Patents
Literature
132 results about "Epstein–Barr virus" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
The Epstein–Barr virus (EBV, sometimes abbreviated as EPV), formally called Human gammaherpesvirus 4, is one of eight known human herpesvirus types in the herpes family, and is one of the most common viruses in humans.
Quantitative epstein barr virus PCR rapid assay
InactiveUS6790952B2Sensitive and accurateSugar derivativesMicrobiological testing/measurementEpstein bar virusNucleotide
The present invention provides novel compositions comprising Epstein-Barr virus-specific oligonucleotides that are useful as primers to amplify particular regions of the genome during enzymatic nucleic acid amplification. The invention also provides a rapid, sensitive and specific method for the detection and quantitation of the virus which may be present in a clinical specimen, using the virus-specific primers and enzymatic nucleic acid amplification; hybridization of amplified target sequences, if present, with one or more Epstein-Barr virus-specific oligonucleotide probes which are labeled with a detectable moiety; and detection of the detectable moiety of labeled oligonucleotide probe hybridized to amplified target sequences of Epstein-Barr virus DNA.
Owner:CHILDRENS HOSPITAL RES FOUND
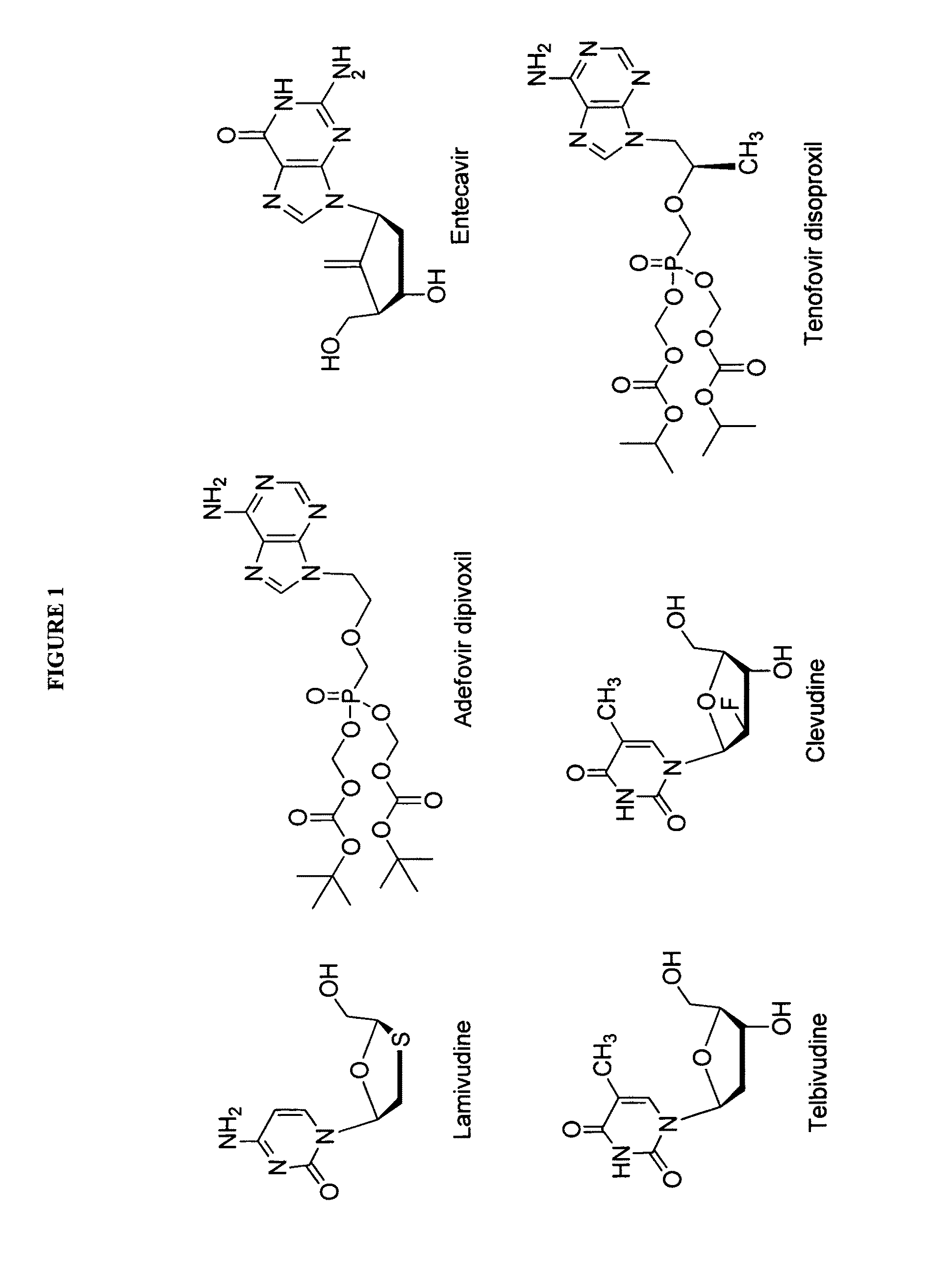
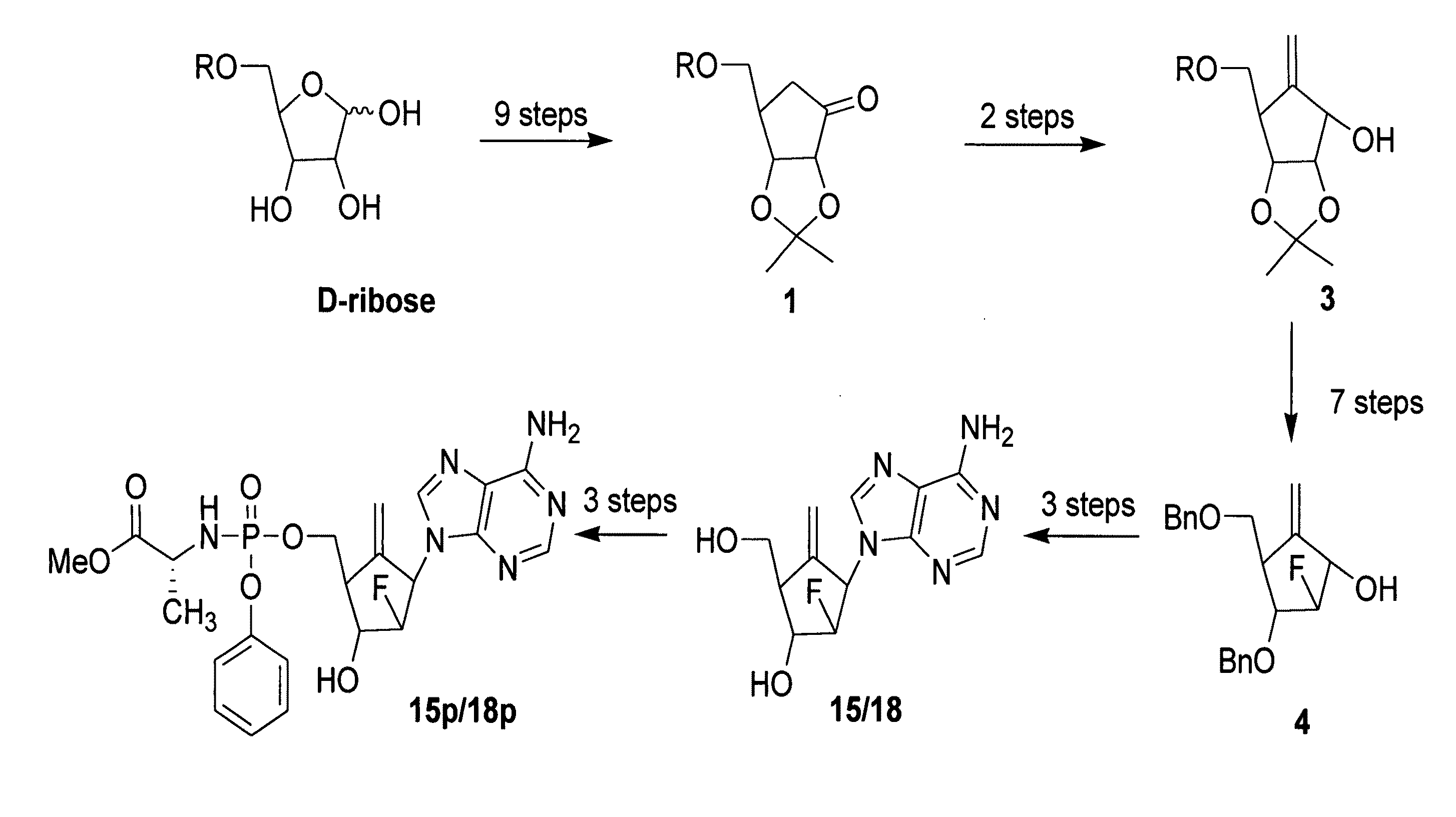
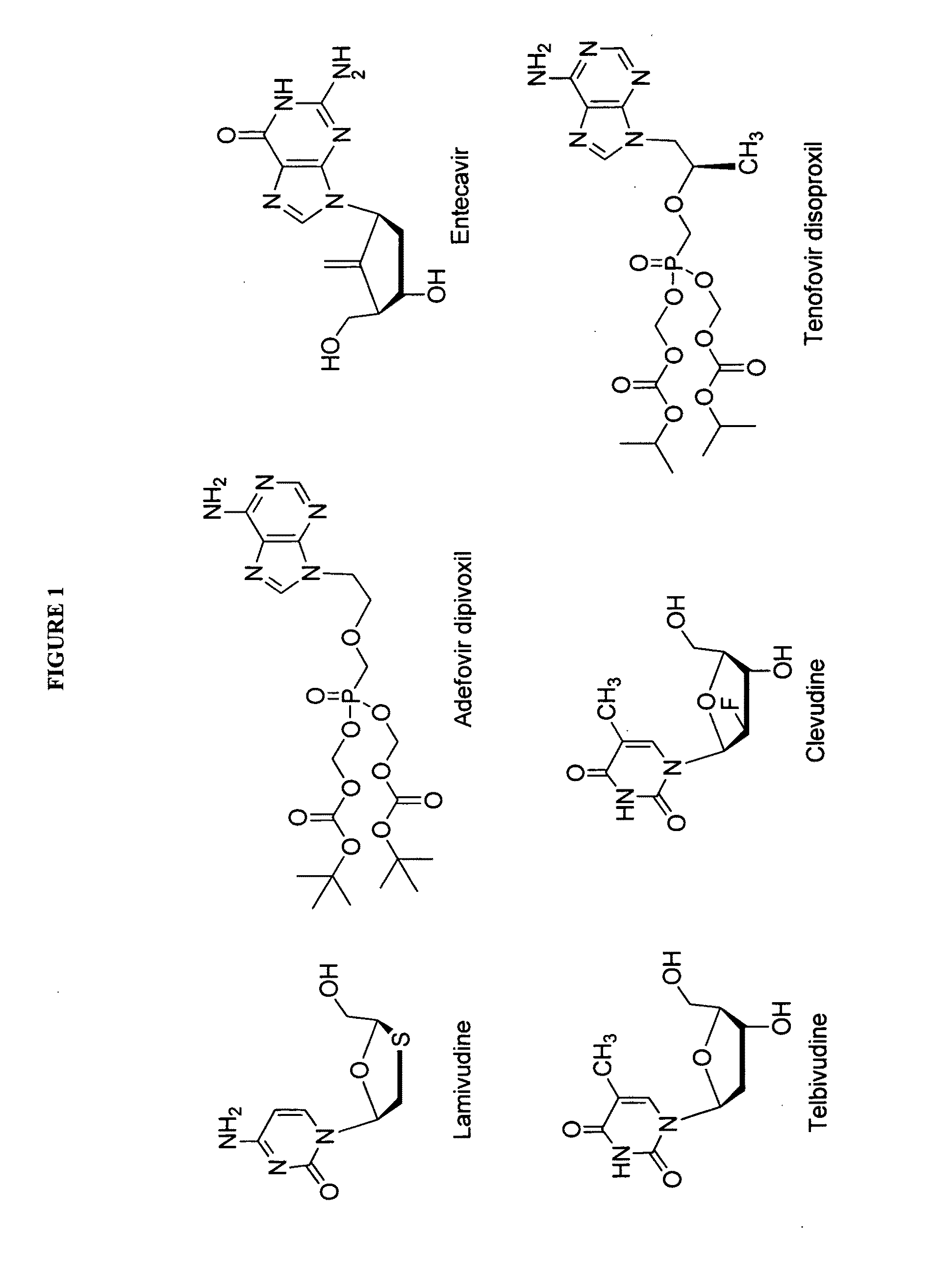
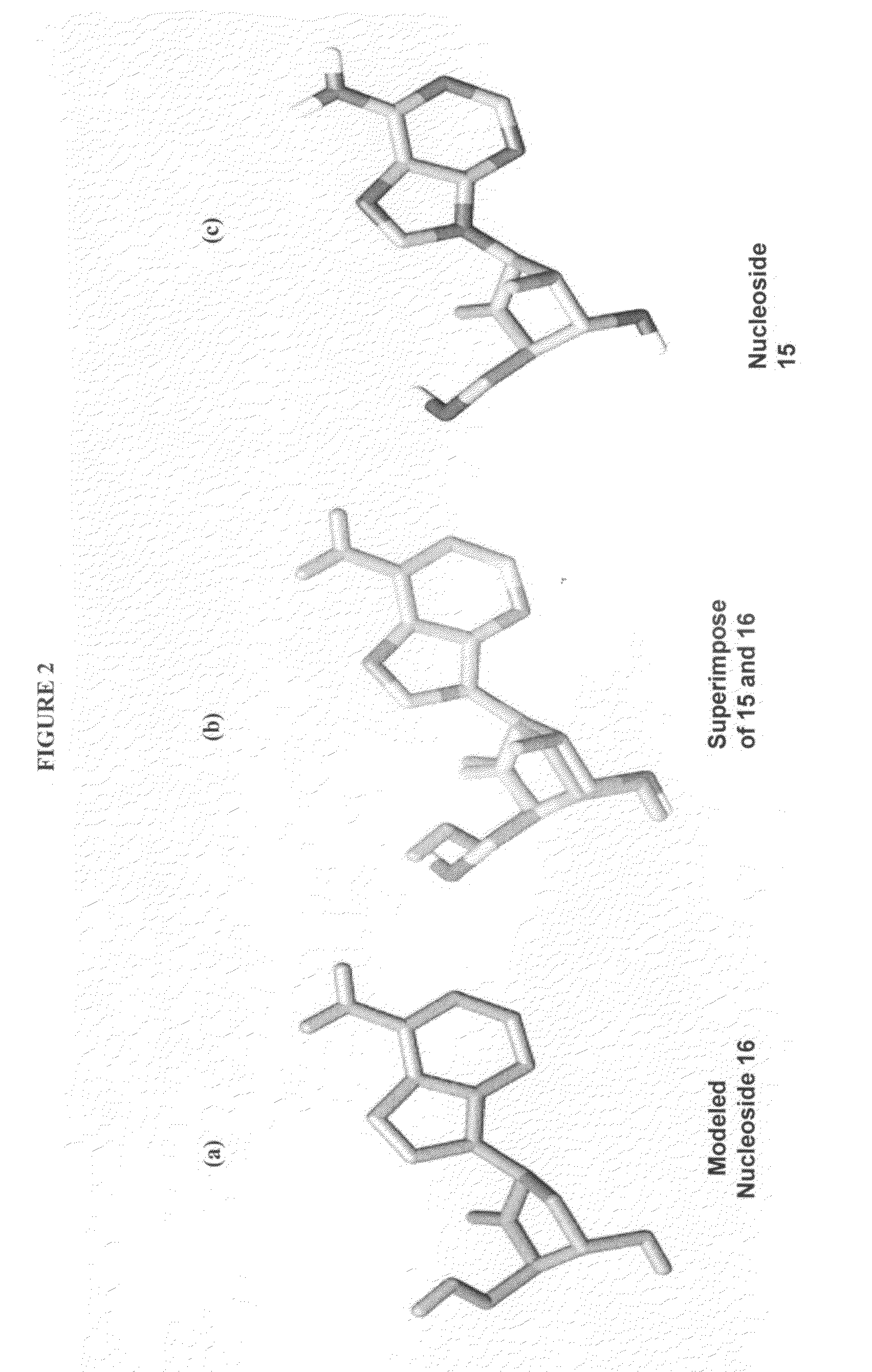
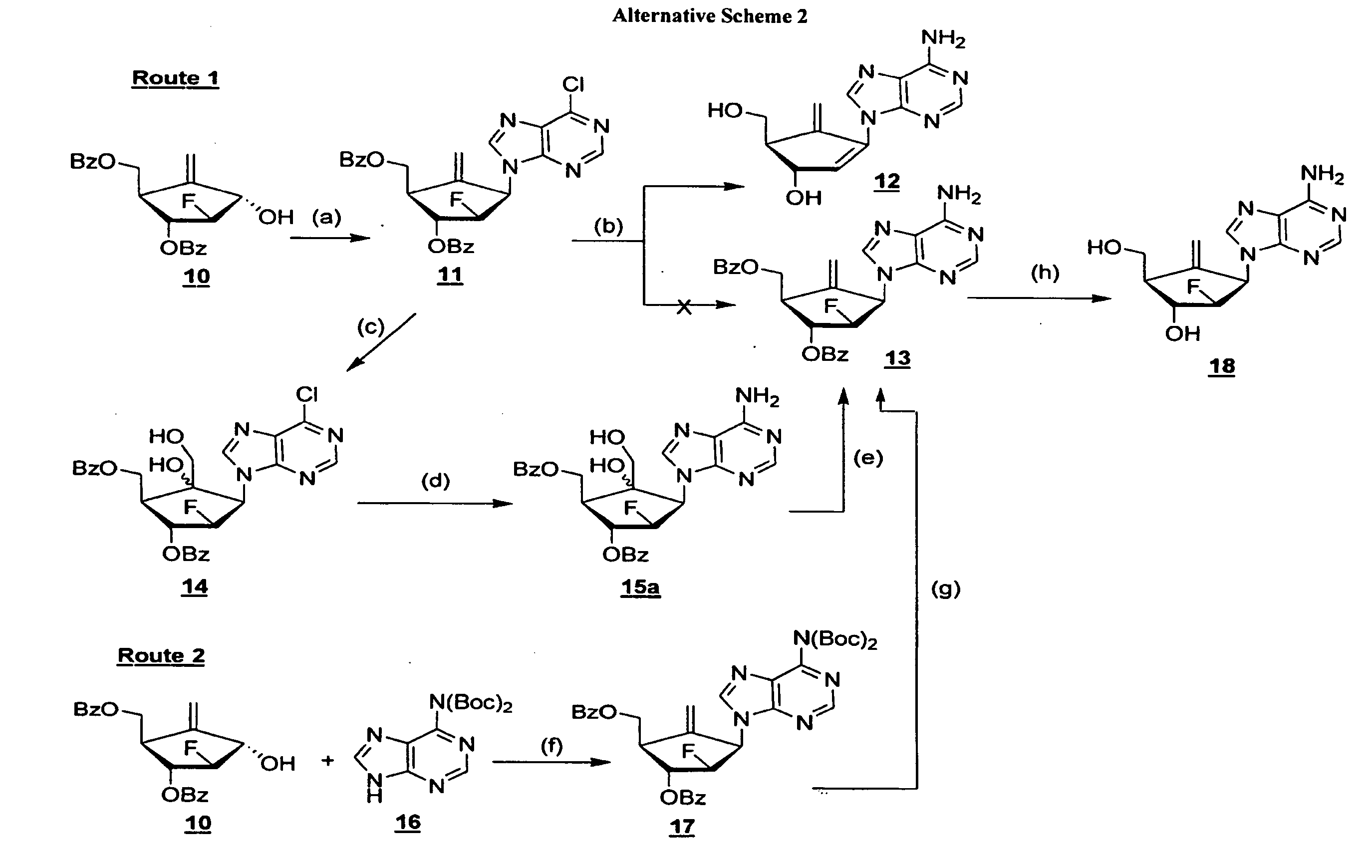
2′-fluoro-6′-methylene carbocyclic nucleosides and methods of treating viral infections
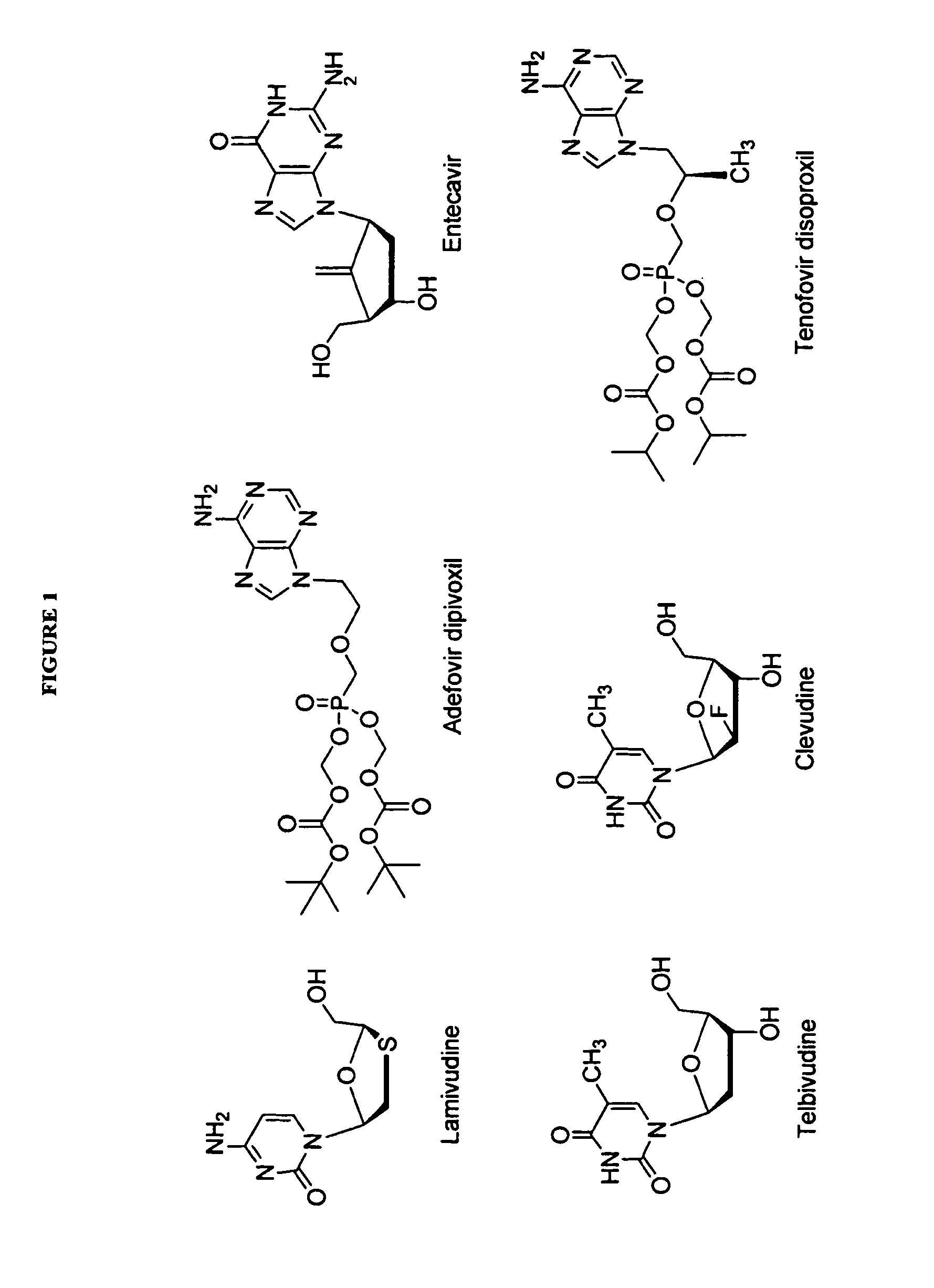
The present invention relates to 2′-Fluoro-6′-methylene carbocyclic nucleosides, pharmaceutical compositions containing these nucleosides and their use in the treatment or prophylaxis of a number of viral infections and secondary disease states and conditions thereof, especially including Hepatitis B virus (HBV) and secondary disease states and conditions thereof (cirrhosis and liver cancer), Heptatitis C virus (HCV), Herpes Simplex virus I and II (HSV-1 and HSV-2), cytomegalovirus (CMV), Varicella-Zoster Virus (VZV) and Epstein Barr virus (EBV) and secondary cancers which occur thereof (lymphoma, nasopharyngeal cancer, including drug resistant (especially including lamivudine and / or adefovir resistant) and other mutant forms of these viruses.
Owner:UNIV OF GEORGIA RES FOUND INC
2′-fluoro-6′methylene carbocyclic nucleosides and methods of treating viral infections
ActiveUS8946244B2Reduce the possibilityBiocideSugar derivativesHerpes zoster virusNasopharyngeal cancer
The present invention relates to 2′-Fluoro-6′-methylene carbocyclic nucleosides, pharmaceutical compositions containing these nucleosides and their use in the treatment or prophylaxis of a number of viral infections and secondary disease states and conditions thereof, especially including Hepatitis B virus (HBV) and secondary disease states and conditions thereof (cirrhosis and liver cancer), Hepatitis C virus (HCV), Herpes Simplex virus I and II (HSV-1 and HSV-2), cytomegalovirus (CMV), Varicella-Zoster Virus (VZV) and Epstein Barr virus (EBV) and secondary cancers which occur thereof (lymphoma, nasopharyngeal cancer, including drug resistant (especially including lamivudine and / or adefovir resistant) and other mutant forms of these viruses.
Owner:UNIV OF GEORGIA RES FOUND INC
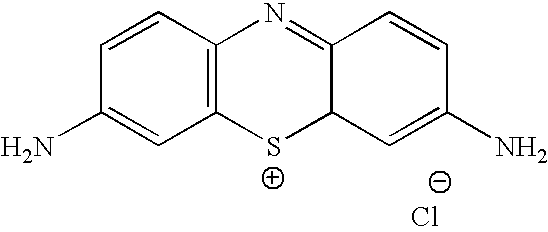
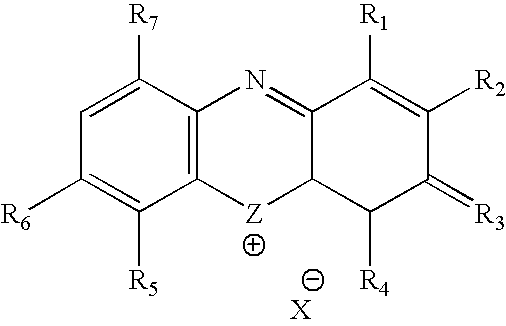
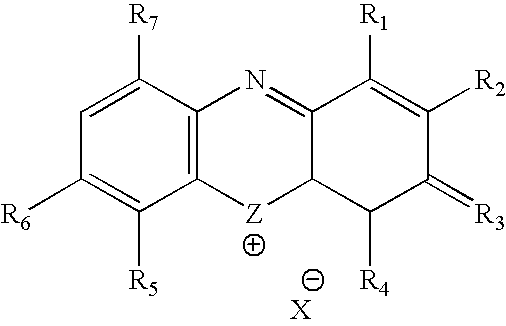
Methylene Blue Therapy of Viral Disease
InactiveUS20060264423A1Avoid infectionHeterocyclic compound active ingredientsAgainst vector-borne diseasesVirus typeHerpes zoster virus
A method for using thiazine dyes, especially methylene blue or methylene blue derivatives, in an immediate or controlled release formulation, alone or in combination with low levels of light or other drugs, to selectively inactivate or inhibit hepatitis infection, has been developed. Clinical trial results demonstrate efficacy in a human clinical trial for treatment of hepatitis C by oral administration of methylene blue immediate release formulation, in a dosage of 65 mg twice daily, over a period of at least 100 days. A method for using thiazine dyes, especially methylene blue or methylene blue derivatives, in an immediate or controlled release formulation, along or in combination with low levels of light or other drugs, to prevent or decrease reactivation of viruses, is also described. The preferred class of patient is infected with, or has been exposed to, viruses such as Herpes simplex virus type 1 & 2, Varicella zoster virus, Epstein-Barr virus, Cytomegalovirus, and Herpes virus type 6 & 7, Adenovirus, and Human polyoma viruses, e.g. JC virus and BK virus. In one embodiment the thiazine dye is administered to a patient experiencing symptoms or disease caused by reactivation of a virus. In a preferred embodiment the thiazine dye is administered to a patient at risk for or experiencing symptoms or disease caused by reactivation of a virus, prior to or during immunosuppression or chemotherapy.
Owner:BIOENVISION
HLA binding peptides and their uses
InactiveUS7252829B1Effective combinationTumor rejection antigen precursorsSsRNA viruses positive-senseImmunodeficiency virusBinding peptide
The present invention provides the means and methods for selecting immunogenic peptides and the immunogenic peptide compositions capable of specifically binding glycoproteins encoded by HLA alleles and inducing T cell activation in T cells restricted by the allele. The peptides are useful to elicit an immune response against a desired antigen. The immunogenic peptide compositions of the present invention comprise immunogenic peptides having an HLA binding motif, where the peptide is from a target antigen. Target antigens of the present invention include prostate specific antigen (PSA), hepatitis B core and surface antigens (HBVc, HBVs) hepatitis C antigens, Epstein-Barr virus antigens, melanoma antigens (e.g., MAGE-1), human immunodeficiency virus (HIV) antigens, human papilloma virus (HPV) antigens, Lassa virus, mycobacterium tuberculosis (MT), p53, CEA, trypanosome surface antigen (TSA) and Her2 / neu. An example of an immunogenic peptide of the present invention corresponds to a peptide less than about 15 amino acids in length that comprises an HLA-A2.1 binding motif, where the peptide comprises the p53 sequence SMPPPGTRV.
Owner:OSE PHARMA INT
2'-Fluoro-6'-Methylene Carbocyclic Nucleosides and Methods of Treating Viral Infections
The present invention relates to 2′-Fluoro-6′-methylene carbocyclic nucleosides, pharmaceutical compositions containing these nucleosides and their use in the treatment or prophylaxis of a number of viral infections and secondary disease states and conditions thereof, especially including Hepatitis B virus (HBV) and secondary disease states and conditions thereof (cirrhosis and liver cancer), Heptatitis C virus (HCV), Herpes Simplex virus I and II (HSV-1 and HSV-2), cytomegalovirus (CMV), Varicella-Zoster Virus (VZV) and Epstein Barr virus (EBV) and secondary cancers which occur thereof (lymphoma, nasopharyngeal cancer, including drug resistant (especially including lamivudine and / or adefovir resistant) and other mutant forms of these viruses.
Owner:UNIV OF GEORGIA RES FOUND INC
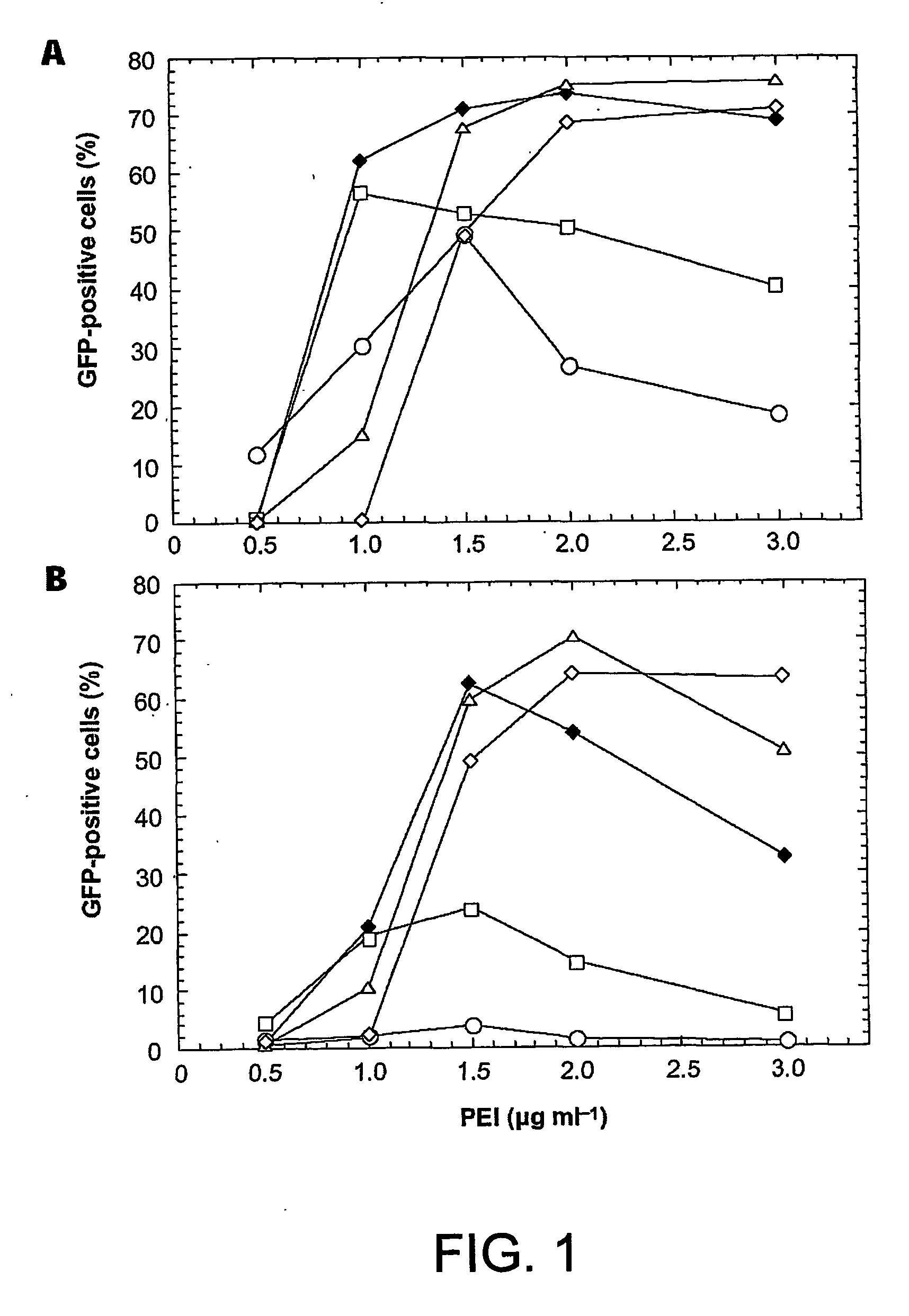
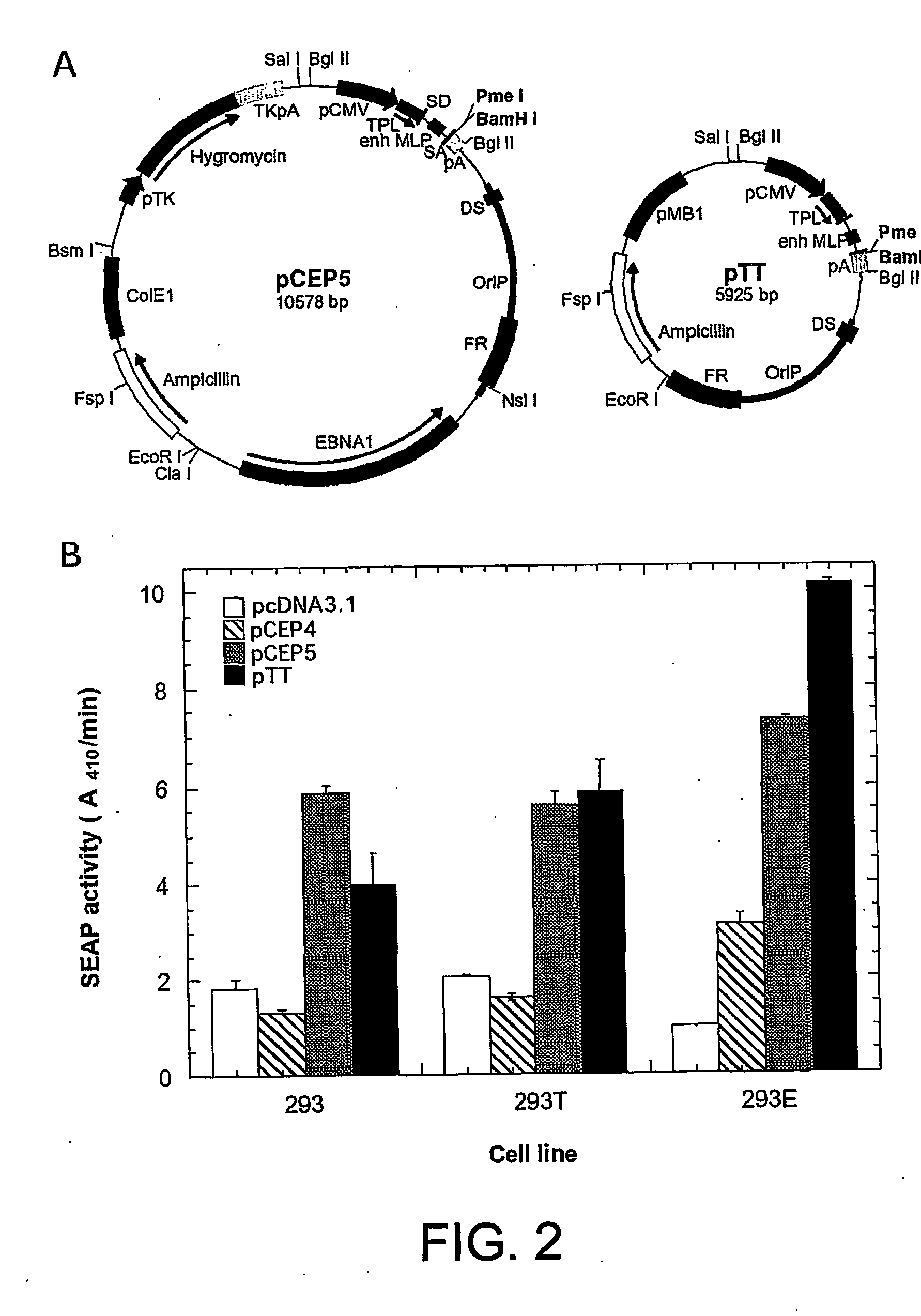
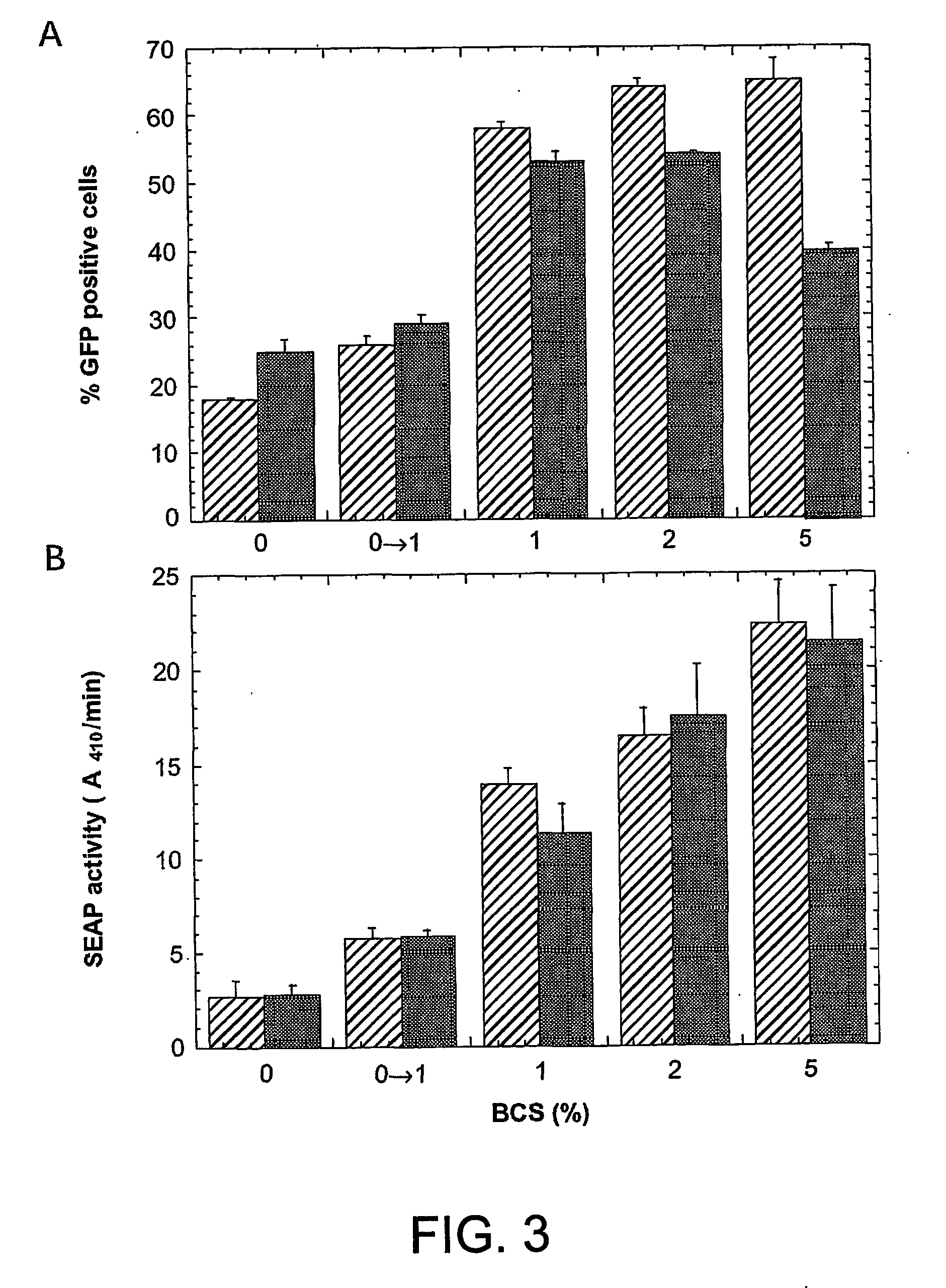
Enhanced production of recombinant proteins by transient transfection of suspension-growing mammalian cells
ActiveUS20050170450A1Increase transcriptional activityIncreasing nuclear importGenetically modified cellsVirus peptidesEpstein–Barr virusGene Variant
Disclosed is a new process for the production of recombinant proteins, by transient transfection of suspension-grown human embryonic kidney cells (293 cell line and its genetic variants) with an expression vector, using polyethylenimine (PEI) as a transfection reagent. In a preferred embodiment, the process uses 293E cells expressing the Epstein-Barr virus (EBV) EBNA 1 protein, in combination with an oriP-based episomal expression vector having an improved cytomegalovirus expression cassette comprising the CMV5 promoter. The process combines in a single step the cell growth, transfection and protein expression, is carried out without changing the culture medium, and allows to achieve high expression levels in a short period of time. The process may be carried out in a serum-free, low-protein culture medium, is easily scalable, compatible with continuous production processes, and fully adapted to high-throughput production of milligram quantities of recombinant proteins.
Owner:NAT RES COUNCIL OF CANADA
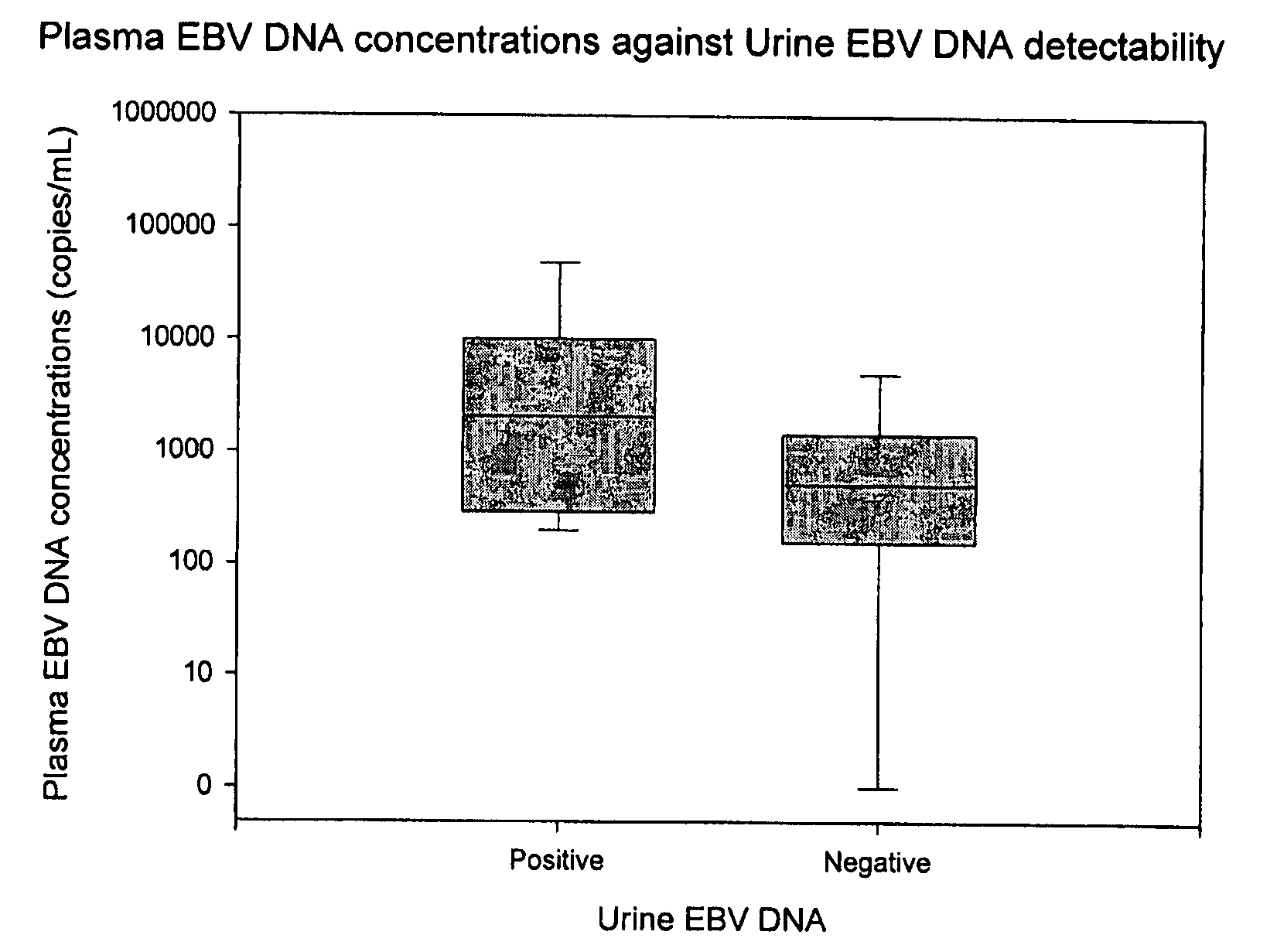
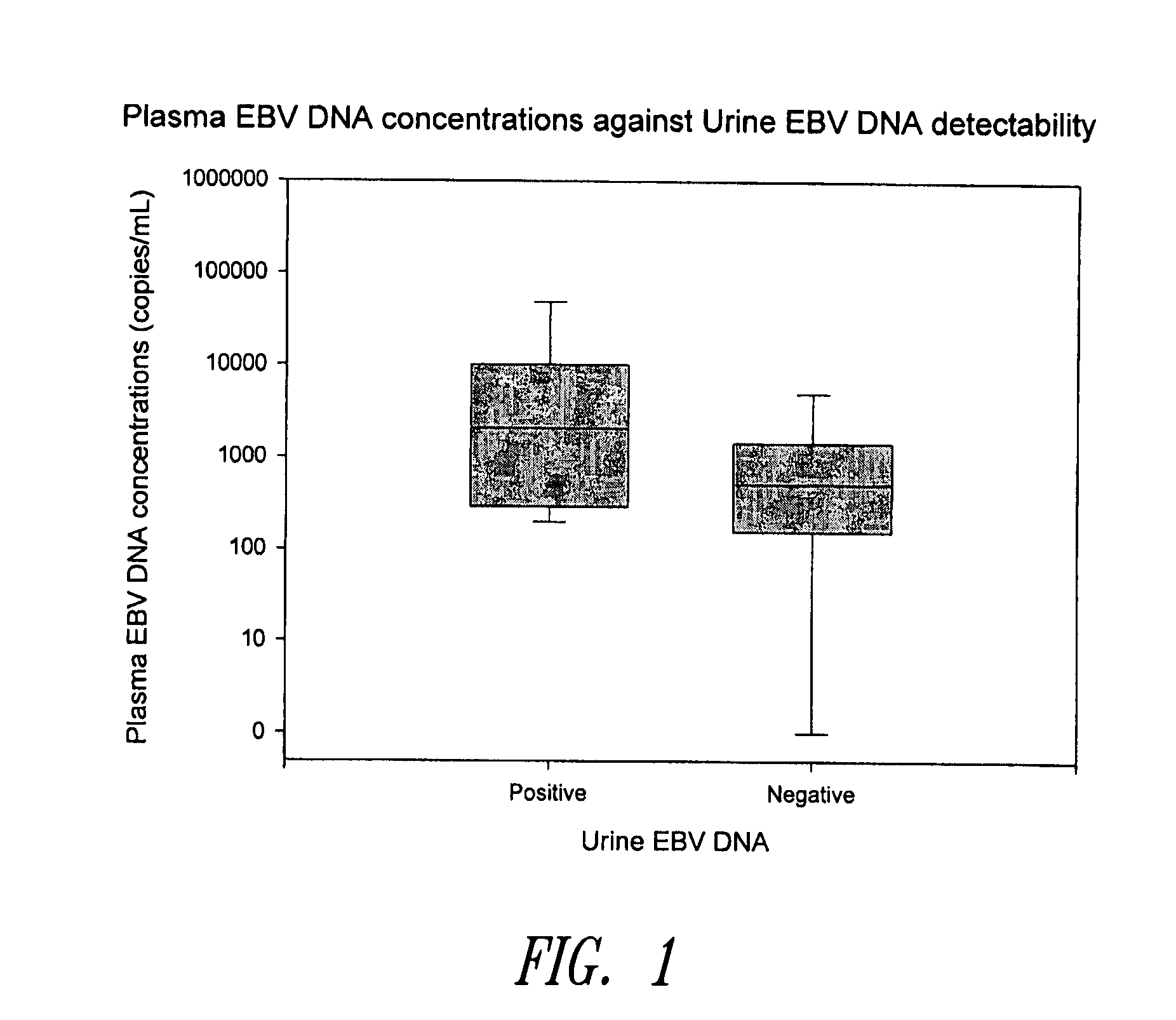
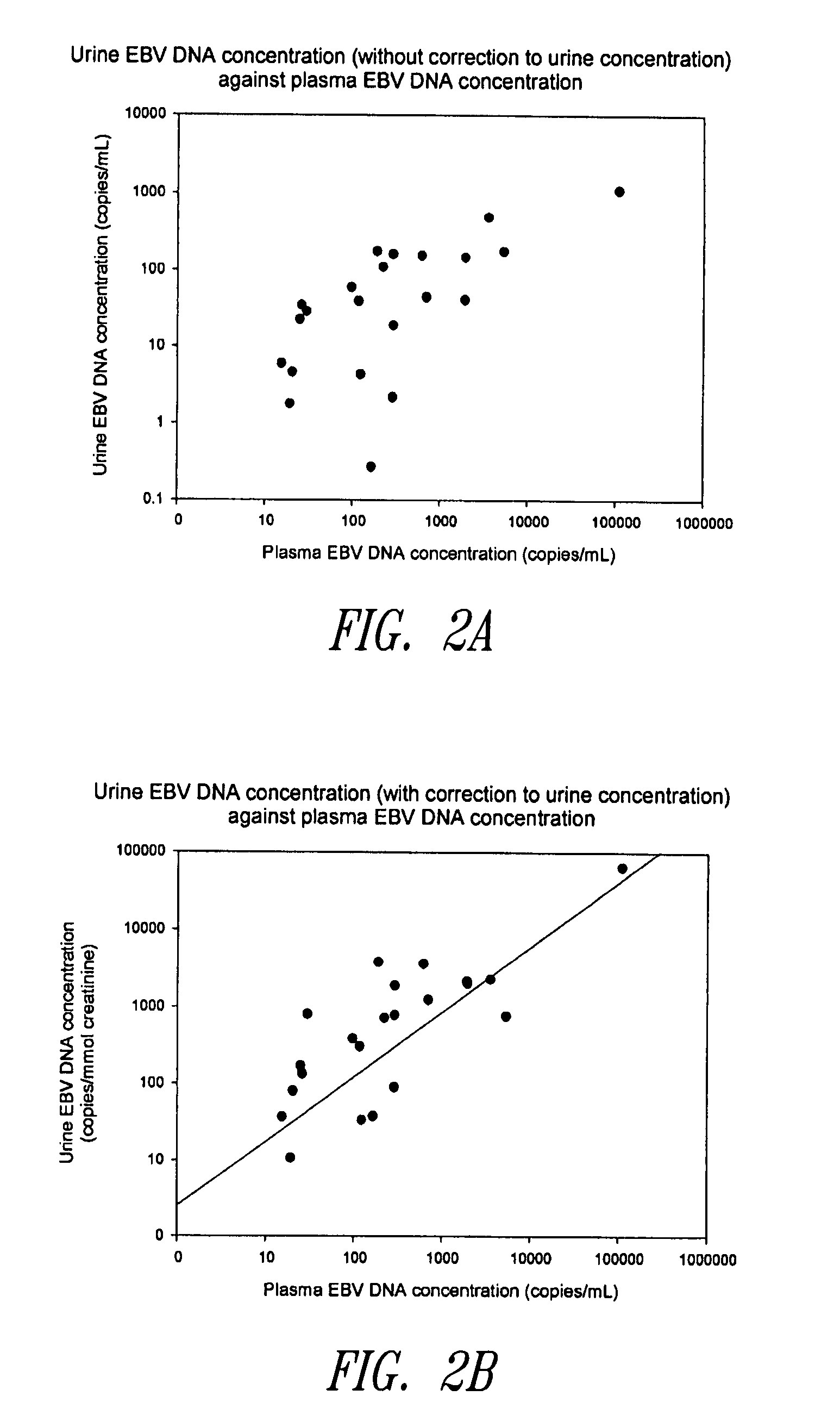
Methods and kits for diagnosis, prognosis or monitoring of Epstein-Barr virus (EBV)-associated cancer
InactiveUS20080206749A1Lower Level RequirementsEliminate false positive caseMicrobiological testing/measurementFermentationBacteriuriaNon invasive
Disclosed is a non-invasive method for diagnosis, prognosis or monitoring of Epstein-Barr virus (EBV)-associated cancer by detecting and / or quantifying EBV associated nucleic acid fragments in a urine sample from an individual. Kits for diagnosis, prognosis or monitoring of cancer are also disclosed.
Owner:HONG KONG THE CHINESE UNIV OF
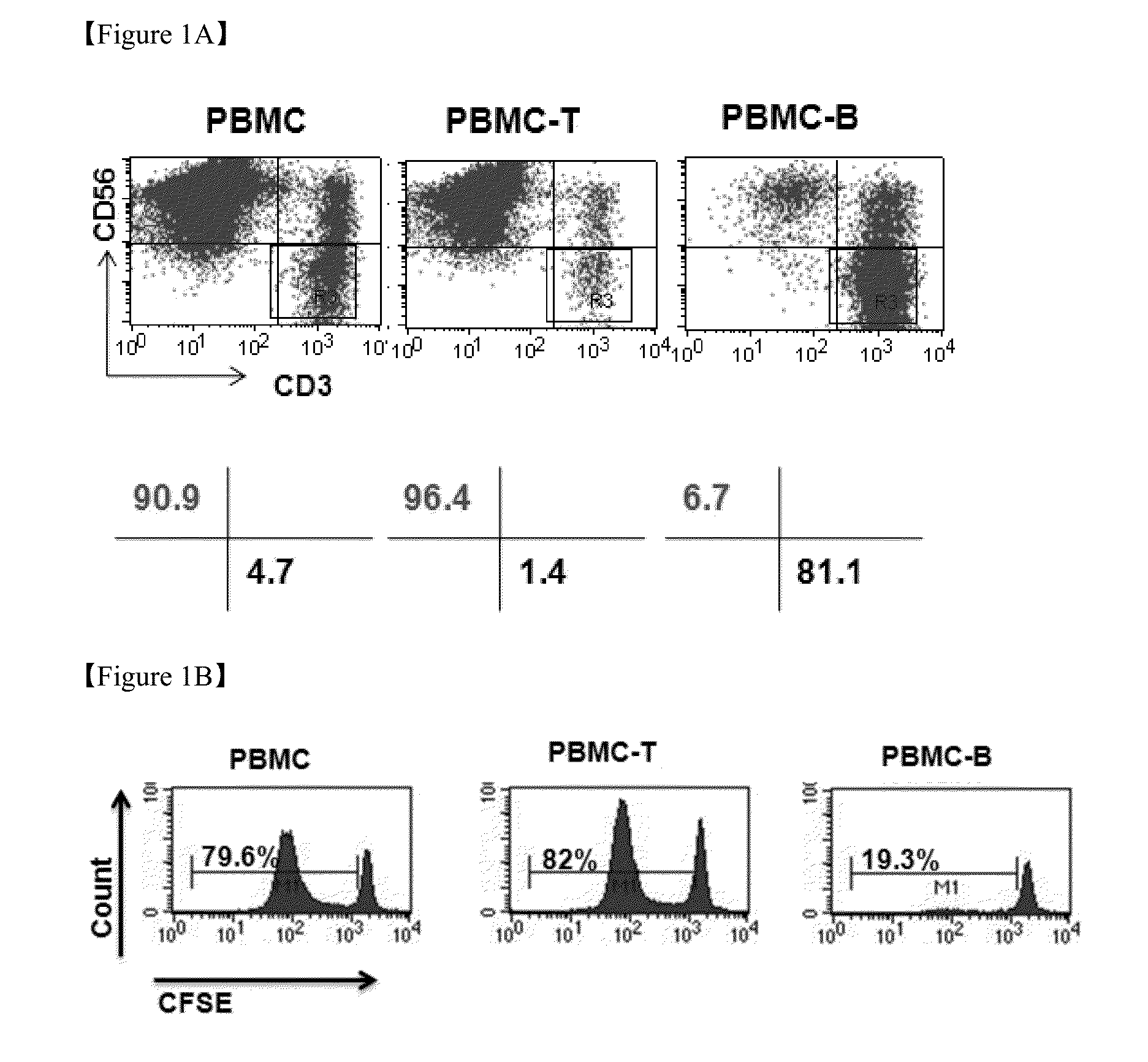
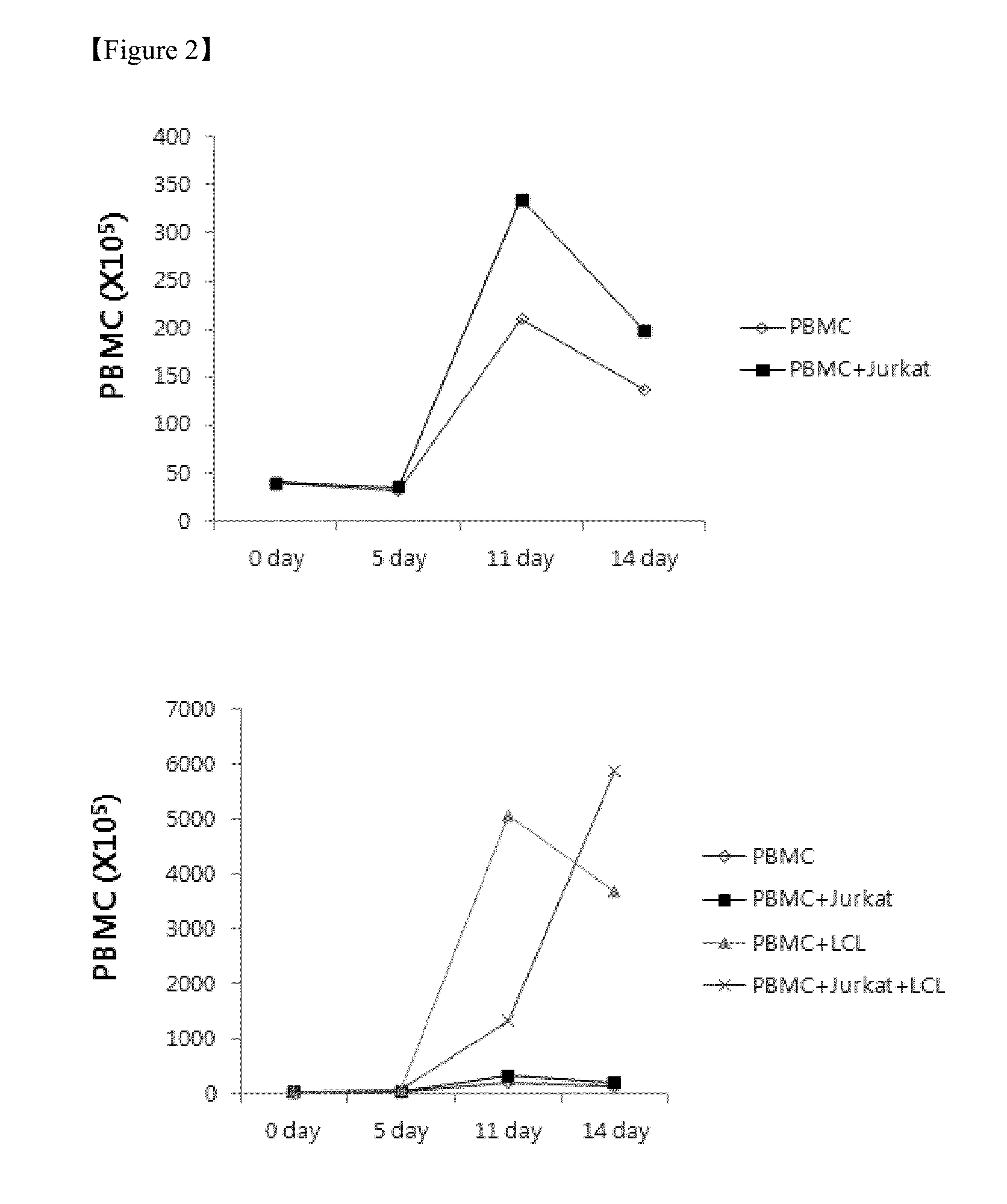
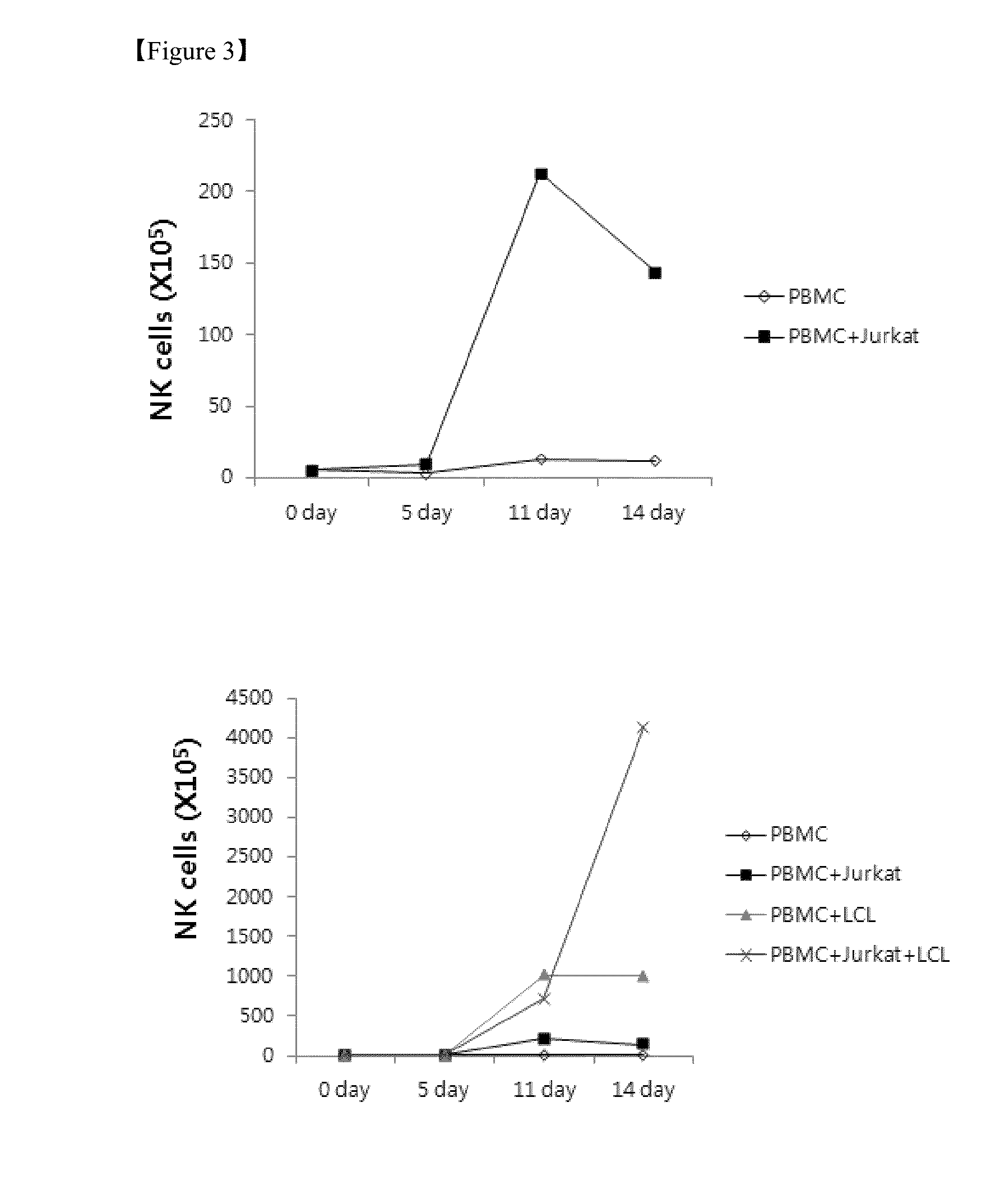
Method for the induction and expansion of natural killer cells derived from peripheral blood mononuclear cells
ActiveUS20150152387A1Improve efficiencyImprove efficacyBiocideMammal material medical ingredientsNatural Killer Cell FunctionNatural killer cell
The present invention relates to a method for inducing and expanding natural killer cells derived from peripheral blood mononuclear cells, which comprises co-culturing, as feeder cells, irradiated Jurkat cells and irradiated Epstein-Barr virus transformed lymphocyte continuous line (EBV-LCL) cells in the presence of cytokines, along with peripheral blood mononuclear cells. According to the present invention, a large quantity of natural killer cells can be induced and proliferated from a small quantity of peripheral blood mononuclear cells even without the use of high-cost equipment or various kinds of expensive cytokines, thereby making it possible to significantly improve the efficiency and efficacy of the prevention and treatment of cancer using the natural killer cells.
Owner:NKMAX CO LTD
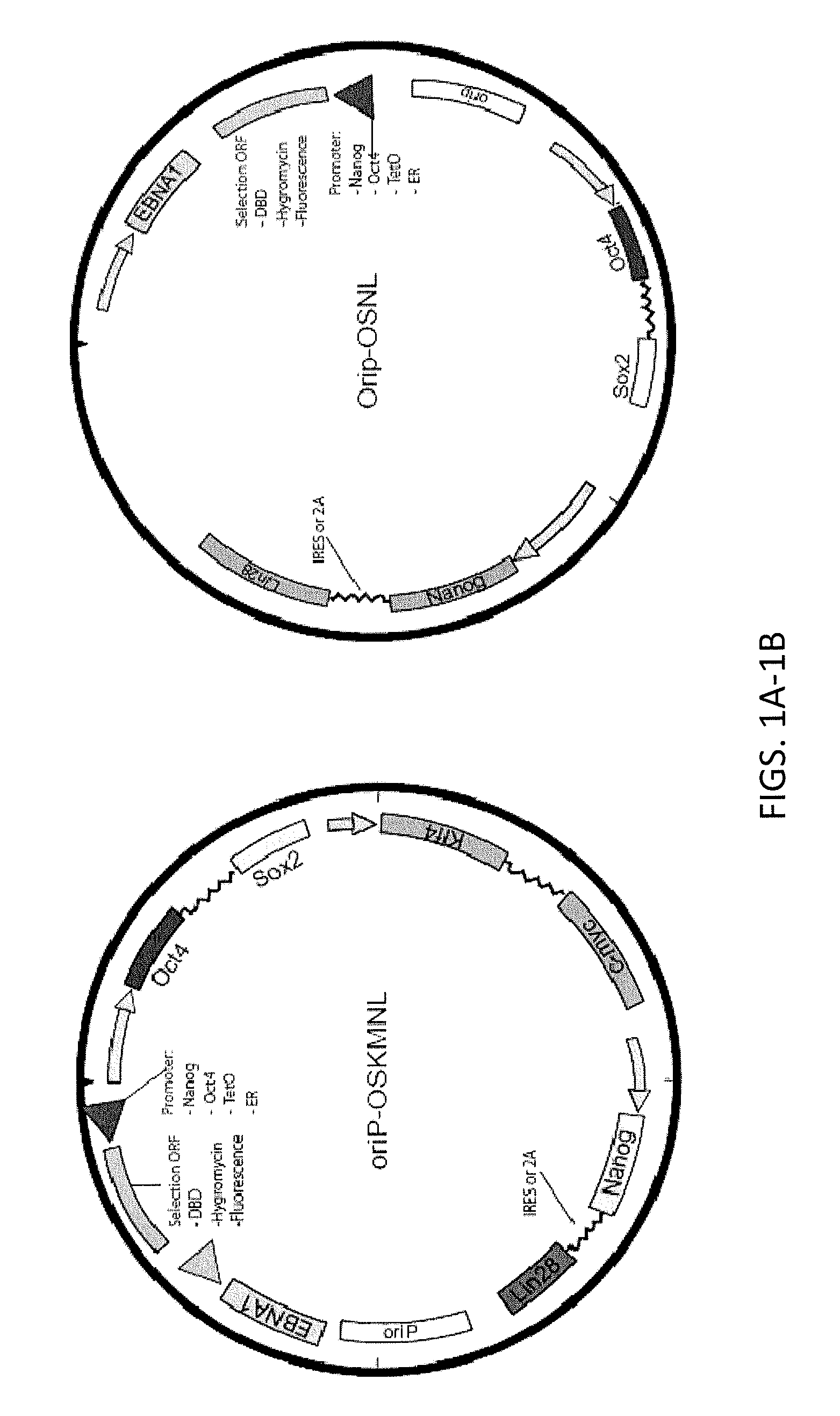
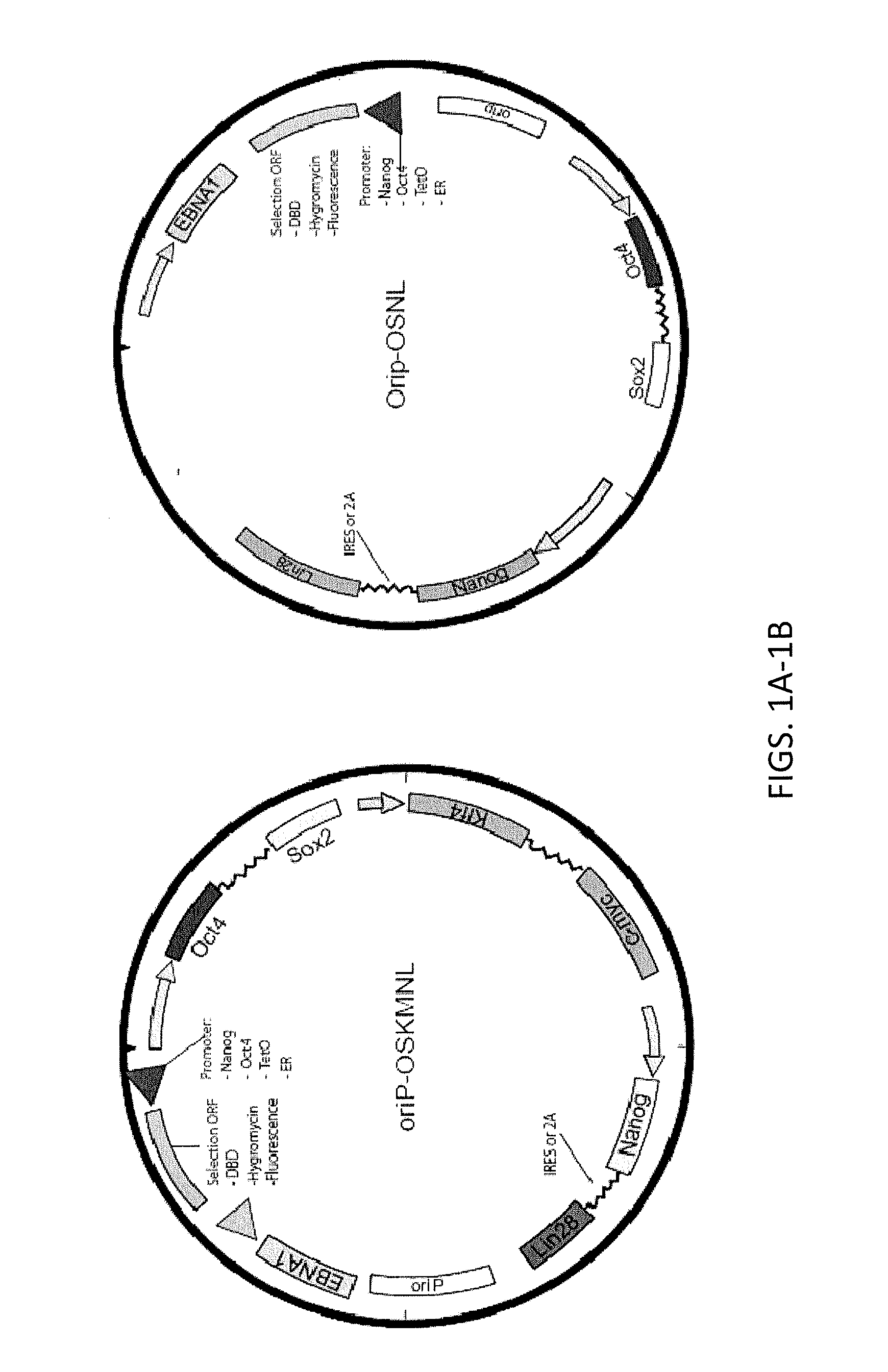
Reprogramming immortalized B-cells to induced pluripotent stem cells
ActiveUS8765470B2Minimize impactHigh reprogramming efficiencyGenetically modified cellsArtificial cell constructsLymphocyteB cell
Methods and composition for providing induced pluripotent stem (iPS) cells are provided. For example, in certain aspects methods including reprogramming B lymphocytes transformed by episomal vectors such as Epstein-Barr virus-based vectors are described. Furthermore, the invention provides induced pluripotent stem cells essentially free of exogenous elements and having B cell immunoglobin variable region rearrangement.
Owner:FUJIFILM CELLULAR DYNAMICS INC
Reprogramming immortalized b cells
ActiveUS20120058562A1High reprogramming efficiencySave stepsGenetically modified cellsArtificial cell constructsLymphocyteB cell
Methods and composition for providing induced pluripotent stem (iPS) cells are provided. For example, in certain aspects methods including reprogramming B lymphocytes transformed by episomal vectors such as Epstein-Barr virus-based vectors are described. Furthermore, the invention provides induced pluripotent stem cells essentially free of exogenous elements and having B cell immunoglobin variable region rearrangement.
Owner:FUJIFILM CELLULAR DYNAMICS INC
Treatment of EBV and KHSV infection
InactiveUS7211570B2Growth inhibitionIncreases activity and bioavailability and stabilityBiocideGenetic material ingredientsMedicineDiluent
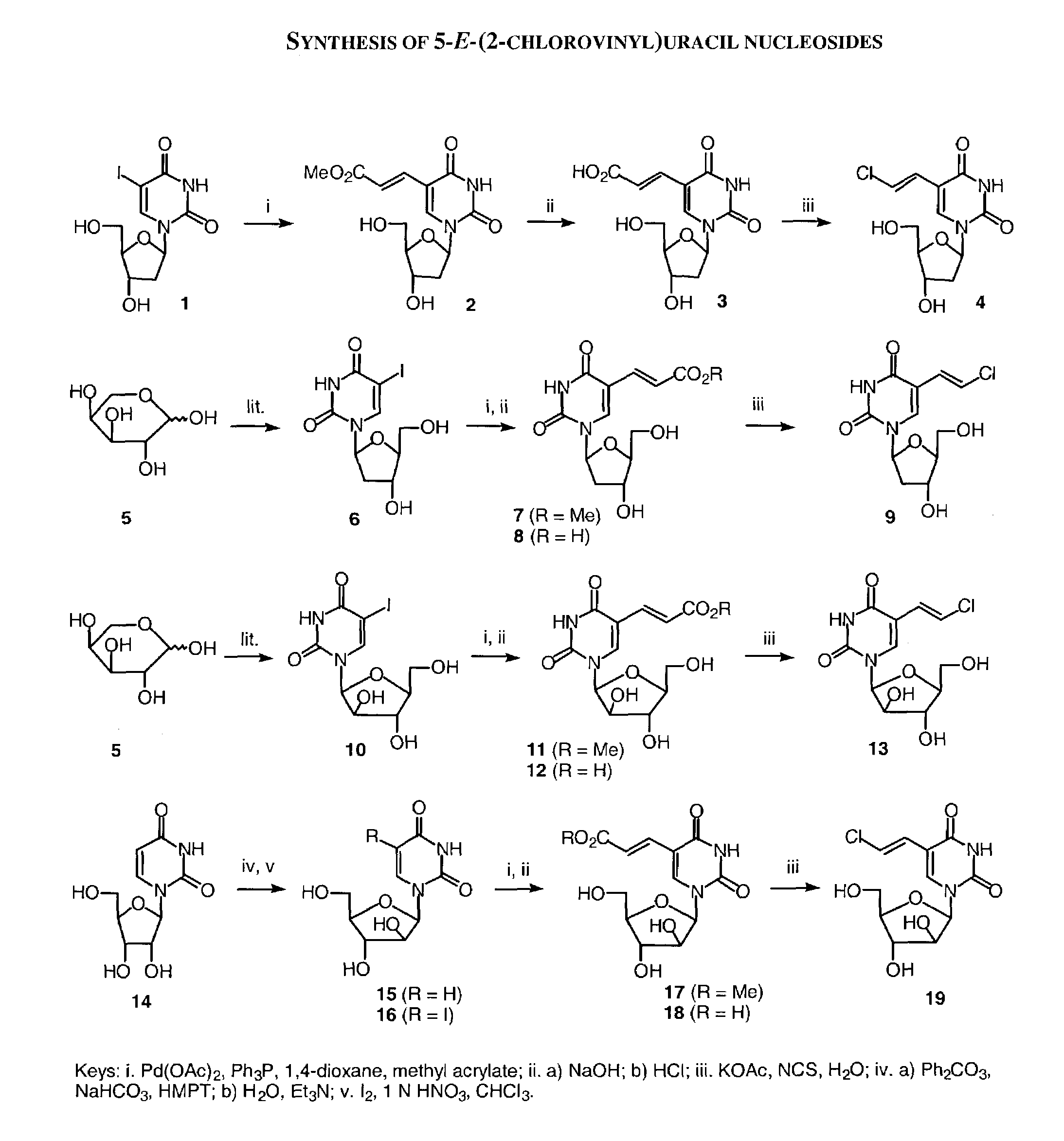
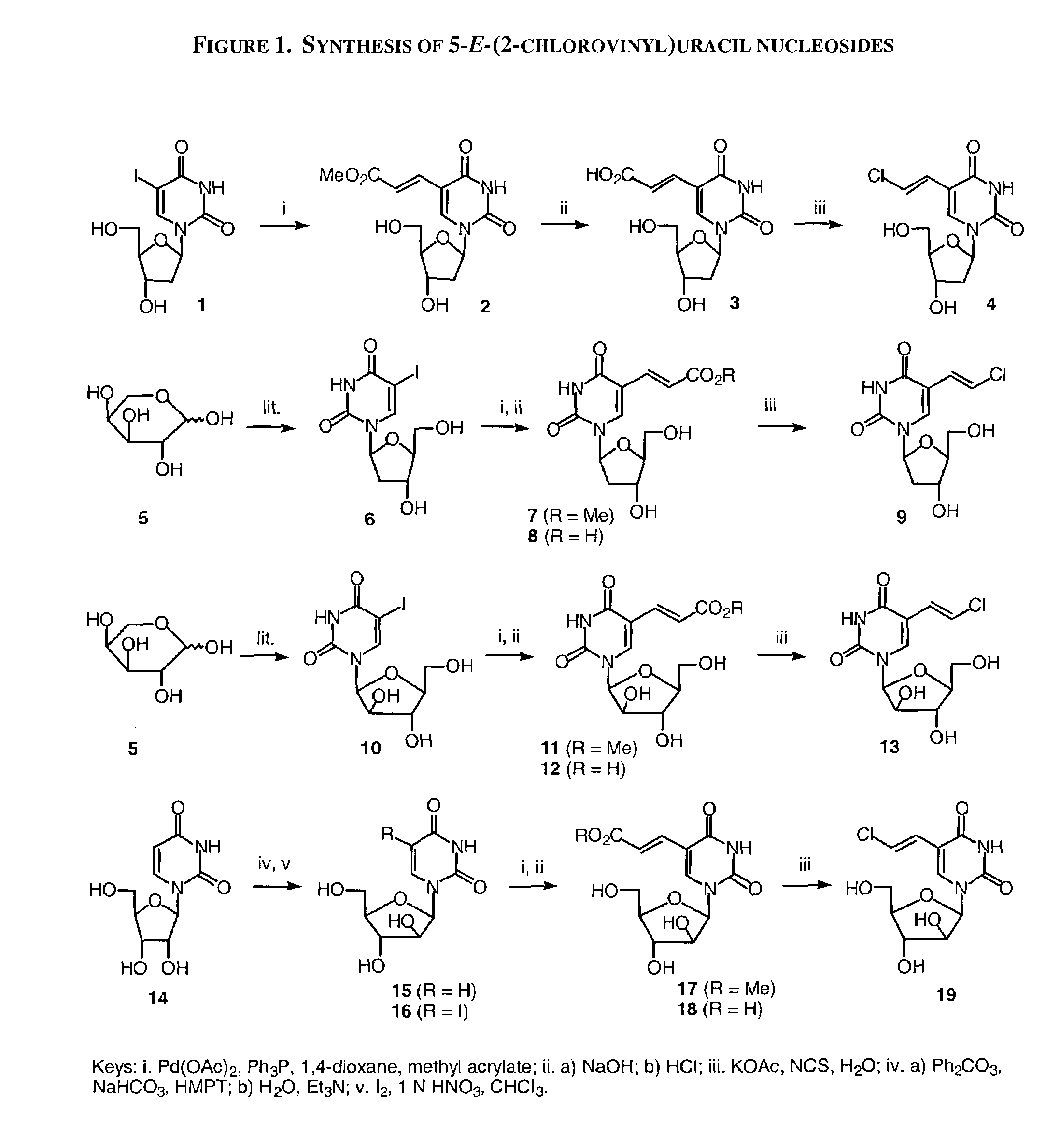
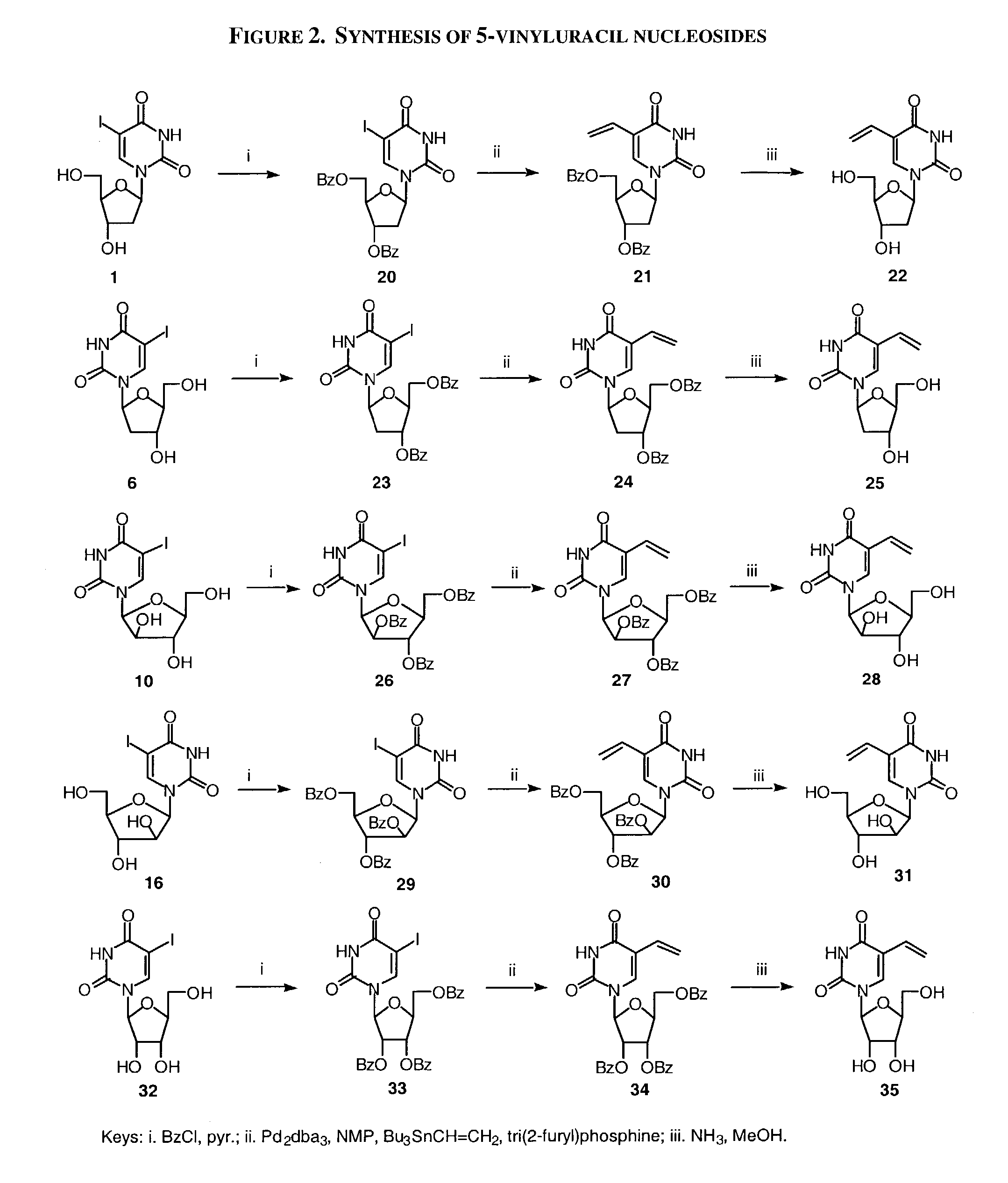
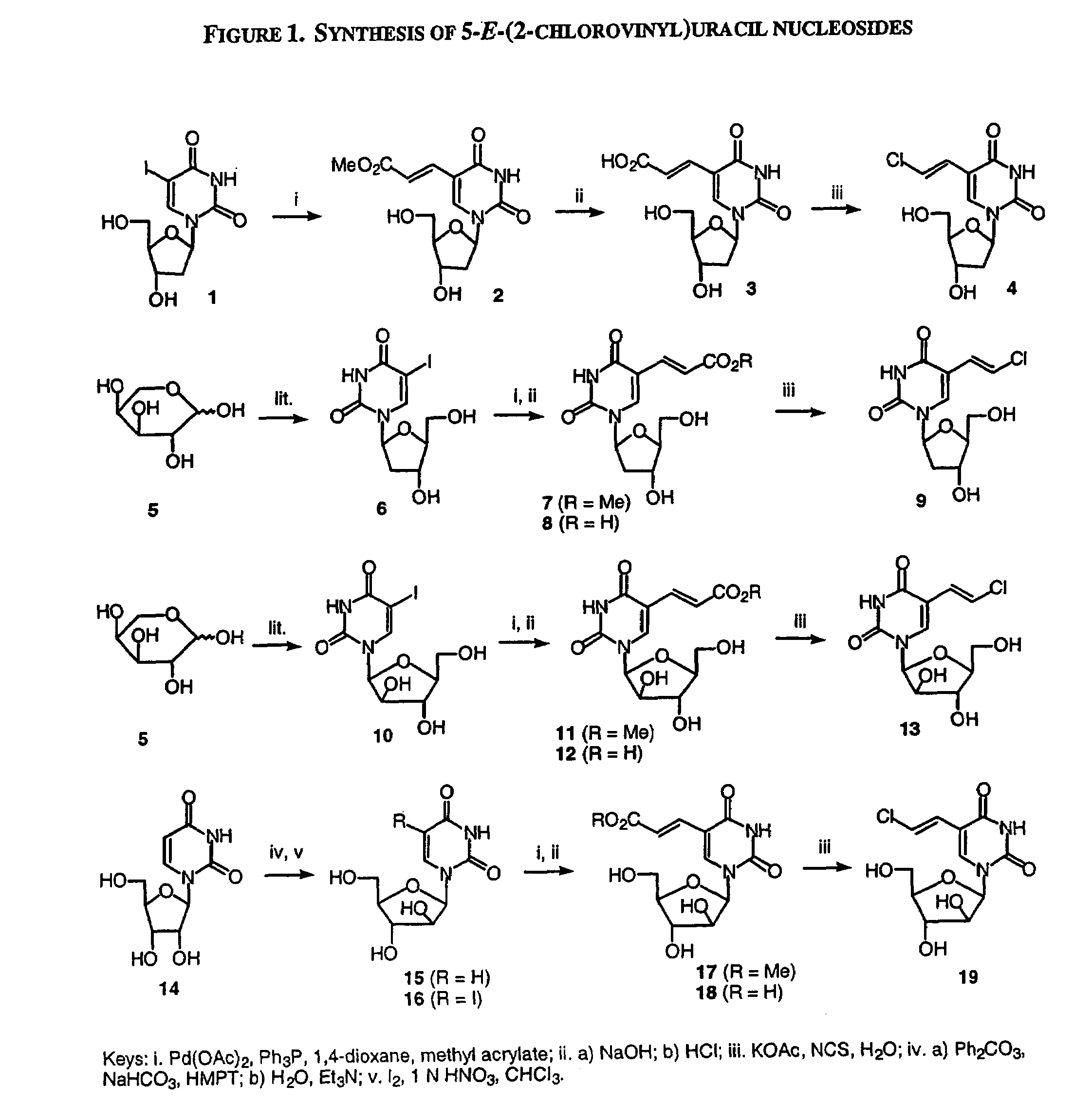
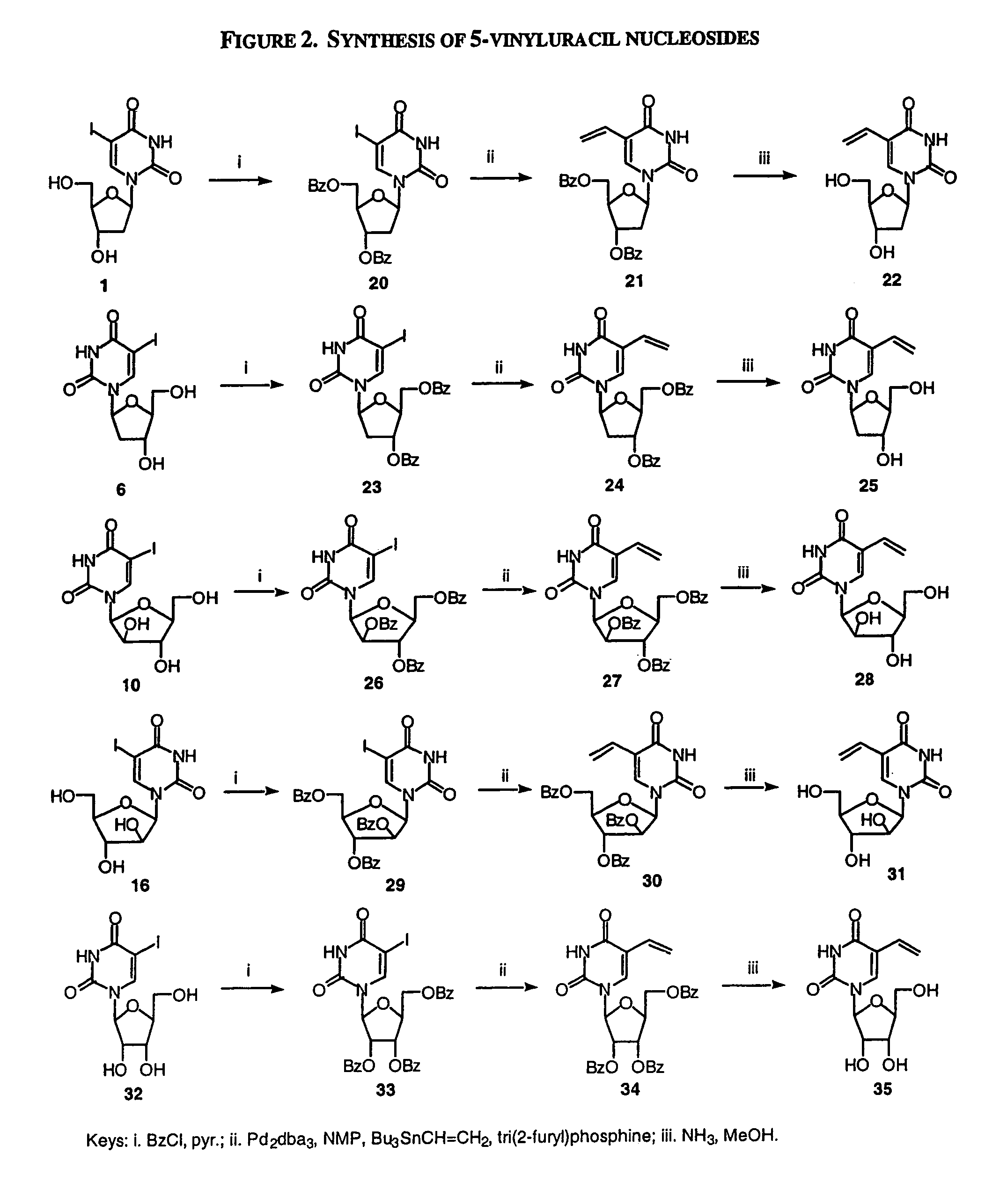
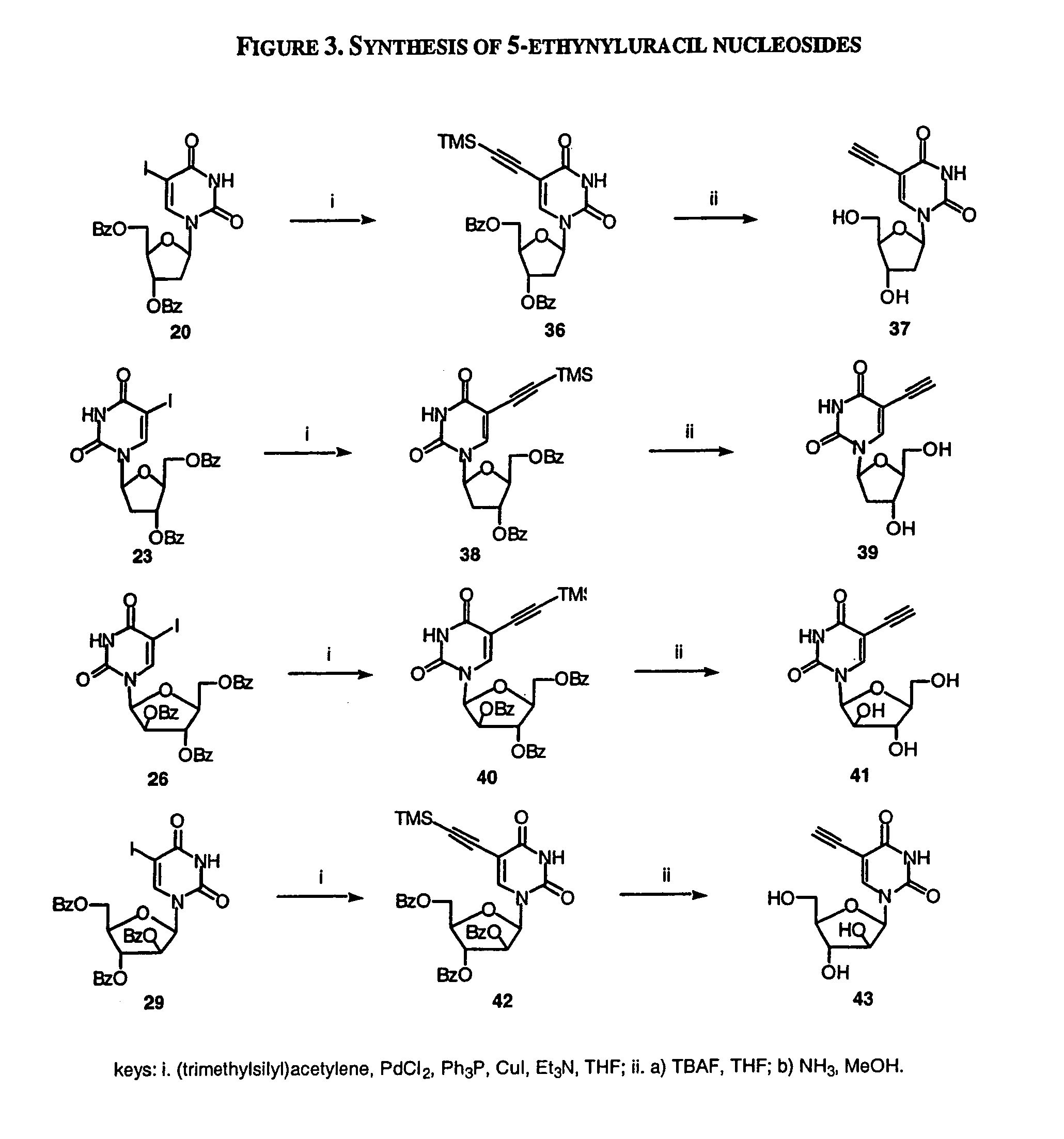
A method and composition for the treatment, prevention and / or prophylaxis of a host, and in particular, a human, infected with Epstein-Barr virus (EBV), is provided that includes administering an effective amount of a 5-substituted uracil nucleoside or its pharmaceutically acceptable salt or prodrug, optionally in a pharmaceutically acceptable diluent or excipient.
Owner:EMORY UNIVERSITY +1
Carcinogenesis inhibitors
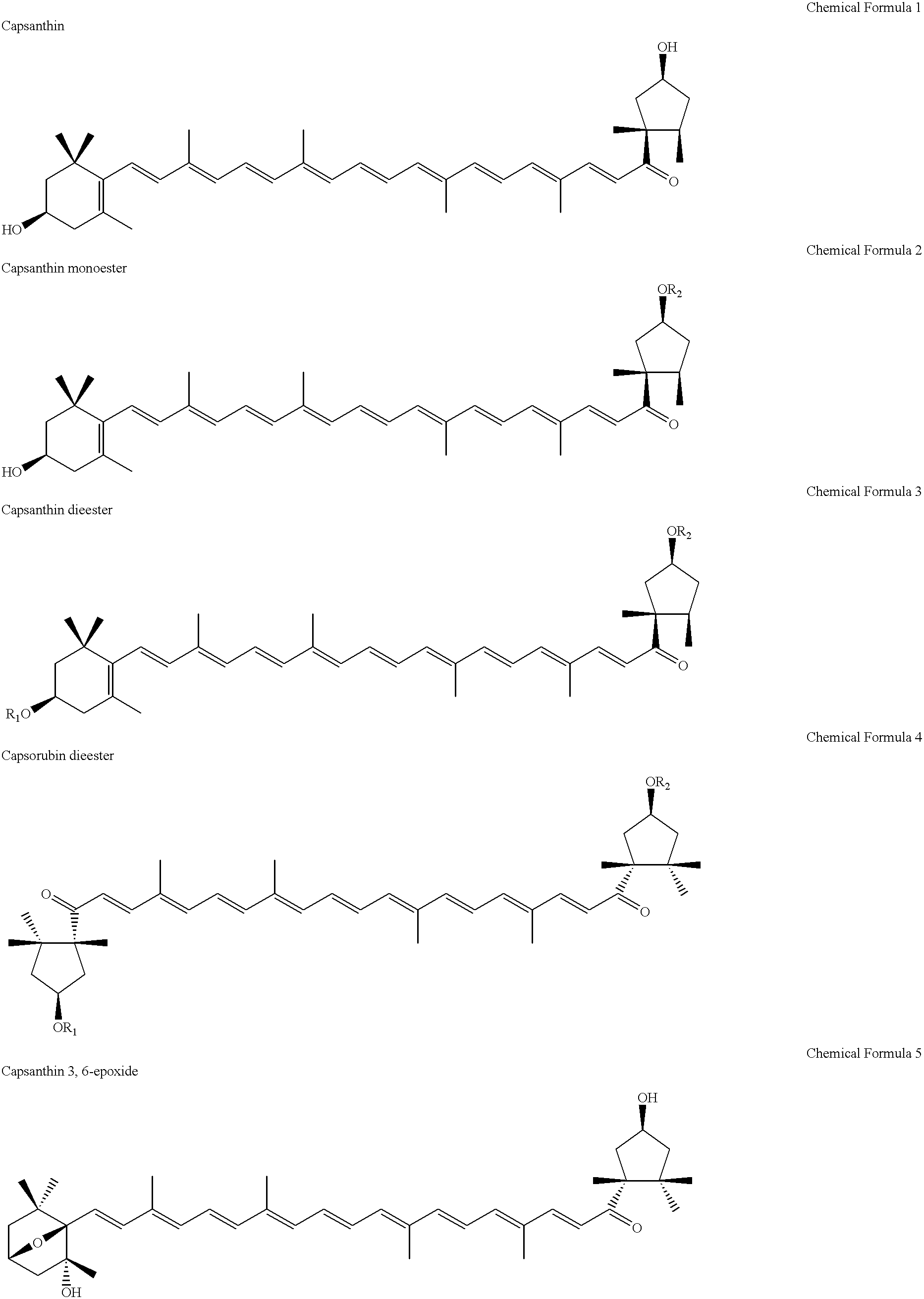
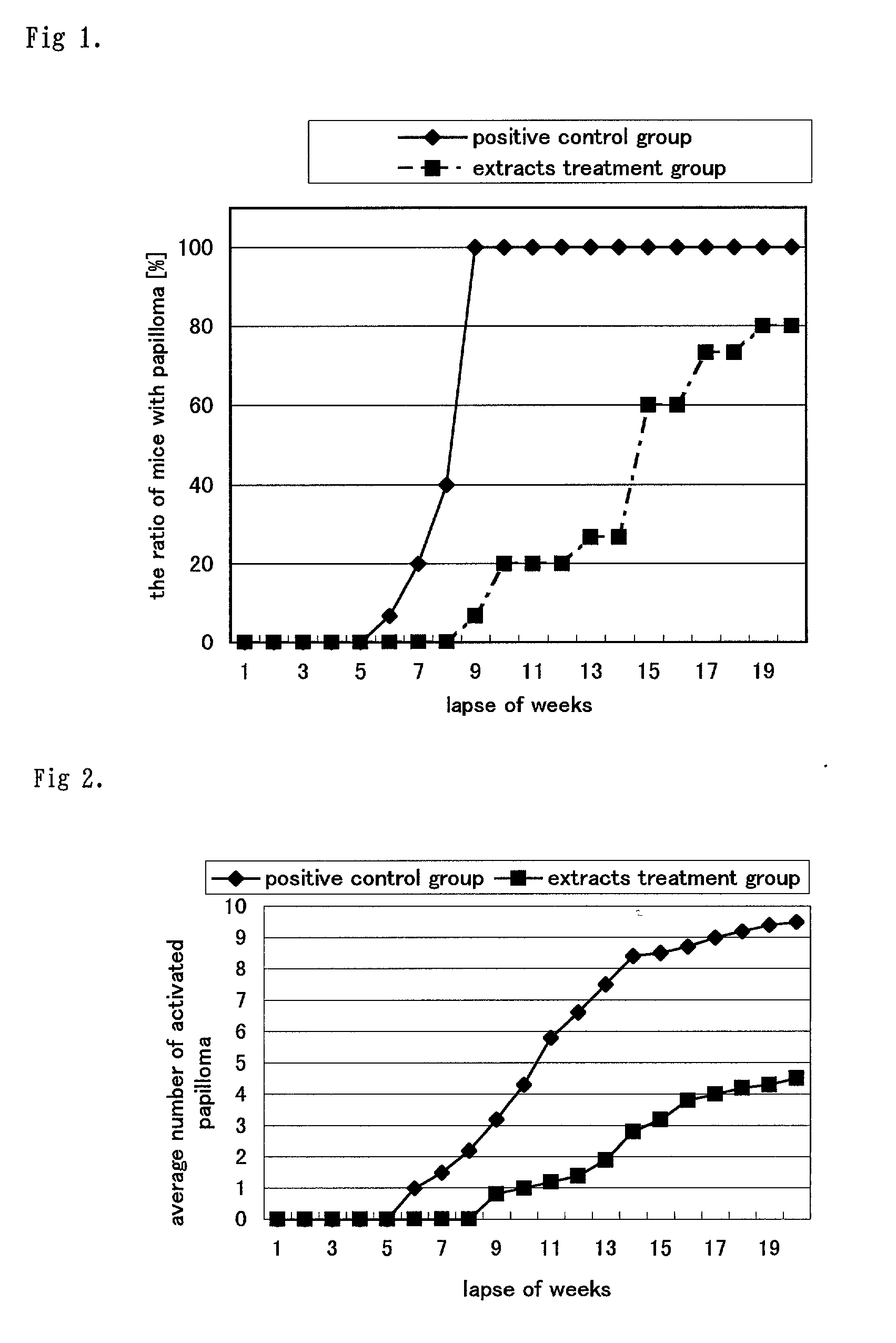
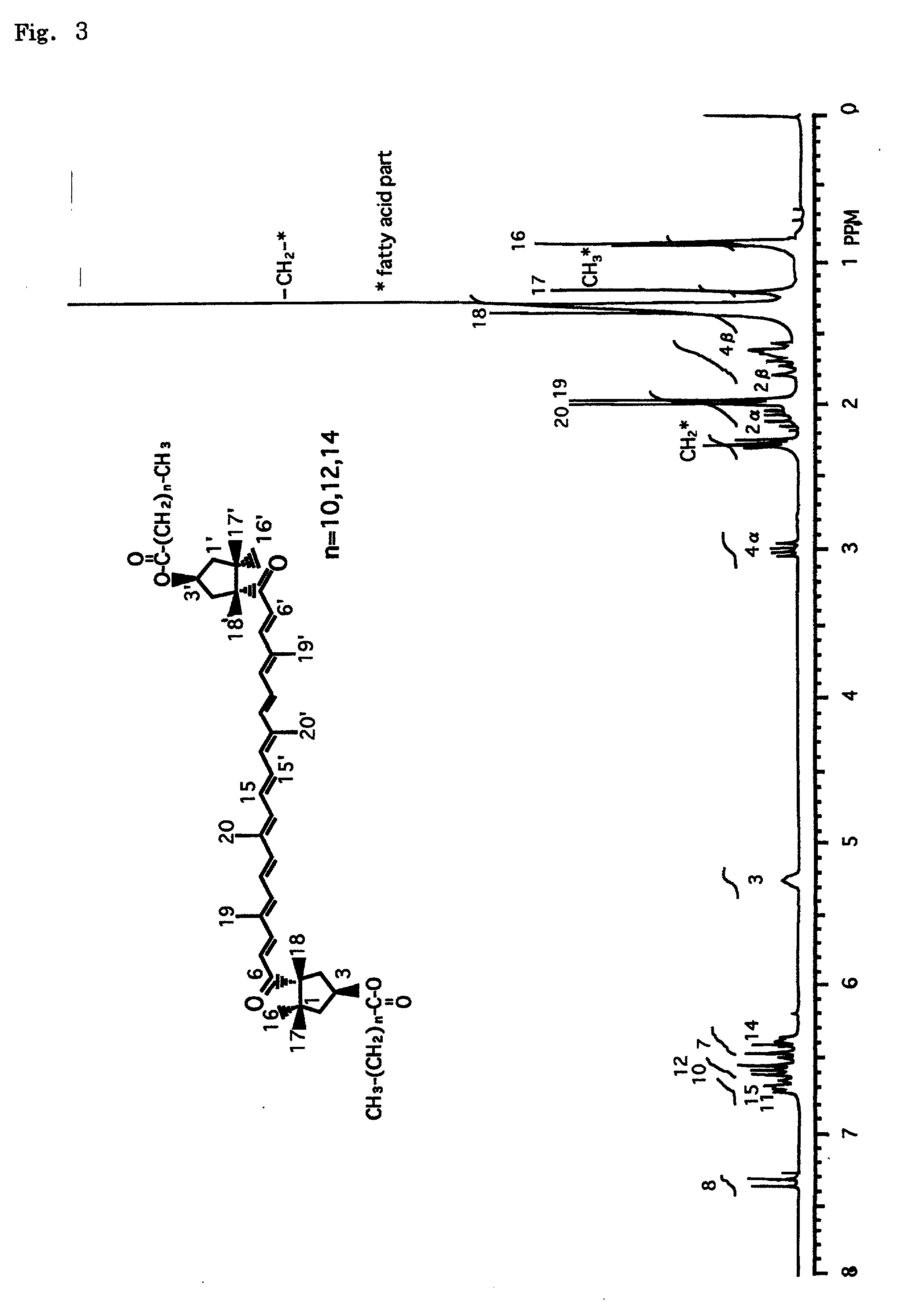
Carcinogenesis inhibitors containing as the active ingredient carotenoids extracted from the pure-line species of paradicsom paprika (species classified as Capsicum annuum L. var. grossum), etc., such as capsanthin, its fatty acid esters, capsorubin diesters, capsanthin 3,6-epoxide, capsorubin and cucurbitaxanthine A-3' ester. These carcinogenesis inhibitors and paradicsom paprika extracts originate in natural substances and, therefore, make it possible to provide excellent Epstein-Barr virus genome inactivating agents. Thus, they are expected as being useful in preventing carcinogenesis and, based on their effects, applicable in various fields including drugs, cosmetics and health foods.
Owner:DESIGNER FOODS ASSOC
2'-fluoro-6'-methylene carbocyclic nucleosides and methods of treating viral infections
ActiveUS20130005677A1Reduce infectious virus titerReduce cell viabilityBiocideSugar derivativesHerpes simplex diseaseCirrhosis
The present invention relates to 2′-Fluoro-6′-methylene carbocyclic nucleosides, pharmaceutical compositions containing these nucleosides and their use in the treatment or prophylaxis of a number of viral infections and secondary disease states and conditions thereof, especially including Hepatitis B virus (HBV) and secondary disease states and conditions thereof (cirrhosis and liver cancer), Heptatitis C virus (HCV), Herpes Simplex virus I and II (HSV-1 and HSV-2), cytomegalovirus (CMV), Varicella-Zoster Virus (VZV) and Epstein Barr virus (EBV) and secondary cancers which occur thereof (lymphoma, nasopharyngeal cancer, including drug resistant (especially including lamivudine and / or adefovir resistant) and other mutant forms of these viruses.
Owner:UNIV OF GEORGIA RES FOUND INC
HLA binding peptides and their uses
InactiveUS20090012004A1Effective combinationSsRNA viruses positive-senseTumor rejection antigen precursorsImmunodeficiency virusBinding peptide
The present invention provides the means and methods for selecting immunogenic peptides and the immunogenic peptide compositions capable of specifically binding glycoproteins encoded by HLA alleles and inducing T cell activation in T cells restricted by the allele. The peptides are useful to elicit an immune response against a desired antigen. The immunogenic peptide compositions of the present invention comprise immunogenic peptides having an HLA binding motif, where the peptide is from a target antigen. Target antigens of the present invention include prostate specific antigen (PSA), hepatitis B core and surface antigens (HBVc, HBVs) hepatitis C antigens, Epstein-Barr virus antigens, melanoma antigens (e.g., MAGE-1), human immunodeficiency virus (HIV) antigens, human papilloma virus (HPV) antigens, Lassa virus, mycobacterium tuberculosis (MT), p53, CEA, trypanosome surface antigen (TSA) and Her2 / neu.
Owner:OSE PHARMA INT
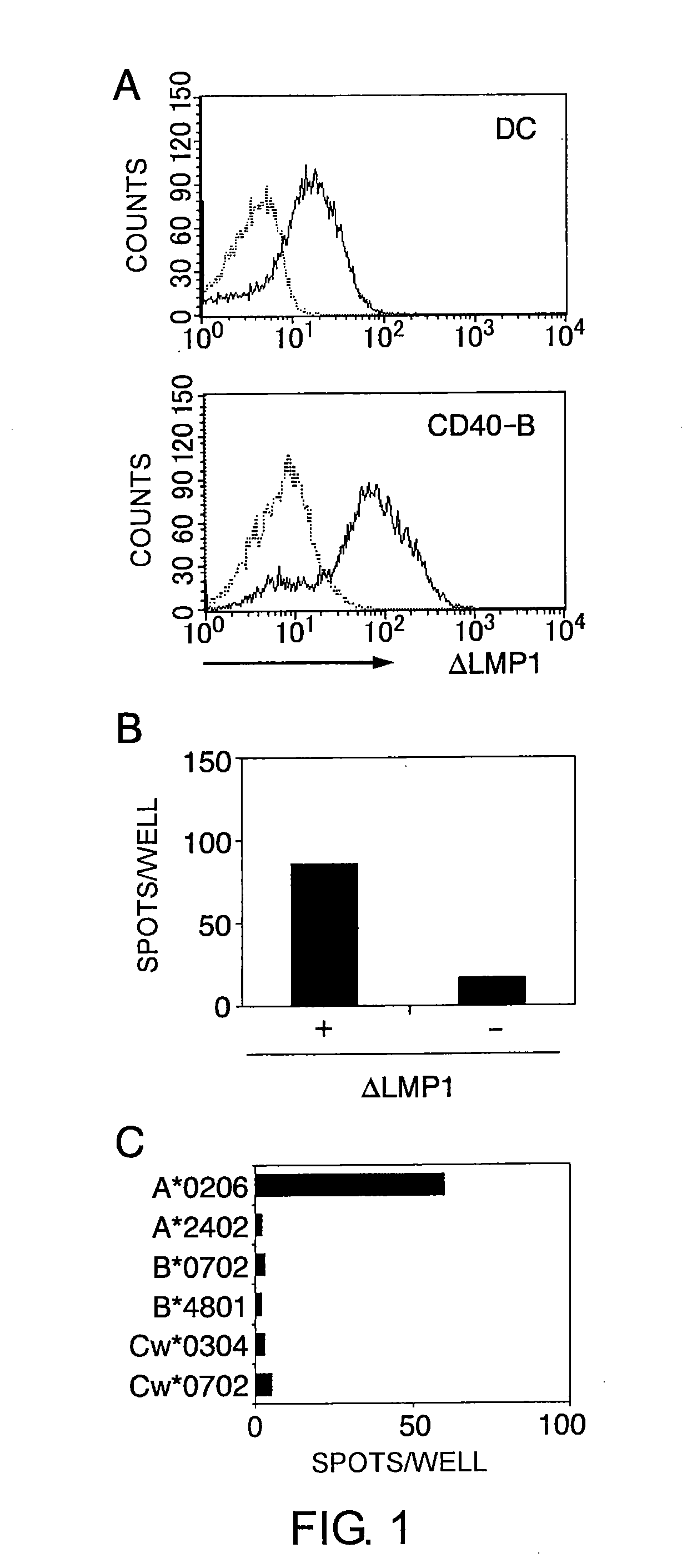
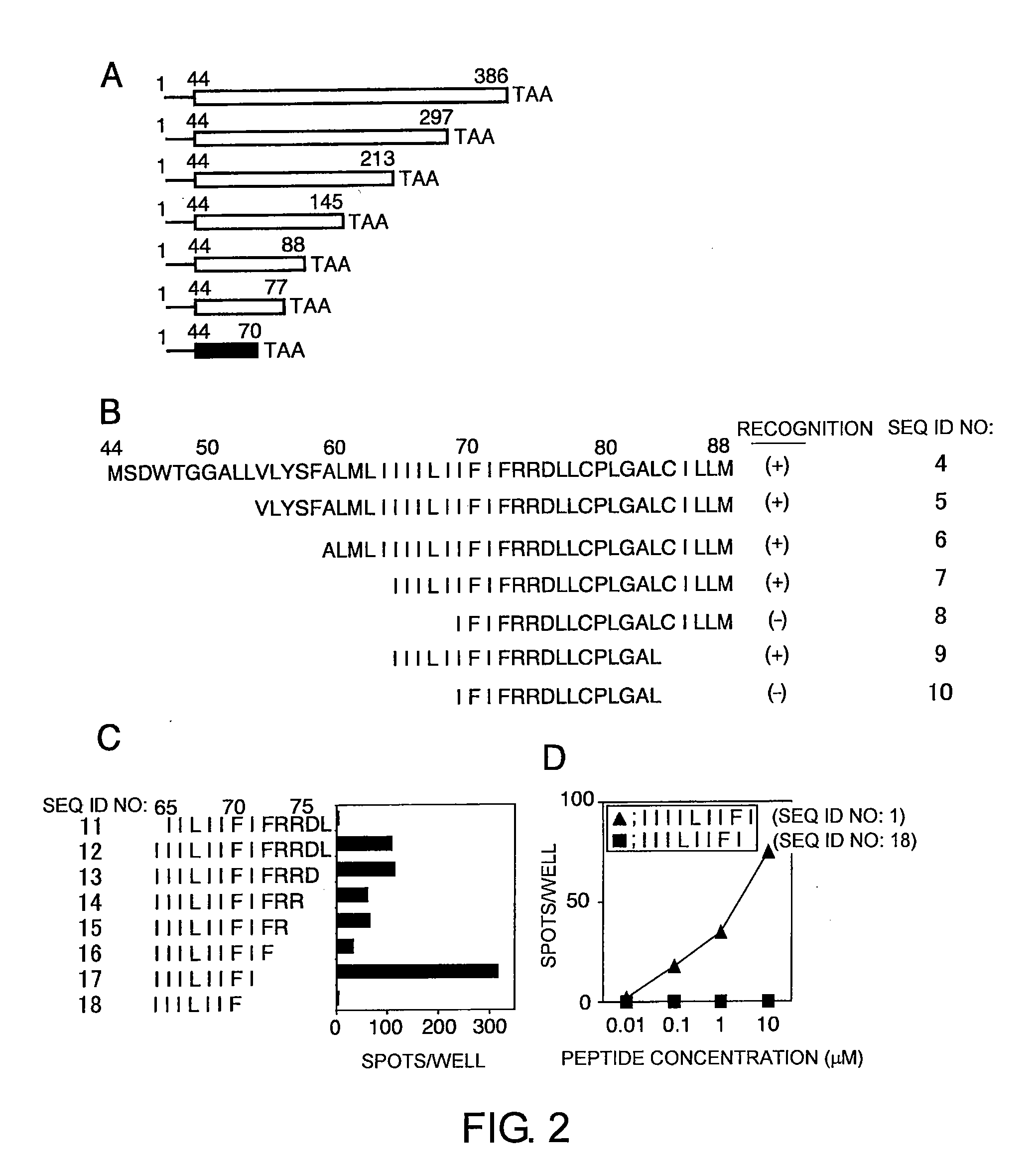
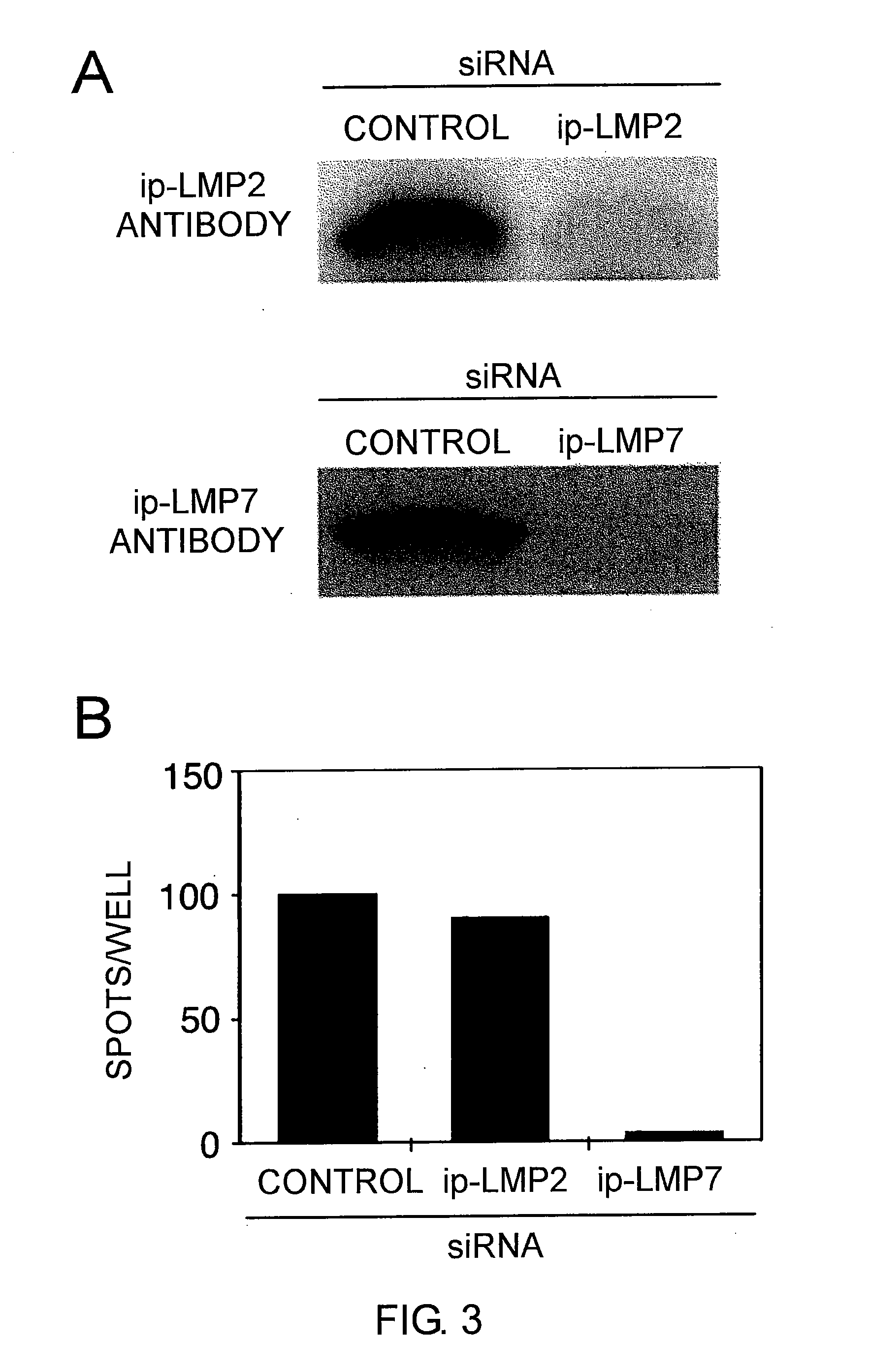
Cytotoxic t-cell epitope peptides that specifically attack epstein-barr virus-infected cells and uses thereof
InactiveUS20090305324A1Peptide/protein ingredientsGenetic material ingredientsEpitopeNatural Killer Cell Inhibitory Receptors
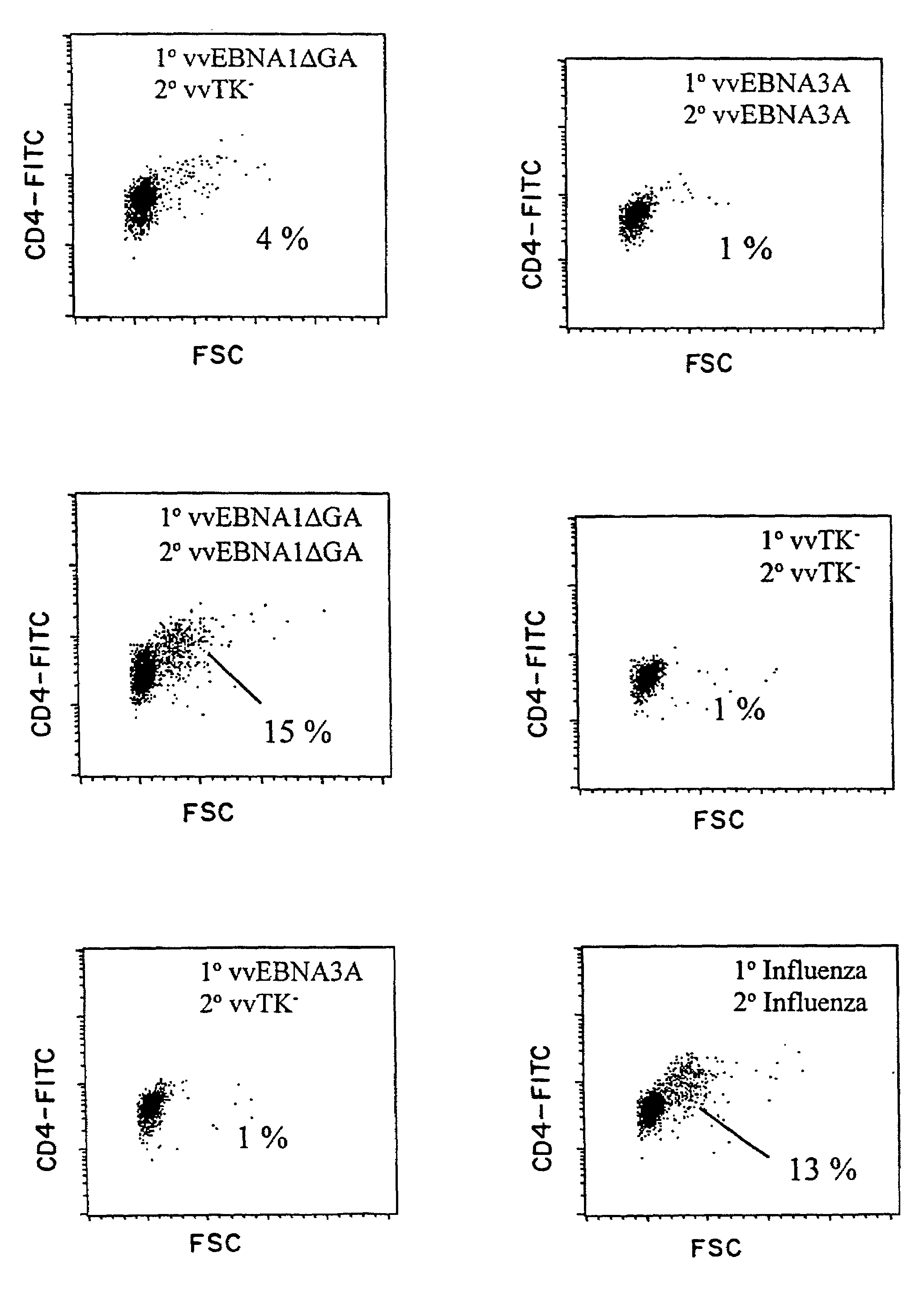
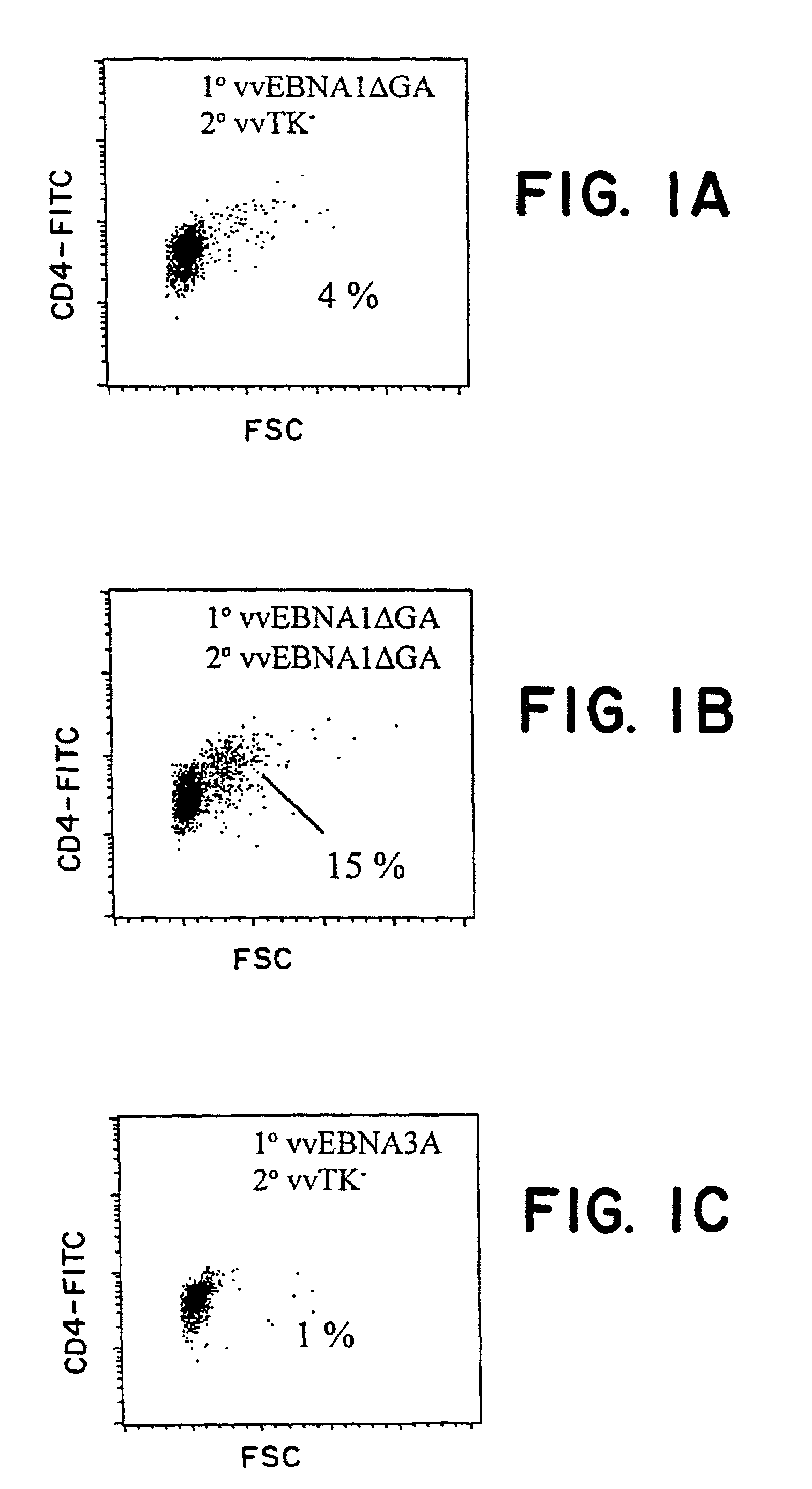
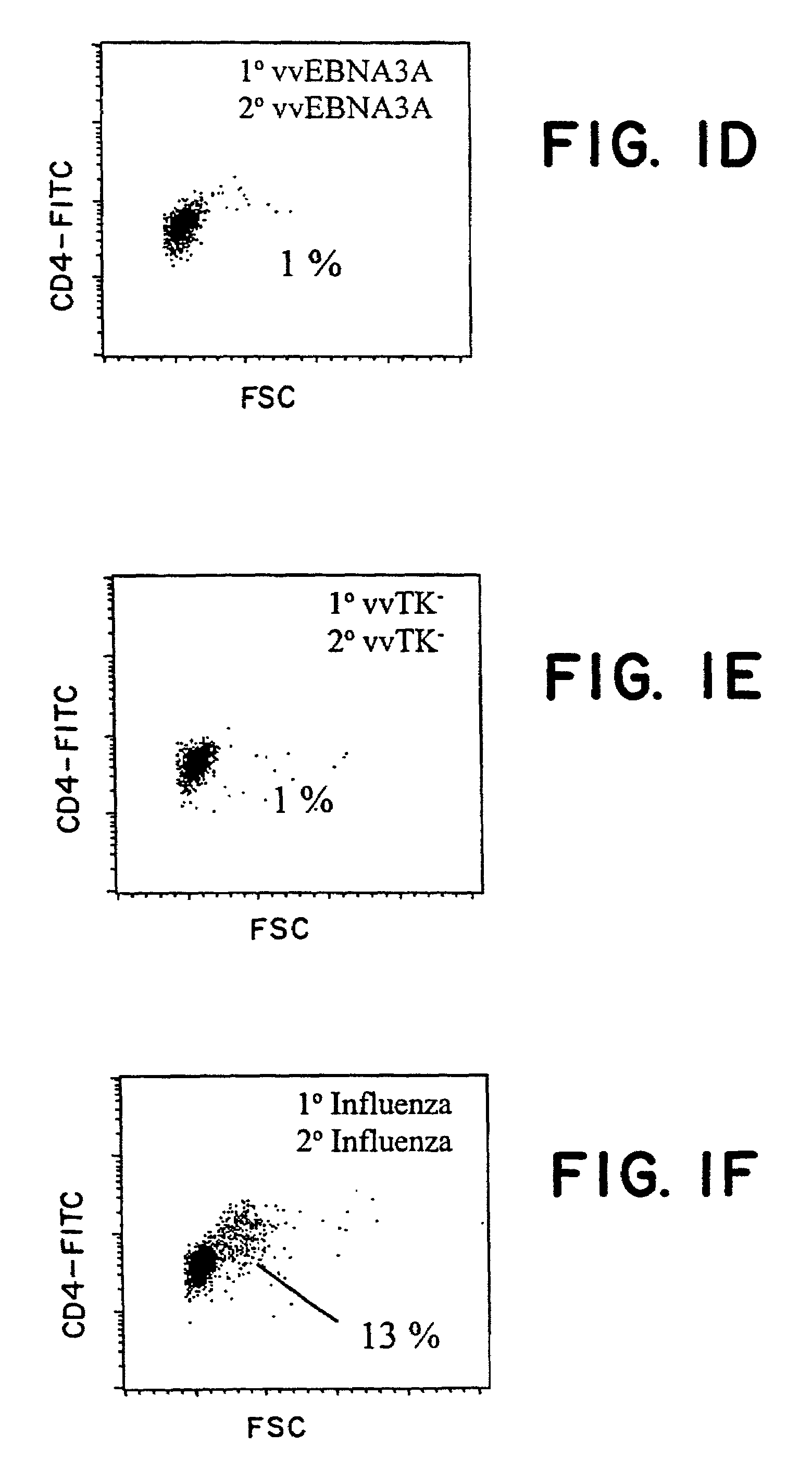
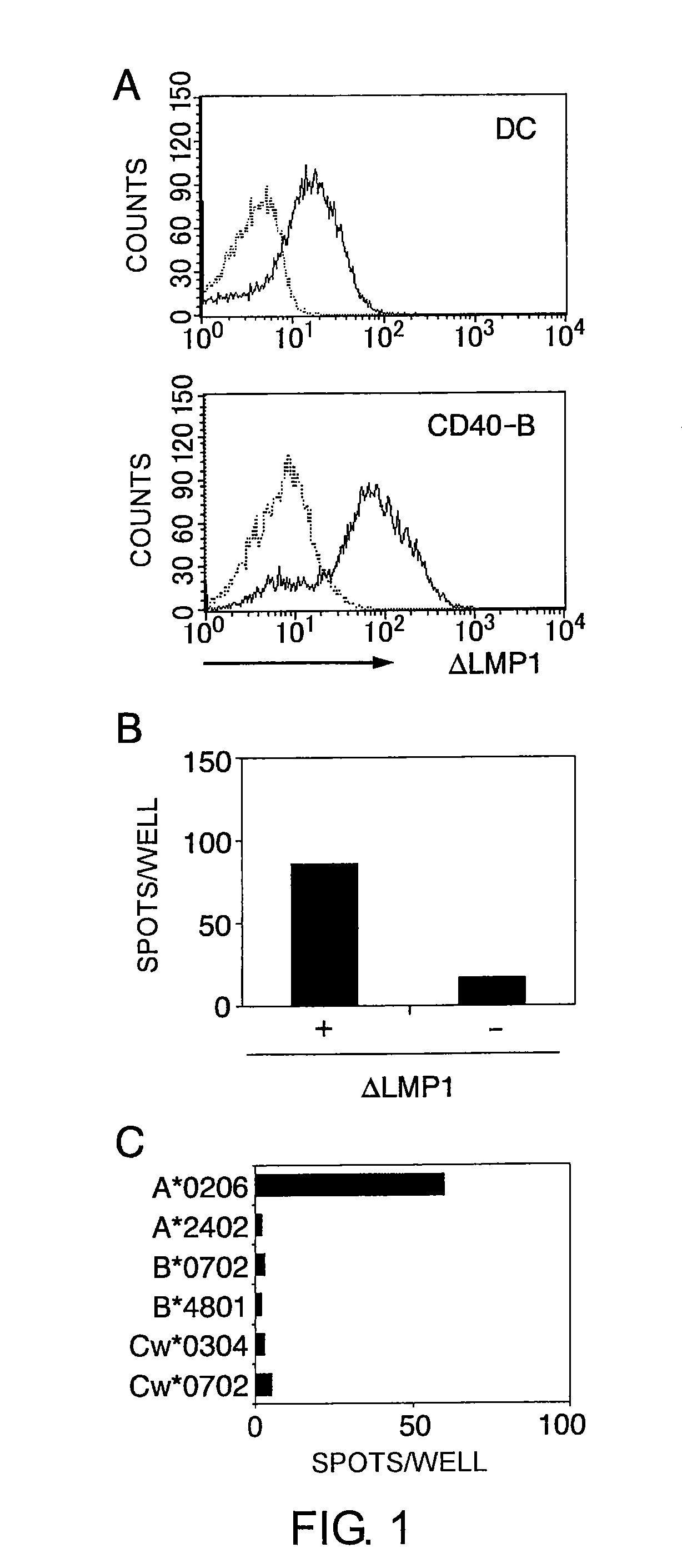
The present inventors introduced mRNAs for the Epstein-Barr virus proteins LMP1 and EBNA1 into antigen-presenting cells, and as a result, demonstrated that the cells induced Epstein-Barr virus-specific cytotoxic T cells. The present inventors also demonstrated that the cytotoxic T cells recognized epitope peptides presented via HLA-A*0206, HLA-Cw*0303, or HLA-Cw*0304, inhibited the outgrowth of Epstein-Barr virus-infected B cells, and lysed Epstein-Barr virus-infected NK lymphomas and NK cells.
Owner:AICHI PREFECTURE +1
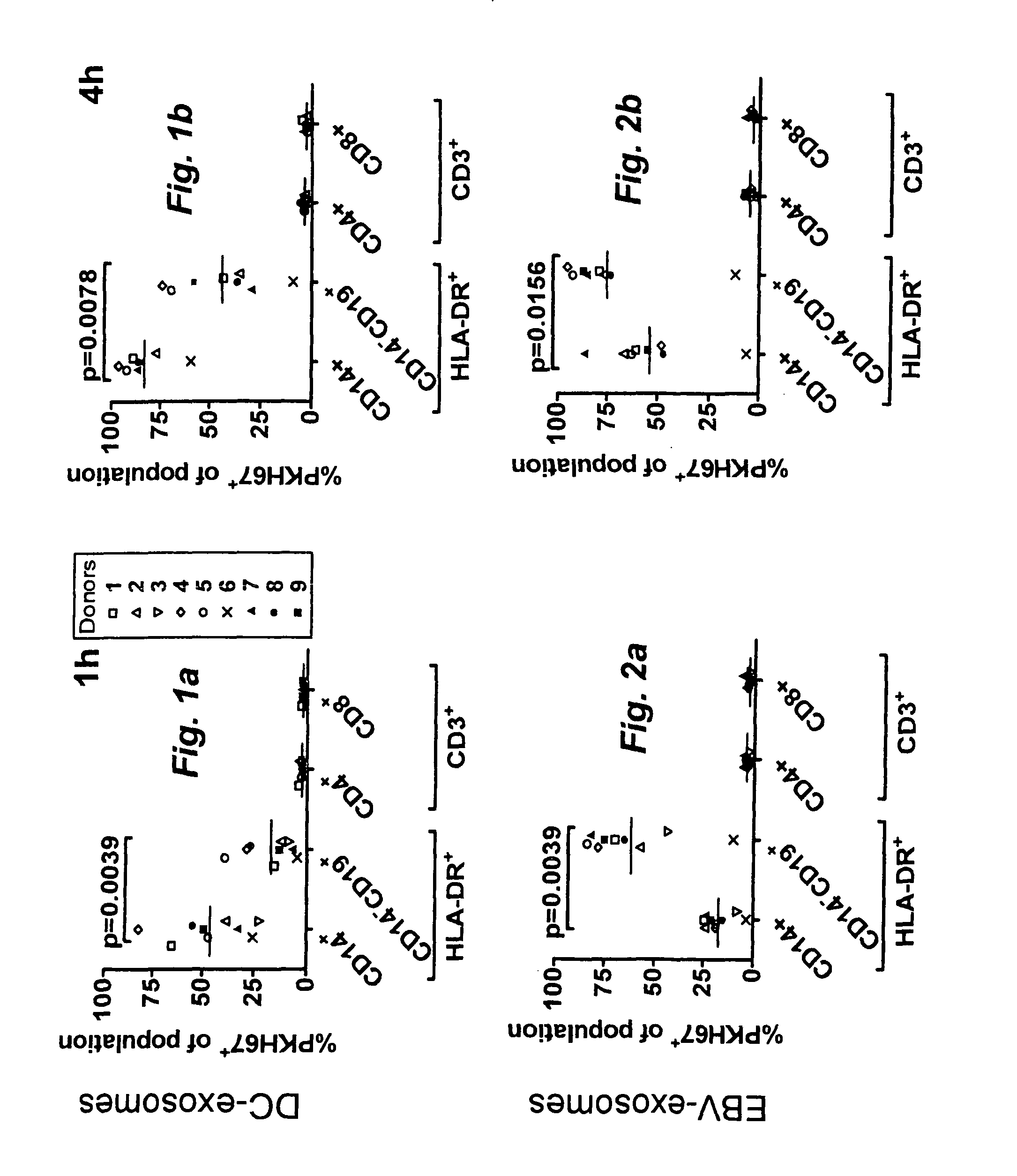
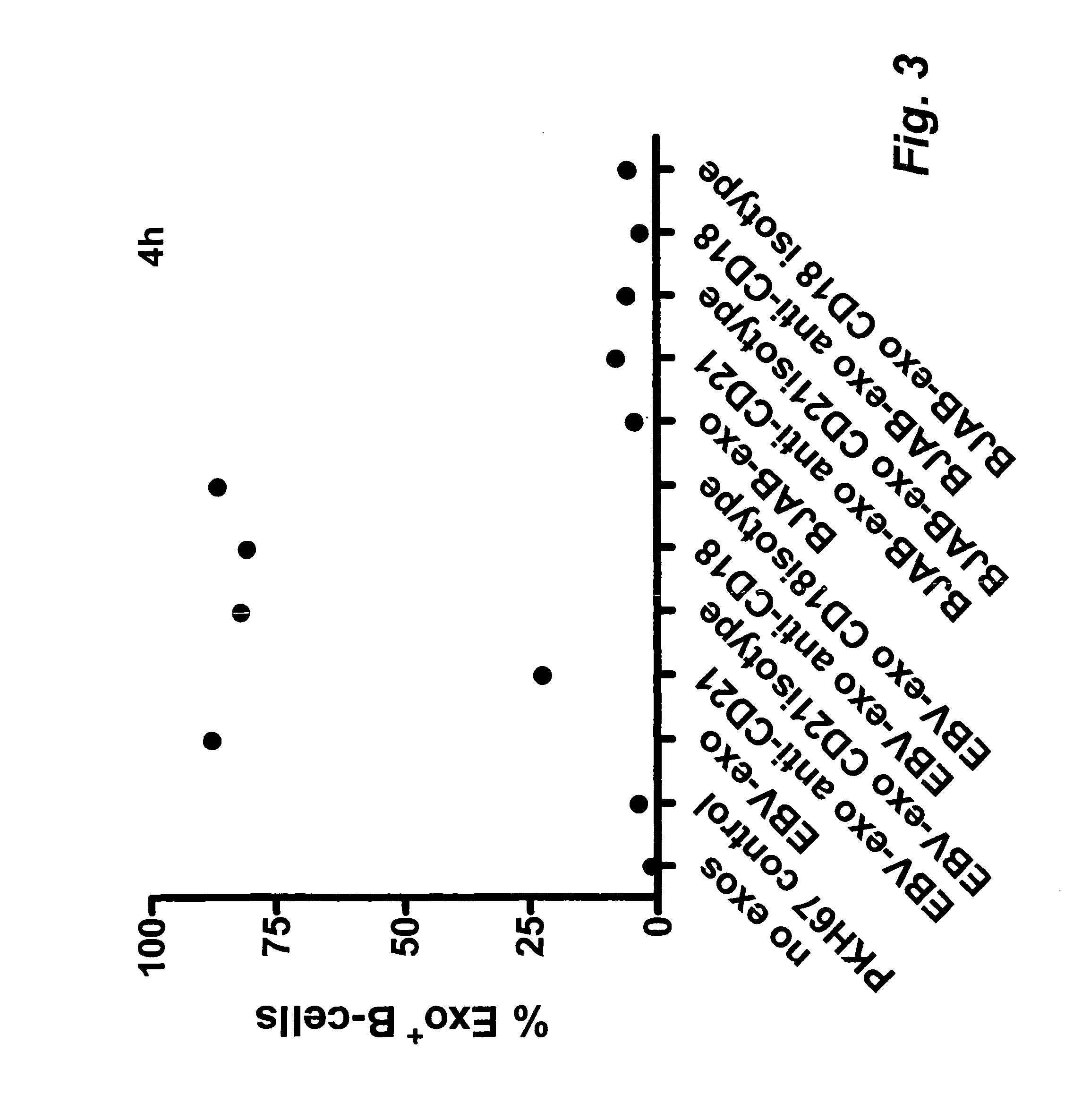
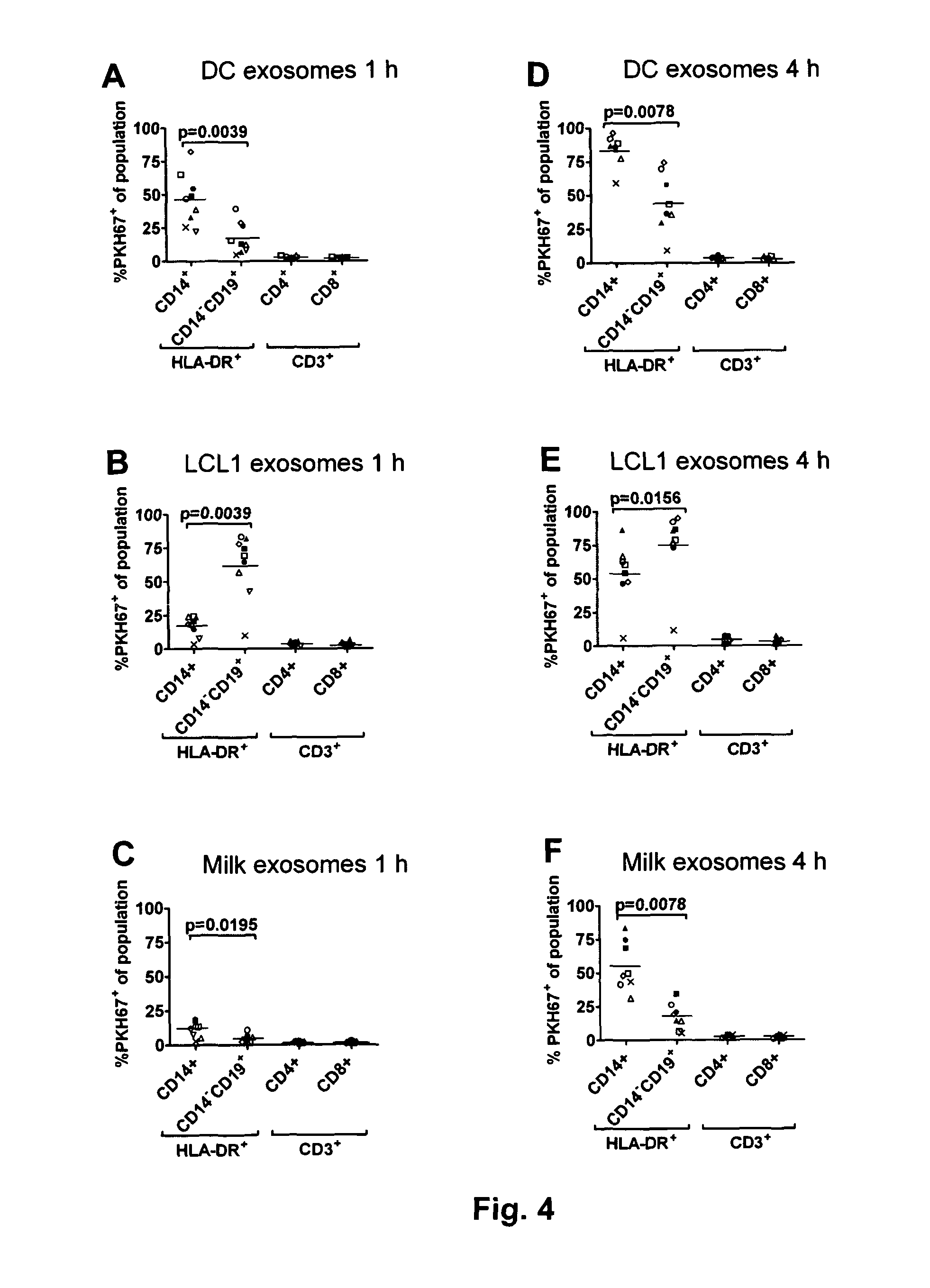
Exosome based treatment of cancer
ActiveUS20120183575A1Efficient and specific immune responseStrong T-cell responseAntibacterial agentsBacterial antigen ingredientsAntigenCancer antigen
A method of treating cancer in a patient comprises immortalizing B cells collected from the patient by infection with Epstein Barr virus, transforming the cells to a latent stage, culturing the cells in the presence of a cancer antigen, harvesting exosomes released from the cells, administering the exosomes to the patient. Alternatively the harvested exosomes are loaded with cancer antigen.
Owner:ITH IMMUNE THERAPY HLDG
Treatment of EBV and KHSV infection and associated abnormal cellular proliferation
InactiveUS7638502B2Increases activity and bioavailability and stabilityEffective destructionBiocidePeptide/protein ingredientsMedicineDiluent
A method and composition for the treatment, prevention and / or prophylaxis of a host, and in particular, a human, infected with Epstein-Barr virus (EBV), is provided that includes administering an effective amount of a 5-substituted uracil nucleoside or its pharmaceutically acceptable salt or prodrug, optionally in a pharmaceutically acceptable diluent or excipient.
Owner:GILEAD PHARMASSET LLC
Vectors, cell lines and their use in obtaining extended episomal maintenance replication of hybrid plasmids and expression of gene products
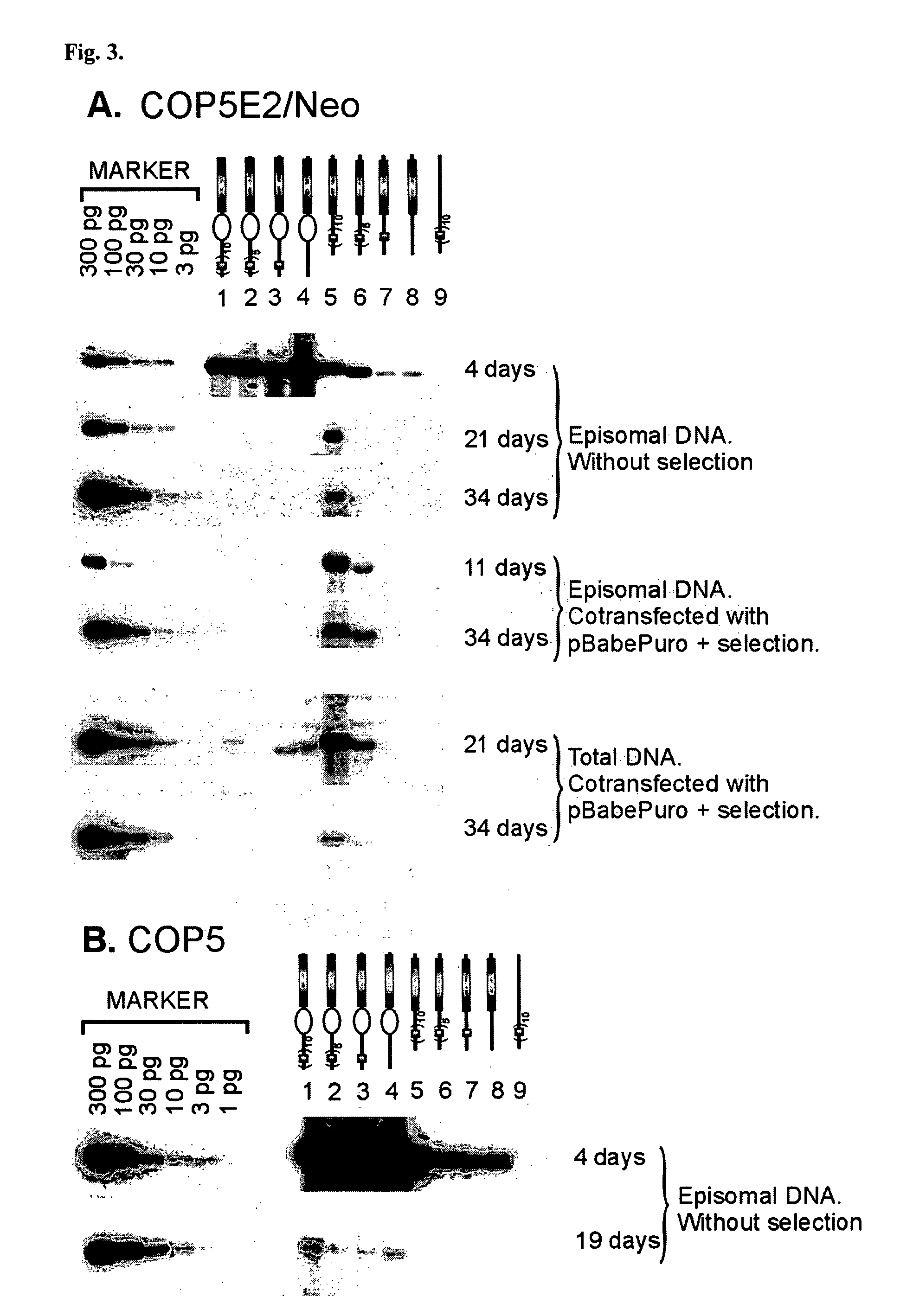
This disclosure shows that the BPV-1 E2 protein-dependent minichromosome maintenance element (MME) comprised of E2 multimeric binding sites can provide the stable maintenance replication function to the mouse polyomavirus (PyV) core origin plasmids in the presence of BPV-1 E2 protein and PyV large T-antigen (LT). MME dependent plasmids are lost with the frequency of 6% per generation. Significantly long stable maintenance replication can also be provided without selection pressure. We also demonstrate that PyV core origin maintenance function / replication activation could be provided by Epstein-Barr virus Family of repeats and EBNA1 protein. The maintenance of the Polyomavirus core origin plasmid was characterized by 13% loss of the plasmid during one cell generation in the case of EBV FR harboring plasmids. Our data clearly indicate that maintenance functions from different viruses can provide segregation / partitioning function to different heterologous origins in variety of cells and can be used in expression of gene products.
Owner:RAKUVABRIK LTD
Composition For Promoting Apoptosis Or Inhibiting Cell Growth, Comprising Epstein-Barr Virus Microrna
ActiveUS20140296319A1Promote apoptosisInhibit cell growthOrganic active ingredientsPeptide/protein ingredientsApoptosisMicroRNA
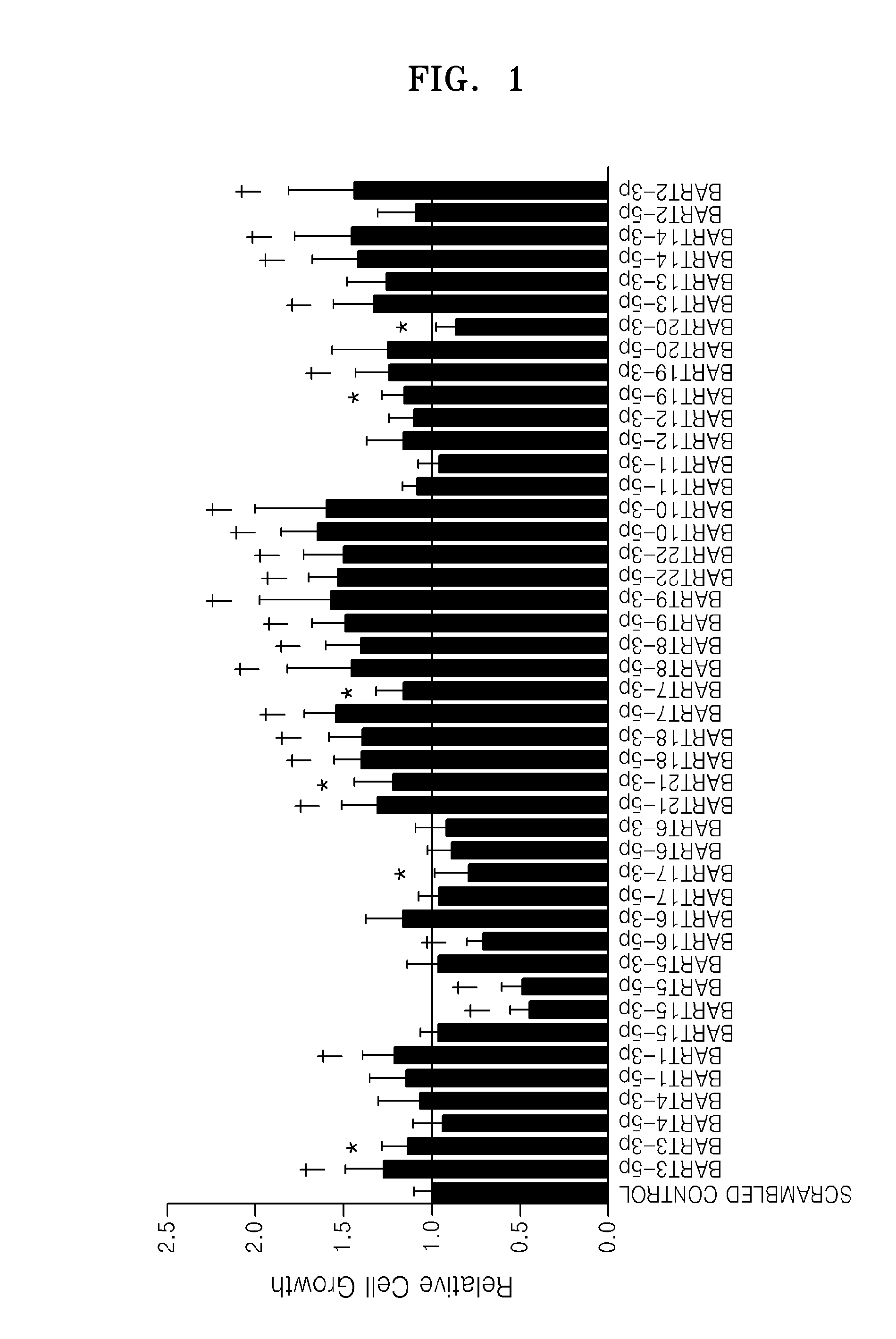
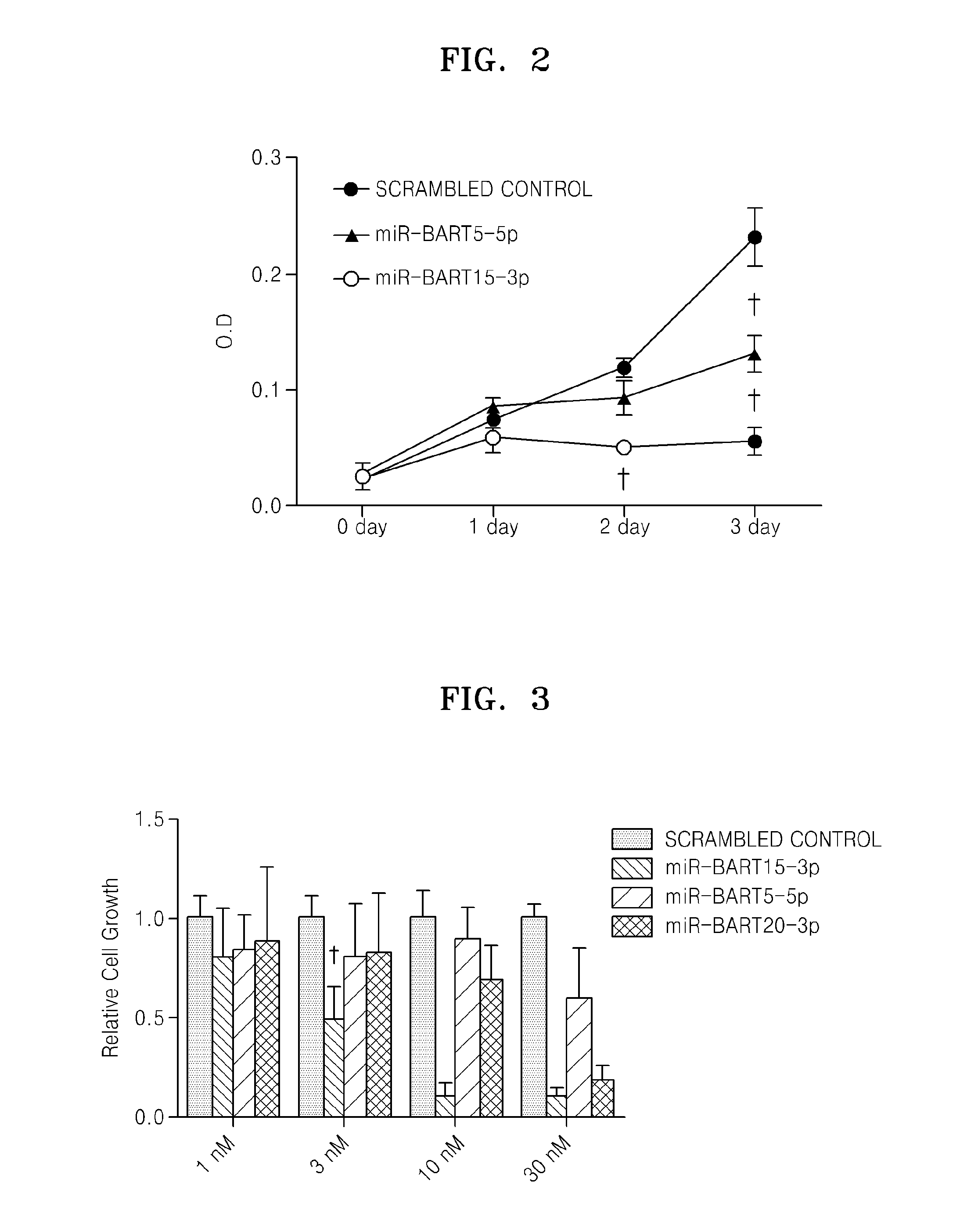
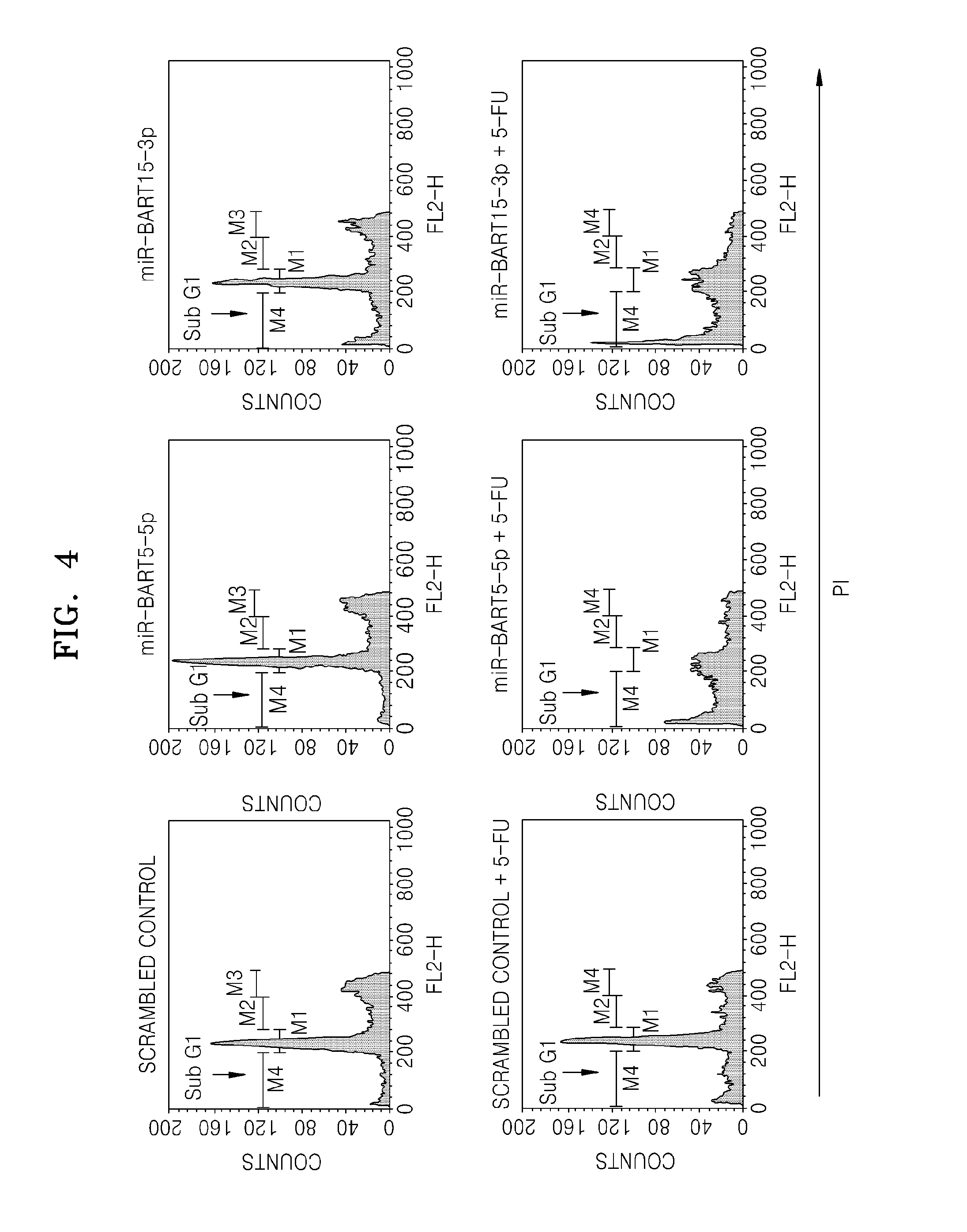
Provided is a use of an miRNA of an Epstein-Barr virus (EBV), more specifically, EBV miR-BART4-5p, miR-BART4-3p, miR-BART1-5p, miR-BART15-3p, miR-BART5-5p, miR-BART5-3p, miR-BART16-5p, miR-BART16-3p, miR-BART17-3p, miR-BART21-3p, miR-BART18-5p, miR-BART7-5p, miR-BART9-5p, miR-BART22-5p, miR-BART20-3p, miR-BART13-5p, miR-BART13-3p, miR-BART2-3p, and mimics thereof for promoting apoptosis or inhibiting cell growth.
Owner:THE CATHOLIC UNIV OF KOREA IND ACADEMIC COOPERATION FOUND
Methods to prevent vertical transmission of infectious diseases
InactiveUS20080269205A1Prevent vertical transmissionPrevent the spread of diseaseAntiviralsRadiation therapyEscherichia coliMonilinia laxa
The present invention presents a method of preventing vertical transmission of an infectious disease comprising: (a) applying a photosensitizing composition to host tissues of birth canal of a mother during the intrapartum period; and (b) applying light to the host tissues after the step (a) at a wavelength absorbed by the photosensitizing composition so as to inhibit or eliminate infectious disease microorganisms that come into contact with the host tissues. The infectious disease may be caused by human immunodeficiency virus type 1, hepatitis B virus, hepatitis C virus, Group B Streptococcus, cytomegalovirus, Listeria monocytogenes, Chiamydia trachomatis, Escherichia coli, herpes simplex virus, Epstein-Barr virus, Toxoplasma gondii, human papilloma virus, and Candida.
Owner:ONDINE INT
Protective antigen of Epstein Barr Virus
The present invention relates to the identification of a subunit vaccine to prevent or treat infection of Epstein Barr Virus. In particular, EBNA-1 was identified as a vaccine antigen. In a specific embodiment, a purified protein corresponding to EBNA-1 elicited a strong CD4+ T cell response. The responsive CD4+ T cell are primarily TH1 in function. EBNA-1 is an attractive candidate for a protective vaccine against EBV, and for immunotherapy of EBV infection and neoplasms, particularly with dendritic cells charged with EBNA-1.
Owner:THE ROCKEFELLER UNIV
Cytotoxic T-cell epitope peptides that specifically attack epstein-barr virus-infected cells and uses thereof
The present inventors introduced mRNAs for the Epstein-Barr virus proteins LMP1 and EBNA1 into antigen-presenting cells, and as a result, demonstrated that the cells induced Epstein-Barr virus-specific cytotoxic T cells. The present inventors also demonstrated that the cytotoxic T cells recognized epitope peptides presented via HLA-A*0206, HLA-Cw*0303, or HLA-Cw*0304, inhibited the outgrowth of Epstein-Barr virus-infected B cells, and lysed Epstein-Barr virus-infected NK lymphomas and NK cells.
Owner:AICHI PREFECTURE +1
Immunoreactive peptides from epstein-barr virus
Owner:ORTHO-CLINICAL DIAGNOSTICS
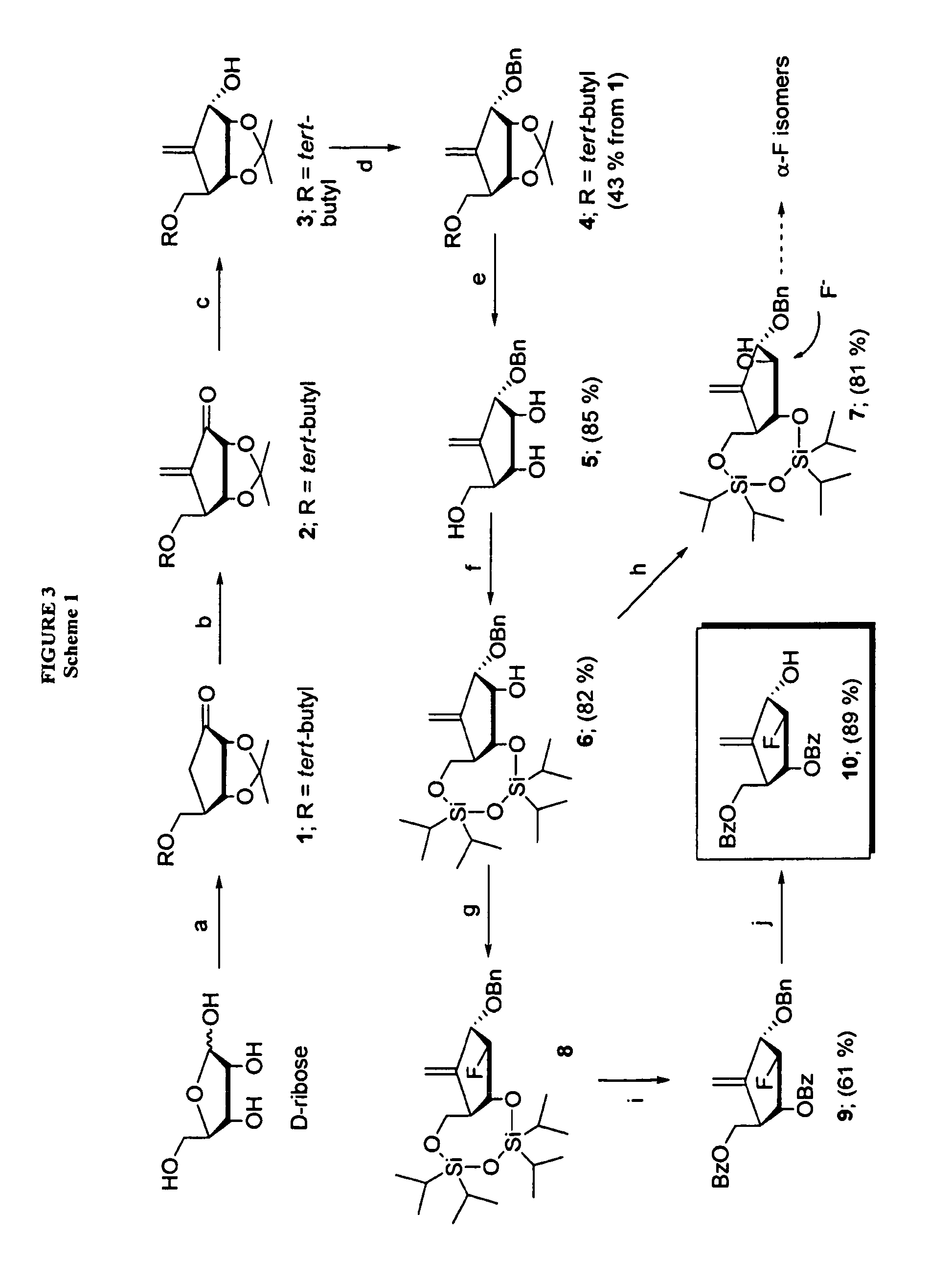
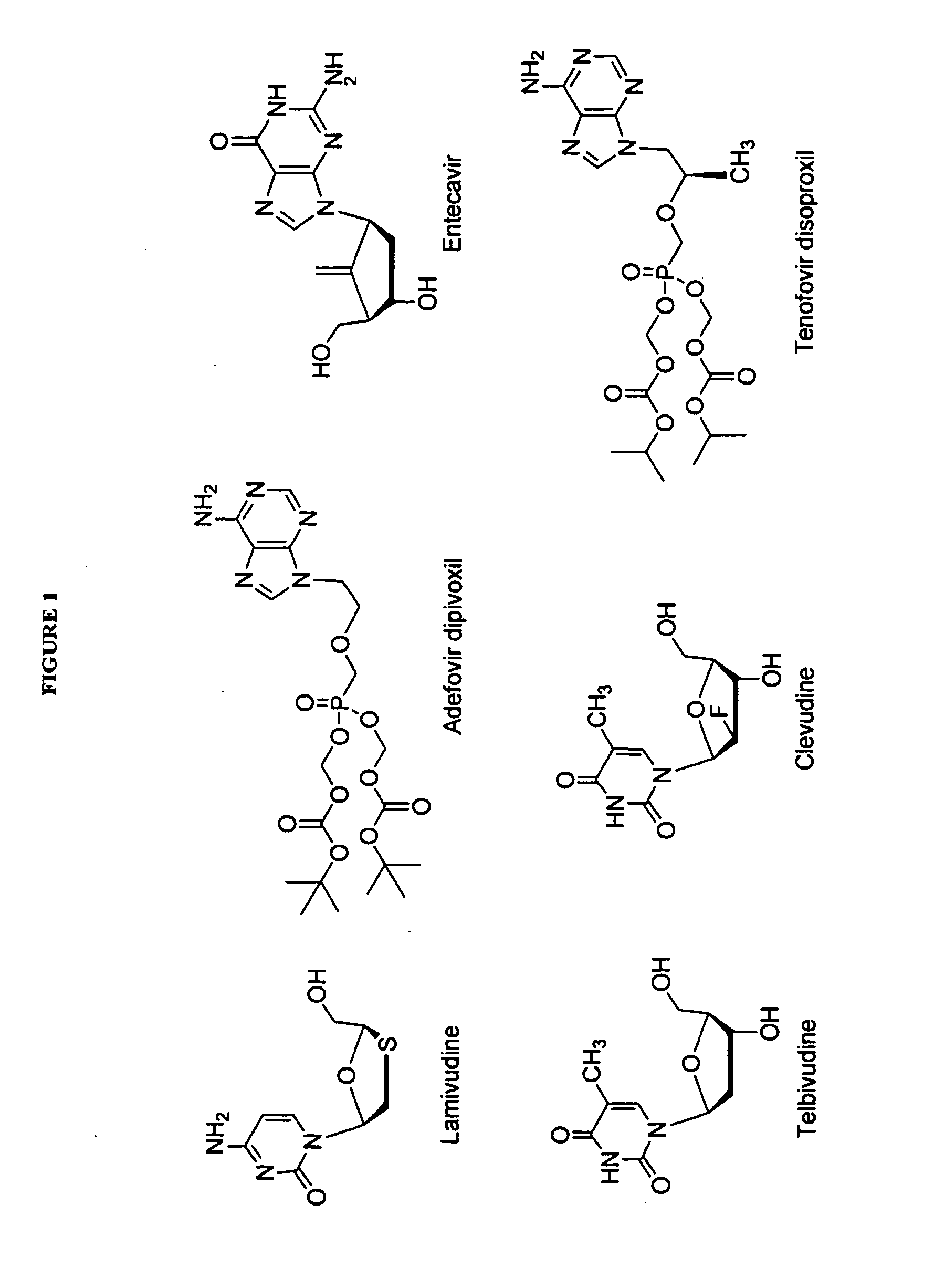
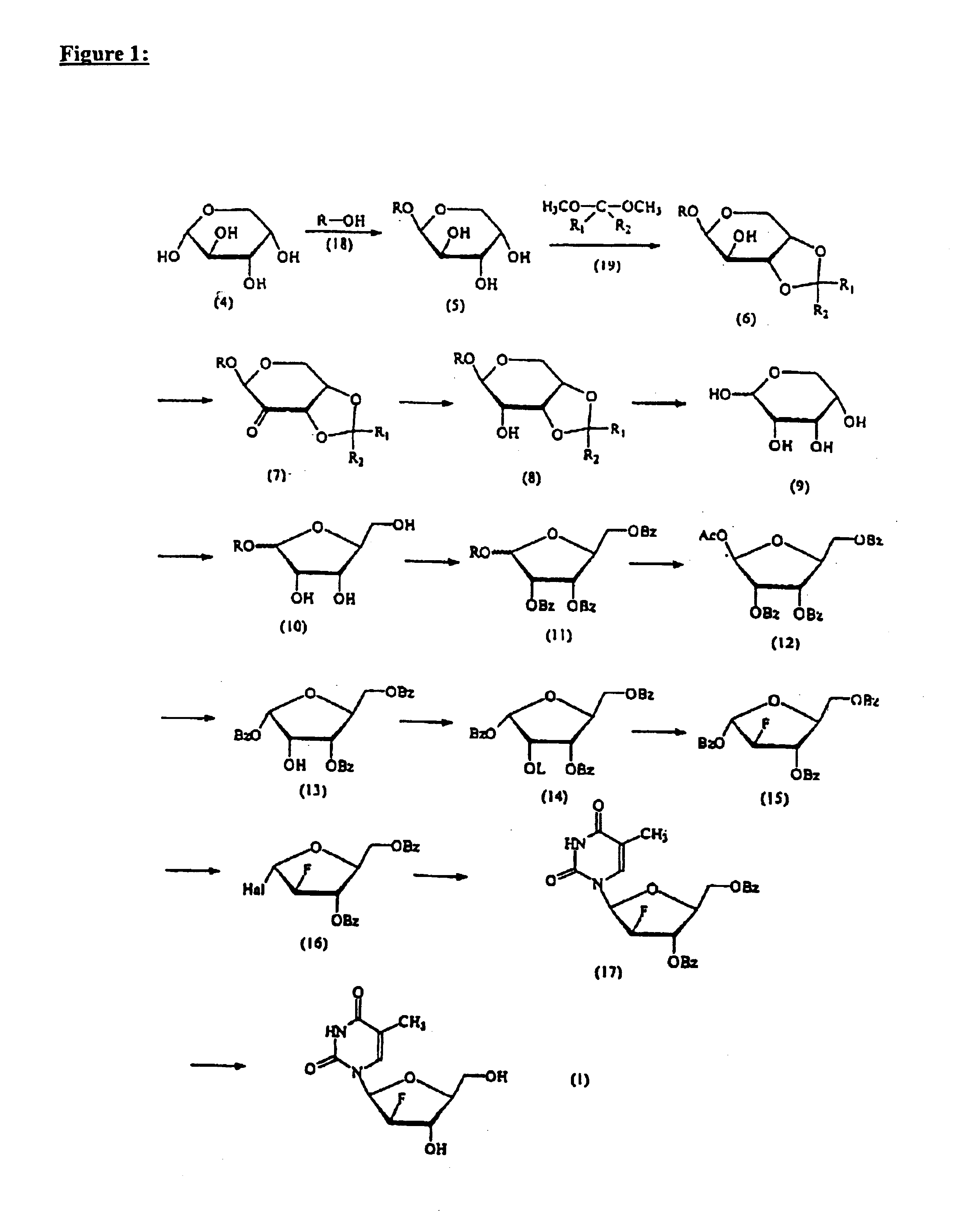
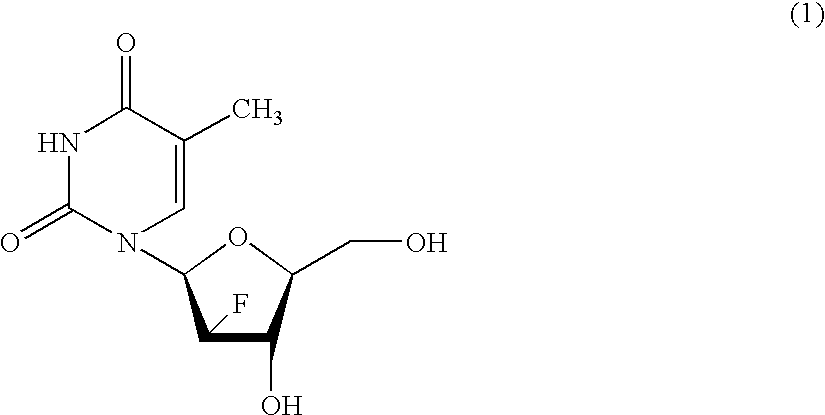
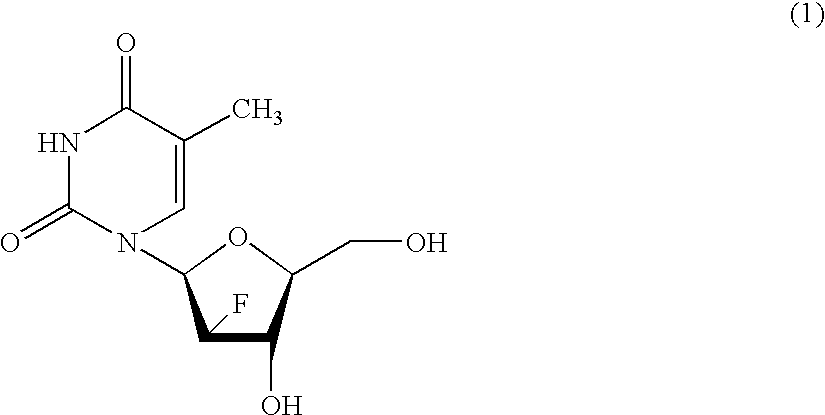
Process for the preparation of 2'-fluoro-5-methyl-beta-L-arabino-furanosyluridine
InactiveUS6894159B2Speed up the processSugar derivativesMonosaccharidesHepatitis B virusMethyl group
The present invention relates to a novel and improved process for preparing 2′-fluoro-5-methyl-β-L-arabinofuranosyluridine represented by formula (1) which shows anti-viral activity, especially potent anti-viral activity against hepatitis B-virus and Epstein-Barr virus:
Owner:UNIV OF GEORGIA RES FOUND INC
Antiviral nucleosides
InactiveUS7589077B2Superior anti-HBV activityHigh anti-EBV propertyBiocideSugar derivativesHerpes simplex virus DNAHepatitis B virus
Disclosed are nucleosides which are useful in diagnosing and treating viral infections, for example, infections caused by hepatitis B virus (HBV), and herpes viruses including Epstein Barr virus.
Owner:KUMAR RAKESH
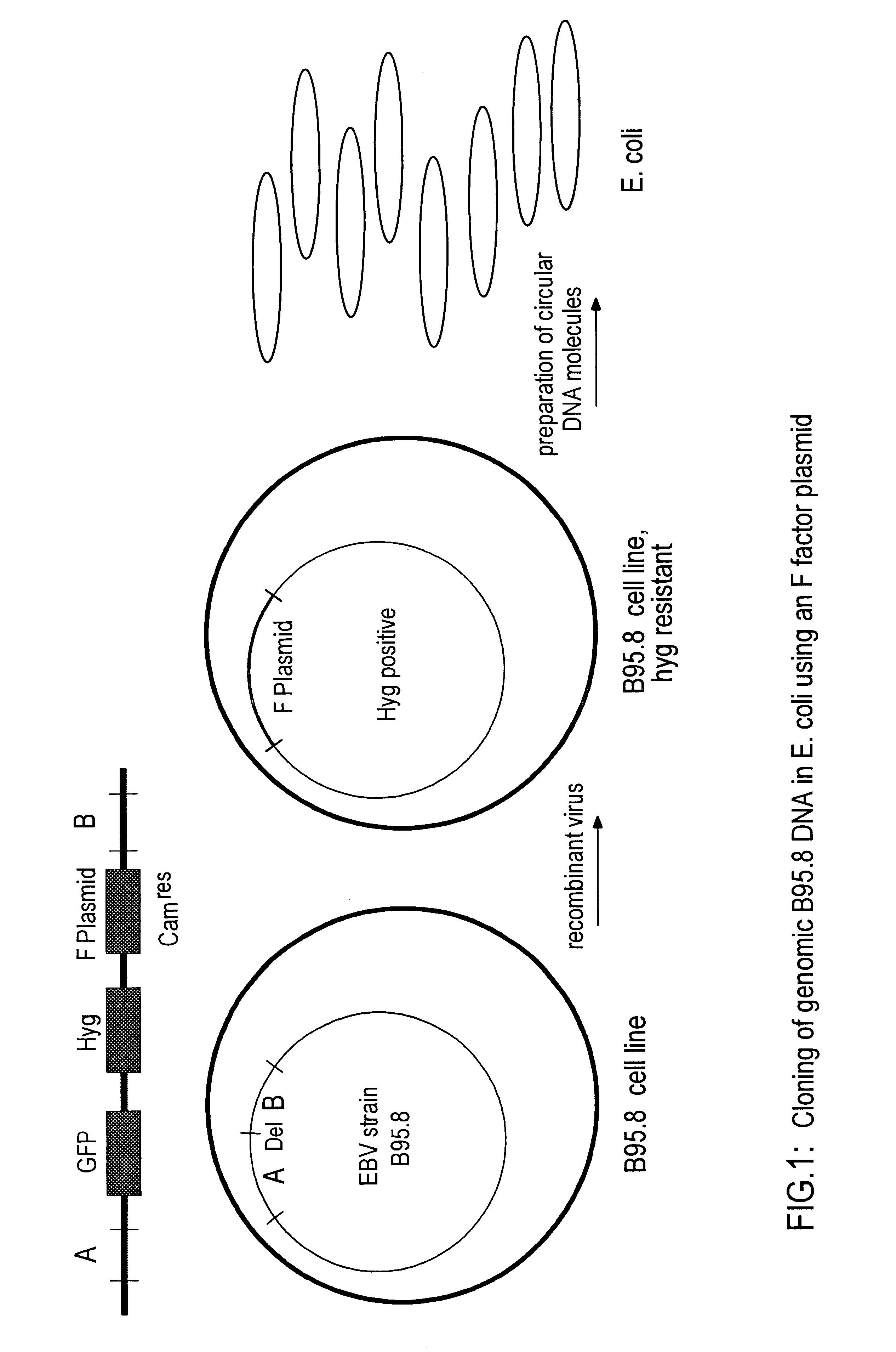
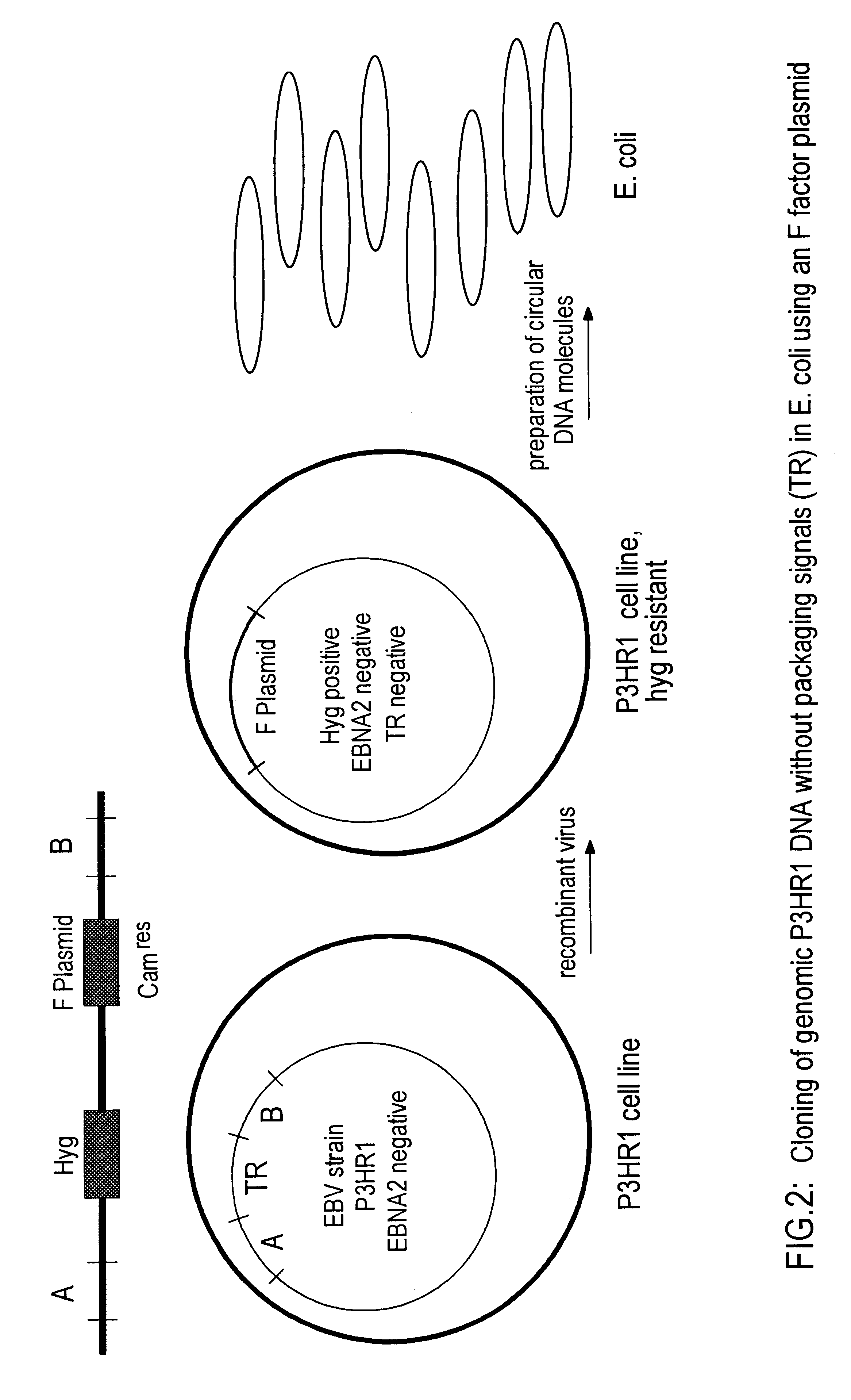
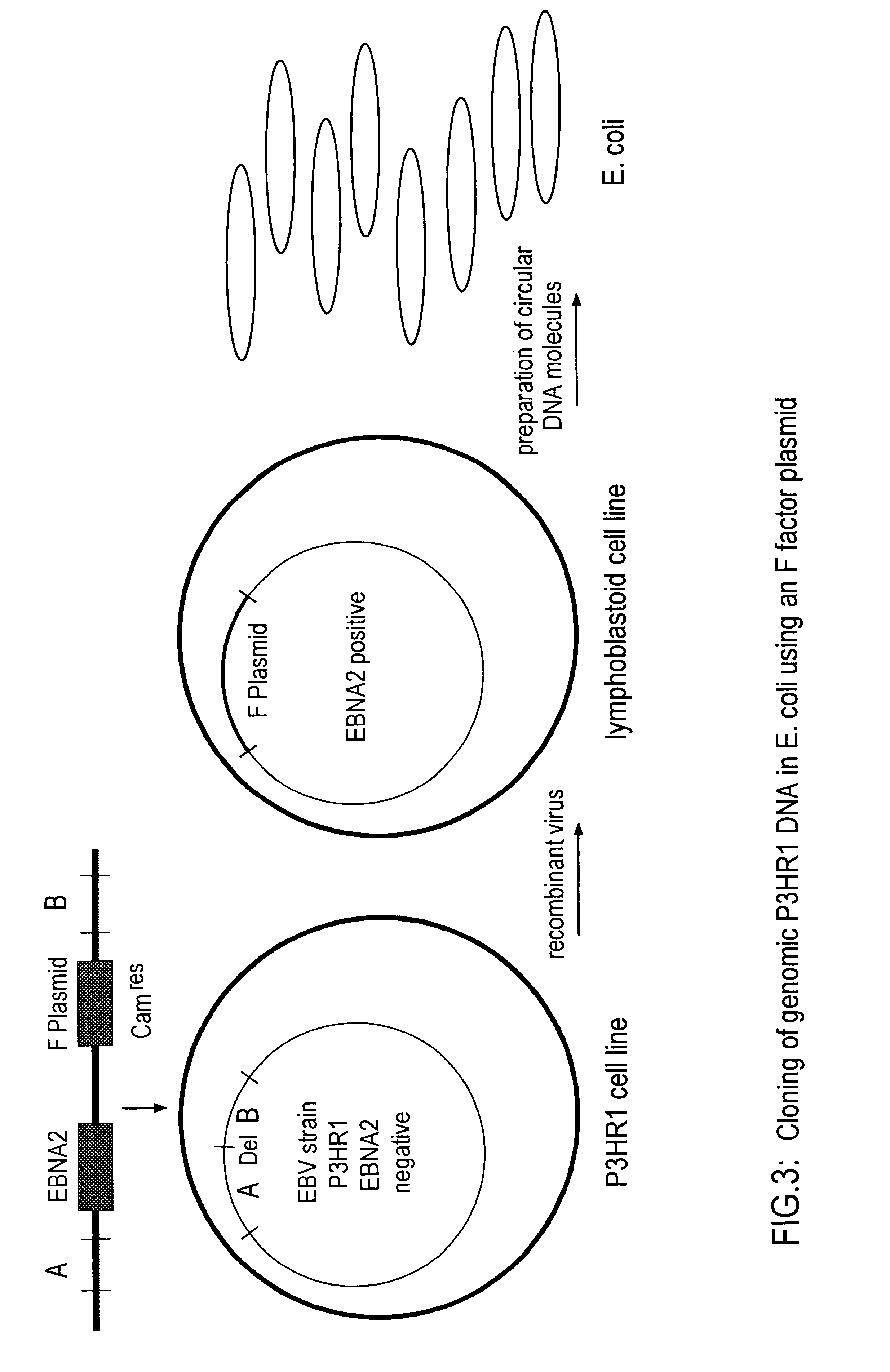
DNA virus vectors and methods for their preparation
InactiveUS6291246B1Improve efficiencyDifficult to controlBacteriaGenetic material ingredientsDna viralVirology
The invention relates to a method for the preparation of DNA virus vectors capable of replication in eukaryotic as well as in prokaryotic cells as well as to DNA virus vectors prepared by this method. Preferably, the method is used to prepare Epstein-Barr virus vectors.
Owner:GSF FORSCHUNGSZENT FUR UMWELT & GESUNDHEIT
Herpes virus EBV (Epstein-Barr Virus) detection kit
ActiveCN103060473AEasy to operateSimple methodMicrobiological testing/measurementFluorescence/phosphorescenceEBV InfectionsPotassium
The invention provides a herpes virus EBV (Epstein-Barr Virus) detection kit. The kit comprises a nucleic acid releasing agent and PCR (polymerase chain reaction) reaction solution, wherein the nucleic acid releasing agent comprises 0.01-0.5 mM / L of surfactin, 20-300 mM / L of potassium chloride, 0.01-2% of sodium dodecyl sulphate and 0.05-1% of ethanol; and the PCR reaction solution comprises an upstream primer and a downstream primer used for target polynucleotide amplification, and a probe used for target polynucleotide detection. The detection result of the method for releasing nucleic acid by the nucleic acid releasing agent in the kit disclosed by the invention has no obvious difference with the detection result of a boiling method, a strong protein denaturing agent is used during nucleic acid extraction in the kit disclosed by the invention for rapidly breaking the coat protein structure of a pathogen and releasing the nucleic acid of the pathogen, and release and extraction for DNA (deoxyribonucleic acid) can be rapidly finished without heating; the sensitivity of the EBV detection of the kit disclosed by the invention can achieve 400 copies / ml, and the quantitative linear range is 400-4.00E+09 copies / ml; by applying the kit, rapid and accurate detection can be performed on EBV-DNA in the unknown samples of blood plasma, throat swab, peripheral blood and the like, and reliable experimental basis is provided for diagnosing EBV infection.
Owner:SANSURE BIOTECH INC
Treatment of epstein-barr virus-associated diseases
InactiveUS20090202584A1Improve stabilityImprove translationSugar derivativesViral antigen ingredientsVirologyDisease cause
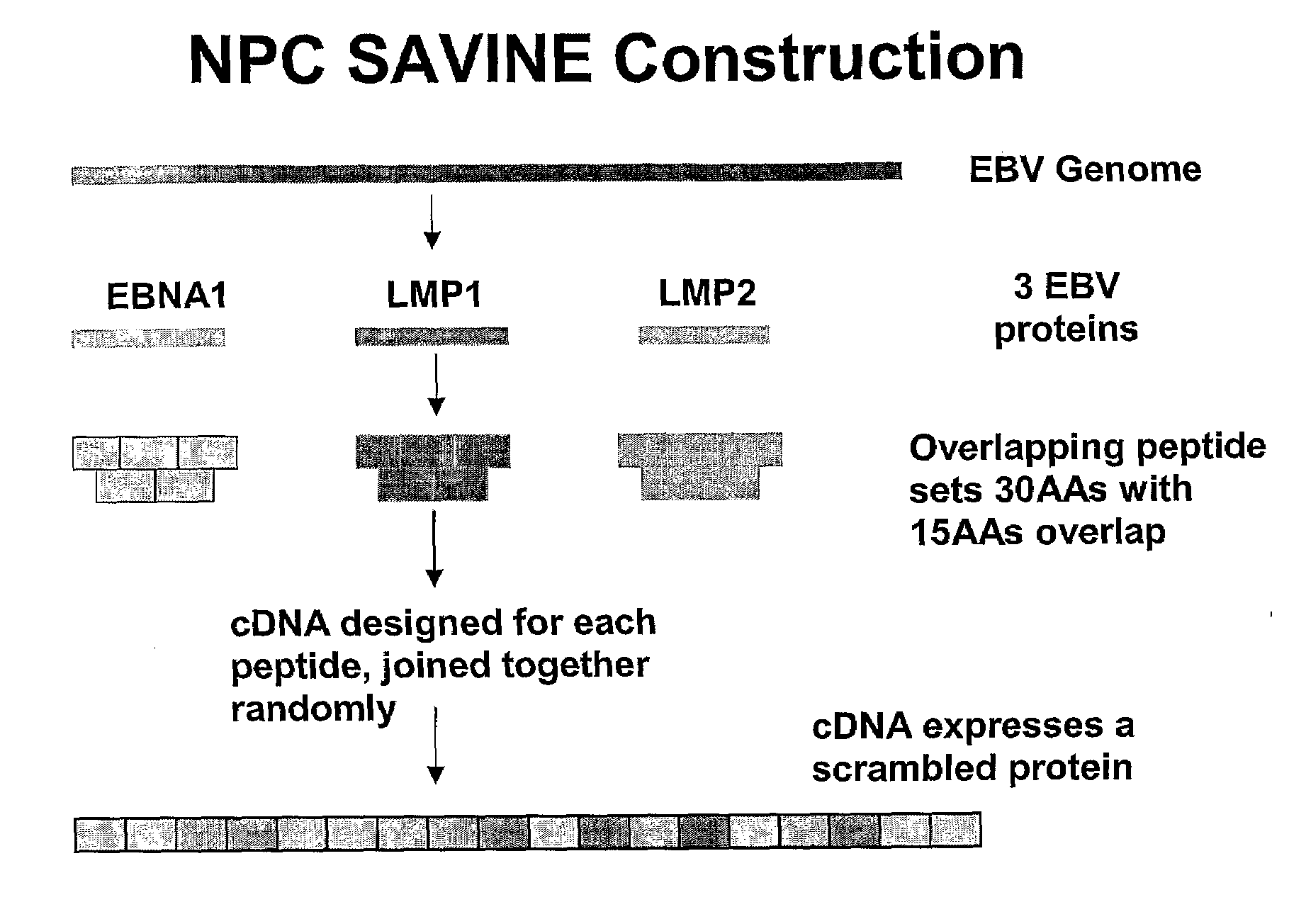
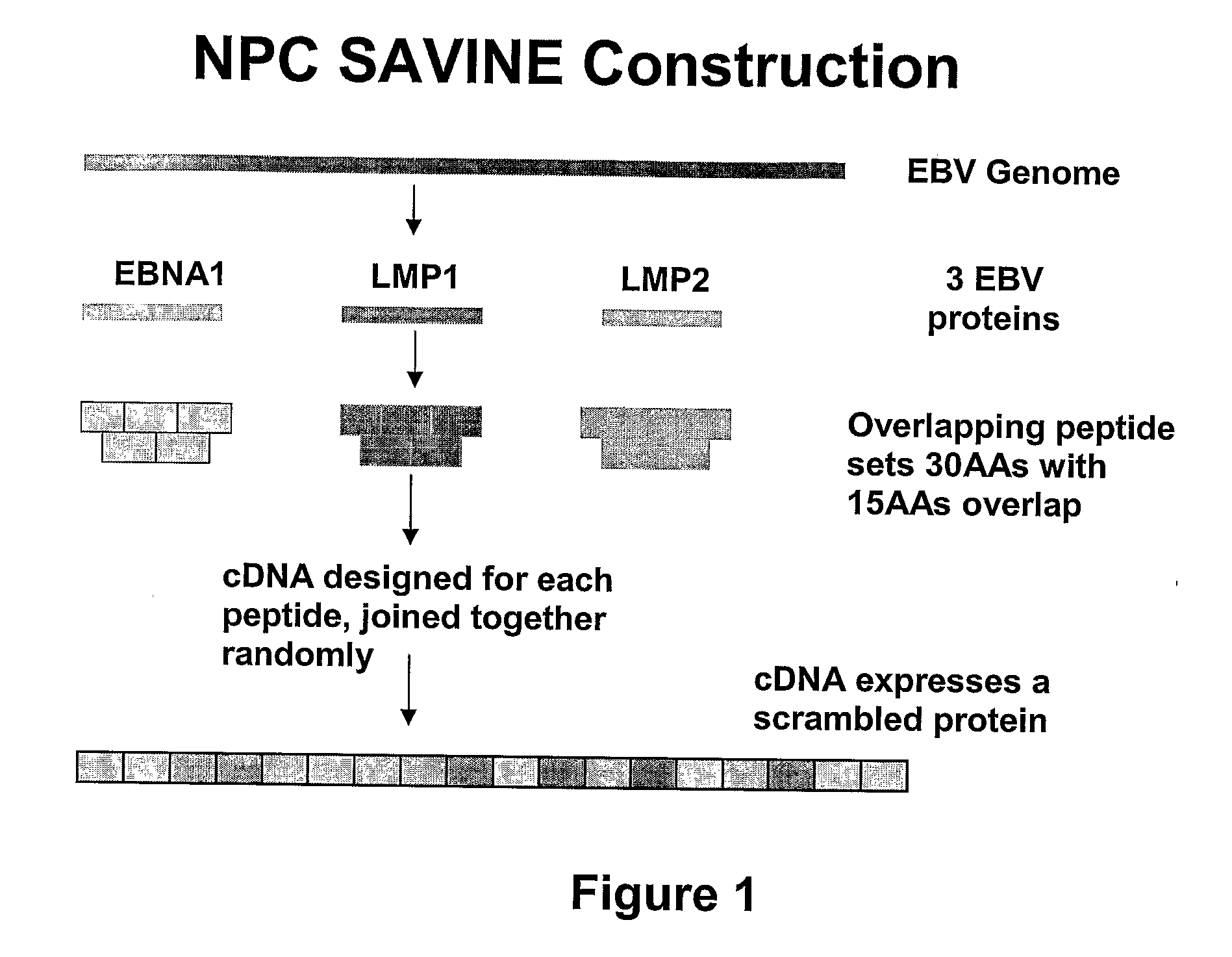
The present invention relates to a vaccine for the treatment or prevention of an EBV-associated disease in a subject, wherein said vaccine comprises a synthetic polypeptide comprising a plurality of different segments of at least one parent EBV polypeptide, and wherein the segments are linked together in a different relationship relative to their linkage in the at least one parent EBV polypeptide.
Owner:SAVINE THERAPEUTICS
Diagnostics and therapy of epstein-barr virus in autoimmune disorders
InactiveUS7192715B2Encourage autoimmunityVirusesPeptide/protein ingredientsAutoimmune conditionAutoimmune responses
Data consistent with autoimmune disease being caused by Epstein-Barr virus are shown. Based on this evidence, an effective vaccine would prevent the autoimmune disease in those vaccinated, modified or administered so that the vaccine is not itself capable of inducing autoimmune disease. In the case of anti-Sm, structures to be avoided in an Epstein-Barr virus-derived vaccine have been identified. Differences have been identified in the immune responses to Epstein-Barr infection between individuals who develop a specific autoimmune disease and those who do not. These differences are used to distinguish those who are at greater risk for developing specific autoimmune diseases from those who are a lesser risk. Assuming Epstein-Barr virus causes autoimmune disease and that Epstein-Barr virus remains latent in the patient for life, reactivation of the virus from the latent state is important in generating or maintaining the autoimmune response that culminates in autoimmune disease. Cells infected with latent virus may also encourage autoimmunity. Based on the understanding that reactivation or latency are important to produce or sustain autoimmunity, then therapies directed against Epstein-Barr virus will also be effective therapies for the autoimmune manifestations of disease for which Epstein-Barr virus is responsible.
Owner:HARLEY JOHN B +1