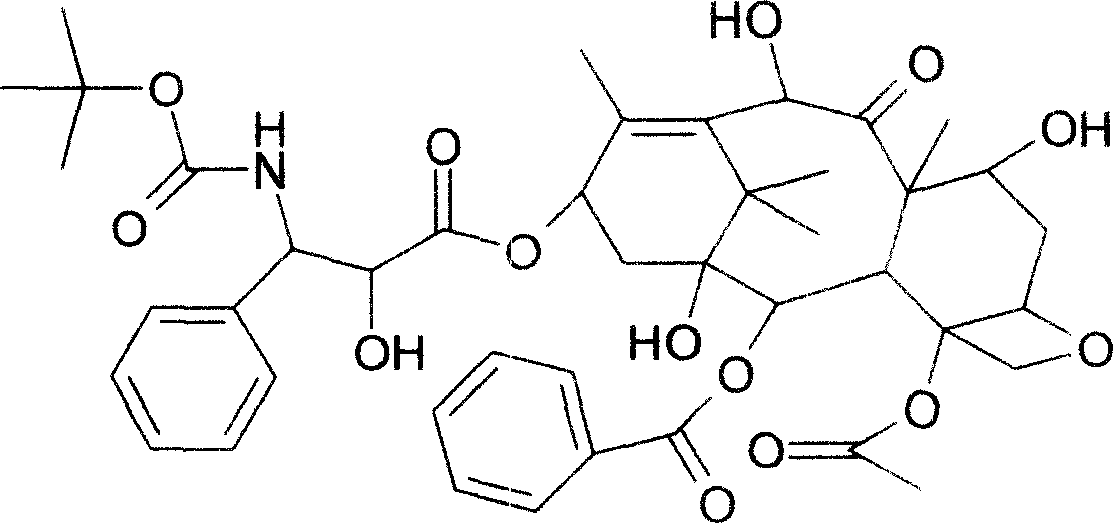
Patents
Literature
735 results about "Docetaxel" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
This medication is used to treat cancer (such as breast, lung, prostate, stomach, and head/neck cancer).
Treatment of metastatic breast cancer
InactiveUS20090317387A1Improve survivalOrganic active ingredientsAntibody ingredientsBiologic therapiesDocetaxel-PNP
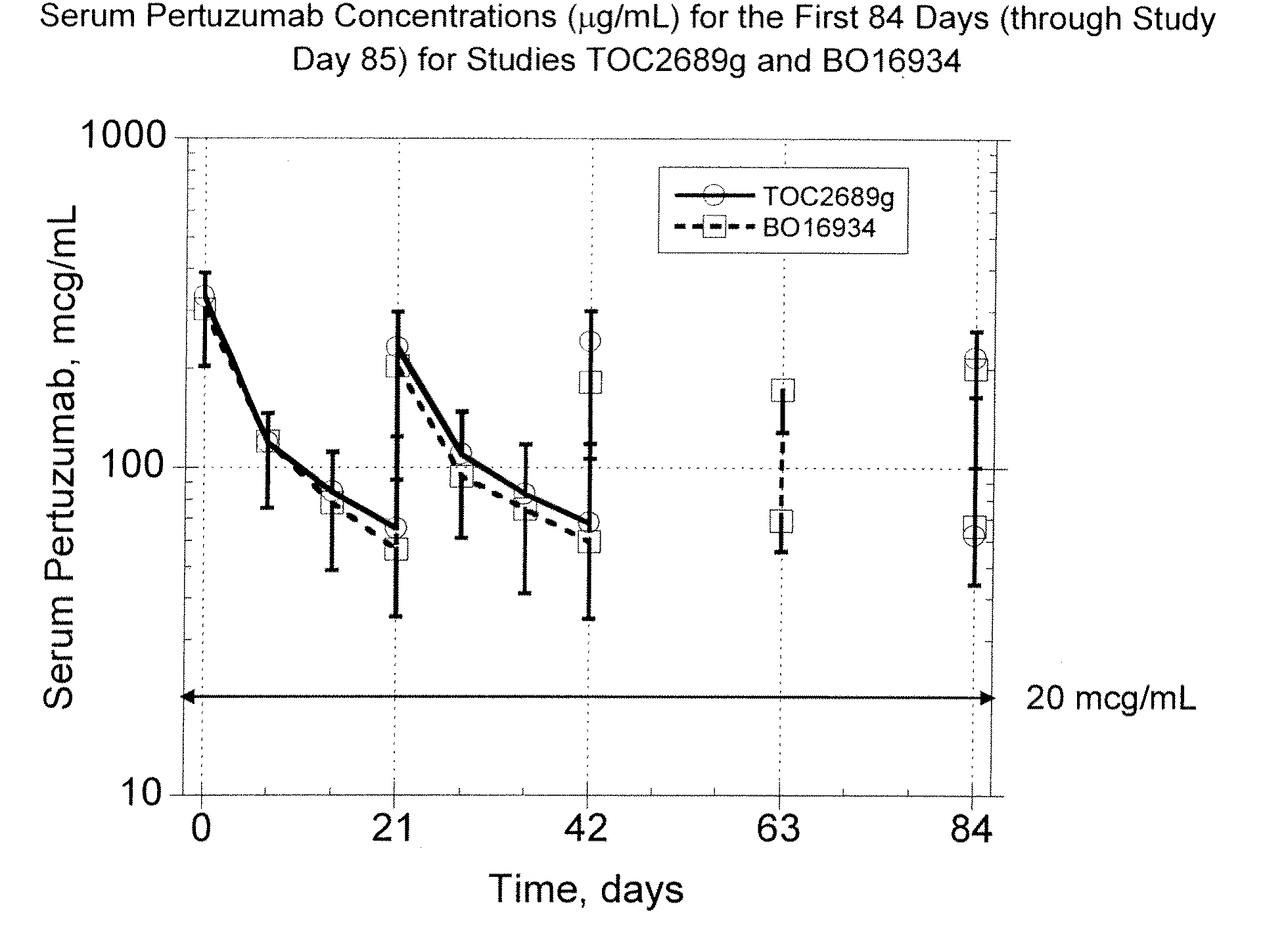
The present invention concerns treatment of previously untreated HER2-positive metastatic breast cancer with a combination of a growth inhibitory HER2 antibody, a HER2 dimerization inhibitor antibody and a taxane. In particular, the invention concerns the treatment of HER2-positive metastatic breast cancer in patients who did not receive prior chemotherapy or biologic therapy with a HER2 antibody binding essentially to epitope 2C4, a HER2 antibody binding essentially to epitope 4D5, and a taxane. The invention further comprises extending survival of such patients by the combination therapy of the present invention. In a preferred embodiment, the treatment involves administration of trastuzumab, pertuzumab and docetaxel.
Owner:GENENTECH INC
Compositions and methods for preparation of poorly water soluble drugs with increased stability
The present invention provides stable pharmaceutical compositions of poorly water soluble pharmaceutical agents and stabilizing agents which function to increase stability of the compositions. The use of stabilizing agents provide extended stability of nanoparticle suspensions and other formulations of poorly water soluble pharmaceutical agents such as docetaxel under certain conditions, for example upon dilution for administration.
Owner:ABRAXIS BIOSCI LLC
Lyophilized solid taxane composition, a process for preparing said solid composition, a pharmaceutical formulation and a kit for said formulation
InactiveUS20090215882A1High level of chemical degradationImprove solid solubilityOrganic active ingredientsBiocideDocetaxel-PNPDocetaxel
A lyophilized solid composition of taxane (preferably docetaxel and paclitaxel), is suitable to prepare a pharmaceutical formulation to be administered to mammals, particularly humans, comprising a taxane, a tensoactive, a lyophilizing excipient, and acid; also essentially free from organic solvents. The solid composition is free from polysorbate 80 and polyoxyethylated castor oil; it is sterile; it is soluble in aqueous solutions in the absence of organic solvent and it has an apparent density from 0.05 g / ml to 0.45 g / ml. A procedure of double lyophilization obtains a solid composition of taxane. A pharmaceutical formulation of a taxane comprises a solid composition of lyophilized taxane and a solubilizing composition. A kit comprises the compositions and a syringe.
Owner:ERIOCHEM SA
Water soluble paclitaxel derivatives
InactiveUS6884817B2Surprising antitumor activityImprove efficacyHeavy metal active ingredientsBiocideDocetaxel-PNPDocetaxel
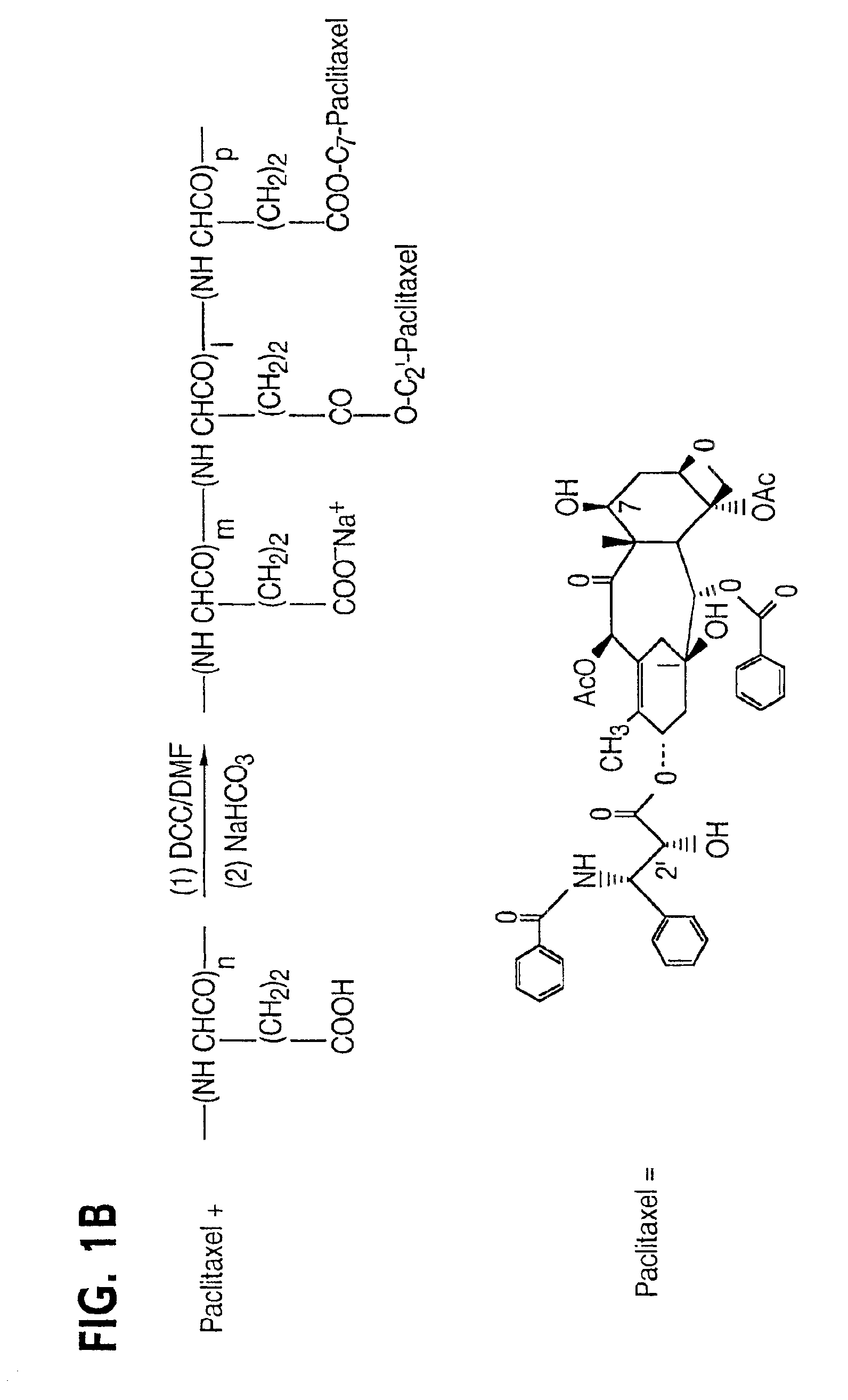
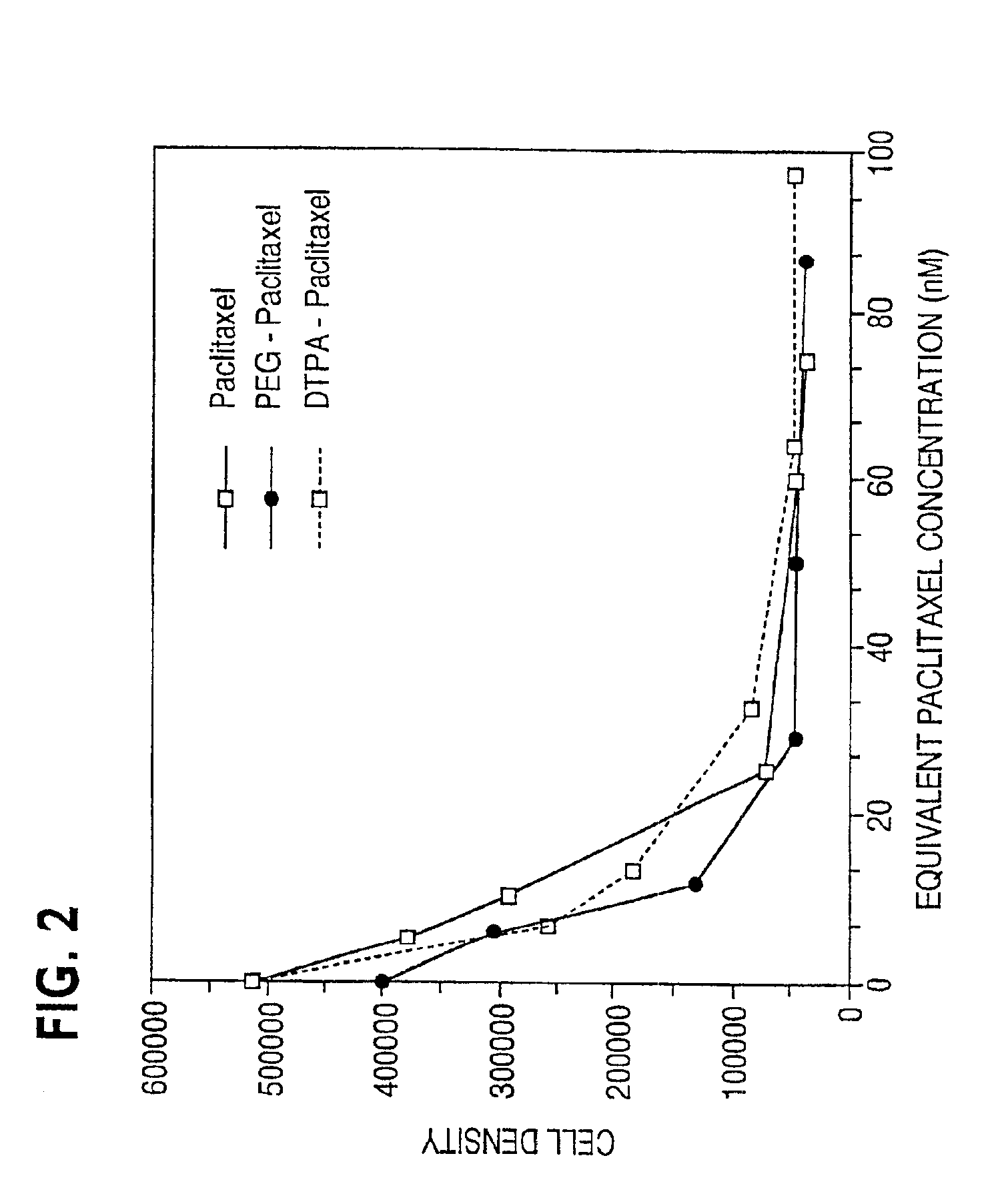
Disclosed are water soluble compositions of paclitaxel and docetaxel formed by conjugating the paclitaxel or docetaxel to a water soluble polymer such as poly-glutamic acid, poly-aspartic acid or poly-lysine. Also disclosed are methods of using the compositions for treatment of tumors, auto-immune disorders such as rheumatoid arthritis. Other embodiments include the coating of implantable stents for prevention of restenosis.
Owner:PG TXL COMPANY
Antineoplastic conjugates of transferrin, albumin and polyethylene glycol
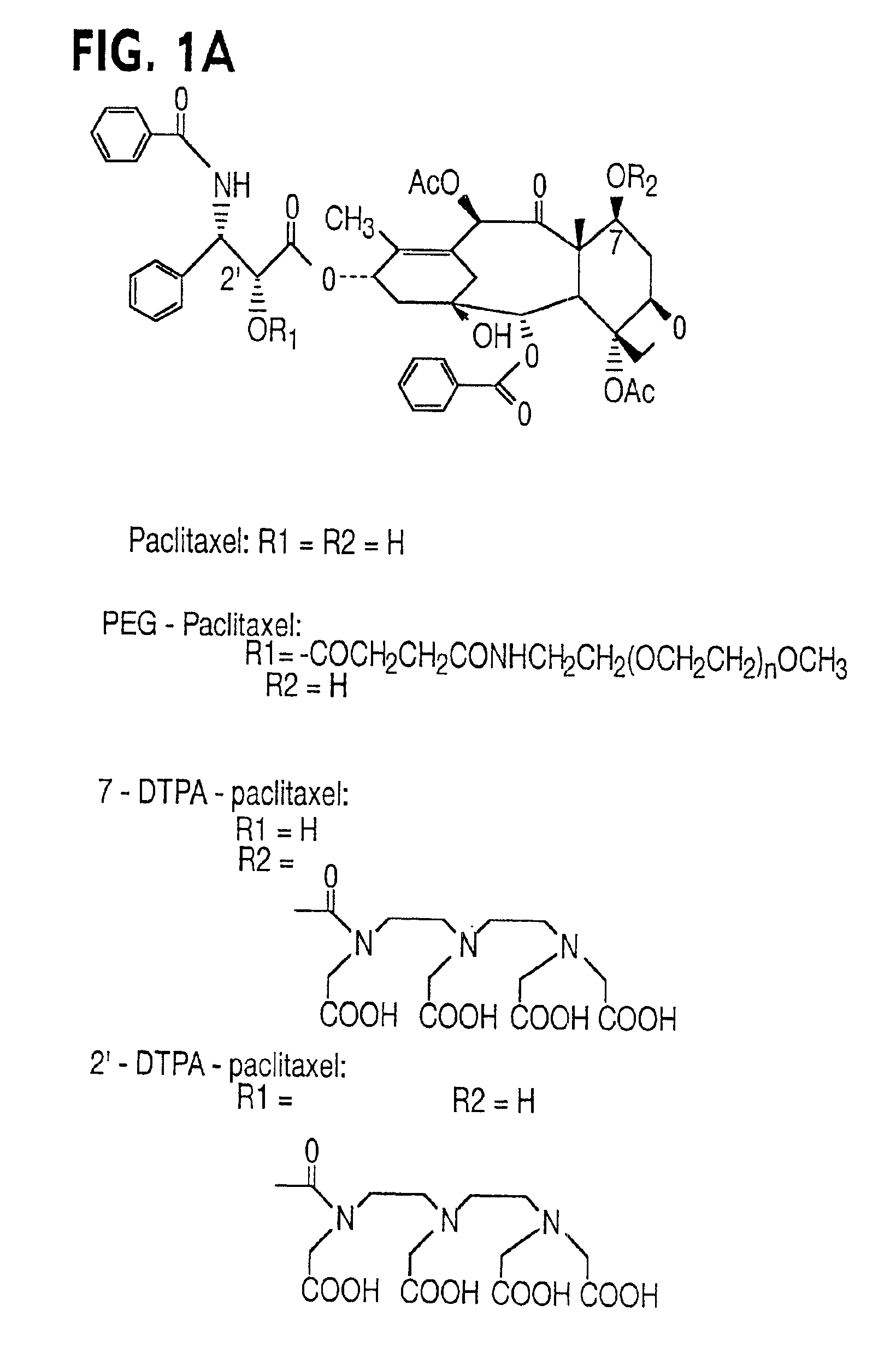
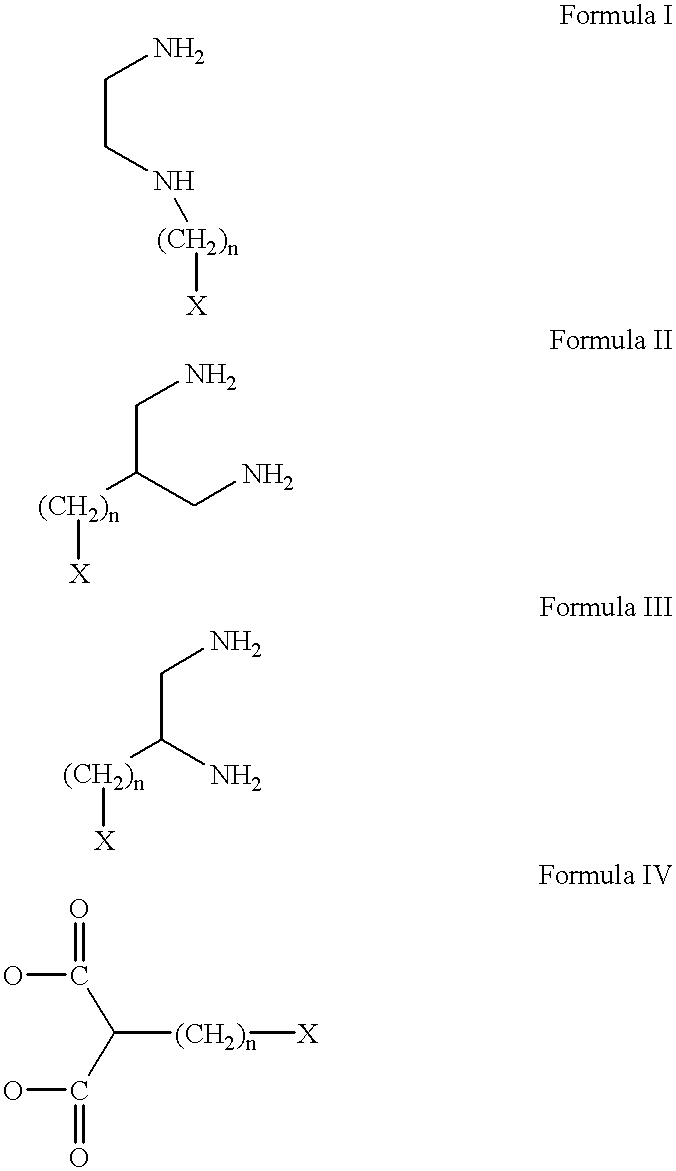
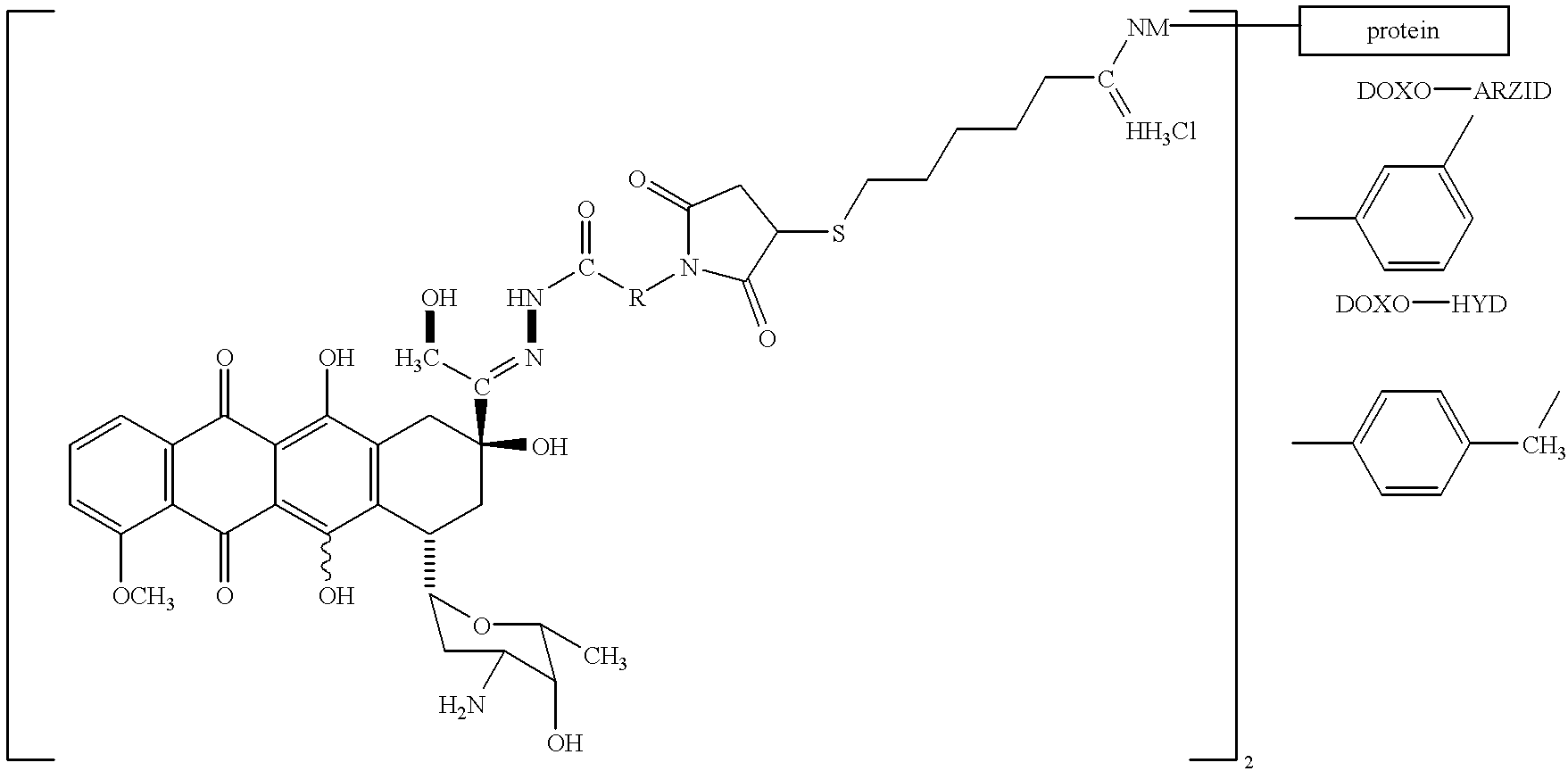
Conjugates of transferrin, albumin and polyethylence glycol consisting of native or thiolated transferrin or albumin or of polyethylene glycol (MW between approximately 5,000 and 20,0000) with at least one HS-, HO- or H2N group and cytostatic compounds derived through maleinimide or N-hydroxysuccinimide ester compounds, such as doxorubicin, daunorubicin, epirubicin, idarubicin, mitoxandrone, chloroambucil, melphalan, 5-fluorouracyl, 5'-desoxy-5-fluorouridine, thioguanine, methotrexate, paclitaxel, docetaxel, topotecan, 9-aminocamptothecin, etoposide, teniposide, mitopodoside, vinblastine, vincristine, vindesine, vinorelbine or a compound of general formula A, B, C or D, where n=0-6, X=-NH2, -OH, -COOH, -O-CO-R-COR*, -NH-CO-R-COR*, where R is an aliphatic carbon chain with 1-6 carbon atoms or a substituted or unsubstituted phenylene group and R* H, phenyl, alkyl with 1-6 carbon atoms.
Owner:KRATZ FELIX
Solid nanoparticle formulation of water insoluble pharmaceutical substances with reduced ostwald ripening
ActiveUS8728527B2Good dispersionReduced and substantially no particle growthBiocideOrganic active ingredientsDocetaxel-PNPDocetaxel
The present invention belongs to the fields of pharmacology, medicine and medicinal chemistry. The present invention provides novel pharmaceutical compositions composed of solid nanoparticles dispersed in aqueous medium of substantially water insoluble pharmaceutical substances such as docetaxel with reduced Ostwald ripening.
Owner:AUSTIN BIOSCIENCES CORP
Method of treating cancer, especially soft tissue sarcoma utilizing gemcitabine in combination with docetaxel and anti-VEGF therapy (bevacizumab)
InactiveUS20070065449A1Great likelihoodLong median survivalBiocideGenetic material ingredientsAbnormal tissue growthLymphatic Spread
The present invention relates to a pharmaceutical cocktail, in particular, effective amounts of gemcitabine, in combination with effective amounts of docetaxel and angiogenesis inhibitor, especially a vascular endothelial growth factor (VEGF) inhibitor, such as bevacizumab for the treatment of cancer, in particular sarcoma, especially soft tissue sarcoma. Pharmaceutical compositions and methods of treating cancer, including sarcoma, especially soft tissue sarcoma (prolonging the patient's life, eliminating the tumor, improving the patient's quality of life, shrinking the tumor, prolonging survival and / or preventing the tumor's metastases) are additional aspects of the present invention.
Owner:STC UNM
Compositions and methods for preparation of poorly water soluble drugs with increased stability
Owner:ABRAXIS BIOSCI LLC
Therapeutic compounds
InactiveUS20040048923A1Improve pharmacological activityGood curative effectBiocideOrganic compound preparationDocetaxel-PNPTreatment effect
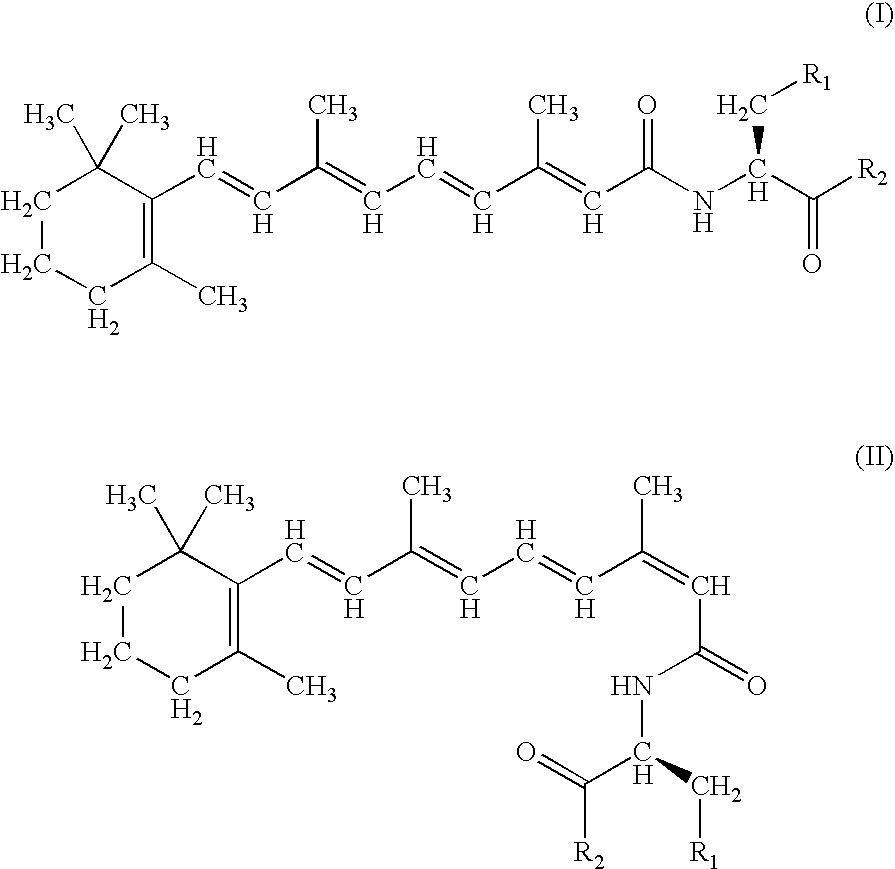
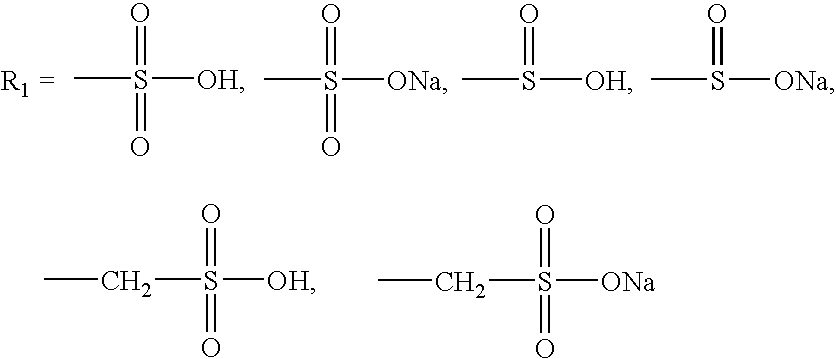
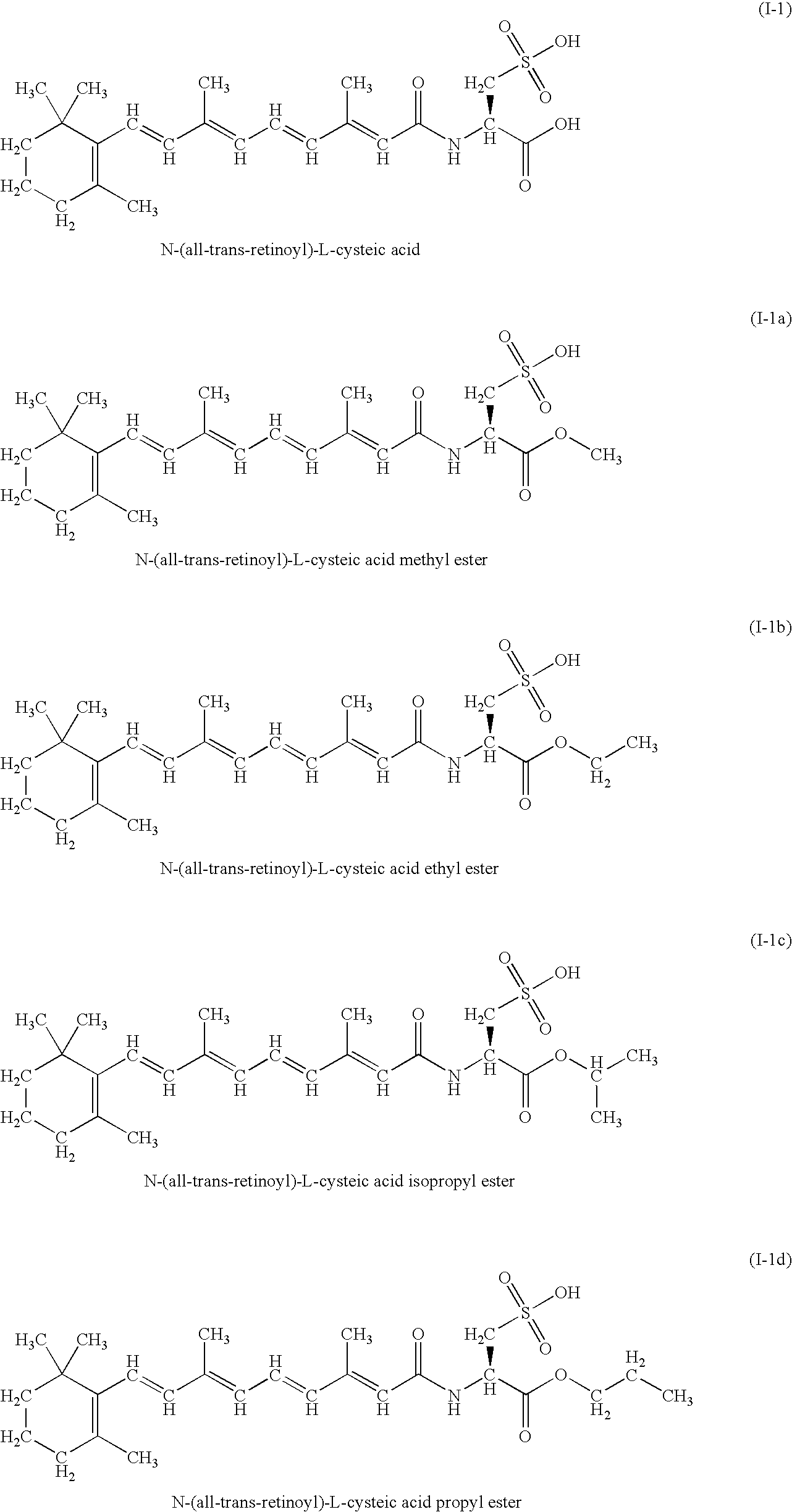
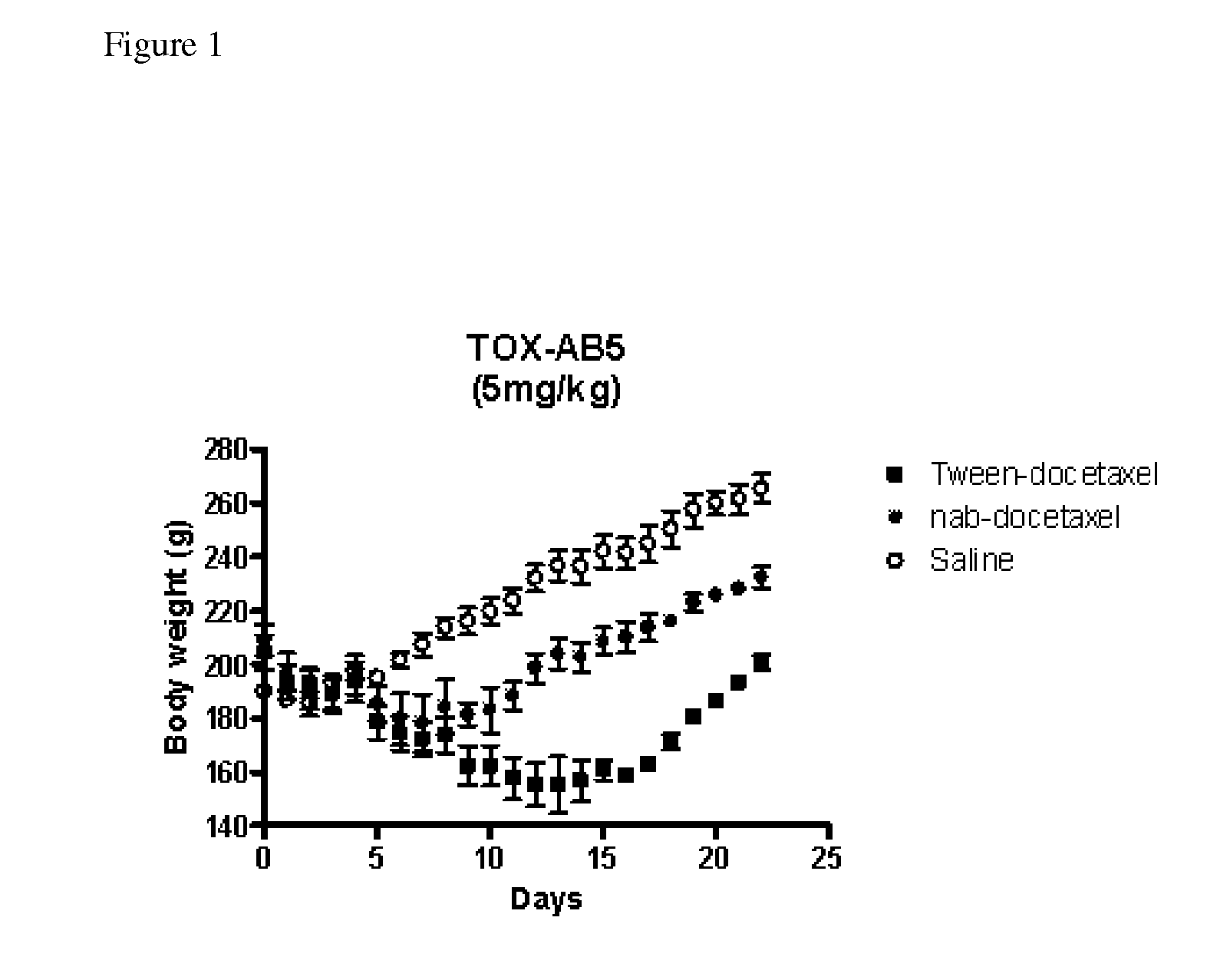
A group of new compounds, N-(all-trans-Retinoyl)-L-cysteic acid, N-(13-cis-Retinoyl)-L-cysteic acid, N-(all-trans-Retinoyl)-L-cysteinesulfinic acid, N-(13-cis-Retinoyl)-L-cysteinesulfinic acid, N-(all-trans-Retinoyl)-L-homocysteic acid, N-(13-cis-Retinoyl)-L-homocysteic acid, and sodium salts of these compounds, including sodium salts of their esters and amides, is shown to exhibit therapeutic effects per se, and which compounds in combination with cytotoxic compounds, such as docetaxel, paclitaxel, doxorubicin and mitoxantrone, exhibit a synergistic effect. These compounds make it possible to manufacture new formulations of poorly soluble pharmaceutical compounds, and the present invention discloses a process of manufacturing water-soluble formulations of such compounds, exemplified by docetaxel, and paclitaxel, exhibiting enhanced pharmacological activity, and formulations of water-soluble pharmaceuticals exemplified by doxorubicin and mitoxantrone, exhibiting improved therapeutic efficacy.
Owner:VIVESTO AB
Methods of treating cancer
ActiveUS20120070502A1High elongationLow toxicityPowder deliveryOrganic active ingredientsCarboplatinPlatinum
The present invention provides methods and compositions for treating non-small-cell lung cancer (NSCLC) by administering a) a composition comprising nanoparticles that comprise paclitaxel and an albumin and b) a platinum-based agent (e.g., carboplatin). The present application also provides methods of treating prostate cancer by administering to the individual a) an effective amount of a composition comprising nanoparticles comprising docetaxel and an albumin; and b) an effective amount of a steroid.
Owner:ABRAXIS BIOSCI LLC
Methods of treating cancer
ActiveUS20140072643A1Many symptomShorten the progressOrganic active ingredientsHeavy metal active ingredientsCarboplatinDocetaxel-PNP
The present invention provides methods and compositions for treating non-small-cell lung cancer (NSCLC) by administering a) a composition comprising nanoparticles that comprise paclitaxel and an albumin and b) a platinum-based agent (e.g., carboplatin). The present application also provides methods of treating prostate cancer by administering to the individual a) an effective amount of a composition comprising nanoparticles comprising docetaxel and an albumin; and b) an effective amount of a steroid.
Owner:ABRAXIS BIOSCI LLC
Antibodies against csf-1r
ActiveUS20110243947A1Prevent dimerizationInhibit tumor growthImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsDocetaxel-PNPDocetaxel
The invention provides a human antibody that binds human CSF-1R with high affinity. Antibodies of the present invention have significant advantages over the antibodies known in the art by being multifunctional: inhibiting signaling of CSF-1R, internalizing and inducing CSF-1R degradation and stimulating ADCC in cell including tumors, macrophages and monocytes. They are also shown to be effective in treating leukemia, breast, endometrial and prostate cancer alone or in combination with docetaxel, paclitaxel, Herceptin® or doxorubicin.
Owner:IMCLONE SYSTEMS
Compositions and methods for preparation of poorly water soluble drugs with increased stability
The present invention provides stable pharmaceutical compositions of poorly water soluble pharmaceutical agents and stabilizing agents which function to increase stability of the compositions. The use of stabilizing agents provide extended stability of nanoparticle suspensions and other formulations of poorly water soluble pharmaceutical agents such as docetaxel under certain conditions, for example upon dilution for administration.
Owner:ABRAXIS BIOSCI LLC
Oral pharmaceutical compositions containing taxanes and methods of treatment employing the same
Pharmaceutical compositions for oral administration to mammalian subjects comprise a taxane or taxane derivative (e.g., paclitaxel or docetaxel) as active ingredient and a vehicle comprising at least 30% by weight of a carrier for the taxane, said carrier having an HLB value of at least about 10. The compositions may also comprise 0–70% of a viscosity-reducing co-solubilizer. The compositions may be incorporated into conventional oral pharmaceutical dosage forms, or can be in the form of a two-part medicament wherein the first part includes the taxane in a solubilizing vehicle and the second part comprises a carrier for the taxane to promote oral absorption. Methods of treatment of taxane-responsive disease conditions employing the novel compositions are also disclosed, whereby the compositions can be administered alone or in association with an oral bioavailability enhancing agent.
Owner:BAKER NORTON PHARMA INC
Novel improved compositions for cancer therapy
InactiveUS20100166872A1Reduced chemotherapy-induced side-effectsOrganic active ingredientsBiocideDocetaxel-PNPSide effect
The present invention relates to novel and improved compositions of anticancer drugs, preferably taxanes, such as paclitaxel and docetaxel, their derivatives or their analogues, methods of manufacturing these compositions and methods of fractionating the particles in particular size range and methods of treating cancer patients with these compositions, which provide reduced chemotherapy-induced side-effects especially reduced chemotherapy-induced-alopecia. The composition is such that there is substantially no free drug in the said composition.
Owner:PANACEA BIOTEC
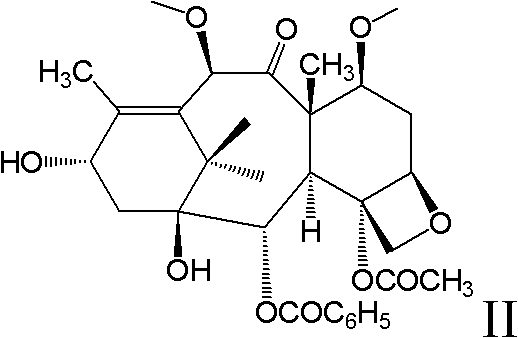
Docetaxel polymeric micelle medicine composition taking amino acid as stabilizing agent
ActiveCN101972480AObserve stabilityOrganic active ingredientsPharmaceutical delivery mechanismDocetaxelAmino acid composition
The invention relates to polymeric micelle medicine carrying system taking amino acid as stabilizing agent and a preparation method thereof. The medicine carrying system can be acted as a medicine carrying composition which is composed of amphipathy block copolymer and amino acid; wherein the amphipathy block copolymer comprises diblock copolymer and triblock copolymer, amino acid is selected from twenty common amino acids, the ratio (w / w) of amphipathy block copolymer and amino acid is 100-1.00: 0.01-99.9.
Owner:FANTAI INST OF CHEM MEDICINES NANJING +2
Vitamin E succinate stabilized pharmaceutical compositions, methods for the preparation and the use thereof
The present invention provides vitamin E succinate (VES)-stabilized compositions, methods for the preparation thereof and methods useful for the in vivo delivery of substantially water insoluble and optionally chemically unstable pharmacologically active agents (such as docetaxel).
Owner:CHEN ANDREW XIAN
Vitamin E succinate stabilized pharmaceutical compositions, methods for the preparation and the use thereof
The present invention provides vitamin E succinate (VES)-stabilized compositions, methods for the preparation thereof and methods useful for the in vivo delivery of substantially water insoluble and optionally chemically unstable pharmacologically active agents (such as docetaxel).
Owner:CHEN ANDREW XIAN
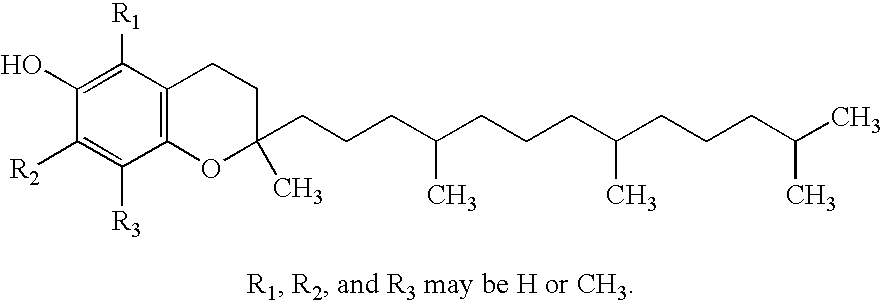
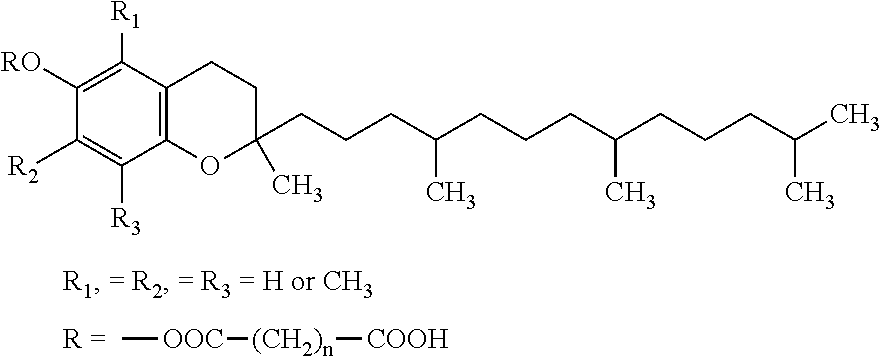
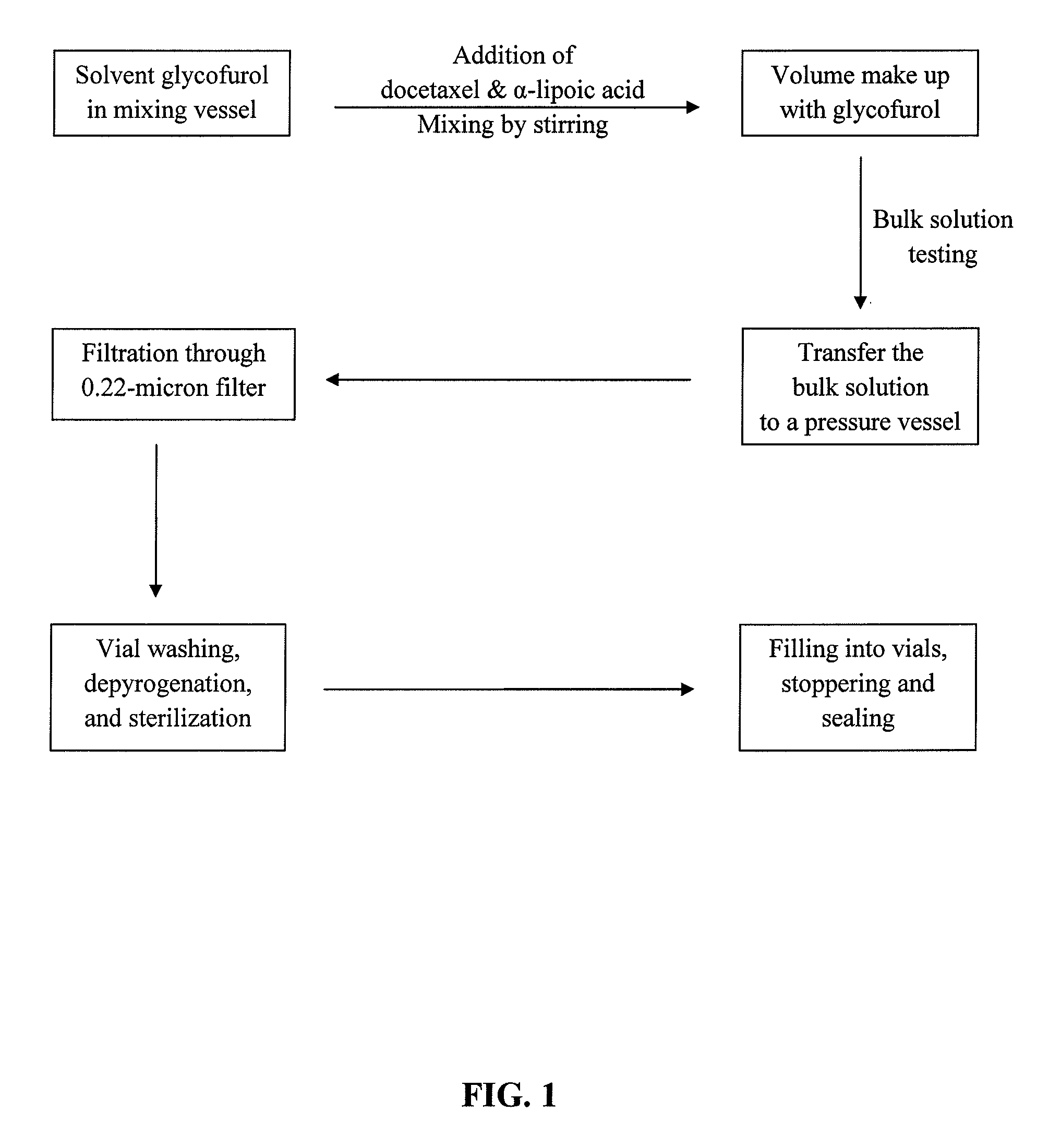
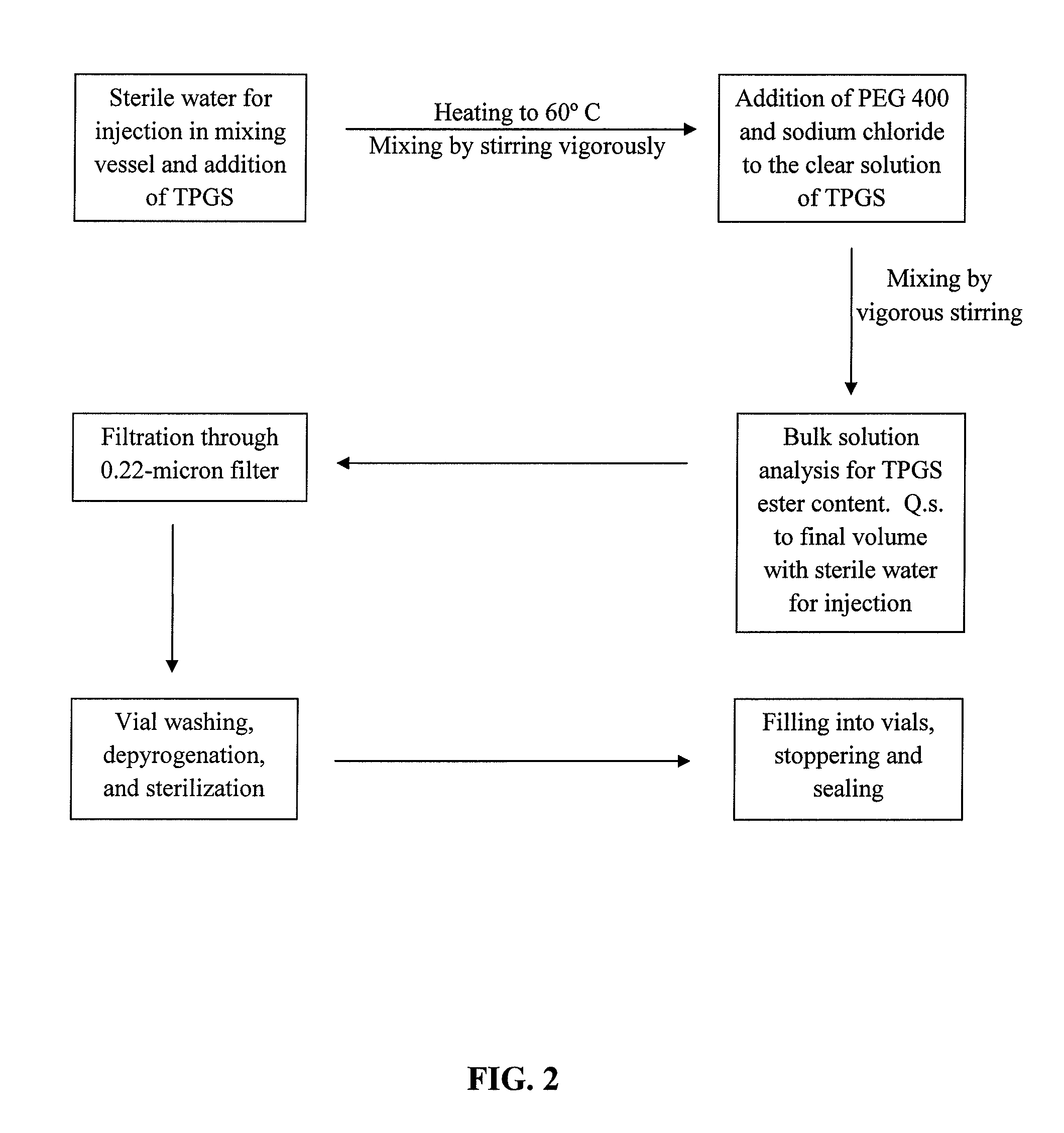
Docetaxel formulations with lipoic acid
Pharmaceutical formulations comprising docetaxel, solubilizer, and α-lipoic acid, wherein the formulation is substantially free of polysorbates and polyethoxylated castor oil. The solubilizer may comprise glycofurol, acetic acid, benzyl alcohol, or ethanol. The α-lipoic acid, at certain concentrations, may impart stability and prevent degradation of docetaxel while the formulations are in storage. The formulations may be combined with a diluent, which comprises one or more hydrotropes such as tocopherol polyethylene glycol succinate and polyethylene glycol. The formulations combined with the diluent also exhibit stability after storage. Methods of administering docetaxel comprise preparing the formulation comprising docetaxel, solubilizer, and α-lipoic acid; mixing the formulation with a diluent; diluting the resulting formulation in saline, water for injection, or the like; and then injecting the formulations into patients in need thereof.
Owner:SCIDOSE
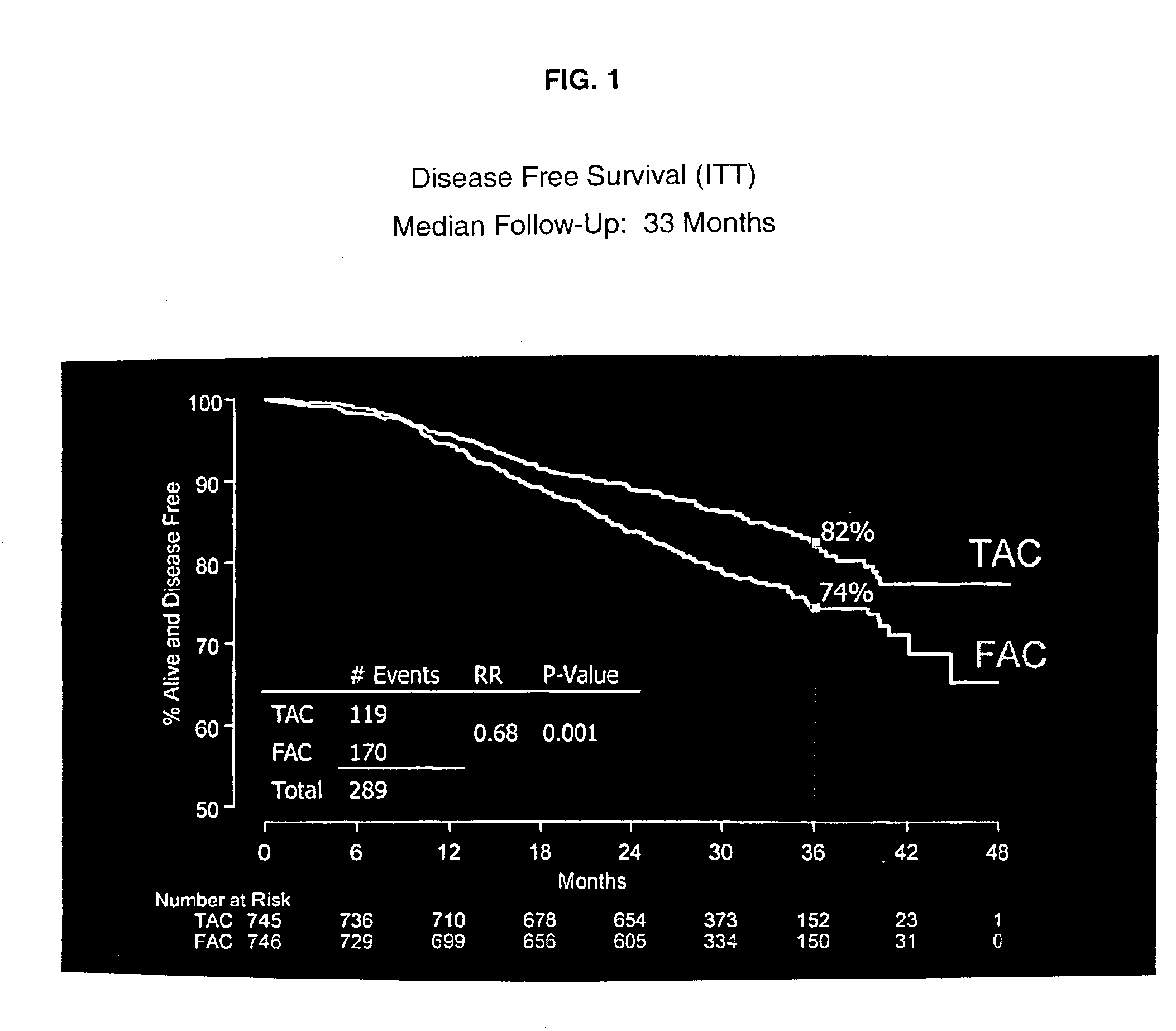
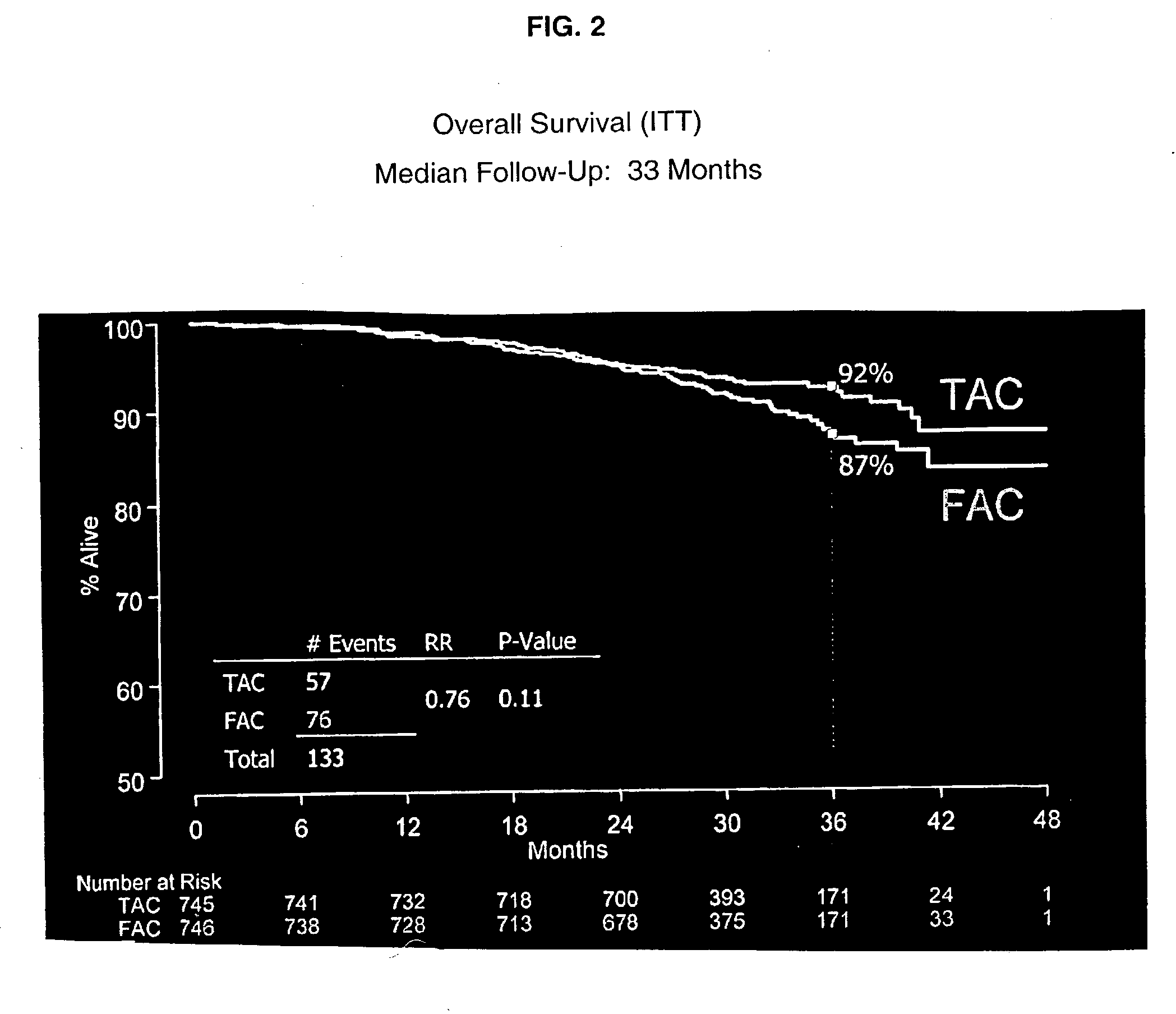
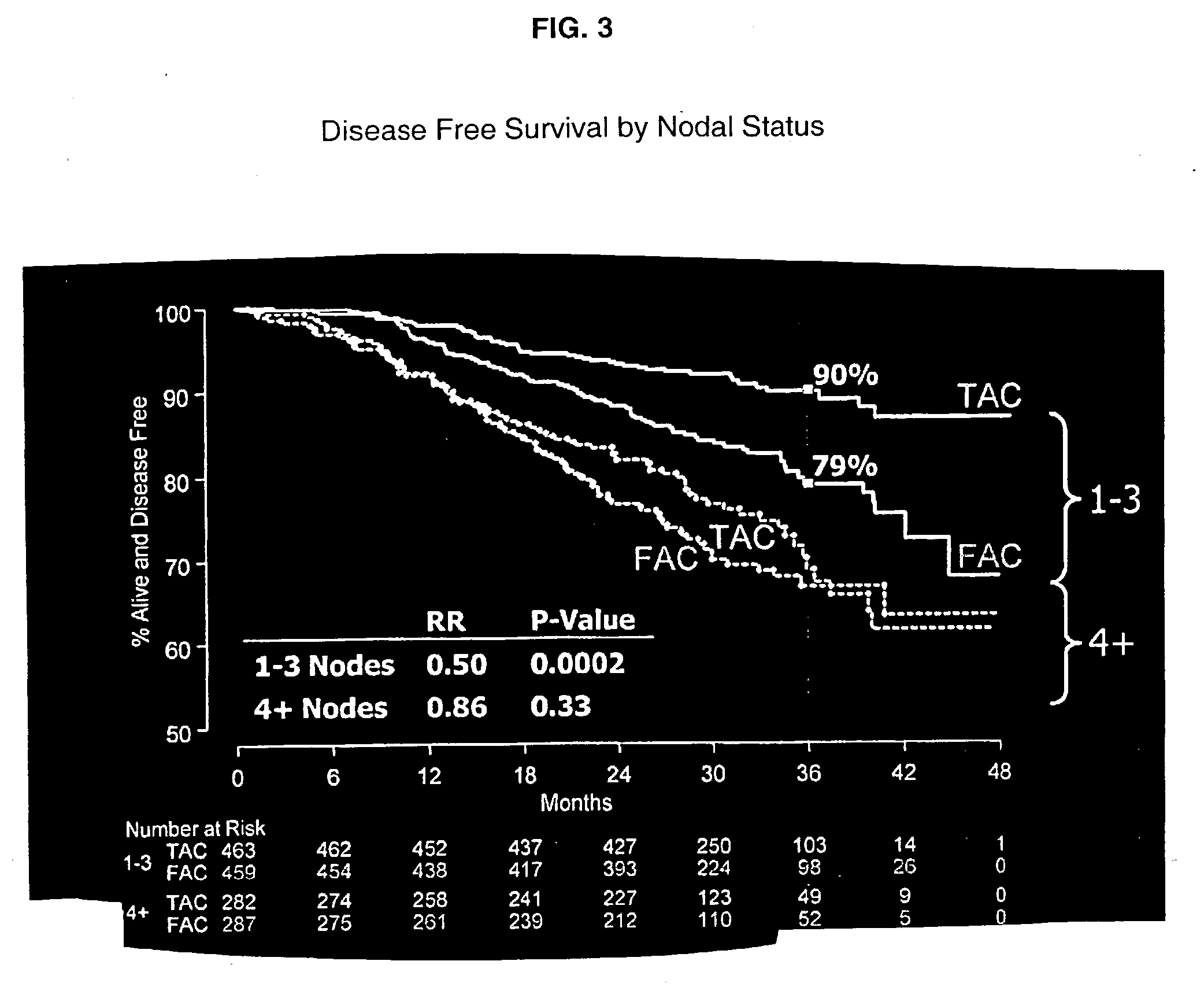
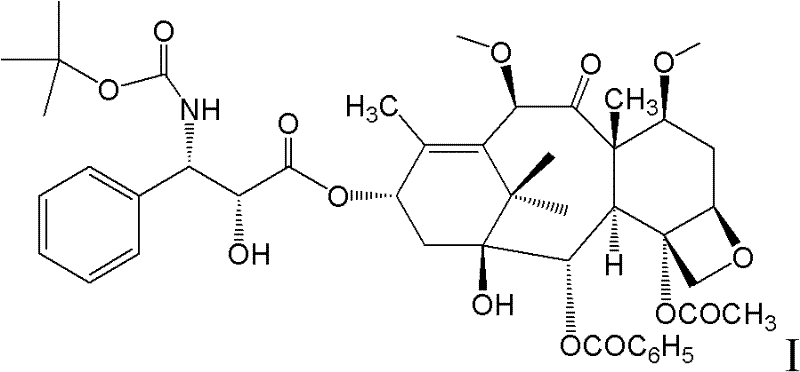
Use of docetaxel/doxorubicin/cyclophosphamide in adjuvant therapy
The present invention relates to a new method of adjuvant therapy in the treatment of early breast cancer, comprising administering six cycles of docetaxel, doxorubicin and cyclophosphamide to a patient in need thereof, wherein said dosages have a marked therapeutic effect when compared to other adjuvant therapies.
Owner:AVENTIS PHARMA SA (US)
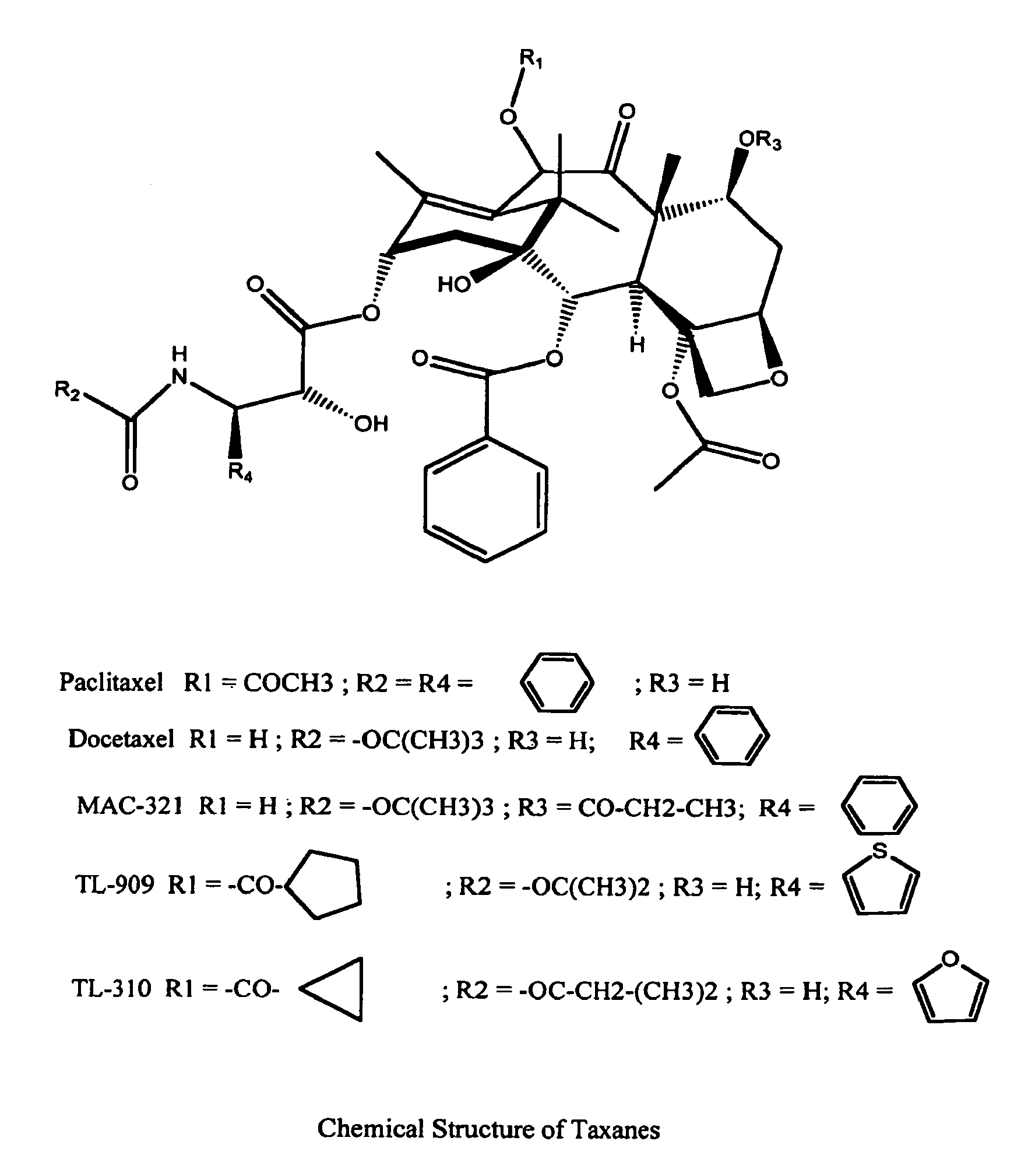
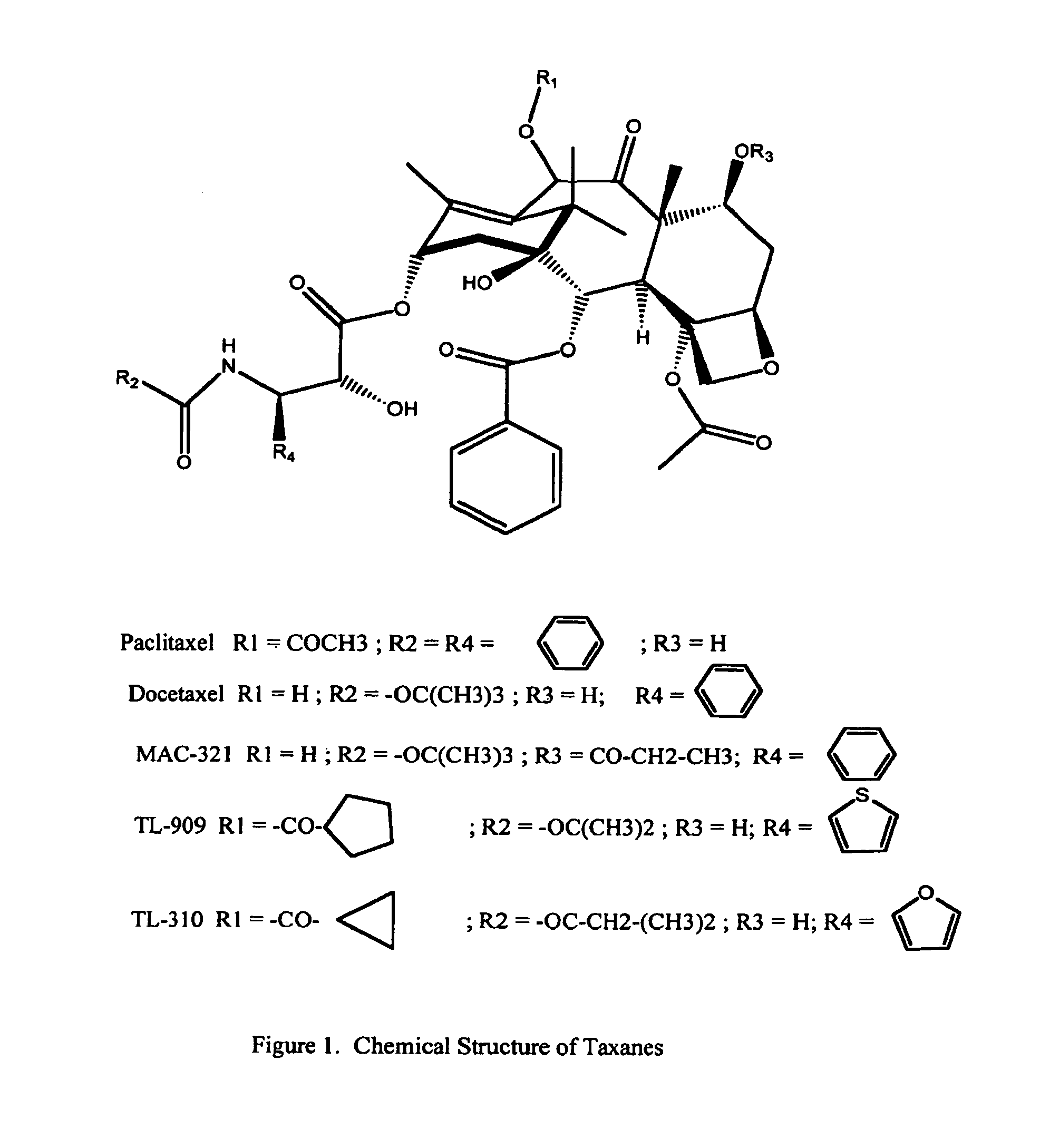
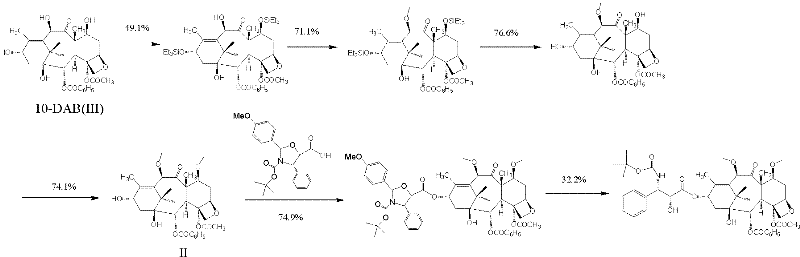
New taxane derivative and preparation method thereof
The invention provides a new taxane derivative and a preparation method thereof as well as a method of preparing antitumor medicaments (cabazitaxel, docetaxel and taxol) by utilizing new taxane isoserine ester.
Owner:SHANGHAI BIOMAN PHARMA
Stable Pharmaceutical Composition Containing Docetaxel and a Method of Manufacturing the Same
InactiveUS20090163574A1Improve stabilityEasily prepared into injectionOrganic active ingredientsBiocideDocetaxel-PNPDocetaxel
The present invention relates to a stable pharmaceutical composition for injection containing docetaxel and a method of preparing the same. More particularly, the present invention relates to a pharmaceutical composition for injection containing docetaxel having better storage stability than conventional medications, which is prepared by dissolving docetaxel, a water-insoluble compound, in distilled water after mixing it with cyclodextrin (CD) and a water-soluble polymer such as hydroxypropyl methylcellulose (HPMC), polyethylene glycol (PEG) or polyvinylpyrrolidone (PVP) and lyophilizing the mixture, and a method of preparing the same.
Owner:SK CHEM CO LTD
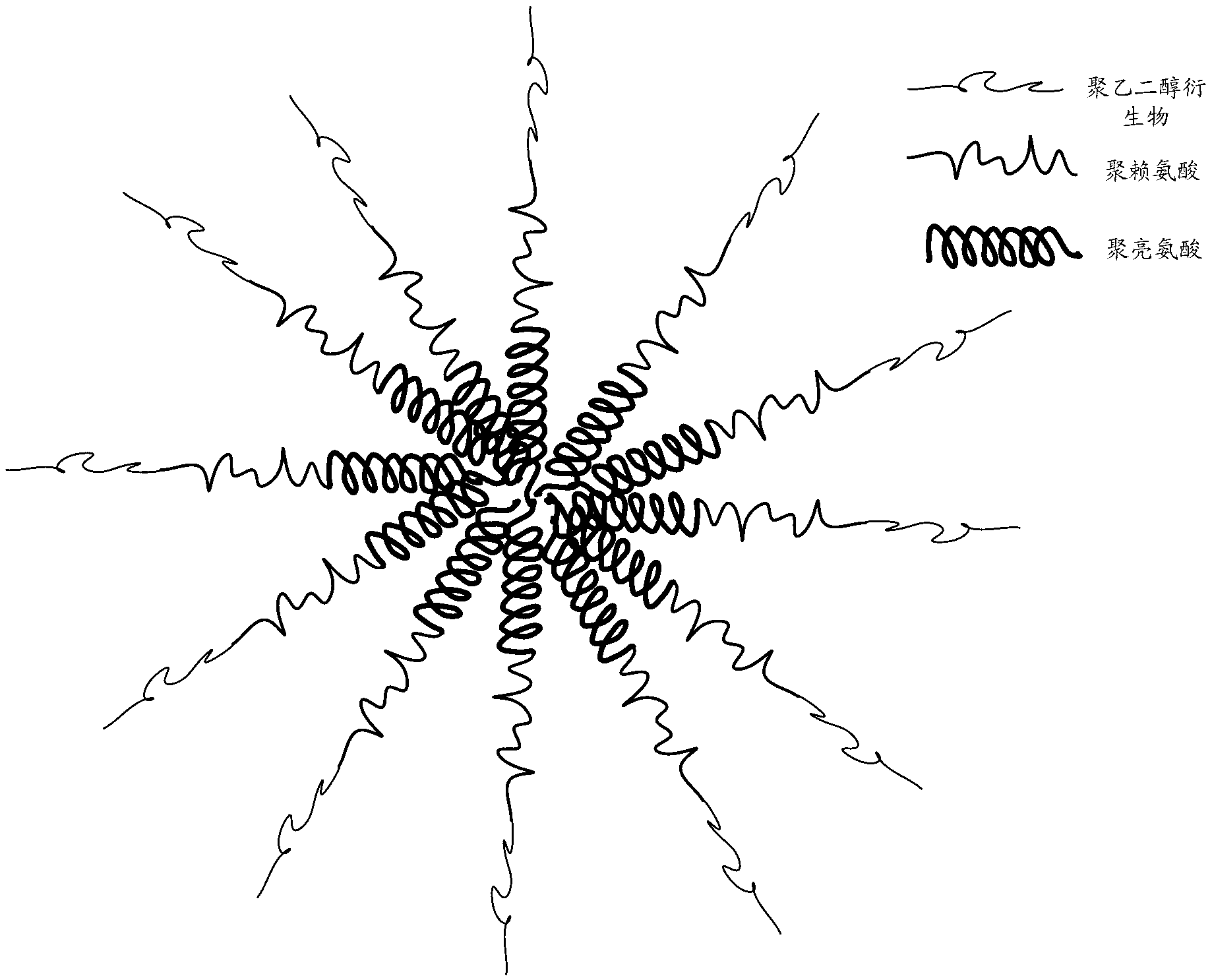
Drug-loading nano-micelles, and preparation method and application thereof
ActiveCN102920649AEasy to prepareEasy to operate and promoteOrganic active ingredientsGenetic material ingredientsDocetaxelPolyethylene glycol
The invention relates to drug-loading nano-micelles, and a preparation method and an application thereof. According to the to drug-loading nano-micelles, a polyethylene glycol derivative-polylysine-polyleucine amphiphilic triblock copolymer is adopted as a carrier, and self-assembly is carried out in a water solution, such that nano-micelles with a three-layer structure transferring gene and medicine are formed. With the hydrophobic effect between polyleucine, the copolymer forms a core of the micelles in water, such that a hydrophobic medicine docetaxel is entrapped. With polylysine, the nano-micelles are positively charged, such that negatively charged anti-apoptotic protein small interfering RNA can be bonded. Polyethylene glycol is used for protecting the stability of the nano-micelles loading the medicines and gene, and a cycle time of the nano-micelles in vivo is prolonged. The nano-micelles are used for loading both an anti-apoptotic protein small interfering RNA gene medicine and a docetaxel medicine, such that the gene treatment medicine and the chemotherapeutic medicine cooperates in treating cancer, and a synergetic effect of the two medicines can be developed. Therefore, a direction is provided for better treatment of breast cancer.
Owner:SHENZHEN INST OF ADVANCED TECH CHINESE ACAD OF SCI
Process of purification of paclitaxel and docetaxel
InactiveUS20030225291A1Simple and cost-effectiveImprove scalabilityOrganic chemistryAlkaneDocetaxel-PNP
A process for the purification of paclitaxel comprising: (a) mixing crude paclitaxel with a mixture of solvents such as alkane and chlorinated alkane, filtering the solid followed by drying to obtain paclitaxel of increased purity; (b) repeating step (a) one or more times to obtain paclitaxel of increased purity; (c) dissolving the solid obtained from step (b) in alkyl ketone followed by adding alkane thereto, filtering and drying the solid thus formed to obtain paclitaxel of increased purity; (d) repeating step (c) one or more times to increase the purity of paclitaxel; (e) dissolving the paclitaxel obtained from step (d) in alkanol and then adding water, filtering and drying the solid thus formed, to obtain paclitaxel of increased purity; and (f) dissolving the solid obtained from step (e) in alkyl ketone, filtering, followed by adding alkane to the filtrate, filtering and drying the solid thus formed to obtain pure paclitaxel.
Owner:DABUR PHARM LTD
Fat emulsion containing docetaxel and its preparing method
InactiveCN1709236ASolve the existing technology deficiencies of low solubilityImprove securityOrganic active ingredientsDiseaseVegetable oil
The present invention relates to an infatmul containing docetaxel and its preparation method. Said preparation can be directly used for intravenous injection. At the same time of that it is used as medicine for curing tumor disease said preparation also can provide nutrients for patient. The weight volume concentration range of docetaxel contained by said preparation is 0.1-1.0 mg / ml, and its specification is that 1-500 mg of active component docetaxel is contained. Said infatmul composition includes docetaxel, vegetable oil, phospholipids and injection water, at the same time the components of glycerin and group emulsion, etc. can be added, in which the optimum group emulsion is poloxamer 188. Besides, said invention also provides the concrete steps of its preparation method and its application in preparation of medicine for resisting tumor.
Owner:CISEN PHARMA
Prediction of Breast Cancer Response to Taxane-Based Chemotherapy
InactiveUS20090239223A1High degree of correlationReliable distinctionMicrobiological testing/measurementDocetaxel-PNPDocetaxel
The invention relates to methods and kits for the prediction of a likely outcome of chemotherapy in a cancer patient. More specifically, the invention relates to the prediction of tumour response to chemotherapy based on measurements of expression levels of a small set of marker genes. The set of marker genes is useful for the identification of breast cancer subtypes responsive to taxane based chemotherapy, such as e.g. a taxane-anthracycline-cyclophosphamide-based (e.g. Taxotere (docetaxel)-Adriamycin (doxorubicin)-cyclophosphamide, i.e. (TAC)-based) chemotherapy.
Owner:SIEMENS HEALTHCARE DIAGNOSTICS INC
Long-circulating solid lipid docetaxel nanoparticles and preparation method thereof
InactiveCN101653414AGood water solubilityImprove stabilityOrganic active ingredientsAntineoplastic agentsLipid formationSolubility
The invention discloses long-circulating solid lipid docetaxel nanoparticles and a preparation method thereof. The long-circulating solid lipid docetaxel nanoparticles comprise the following materialsin therapeutic effective dose: docetaxel, lipid materials, long-circulating auxiliary materials and an emulsifier. The long-circulating solid lipid docetaxel nanoparticles have small particle size, high encapsulation rate and good stability, and not only improve the solubility and the stability of the docetaxel, reduce the toxicity of the docetaxel, but also prolong the circulating time of a medicament in blood, and improve the therapeutic index of the medicament, so that the preparation has the characteristics of low toxicity, low allergy, high efficiency and targeting in clinical application.
Owner:SHANGHAI INST OF MATERIA MEDICA CHINESE ACAD OF SCI
Docetaxel solid lipid nanoparticle and preparation method thereof
InactiveCN102579341AGood chemical stabilityImprove stabilityPowder deliveryOrganic active ingredientsTumour targetingDocetaxel
The invention belongs to the field of medicine preparation and particularly relates to a docetaxel solid lipid nanoparticle and a preparation method thereof. The docetaxel solid lipid nanoparticle consists of an effective curative dose of docetaxel, solid lipid materials, liquid lipid materials, an emulsifying agent, additives, long-circulating auxiliary materials and water for injection. The docetaxel solid lipid nanoparticle provided by the invention has the characteristics of stable docetaxel structure, phagocytosis prevention of an in-vivo reticuloendothelial system and active tumor targeting, and can be stably stored.
Owner:SHENYANG PHARMA UNIVERSITY
Mesoporous silica nano-preparation and its preparation method and use
InactiveCN103990130ANo releaseQuick releaseNanomedicinePharmaceutical non-active ingredientsDocetaxel-PNPDocetaxel
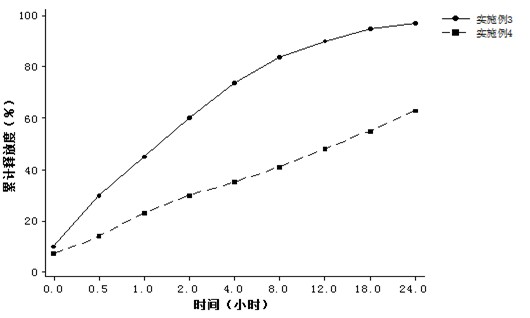
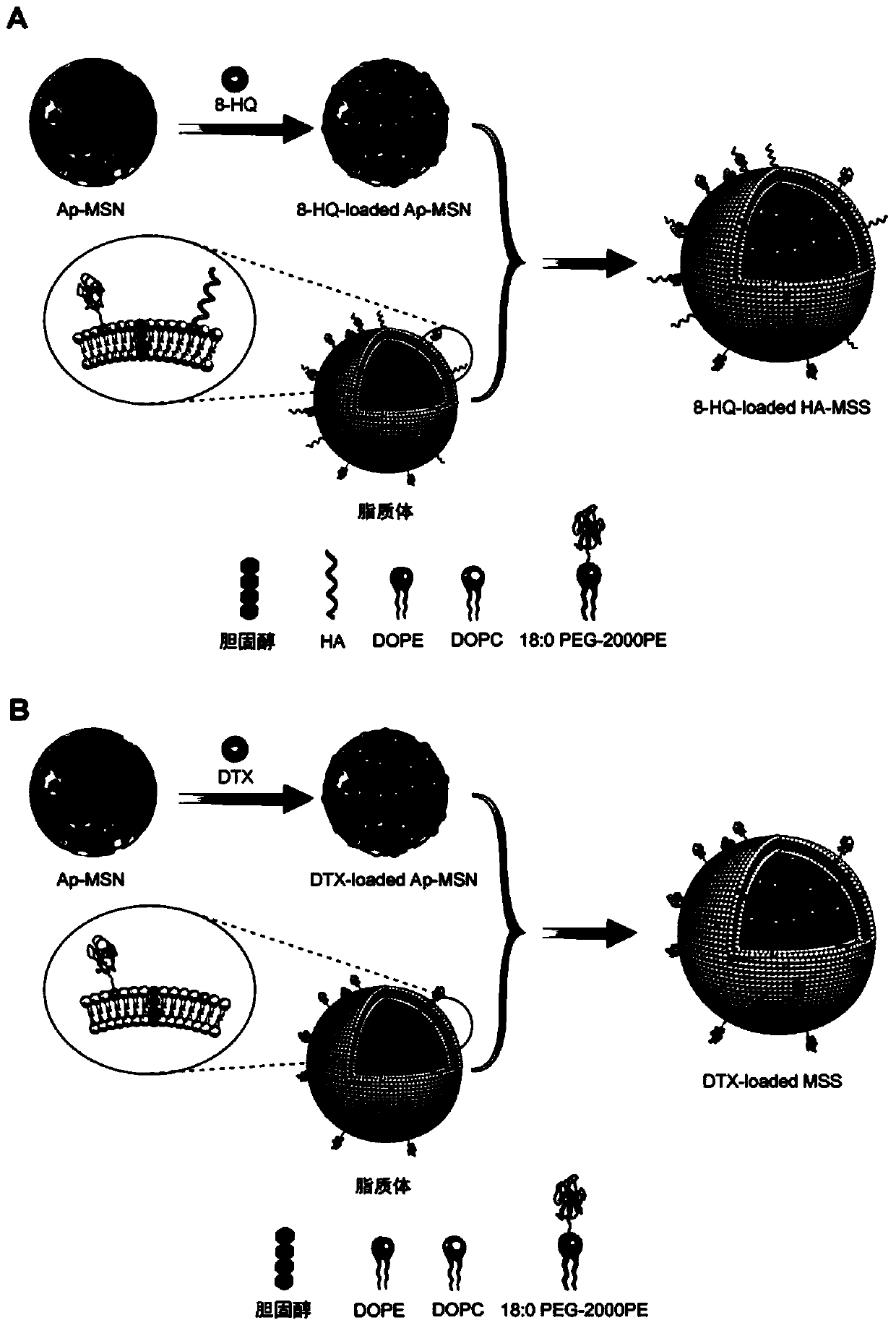
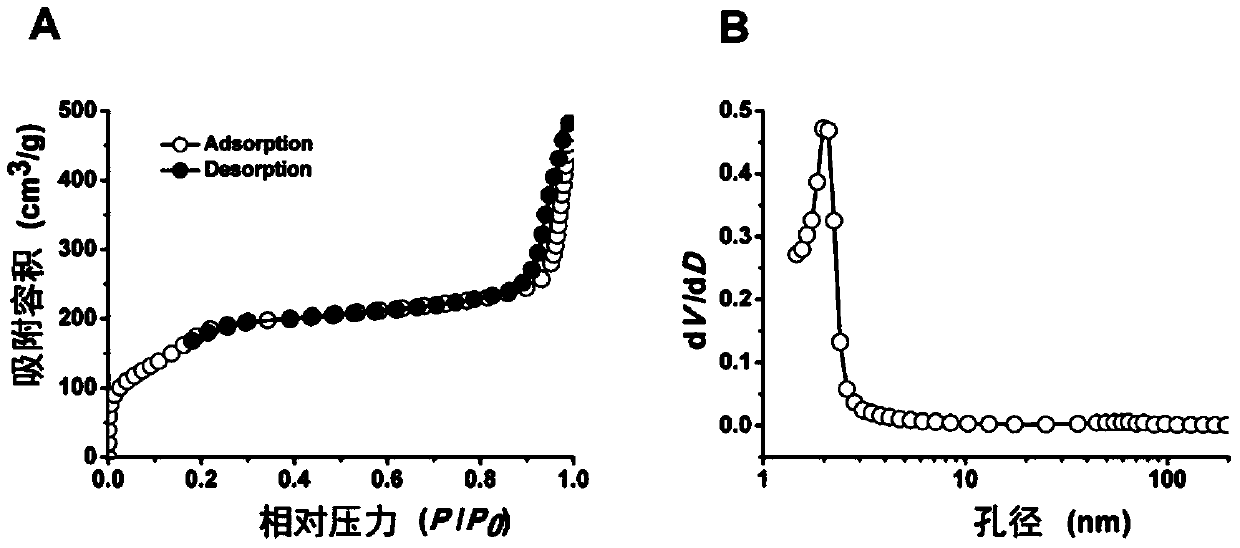
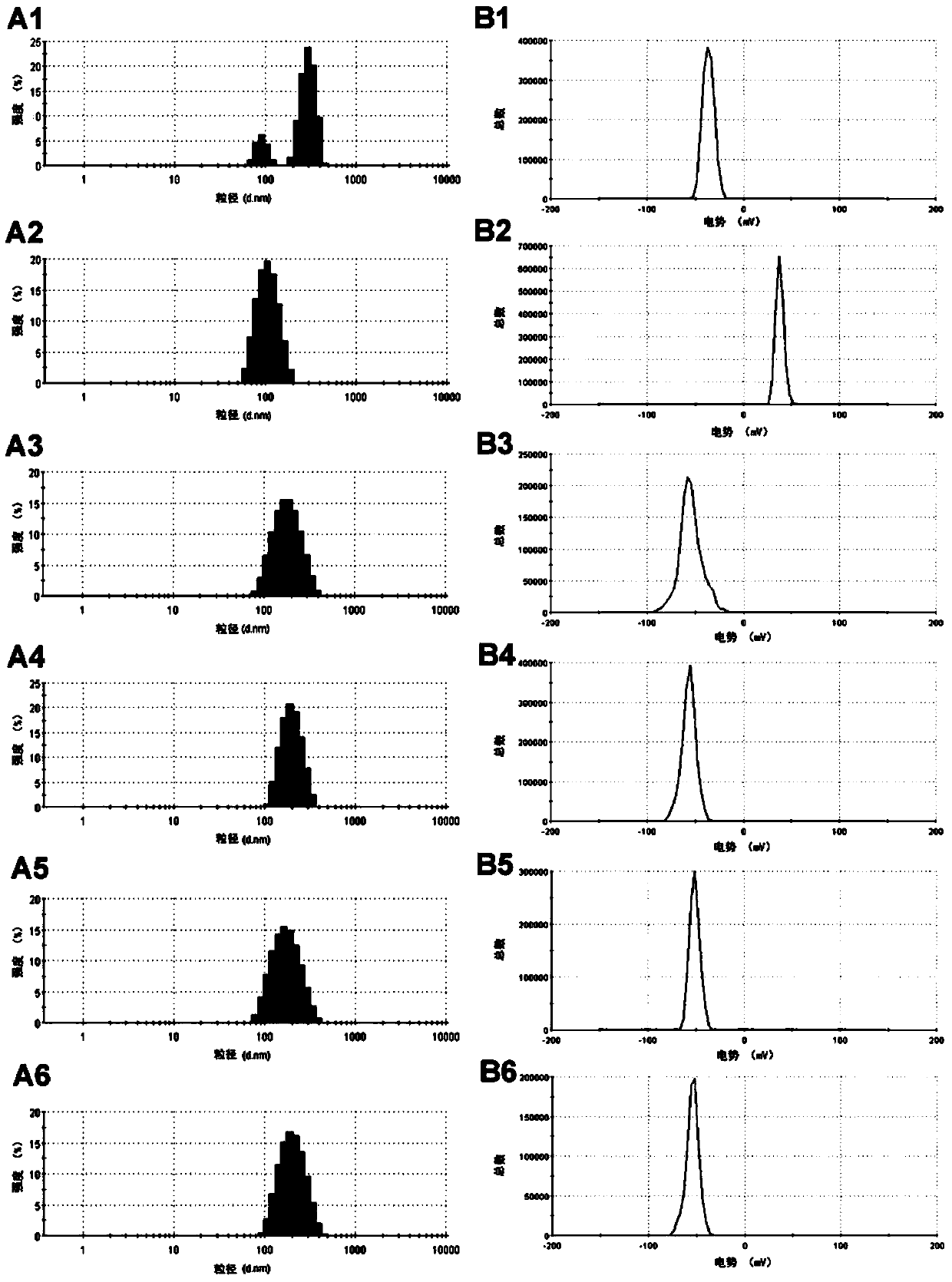
The invention discloses a mesoporous silica nano-preparation. The mesoporous silica nano-preparation comprises docetaxel or 8-hydroxyquinoline, mesoporous silica nanoparticles, and a lipid double-molecule layer. The mesoporous silica nanoparticles carry docetaxel or 8-hydroxyquinoline. The surfaces of the mesoporous silica nanoparticles carrying docetaxel or 8-hydroxyquinoline are coated with the lipid double-molecule layer. The invention also discloses a preparation method and a use of the mesoporous silica nano-preparation. The mesoporous silica nano-preparation carrying docetaxel or 8-hydroxyquinoline can substantially improve antitumor activity of a drug, can effectively reduce tumor recurrence and nonspecific toxicity and has a very wide application prospect.
Owner:SECOND MILITARY MEDICAL UNIV OF THE PEOPLES LIBERATION ARMY
Taxanes medicine preparation for intravenous injection and preparation method thereof
ActiveCN101288642AGood biocompatibilityHigh tolerance in vivoOrganic active ingredientsSolution deliveryDrugs solutionDocetaxel
The invention relates to the technical field of medicine, which is a preparation of a taxane drug for intravenous drug delivery, consisting of two parts of drug solution and an emulsion. The drug solution is composed of paclitaxel or docetaxel, a pH regulator and a solvent for injection, wherein, the solvent for injection is an organic solvent; the emulsion comprises a fat emulsion and is composed of oil for injection, an emulsifier, an antioxidant, an isotonic regulator, a stabilizer, a pH regulator and water for injection. When in use, the drug solution can be added and evenly mixed in the emulsion for direct intravenous drip according to the clinical drug dosage and can also be firstly added in the emulsion with the volume that is not less than 5 times of the volume of the drug solution according to the clinical drug dosage and then added with a certain amount of physiological saline or glucose injection for intravenous drip. The preparation of the invention does not contain solubilizer and has the advantages of little toxicity, safety, effectivity, stability and economy. The fat emulsion can also be taken as a nutrition replenisher for a patient, thus achieving better treatment effects. The physiological saline or the glucose injection can also replace a certain amount of the emulsion, so the storage and the transportation are convenient, and the preparation is more economical.
Owner:JIANGSU TASLY DIYI PHARMA CO LTD