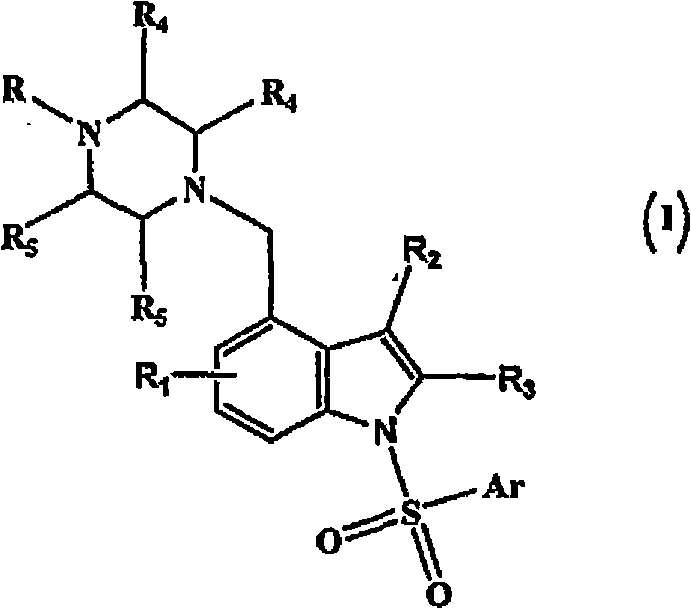
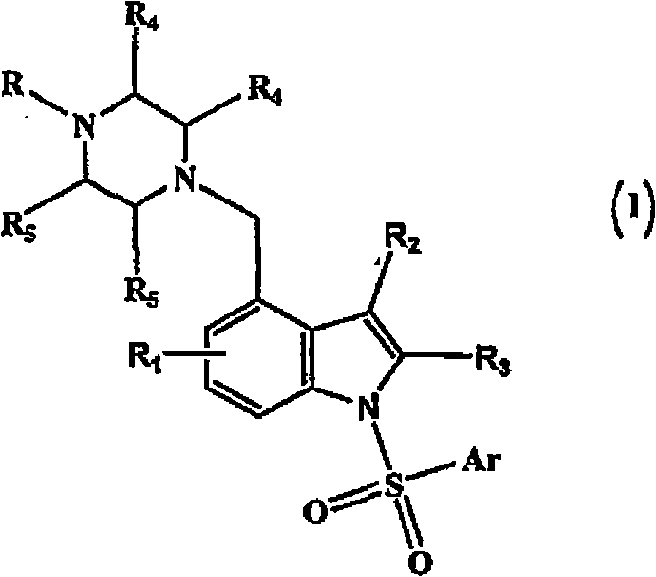
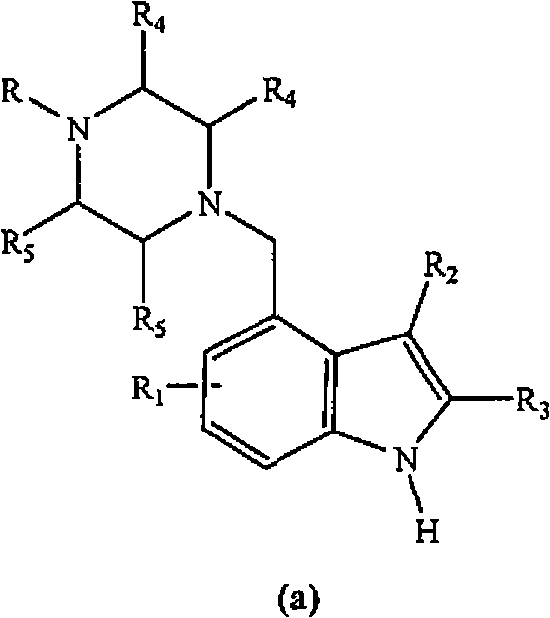
4-(heterocyclyl)alkyl-n-(arylsulfonyl) indole compounds and their use as 5-ht6 ligands
A compound, alkyl technology, applied in the field of pharmaceutical compositions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0153] Embodiment 1: Preparation of (4-methylpiperazin-1-ylmethyl)-1H-indole
[0154] Step (i): Preparation of (2-methyl-3-nitrophenyl)-(4-methylpiperazin-1-yl)methanone
[0155] 2-Methyl-3-nitrobenzoic acid (5.525 mmol, 1.0 g) was placed in a 25 mL two-neck round bottom flask connected to a condenser (with a protective tube). Thionyl chloride (6.07mmol, 0.735g) and 1,2-dichloroethane (5mL) were added thereto, and the solution was refluxed for 3 hours. The reaction mixture was added to another 100 mL flask containing a solution of N-methylpiperazine (16.57 mmol, 1.66 g) in 10 mL of 1,2-dichloroethane, where the temperature was maintained below 5°C. The reaction mixture was then stirred at 25°C for 0.5 hours. After the reaction was complete, the reaction mixture was poured into 50 mL of water. The 1,2-dichloroethane layer was collected, washed with water (2×10 mL), brine (10 mL), and dried over anhydrous sodium sulfate. The volatiles were removed under reduced pressure to ...
Embodiment 2
[0162] Example 2: Preparation of 1-benzenesulfonyl-4-(4-methylpiperazin-1-ylmethyl)-1H-indole
[0163] (4-Methylpiperazin-1-ylmethyl)-1H-indole (0.8733 mmol, 0.2 g) (from Example 1) was dissolved in 2 mL of N,N-dimethylformamide. The above solution was then added slowly to a 25 mL flask containing a suspension of sodium hydride (1.31 mmol, 31.4 mg) in 1 mL DMF under nitrogen atmosphere while maintaining the temperature below 10 °C. The reaction mixture was then stirred at 25°C for 1 hour. Benzenesulfonyl chloride (1.31 mmol, 0.2312 g) was slowly added to the well stirred solution while maintaining the temperature below 10°C. The reaction mixture was stirred for an additional 2 hours. After the reaction was completed, the reaction mixture was poured onto 20 g of ice-water mixture with stirring, and the resulting mixture was extracted with ethyl acetate (2×20 mL). The combined ethyl acetate extracts were then washed with water (20 mL), brine (20 mL), and dried over anhydrous ...
Embodiment 3
[0168] Following the procedure described in Example 2 with some non-critical modifications, the following compounds (2-42) of the present invention were prepared.
[0169] 2
[0170] 1 H-NMR (ppm): 2.26 (3H, s), 2.43 (8H, bs), 3.68 (2H, s),
[0171] 6.89-6.89 (1H, d), 7.16-7.17 (2H, m), 7.43-7.58 (4H, m),
[0172] 1-ylmethyl)-1H-indole
[0173] 27
[0174] indole
[0175] 40
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap