Platinum (II) complex using chiral bicyclodiamine as carrier ligand and preparation method and application of platinum (II) complex
A complex and carrier technology, applied in the field of new platinum complexes and their preparation, can solve the problems of cisplatin drug treatment failure and other problems, and achieve the effect of overcoming cisplatin resistance
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0036] Example 1: Preparation of Ligands LR and LS.
[0037] (1) Synthesis of bicyclo[2,2,2]octane-2-alkene-7,8-trans diacid chloride
[0038] Add 4.1g (50mmol) of 1,3-cyclohexadiene into a 50mL single-necked round-bottomed flask, and slowly add 8.8g (55mmol) of fumaryl chloride dropwise at 0°C. After 12 hours of reaction, the target product bicyclo[2,2,2]octane-2-alkene-7,8-trans-diacid chloride was obtained.
[0039] (2) Synthesis of bicyclo[2,2,2]octane-2-alkene-7,8-trans diamine
[0040] 13.0g (200mmol) NaN 3and 50mL of water into a 250mL single-necked round bottom flask. Slowly add 100 mL of toluene solution of bicyclo[2,2,2]octane-2-alkene-7,8-trans-diacid chloride, the step product, into the reaction liquid at 0°C, and stir the reaction at this temperature for 3 h . After the reaction was completed, the reaction liquid was extracted with toluene (3×150 mL), and the organic phase was collected and dried over anhydrous sodium sulfate. Add the dried toluene solution ...
Embodiment 2
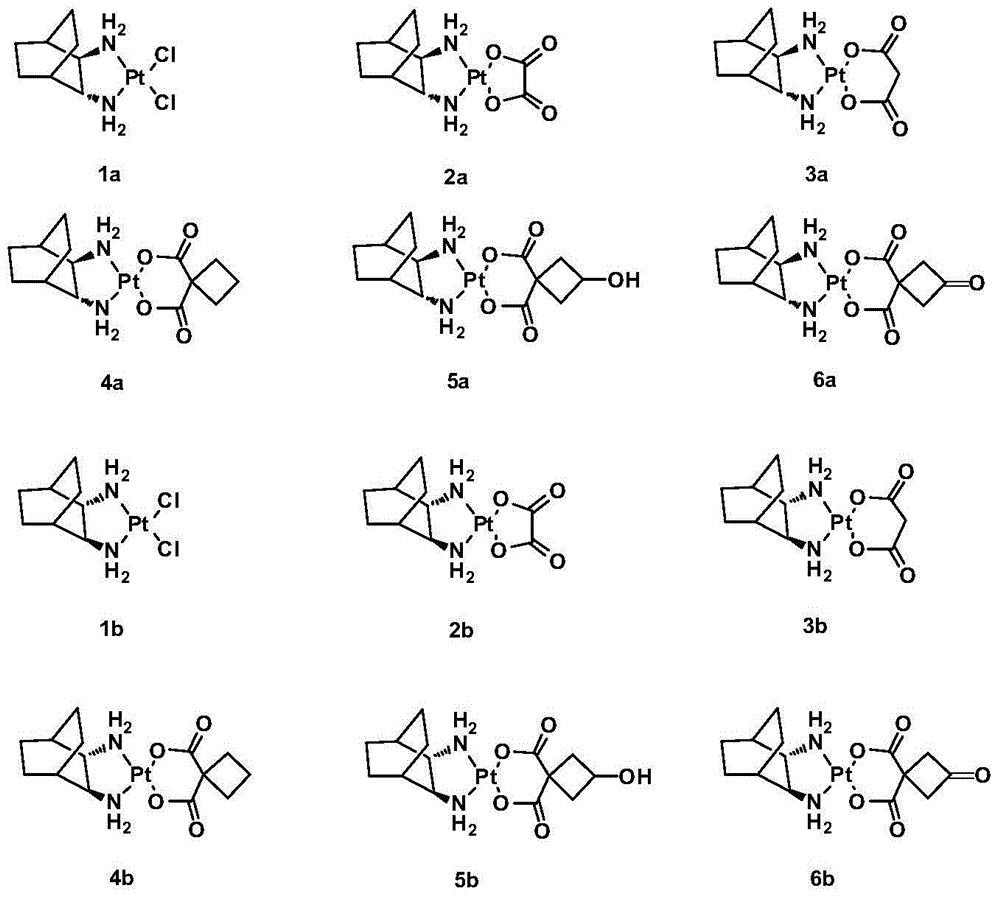
[0048] Example 2: Synthesis of complex 1a.
[0049] Dissolve 2.50g (6.00mmol) of potassium tetrachloroplatinate and 0.84g (6.00mmol) of LR in 50mL of deionized water, stir and react at 30°C for 12 hours in the dark, a bright yellow precipitate is formed, filter it, wash it repeatedly with water, and dry it brightly Yellow solid powder 2.43g, yield 89%. Elem anal. Calcd for C 8 h 16 Cl 2 N 2 Pt: C, 23.66; H, 3.98; N, 6.90. Found: C, 23.41; H, 4.25; N, 7.28. IR (KBr, cm -1 ): 3194, 2939, 2916, 2869, 477, 411; 1 H NMR (300MHz, d 6 -DMSO): δ1.19-1.77 (m, 8H, CH 2 of LR), 1.80-1.91 (m, 2H, CH of LR), 2.53(d, 2H, J=5.2Hz, 2×NH 2 CH ), 5.80-6.51 (m, 4H, 2×CH NH 2 ); 13 C NMR(d 6 -DMSO / TMS, ppm): δ19.15, 19.40, 25.99, 26.03, 31.24, 63.10, 64.07.
Embodiment 3
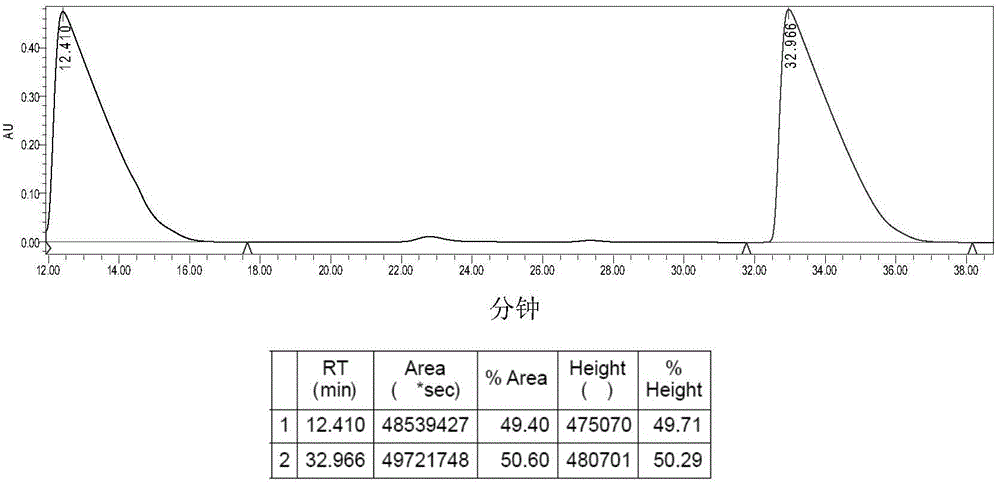
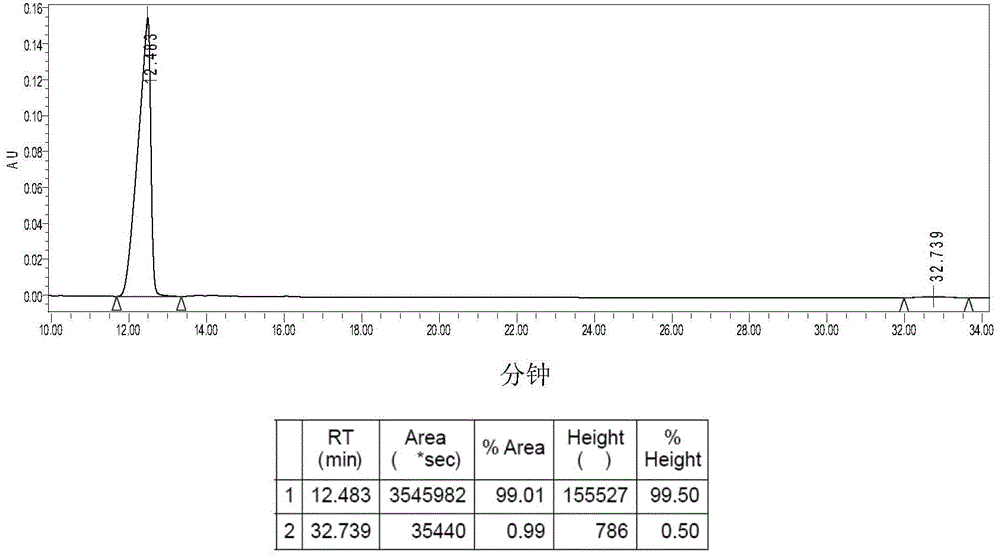
[0050] Example 3: Synthesis of Complex 2a.
[0051] Suspend 0.41g (1.00mmol) of 1a in 120mL of deionized water, stir, add 0.30g (1.00mmol) of silver oxalate, react at 38°C in the dark for 24h, stop the reaction, and diatomaceous earth-assisted filtration to obtain a clear solution. The filtrate was concentrated to 10 mL, a large amount of solids precipitated out, refrigerated at 4°C, filtered, and dried in vacuo to obtain 0.30 g of 2a white solid with a yield of 71%. [α] D 30 =140.0° (c=0.20, DMF:H 2 O=1:1).Elem anal.Calcd for C 10 h 16 N 2 o 4 Pt: C, 28.37; H, 3.81; N, 6.62. Found: C, 28.63; H, 3.57; N, 6.83. IR (KBr, cm -1 ): 3274 (NH), 3131, 2943, 1704, 1660, 1589, 1372, 1153, 807; 1 H NMR (300MHz, d 6 -DMSO): δ1.17-1.65 (m, 8H, CH 2 of LR), 1.69-1.72 (m, 2H, CH of LR), 2.26(d, 2H, J=9.0Hz, 2×NH 2 CH ), 5.31-5.69 (m, 4H, 2×CH NH 2 ); 13 C NMR(d 6 -DMSO / TMS, ppm): δ19.22, 25.99, 31.17, 64.46, 165.87; ESI-MS: m / z [M+Na] + =446(100%).
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap