Patents
Literature
406 results about "Laminin" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Laminins are high-molecular weight (~400 to ~900 kDa) proteins of the extracellular matrix. They are a major component of the basal lamina (one of the layers of the basement membrane), a protein network foundation for most cells and organs. The laminins are an important and biologically active part of the basal lamina, influencing cell differentiation, migration, and adhesion.
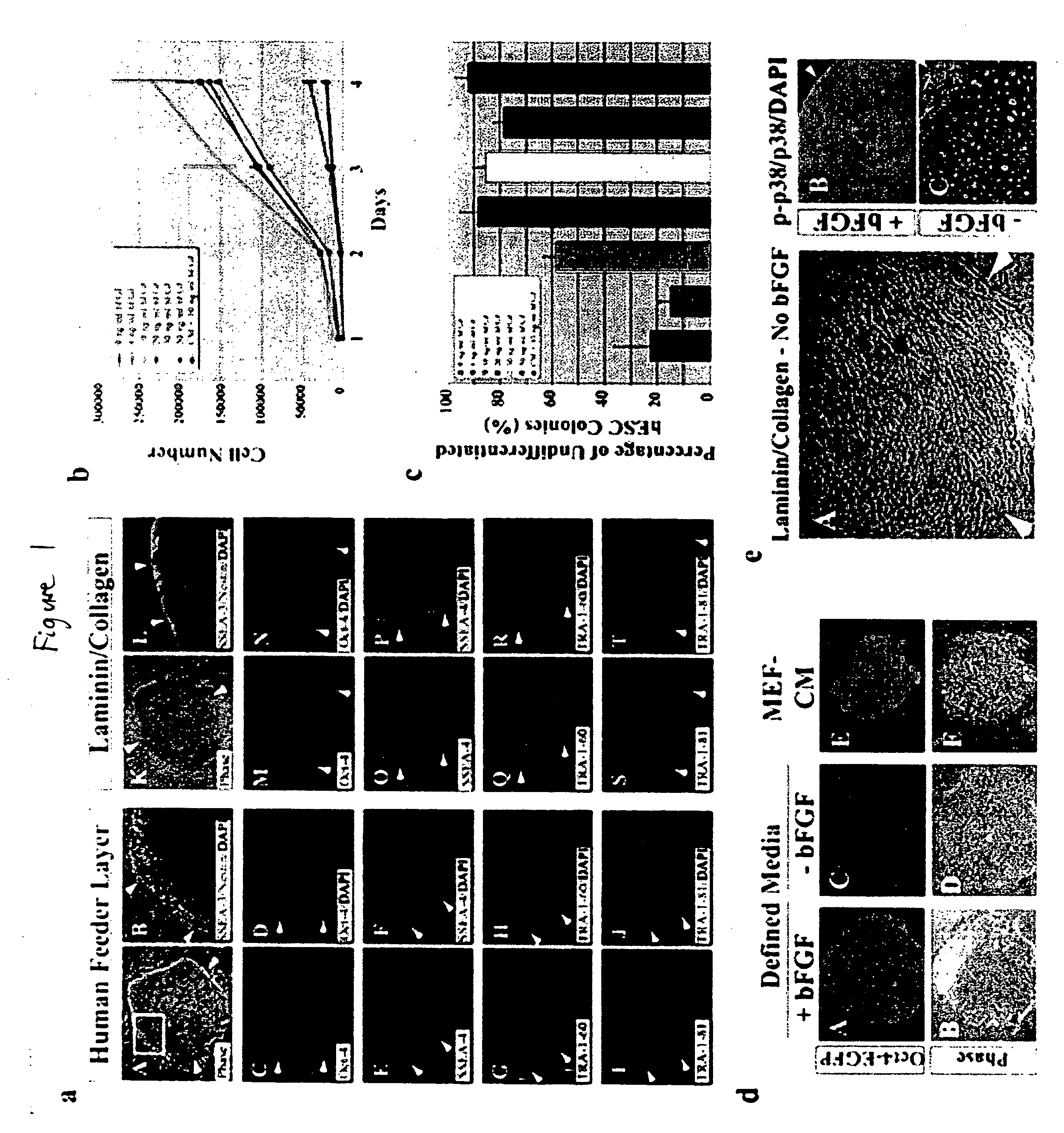
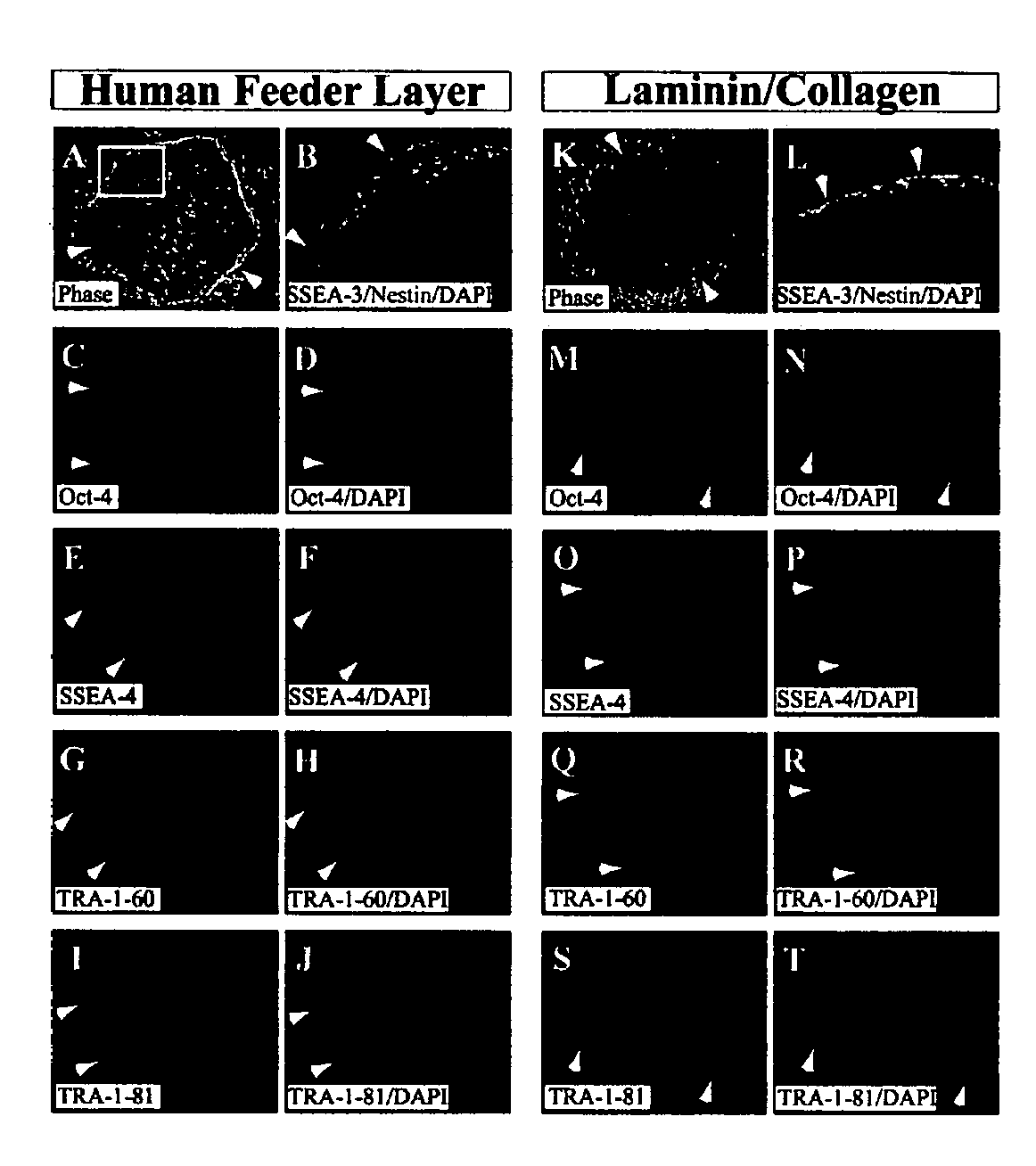
Defined media for stem cell culture
Stem cells, including mammalian, and particularly primate primordial stem cells (pPSCs) such as human embryonic stem cells (hESCs), hold great promise for restoring cell, tissue, and organ function. However, cultivation of stem cells, particularly undifferentiated hESCs, in serum-free, feeder-free, and conditioned-medium-free conditions remains crucial for large-scale, uniform production of pluripotent cells for cell-based therapies, as well as for controlling conditions for efficiently directing their lineage-specific differentiation. This instant invention is based on the discovery of the formulation of minimal essential components necessary for maintaining the long-term growth of pPSCs, particularly undifferentiated hESCs. Basic fibroblast growth factor (bFGF), insulin, ascorbic acid, and laminin were identified to be both sufficient and necessary for maintaining hESCs in a healthy self-renewing undifferentiated state capable of both prolonged propagation and then directed differentiation. Having discerned these minimal molecular requirements, conditions that would permit the substitution of poorly-characterized and unspecified biological additives and substrates were derived and optimized with entirely defined constituents, providing a “biologics”-free (i.e., animal-, feeder-, serum-, and conditioned-medium-free) system for the efficient long-term cultivation of pPSCs, particularly pluripotent hESCs. Such culture systems allow the derivation and large-scale production of stem cells such as pPSCs, particularly pluripotent hESCs, in optimal yet well-defined biologics-free culture conditions from which they can be efficiently directed towards a lineage-specific differentiated fate in vitro, and thus are important, for instance, in connection with clinical applications based on stem cell therapy and in drug discovery processes.
Owner:THE BURNHAM INST
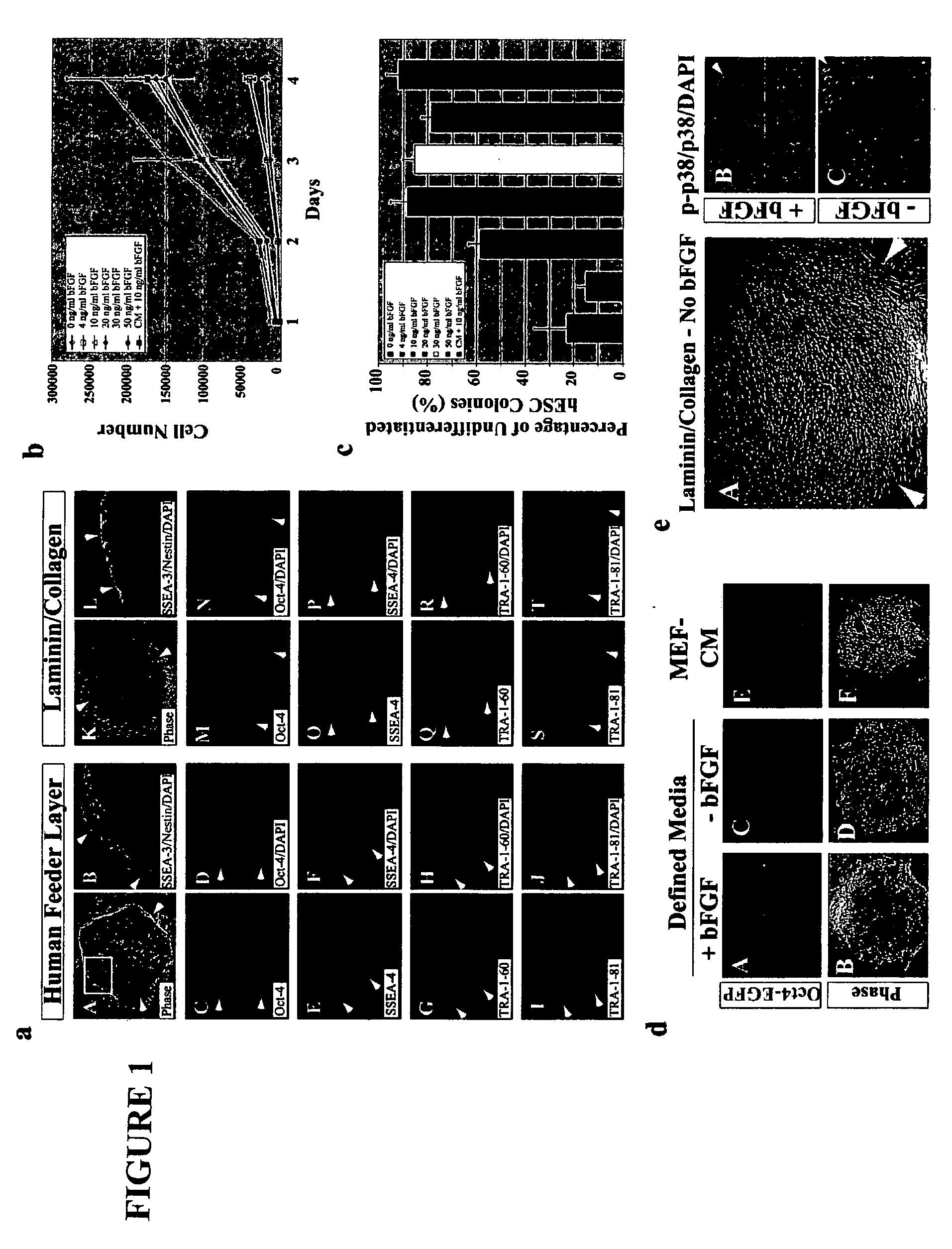
Defined media for pluripotent stem cell culture
Stem cells, including mammalian, and particularly primate primordial stem cells (pPSCs) such as human embryonic stem cells (hESCs), hold great promise for restoring cell, tissue, and organ function. However, cultivation of stem cells, particularly undifferentiated hESCs, in serum-free, feeder-free, and conditioned-medium-free conditions remains crucial for large-scale, uniform production of pluripotent cells for cell-based therapies, as well as for controlling conditions for efficiently directing their lineage-specific differentiation. This instant invention is based on the discovery of the formulation of minimal essential components necessary for maintaining the long-term growth of pPSCs, particularly undifferentiated hESCs. Basic fibroblast growth factor (bFGF), insulin, ascorbic acid, and laminin were identified to be both sufficient and necessary for maintaining hESCs in a healthy self-renewing undifferentiated state capable of both prolonged propagation and then directed differentiation. Having discerned these minimal molecular requirements, conditions that would permit the substitution of poorly-characterized and unspecified biological additives and substrates were derived and optimized with entirely defined constituents, providing a “biologics”-free (i.e., animal-, feeder-, serum-, and conditioned-medium-free) system for the efficient long-term cultivation of pPSCs, particularly pluripotent hESCs. Such culture systems allow the derivation and large-scale production of stem cells such as pPSCs, particularly pluripotent hESCs, in optimal yet well-defined biologics-free culture conditions from which they can be efficiently directed towards a lineage-specific differentiated fate in vitro, and thus are important, for instance, in connection with clinical applications based on stem cell therapy and in drug discovery processes.
Owner:THE BURNHAM INST
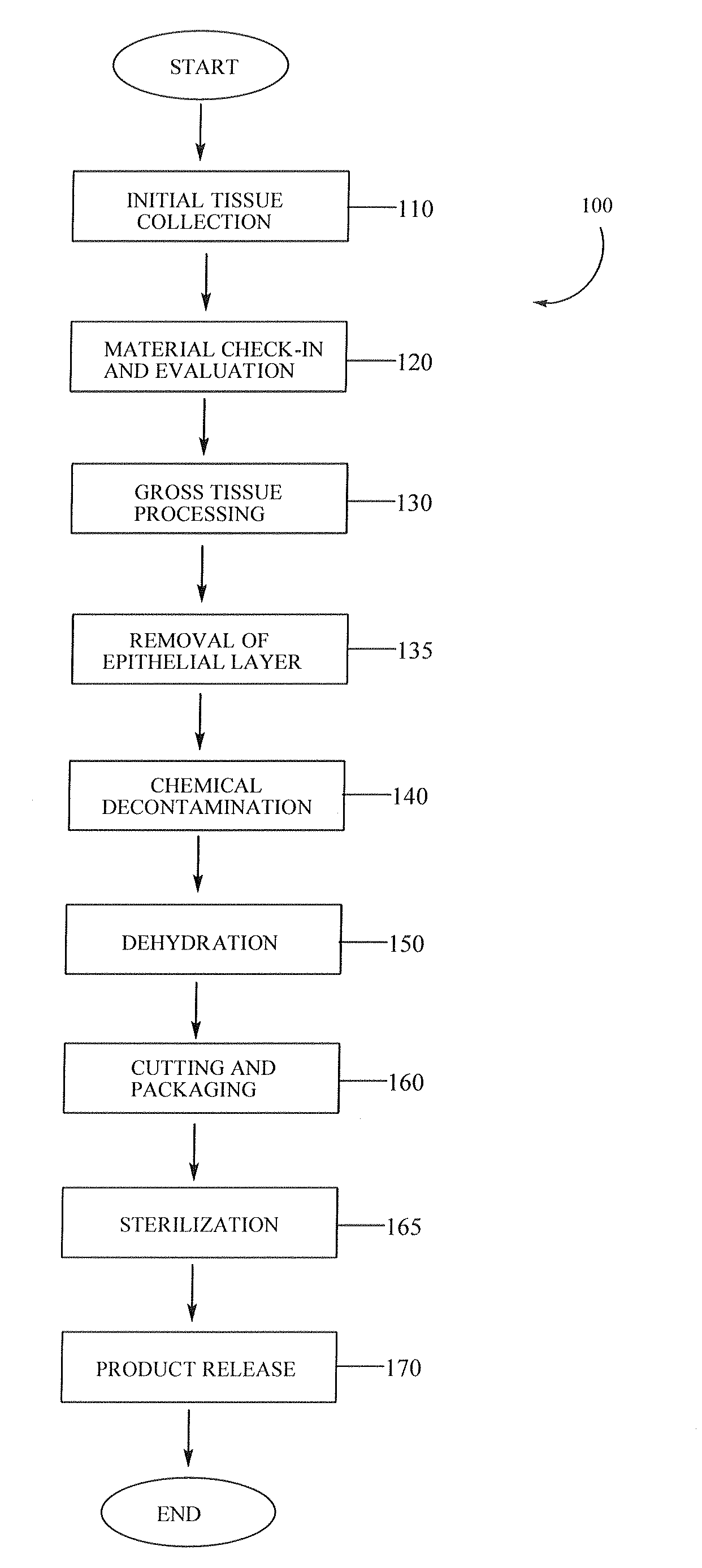
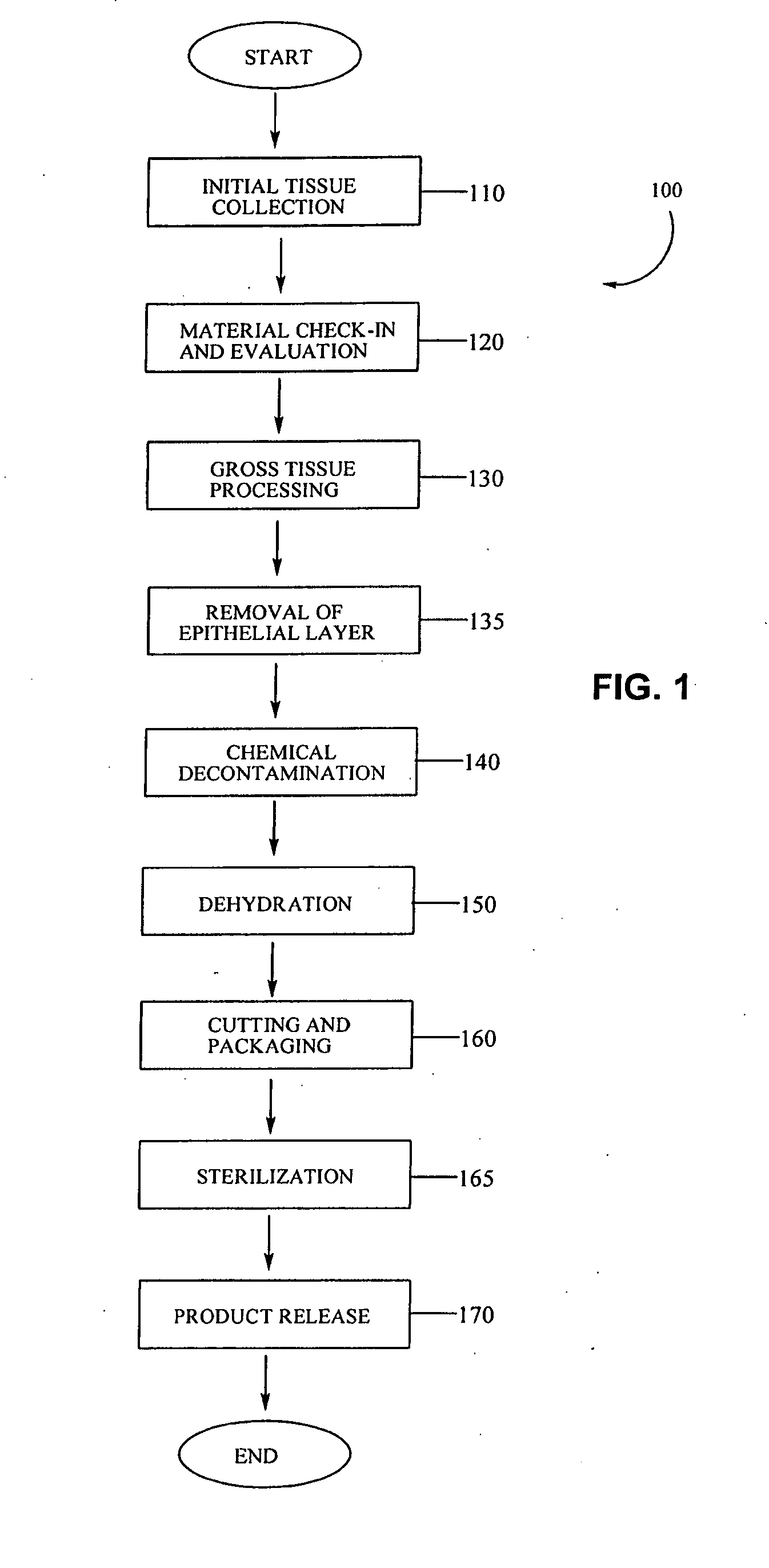
Placental tissue grafts and improved methods of preparing and using the same
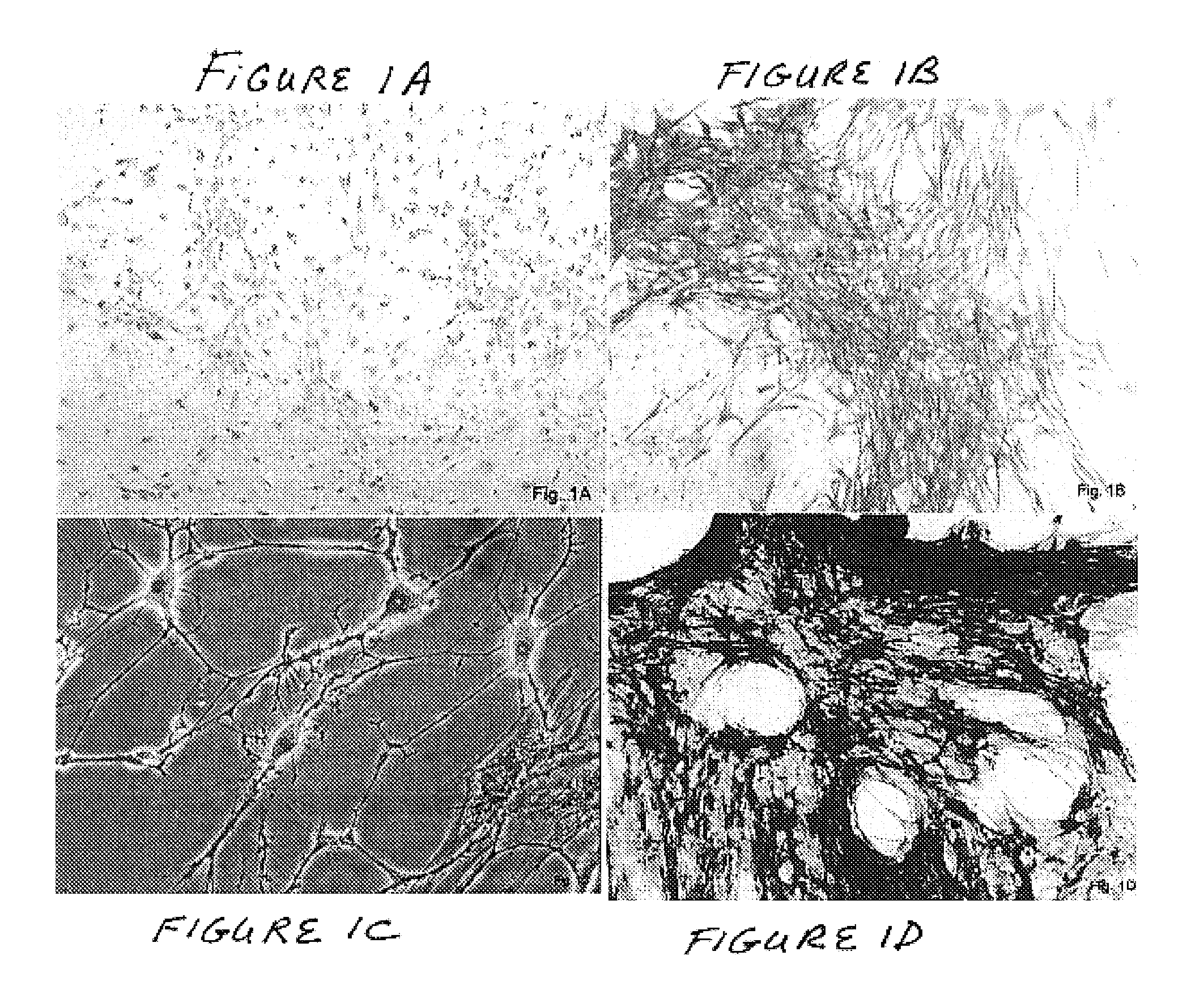
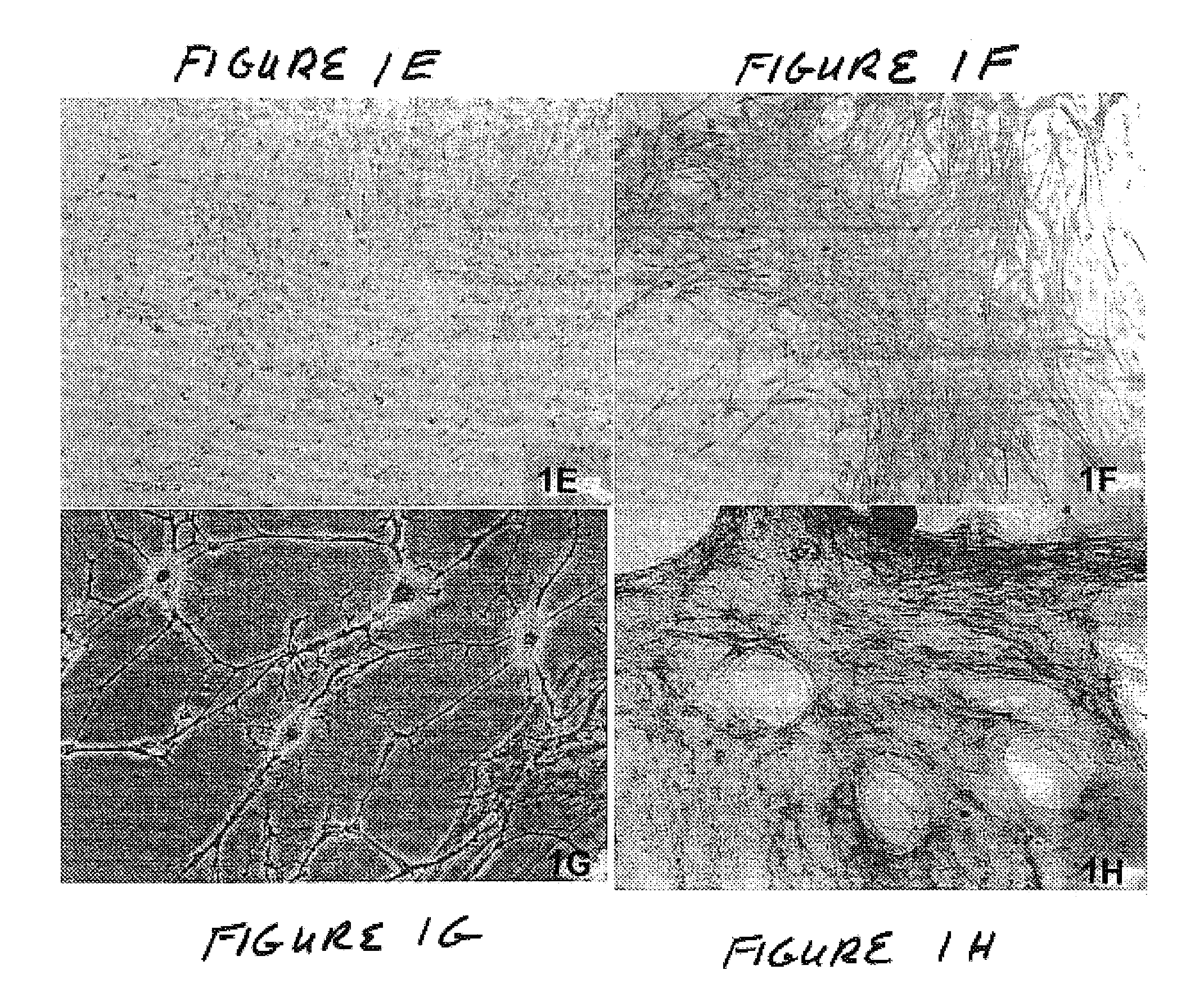
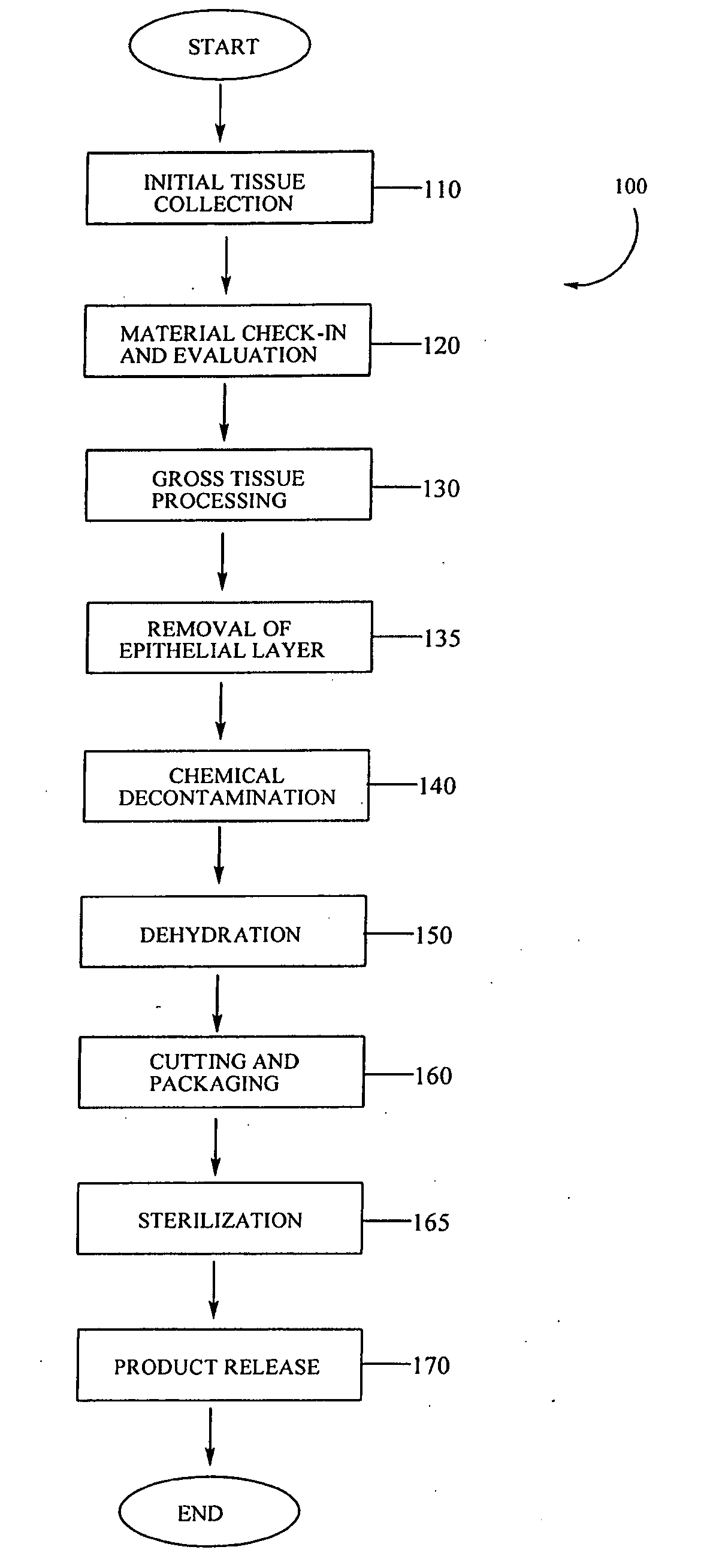
Described herein are tissue grafts derived from the placenta. The grafts are composed of at least one layer of amnion tissue where the epithelium layer has been substantially removed in order to expose the basement layer to host cells. By removing the epithelium layer, cells from the host can more readily interact with the cell-adhesion bio-active factors located onto top and within of the basement membrane. Also described herein are methods for making and using the tissue grafts. The laminin structure of amnion tissue is nearly identical to that of native human tissue such as, for example, oral mucosa tissue. This includes high level of laminin-5, a cell adhesion bio-active factor show to bind gingival epithelia-cells, found throughout upper portions of the basement membrane.
Owner:MIMEDX GROUP
Biodegradable polymer coils for intraluminal implants
An endovascular cellular manipulation and inflammatory response are elicited from implantation in a vascular compartment or any intraluminal location of a separable coil comprised at least in part of at least one biocompatible and absorbable polymer or protein and growth factors. Typically a catheter associated with the separable coil is used to dispose the coil into a selected body lumen. The biocompatible and absorbable polymer or protein is thrombogenic. The coil further is comprised at least in part of a growth factor or more particularly a vascular endothelial growth factor, a basic fibroblast growth factor or other growth factors. The biocompatible and absorbable polymer is in the illustrated embodiment at least one polymer selected from the group consisting of polyglycolic acid, poly~glycolic acid poly-L-lactic acid copolymers, polycaprolactive, polyhydroxybutyrate / hydroxyvalerate copolymers, poly-L-lactide. Polydioxanone, polycarbonates, and polyanhydrides. The biocompatible and absorbable protein is at least one protein selected from the group consisting of collagen, fibrinogen, fibronectin, vitronectin, laminin, and gelatin. In one embodiment the coil is composed of the biocompatible and absorbable polymer or protein with a radio-opaque material is disposed thereon. Alternatively, the coil is composed of a radio-opaque material, and the biocompatible and absorbable polymer or protein is disposed thereon. This apparatus may be positioned within intracranial aneurysms or any aneurysm in the body as well as within other body cavities.
Owner:RGT UNIV OF CALIFORNIA
Hydrogel bioscaffoldings and biomedical device coatings
Bioscaffoldings formed of hydrogels that are crosslinked in situ in an infarcted region of the heart (myocardium) by a Michael's addition reaction or by a disulfide bond formed by an oxidative process are described. Each of the bioscaffoldings described includes hyaluronan as one of the hydrogel components and the other component is selected from collagen, collagen-laminin, poly-l-lysine, and fibrin. The bioscaffolding may further include an alginate component. The bioscaffoldings may have biofunctional groups such as angiogenic factors and stem cell homing factors bound to the collagen, collagen-laminin, poly-l-lysine, or fibrinogen hydrogel component. In particular, the biofunctional groups may be PR11, PR39, VEGF, bFGF, a polyarginine / DNA plasmid complex, or a DNA / polyethyleneimine (PEI) complex. Additionally, the hydrogel components may be injected into the infarct region along with stem cells and microspheres containing stem cell homing factors. The bioscaffolding may be formed on a stent or a cardiac medical device.
Owner:ABBOTT CARDIOVASCULAR
Methods and compositions for smooth muscle reconstruction
This invention also provides a purified or isolated population of ADSCs that can differentiate into a cell of the leiomyogenic lineage, e.g., smooth muscle or skeletal muscle. In yet another aspect, the population additionally can be differentiated into a lineage selected from the group consisting of osteogenic, adipogenic, chondrogenic, myogenic and neuronal. This invention further provides a composition comprising a substantially homogeneous expanded population of smooth muscle cells. This invention provides a composition comprising a substantially homogeneous expanded population of skeletal muscle cells. Also provided herein is an isolated composition comprising a purified adipose-derived stem cell (ADSC) or progeny of said ADSC and an effective amount of laminin or heparin, effective to induce leiomyogenic differentiation. Diagnositic and therapeutic uses for these compositions are provided herein.
Owner:RGT UNIV OF CALIFORNIA
Placental tissue grafts and improved methods of preparing and using the same
Described herein are tissue grafts derived from the placenta. The grafts are composed of at least one layer of amnion tissue where the epithelium layer has been substantially removed in order to expose the basement layer to host cells. By removing the epithelium layer, cells from the host can more readily interact with the cell-adhesion bio-active factors located onto top and within of the basement membrane. Also described herein are methods for making and using the tissue grafts. The laminin structure of amnion tissue is nearly identical to that of native human tissue such as, for example, oral mucosa tissue. This includes high level of laminin-5, a cell adhesion bio-active factor show to bind gingival epithelia-cells, found throughout upper portions of the basement membrane.
Owner:MIMEDX GROUP +1
Compositions and methods for making and using laminin nanofibers
ActiveUS20110236974A1Low costHigh manufacturing requirementsElectric discharge heatingNanomedicineMicrosphereBiopolymer
The present invention provides methodologies and parameters for fabrication of the hybrid biomaterial by blending pure laminin or complex extracts of tissues containing laminin with biopolymers such as polycaprolactone (PCL), polylactic / polyglycolic acid copolymer (PLGA) or Polydioxanone (PDO) in fluoroalcohols (HFP, TFA), fabrication of substrates and scaffolds and devices from the hybrid biomaterial in forms such as films, nanofibers by electrospinning or microspheres, and the biological or biomedical use of the material or devices derived from it.
Owner:UNIV OF VIRGINIA ALUMNI PATENTS FOUND
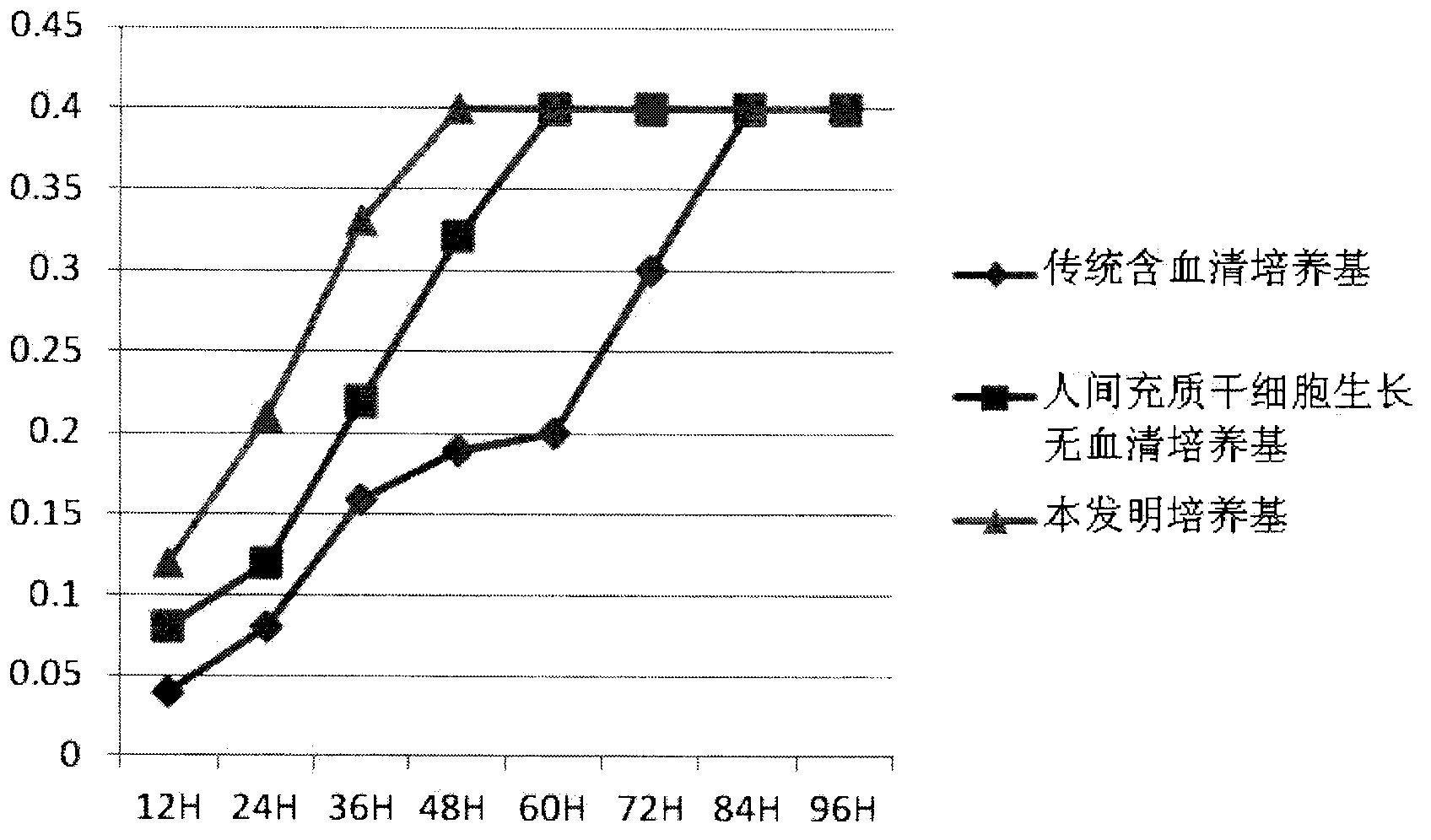
Clinical-grade human mesenchymal stem cell serum-free complete medium
ActiveCN103243071APromote growthLow toxicitySkeletal/connective tissue cellsInsulin-like growth factorCuticle
The invention relates to a human mesenchymal stem cell culture medium. According to the culture medium, the basal culture medium comprises the following components based on the final concentration: 1-2g / L of human serum albumin, 5-10mg / L of transferring, 2-8mg / L of fibronectin, 1-4mg / L of laminin, 50g / L of Fe(NO3)3.9H2O, 417g / L of FeSO4.7H2O, 1-3mu g / L of estradiol, 2-5mu g / L of testosterone, 1-3mu g / L of progesterone, 39.25-117.74 mu g / L of dexamethasone, 5-10mg / L of insulin, 376.36mg / L of riboflavin, 80.96-242.87mg / L of coenzyme A, 4.41-6.17mg / L of butanediamine, 1-2mg / L of taurine, 0.61-1.85mg / L of aminoethanol, 8.81-26.42mg / L of pyruvic acid, 3.78-7.56mu g / L of sodium selenate, 292.3-584.6mg / L of L-glutamine, 2-8mu g / L of vascular endothelial growth factor, 4-10mu g / L of epidermal growth factor, 4-10mu g / L of basic fibroblast growth factor, 1-5mu g / L of leukaemia inhibitory factor, 1-5mu g / L of insulin-like growth factor-I and 2-8mu g / L of stem cell factor. The culture medium does not contain the animal serum, the potential animal endogenous endotoxin or virus of the animal serum is eliminated, and the culture medium is conveniently applied to clinics.
Owner:QINGDAO RESTORE BIOTECHNOLOGY CO LTD
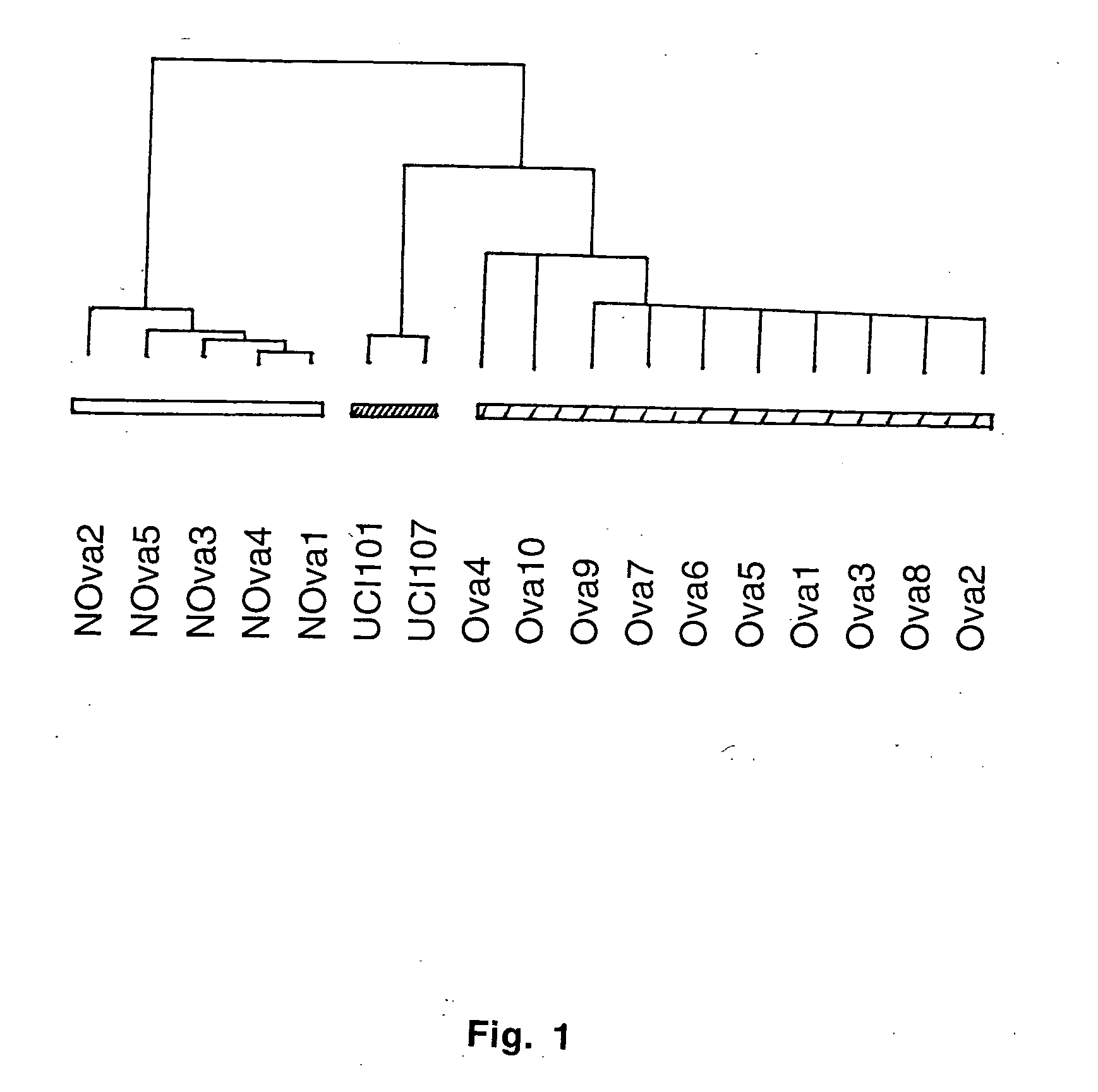
Gene expression profiling in primary ovarian serous papillary tumors and normal ovarian epithelium
InactiveUS20060078941A1Highligthing the divergence of gene expressionMicrobiological testing/measurementBiological testingKallikrein-10Gene family
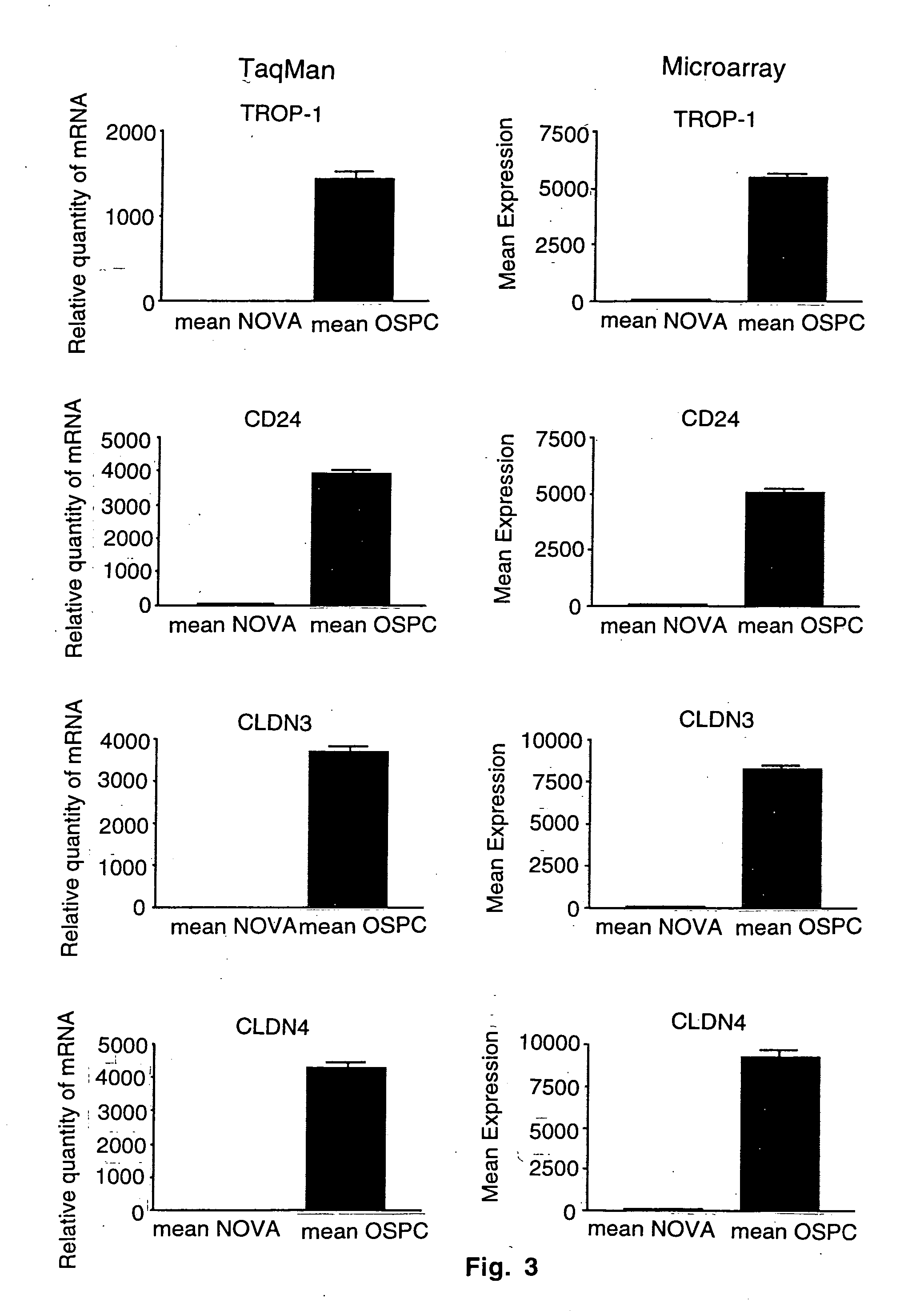
Gene expression profiling and hierarchial clustering analysis readily distinguish normal ovarian epithelial cells from primary ovarian serous papillary carcinomas. Laminin, tumor-associated calcium signal transducer 1 and 2 (TROP-1 / Ep-CAM; TROP-2), claudin 3, claudin 4, ladinin 1, S100A2, SERPIN2 (PAI-2), CD24, lipocalin 2, osteopontin, kallikrein 6 (protease M), kallikrein 10, matriptase and stratifin were found among the most highly overexpressed genes in ovarian serous papillary carcinomas, whereas transforming growth factor beta receptor III, platelet-derived growth factor receptor alpha, SEMACAP3, ras homolog gene family, member I (ARHI), thrombospondin 2 and disabled-2 / differentially expressed in ovarian carcinoma 2 (Dab2 / DOC2) were significantly down-regulated. Therapeutic strategy targeting TROP-1 / Ep-CAM by monoclonal chimeric / humanized antibodies may be beneficial in patients harboring chemotherapy-resistant ovarian serous papillary carcinomas. Claudin-3 and claudin-4 being receptors for Clostridium Perfringens enterotoxin, this toxin may be used as a novel therapeutic agent to treat ovarian serous papillary tumors.
Owner:THE BOARD OF TRUSTEES OF THE UNIV OF ARKANSAS
Biodegradable drug delivery vascular stent
A stent includes a main body of a generally tubular shape for insertion into a lumen of a vessel of a living being. The tubular main body includes a substantially biodegradable matrix having collagen IV and laminin that enclose voids within the matrix. The tubular main body also includes a biodegradable strengthening material in contact with the matrix to strengthen the matrix. The tubular main body is essentially saturated with drugs.
Owner:BOSTON SCI SCIMED INC
Methods and compositions for smooth muscle reconstruction
This invention also provides a purified or isolated population of ADSCs that can differentiate into a cell of the leiomyogenic lineage, e.g., smooth muscle or skeletal muscle. In yet another aspect, the population additionally can be differentiated into a lineage selected from the group consisting of osteogenic, adipogenic, chondrogenic, myogenic and neuronal. This invention further provides a composition comprising a substantially homogeneous expanded population of smooth muscle cells. This invention provides a composition comprising a substantially homogeneous expanded population of skeletal muscle cells. Also provided herein is an isolated composition comprising a purified adipose-derived stem cell (ADSC) or progeny of said ADSC and an effective amount of laminin or heparin, effective to induce leiomyogenic differentiation. Diagnositic and therapeutic uses for these compositions are provided herein.
Owner:RGT UNIV OF CALIFORNIA
Environments that maintain function of primary liver cells
InactiveUS20050059150A1Good adhesionEasy maintenanceHepatocytesArtificial cell constructsECM ProteinL-Ornithine
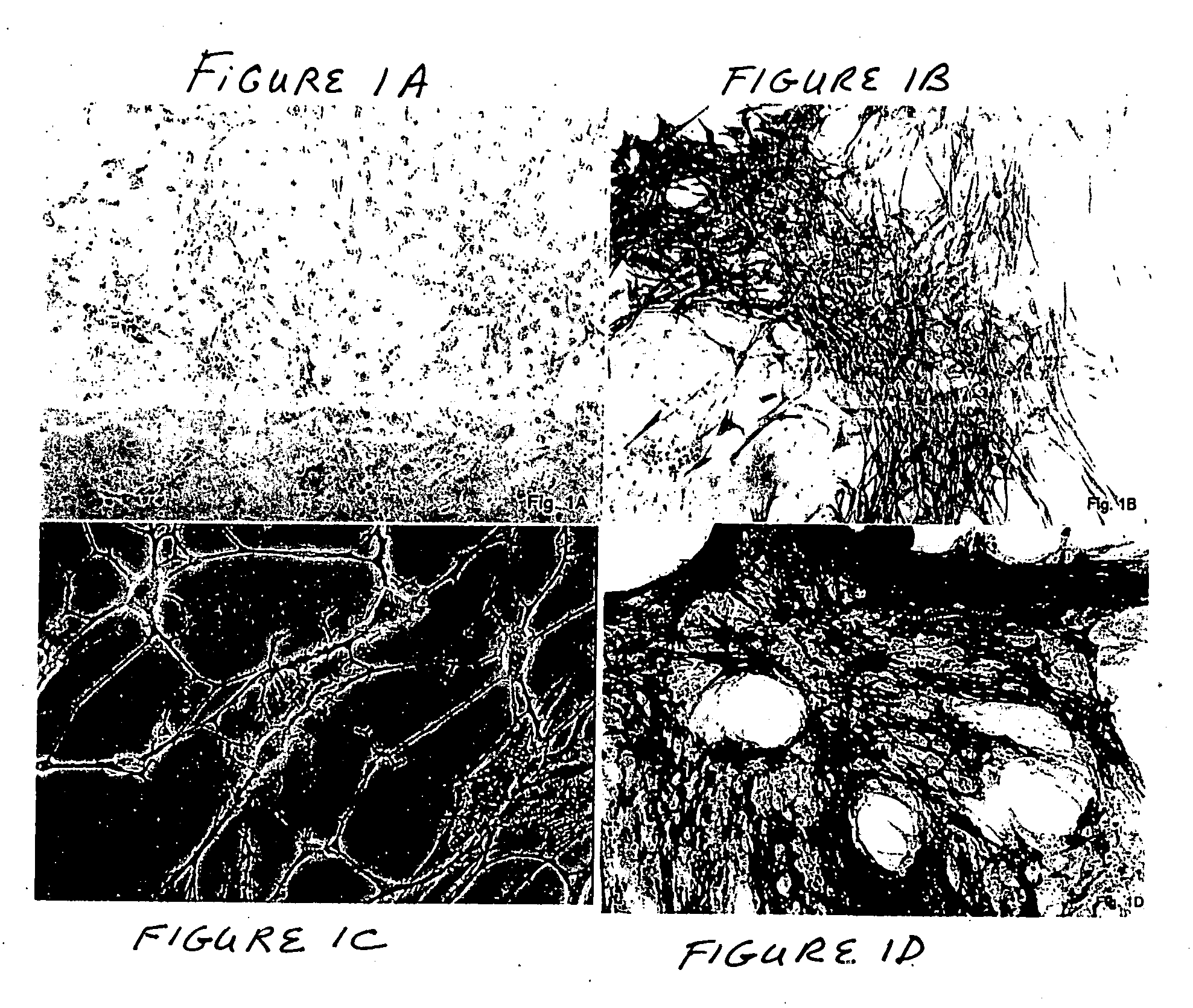
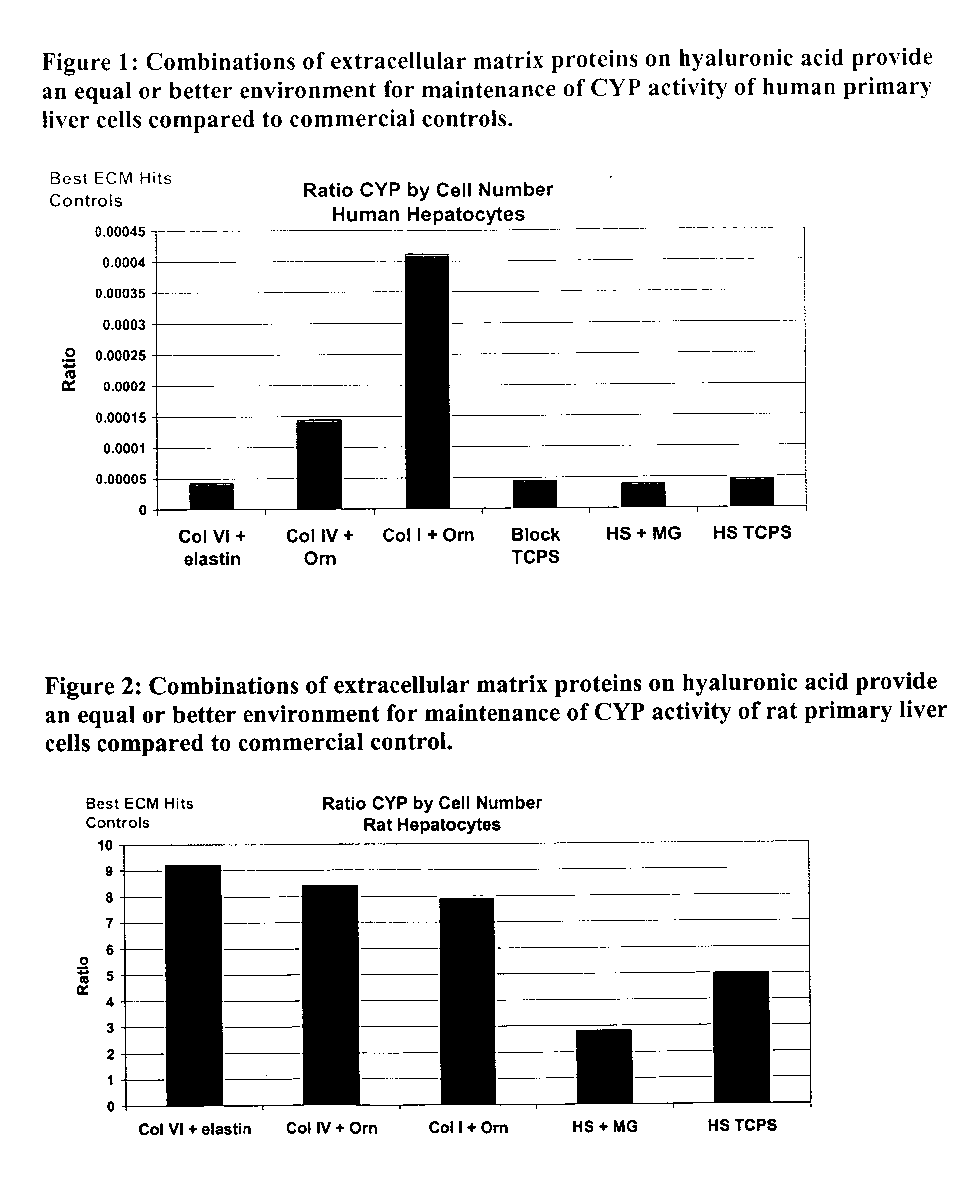
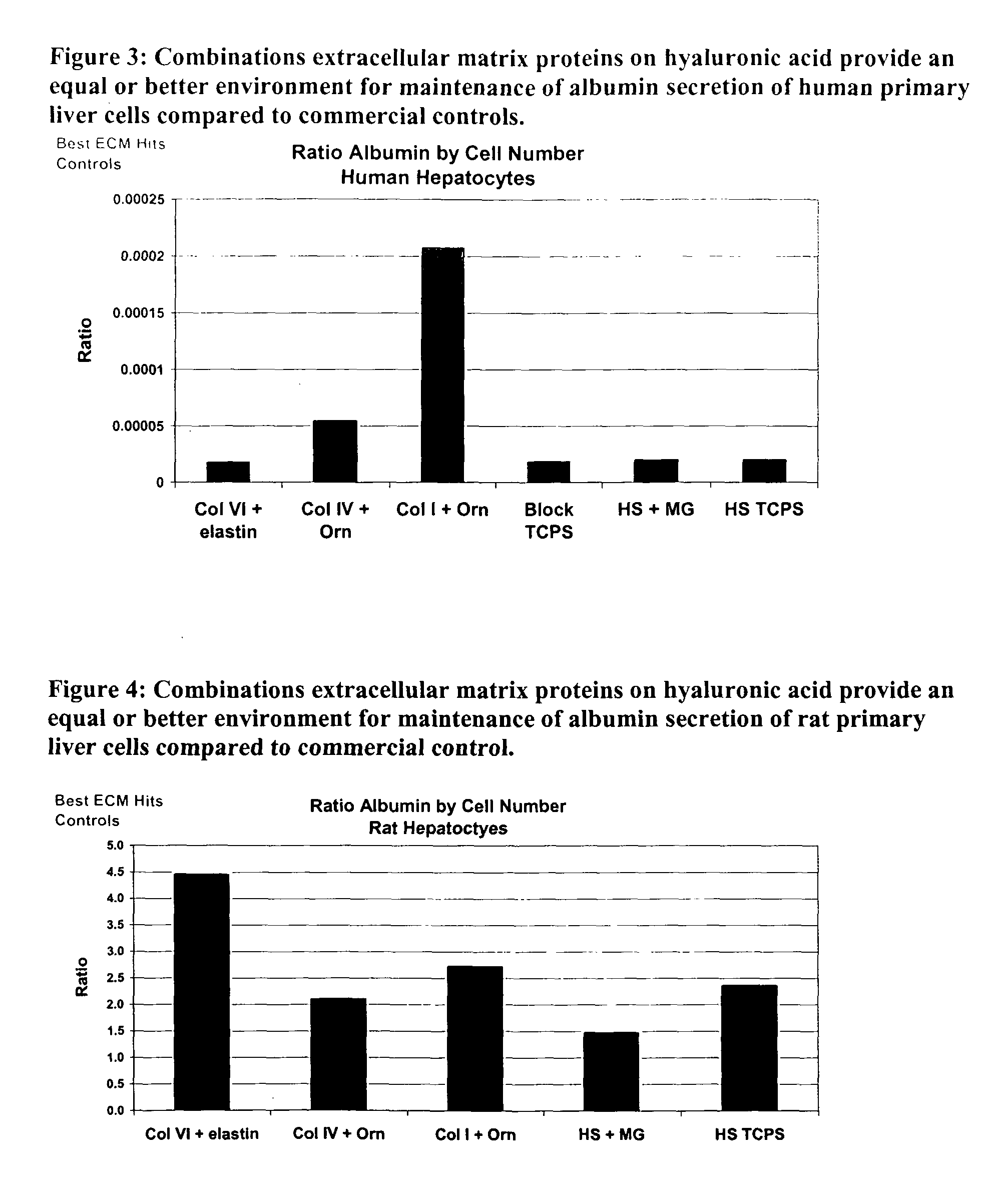
Surfaces useful for cell culture comprise a support to which is bound a CAR material, and, bound to the CAR material, an ECM protein, or a biologically active fragment or variant thereof such as elastin, fibronectin, vitronectin, laminin, collagen I, collagen III, collagen IV, and collagen VI. Also, optionally present on the surface is an active factor, preferably a polycationic polymer or a biologically active fragment or variant thereof, such as polyethyleneimine (PEI), poly-D-lysine (PDL), poly-L-lysine (PLL), poly-D-ornithine (PDO) or poly-L-ornithine (PLO). This surface is used in cell culture to promote cell attachment, survival, and / or proliferation of primary liver cells. The invention also relates to methods utilizing this surface, such as methods for attachment, survival, and / or proliferation of cells. Further disclosed is the use of the surface in cell culture with serum-free medium. Methods of screening using the surface of the invention are also disclosed.
Owner:BECTON DICKINSON & CO
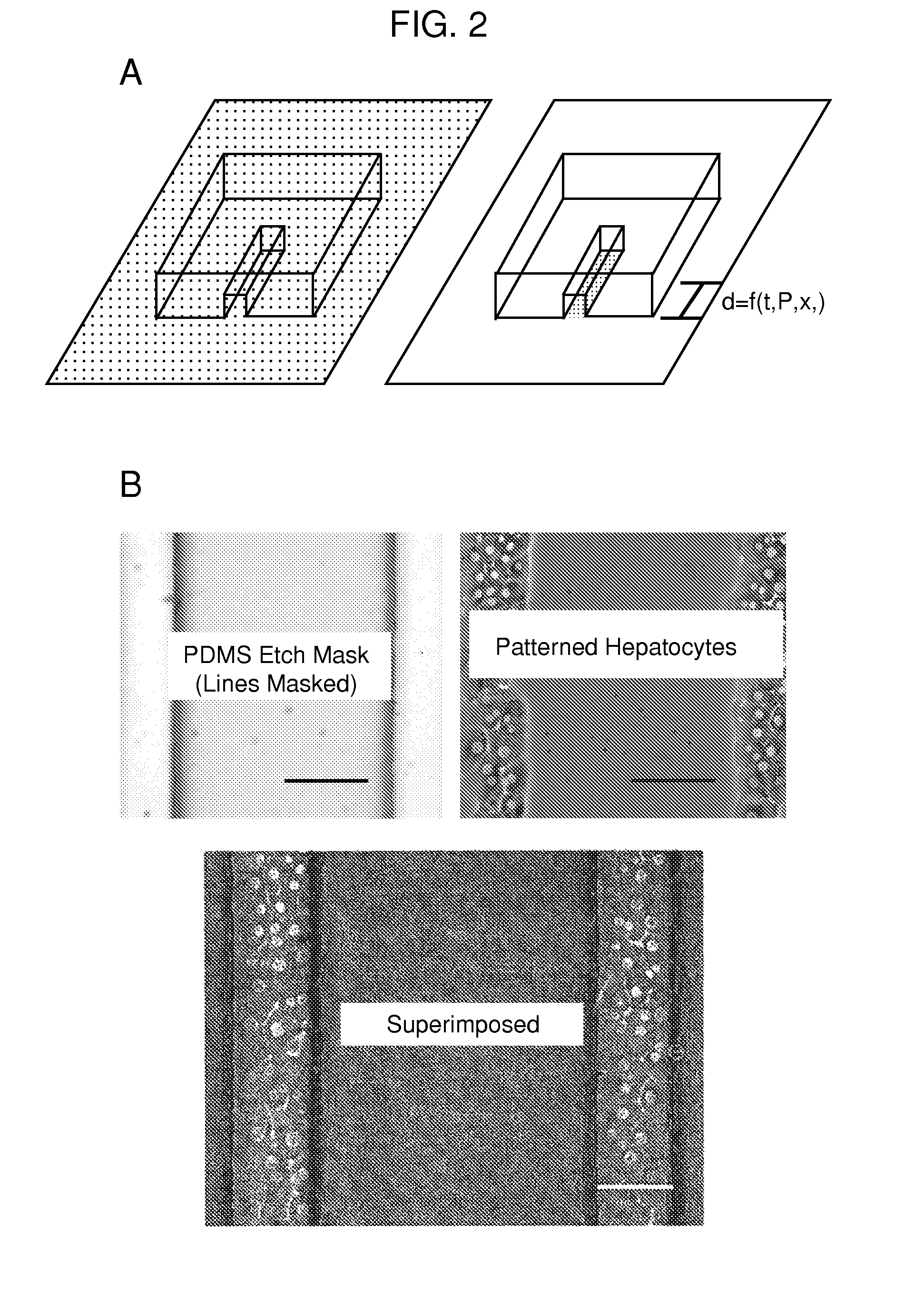
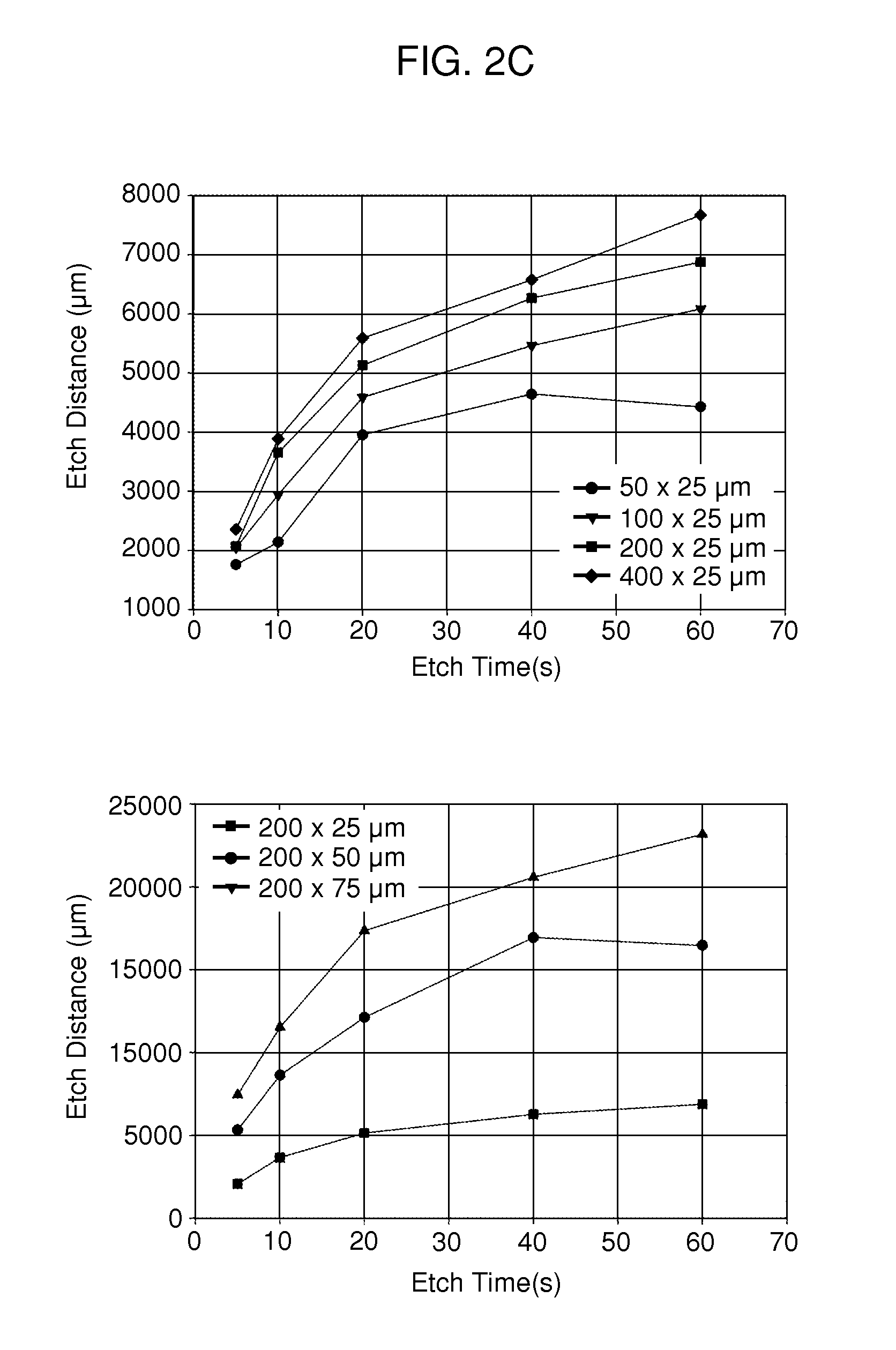
Multi-well micropatterning by ablation
InactiveUS20080220516A1Without increasing experimental complexitySolve the practicality is not highBioreactor/fermenter combinationsSequential/parallel process reactionsMatrigelAlternative methods
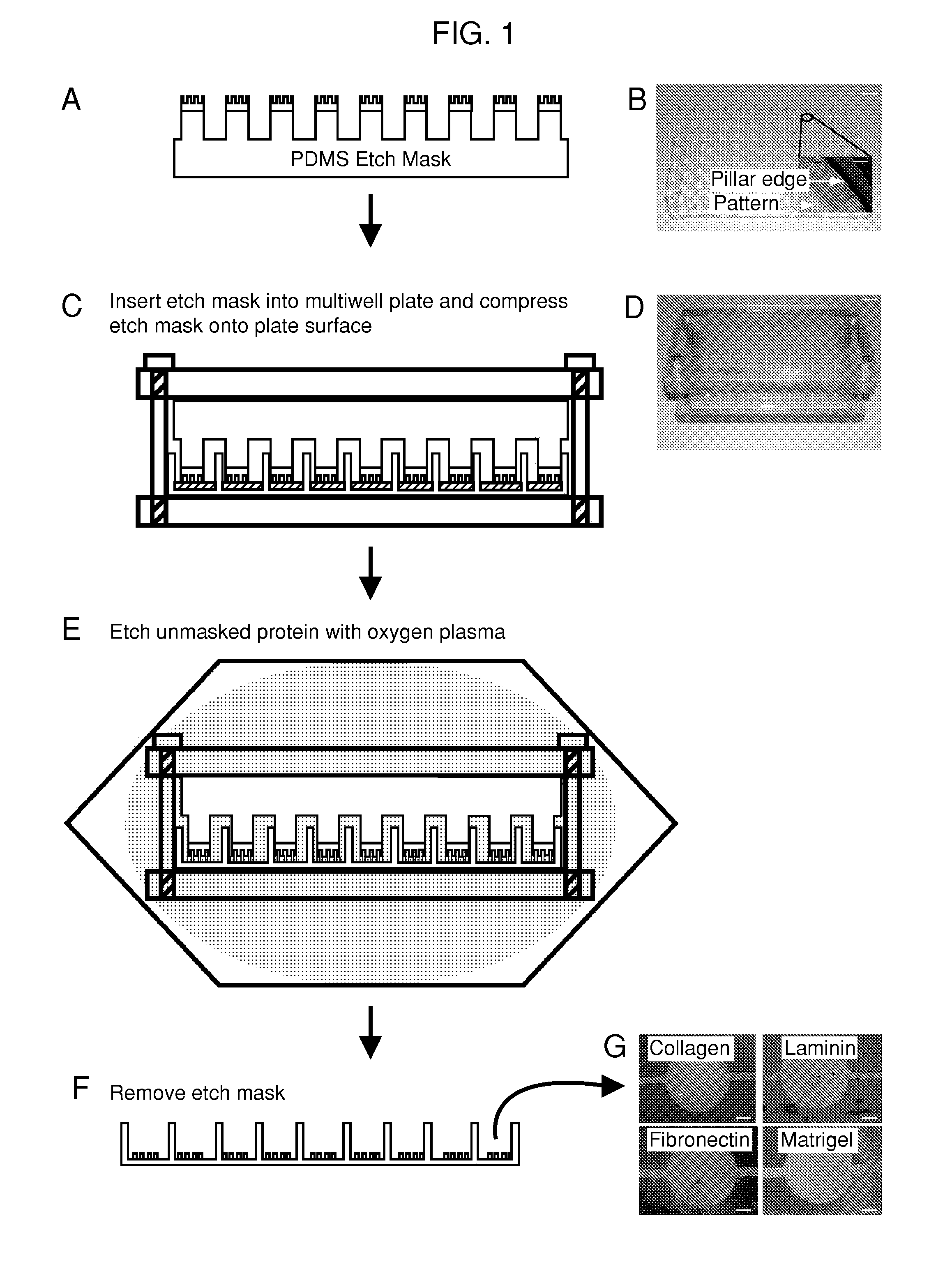
The present invention is drawn to the generation of micropatterns of biomolecules and cells on standard laboratory materials through selective ablation of a physisorbed biomolecule with oxygen plasma. In certain embodiments, oxygen plasma is able to ablate selectively physisorbed layers of biomolecules (e.g., type-I collagen, fibronectin, laminin, and Matrigel) along complex non-linear paths which are difficult or impossible to pattern using alternative methods. In addition, certain embodiments of the present invention relate to the micropatterning of multiple cell types on curved surfaces, multiwell plates, and flat bottom flasks. The invention also features kits for use with the subject methods.
Owner:MASSACHUSETTS INST OF TECH
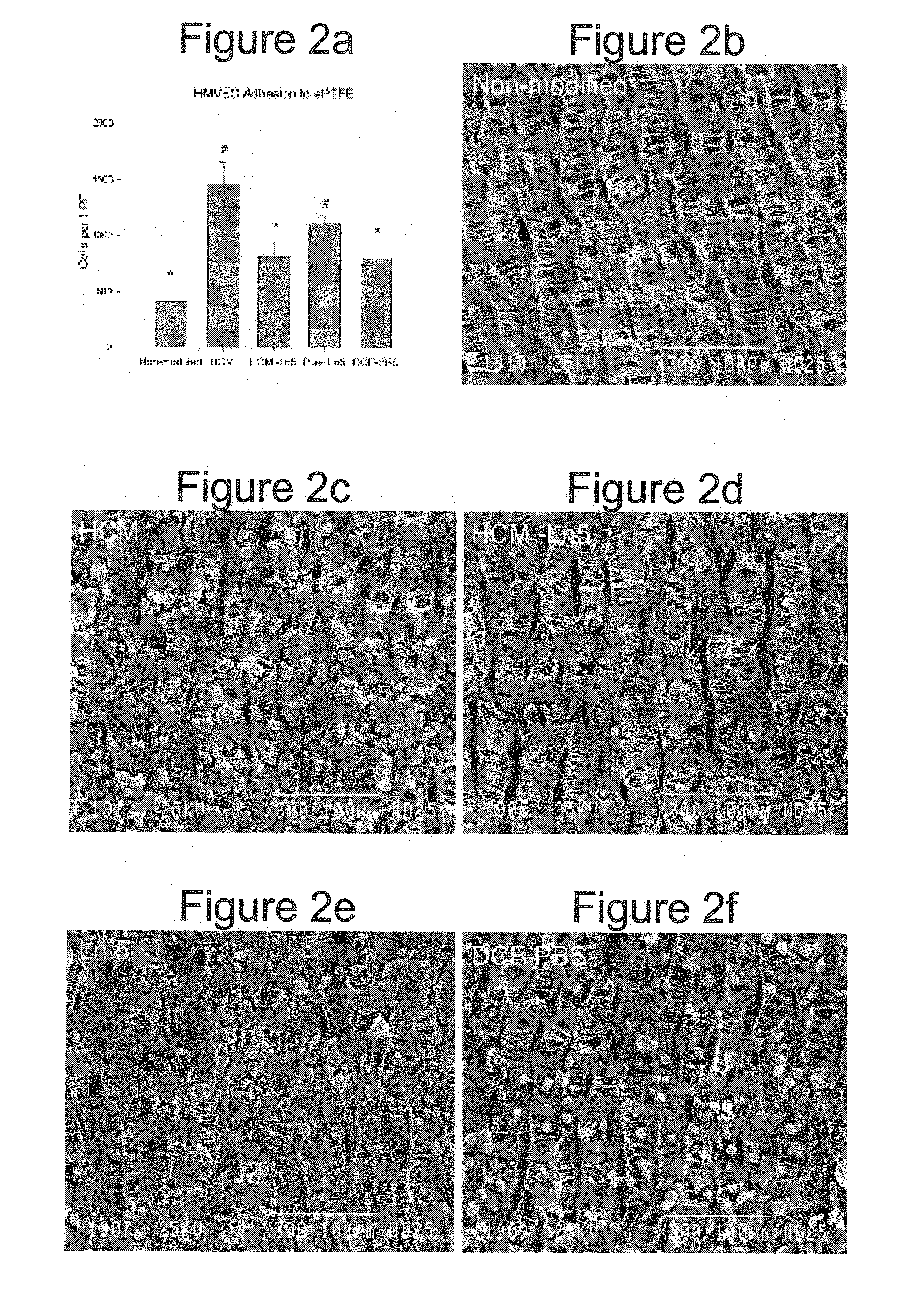
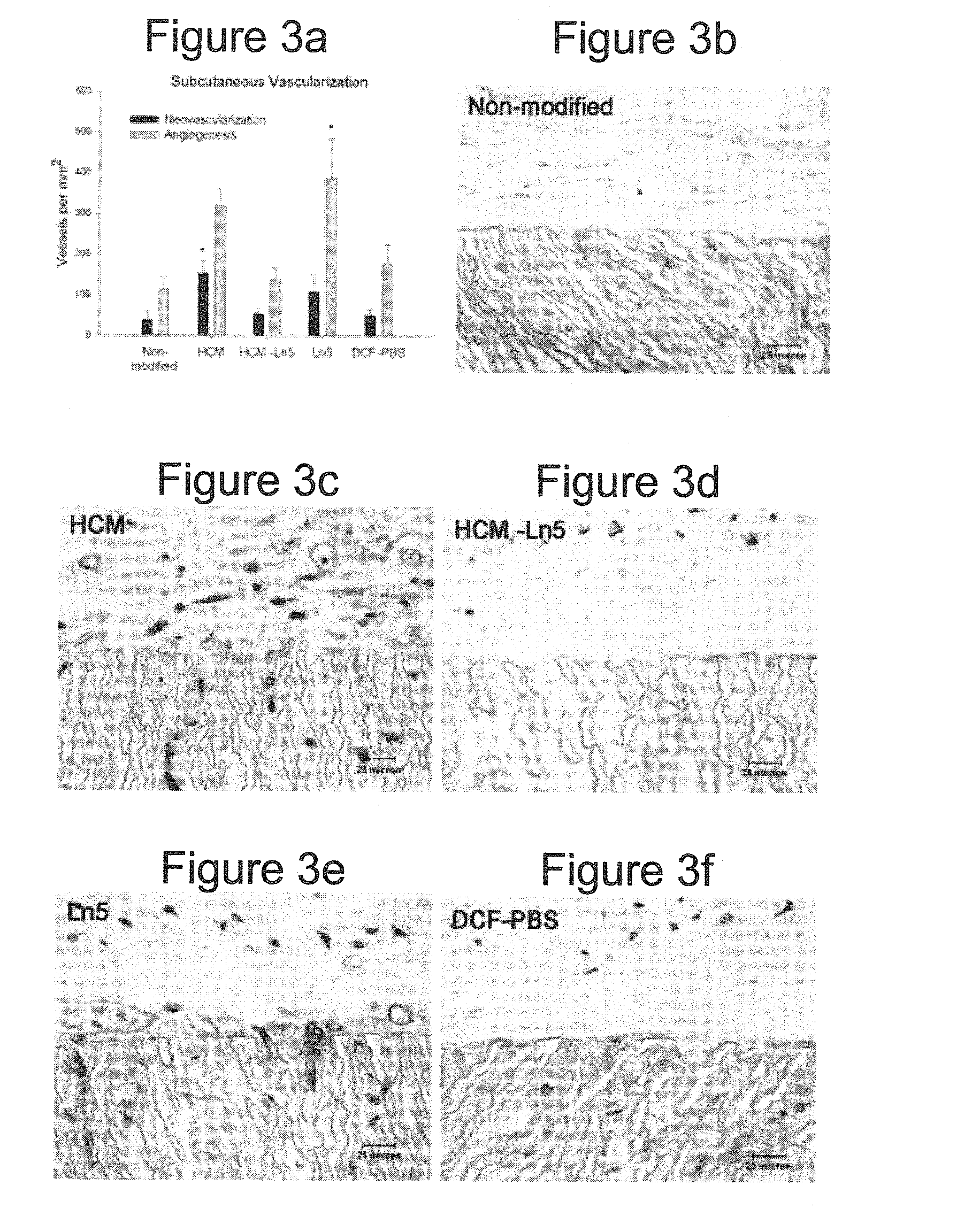
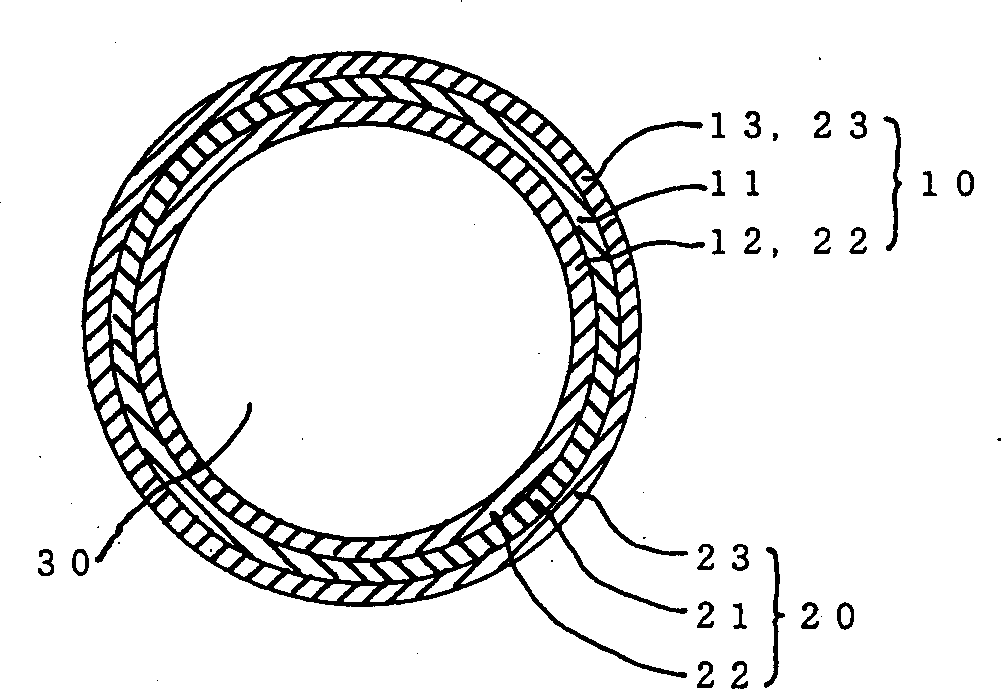
Implantable medical articles having laminin coatings and methods of use
InactiveUS20110244014A1Function increasePromote blood vessel formationSaccharide peptide ingredientsCoatingsLamininMedical device
Owner:WILLIAMS STUART K +3
Enzyme-mediated modification of fibrin for tissue engineering: fibrin formulations with peptides
InactiveUS7241730B2Efficacious platformEnhanced andPeptide/protein ingredientsTransferasesCell Surface ProteinsADAMTS Proteins
Heparin-binding regions of several proteins, such as neural cell adhesion molecule, fibronectin, laminin, midkine, and anti-thrombin III have been shown to promote neurite extension on two-dimensional surfaces. The effect of heparin-binding peptides on neurite extension through three-dimensional matrices was investigated by culturing embryonic chick dorsal root ganglia (DRG) within fibrin gels containing chemically attached heparin-binding peptide (HBP). The length of neurites within fibrin gels containing cross-linked HBP was increased by more than 70% over extension through fibrin gels containing no peptide. The HBP sequence of antithrombin III was incorporated into the fibrin gel as the C-terminal domain of a bidomian, chimeric peptide; the N-terminal second domain of this peptide contained the ∀2-plasmin inhibitor substrate for Factor XIIIa. Factor XIIIa, a transglutaminase, was used to chemically attach the HBP-containing chimeric peptide to the fibrin gels during polymerization. The amount of HBP cross-linked into the fibrin gels was determined, after degradation by plasmin using gel permeation chromatography, to be approximately 8 moles of peptide per mole fibrinogen. A peptide (HBP), where the cross-linking glutamine was replaced with glycine, showed no increase in extension in comparison with fibrin gels. The additional of heparin to the gel percursors resulted in no increase in neurite extension in comparison with fibrin gels. HBPs promote neurite extension by binding to cell surface proteoglycans on the DRG.
Owner:UNIV ZURICH +1
Cross-linked hyaluronic acid-laminin gels and use thereof in cell culture and medical implants
InactiveUS20060024373A1Easy to useConvenient environmentStentsPowder deliveryCross-linkTransplantation
The present invention concerns universal biocompatible matrices comprising cross-linked hyaluronic acid-laminin gels useful for clinical applications including as implants, for tissue engineering as well as in biotechnology. The gel matrices according to the present invention may be used clinically either per se or as a cell bearing implant, e.g., stem cells or endothelial cells, optionally containing bioactive molecules or drugs. The gels are particularly useful for use for transplantation or as a coating or a scaffold for use on medical devices including stents.
Owner:N V R LABS
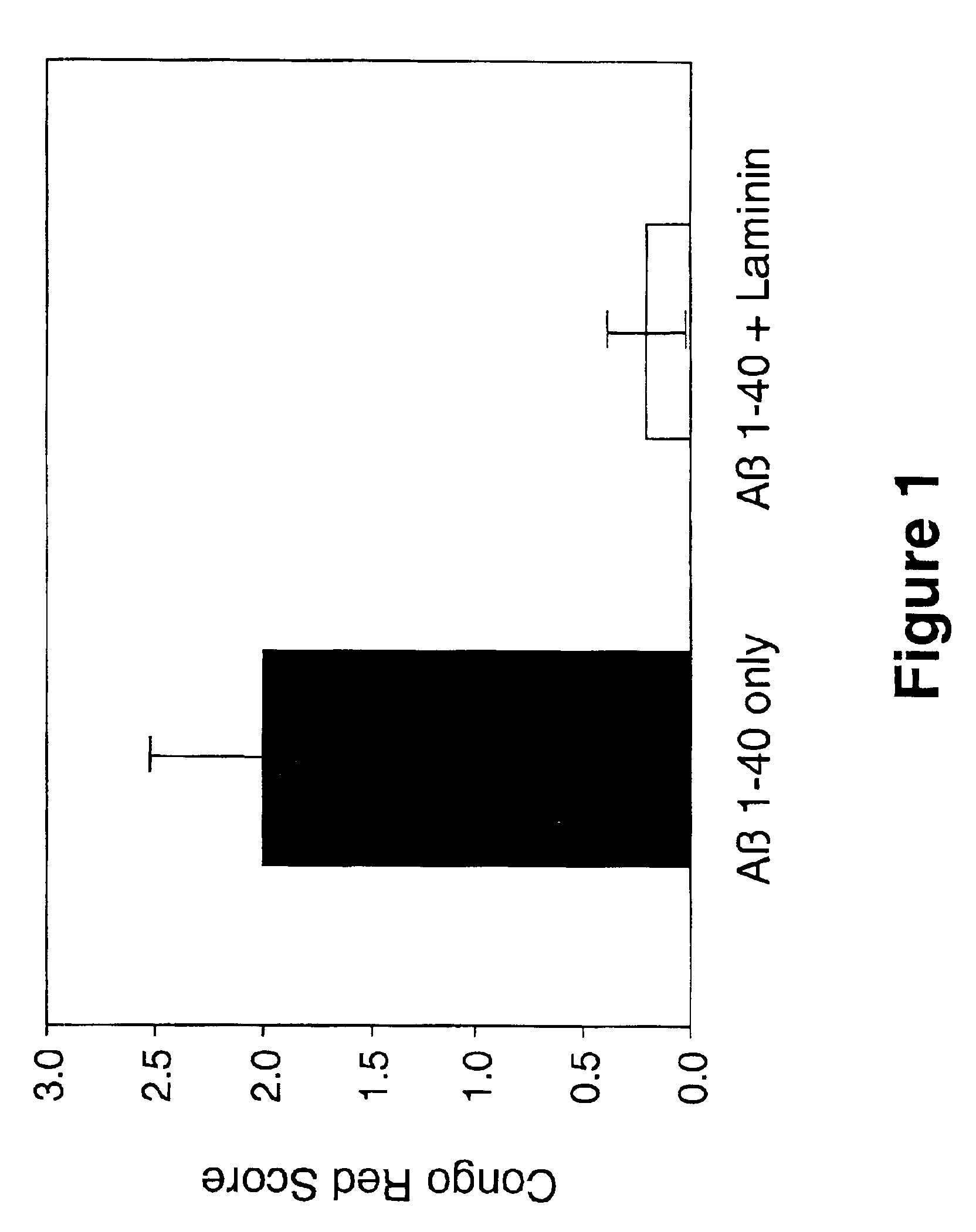
Therapeutic applications of laminin and laminin-derived protein fragments
InactiveUS20020111309A1Inhibiting amyloidosisPreventing amyloid depositionConnective tissue peptidesPeptide/protein ingredientsAmyloid betaKilodalton
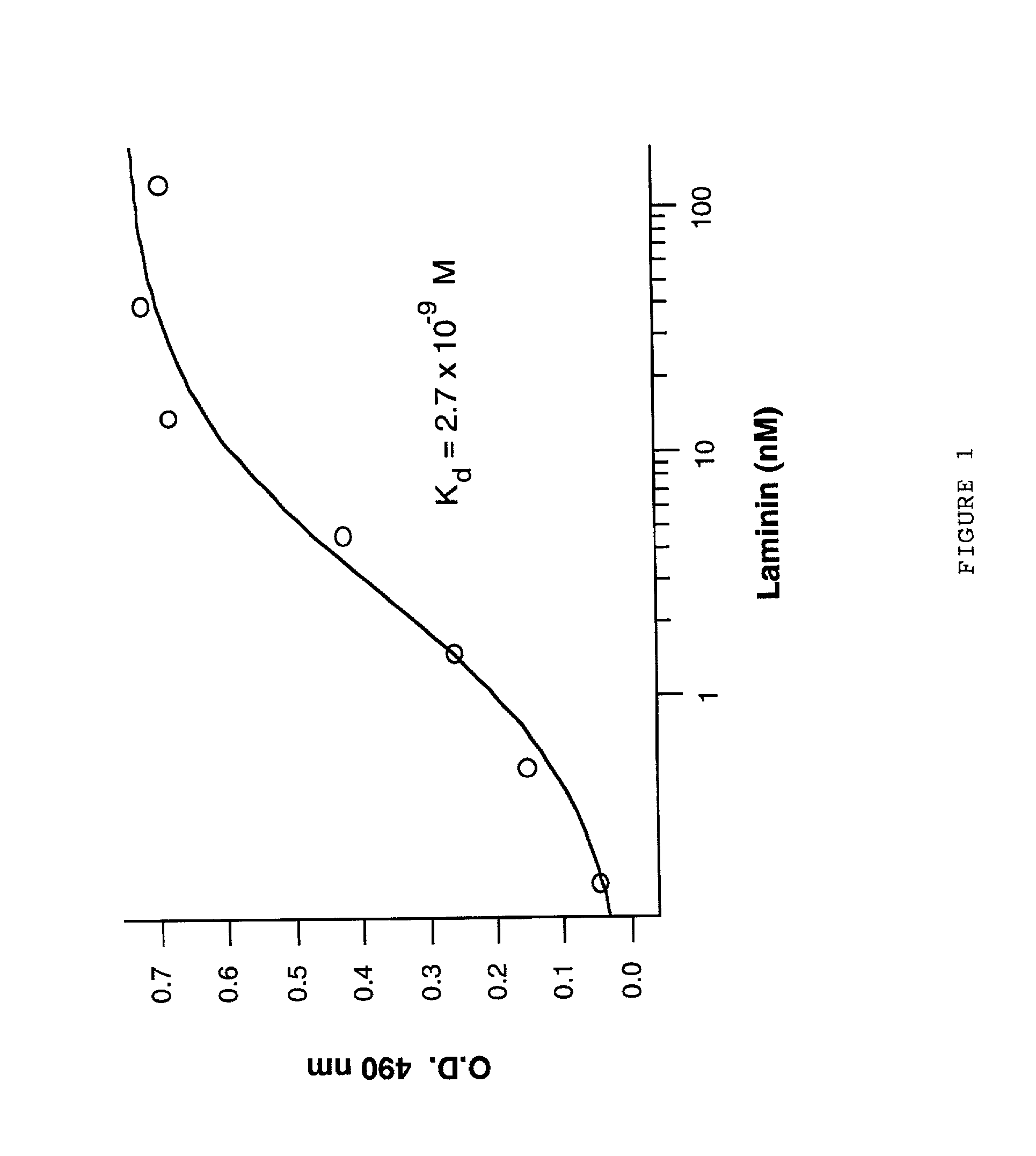
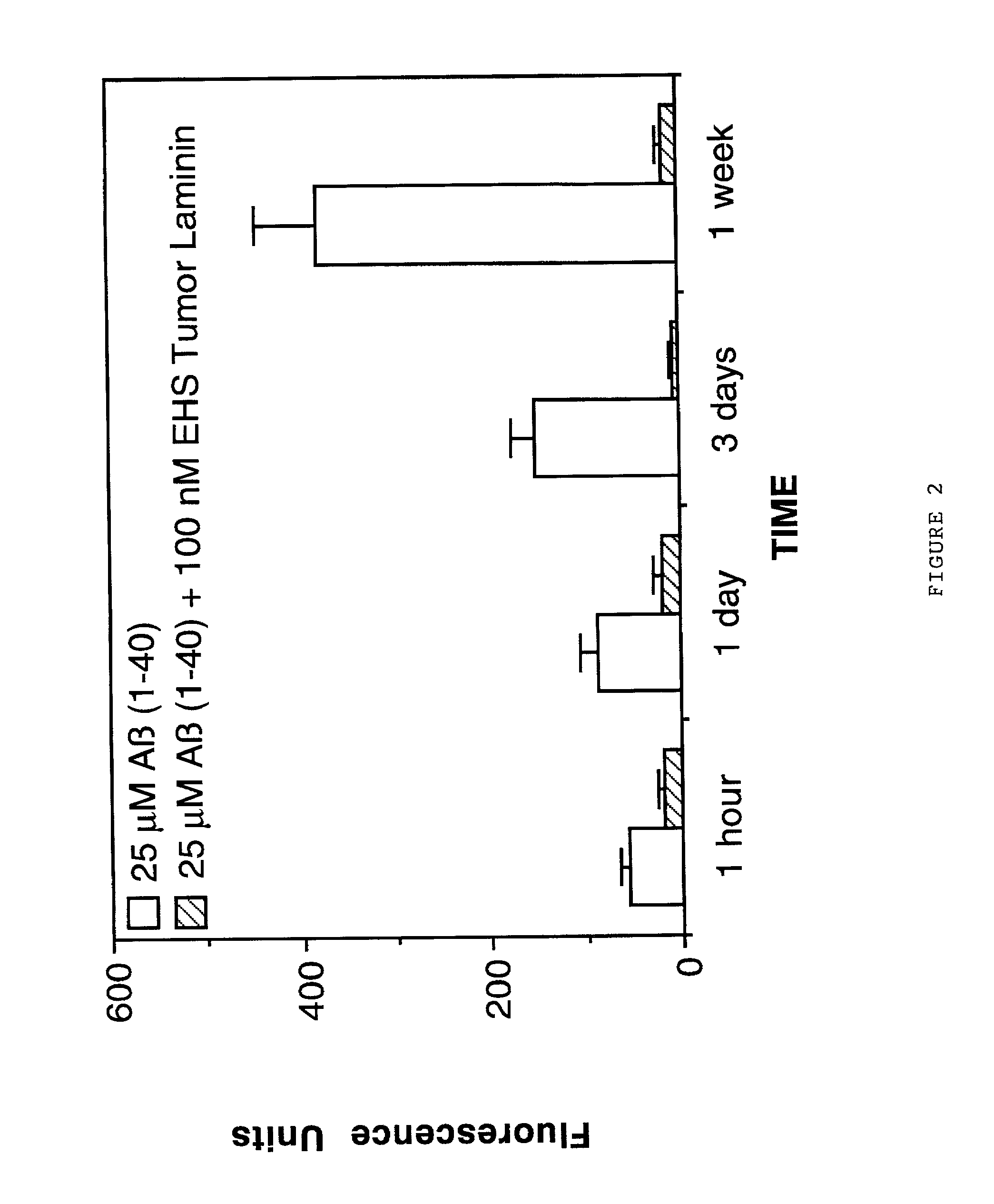
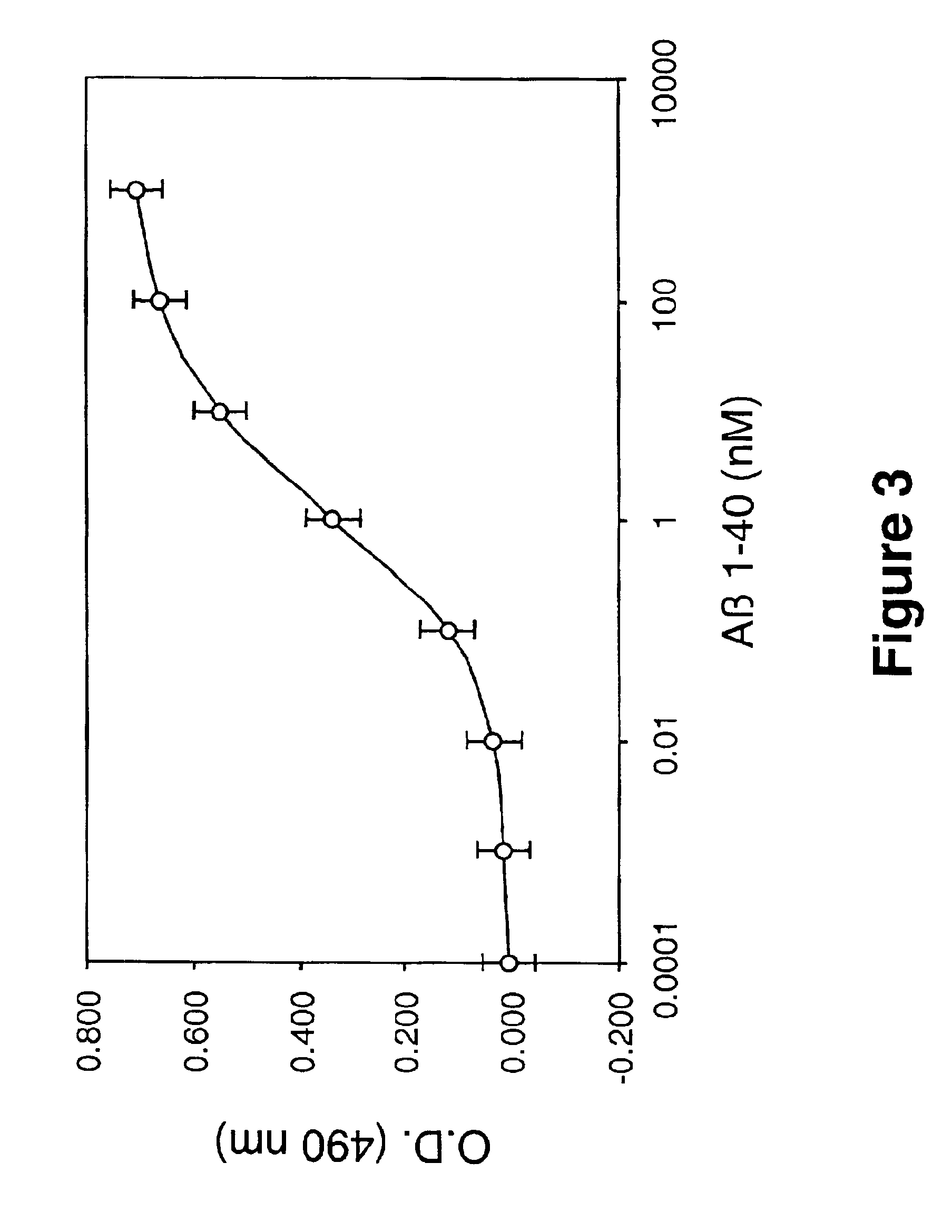
Laminin and specific laminin-derived protein fragments are disclosed as potent inhibitors of Alzheimer's disease type amyloidoses. A specific region is identified within laminin which interacts with the Alzheimer's disease beta-amyloid protein and contributes to the observed inhibitory and therapeutic effects. A prominent ~130 kilodalton band in laminin was found in human serum and cerebrospinal fluid which primarily interacted with Abeta as determined by ligand blotting methodology. This ~130 kilodalton laminin fragment is known as the E8 fragment and is also believed to consist of the globular domains of the laminin A chain. The interaction of specific laminin fragments such as the newly discovered ~130 kDa protein is believed to bind Abeta in biological fluids and keep it in a soluble state.
Owner:UNIV OF WASHINGTON
Hydrogel bioscaffoldings and biomedical device coatings
Bioscaffoldings formed of hydrogels that are crosslinked in situ in an infarcted region of the heart (myocardium) by a Michael's addition reaction or by a disulfide bond formed by an oxidative process are described. Each of the bioscaffoldings described includes hyaluronan as one of the hydrogel components and the other component is selected from collagen, collagen-laminin, poly-1-lysine, and fibrin. The bioscaffolding may further include an alginate component. The bioscaffoldings may have biofunctional groups such as angiogenic factors and stem cell homing factors bound to the collagen, collagen-laminin, poly-1-lysine, or fibrinogen hydrogel component. In particular, the biofunctional groups may be PR11, PR39, VEGF, bFGF, a polyarginine / DNA plasmid complex, or a DNA / polyethyleneimine (PEI) complex. Additionally, the hydrogel components may be injected into the infarct region along with stem cells and microspheres containing stem cell homing factors. The bioscaffolding may be formed on a stent or a cardiac medical device.
Owner:ABBOTT CARDIOVASCULAR
Peptides for the treatment of Alzheimer's disease and other beta-amyloid protein fibrillogenesis disorders
InactiveUS6933280B2Enhance clearance and removalConnective tissue peptidesNervous disorderAβ amyloidMedicine
A pharmaceutical composition, or pharmaceutical agent for treating Aβ amyloidosis in a patient, having at least one selected laminin peptide or fragment thereof from the group AG73 (SEQ ID NO:1), C-16 (SEQ ID NO:2), A-13 (SEQ ID NO:3), HA3G47 (SEQ ID NO:4), HA3G58 (SEQ ID NO:5), HA3G67 (SEQ ID NO:6), HA3G74 (SEQ ID NO:7), HA3G76 (SEQ ID NO:8), HA3G79 (SEQ ID NO:9), HA3G83 (SEQ ID NO:10), A4G82 (SEQ ID NO:11), A5G15 (SEQ ID NO:12), A5G56 (SEQ ID NO:13), A5G80 (SEQ ID NO:14), A5G81 (SEQ ID NO:15), A5G82 (SEQ ID NO: 16), A5G84 (SEQ ID NO:17), A5G101 (SEQ ID NO:18), and A5G109 (SEQ ID NO:19) (Sequence Group A); and a method for enhancing Aβ amyloid fibril formation, or of forming amyloid-plaque like deposits in vitro, including incubating Aβ 1-40 or Aβ 1-42 with a selected laminin-derived polypeptide from the group A-13 (SEQ ID NO:3), HA3G47 (SEQ ID NO:4), HA3G58 (SEQ ID NO:5), HA3G83 (SEQ ID NO:10), LAM-L (SEQ ID NO:20), A4G10 SEQ ID NO:21), A4G46 (SEQ ID NO:22), A4G47 (SEQ ID NO:23), A4G84 (SEQ ID NO:24), A4G92 (SEQ ID NO:25), A4G107 (SEQ ID NO:26), A5G3 (SEQ ID NO:27), A5G10 (SEQ ID NO:28), A5G27 (SEQ ID NO:29), A5G33 (SEQ ID NO:30), A5G65 (SEQ ID NO:31), A5G77 (SEQ ID NO:32), A5G87 (SEQ ID NO:33), A5G90 (SEQ ID NO:34) and A5G111 (SEQ ID NO:35) (Sequence Group C).
Owner:UNIV OF WASHINGTON
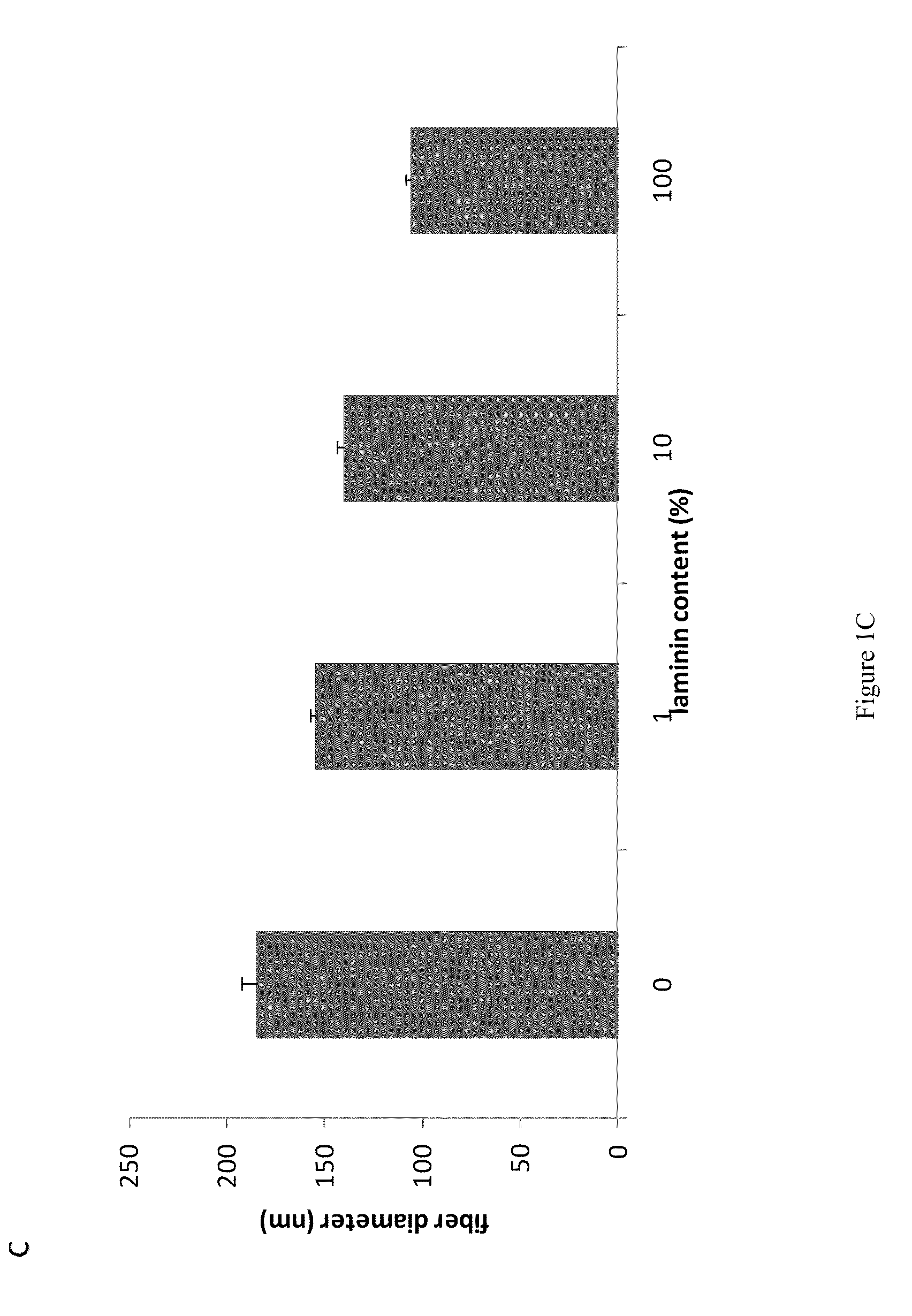
Compositions and Methods for Making and Using Laminin Nanofibers
InactiveUS20100120115A1Extended shelf lifeHigh tensile strengthMonocomponent protein artificial filamentBioreactor/fermenter combinationsSpinningElectrospinning
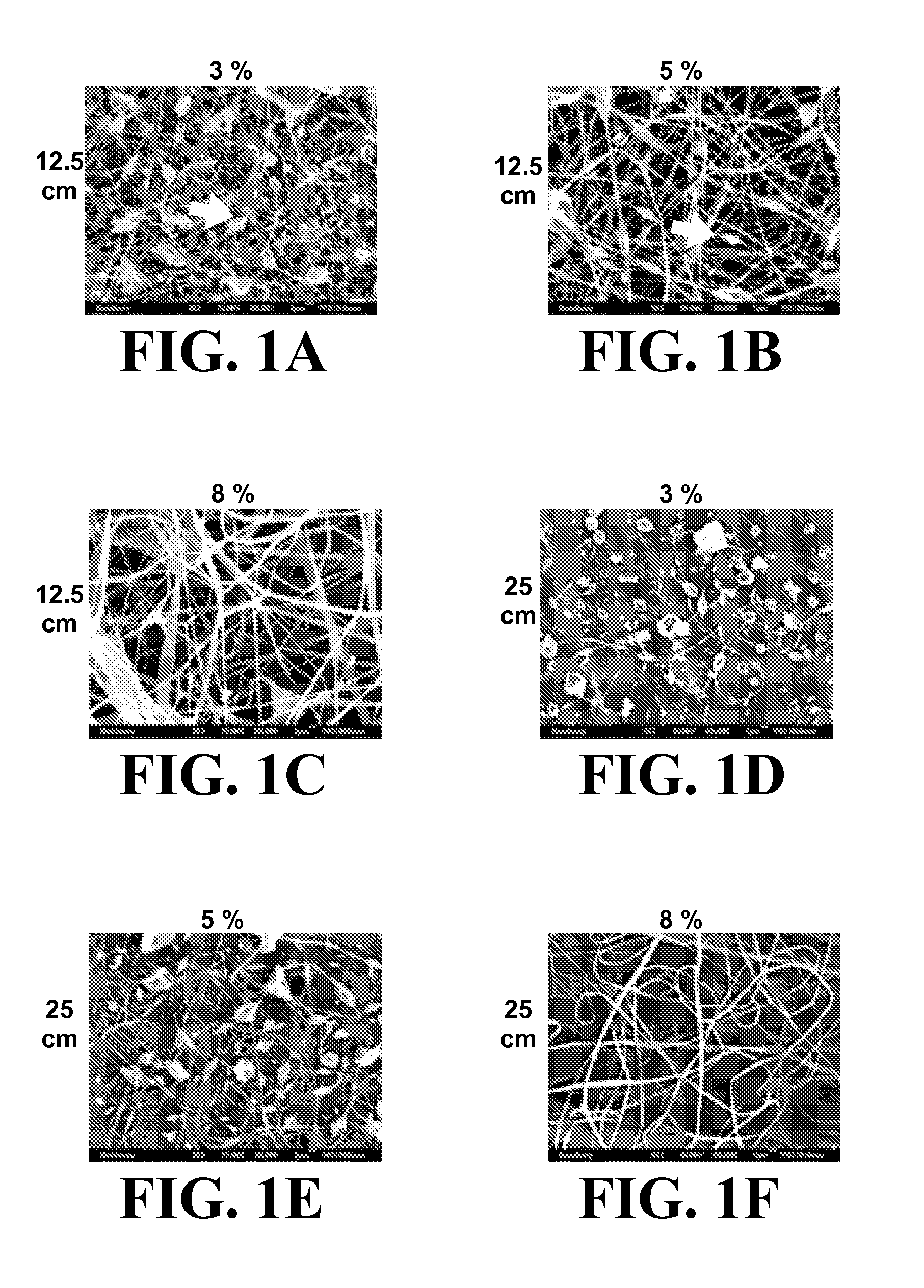
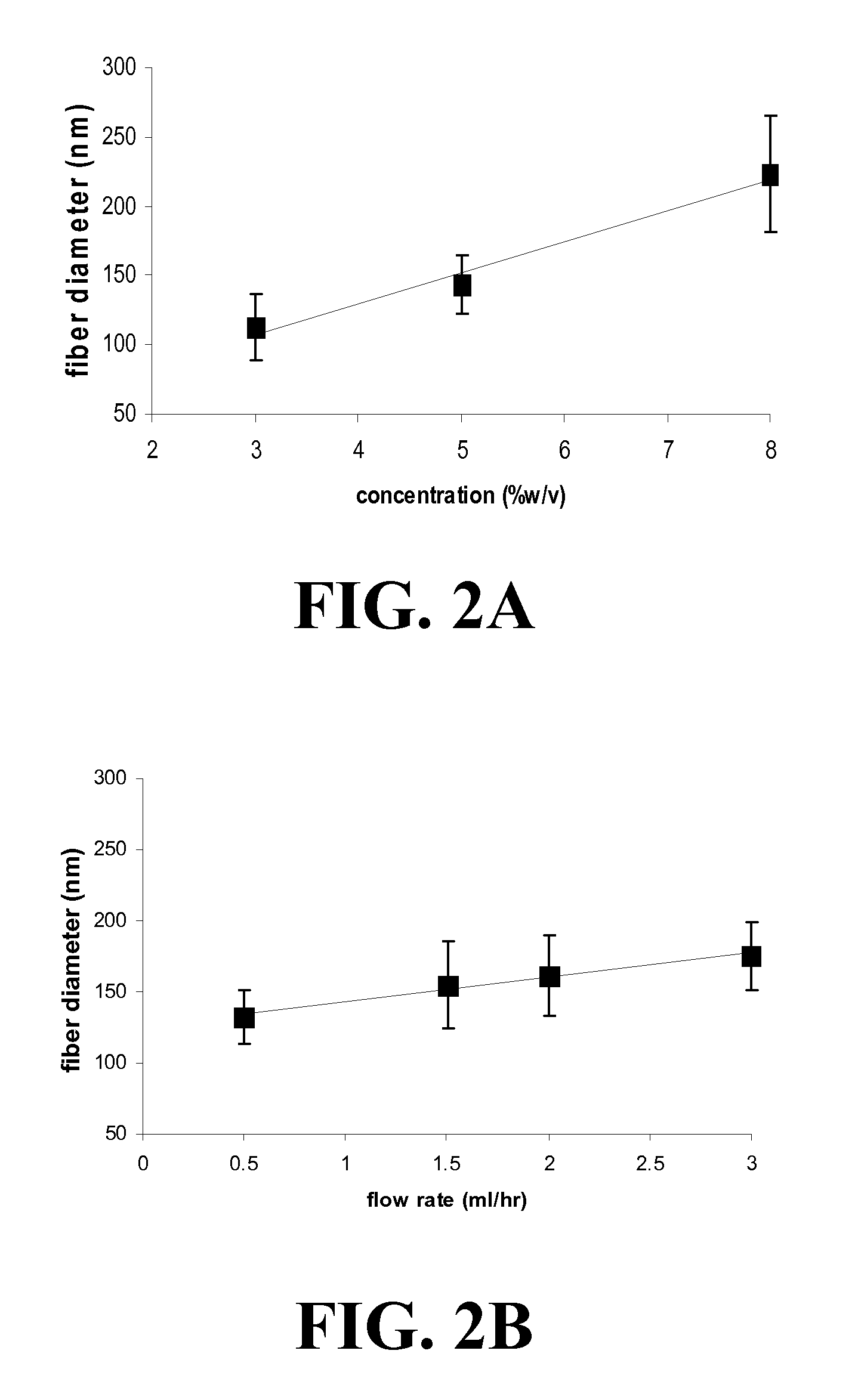
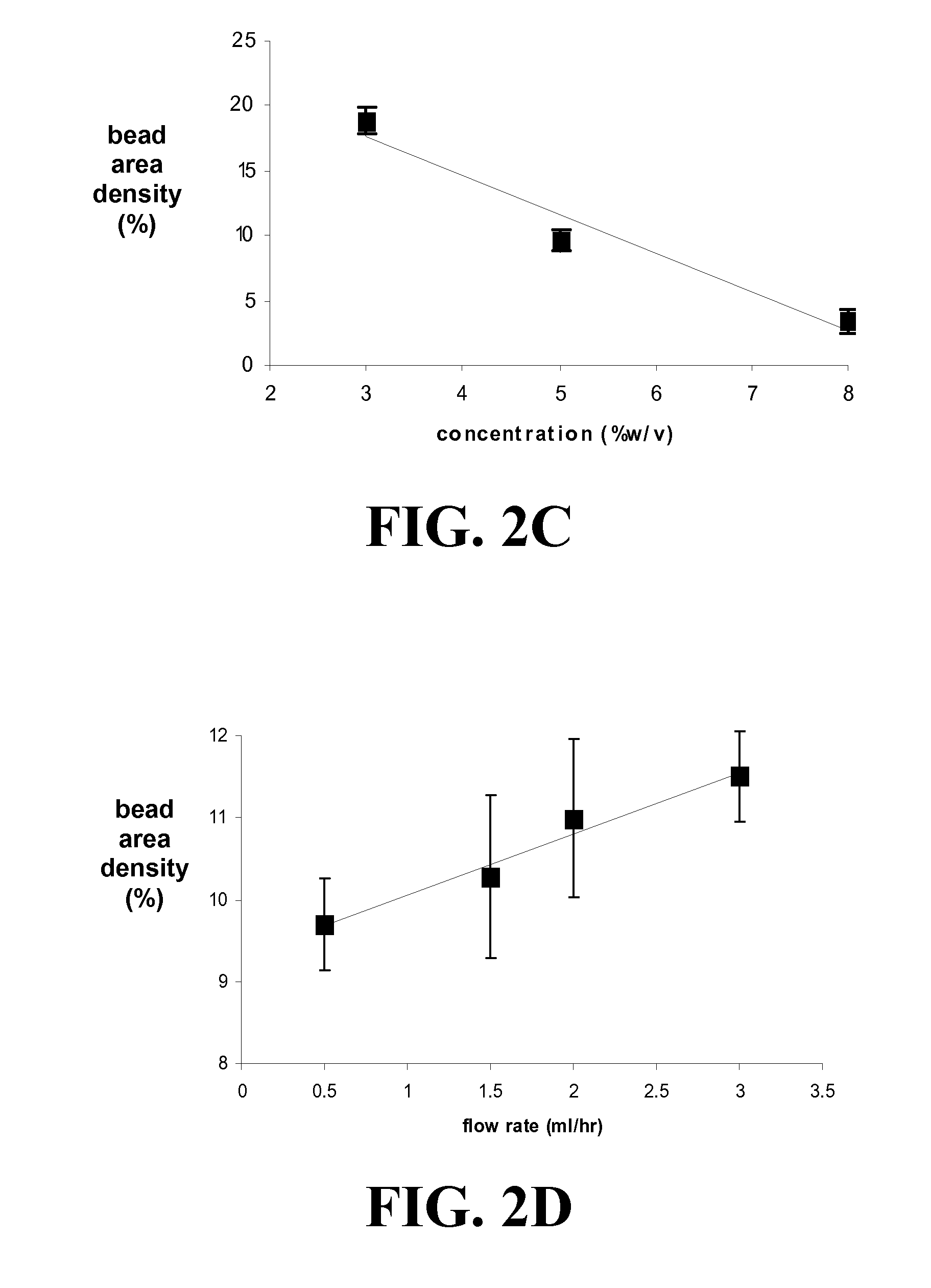
The present invention encompasses methodologies and parameters for the formation of nanofibrous (to microfibrous) laminin via electrospinning. The present application discloses conditions and appropriate parameters to synthesize laminin fibers from a diameter of about 10 nM to a diameter of over 1,000 nM via electrospinning.
Owner:UNIV OF VIRGINIA ALUMNI PATENTS FOUND
Artificial neural tube
The present invention is to provide an artificial tube for nerve which remains in a body until a nerve regenerates, does not remain in the body as a foreign body after regeneration of the nerve, induces axons regenerated from severed nerve stumps, promotes infiltration of blood capillaries from the body and promotes regeneration of nerve tissue, and a method for producing the same. The artificial tube for nerve has fine fibrous collagen bodies (30) in the lumen of a tube comprised of a biodegradable and absorbable material, the voids inside the fine fibrous collagen bodies being filled with laminin.
Owner:TAPIC INT +1
Compositions and Methods for Stem Cell Expansion and Differentiation
InactiveUS20070280989A1Free from damagePositively chargedBiocideGenetic material ingredientsCross-linkProgenitor
The present invention relates to compositions comprising stem cells and partially committed progenitor cells and to methods of controlling cell proliferation and differentiation, which can be used for expansion of stem cells and their subsequent differentiation. The present invention provides expanded population of essentially undifferentiated stem cells, which are useful in clinical procedures involving stem cell therapy, and a population derived thereof of which at least part of the cells are differentiated. The cells can be used per se, as a part of a cell-bearing composition comprising cross-linked hyaluronic acid-laminin gels or as a part of a composite implant for tissue regeneration.
Owner:NVR LABS
Method and composition for angiogenesis inhibition
InactiveUS20050271650A1Inhibit angiogenesisReduced affinitySenses disorderPeptide/protein ingredientsLamininAngiogenesis Inhibition
The invention features methods and compositions for inhibiting angiogenesis in a tissue or detecting angiogenesis using antagonists of denatured or proteolyzed forms of laminin.
Owner:CELL MATRIX
Blocking the migration or metastasis of cancer cells by affecting adhesion proteins and the uses of new compounds thereof
ActiveUS20100004190A1Growth inhibitionReduce adhesionBiocideSugar derivativesDiseaseLymphatic Spread
This invention provides methods, processes, compounds and compositions for modulating the gene expression and modulating the secretion, expression, or synthesis of adhesion proteins or their receptors to cure disease, wherein the modulating comprises positive and negative regulating; wherein comprises inhibiting cancer growth, wherein the adhesion proteins or receptors comprise fibronectin, integrins family, Myosin, vitronectin, collagen, laminin, Glycosylation cell surface proteins, polyglycans, cadherin, heparin, tenascin, CD 54, CAM, elastin and FAK; wherein the methods, processes, compounds and compositions are also for anti-angiogenesis; wherein the cancers comprise breast cancer, leukocyte cancer, liver cancer, ovarian cancer, bladder cancer, prostate cancer, skin cancer, bone cancer, brain cancer, leukemia cancer, lung cancer, colon cancer, CNS cancer, melanoma cancer, renal cancer or cervix cancer.
Owner:PACIFIC ARROW
Implantable medical articles having laminin coatings and methods of use
InactiveUS20060210603A1Function increasePromote blood vessel formationSenses disorderPeptide/protein ingredientsLamininFibrosis
Laminin-containing coatings for the surfaces of implantable medical devices are disclosed. The coatings promote the formation of vessels in association with the coated surfaces with minimal fibrotic resonse.
Owner:WILLIAMS STUART K +3
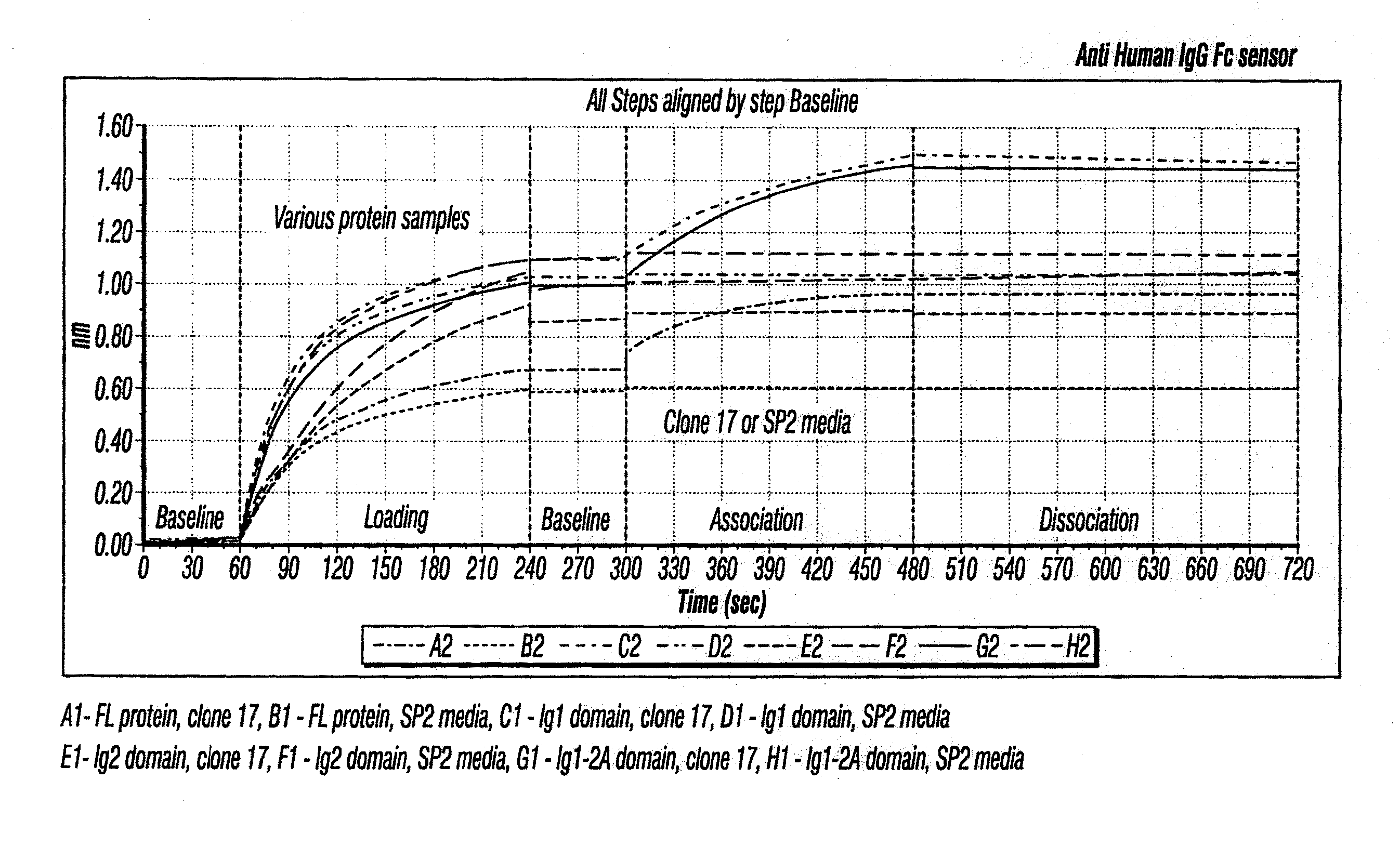
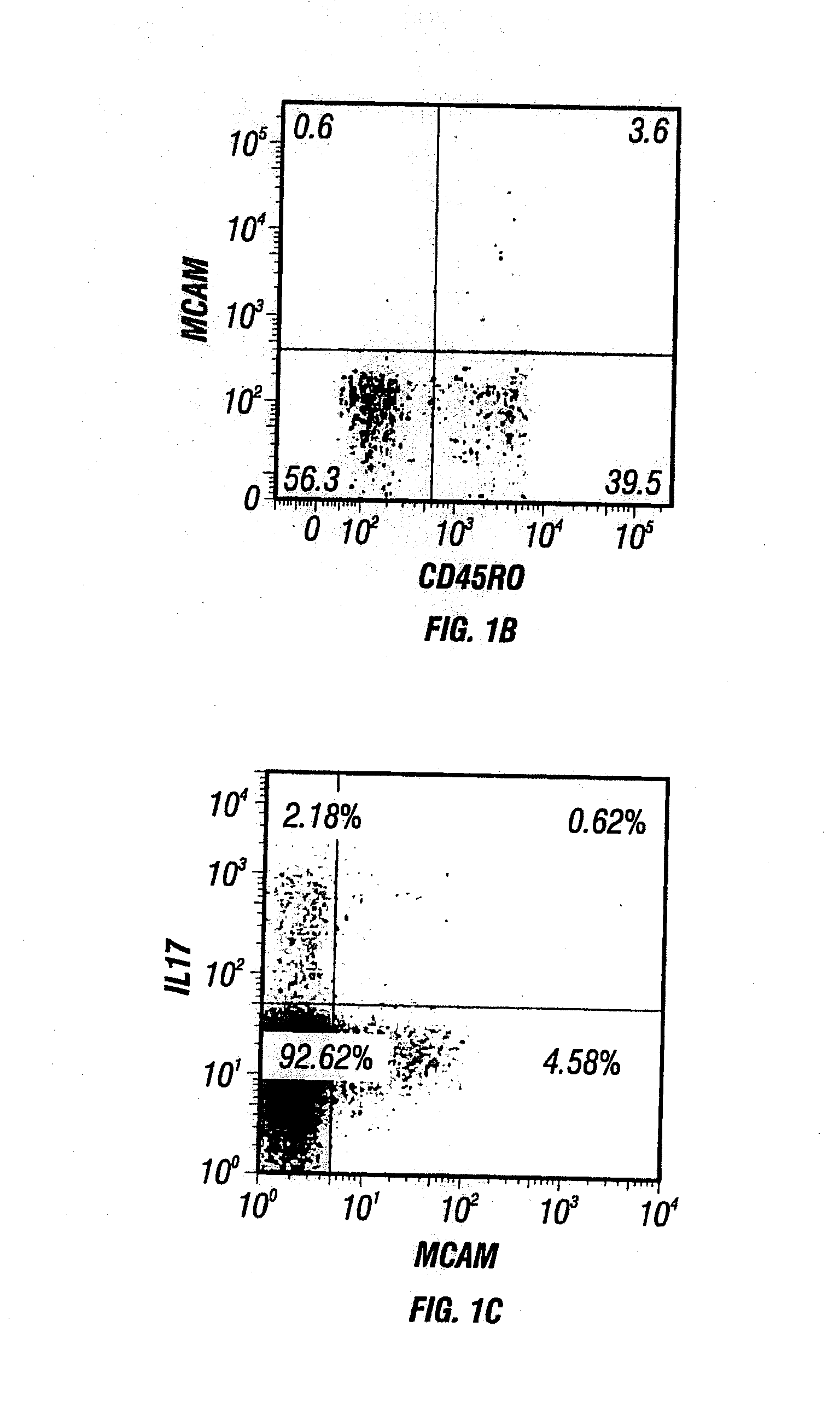
Anti-MCAM Antibodies And Associated Methods Of Use
Described herein are anti-MCAM antibodies and antigen binding fragments thereof that are capable of inhibiting the interaction between MCAM and its ligand, a protein comprising a laminin α-4 chain. These anti-MCAM antibodies and antigen binding fragments thereof may be useful for, for example, treating inflammatory conditions characterized by the infiltration of MCAM-expressing cells into a site of inflammation in the body.
Owner:PROTHENA BIOSCI LTD
Methods and compositions for growing corneal endothelial and related cells on biopolymers and creation of artifical corneal transplants
This invention discloses methods to attach and grow a monolayer of cultured human corneal endothelial cells onto the endothelial side of the stroma synthesized from biopolymer to generate a more bio-equivalent artificial cornea. The approaches will include the use of attachment and growth promoting agents such as fibronectin, laminin, RGDS, collagen type IV, bFGF conjugated with polycarbophil, and EGF conjugated with polycarbophil. The patent also describes a method to create a self-sustaining polymer containing adhesive molecules and growth factors to support the attachment and proliferation of cultured human corneal endothelial cells for corneal transplantation either as a half-thickness device or full-thickness button replacement. An approach for the implantation of cultured retinal pigment epithelial (RPE) cells into the sub-retinal space for treatment of age-related macular degeneration (ARMD) is disclosed in this invention. This method will enable the delivery of the transplanted RPE in a sheet of monolayer cells and will be better suited to perform their physiological function.
Owner:CELLULAR BIOENG
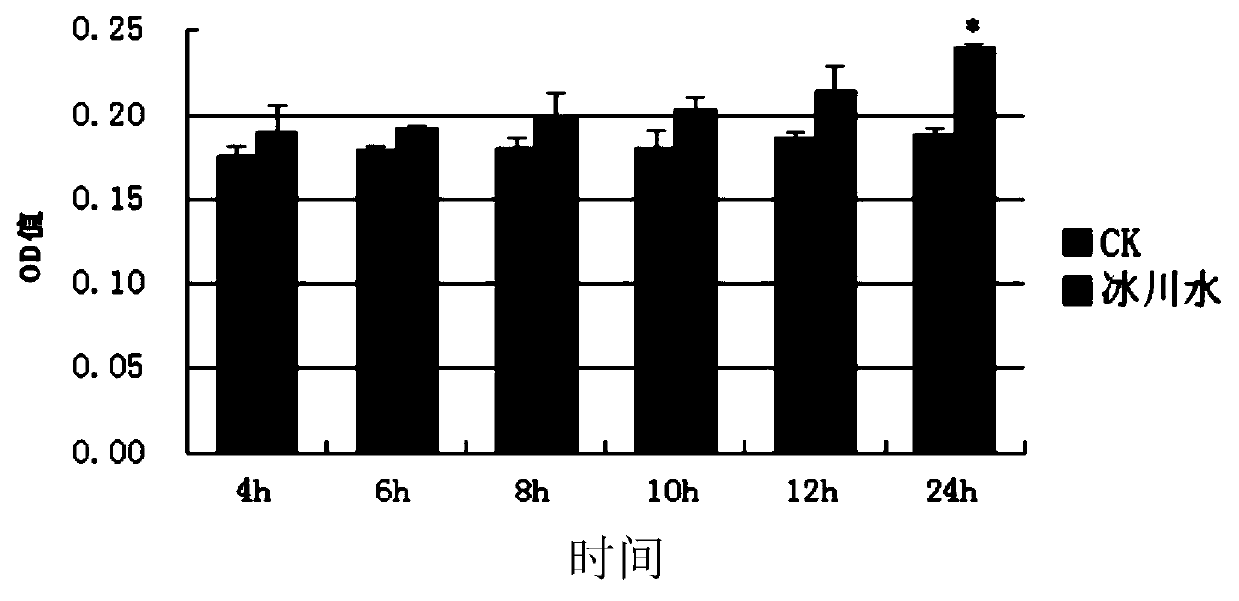
Application of glacial water in preparation of externally applied agent for skin and glacial water-containing externally applied agent for skin
InactiveCN105496827APromote proliferationStrong functional activityCosmetic preparationsToilet preparationsFiberHypopigmentation
The invention discloses application of glacial water in preparation of an externally applied agent for the skin and a glacial water-containing externally applied agent for the skin. It is found for the first time that glacial water can obviously promote propagation of fibroblasts, improve synthesis of fibroblast collagen I and elastin laminin of the skin and substantially inhibit propagation activity of human epidermal melanocytes (HEM-m) and formation of melanin. Glacial water is used as one or more selected from the group consisting of an anti-aging active component, a whitening active component and a moisture-retaining component of the externally applied agent for the skin; so the glacial water-containing externally applied agent for the skin is natural and safe and is capable of improving skin elasticity, resisting crease, repairing impaired skin, reducing hyperpigmentation and whitening the skin.
Owner:JALA GROUP CORPORATION
Recombinant laminin-521
ActiveUS20120156254A1Fast and economically efficient scale-upFacilitate scientificBioreactor/fermenter combinationsPeptide/protein ingredientsSingle cell suspensionCell culture media
The present disclosure related to isolated laminin-521, methods for making recombinant laminin-521, host cells that express recombinant laminin-521, and compositions containing laminin-521. Laminin-521 can maintain stem cells in vitro pluripotency, enable self-renewal, and enable single cell survival of human embryonic stem cells. When pluripotent human embryonic stem cells are cultured on plates coated with a matrix of recombinant laminin-521 (laminin 11), in the absence of differentiation inhibitors or feeder cells, the embryonic stem cells proliferate and maintain their pluripotency. It has also been discovered that human recombinant laminin-521 (laminin-11) provides single cell survival of stem cells after complete dissociation into a single cell suspension. Useful cell culture mediums containing at most 3.9 ng / ml of beta fibroblast growth factor (bFGF) are also described herein.
Owner:BIOLAMINA