Piplartine analogue, preparation method therefor and applications
A technology of perylene amide and analogs, applied in the field of medicinal chemistry, can solve the problems of limiting clinical application, large size, which can cause edema of adjacent renal tubules and glomerular epithelial cells, renal tubular hemorrhage, resistance, etc., and achieve good inhibitory activity , the effect of good medicinal prospects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
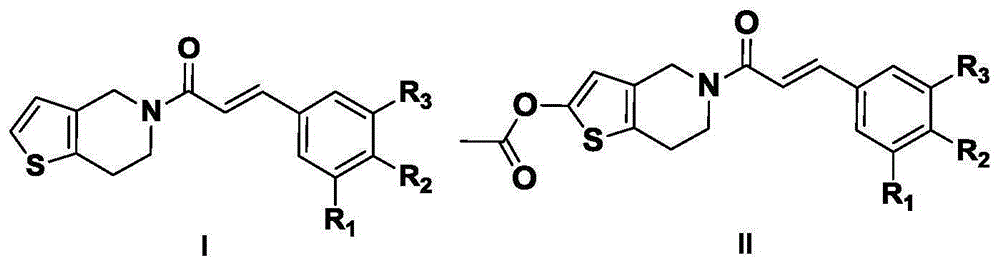
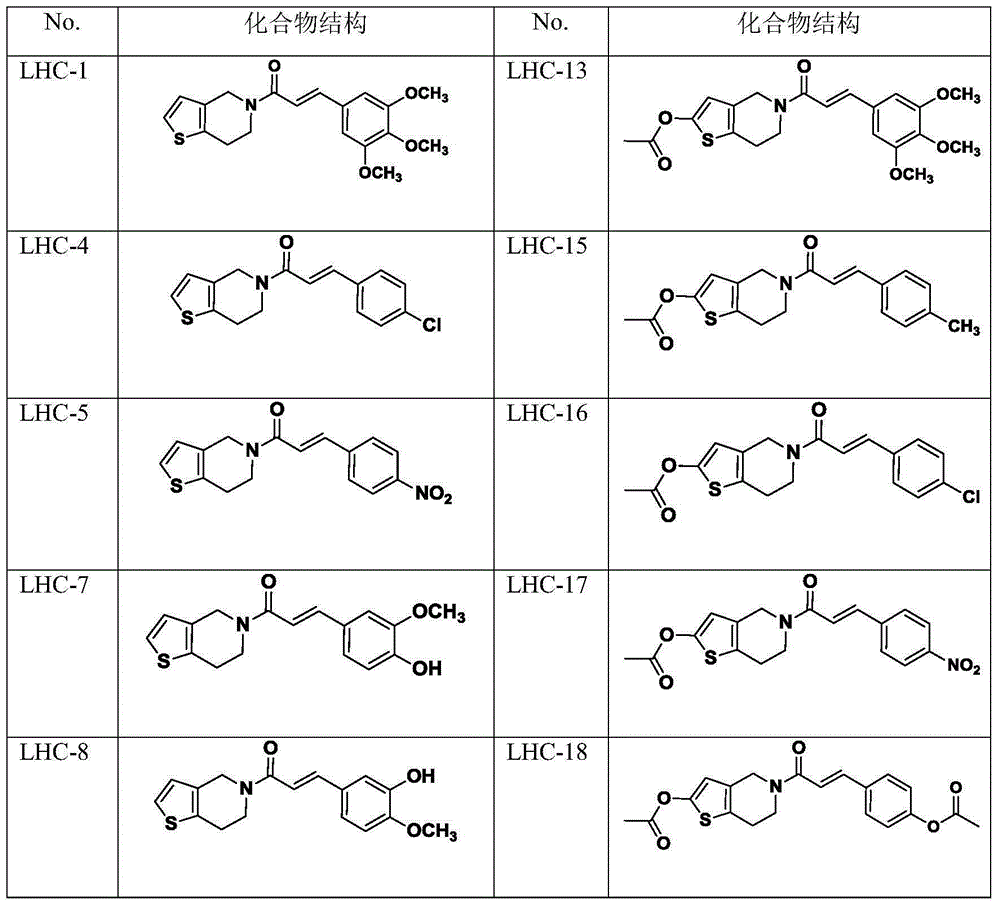
[0030] (E)-1-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)-3-(3,4,5-trimethoxyphenyl)propan-2 Synthesis of -en-1-one (LHC-1)
[0031] 1.1 Synthesis of (E)-3-(3,4,5-trimethoxyphenyl)acrylic acid
[0032]
[0033] In a 500mL three-necked flask, add 3,4,5-trimethoxybenzaldehyde (10.0g, 51.0mmol), malonic acid (6.41g, 61.5mmol), pyridine (35mL), piperidine (2mL) and benzene (150mL), install an oil-water separator, and reflux at 110°C for 6h. TLC [V (chloroform): V (methanol) = 10:1 is the developer] detection shows that the reaction is basically complete, cooled to room temperature, added 75 (mL) saturated aqueous sodium carbonate solution, continued to stir for 30 min, separated, took the water phase, the water phase Adjust the pH to 4 with 3 mol / L hydrochloric acid, and a large amount of white solid precipitated; suction filtered, the filter cake was recrystallized with absolute ethanol, and dried to obtain 7.92 g of white solid, yield 65.2%, m.p.124.3-125.7°C.
[0034] 1.2(E...
Embodiment 2
[0039] (E)-1-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)-3-(4-methoxyphenyl)prop-2-ene-1 -Synthesis of ketone (LHC-2)
[0040]
[0041] (E)-3-(4-methoxyphenyl)acrylic acid was synthesized according to the method 1.1 in Example 1, and (E)-1-(6,7-dihydrothiophene was obtained by operating according to the method 1.2 in Example 1 And[3,2-c]pyridin-5(4H)-yl)-3-(4-methoxyphenyl)prop-2-en-1-one (LHC-2) light yellow solid, m.p.131.4~ 132.7°C; 1 H-NMR (CDCl 3 ,400MHz)δ:7.67(d,J=15.2Hz,1H,ArCH=),7.50(d,J=8.0Hz,2H,ArH),7.15(d,J=5.2Hz,1H,ThH),6.90( d,J=8.4Hz,2H,ArH),6.83-6.79(m,2H,ThH and-CH=),4.76(s,2H,Py-CH 2 ),3.97(br s,2H,Py-CH 2 ),3.84(s,3H,OCH 3 ),2.94(br s,2H,Py-CH 2 ); 13 C-NMR (CDCl 3 ,100MHz)δ:166.3,160.9,142.6,132.3,129.4,127.9,125.2,124.5,123.5,114.9,114.2,55.3,45.9,43.3,26.0; IR(KBr,cm -1 )υ: 3004.3, 2962.5, 2905.6, 1649.8, 1593.0, 1506.3, 1437.5, 1249.2, 1165.5, 1060.8, 1027.9, 980.1, 896.4, 824.6, 716.9; ESI-Mass for C 17 h 17 NO 2 S:m / z(M + +H)300.13.
Embodiment 3
[0043] (E)-1-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)-3-(4-methylphenyl)prop-2-en-1- Synthesis of Ketones (LHC-3)
[0044]
[0045] (E)-3-(4-methylphenyl)acrylic acid was synthesized according to the method 1.1 in Example 1, and (E)-1-(6,7-dihydrothieno) was obtained according to the method 1.2 in Example 1 [3,2-c]pyridin-5(4H)-yl)-3-(4-methylphenyl)prop-2-en-1-one (LHC-3) white solid, m.p.145.8~147.2℃; 1 H-NMR (CDCl 3 ,400MHz) δ:7.69(d,J=15.6Hz,1H,ArCH=),7.45(d,J=7.6Hz,2H,ArH),7.19(d,J=8.0Hz,2H,ArH),7.15( d,J=5.2Hz,1H,ThH),6.89(d,J=15.6Hz,1H,-CH=),6.83(d,J=4.8Hz,1H,ThH),4.77(s,2H,Py- CH 2 ),3.97(br s,2H,Py-CH 2 ),2.95(br s,2H,Py-CH 2 ),2.37(s,3H,CH 3 ); 13 C-NMR (CDCl 3 ,100MHz)δ:166.2,142.9.139.9,132.4,129.5,127.6,125.2,124.5,123.5,116.5,116.2,45.9,43.2,25.9,21.4; IR(KBr,cm -1 )υ: 3076.1, 2923.6, 2833.9, 1643.9, 1596.0, 1518.3, 1455.5, 1452.5, 1323.9, 1219.3, 1054.8, 1013.0, 968.1, 812.6, 737.9, 660.1; ESI-Mass for C 17 h 17 NOS:m / z(M + +H)284.08.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap