Patents
Literature
6899 results about "Urine" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Urine is a liquid by-product of metabolism in humans and in many other animals. Urine flows from the kidneys through the ureters to the urinary bladder. Urination results in urine being excreted from the body through the urethra.
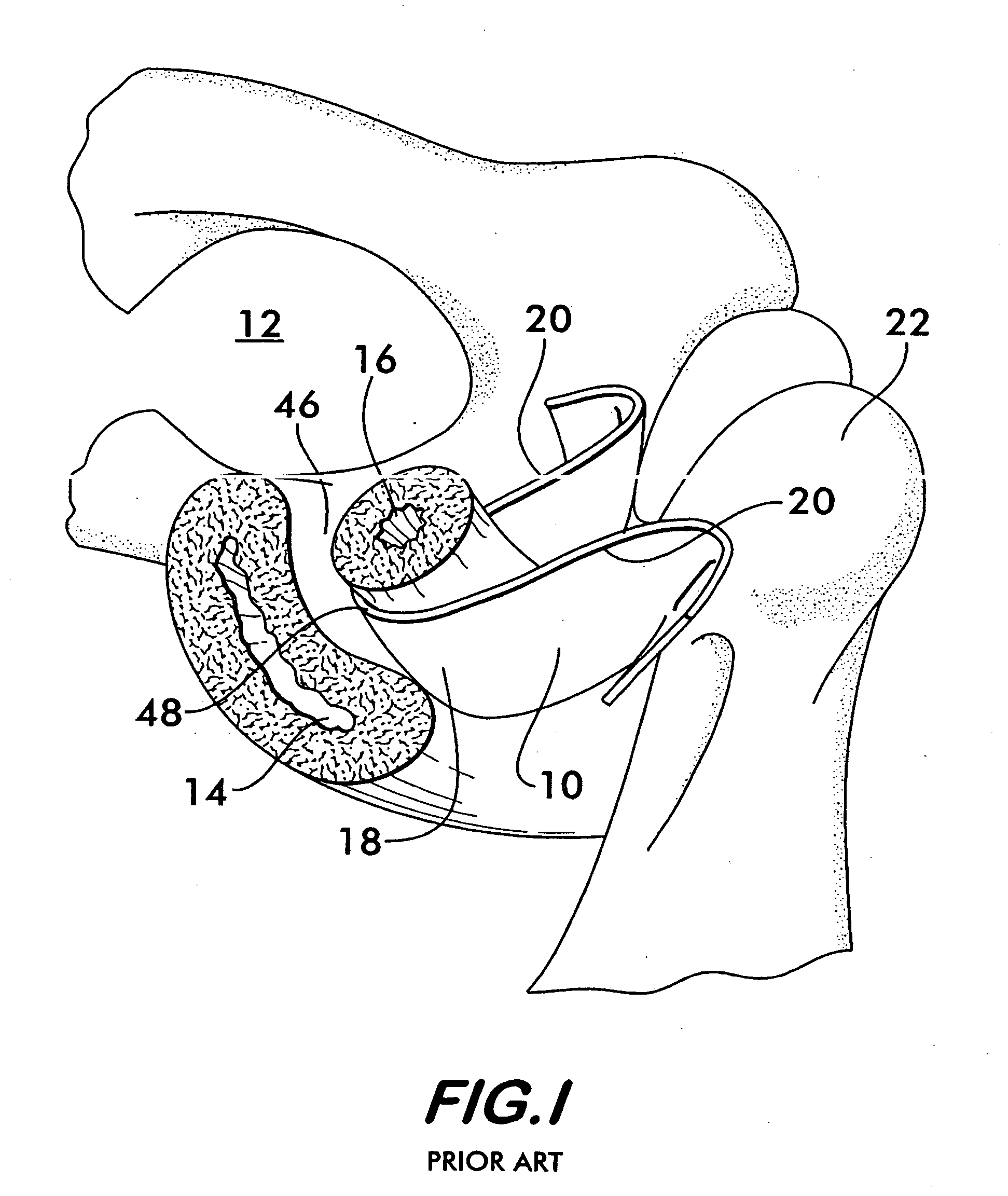
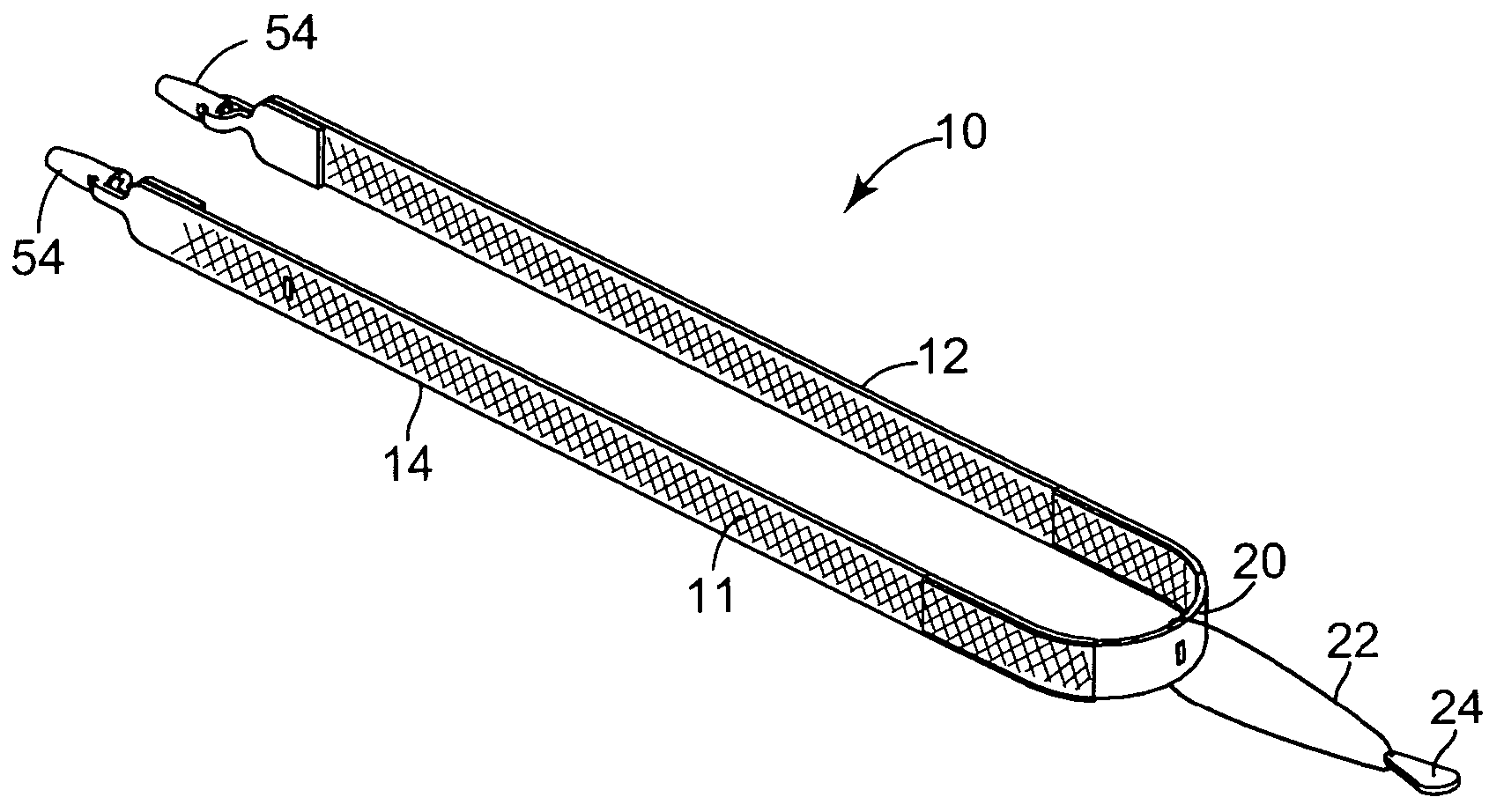
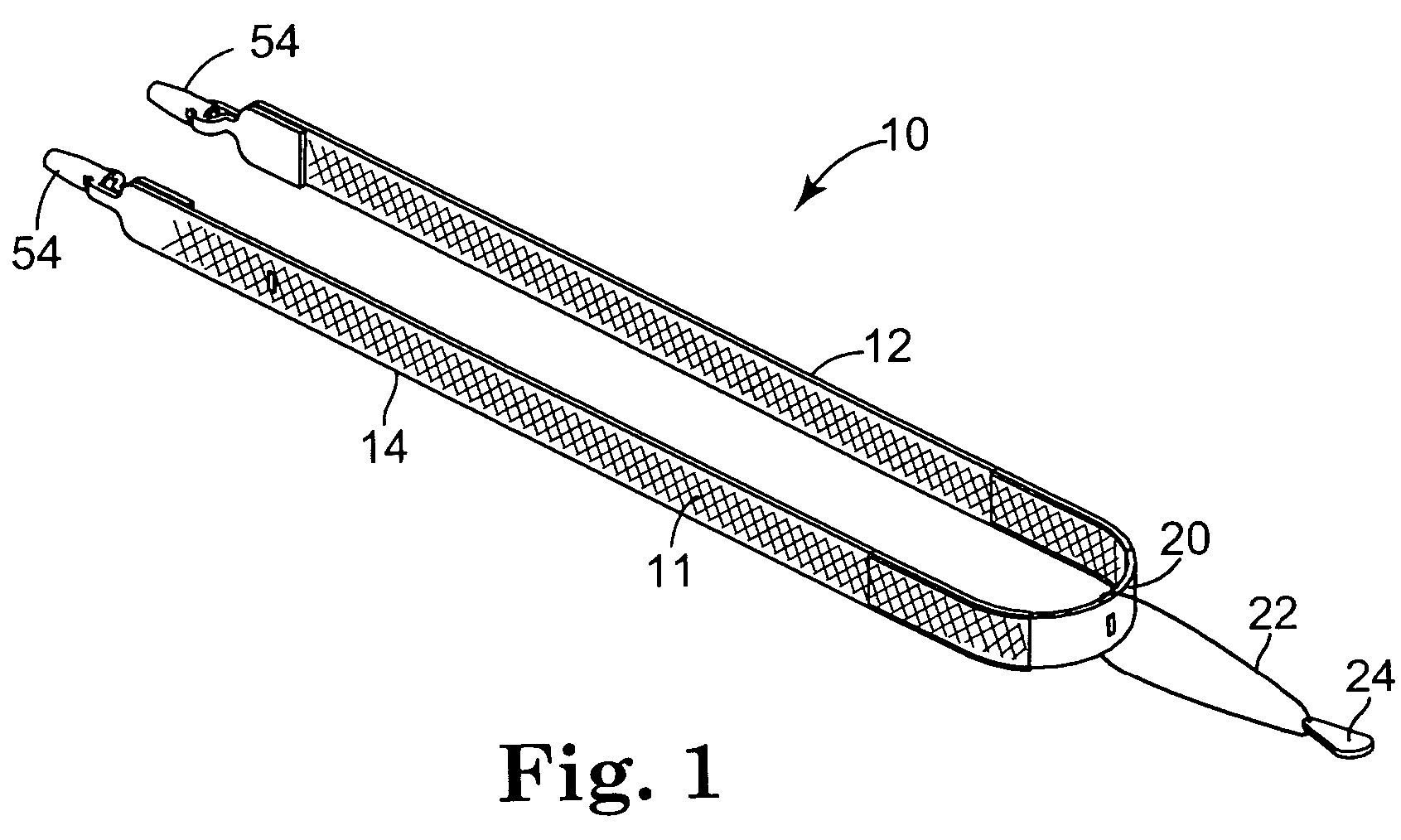
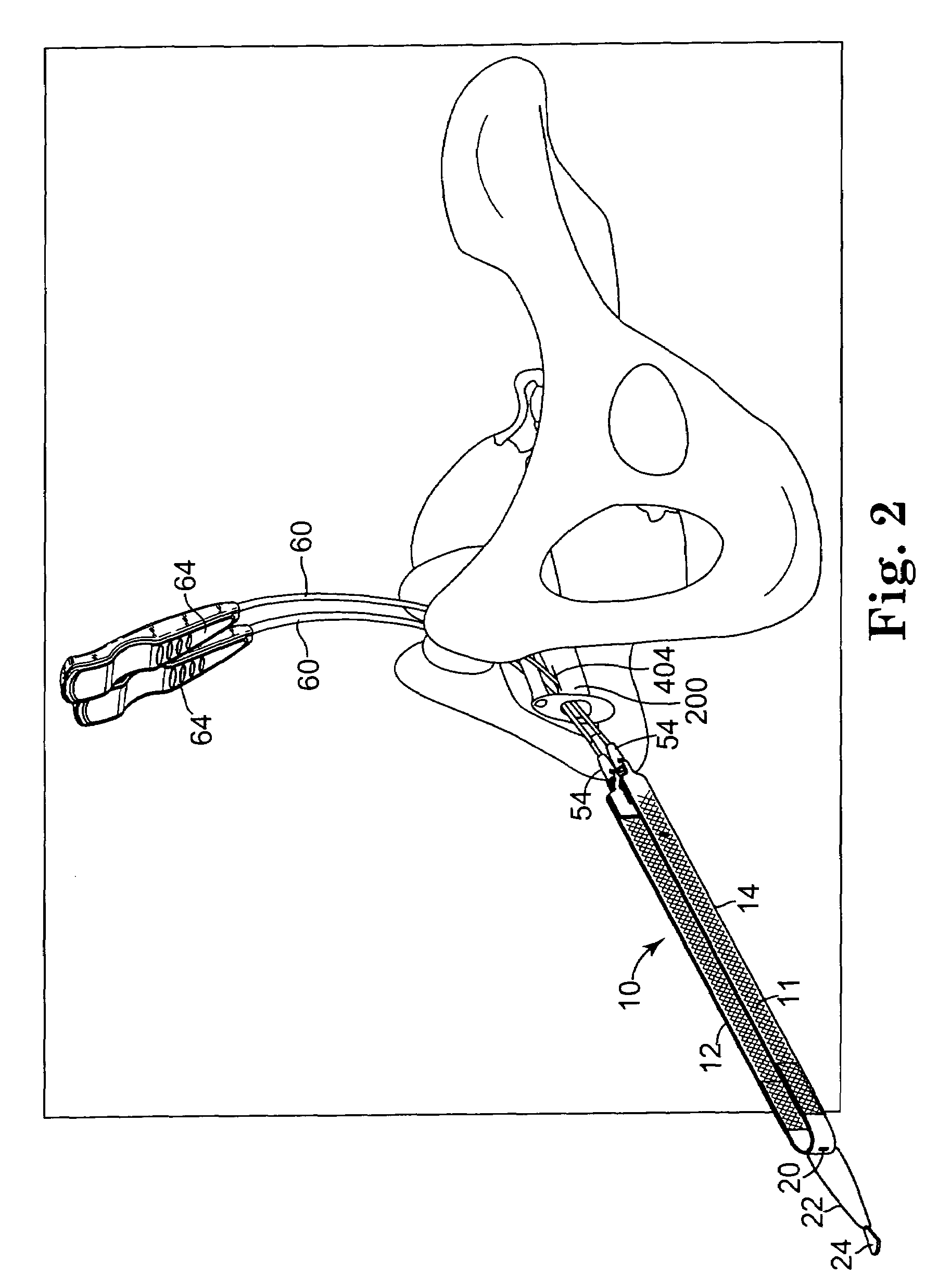
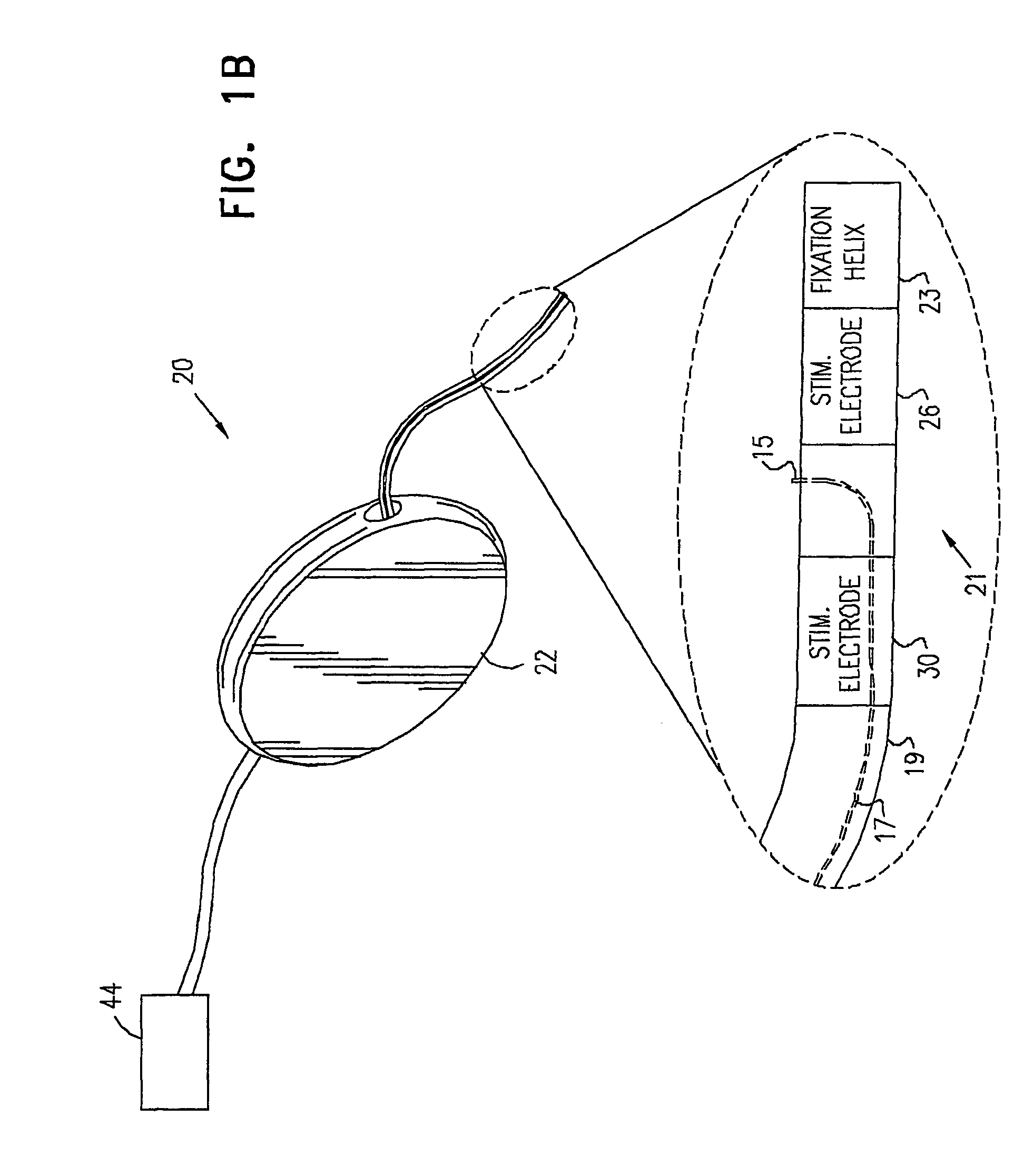
Pelvic disorder treatment device
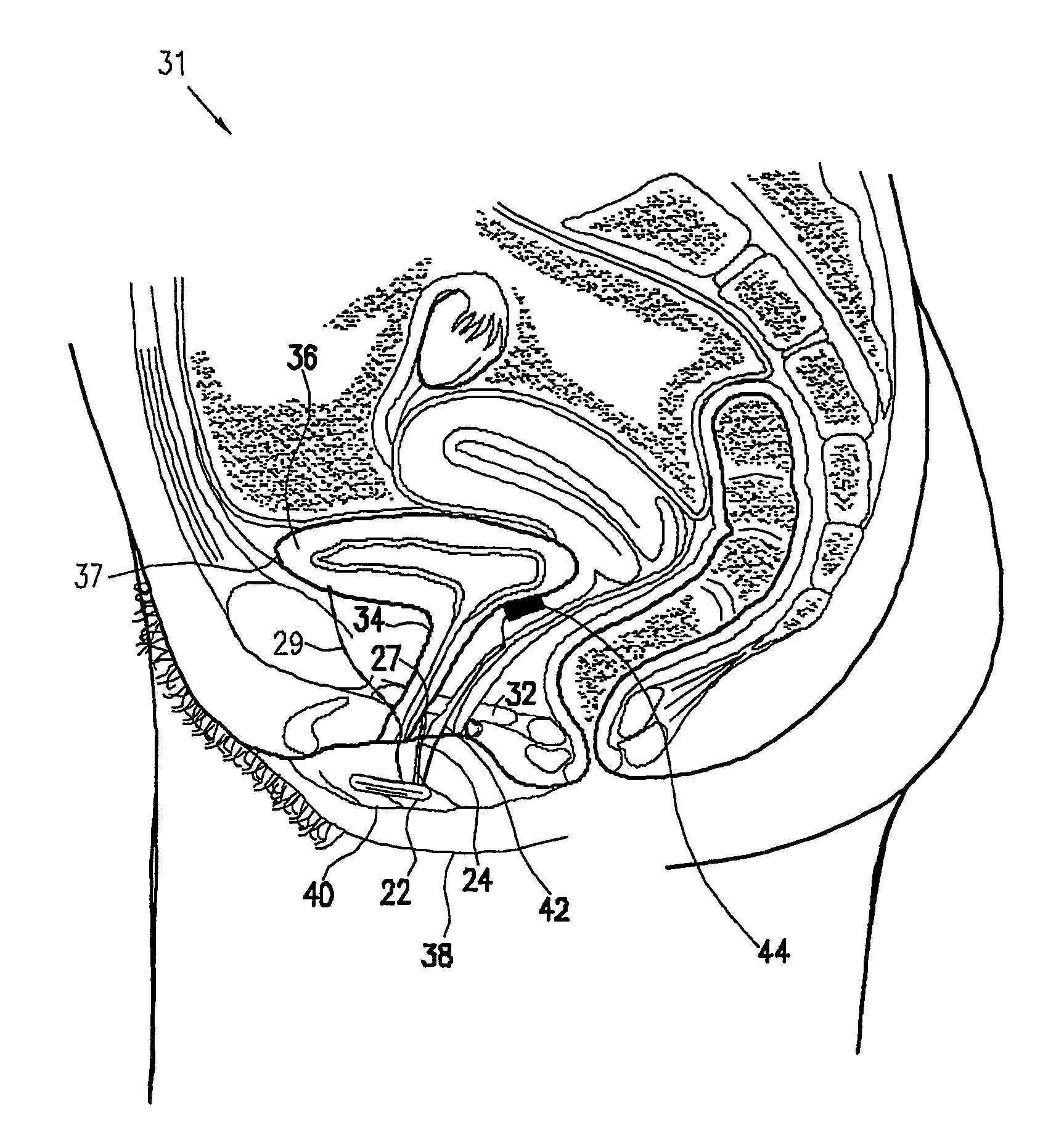
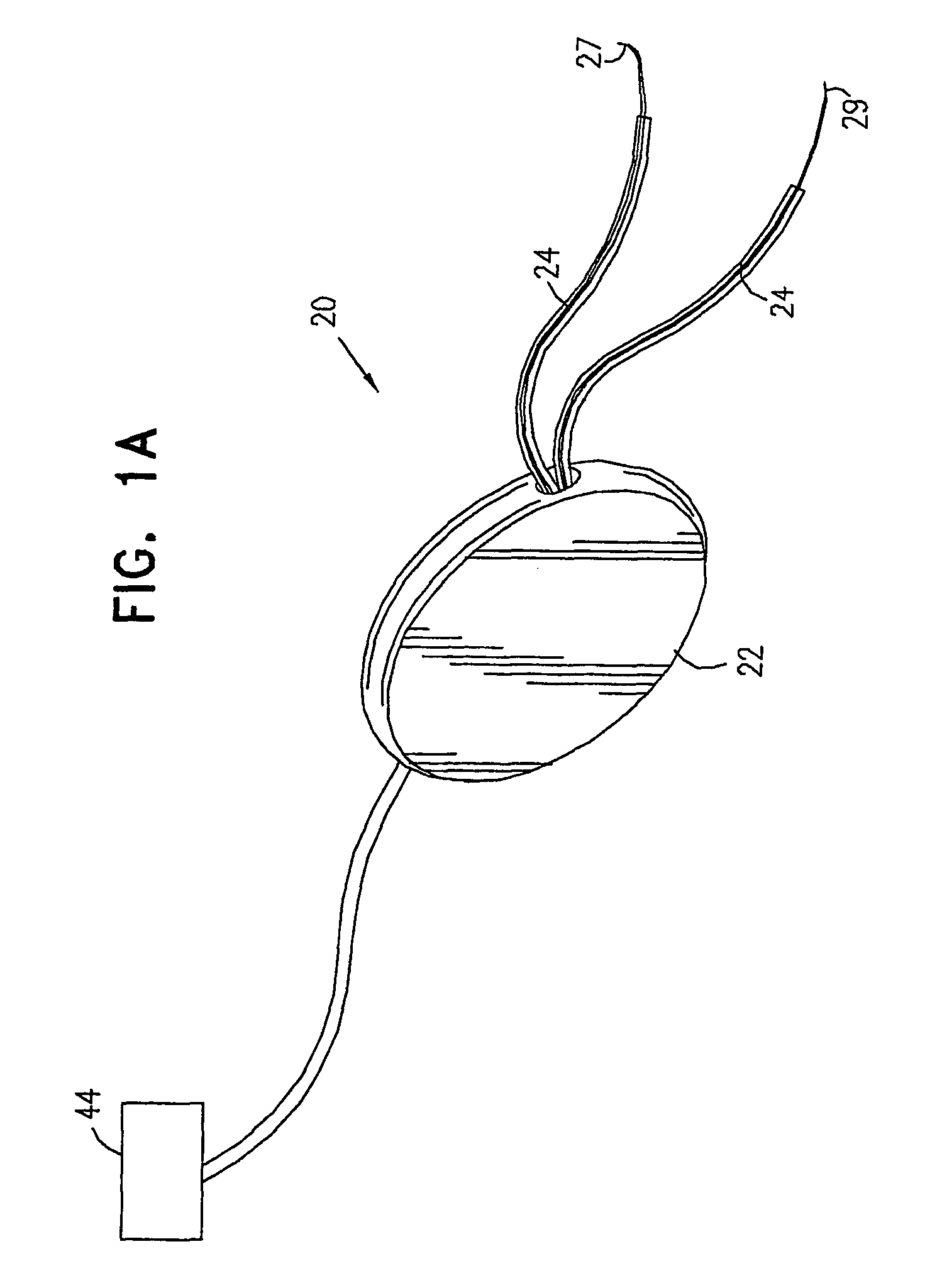
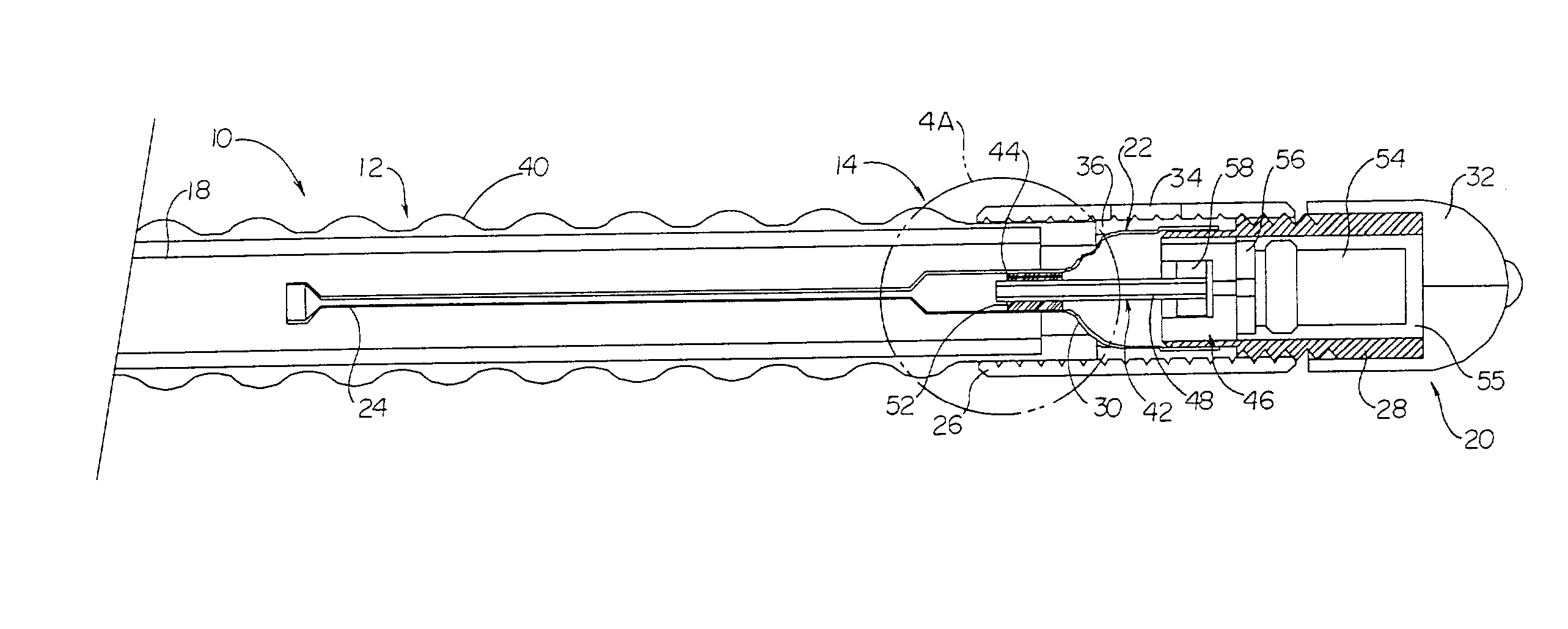
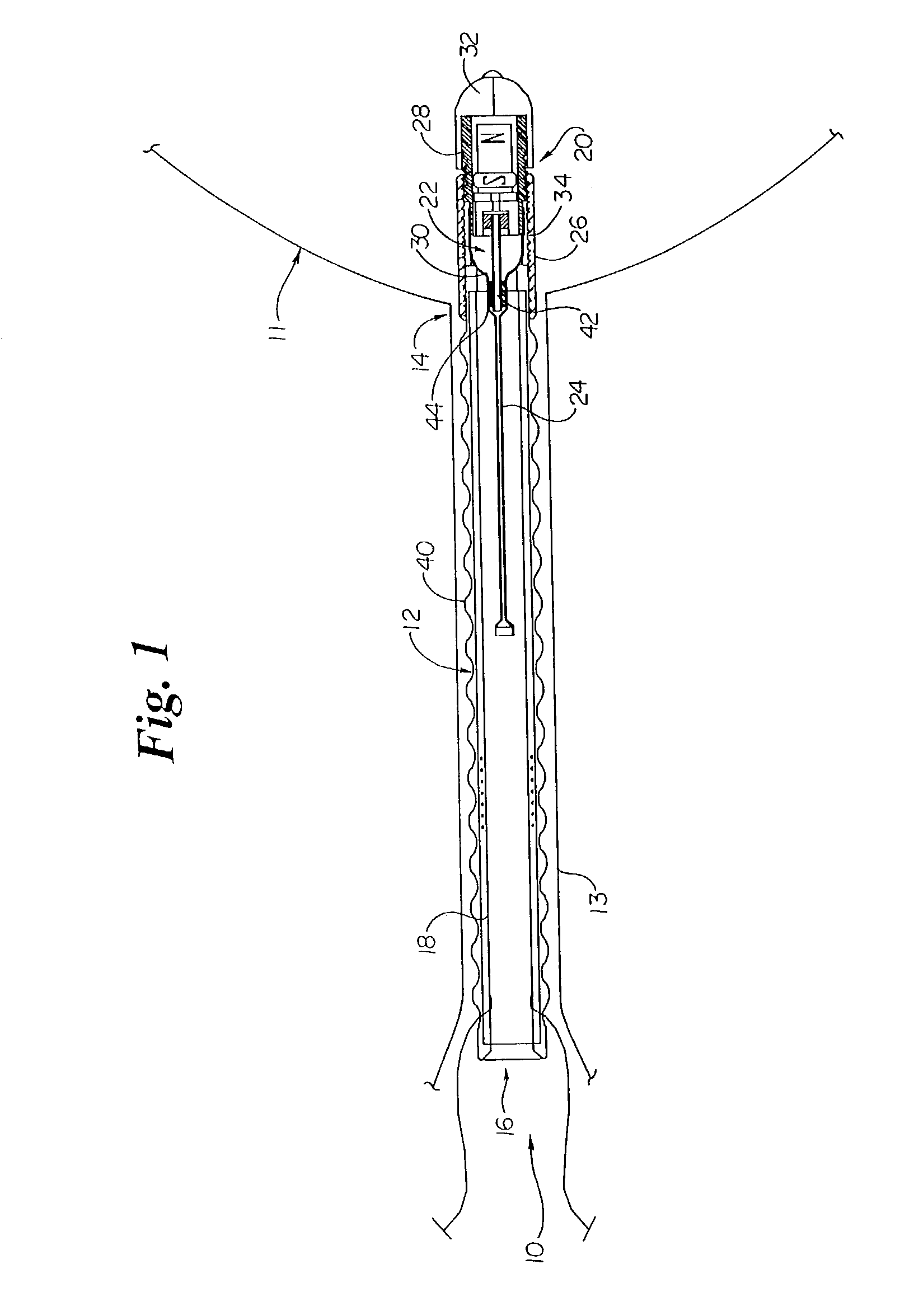
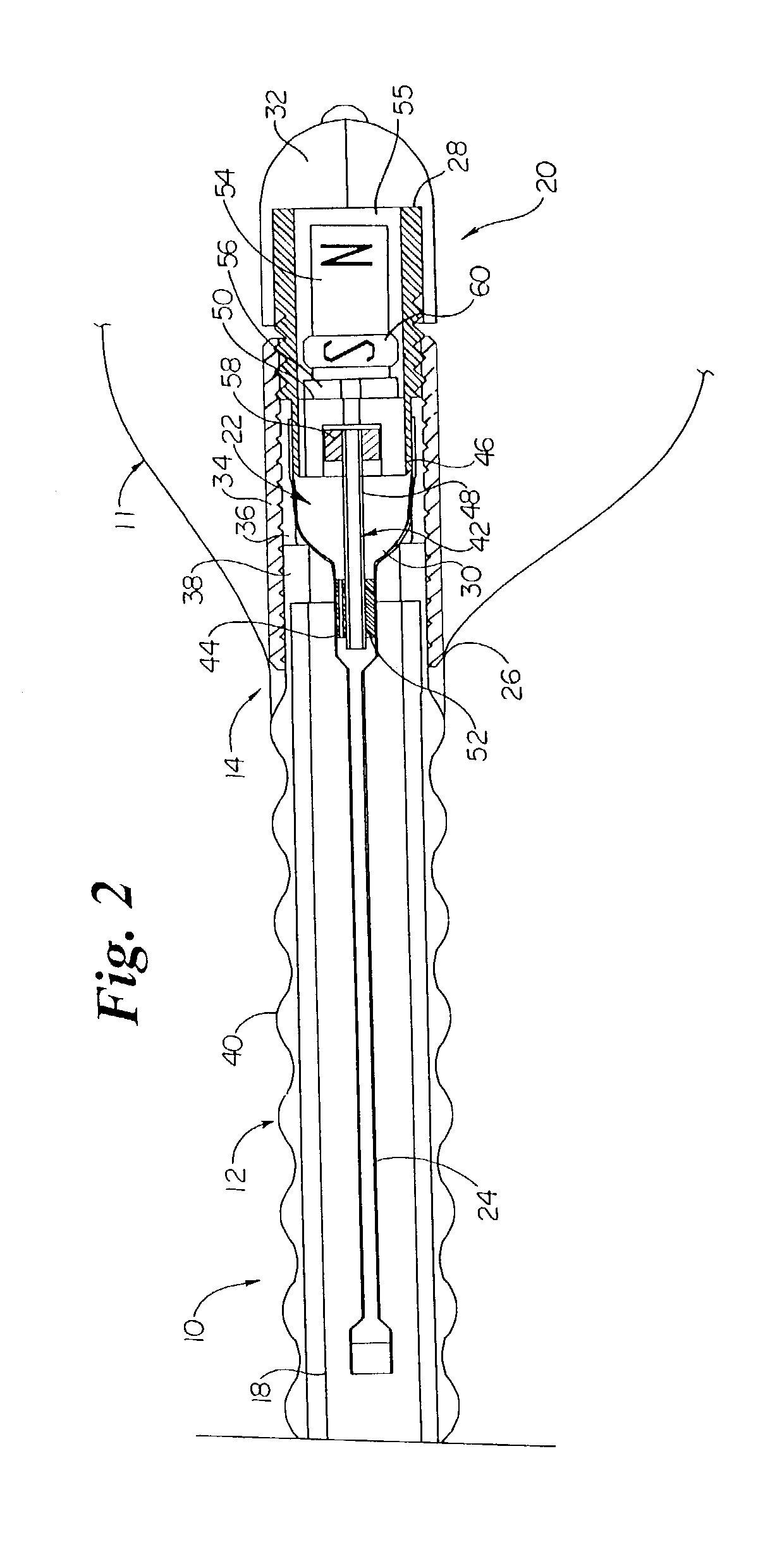
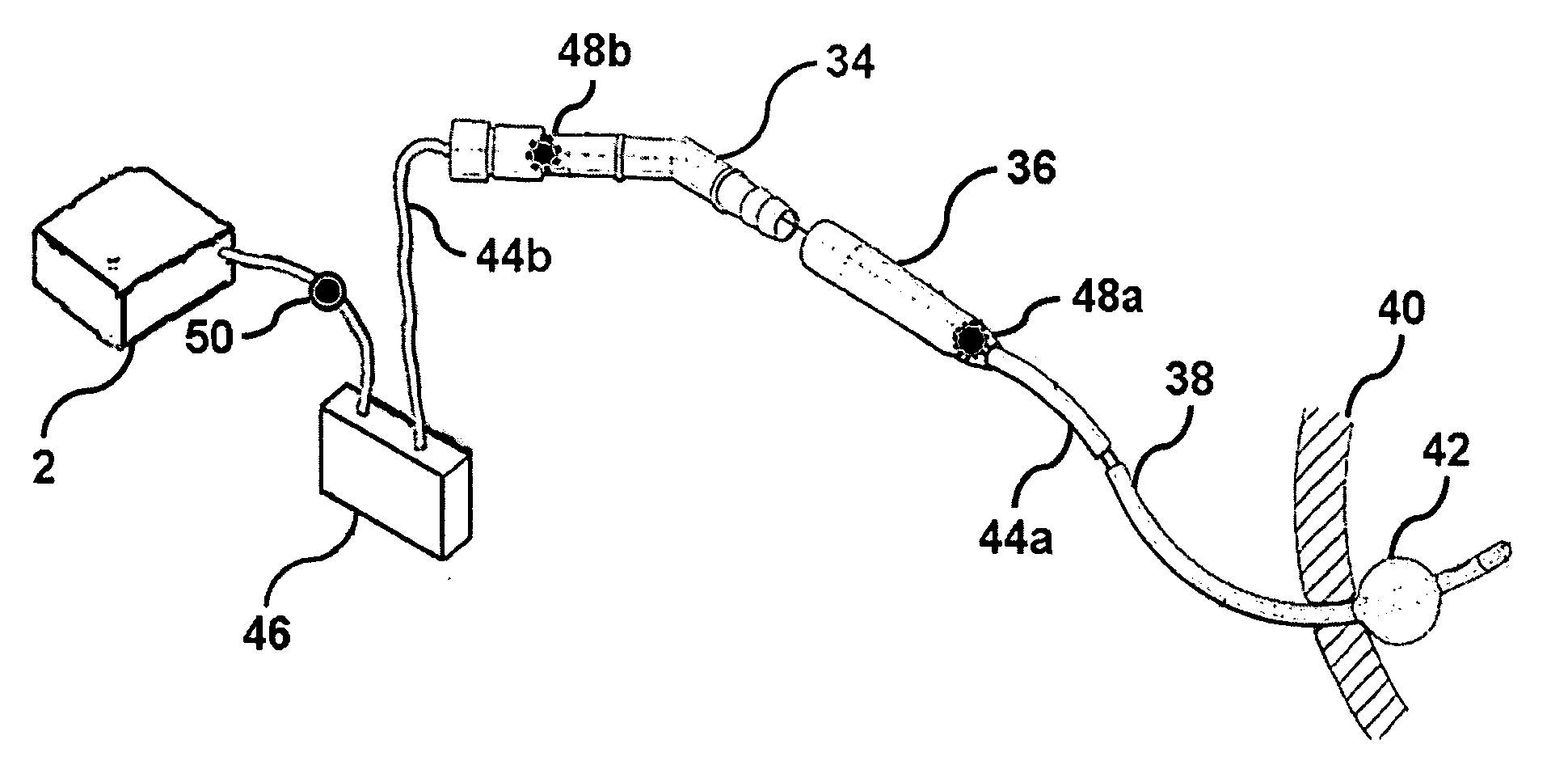
InactiveUS6862480B2Reduce decreaseRelieving pelvic painUltrasonic/sonic/infrasonic diagnosticsElectrotherapyInterstitial cystitisFecal incontinence
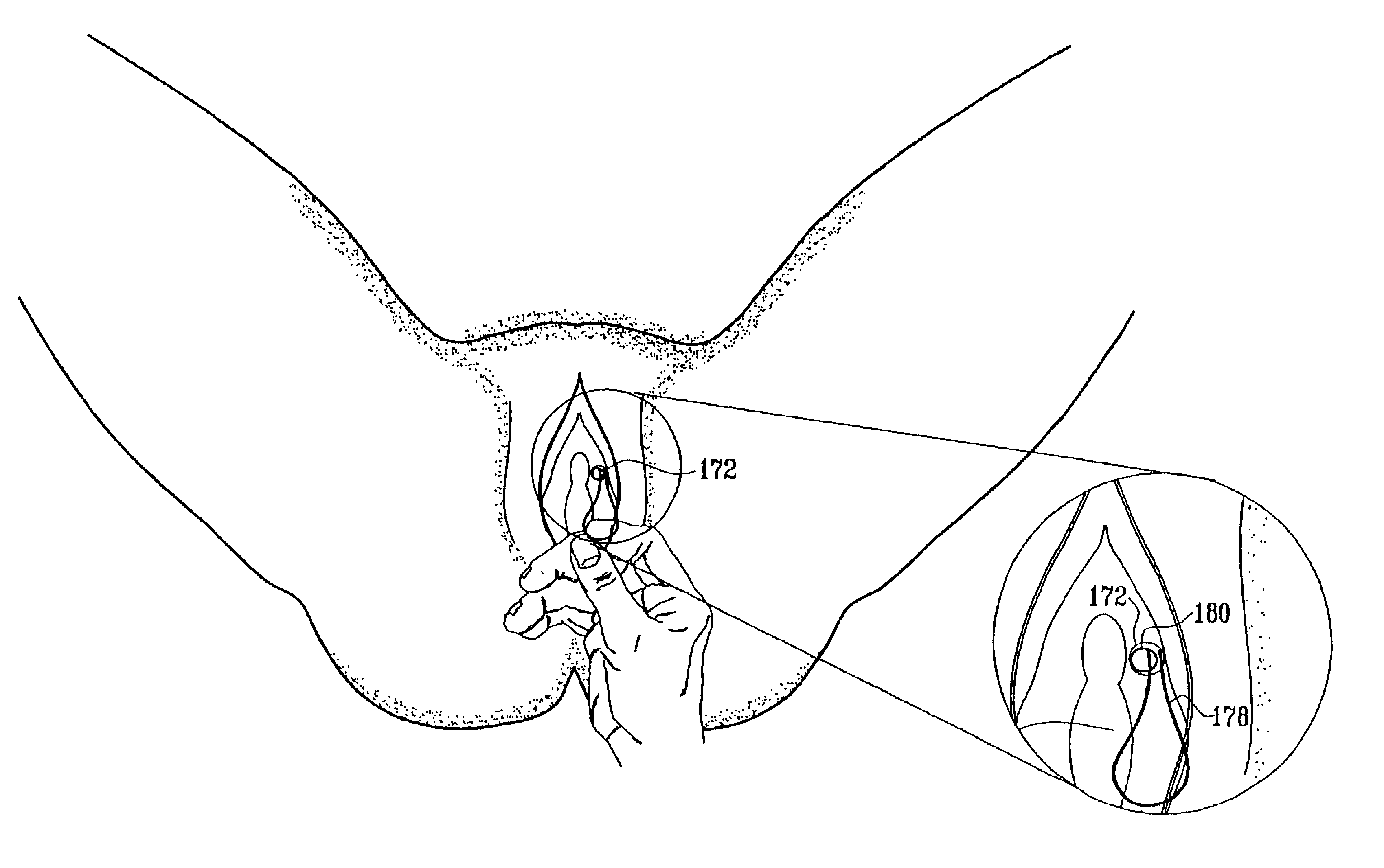
A device for treating a medical condition is provided, and a surgical procedure for implanting the device is disclosed. The device includes a sensor, which is adapted to generate a signal responsive to a state of a patient, and at least one electrode, which is adapted to be coupled to a pelvic site of the patient. A control unit is adapted to receive the signal, to analyze the signal so as to distinguish between an imminent stress incontinence event and an imminent urge event, and, responsive to analyzing the signal, to apply an electrical waveform to the at least one electrode. In various configurations, the device may be used alternatively or additionally to treat fecal incontinence, interstitial cystitis, chronic pelvic pain, or urine retention.
Owner:ASTORA WOMENS HEALTH
Incontinence sling
A sling for controlling urinary incontinence is disclosed. The sling is formed from a tube having substantially lengthwise inextensible end portions and an elastically lengthwise extensible intermediate portion between the end portions. The intermediate portion has a U-shape which cradles the urethra. The end portions extend through the abdominal tissue to anchor the intermediate portion in position. The sling places the urethra under a transverse compressive load to hold it closed and prevent inadvertent urination. The tube is formed from interlaced filamentary members. Interlacing may be by warp knitting, weaving using a leno weave or braiding using a tri-axial braid structure. The end portions have a rough texture to facilitate anchoring in the tissue of the abdominal wall. The intermediate portion is smooth and soft to prevent tissue erosion.
Owner:STOUT MEDICAL GROUP
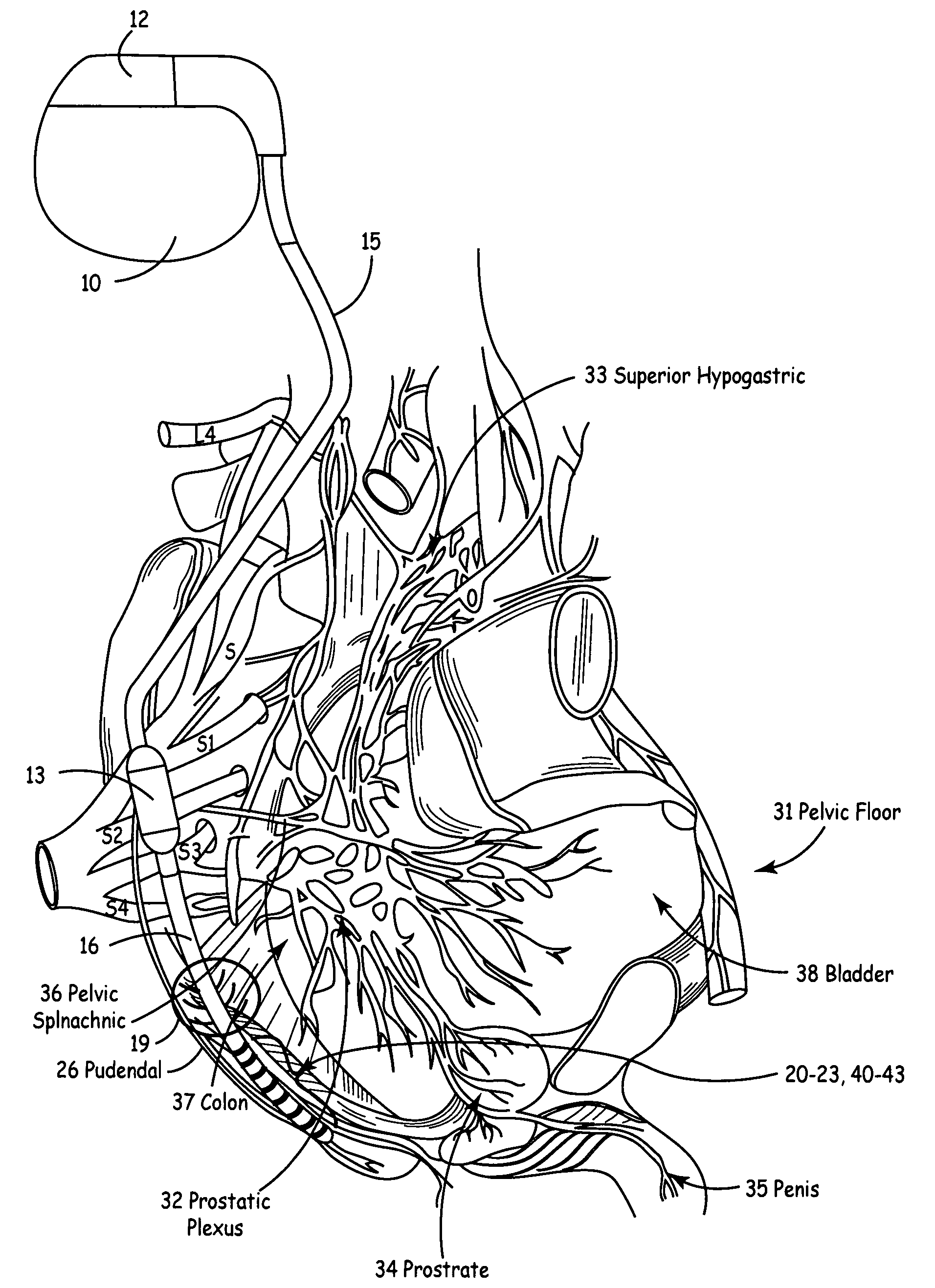
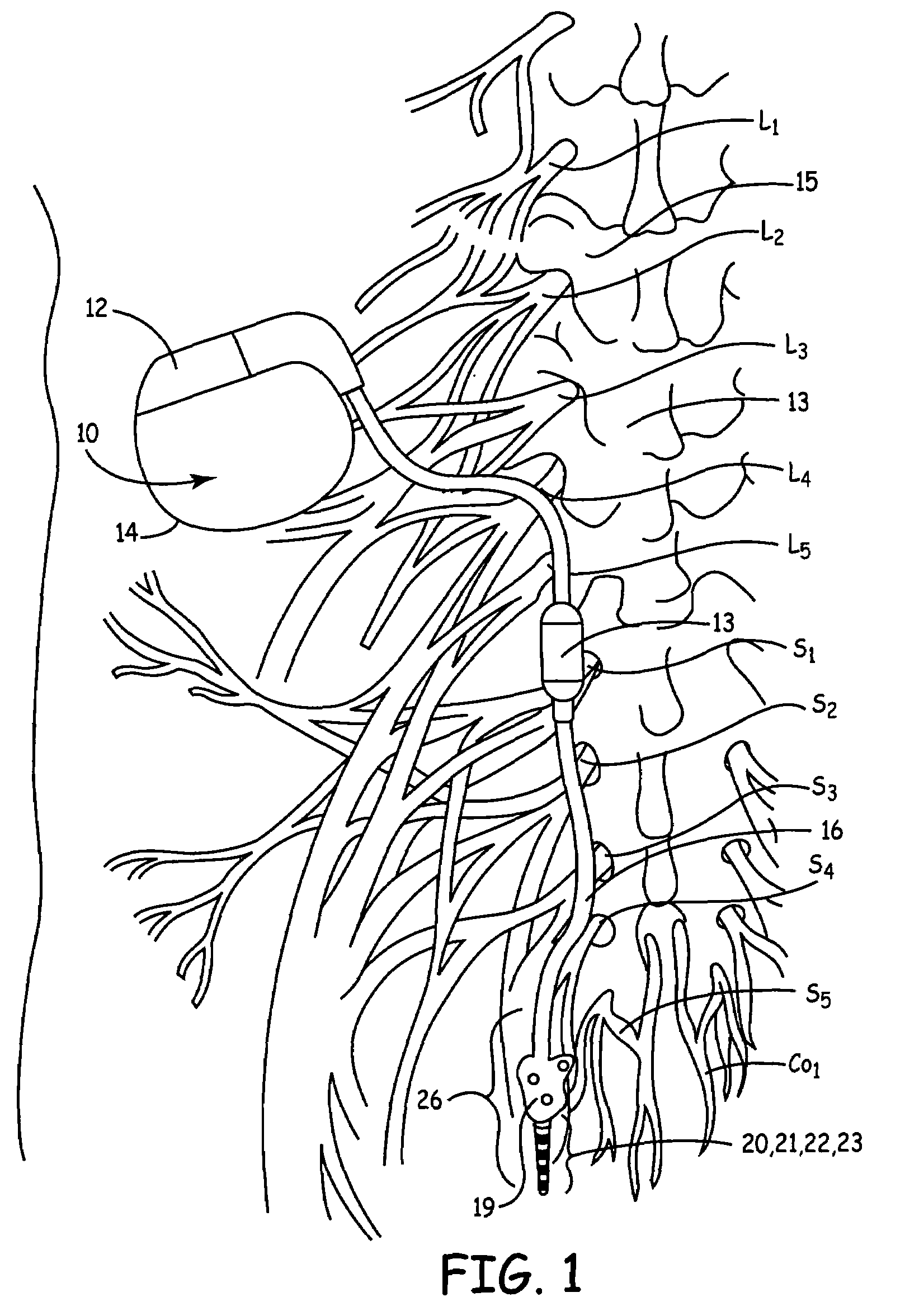
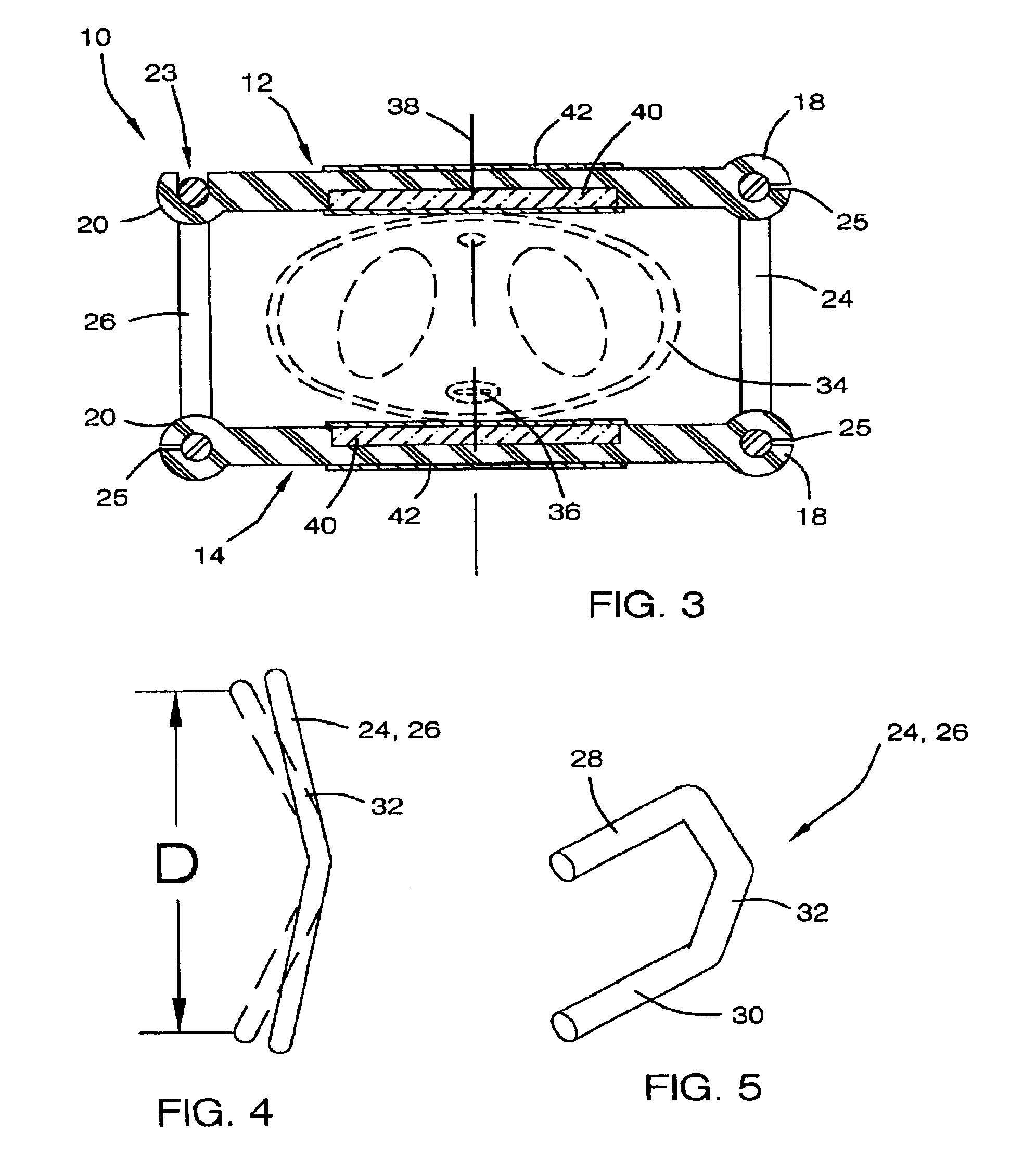
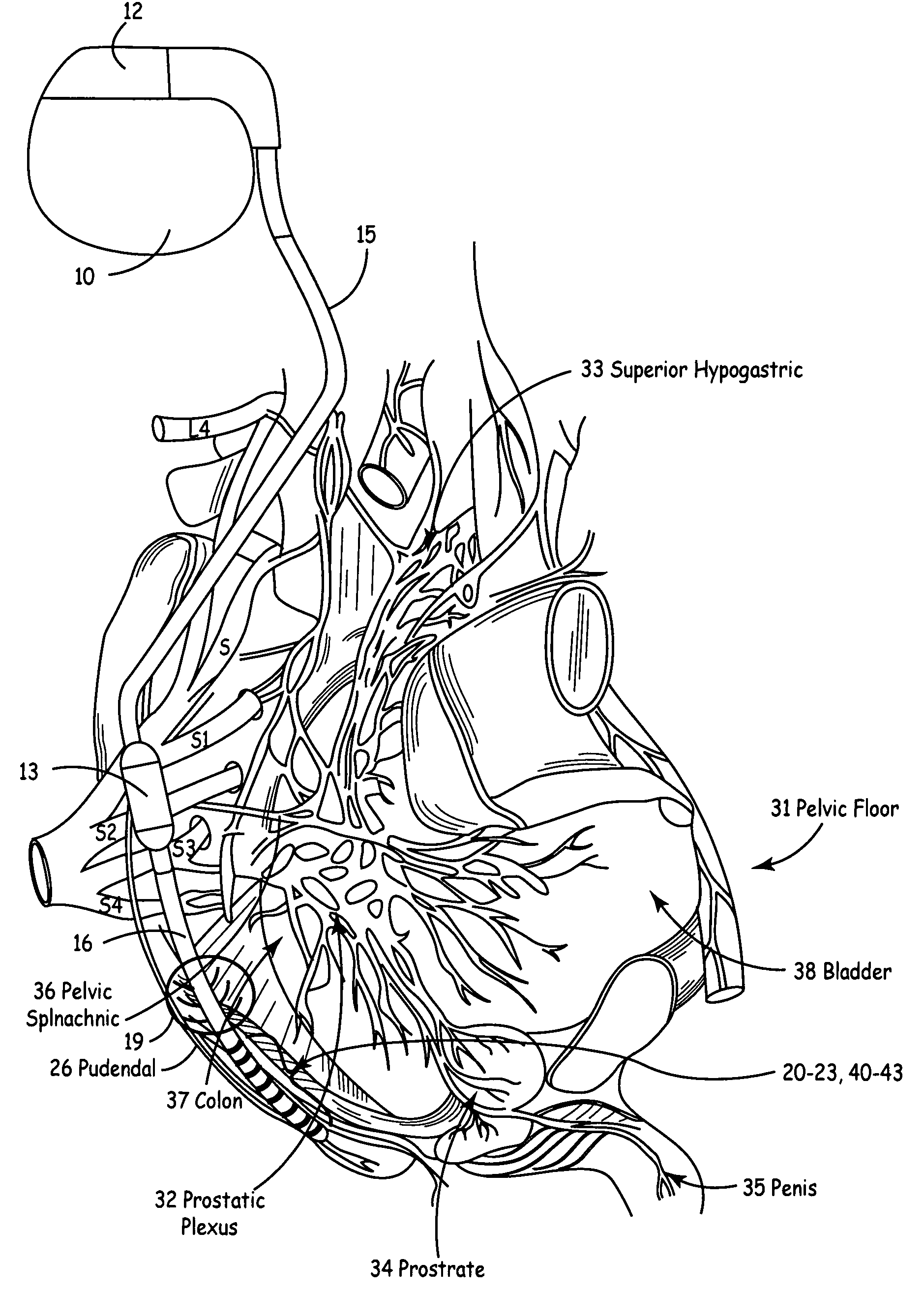
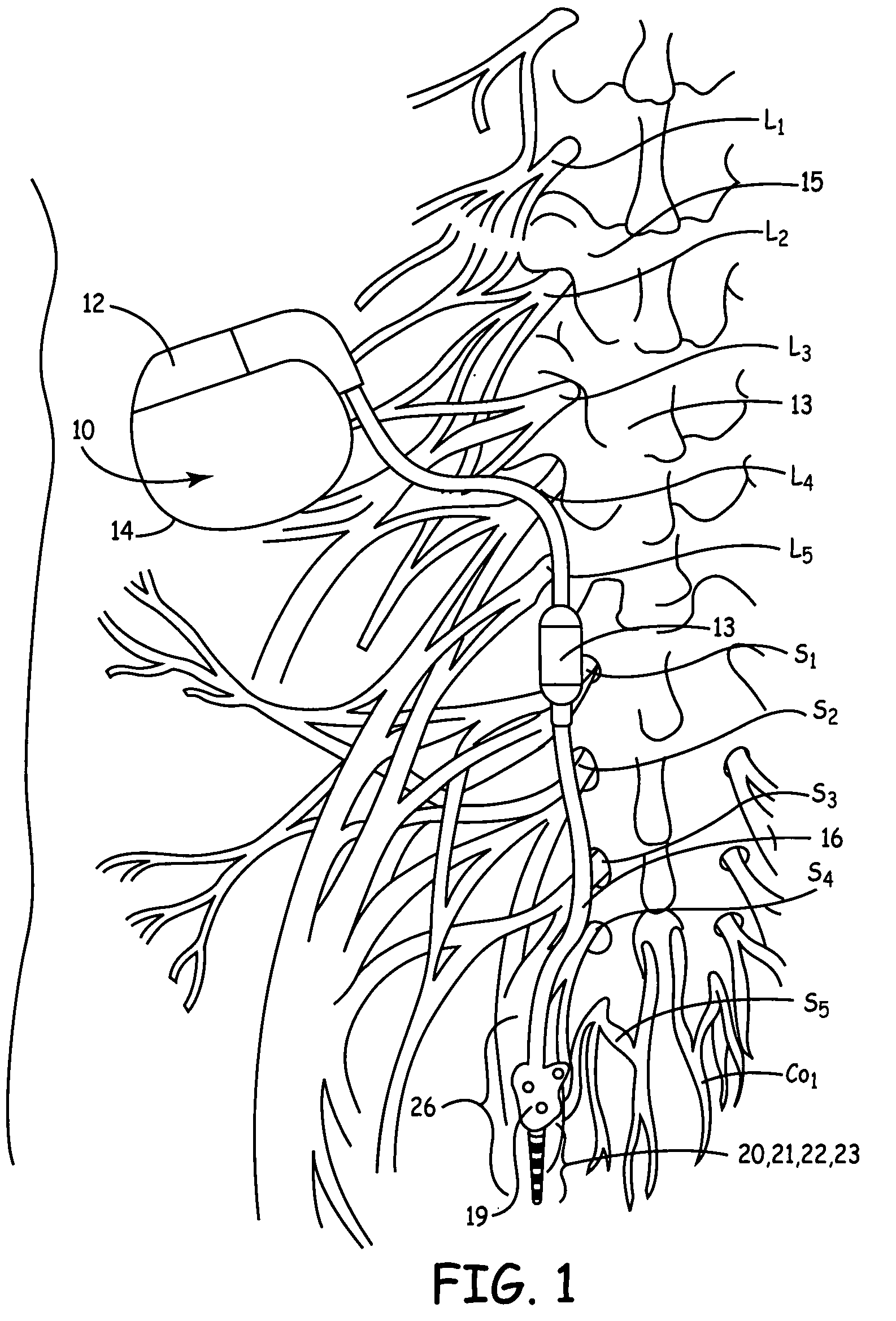
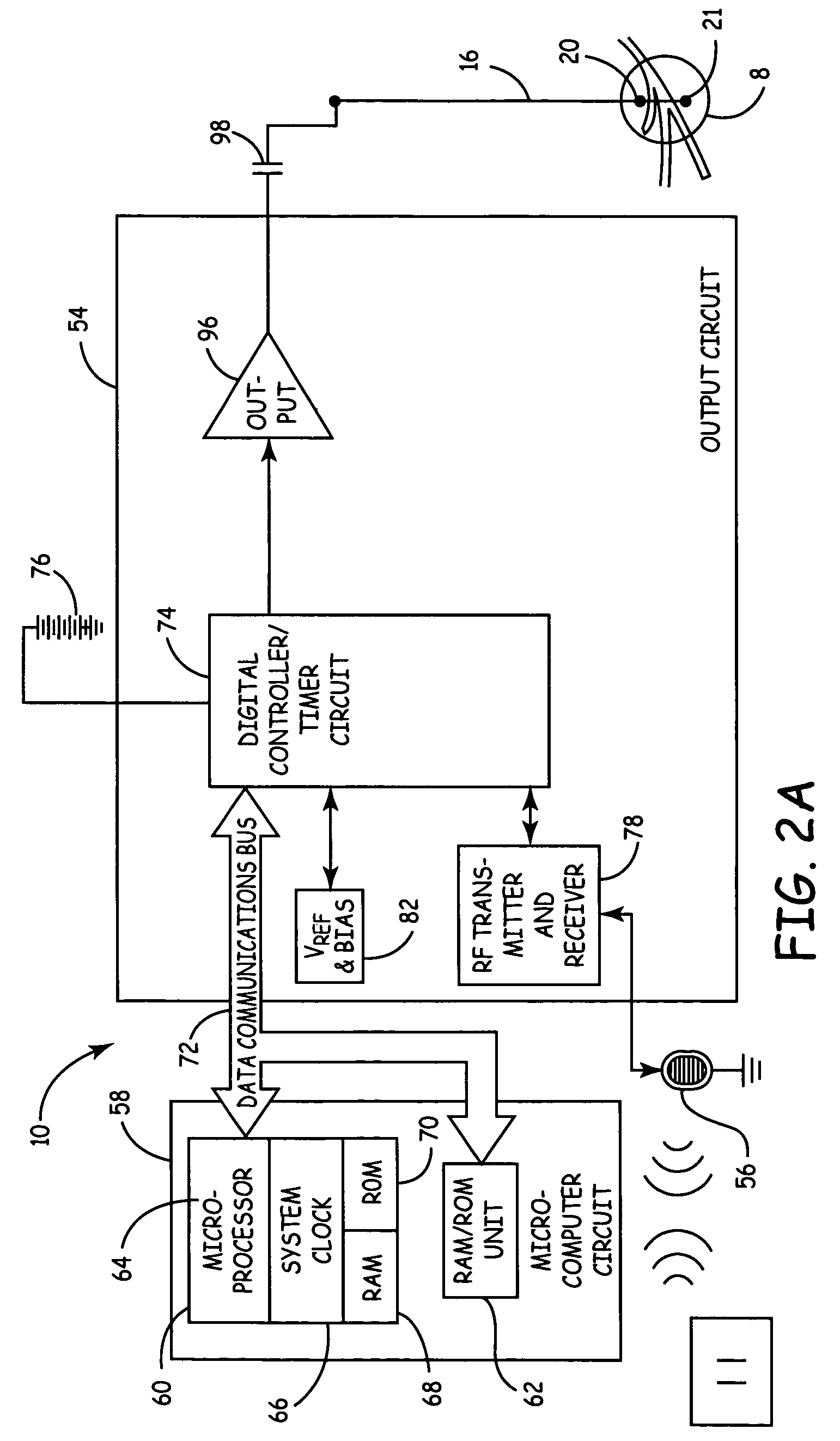
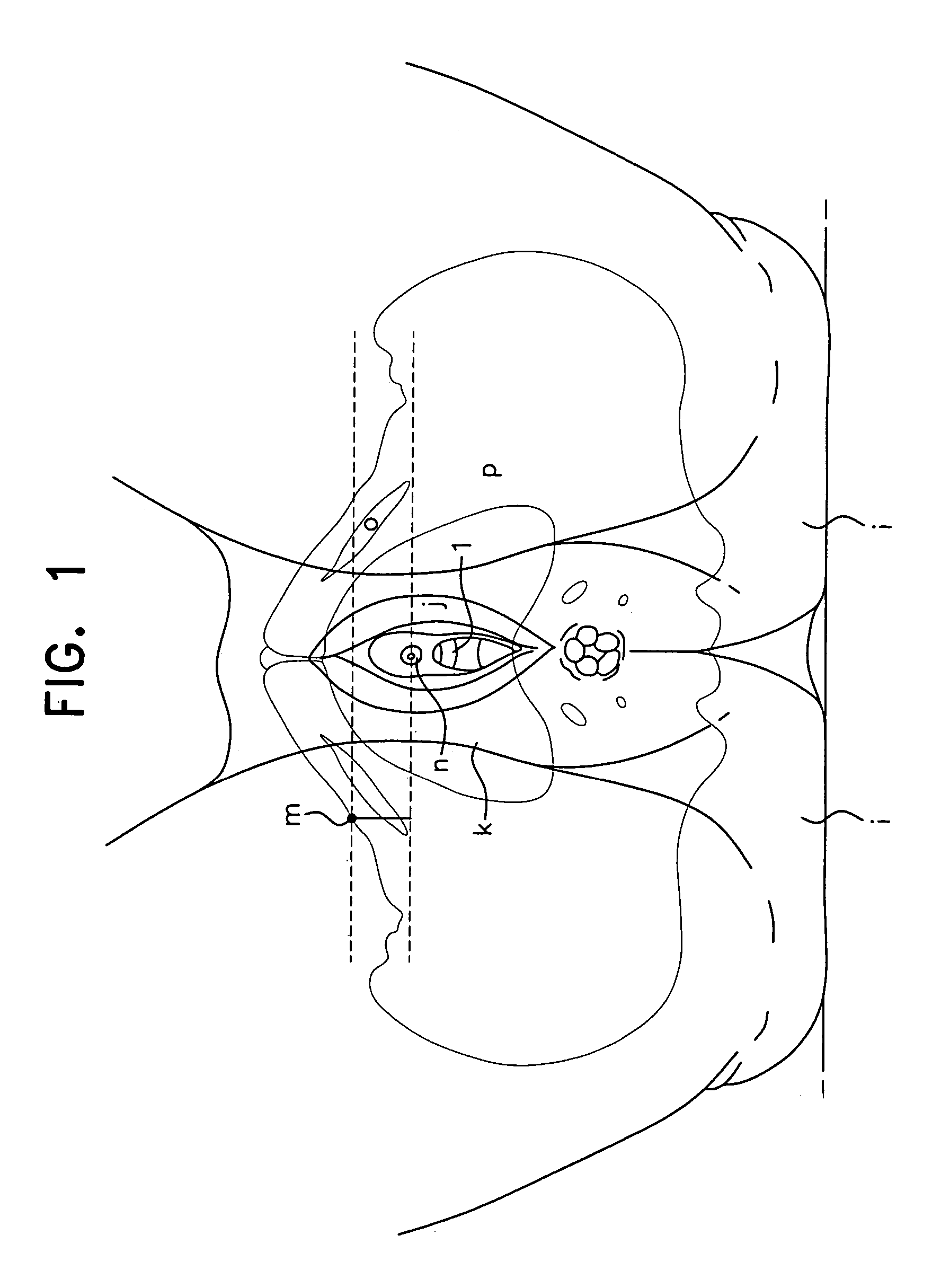
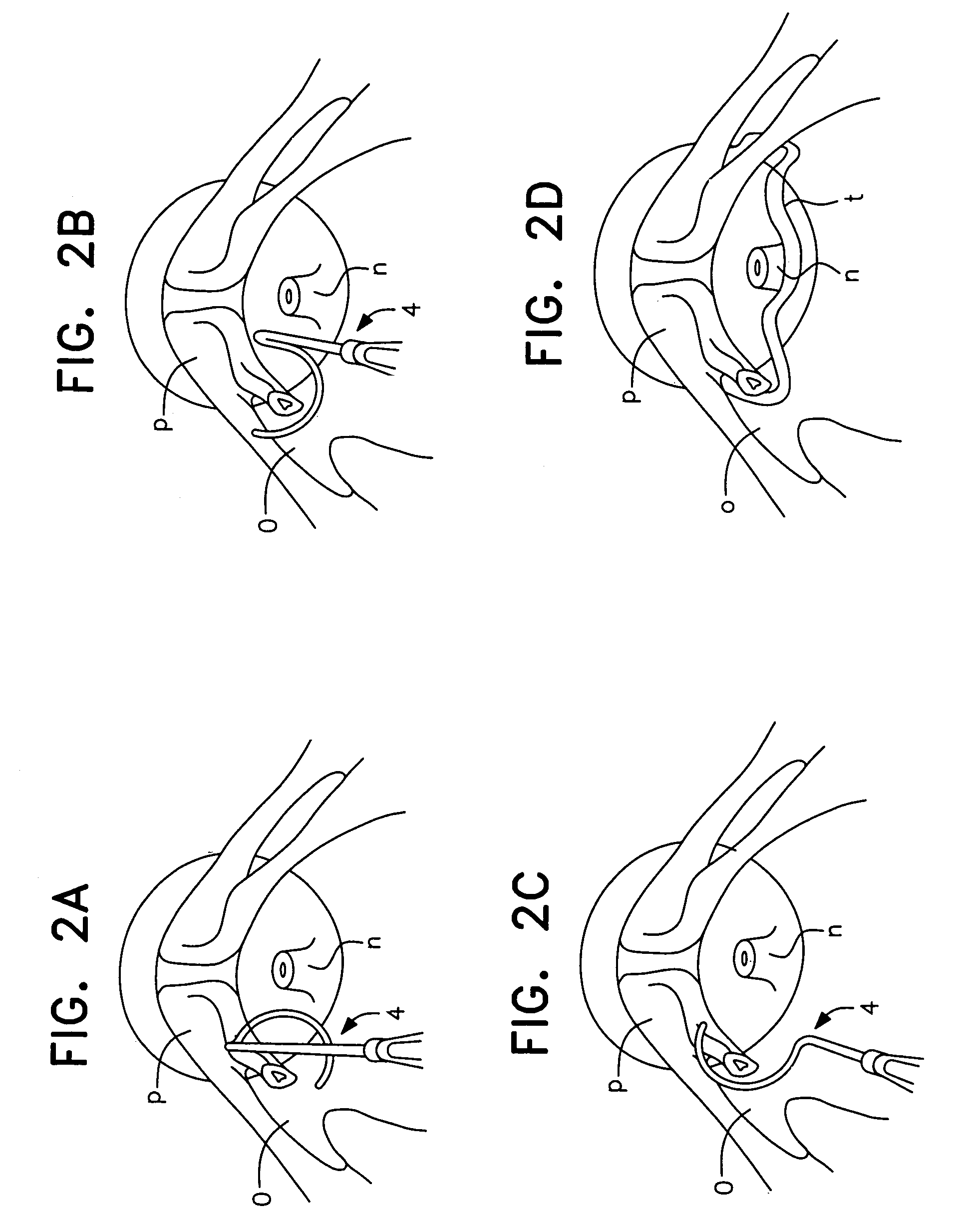
Method, system and device for treating disorders of the pelvic floor by means of electrical stimulation of the pudendal and associated nerves, and the optional delivery of drugs in association therewith
ActiveUS7328068B2Undesirable side effects of sacral nerve stimulation may be avoided or minimizedUndesirable side-effectDigestive electrodesGenital electrodesDiseaseProstatalgia
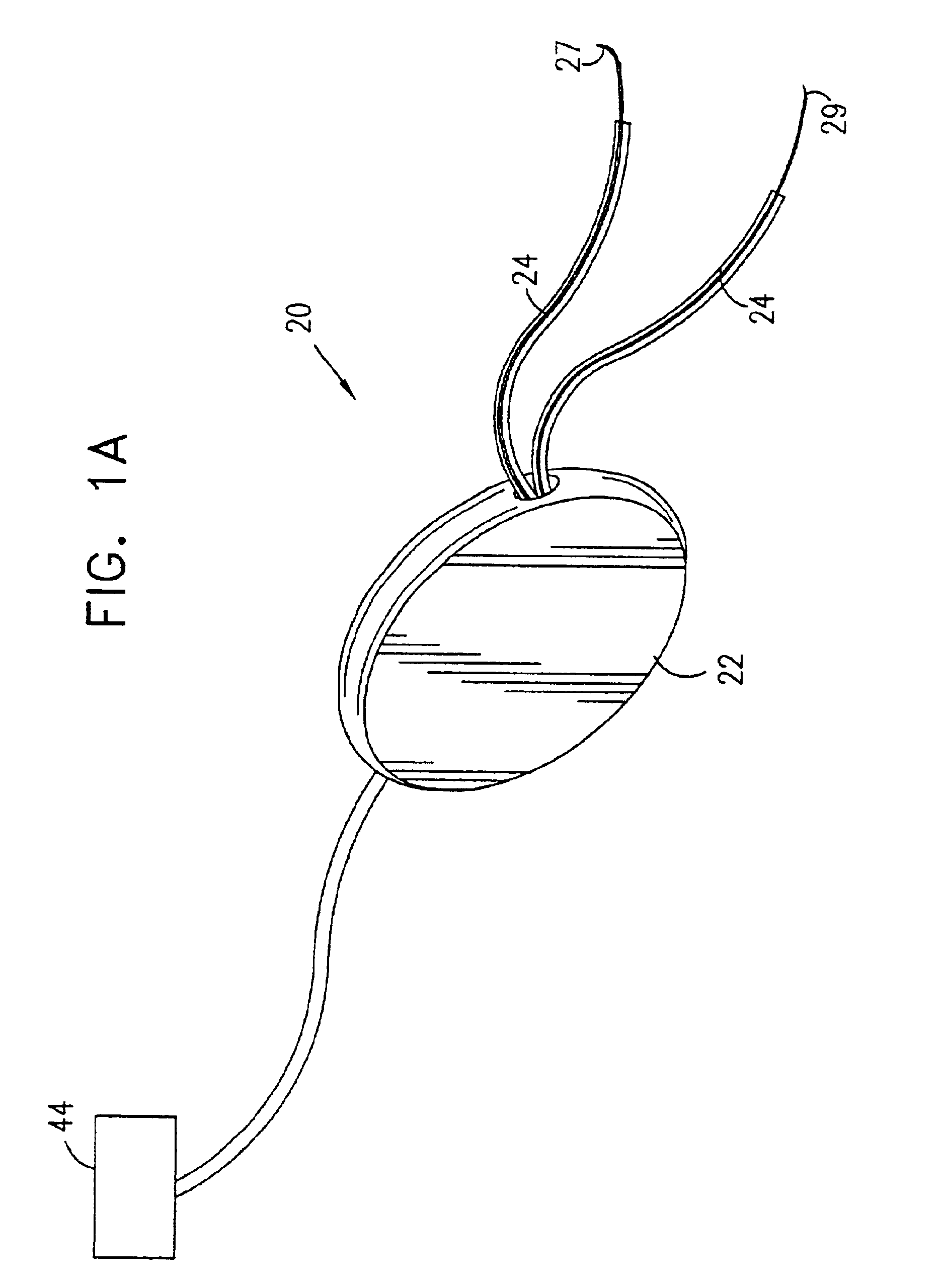
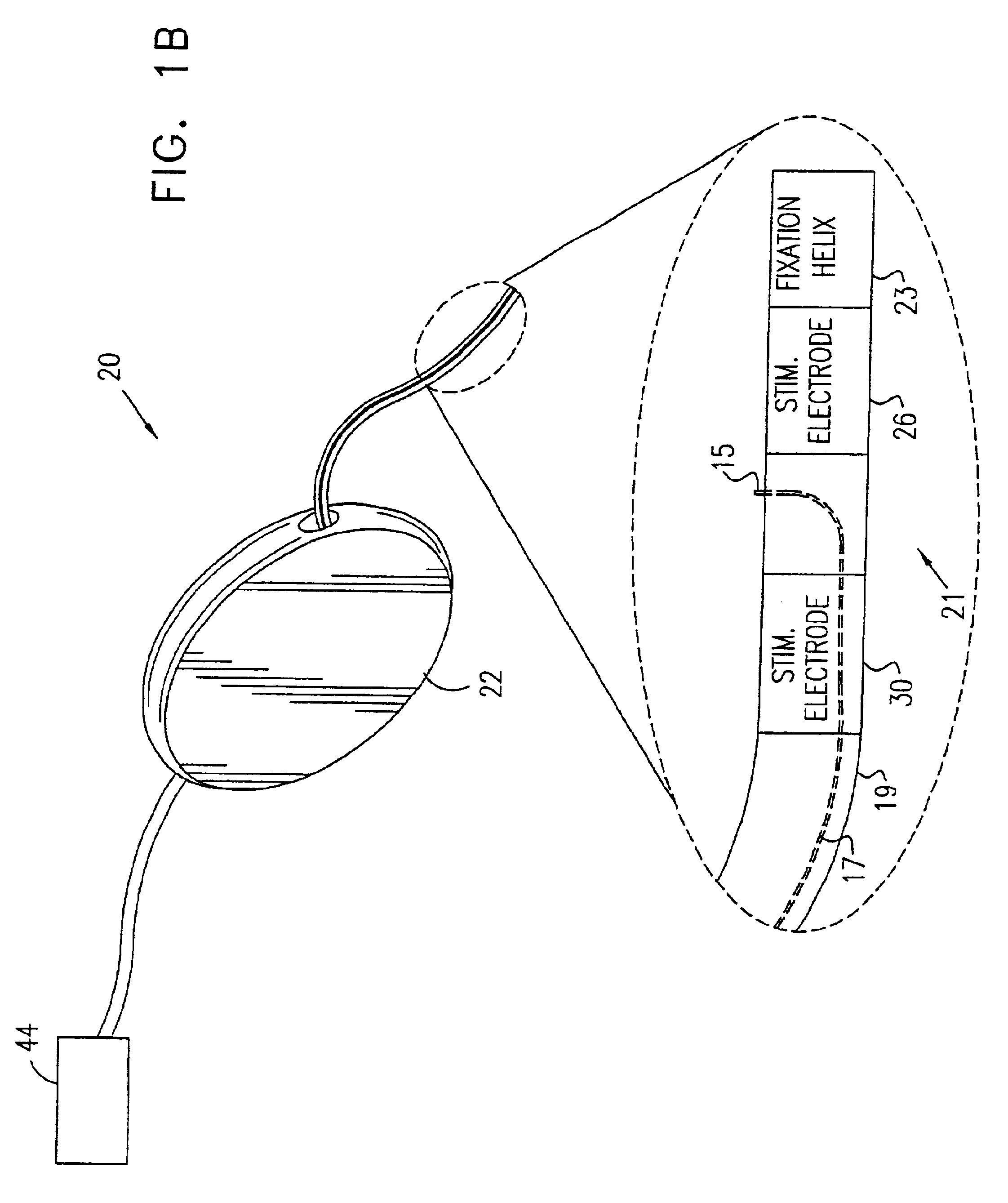
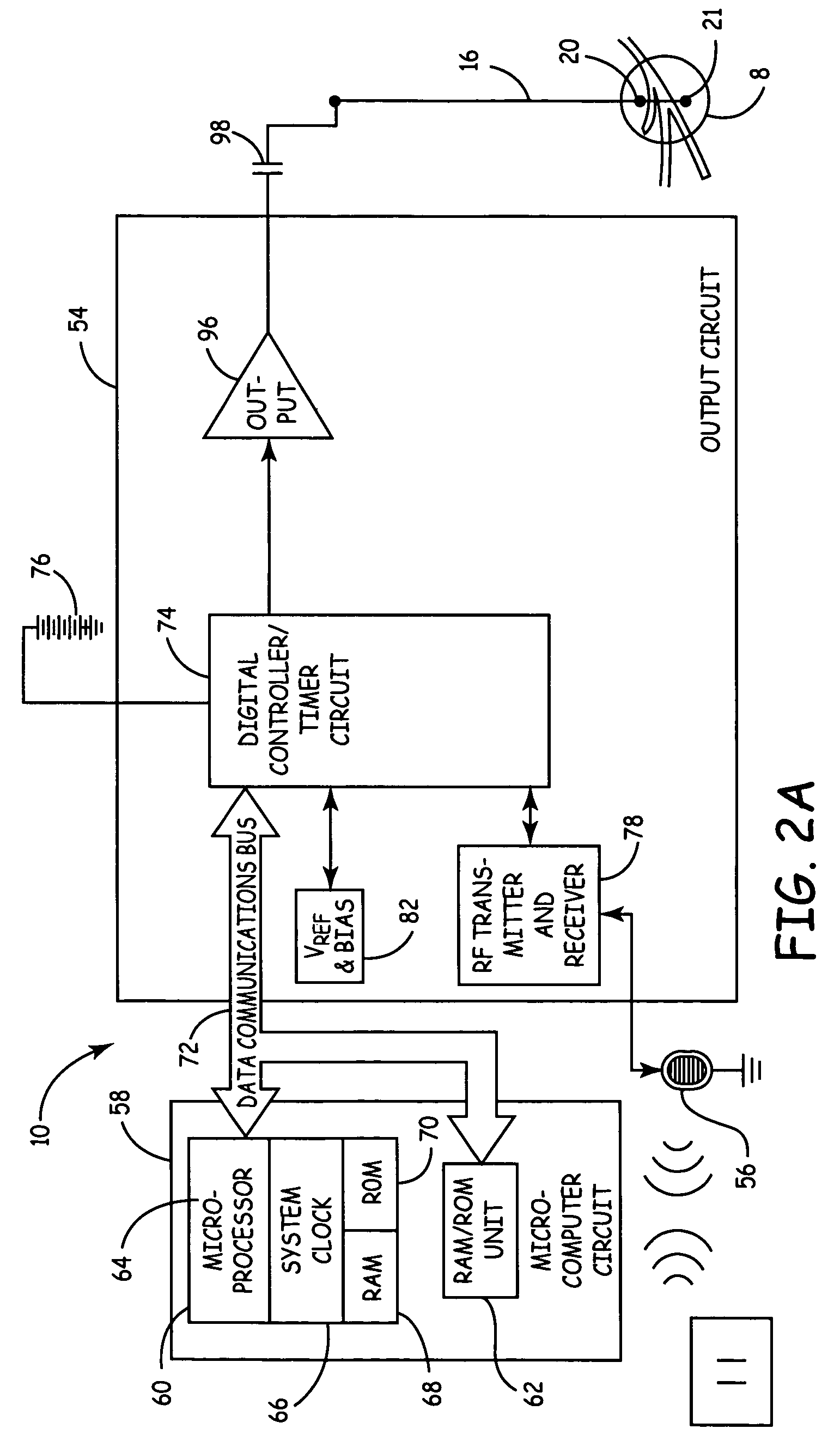
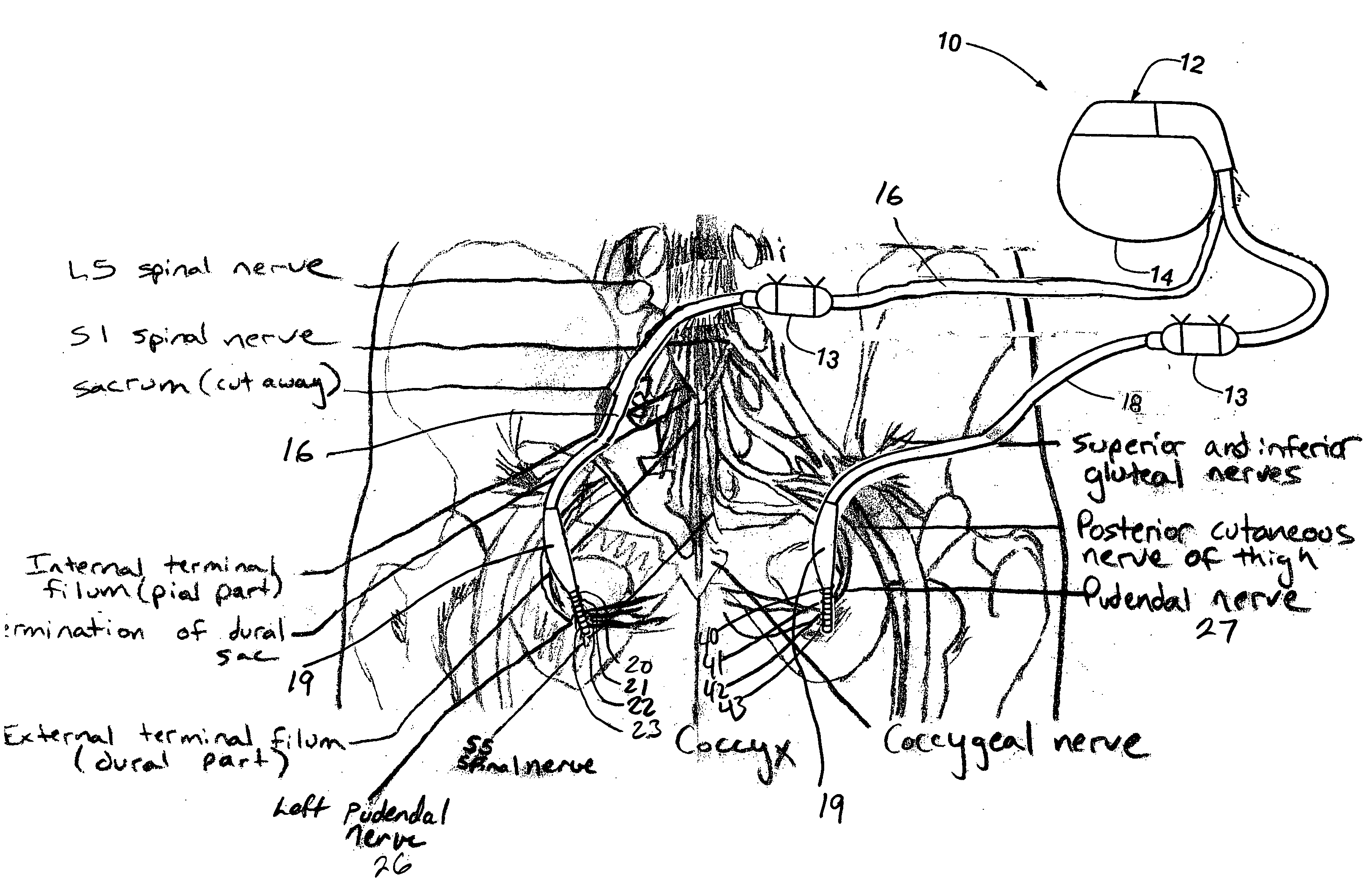
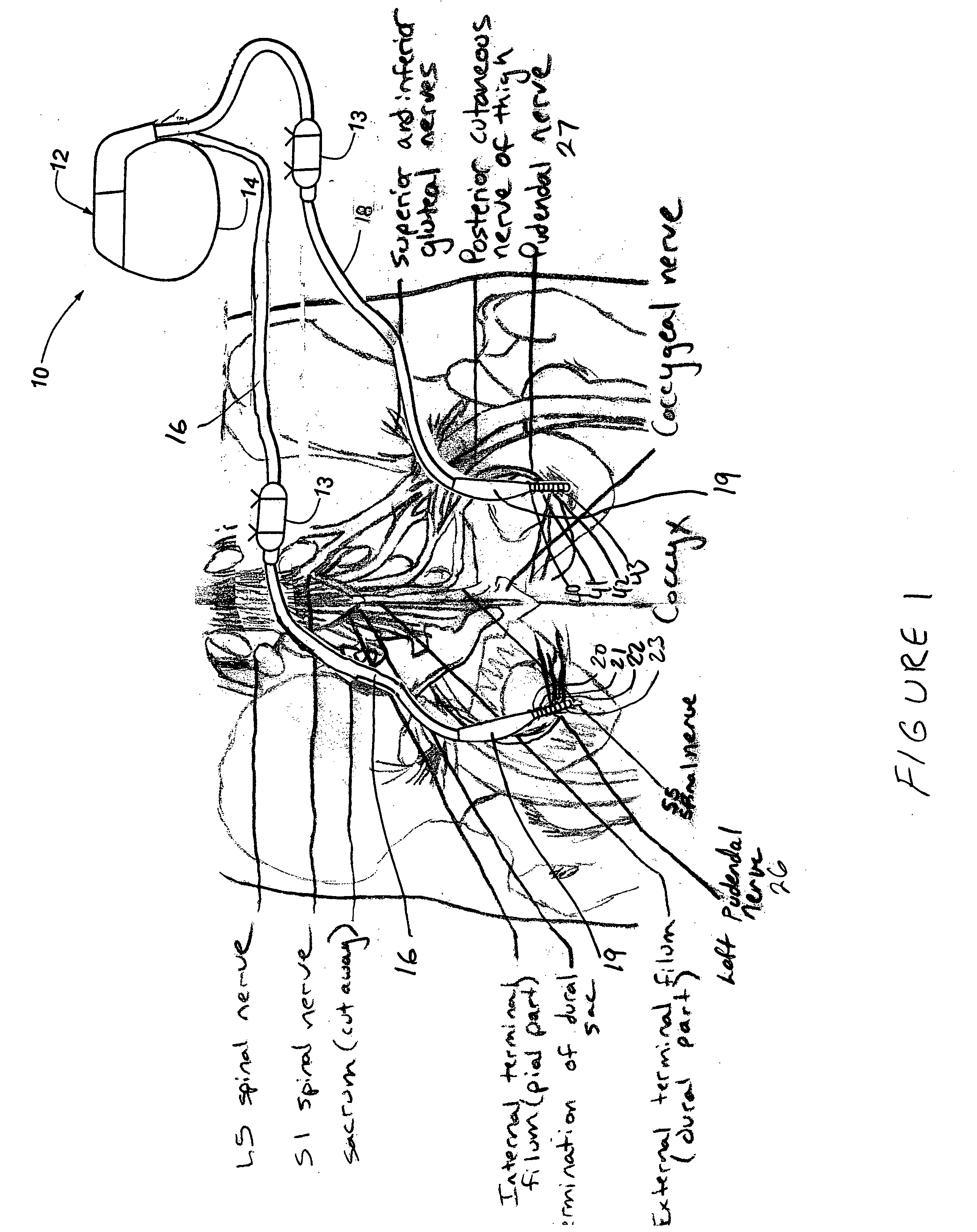
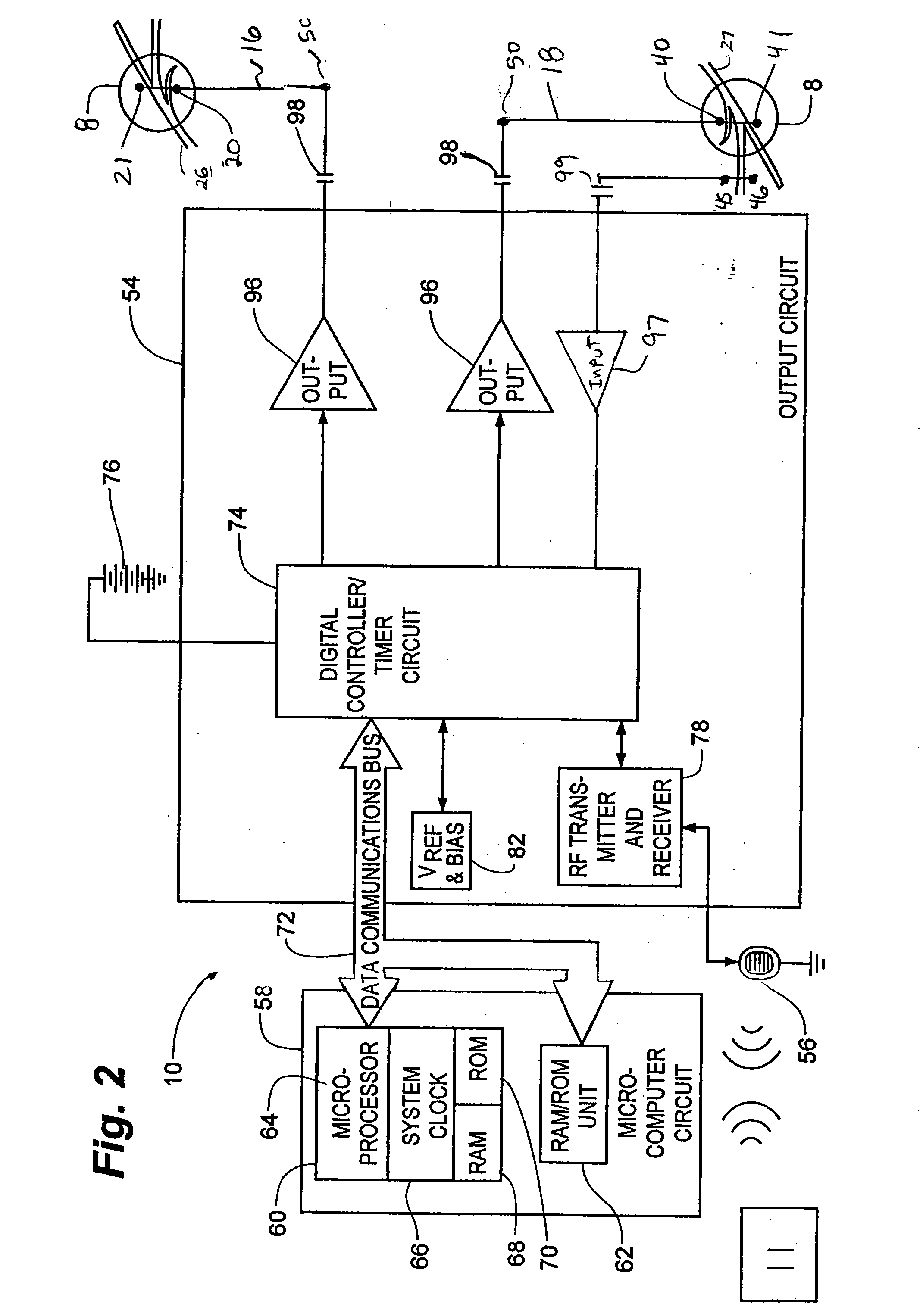
Described are implantable devices and methods for treating various disorders of the pelvic floor by means of electrical stimulation of the pudendal or other nerves, and optional means for delivering drugs in association therewith. A method of precisely positioning and implanting a medical electrical lead so as to provide optimal stimulation of the pudendal nerve or a portion thereof is also described. Placement of a stimulation lead next to or on the pudendal nerve may be performed using conventional prior art techniques through gross anatomical positioning, but usually does not result in truly optimal lead placement. One method of the present invention utilizes neurophysiological monitoring to assess the evoked responses of the pudendal nerve, and thereby provide a method for determining the optimal stimulation site. Additionally, one or more electrical stimulation signals are applied, and optionally one or more drugs are infused, injected or otherwise administered, to appropriate portions of a patient's pelvic floor and pudendal nerve or portions thereof in an amount and manner effective to treat a number of disorders, including, but not limited to, urinary and / or fecal voiding dysfunctions such as constipation, incontinence disorders such as urge frequency and urinary retention disorders, sexual dysfunctions such as orgasmic and erectile dysfunction, pelvic pain, prostatitis, prostatalgia and prostatodynia.
Owner:MEDTRONIC INC
Method, system and device for treating various disorders of the pelvic floor by electrical stimulation of the left and right pudendal nerves
InactiveUS20040193228A1Directly and effectively stimulatingUndesirable side effects of sacral nerve stimulation may be avoided or minimizedElectrotherapyDiseaseProstatalgia
Described are implantable devices and methods for treating various disorders of the pelvic floor by means of electrical stimulation of the pudendal and sacral nerves, or portions thereof, and optional means for delivering drugs in association therewith. Two or more electrical stimulation regimes are applied on a continuous, alternating, intermittent or other basis to the sacral and pudendal nerves, and optionally one or more drugs are infused, injected or otherwise administered, to appropriate portions of a patient's pelvic floor and pudendal nerve and / or sacral nerve, or portions thereof, in an amount and manner effective to treat a number of disorders, including, but not limited to, urinary and / or fecal voiding dysfunctions such as constipation, incontinence disorders such as urge frequency and urinary retention disorders, sexual dysfunctions such as orgasmic and erectile dysfunction, pelvic pain, prostatitis, prostatalgia and prostatodynia.
Owner:GERBER MARTIN T
Wetness monitoring system
InactiveUS7250547B1Reduce operating costsLower annual operating costsBaby linensAlarmsData acquisitionMonitoring system
The present invention relates to a wetness monitoring system that includes a data collection device that sends wetness measurement data to a central computer that detects changes in wetness measurement data caused by the presence of urine or other dielectric fluids. The data collection device includes a semi-reusable sensor and reusable data collector that are worn on a garment of the person. The data collector includes an internal power source so that the person can live a normal ambulatory life. The data collector has an electrical circuit that uses the changing resistance characteristics in the sensor to gather wetness measurement data. The data collector periodically generates and transmits a signal containing the actual wetness measurement data. The signals are coded to identify the particular data collector or person sending the signal. The data collector is programmed to conserve power by sending signals less frequently during periods when the sensor is clearly dry. Signals are sent more frequently when the sensor is damp or a wetness event may have occurred. The central computer receives the signals containing the wetness measurement data and compares the measurement data to an adjustable wetness sensitivity level to determine if a wetness event has occurred. When the central computer determines that a wetness event has occurred, the computer displays the name of the particular person wearing the data collector and the approximate time that the wetness event occurred. The system then pages an appropriate healthcare worker to inform them that the particular individual needs attention and tracks the approximate response times to ensure that the patient is continuously receiving prompt care.
Owner:RF TECH
Process for discriminating between biological states based on hidden patterns from biological data
The invention describes a process for determining a biological state through the discovery and analysis of hidden or non-obvious, discriminatory biological data patterns. The biological data can be from health data, clinical data, or from a biological sample, (e.g., a biological sample from a human, e.g., serum, blood, saliva, plasma, nipple aspirants, synovial fluids, cerebrospinal fluids, sweat, urine, fecal matter, tears, bronchial lavage, swabbings, needle aspirantas, semen, vaginal fluids, pre-ejaculate.), etc. which is analyzed to determine the biological state of the donor. The biological state can be a pathologic diagnosis, toxicity state, efficacy of a drug, prognosis of a disease, etc. Specifically, the invention concerns processes that discover hidden discriminatory biological data patterns (e.g., patterns of protein expression in a serum sample that classify the biological state of an organ) that describe biological states.
Owner:ASPIRA WOMENS HEALTH INC +1
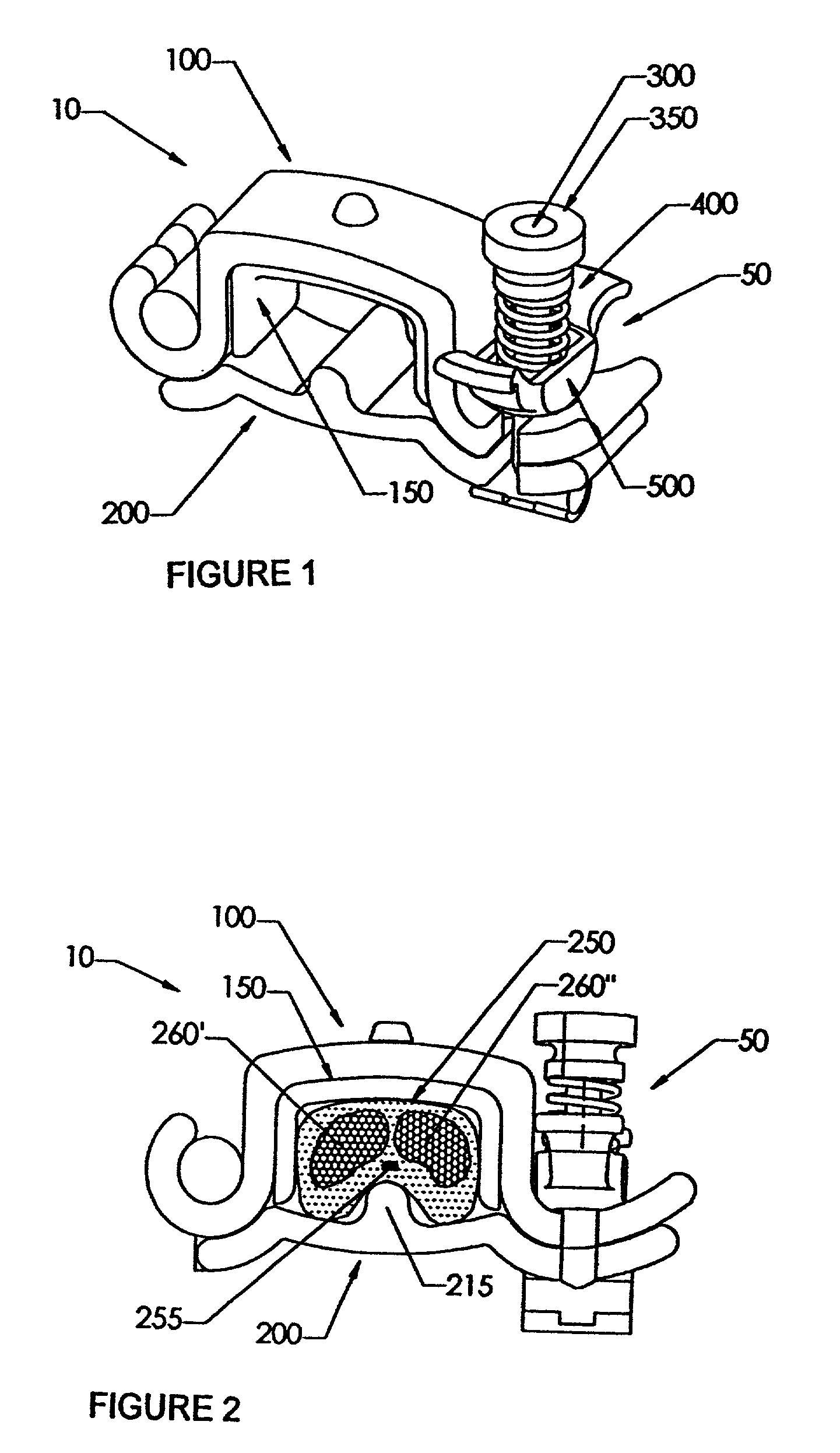
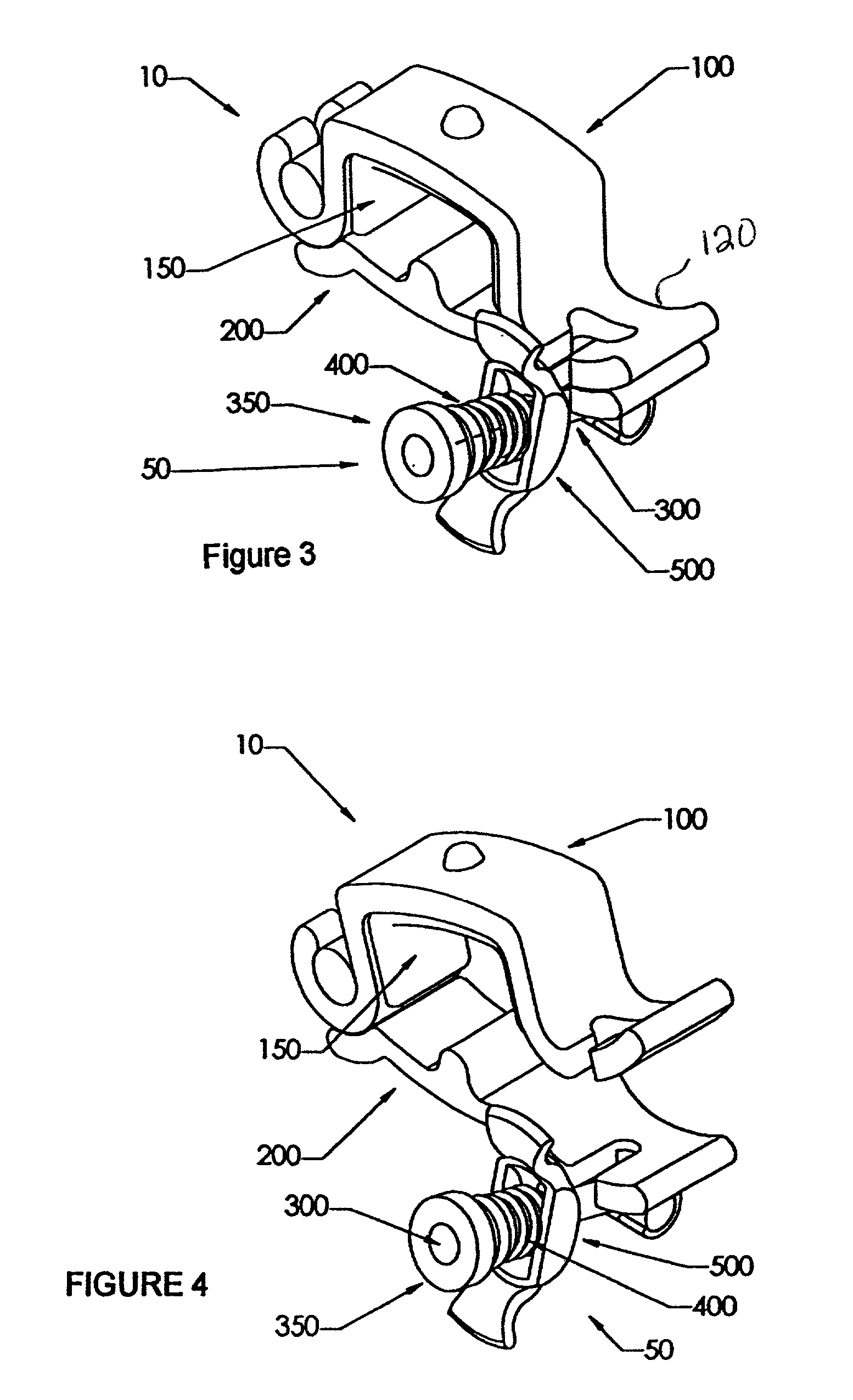
Urological device for the incontinent male
The present invention features a device to prevent the unwanted flow of urine in incontinent human males. This penile clamp is designed to provide an external pressure applied to the underside of the penis at the base thereof to close the urethral canal. The apparatus includes a rigid projection on the lower member of the device that applies a concentrated force on the canal. The upper bar of the device provides an attachment for a pad as an integral part of an easily disinfected clamp that is hinged at one end and provided with an adjustable tensioning closure device at its other end. The closure device is easily removable to allow one hand release of the penis restriction. A screw-type adjustment is easily set and is easily disassembled by a quick-release apparatus. While the clamp can be closed over the penis of an incontinent male to prevent urination, it does not appreciably restrict blood flow therein.
Owner:BIOENG CONSULTANTS LTD
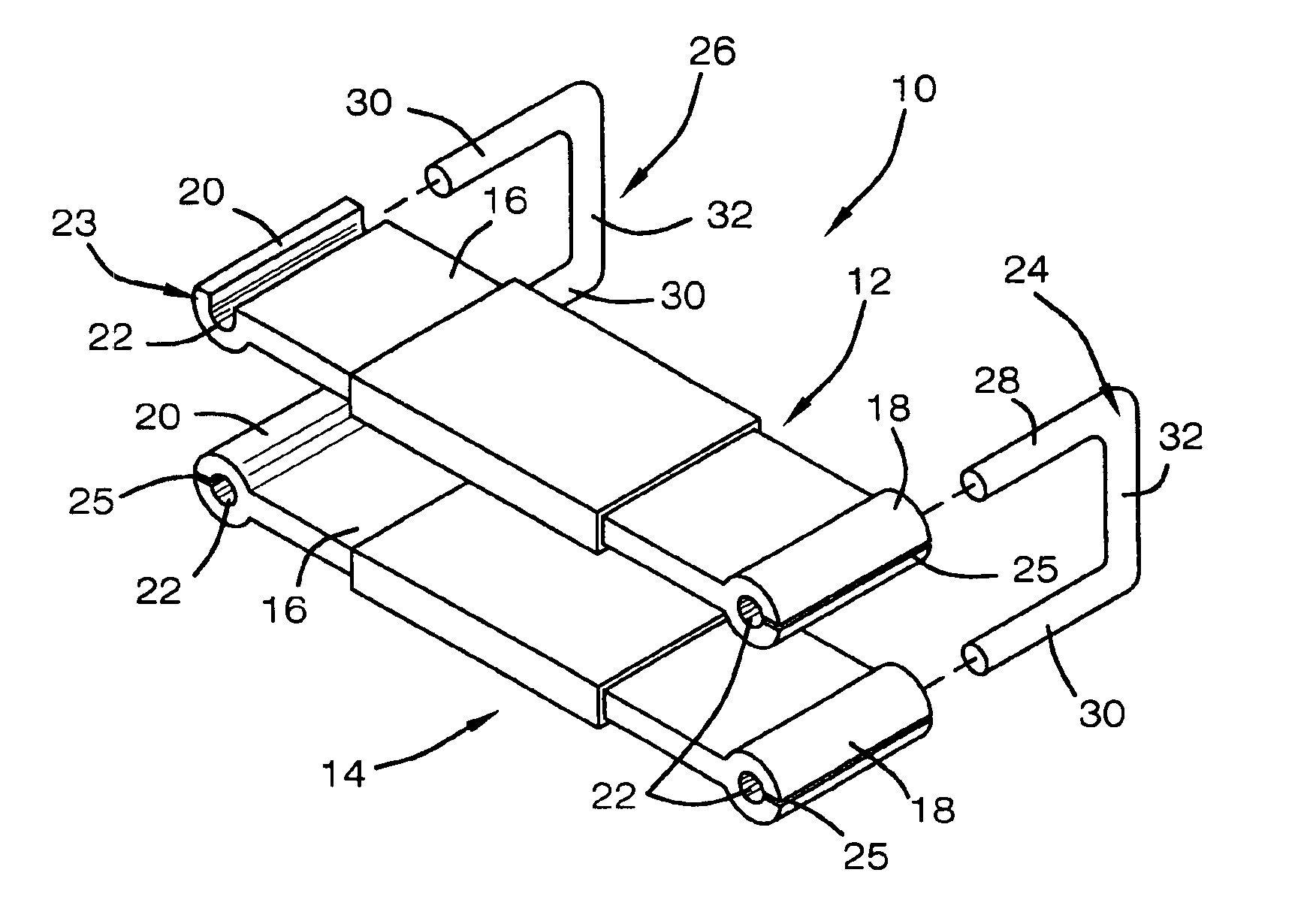
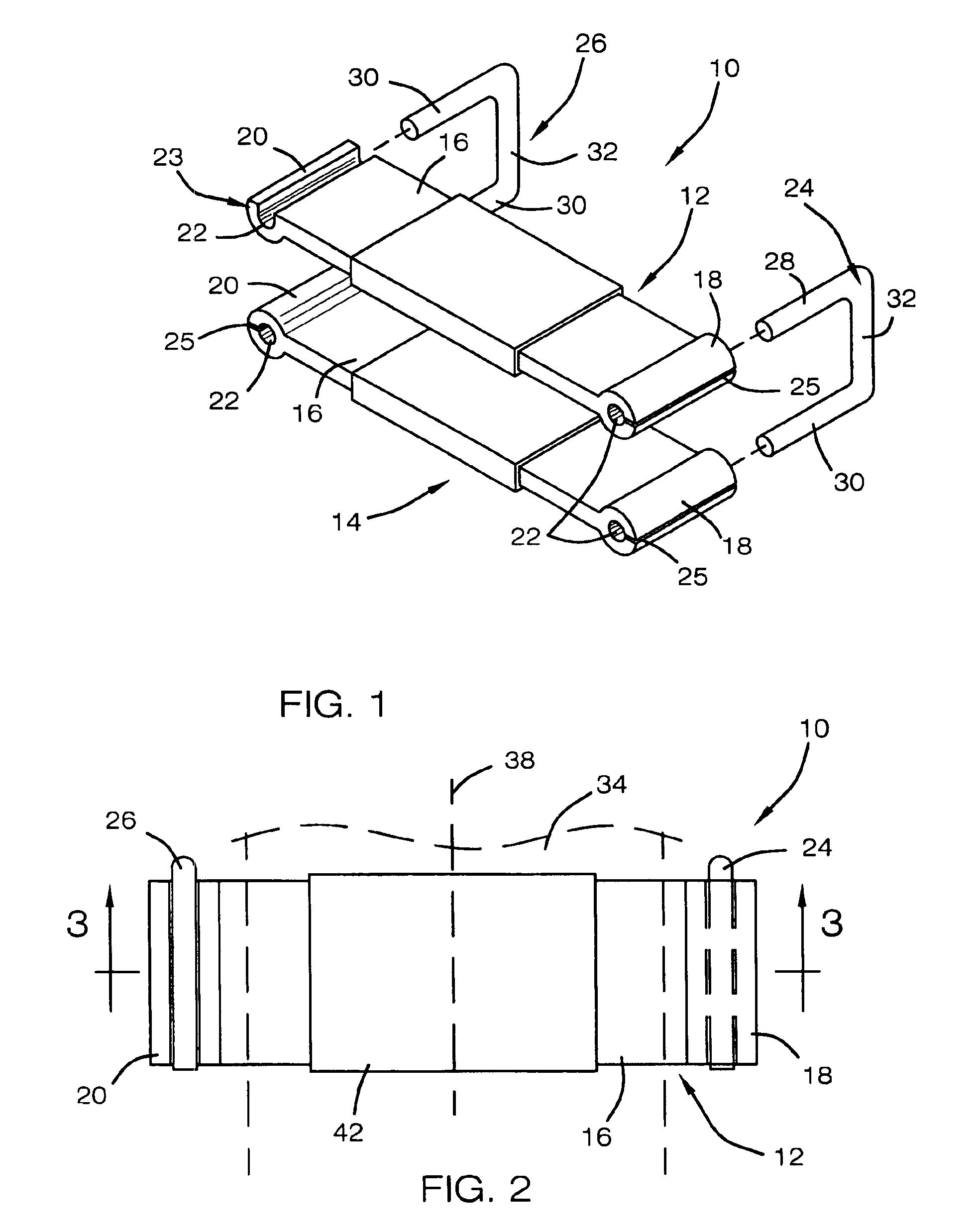
External incontinence clamp
InactiveUS6960218B2Improve wearing comfortEliminate slippageSuture equipmentsMale contraceptivesPenisEngineering
An external incontinence clamp for stopping involuntary voiding of urine is provided. The clamp includes a first rigid member having opposed ends, each end defining a passage extending therethrough, a second rigid member having opposed ends, each end defining a passage extending therethrough, and two pins of generally u-shape. The pins are adapted to be received by the passages defined by the ends of the first rigid member and of the second rigid member so as to secure the first rigid member in a spaced and generally parallel relationship to the second rigid member. The penis is positioned between the first and second members so that the underside of the penis is in contact with the second member and the top surface of the penis is in contact with the first member, thereby stopping involuntary voiding of urine. A clamp kit includes a plurality of pins of different width to vary the force applied to the penis.
Owner:RENNICH HENRY
Method, system and device for treating disorders of the pelvic floor by means of electrical stimulation of the pudenal and associated nerves, and the optional delivery of drugs in association therewith
ActiveUS20050113877A1Reduce traumaAvoid damageDigestive electrodesArtificial respirationDiseaseProstatalgia
Described are implantable devices and methods for treating various disorders of the pelvic floor by means of electrical stimulation of the pudendal or other nerves, and optional means for delivering drugs in association therewith. A method of precisely positioning and implanting a medical electrical lead so as to provide optimal stimulation of the pudendal nerve or a portion thereof is also described. Placement of a stimulation lead next to or on the pudendal nerve may be performed using conventional prior art techniques through gross anatomical positioning, but usually does not result in truly optimal lead placement. One method of the present invention utilizes neurophysiological monitoring to assess the evoked responses of the pudendal nerve, and thereby provide a method for determining the optimal stimulation site. Additionally, one or more electrical stimulation signals are applied, and optionally one or more drugs are infused, injected or otherwise administered, to appropriate portions of a patient's pelvic floor and pudendal nerve or portions thereof in an amount and manner effective to treat a number of disorders, including, but not limited to, urinary and / or fecal voiding dysfunctions such as constipation, incontinence disorders such as urge frequency and urinary retention disorders, sexual dysfunctions such as orgasmic and erectile dysfunction, pelvic pain, prostatitis, prostatalgia and prostatodynia.
Owner:MEDTRONIC INC
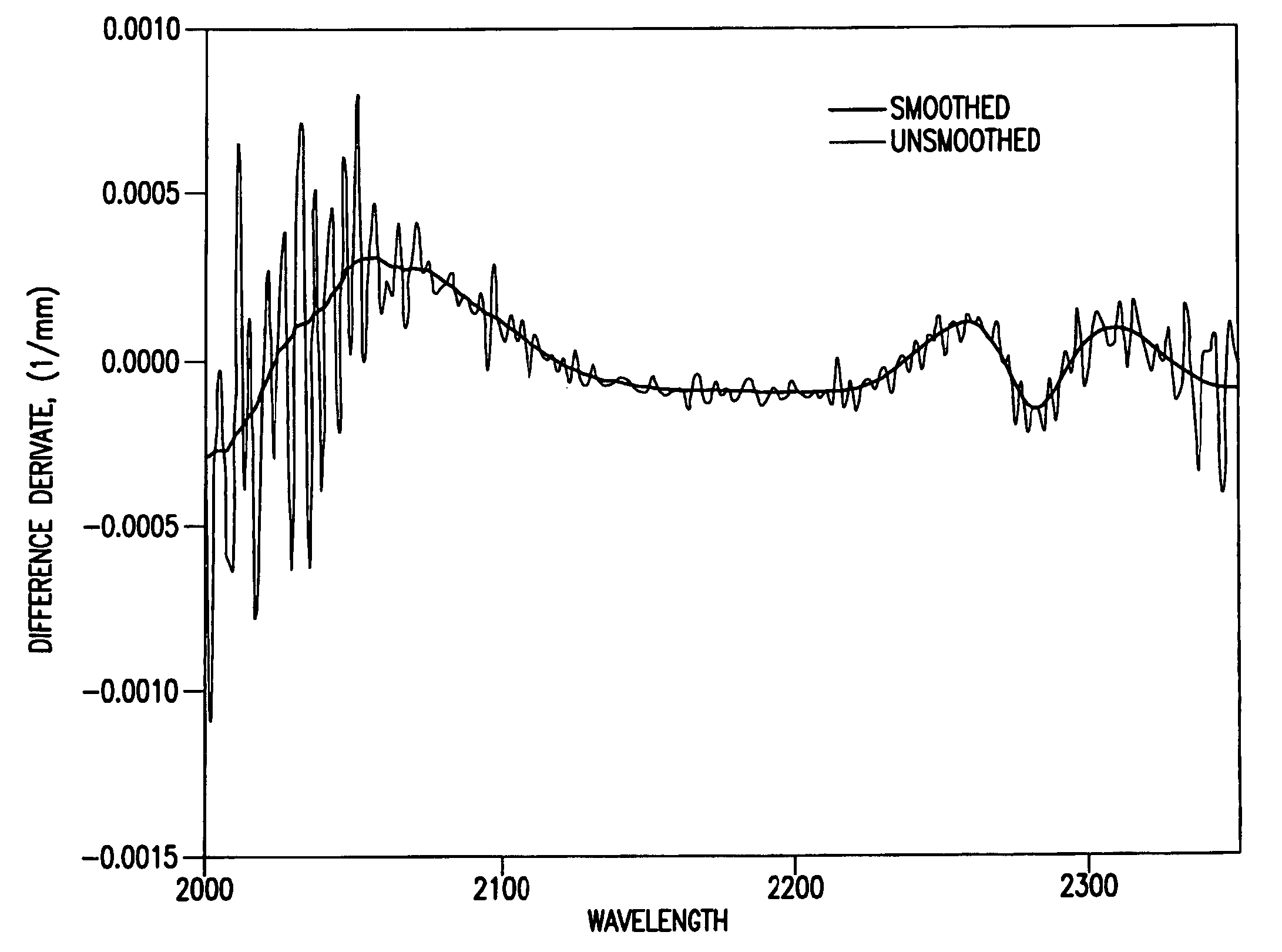
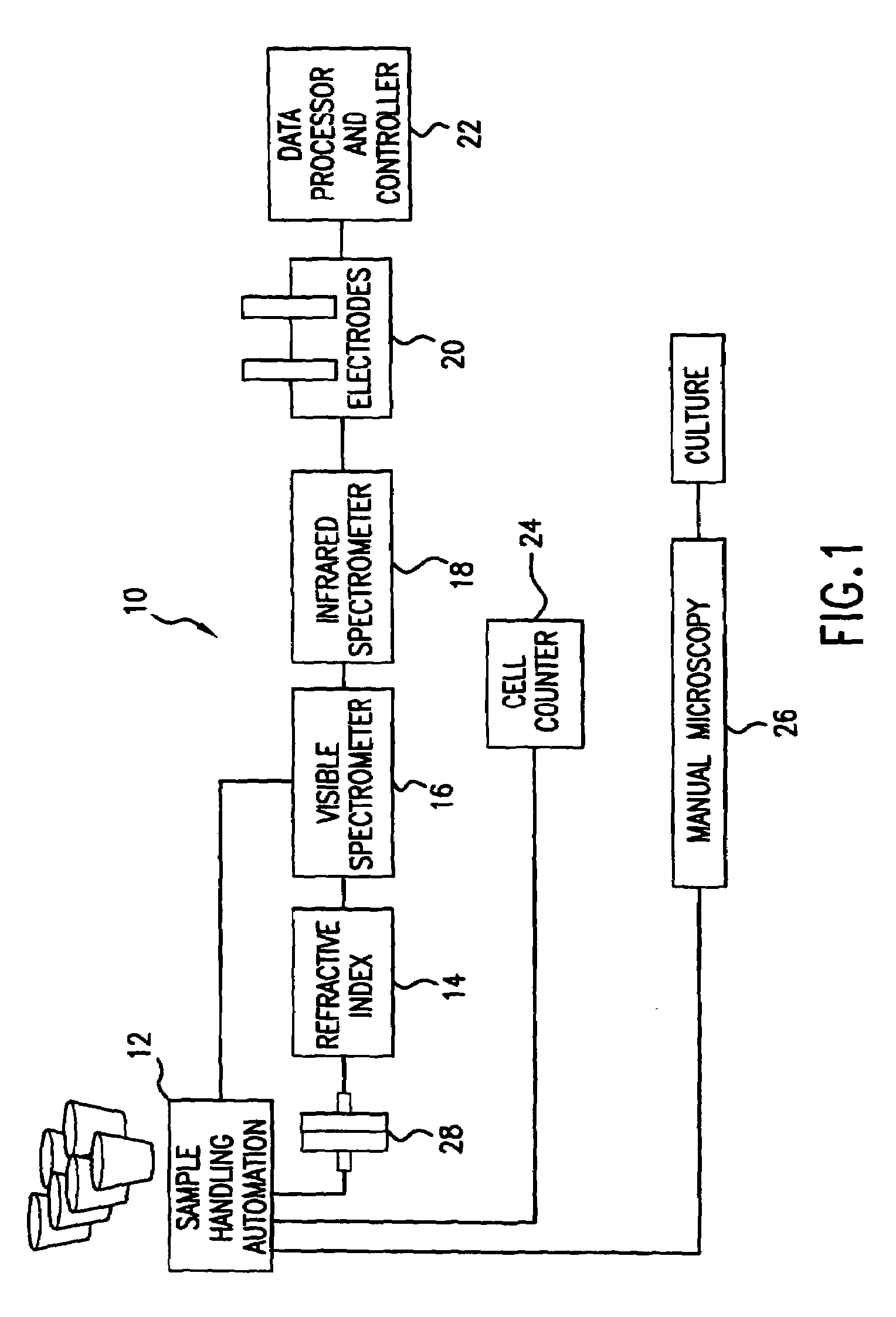
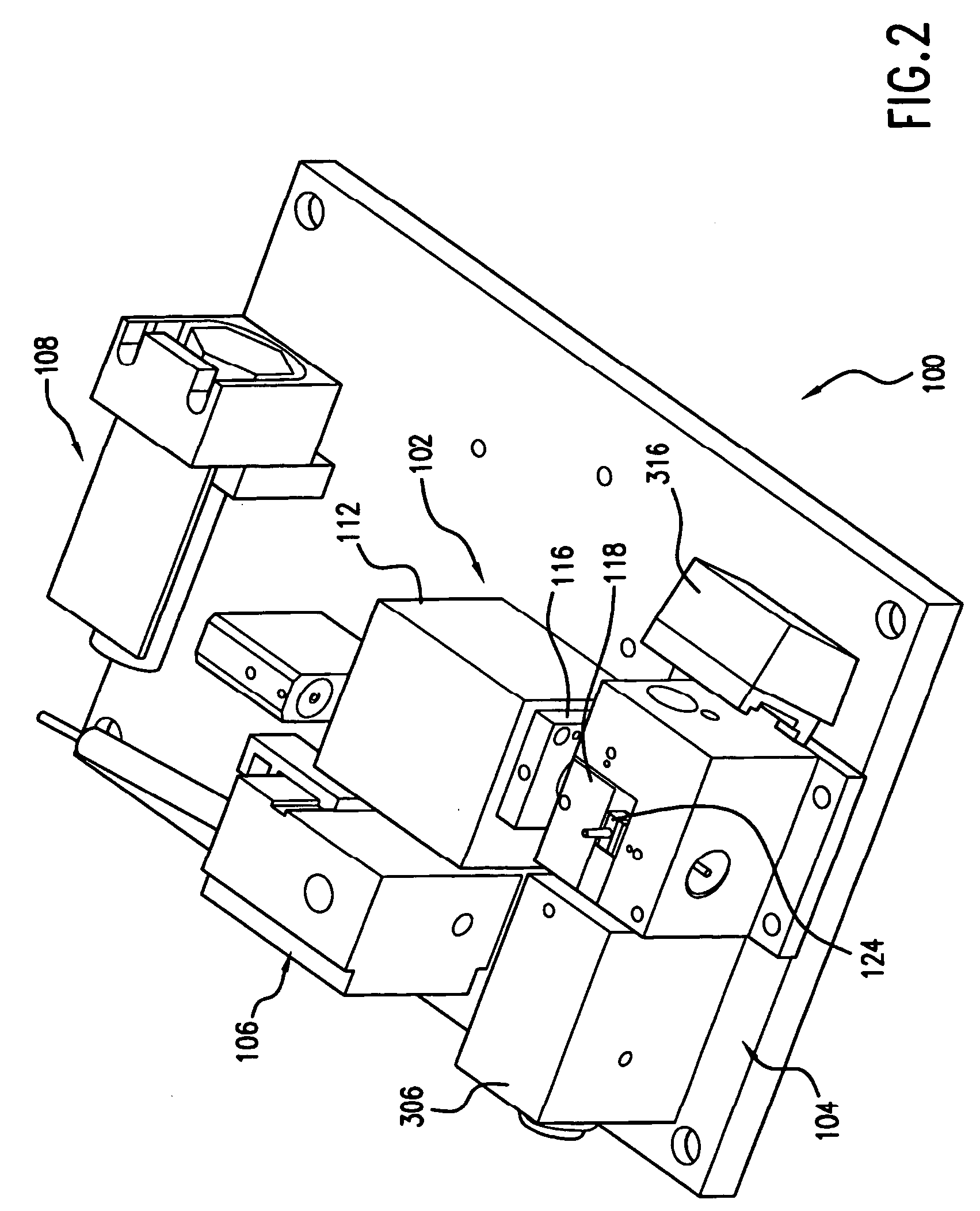
Reagentless analysis of biological samples by applying mathematical algorithms to smoothed spectra
InactiveUS7303922B2Improve accuracyImprove automationPhase-affecting property measurementsRaman scatteringCreatinine riseRefractive index
Apparatus and method for determining at least one parameter, e. g., concentration, of at least one analyte, e. g., urea, of a biological sample, e. g., urine. A biological sample particularly suitable for the apparatus and method of this invention is urine. In general, spectroscopic measurements can be used to quantify the concentrations of one or more analytes in a biological sample. In order to obtain concentration values of certain analytes, such as hemoglobin and bilirubin, visible light absorption spectroscopy can be used. In order to obtain concentration values of other analytes, such as urea, creatinine, glucose, ketones, and protein, infrared light absorption spectroscopy can be used. The apparatus and method of this invention utilize one or more mathematical techniques to improve the accuracy of measurement of parameters of analytes in a biological sample. The invention also provides an apparatus and method for measuring the refractive index of a sample of biological fluid while making spectroscopic measurements substantially simultaneously.
Owner:ABBOTT LAB INC
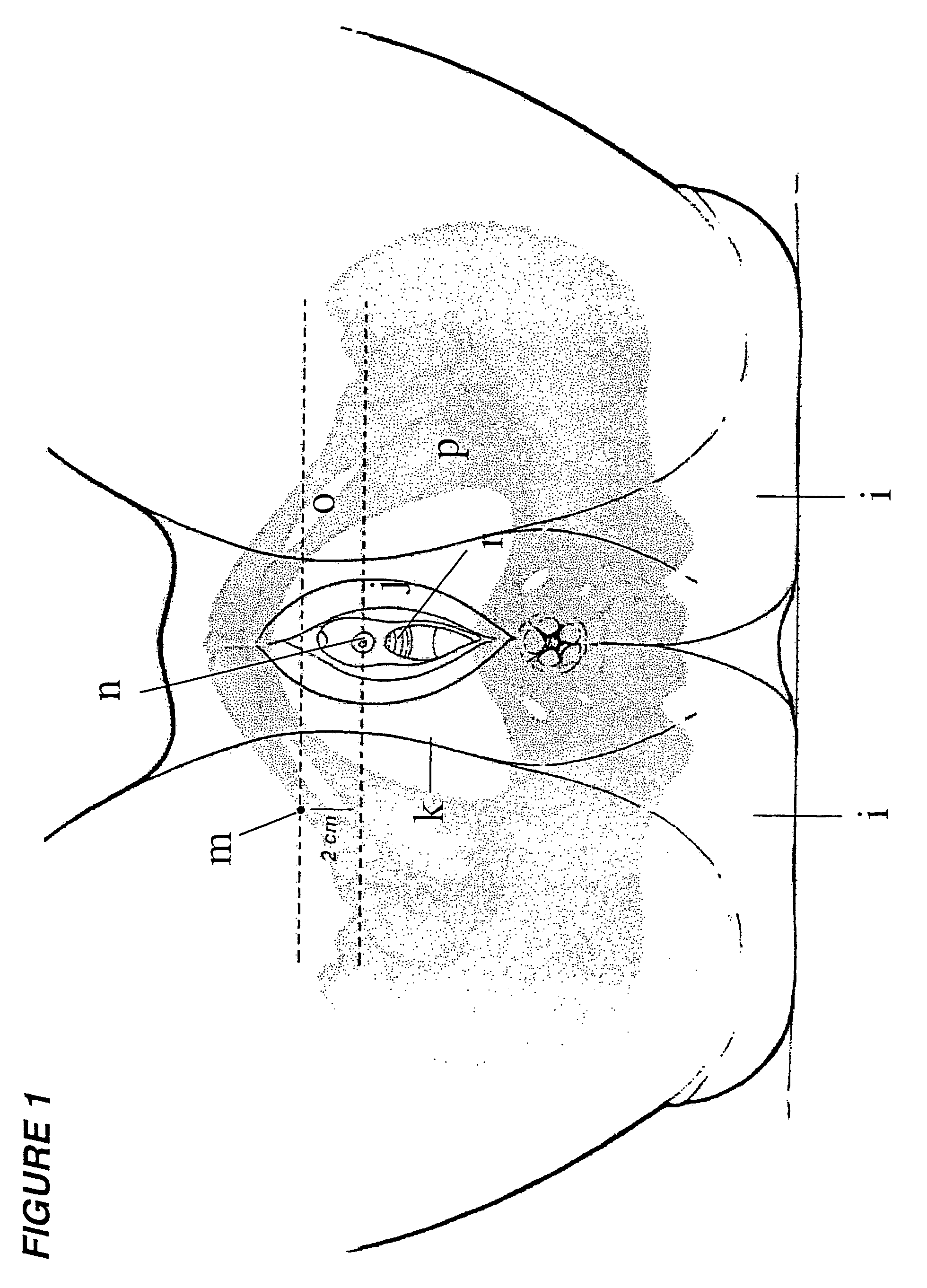
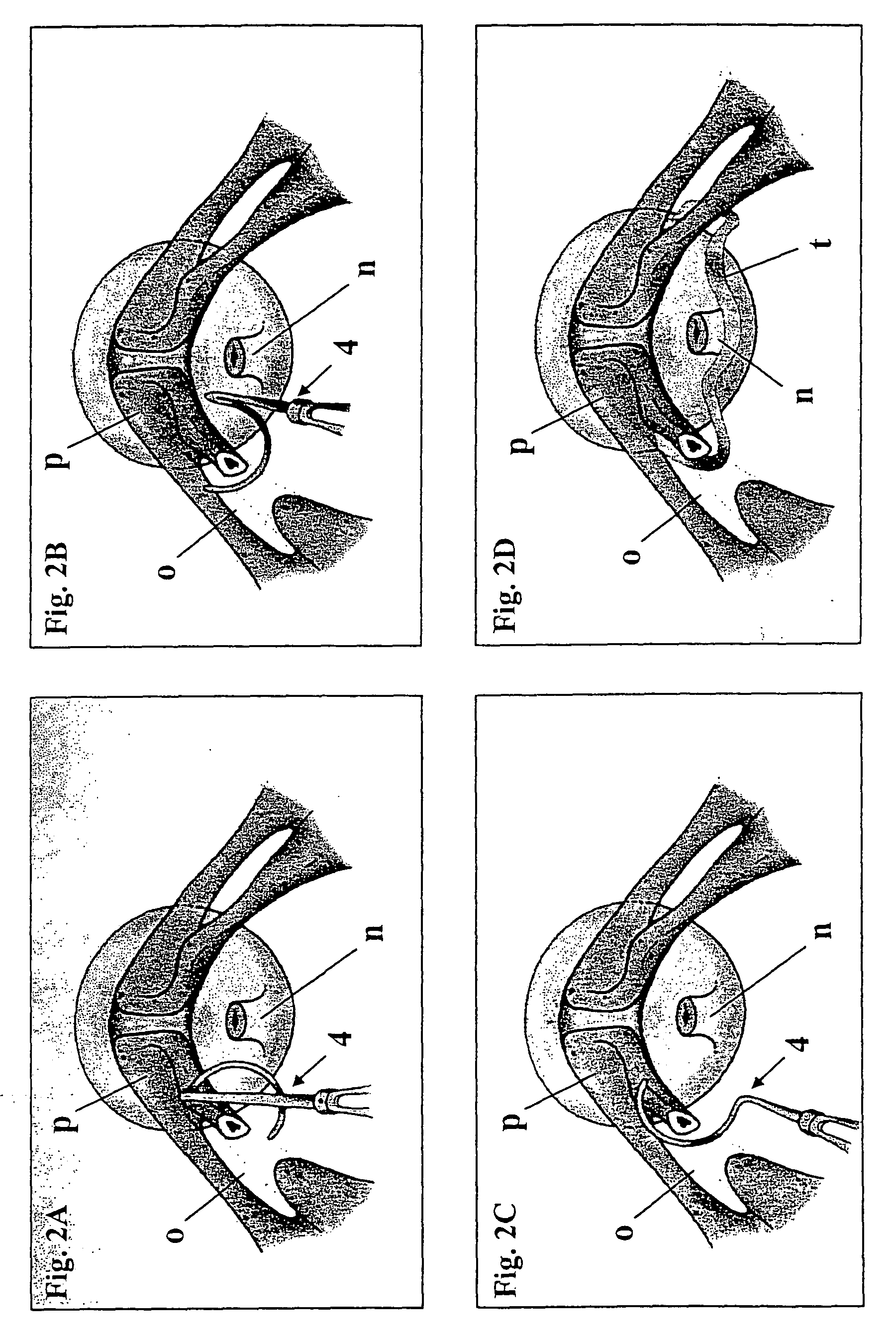
Surgical procedure for the treatment of female urinary incontinence: tension-free inside-out transobturator urethral suspension
A new, quick, simple, efficient, safe, and reproducible surgical technique for the treatment of female urinary incontinence, in which the posterior urethra is suspended using a tape passed through the obturator orifices from inside (underneath the urethra) to outside (thigh folds). A variety of specifically designed surgical instruments are utilized to perform this operative procedure.
Owner:UNIV LIEGE +1
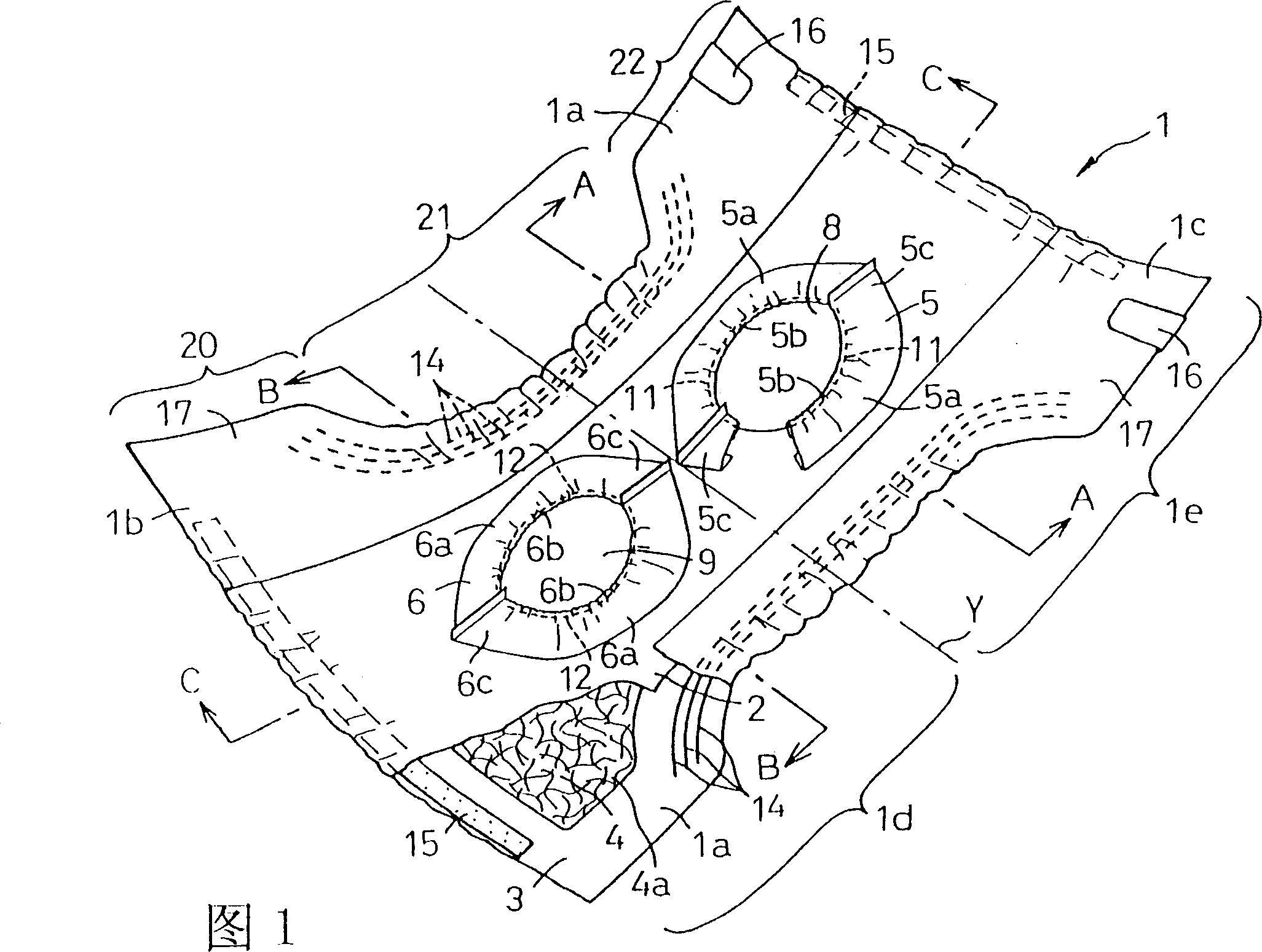
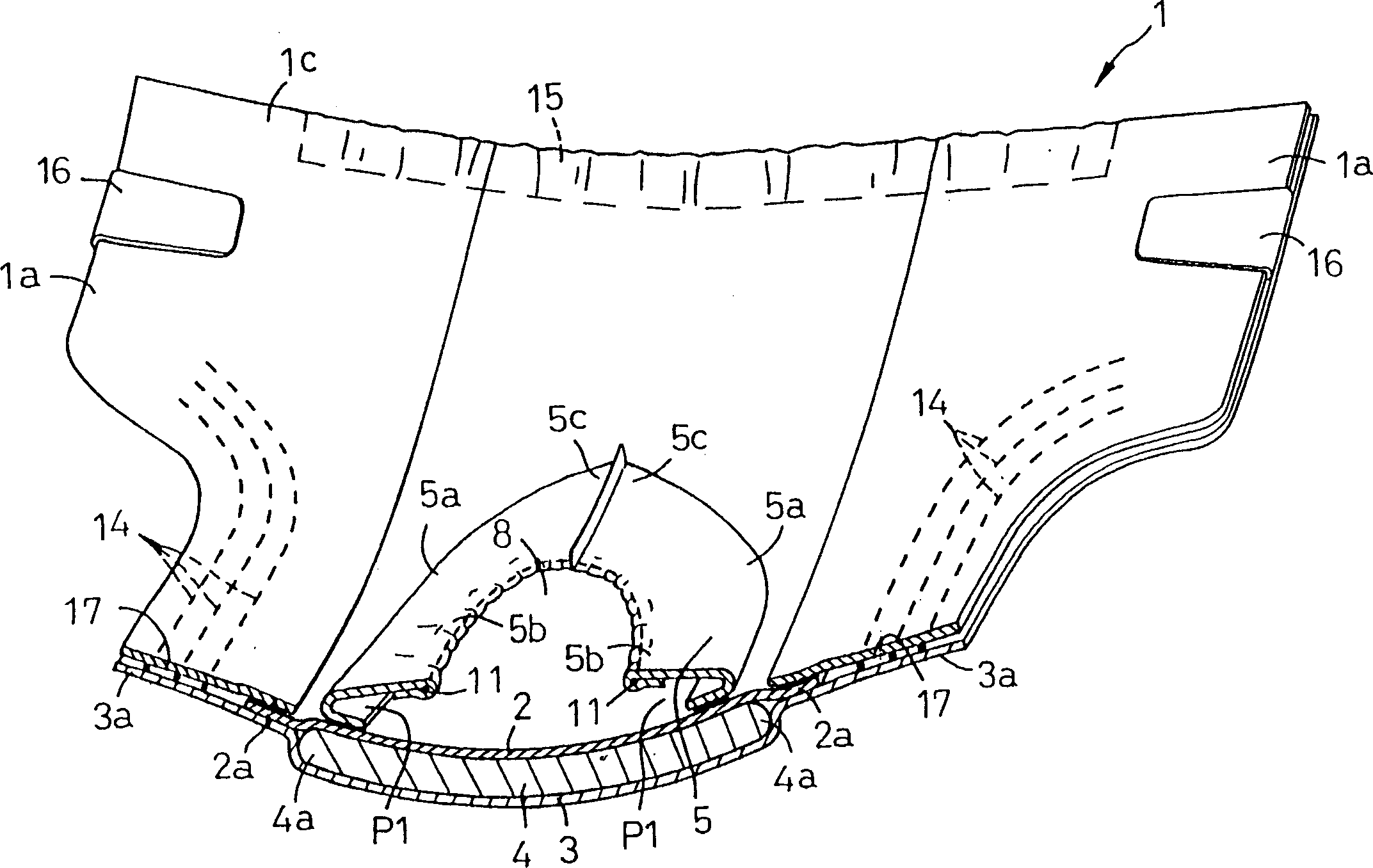
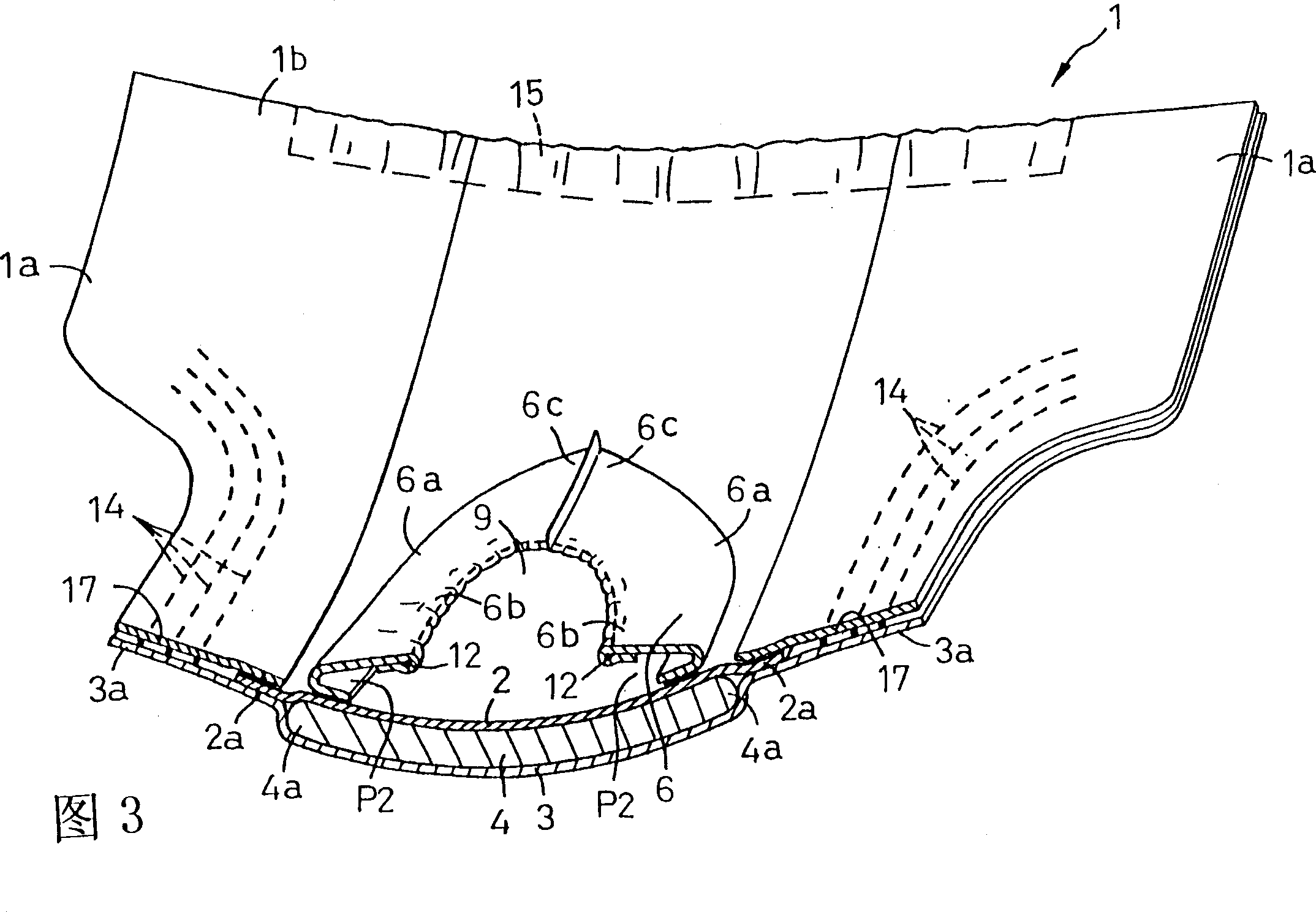
Disposable diaper
In order to provide a disposable diaper that can prevent excreted urine and feces from being mixed and avoid uncomfortableness. A diaper is constituted of a laminated panel made by interposing a core between a top sheet and a back sheet and providing a first leakage barrier sheet extending in an annular shape on an outer face of the top sheet, and has a first opening 8 in an approximately central part of the first leakage barrier sheet. The first leakage barrier sheet is located at a rear half part 1e of the panel and a second leakage barrier sheet 6 is located at a front half part 1d of the panel and a second opening is defined in an approximately central part of the second leakage barrier sheet.So that urine discharged into the second opening may be absorbed by the core through the topsheet and feces discharged into the first opening may be absorbed by the core through the topsheet.
Owner:UNI CHARM CORP
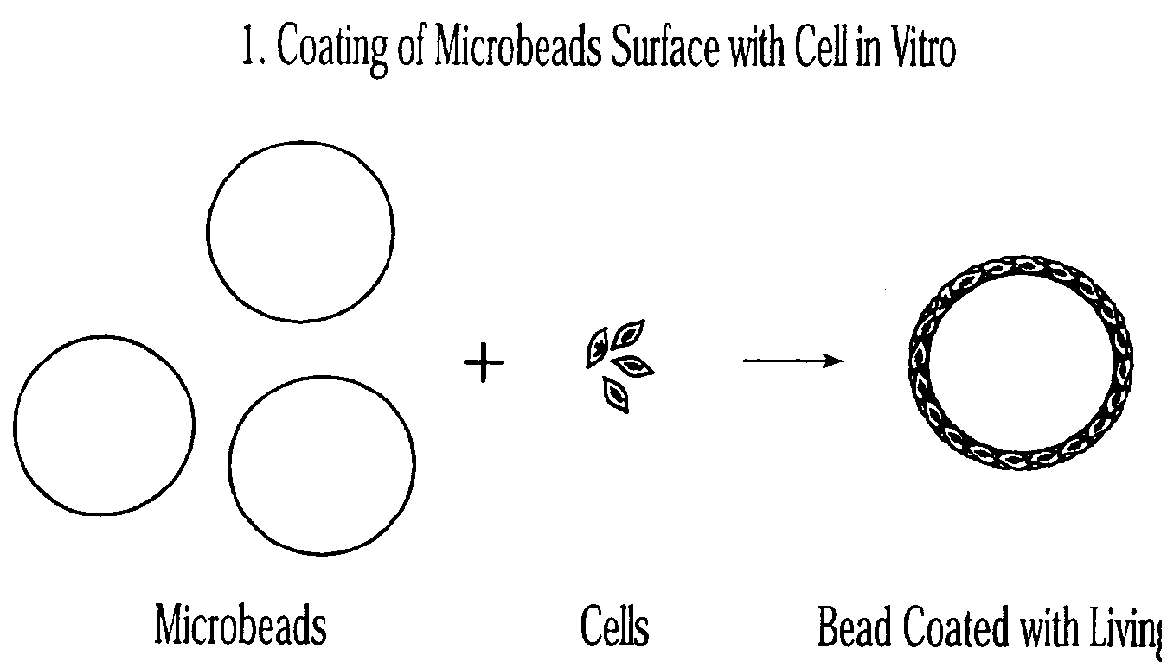
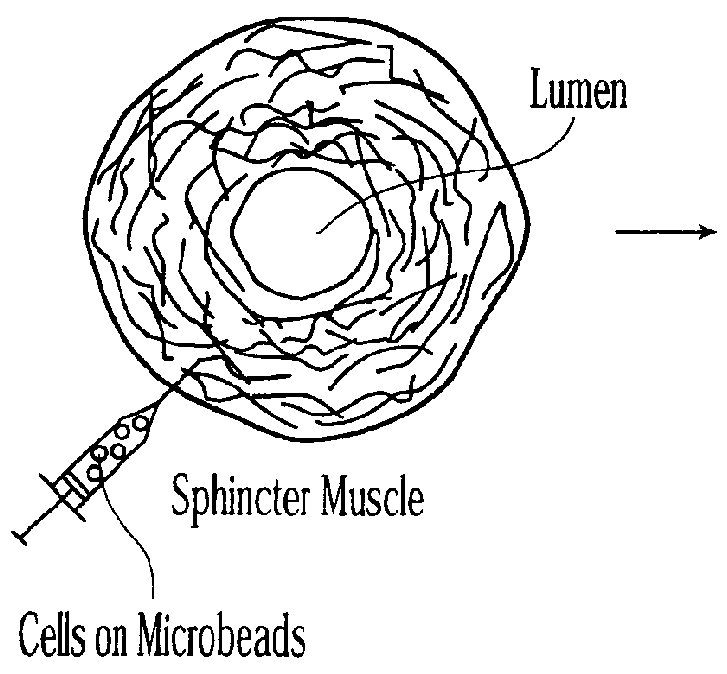
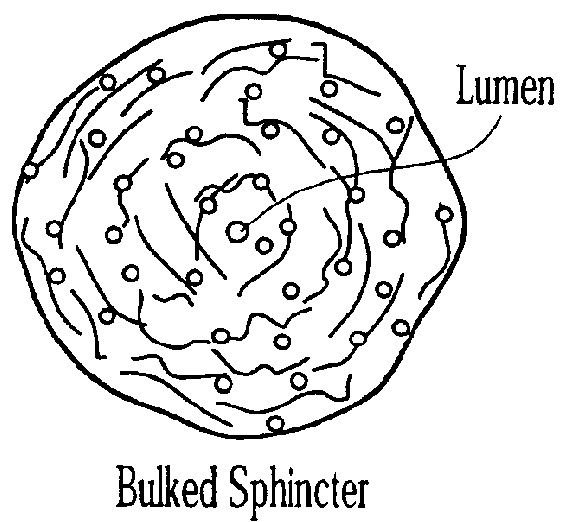
Injectable microspheres for dermal augmentation and tissue bulking
The present invention relates to elastic, hydrophilic and substantially spherical microspheres useful for dermal augmentation and tissue bulking. The invention provides injectable compositions comprising the microspheres and a biocompatible carrier for use in dermal augmentation. The present invention further provides methods of dermal augmentation and tissue bulking, particularly for the treatment of skin contour deficiencies, Gastro-esophageal reflux disease, urinary incontinence, and urinary reflux disease, using the injectable compositions.
Owner:BIOSPHERE MEDICAL INC
Surgical article and methods for treating female urinary incontinence
ActiveUS7303525B2Easy to disassembleReduce the amount requiredAnti-incontinence devicesSurgeryCo operativeSling procedure
A sling assembly including a surgical sling configured to be implanted during a surgical sling procedure. The sling includes first and second regions and a central portion. The sling assembly further includes a removable sheath assembly situated about the surgical sling. The removable sheath assembly includes first and second upper sheaths. The first upper sheath is configured to be situated about the first region of the surgical sling, and the second upper sheath is configured to be situated about the second region of the surgical sling. The removable sheath assembly further includes a lower sheath. The lower sheath is configured to be situated about the central portion of the surgical sling and to be in cooperative association with both the first and second upper sheaths.
Owner:ASTORA WOMENS HEALTH
Surgical procedure for the treatment of female urinary incontinence: tension-free inside-out transobturator urethral suspension
A new, quick, simple, efficient, safe, and reproducible surgical technique for the treatment of female urinary incontinence, in which the posterior urethra is suspended using a tape passed through the obturator orifices from inside (underneath the urethra) to outside (thigh folds). A variety of specifically designed surgical instruments are utilized to perform this operative procedure.
Owner:UNIV LIEGE +1
Outlet tube device for urinary drainage bag
InactiveUS6132407AReduce manufacturing costEasy to operateClosuresCheck valvesUrinary drainageResidual urine
An improved outlet tube device for a urinary drainage bag is disclosed wherein an outlet valve mechanism is contained with a rigid housing fixed securely to the drainage bag, rather than just being clamped to the outlet tubing, and thus cannot become separated from the bag. The outlet valve can be opened only by positive action on the part of the medical care personnel, thereby assuring that urine will be discharged only in a controlled manner. Further, because the valve mechanism does not require that a substantial length of tubing with a freely movable end be used in conjunction with the outlet port, there is no need to engage the free end of the tubing with a keeper. Thus the possibility of the tubing springing back to its straight configuration and flicking residual urine on the attending medical personnel is eliminated. In one embodiment the outlet tube device comprises a pinchcock-type valve which is fixedly mounted to the bag and which is normally operative to clamp off the outlet tube. In a second embodiment the outlet tube device comprises a syringetype fitting which is fixedly mounted to the bag. Depressing the syringe plunger closes the valve, and retracting the plunger opens the valve to permit fluid to be discharged through the outlet tube. In a third embodiment, a hose clamp is contained within a rigid housing and is selectively operative to clamp a hose to prevent fluid flow therethrough.
Owner:CR BARD INC
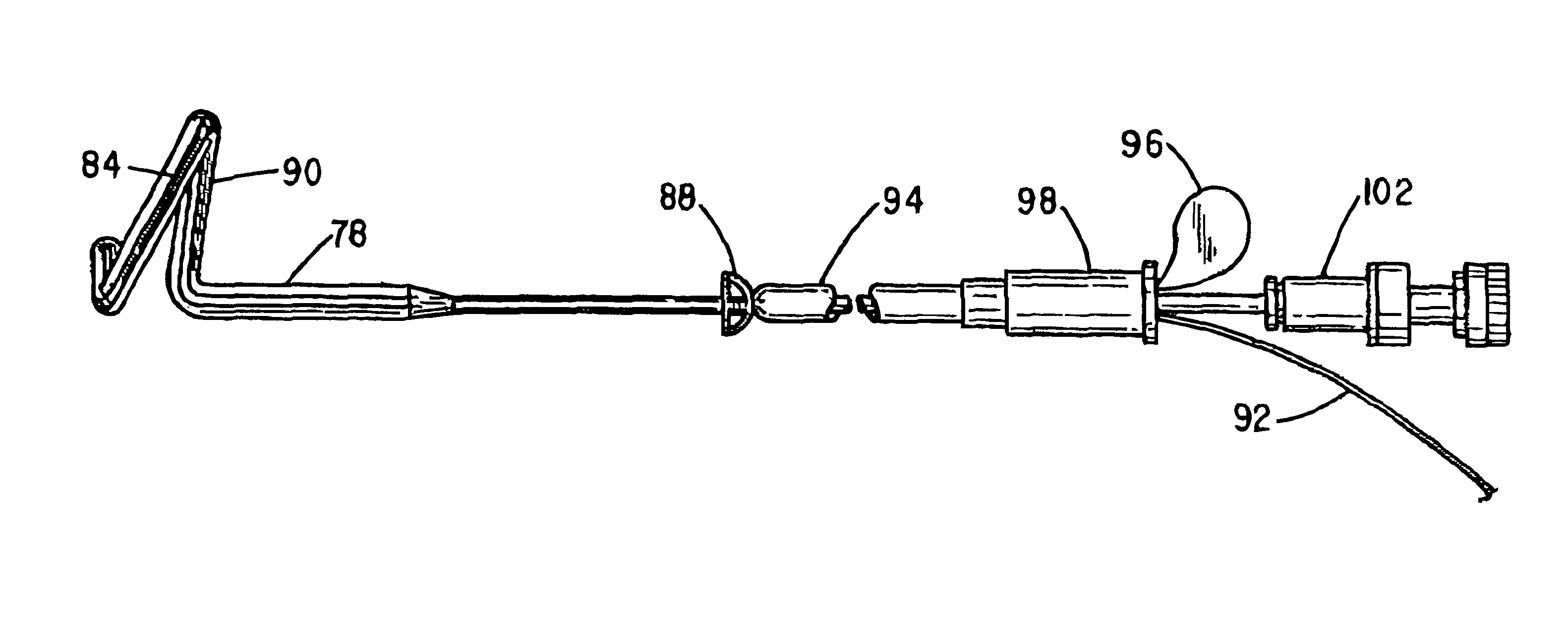
Pelvic disorder treatment device
ActiveUS7613516B2Relieving pelvic painReliable controlUltrasonic/sonic/infrasonic diagnosticsElectrotherapyInterstitial cystitisFecal incontinence
Owner:MEDTRONIC INC +1
Systems and methods for analyzing body fluids
ActiveUS20110070606A1Improve accuracyLow costBioreactor/fermenter combinationsBiological substance pretreatmentsBody fluidVaginal tissue
Systems and methods analyzing body fluids contain cells including blood, bone marrow, urine, vaginal tissue, epithelial tissue, tumors, semen, and spittle are disclosed. The systems and methods utilize an improved technique for applying a monolayer of cells to a slide and generating a substantially uniform distribution of cells on the slide. Additionally aspects of the invention also relate to systems and method for utilizing multi-color microscopy for improving the quality of images captured by a light receiving device.
Owner:ROCHE DIAGNOSTICS HEMATOLOGY INC
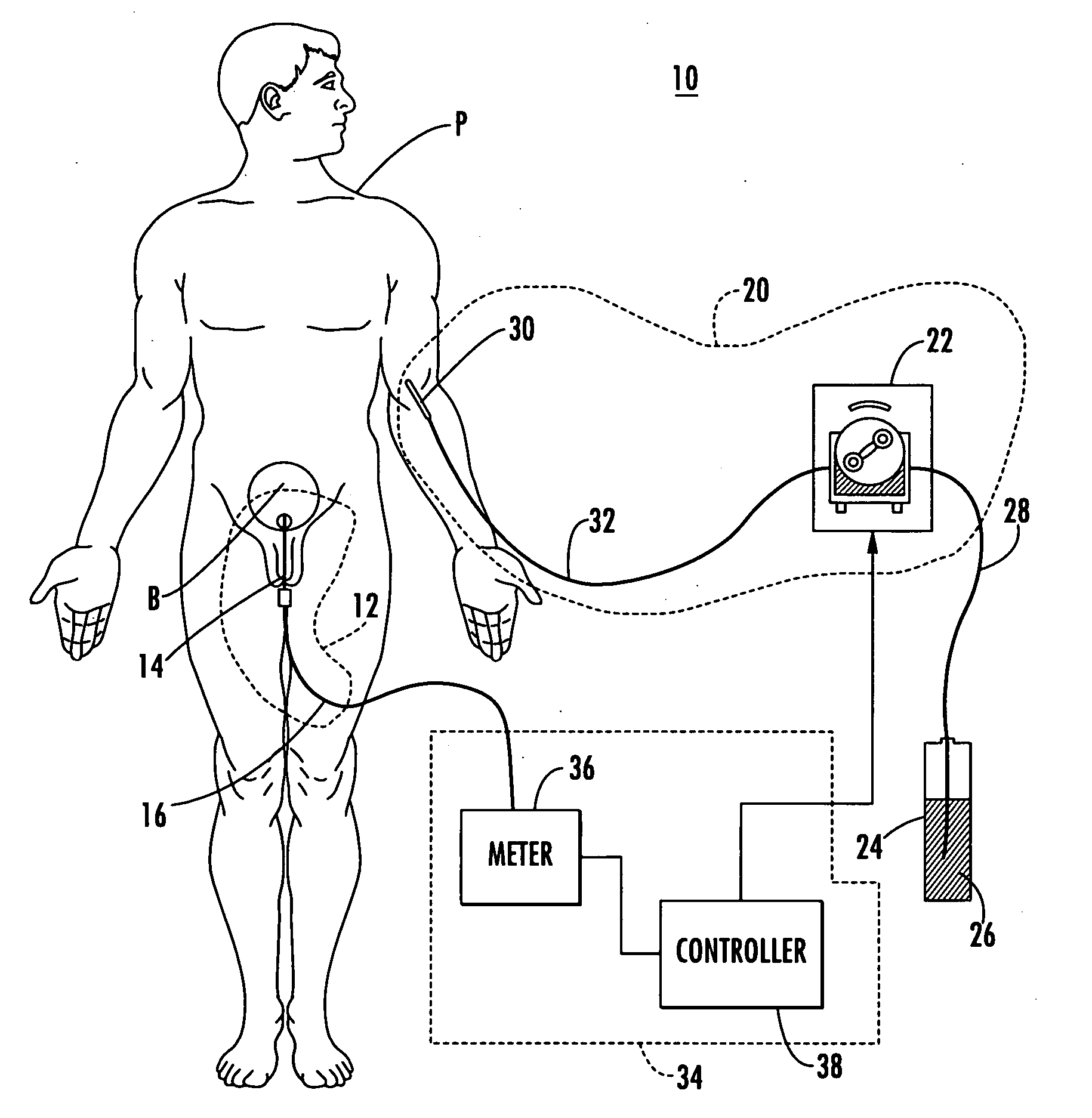
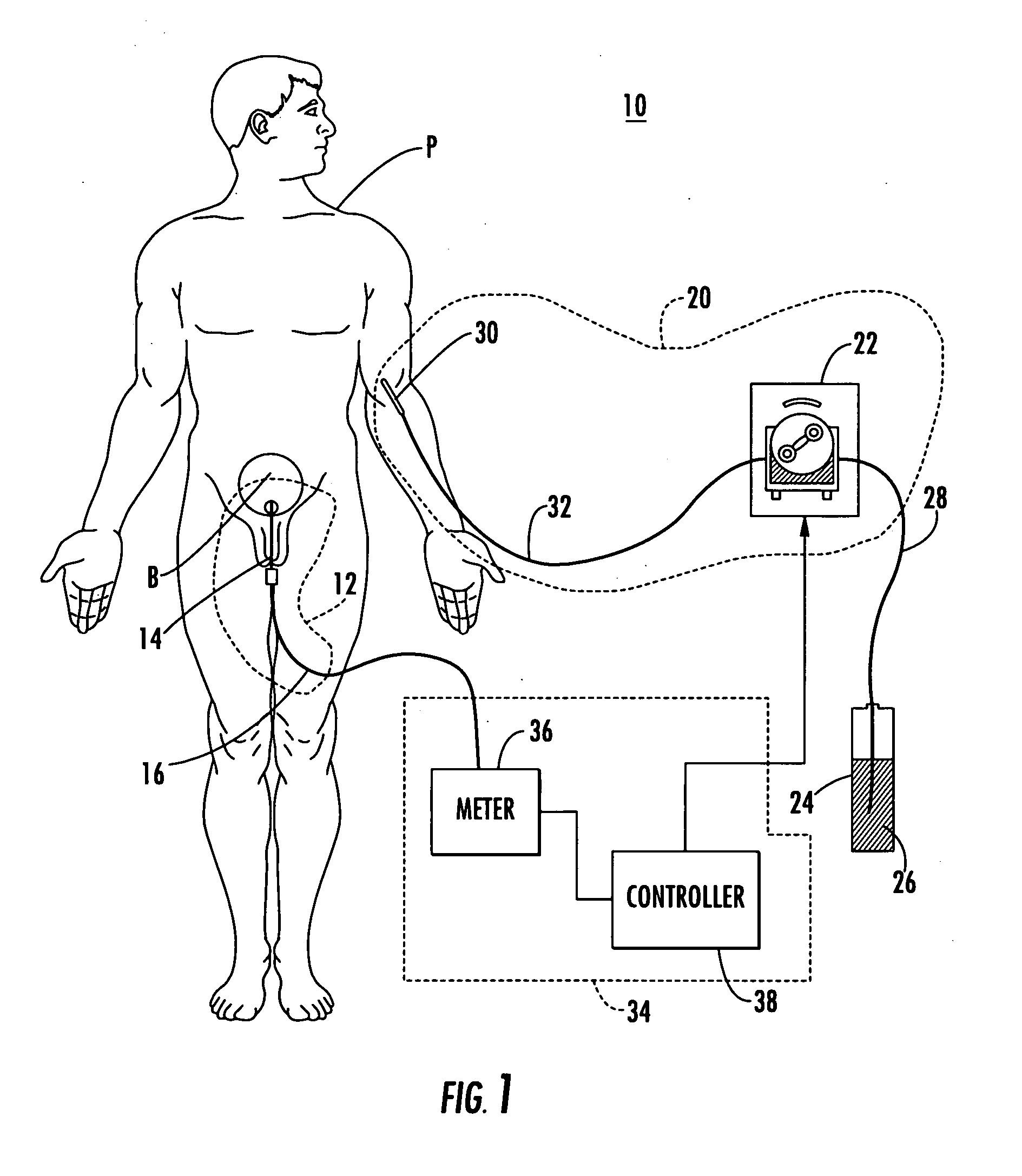
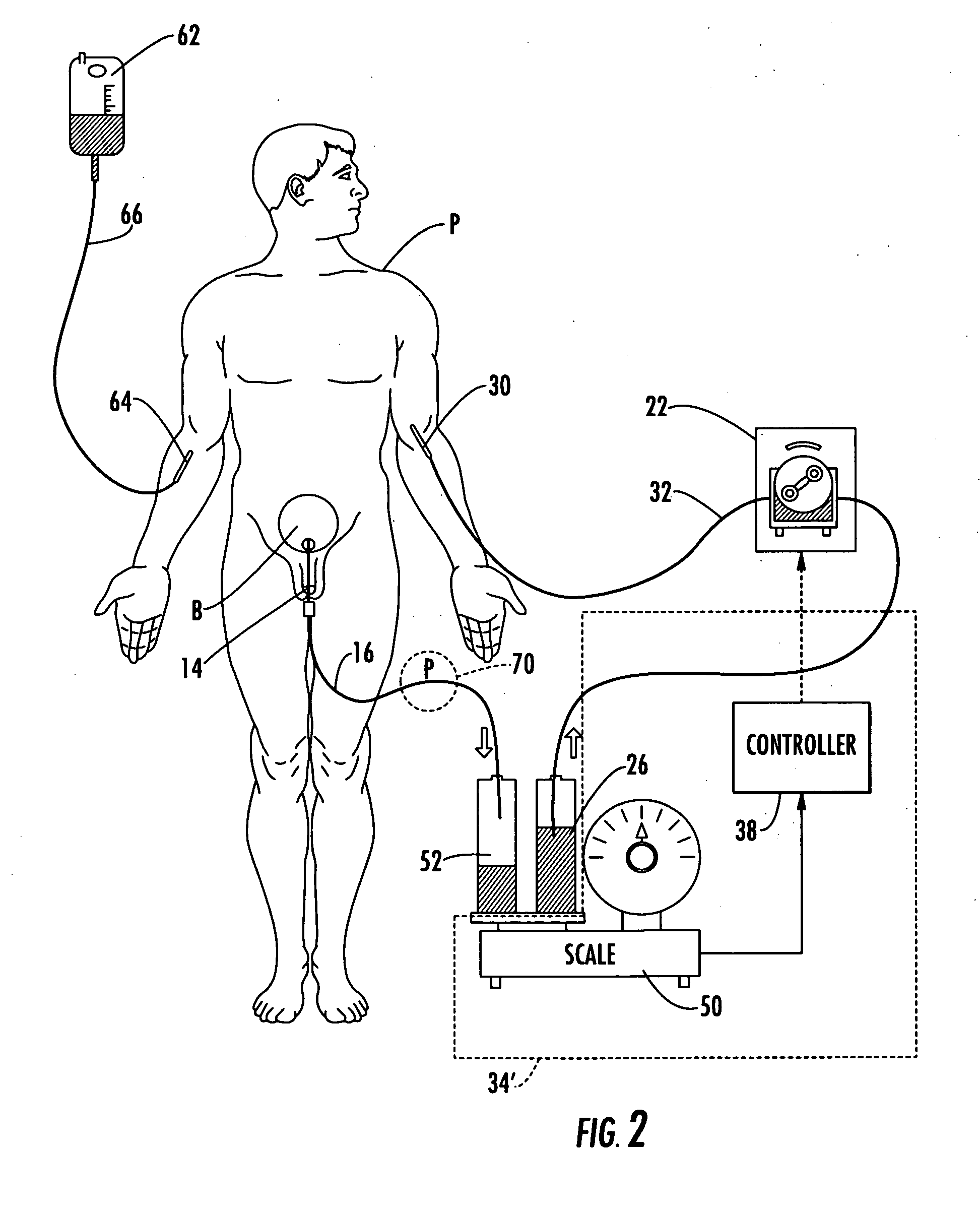
Patient hydration system with abnormal condition sensing
ActiveUS20060235353A1Prevent kidney damagePrevent dehydrationMedical devicesPressure infusionUrine outputIntensive care medicine
A patient hydration system and method wherein hydration fluid is administered to a patient and the patient's urine output is measured either by weight or some other suitable way. A controller is responsive to the patient's urine output measurement and configured to control the infusion rate to hydrate the patient based on the patient's urine output. The controller also detects abnormal patient urine output measurements and initiates corrective action in response.
Owner:MEDICAL SYST
System for monitoring the health of an individual and method for use thereof
InactiveUS20050261605A1Easy to installReadily attachableSurgeryVaccination/ovulation diagnosticsTelecommunications linkUrine
A medical analysis system for measuring properties and constituents of urine. The system includes a toilet unit mountable on a toilet bowl, readily attachable and detachable therefrom. The toilet unit includes a collection and sampling sub-unit for collecting and measuring the characteristics of urine and an electronics sub-unit for data collection and processing of the measured characteristics. The toilet unit also includes a communications link for data transfer between the collection and sampling and electronics sub-units. It also includes a means for removably fastening the collection and sampling sub-unit inside a toilet bowl, permitting ready attachment and removal of the toilet unit from a toilet bowl as desired. A wall unit, having a communications link for data transfer between the wall and toilet units, also includes means for storing and further processing the data for determining if changes have occurred in the measured characteristics of urine over time.
Owner:S A E AFIKIM COMPIZED DAIRY MANAGEMENT SYST
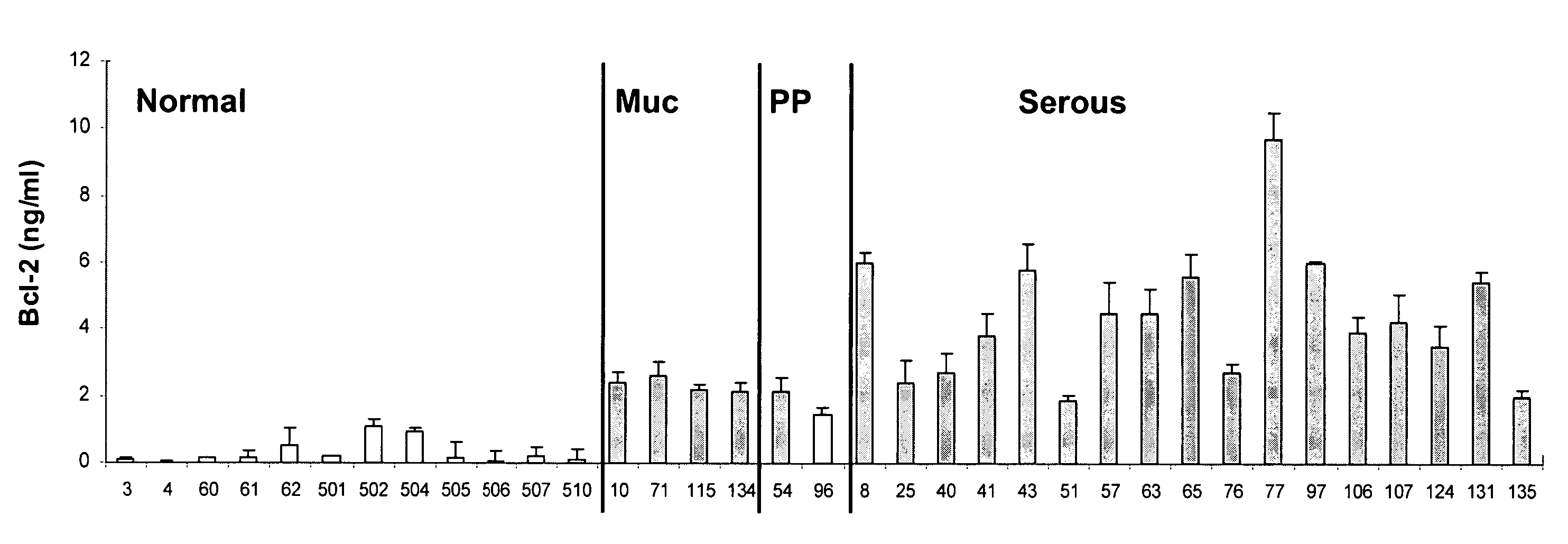
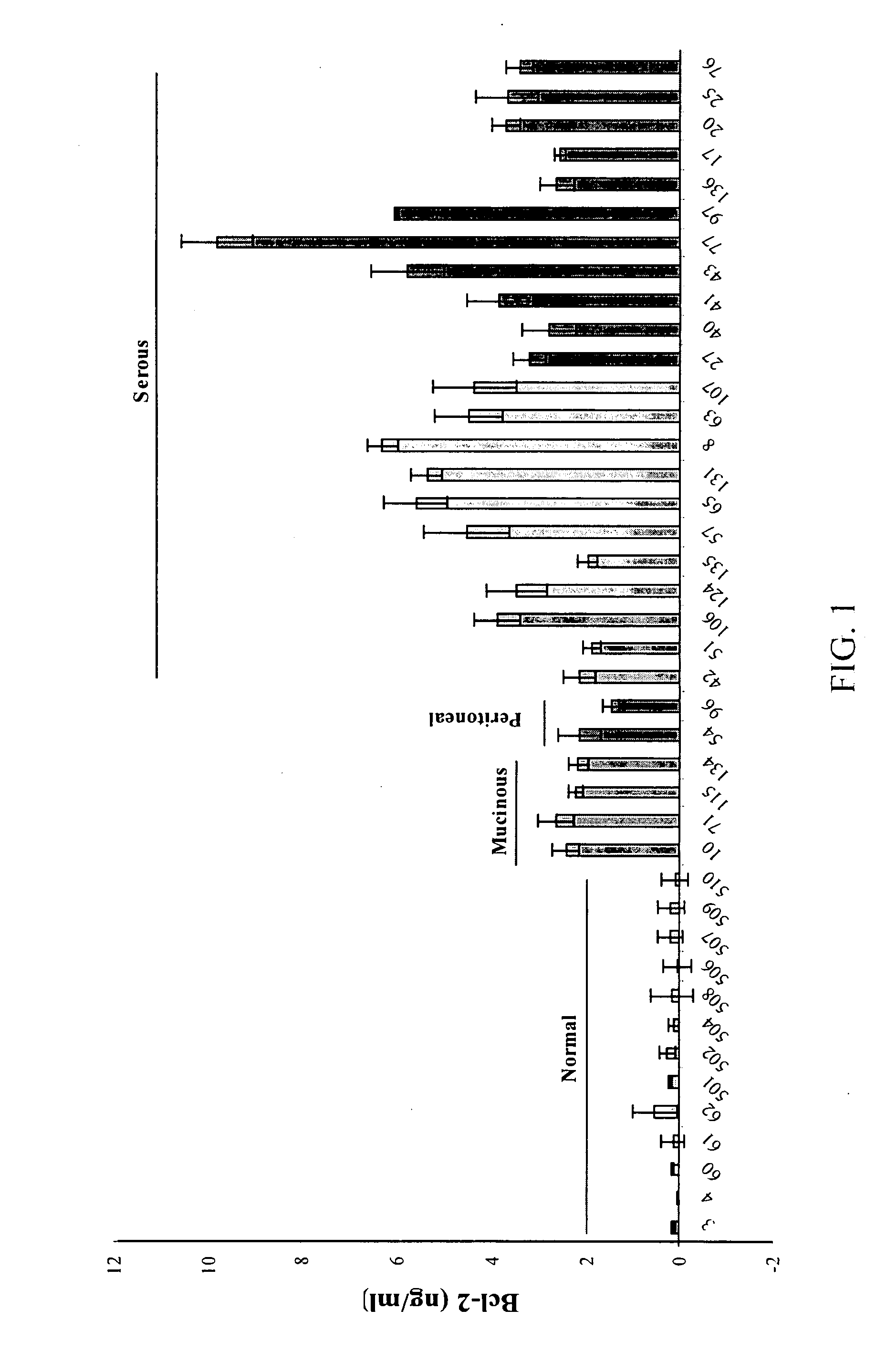
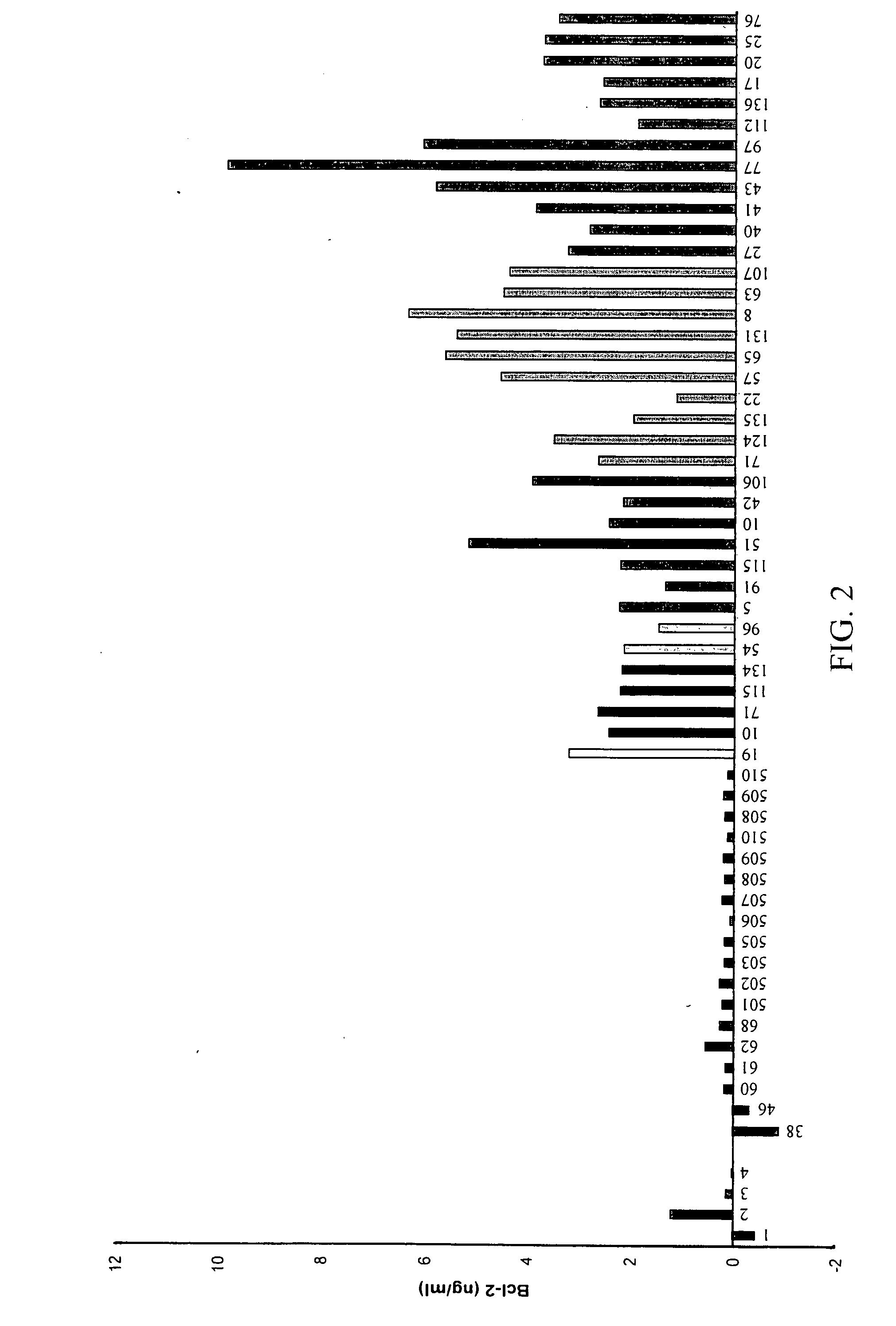
Detection of cancer by elevated levels of BCL-2
ActiveUS20080009005A1Lower Level RequirementsSimple and rapid and reliable and accurate and cost-effectiveBioreactor/fermenter combinationsBiological substance pretreatmentsOncologyCancer research
The present invention relates to a method for the diagnosis, prognosis, and monitoring of cancer, such as early or late stage ovarian cancer, in a subject by detecting Bcl-2 in a biological sample from the subject, preferably a urine or blood sample. Bcl-2 may be measured using an agent that detects or binds to Bcl-2 protein or an agent that detects or binds to encoding nucleic acids, such as antibodies specifically reactive with Bcl-2 protein or a portion thereof. The invention further relates to kits for carrying out the methods of the invention. The invention further relates to a device for the rapid detection of Bcl-2 in a bodily fluid and methods for rapidly measuring Bcl-2 in a bodily fluid.
Owner:UNIV OF SOUTH FLORIDA
Wetness Indicator Having Varied Hues
ActiveUS20130066289A1Component separationMaterial analysis by observing effect on chemical indicatorChange colorHue
A wetness indicator material may be used on a substrate to form a wetness sensor. The sensor may show either the presence or absence of an aqueous-based fluid or water-containing medium, such as vaginal fluid or urine in a personal hygiene article. The wetness sensor may be incorporated into the article. The wetness indicator material includes a standard colorant that does not change color when wetted. The standard colorant increases the range of hues exhibited by the wetness indicator material.
Owner:KIMBERLY-CLARK WORLDWIDE INC
Urinary flow control device and method
InactiveUS7001327B2Control displacementIncrease pressureAnti-incontinence devicesSurgeryBladder pressureUrinary flow
A urinary flow control device having a bladder responsive flow control assembly operatively engaged with an elongate member having proximal and distal ends, and a lumen capable of discharging urine therethrough is provided. The flow control assembly includes first and second fluid containing compartments adapted to be in fluid communication. The flow control assembly is adapted to receive and contain urine from a bladder so as to impinge upon the first fluid containing compartment. The flow control assembly is responsive to increased bladder pressure such that fluid is displaced from the first fluid containing compartment to the second fluid containing compartment to thereby permit select passage of received urine to the lumen of the elongate member for discharge therethrough. A method of controlling urinary flow is likewise provided.
Owner:SRS MEDICAL SYST INC
Device and systems for the intermittent drainage of urine and other biological fluids
This invention provides a drainage device for biological fluids. The device typically comprises a port for attachment of the device to a catheter or to a waste collection device; and a means for applying a negative pressure to said port, wherein said negative pressure is an intermittent substantially constant negative pressure.
Owner:RGT UNIV OF CALIFORNIA
Film-forming compositions for protecting skin from body fluids and articles made therefrom
The present invention relates to a film-forming barrier composition useful for protecting the skin from body fluids, such as urine, feces, menses. The composition comprises a skin conditioning agent, a viscosity enhancing agent and an oleophilic film-forming agent. Optionally, the lotion may comprise skin care ingredients for treating or maintaining skin health. The composition may be applied to the skin by suitable delivery vehicles, such as absorbent articles or components thereof, sheet materials, pads, bandages, sponges, foam pads, pumps, sprays, depending on the viscosity of the composition.
Owner:THE PROCTER & GAMBLE COMPANY
Integrated body fluid collection and analysis device with sample transfer component
A single, integrated device in which a body fluid (e.g., blood) of a human or animal can be both collected and analyzed easily and without risk of contamination is disclosed. The collection portion and analysis portion of the device are permanently joined to permit movement of small quantities of body fluid under controlled conditions, to minimize any waste of the body fluid, to ensure that no contamination reaches the main body fluid volume, and to create a permanent physical record of the results of the analysis in association with the fluid sample itself: A wide variety of different body fluid components which may be indicative of various diseases, dysfunctions and abnormalities of the human or animal or the body fluid itself can be tested for. The device includes a container for collecting human or animal body fluid, one or more testing chambers containing one or more analysis units activated by body fluid; a transfer pump or vacuum assembly to transfer one or more samples into the analysis units; and one-way valves or the equivalent to prevent any portion of the withdrawn sample from being returned to the collection container. The body fluid acted upon may be blood, blood plasma, urine, bile, pleural fluid, ascites fluid, stomach or intestine fluid, colostrom, milk or lymph.
Owner:AALTO SCI
Self-cleansing bladder drainage device
An urethral drain having deep external drainage channels, a low-profiled bladder retention segment, and a reversibly detachable collection segment, facilitates the draining of urine and fluids from the bladder. The low-profiled retention means minimizes bladder irritations and the deep external channels reduce the occurrence of infections. Incorporation of a reduced diameter smooth segment on the catheter, proximate the location of the external urethral sphincter allows the patient to void normally and at will. Modifying the size of this smooth segment aids the function of a defective sphincter in controlling urine leakage. The drain can be worn concealed within the urethra. Flushing action from normal voiding washes out particulate matters in the urethra and the concealed drain further minimizes contamination. Together, these features improve quality of life for patients needing catheterization.
Owner:CONSERT INC
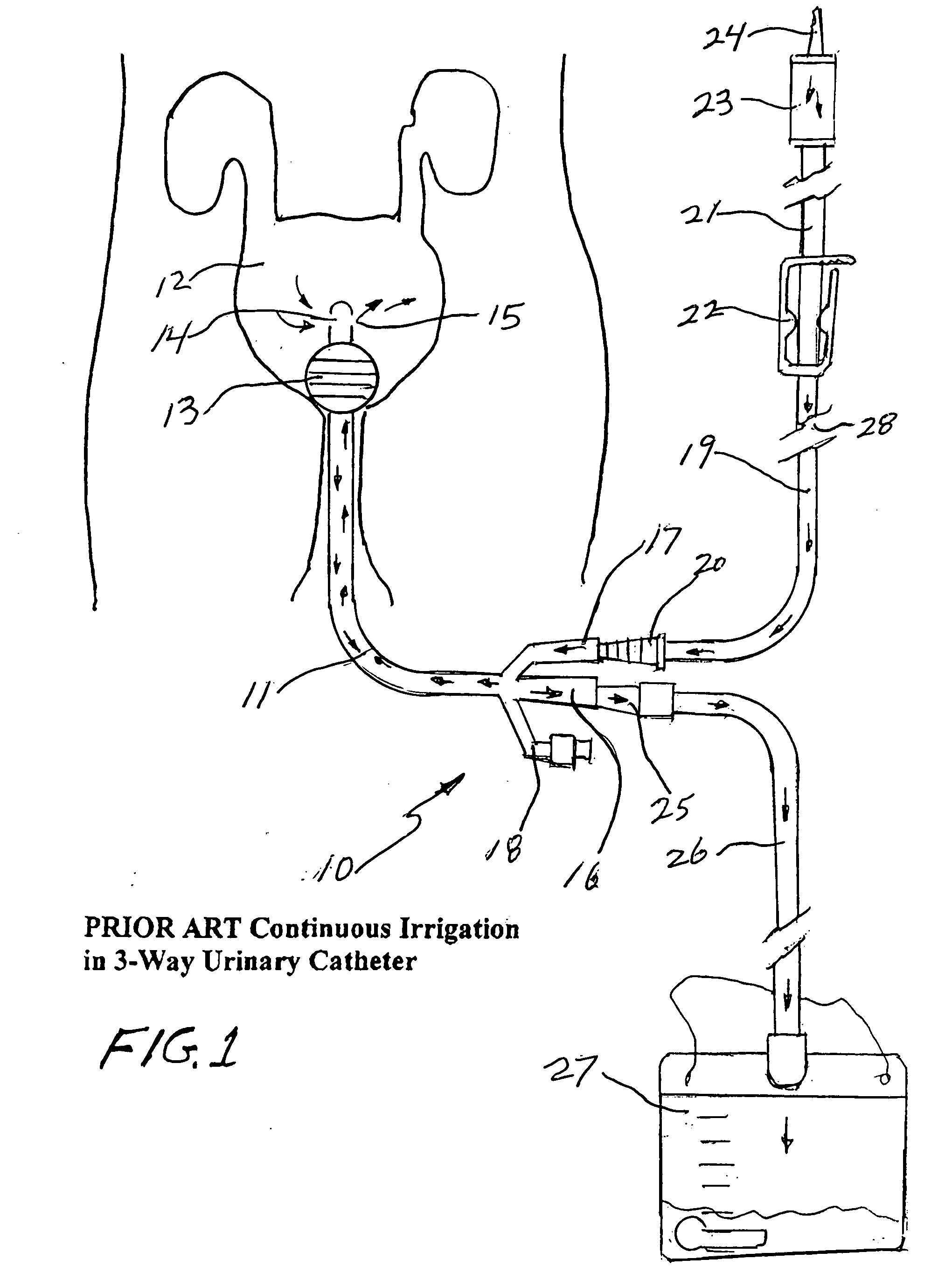
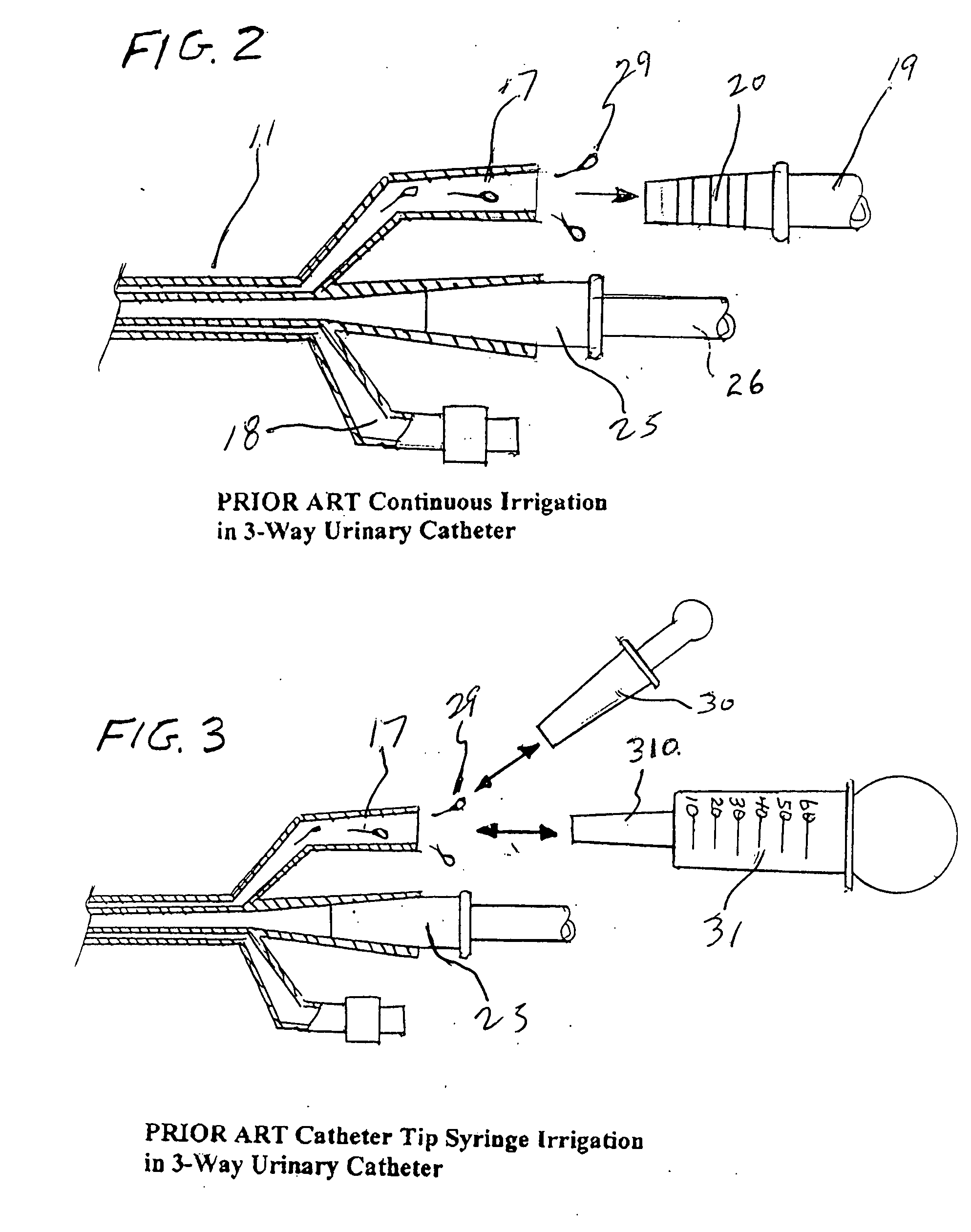
Closed system irrigation connector for urinary catheters
ActiveUS20060064065A1Reducing potential urinary tract infectionReduce leakageInfusion devicesSurgeryUrinary catheterUpper urinary tract infection
A closed system irrigation connector for use with both 3-way and 2-way urinary catheters is disclosed. The connector fits all urinary catheters and accepts continuous irrigation sets as well as all bulb, piston, and Toomey catheter tip irrigation syringes. The connector eliminates the use of catheter plugs and reduces the mess and leakage often associated with present urinary catheter irrigation procedures. The connector converts an open procedure to a closed system procedure protecting the patient from outside contamination to reduce urinary tract infections as well as the clinician from potentially infectious body fluids. It comprises an assembly of a plastic housing with an internal silicone diaphragm slit valve and a plastic entrance port that are unitized in assembly by sonic welding. The entrance port forms a tapered wedge lock engagement with irrigation sets as well as catheter tip irrigation syringes.
Owner:DALE MEDICAL PRODS
Disposable urinal system
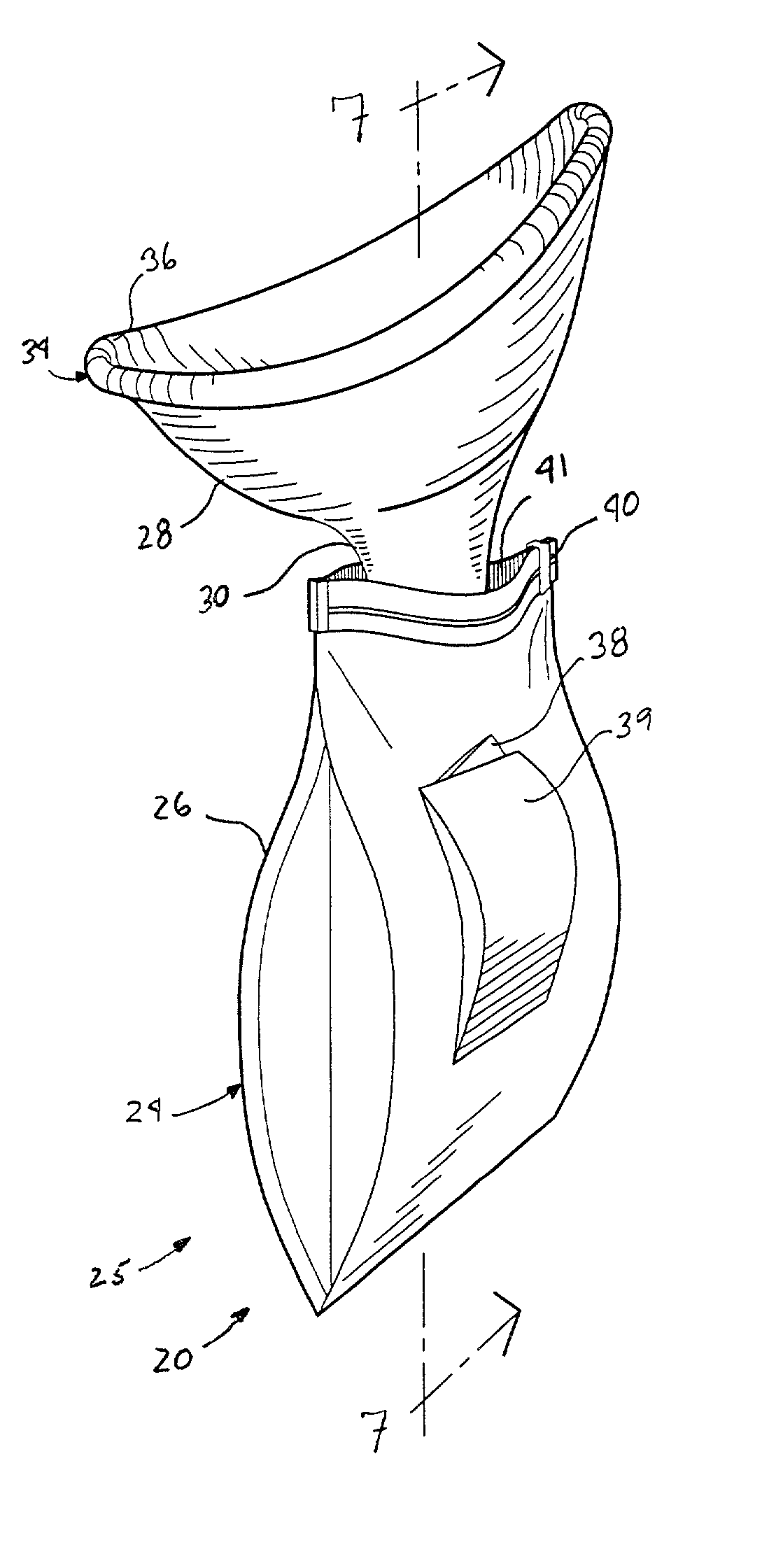
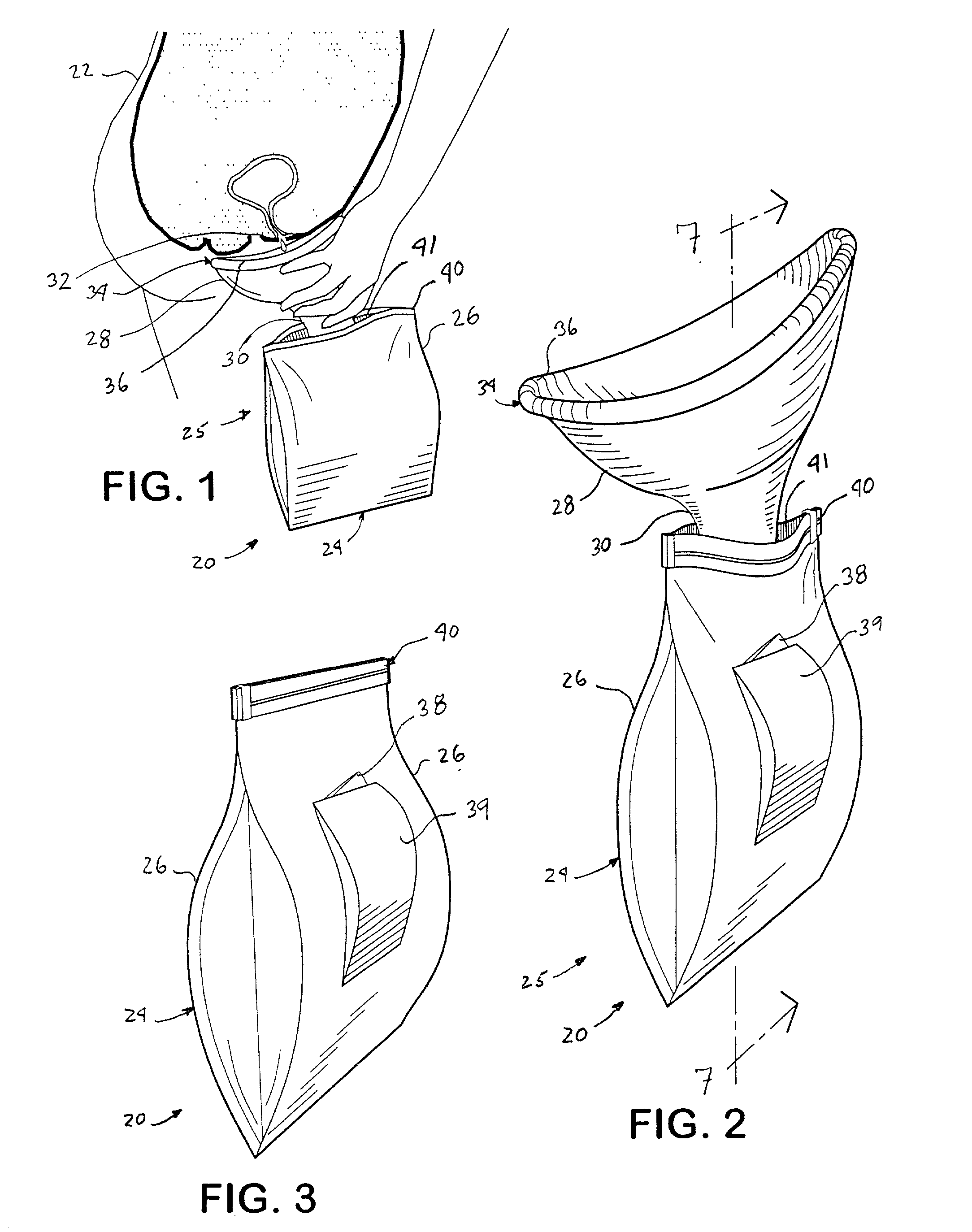
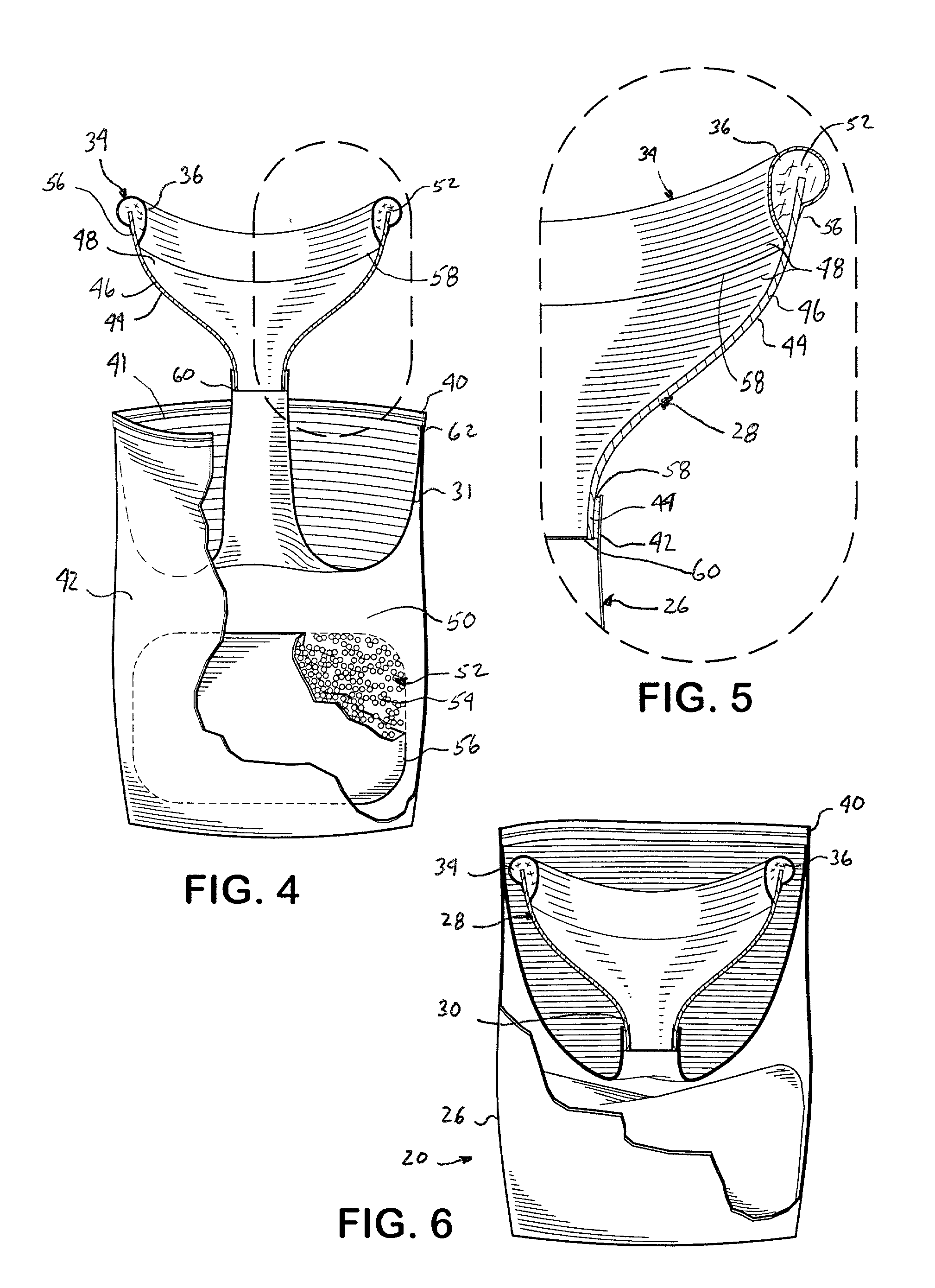
InactiveUS20020193762A1Easy to storeEasy to useNursing urinalsNon-surgical orthopedic devicesHydrophobic polymerHand held
A hand-held disposable portable urinal device particularly well-suited for female use. The easy-to-use device has a leak and splash resistant soft upper seal attached to a retractable funnel, which easily folds back into a sealable bag. The funnel has a neck portion which allows the user to comfortably hold the urinal close to the ureter during use. The disposable urinal bag is odor- and vapor-sealable, and has a hydrophobic polymer containing inner bag for absorbing the urine. In addition, the disposable urinal has a deodorizer activated by the urine upon contact. The disposable urinal is easily transported in a folded state, and preferably comes three in a box. After use, the disposable urinal may be sealed and thrown away.
Owner:SUYDAM KRISTEN V
Tea-derived feed additive and animal feed containing the same
InactiveUS6068862AReduce generationQuality improvementSolid waste disposalAnimal feeding stuffFecesAdditive ingredient
The present invention relates to a feed additive for animals comprising tea extract, to a feed additive for animals of which active ingredient is tea leaf fiber, and to an animal feed comprising the feed additive for animals. The tea extract in the present invention exerts great effects in preventing and treating noninfectious and infectious diarrhea in domestic animals, poultry, and pet animals, improving milk quality of fresh milk, increasing milk yield, and improving reproductive efficiency. Also, the tea leaf fiber of the present invention improves intestinal function in domestic animals, decreases the occurrence of diarrhea, increases milk yield, improves feed efficiency and removes malodor of feces and urine.
Owner:TAIYO KAGAKU CO LTD