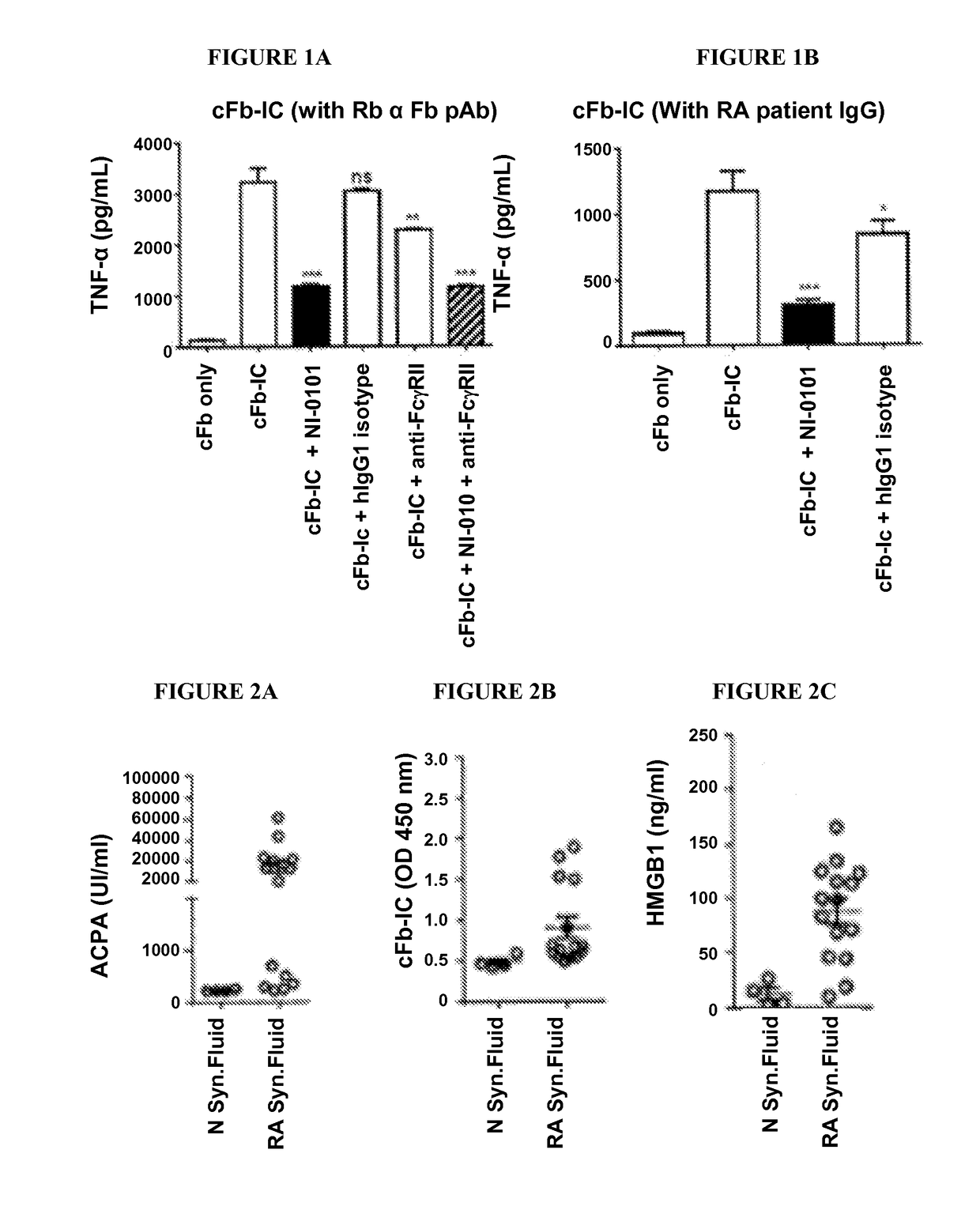
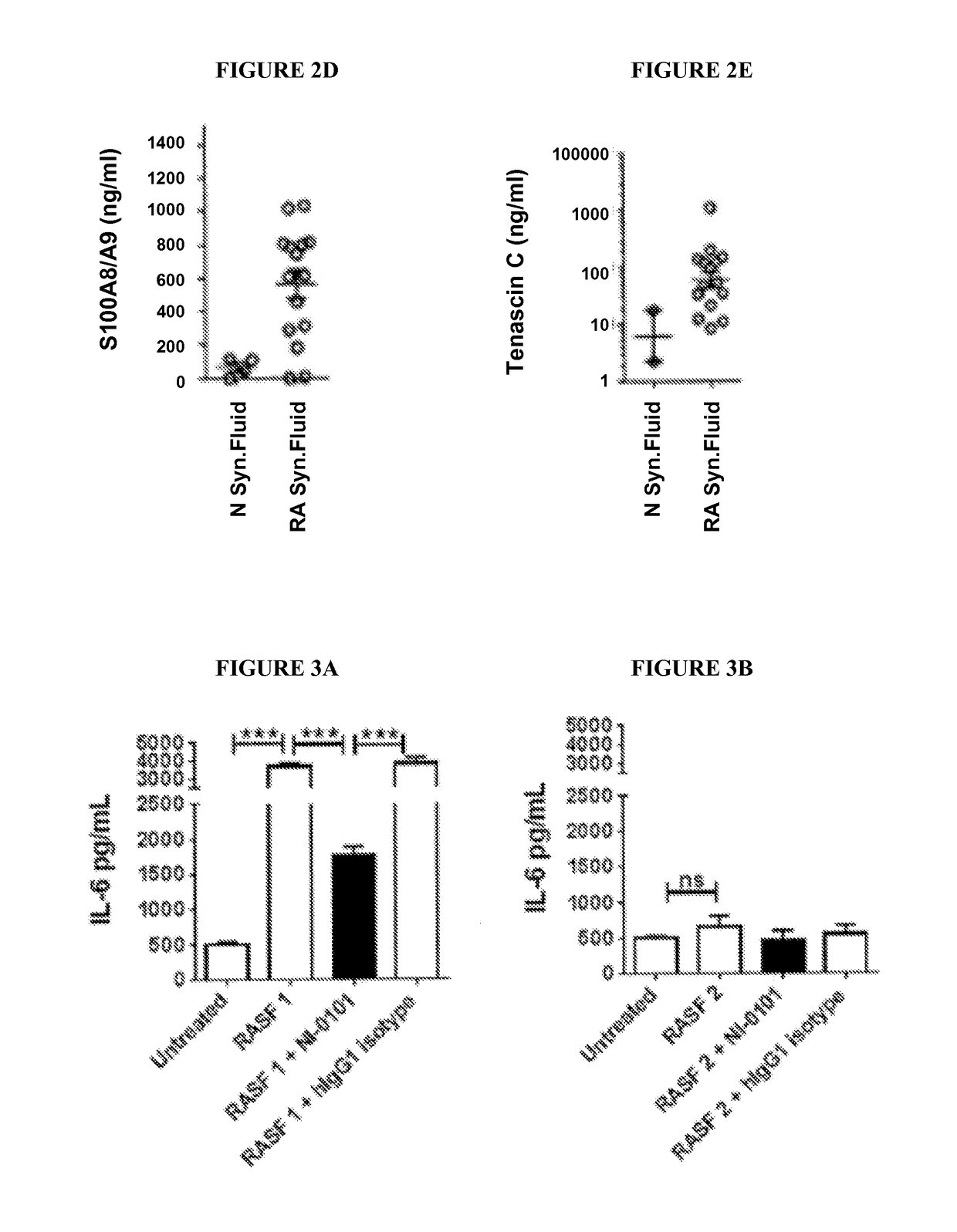
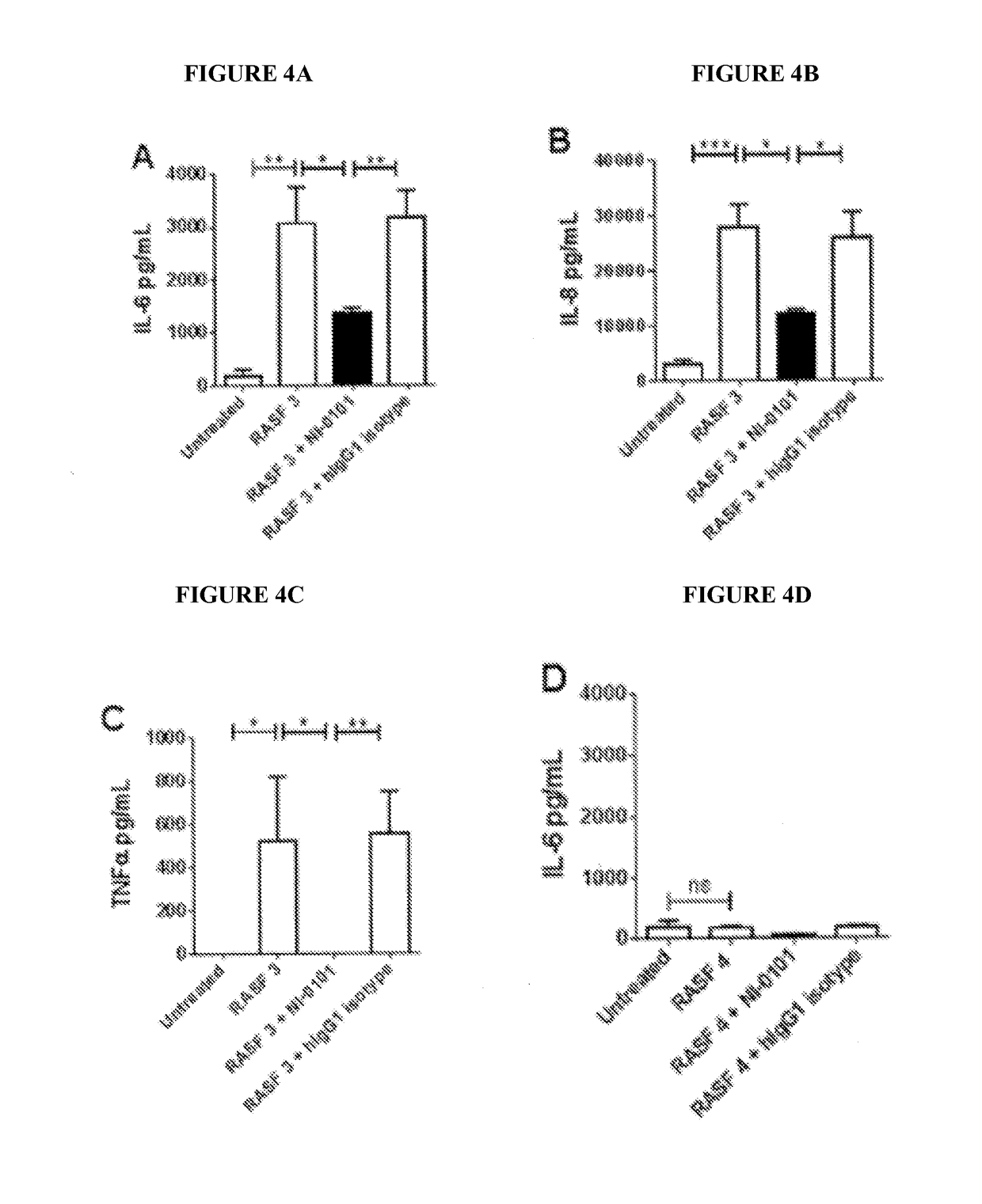
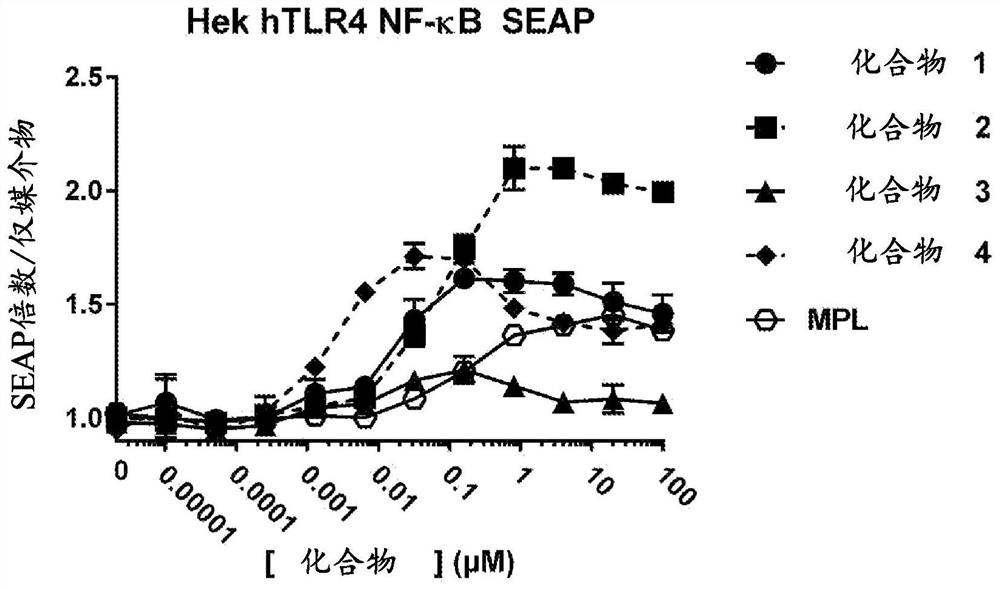
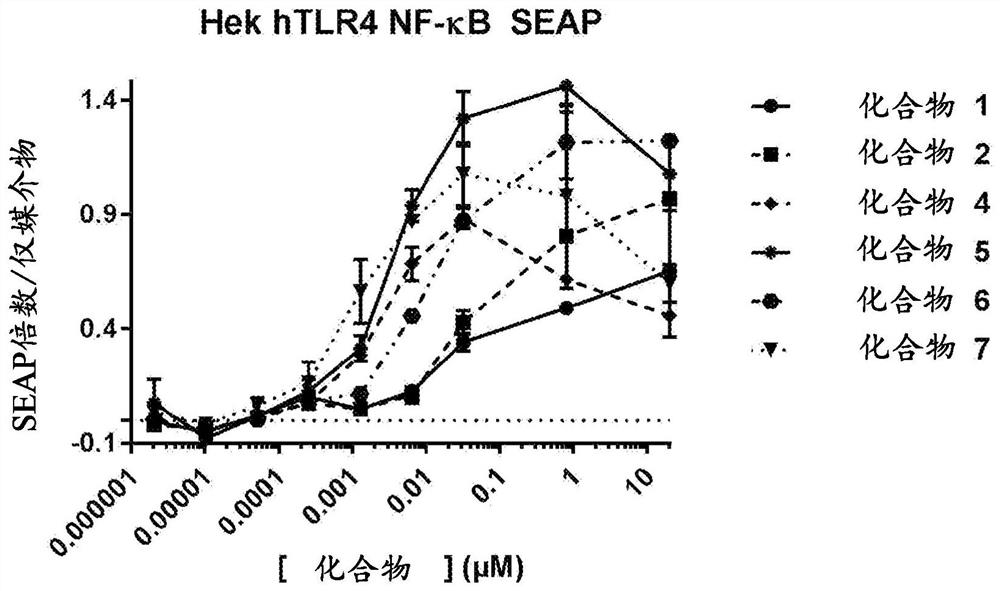
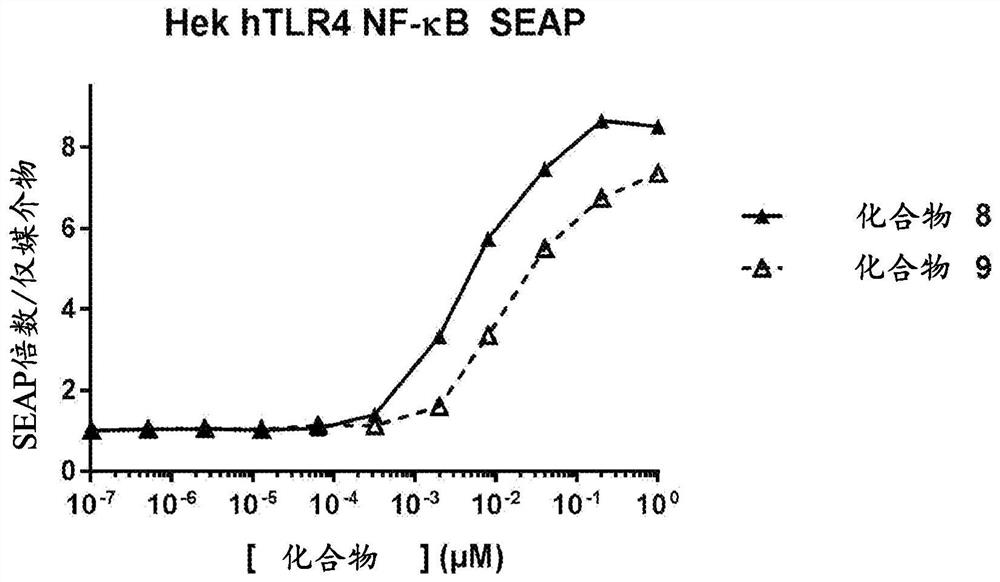
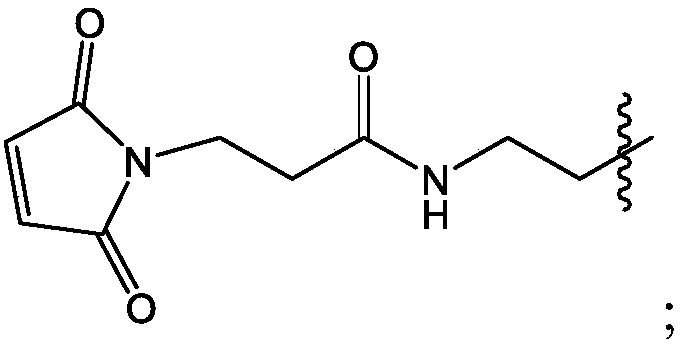
Patents
Literature
75 results about "Toll" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
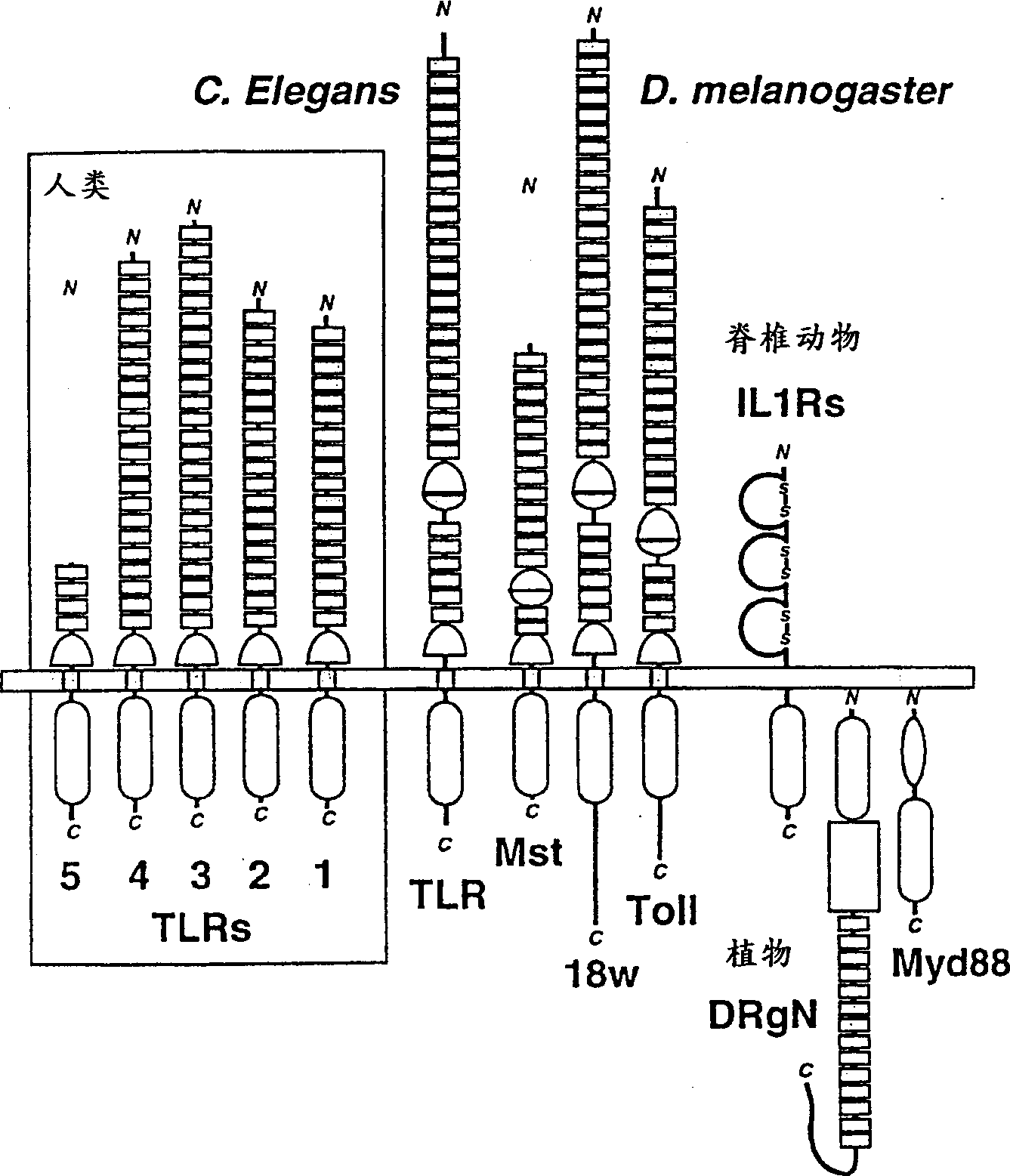
The toll genes encode members of the toll-like receptor class of proteins. Mutants in the toll gene were originally identified by 1995 Nobel Laureates Christiane Nüsslein-Volhard and Eric Wieschaus and colleagues in the fruit fly Drosophila melanogaster in 1985, and cloned by the laboratory of Kathryn Anderson in 1988. Since then, thirteen mammalian toll genes have been identified. In flies, toll was first identified as a gene important in embryogenesis in establishing the dorsal-ventral axis. In 1996, toll was found to have a role in the fly's immunity to fungal infections. Both mammalian and invertebrate toll genes are required for innate immunity. Toll-like receptors in mammals were identified in 1997 at Yale University by Ruslan Medzhitov and Charles Janeway. Concurrently, two separate studies, led by Shizuo Akira, Bruce A. Beutler and their respective colleagues discovered that the Toll-like receptors act as the principal sensors of infection in mammals. The name of the gene family derives from Christiane Nüsslein-Volhard's 1985 exclamation, "Das ist ja toll!" The exclamation, which translates as "That's amazing!"
8-Substituted Benzoazepines as Toll-Like Receptor Modulators
Provided are compositions and methods useful for modulation of signaling through the Toll-like receptors TLR7 and / or TLR8. The compositions and methods have use in the treatment of autoimmunity, inflammation allergy, asthma, graft rejection, graft versus host disease, infection, sepsis, cancer and immunodeficiency.
Owner:ARRAY BIOPHARMA
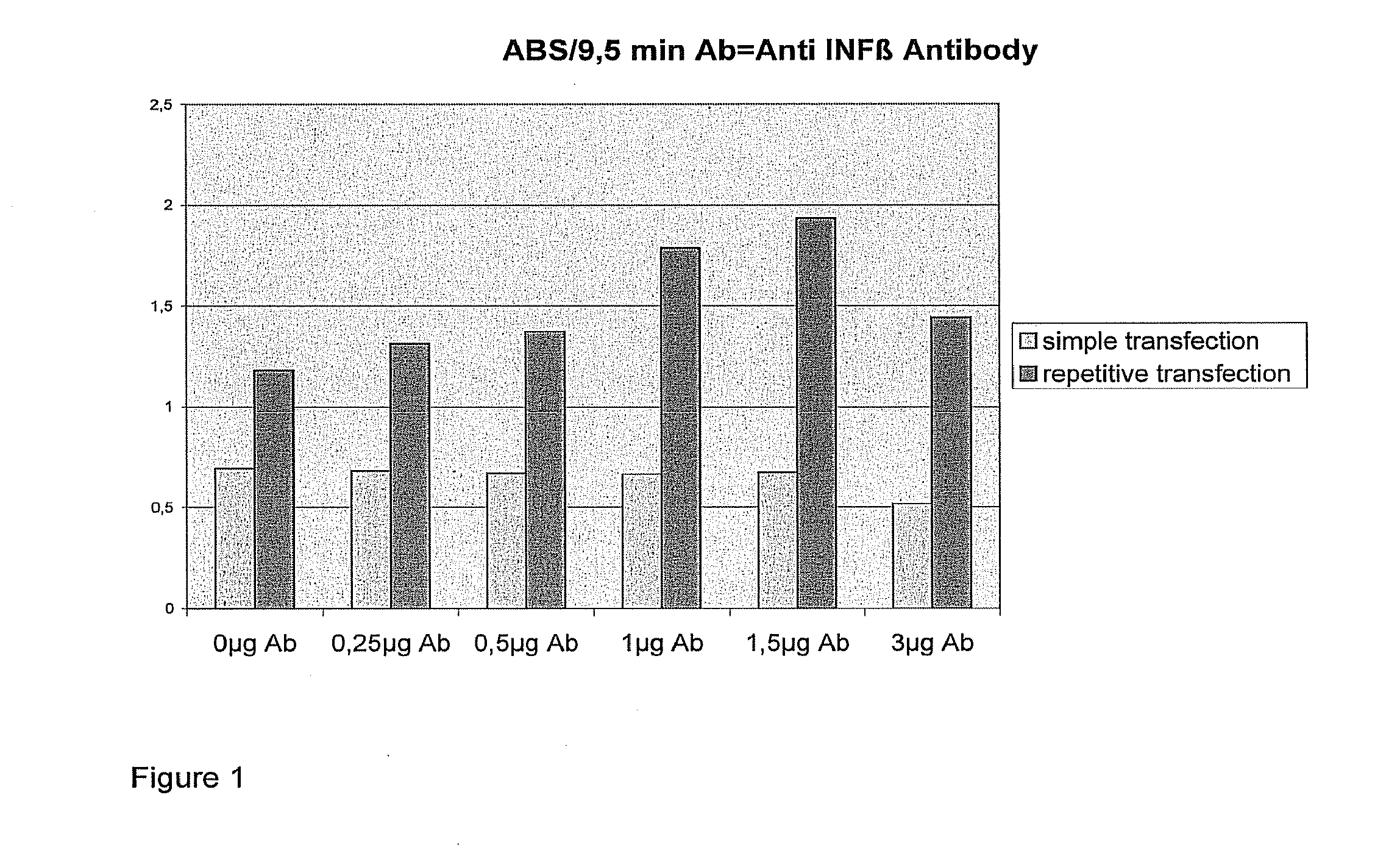
Transfection results of non-viral gene delivery systems by influencing of the innate immune system
The innate immune system of eukaryotes is able to recognise foreign genetic material by means of Toll-like receptors and to initiate signal transduction cascades that trigger an antiviral state of cell populations by way of an interferon response. That antiviral state is also a barrier for non-viral gene delivery systems. If the signal transduction cascade is interrupted intracellularly or intercellularly, transfection efficiencies of non-viral gene delivery systems can be increased and undesirable changes in the expression profile can be avoided. Since RNA-interference is to be attributed to the antiviral state, the RNAi machinery is likewise activated after activation of the innate immune system. In that way, knock-down efficiencies on transfection with siRNA can be increased.
Owner:BIONTEX LAB
Immunoglobulin and/or Toll-Like Receptor Proteins Associated with Myelogenous Haematological Proliferative Disorders and Uses Thereof
InactiveUS20110044894A1Reduced expression levelAddress bad outcomesIn-vivo radioactive preparationsMicrobiological testing/measurementReceptorMyelogenous
The disclosure relates to methods and compositions effective in the diagnosis, prognosis and treatment of human hematopoietic cancers. In particular, the disclosure provides tumor-associated genes that encode for members of the immunoglobulin (Ig) and / or toll-like receptor superfamilies that are differentially expressed in hematopoietic tumor cells of myeloid origin compared with other cells, e.g., normal stem cells.
Owner:CELLERANT THERAPEUTICS INC
Lyophilised antigen composition
InactiveUS20090035360A1Cost benefitSuitable for useOrganic active ingredientsAntibody mimetics/scaffoldsVaccinationMedicine
The present invention provides lyophilised compositions comprising an antigen and a Toll-like receptor (TLR) 9 agonist. Such compositions may be reconstituted into immunogenic compositions for use in vaccination with a carrier selected from the group of particulate carriers consisting of liposomes, mineral salts, emulsions, polymers and ISCOMs. Methods of making immunogenic compositions from the lyophilised compositions of the invention and use of the same in immunisation are also herein provided.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
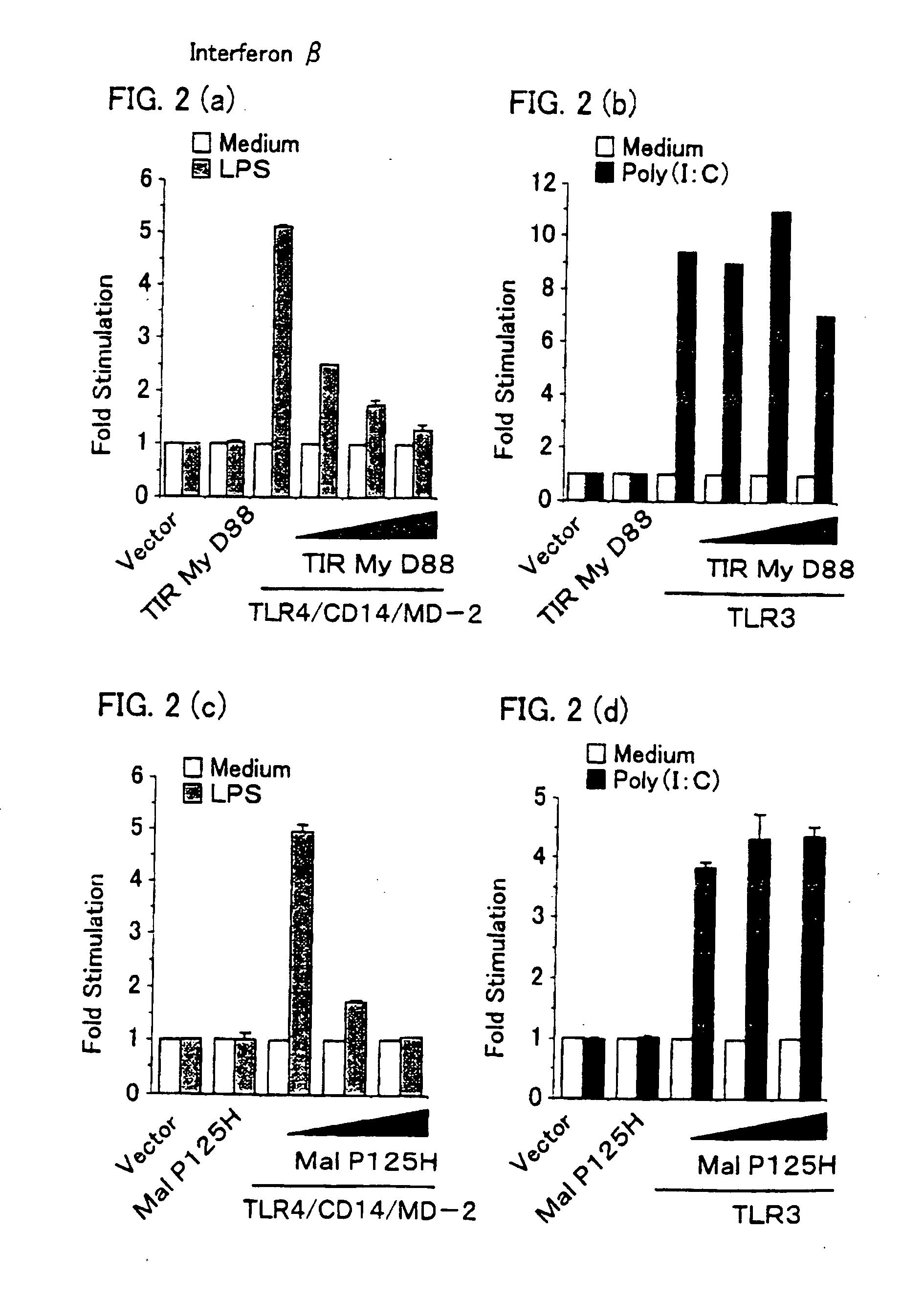
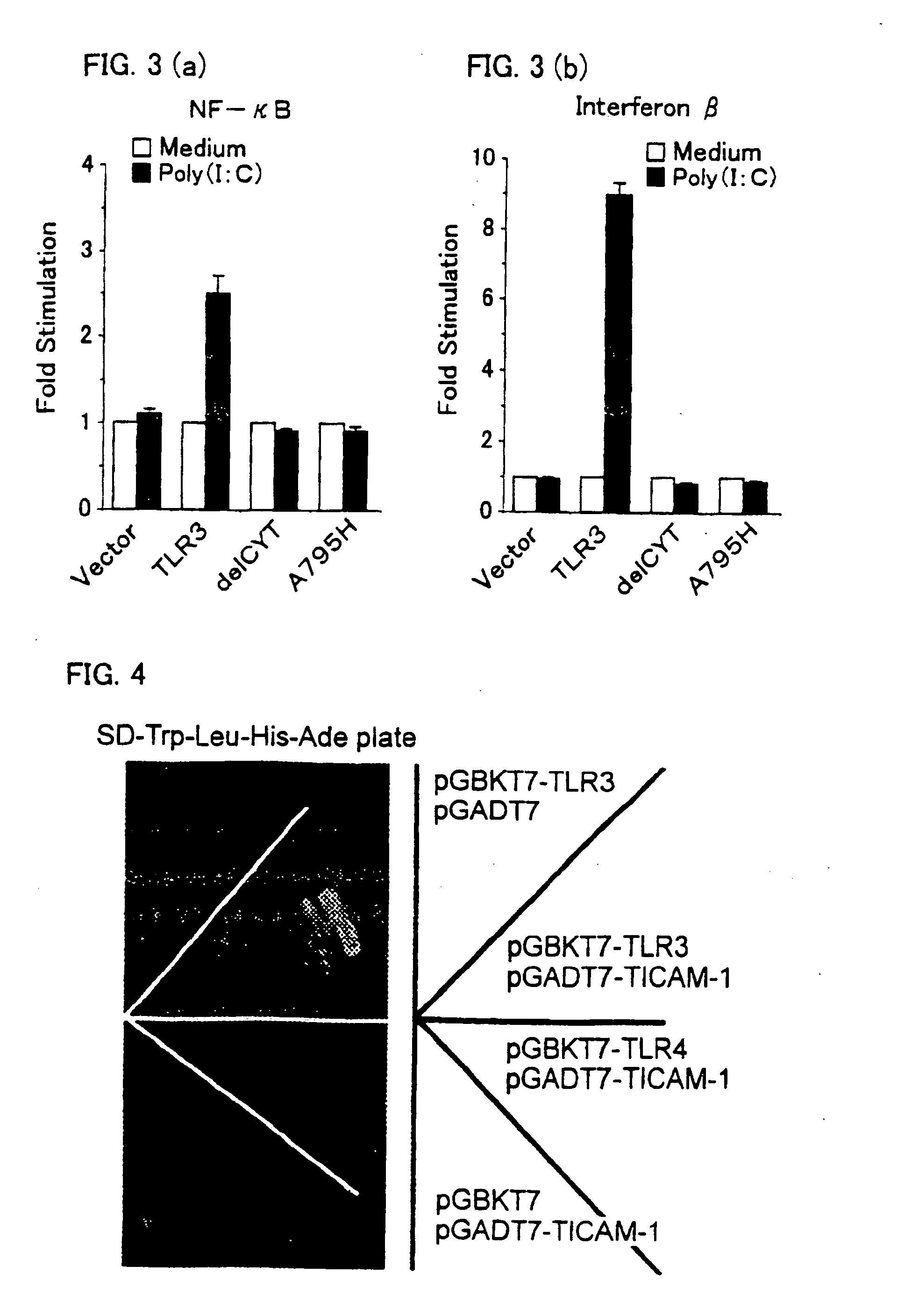
Novel adaptor protein binding to mammalian toll-like receptor 3, and gene thereof
InactiveUS20060134128A1Enhance mammalian type I interferon productionInhibit productionPeptide/protein ingredientsAntiviralsViral infectious diseaseMutant
A novel adaptor protein and its gene are provided. The novel adaptor protein has a property of binding to mammalian Toll-like receptor 3, which controls type I interferon production that is effective for prevention / treatment of viral infectious disease such as hepatitis B, hepatitis C, and the like, treatment of tumors, and the other purposes. Novel adaptor protein TICAM-1, which has an amino acid sequence set forth in SEQ ID NO: 2 or NO: 4, has the property of specifically binding to the mammalian Toll-like receptor 3 and a property of inducing type I interferon production. A mutant of the adaptor protein TICAM- 1 has similar properties, provided that it has TIR domain (an amino acid sequence ranging from 394-position to 532-position in the amino acid sequence set forth in SEQ ID NO: 2 or an amino acid sequence ranging 396-position to 534-position in the amino acid sequence set forth in SEQ ID NO: 4. The gene is a gene encoding the adaptor protein TICAM-1.
Owner:JAPAN SCI & TECH CORP
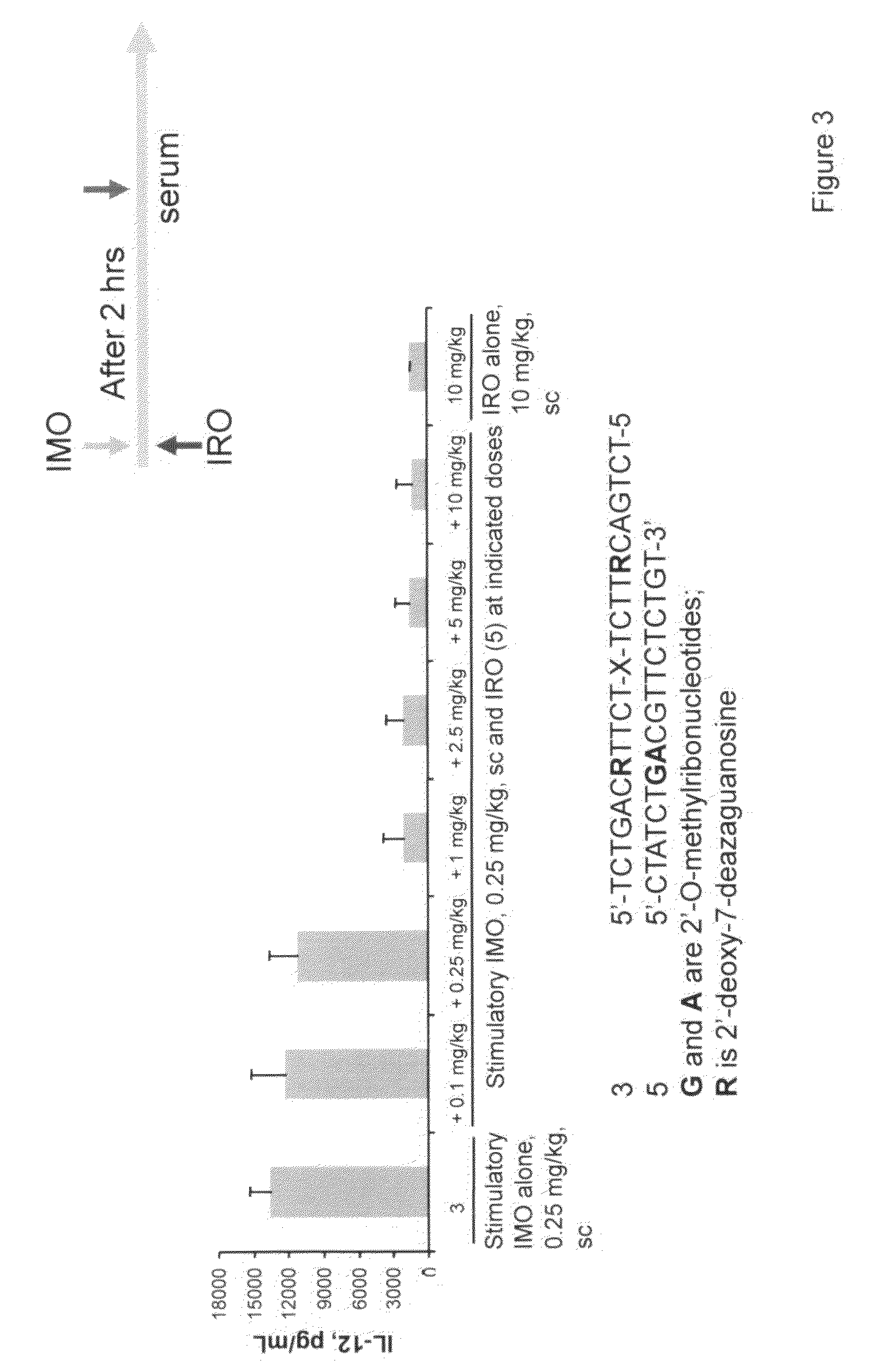
Immune regulatory oligonucleotide (IRO) compounds to modulate toll-like receptor based immune response
Owner:IDERA PHARMA INC
Polypeptides and use thereof
InactiveUS20090054321A1Symptoms improvedAlleviate the conditionPeptide/protein ingredientsLibrary screeningPeptideAmino acid
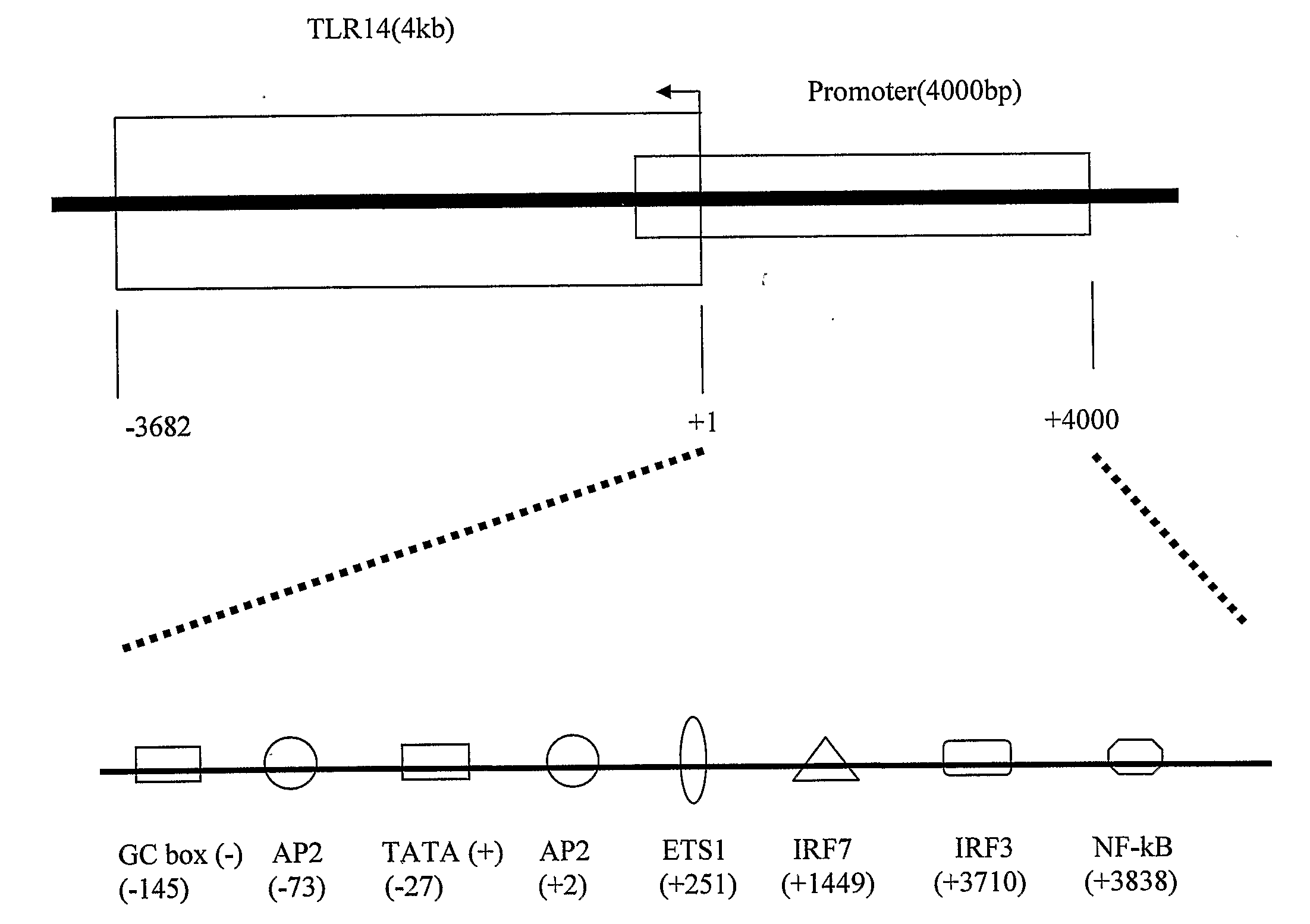
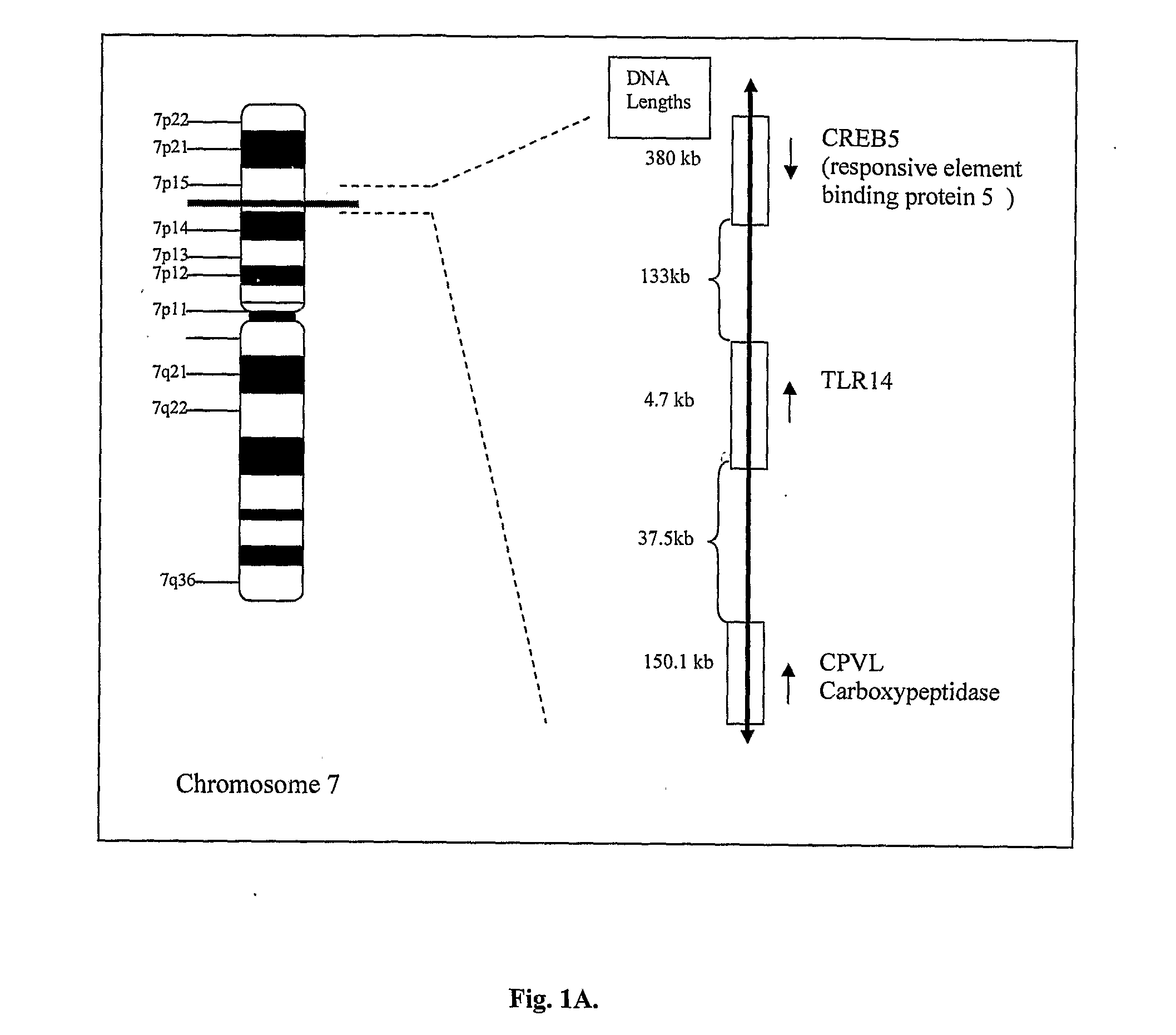
An isolated polypeptide comprises an amino acid sequence of SEQ ID No. 1 or 2 or a variant or fragment thereof. The variant may comprise an amino acid sequence that is at least 70% or 95% identical to the amino acid sequence of SEQ ID No. 1 or 2. A fragment thereof may be a peptide comprising at least 12 contiguous amino acids of SEQ ID No. 1 or 2. The polypeptide exhibits toll-like receptor activity. The TLR has been named TLR1 4. TLR receptors recognise a range of ligands and activate a series of signalling pathways that lead to the induction of immune and inflammatory genes.
Owner:OPSONA THERAPEUTICS
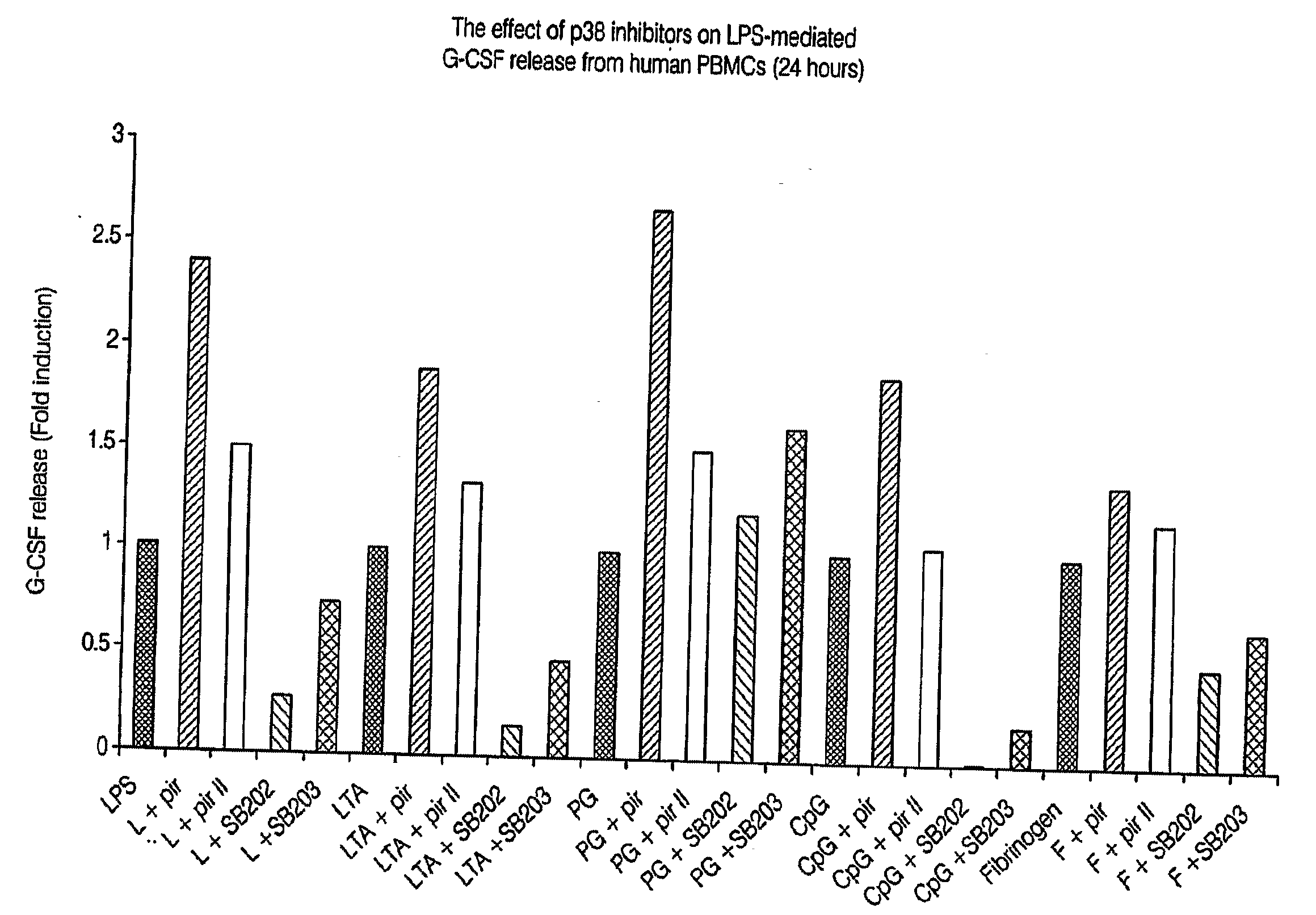
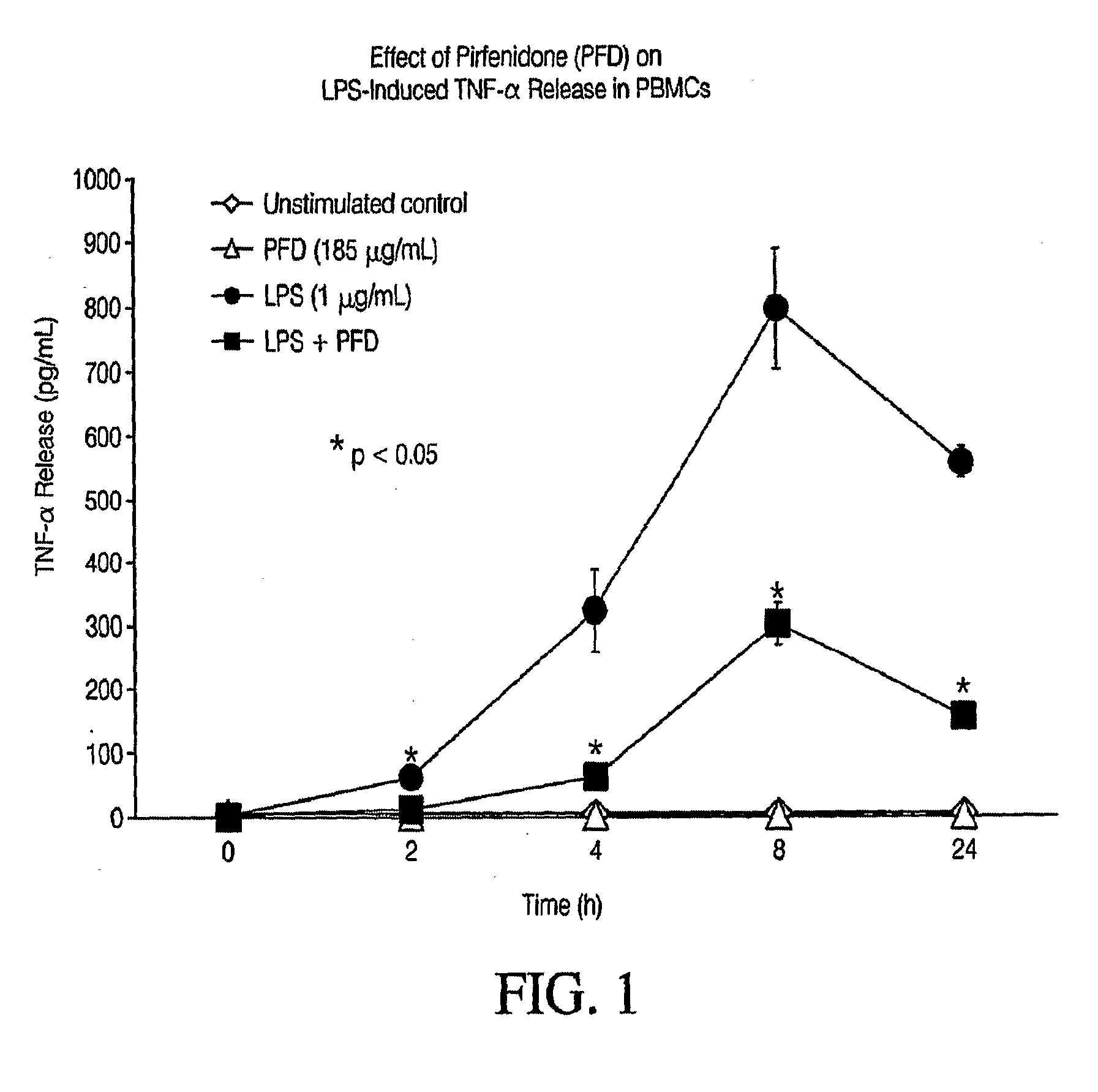
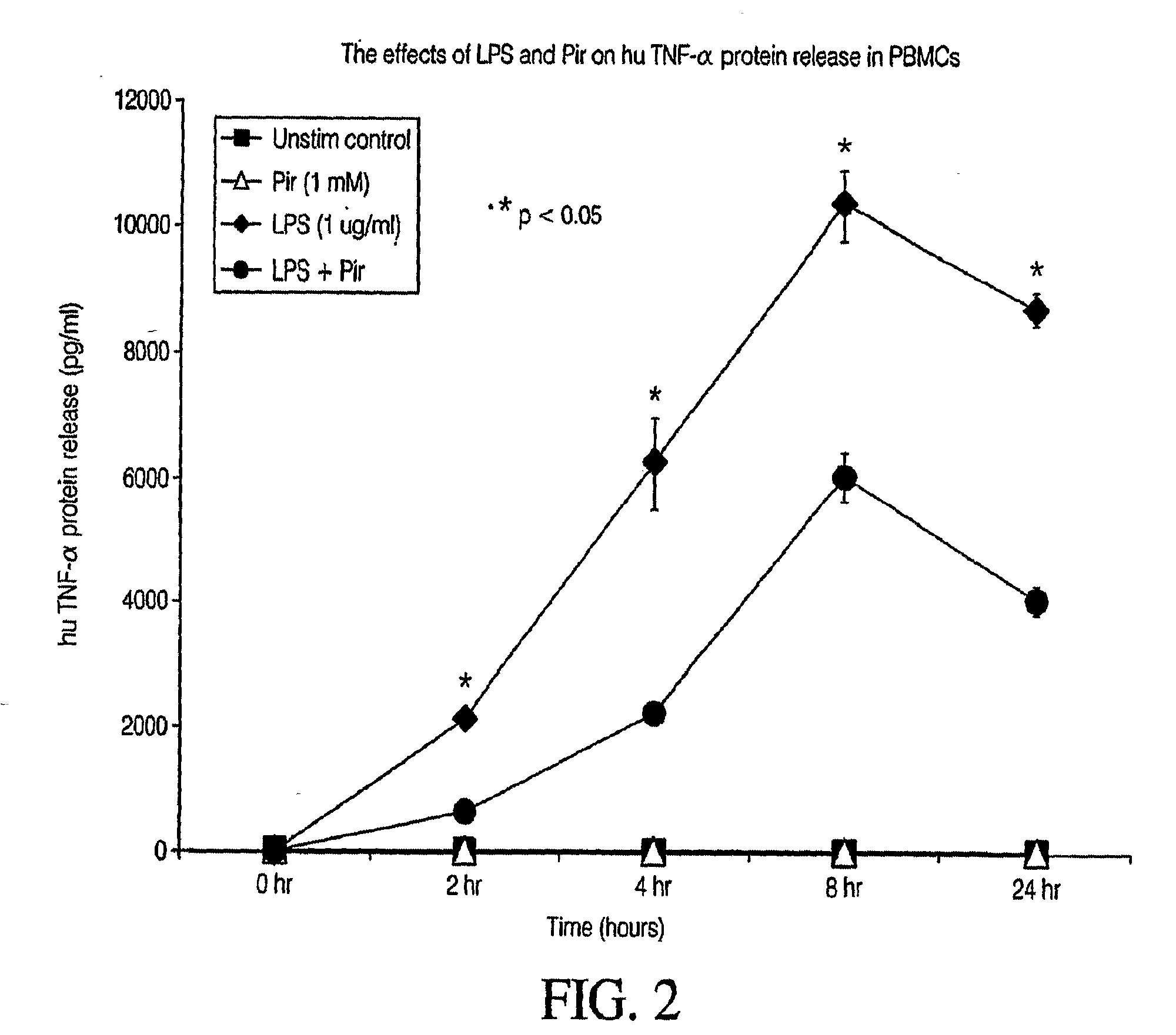
Pirfenidone/toll-like receptor (TLR) agonist compositions and methods for using them to stimulate production of granulocyte colonizing stimulating factor (g-csf)
InactiveUS20090170804A1Increase the number ofBiocideCarbohydrate active ingredientsPirfenidoneToll-like receptor
The invention disclosed herein relates to compositions and methods for treating subjects suffering from or at risk of developing neutropenia. In some embodiments, the methods comprise administering to a subject suffering from or at risk of developing neutropenia, an effective amount of pirfenidone and one or more toll-like receptor (TLR) agonists.
Owner:INTERMUNE INC
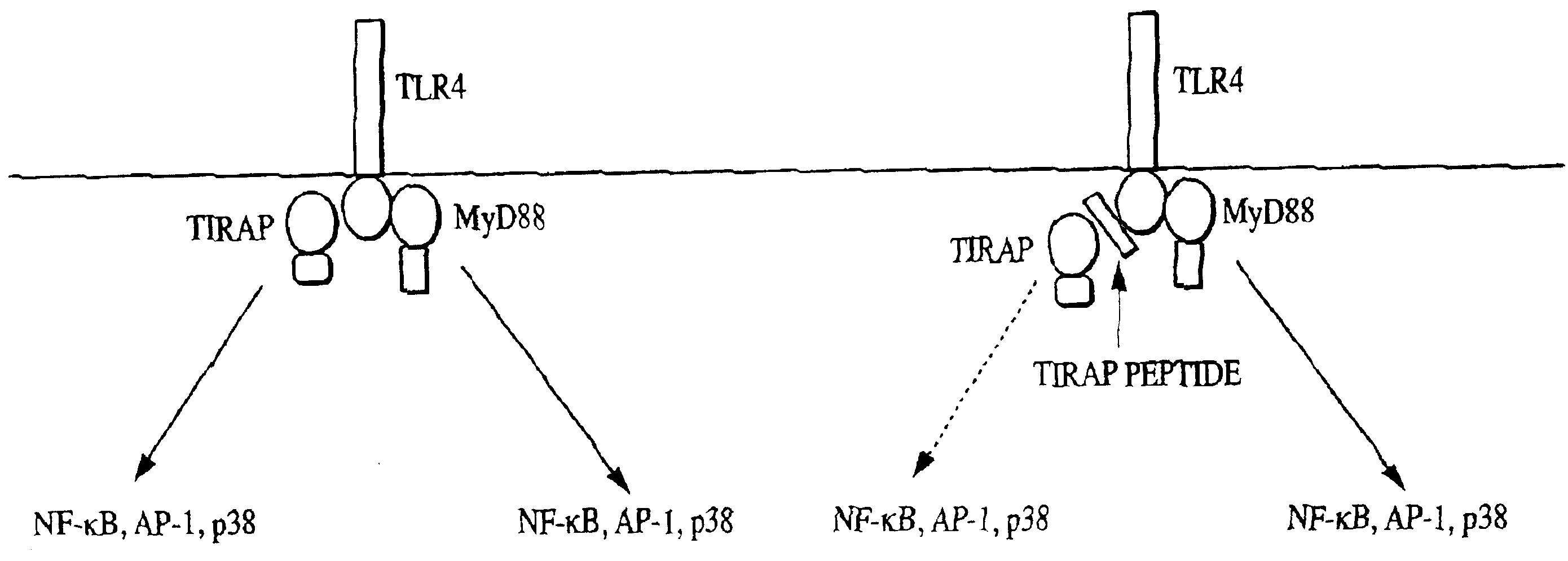
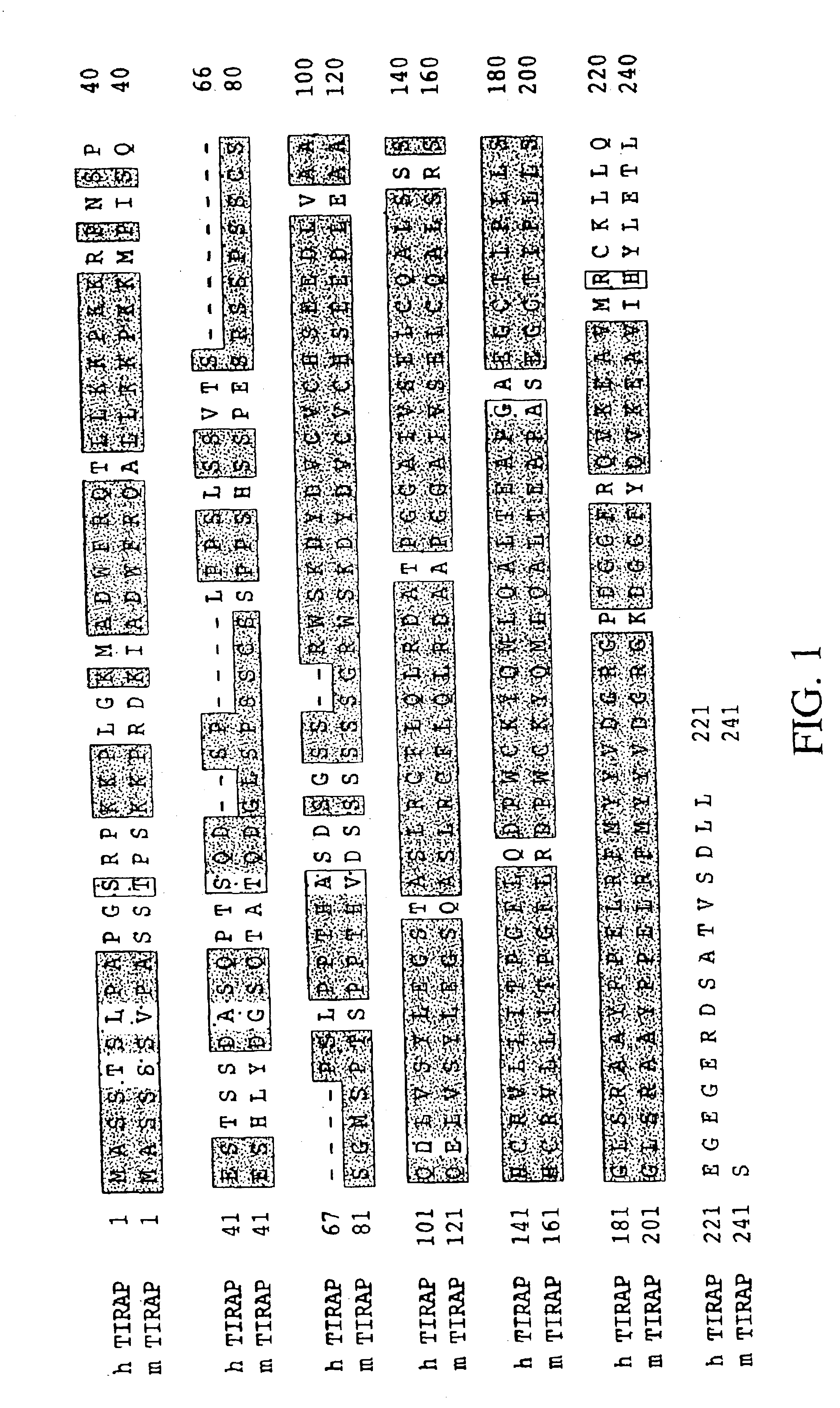
Toll/interleukin-1 receptor adapter protein (TIRAP)
Owner:CURAEN CORP
Method of transfection and compositions therefor
Owner:ENA RESPIRATORY PTY LTD
Toll-like receptor 8 (TLR8)-specific antagonists and methods of making and uses thereof
PendingCN111542515AOrganic chemistryHeterocyclic compound active ingredientsTLR8Autoimmune condition
Toll-like receptor 8 (TLR8)-specific inhibitors and methods of using the same in individuals having an autoimmune disease or an inflammatory disorder.
Owner:UNIV OF COLORADO THE REGENTS OF
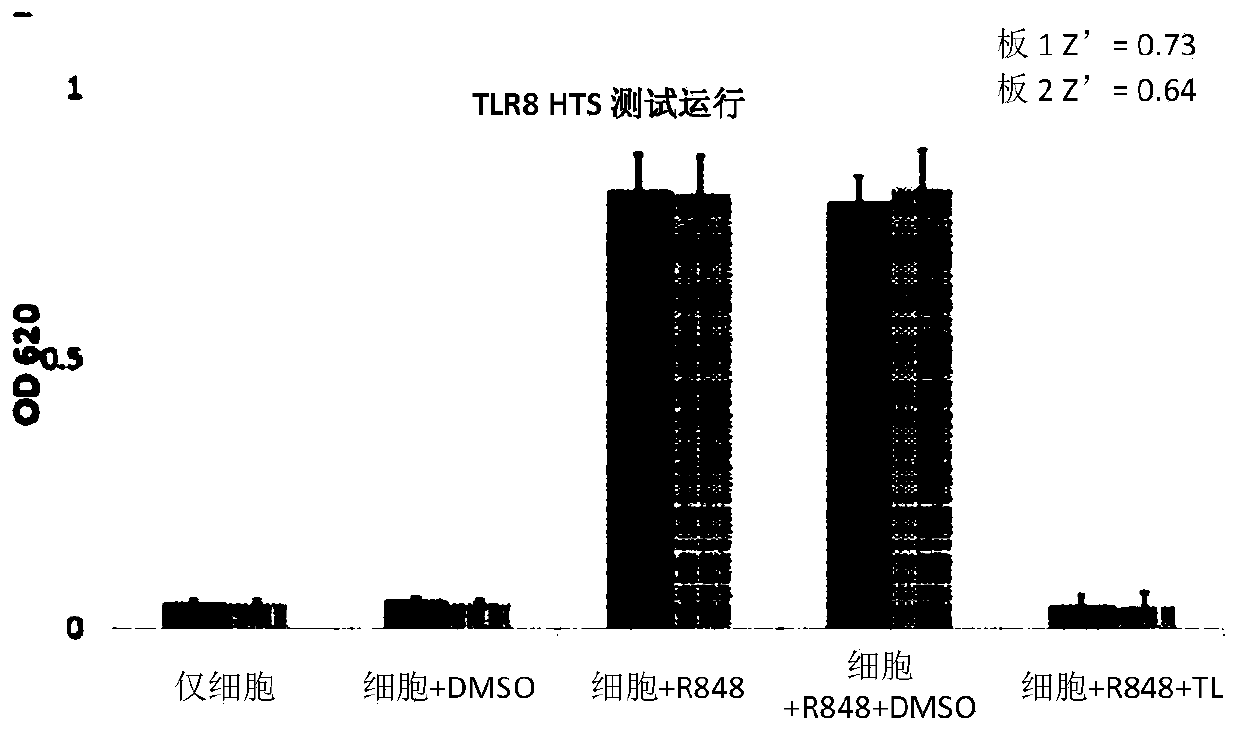
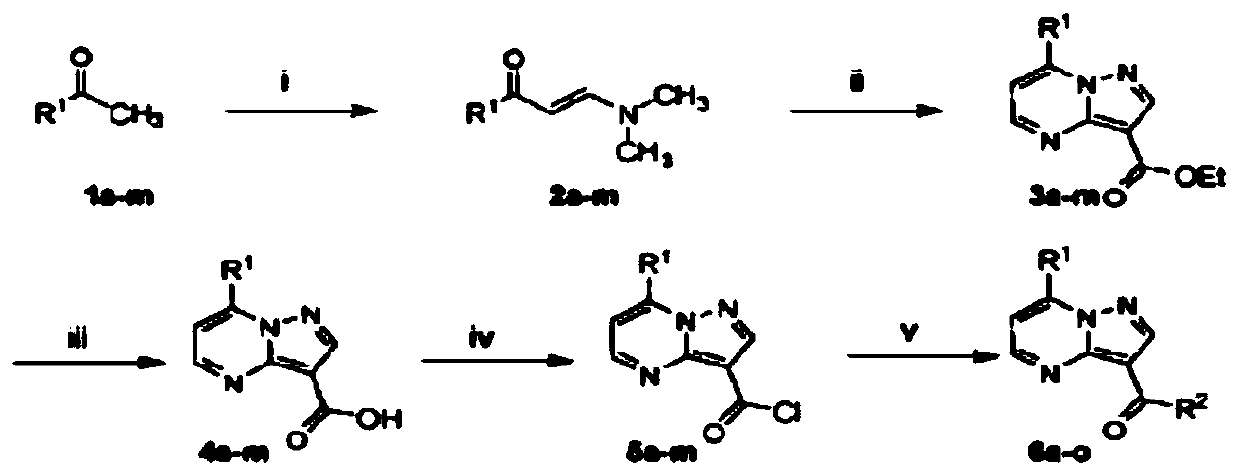
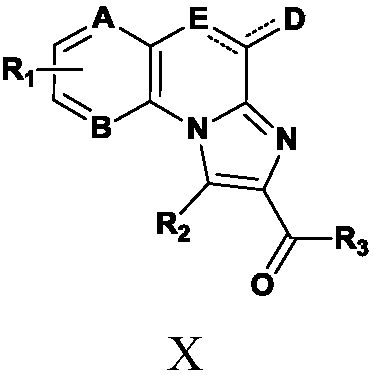
TLR (Toll-like receptor) modulators, pharmaceutical composition thereof, preparation method and application
ActiveCN108794485AGood TLR7/8 regulatory activityStrong cytotoxicityOrganic active ingredientsNervous disorderDrugToll-like receptor
The invention belongs to the field of pharmaceutical chemistry, particularly relates to compounds with TLR (Toll-like receptor) modulation effect as well as a preparation method and an application ofthe compounds, and provides compounds represented by the following formula X in the description. The compounds of formula X can be used as the TLR modulators and have higher activity. Besides, the compounds have the characteristics of high efficiency, low toxicity, resistance to drug resistance and the like, and have clinical application value. Moreover, synthesis steps of the compounds are simple, so that the compounds have higher economic utilization value.
Owner:北京施安泰医药技术开发有限公司
Toll-like receptor 9 effector agents and uses thereof
InactiveUS20050244410A1Genetic material ingredientsImmunoglobulins against cell receptors/antigens/surface-determinantsAntibodyToll-Like Receptor 9
Cell surface TLR9 and TLR9 ligand binding agents are disclosed. The binding agents include antibodies and other proteins. The binding agents are useful as therapeutics, diagnostics or research reagents.
Owner:BASSIRI ASHLYN +9
Humanised Antibodies to Toll-Like Receptor 2 and Uses Thereof
ActiveUS20120164159A1Strong binding specificityGood curative effectSenses disorderFungiHeavy chainAutoimmune disease
A fully humanised antibody having binding specificity to Toll-like Receptor 2 comprises a light chain and a heavy chain entirely comprised of amino acid sequence of human origin. The variable region of the light chain comprises an amino acid sequence which is substantially homologous with the sequence of SEQ ID NO:1, while the variable region of the heavy domain comprises an amino acid sequence which is substantially homologous with the sequence of SEQ ID NO:4. Also provided are nucleic acids encoding such antibodies, as well as the use of the antibodies in medicine, in particular for the treatment of inflammatory and autoimmune diseases which are mediated by Toll-like Receptor 2 activation and signalling.
Owner:NEURAMEDY CO LTD
Methods of Treating, Reducing, or Preventing Autoimmune Conditions
InactiveUS20080020991A1Reduce or prevent conditionReduce effectOrganic active ingredientsSugar derivativesScreen methodToll-like receptor
The present invention relates to methods and kits for treating, reducing, or preventing autoimmune conditions such as rheumatoid arthritis by administering to a mammal in need thereof an agent that modulates the expression level or biological activity of Toll-like receptor-9 (TLR-9). Also disclosed are screening methods that make use of TLR-9 for the identification of novel therapeutics for autoimmune conditions.
Owner:JOSLIN D ABETES CENTER INC
Application of Toll-like receptor ligand protein in resisting bacterial infection
PendingCN110714000AEnhanced phagocytosisIncrease lethalityAntibacterial agentsBacterial antigen ingredientsCytokineImmunity response
The invention provides an application of a Toll-like receptor ligand protein in resisting bacterial infection. According to the application, streptococcus pneumoniae endopeptidase O (PepO) is a ligandof a Toll-like receptor 2 and a Toll-like receptor 4, can remarkably enhance phagocytosis and killing effects of macrophages on pathogenic bacteria, and can up-regulate secretion of related cytokinesand chemokines, thereby inducing strong innate immune response and enhancing resistance of respiratory tracts to pathogenic bacteria infection.
Owner:CHONGQING MEDICAL UNIVERSITY
Anti-TLR4 antibodies and methods of use thereof
ActiveUS8734790B2Immunoglobulins against cell receptors/antigens/surface-determinantsMammal material medical ingredientsAntiendomysial antibodiesTransplant rejection
This invention relates generally to antibodies that specifically bind Toll-like Receptor 4 (TLR-4), and to methods of using the anti-TLR4 antibodies as therapeutics and to methods of using the anti-TLR4 antibodies in methods of preventing transplant rejection and / or prolonging survival of transplanted biological material.
Owner:NOVIMMUNE
Human TOLL-like receptor proteins, related reagents and methods
InactiveCN1263555AFungiCell receptors/surface-antigens/surface-determinantsToll-like receptorPolyclonal antibodies
Nucleic acids encoding nine human receptors, designated DNAX Toll-like receptors 2-10 (DTLR2-10), homologous to the Drosophila Toll receptor and the human IL-1 receptor, purified DTLR proteins and fragments thereof, mono- / polyclonal antibodies against these receptors, and methods for diagnostic and therapeutic use.
Owner:SCHERING AG
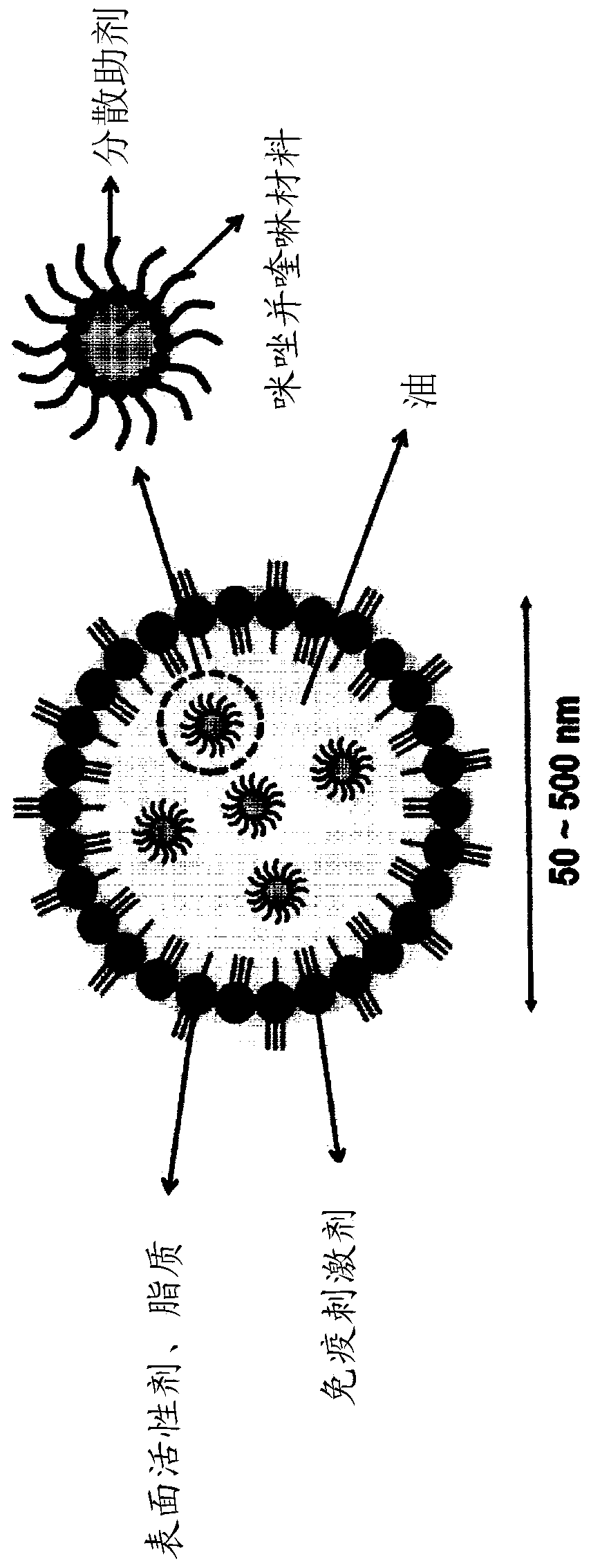
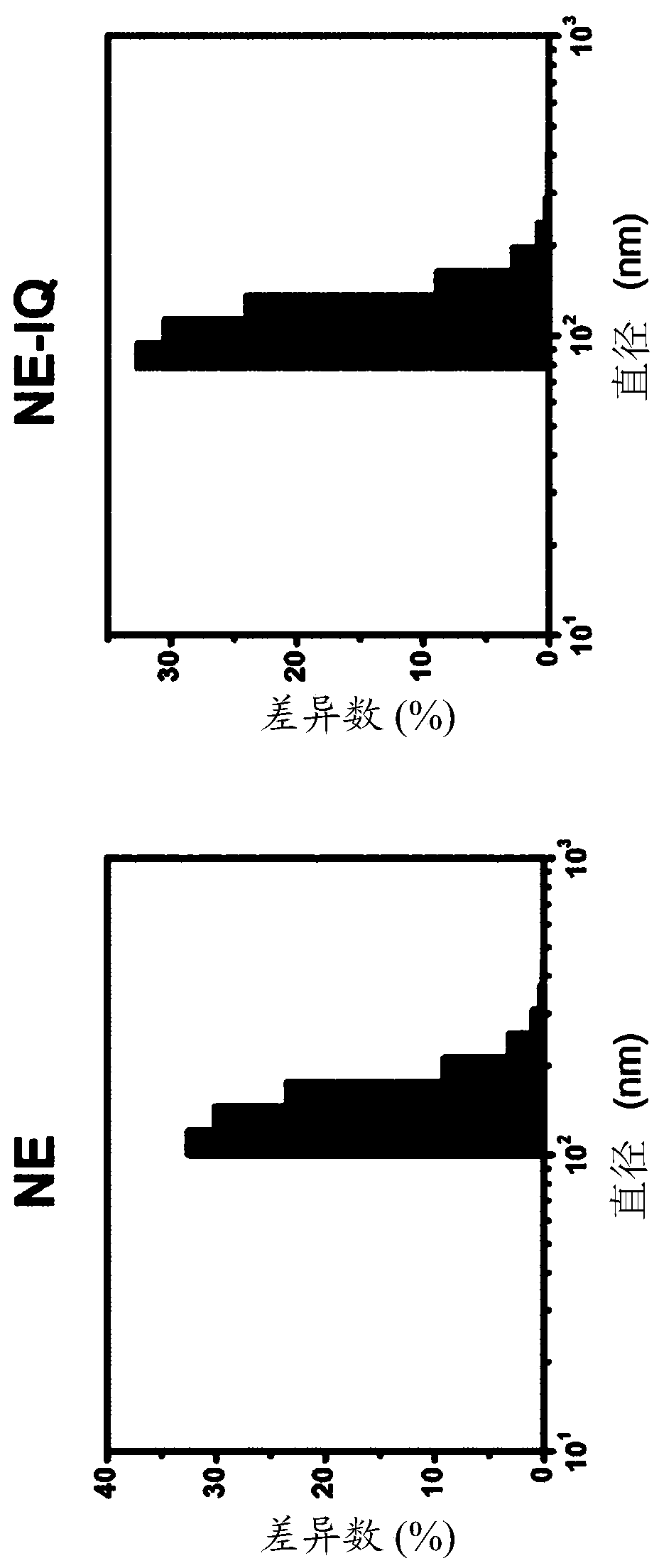
Nanoemulsion comprising imidazoquinoline-based material and use thereof
InactiveCN110430868AGood dispersionImprove immunityOrganic non-active ingredientsAntiinfectivesQuinolineTGE VACCINE
Owner:DANDI BIOSCI INC
ACTIVATION OF INNATE IMMUNITY FOR ENHANCED NUCLEAR REPROGRAMMING OF SOMATIC CELLS WITH mRNA
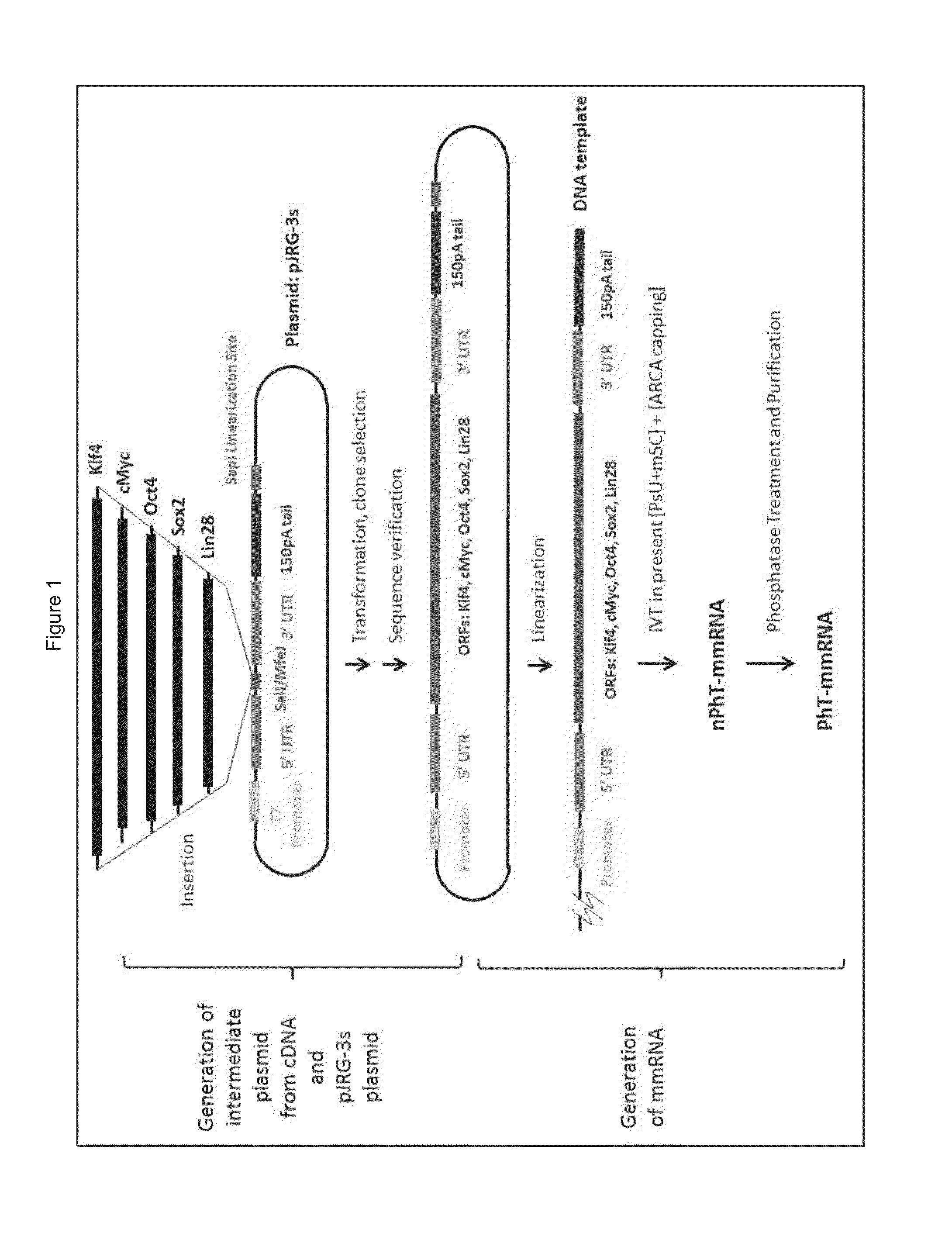
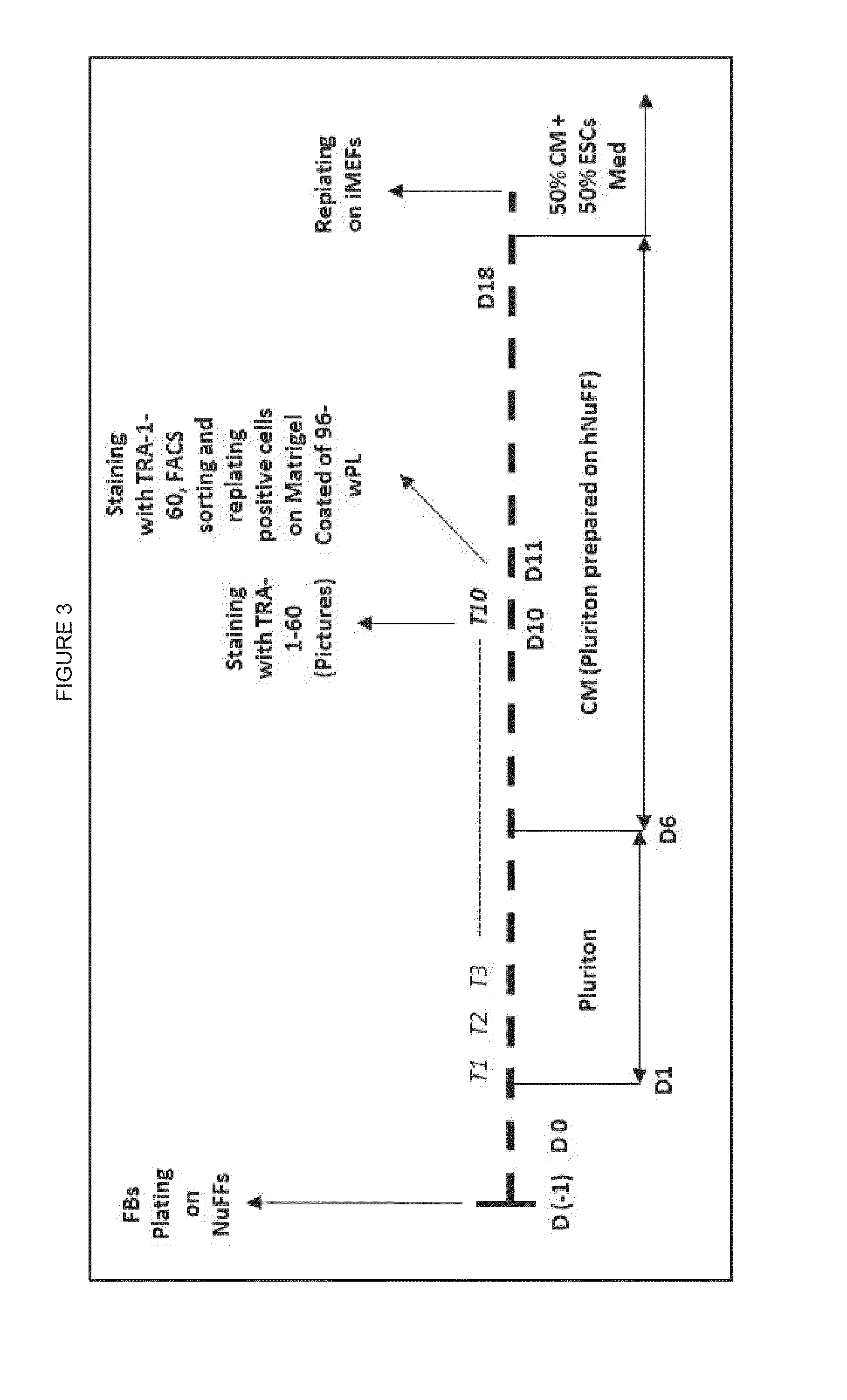
ActiveUS20150225699A1Enhance transdifferentiationGenerate efficientlyEpidermal cells/skin cellsArtificially induced pluripotent cellsToll-like receptorNuclear reprogramming
The nuclear reprogramming of somatic cells with mRNA encoding reprogramming factors is shown to be greatly accelerated by activation of innate immune responses in the somatic cell. Methods of activating innate immunity include activation of PKR, of toll-like receptors, e.g. TLR3, etc. In some embodiments the mRNA provides the activator of innate immunity.
Owner:THE BOARD OF TRUSTEES OF THE LELAND STANFORD JUNIOR UNIV
Modulators of toll-like receptors for the treatment of hiv
Owner:GILEAD SCI INC
Synthetic gene
InactiveUS20090274726A1Enhance immune responseImprove responsivenessAntibacterial agentsSenses disorderDiseaseNucleotide
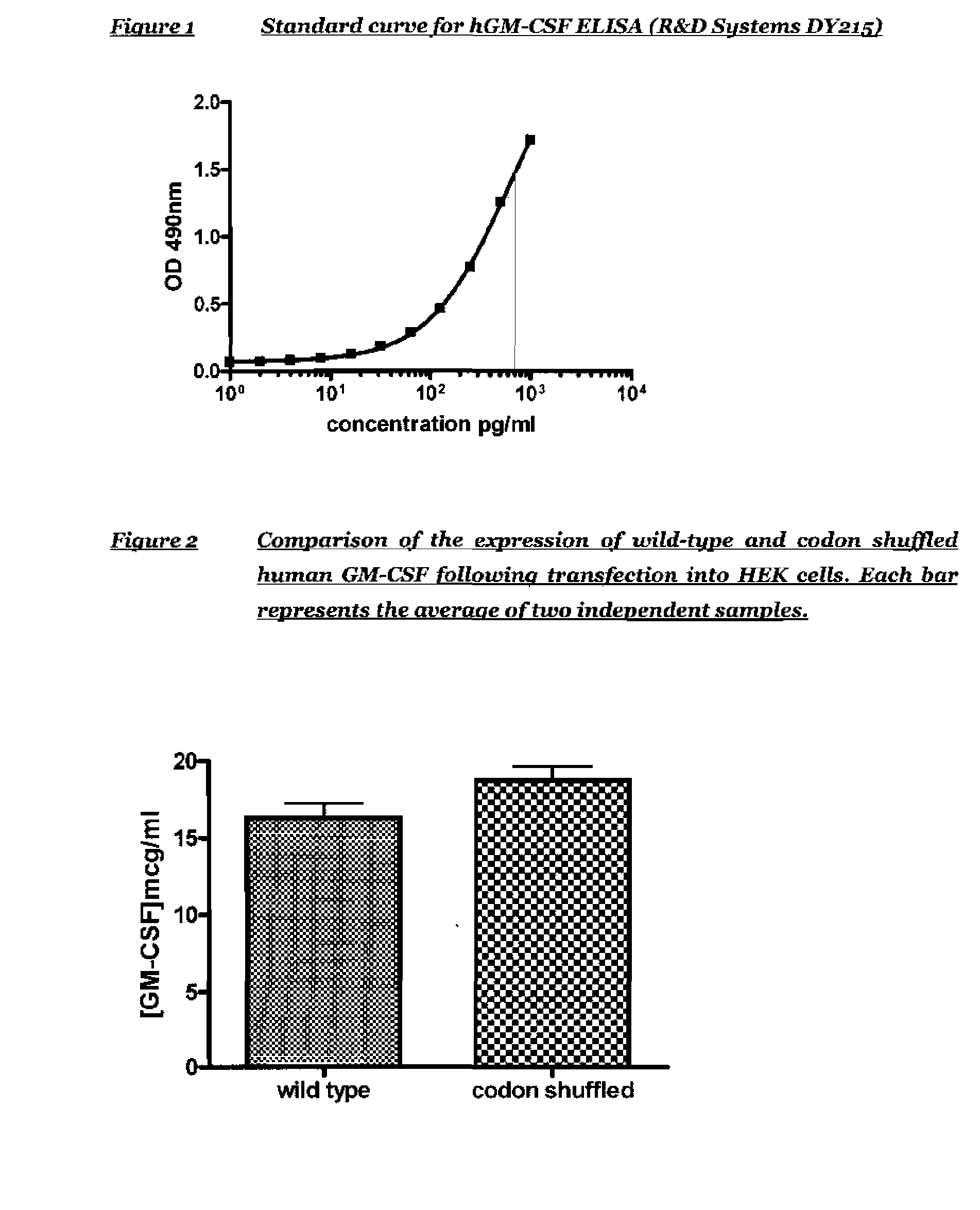
The present invention relates to synthetic genes, processes for designing said synthetic genes and their uses in gene therapy and improved DNA vaccination. The novel synthetic genes and processes are codon shuffled so that they have reduced homology relative to a naturally occurring gene encoding the same protein without altering the overall codon usage frequency of the gene. In particular the present invention relates to improved polynucleotides and methods for the treatment or prevention of disease comprising codon-shuffled GM-CSF nucleic acid sequences. Nucleic acid vaccines of the present invention may comprise a combination of a nucleotide sequence encoding codon-shuffled GM-CSF, a nucleotide encoding an antigen against which it is desired to raise an immune response and a toll-like receptor (TLR) agonist.
Owner:GLAXO GROUP LTD
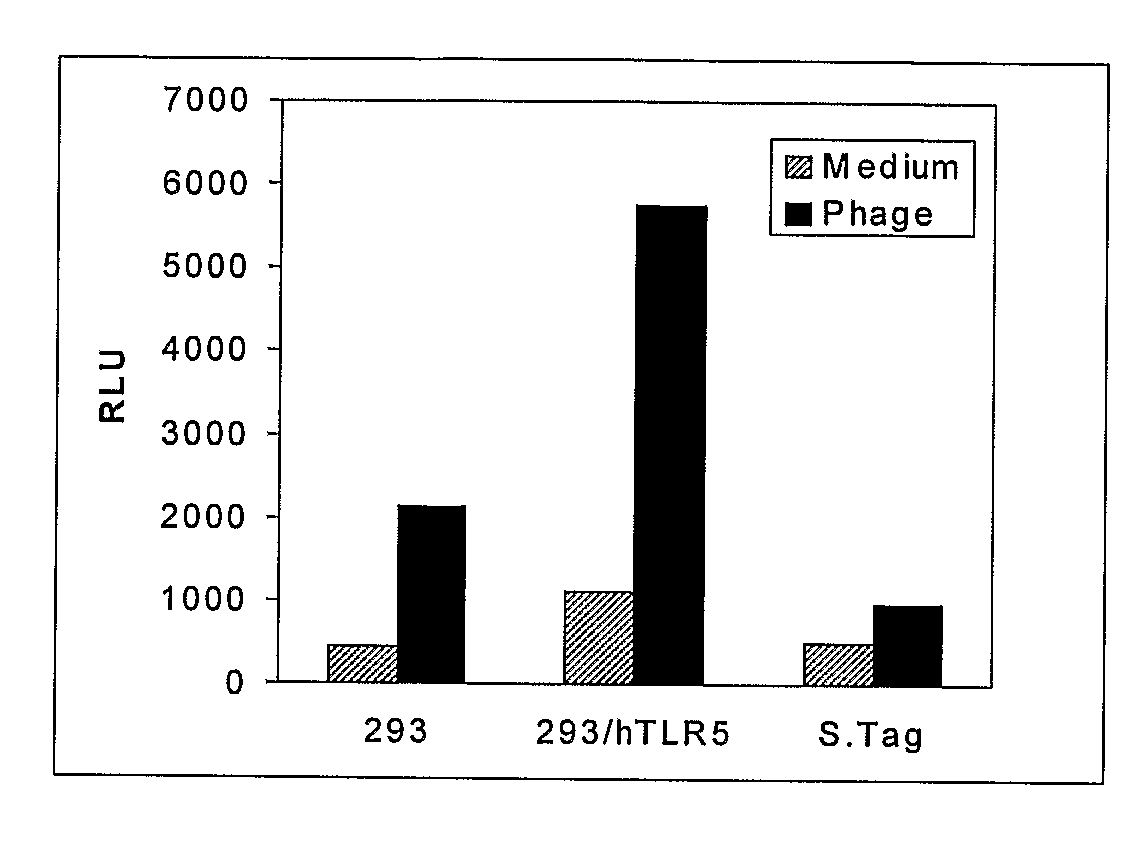
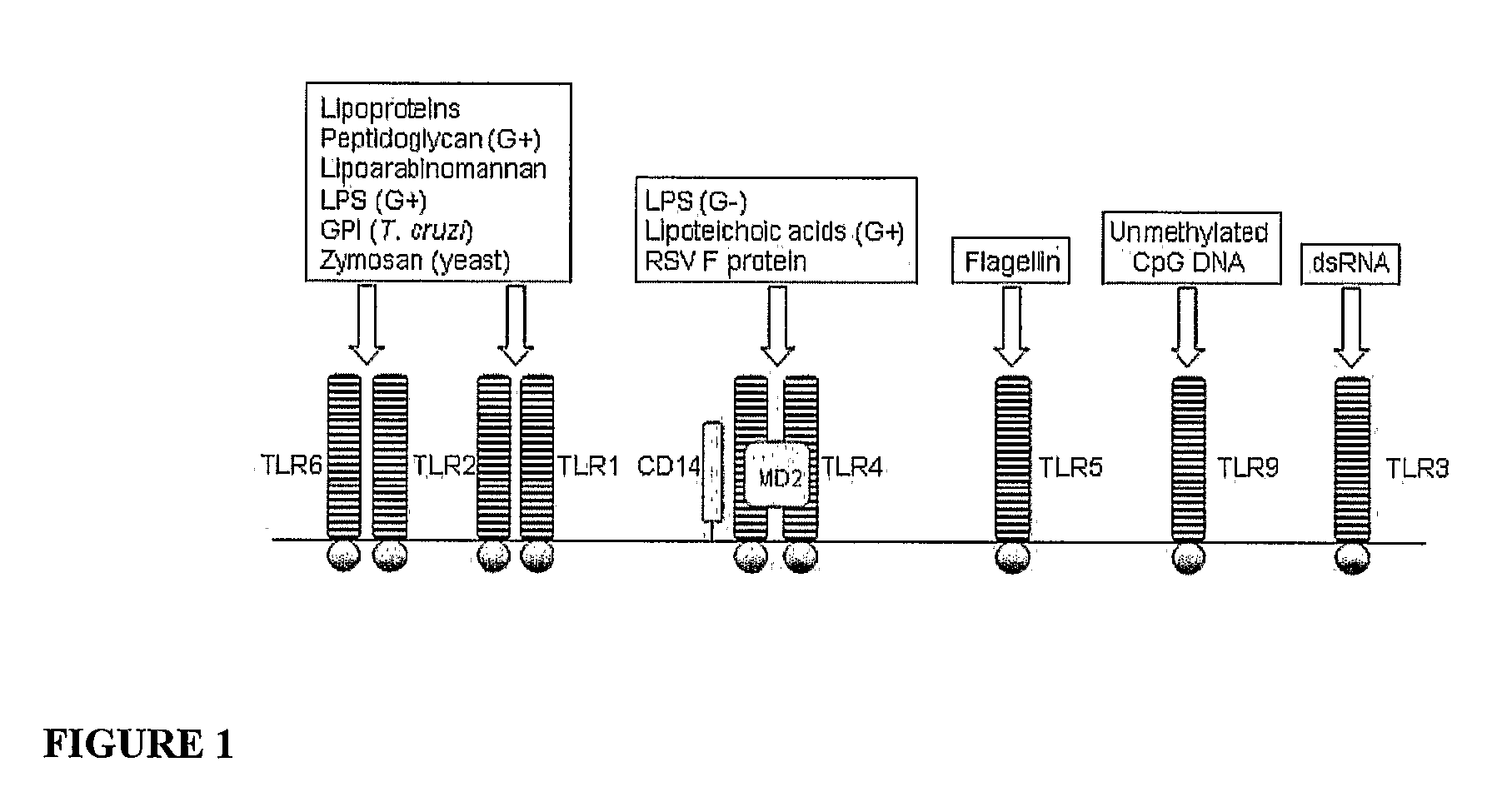
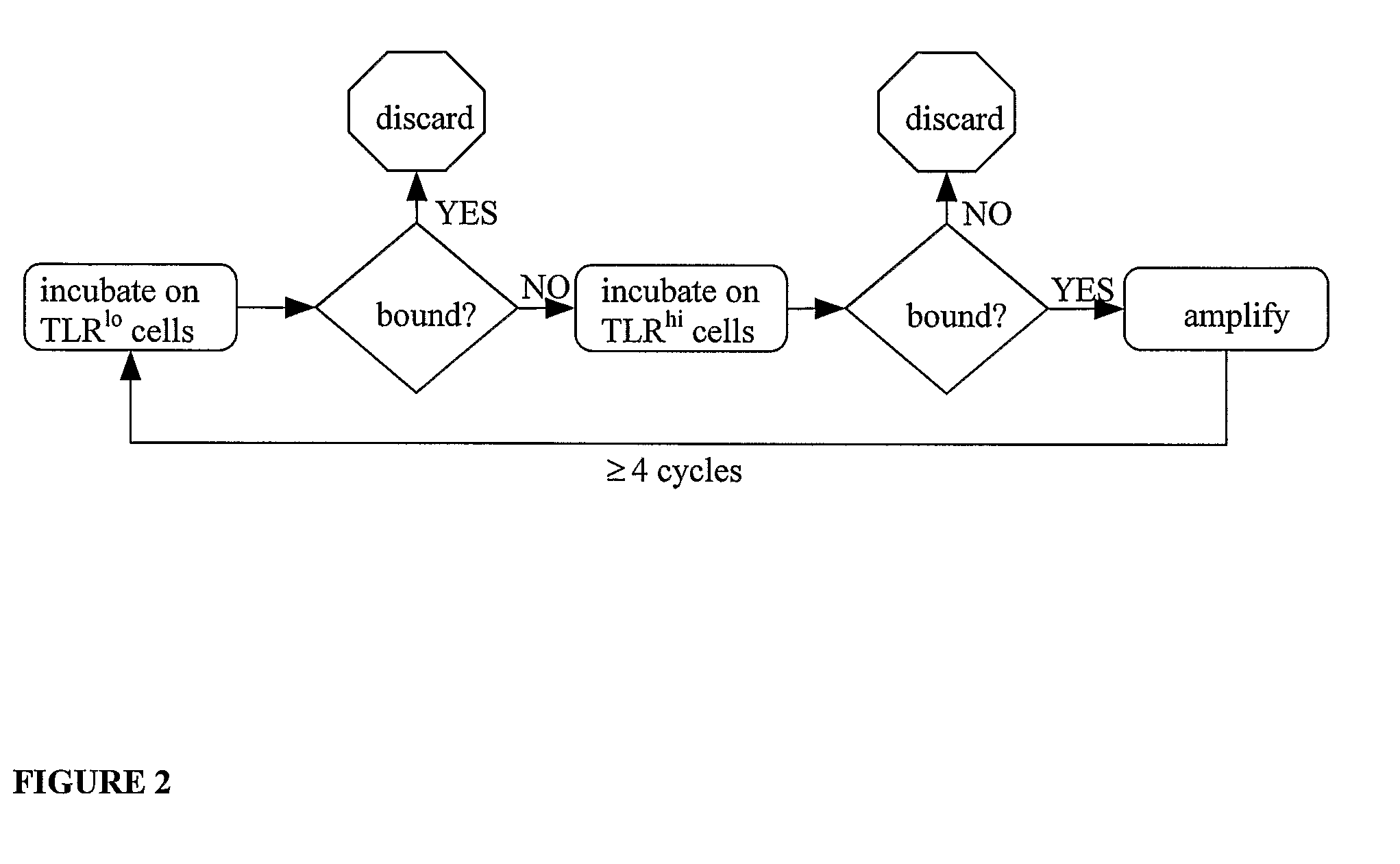
Method to identify polypeptide toll-like receptor (TLR) ligands
The present invention provides novel methods to identify polypeptide ligands for Toll-like Receptors (TLRs), such as TLR2, TLR4 and TLR5. The method involves the use of phage display technology in an iterative biopanning procedure. The invention also provides polypeptide TLR ligands identified by the methods of the invention. In preferred embodiments, the polypeptide TLR ligands so identified modulate TLR signaling and thereby regulate the Innate Immune Response. The invention also provides vaccines comprising a polypeptide TLR ligand identified by the methods of the invention and an antigen. The invention also provides methods of modulating TLR signaling using the polypeptide TLR ligands and vaccines of the invention.
Owner:VAXINNATE
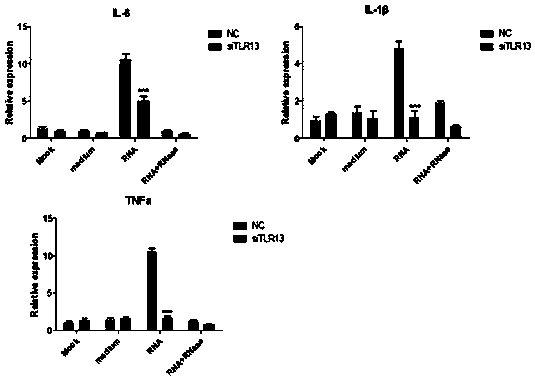
Innate immune TLR13 (toll-like receptor 13) gene of epinephelus coioides as well as eukaryotic expression vectors and application of innate immune TLR13 gene
ActiveCN107557366AHighlight Substantial ProgressFermentationAnimals/human peptidesInflammatory factorsNucleotide
The invention discloses an innate immune TLR13 (toll-like receptor 13) gene of epinephelus coioides as well as eukaryotic expression vectors and an application of the innate immune TLR13 gene for thefirst time. The nucleotide sequence of the TLR13 gene is shown as SEQ. ID.NO:1, the full-length cDNA sequence of the TLR13 gene is shown as SEQ. ID.NO:2, sequences of a pair of primers contained on the full-length cDNA sequence are shown as SEQ. ID.NO:4-5, and the amino acid sequence of the TLR13 protein is shown as SEQ. ID.NO:3. The two eukaryotic expression vectors containing the full-length cDNA sequence of the TLR13 gene are provided, namely, pEGFP-N3-TLR13 and pcDNA3.1-TLR13. Besides, the TLR13 gene or the TLR13 protein is applied to research of regulation of inflammatory factor expression, recognition of bacterial RNA, recognition of vibrio parahaemolyticus RNA and regulation of inflammatory factors.
Owner:SUN YAT SEN UNIV
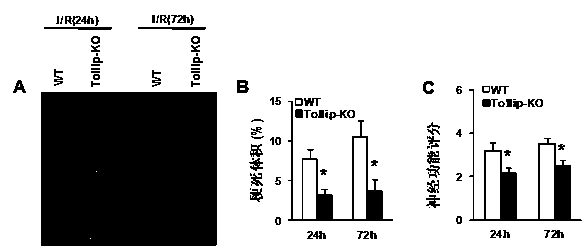
Application of Toll interacting protein (Tollip) in stroke disease
ActiveCN103990147AExacerbating the role of strokeNervous disorderGenetic material ingredientsDiseaseNervous system
The invention discloses a function and application of a Toll interacting protein (Toll-interacting protein, Tollip) gene in a stroke disease, and belongs to the fields of functions and application of genes. The Tollip gene knockout mice are taken as experiment objects, and the model is perfused by middle cerebral artery occlusion. The result shows that the brain infarction volume of the Tollip gene knockout mice is obviously reduced in comparison with that of wild-type control mice, the neurological function is obviously improved, and the cell quantity of brain death is also obviously reduced, so as to find that the Tollip gene can promote deterioration of the nervous system, and aggravate and promote occurrence and development of the stroke. The Tollip can be used as a drug target for screening a drug of treating the stroke, and a Tollip inhibitor can be applied to preparation of the drug for treating the stroke.
Owner:WUHAN UNIV
Selective agonist of toll-like receptor 3
InactiveUS20100310600A1Disease in the subject is reduced or eliminatedImprove morbidityAntibacterial agentsBiocideAgonistAntiproliferative Agents
A mismatched double-stranded ribonucleic acid, which is an agonist for Toll-like receptor 3 (TLR3), is used in vitro or in vivo as an antimicrobial agent, antiproliferative agent, and / or immunostimulant. Poly(l:C11-14U) is a more selective agonist of TLR3 as compared to poly(l:C) even though the both double-stranded RNA are structurally analogous.
Owner:CARTER WILLIAM A +1
Use of Toll-Like Receptor 4 Agonists to Treat Inflammation and Tissue Injury
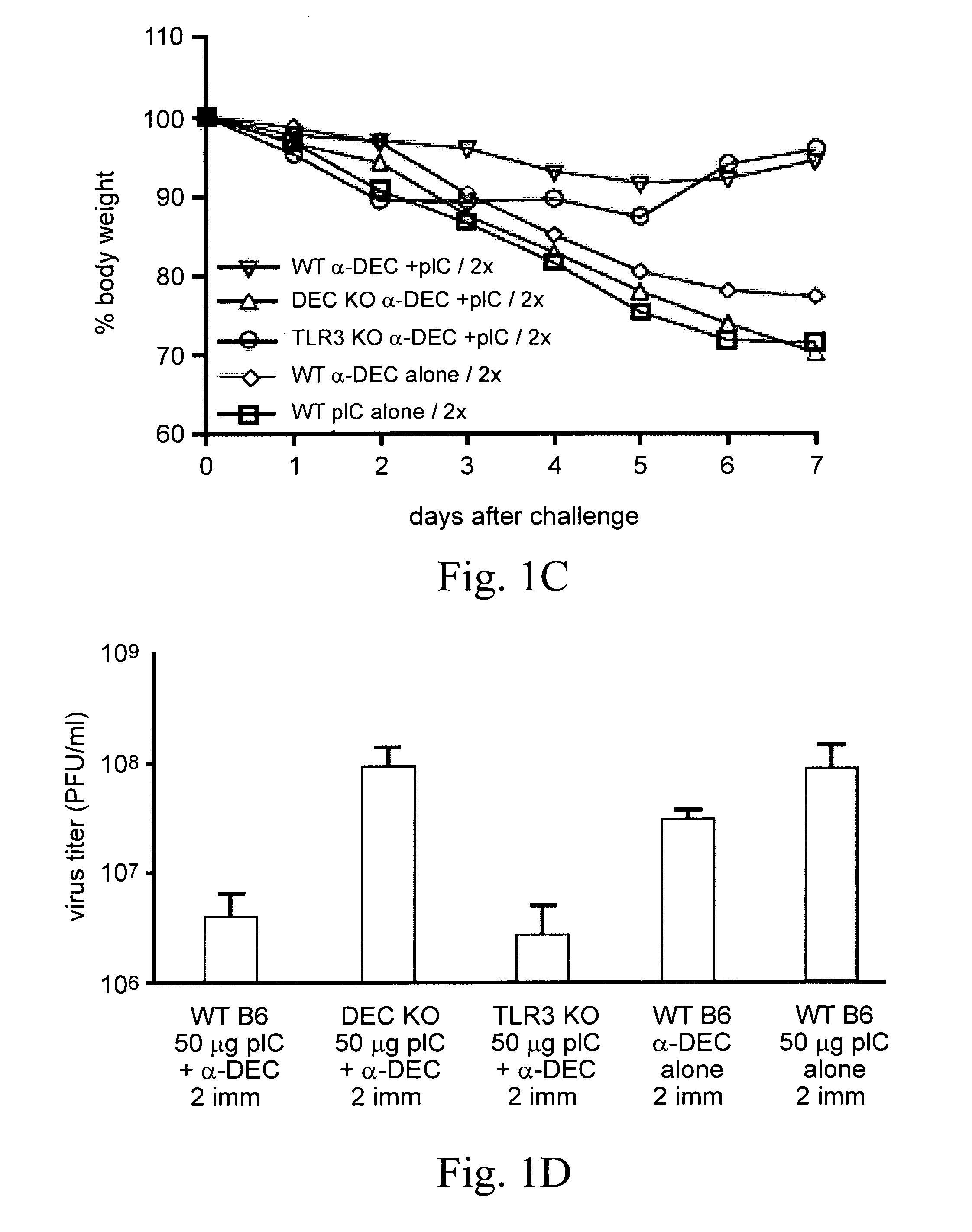
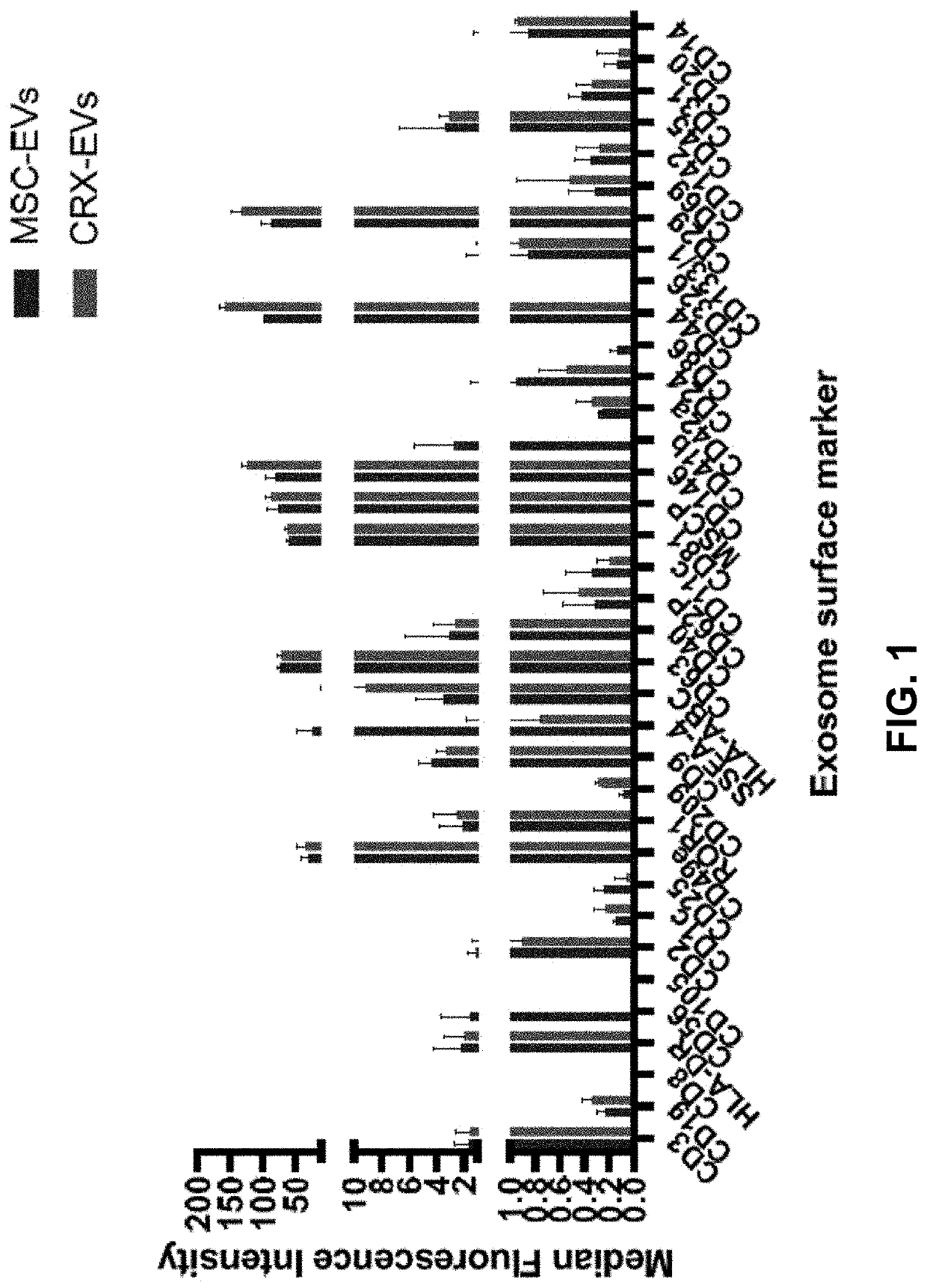
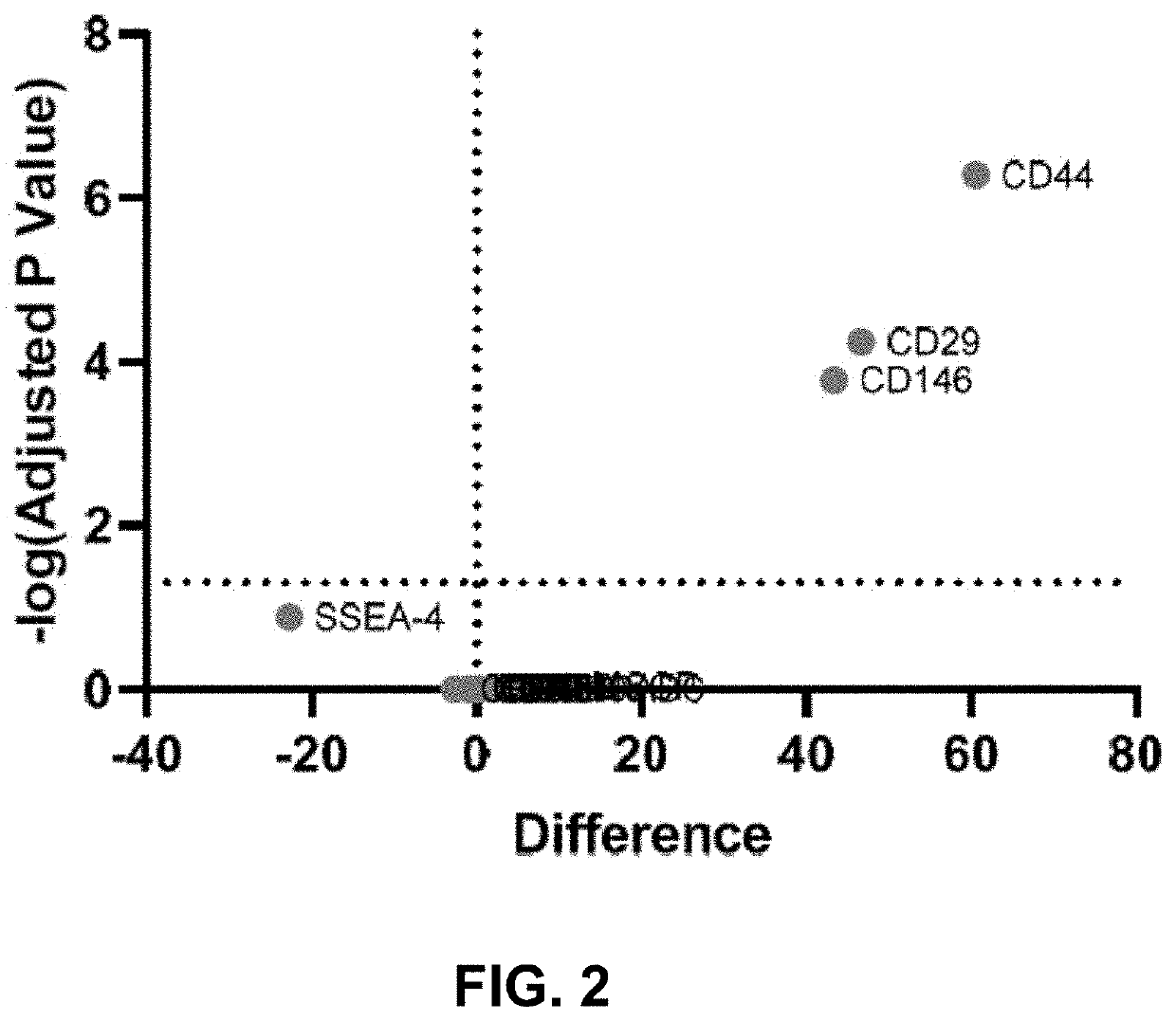
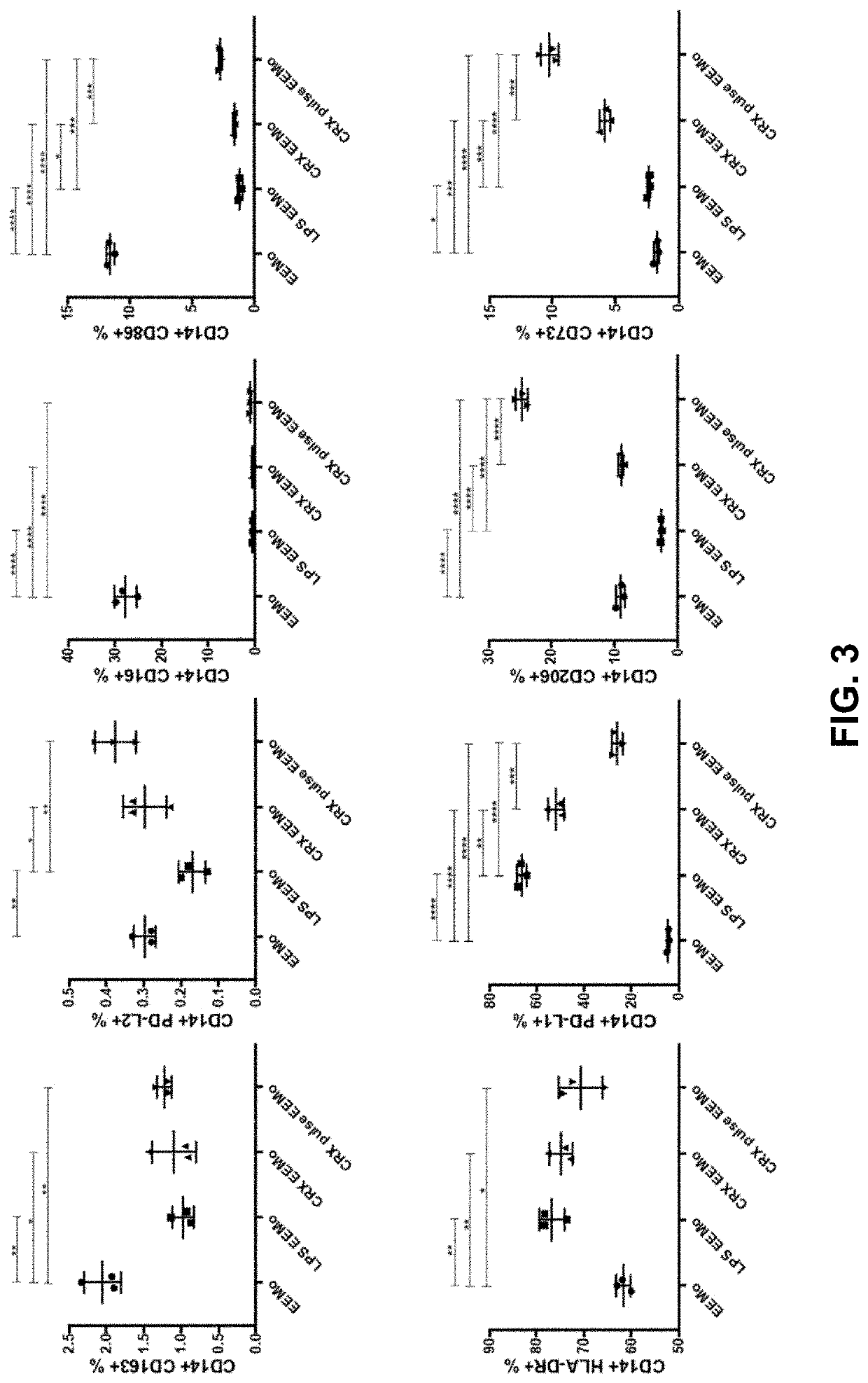
The disclosure relates to populations of educated macrophages and monocytes generated ex vivo or in vivo, and methods of making and using the same using lipid A aminoalkylglucosaminide phosphate molecules, such as CRX molecules or extracellular vesicles (EVs) from mesenchymal stromal cells (MSC) stimulated with CRX molecules. Also described are EVs and methods for making and using the same from MSCs exposed to CRX.
Owner:WISCONSIN ALUMNI RES FOUND
Methods and compositions for diagnosis and treatment of disorders in patients with elevated levels of TLR4 ligands and other biomarkers
This invention relates generally to methods and compositions for diagnosing and treating disorders associated with elevated levels of Toll-like Receptor 4 (TLR4) ligands and other biomarkers. The invention also relates to methods of treating, delaying the progression of, or otherwise ameliorating a symptom of a disorder associated with elevated levels of TLR4 ligands and other biomarkers using agents that interfere with or otherwise antagonize TLR4 signaling, including neutralizing anti-TLR4 antibodies.
Owner:NOVIMMUNE
Toll-like receptor ligands
Toll-like receptor (TLR) ligands having an allose-based core are stable in aqueous formulation and are useful in treating, preventing, or reducing susceptibility to diseases or conditions mediated byTLRs, such as cancer, infectious disease, allergy, autoimmune disease, sepsis, and ischemia reperfusion.
Owner:英尼穆内公司
Intratumoral administration of particles containing a toll-like receptor 9 agonist and a tumor antigen for treating cancer
The present disclosure relates to methods for treating cancer by intratumoral delivery of particles containing a Toll-like receptor 9 agonist (TLR9) and a tumor antigen, in which the TLR9 agonist is apolynucleotide or a chimeric compound thereof. The methods of the present disclosure involve injection of the particles into at least one tumor, and are effective for treating both injected and uninjected tumors of a mammalian subject. Additionally, the present disclosure provides immunogenic compositions containing the particles, as well as methods of manufacture thereof.
Owner:DYNAVAX TECH CORP