Active peptide derived from Caprin1 and application thereof
An active peptide, stress technology, applied in the field of biomedicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
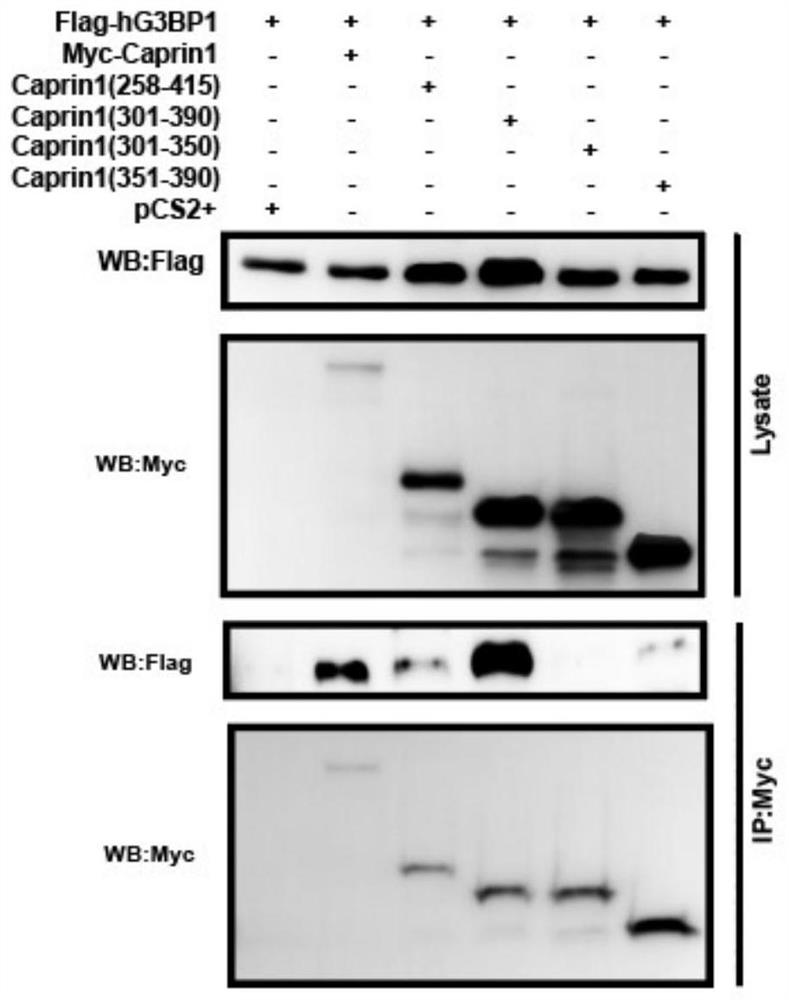
[0042] Example 1 Design of polypeptide SGs inhibitors based on blocking the interaction between Caprin1 and G3BP1
[0043] 1. According to research reports, the interaction between the two nucleating proteins Caprin1 of SGs and G3BP1 will promote the assembly of SGs. The present invention explores the minimum G3BP1 binding region of the Caprin1 protein, and designs four polypeptide fragments based on the Caprin1 protein: Caprin1(258-415), Caprin1(301-390), Caprin1(301-350), and Caprin1(351-390). Entrust Shanghai Gil Biochemical Company to synthesize the above polypeptide fragments.
[0044] 2. Construct Caprin1 and hG3BP1 expression plasmids, design and synthesize the following primers:
[0045] G3BP1:
[0046] G3BP1-F 5'-atggtgatggagaagcctagtcccctgct-3' (SEQ ID No: 14);
[0047] G3BP1-Xba-R 5'-ccatctagattcactgccgtggcgcaagcc-3' (SEQ ID No: 15);
[0048] Caprin1:
[0049] Caprin1-F 5'-atgccctcggccaccagccaca-3' (SEQ ID No: 16);
[0050] Caprin1-Xba-R 5'-tcttctagaaacatattat...
Embodiment 2
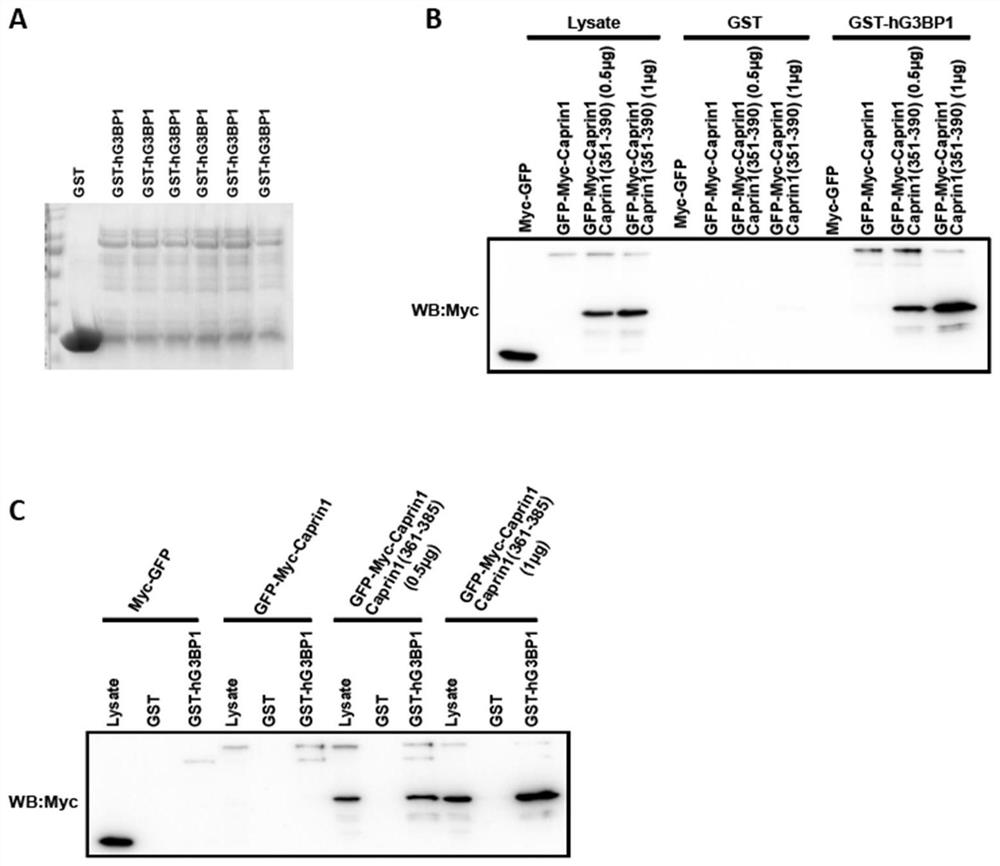
[0058] Example 2 investigates the ability of the active peptide derived from Caprin1 of the present invention to compete with the protein Caprin1 for binding to G3BP1
[0059] Based on the research results of Example 1, Caprin1 (361-385) with a shorter sequence was designed on the basis of Caprin1 (351-390).
[0060] 1. Construction of Caprin1(361-385), Caprin1(351-390) expression plasmids
[0061] The following primers were designed and synthesized:
[0062] Caprin1 (351-390):
[0063] Caprin1-351F 5'-gatccccttgtgagaagacagcgagtacaa-3' (SEQ ID No: 18);
[0064] Caprin1-Xba-390R 5'-ctgtctagatacaatggcaggatcaagtgt-3' (SEQ ID No: 19);
[0065] Caprin1 (361-385):
[0066] Caprin1-361F 5'-gaccttatggcacaaatgcagggtccctat-3' (SEQ ID No: 20);
[0067] Caprin1-Xba-385R 5'-tgatctagaaagtgtctgattttcaaaatc-3' (SEQ ID No: 21).
[0068] Referring to "Molecular Cloning Experimental Guide (Fourth Edition)" (Cold Spring Harbor Laboratory Press / Science Press), use the pCS2-Myc-hCaprin1 prepa...
Embodiment 3
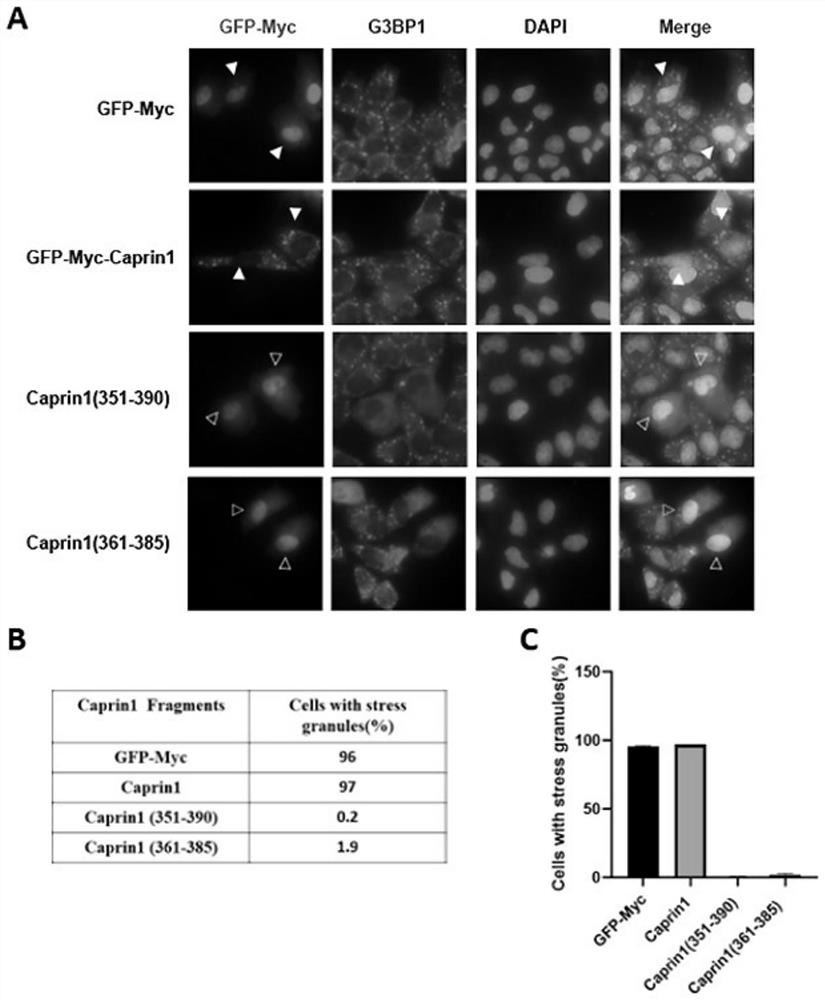
[0083] Example 3 To investigate the activity of Caprin1(361-385) and Caprin1(351-390) in inhibiting SGs HeLa cell line was seeded in 6-well plate, and when the cell confluency reached about 60%, the expression of Caprin1 constructed in Example 2 was carried out. (361-385) and Caprin1(351-390) plasmids were transfected into HeLa cells and overexpressed in the cells. When the cell confluency reaches about 80%, take 12 mL of cell culture base into a 15 mL centrifuge tube, add 12 μL of 0.5 mM AS, mix well, replace the medium in the 6-well plate, and stimulate HeLa cells to form SGs (use endogenous G3BP1 as the SGs Mark). Put into the carbon dioxide cell incubator and continue to cultivate for 45min. Remove the 6-well plate for immunofluorescence experiments.
[0084] The result is as image 3 As shown, the results indicated that overexpression of Caprin1(351-390), Caprin1(361-385) inhibited the SGs induced by AS compared with the control group ( image 3 A). In order to ensur...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap