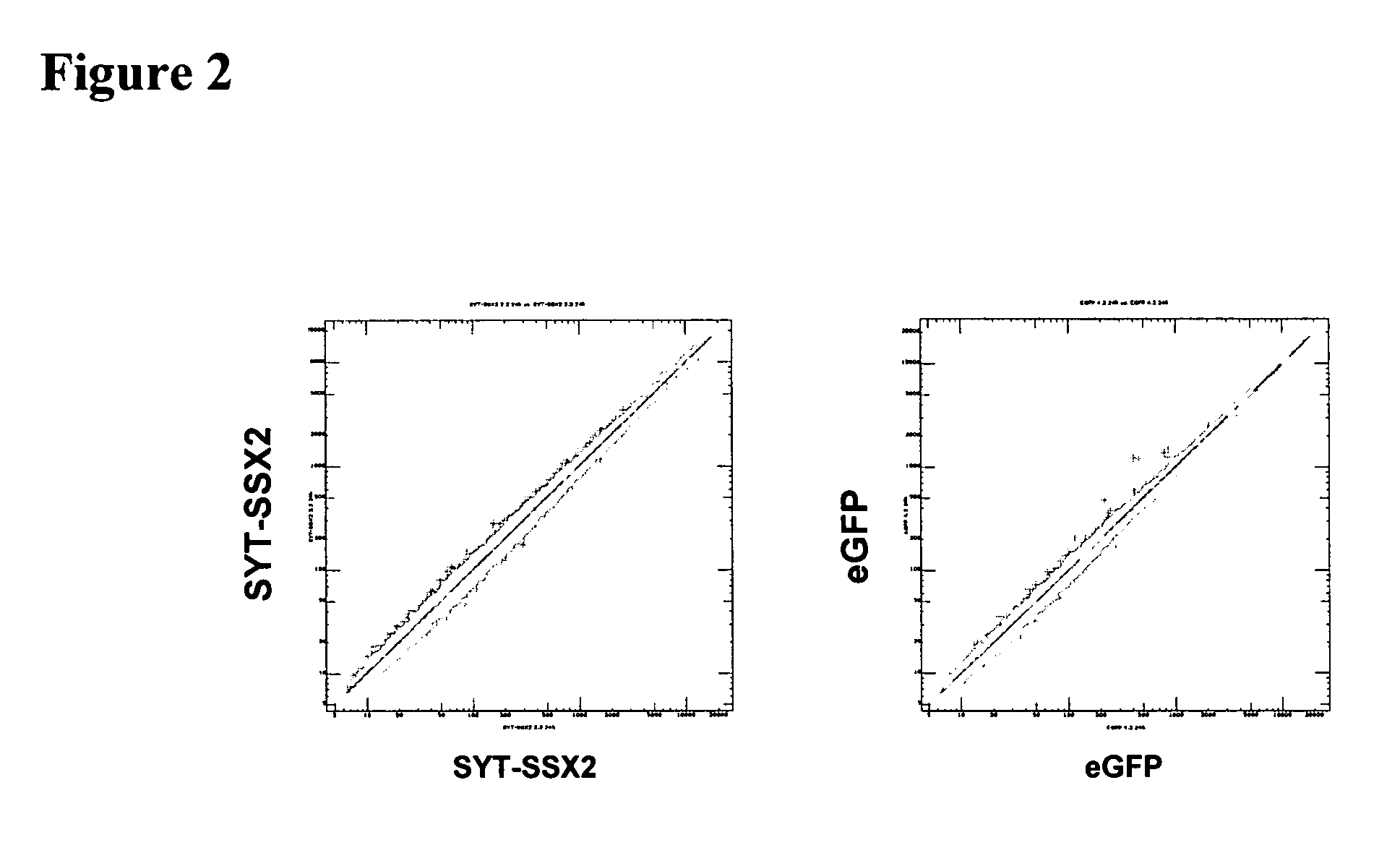
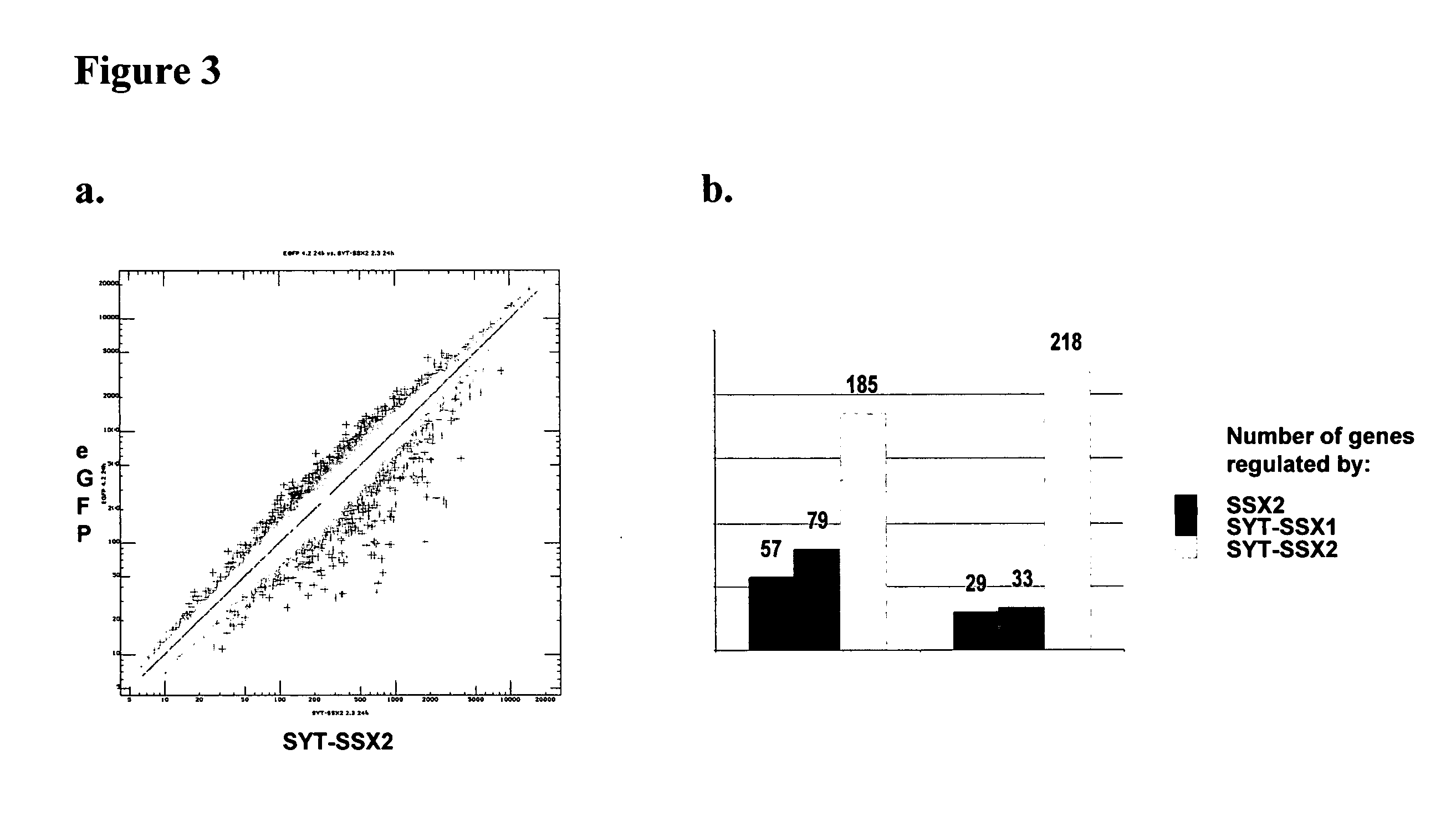
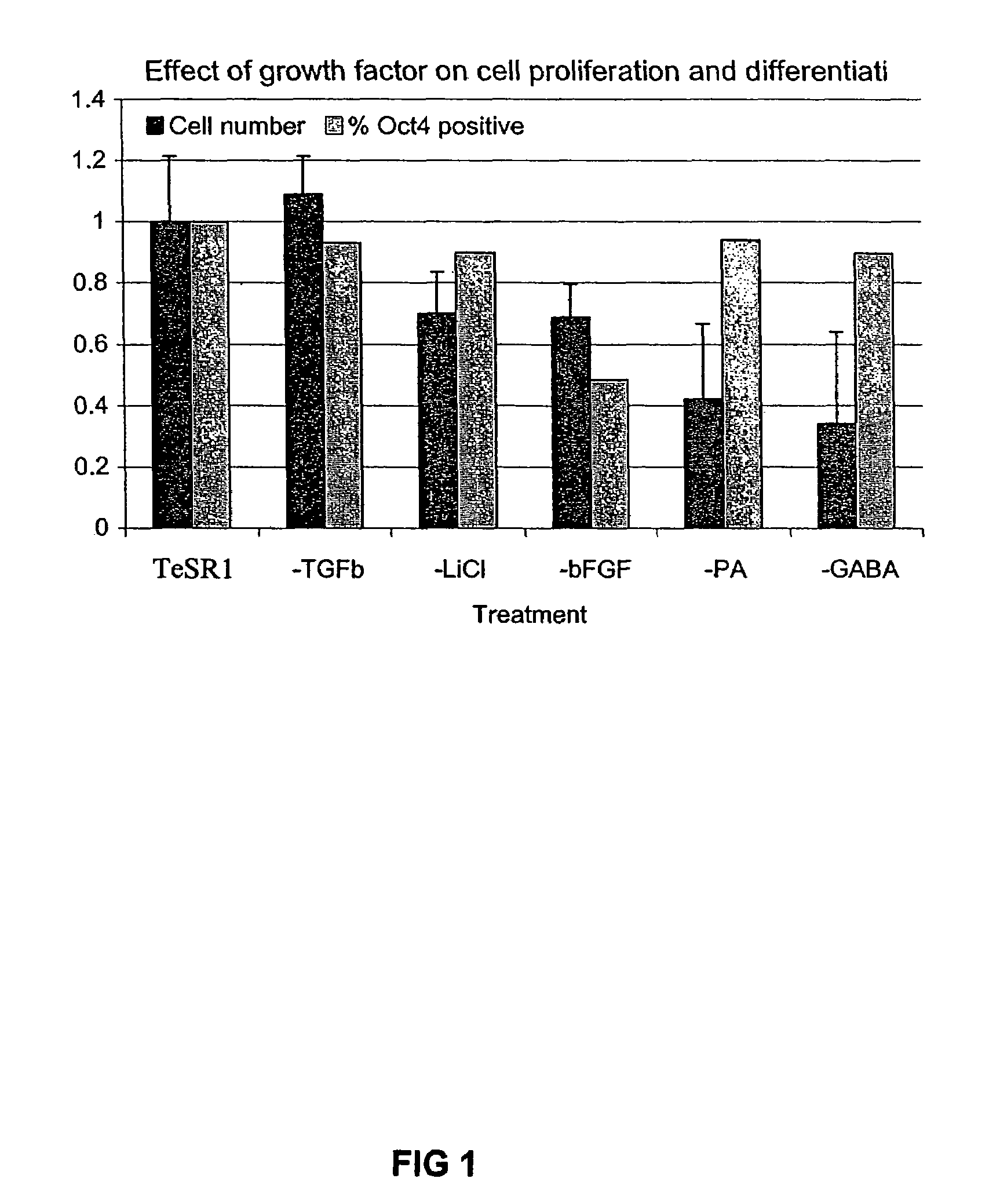
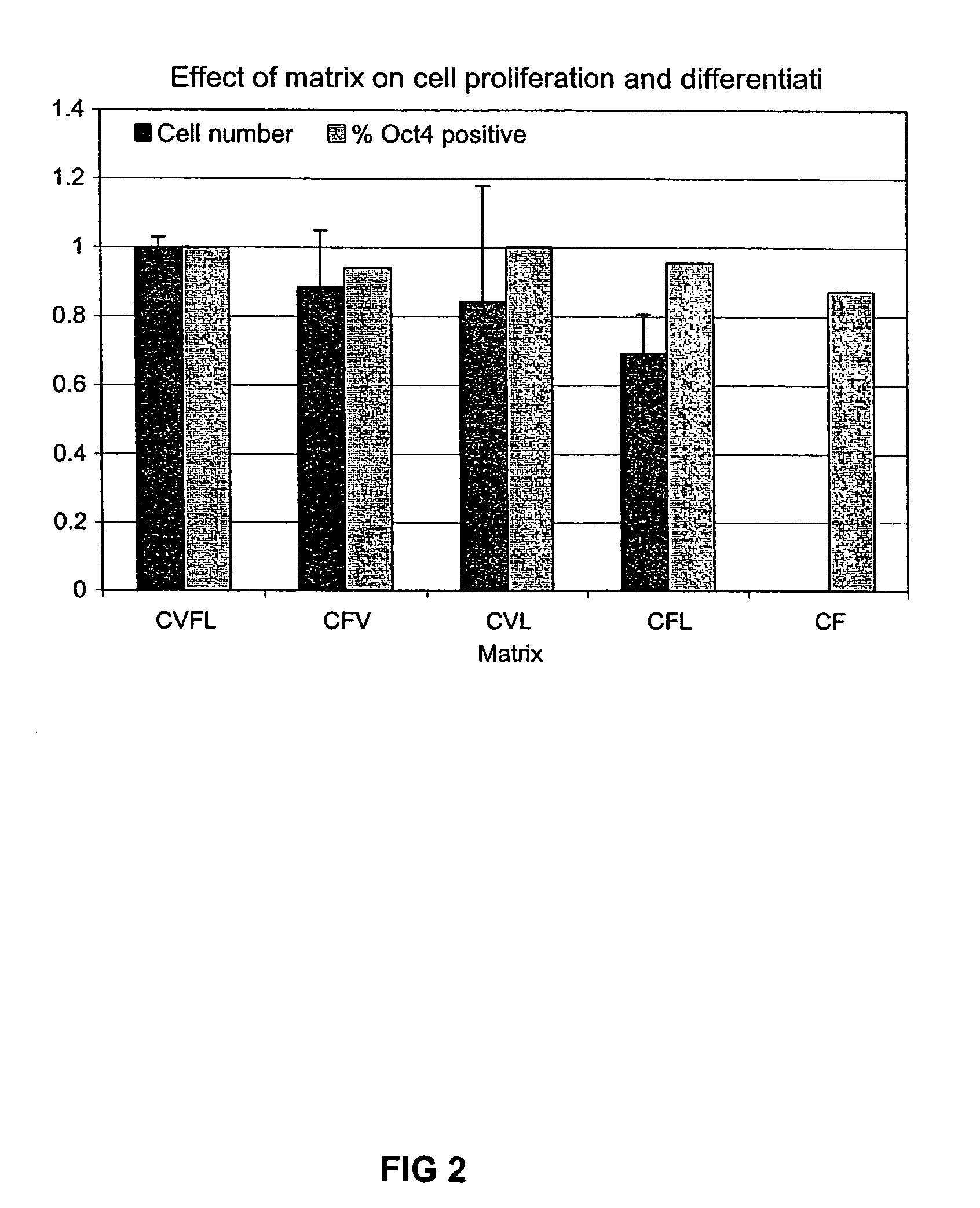
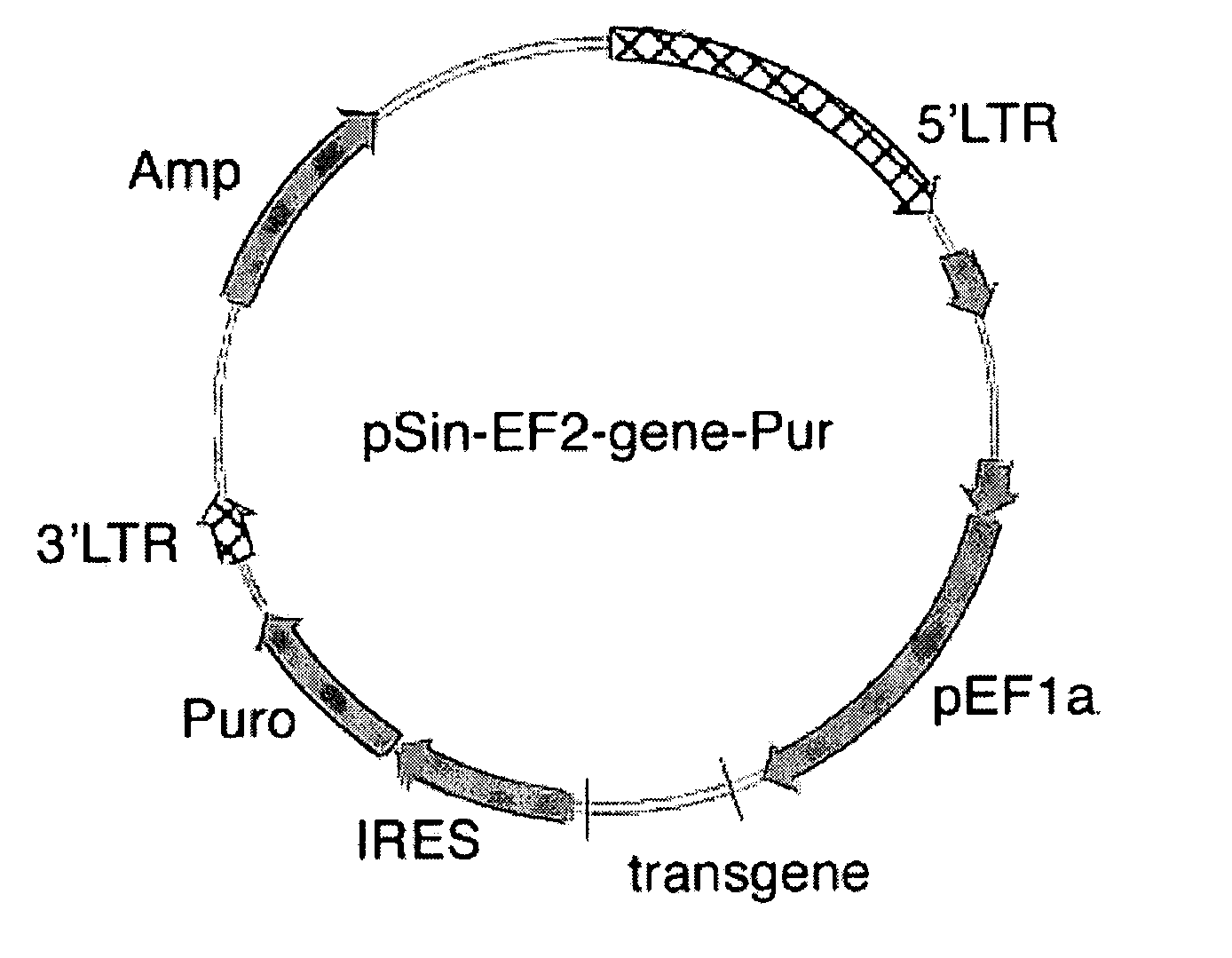
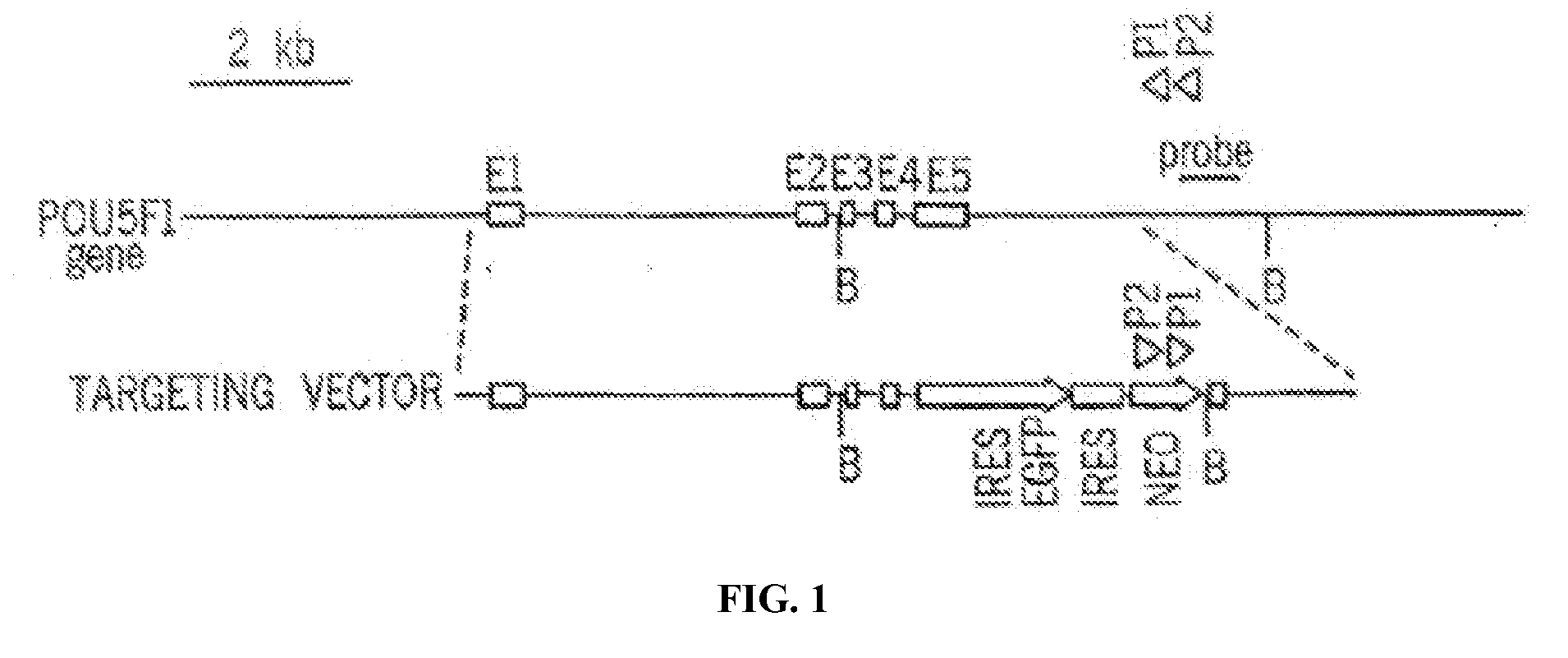
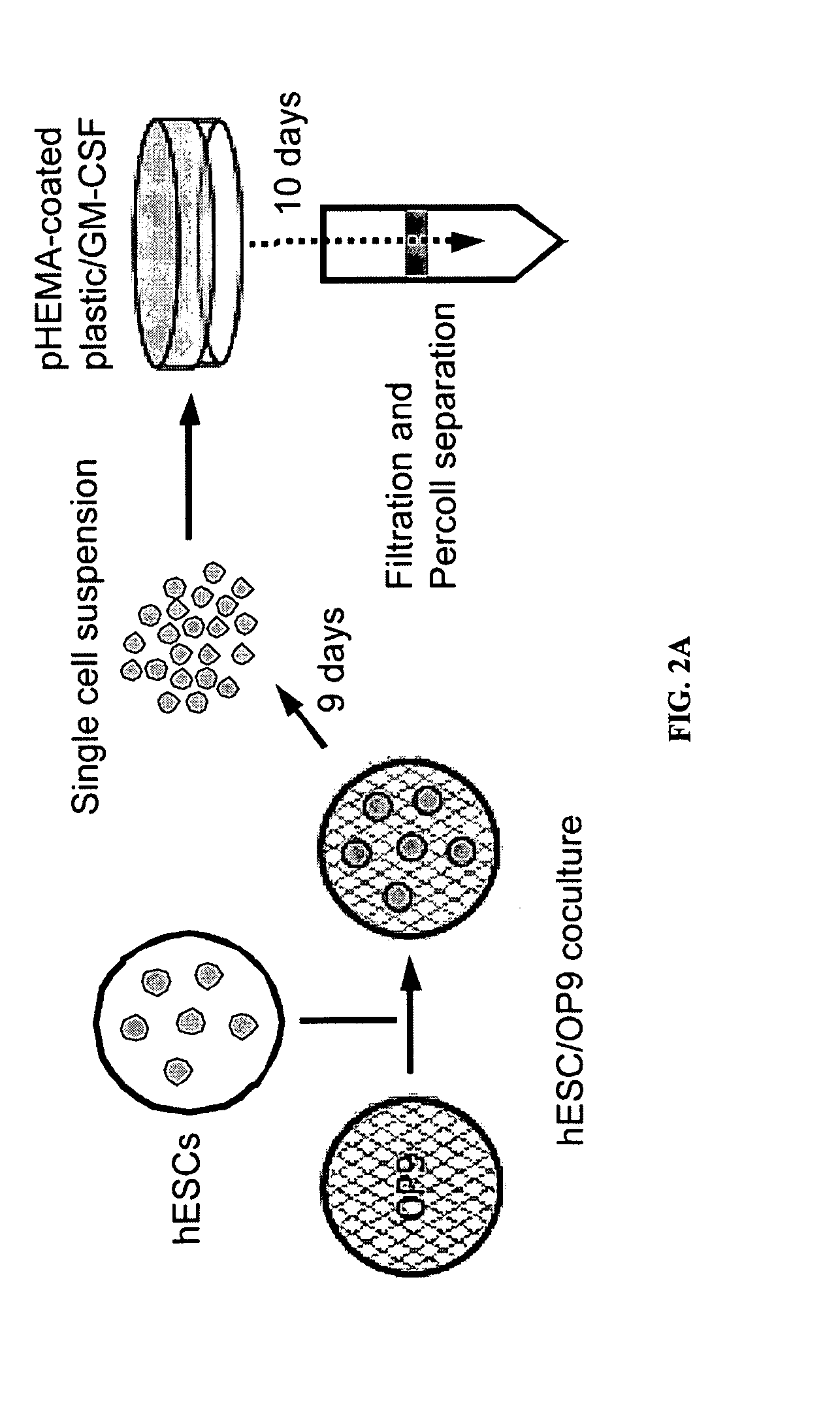
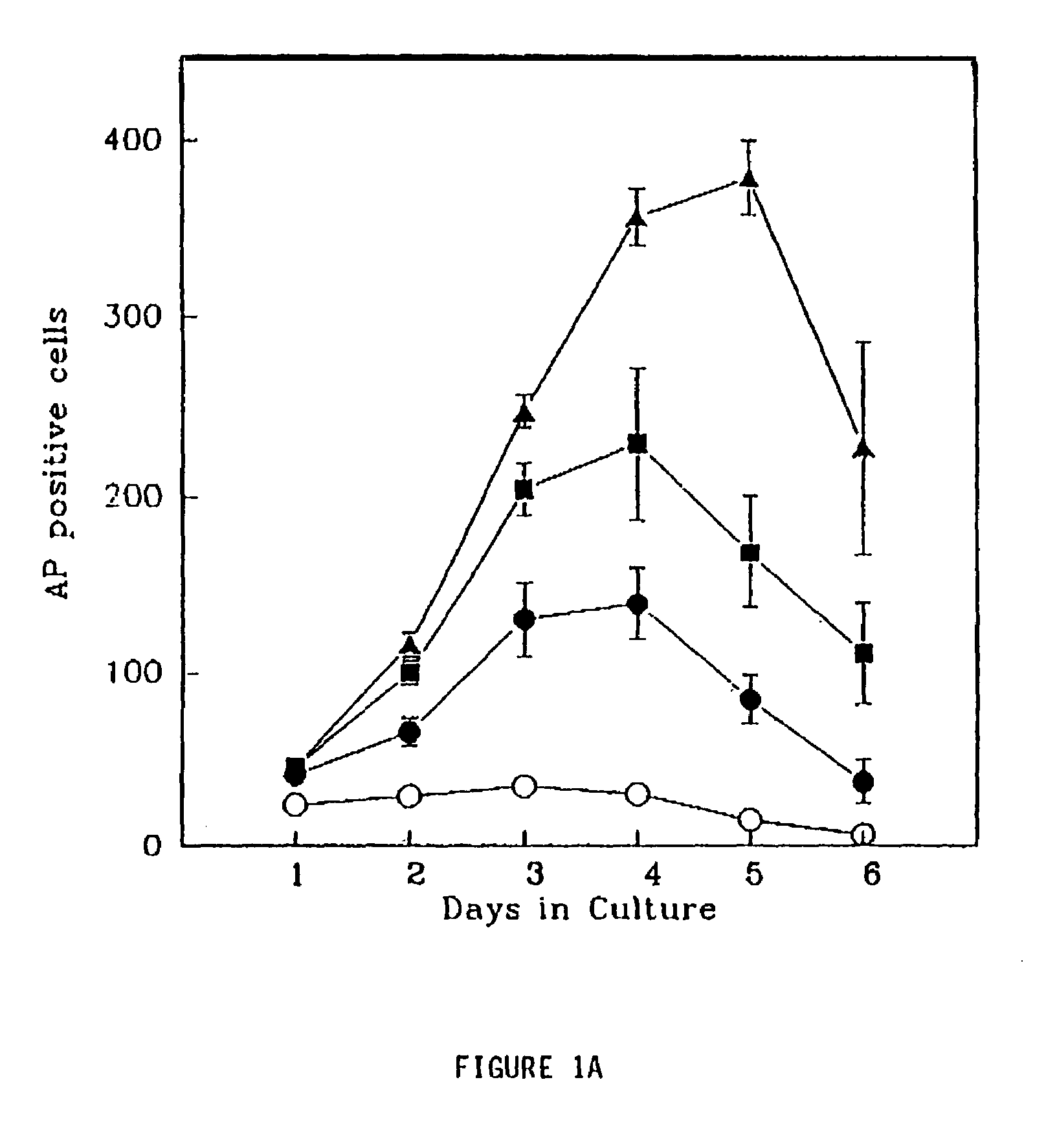
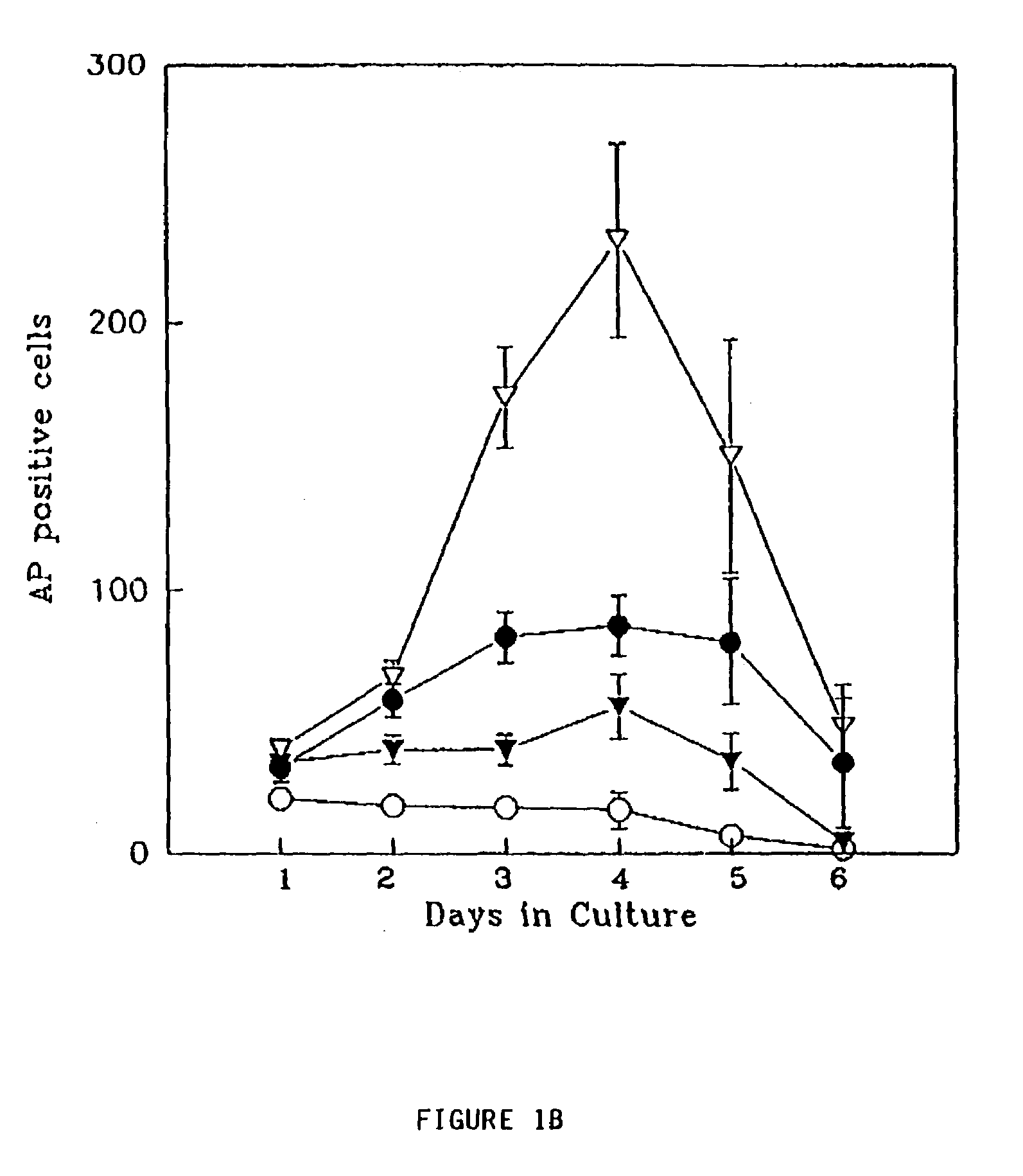
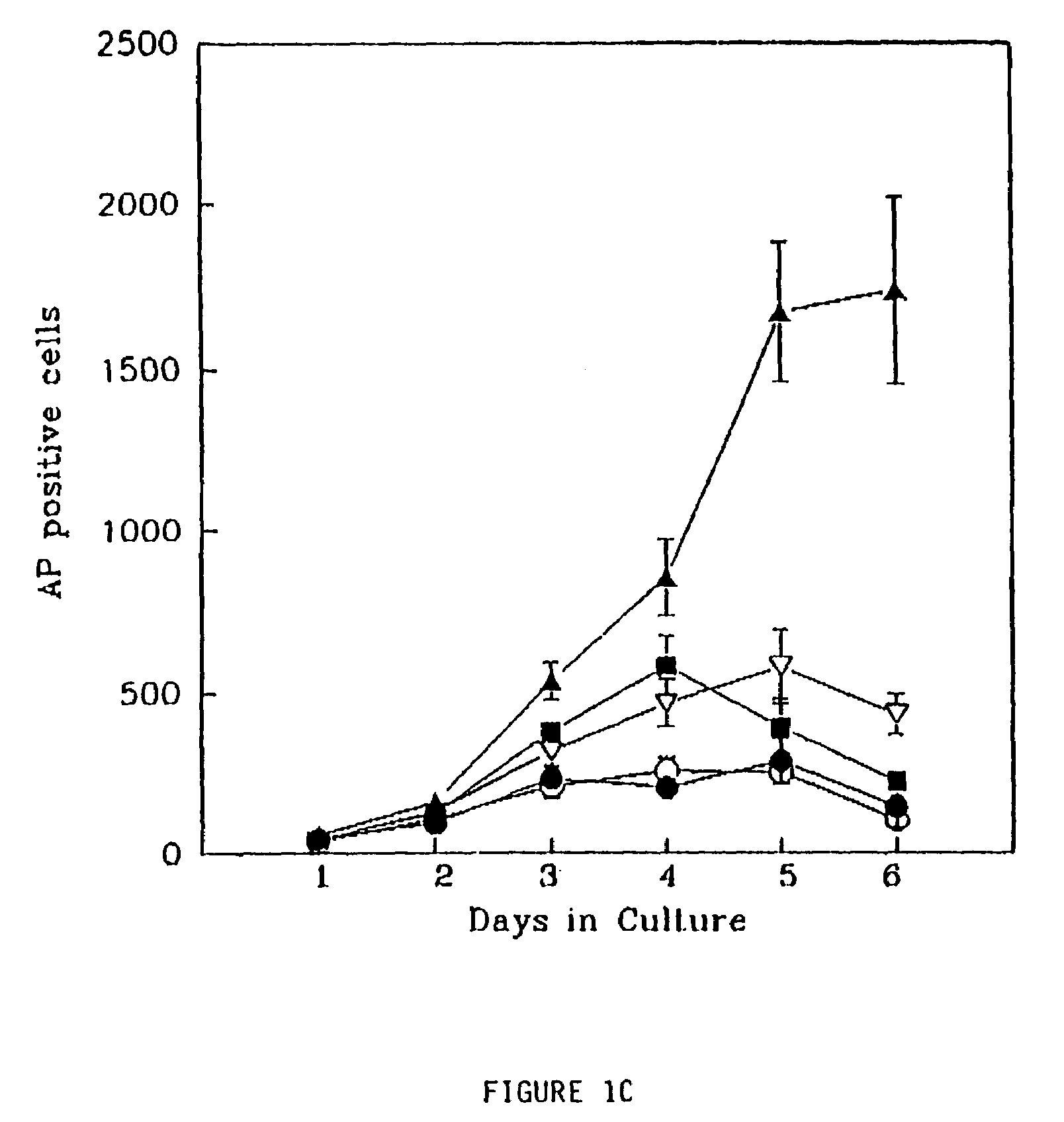
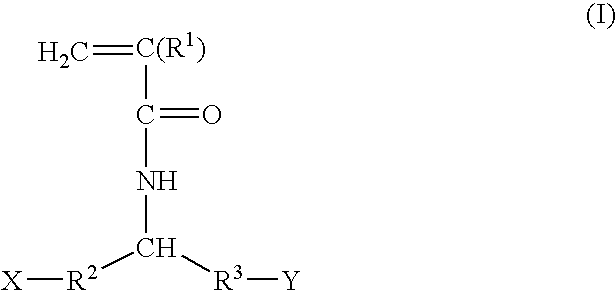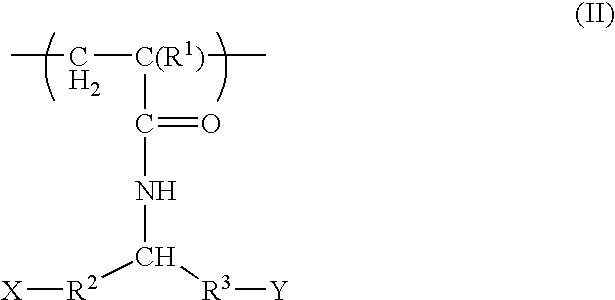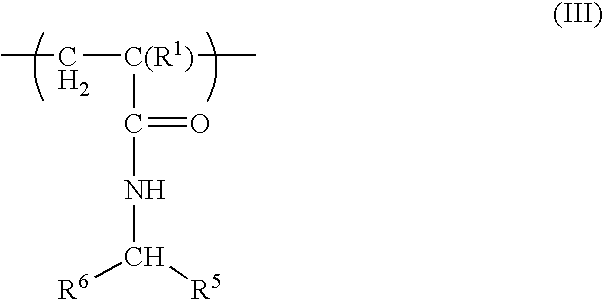Patents
Literature
3934results about "Artificially induced pluripotent cells" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Defined media for stem cell culture
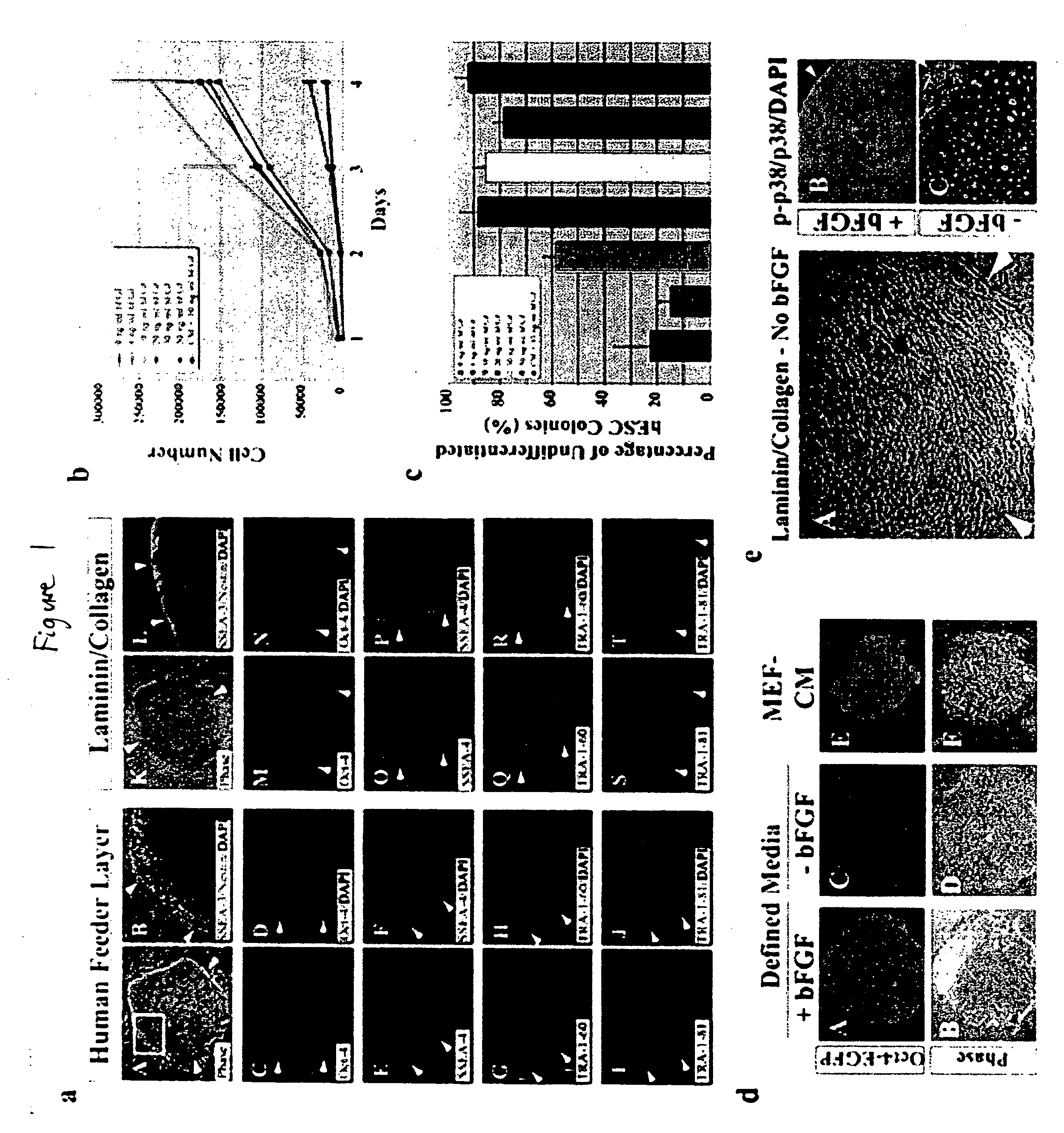
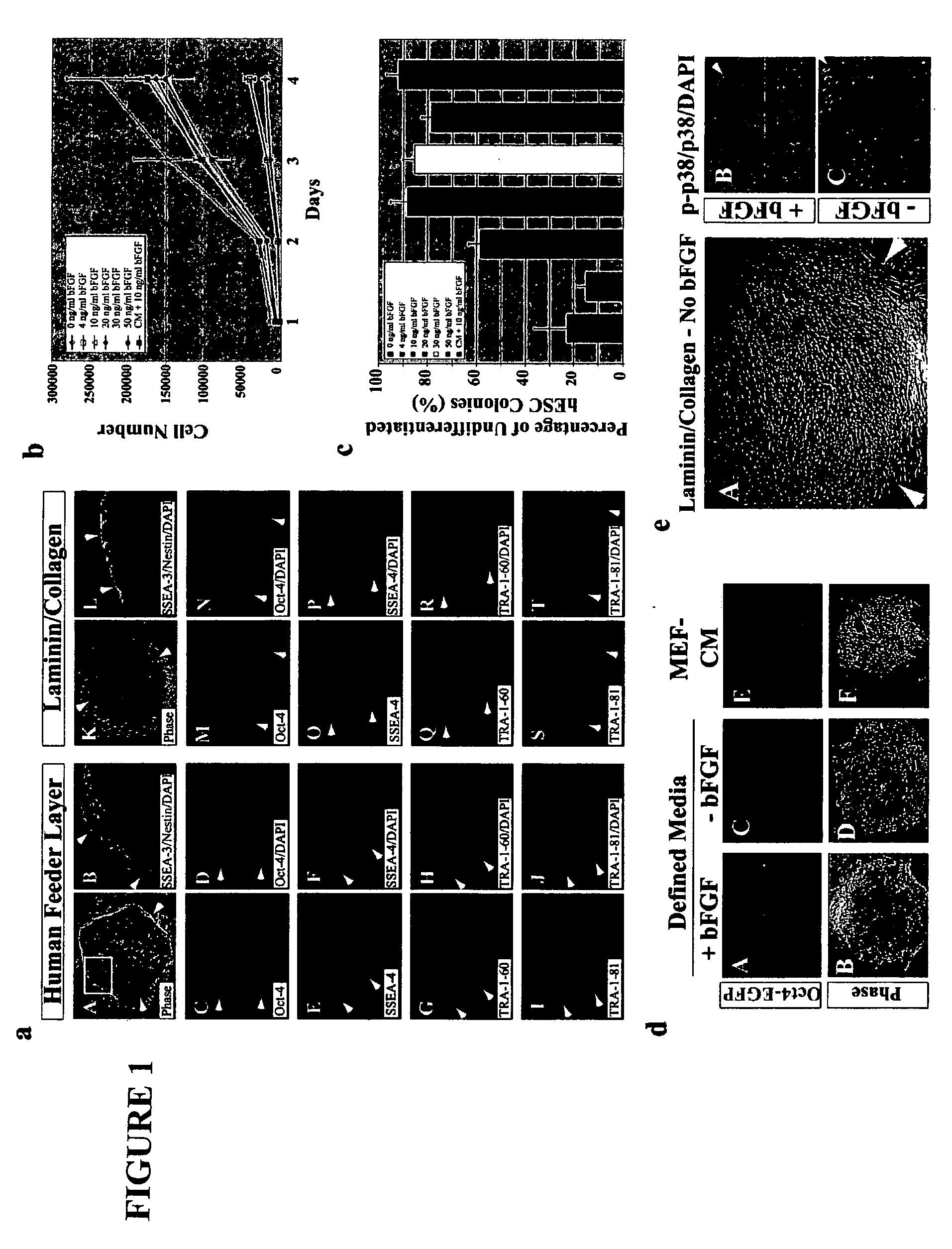
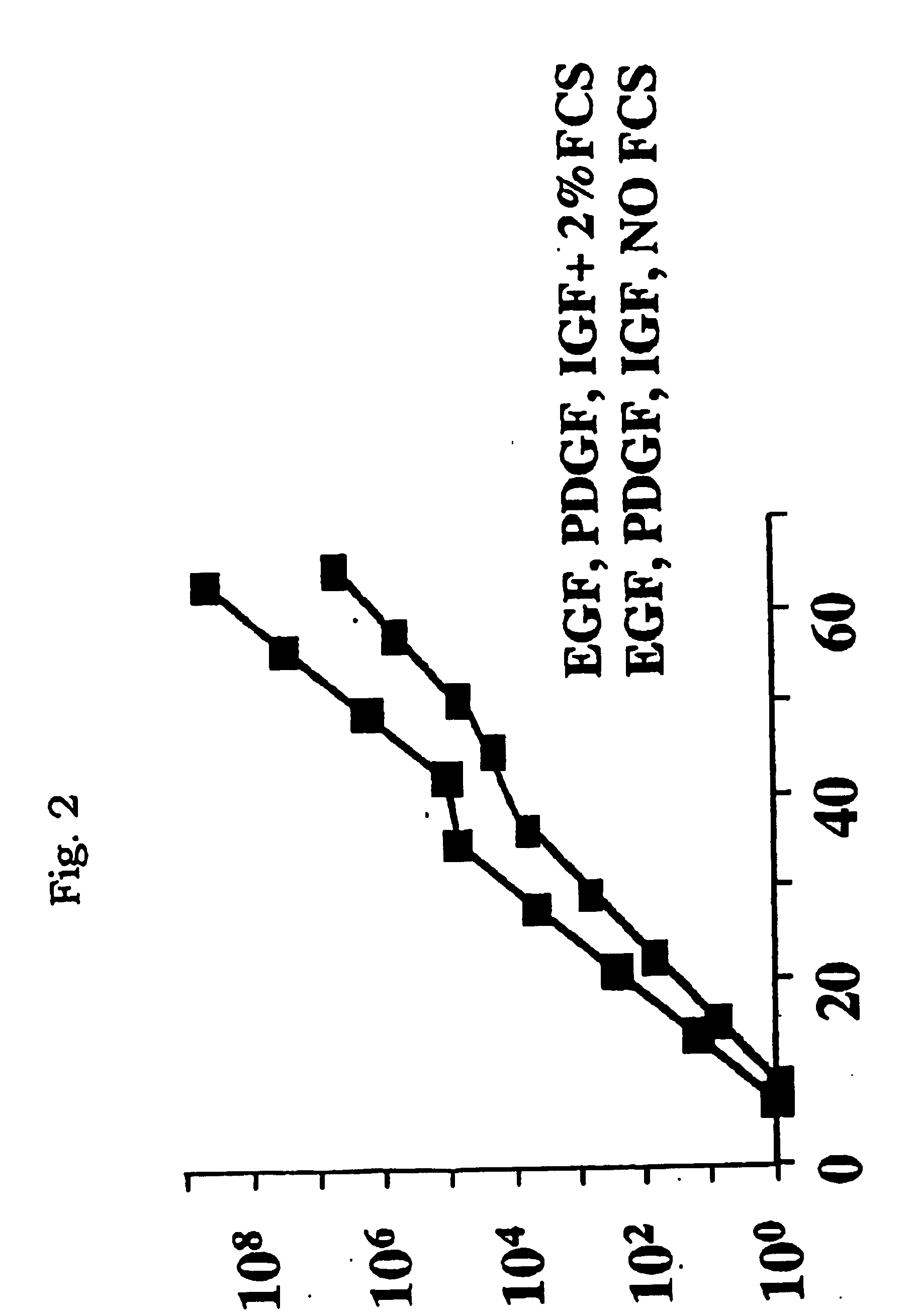
Stem cells, including mammalian, and particularly primate primordial stem cells (pPSCs) such as human embryonic stem cells (hESCs), hold great promise for restoring cell, tissue, and organ function. However, cultivation of stem cells, particularly undifferentiated hESCs, in serum-free, feeder-free, and conditioned-medium-free conditions remains crucial for large-scale, uniform production of pluripotent cells for cell-based therapies, as well as for controlling conditions for efficiently directing their lineage-specific differentiation. This instant invention is based on the discovery of the formulation of minimal essential components necessary for maintaining the long-term growth of pPSCs, particularly undifferentiated hESCs. Basic fibroblast growth factor (bFGF), insulin, ascorbic acid, and laminin were identified to be both sufficient and necessary for maintaining hESCs in a healthy self-renewing undifferentiated state capable of both prolonged propagation and then directed differentiation. Having discerned these minimal molecular requirements, conditions that would permit the substitution of poorly-characterized and unspecified biological additives and substrates were derived and optimized with entirely defined constituents, providing a “biologics”-free (i.e., animal-, feeder-, serum-, and conditioned-medium-free) system for the efficient long-term cultivation of pPSCs, particularly pluripotent hESCs. Such culture systems allow the derivation and large-scale production of stem cells such as pPSCs, particularly pluripotent hESCs, in optimal yet well-defined biologics-free culture conditions from which they can be efficiently directed towards a lineage-specific differentiated fate in vitro, and thus are important, for instance, in connection with clinical applications based on stem cell therapy and in drug discovery processes.
Owner:THE BURNHAM INST
RNA preparations comprising purified modified RNA for reprogramming cells
ActiveUS20110143397A1Promote growthArtificial cell constructsCell culture active agentsSingle strandSomatic cell
The present invention provides compositions and methods for reprogramming somatic cells using purified RNA preparations comprising single-strand mRNA encoding an iPS cell induction factor. The purified RNA preparations are preferably substantially free of RNA contaminant molecules that: i) would activate an immune response in the somatic cells, ii) would decrease expression of the single-stranded mRNA in the somatic cells, and / or iii) active RNA sensors in the somatic cells. In certain embodiments, the purified RNA preparations are substantially free of partial mRNAs, double-stranded RNAs, un-capped RNA molecules, and / or single-stranded run-on mRNAs.
Owner:THE TRUSTEES OF THE UNIV OF PENNSYLVANIA
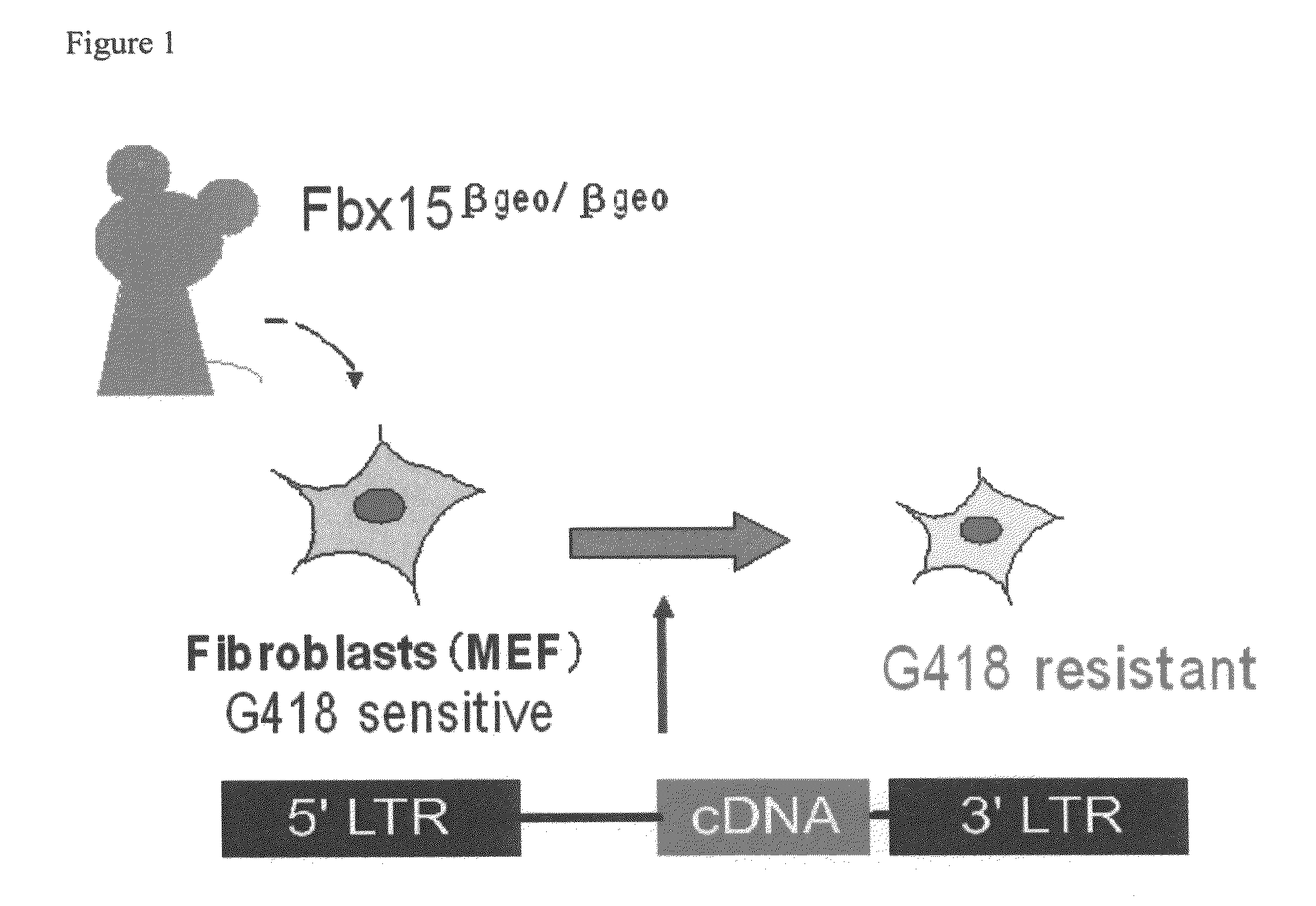
Nuclear Reprogramming Factor
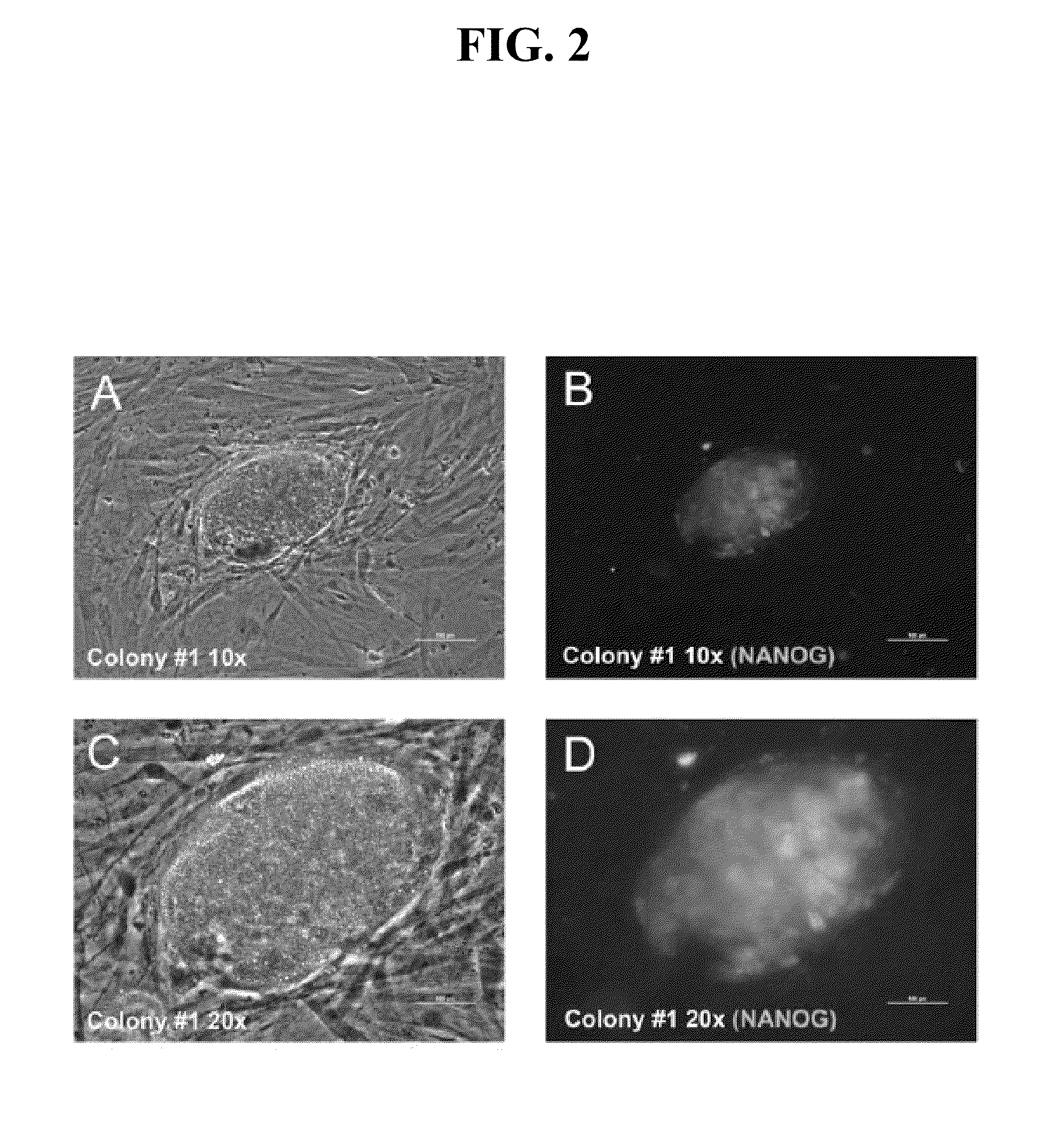
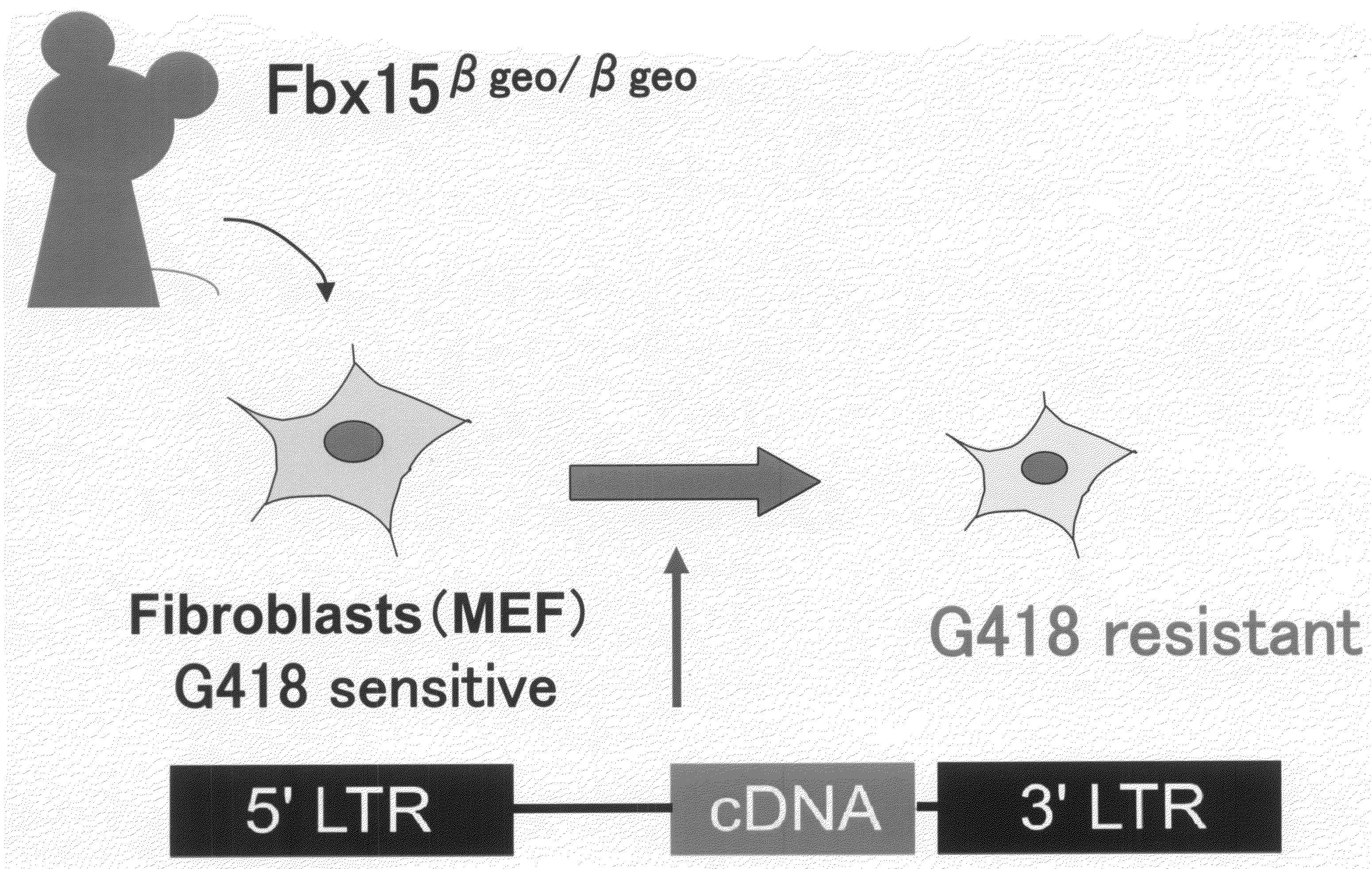
There is provided a nuclear reprogramming factor for a somatic cell, which comprises a gene product of each of the following three kinds of genes: an Oct family gene, a Klf family gene, and a Myc family gene, as a means for inducing reprogramming of a differentiated cell to conveniently and highly reproducibly establish an induced pluripotent stem cell having pluripotency and growth ability similar to those of ES cells without using embryo or ES cell.
Owner:KYOTO UNIV
Embryonic-like stem cells derived from post-partum mammalian placenta, and uses and methods of treatment using said cells
Owner:CELULARITY INC
Methods of using JNK or MKK inhibitors to modulate cell differentiation and to treat myeloproliferative disorders and myelodysplastic syndromes
InactiveUS20040028660A1Increase productionPromote recoveryBiocideGenetic material ingredientsCord blood stem cellMyeloproliferative Disorders
The present invention provides methods of modulating mammalian, particularly human, stem cell and progenitor cell differentiation to regulate and control the differentiation and maturation of these cells along specific cell and tissue lineages. The methods of the invention relate to the use of certain small organic molecules to modulate the differentiation of stem cell populations along specific cell and tissue lineages, particularly embryonic-like stem cells originating from a postpartum placenta or stem cells isolated form sources such as cord blood. The invention also relates to the treatment or prevention of myelodysplastic syndrome or myeloproliferative syndrome, or symptoms thereof, comprising administration of JNK or MKK inhibitors, alone or in combination, as well as with or without the use of unconditioned cells or cells conditioned in accordance with other aspects of the invention. Finally, the invention relates to the use of such differentiated stem cells in transplantation and other medical treatments.
Owner:ANTHROGENESIS CORP +1
Multipotent amniotic fetal stem cells
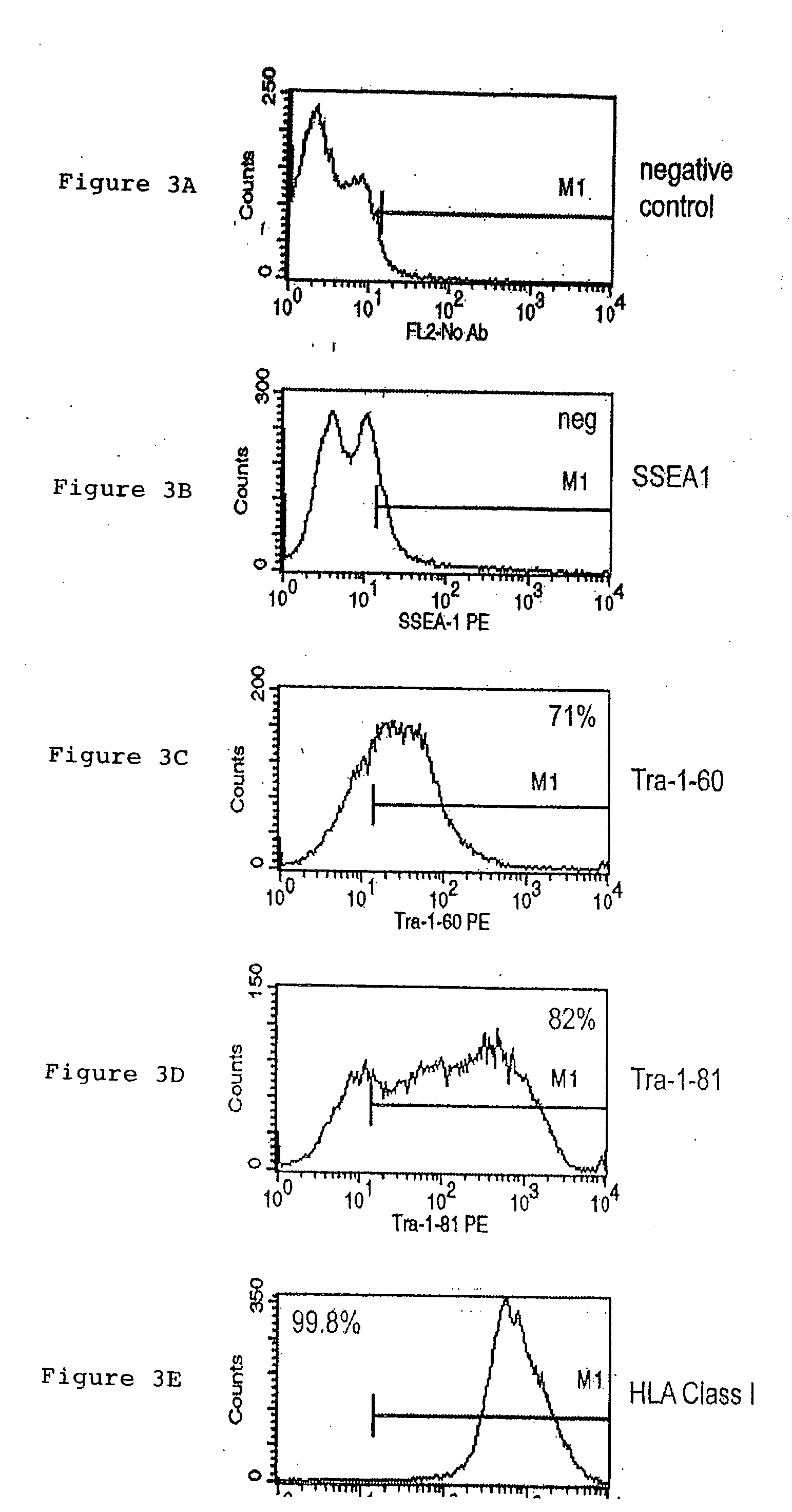
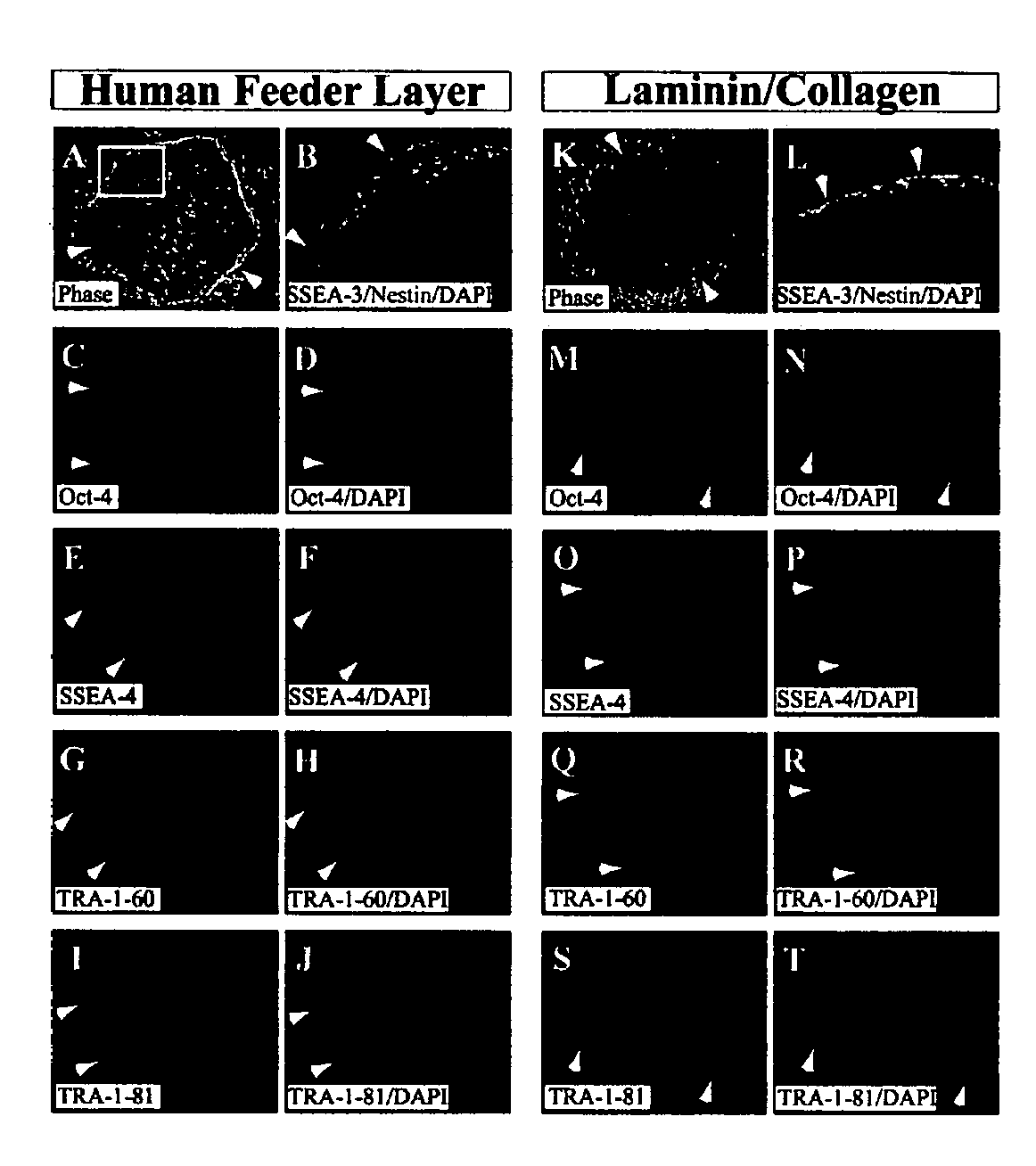
A source of multipotent amniotic fluid / fetal stem cells (MAFSCs) is disclosed. MAFSC are of fetal origin and have a normal diploid karyotype. These cells are characterized by the following cell surface markers: SSEA3, SSEA4, Tra-1-60, Tra-1-81, Tra-2-54, HLA class I, CD13, CD44, CD49b, CD105 and are distinguished by the absence of the antigen markers CD34, CD45, and HLA Class II, but are distinguished from mouse embryonic stem cells in that these cells do not express the cell surface marker SSEA1. MAFSC express the stem cell transcription factor Oct-4. MAFSC cells can be propagated for an indefinite period of time in continuous culture in an undifferentiated state. The MAFSCs have the ability to differentiate in culture in a regulated manner, into three or more subphenotypes. Cells can then be differentiated into endodermal, mesodermal and ectodermal derived tissues in vitro and in vivo. A method for isolating, identifying, expanding and differentiating MAFSCs is disclosed.
Owner:RGT UNIV OF CALIFORNIA
Methods and compositions for the treatment and management of hemoglobinopathy and anemia
InactiveUS20050143420A1Good effectRelieve symptomsBiocidePeptide/protein ingredientsRed blood cellThalassemia
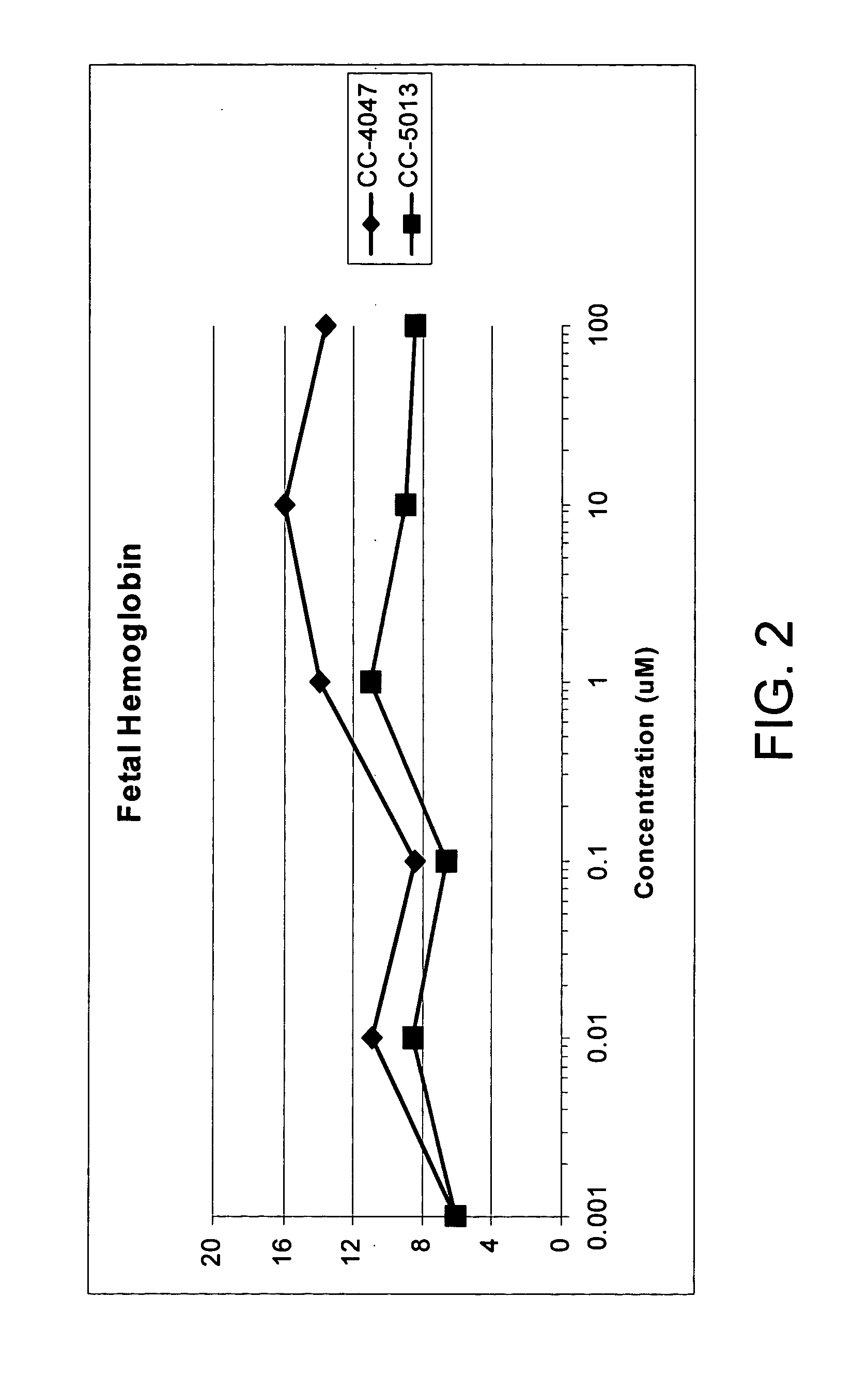
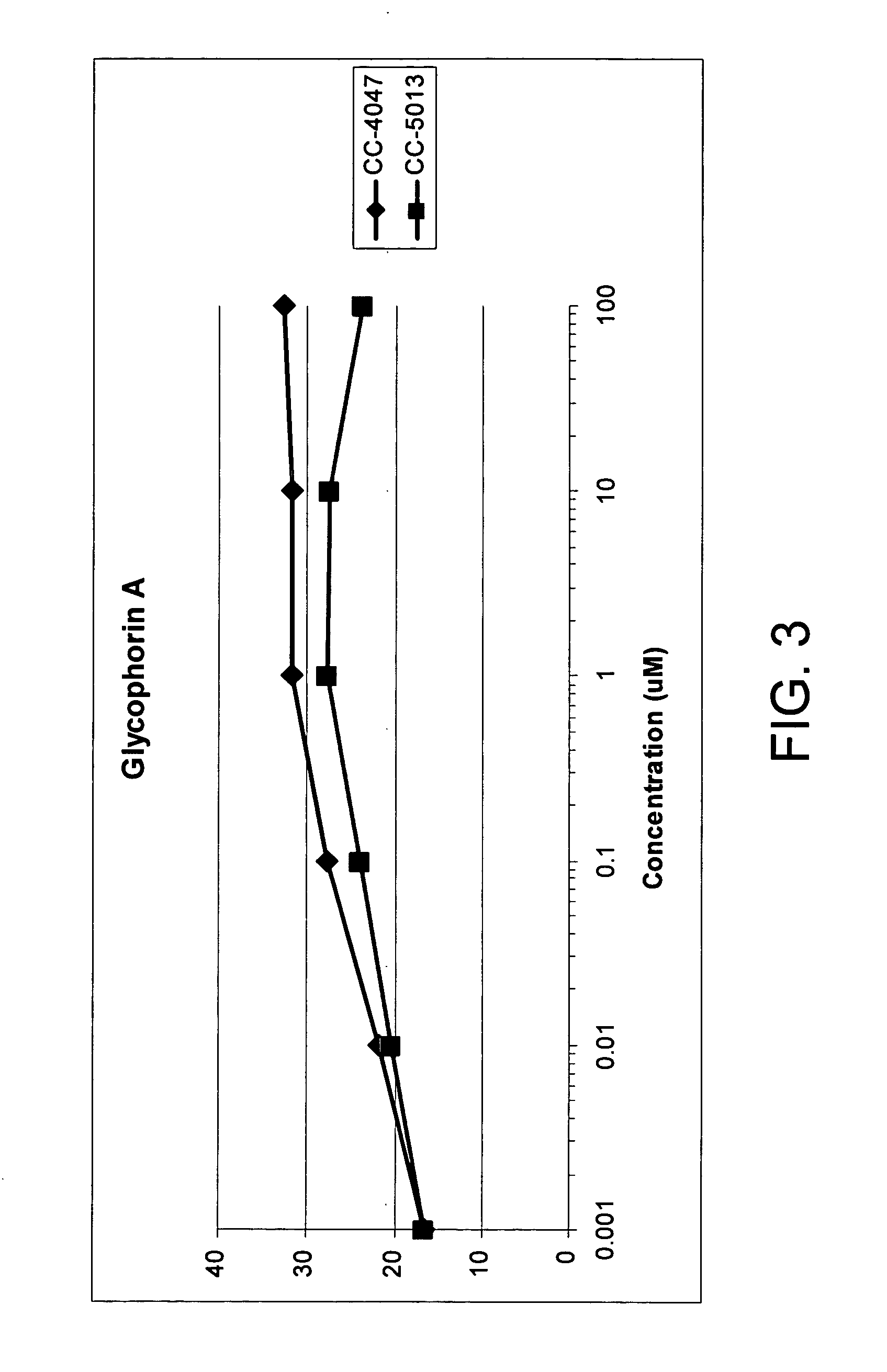
The present invention is directed to the use of immunomodulatory compounds, particularly members of the class of compounds known as IMiDs™, and more specifically the compounds 4-(Amino)-2-(2,6-dioxo(3-piperidyl))-isoindoline-1,3-dione and 3-(4-amino-1-oxo-1,3-dihydroisoindol-2-yl)-piperidine-2,6-dione, to induce the expression of fetal hemoglobin genes, genes essential for erythropoiesis, and genes encoding alpha hemoglobin stabilizing protein, within a population of CD34+ cells. These compounds are used to treat hemoglobinopathies such as sickle cell anemia or β-thalassemia, or anemias caused by disease, surgery, accident, or the introduction or ingestion of toxins, poisons or drugs.
Owner:SIGNAL PHARMA LLC
Defined media for pluripotent stem cell culture
Stem cells, including mammalian, and particularly primate primordial stem cells (pPSCs) such as human embryonic stem cells (hESCs), hold great promise for restoring cell, tissue, and organ function. However, cultivation of stem cells, particularly undifferentiated hESCs, in serum-free, feeder-free, and conditioned-medium-free conditions remains crucial for large-scale, uniform production of pluripotent cells for cell-based therapies, as well as for controlling conditions for efficiently directing their lineage-specific differentiation. This instant invention is based on the discovery of the formulation of minimal essential components necessary for maintaining the long-term growth of pPSCs, particularly undifferentiated hESCs. Basic fibroblast growth factor (bFGF), insulin, ascorbic acid, and laminin were identified to be both sufficient and necessary for maintaining hESCs in a healthy self-renewing undifferentiated state capable of both prolonged propagation and then directed differentiation. Having discerned these minimal molecular requirements, conditions that would permit the substitution of poorly-characterized and unspecified biological additives and substrates were derived and optimized with entirely defined constituents, providing a “biologics”-free (i.e., animal-, feeder-, serum-, and conditioned-medium-free) system for the efficient long-term cultivation of pPSCs, particularly pluripotent hESCs. Such culture systems allow the derivation and large-scale production of stem cells such as pPSCs, particularly pluripotent hESCs, in optimal yet well-defined biologics-free culture conditions from which they can be efficiently directed towards a lineage-specific differentiated fate in vitro, and thus are important, for instance, in connection with clinical applications based on stem cell therapy and in drug discovery processes.
Owner:THE BURNHAM INST
Nuclear reprogramming factor and induced pluripotent stem cells
InactiveUS20090227032A1Easy to prepareEffective isolationGenetically modified cellsPeptidesNuclear reprogrammingCell therapy
The present invention relates to a nuclear reprogramming factor having an action of reprogramming a differentiated somatic cell to derive an induced pluripotent stem (iPS) cell. The present invention also relates to the aforementioned iPS cells, methods of generating and maintaining iPS cells, and methods of using iPS cells, including screening and testing methods as well as methods of stem cell therapy. The present invention also relates to somatic cells derived by inducing differentiation of the aforementioned iPS cells.
Owner:KYOTO UNIV
Isolation, cultivation and uses of stem/progenitor cells
The present invention relates to a method for isolating stem / progenitor cells from the amniotic membrane of umbilical cord, wherein the method comprises separating the amniotic membrane from the other components of the umbilical cord in vitro, culturing the amniotic membrane tissue under conditions allowing cell proliferation, and isolating the stem / progenitor cells from the tissue cultures. The isolated stem cell cells can have embryonic stem cell-like properties and can be used for various therapeutic purposes. In one embodiment, the invention relates to the isolation and cultivation of stem cells such as epithelial and / or mesenchymal stem / progenitor cells under conditions allowing the cells to undergo mitotic expansion. Furthermore, the invention is directed to a method for the differentiation of the isolated stem / progenitor cells into epithelial and / or mesenchymal cells.
Owner:CELLRESEARCH CORP PTE LTD
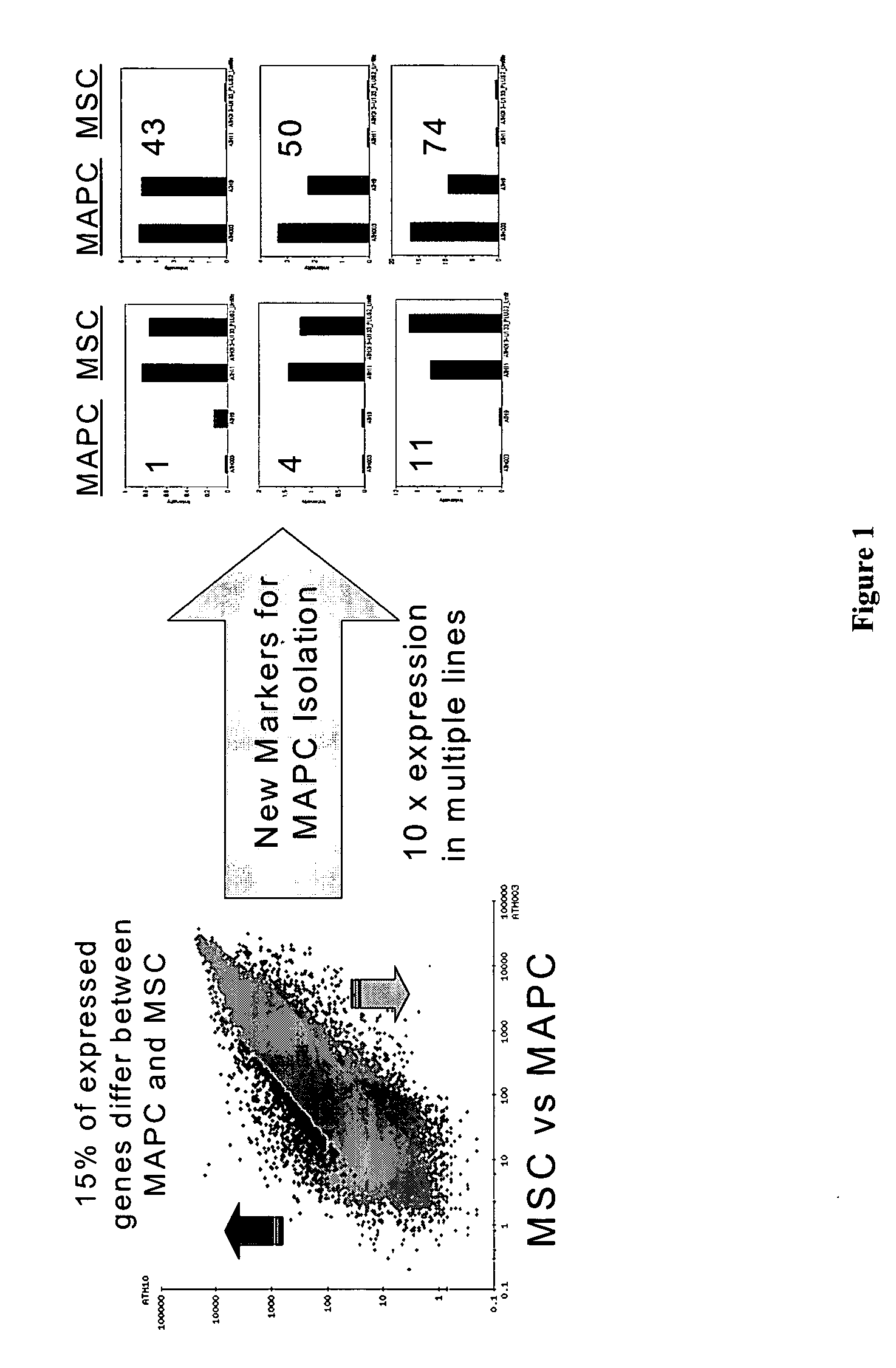
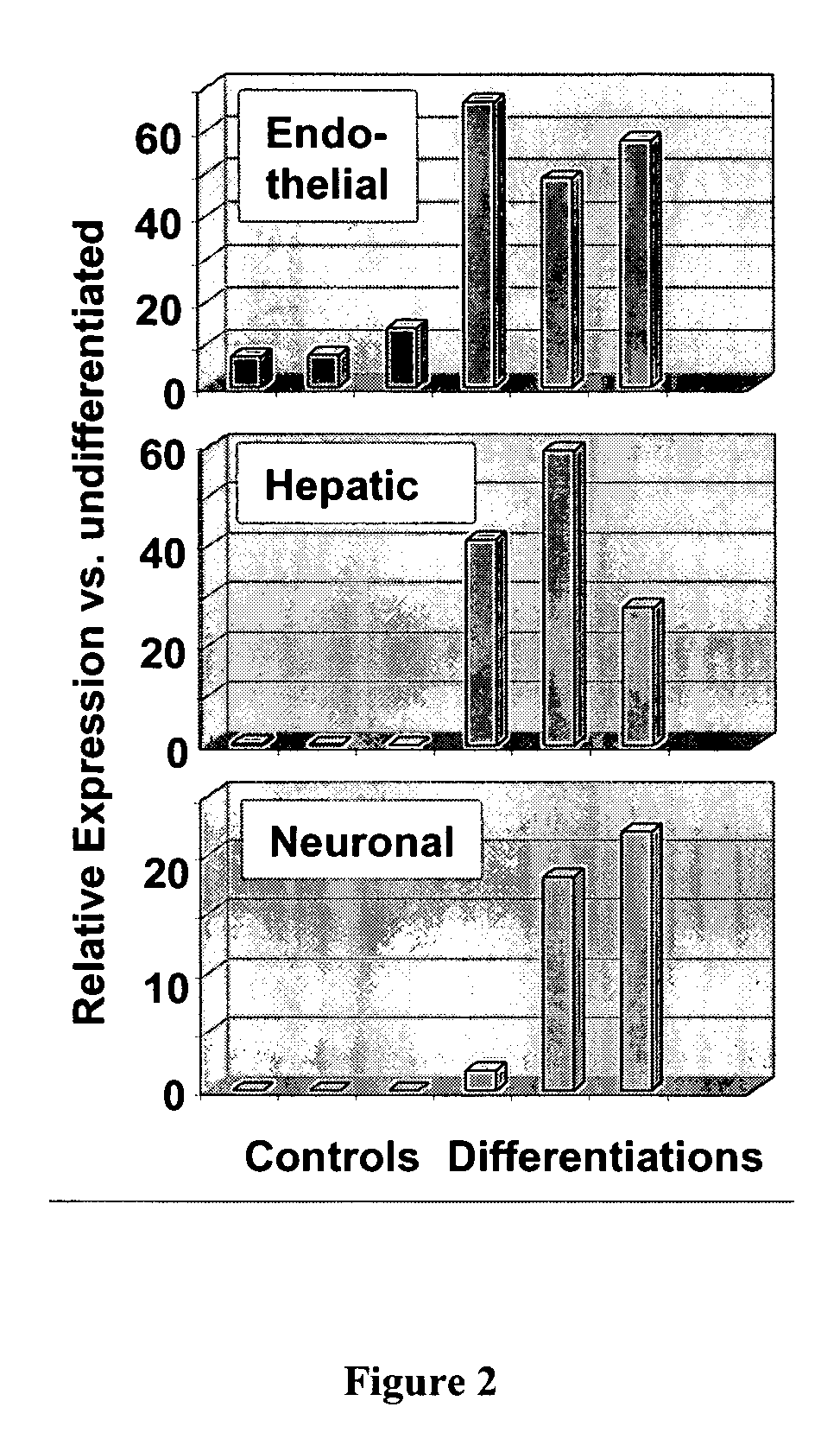
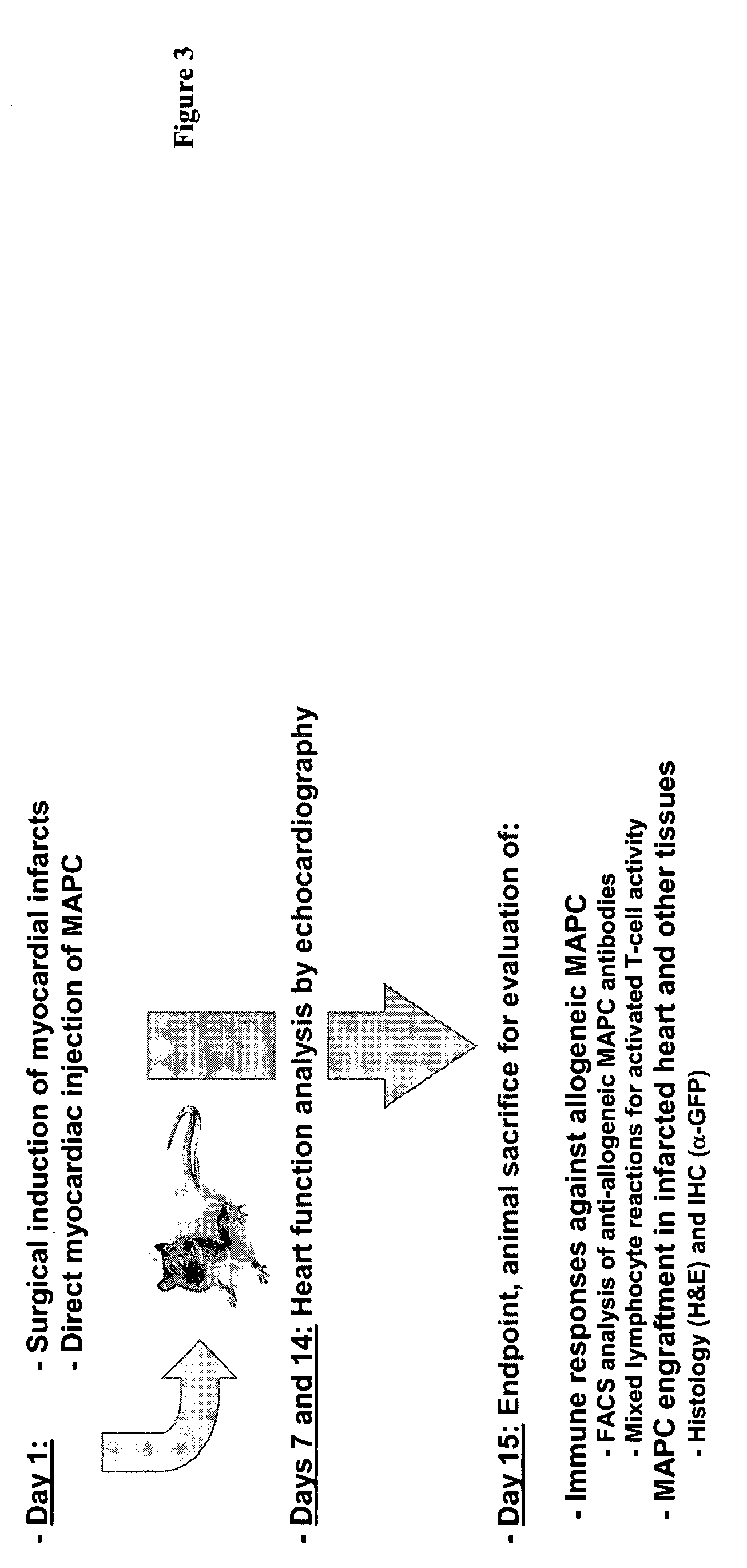
Use of multipotent adult stem cells in treatment of myocardial infarction and congestive heart failure
ActiveUS20060008450A1Restore and enhance cardiac muscle functionRapid responseBiocideGenetic material ingredientsTissues typesEmbryo
Owner:RGT UNIV OF MINNESOTA
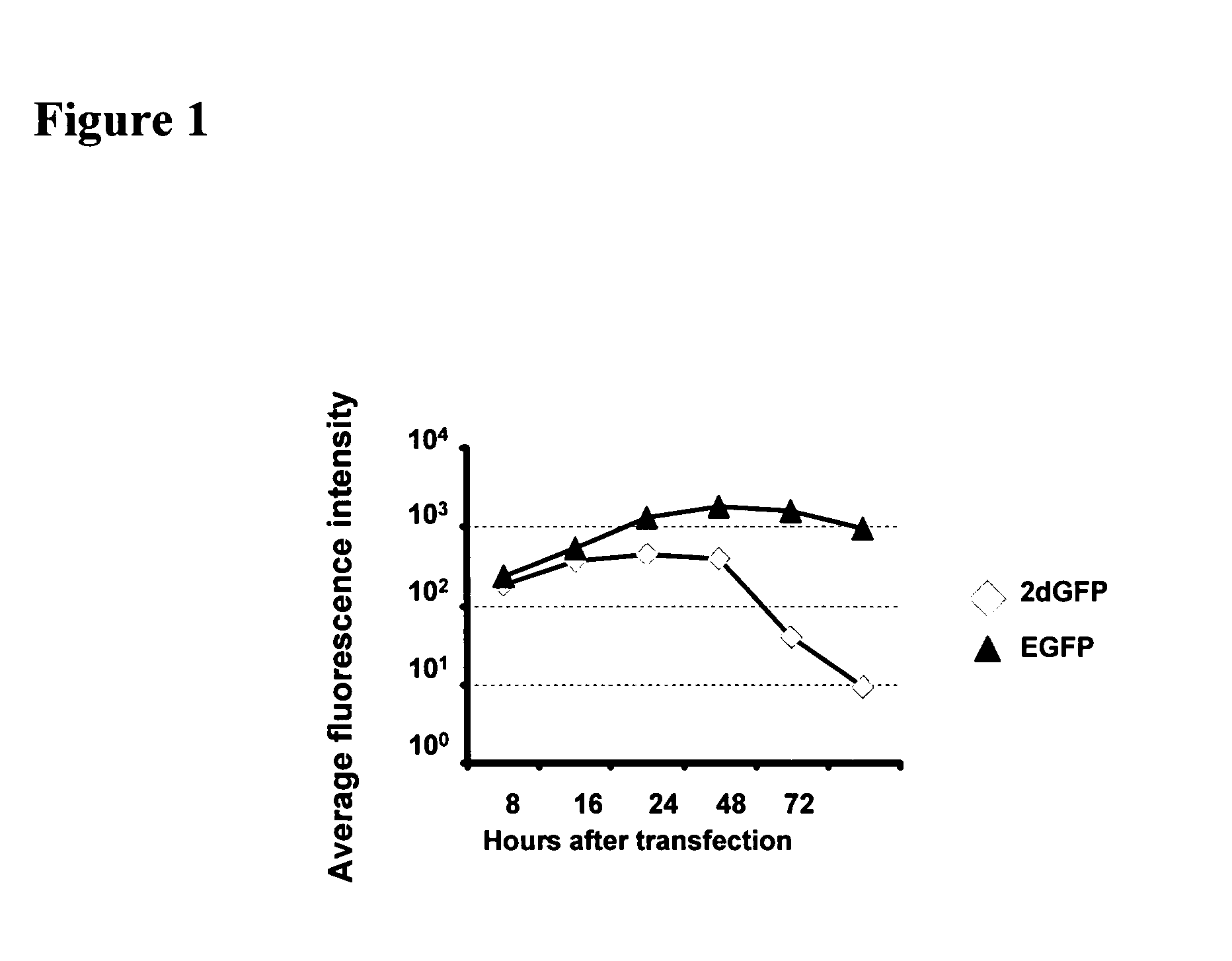
Use of RNA for reprogramming somatic cells
InactiveUS20110065103A1Promotes reprogrammingBlood/immune system cellsDrug compositionsReprogrammingSomatic cell
The present invention provides methods for de-differentiating somatic cells into stem-like cells without generating embryos or fetuses. More specifically, the present invention provides methods for effecting the de-differentiation of somatic cells to cells having stem cell characteristics, in particular pluripotency, by introducing RNA encoding factors inducing the de-differentiation of somatic cells into the somatic cells and culturing the somatic cells allowing the cells to de-differentiate.
Owner:BIONTECH AG +2
Culturing human embryonic stem cells in medium containing pipecholic acid and gamma amino butyric acid
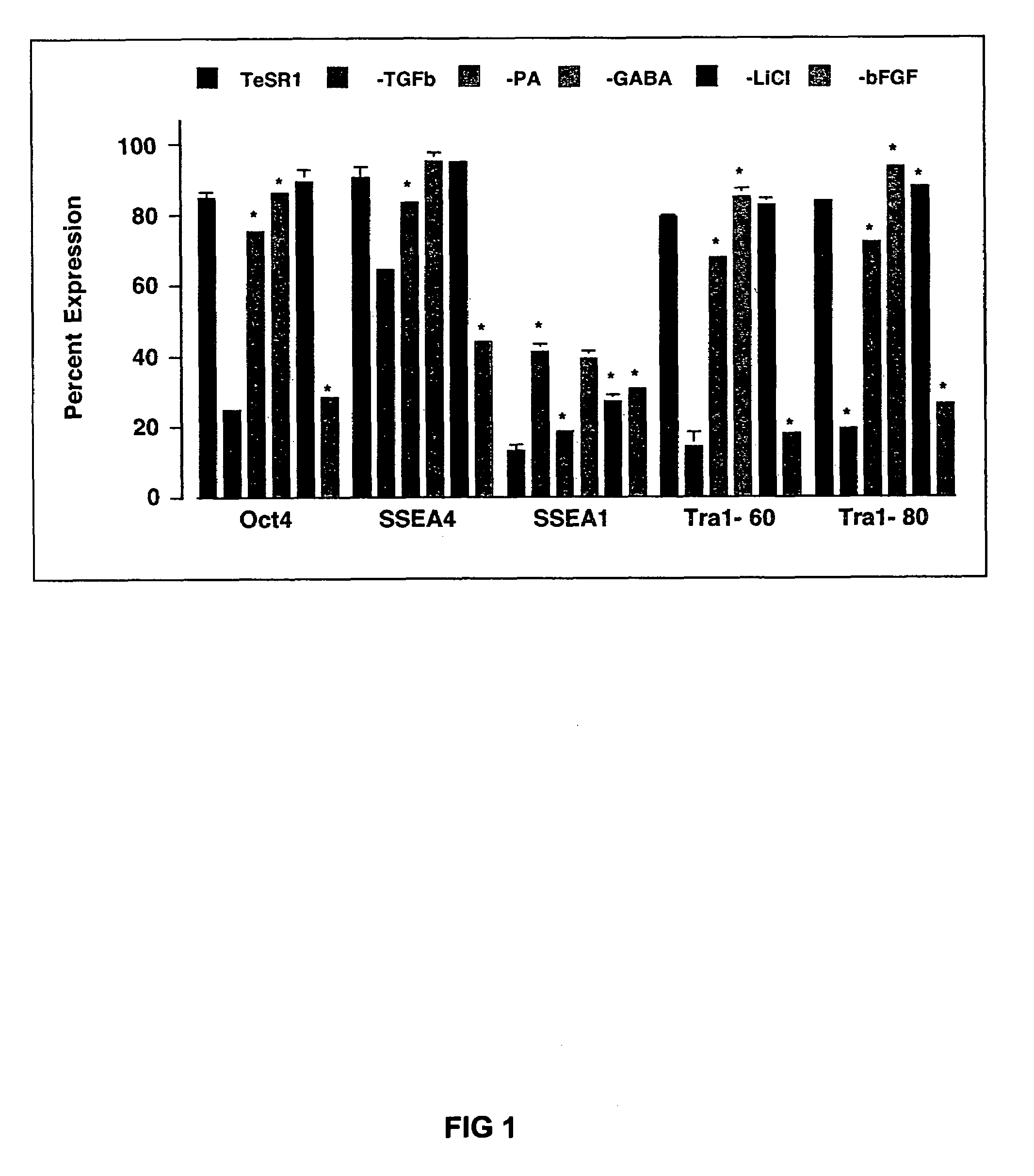
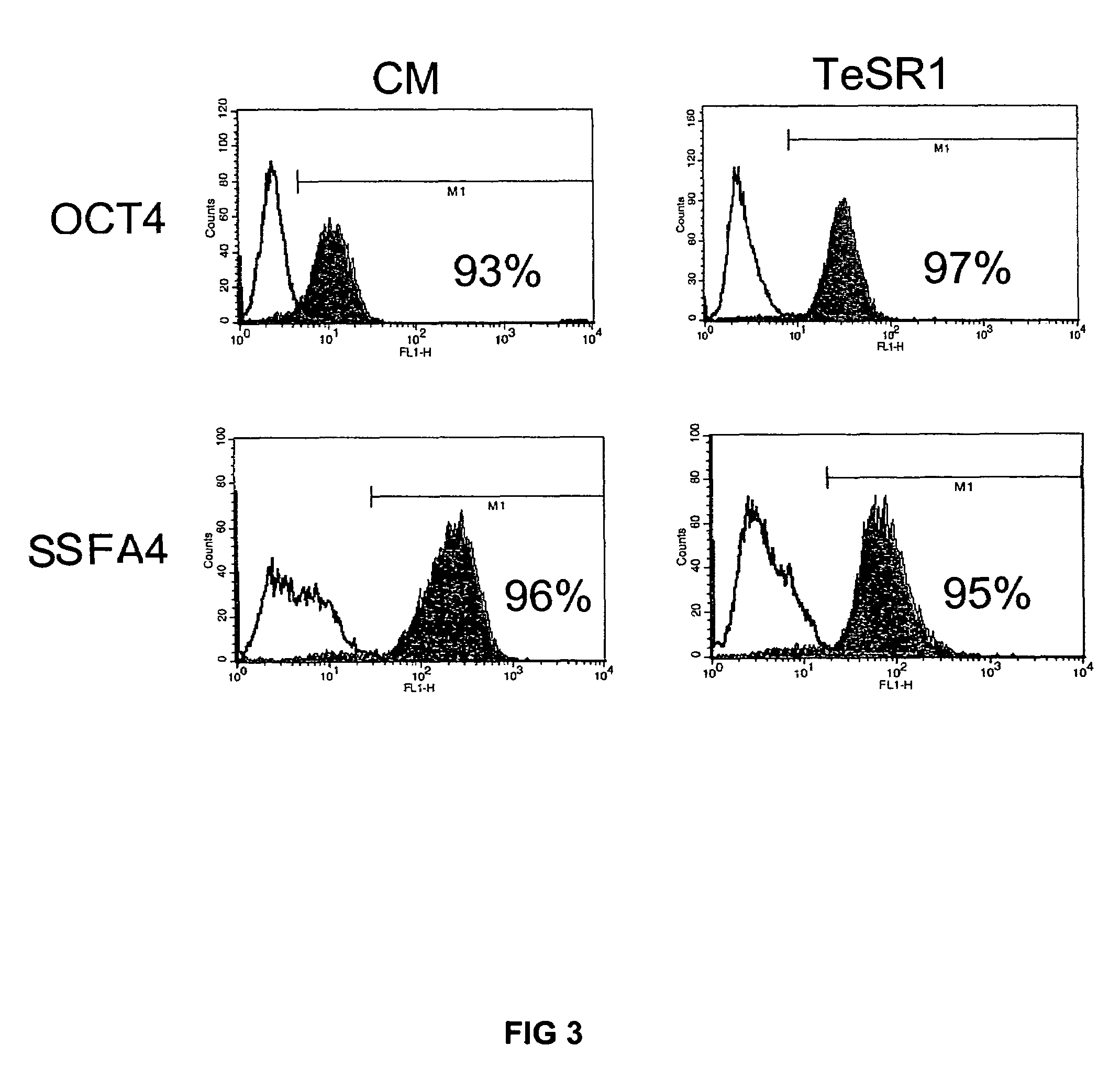
Previous methods for culturing human embryonic stem cells have required either fibroblast feeder cells or a medium which has been exposed to fibroblast feeder cells in order to maintain the stem cells in an undifferentiated state. It has now been found that if high levels of fibroblast growth factor are used in a medium with gamma amino butyric acid, pipecholic acid, lithium and lipids, the stem cells will remain undifferentiated indefinitely through multiple passages, even without feeder cells or conditioned medium. A humanized matrix of human proteins can be used as a basement matrix to culture the cells. New lines of human embryonic stem cells made using these culture conditions, the medium and the matrix, will never have been exposed to animal cells, animal products, feeder cells or conditioned medium.
Owner:WISCONSIN ALUMNI RES FOUND
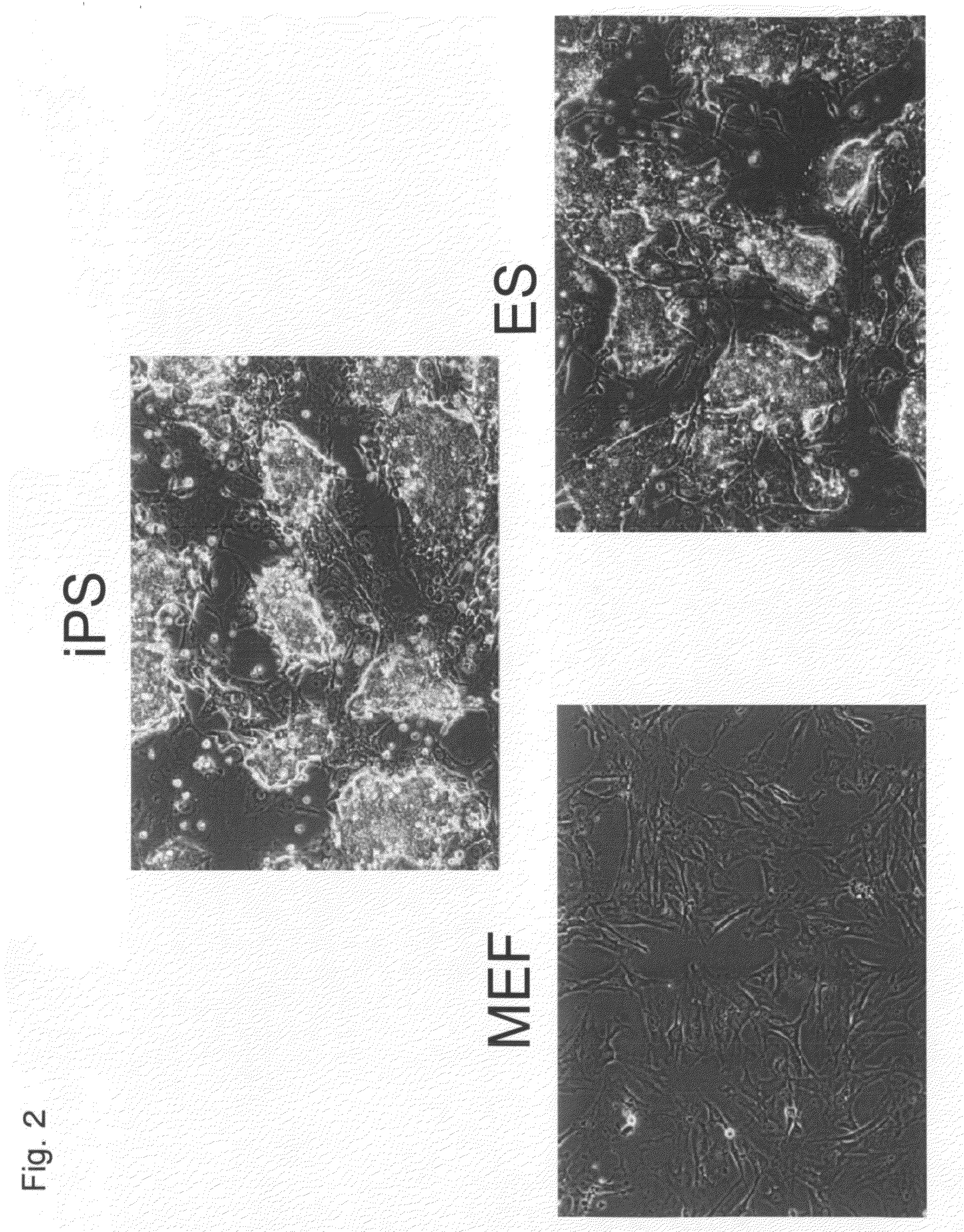
Somatic cell reprogramming
ActiveUS20080233610A1High nucleus to cytoplasm ratioImprove effectivenessNervous disorderVirusesSomatic cellCell biology
The present invention relates to methods for reprogramming a somatic cell to pluripotency by administering into the somatic cell at least one or a plurality of potency-determining factors. The invention also relates to pluripotent cell populations obtained using a reprogramming method.
Owner:WISCONSIN ALUMNI RES FOUND
Pluripotential embryonic stem cells and methods of making same
The present invention provides a non-mouse, including human, pluripotential embryonic stem cell which can:(a) be maintained on feeder layers for at least 20 passages; and(b) give rise to embryoid bodies and multiple differentiated cell phenotypes in monolayer culture.The invention further provides a method of making a pluripotential embryonic stem cell comprising culturing germ cells and germ cell progenitors in a composition comprising a growth enhancing amount of basic fibroblast growth factor, leukemia inhibitory factor, membrane associated steel factor, and soluble steel factor to primordial germ cells under cell growth conditions, thereby making a pluripotential embryonic stem cell.Also provided are compositions useful to produce the pluripotent embryonic stem cells and methods of screening associated with the method of making the embryonic stem cell.
Owner:VANDERBILT UNIV
Postpartum cells derived from placental tissue, and methods of making, culturing, and using the same
InactiveUS20060166361A1Artificial cell constructsCell culture supports/coatingPlacental tissueCell biology
Cells derived from postpartum placenta and methods for their isolation are provided by the invention. The invention further provides cultures and compositions of the placenta-derived cells. The placenta-derived cells of the invention have a plethora of uses, including but not limited to research, diagnostic, and therapeutic applications.
Owner:ETHICON INC
Treatment of stroke and other acute neuraldegenerative disorders using postpartum derived cells
InactiveUS20060233765A1BiocideMammal material medical ingredientsPhysiologyNeuro-degenerative disease
Cells derived from postpartum tissue such as the umbilical cord and placenta, and methods for their use to regenerate, repair, and improve neural tissue, and to improve behavior and neurological function in stroke patients are disclosed.
Owner:DEPUY SYNTHES PROD INC
Nuclear reprogramming factor
ActiveUS8048999B2Improve abilitiesPromote growthNervous disorderVirusesGene productNuclear reprogramming
There is provided a nuclear reprogramming factor for a somatic cell, which comprises a gene product of each of the following three kinds of genes: an Oct family gene, a Klf family gene, and a Myc family gene, as a means for inducing reprogramming of a differentiated cell to conveniently and highly reproducibly establish an induced pluripotent stem cell having pluripotency and growth ability similar to those of ES cells without using embryo or ES cell.
Owner:KYOTO UNIV
Isolation of bone marrow fraction rich in connective tissue growth components and the use thereof to promote connective tissue formation
InactiveUS20050130301A1Peptide/protein ingredientsSkeletal disorderConnective tissue fiberTissue defect
A bone marrow isolate rich in one or more connective tissue growth components, methods of forming the isolate, and methods of promoting connective tissue growth using the isolate are described. A biological sample comprising bone marrow is centrifuged to separate the sample into fractions including a fraction rich in connective tissue growth components. The fraction rich in connective tissue growth components is then isolated from the separated sample. The isolate can be used directly or combined with a carrier and implanted into a patient at a tissue (e.g., bone) defect site. The biological sample can comprise bone marrow and whole blood. The isolate can be modified (e.g., by transfection with a nucleic acid encoding an osteoinductive polypeptide operably linked to a promoter) prior to application to the tissue defect site. The isolate can be made and applied to the tissue defect site in a single procedure (i.e., intraoperatively).
Owner:MCKAY WILLIAM F +1
Methods for the production of ips cells using non-viral approach
ActiveUS20100003757A1Promote generationExpand the populationGenetically modified cellsVirus peptidesVector elementCell type
Methods and composition of induction of pluripotent stem cells and other desired cell types are disclosed. For example, in certain aspects methods for generating essentially vector-free induced pluripotent stem cells are described. Furthermore, the invention provides induced pluripotent stem cells and desired cell types essentially free of exogenous vector elements with the episomal expression vectors to express differentiation programming factors.
Owner:FUJIFILM CELLULAR DYNAMICS INC
Somatic cell reprogramming by retroviral vectors encoding Oct3/4. Klf4, c-Myc and Sox2
ActiveUS8129187B2Easy to prepareEffective isolationGenetically modified cellsArtificial cell constructsNuclear reprogrammingCell therapy
The present invention relates to a nuclear reprogramming factor having an action of reprogramming a differentiated somatic cell to derive an induced pluripotent stem (iPS) cell. The present invention also relates to the aforementioned iPS cells, methods of generating and maintaining iPS cells, and methods of using iPS cells, including screening and testing methods as well as methods of stem cell therapy. The present invention also relates to somatic cells derived by inducing differentiation of the aforementioned iPS cells.
Owner:KYOTO UNIV
Postpartum cells derived from umbilical cord tissue, and methods of making and using the same
Cells derived from human umbilical cords are disclosed along with methods for their therapeutic use. Isolation techniques, culture methods and detailed characterization of the cells with respect to their cell surface markers, gene expression, and their secretion of trophic factors are described.
Owner:DEPUY SYNTHES PROD INC
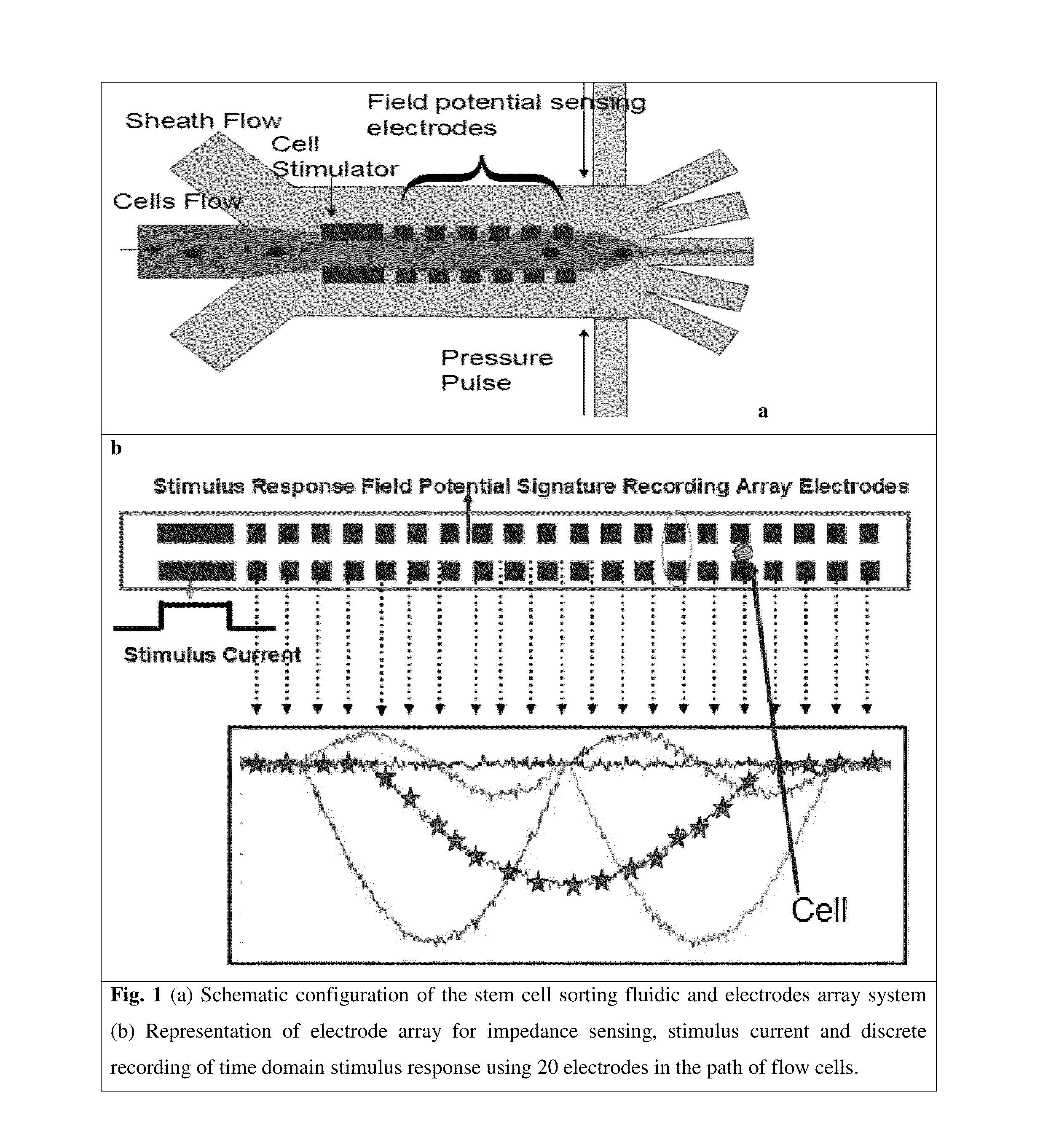
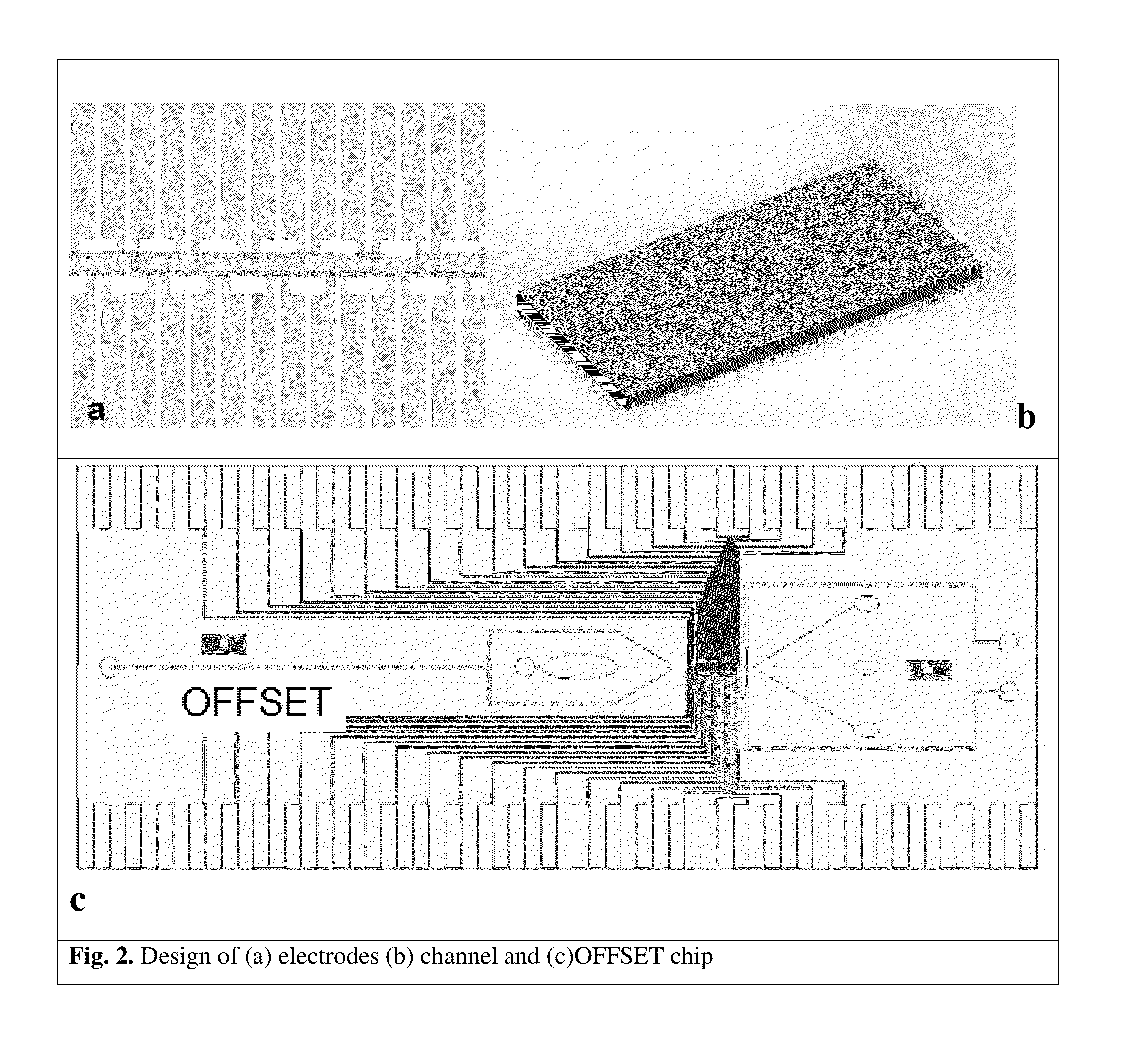
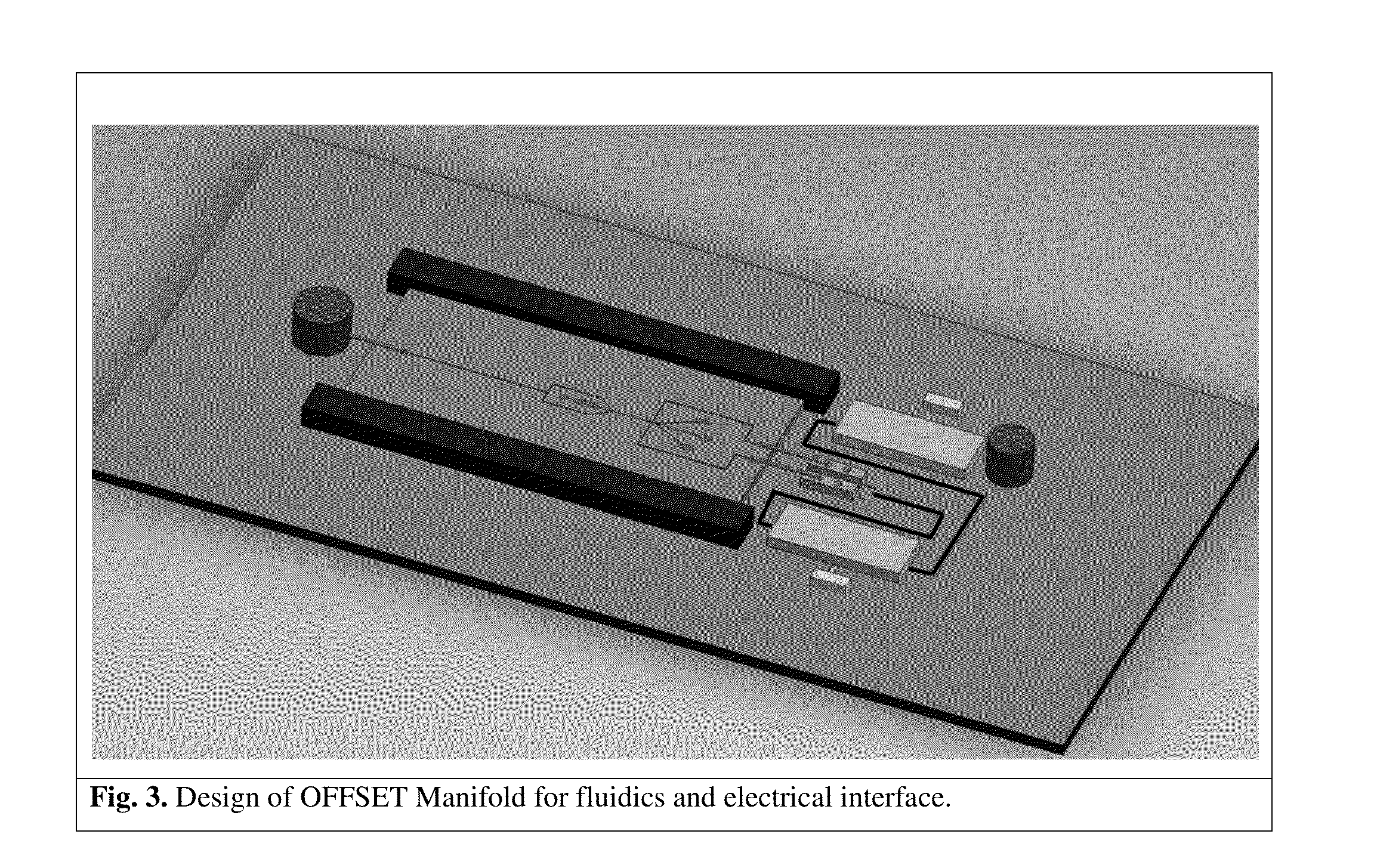
Microfluidic devices and methods for cell sorting, cell culture and cells based diagnostics and therapeutics
ActiveUS20140248621A1Uniform pressure distributionReduce widthHeating or cooling apparatusMicrobiological testing/measurement3D cell cultureTumor cells
Microfluidic devices and methods that use cells such as cancer cells, stem cells, blood cells for preprocessing, sorting for various biodiagnostics or therapeutical applications are described. Microfluidics electrical sensing such as measurement of field potential or current and phenomena such as immiscible fluidics, inertial fluidics are used as the basis for cell and molecular processing (e.g., characterizing, sorting, isolation, processing, amplification) of different particles, chemical compositions or biospecies (e.g., different cells, cells containing different substances, different particles, different biochemical compositions, proteins, enzymes etc.). Specifically this invention discloses a few sorting schemes for stem cells, whole blood and circulating tumor cells and also extracting serum from whole blood. Further medical diagnostics technology utilizing high throughput single cell PCR is described using immiscible fluidics couple with single or multi cells trapping technology.
Owner:BIOPICO SYST
Pluripotent stem cells derived without the use of embryos or fetal tissue
InactiveUS20030113910A1New breed animal cellsArtificial cell constructsPluripotential stem cellGerm layer
Owner:STEMA
Use of umbilical cord blood to treat individuals having a disease, disorder or condition
The present invention provides methods of using cord blood and cord blood-derived stem cells in high doses to treat various conditions, diseases and disorders. The high-dose cord blood and cord blood-derived stem cells have a multitude of uses and applications, including but not limited to, therapeutic uses for transplantation and treatment and prevention of disease, and diagnostic and research uses. In particular, the cord blood or cord blood-derived stem cells are delivered in high doses, e.g., at least 3 billion nucleated cells per treatment, where treatment may comprise a single or multiple infusions. The invention also provides for the use of cord blood or cord blood-derived stem cells from multiple donors without the need for HLA typing.
Owner:CELULARITY INC
Multipotent adult stem cells and methods for isolation
ActiveUS20050181502A1Improve the level ofLong telomereGenetically modified cellsDiagnosticsTissues typesEmbryo
Owner:ABT HOLDING COMPANY +1
Medium containing pipecholic acid and gamma amino butyric acid and culture of embryonic stem cells
Previous methods for culturing human embryonic stem cells have required either fibroblast feeder cells or a medium which has been exposed to fibroblast feeder cells in order to maintain the stem cells in an undifferentiated state. It has now been found that if high levels of fibroblast growth factor, gamma amino butyric acid, pipecholic acid, lithium and transforming growth factor beta are added to the medium in which the stem cells are cultured, the stem cells will remain undifferentiated indefinitely through multiple passages, even without feeder cells or conditioned medium.
Owner:WISCONSIN ALUMNI RES FOUND
Methods for reprogramming somatic cells
Owner:WHITEHEAD INST FOR BIOMEDICAL RES
Banking of multipotent amniotic fetal stem cells
Stem cells, including those designated as multipotent amniotic fluid stem cells (MAFSC) cells are found in the amniotic fluid of mammals, including humans. MAFSCs are fetal, multipotent stem cells that can be used for any desired stem cell utility, including treatment of individuals in need of tissue replacement or gene therapy. Methods of banking MAFSCs derived from the amniotic fluid cells of pregnant individuals are disclosed. Amniotic fluid-derived cells are banked for the purpose of access to transplantation antigen-compatible or syngeneic multipotent stem cells.
Owner:HAAS MARTIN
Medium for growing human embryonic stem cells
InactiveUS7297539B2Rapid productionExpanding primate pluripotent stem (pPS) cellsHepatocytesGastrointestinal cellsGerm layerFiber
This disclosure provides an improved system for culturing human pluripotent stem cells. Traditionally, pluripotent stem cells are cultured on a layer of feeder cells (such as mouse embryonic fibroblasts) to prevent them from differentiating. In the system described here, the role of feeder cells is replaced by components added to the culture environment that support rapid proliferation without differentiation. Effective features are a suitable support structure for the cells, and an effective medium that can be added fresh to the culture without being preconditioned by another cell type. Culturing human embryonic stem cells in fresh medium according to this invention causes the cells to expand surprisingly rapidly, while retaining the ability to differentiate into cells representing all three embryonic germ layers. This new culture system allows for bulk proliferation of pPS cells for commercial production of important products for use in drug screening and human therapy.
Owner:ASTERIAS BIOTHERAPEUTICS INC
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com