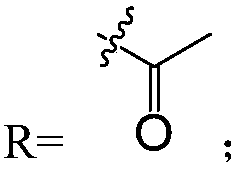
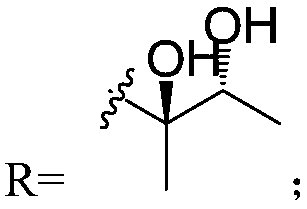
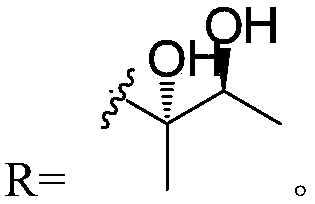
Three piericidin natural products and application thereof to preparation of anti-kidney cancer medicines
A pteromycin and kidney cancer technology, applied in the field of microbial natural products, can solve the problems of weak research on the mechanism of action, anti-tumor potential to be further explored, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0023] Embodiment 1: the preparation of piericidins G, I, and J of piericidins compound
[0024] 1. Solid culture of Streptomyces sp.HBERC-58855
[0025] Streptomyces sp.HBERC-58855 (preservation number is CCTCC NO:M 2017186) is isolated from the mangrove bottom mud, and the strain is preserved on the slant of ISP-2 medium. The composition of ISP-2 medium is: yeast extract powder 4g, glucose 4g, malt extract powder 10g, coarse sea salt 30g, agar powder 20g, water 1000mL, pH 7.2-7.4, sterilized for later use.
[0026] 2. Amplified fermentation of Streptomyces sp.HBERC-58855
[0027] Take a small amount of Streptomyces sp.HBERC-58855 slant strains for seed fermentation culture. The medium is 20 grams of mannitol, 10 grams of soybean peptone, 2.5 grams of soybean oil, 0.35 grams of dipotassium hydrogen phosphate, 950 ml of deionized water, and adjust the pH to 7.0 , dilute to 1000 ml, fill 100 ml in a 500 ml Erlenmeyer bottle, and sterilize for later use. The culture condition...
Embodiment 2
[0030] Example 2: Structural identification of Piericidins G, I, and J
[0031] 1.Piericidins G, I, and J physicochemical property data
[0032] Compounds Piericidins G, I, and J were subjected to structural analysis and testing, and the following physical and chemical property data were obtained:
[0033] Compound piericidin G: colorless oil, -5.5(c 0.218, MeOH); IR(ATR)ν max 3312,2945,2835,1635,1472,1417,1126,1018,667,600,562cm -1 ; CD (0.2mg / ml, MeOH), λmax (Δε) 240 (5.8), 223 (-7.8); 1 H and 13 C NMR data in Table 1 and Table 2; (+)-HR-ESIMS m / z 414.2657[M+H] + (calcd for C 25 h 36 NO 4 414.2600).
[0034] Compound piericidin I: colorless oil, 0.08(c 0.12, MeOH); IR(ATR)ν max 3335,2945,2833,1448,1416,1020,667,600,552cm -1 ; CD (0.342mg / ml, MeOH), λmax (Δε) 237 (-1.5), 205 (3.5); 1 H and 13 C NMR data in Table 1 and Table 2; (+)-HR-ESIMS m / z 504.3320[M+H] + (calcdforC 29 h 45 NO 6 504.3280).
[0035] Compound piericidin J: colorless oil, 2.22 (c 0...
Embodiment 3
[0052] Example 3: Inhibitory Activity of Piericidins G, I, and J on Renal Cancer Cells
[0053] Two human kidney cancer cell lines were ordered from the Shanghai Cell Resource Center of the Chinese Academy of Sciences: ACHN human kidney cancer cell line (Cat#TCHu199); OS-RC-2 human kidney cancer cell line (Cat#TCHu40); human renal tubular epithelial cell HK- 2Provided by School of Pharmacy, Southern Medical University.
[0054] The same type of natural product piericidin A (PiericidinA) and the marketed drug Sorafenib for the treatment of renal cancer were used as positive controls.
[0055] The cell inhibitory activity experiment adopts CCK-8 detection method. Collect the cells in the logarithmic growth phase, count them, resuspend the cells with complete medium, adjust the cell concentration to an appropriate concentration (determined according to the results of the cell density optimization test), inoculate a 96-well plate, and add 100 μl of cell suspension to each well. ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap