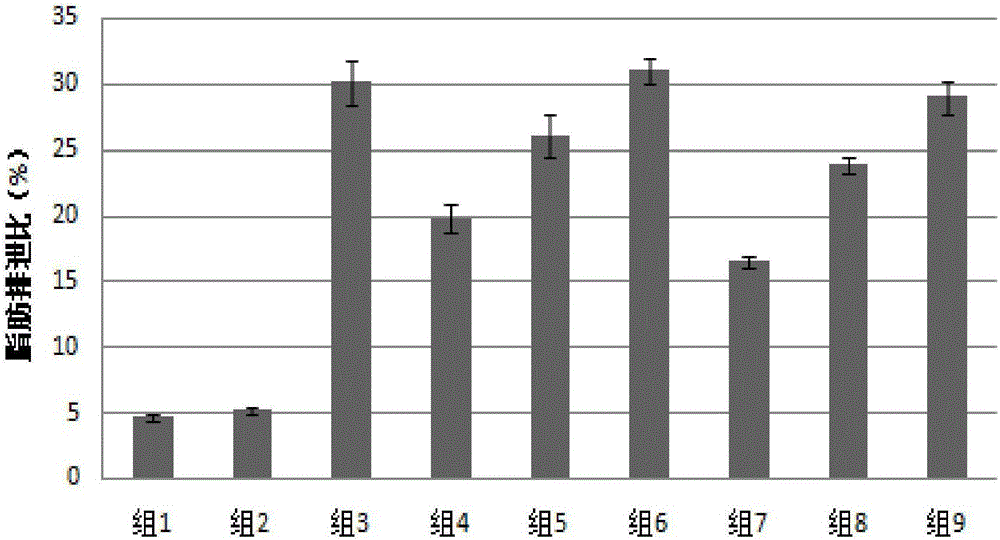
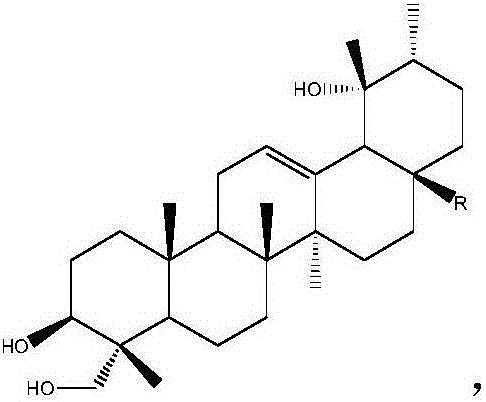
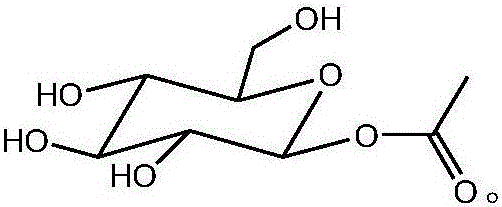
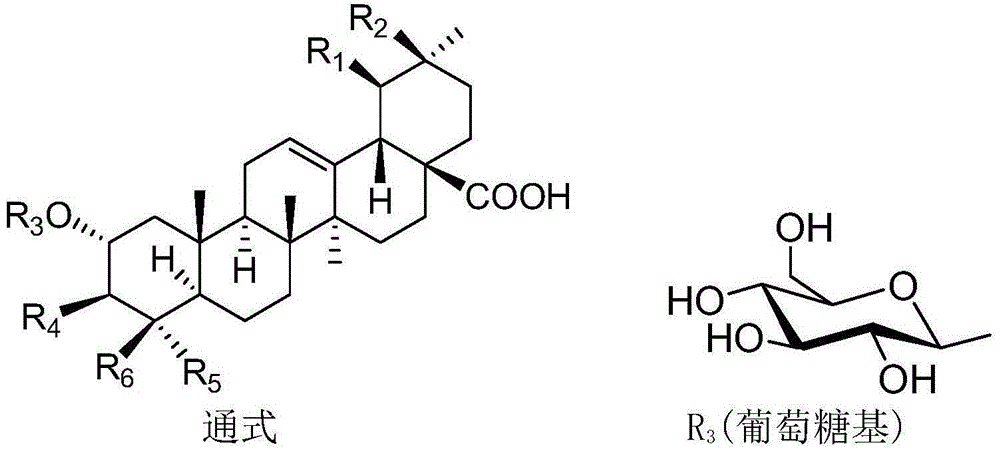
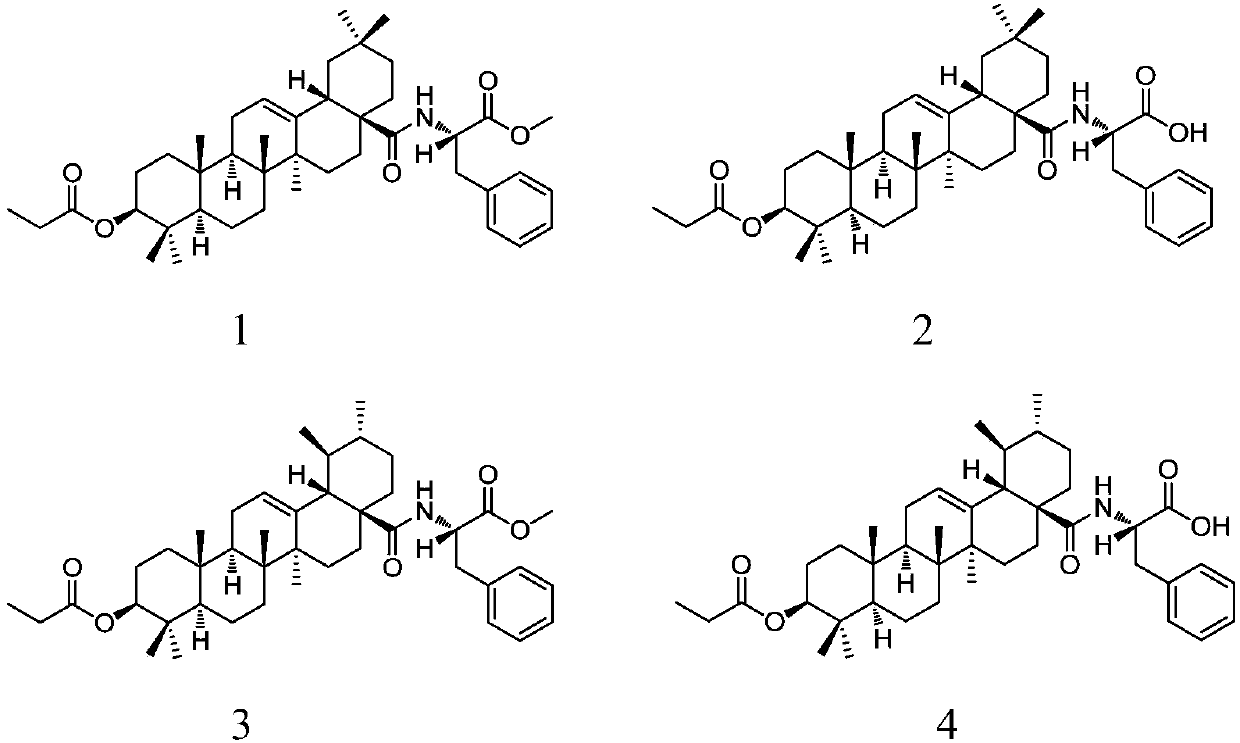
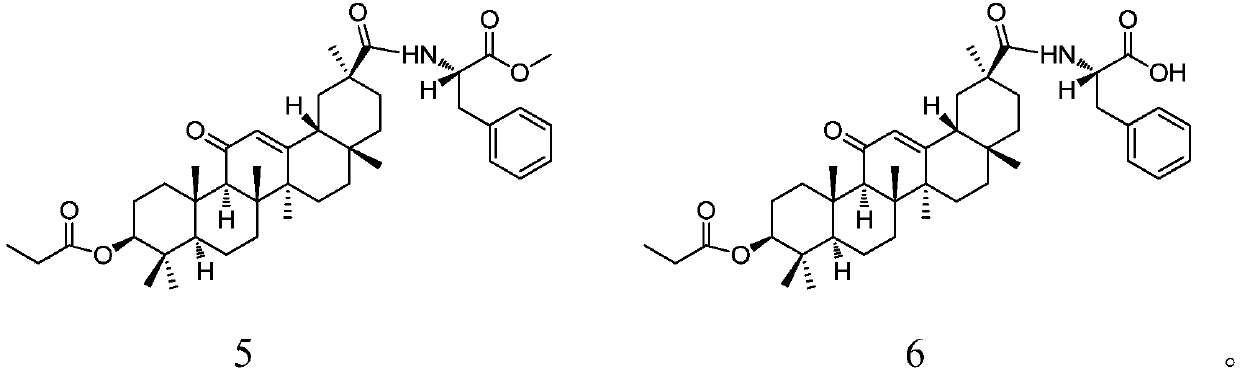
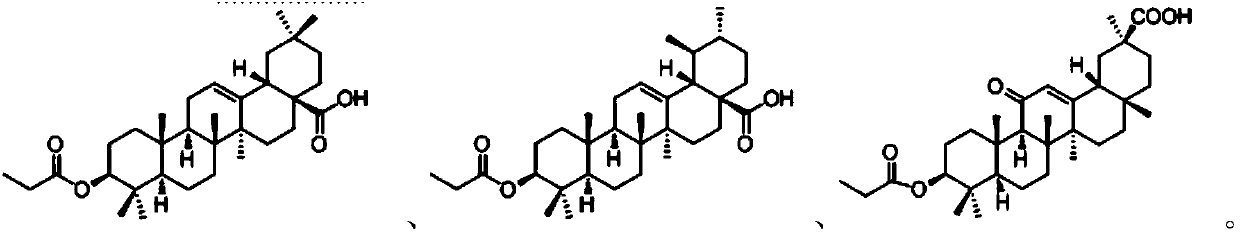
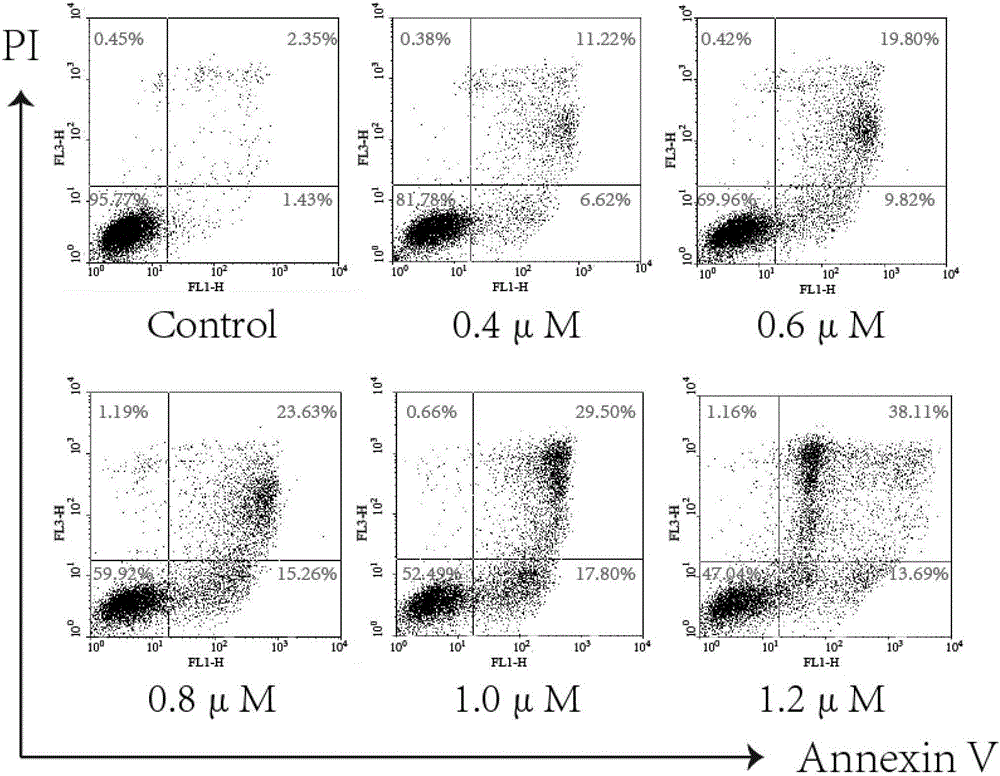
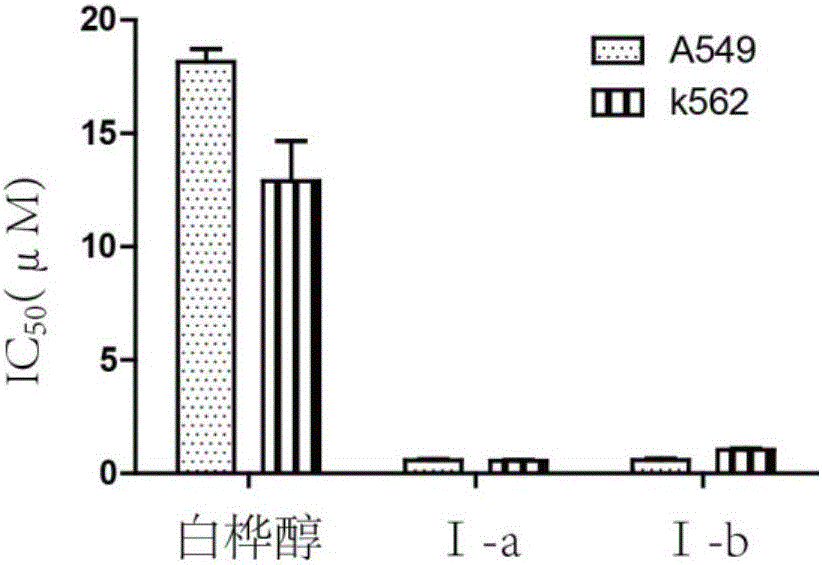
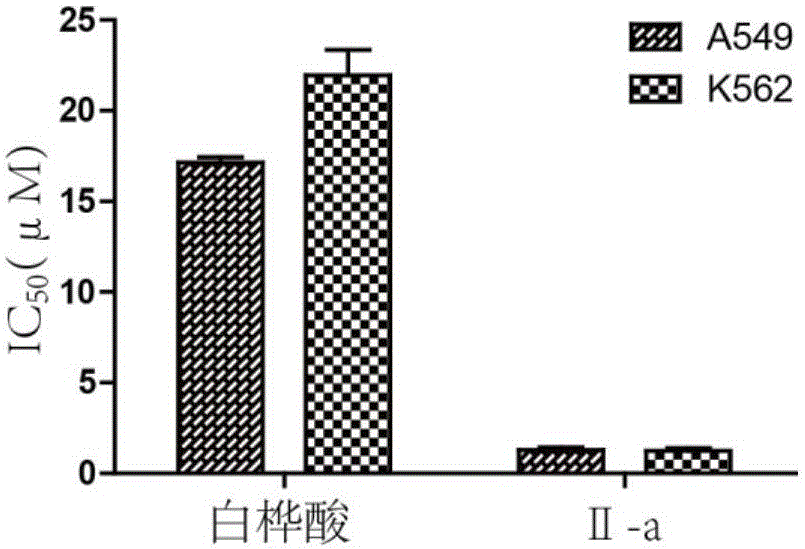
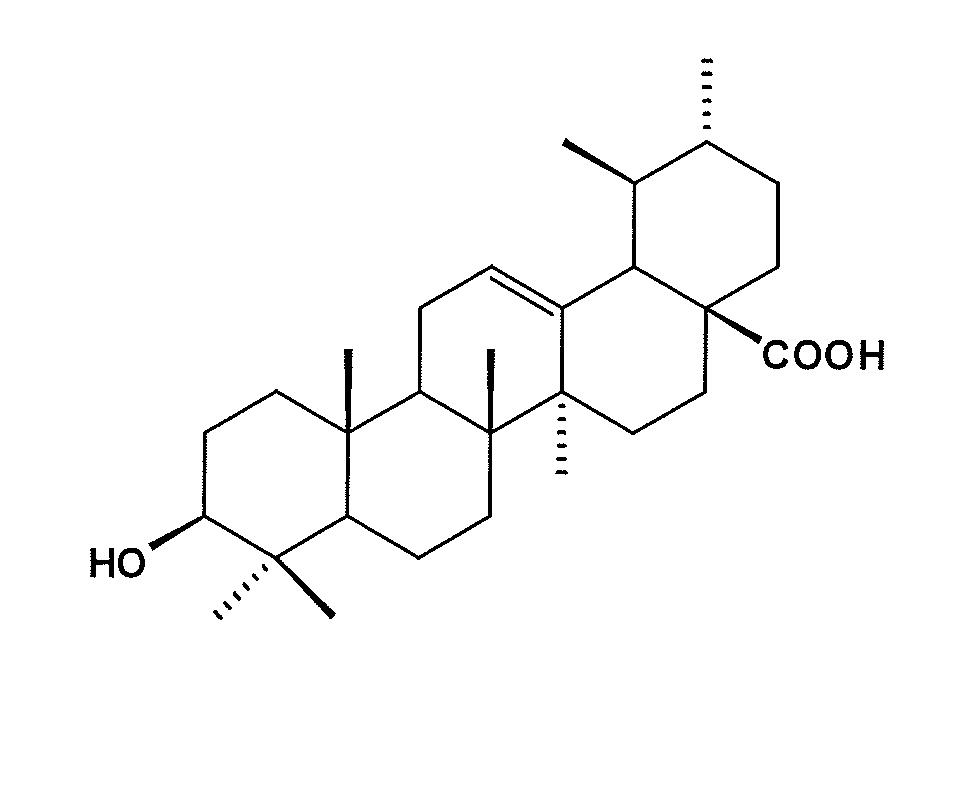
Patents
Literature
31 results about "Pentacyclic Triterpenes" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
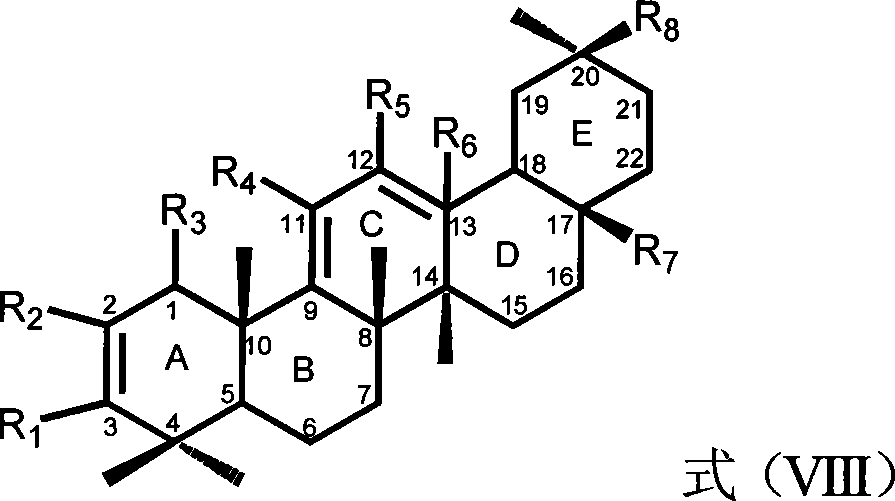
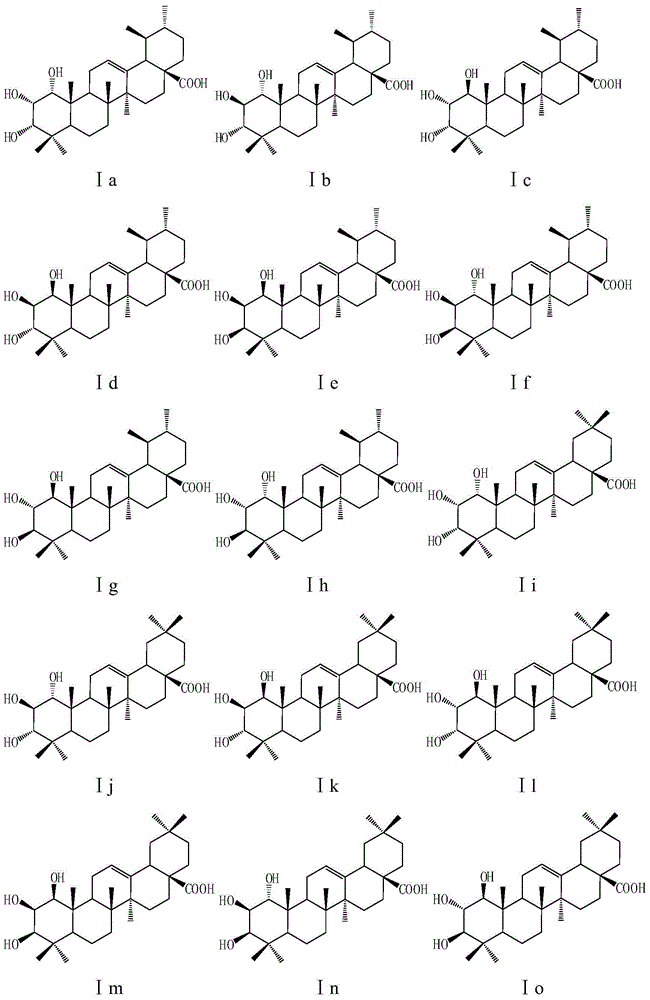
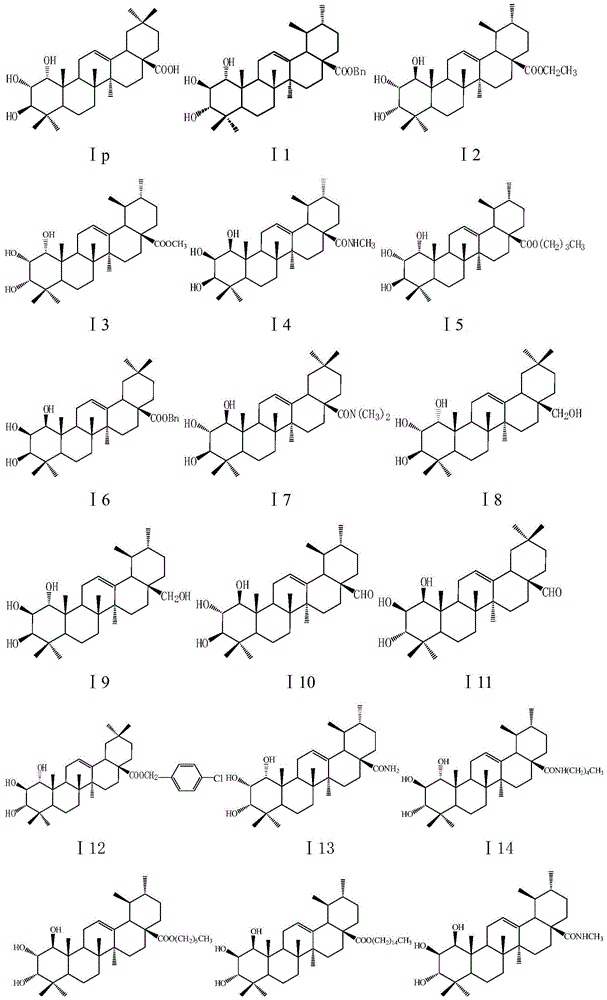
Five-ring derivatives of dammarane having a chair-chair-chair-boat configuration. They include the lupanes, oleananes, amyrins, GLYCYRRHIZIC ACID, and soyasaponins.
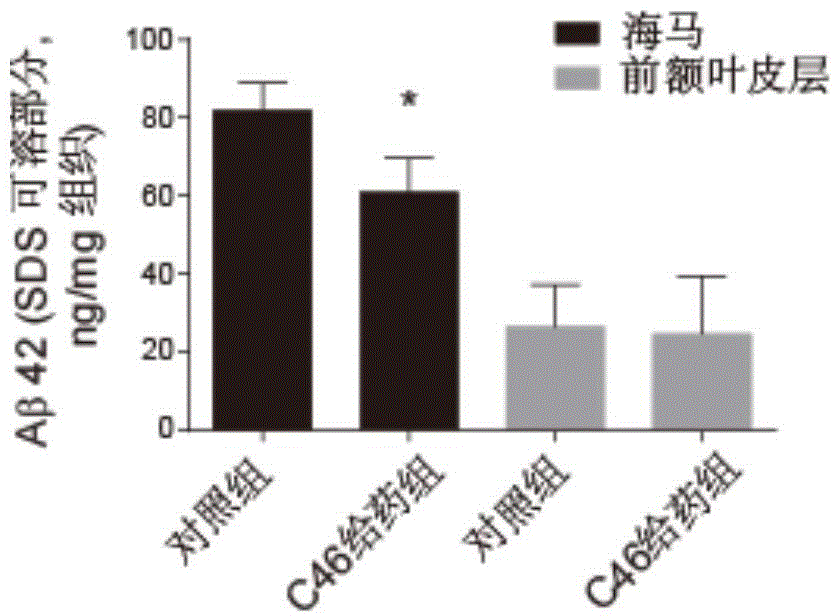
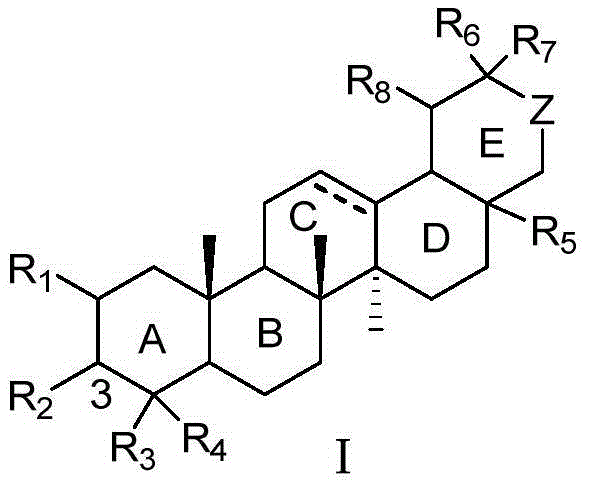
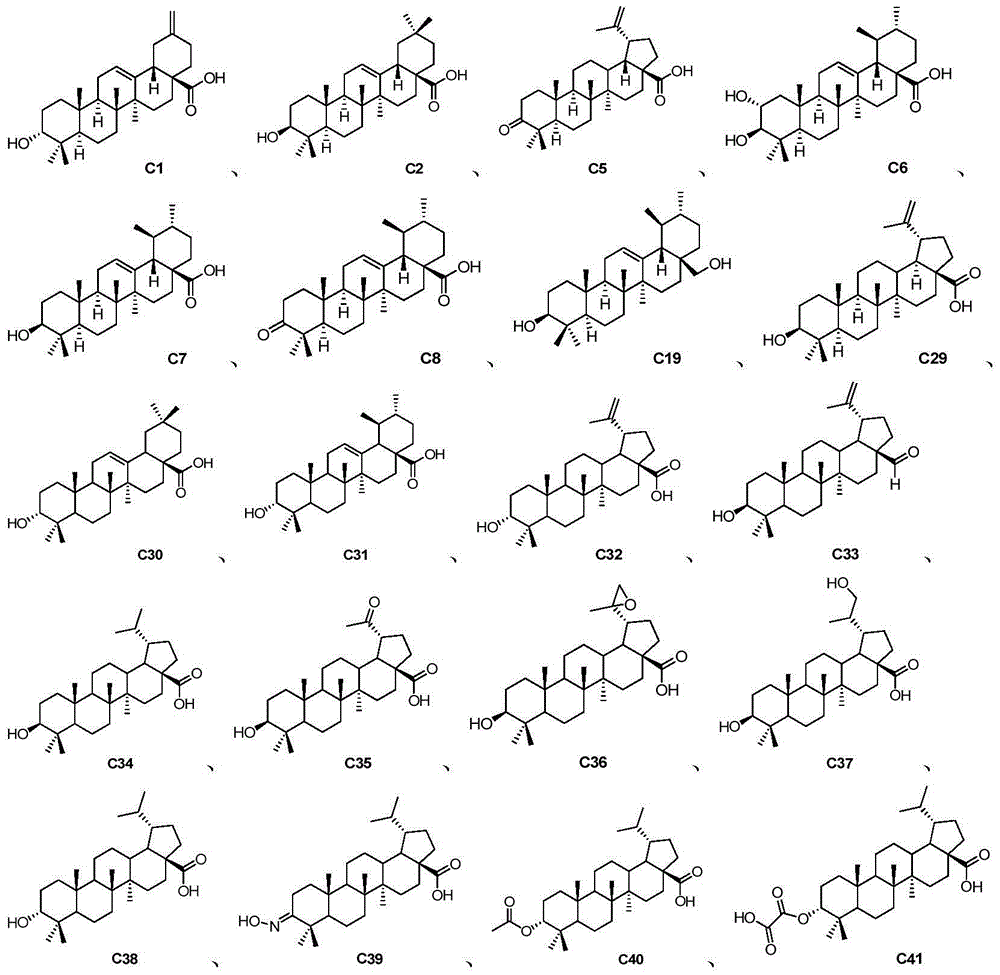
Pentacyclic triterpene compound and application of pentacyclic triterpene compound in preparation of medicine for treating Alzheimer's disease
ActiveCN104910238AActivity that inhibits the interactionReduce generationOrganic active ingredientsNervous disorderDiseasePentacyclic Triterpenes
Disclosed are a pentacyclic triterpene compound represented by general formula (I) and pharmaceutical compositions containing the compound. Also disclosed is use of the compound in preparation of a PS1 / BACE1 interaction inhibitor, and uses thereof in preparation of drugs for treating Alzheimer's disease. Disclosed for the first time is that a pentacyclic triterpene compound represented by general formula (I) can be used as a PS1 / BACE1 inhibitor, having activity of inhibiting PS1 / BACE1 interaction, and can reduce generation of Aβ and be used in preparation of drugs for treating Alzheimer's disease.
Owner:SHANGHAI INST OF MATERIA MEDICA CHINESE ACAD OF SCI
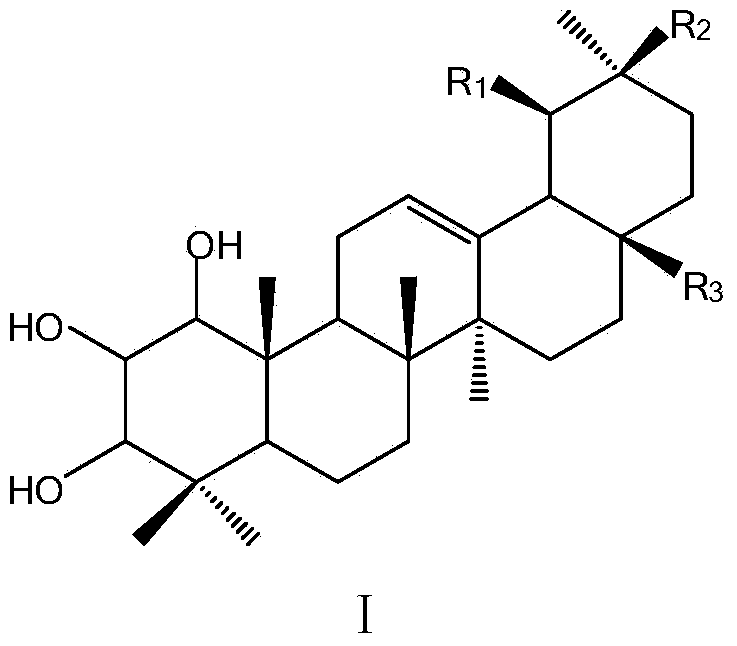
Application of A-ring trihydroxyl substituted pentacyclic triterpene compound to pharmacy
ActiveCN103919784AStrong inhibitory activityGood treatment effectAntibacterial agentsBiocidePhytophthora sp.Tobacco mosaic virus
The invention discloses an application of an A-ring trihydroxyl substituted pentacyclic triterpene compound to preparation of an antibacterial or anti-tobacco mosaic virus drug. The general formula of the compound is as shown in the specification, wherein hydroxyls of C1, C2 and C3 are respectively in alpha configuration or beta configuration; R1 and R2 are selected from hydrogen or methyl and are different from each other; R3 is selected from -COOH, -CH2OH, -CHO, -COOR1, -CONH2, -CONHR1 and -CONR1R2; R1 and R2 are selected from alkyl containing 1-15 carbon atoms, substituted or unsubstituted phenyl and substituted or unsubstituted phenyl alkyl; and the substituent group is selected from halogen, hydroxyl, cyan, amino, nitryl, sulfydryl or phenyl, acyl, aryl, alkoxy and alkyl containing 1-15 carbon atoms. The compounds have antibacterial or anti-tobacco mosaic virus activity, have extremely high bacteriostatic activity especially to gram positive bacteria, phytophthora nicotiana and tobacco mosaic virus, and have a good application prospect in fields of medicines and pesticides.
Owner:THE KEY LAB OF CHEM FOR NATURAL PROD OF GUIZHOU PROVINCE & CHINESE ACADEMY OF SCI
Mitochondrion-targeted antitumor pentacyclic triterpene derivatives, and preparation method and application thereof
ActiveCN105924492AEasy to prepareProcess stabilityOrganic active ingredientsSteroidsDrugPentacyclic Triterpenes
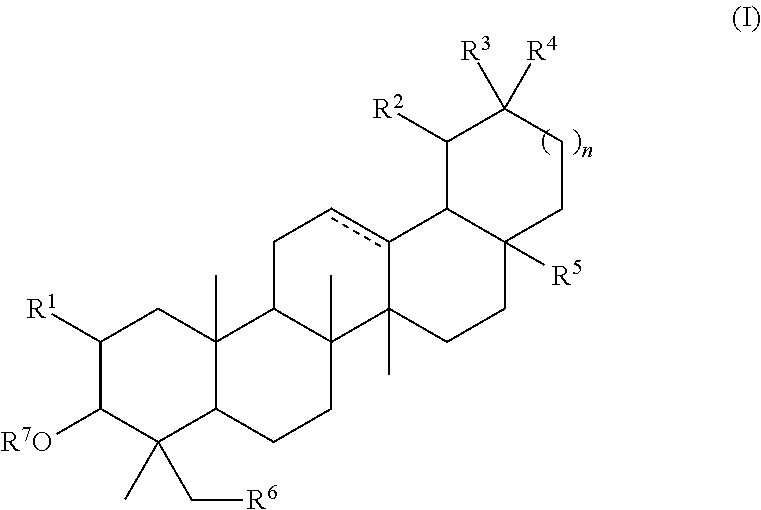
The invention discloses mitochondrion-targeted antitumor pentacyclic triterpene derivatives of which the structural formula are disclosed as Formula (I), Formula (II), Formula (III), Formula (IV) or Formula (V), wherein R1 is hydrogen, formacyl, acetyl or a group (n=1-19) disclosed in the specification; R2 is disclosed in the specification (n=1-19); R3 is hydroxy, methoxy or ethoxy; and R4 is disclosed in the specification (n=1-19). The compounds have favorable antitumor activity; and the natural compounds are targeted to the mitochondrion, so that the antitumor pentacyclic triterpene derivatives can be better applied to the development of antitumor drugs. The compounds are salts, thereby greatly enhancing the water solubility of drugs and improving the pharmacokinetic parameters.
Owner:SHANDONG UNIV
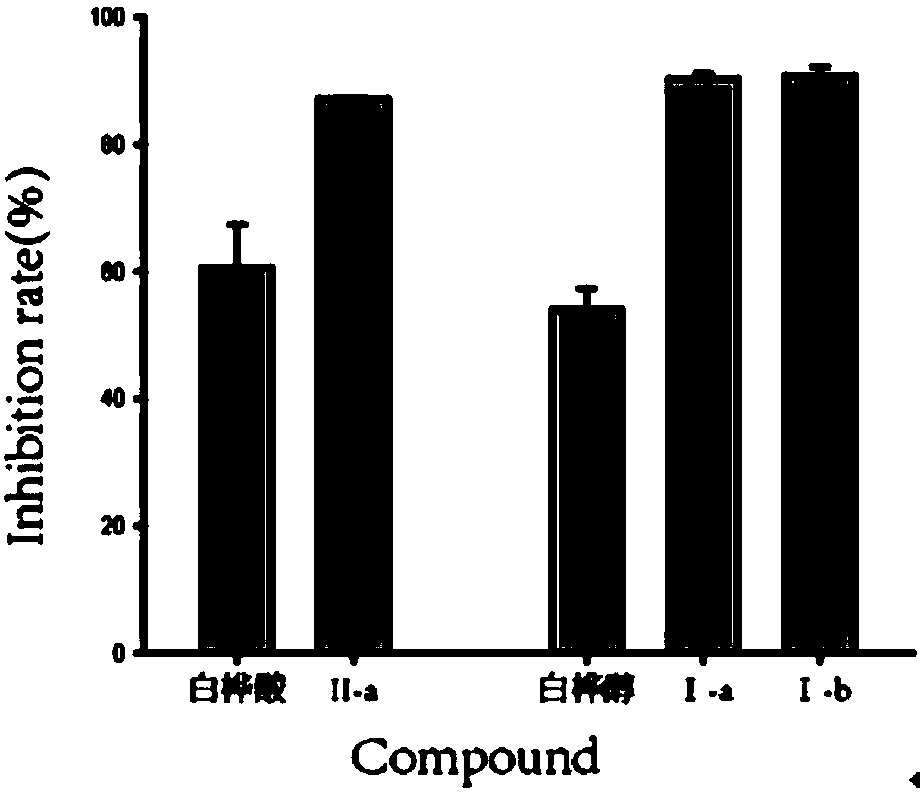
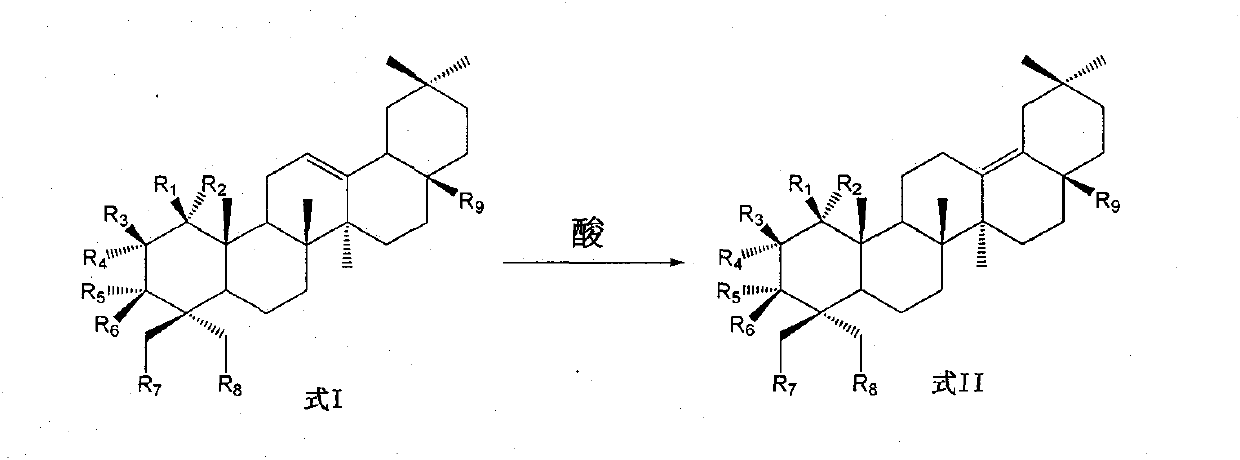
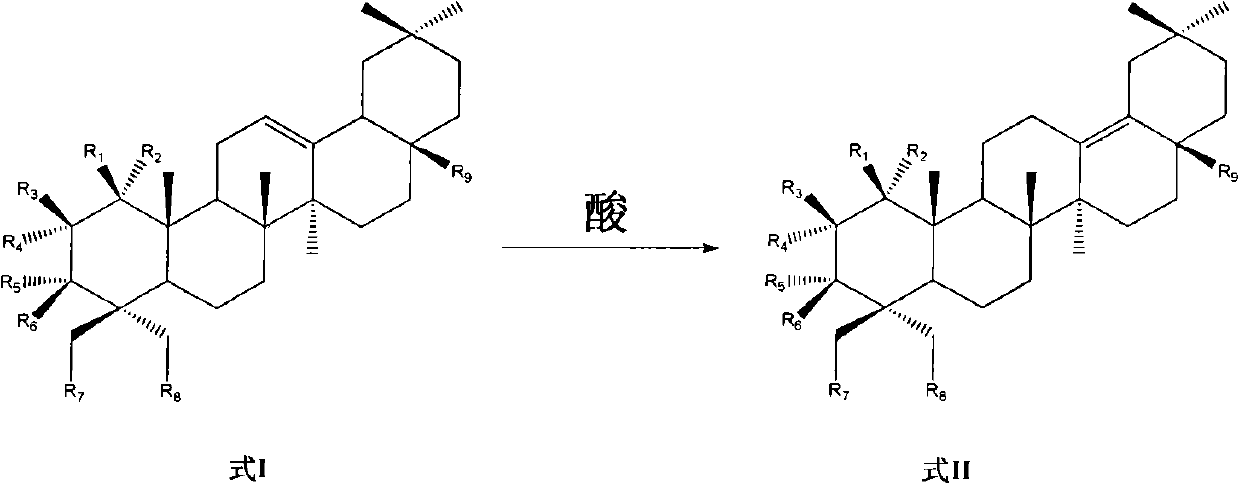
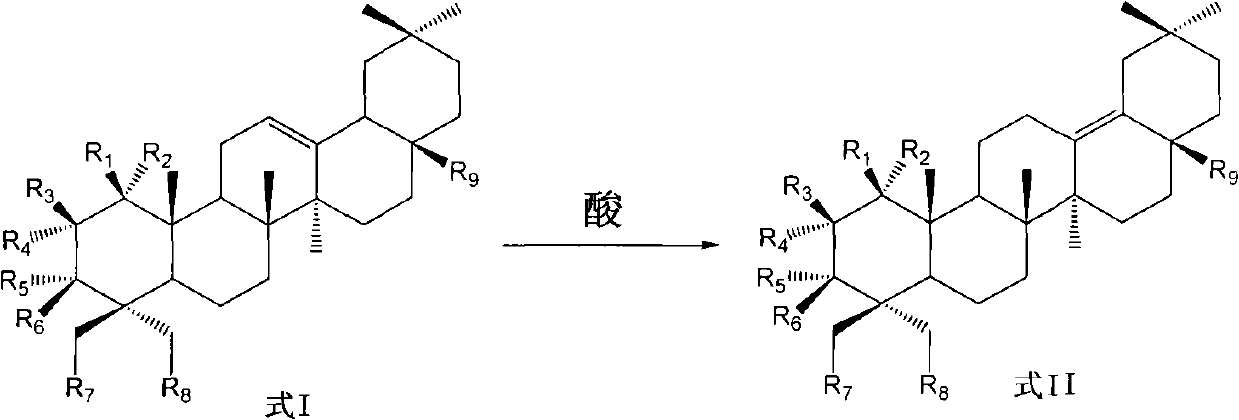
Preparation of 13 (18)-oleanane-type pentacyclic triterpene and derivatives of 13(18)-oleanane-type pentacyclic triterpene
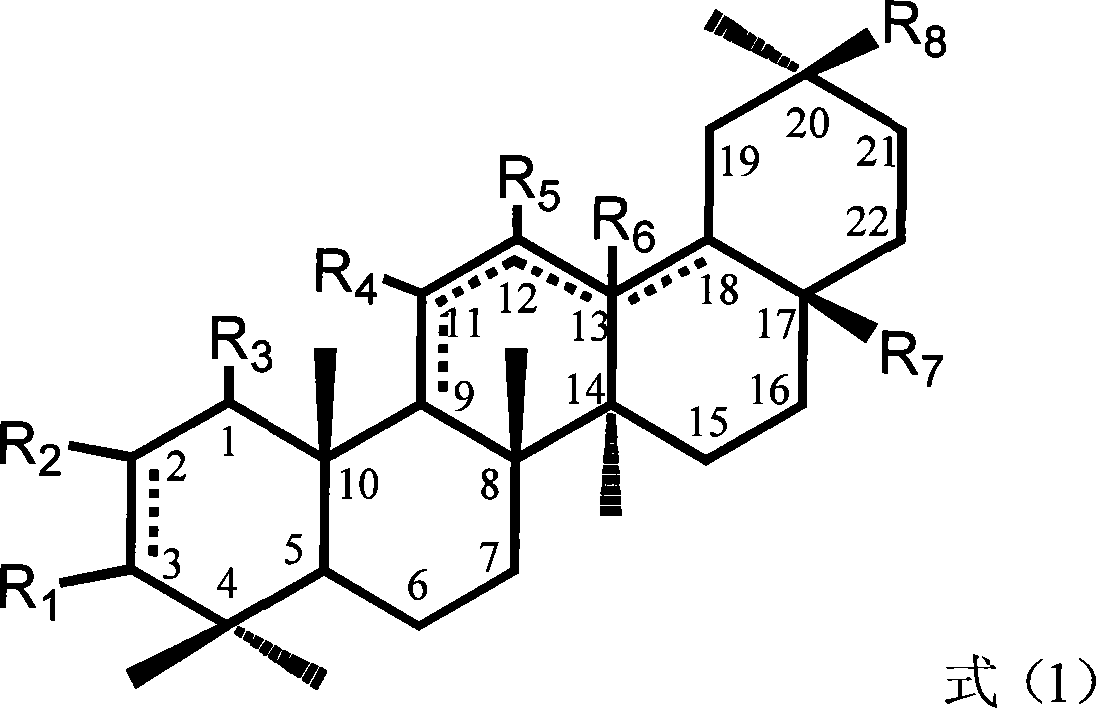
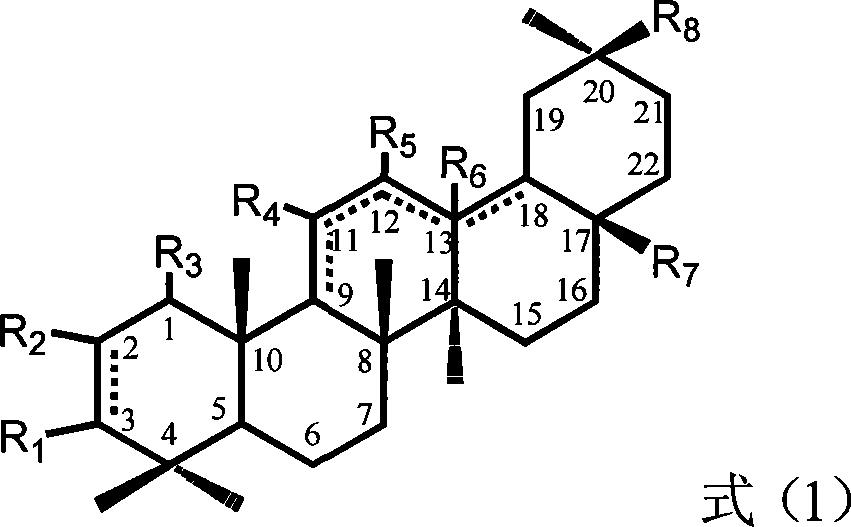
The invention relates to the field of pharmacy, in particular to a synthesis method of 13 (18)-oleanane-type pentacyclic triterpene as shown in a formula II and derivatives or pharmaceutically acceptable salts or esters of the 13 (18)-oleanane-type pentacyclic triterpene by 12-oleanane-type pentacyclic triterpene as shown in a formula I and derivatives of the 12-oleanane-type pentacyclic triterpene. Definitions of R1-9 are as shown in specification.
Owner:CHINA PHARM UNIV
Polysubstitution hydrogenated naphthalene compounds, producing method and uses of the same
The present invention belongs to the field of organic chemistry and relates to a multi-substituted hydrogenated naphthalene compound, the preparation methods and uses. Specifically, the present invention relates to a chiral multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound, the synthetic method and uses. The present invention aims to provide a chiral multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound; the oleanane-type or usu-type five-ring triterpenoid compound is used as a raw material for preparing the compound. The multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound of the present invention can be used for synthesis of drugs or spices containing multi-hydrogen naphthalene fragments and the analogues. The method of the present invention is simple and easy, low in cost, high in production rate, and can realize industrialization.
Owner:CHENGDU INST OF BIOLOGY CHINESE ACAD OF S
Pentacyclic triterpene saponin and application thereof
InactiveCN102453074AImprove protectionPrevention and treatment of senile dementiaOrganic active ingredientsNervous disorderDiseaseStructural formula
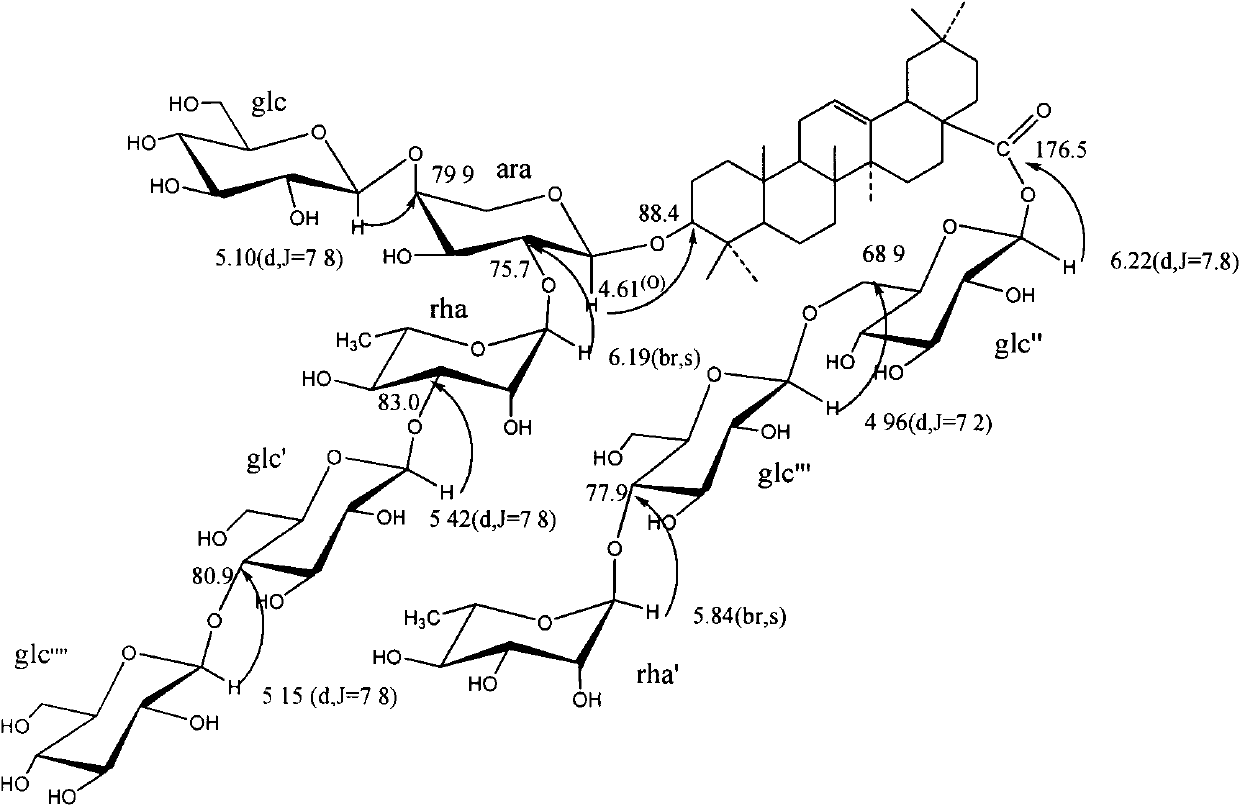
The invention relates to the medical technical field, in particular to a pentacyclic triterpene compound which is separated to be obtained from a ranunculaceae plant of pulsatilla cernua (Thunb.) Bercht.et Opiz, and the chemical structural formula is shown in the accompanying drawing, wherein glc, rha and ara respectively represent beta-D-glucopyranose, alpha-L-pyrane rhamnose and alpha-L-pyrane arabinose, and the compounds A and B are respectively a new compound. Cell experiments in vitro show that the compound has a remarkable protection function to neuroblastoma cell strain SH-SY5Y induced by A beta (25-35). The compound disclosed by the invention has the advantages of simpleness in preparation and remarkable activity. The invention provides the new compound for preventing and curing AD (Alzheimer's Disease). In the invention, the glc, rha and ara respectively represent beta-D-glucopyranose, alpha-L-pyrane rhamnose and alpha-L-pyrane arabinose.
Owner:SHENYANG PHARMA UNIVERSITY
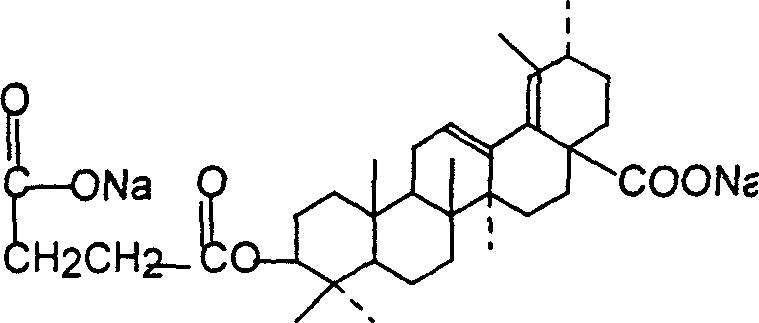
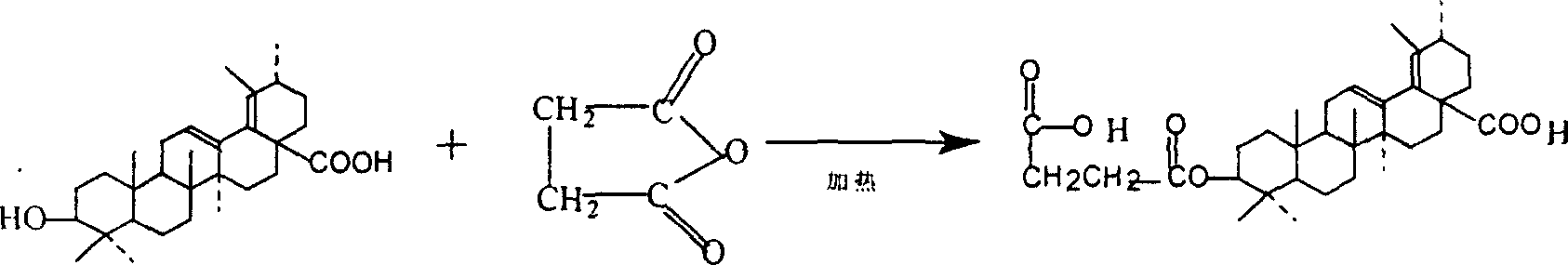
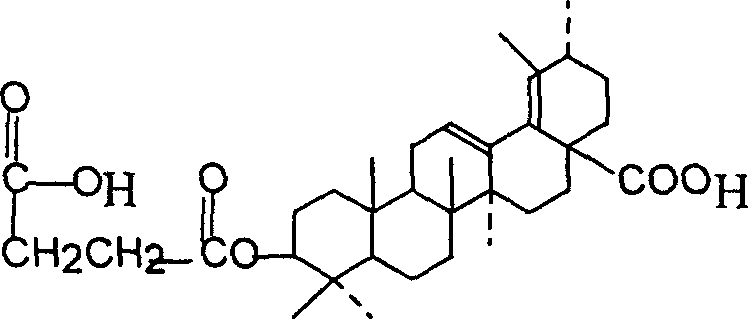
Preparation method of 3š�-succinyl-18-disodium salt dehydro ursolic acid
A process for preparing 3 beta-succinyl-18-disodium dehydroursolate includes reaction between 18-dehydroursolic acid and succinic acid anhydride in alkaline organic solvent at 80-202 deg.C for 5-12 hr, adding diluted acid, filter, water washing to remove pyridine, dissolving in C1-14 emtrol or ethyl acetate, adding sodium hydroxide solution, filter, recrystallizing and drying. Its advantage is high purity (more than 95%).
Owner:GUANGZHOU HANFANG PHARMA
Preparation method and application of derivatives of pentacyclic triterpene structural compound
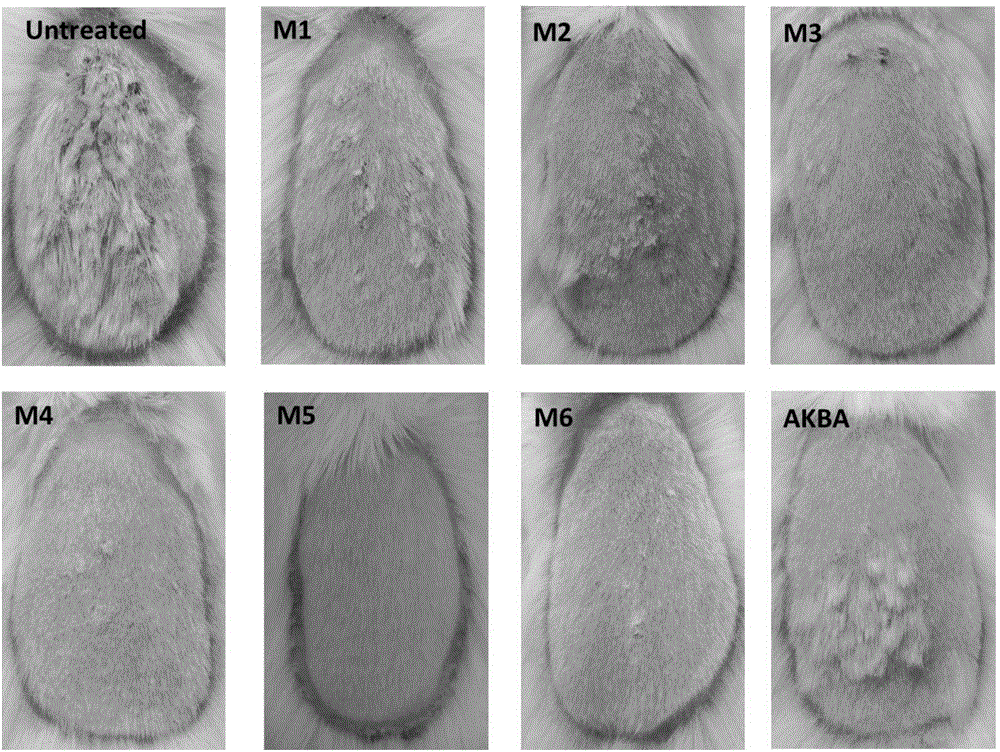
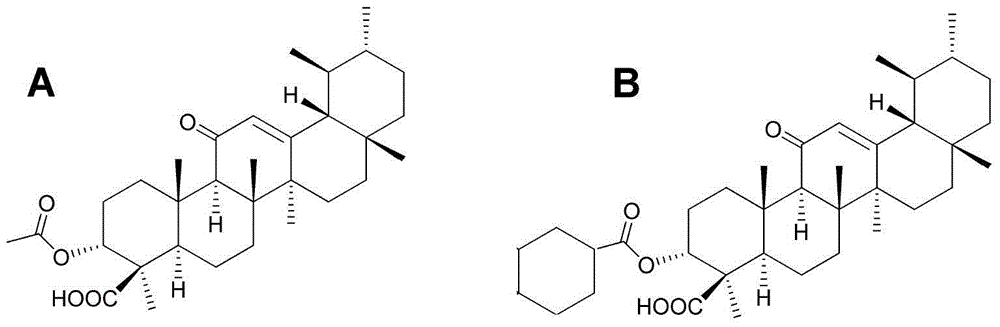
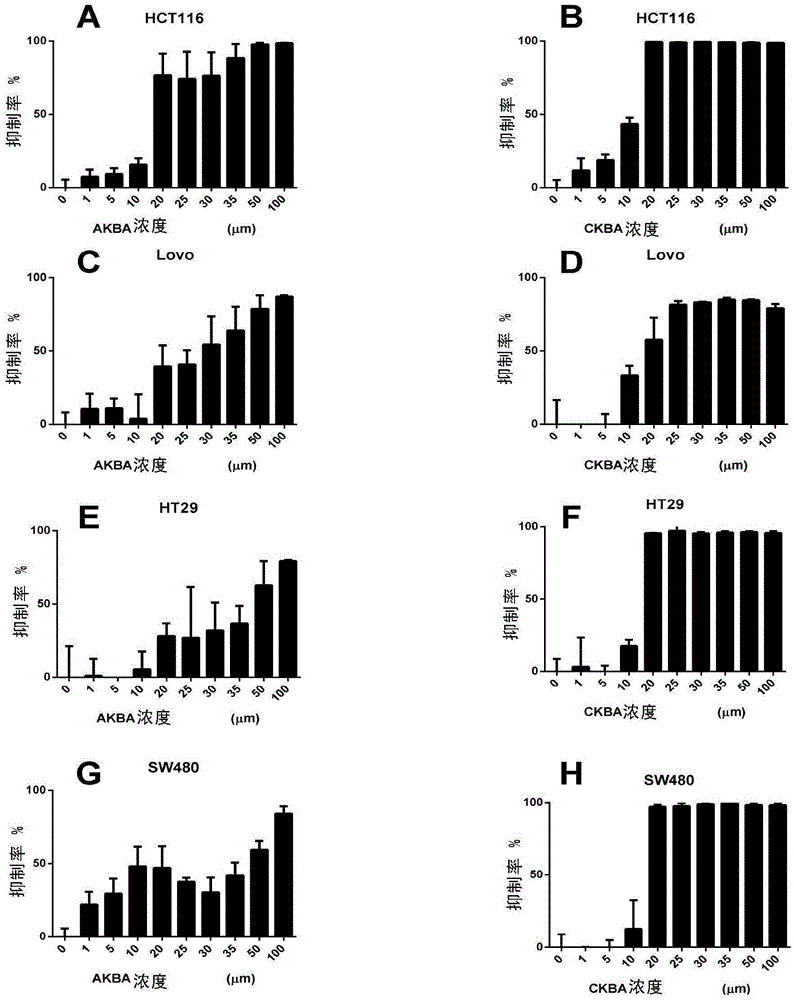
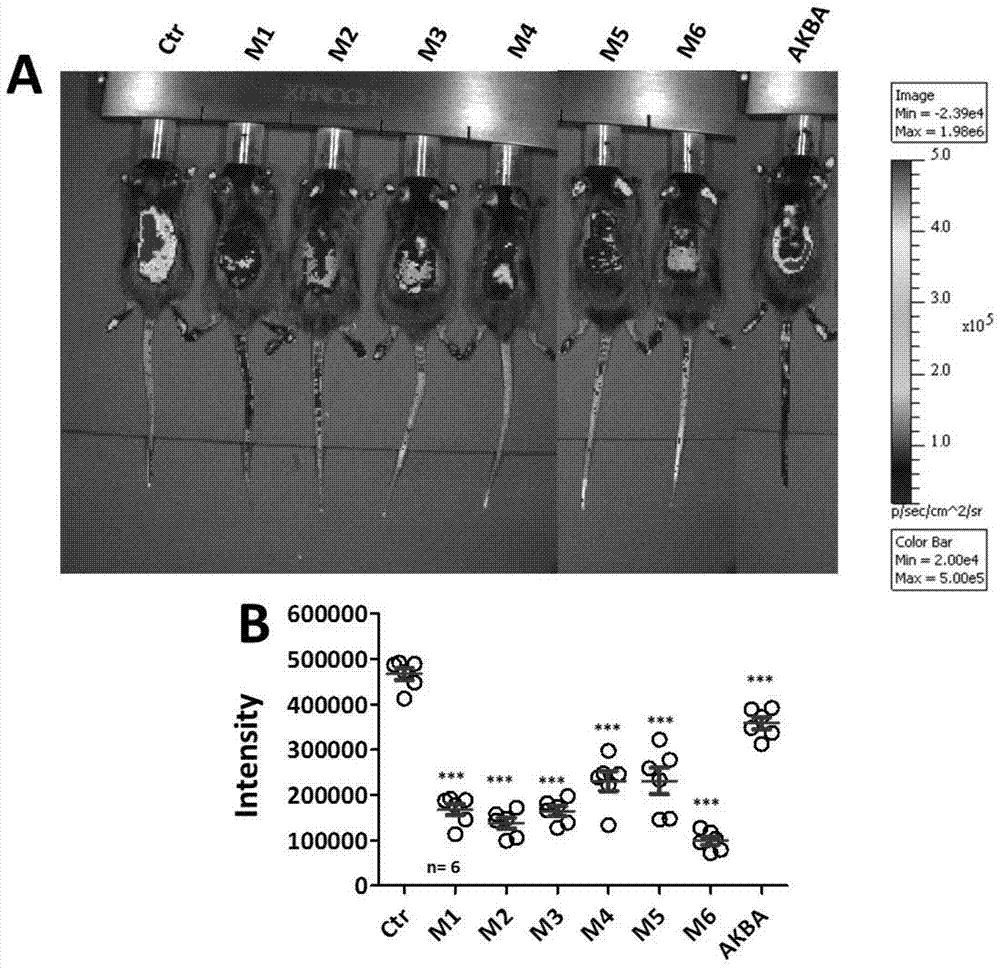
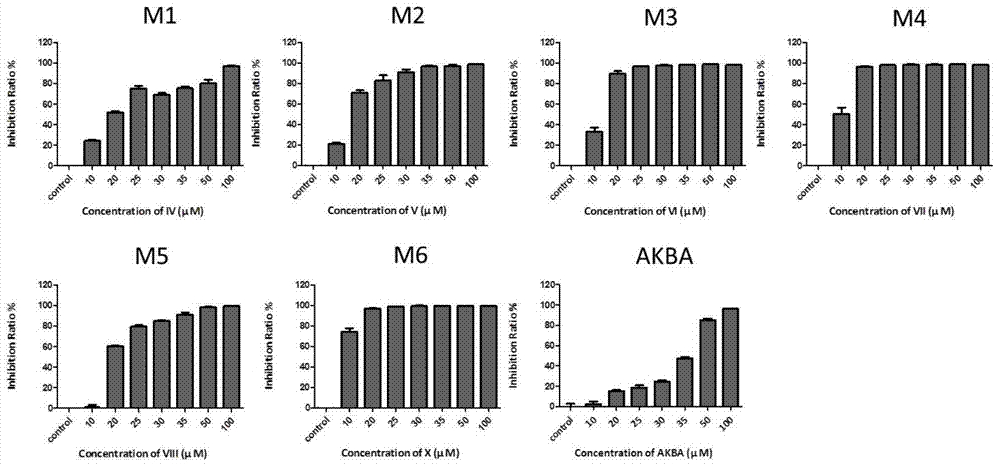
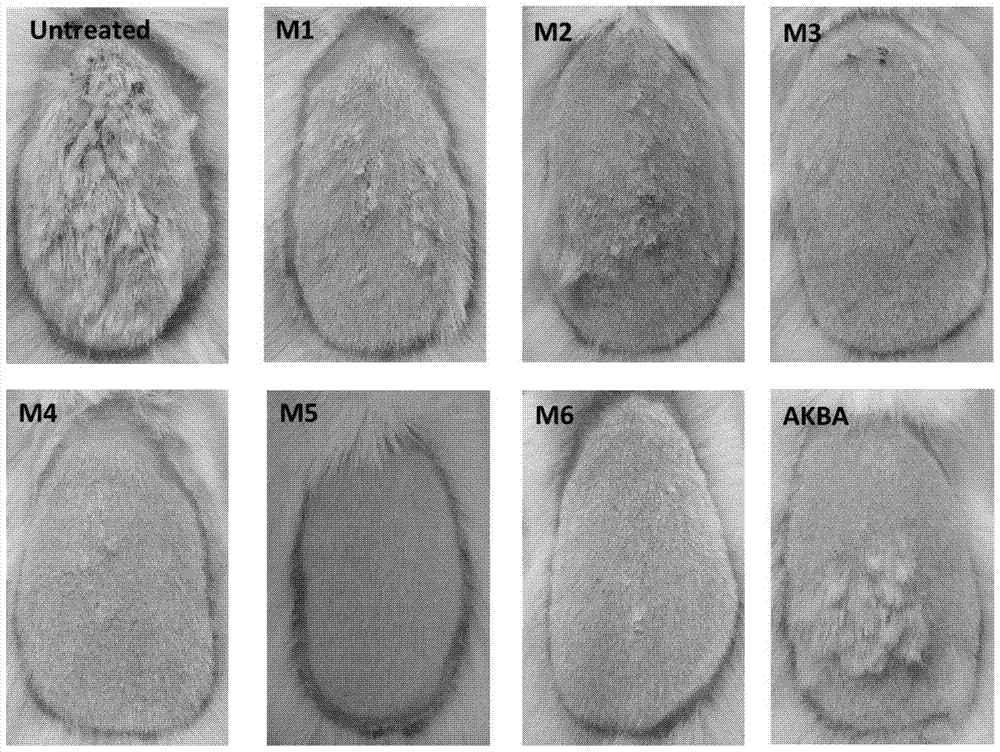
ActiveCN104974213AInhibitory activityReduce skin thicknessOrganic active ingredientsSteroidsBeta-boswellic acidEsterification reaction
The invention relates to six new compounds for treating psoriasis, a preparation method and an application thereof. The six new compounds belong to a series of derivatives from a mother compound: acetyl-11-keto-[beta]-boswellic acid (AKBA) through modification of a hydrolysis reaction and an esterification reaction. Compared with the mother compound AKBA before chemical modification, the six compound, of which the structure are represented as the formulas (II-VII), can inhibit division and cell proliferation of keratinocyte in in-vitro experiments and can treat the psoriasis on mice more effectively in a live animal test. The new compounds can be used for preparing the medicine for treating the psoriasis.
Owner:SHANGHAI JIAOTONG UNIV SCHOOL OF MEDICINE
Method for synthesizing 3-O-glucose-based oleanolic acid and cellobiose oleanolic acid by using saccharomyces cerevisiae
ActiveCN106318966AIncrease productionThe fermentation process is simpleFungiMicroorganism based processesSolubilityUdp glucosyltransferase
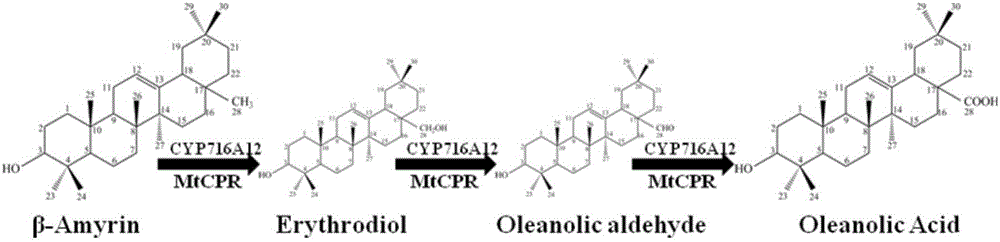
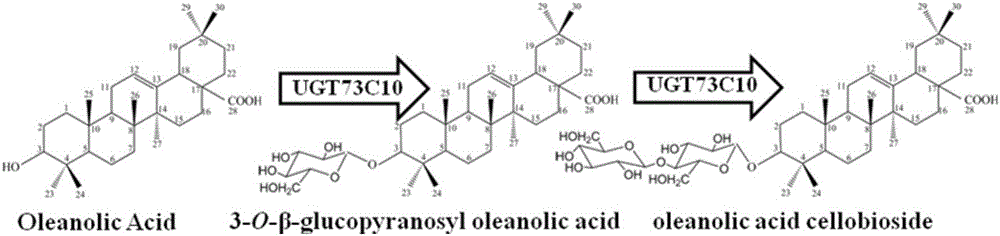
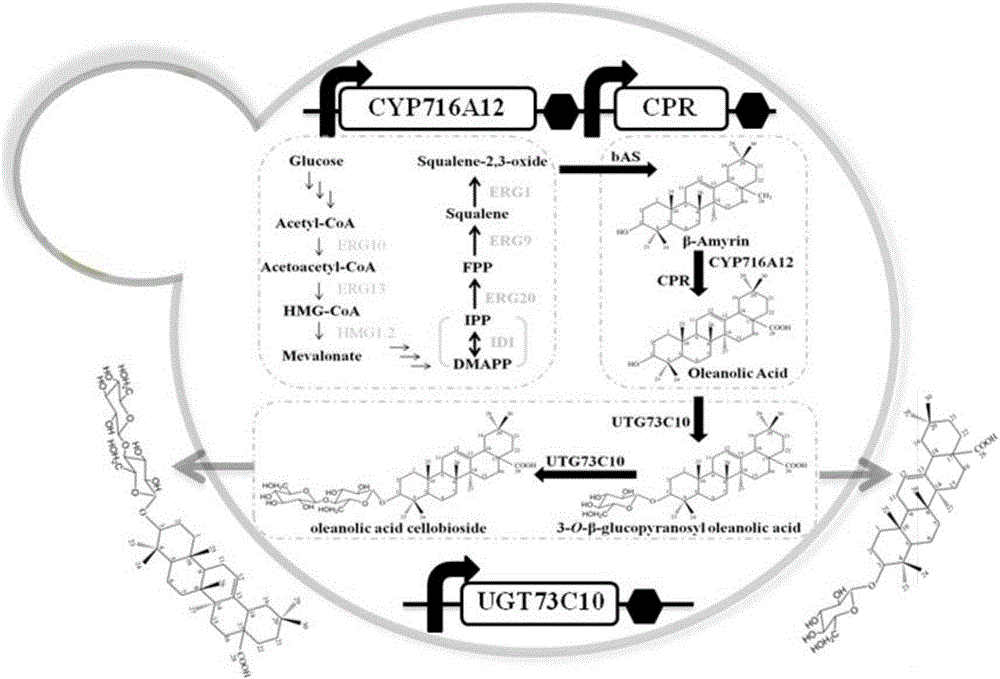
The invention provides a method for synthesizing 3-O-glucose-based oleanolic acid and cellobiose oleanolic acid by using saccharomyces cerevisiae engineering bacteria, and belongs to the field of bioengineering. The method comprises the following steps: synthesizing a codon optimized P450 cytochrome monooxygenase gene, a cytochrome reductase gene and a UDP-glucosyltransferase gene by a chemical method; constructing corresponding gene expression boxes by combining a saccharomyces cerevisiae promoter with a terminator; constructing gene expression vectors by a DNA (Deoxyribonucleic Acid) klenow fragment assembling method, and importing the gene expression vectors into the saccharomyces cerevisiae capable of producing beta-amyrin. Direct synthesis of the 3-O-glucose-based oleanolic acid and the cellobiose oleanolic acid serving as plant secondary metabolites in the saccharomyces cerevisiae is realized for the first time; in addition, two synthesized compounds can span cytomembrane of the saccharomyces cerevisiae engineering bacteria, and a downstream separation and extraction process is simplified, so that a new idea is provided for producing pentacyclic triterpene compounds with low water solubility and difficulty in spanning membranes by using the saccharomyces cerevisiae. The method is simple in process and can be used for producing the 3-O-glucose-based oleanolic acid and the cellobiose oleanolic acid by fermenting.
Owner:BEIJING INSTITUTE OF TECHNOLOGYGY
Ursolic acid and amine cyclodextrin clathrate compound
InactiveCN104857003ATargeted optimizationImprove securityOrganic active ingredientsPharmaceutical non-active ingredientsSolubilityUrsolic acid
The invention discloses a ursolic acid and amine cyclodextrin clathrate compound. The amine cyclodextrin is an amino-substituted beta-cyclodextrin, and can generate clathration with ursolic acid; and the amino on the amine cyclodextrin and the carboxyl on ursolic acid can produce ionic interaction, thus greatly improving the water solubility of ursolic acid, and facilitating the formation of a liquid ursolic acid preparation. The clathrate compound prepared by the method provided by the invention has good water-solubility, good stability, and good anti-tumor activity in vitro. Meanwhile, the preparation method is simple, mild in conditions and easy to operate, can be used to develop novel ursolic acid preparation and has important practical value.
Owner:KUNMING UNIV OF SCI & TECH
Diene oleanolic acid pentacyclic triterpenes derivatives and use thereof
The invention provides a diene oleanolic acid pentacyclic triterpenoid derivative, relative medicine salt or solvate, represented as formula (1). The inventive compound can inhibit the growth activity of tumor cell to be applied in anti-tumor drug and can inhibit mice ear swelling caused by dimethylbenzene to be applied in anti-inflammatory agent. The formula (1) is represented as above.
Owner:ZHEJIANG UNIV
Pentacyclic triterpenes compound with ACC1 protein regulation effect and use of pentacyclic triterpenes compound
The invention relates to a pentacyclic triterpenes compound with an ACC1 protein regulation effect and a use of the pentacyclic triterpenes compound. The compound can directly act on ACC1 proteins to play an effect of inhibiting the tumor growth, and the inhibition effect is significant.
Owner:SUZHOU BOTANY BIOMEDICALS
Pentacyclic triterpenoid compound with modified structure and preparation method and use thereof
ActiveUS9725482B2Efficacy of treatmentInhibition of activationSteroidsImmunological disordersAutoimmune conditionMedicine
The present invention relates to new pentacyclic triterpenes, their preparation method and use. The compounds of the present invention could effectively treat psoriasis and selectively inhibit in vitro differentiation of the TH1 and TH17 cells, thereby could be used to treat the TH1- or TH17-mediated autoimmune diseases.
Owner:SUZHOU BOTANY BIOMEDICALS
Pentacyclic triterpene saponin compounds with anti-breast cancer activity in spina gleditsiae and extraction method thereof
ActiveCN107383151ASignificant anti-breast cancer activitySteroidsSexual disorderGradient elutionEthyl acetate
The invention discloses pentacyclic triterpene saponin compounds with anti-breast cancer activity in spina gleditsiae and an extraction method thereof. The extraction method comprises the following steps: (1) grinding spina gleditsiae, and using an organic extracting agent to carry out heating reflux extraction and concentration to obtain a crude extract of spina gleditsiae; (2) dispersing the crude extract obtained in the step (1) into water, extracting the crude extract by petroleum ether and ethyl acetate in sequence to obtain a water phase, and concentrating the water phase to obtain a water phase part; (3) diluting obtained water phase part by water, making diluted water phase part go through an adsorbent resin chromatographic column to carry out adsorption, then eluting the adsorbent resin chromatographic column by an ethanol-water system in a gradient elution mode, and concentrating each elution part until no water is left; (4) making the 60% ethanol-water elution part go through a silica gel chromatographic column to carry out adsorption, and eluting the silica gel chromatographic column by a CH2Cl2-MeOH system in a gradient elution mode; and (5) making the elution part with a volume ratio of CH2Cl2 to MeOH of 1:1 go through a RP-C18 medium pressure chromatographic column, eluting the RP-C18 medium pressure chromatographic column by a methanol-water system in a gradient elution mode, and saving the 80% methanol-water elution part to prepare spina gleditsiae saponin A by using CH3CN / H2O (26:74 v / v).
Owner:SHANDONG ANALYSIS & TEST CENT
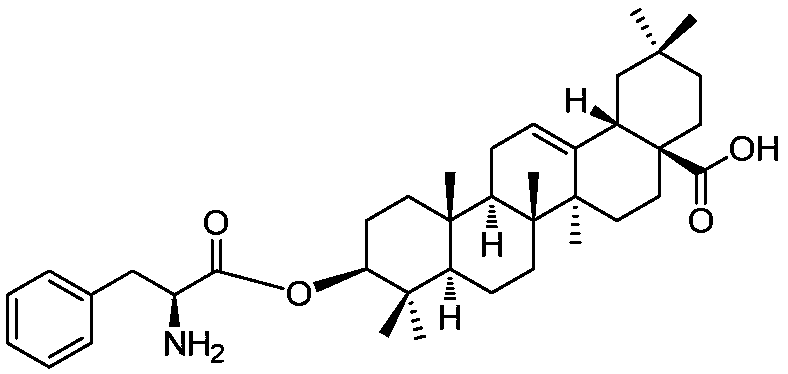
3-(L-phenylalanine)-pentacyclic triterpene derivatives as well as synthetic method and application thereof
InactiveCN109503697AStrong antiproliferative activityGood potential medicinal valueOrganic active ingredientsSteroidsSynthesis methodsIn vitro test
The invention discloses a series of 3-(L-phenylalanine)-pentacyclic triterpene derivatives as well as a synthetic method and an application thereof. The pentacyclic triterpene derivatives comprise two3-(L-phenylalanine)-oleanolic acid derivatives and two 3-(L-phenylalanine)-glycyrrhetinic acid derivatives, specially, one 3-(L-phenylalanine)-oleanolic acid derivative or one 3-(L-phenylalanine)-glycyrrhetinic acid derivative is obtained from oleanolic acid / glycyrrhetinic acid, Boc-L-phenylalanine, dicyclohexylcarbodiimide and 4-dimethylaminopyridine by a reaction and subjected to a reaction with trifluoroacetic acid, and the other two derivatives are obtained. In-vitro test results prove that the 3-(L-phenylalanine)-pentacyclic triterpene derivatives have good proliferation inhibition activity on certain tumor cell lines, have better potential medicinal values and are expected to be applied to preparation of various antitumor drugs.
Owner:GUANGXI NORMAL UNIV
Preparation method and application of pentacyclic triterpene derivatives
ActiveCN104974213BInhibitory activityReduce skin thicknessOrganic active ingredientsSteroidsBeta-boswellic acidEsterification reaction
The invention relates to six new compounds for treating psoriasis, a preparation method and an application thereof. The six new compounds belong to a series of derivatives from a mother compound: acetyl-11-keto-[beta]-boswellic acid (AKBA) through modification of a hydrolysis reaction and an esterification reaction. Compared with the mother compound AKBA before chemical modification, the six compound, of which the structure are represented as the formulas (II-VII), can inhibit division and cell proliferation of keratinocyte in in-vitro experiments and can treat the psoriasis on mice more effectively in a live animal test. The new compounds can be used for preparing the medicine for treating the psoriasis.
Owner:SHANGHAI JIAOTONG UNIV SCHOOL OF MEDICINE
Application of pentacyclic triterpene compounds to preparation of medicine for treating adiposis
ActiveCN106349318AEnhanced inhibitory effectReduced Risk of Nonalcoholic Fatty Liver DiseaseMetabolism disorderSteroidsOrlistatIrritation
The invention discloses application of pentacyclic triterpene compounds to preparation of medicine for treating adiposis and weight losing functional food. The pentacyclic triterpene compounds comprise pedunculoside and rotundic acid. The compounds have higher inhibition effect on lipase. Compared with synthetic medicine of Orlistat, the pentacyclic triterpene compounds have the advantages that the effect is mild; no irritation effect exists on the gastrointestinal tracts; the medicine is suitable for being taken for a long time.
Owner:SUN YAT SEN UNIV
Polyglycosidated pentacyclic triterpene-28-acid, and preparation method and application thereof
InactiveCN105175477AGood water solubilityMild effectSugar derivativesMetabolism disorderSolubilitySide effect
The invention discloses a polyglycosidated pentacyclic triterpene-28-acid, and a preparation method and an application thereof. The water solubility of polyhydroxypentacyclic triterpene-28-acid is further enhanced on the basis of maintaining or improving the activity of the polyhydroxypentacyclic triterpene-28-acid, and a plurality of glycosyl groups are introduced to the hydroxyl position of polyhydroxypentacyclic triterpene-28-acid and are glycosidated to synthesize a series of the polyglycosidated pentacyclic triterpene-28-acid with good water solubility. Researches show that like compounds have good in vitro alpha-glucosidase inhibition activity. The invention provides a method for screening an alpha-glucosidase inhibitor with the advantages of good water solubility, high inhibition activity, mild effects and small toxic and side effects. The screening method can be widely used in subsequent anti-diabetic clinic fields.
Owner:JIANGXI AGRICULTURAL UNIVERSITY
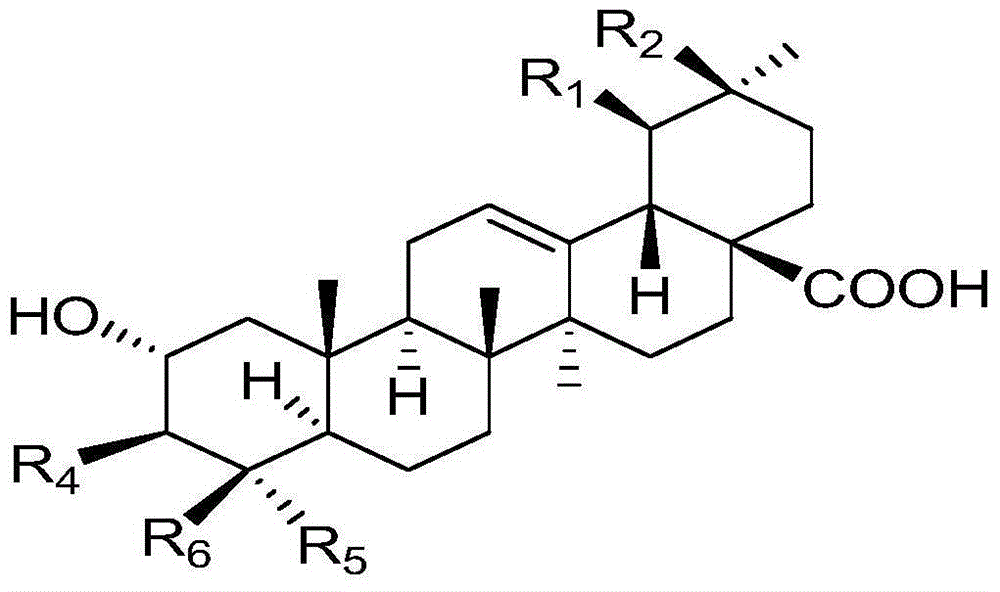
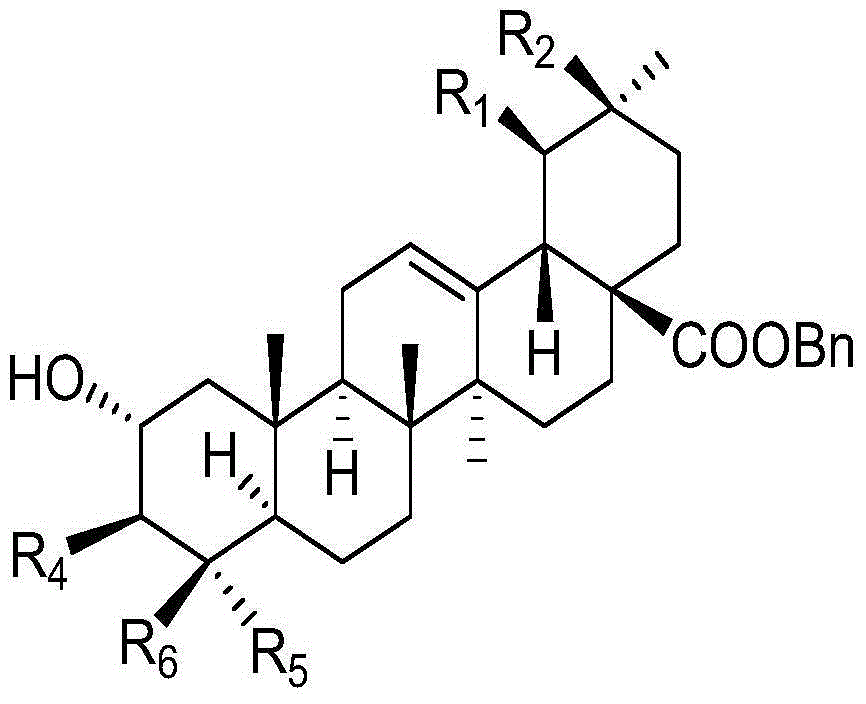
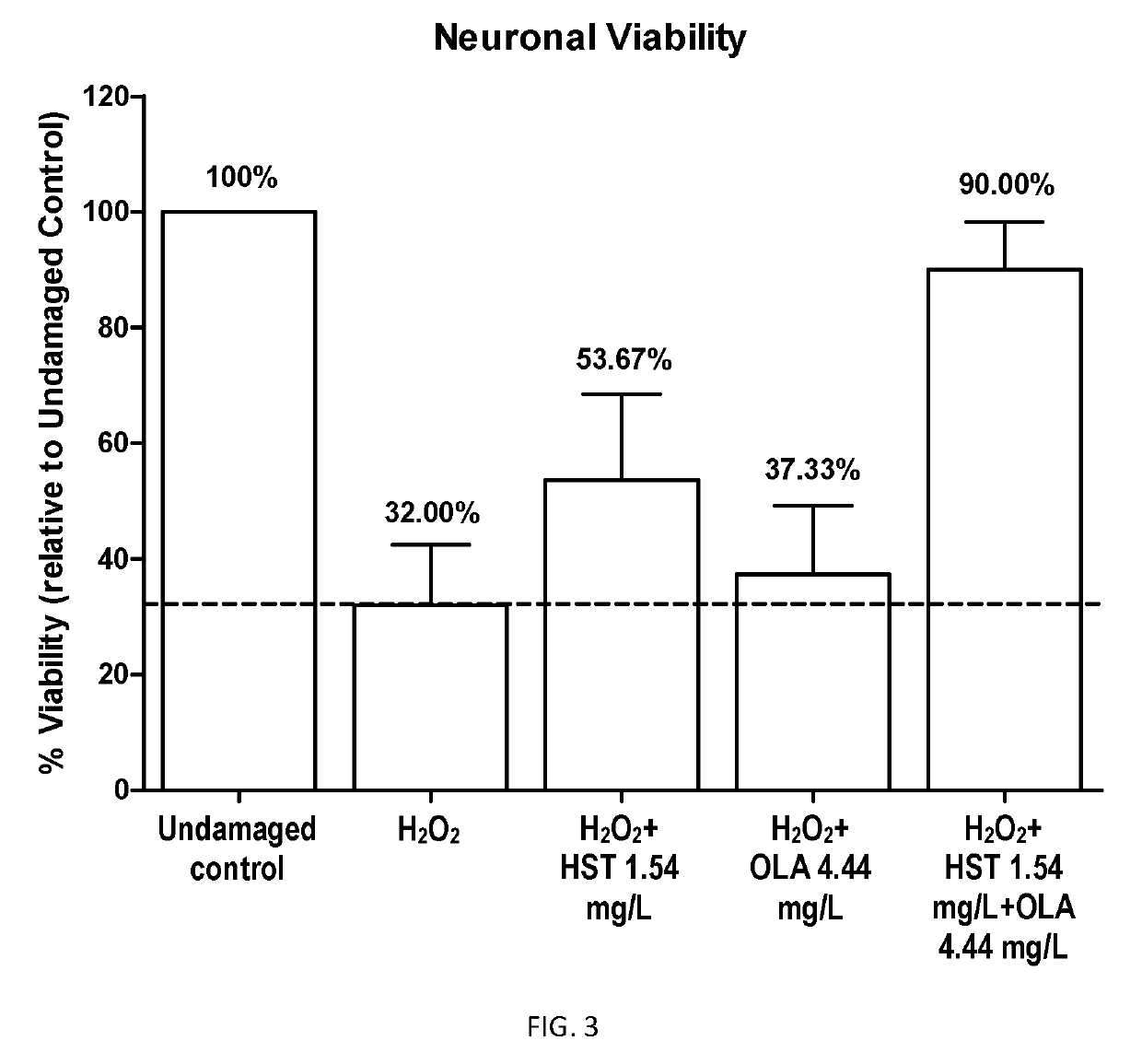
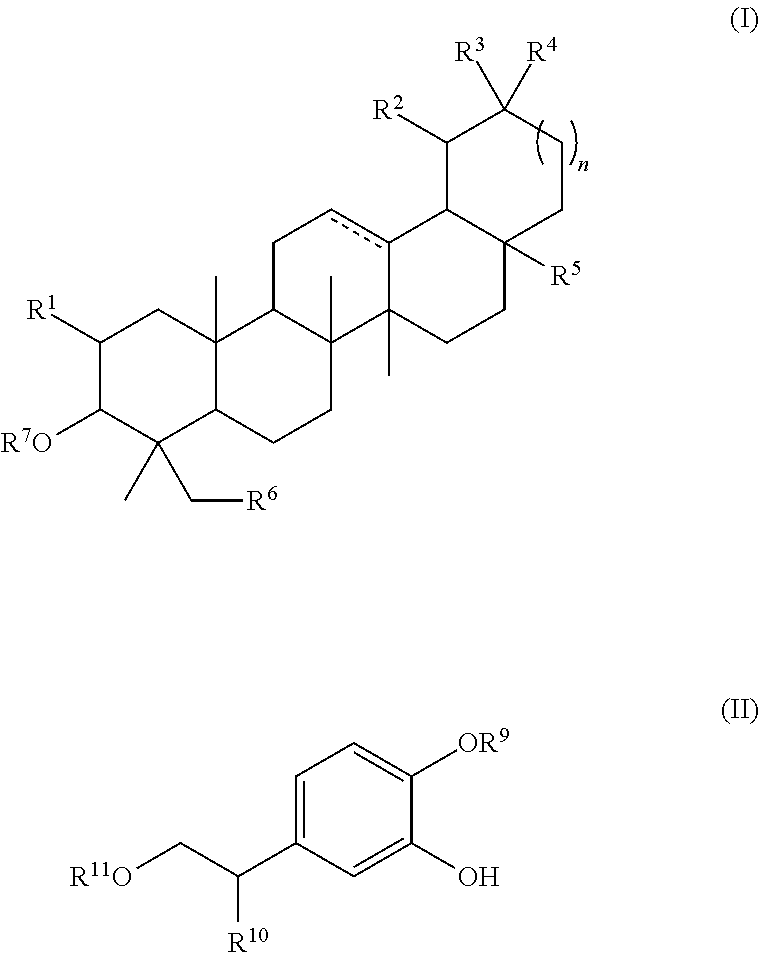
Combination of pentacyclic triterpenes and hydroxytyrosol and derivatives thereof
The present invention is related to a product comprising a pentacyclic triterpene and hydroxytyrosol or derivatives thereof, uses, and pharmaceutical composition thereof.
Owner:NATAC BIOTECH
Preparation of 3beta-succinyl-18-disodium salt dehydro ursolic acid
InactiveCN1216066CThe synthesis process is simpleEasy to separate and purifySteroidsUrsolic acidSuccinic acid
A process for preparing 3 beta-succinyl-18-disodium dehydroursolate includes reaction between 18-dehydroursolic acid and succinic acid anhydride in alkaline organic solvent at 80-202 deg.C for 5-12 hr, adding diluted acid, filter, water washing to remove pyridine, dissolving in C1-14 emtrol or ethyl acetate, adding sodium hydroxide solution, filter, recrystallizing and drying. Its advantage is high purity (more than 95%).
Owner:GUANGZHOU HANFANG PHARMA
Nanoparticle mediated therapy
PendingUS20220168230A1Induce hypoglycemiaImproved mouse survivalSulfonylurea active ingredientsPharmaceutical non-active ingredientsNatural sourcePlant sterol
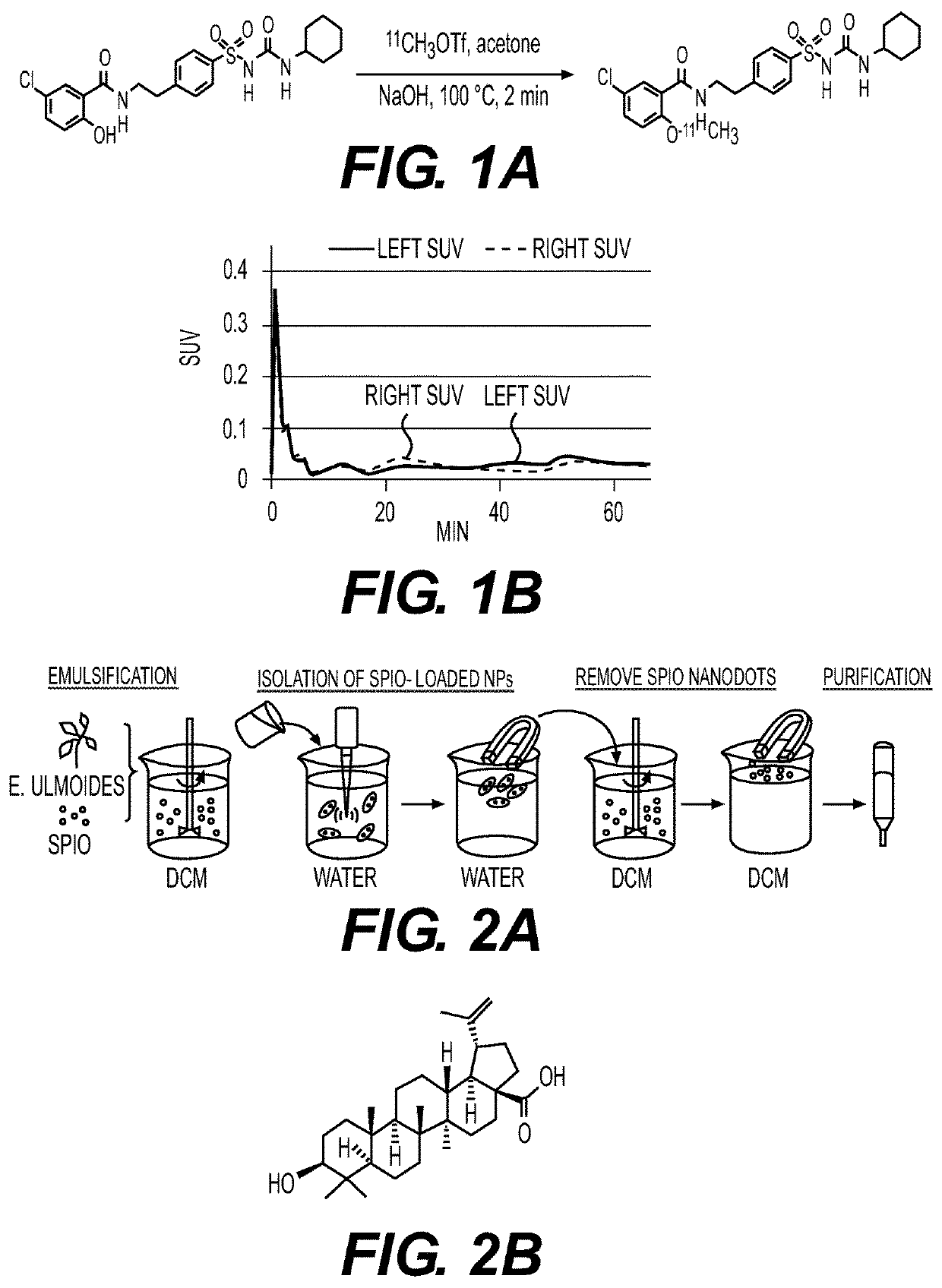
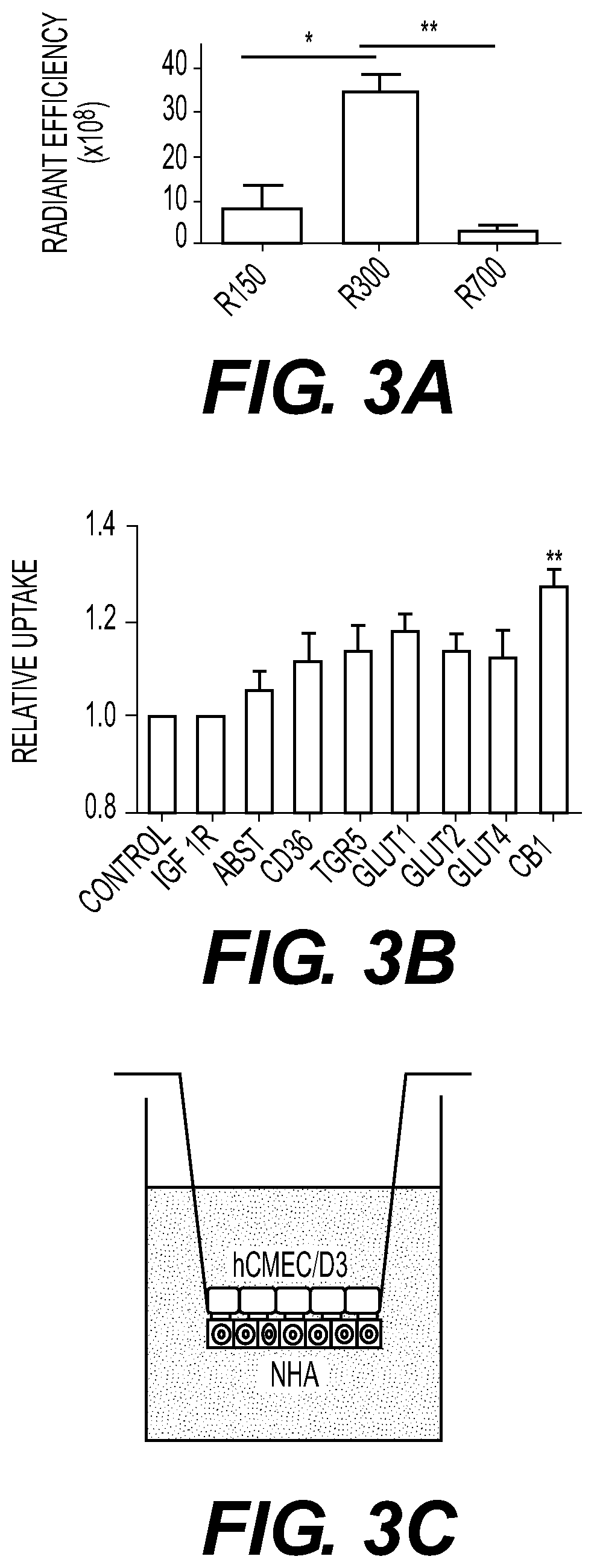
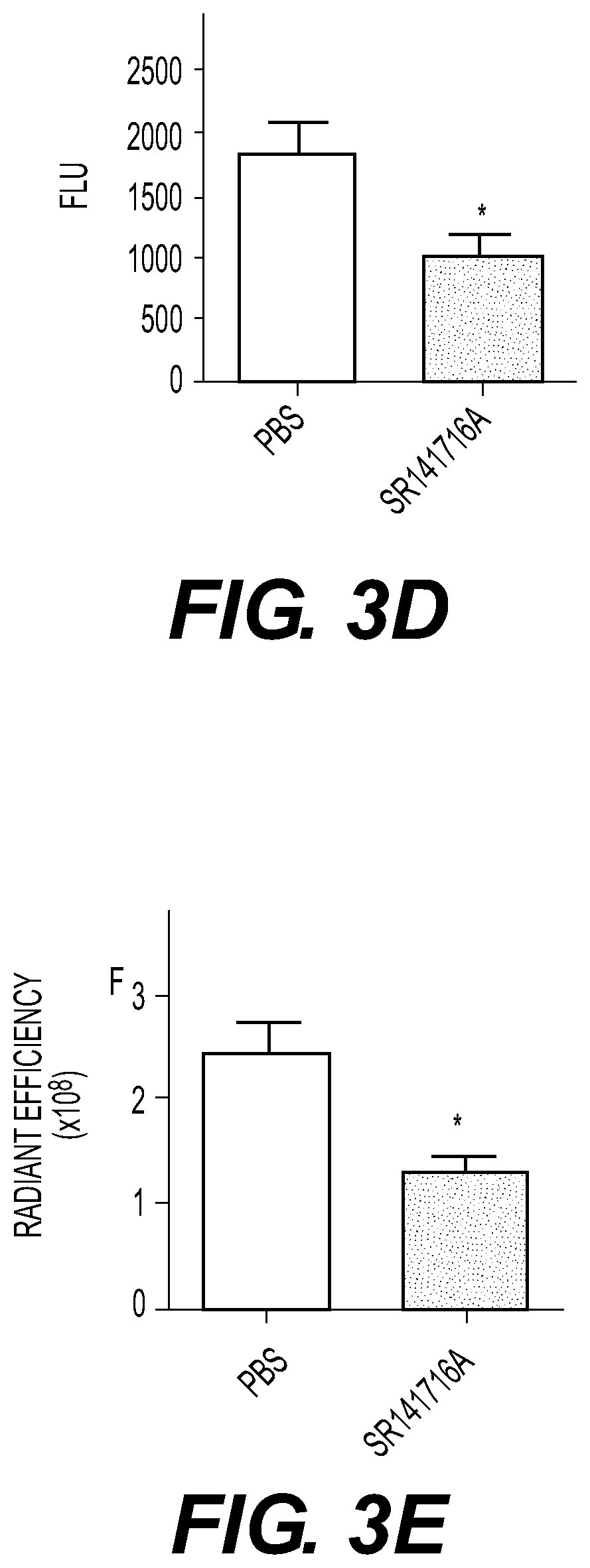
At least five classes of MNP-based compounds have been demonstrated to form supramolecular particles for effective delivery by injection or topically of different types of therapeutic, prophylactic, or diagnostic agents. These compounds are isolated from natural sources such as plants. Exemplary MNP-based compounds, from which synthetic analogs or derivatives are made and appreciated to function similarly, e.g., capable of forming supramolecular particles include diterpene resin acids (e.g., abietic acid and pimaric acid), phytosterols (e.g., stigmasterol and β-sitosterol), lupane-type pentacyclic triterpenes (e.g., lupeol and betulinic acid), oleanane-type pentacyclic tritepenes (e.g., glycyrrhetic acid and sumaresinolic acid), and lanostane-type triterpenes and derivatives (e.g., dehydrotrametenolic acid and poricoic acid A). In some cases the MNP-based compounds are therapeutically effective in the absence of added therapeutic, prophylactic or diagnostic agent. Betulinic acid (BA) NPs were capable of efficiently penetrating ischemic brains and effectively promoting functional recovery as antioxidant agents.
Owner:YALE UNIV
Combination of pentacyclic triterpenes and hydroxytyrosol and derivatives thereof
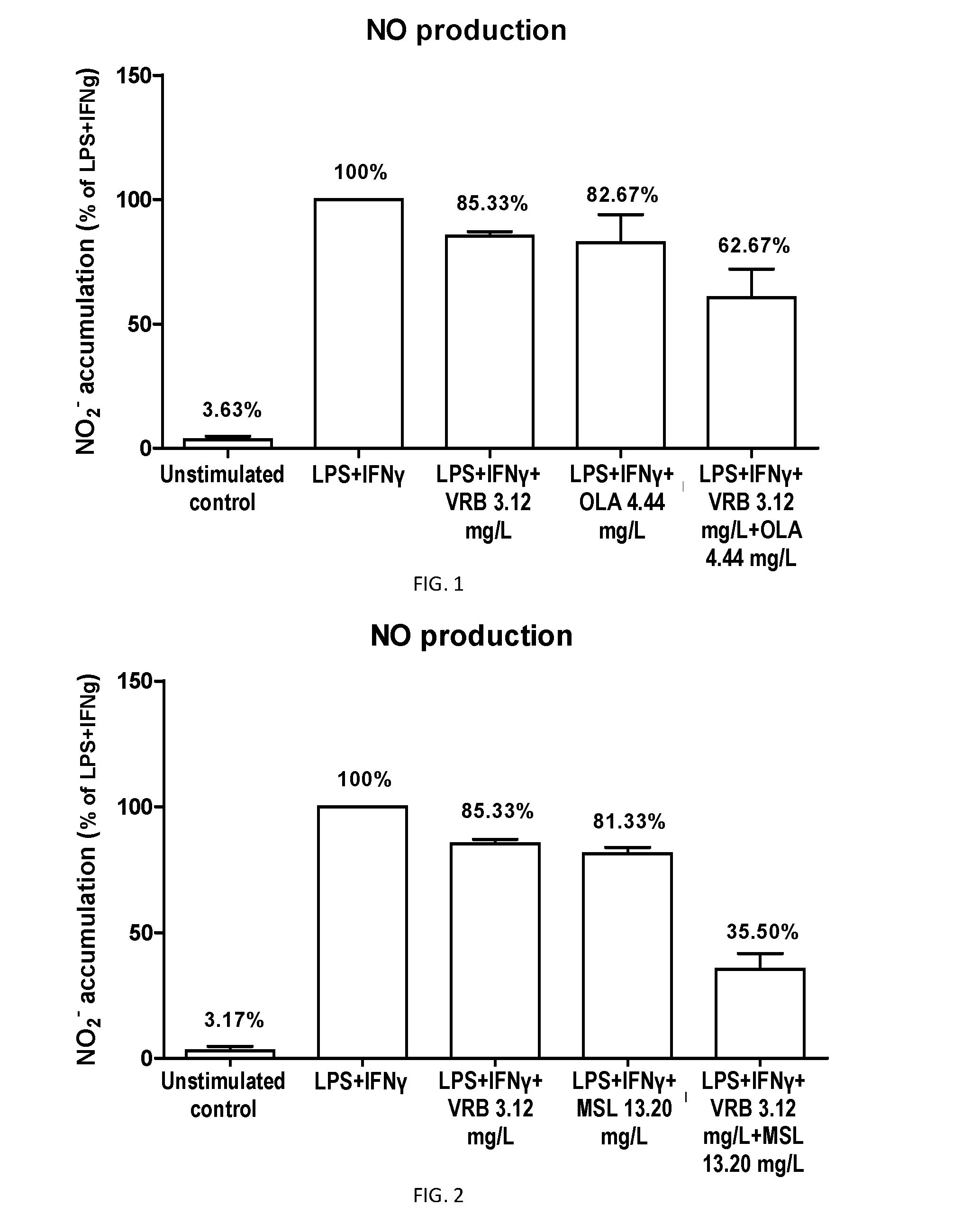
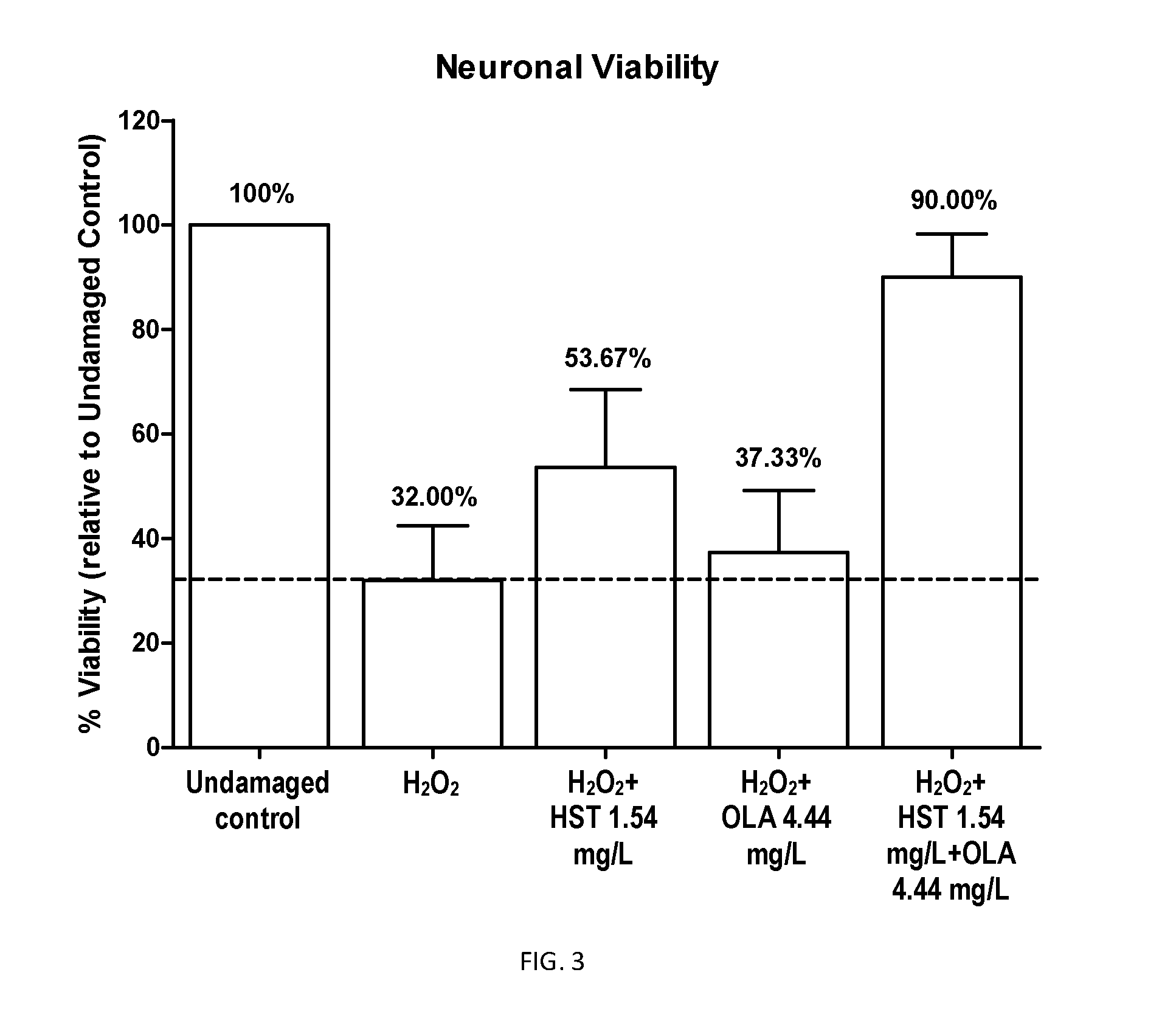
The present invention is related to a product comprising a pentacyclic triterpene of formula (I) and a hydroxytyrosol or derivatives thereof of formula (II), pharmaceutical compositions thereof and their uses as antioxidants, anti-inflammatories and neuron protectors.
Owner:NATAC BIOTECH
Fast isolation method for the natural scaffold ursolic acid from diospyros melanoxylon
The present invention relates to a process for the fast isolation of ursolic acid, a highly potent natural scaffold, from the leaves of Diospyros melanoxylon. The present invention also provides an improved and fast isolation process of the title compound, which is a pentacyclic triterpenic acid highly useful for the synthesis of a wide range of novel and potent bio-active molecules.
Owner:COUNCIL OF SCI & IND RES
Method for separating high-purity saponin monomers from tea seeds
The invention specifically relates to a method for separating high-purity saponin monomers from tea seeds, belonging to the field of separation and purification of natural compounds. In particular, the method involves separation and purification of six high-purity pentacyclic triterpene oleanane saponins in tea seeds. Compared with the prior art, the method provided by the invention has the following beneficial effects: (a) the purity of the compounds are high, as high as 91 to 99%; (b) separation process is simple, and six main monomeric saponin substances in tea seeds can be obtained at thesame time; and (c) the method has good repeatability and is well targeted.
Owner:ZHEJIANG UNIV +1
Application of a ring trihydroxyl substituted pentacyclic triterpene compound in pharmacy
ActiveCN103919784BStrong inhibitory activityGood treatment effectAntibacterial agentsOrganic active ingredientsTobacco mosaic virusPhytophthora sp.
The invention discloses an application of an A-ring trihydroxyl substituted pentacyclic triterpene compound to preparation of an antibacterial or anti-tobacco mosaic virus drug. The general formula of the compound is as shown in the specification, wherein hydroxyls of C1, C2 and C3 are respectively in alpha configuration or beta configuration; R1 and R2 are selected from hydrogen or methyl and are different from each other; R3 is selected from -COOH, -CH2OH, -CHO, -COOR1, -CONH2, -CONHR1 and -CONR1R2; R1 and R2 are selected from alkyl containing 1-15 carbon atoms, substituted or unsubstituted phenyl and substituted or unsubstituted phenyl alkyl; and the substituent group is selected from halogen, hydroxyl, cyan, amino, nitryl, sulfydryl or phenyl, acyl, aryl, alkoxy and alkyl containing 1-15 carbon atoms. The compounds have antibacterial or anti-tobacco mosaic virus activity, have extremely high bacteriostatic activity especially to gram positive bacteria, phytophthora nicotiana and tobacco mosaic virus, and have a good application prospect in fields of medicines and pesticides.
Owner:THE KEY LAB OF CHEM FOR NATURAL PROD OF GUIZHOU PROVINCE & CHINESE ACADEMY OF SCI
Polysubstitution hydrogenated naphthalene compounds, producing method and uses of the same
The present invention belongs to the field of organic chemistry and relates to a multi-substituted hydrogenated naphthalene compound, a synthesis method and use thereof. In particular, the present invention relates to a chiral multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound, the synthetic method and uses. The present invention aims to provide a chiral multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound; the oleanane-type or usu-type five-ring triterpenoid compound is used as a raw material for preparing the compound. The multi-substituted ten-hydrogen and / or eight-hydrogen naphthalene compound of the present invention can be used for synthesis of drugs or spices containing multi-hydrogen naphthalene fragments and the analogues. The method of the present invention is simple and easy, low in cost, high in production rate, and can realize industrialization.
Owner:CHENGDU INST OF BIOLOGY CHINESE ACAD OF S
Pentacyclic triterpenes in treatment of vitiligo
The present invention relates to a composition comprising at least one pentacyclic triterpene or one of the pharmaceutically acceptable salts thereof for use in the treatment and / or prevention of vitiligo.
Owner:PIERRE FABRE DERMO COSMETIQUE CORP
28-(L-phenylalanine)-pentacyclic triterpene derivatives as well as synthesis methods and application thereof
InactiveCN109517025AStrong antiproliferative activityGood potential medicinal valueOrganic active ingredientsSteroidsSynthesis methodsAcid derivative
The invention discloses a series of 28-(L-phenylalanine)-pentacyclic triterpene derivatives as well as synthesis methods and application thereof. The pentacyclic triterpene derivatives particularly comprises two 28-(L-phenylalanine)-oleanolic acid derivatives, two 28-(L-phenylalanine)-ursolic acid derivatives and two 28-(L-phenylalanine)-glycyrrhetinic acid derivatives. Shown by results of in vitro tests by the applicant, the derivatives have favorable proliferation inhibition activity to certain tumor cell strains, have better potential medicinal value, and are expected to be used for preparing various anti-tumor drugs.
Owner:GUANGXI NORMAL UNIV
Mitochondria-targeted anti-tumor pentacyclic triterpene derivatives, preparation method and application thereof
ActiveCN105924492BEasy to prepareProcess stabilityOrganic active ingredientsSteroidsSolubilityHydrogen
The invention discloses mitochondrion-targeted antitumor pentacyclic triterpene derivatives of which the structural formula are disclosed as Formula (I), Formula (II), Formula (III), Formula (IV) or Formula (V), wherein R1 is hydrogen, formacyl, acetyl or a group (n=1-19) disclosed in the specification; R2 is disclosed in the specification (n=1-19); R3 is hydroxy, methoxy or ethoxy; and R4 is disclosed in the specification (n=1-19). The compounds have favorable antitumor activity; and the natural compounds are targeted to the mitochondrion, so that the antitumor pentacyclic triterpene derivatives can be better applied to the development of antitumor drugs. The compounds are salts, thereby greatly enhancing the water solubility of drugs and improving the pharmacokinetic parameters.
Owner:SHANDONG UNIV
Fast isolation method for the natural scaffold ursolic acid from Diospyros melanoxylon
Owner:COUNCIL OF SCI & IND RES