Patents
Literature
3571 results about "Allergy" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Allergies, also known as allergic diseases, are a number of conditions caused by hypersensitivity of the immune system to typically harmless substances in the environment. These diseases include hay fever, food allergies, atopic dermatitis, allergic asthma, and anaphylaxis. Symptoms may include red eyes, an itchy rash, sneezing, a runny nose, shortness of breath, or swelling. Food intolerances and food poisoning are separate conditions. Common allergens include pollen and certain food. Metals and other substances may also cause problems. Food, insect stings, and medications are common causes of severe reactions. Their development is due to both genetic and environmental factors.
Avenanthramide-containing compositions
Methods and compositions for treating or preventing a skin condition, an inflammation, an irritation or an allergy associated with an ectoparasitic infection or infestation on an animal. The methods involve applying to the skin of the animal a pharmaceutical composition that contains a therapeutically effective amount of one or more than one avenanthramide, an optional ecto and / or endo-parasiticidal agent, and a pharmaceutically acceptable diluent or carrier
Owner:CEAPRO
Method for the generation of antigen-specific lymphocytes
InactiveUS20070116690A1Function increaseEnhancing function of T cellBiocideVirusesAutoimmune conditionAutoimmune disease
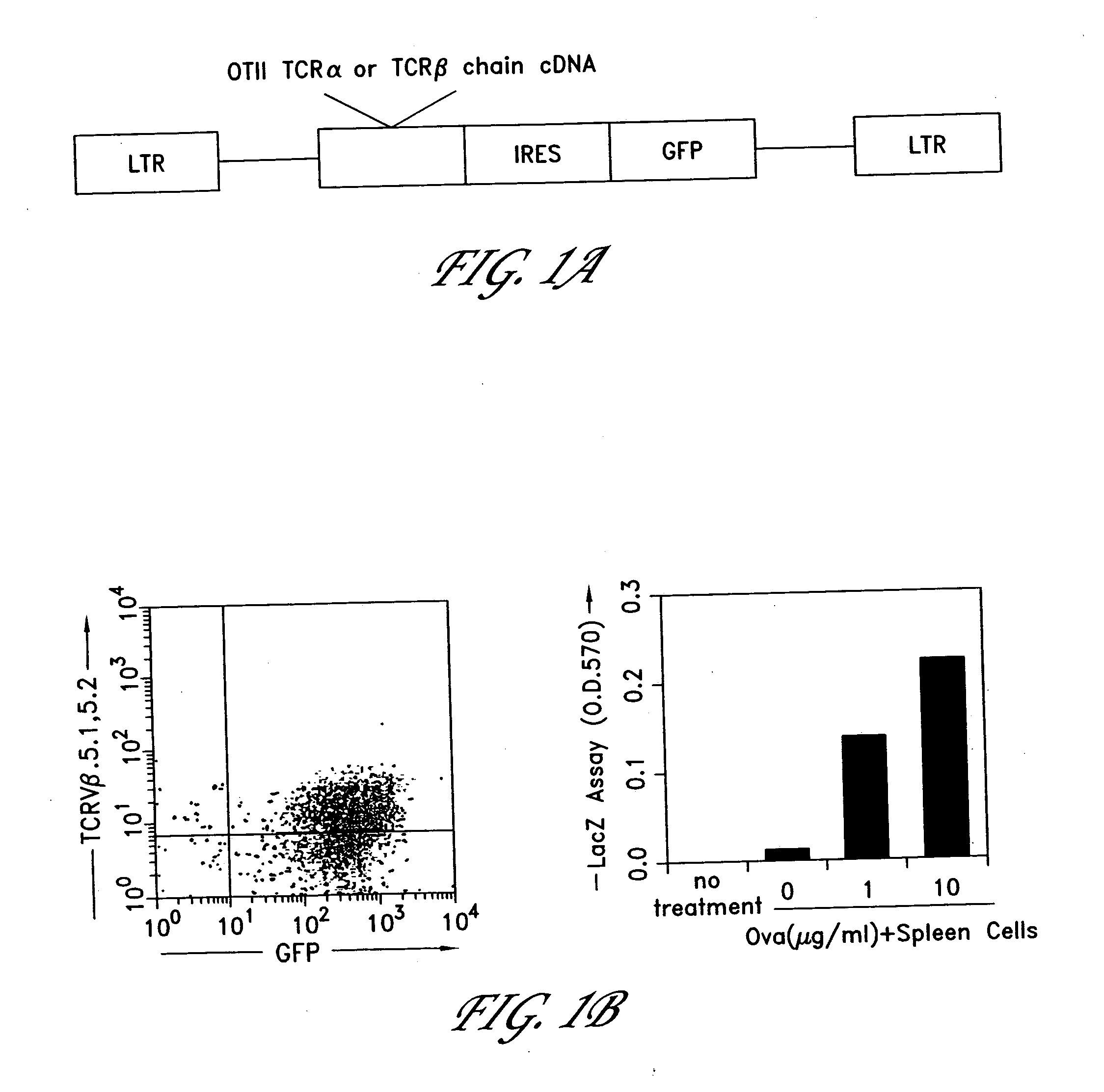
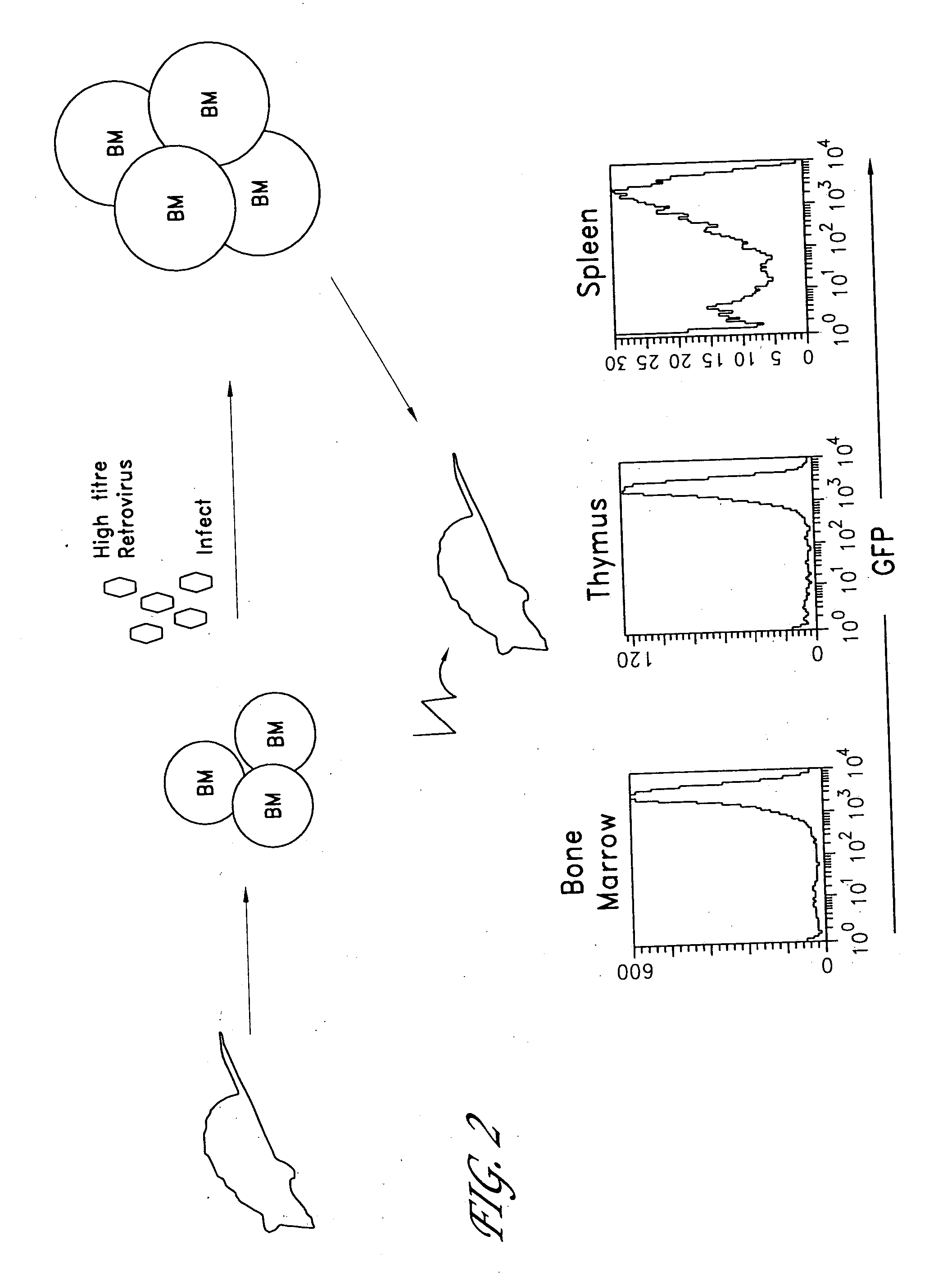
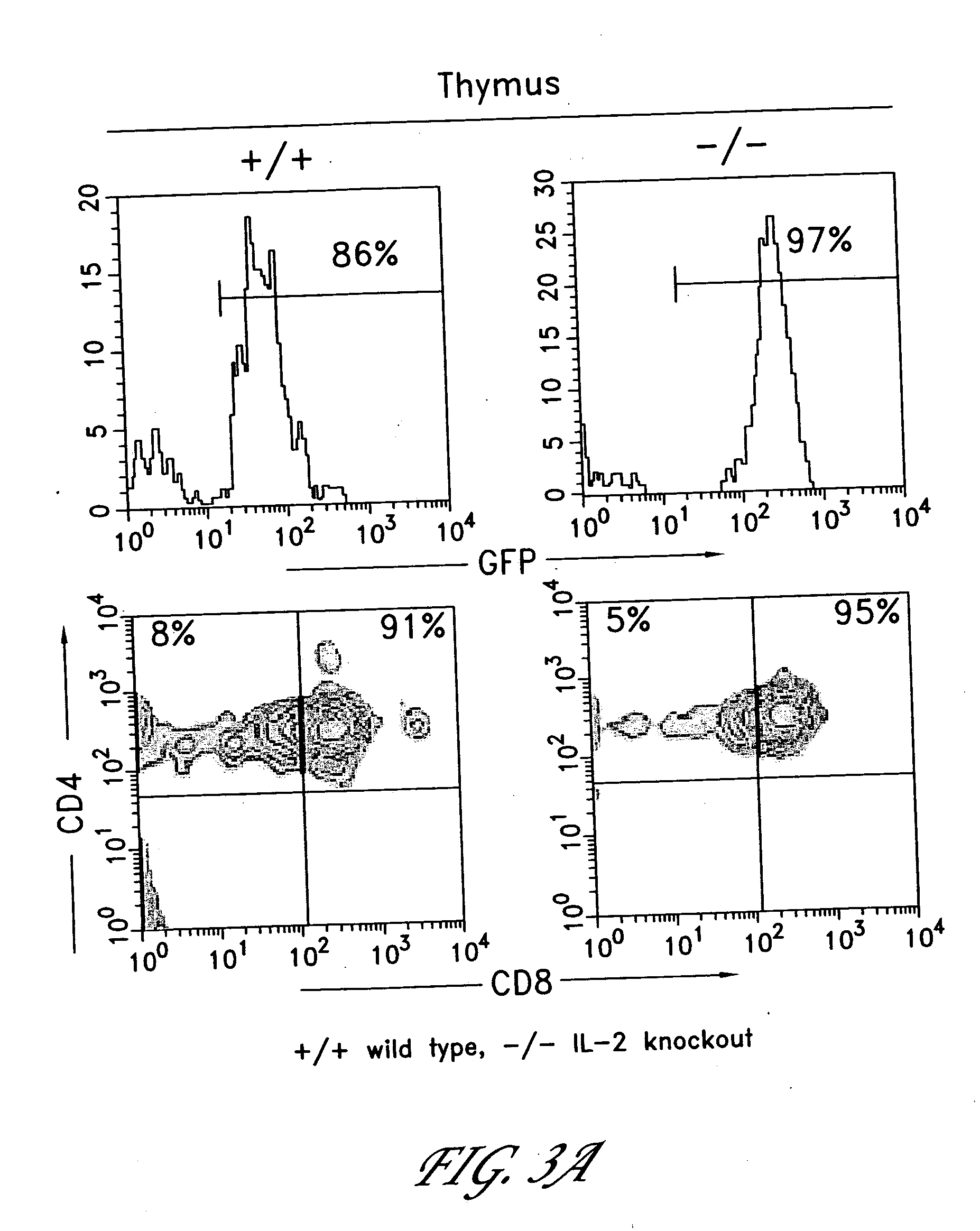
The invention provides systems and methods for the generation of lymphocytes having a unique antigen specificity. In a preferred embodiment, the invention provides methods of virally infecting cells from bone marrow with one or more viral vectors that encode antigen-specific antibodies for the production of, for example B cells and T cells. In some embodiments, the viral vectors include an IRES or 2A element to promote separation of, for example, the α subunit and β subunit of a T cell receptor (TCR) or heavy and light chains of a B-cell antibody. The resulting lymphocytes, express the particular antibody that was introduced in the case of B cells and TCR in the case of T cells. The lymphocytes generated can be used for a variety of therapeutic purposes including the treatment of various cancers and the generation of a desired immune response to viruses and other pathogens. The resulting cells develop normally and respond to antigen both in vitro and in vivo. We also show that it is possible to modify the function of lymphocytes by using stem cells from different genetic backgrounds. Thus our system constitutes a powerful tool to generate desired lymphocyte populations both for research and therapy. Future applications of this technology may include treatments for infectious diseases, such as HIV / AIDS, cancer therapy, allergy, and autoimmune disease.
Owner:CALIFORNIA INST OF TECH
8-Substituted Benzoazepines as Toll-Like Receptor Modulators
Provided are compositions and methods useful for modulation of signaling through the Toll-like receptors TLR7 and / or TLR8. The compositions and methods have use in the treatment of autoimmunity, inflammation allergy, asthma, graft rejection, graft versus host disease, infection, sepsis, cancer and immunodeficiency.
Owner:ARRAY BIOPHARMA
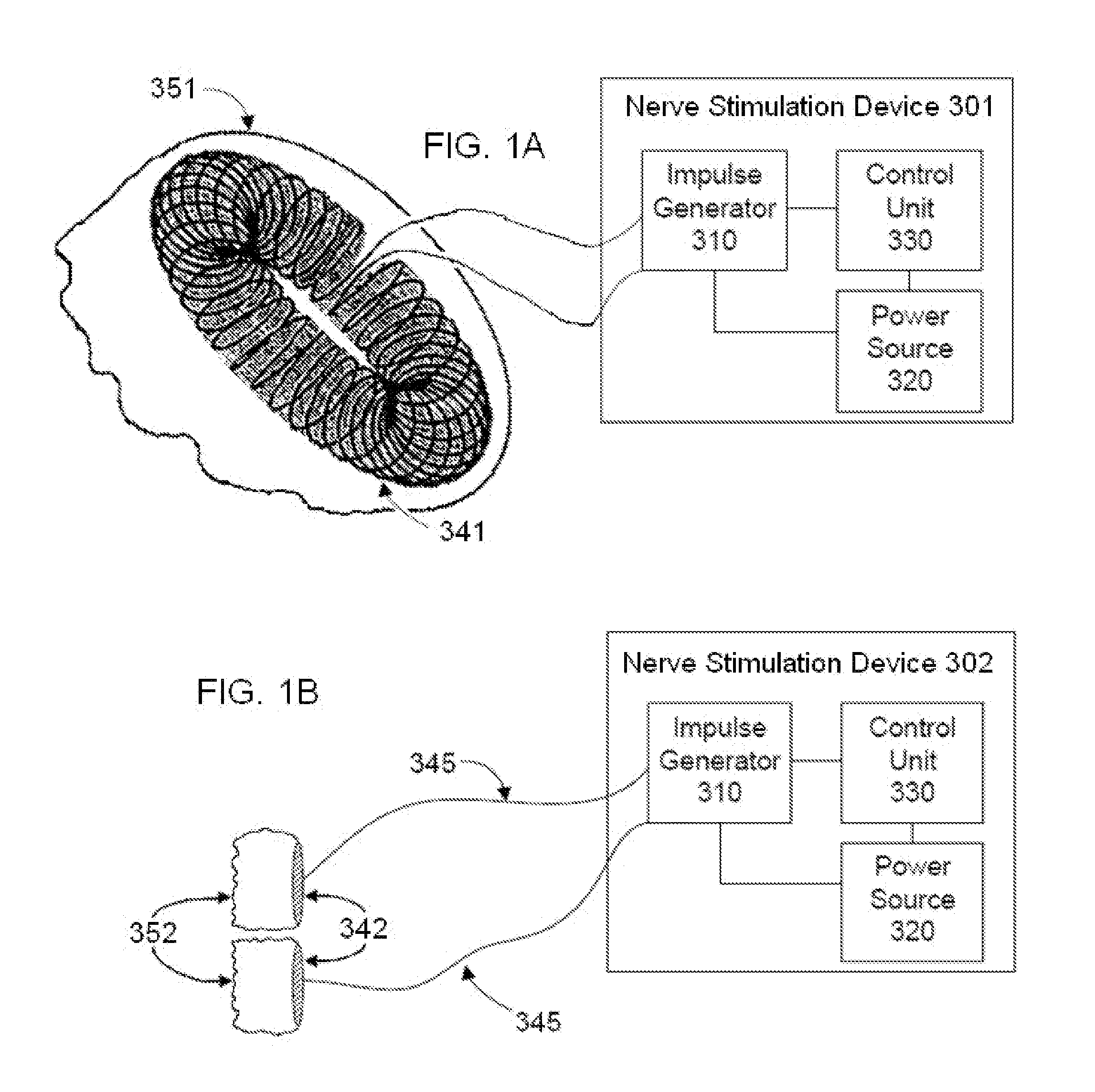
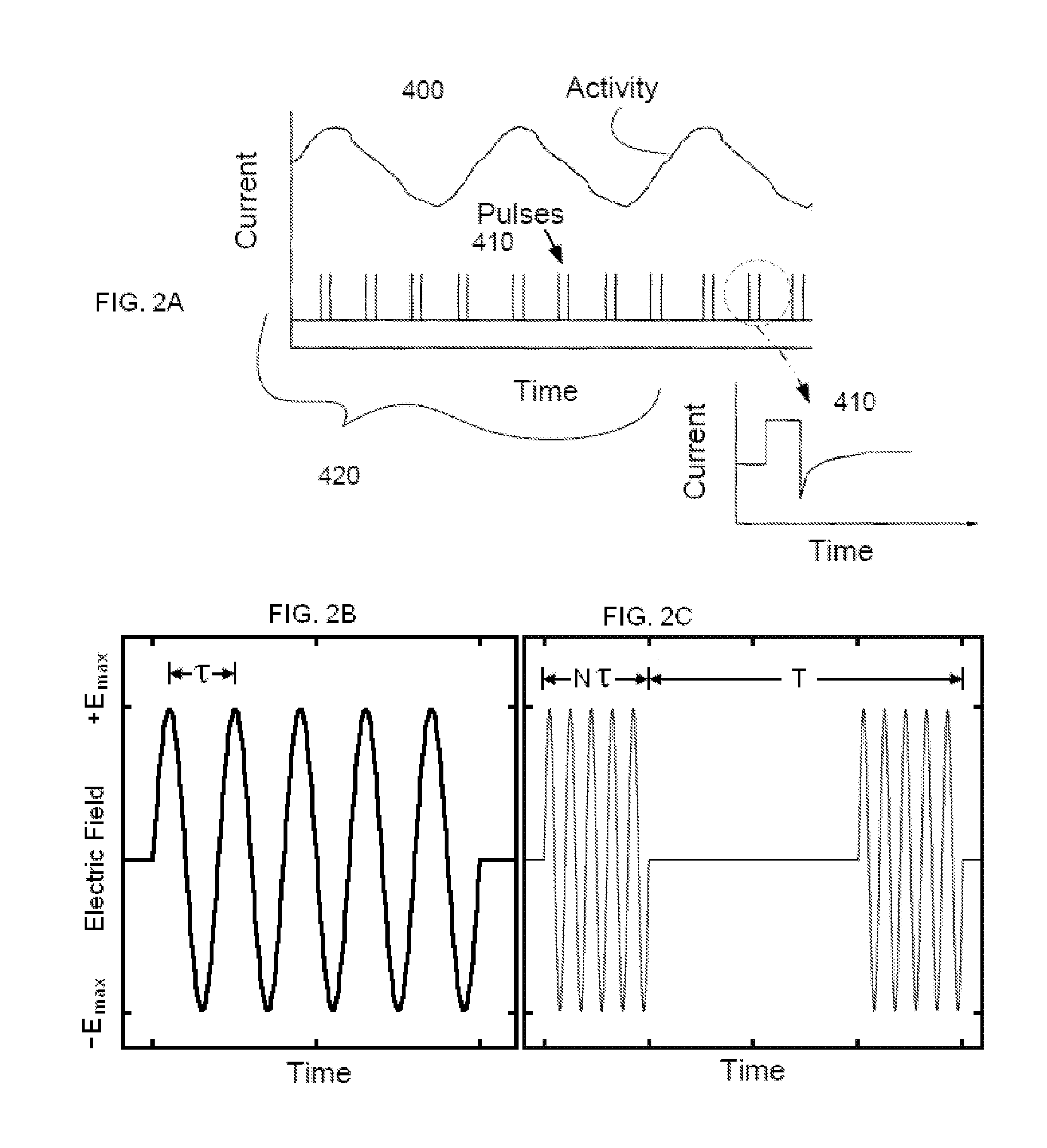
Electrical and magnetic stimulators used to treat migraine/sinus headache and comorbid disorders
Non-invasive electrical nerve stimulation devices and magnetic stimulation devices are disclosed, along with methods of treating medical disorders using energy that is delivered noninvasively by such devices. The disorders comprise migraine and other primary headaches such as cluster headaches, including sinus symptoms that resemble an immune-mediated response (“sinus” headaches), irrespective of whether those symptoms arise from an allergy that is co-morbid with the headache. The disclosed methods may also be used to treat other disorders that may be co-morbid with migraine headaches, such as anxiety disorders. In preferred embodiments of the disclosed methods, one or both of the patient's vagus nerves are stimulated non-invasively. In other embodiments, parts of the sympathetic nervous system and / or the adrenal glands are stimulated.
Owner:ELECTROCORE
Small molecule toll-like receptor (TLR) antagonists
The invention provides methods and compositions useful for modulating signaling through Toll-like receptors. The methods involve contacting a TLR-expressing cell with a small molecule having a core structure including at least two rings. Certain of the compounds are 4-primary amino quinolines. Many of the compounds and methods are useful specifically for inhibiting immune stimulation involving at least one of TLR9, TLR8, TLR7, and TLR3. The methods may have use in the treatment of autoimmunity, inflammation, allergy, asthma, graft rejection, graft versus host disease, infection, sepsis, cancer, and immunodeficiency.
Owner:COLEY PHARMA GMBH +1
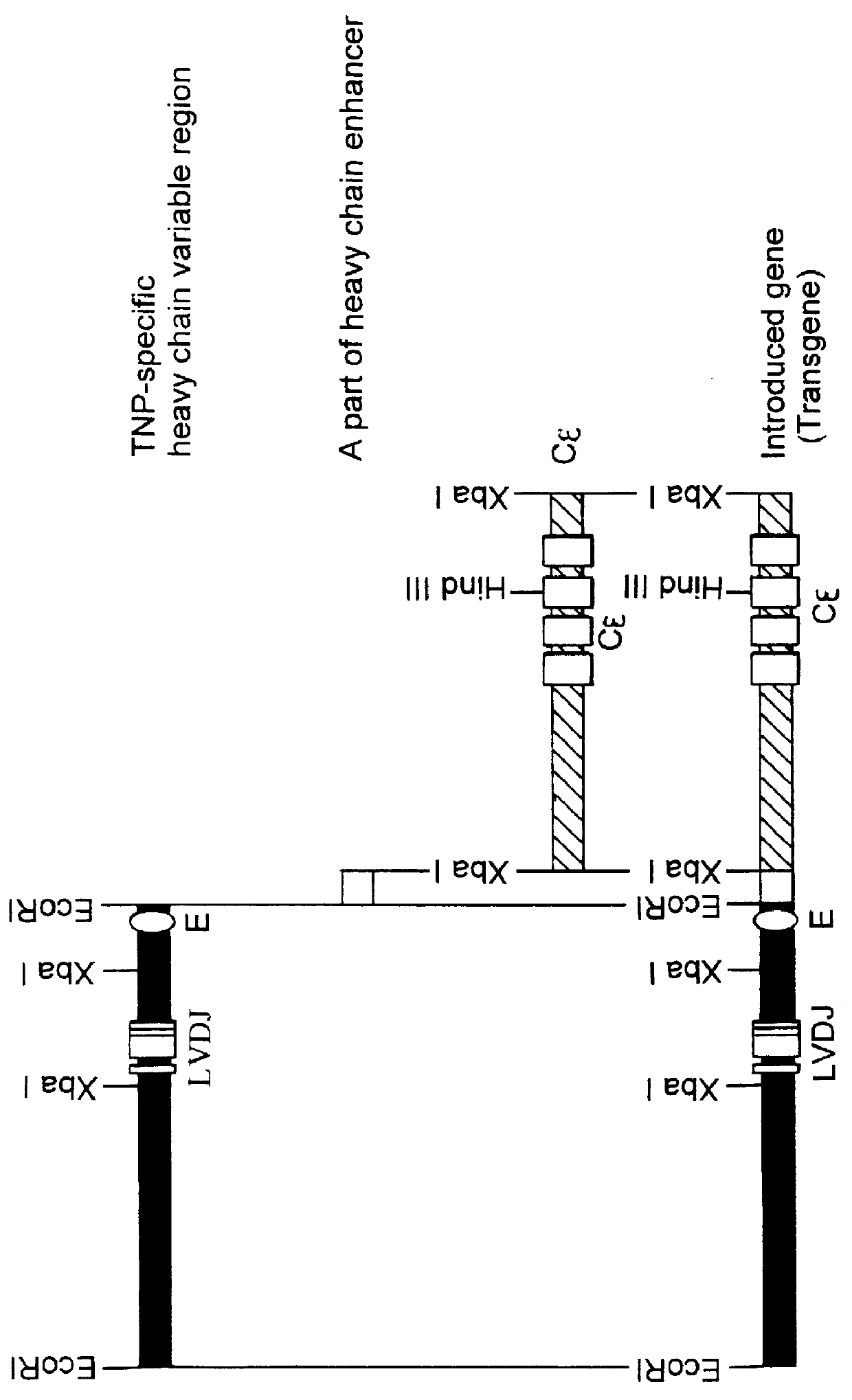
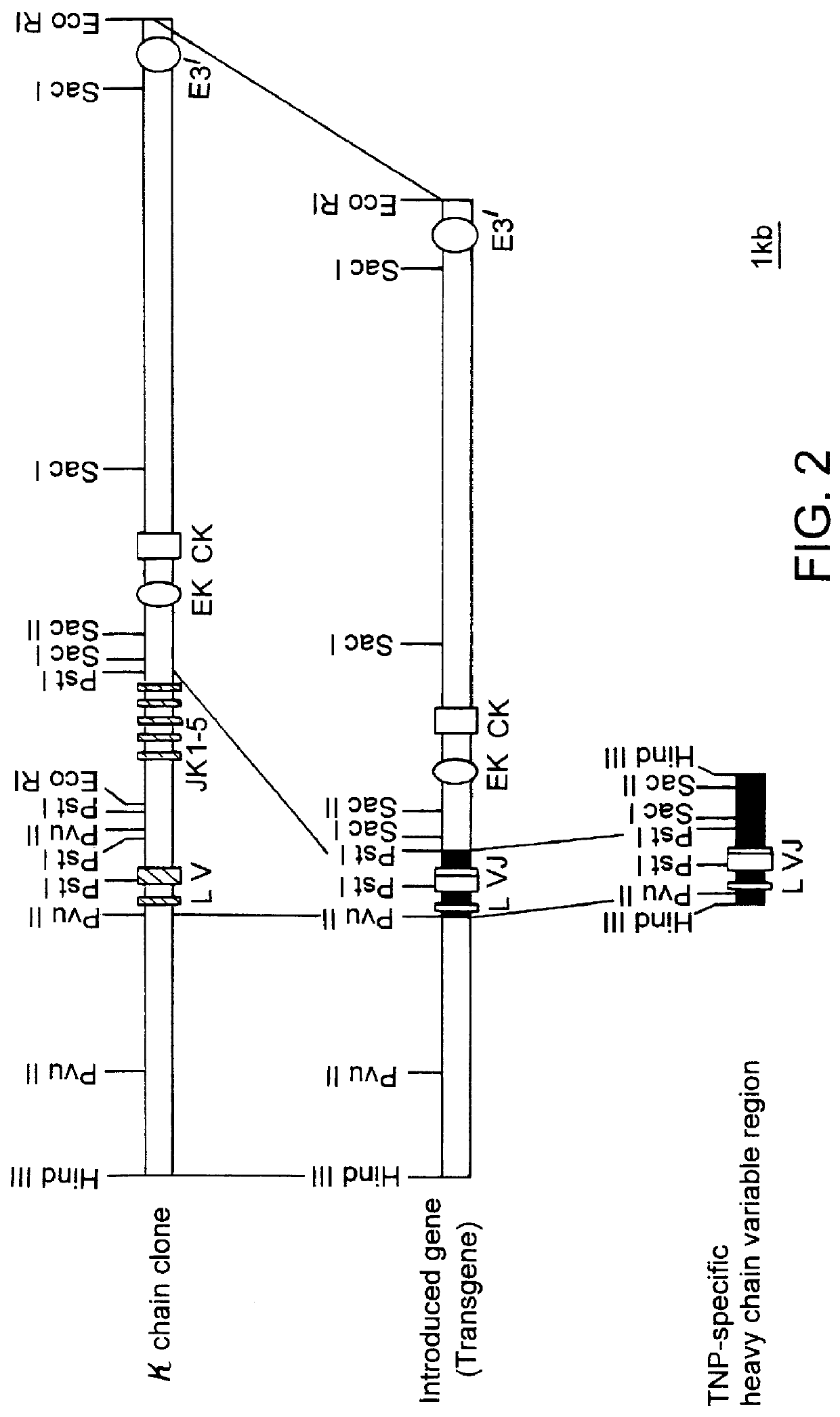
Transgenic mouse allergy models and methods for their use
InactiveUS6118044AImprove import efficiencySufficient amountMicrobiological testing/measurementTissue cultureAntibody typesAllergy
Transgenic mice which constitutively express an antibody-type molecule encoded by the transgene and which has an IgE heavy chain constant region and is specific for a pre-defined antigen, provide an allergic reaction to that antigen without prior sensitization and are useful as allergy models.
Owner:SANKYO CO LTD +1
Aminodiazepines as Toll-Like Receptor Modulators
Provided are compositions and methods useful for modulation signaling through the Toll-like receptor TLR8. The compositions and methods have use in the treatment of autoimmunity, inflammation allergy, asthma, graft rejection, graft versus host disease, infection, sepsis, cancer and immunodeficiency.
Owner:ARRAY BIOPHARMA
Chemical modifications of monomers and oligonucleotides with cycloaddition
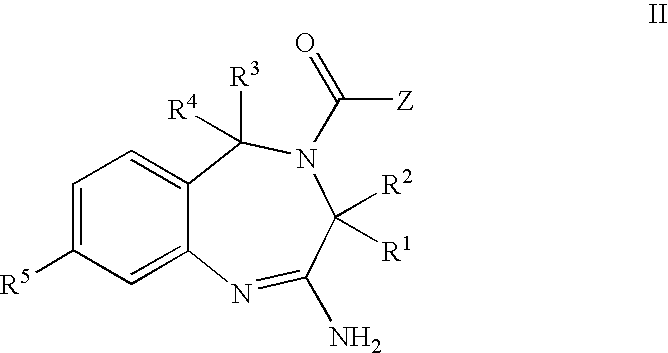
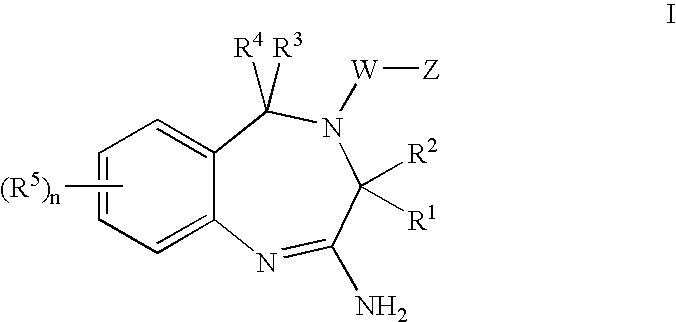
The invention features compounds of formula I or II:In one embodiment, the invention relates compounds and processes for conjugating ligand to oligonucleotide. The invention further relates to methods for treating various disorders and diseases such as viral infections, bacterial infections, parasitic infections, cancers, allergies, autoimmune diseases, immunodeficiencies and immunosuppression.
Owner:ALNYLAM PHARMA INC
Compositions, formulations and kit with anti-sense oligonucleotide and anti-inflammatory steroid and/or obiquinone for treatment of respiratory and lung disesase
InactiveUS20070021360A1Decreased airwayOrganic active ingredientsBiocideDiseaseAntiendomysial antibodies
A pharmaceutical composition and formulations comprise preventative, prophylactic or therapeutic amounts of an oligo(s) anti-sense to a specific gene(s) or its corresponding mRNA(s), and a glucocorticoid and / or non-glucocorticoid steroid or a ubiquinone or their salts. The agents, composition and formulations are used for treatment of ailments associated with impaired respiration, bronchoconstriction, lung allergy(ies) or inflammation, and abnormal levels of adenosine, adenosine receptors, sensitivity to adenosine, lung surfactant and ubiquinone, such as pulmonary fibrosis, vasoconstriction, inflammation, allergies, allergic rhinitis, asthma, impeded respiration, lung pain, cystic fibrosis, bronchoconstriction, COPD, RDS, ARDS, cancer, and others. The present treatment is effectively administered by itself for conditions without known therapies, as a substitute for therapies exhibiting undesirable side effects, or in combination with other treatments, e.g. before, during and after other respiratory system therapies, radiation, chemotherapy, antibody therapy and surgery, among others. Each of the agents of this invention may be administered directly into the respiratory system so that they gain direct access to the lungs, or by other effective routes of administration. A kit comprises a delivery device, the agents and instructions for its use.
Owner:EPIGENESIS PHARMA LLC
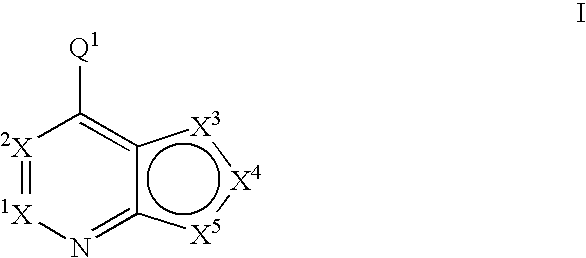
6,6-Bicyclic ring substituted heterobicyclic protein kinase inhibitors
ActiveUS20060235031A1Treatment and/or prevention of hyperproliferative diseasesBiocideSenses disorderDiseasePTK Inhibitors
Compounds of the formula and pharmaceutically acceptable salts thereof, wherein X1, X2, X3, X4, X5, X6, X7, R1, and Q1 are defined herein, inhibit the IGF-1R enzyme and are useful for the treatment and / or prevention of hyperproliferative diseases such as cancer, inflammation, psoriasis, allergy / asthma, disease and conditions of the immune system, disease and conditions of the central nervous system.
Owner:ACERTA PHARMA BV
Compositions and methods for modulating immune responses
ActiveUS20100151000A1Lower Level RequirementsDecreases functional activityPowder deliveryAntipyreticDiseaseAutoimmune responses
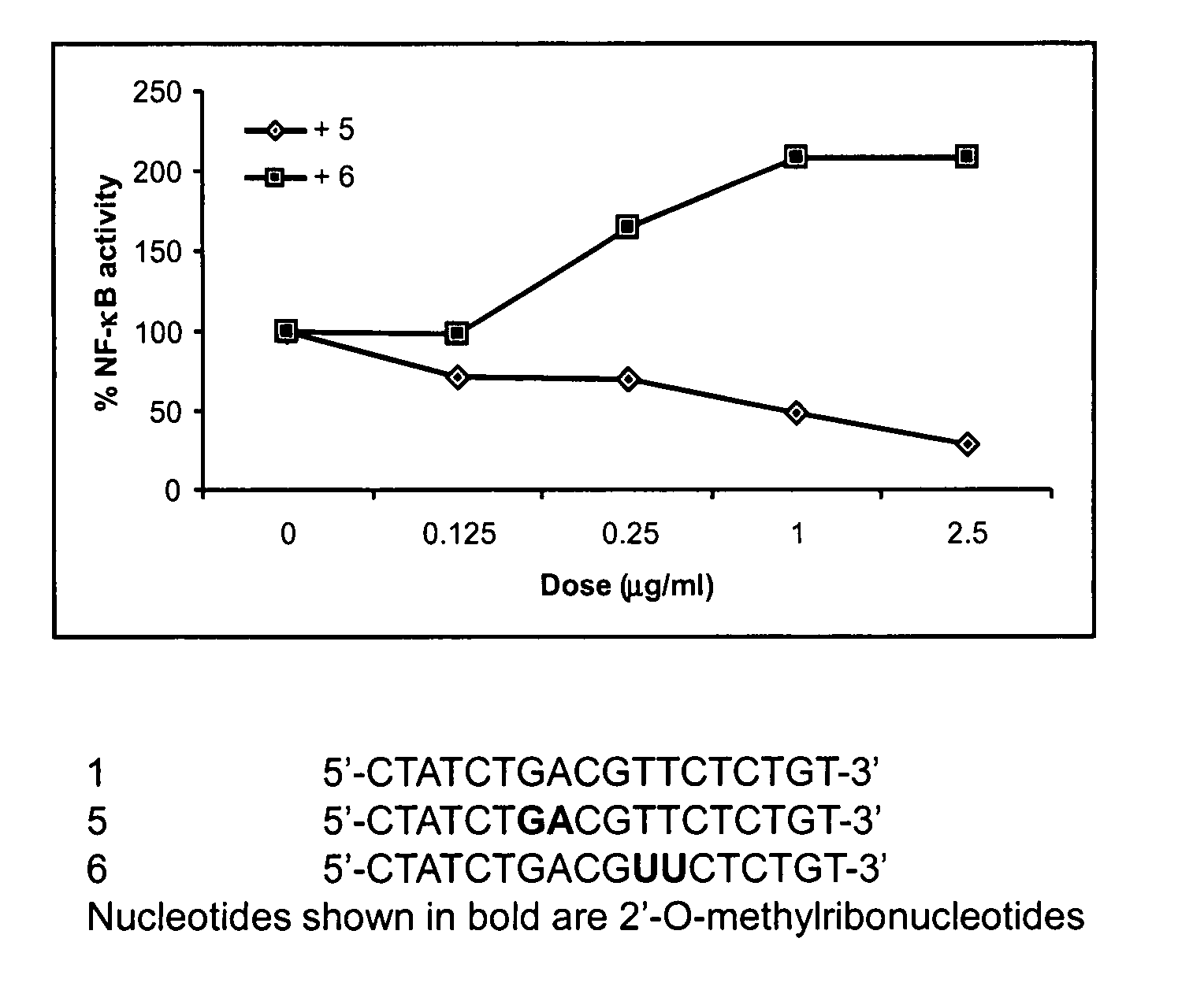
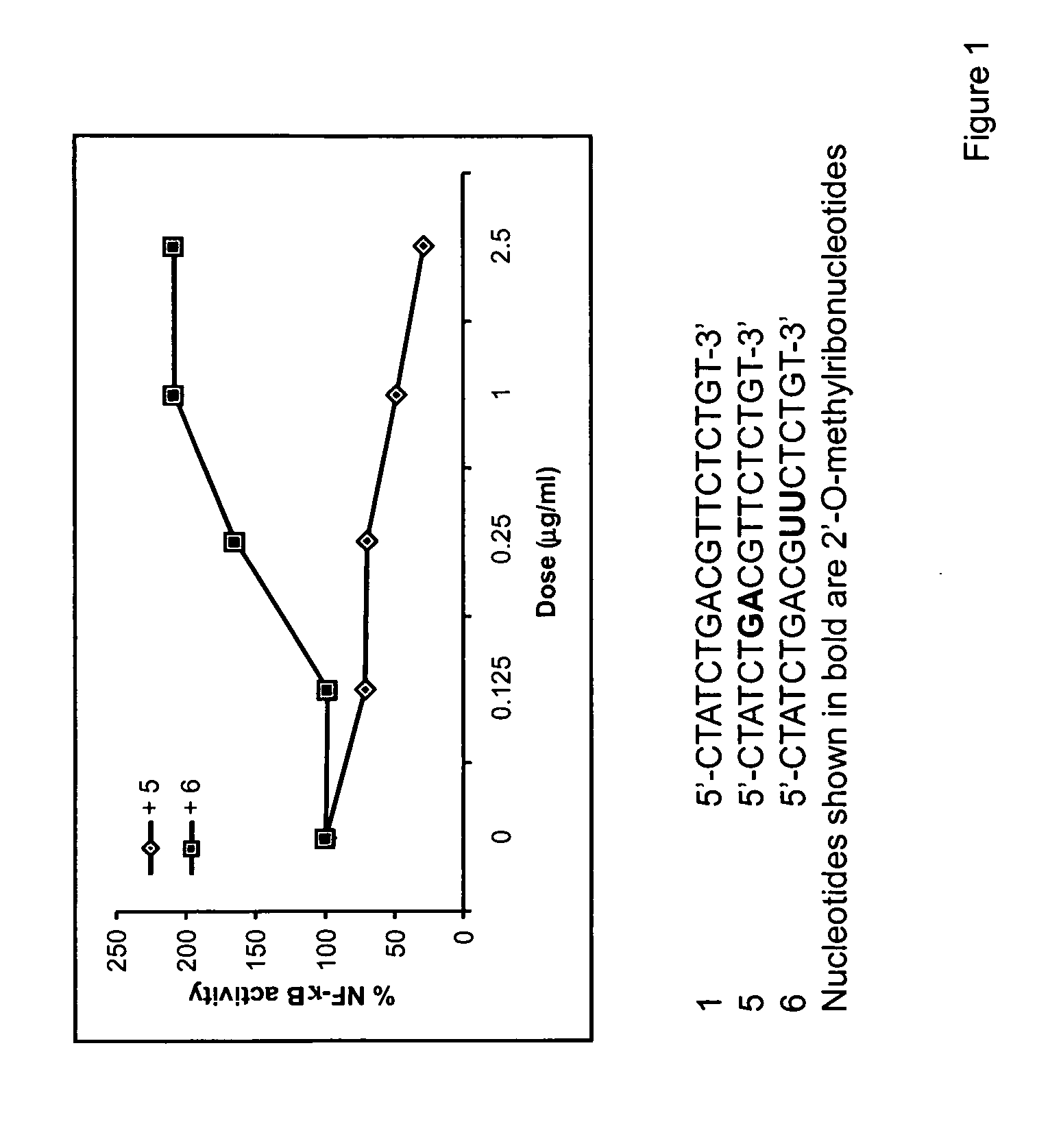
This invention discloses methods and compositions for modulating immune responses, which involve particulate delivery of agents to immune cells, wherein the agents comprise an inhibitor of the NF-κB signaling pathway and an antigen that corresponds to a target antigen. The methods and compositions of the present invention are particularly useful in the treatment or prophylaxis of an undesirable immune response associated with the target antigen, including autoimmune diseases, allergies and transplantation associated diseases.
Owner:THE UNIV OF QUEENSLAND
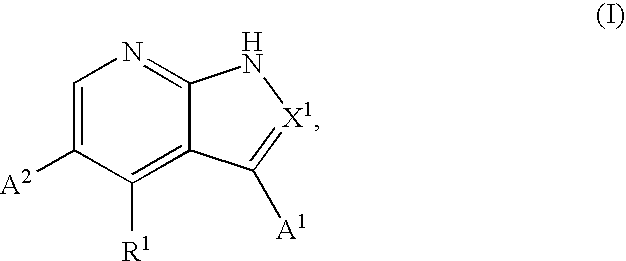
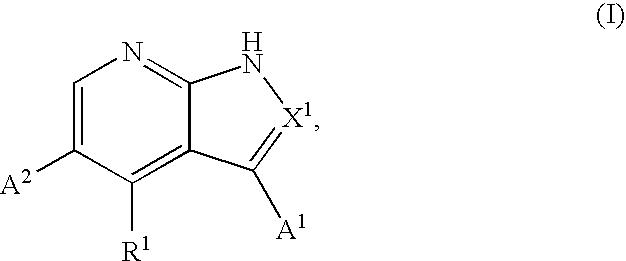
Substituted pyrrolopyridines and pyrazolopyridines as kinase modulators
Provided herein are substituted pyrrolopyridine heterocycles and substituted pyrazolopyridine heterocycles, pharmaceutical compositions comprising said heterocycles and methods of using said heterocycles in the treatment of disease. The heterocycles disclosed herein function as kinase modulators and have utility in the treatment of diseases such as cancer, allergy, asthma, inflammation, obstructive airway disease, autoimmune diseases, metabolic disease, infection, CNS disease, brain tumor, obesity, asthma, hematological disorder, degenerative neural disease, cardiovascular disease, or disease associated with angiogenesis, neovascularization, or vasculogenesis.
Owner:SGX PHARMA INC
Molecular antigen array
InactiveUS20060222652A1Improve efficiencyPeptide/protein ingredientsAntibody mimetics/scaffoldsAntigenAllergy
The invention provides compositions and processes for the production of ordered and repetitive antigen or antigenic determinant arrays. The compositions of the invention are useful for the production of vaccines for the prevention of infectious diseases, the treatment of allergies and the treatment of cancers. Various embodiments of the invention provide for a core particle that is coated with any desired antigen in a highly ordered and repetitive fashion as the result of specific interactions.
Owner:CYTOS BIOTECHNOLOGY AG
Cyclic-Dinucleotides and Its Conjugates as Adjuvants and Their Uses in Pharmaceutical Compositions
ActiveUS20080286296A1Severe formLess immunomodulatory effectOrganic active ingredientsAntipyreticDiseaseAutoimmune responses
The present invention relates to new adjuvants and the uses in pharmaceutical compositions, like in vaccines. In particular, the present invention provides new compounds useful as adjuvants and / or immunomodulators for prophylactic and / or therapeutic vaccination in the treatment of infectious diseases, inflammatory diseases, autoimmune diseases, tumors, allergies as well as for the control of fertility in human or animal populations. The compounds are particularly useful not only as systemic, but preferably as mucosal adjuvants. In addition, the invention relates to its uses as active ingredients in pharmaceutical compositions.
Owner:GESELLSCHAFT FUR BIOTECHNOLOGISCHE FORSCHUNG MBH GBF
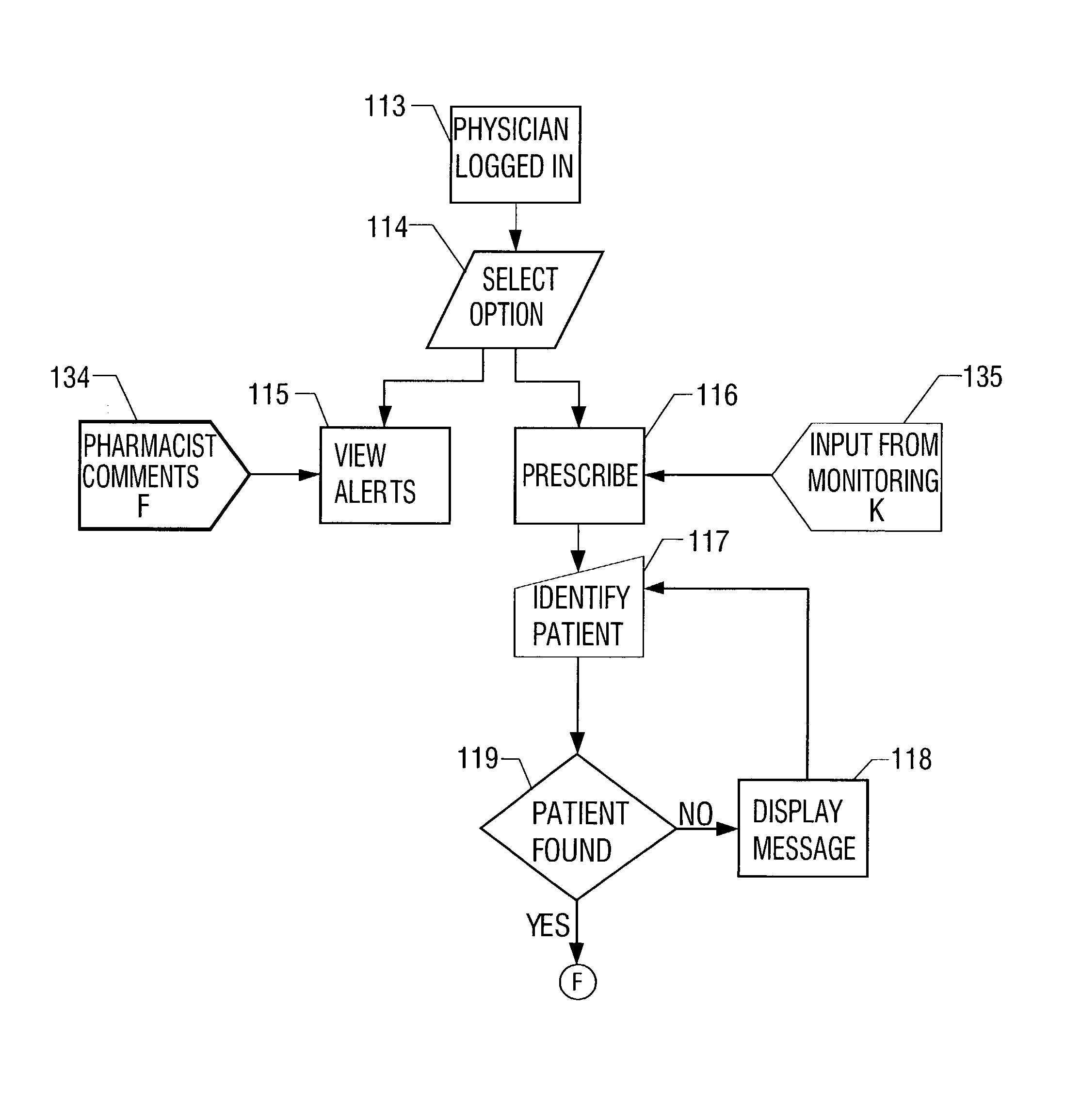
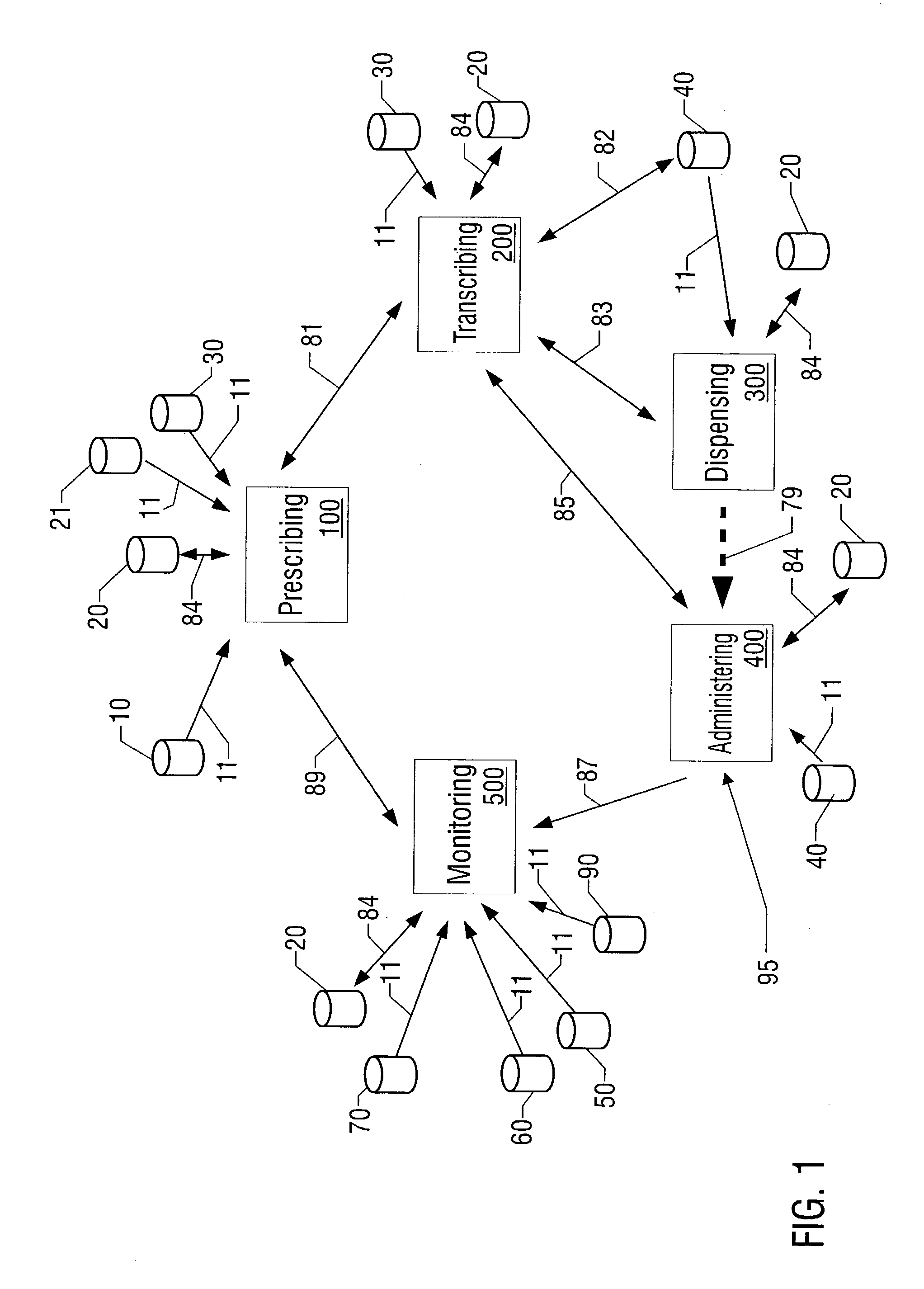
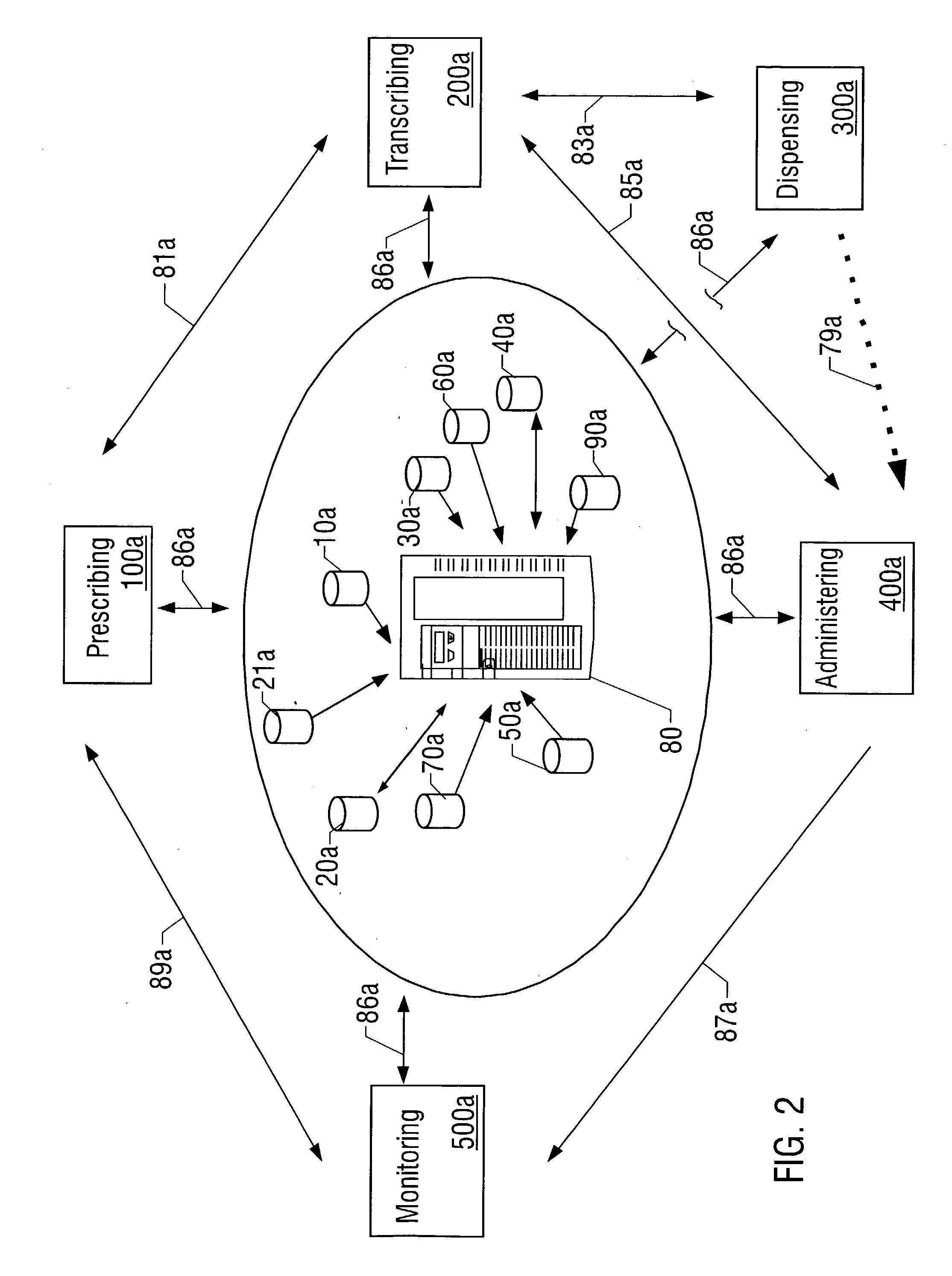
Closed loop medication use system and method
ActiveUS20030236683A1Data processing applicationsDrug and medicationsMedication informationGuideline
A closed loop medication use system and method includes selecting a medication to prescribe to a patient based on patient information, such as laboratory results, radiology results, and patient allergies, healthcare industry practices, patient-care site specific guidelines, and medication information. The selected medication is prescribed on an unverified prescription order that is then transcribed. Transcribing includes performing multiple crosschecks of the prescription order to real-time patient information, healthcare industry practices, and medication information to generate a verified prescription order. After transcribing, the appropriate dispensing method is determined for the prescription order and dispensed. The dispensed medication is administered after confirmation by the administering clinician of the right patient, right medication, right dosage, right route, and right time. The whole process of medication use described above is monitored continuously in real-time. The monitored information is communicated to the prescribing, transcribing, dispensing and administering parts of the system.
Owner:HARRIS DAWN HLDG INC +1
Mixture of at Least 6 Species of Lactic Acid Bacteria and/or Bifidobacteria in the Manufacture of Sourdough
A mixture of at least 6 species of lactic acid bacteria and / or Bifidobacteria is disclosed for use in bakery and medical field. The preferred mixture comprises Streptococcus thermophilus, Bifidobacterium infantis, Bifidobacterium longum, Bifidobacterium breve, Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus casei, Lactobacillus delbrueckii subsp. bulgaricus. Said mixture is useful for a sourdough, a leavening composition. Baked goods and other food products obtained therefrom are disclosed. These goods have low or no gluten content and are suitable for the integration of the diet of a subject suffering from celiac disease, for decreasing the risk of allergies due to wheat flour albumins and globulins, for the treatment of schizophrenic symptoms, in the preparation of products for enteric diet.
Owner:VSL PHARMA INC
Toxin peptide therapeutic agents
ActiveUS20070071764A1Avoid it happening againRelieve symptomsNervous disorderAntipyreticHalf-lifeSjögren syndrome
Disclosed is a composition of matter of the formula (X1)a—(F1)d—(X2)b—(F2)e—(X3)c (I) and multimers thereof, in which F1 and F2 are half-life extending moieties, and d and e are each independently 0 or 1, provided that at least one of d and e is 1; X1, X2, and X3 are each independently -(L)f-P-(L)g-, and f and g are each independently 0 or 1; P is a toxin peptide of no more than about 80 amino acid residues in length, comprising at least two intrapeptide disulfide bonds; L is an optional linker; and a, b, and c are each independently 0 or 1, provided that at least one of a, b and c is 1. Linkage to the half-life extending moiety or moieties increases the in vivo half-life of the toxin peptide, which otherwise would be quickly degraded. A pharmaceutical composition comprises the composition and a pharmaceutically acceptable carrier. Also disclosed are a DNA encoding the inventive composition of matter, an expression vector comprising the DNA, and a host cell comprising the expression vector. Methods of treating an autoimmune disorder, such as, but not limited to, multiple sclerosis, type 1 diabetes, psoriasis, inflammatory bowel disease, contact-mediated dermatitis, rheumatoid arthritis, psoriatic arthritis, asthma, allergy, restinosis, systemic sclerosis, fibrosis, scleroderma, glomerulonephritis, Sjogren syndrome, inflammatory bone resorption, transplant rejection, graft-versus-host disease, and lupus and of preventing or mitigating a relapse of a symptom of multiple sclerosis are also disclosed.
Owner:AMGEN INC
Benzimidazole derivatives and their use as KDR kinase protein inhibitors
The invention discloses and claims benzimidazole compounds of formula (I): wherein X is C—R2; Y is C—R2 or C—R3; W and Z are each C—R3; R1 is an optionally substituted aryl, heteroaryl or a saturated 5- or 6-membered monocyclic heterocyclic radical or a bicyclic heterocyclic radical; and A5 is H or alkyl; or a stereoisomer, a racemate, an enantiomer or a diastereoisomer of said compound of formula (I) or a pharmaceutically acceptable salt thereof; the use of compounds of formula (I) for the treatment of a disorder of proliferation of blood vessels, uncontrolled angiogenesis, a fibrotic disorder, a disorder of proliferation of mesangial cells, a metabolic disorder, allergy, asthma, thrombosis, a disease of the nervous system, retinopathy, psoriasis, rheumatoid arthritis, diabetes, muscle degeneration, solid tumors and cancers, pharmaceutical compositions comprising a compound of formula (I) and one or more pharmaceutically acceptable adjuvants or diluents and pharmaceutical compositions comprising a compound of formula (I) and one or more. antimitiotic agents.
Owner:AVENTIS PHARMA SA (US)
Semi-soft C-class immunostimulatory oligonucleotides
The invention relates to specific C-Class semi-soft CpG immunostimulatory oligonucleotides that are useful for stimulating an immune response. In particular the oligonucleotides are useful for treating allergy, such as allergic rhinitis and asthma, cancer and infectious disease, such as hepatitis B and hepatitis C.
Owner:COLEY PHARMA GRP INC +1
Compositions & formulations with an epiandrosterone or a ubiquinone & kits & their use for treatment of asthma symptoms & for reducing adenosine/adenosine receptor levels
A composition and various formulations comprise preventative or therapeutic amounts of an epiandrosterone, analogue thereof or salt thereof, and / or a ubiquinone or salt thereof, and a pharmaceutically or veterinarily acceptable carrier or diluent. The composition and formulations are useful for treating bronchoconstriction, respiratory tract inflammation and allergies, asthma, and cancer. A method of treating diseases associated with low adenosine levels or adenosine depletion comprises administering folinic acid or a pharmaceutically acceptable salt hereof in a preventative or therapeutic amount, or an amount effective to treat adenosine depletion.
Owner:EAST CAROLINA UNIVERISTY
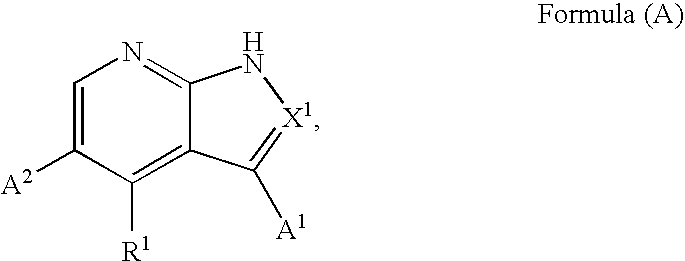
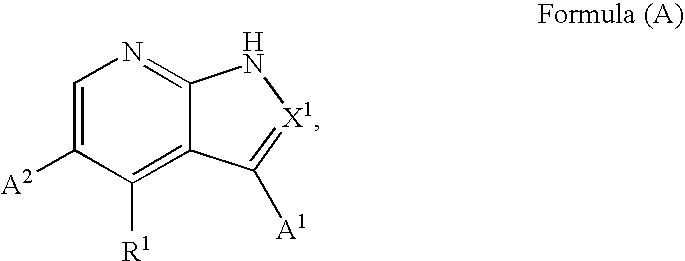
Substituted pyrrolopyridines and pyrazolopyridines as kinase modulators
Provided herein are substituted pyrrolopyridine heterocycles and substituted pyrazolopyridine heterocycles, pharmaceutical compositions comprising said heterocycles and methods of using said heterocycles in the treatment of disease. The heterocycles disclosed herein function as kinase modulators and have utility in the treatment of diseases such as cancer, allergy, asthma, inflammation, obstructive airway disease, autoimmune diseases, metabolic disease, infection, CNS disease, brain tumor, obesity, asthma, hematological disorder, degenerative neural disease, cardiovascular disease, or disease associated with angiogenesis, neovascularization, or vasculogenesis.
Owner:SGX PHARMA INC
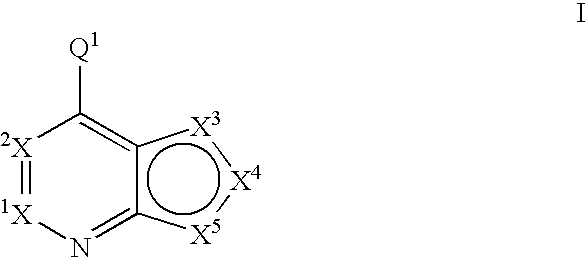
Fused heterobicyclic kinase inhibitors
InactiveUS20070208053A1Low toxicityDisrupting virus life-cycleAntibacterial agentsBiocideNervous systemAllergy
Compounds of the formula and pharmaceutically acceptable salts thereof, wherein X1, X2, X3, X4, X5, X5, X7, R1, and Q1 are defined herein, inhibit kinase enzymes and are useful for the treatment and / or prevention of hyperproliferative diseases such as cancer. The compounds are also useful in the treatment of inflammation, allergy, asthma, disease and conditions of the immune system, disease and conditions of the nervous system, cardiovascular diseases, disease and conditions of the eye, dermatological diseases, osteoporosis, diabetes, multiple sclerosis, and infections.
Owner:OSI PHARMA INC
Immunostimulatory nucleic acids for the treatment of asthma and allergy
InactiveUS20060154890A1Avoid allergiesPrevent asthmaBiocideElcosanoid active ingredientsTime scheduleAllergy
The invention involves administration of an immunostimulatory nucleic acid alone or in combination with an asthma / allergy medicament for the treatment or prevention of asthma and allergy in subjects. The combination of drugs are administered in synergistic amounts or in various dosages or at various time schedules. The invention also relates to kits and compositions concerning the combination of drugs.
Owner:COLEY PHARM GRP INC
Use of fumaric acid derivatives for treating cardiac insufficiency, and asthma
According to a first aspect the invention relates to the use of fumaric acid derivatives selected from the group consisting of dialkyl fumarates, monoalkyl hydrogen fumarates, fumaric acid monoalkyl ester salts, fumaric acid monoamides, monoamido fumaric acid salts, fumaric acid diamides, monoalkyl monoamido fumarates, carbocyclic and oxacarbocyclic oligomers of these compounds and mixtures thereof for preparing a drug for the treatment or prevention of cardiac insufficiency, in particular left ventricular insufficiency, myocardial infarction and angina pectoris. According to a second aspect the invention relates to the use of fumaric acid derivatives, selected from the group consisting of dialkyl fumarates, monoalkyl hydrogen fumarates, fumaric acid monoalkyl ester salts, fumaric acid monoamides, monoamido fumaric acid salts, fumaric acid diamides, monoalkyl monoamido fumarates, carbocyclic and oxacarbocyclic oligomers of these compounds and mixtures thereof for preparing a drug for the treatment of asthma and chronic obstructive pulmonary diseases, especially asthma caused by allergies, infections, analgesics, job conditions or physical effort, mixed forms of asthma, or asthma cardiale.
Owner:FUMAPHAM AG
Small molecule toll-like receptor (TLR) antagonists
The invention provides methods and compositions useful for modulating signaling through Toll-like receptors. The methods involve contacting a TLR-expressing cell with a small molecule having a core structure including at least two rings. Certain of the compounds are 4-primary amino quinolines. Many of the compounds and methods are useful specifically for inhibiting immune stimulation involving at least one of TLR9, TLR8, TLR7, and TLR3. The methods may have use in the treatment of autoimmunity, inflammation, allergy, asthma, graft rejection, graft versus host disease, infection, sepsis, cancer, and immunodeficiency.
Owner:COLEY PHARMA GMBH +1
Immunoregulator
InactiveUS20020064501A1Prevent rejectionImmunological interactionMicrobiological testing/measurementTetrapeptide ingredientsAutoimmune responsesAllergy
The invention relates to the field of immunology. Specifically, the invention relates to the field of immune-mediated disorders such as allergies, auto-immune disease, transplantation-related disease or inflammatory disease. The invention provides for an immunoregulator (IR), use of an IR in preparing a pharmaceutical composition for treating an immune-mediated disorder and a method for treating an immune-mediated disorder.
Owner:BIOTEMPT
Compositions and methods for characterizing and restoring gastrointestinal, skin, and nasal microbiota
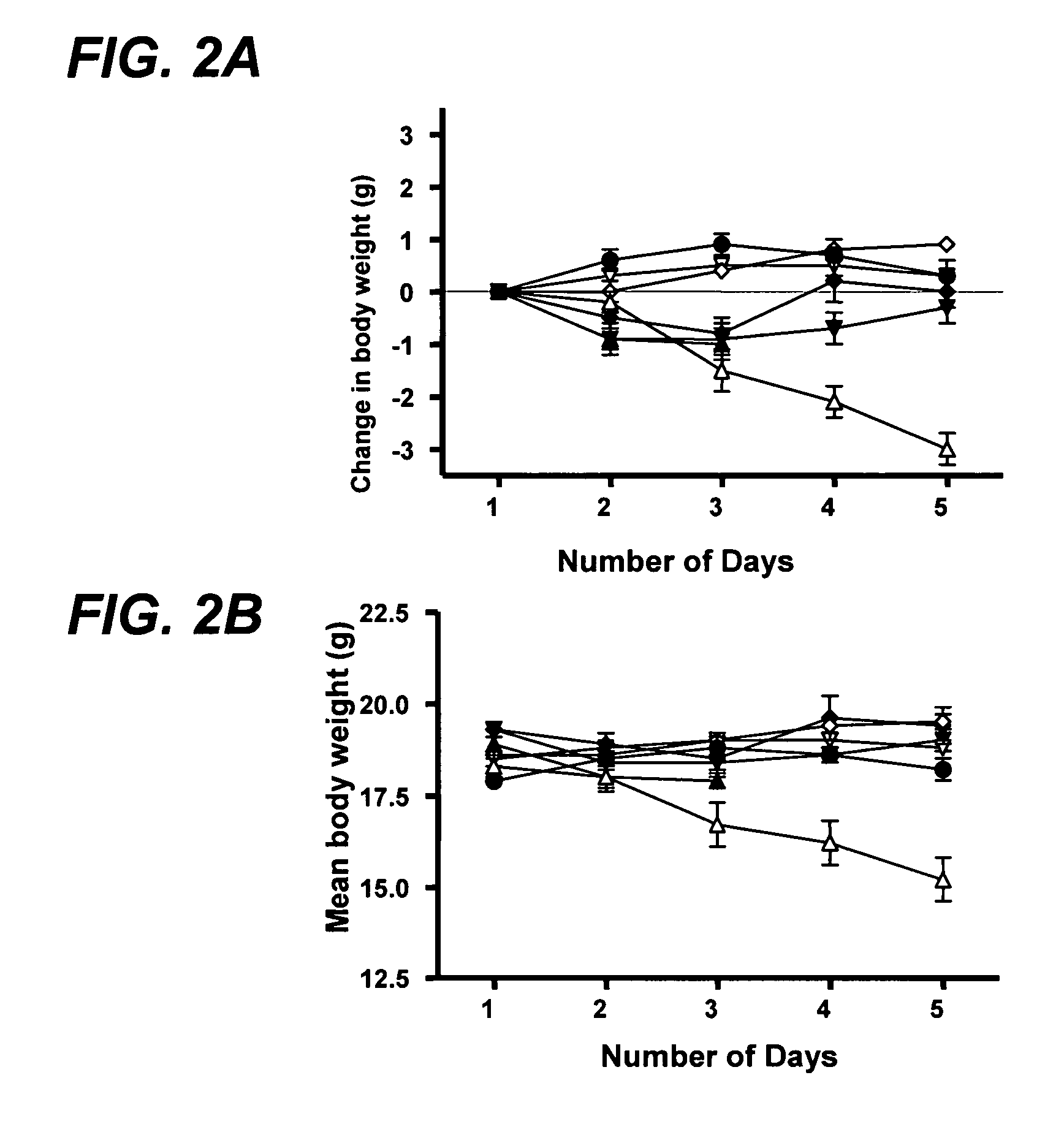
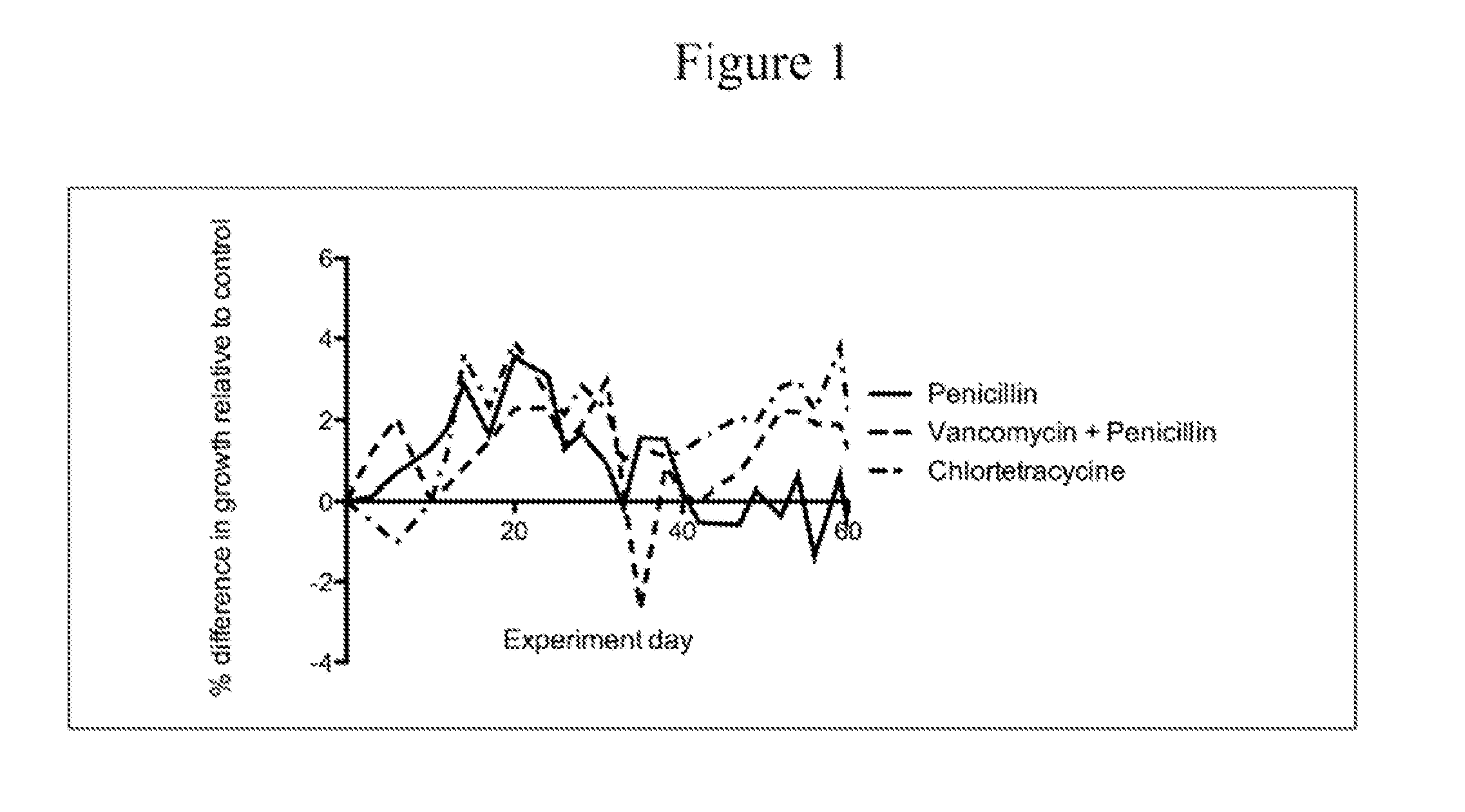
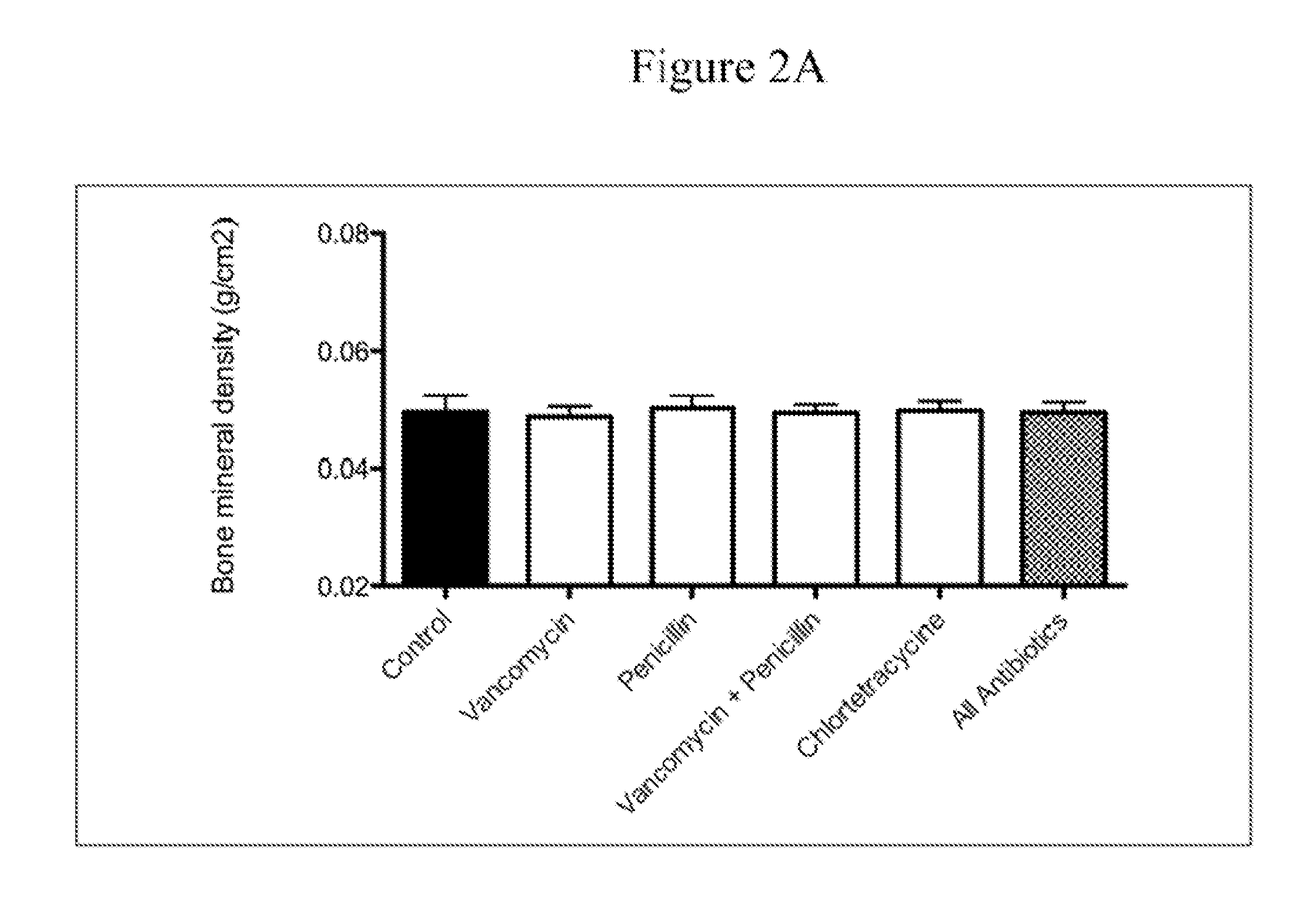
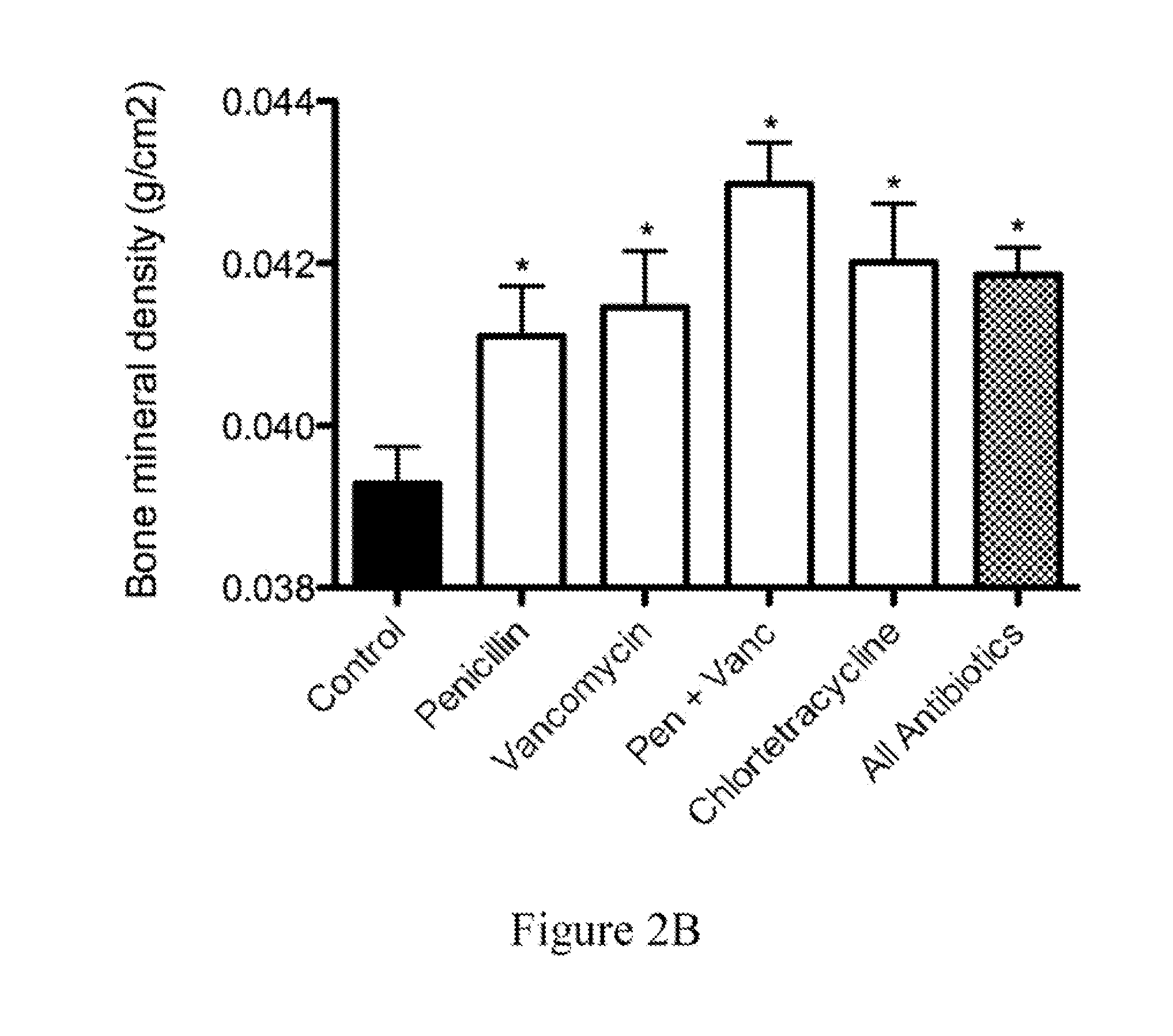
ActiveUS20100074872A1Growth inhibitionFacilitate calorie uptakeBiocideMetabolism disorderBacteroidesDisease
The present invention relates to characterizing changes in mammalian bacterial gastrointestinal, cutaneous and nasal microbiota associated with antibiotic treatment and various disease conditions (such as asthma, allergy, obesity, metabolic syndrome, gastrointestinal reflux disease (GERD), eosinophilic esophagitis, gastro-esophageal junction adenocarcinomas (GEJAC), infections due to bacteria that are resistant to antibiotics, including Methicillin-resistant Staphylococcus aureus (MRSA), Clostridium difficile, vancomycin-resistant enterococci, etc.) and related diagnostic and therapeutic methods. Therapeutic methods of the invention involve the use of live bacterial inoculants that are capable of restoring healthy mammalian bacterial gastrointestinal, skin, and nasal microbiota.
Owner:NEW YORK UNIV
Immune regulatory oligonucleotide (IRO) compounds to modulate toll-like receptor based immune response
ActiveUS20090060898A1Avoid immune responseInhibitory activityOrganic active ingredientsAntipyreticTlr agonistsAutoimmune disease
The invention provides novel immune regulatory oligonucleotides (IRO) as antagonist of TLRs and methods of use thereof. These IROs have unique sequences that inhibit or suppress TLR-mediated signaling in response to a TLR ligand or TLR agonist. The methods may have use in the prevention and treatment of cancer, an autoimmune disorder, airway inflammation, inflammatory disorders, infectious disease, skin disorders, allergy, asthma or a disease caused by a pathogen.
Owner:IDERA PHARMA INC
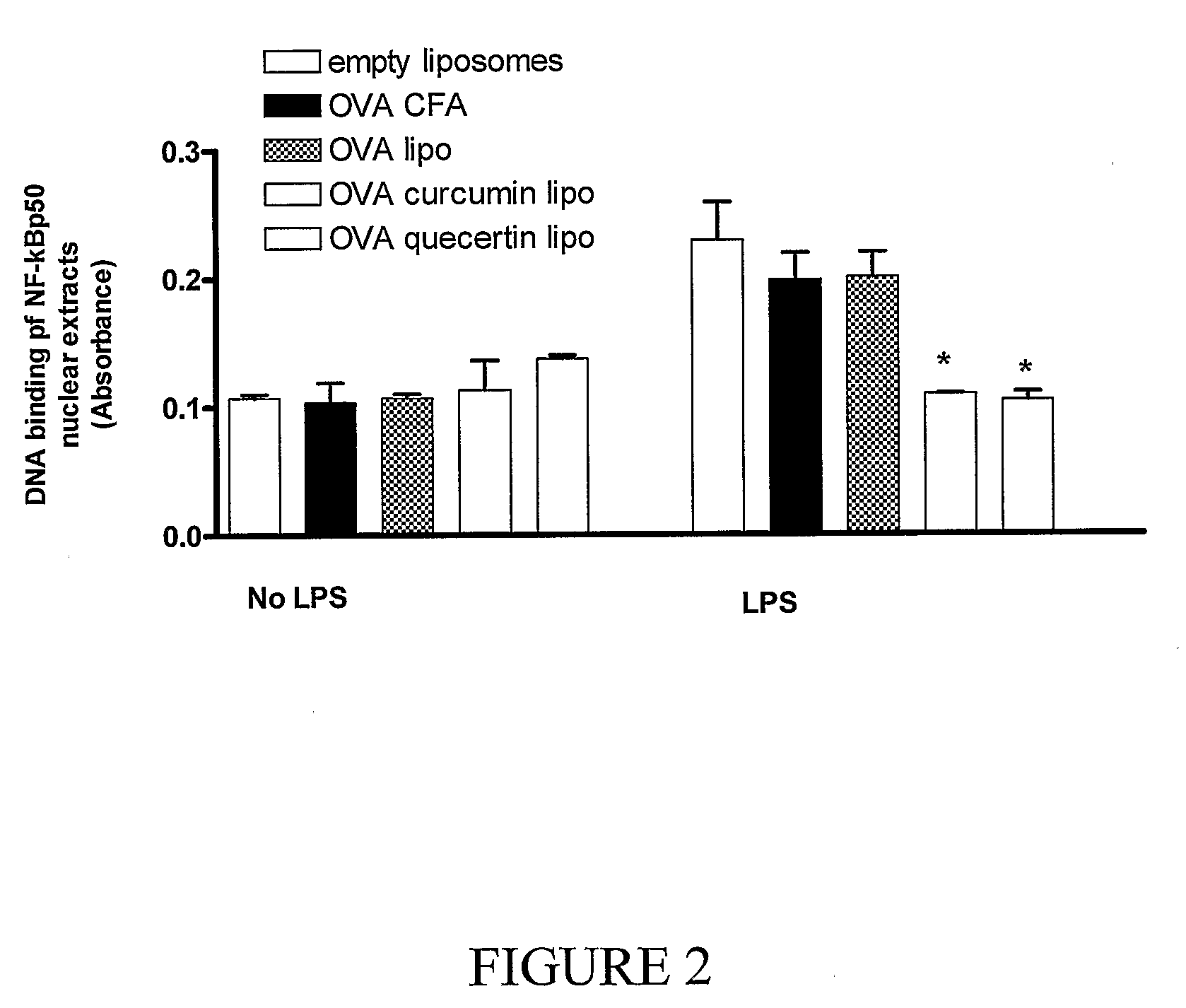
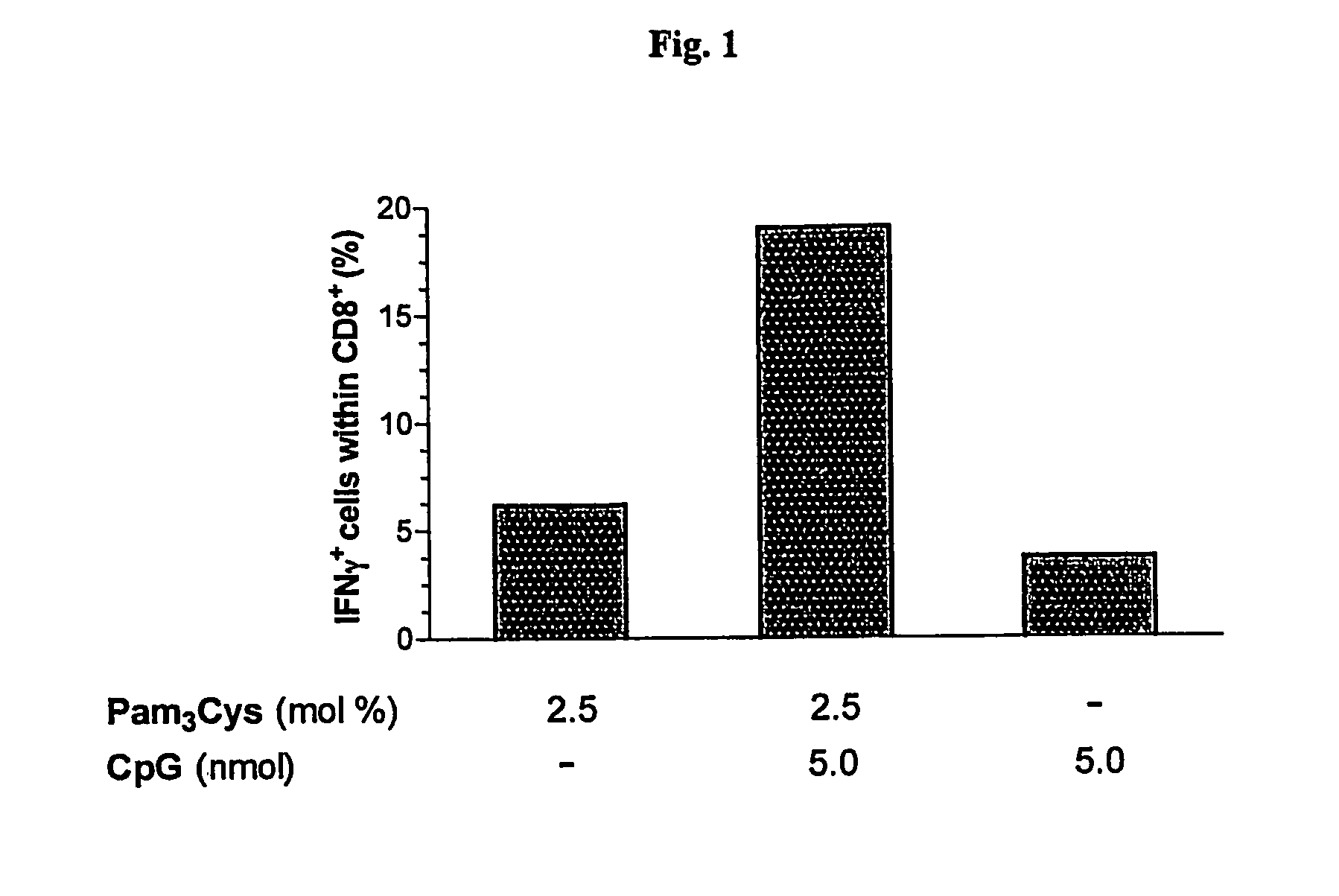
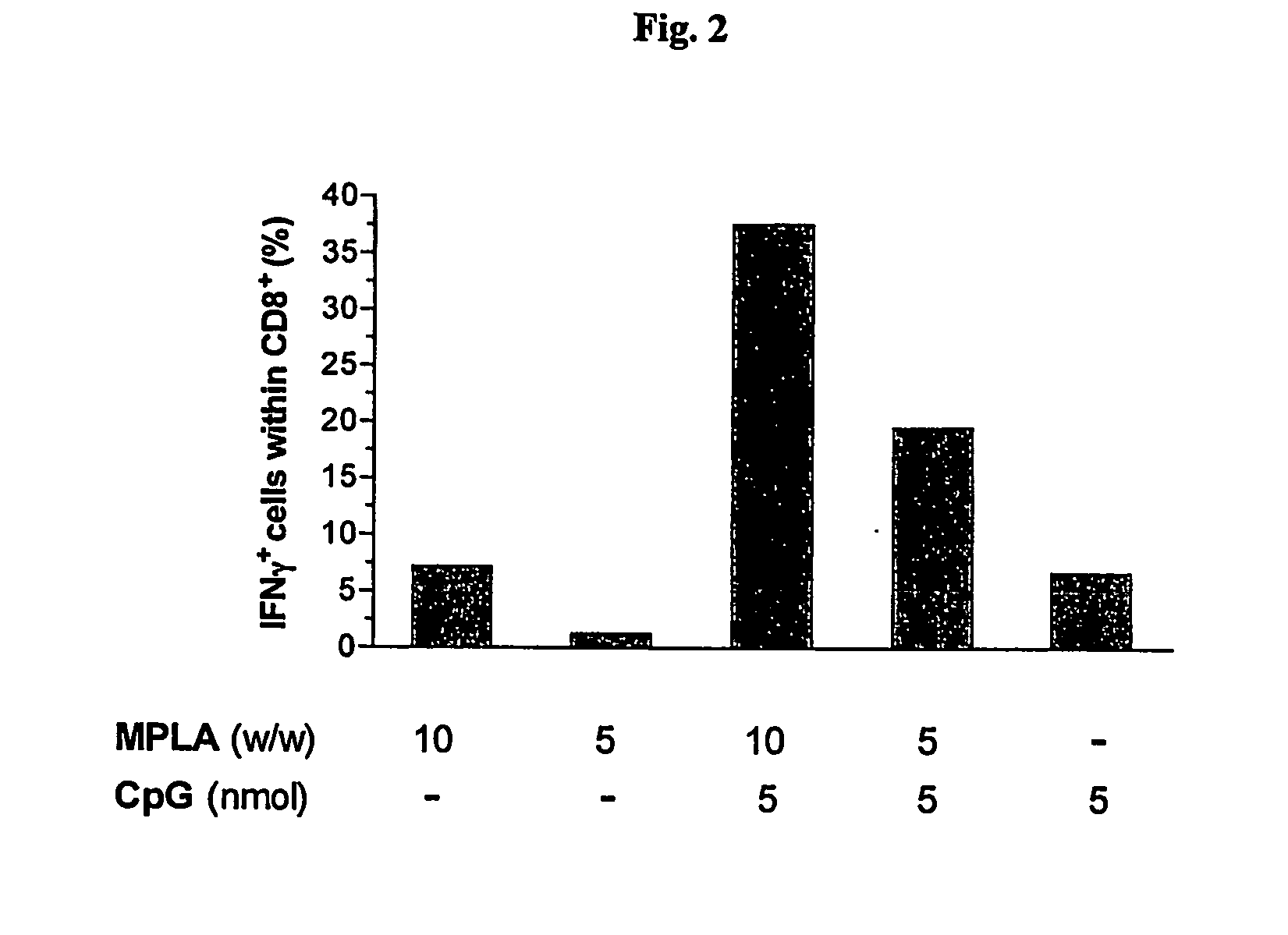
Synergistic Liposomal Adjuvants
InactiveUS20070298093A1Improve actionEnhance immune responseAntibacterial agentsAntimycoticsVascular diseaseAdjuvant
The present invention relates to liposome, mixtures or liposomes and liposomal compositions comprising at least two different adjuvants and a therapeutic agent, their production and use for the prevention and therapy of proliferative diseases, infectious diseases, vascular diseases, rheumatoid diseases, inflammatory diseases, immune diseases, in particular autoimmune diseases and allergies.
Owner:PHARMEXA
Structure and use of 5' phosphate oligonucleotides
Oligonucleotides bearing free, uncapped 5′ phosphate group(s) are recognized by RIG-I, leading to the induction of type I IFN, IL-18 and IL-1β production. Bacterial RNA also induces type I IFN production. 5′ phosphate oligonucleotides and bacterial RNA can be used for inducing an anti-viral response or an anti-bacterial response, in particular, type I IFN and / or IL-18 and / or IL-1β production, in vitro and in vivo and for treating various disorders and diseases such as viral infections, bacterial infections, parasitic infections, tumors, allergies, autoimmune diseases, immunodeficiencies and immunosuppression. Single-stranded 5′ triphosphate RNA can be used for inducing an anti-viral response, an anti-bacterial response, or an anti-tumor response, in particular, type I IFN and / or IL-18 and / or IL-1β production, in a target cell-specific manner.
Owner:UNIVERSITY OF BONN