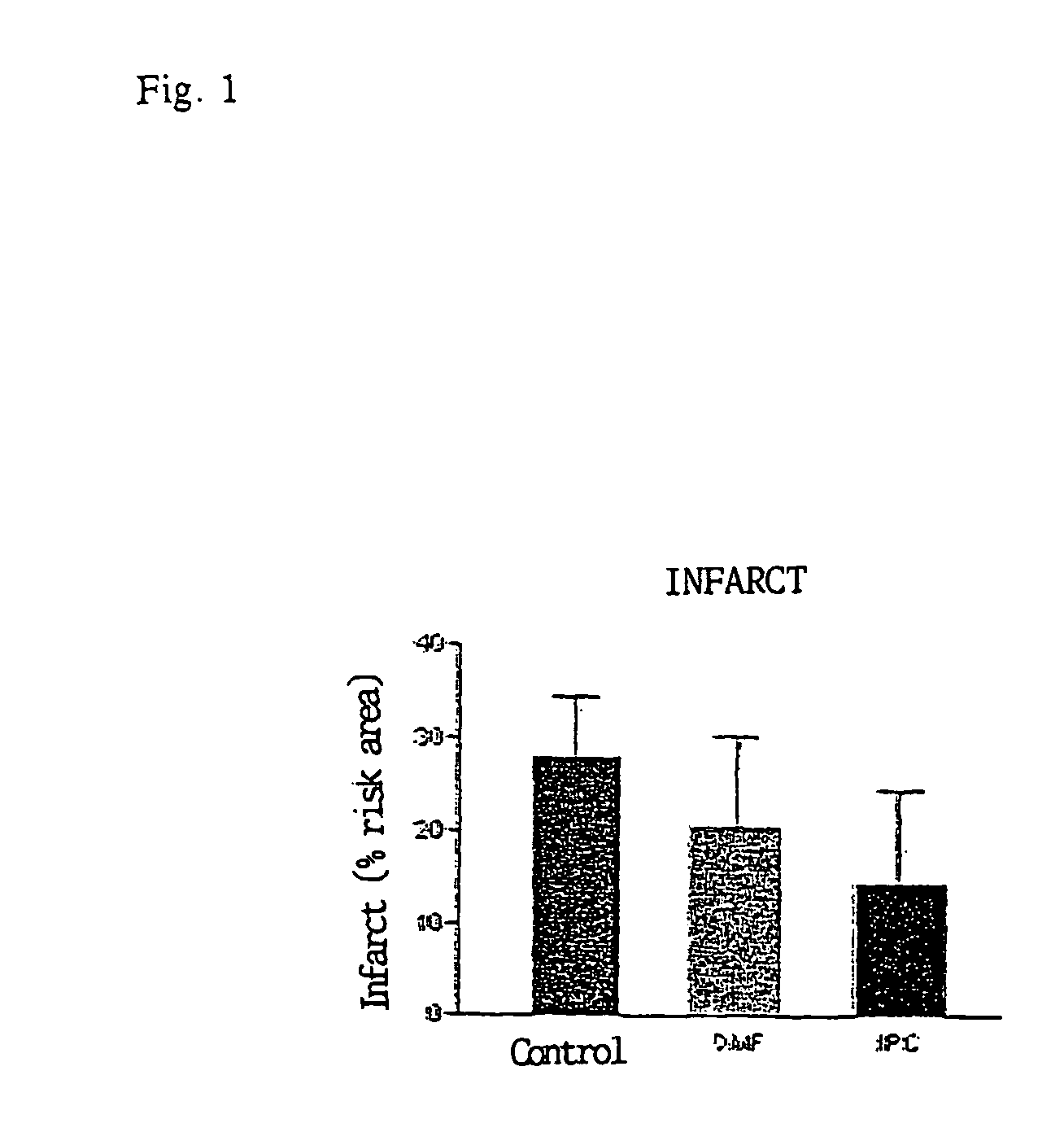
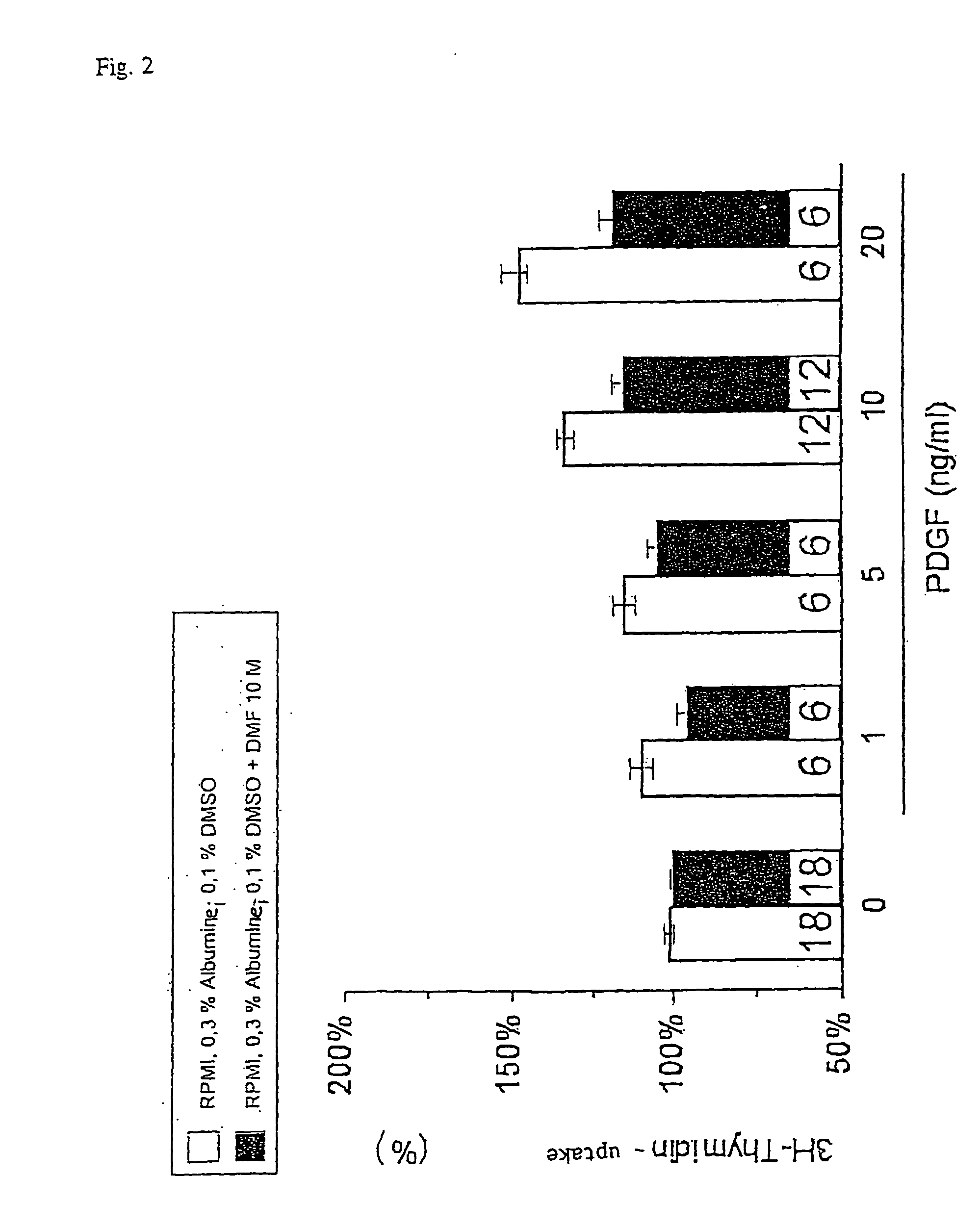
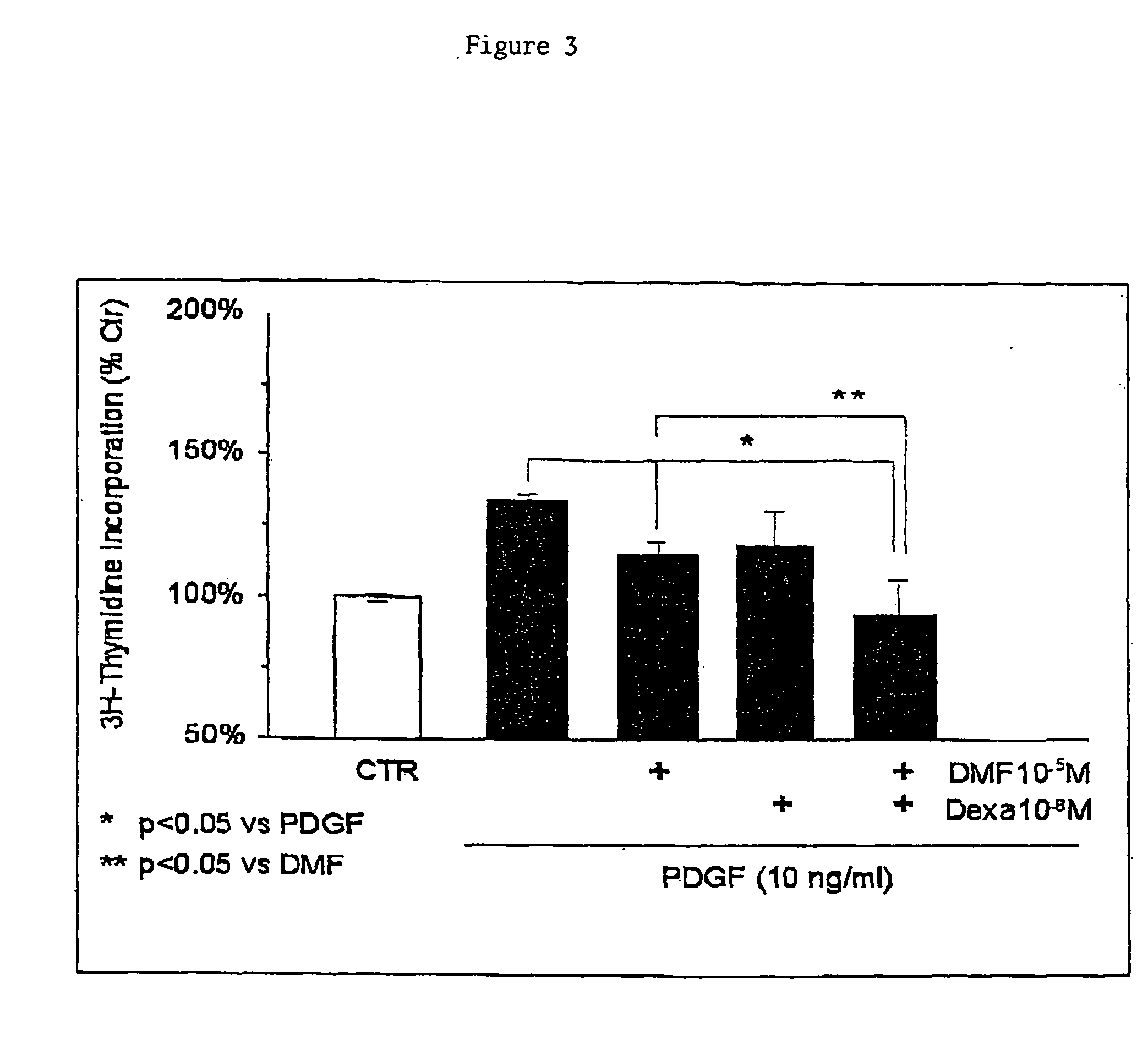
Use of fumaric acid derivatives for treating cardiac insufficiency, and asthma
a technology of fumaric acid and derivatives, which is applied in the direction of cardiovascular disorders, drug compositions, peptide/protein ingredients, etc., can solve the problems of respiratory distress
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples of preparation
[0062] To explain the use according to the invention, various examples for the preparation of preferred pharmaceutical preparations are given below. The examples are for illustrations purposes only, but not to restrict the invention.
example 1
Preparation of Film Tablets with an Enteric Coating Containing 100.0 mg of Monomethyl Fumarate-Ca Salt, which Corresponds to 78 mg of Fumaric Acid
