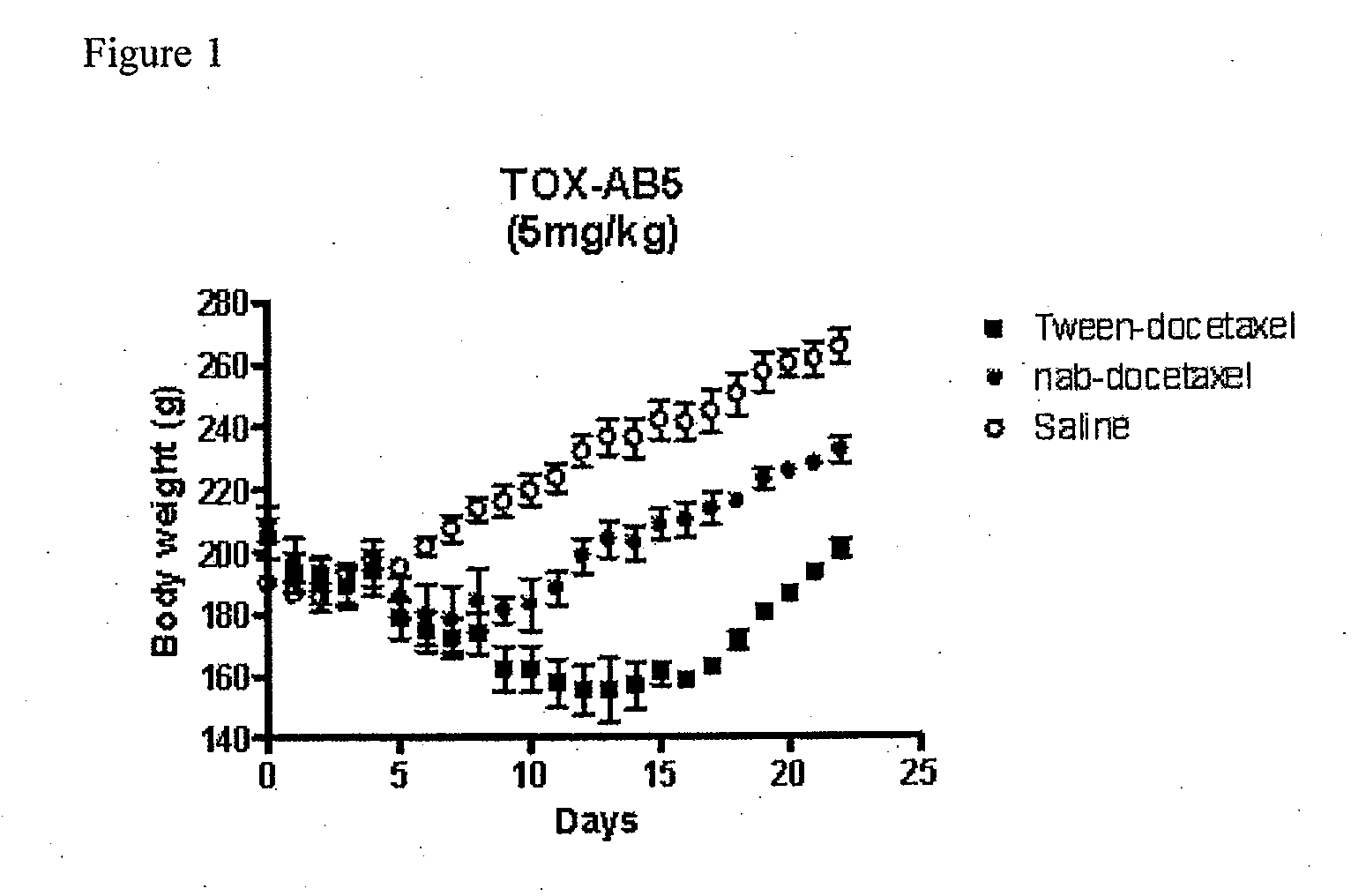
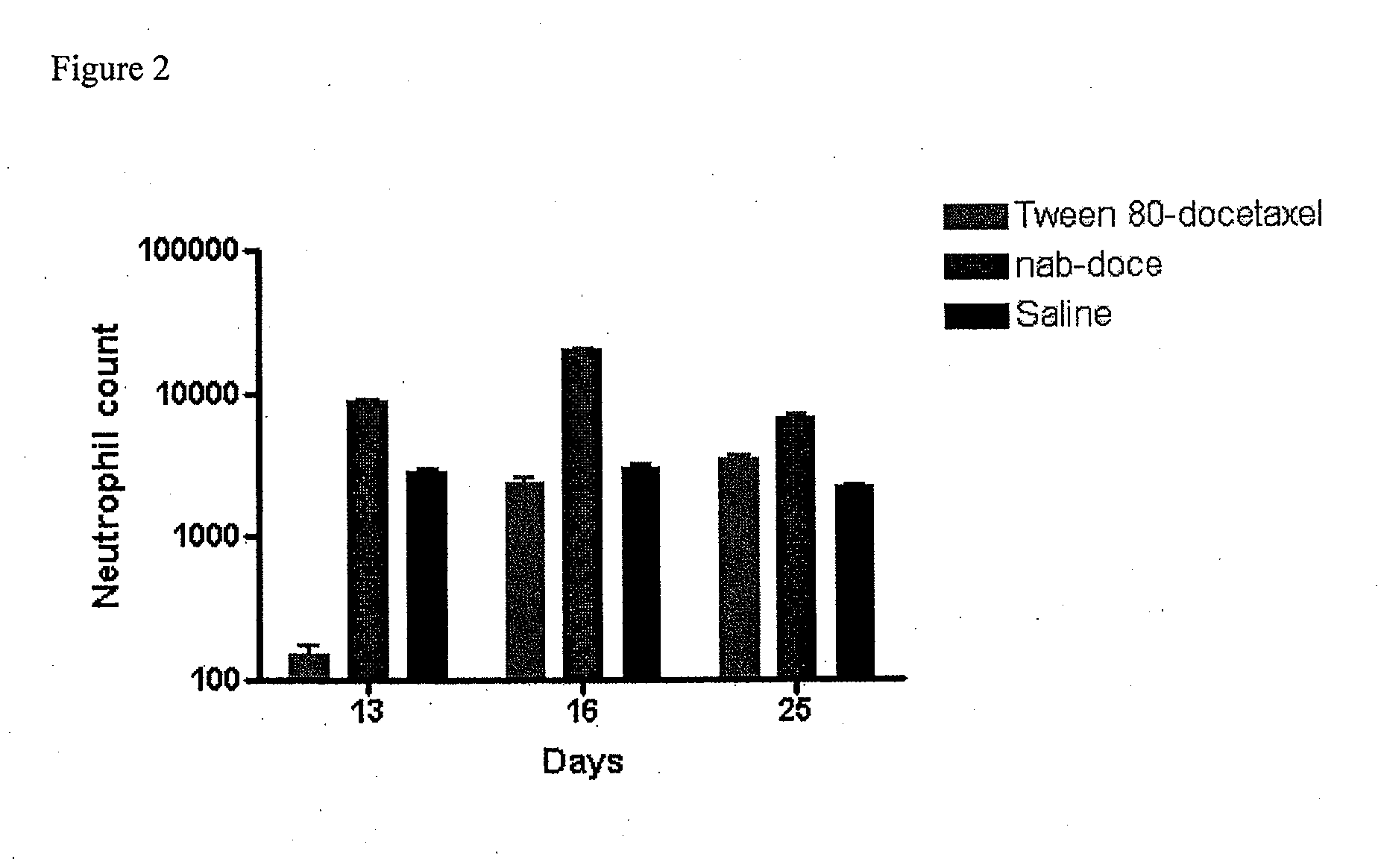
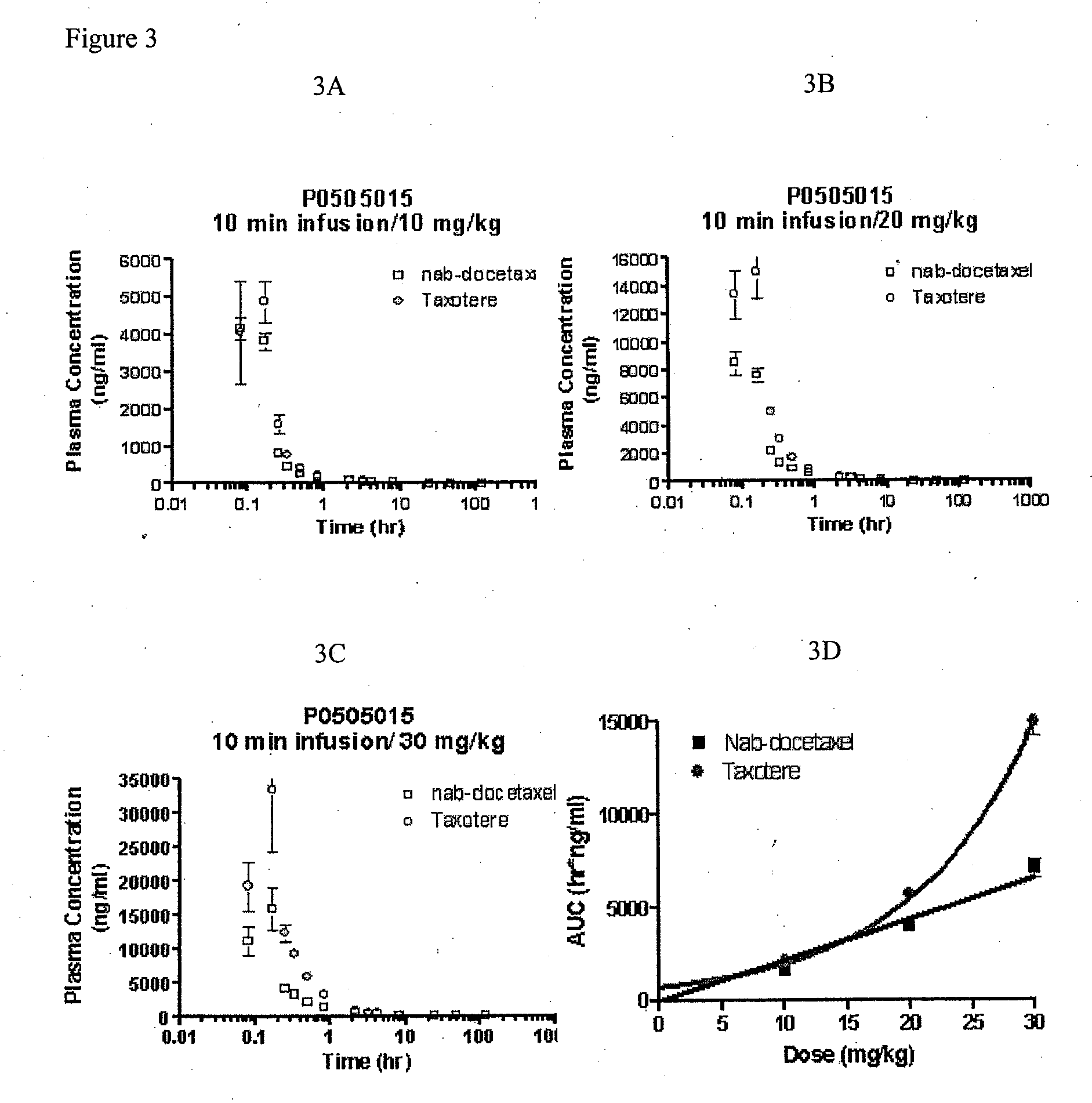
Compositions and methods for preparation of poorly water soluble drugs with increased stability
a technology compositions, applied in the field of compositions and methods for the preparation of poorly water soluble drugs with increased stability, can solve the problems of increasing the number of pharmaceutical drugs being formulated that are poorly soluble or insoluble in aqueous solutions, compositions tend to be unstable, and paclitaxel is very poorly water soluble, so as to achieve enhanced stability of compositions.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
[0152]This example demonstrates the instability of a preparation of pharmaceutical compositions comprising docetaxel and albumin prepared as described in U.S. Patent Publication 2005 / 0004002 A1.
[0153]30 mg of docetaxel was dissolved in 2 mL chloroform / ethanol. The solution was then added into 27.0 mL of HSA solution (3% w / v). The mixture was homogenized for 5 minutes at low RPM (Vitris homogenizer model Tempest I.Q.) in order to form a crude emulsion, and then transferred into a high pressure homogenizer (Avestin). The emulsification was performed at 9000-40,000 psi. The resulting system was transferred into a Rotavap and solvent was rapidly removed at reduced pressure. The resulting dispersion was translucent and the typical average diameter of the resulting particles was in the range 50-220 nm (Z-average, Malvern Zetasizer). The dispersion was further lyophilized for 48 hours. The resulting cake was easily reconstituted to the original dispersion by addition of sterile water or sa...
example 2
[0154]This example demonstrates the instability of docetaxel nanoparticles prepared by sonication.
[0155]25.9 mg of docetaxel was added to a 20-mL scintillation vial and dissolved in 0.3 mL of chloroform. 4.7 mL of HSA (3.0%, w / v) was added to the docetaxel dissolved mixture. The composition was sonicated (Sonic Dismembrator, model 550, Fisher Scientific Company, Pittsburgh, Pa. 155275) is at 50% power for 1 min. The mixture was transferred into a rotary evaporator, and chloroform-ethanol was rapidly removed at 45° C., at reduced pressure. The diameter of the resulting docetaxel particles was 250-300 nm (Z-average, Malvern Zetasizer). The suspension precipitated in less than 1 day.
example 3
[0156]This example demonstrates the instability of docetaxel nanoparticles prepared by sonication testing soybean oil as a stabilizer.
[0157]18.0 mg of docetaxel was added to a 20-mL scintillation vial and dissolved in 0.1 mL of chloroform-ethanol mixture. 0.05 mL of soybean oil and 2.35 mL of HSA (5.0%, w / v) was added to the above organic solvent. The sample was sonicated (Sonic Dismembrator, model 550, Fisher Scientific Company, Pittsburgh, Pa. 155275) for 2 min. The mixture is transferred into a rotary evaporator, and chloroform-ethanol is rapidly removed at 45° C., at reduced pressure. The diameter of the resulting docetaxel particles was ˜270 nm (Z-average, Malvern Zetasizer). The suspension precipitated in less than 1 day.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More