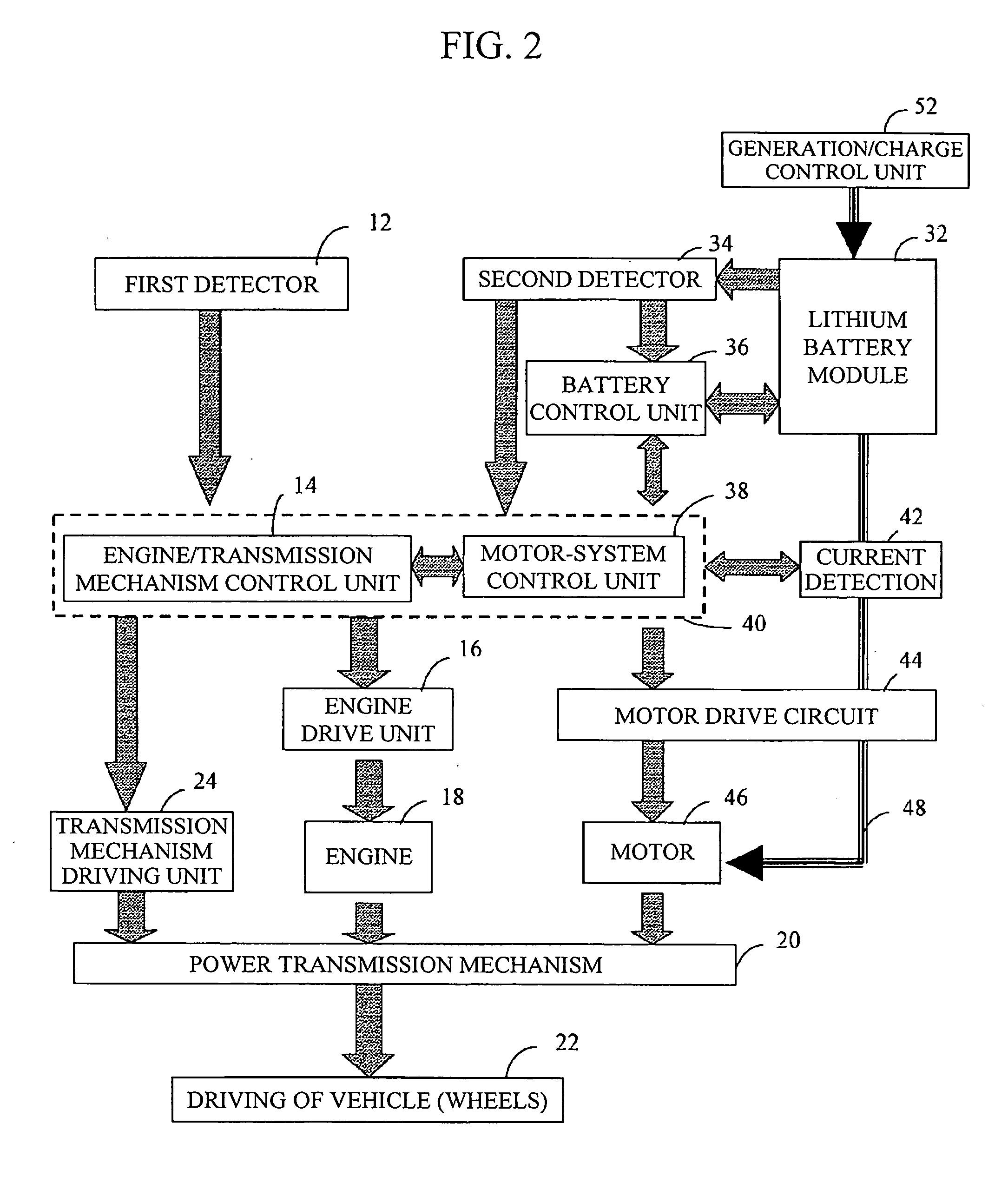
Battery module having lithium battery, and vehicle control system employing battery module having lithium battery
InactiveUS20050110460A1Improve cooling effectLess mounting spaceBatteries circuit arrangementsCell temperature controlControl systemCooling effect
A battery module with an excellent cooling effect includes a battery unit 194 and a circuit unit 192. The battery unit 194 includes a casing main body 195 having an external frame 195a formed of a plate-like member in a rectangular shape and an internal member 197 supported inside the external frame 195a. A plurality of lithium single cells 202 to 208 are connected in series to form a longitudinal tandem cell 200. A plurality of the tandem cells 200 are disposed on either side of the internal member in parallel, and the entire tandem cells are connected in series by connecting metal fittings that are disposed on either side of the tandem cells 200. A cover 198 with ventilation opening is mounted on the casing main body 195. The circuit unit 192 is disposed on one end of the battery unit 194 and includes a control circuit that detects the voltage of each of the lithium single cells 202 to 208 and transmits the result of detection using a control means.
Owner:SHIN KOBE ELECTRIC MASCH CO LTD
Electrode for use in lithium battery and rechargeable lithium battery
InactiveUS7235330B1High charge and discharge capacityImprove featuresElectrode manufacturing processesSmall-sized cells cases/jacketsAmorphous siliconSurface roughness
Owner:SANYO ELECTRIC CO LTD
Methods and systems for making electrodes having at least one functional gradient therein and devices resulting therefrom
InactiveUS20110123866A1Improve performanceElectrode manufacturing processesElectric shock equipmentsHigh fluxEngineering
The invention disclosed herein provides for methods and apparatuses that yield electrodes having at least one functional gradient therein. In many embodiments, the electrodes comprise an electrode matrix having a plurality of layers, where at least two of the layers differs functionally, in composition, structure, or, organization. High-throughput electrode screening apparatuses are disclosed that include array formers and testers. Electrodes and battery cells arising from the methods and apparatuses disclosed herein are likewise disclosed. The methods, apparatuses, and resulting electrode and cell devices are, in some embodiments, ideally suited for use in lithium-ion batteries.
Owner:MOLECULAR NANOSYST
Method for detecting electrical property consistency of lithium ion battery
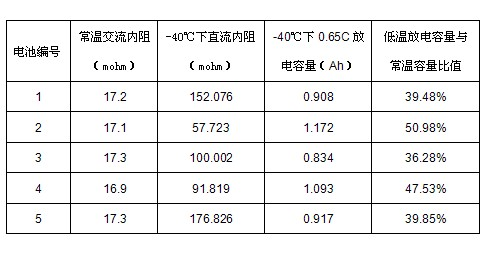
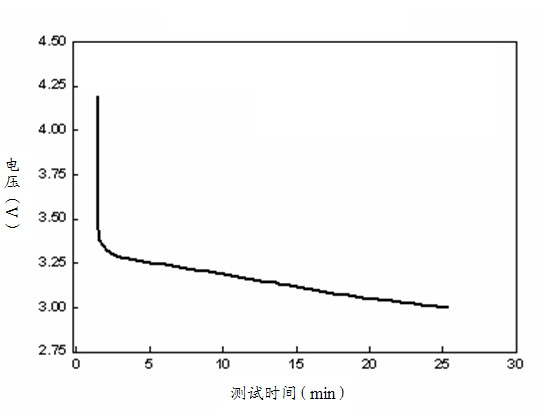
ActiveCN101907688AElectrical performance consistency solutionEasy to useElectrical testingElectricityInternal resistance
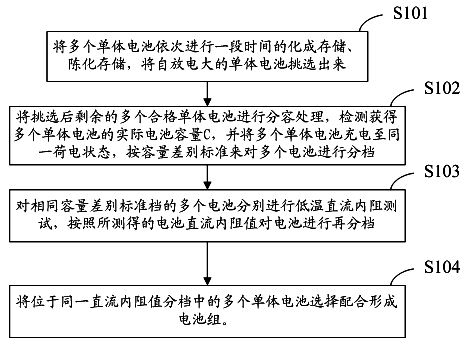
The invention discloses a method for detecting the electrical property consistency of lithium ion battery. The method comprises the following steps of: performing formation storage and ageing storage on a plurality of monomer batteries for a long time one by one and selecting the battery with large self discharge; performing capacity grading treatment on the rest of qualified monomer batteries to obtain the actual battery volume C of the plurality of monomer batteries, charging the plurality of monomer batteries to the same charging state and classifying the plurality of batteries according to the volume difference standard; performing low-temperature direct-current internal resistance test on the plurality of batteries with the same volume difference standard grade and reclassifying the battery according to the direct-current internal resistance of the battery; and selecting and matching the plurality of batteries in the same direct-current internal resistance grade to form the battery pack. The invention can detect the electrical property consistency of the lithium ion battery under the low-temperature condition, effectively solve the problem that the battery has good electrical property consistency under the normal-temperature condition and differed electrical property consistency under the low-temperature condition and is suitable for detecting a batch of batteries and selecting qualified monomer batteries to form a battery pack.
Owner:TIANJIN LISHEN BATTERY +1
High-capacity lithium-ion electrolyte, battery and preparation method of battery
ActiveCN101771167AIncrease capacityLow costFinal product manufactureCell electrodesElectrolytic agentPhysical chemistry
The invention relates to a high-capacity lithium-ion electrolyte, a battery and a preparation method of a battery, in particular to the high-capacity lithium-ion electrolyte and the battery using the high-capacity lithium-ion electrolyte and the preparation method of the battery. The electrolyte disclosed by the invention comprises lithium salt and non-aqueous organic solvent, and also consists of the following components in weight percent in terms of the total weight of the electrolyte: 0.5-7% of film-forming additive, 0-15% of flame-retardant additive, 2-10% of antiovefill additive, 0.01-2% of stabilizer and 0.01-1% of wetting agent; the electrolyte can enable the anode with high Ni content to work stably, and reduce the battery cost; the high-capacity lithium-ion battery can perform high ratio capacity and excellent safety and high temperature property and cyclic life fully due to the addition and synergetic functions of various functional additives.
Owner:JIUJIANG TINCI ADVANCED MATERIALS CO LTD