[0063] Taking the necessary precautions (breathing mask, gloves, protective clothing, etc.), 10 kg of monomethyl fumarate-Ca salt are crushed, mixed intensely and homogenized by means of a sieve 800. Then an excipient mixture of the following composition is prepared: 21 kg of starch derivative (STA-RX 1500®), 2 kg of micro-crystalline cellulose (Avicel PH 101®), 0.6 kg of polyvinyl pyrrolidone (PVP, Kollidon® 25), 4 kg of Primogel®, 0.3 kg of colloidal silicic acid (Aerosil®).
[0064] The active ingredient is added to the entire powder mixture, mixed, homogenized by means of a sieve 200 and processed with a 2% aqueous solution of polyvinyl pyrrolidone (PVP, Kollidon® 25) in the usual manner into binder granules, and then mixed with the outer phase in a dry state. The latter consists of 2 kg of a so-called FST complex containing 80% of tal...
example 2
Preparation of Enteric Coated Capsules Containing 86.5 mg of Monoethyl Fumarate-Ca Salt and 110.0 mg of Dimethyl Fumarate, which Corresponds to a Total of 150 mg of Fumaric Acid
[0068] Taking the necessary precautions (breathing mask, gloves, protective clothing, etc.), 8.65 kg of monoethyl fumarate-Ca salt and 11 kg of dimethyl fumarate are intensely mixed with a mixture consisting of 15 kg of starch, 6 kg of lactose Ph. Helv. VII, 2 kg of micro-crystalline cellulose (Avicel®), 1 kg of polyvinyl pyrrolidone (Kollidon® 25) and 4 kg of Primogel® and homogenized by means of a sieve 800.
[0069] Together with a 2% aqueous solution of polyvinyl pyrrolidone (Kollidon® 25) the entire powder mixture is processed in the usual manner into a binder granulate and mixed with the outer phase in the dried state. Said outer phase consists of 0.35 kg of colloidal silicic acid (Aerosil®), 0.5 kg of magnesium stearate and 1.5 kg of talcum Ph. Helv. VII. The homogeneous mixture is then filled in porti...
PUM
| Property | Measurement | Unit |
|---|---|---|
| mean diameter | aaaaa | aaaaa |
| mean diameter | aaaaa | aaaaa |
| mean diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More