Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle
A blood rheology and ephedrine technology, which is applied in the directions of medical preparations containing active ingredients, pharmaceutical formulas, organic active ingredients, etc., can solve the problems of undiscovered pharmaceutical compositions and the like
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
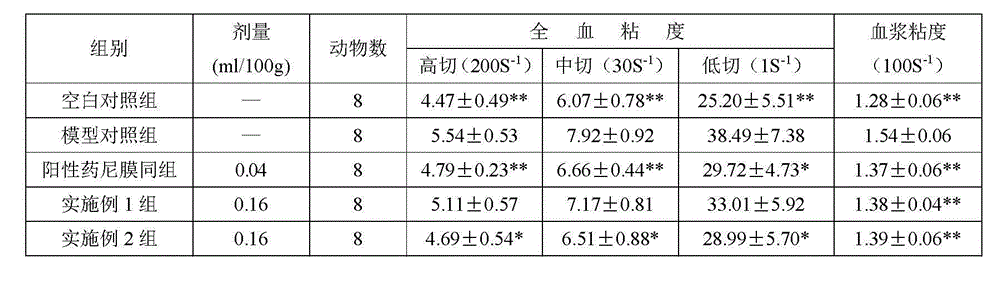
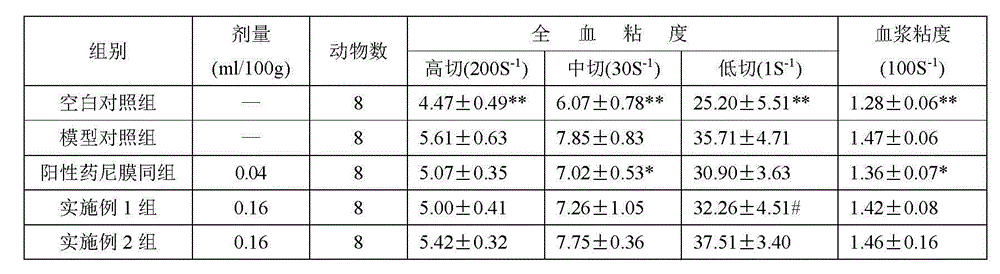
[0010] Embodiment 1: take by weighing 0.16g of seldanolactone of 98% purity, gastrodin 0.024g of 98% purity, 0.24g of ephedrine of 98% purity, add the polyoxyethylene sorbitan of 5% by volume percentage Monooleate 85ml, stir evenly, make the concentration of the composition of described cedanolactone, gastrodin and ephedrine in the polyoxyethylene sorbitan monooleate aqueous solution of 5% (volume ratio) 5mg / ml.
Embodiment 2
[0011] Embodiment 2: Take by weighing 0.16g of seldanolactone of 98% purity, 0.01g of gastrodin of 98% purity, 0.24g of ephedrine of 98% purity, add the polyoxyethylene sorbitan of 5% by volume percentage Monooleate 82ml, stir evenly, make the concentration of the composition of described sedanolactone, gastrodin and ephedrine in the polyoxyethylene sorbitan monooleate aqueous solution of 5% (volume ratio) 5mg / ml.
[0012] Said sedanolactone is prepared according to the method described in the invention patent application of application number 200880001478.9, named as a pharmaceutical composition for preventing and treating cerebral ischemic stroke and its preparation method, gastrodin and ephedrine are commercially available product.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap