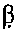Patents
Literature
366 results about "Testosterone" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
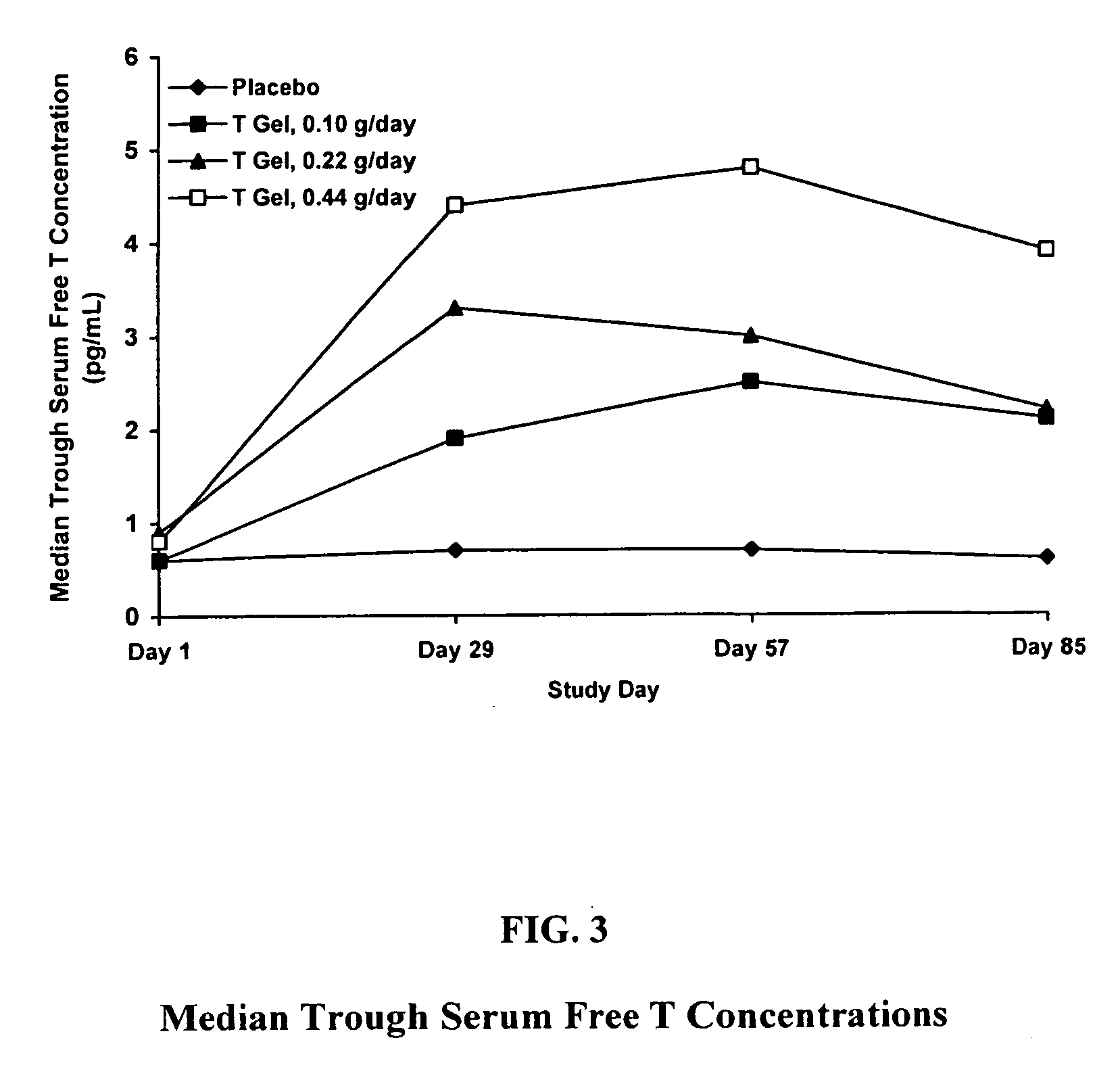
This medicated gel contains testosterone. It is used for hormone replacement in men who are not able to produce enough testosterone (e.g., hypogonadism). This medication is absorbed through the skin, enters your bloodstream, and helps your body reach normal testosterone levels.
Pharmaceutical composition and method for transdermal drug delivery
InactiveUS20050042268A1Improves transdermal penetrationSmall amountOrganic active ingredientsPharmaceutical delivery mechanismHormones regulationTransdermal medication
A pharmaceutical composition for transdermal administration of a hormone (e.g., testosterone), which includes urea and / or a derivative thereof as a penetration enhancer, and methods utilizing same for treating medical conditions in which elevating a hormone serum level is beneficial are disclosed.
Owner:AGIS INDUSTRIES (1983) LTD
Pharmaceutical composition and method for transdermal drug delivery
InactiveUS20050020552A1Increase concentrationImproves transdermal penetrationOrganic active ingredientsAerosol deliveryIsostearic acidHormones regulation
A pharmaceutical composition for transdermal administration of a hormone (e.g., testosterone), which includes isostearic acid as a penetration enhancer, and methods utilizing same for treating medical conditions in which elevating a hormone serum level is beneficial are disclosed.
Owner:AGIS INDUSTRIES (1983) LTD
Pharmaceutical composition and method for transdermal drug delivery
InactiveUS20050025833A1Increase concentrationImproves transdermal penetrationPowder deliveryOrganic active ingredientsAmmonium compoundsCompound (substance)
A pharmaceutical composition for transdermal administration of a hormone (e.g., testosterone), which includes a quaternary ammonium compound as a penetration enhancer, and methods utilizing same for treating medical conditions in which elevating a hormone serum level is beneficial are disclosed.
Owner:AGIS INDUSTRIES (1983) LTD
Method of increasing testosterone and related steriod concentrations in women
InactiveUS20070154533A1Improve bioavailabilityGood chemical stabilityOrganic active ingredientsOintment deliveryPhysiologyTestosterone
The present invention relates to methods, kits, combinations, and compositions for treating, preventing or reducing the risk of developing a testosterone-deficient disorder, or the symptoms associated with, or related to a testosterone-deficient disorder in a subject in need thereof. The present invention also relates to a method of administering a steroid in the testosterone synthetic pathway to a subject in need thereof.
Owner:LABORATORIES BESINS INTERNATIONAL SAS
Compounds and methods for the treatment of urogenital disorders
InactiveUS6987129B2Reduce painLess discomfortBiocidePeptide/protein ingredientsDiseaseFemale Sexual Arousal Disorder
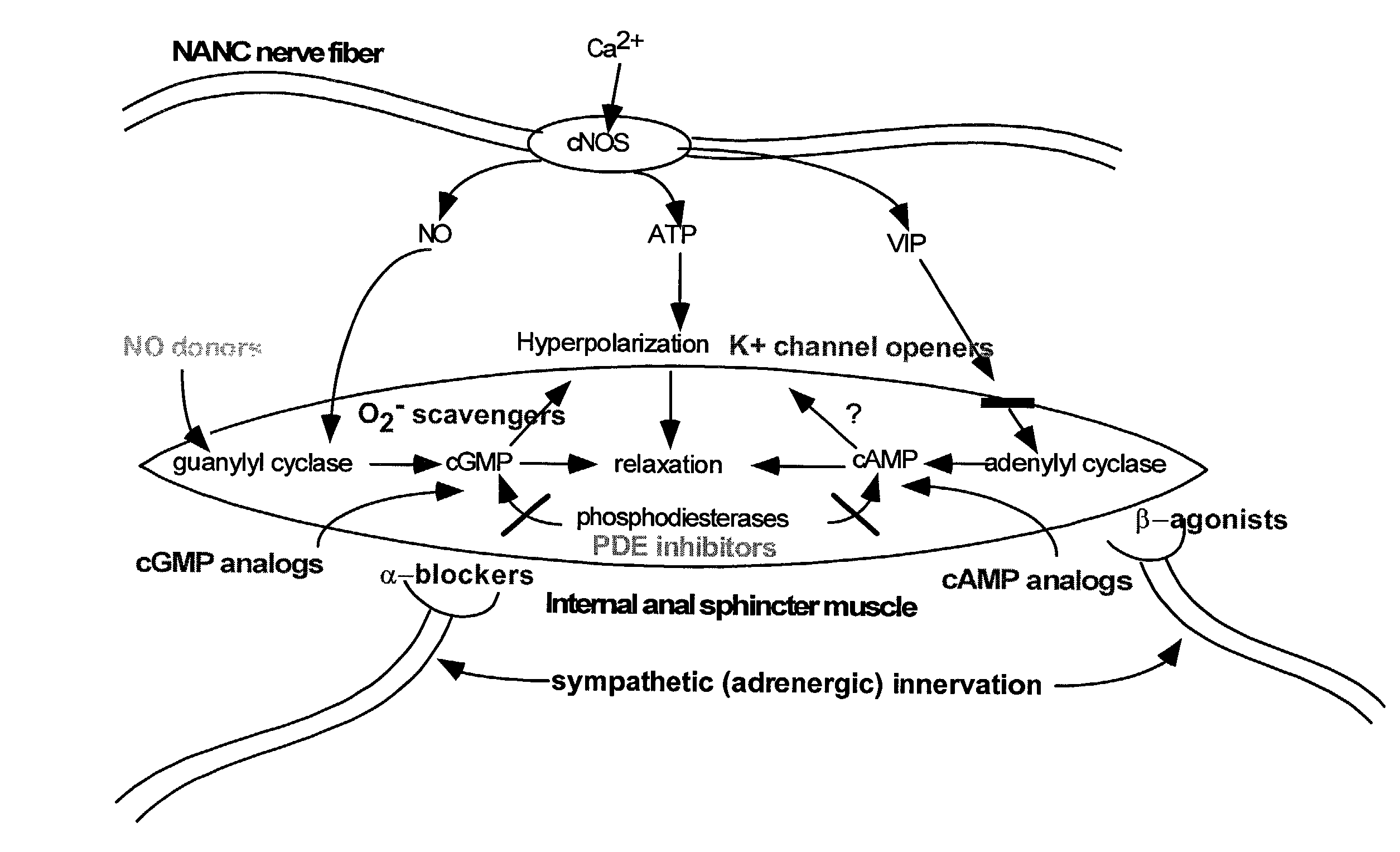
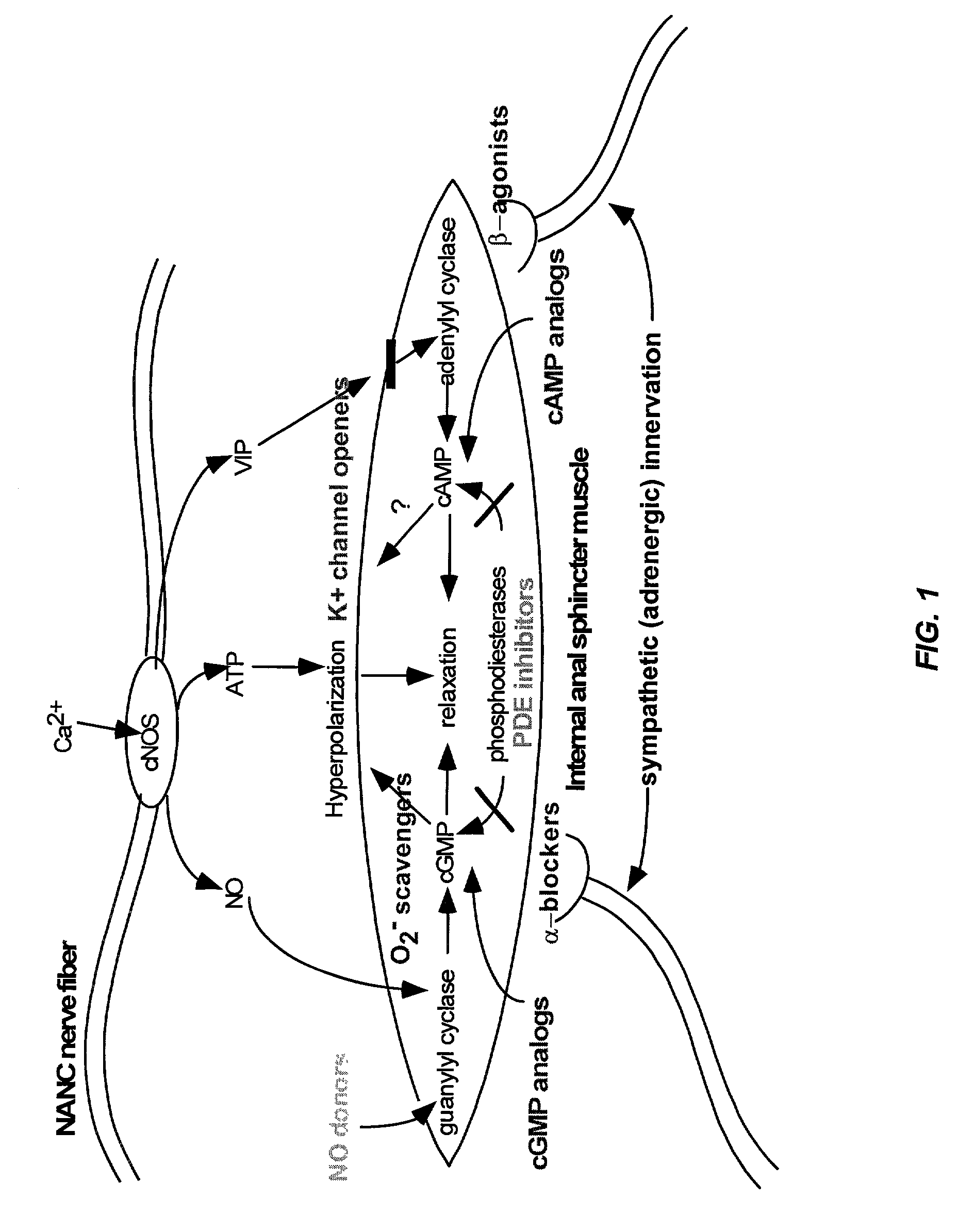
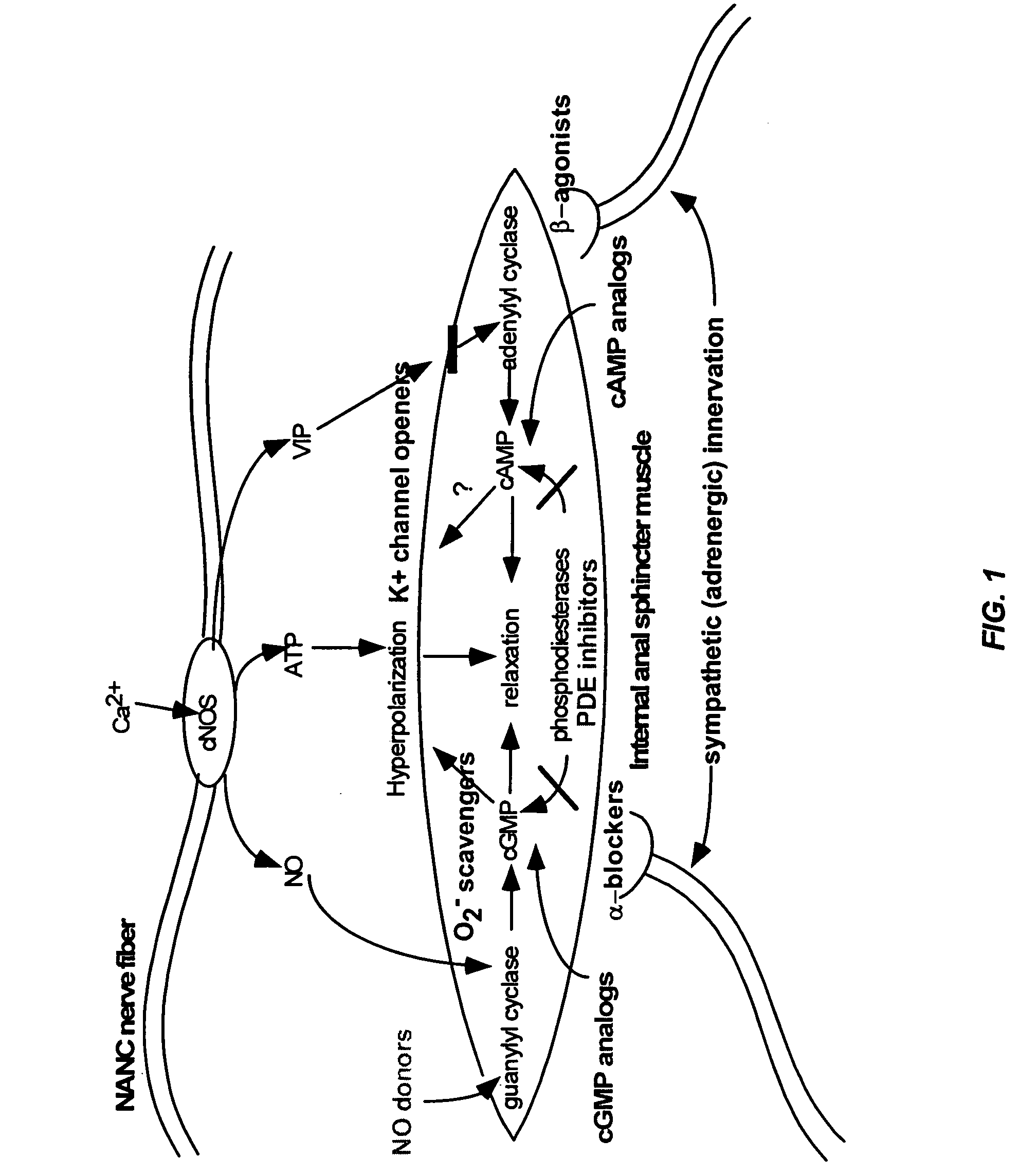
The present invention provides methods for treating a variety of urogenital disorders, such as, for example, vaginismus, dyspareunia, vulvodynia (including vulvar vestibulitis), interstitial cystitis, nonspecific urethriris (i.e., nonspecific pain and / or burning of the urinary tract) and sexual dysfunctions, such as, for example, female sexual arousal disorders and female sexual orgasmic disorders, using a variety of compounds, including, but not limited to, NO donors, calcium channel blockers, cholinergic modulators, α-adrenergic receptor antagonists, β-adrenergic receptor agonists, phosphodiesterase inhibitors, cAMP-dependent protein kinase activators (e.g., cAMP mimetics), superoxide scavengers, potassium channel activators, estrogen-like compounds, testosterone-like compounds, benzodiazepines, adrenergic nerve inhibitors, antidiarrheal agents, HMG-CoA reductase inhibitors, smooth muscle relaxants, adenosine receptor modulators, adenylyl cyclase activators, endothelin receptor antagonists, bisphosphonates and cGMP-dependent protein kinase activators (e.g., cGMP mimetics).
Owner:STREHKEHN INT LTD
Immobilized and activity-stabilized complexes of LHRH antagonists and processes for their preparation
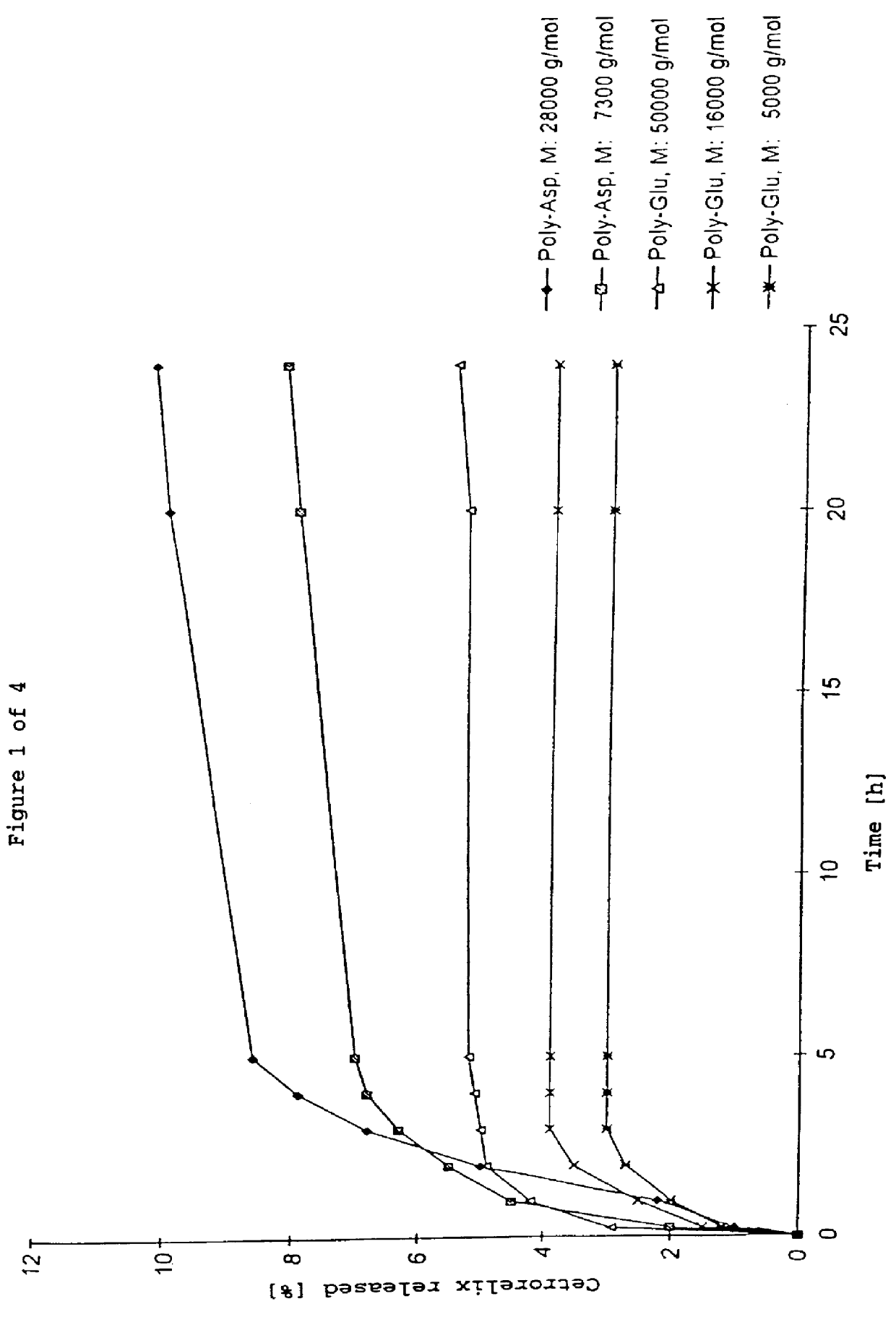
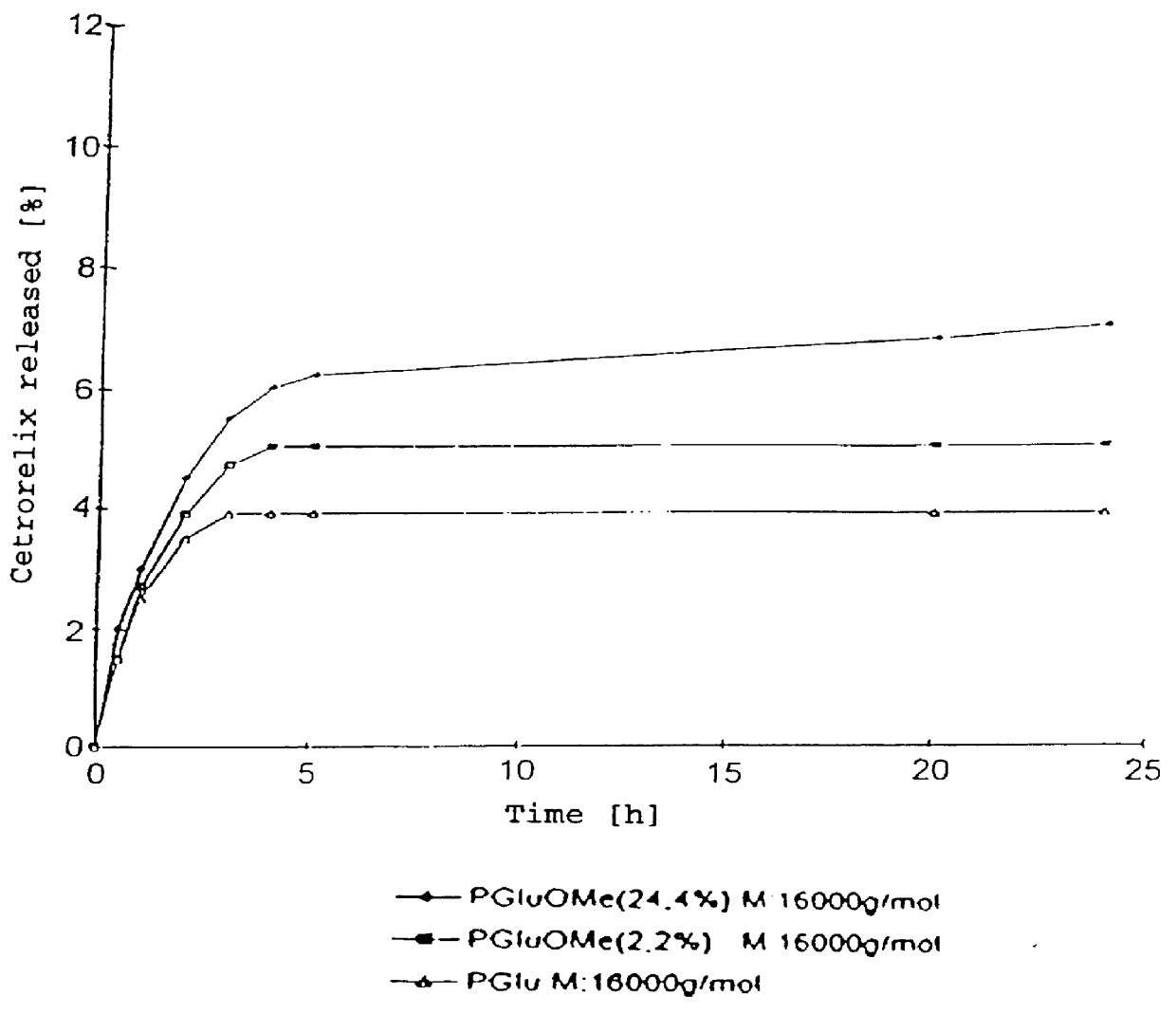
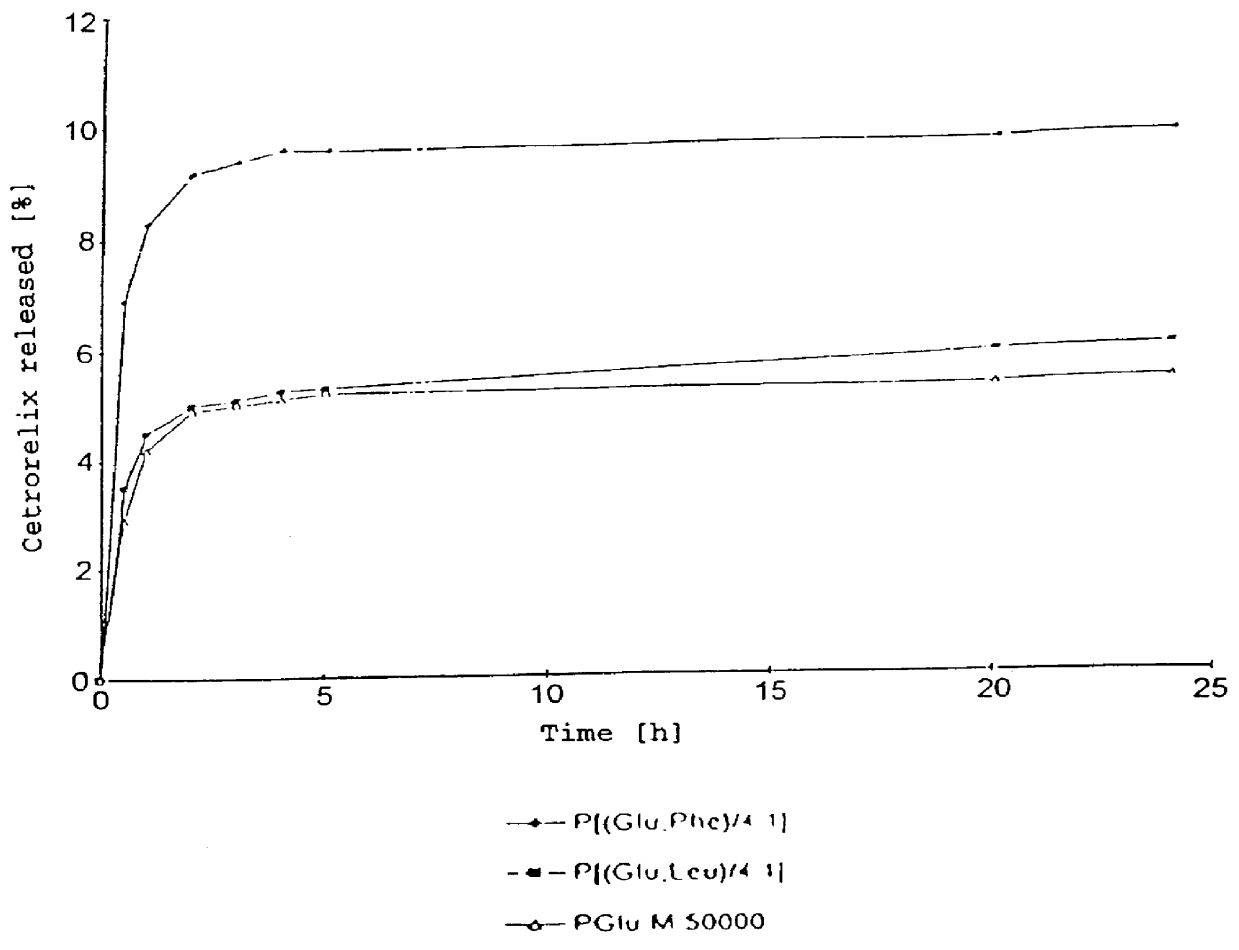
In this invention, a release-delaying system is to be developed for LHRH antagonists, in particular for cetrorelix, which allows the active compound to be released in a controlled manner over several weeks by complexation with suitable biophilic carriers. The acidic polyamino acids polyglutamic acid and polyaspartic acid were selected for complexation with cetrorelix. The cetrorelix polyamino acid complexes are prepared from aqueous solutions by combination of the solutions and precipitation of the complexes, which are subsequently centrifuged off and dried over P2O5 in vacuo. If complexes having a defined composition are to be obtained, lyophilization proves to be a suitable method. The cetrorelix-carboxylic acid complexes were also prepared from the aqueous solutions. In the random liberation system, the acidic polyamino acids poly-Glu and poly-Asp showed good release-delaying properties as a function of the hydrophobicity and the molecular mass of the polyamino acid. In animal experiments, it was possible to confirm the activity of the cetrorelix-polyamino acid complexes as a depot system in principle. It is thus possible by complexation of cetrorelix with polyamino acids to achieve testosterone suppression in male rats over 600 hours. The release of active compound here can be controlled by the nature and the molecular mass of the polymers.
Owner:ZENTARIS GMBH
Buccal, polar and non-polar spray containing testosterone
InactiveUS20050180923A1Fast absorptionRapid onsetAerosol deliveryOrganic non-active ingredientsSolventPharmacology
Buccal aerosol sprays or capsules using polar and non-polar solvent have now been developed which provide testosterone for rapid absorption through the oral mucosa, resulting in fast onset of effect. The buccal polar compositions of the invention comprise formulation I: aqueous polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; formulation II: aqueous polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optionally flavoring agent, and propellant; formulation III: non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; and formulation IV: non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optional flavoring agent, and propellant; formulation V: a mixture of a polar and a non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; formulation VI: a mixture of a polar and a non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optional flavoring agent, and propellant.
Owner:NOVADEL PHARMA
Commercial scale production methods for transdermal hormone formulations
InactiveUS20120046264A1The right amountBiocidePharmaceutical delivery mechanismPhysiologyHormones regulation
Methods for commercial production of transdermal formulations comprising a hormone compound are provided. In particular, methods for commercial scale production under an inert atmosphere of a transdermal formulation comprising a therapeutically effective amount of a hormone, preferably a testosterone compound, useful for the treatment of hypoactive sexual desire disorder (HSDD) in postmenopausal women are provided.
Owner:ANI PHARMA
Emulsion formulations
A SEDDS or SMEDDS or SNEDDS formulation for drug delivery of a lipophilic therapeutic agent, providing enhanced modulation of solubility, stability, absorption, metabolism, and / or pharmacokinetic profile of the therapeutic agent by formulation with a lipophilic surfactant, a hydrophilic surfactant, one or more solubilizers and, optionally, digestible oils, resulting in higher bioavailability of the therapeutic agent administered to a subject in need of such therapeutic agent. Also described are pharmaceutical compositions containing the formulations and methods of making and methods of using the formulations and pharmaceutical compositions. Formulations of the disclosure can be constituted to minimize the synthesis of dihydrotestosterone when the therapeutic agent includes testosterone or testosterone esters.
Owner:MARIUS PHARMA LLC
Trace elements
The invention discloses a trace element solution, which comprises at least one metal selected from the group comprising selenium, copper, zinc, manganese and chromium; and at least one component selected from the group comprising a vitamin, a vaccine, a growth stimulant, a dewormer, iron dextran, an antibiotic and a synchronization preparation. The synchronization preparation is a combination of injectable hormonal preparations, inplantable hormonal preparations, intravaginal hormonal preparation and other slow release hormonal preparation. The antibiotics include oral, injectable and implantable theurapeutic remedies. The vaccine includes antigens and a combination of antigens and adjuvents. The growth stimulants include zeranol, estradiol, testosterone, progesterone and trenbolone acetate. The dewormer includes macrocydic lactones, leramizoles, benzimidazoles and salicylanilides. The macrocydic lactones include doramectin, ivermectin, abamectin and moxidectin.
Owner:WARBURTON TECH
Clinical-grade human mesenchymal stem cell serum-free complete medium
ActiveCN103243071APromote growthLow toxicitySkeletal/connective tissue cellsInsulin-like growth factorCuticle
The invention relates to a human mesenchymal stem cell culture medium. According to the culture medium, the basal culture medium comprises the following components based on the final concentration: 1-2g / L of human serum albumin, 5-10mg / L of transferring, 2-8mg / L of fibronectin, 1-4mg / L of laminin, 50g / L of Fe(NO3)3.9H2O, 417g / L of FeSO4.7H2O, 1-3mu g / L of estradiol, 2-5mu g / L of testosterone, 1-3mu g / L of progesterone, 39.25-117.74 mu g / L of dexamethasone, 5-10mg / L of insulin, 376.36mg / L of riboflavin, 80.96-242.87mg / L of coenzyme A, 4.41-6.17mg / L of butanediamine, 1-2mg / L of taurine, 0.61-1.85mg / L of aminoethanol, 8.81-26.42mg / L of pyruvic acid, 3.78-7.56mu g / L of sodium selenate, 292.3-584.6mg / L of L-glutamine, 2-8mu g / L of vascular endothelial growth factor, 4-10mu g / L of epidermal growth factor, 4-10mu g / L of basic fibroblast growth factor, 1-5mu g / L of leukaemia inhibitory factor, 1-5mu g / L of insulin-like growth factor-I and 2-8mu g / L of stem cell factor. The culture medium does not contain the animal serum, the potential animal endogenous endotoxin or virus of the animal serum is eliminated, and the culture medium is conveniently applied to clinics.
Owner:QINGDAO RESTORE BIOTECHNOLOGY CO LTD
Progesterone/testosterone cream for erectile dysfunction
InactiveUS20070167418A1Optimize their medical careImprove erectile dysfunctionBiocideOrganic active ingredientsHormonal imbalancePhysiology
We have found a consistent pattern of hormonal abnormalities in patients with ERECTILE DYSFUNCTION. Our study group included men from age 21 to 88 years old. By helping to correct the hormonal imbalance of progesterone and testosterone towards normal values, most of the men in our study were able to obtain normal erectile function. By using a progesterone / testosterone cream we were able to help correct the hormonal imbalance commonly found in men with ERECTILE DYSFUNCTION.
Owner:FERGUSON STEVEN WALLACE
Therapeutic vaginal emollient
A topical composition for relief from certain menopausal, peri- and post-menopausal symptoms is disclosed, consisting of a low dosage of progesterone, testosterone and estriol in a pharmaceutically acceptable topical carrier in a weight ratio of from about 1:0.1:0.01 to about 1:0.1:0.02, respectively. In a preferred single dose, the three active ingredients are present as follows: (a) from about 20 to about 35 mg progesterone; (b) from about 2 to about 3.5 mg testosterone; and (c) from about 0.2 to about 0.7 mg estriol. A preferred carrier is cocoa butter, and a preferred single dose form is a vaginal suppository.
Owner:CHATROUX SYLVIA S
Modulation of solubility, stability, absorption, metabolism, and pharmacokinetic profile of lipophilic drugs by sterols
InactiveUS20110160168A1Enhance biological absorptionGood metabolic stabilityBiocideMetabolism disorderSterol esterPharmaceutical drug
A formulation for drug delivery, providing enhanced modulation of solubility, stability, absorption, metabolism, and / or pharmacokinetic profile of a lipophilic therapeutic agent by formulation with sterols and / or sterol esters, resulting in higher bioavailability of a therapeutic agent administered to a subject in need of such therapeutic agent. The formulation contains a therapeutic agent and a sterol or sterol ester, and can, optionally, further contain a solubilizer and / or an enhancing agent. Also described are pharmaceutical compositions containing the formulations and methods of making and methods of using the formulations and pharmaceutical compositions. Formulations of the disclosure can be constituted to minimize the synthesis of dihydrotestosterone when the therapeutic agent includes testosterone or testosterone esters.
Owner:MARIUS PHARMA LLC
Lactobacillus reuteri and its application
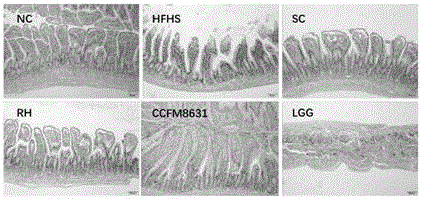
ActiveCN107523526AImprove the level ofImprove triglyceridesMilk preparationNervous disorderDiseaseGut flora
The invention relates to the technical field of microorganism, and discloses a Lactobacillus reuteri and its application. The preservation number of the Lactobacillus reuteri CCFM8631 is CGMCC No.14394; the level of mice peripheral neurotransmitter 5-hydroxytryptamine can be significantlyimproved; the rise of the mice peripheral blood testonsterone level and the abundant abnormity of Blautia, Turicibacter, Oscillospira and Bifidobacterium in the intestinal flora by high glucose and high fatty diets are recovered; the tolerated simulative gastrointestinal fluid is rapidly planted in an intestinal tract, so as to significantly improve the pathological injury of metabolic syndrome mice liver and duodenum and rise of triglyceride and total cholesterol content in the serum by high glucose and high fatty diets are significantly improved; the Lactobacillus reuteri can be used for preventing, delaying or treating metabolic disorder such as metabolic syndrome, irritable bowel syndrome, and anxiety, depression and other metal diseases related to irritable bowel syndrome.
Owner:INFINITUS (CHINA) CO LTD
Testosterone oral dosage formulations and associated methods
InactiveUS20050100608A1Reduce adverse outcomesPowder deliveryOrganic active ingredientsPolyethylene glycolEthylene glycol
Solid oral dosage forms of testosterone and methods for the preparation thereof are disclosed and described. The solid oral dosage form may include a therapeutically effective amount of testosterone in a substantially solid polyethylene glycol carrier. Such a form has been found to alleviate many of the undesirable consequences of undergoing testosterone therapy, such as the pain of injections and problems with patient noncompliance.
Owner:WATSON PHARMA INC
Buccal, polar and non-polar spray containing testosterone
InactiveUS20060210484A1Rapid onsetFast absorptionAerosol deliveryOrganic non-active ingredientsSolventPharmacology
Buccal aerosol sprays or capsules using polar and non-polar solvent have now been developed which provide testosterone for rapid absorption through the oral mucosa, resulting in fast onset of effect. The buccal polar compositions of the invention comprise formulation I: aqueous polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; formulation II: aqueous polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optionally flavoring agent, and propellant; formulation III: non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; and formulation IV: non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optional flavoring agent, and propellant; formulation V: a mixture of a polar and a non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, and optional flavoring agent; formulation VI: a mixture of a polar and a non-polar solvent, testosterone or a pharmaceutically acceptable ester thereof, optional flavoring agent, and propellant.
Owner:NOVADEL PHARMA
Medicine composition for treating impotence and its prepn
The present invention is aimed at strengthening physique and raising sexual function via nourishing Yin, filling life essence and strengthening Yang. The comdposite medicine for treating impotence is prepared with ten kinds of Chinese medicinal materials including dodder seed, wolfberry fruit, schisandra, cnidium fruit, cherokee rose fruit, etc. The pharmacological test shows that the medicine of the present invention can raise the excitement of the penis of emasculated rat to external stimulation, shorten the period for the penis to erect and raise level of testosterone in serum.
Owner:SHIJIAZHUANG YILING PHARMA
Bifidobacterium adolescentis and application thereof
InactiveCN107699517AImprove the level ofImprove oral glucose toleranceMilk preparationNervous disorderDiseaseIrritable bowel
The invention relates to the technical field of microorganisms, and discloses bifidobacterium adolescentis and an application thereof. The collection number of the bifidobacterium adolescentis CCFM8630 is CGMCC No.14395. The bifidobacterium adolescentis CCFM8630 has the advantages that the level of neurotransmitter 5-hydroxytryptamine in peripheral blood of rats can be remarkably upgraded by the bifidobacterium adolescentis CCFM8630; the increased level of hormones such as testosterone in the peripheral blood of the rats due to high-glucose and high-fat diet and anomaly of abundance of Blautiaand Turicibacter in intestinal floras of the rats can be recovered; simulated gastrointestinal fluid can be tolerated, the bifidobacterium adolescentis can be quickly colonized in intestinal tracts,accordingly, rat livers with metabolic syndromes, pathological injury of duodenums and increase of the contents of triglyceride and total cholesterol in serum due to the high-glucose and high-fat dietand oral glucose tolerance can be remarkably improved, and the bifidobacterium adolescentis can be used for preventing, relieving or treating metabolic disorder such as the metabolic syndromes, irritable bowel syndromes and psychological illnesses such as anxiety and depression which are related to the metabolic syndromes.
Owner:INFINITUS (CHINA) CO LTD
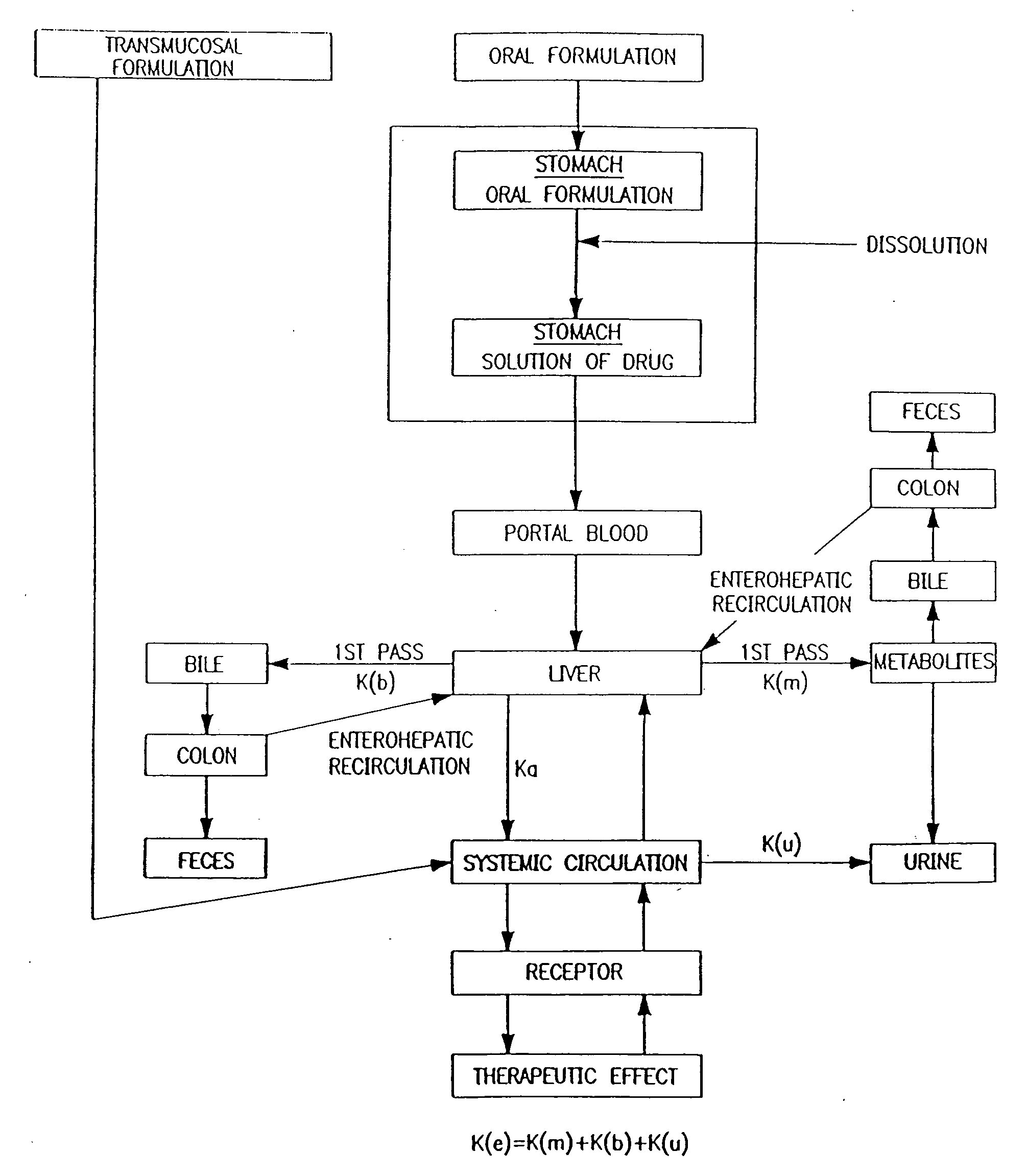
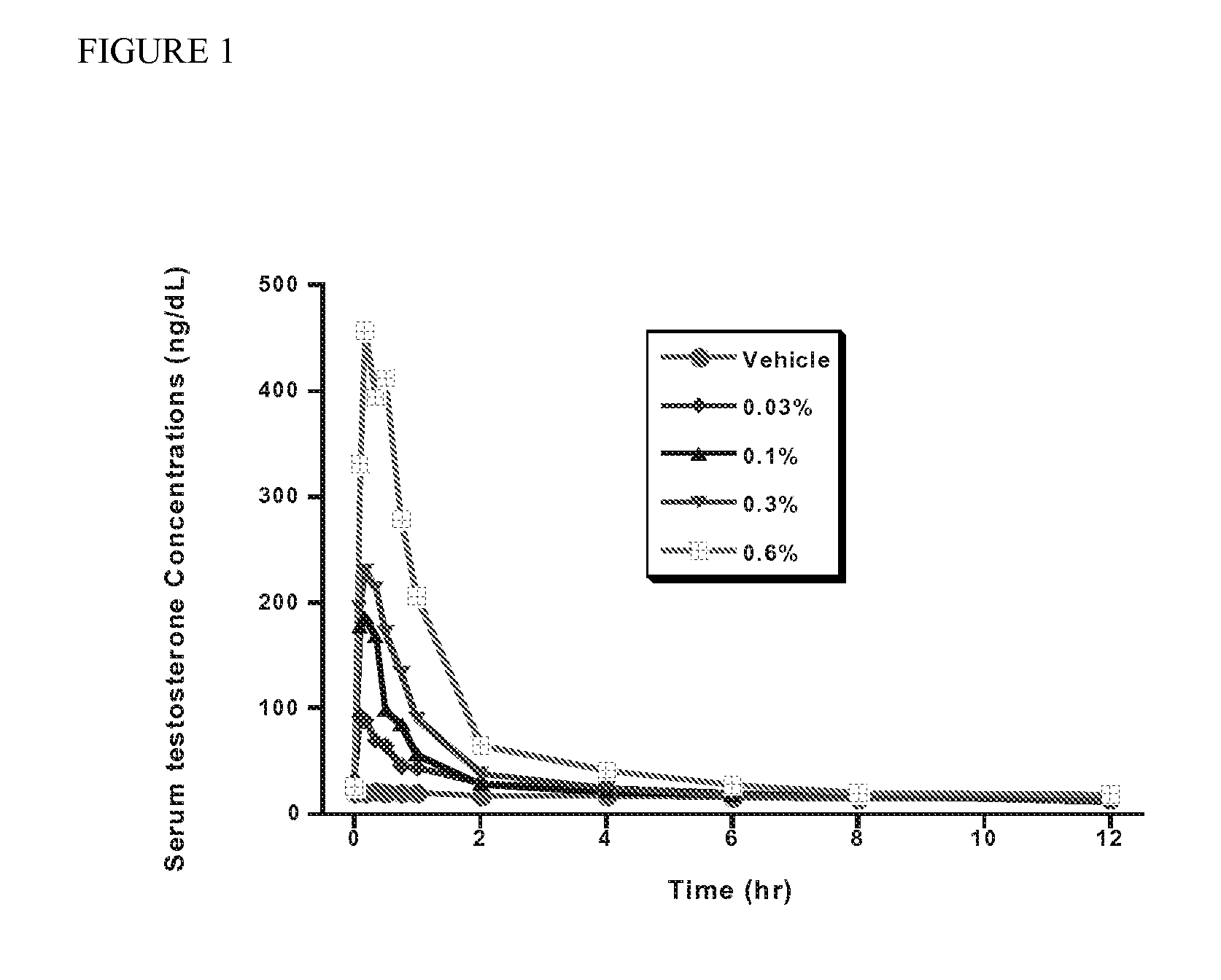
Method and apparatus for transdermal or transmucosal application of testosterone
InactiveUS20060270642A1Effective treatmentDecrease and alleviate clinical symptomBiocideOrganic active ingredientsActive agentMenopausal disorders
Methods, formulations, and devices for providing transdermal or transmucosal delivery of active agents to subjects in need thereof. The formulations and methods treat symptoms of hormonal disorders including hypogonadism, female sexual desire disorder, female menopausal disorder, and adrenal insufficiency.
Owner:ANTARES PHARMA IPL
Compounds and methods for the treatment of urogenital disorders
The present invention provides methods for treating a variety of urogenital disorders, such as, for example, vaginismus, dyspareunia, vulvodynia (including vulvar vestibulitis), interstitial cystitis, nonspecific urethriris (i.e., nonspecific pain and / or burning of the urinary tract) and sexual dysfunctions, such as, for example, female sexual arousal disorders and female sexual orgasmic disorders, using a variety of compounds, including, but not limited to, NO donors, calcium channel blockers, cholinergic modulators, α-adrenergic receptor antagonists, β-adrenergic receptor agonists, phosphodiesterase inhibitors, cAMP-dependent protein kinase activators (e.g., cAMP mimetics), superoxide scavengers, potassium channel activators, estrogen-like compounds, testosterone-like compounds, benzodiazepines, adrenergic nerve inhibitors, antidiarrheal agents, HMG-CoA reductase inhibitors, smooth muscle relaxants, adenosine receptor modulators, adenylyl cyclase activators, endothelin receptor antagonists, bisphosphonates and cGMP-dependent protein kinase activators (e.g., cGMP mimetics).
Owner:STREHKEHN INT LTD
Treatment for dry eye using testosterone and progestagen
InactiveUS20100016264A1Minimizes and avoids systemic treatmentAvoid disadvantagesOrganic active ingredientsBiocideOcular surfaceProgestin
The present invention comprises a composition and methods for treating eye conditions using a composition having a therapeutically effective amount of a progestagen, or a therapeutically effective amount of a progestagen with a testosterone; and pharmaceutically acceptable carrier, wherein the composition is applied to the palpebral part of the eye and / or ocular surface.
Owner:CONNOR CHARLES G +1
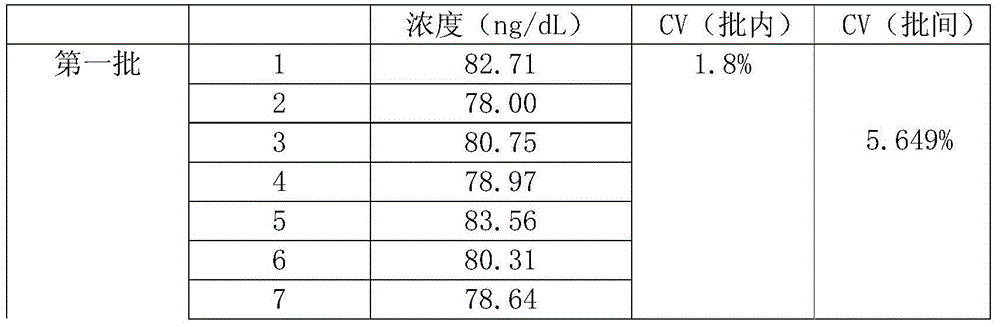
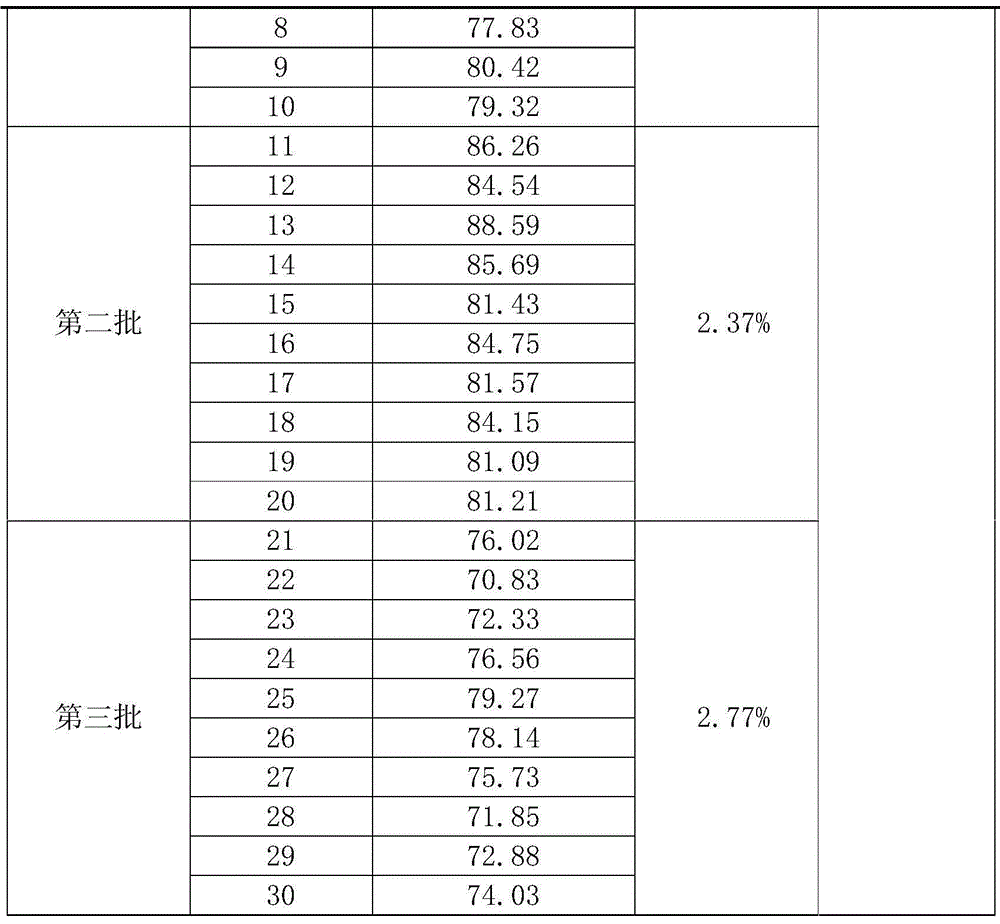
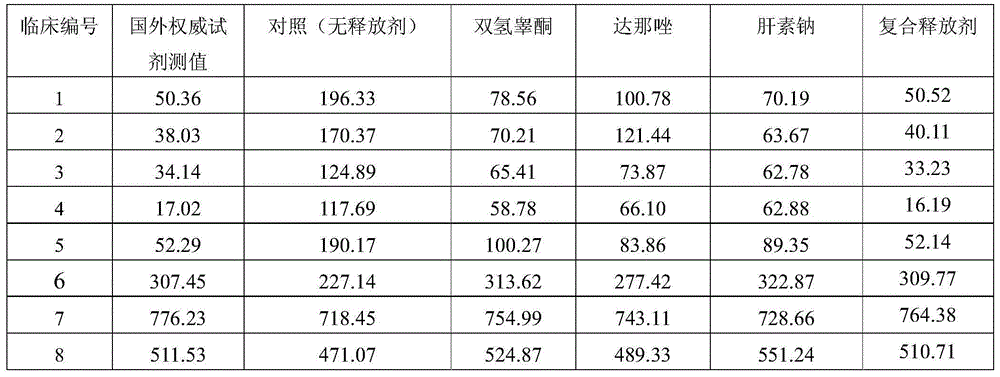
Testosterone detection reagent based on microparticle chemiluminescence immunoassay technology
ActiveCN103954779AFacilitate dissociationReduce the binding forceChemiluminescene/bioluminescenceBiological testingMicrosphereMicroparticle
The invention discloses a testosterone detection reagent based on a microparticle chemiluminescence immunoassay technology; the testosterone detection reagent comprises the following components: 0.002%-0.01% of paramagnetic microspheres, 0.4 mug / ml-1 mug / ml of acridinium ester labeled testosterone antibodies, a composite testosterone release agent, a luminescence liquid A, a luminescence liquid B, 0ng / dl-1500ng / dl of a testosterone calibrator and a cleaning fluid with a certain concentration; and the testosterone release agent is a mixture of dihydrotestosterone, danazol and heparin sodium. The testosterone detection reagent has the advantages of simple operation, high sensitivity, fast detection efficiency, low cost, easy automation and the like, and by combination of the selected composite release agent and different binding globulin, combined-state testosterone in serum can be fully released, so that the content detection of total testosterone is more accurate.
Owner:DIRUI MEDICAL TECH CO LTD
Method and composition for treatment of hair loss
A composition and method for treating hair loss including glyceride of ricinus oil, eucalyptol, and mint oil. The treatment method includes the use of this composition periodically (three times per week) for a predetermined period of time (typically one month). The composition cleans the follicular area, reducing the concentration of DHT (dihydro-testosterone), thereby stimulating the growth of hair.
Owner:DISTRIBUTOR REVOLUTIONS
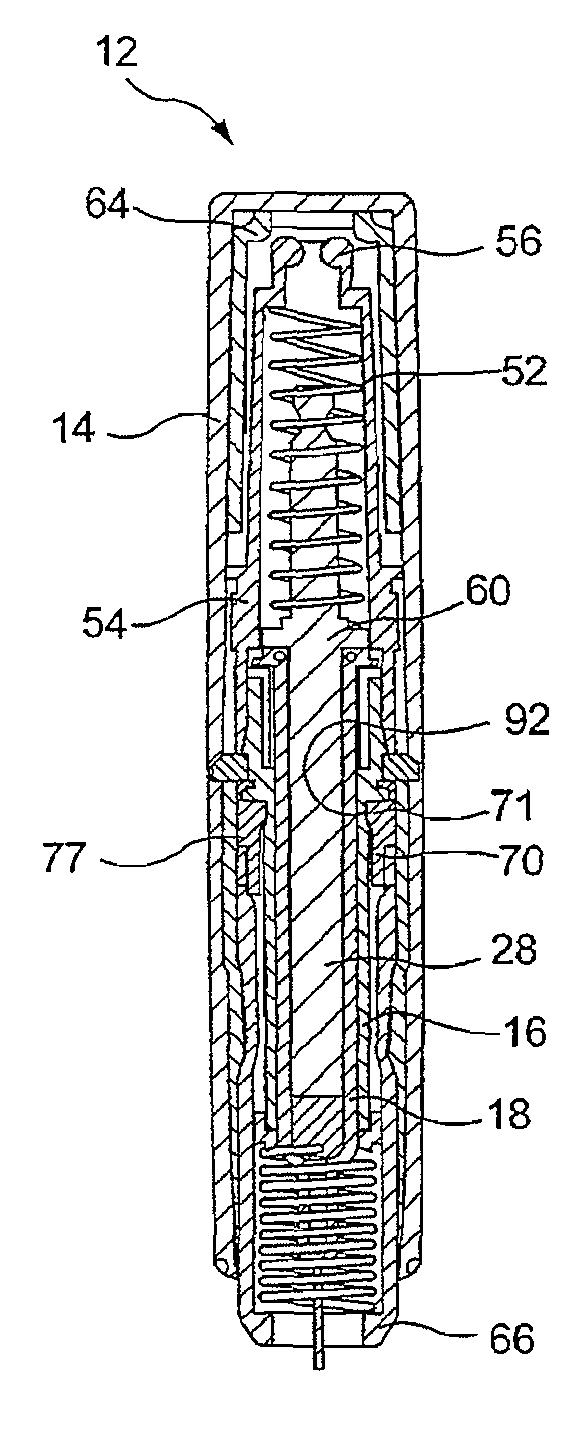
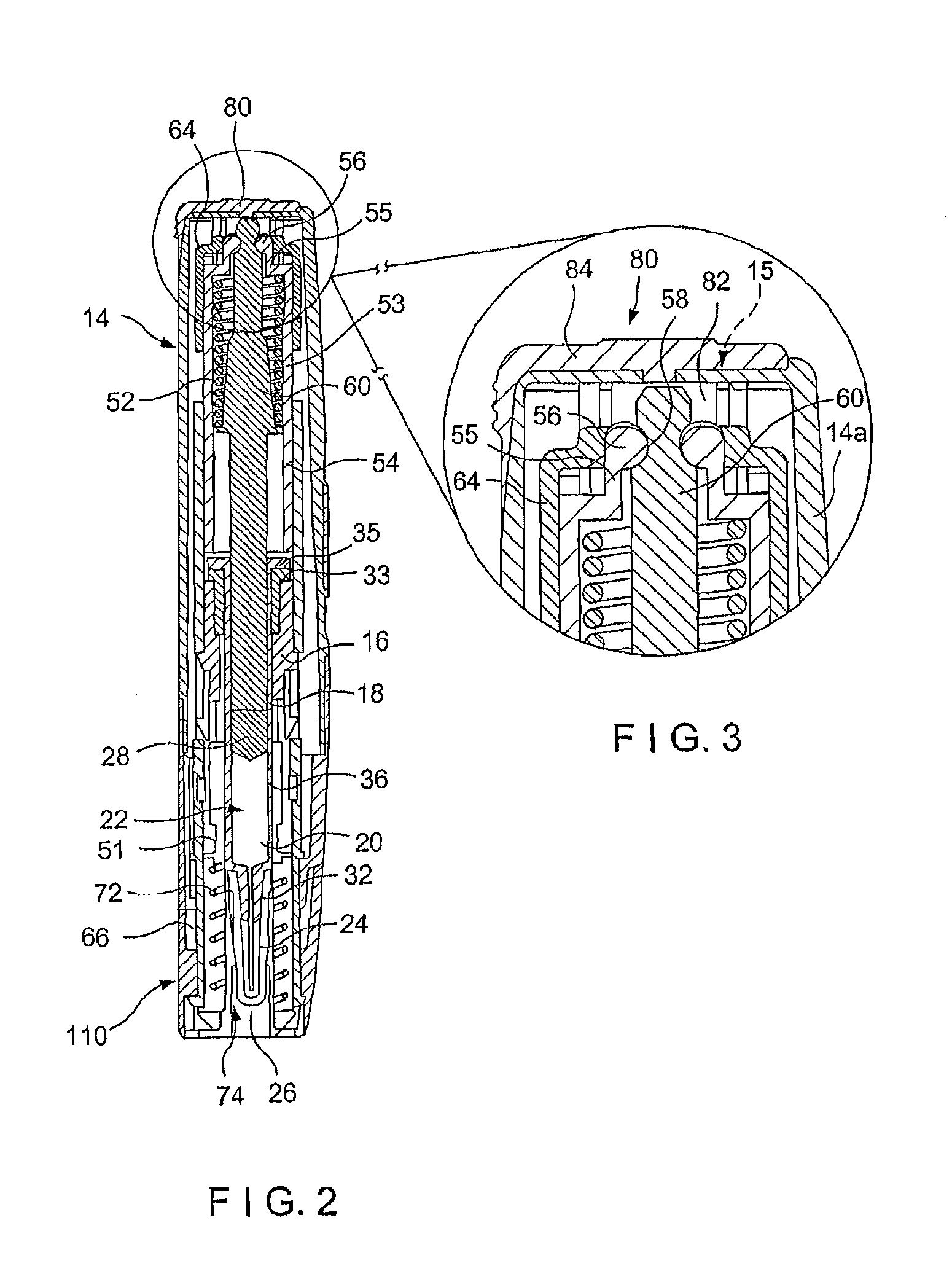
Needle assisted jet injection administration of testosterone compositions
ActiveUS20130303985A1Minimize leak-backOrganic active ingredientsJet injection syringesJet injectionTestosterone
Owner:ANTARES PHARMA
Testosterone formulations
InactiveUS20130295166A1Improve skinGood skin permeabilityOrganic active ingredientsNervous disorderPharmacologyTestosterone
Owner:FERRING BV
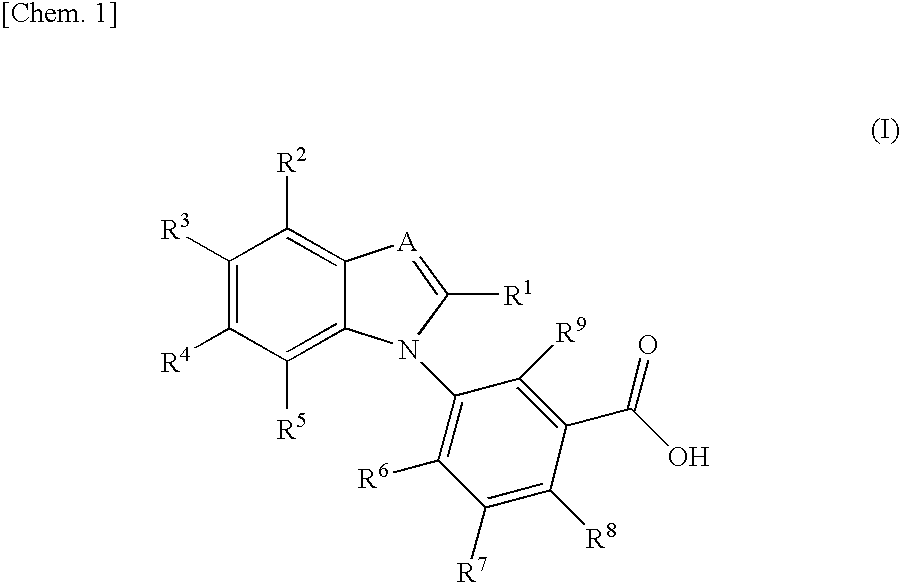
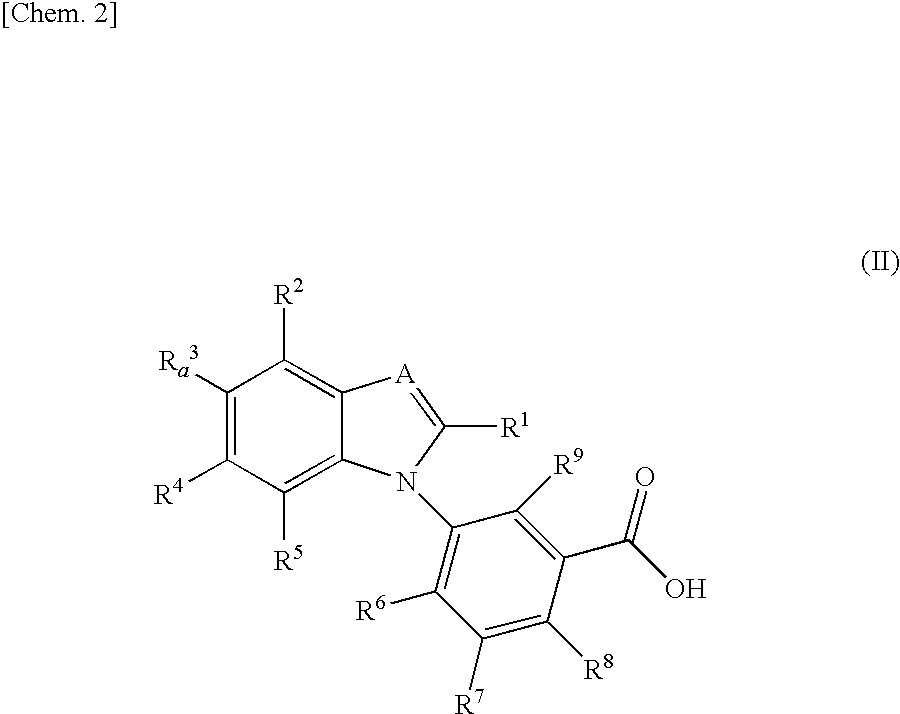
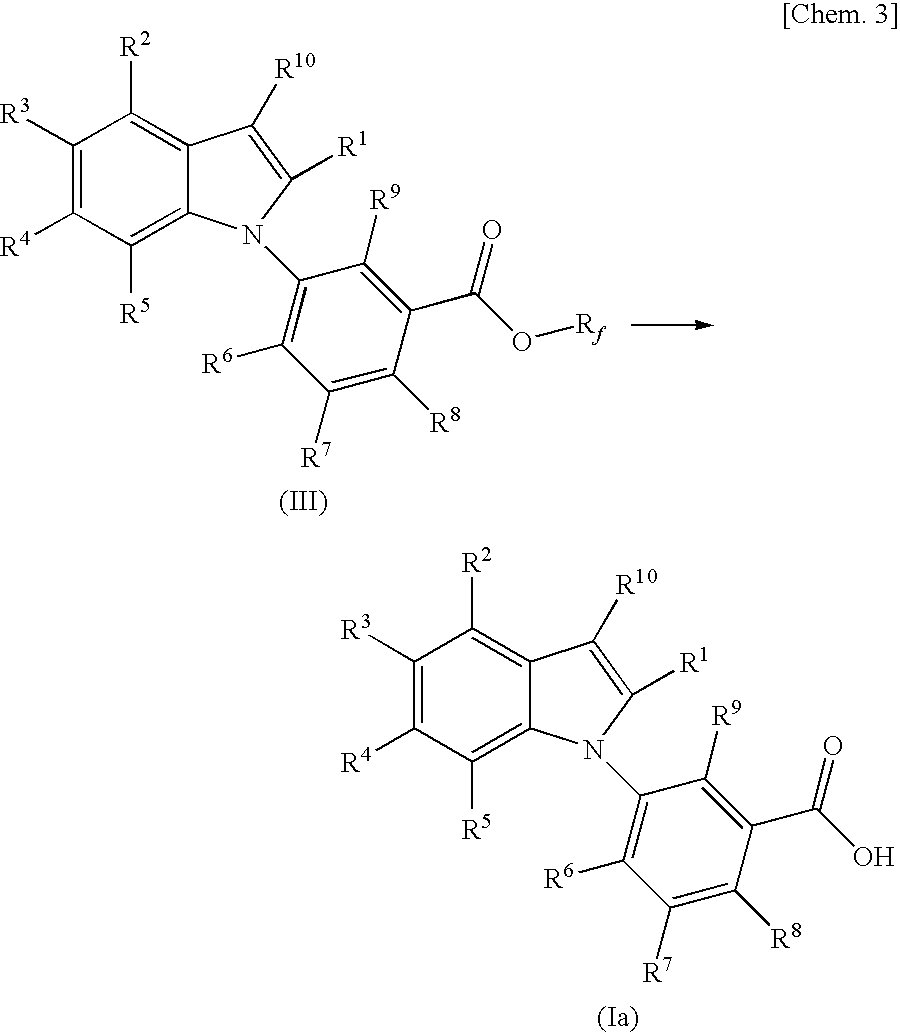
Benzimidazole derivative
InactiveUS20100190826A1Suppress intracrine androgen synthesisBiocideOrganic chemistryDiseaseBenzimidazole derivative
Owner:ASTELLAS PHARMA INC
Method of treating or preventing bone deterioration or osteoporosis
InactiveUS20080058299A1Treating and preventing and reducing riskPrevent and reduce riskOrganic active ingredientsOintment deliverySurgeryOsteoporosis
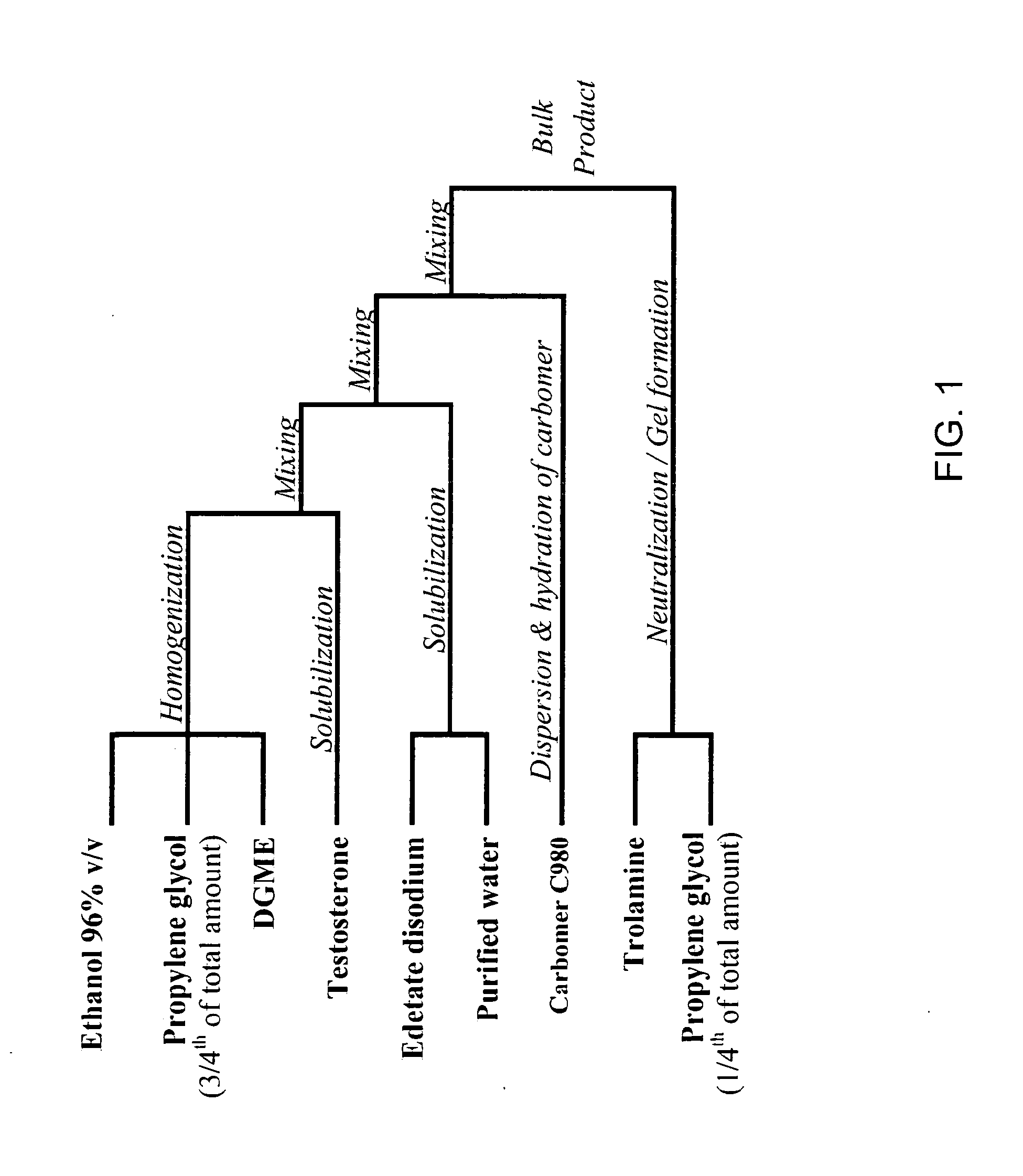
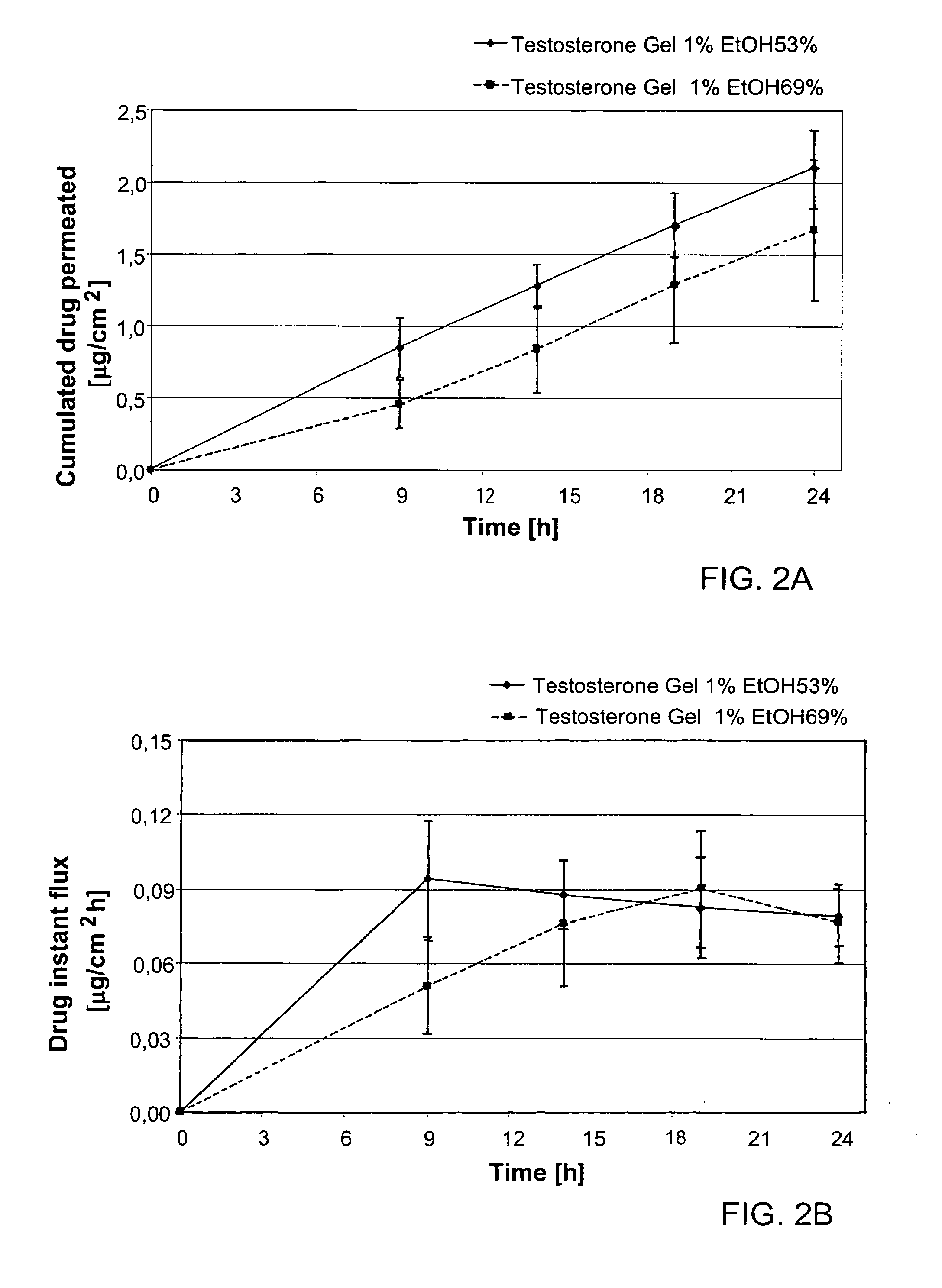
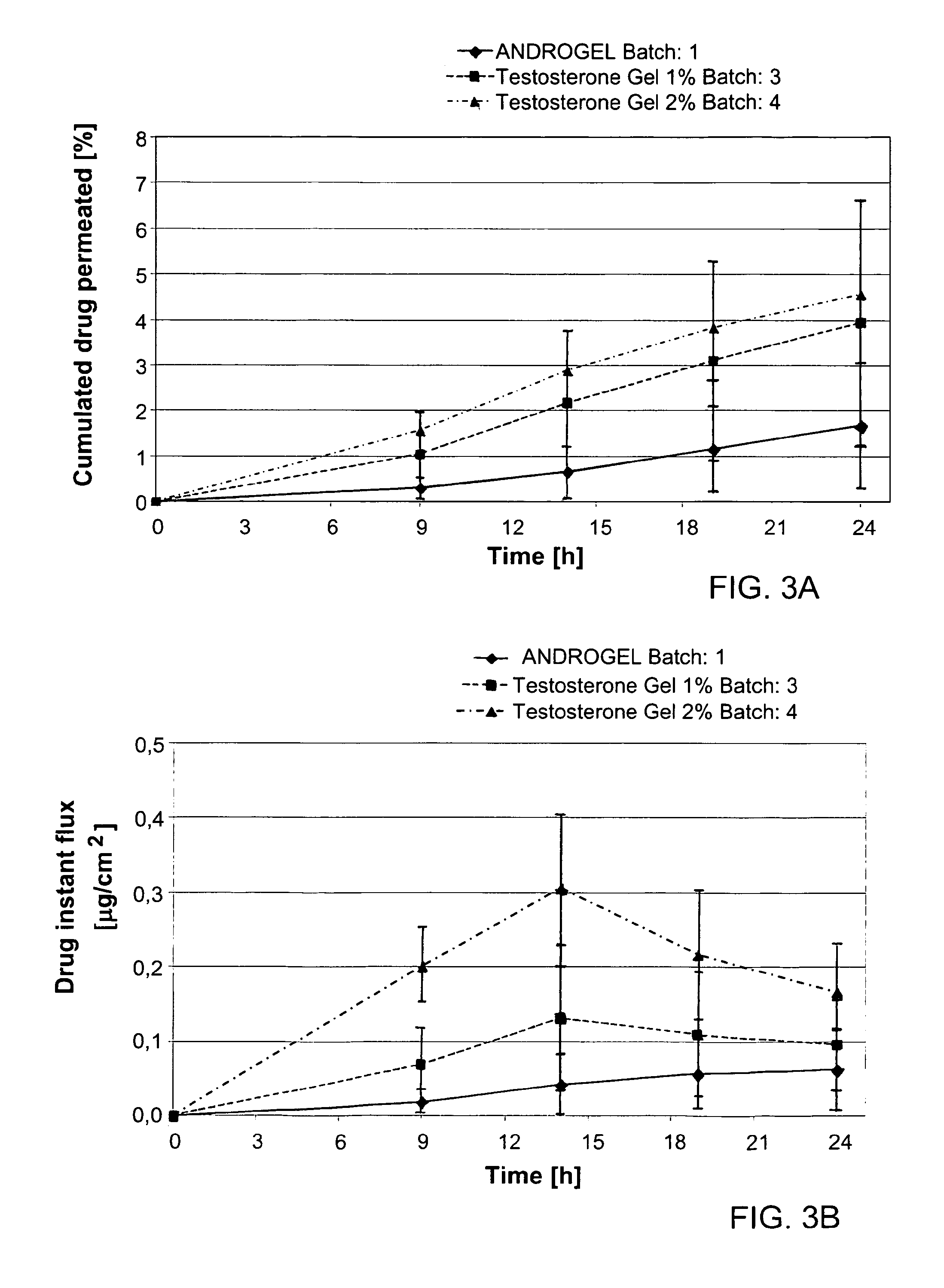
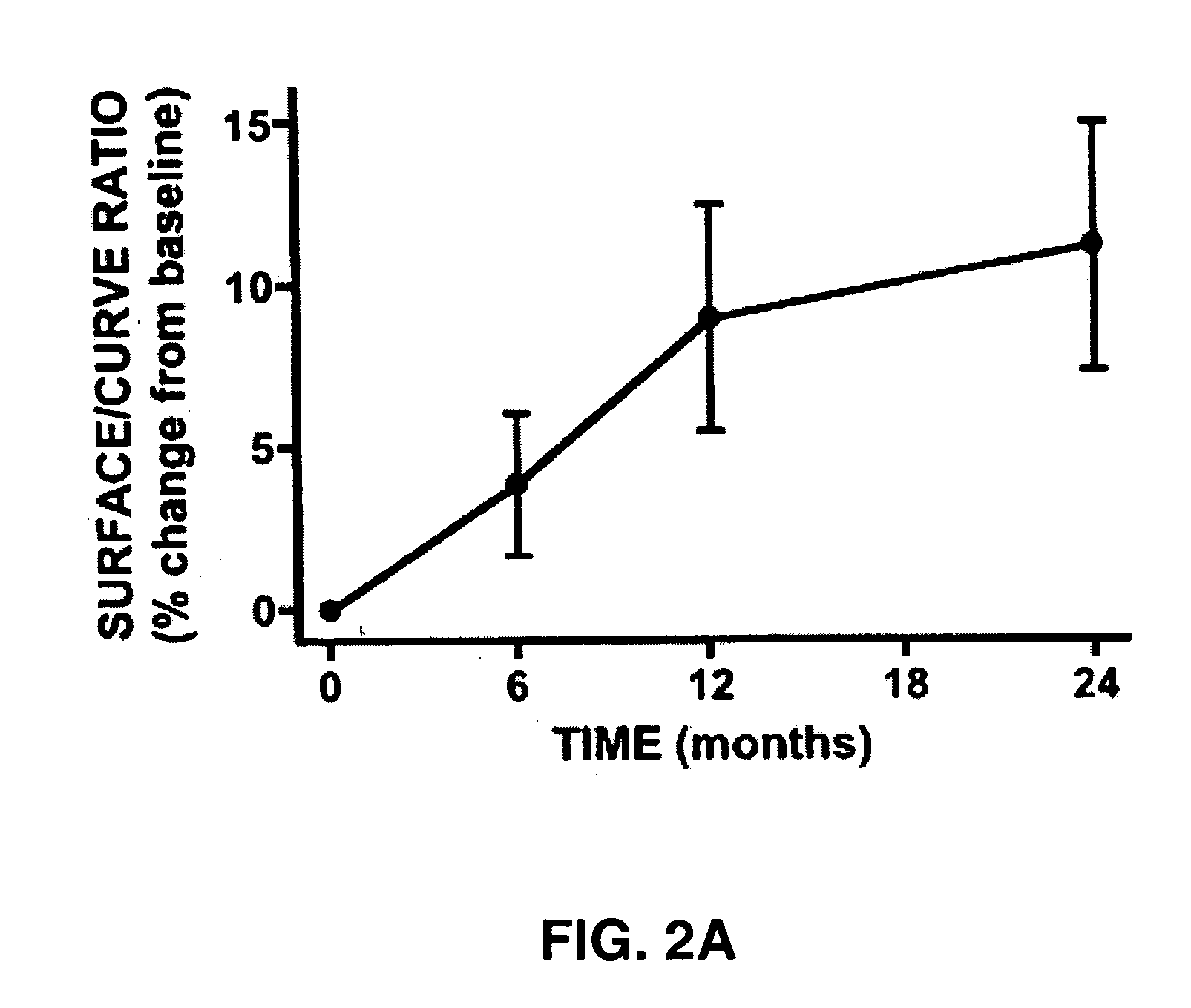
The present invention relates to a transdermal hydroalcoholic testosterone gel formulation and a method for treating, preventing, or reducing the risk of developing deterioration of bone. The present invention also relates to a method for treating, preventing, or reducing the risk of developing osteoporosis.
Owner:LABORATORIES BESINS INTERNATIONAL SAS
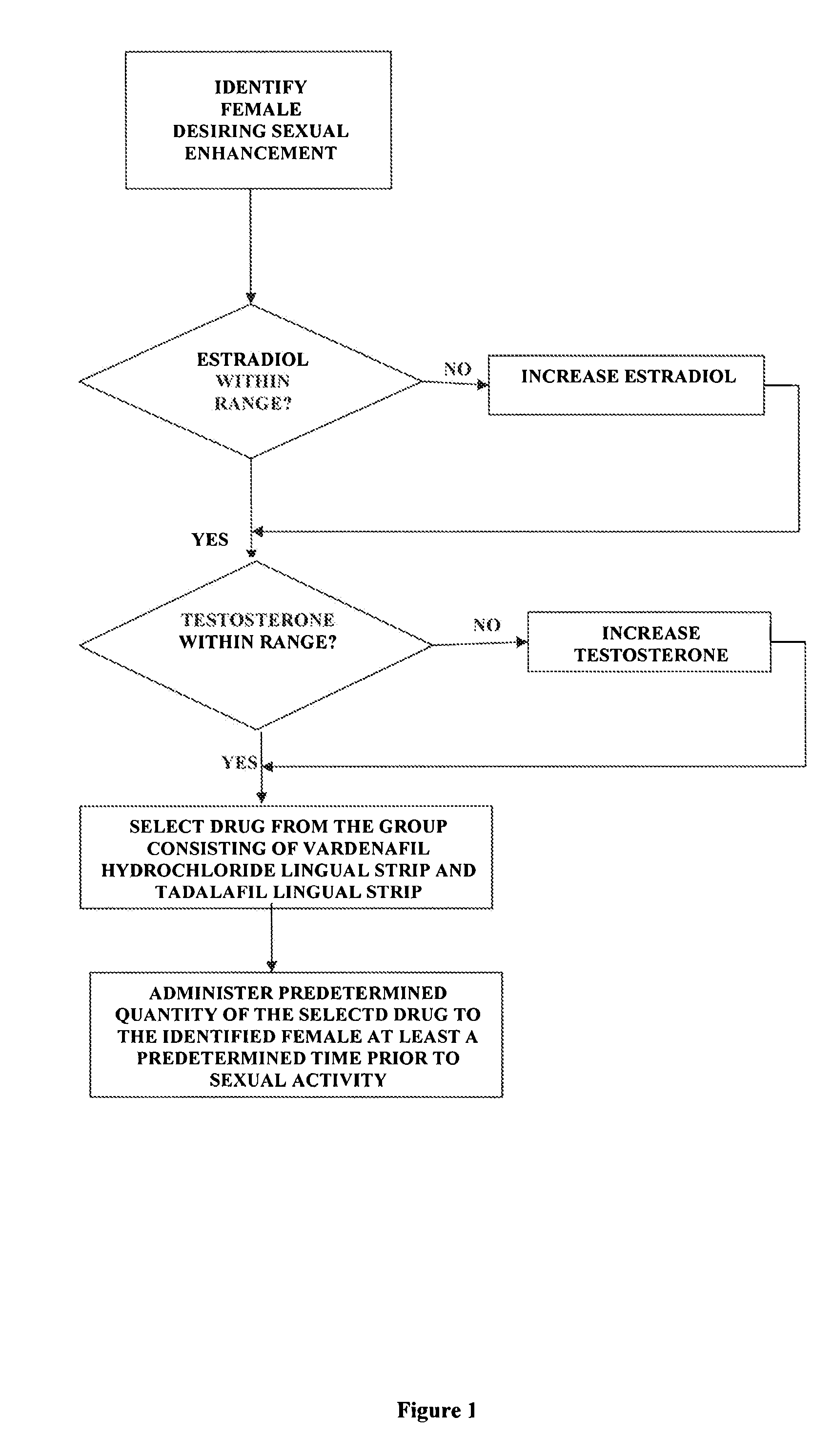
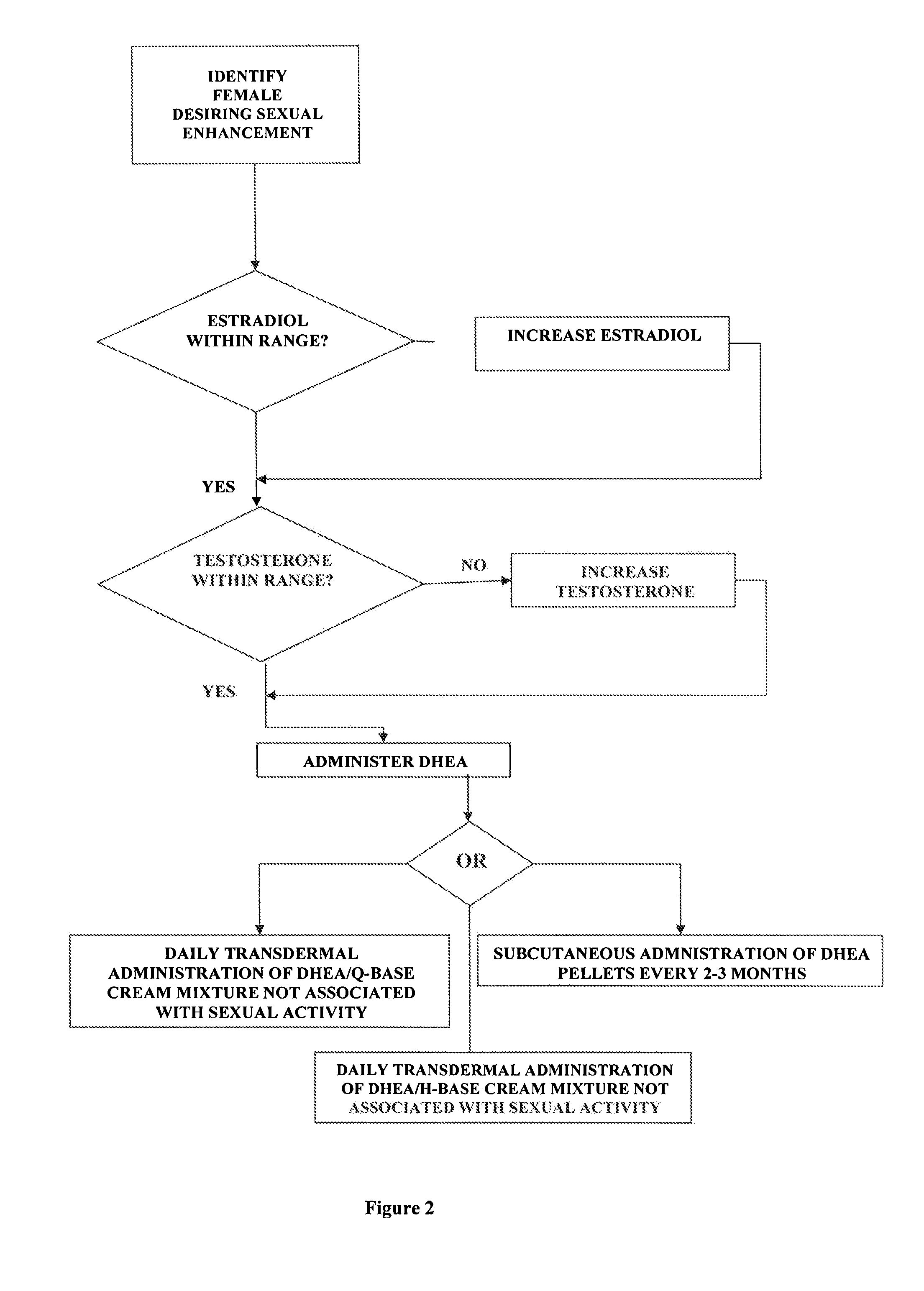
Methods of female sexual enhancement
A method of sexual enhancement in women includes the steps of identifying a woman requesting sexual enhancement, assuring that the woman's blood includes estradiol within a first predetermined range and testosterone within a second predetermined range, and thereafter administrating a drug selected from the group consisting of vardenafil hydrochloride and tadalafil prior to sexual activity. The selected drug may be loaded into a starch strip which is then applied to the woman's tongue. Sexual enhancement in women can also be achieved by transdermal or subcutaneous application of the steroid hormone DHEA.
Owner:LES MEDECINS
Ocular administration of testosterone
InactiveUS20080045486A1Good for healthLower levelBiocideOrganic active ingredientsTherapeutic effectBuccal administration
The present invention provides improving health in a woman or man comprising non-orally administering a testosterone-containing moiety to said woman or man by topically applying a liquid composition including said testosterone-containing moiety to the eye of said woman or man in an amount sufficient to provide a therapeutic effect.
Owner:ALLERGAN INC