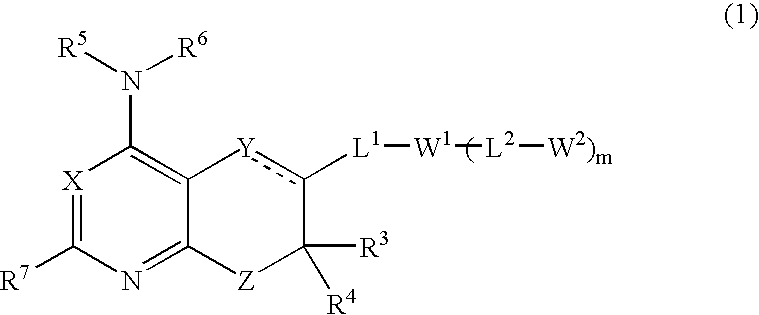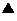Patents
Literature
273 results about "Therapeutic strategy" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Therapeutic strategies, n.pl 1., approaches to treatment based on principles of organization. 2., in nutritional therapy, subcategory of prescriptive dietetics and nutritional pharmacology that addresses the imbalances in biochemistry or disturbances in physiology like tissue or organ dysfunction due to an organic disease.
Therapeutic application of chimeric and radiolabelled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma
Disclosed herein are therapeutic treatment protocols designed for the treatment of B cell lymphoma. These protocols are based upon therapeutic strategies which include the use of administration of immunologically active mouse / human chimeric anti-CD20 antibodies, radiolabeled anti-CD20 antibodies, and cooperative strategies comprising the use of chimeric anti-CD20 antibodies and radiolabeled anti-CD20 antibodies.
Owner:IDEC PHARM CORP
Therapeutic application of chimeric and radiolabelled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma
Disclosed herein are therapeutic treatment protocols designed for the treatment of B cell lymphoma. These protocols are based upon therapeutic strategies which include the use of administration of immunologically active mouse / human chimeric anti-CD20 antibodies, radiolabeled anti-CD20 antibodies, and cooperative strategies comprising the use of chimeric anti-CD20 antibodies and radiolabeled anti-CD20 antibodies.
Owner:BIOGEN INC
Treatment planning with implantable bronchial isolation devices
Disclosed is a treatment planning method that can be used to maximize the effectiveness of minimally invasive treatment on a patient. Pursuant to the treatment planning method, the presence of lung disease, such as emphysema, is first identified, followed by a determination of the distribution and extent of damage of the disease, followed by a determination of whether the patient is suitable for treatment, and a determination of the appropriate strategy for treatment for a suitable patient.
Owner:PULMONX
Compositions and methods for enhancing discriminatory RNA interference
InactiveUS20070259827A1Improved allelic discriminationImprove discriminationOrganic active ingredientsNervous disorderNon targetedGenetic disorder
The present invention provides methods for enhancing discriminatory RNA silencing by RNA silencing agents. In particular, the invention provides methods for generating RNA silencing agents which can discriminate between target and non-target mRNAs that differ in sequence by only one nucleotide. Also provided are improved RNA silencing agents with enhanced discriminatory RNA silencing, e.g., single nucleotide discriminatory RNA silencing. The compositions and methods of the invention are useful in therapeutic strategies for treating genetic disorders associated with dominant, gain-of-function gene mutations.
Owner:UNIV OF MASSACHUSETTS +1
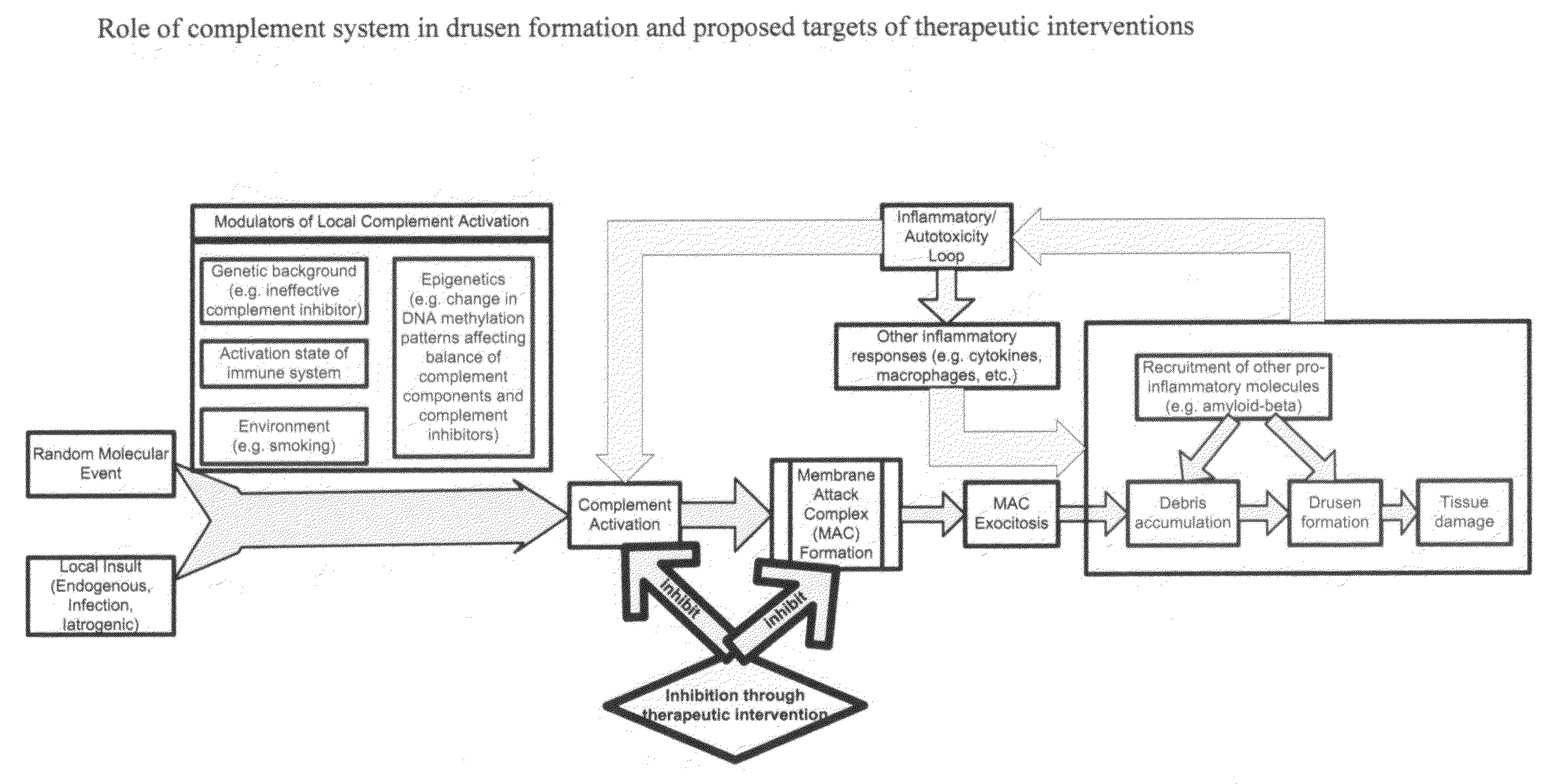
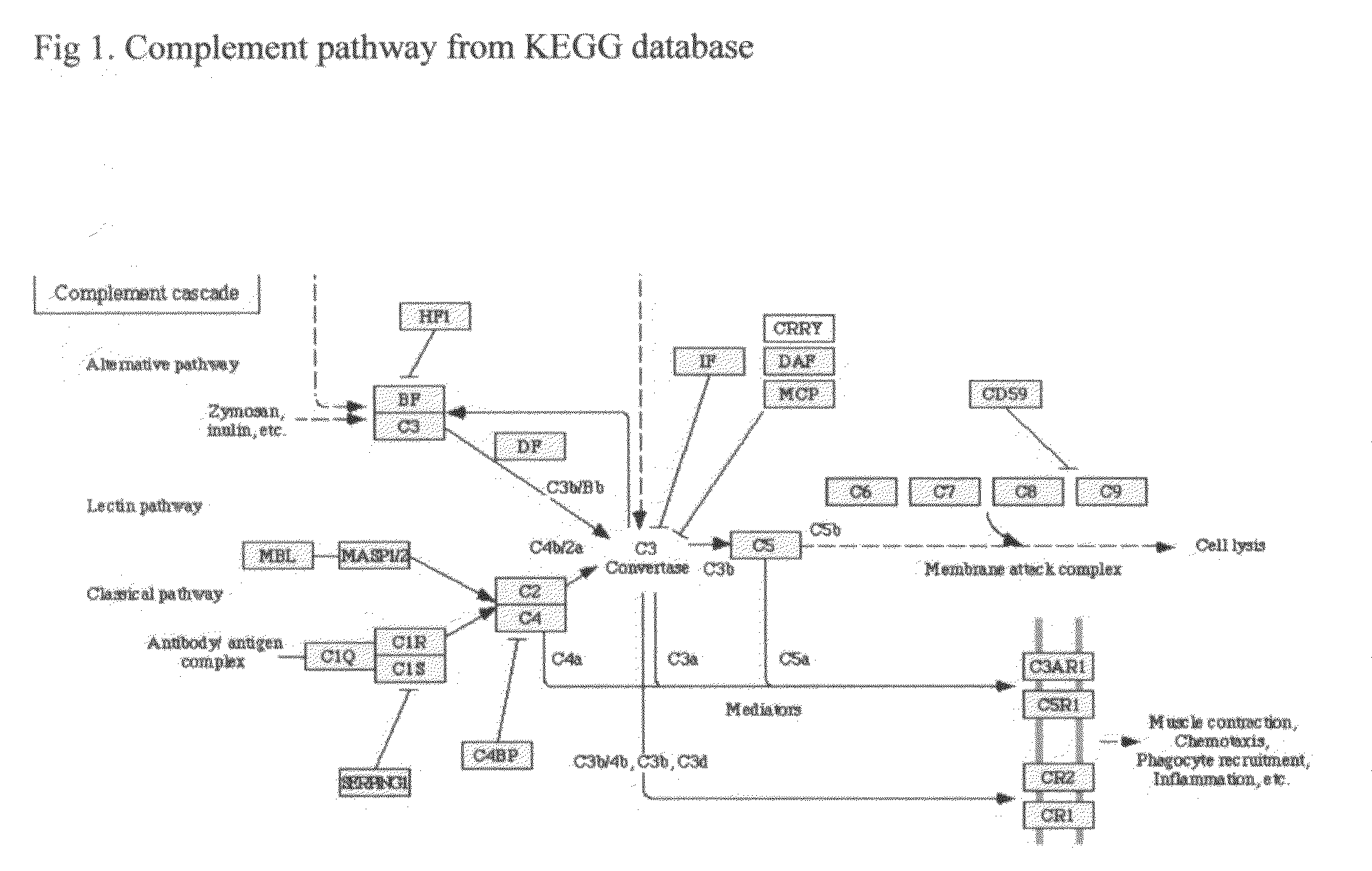
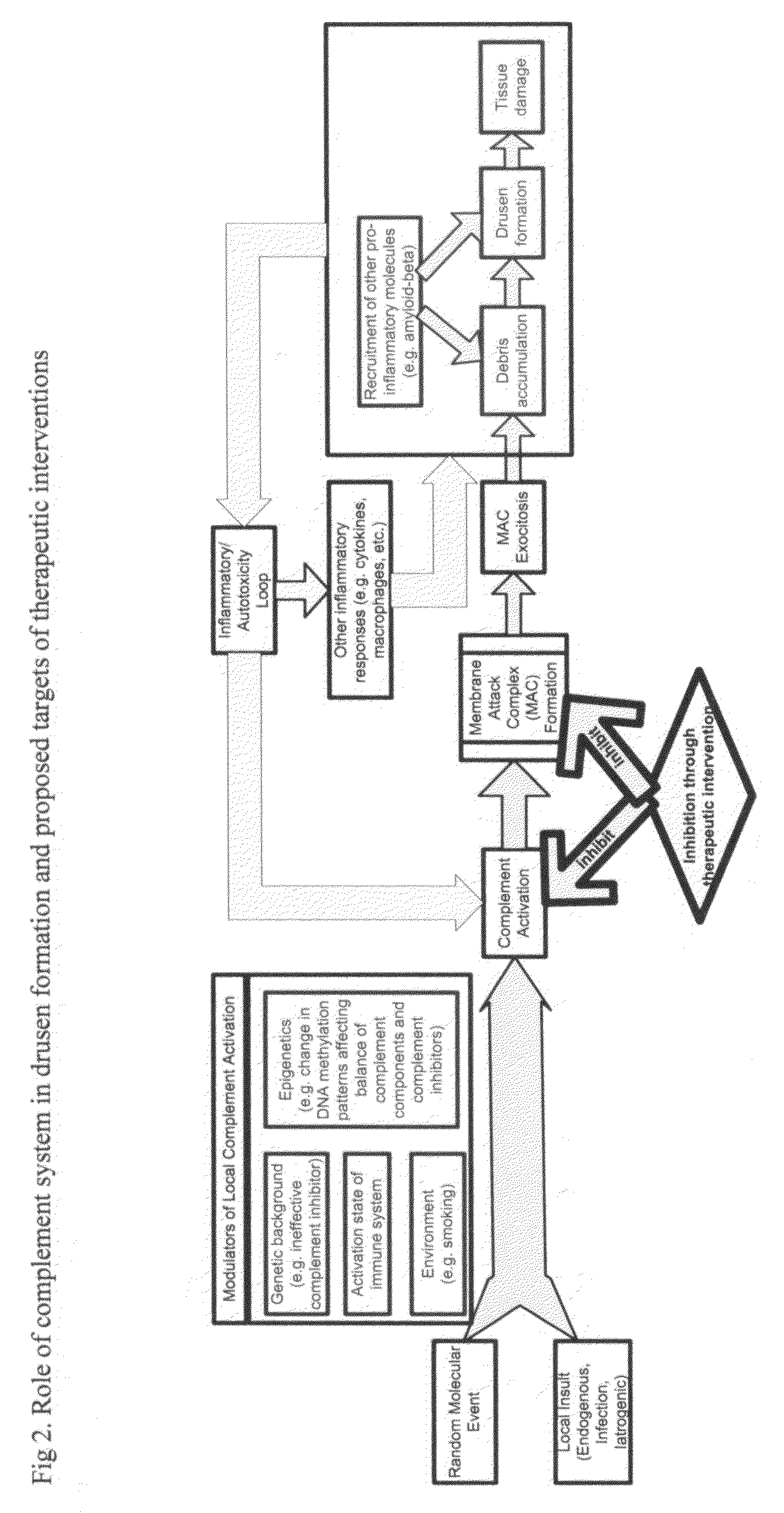
Methods of preventing and treating Alzheimer's disease, age related macular degeneration and other diseases involving extra-cellular debris through the inhibition of the complement system
This invention proposes that the best therapeutic strategy for treating and / or preventing Alzheimer's disease (AD), age related macular degeneration (AMD) and other diseases that exhibit extra-cellular debris deposits, such as atherosclerosis, is the inhibition of the complement pathway. A model for the accumulation of extra-cellular debris through the activation of the complement pathway is presented, and the primary pathogenic role of the debris in the etiology of the disorders is explained. Previously identified complement inhibitors are identified as therapeutic agents for the treatment and / or prevention of AD, AMD and other diseases that exhibit extra-cellular debris deposits.
Owner:DINU VALENTIN
Method of diagnosing small cell lung cancer
InactiveCN101283106AMicrobiological testing/measurementAntineoplastic agentsScreening methodSmall cell
Objective methods for detecting and diagnosing small cell lung cancer (SCLC) are described herein. In one embodiment, the diagnostic method involves determining the expression level of an SCLC-associated gene that discriminates between SCLC cells and normal cells. In another embodiment, the diagnostic method involves determining the expression level of an SCLC-associated gene that distinguishes two major histological types of lung cancer, non-small cell lung cancer (NSCLC) and SCLC. Finally, the present invention provides methods of screening for therapeutic agents useful in the treatment of small cell lung cancer, methods of treating small cell lung cancer and method for vaccinating a subject against small cell lung cancer. Furthermore, the present invention provides chemotherapy resistant lung cancer- or SCLC-associated genes as diagnostic markers and / or molecular targets for therapeutic agent for these cancers. These genes are up-regulated in chemoresistant lung cancer or SCLC. Accordingly, chemoresistant lung cancer or SCLC can be predicted using expression level of the genes as diagnostic markers. As the result, any adverse effects caused by ineffective chemotherapy can be avoided, and more suitable and effective therapeutic strategy can be selected.
Owner:ONCOTHERAPY SCI INC
Modified antigen-presenting cells
InactiveUS20050003484A1Efficient presentation of antigenEffective presentationSsRNA viruses negative-sensePeptide/protein ingredientsAntigenAntigen-presenting cell
The invention relates to antigen-presenting cells having specificity against a selected antigen and methods for making the cells. The invention also relates to a method of selecting efficient antigen-presenting cells using reporter fusion constructs. The highly efficient antigen-presenting cells of the invention will provide a therapeutic strategy of modulating immune responses for a variety of diseases.
Owner:DANA FARBER CANCER INST INC
Soluble Form of Carbonic Anhydrase IX (s-CA IX), Assays to Detect s-CA IX, CA IX's Coexpression with HER-2/neu/c-erbB-2, and CA IX-Specific Monoclonal Antibodies to Non-Immunodominant Epitopes
InactiveUS20080176310A1Improve efficiencyIncrease resourcesOxidoreductasesFermentationKilodaltonWestern blot
Disclosed herein is the discovery of a soluble MN / CA IX (s-CA IX) in body fluids, such as, urine and serum. Said s-CA IX comprises the extracellular domain of CA IX or portions thereof. The predominant s-CA IX species is the extracellular domain comprising a proteoglycan-like (PG) domain and carbonic anhydrase (CA) domain, and having a molecular weight of about 50 / 54 kilodaltons (kd) upon Western blot. A smaller s-CA IX form of about 20 to about 30 kd comprising the CA domain or parts thereof, not linked to the PG domain, has also been found in body fluids. Diagnostic / prognostic methods for precancer and cancer that detect or detect and quantitate said s-CA IX in body fluids, are described. Also disclosed herein is the coexpression of CA IX and HER-2 / neu / c-erbB-2 that provides parallel, alternative and potentially synergistic diagnostic / prognostic and therapeutic strategies for precancer and cancer. Further disclosed are new MN / CA IX-specific antibodies generated from MN / CA IX-deficient mice, preferably monoclonal antibodies and immunoreactive fragments and engineered variants thereof. Such new MN / CA IX-specific antibodies, fragments and variants are useful diagnostically / prognostically and therapeutically for cancer and precancer. Particularly preferred are the new monoclonal antibodies, fragments and variants that are specific for the non-immunodominant epitopes of MN / CA IX, which antibodies are, among other uses, useful to detect soluble MN / CA IX (s-CA IX) in body fluids, alone but preferably in combination with antibodies specific to the immunodominant epitopes of MN / CA IX, for example, in a sandwich assay.
Owner:BIOMEDICAL RES CENT OF THE SLOVAK ACADEMY OF SCI
Soluble Form of Carbonic Anhydrase IX (s-CA IX), Assays to Detect s-CA IX, CA IX's Coexpression with Her-2/neu/c-erbB-2, and CA IX-Specific Monoclonal Antibodies to Non-Immunodominant Epitopes
InactiveUS20080177046A1Good curative effectImprove efficiencyImmunoglobulins against animals/humansBiological material analysisKilodaltonWestern blot
Disclosed herein is the discovery of a soluble MN / CA IX (s-CA IX) in body fluids, such as, urine and serum. Said s-CA IX comprises the extracellular domain of CA IX or portions thereof. The Predominant s-CA IX species is the extracellular domain comprising a proteoglycan-like (PG) domain and carbonic anhydrase (CA) domain, and having a molecular weight of about 50 / 54 kilodaltons (kd) upon Western blot. A smaller s-CA IX form of about 20 to about 30 kd comprising the CA domain or parts thereof, not linked to the PG domain, has also been found in body fluids. Diagnostic / prognostic methods for precancer and cancer that detect or detect and quantitate said s-CA IX in body fluids, are described. Also disclosed herein is the coexpression of CA IX and HER-2 / neu / c-erbB-2 that provides parallel, alternative and potentially synergistic diagnostic / prognostic and therapeutic strategies for precancer and cancer. Further disclosed are new MN / CA IX-specific antibodies generated from MN / CA IX-deficient mice, preferably monoclonal antibodies and immunoreactive fragments and engineered variants thereof. Such new MN / CA IX-specific antibodies, fragments and variants are useful diagnostically / prognostically and therapeutically for cancer and precancer. Particularly preferred are the new monoclonal antibodies, fragments and variants that are specific for the non-immunodominant epitopes of MN / CA IX, which antibodies are, among other uses, useful to detect soluble MN / CA IX (s-CA IX) in body fluids, alone but preferably in combination with antibodies specific to the immunodominant epitopes of MN / CA IX, for example, in a sandwich assay.
Owner:BIOMEDICAL RES CENT OF THE SLOVAK ACADEMY OF SCI
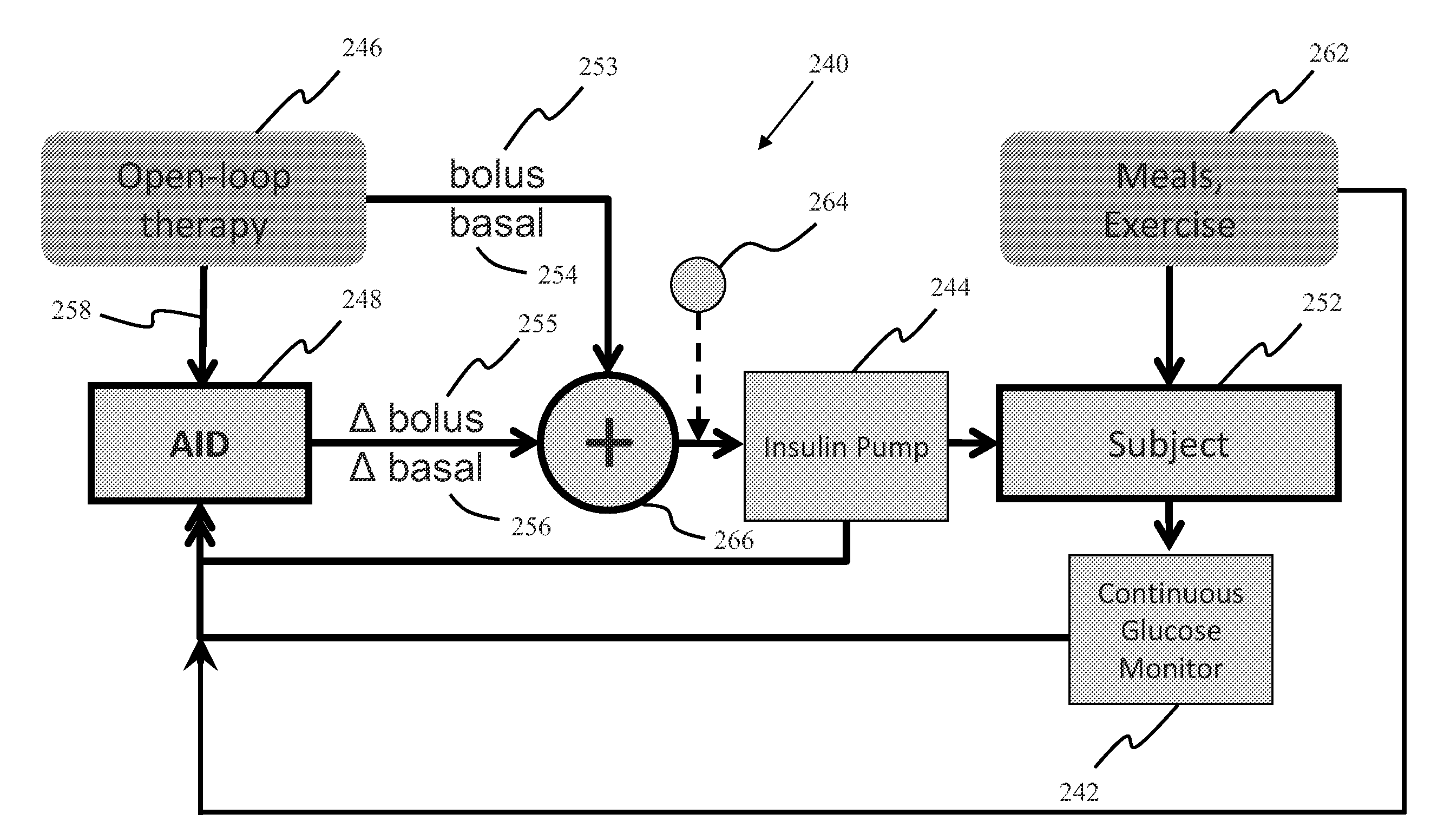
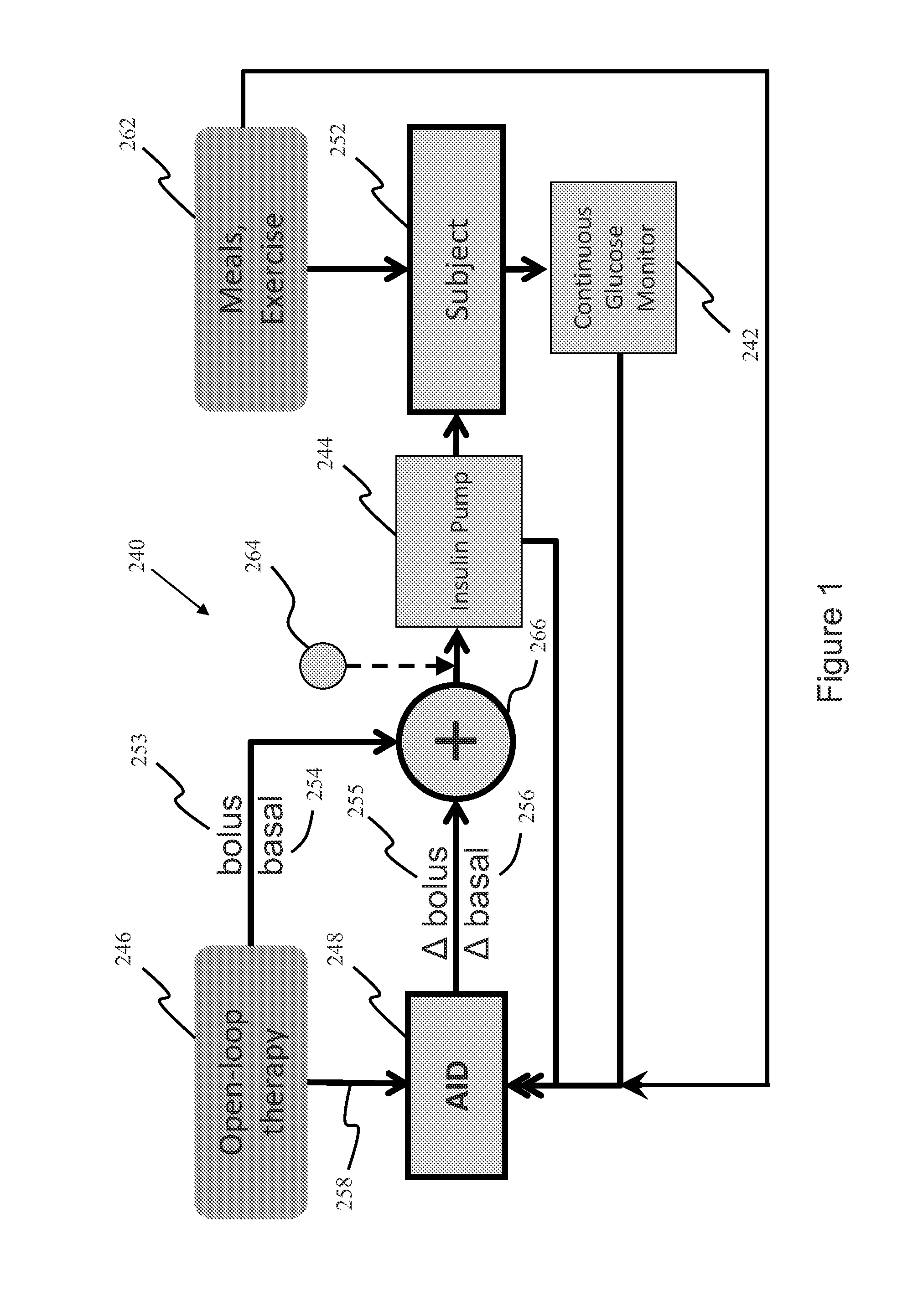
System, Method and Computer Program Product For Adjustment of Insulin Delivery in Diabetes Using Nominal Open-Loop Profiles
InactiveUS20120245556A1Medical simulationPhysical therapies and activitiesGlycemicBlood glucose status
A method, system and computer program product for correcting a nominal treatment strategy of a subject with diabetes. The method, system and computer program product may be configured for providing input whereby the input may include: open-loop therapy settings for the subject, data about glycemic state of the subject; and (optionally) data about meals and / or exercise of the subject. The method, system and computer program product may be configured for providing output, whereby the out-put may include an adjustment (correction) to the open-loop therapy settings for the subject for insulin delivery to the subject.
Owner:UNIV OF VIRGINIA ALUMNI PATENTS FOUND
Soluble form of carbonic anhydrase IX (S-CA IX), assays to detect s-CA IX, CA IX'S coexpression with HER-2/NEU/C-ERBB-2, and CA IX-specific monoclonal antibodies to non-immunodominant epitopes
InactiveUS7816493B2Good curative effectSugar derivativesBiological material analysisKilodaltonC erbb 2
Disclosed herein is the discovery of a soluble MN / CA IX (s-CA IX) found in body fluids, such as, urine and serum. Soluble CA IX comprises the extracellular domain of CA IX or portions thereof. The predominant s-CA IX species is the extracellular domain comprising a proteoglycan-like (PG) domain and carbonic anhydrase (CA) domain, and having a molecular weight of about 50 / 54 kilodaltons. Diagnostic / prognostic methods for precancer / cancer that detect or detect and quantitate s-CA IX in body fluids, are described. Also disclosed is the coexpression of CA IX and HER-2 that provides potentially synergistic diagnostic / prognostic and therapeutic strategies for precancer / cancer. Further disclosed are new MN / CA IX-specific antibodies generated from MN / CA IX-deficient mice, useful diagnostically / prognostically and therapeutically for cancer / precancer. Preferred are new antibodies, specific for non-immunodominant epitopes of MN / CA IX, useful to detect soluble CA IX (s-CA IX) in body fluids, preferably in combination with antibodies specific to immunodominant epitopes of MN / CA IX.
Owner:BIOMEDICAL RES CENT OF THE SLOVAK ACADEMY OF SCI
Compositions and methods for treating proliferative disorders
ActiveUS9447185B2Prevent proliferationReduce inflammationNervous disorderAntipyreticImmunologic disordersNK Cell Receptors
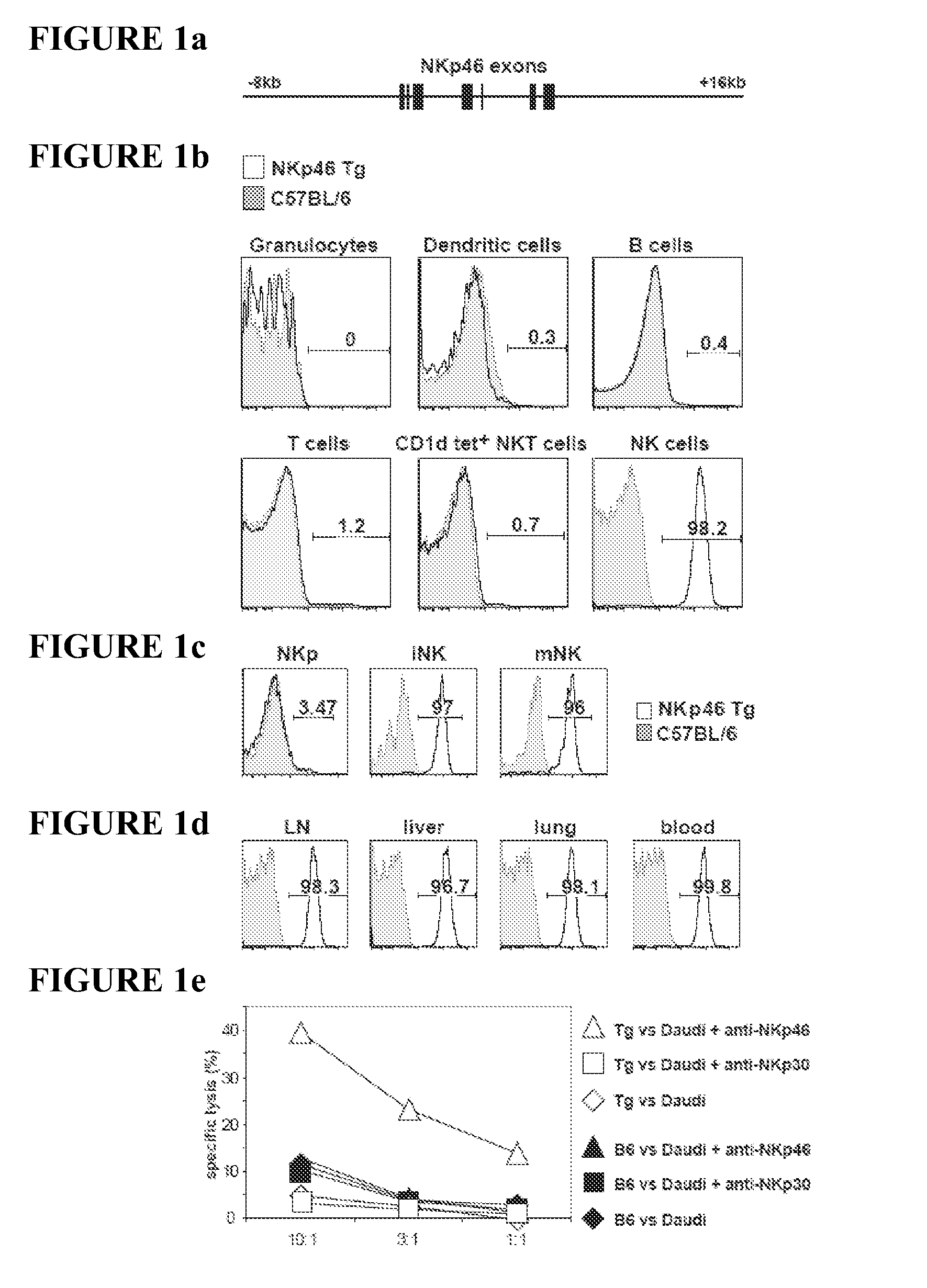
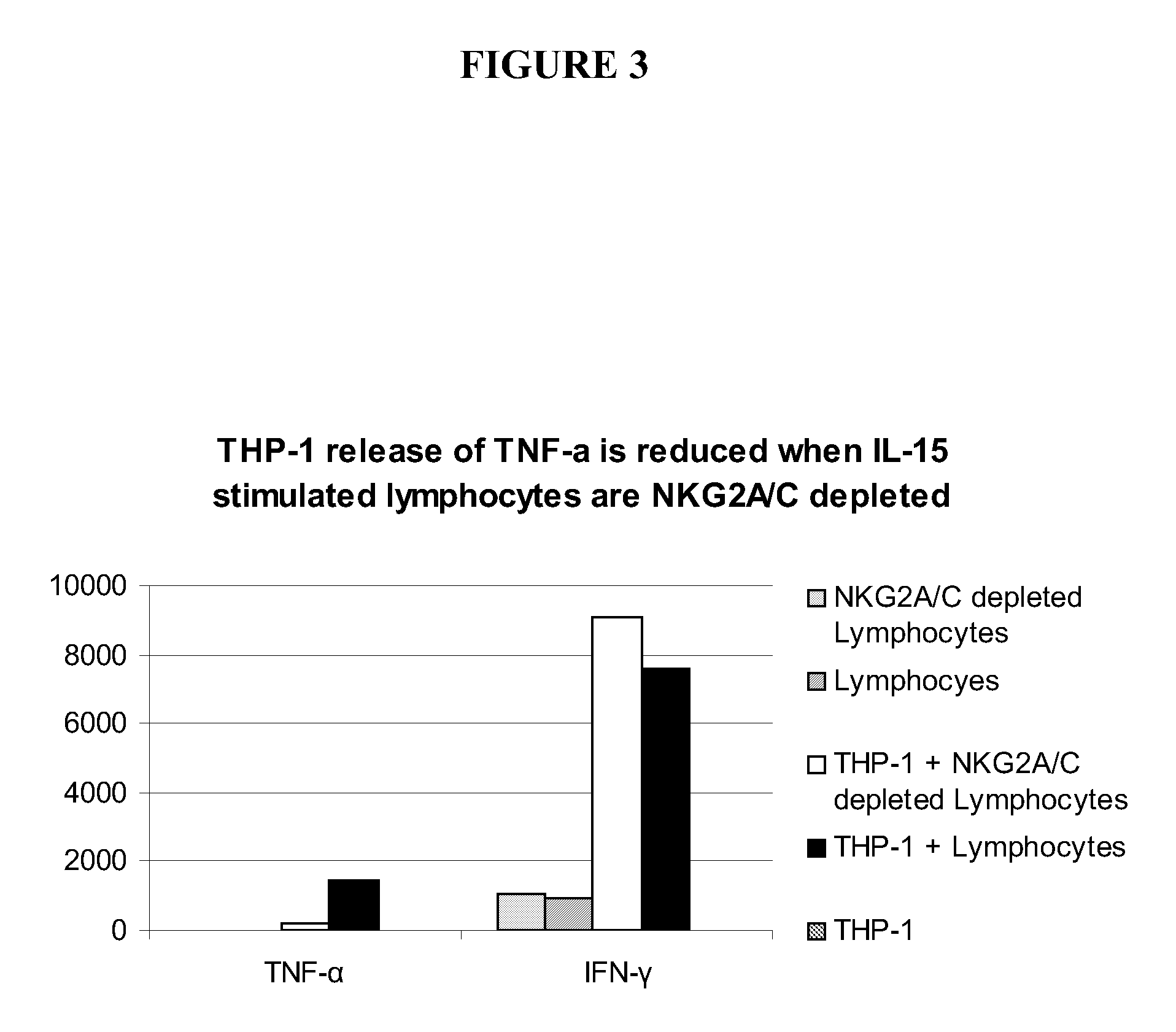
The present invention relates to methods of treating proliferative disorders, particularly immunoproliferative and autoimmune disorders, and methods of producing antibodies which bind NK cell receptors for use in therapeutic strategies for treating such disorders, particularly to deplete cells involved in the immunoproliferative pathology.
Owner:INNATE PHARMA SA +2
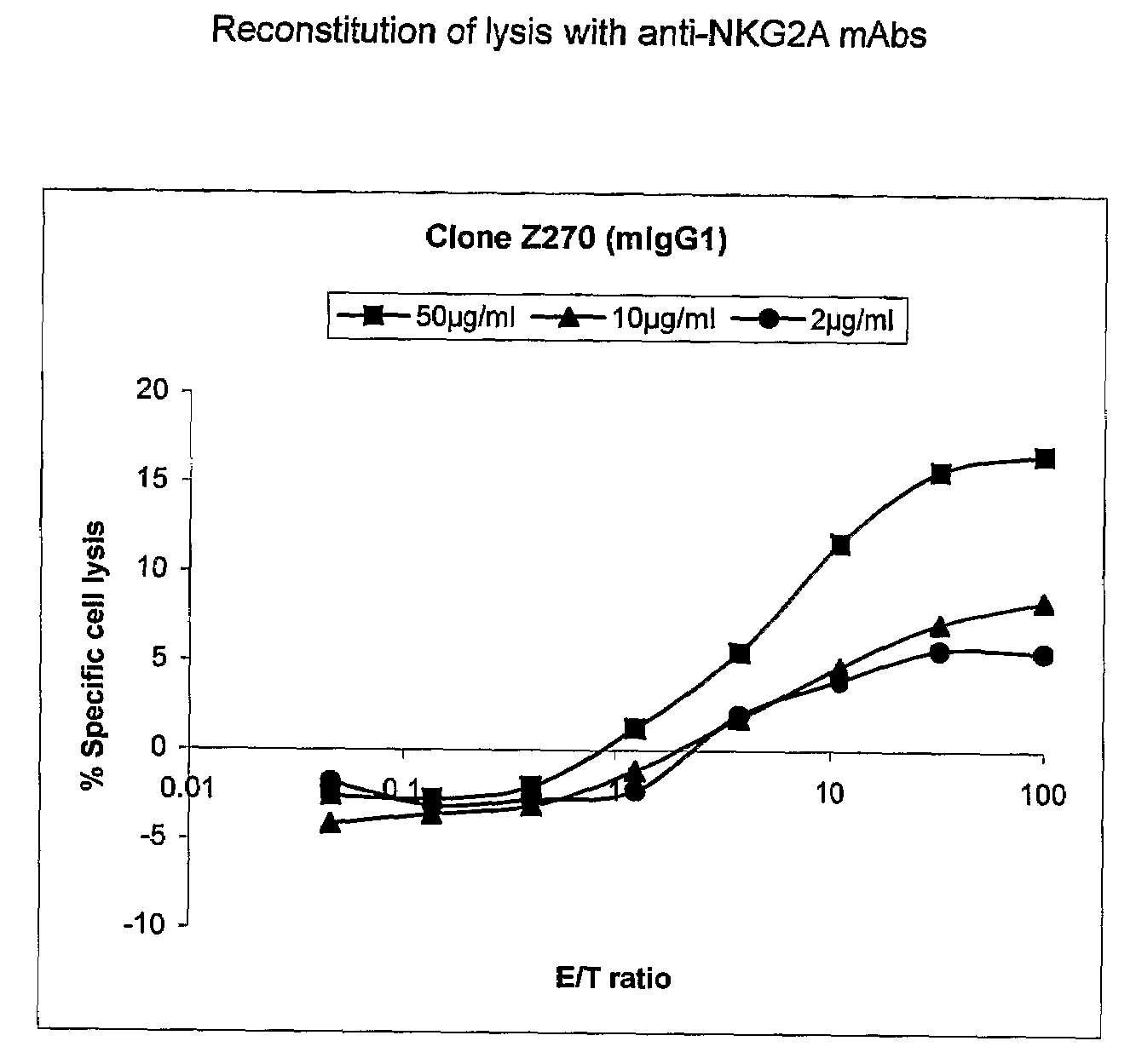
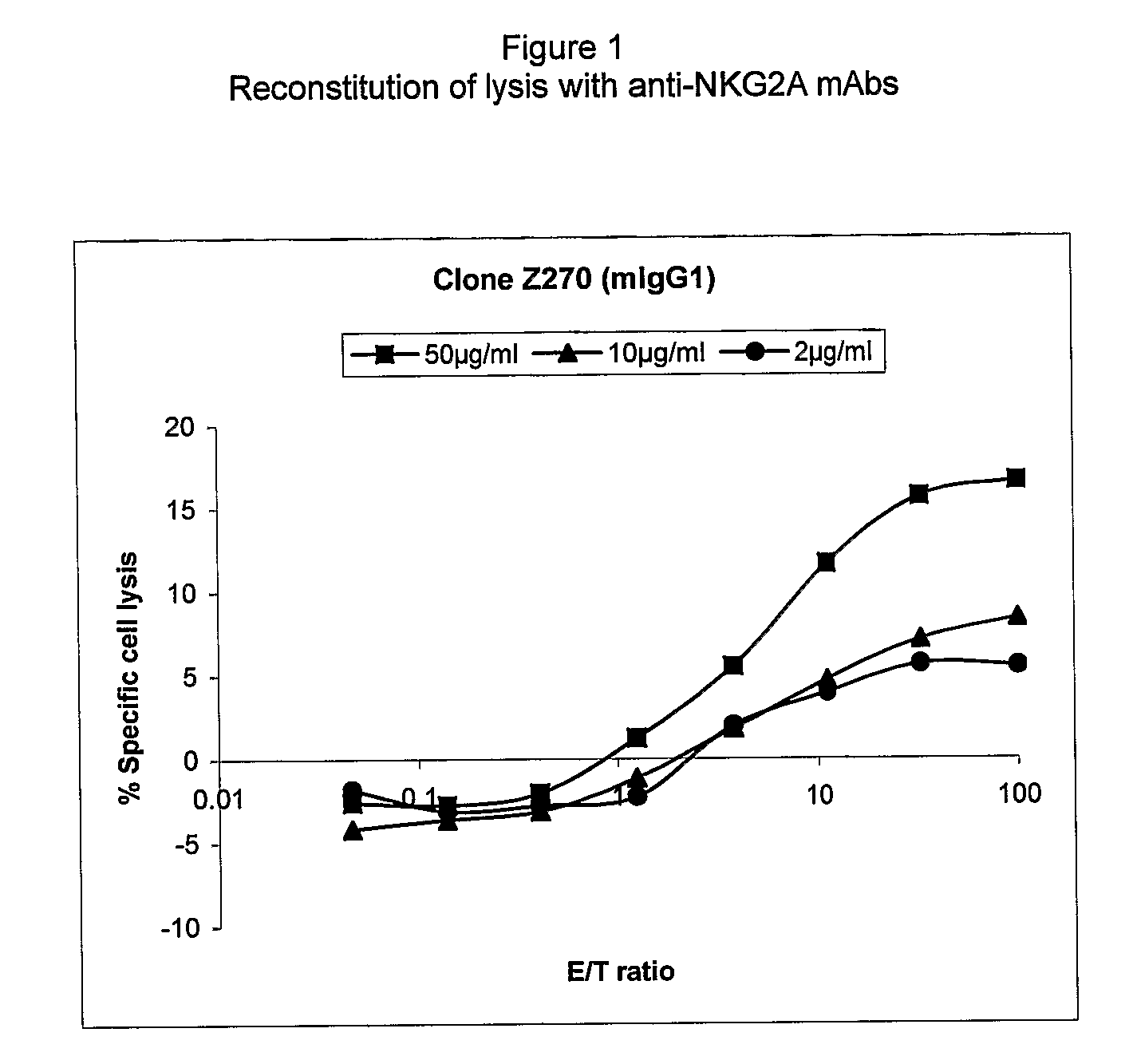
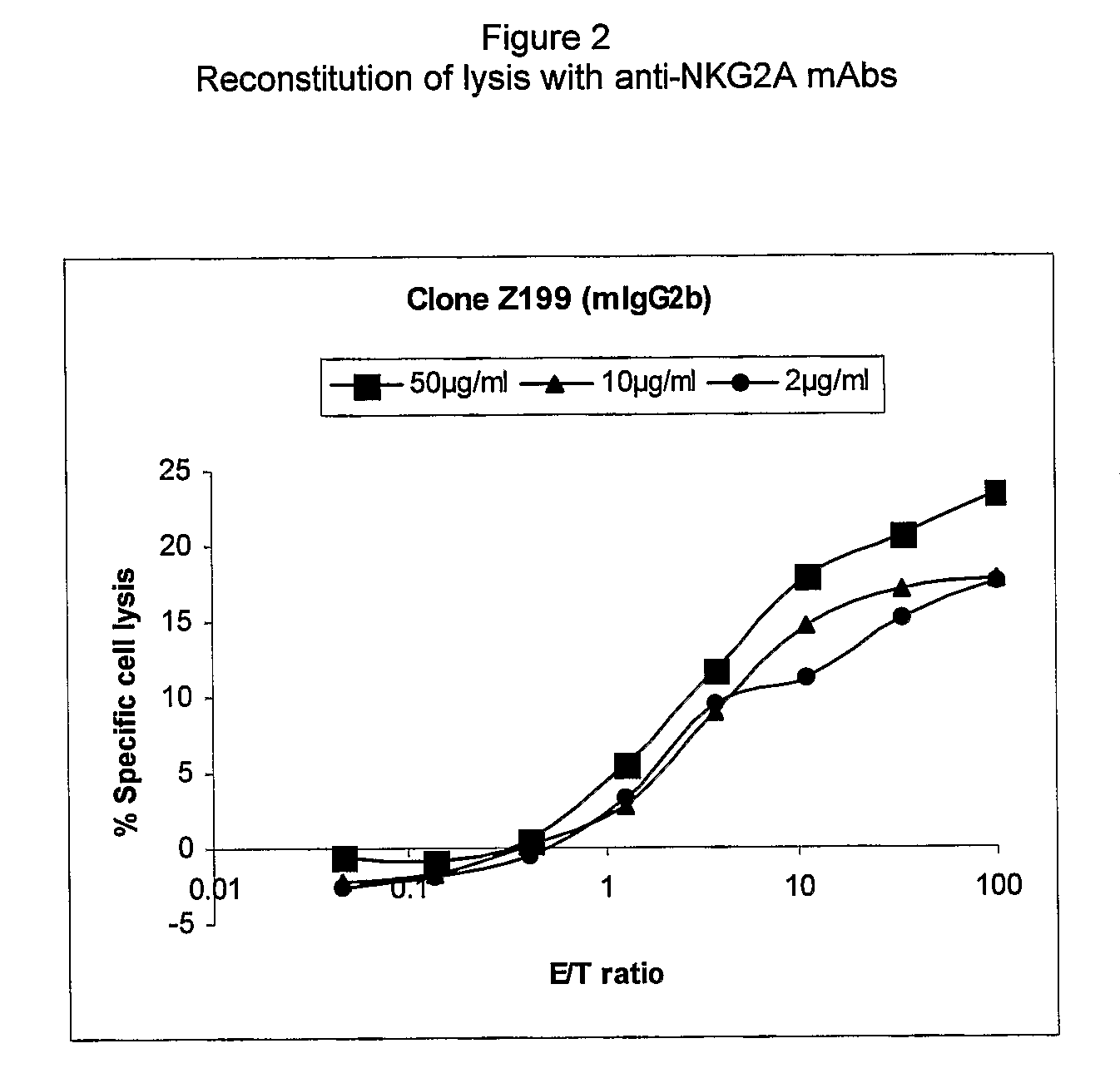
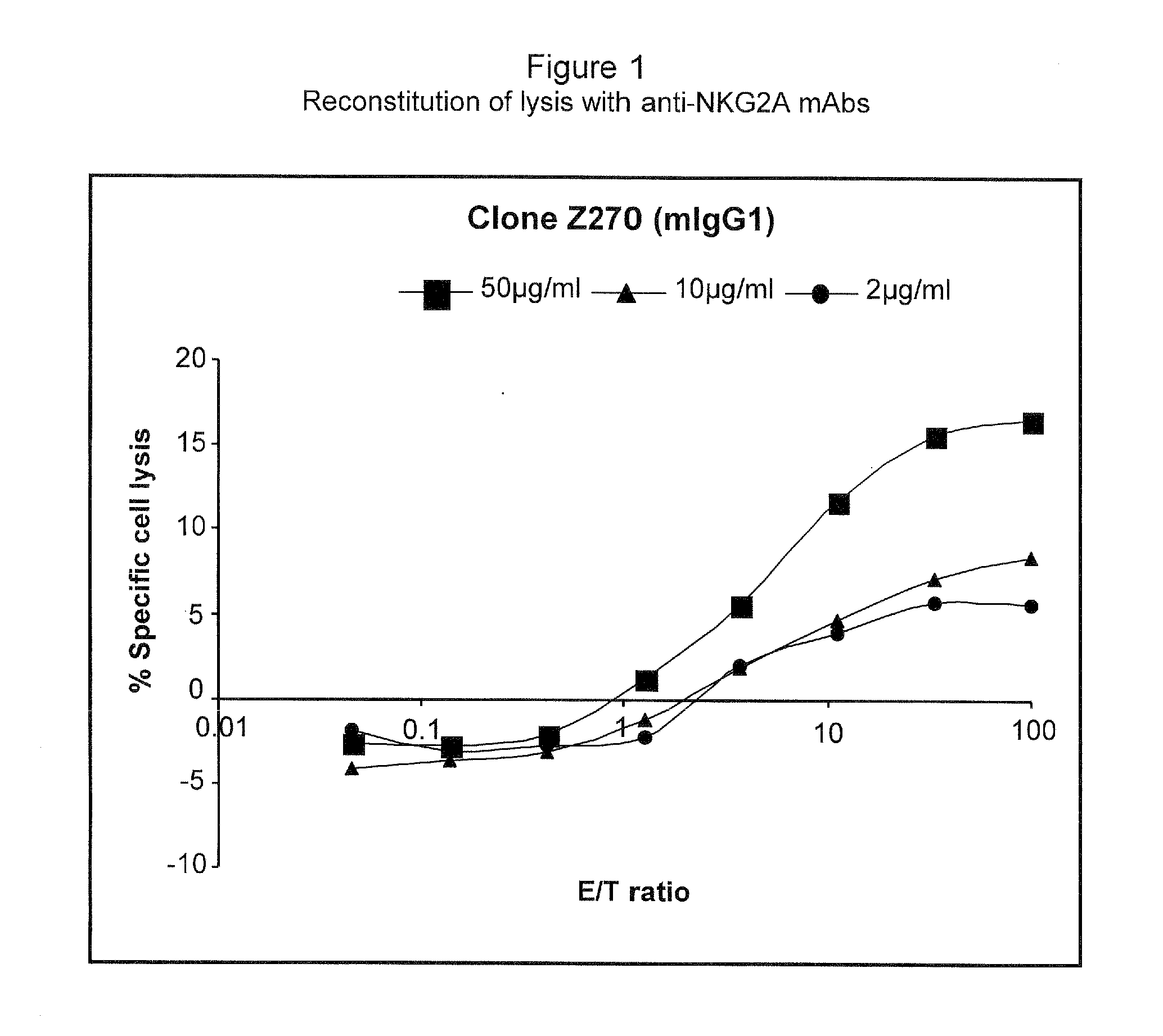
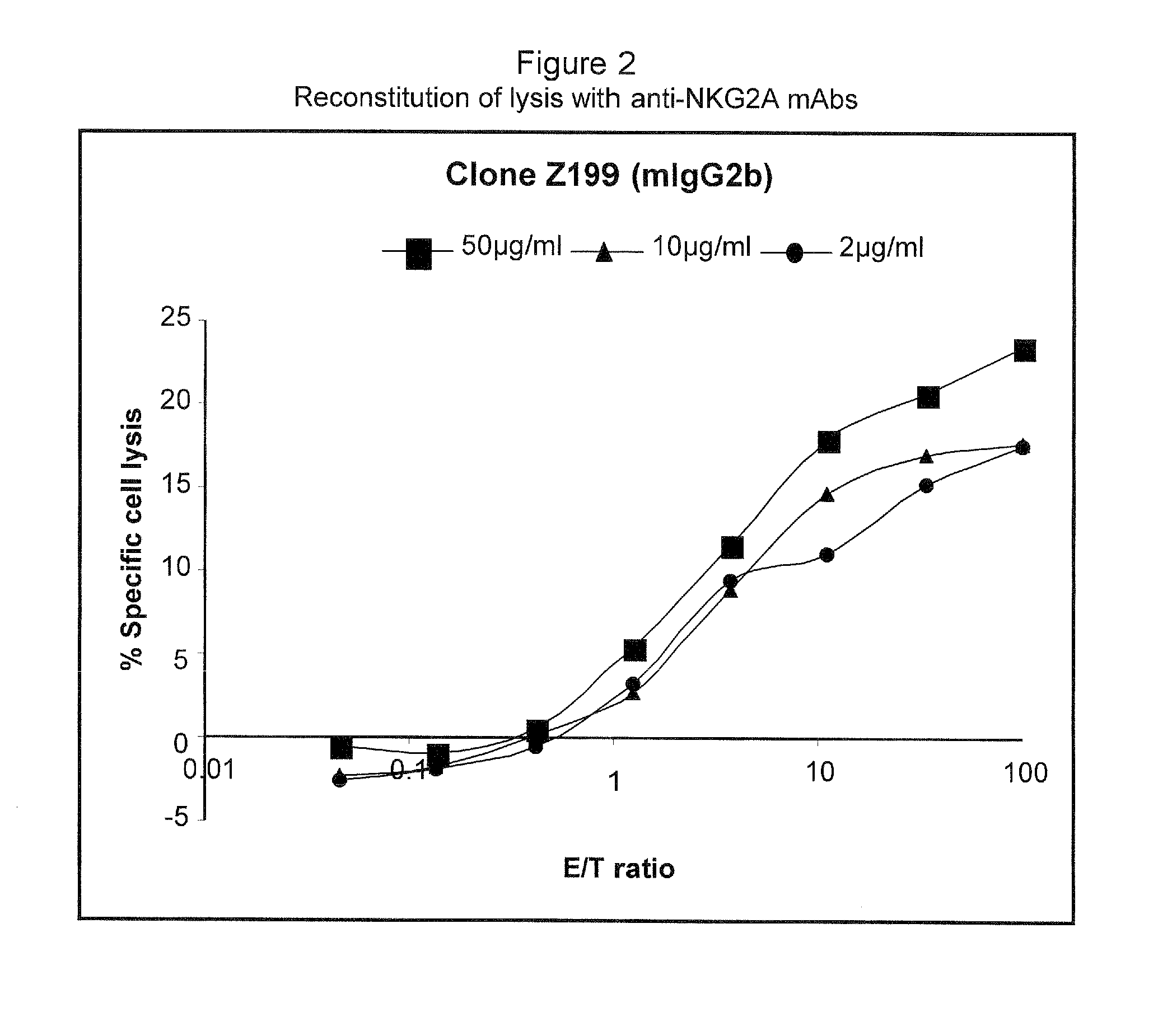
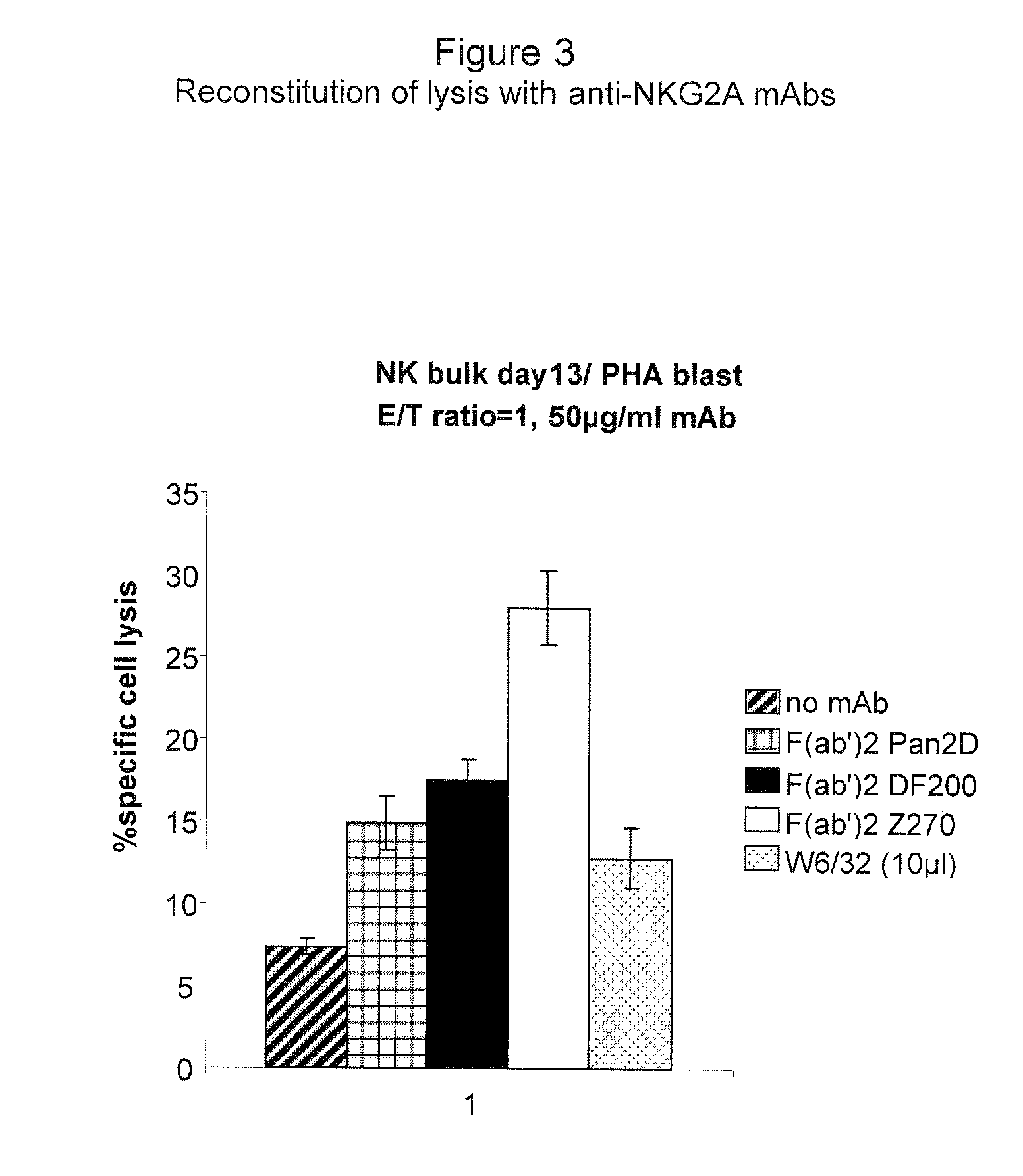
Monoclonal antibodies against nkg2a
ActiveUS20090208416A1Improve bioavailabilityImprove stabilitySenses disorderAntipyreticPresent methodDendritic cell
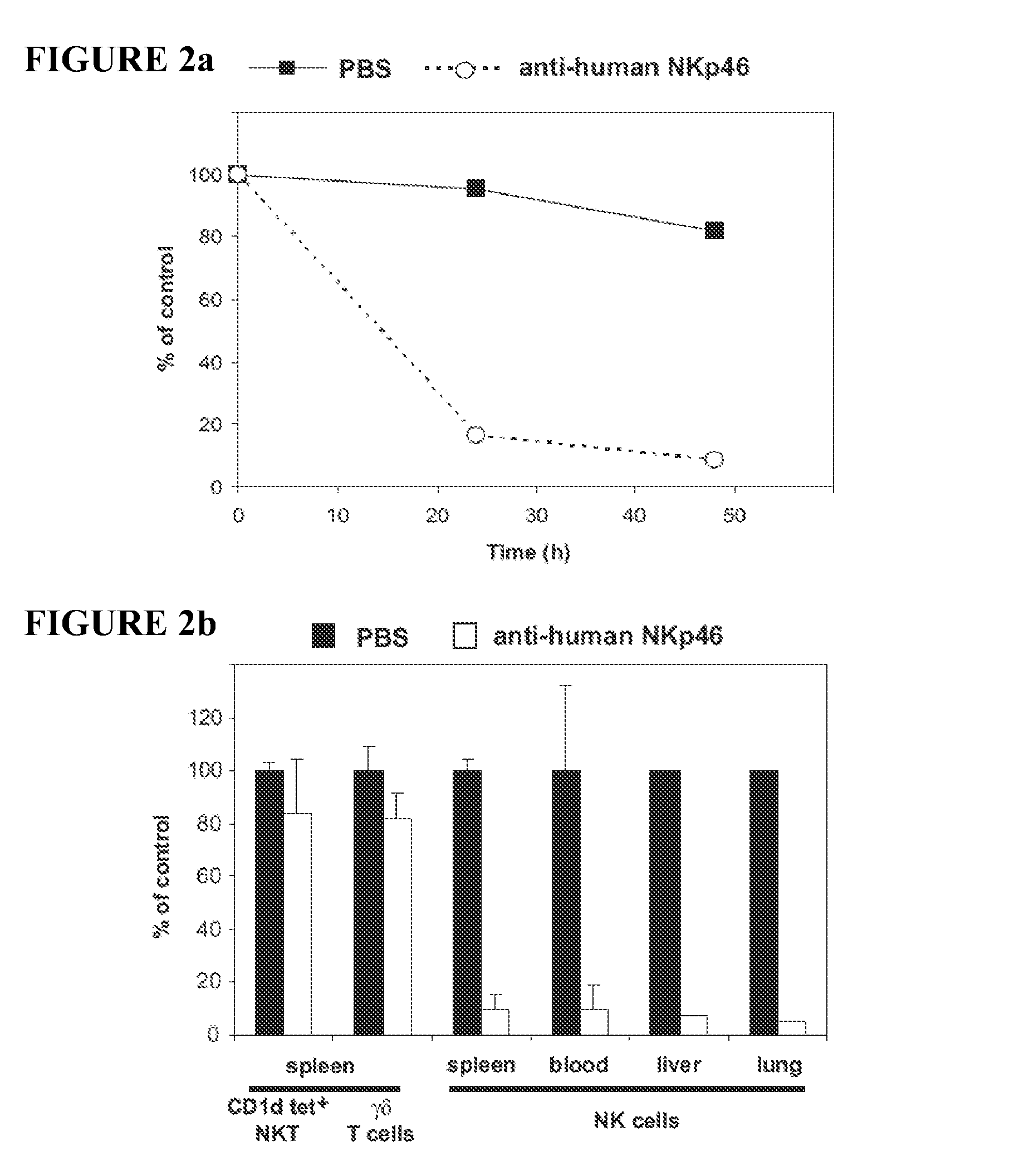
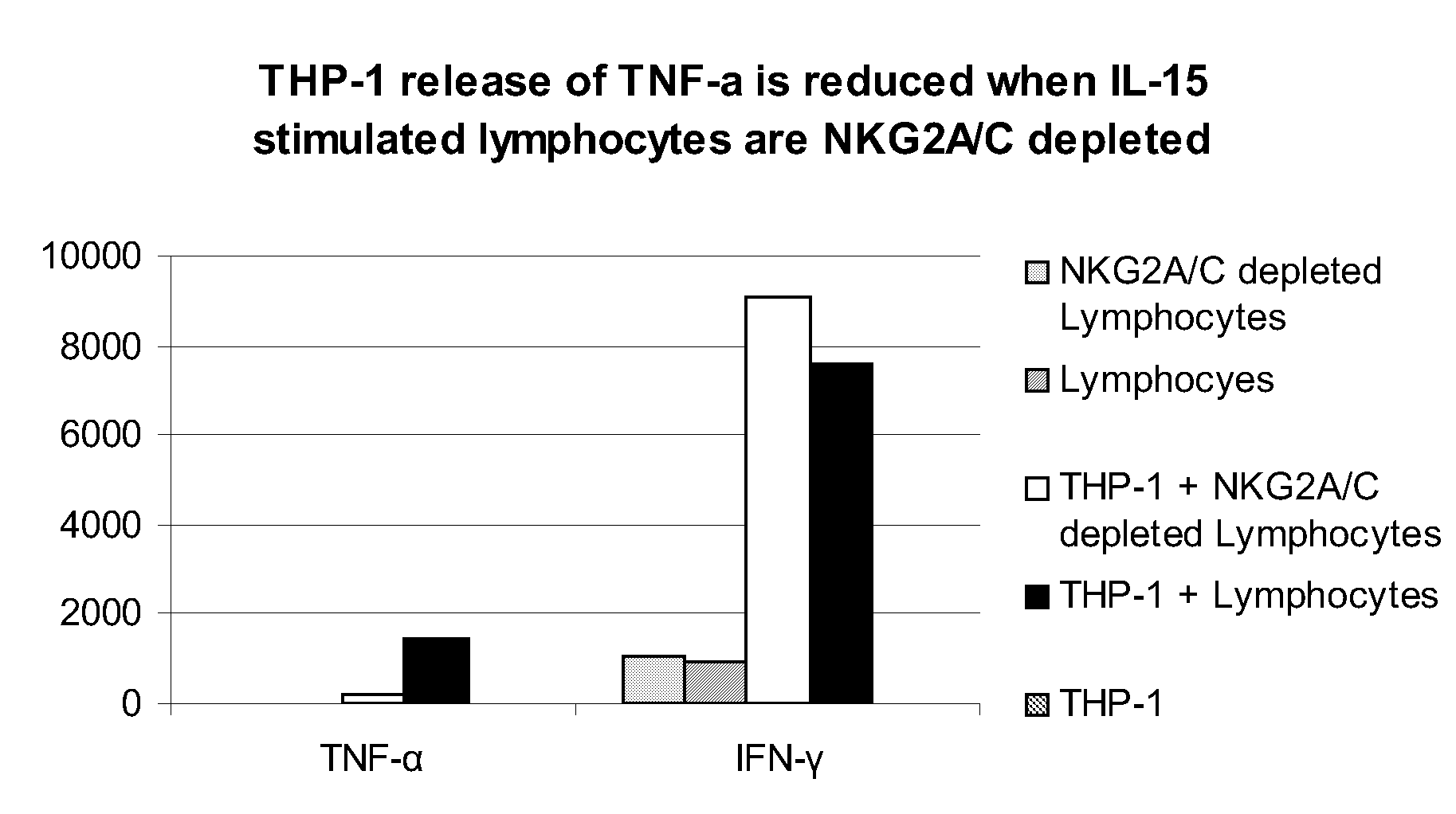
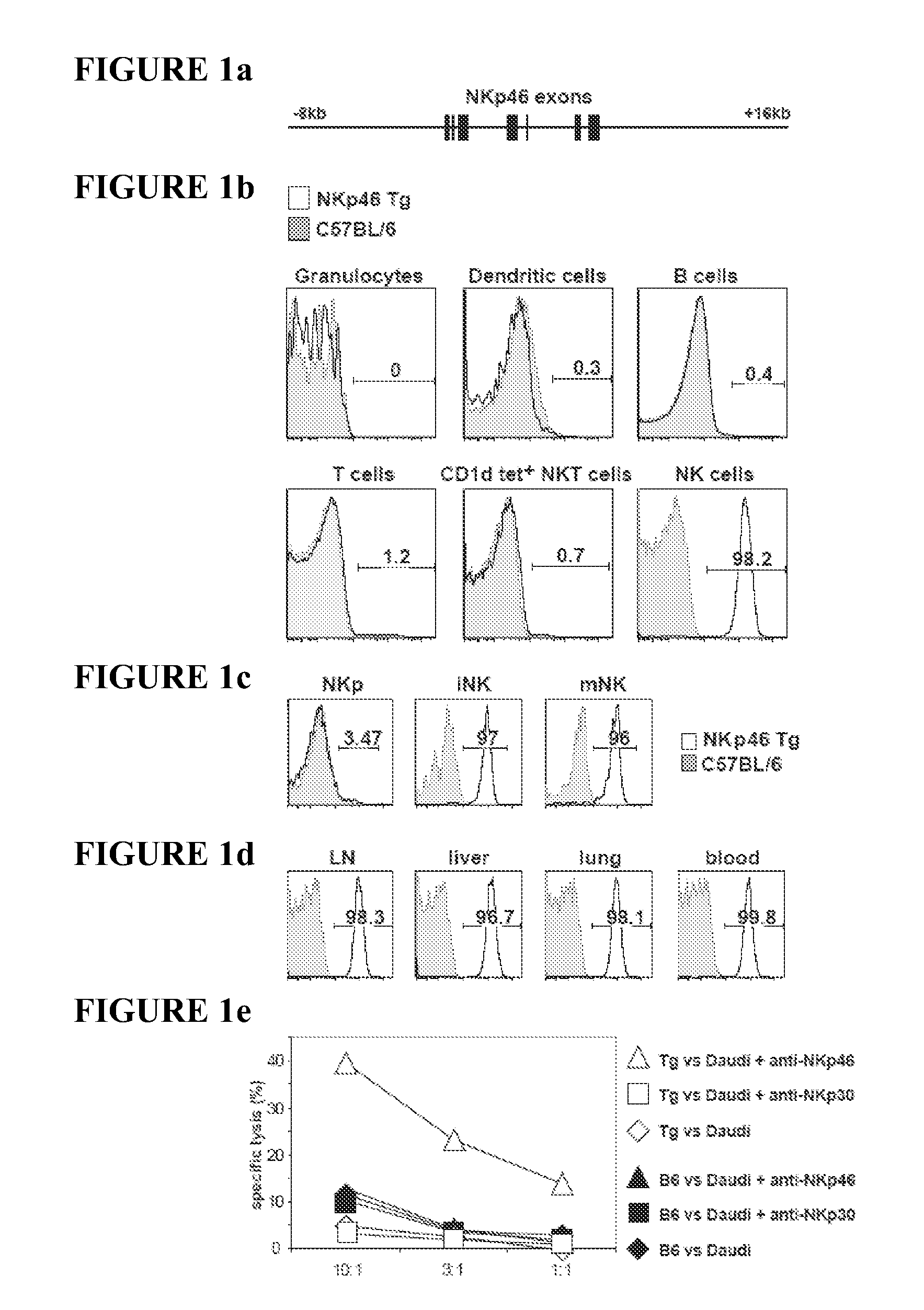
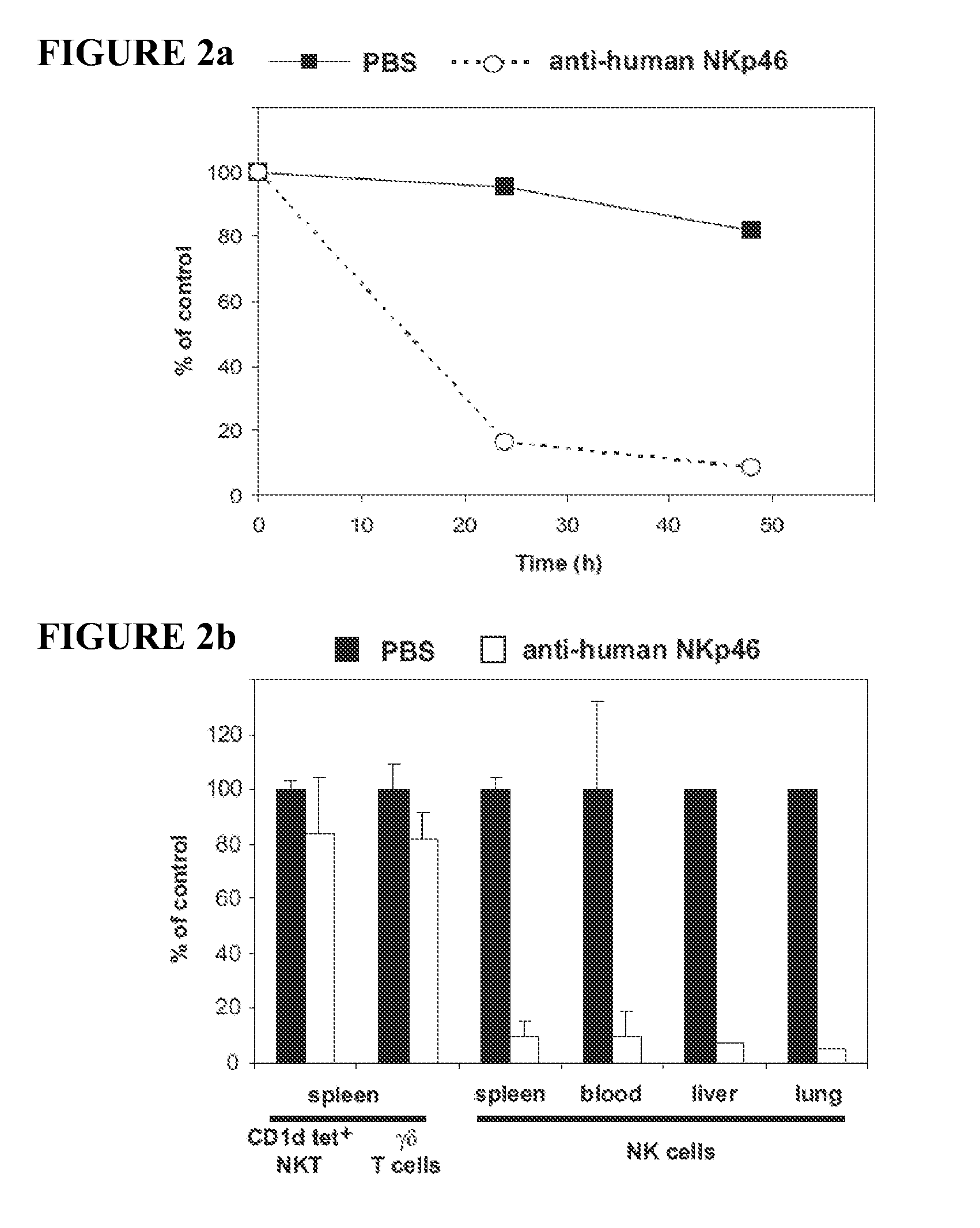
The present invention relates to methods of treating immune disorders, particularly autoimmune or inflammatory disorders, and methods of producing antibodies and other compounds for use in therapeutic strategies for treating such disorders. Generally, the present methods involve the use of antibodies or other compounds that prevent the stimulation of NKG2A receptors on NK cells, leading to the lysis of dendritic cells that contribute to the pathology of the disorders.
Owner:UNIV DEGLI STUDI DI GENOVA GENOVA IT +1
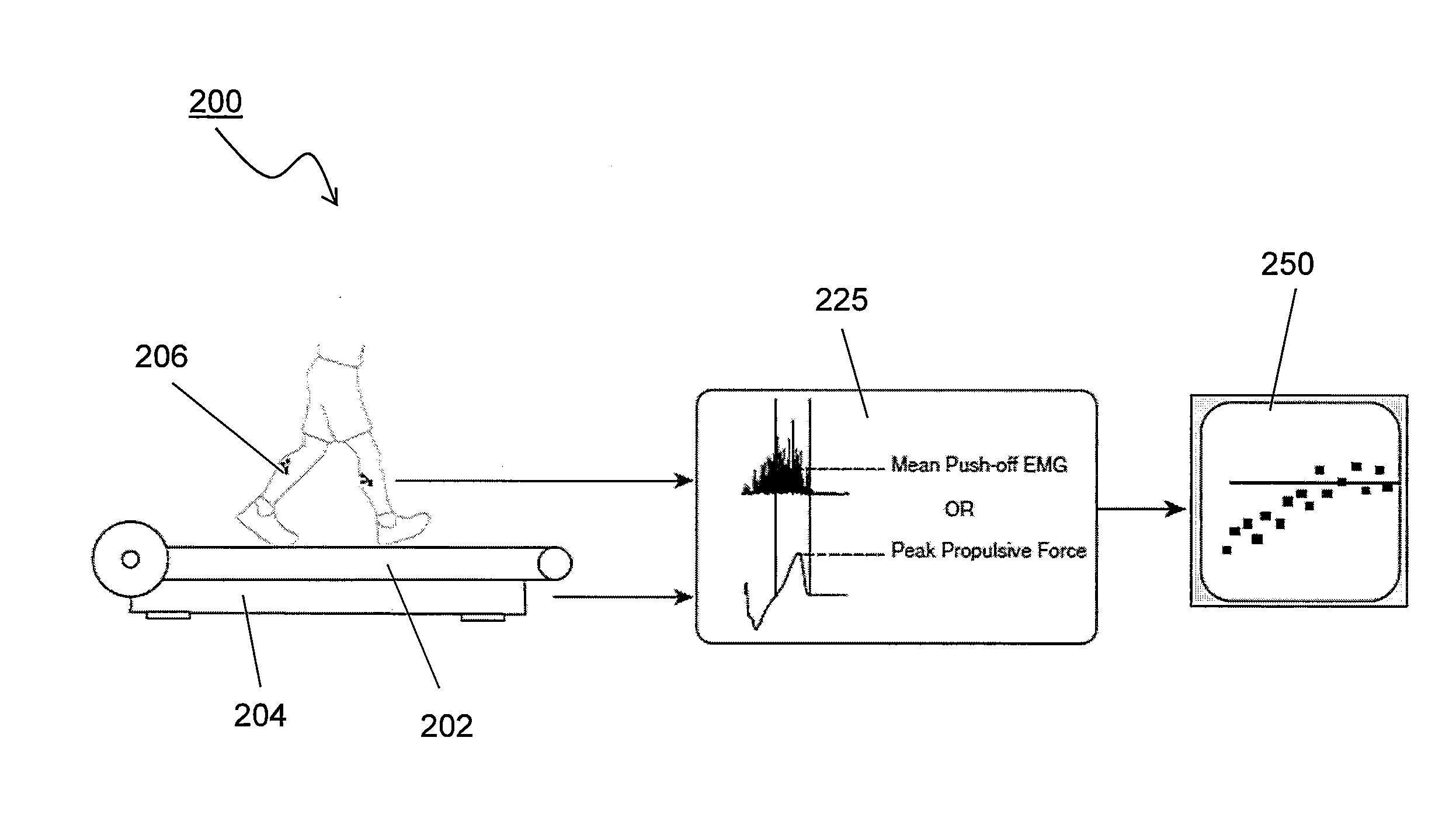
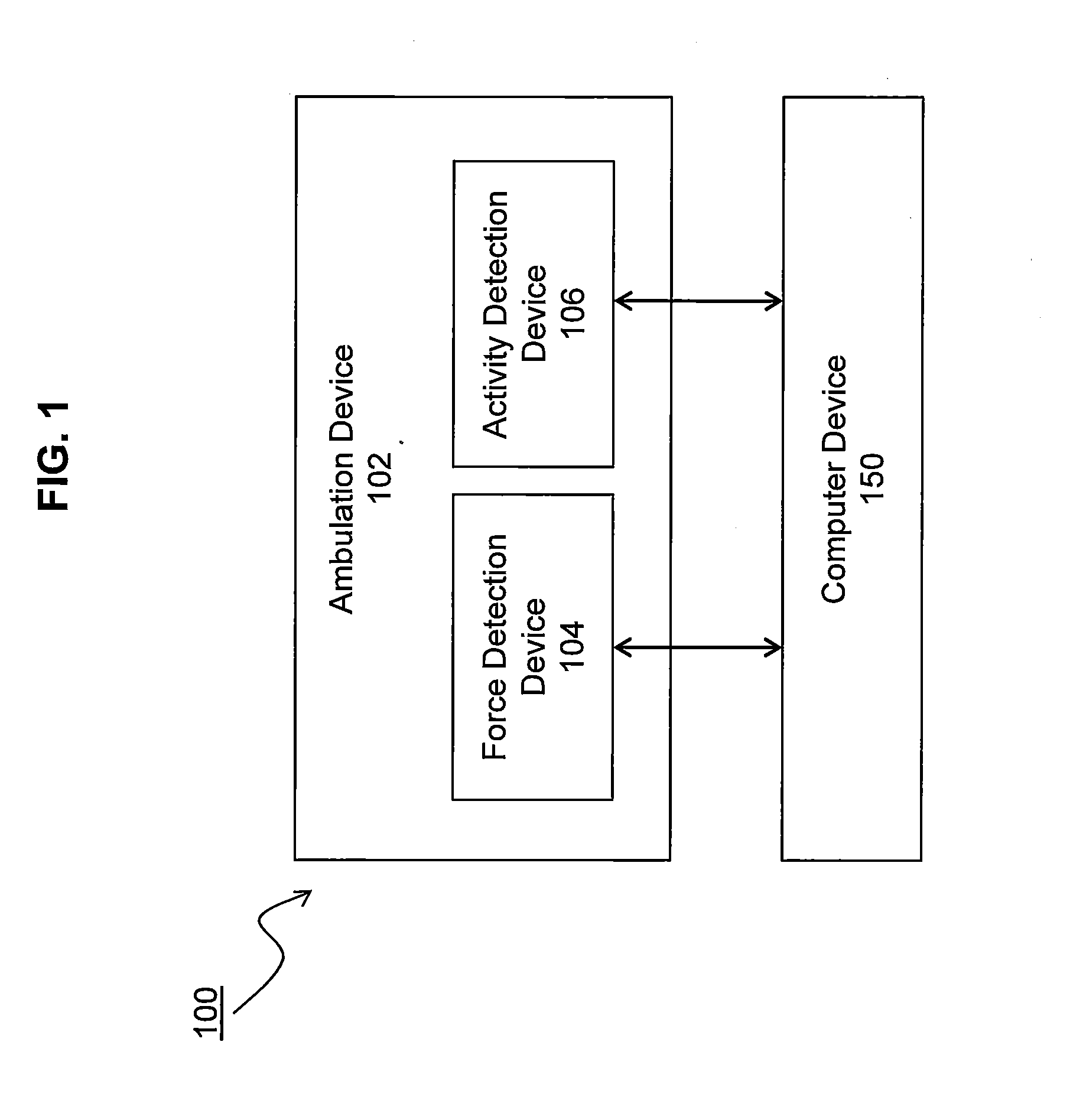
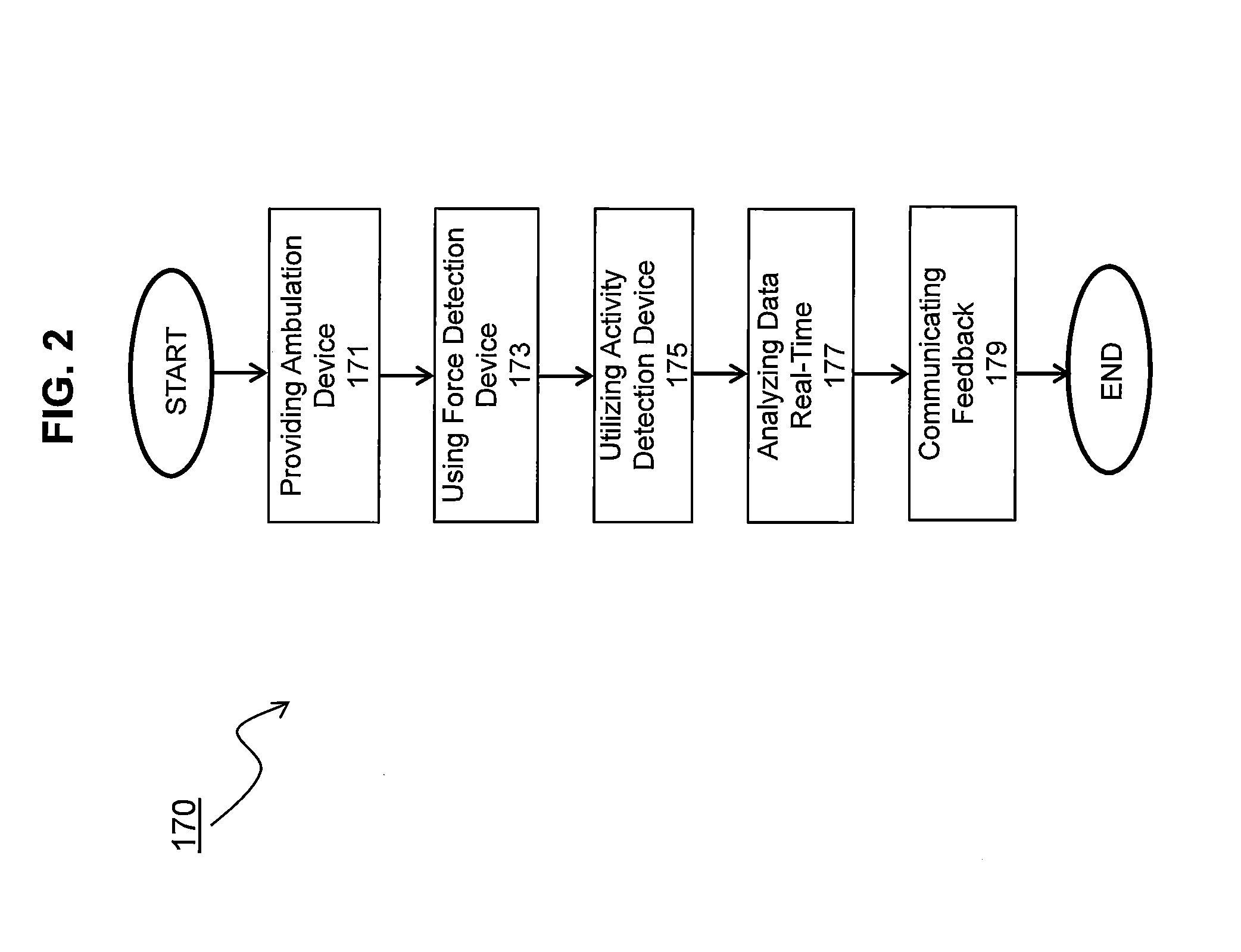
System and methods for measuring propulsive force during ambulation and providing real-time feedback
InactiveUS20140336003A1Increased forward propulsionEasy to understandDiagnostic recording/measuringSensorsPhysical medicine and rehabilitationDuring ambulation
The invention measures propulsive force of an ambulating subject to provide real-time feedback, which may be used for clinical assessment or rehabilitation / training such as that related to walking ability, or any other form of ambulation. Subjects with propulsive deficits have a considerable and underutilized propulsive reserve available during level ambulation. The invention uses real-time propulsive feedback as a therapeutic strategy to encourage a subject to access the propulsive reserve and improve forward propulsion during ambulation.
Owner:UNIV OF COLORADO THE REGENTS OF
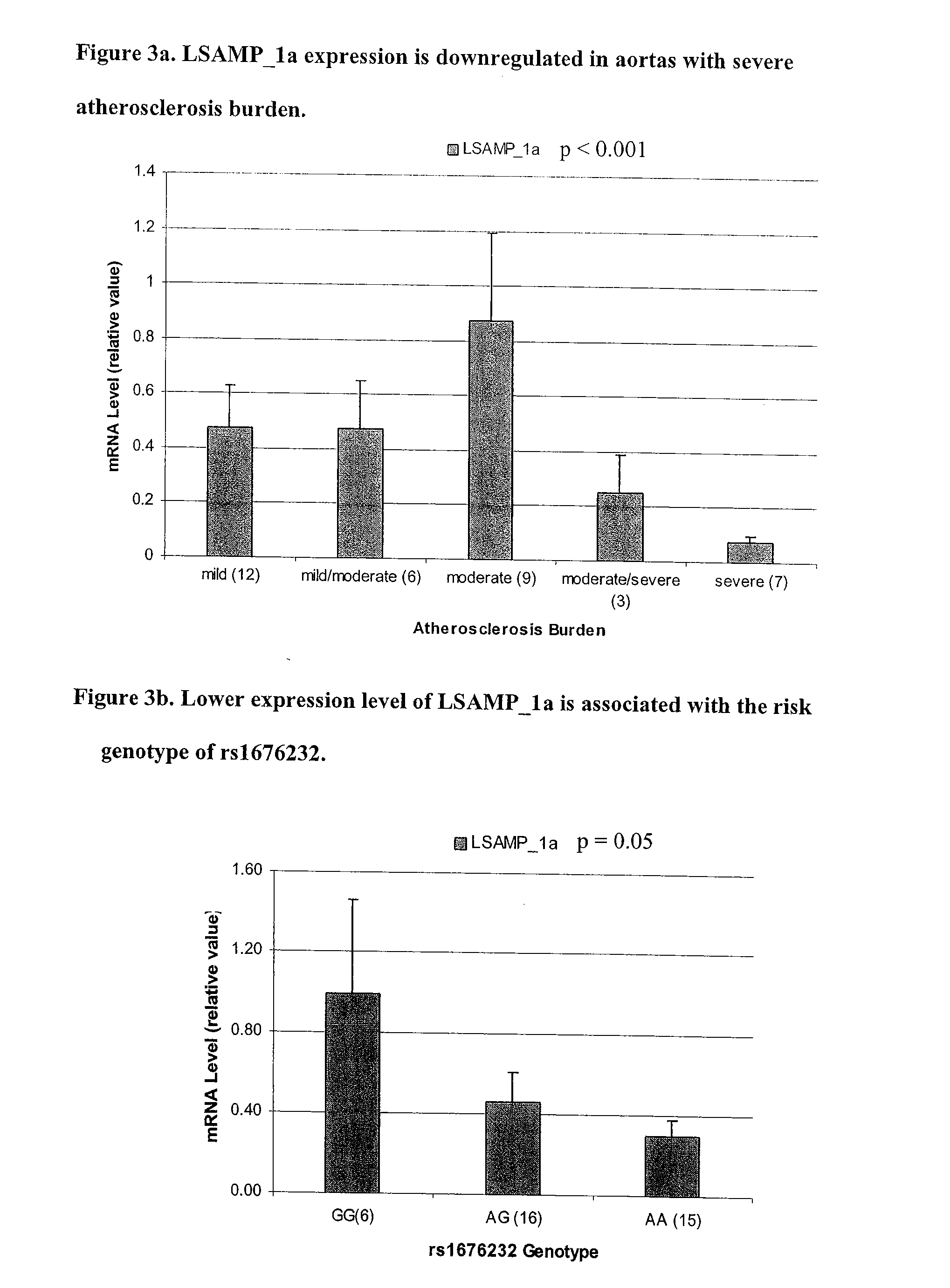
LSAMP Gene Associated With Cardiovascular Disease
InactiveUS20070148661A1Predicting risk of cardiovascularMicrobiological testing/measurementBiological testingGenetic riskEarly initiation
The LSAMP gene can be used for cardiovascular disease risk assessment, in particular Left Main Disease. The genetic risk attributable to LSAMP adds to known cardiovascular disease risk factors. Assessment of risk attributable to LSAMP permits early initiation of preventive and therapeutic strategies. Given the pronounced clinical risk associated with Left Main Disease, such risk assessment should significantly reduce morbidity and mortality.
Owner:DUKE UNIV
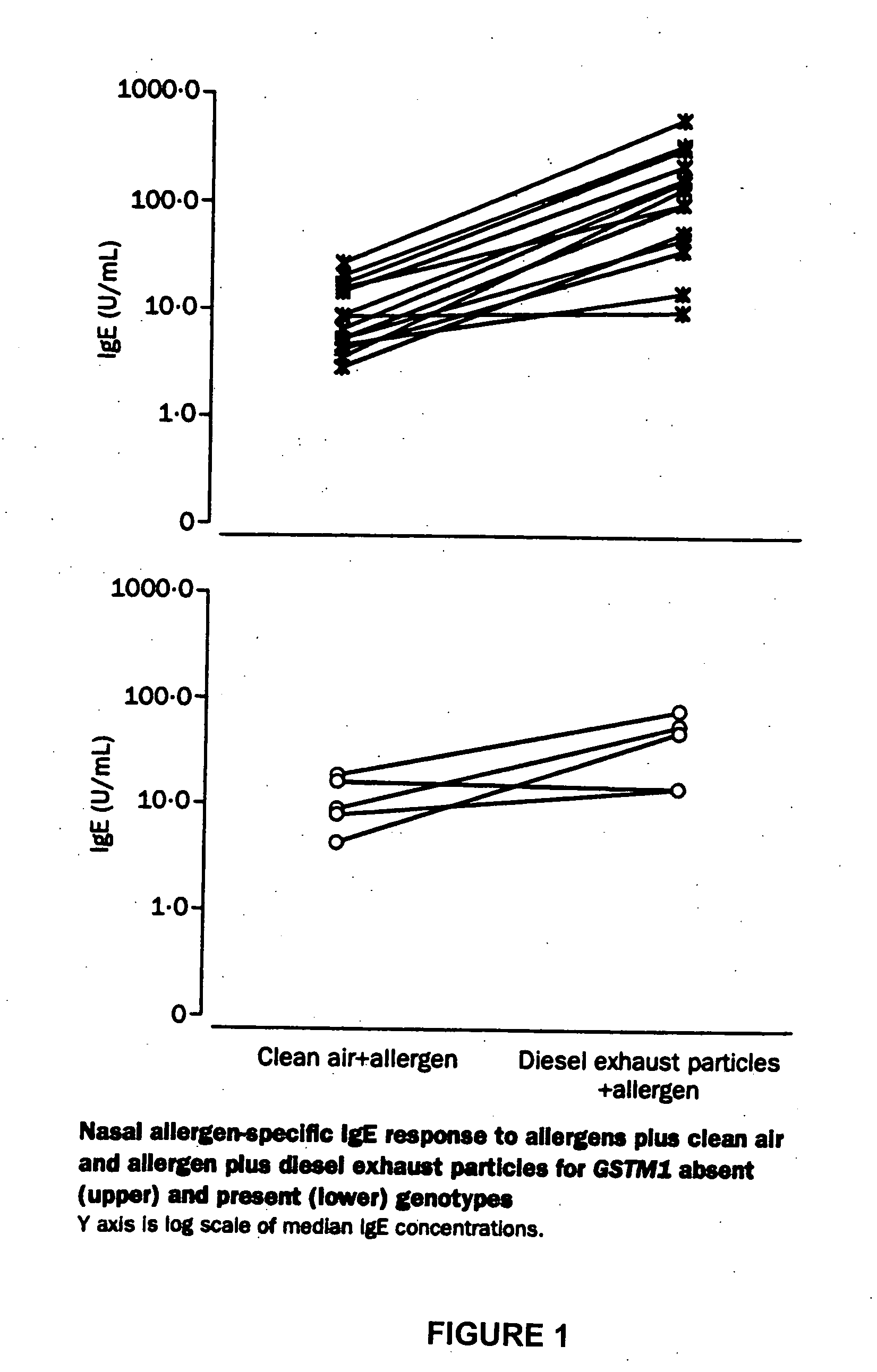
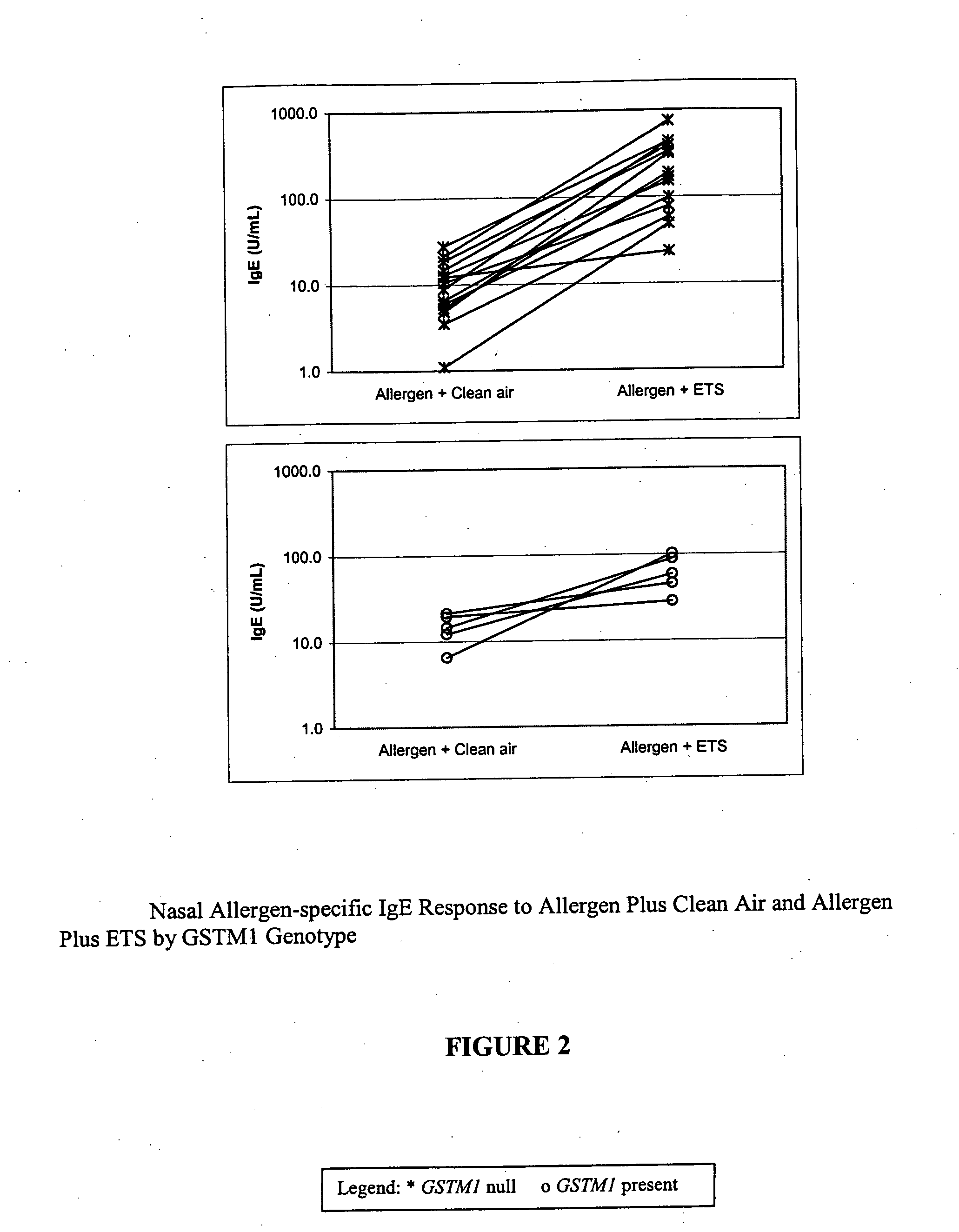
Method for determining susceptibility of an individual to allergen induced hypersensitivity
The present invention is related generally to a method for screening subjects to determine those subjects more likely to exhibit a hypersensitivity to allergen in the presence of airborne pollutants, such subject having a GSTM1 null genotype and / or a GSTP1 Ile / Ile genotype. This invention also provides novel or improved pharmaceutical compositions and therapeutic strategies for the treatment of immune diseases resulting from inappropriate or unwanted immune response such as allergic reactions.
Owner:RGT UNIV OF CALIFORNIA +2
Anorectic
The present invention relates to an anorectic containing a compound having a DGAT inhibitory activity (DGAT1 inhibitory activity) or a prodrug thereof or a pharmaceutically acceptable salt thereof as an active ingredient. The present invention provides an anti-obesity drug which is an anorectic that does not directly act on the central nervous system and is satisfactory in terms of activity, and a therapeutic strategy for preventing or treating obesity.
Owner:JAPAN TOBACCO INC
Combination therapies for hematologic malignancies
InactiveUS20130064812A1Improved profileNo adverse effect on glucose and insulin levelBiocideDipeptide ingredientsHematologic malignancyBendamustine
The invention provides methods that relate to a novel therapeutic strategy for the treatment of hematological malignancies and inflammatory diseases. In particular, the method comprises administration of a compound of formula A,wherein R is H, halo, or C1-C6 alkyl; R′ is C1-C6 alkyl; ora pharmaceutically acceptable salt thereof; andoptionally a pharmaceutically acceptable excipient; andone or more additional therapeutic agents optionally selected from the group consisting of bendamustine, rituximab, and ofatumumab.
Owner:GILEAD CALISTOGA
Combination therapies for treating cancers
InactiveUS20160220573A1Relieve symptomsModulate activityOrganic active ingredientsAntineoplastic agentsOncologyEntospletinib
Provided herein are methods that relate to a therapeutic strategy for treatment of cancer, including hematological malignancies. In particular, the methods include administration entospletinib and a Bcl-2 inhibitor, such as venetoclax, navitoclax, and ABT-737.
Owner:KRONOS BIO INC
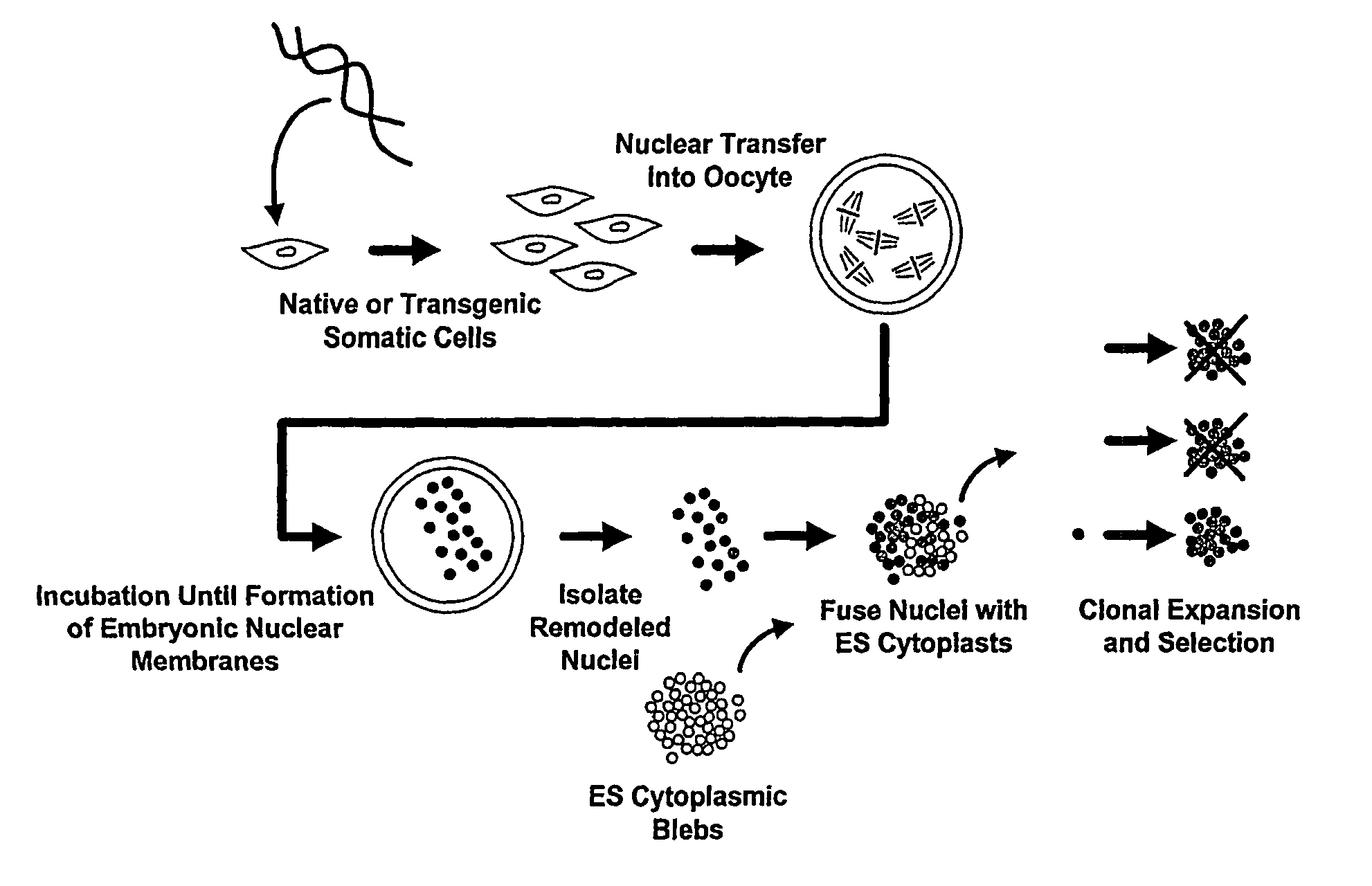
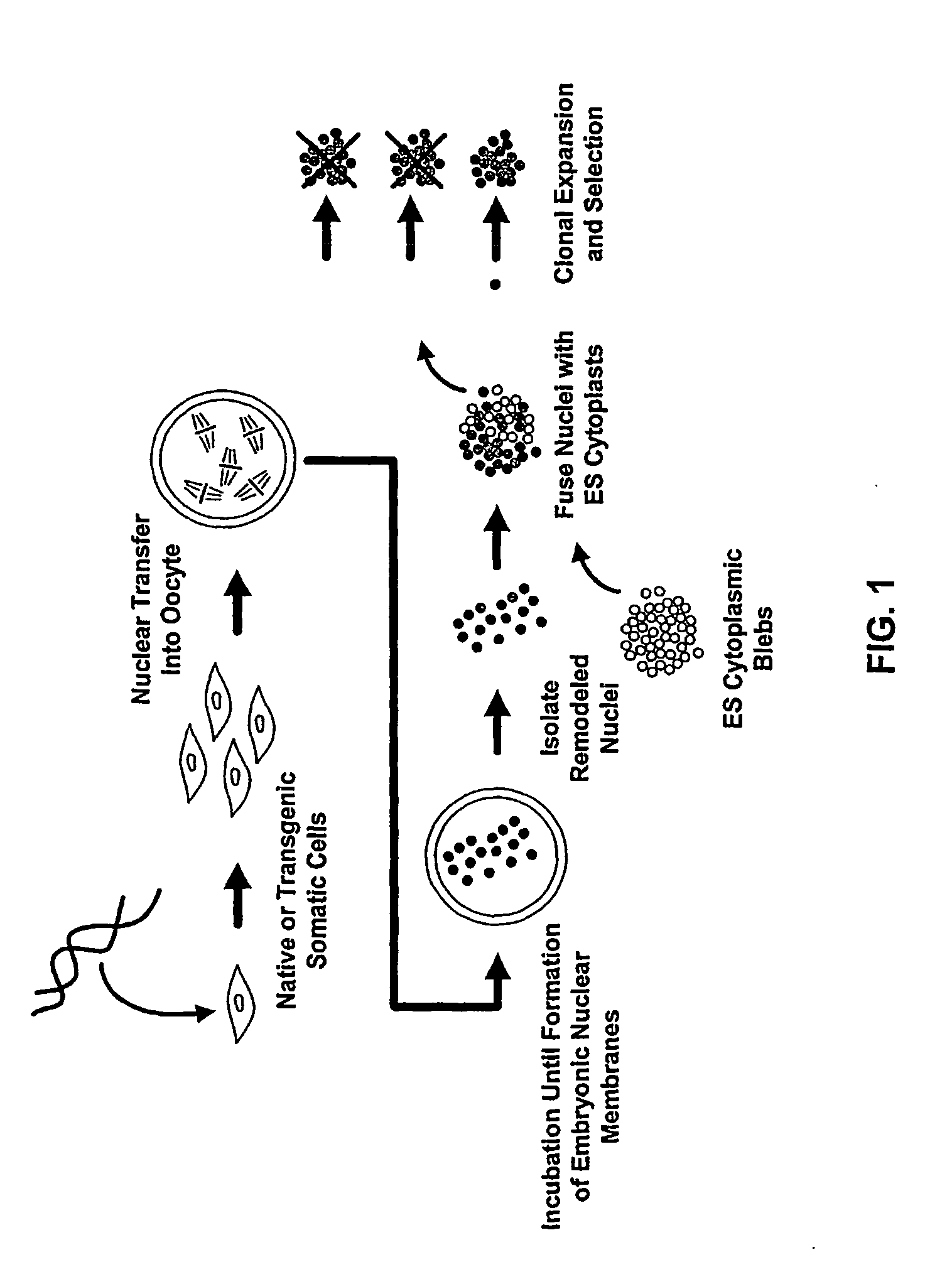
Methods of Reprogramming Animal Somatic Cells
InactiveUS20100167404A1Extend the lifespanExtending telomere lengthCell differentiationMicrobiological testing/measurementMammalEmbryo
This invention generally relates to methods to obtain mammalian cells and tissues with patterns of gene expression similar to that of a developing mammalian embryo or fetus, and the use of such cells and tissues in the treatment of human disease and age-related conditions. More particularly, the invention relates to methods for identifying, expanding in culture, and formulating mammalian pluripotent stem cells and differentiated cells that differ from cells in the adult human in their pattern of gene expression, and therefore offer unique characteristics that provide novel therapeutic strategies in the treatment of degenerative disease.
Owner:ADVANCED CELL TECH INC
Usages of MTHFR gene polymorphisms in predicting homocysteine level, disease risk, and treatment effects and related methods and kit
InactiveUS20070134709A1Improve the level ofBiocideBioreactor/fermenter combinationsDisease riskTreatment effect
This invention features our discovery on usages of Methylenetetrahydrofolate Reductase (MTHFR) gene polymorphisms in predicting homocysteine (Hcy) level and / or incidence and prognosis of diseases associated with increased Hcy level in a subject, as well as predicting treatment effects of medicines in the category of Angiotension Converting Enzyme Inhibihor (ACEI) with and without combination with B Vitamins. This invention also features our discovery on laboratory and analytical methods that are essential to the above described usages of MTHFR gene polymorphisms. In addition, this invention features a kit that has translated the above discoveries into a practical and reliable tool that can be applied to accomplish the above described usages of MTHFR gene polymorphisms. This invention represents an important step in realizing personalized medicine, with the goal to tailor diagnosis, prevention and treatment strategy to meet individual needs.
Owner:XU XIPING +14
Compositions and Methods for Treating Proliferative Disorders
ActiveUS20080274047A1Reduce inflammationReduce in quantityNervous disorderAntipyreticDiseaseNK Cell Receptors
The present invention relates to methods of treating proliferative disorders, particularly immunoproliferative and autoimmune disorders, and methods of producing antibodies which bind NK cell receptors for use in therapeutic strategies for treating such disorders, particularly to deplete cells involved in the immunoproliferative pathology.
Owner:INNATE PHARMA SA +2
Monoclonal Antibodies Against NKG2A
InactiveUS20110229486A1Improve bioavailabilityImprove stabilityCompound screeningSenses disorderDendritic cellPresent method
The present invention relates to methods of treating immune disorders, particularly autoimmune or inflammatory disorders, and methods of producing antibodies and other compounds for use in therapeutic strategies for treating such disorders. Generally, the present methods involve the use of antibodies or other compounds that prevent the stimulation of NKG2A receptors on NK cells, leading to the lysis of dendritic cells that contribute to the pathology of the disorders.
Owner:INNATE PHARMA SA +1
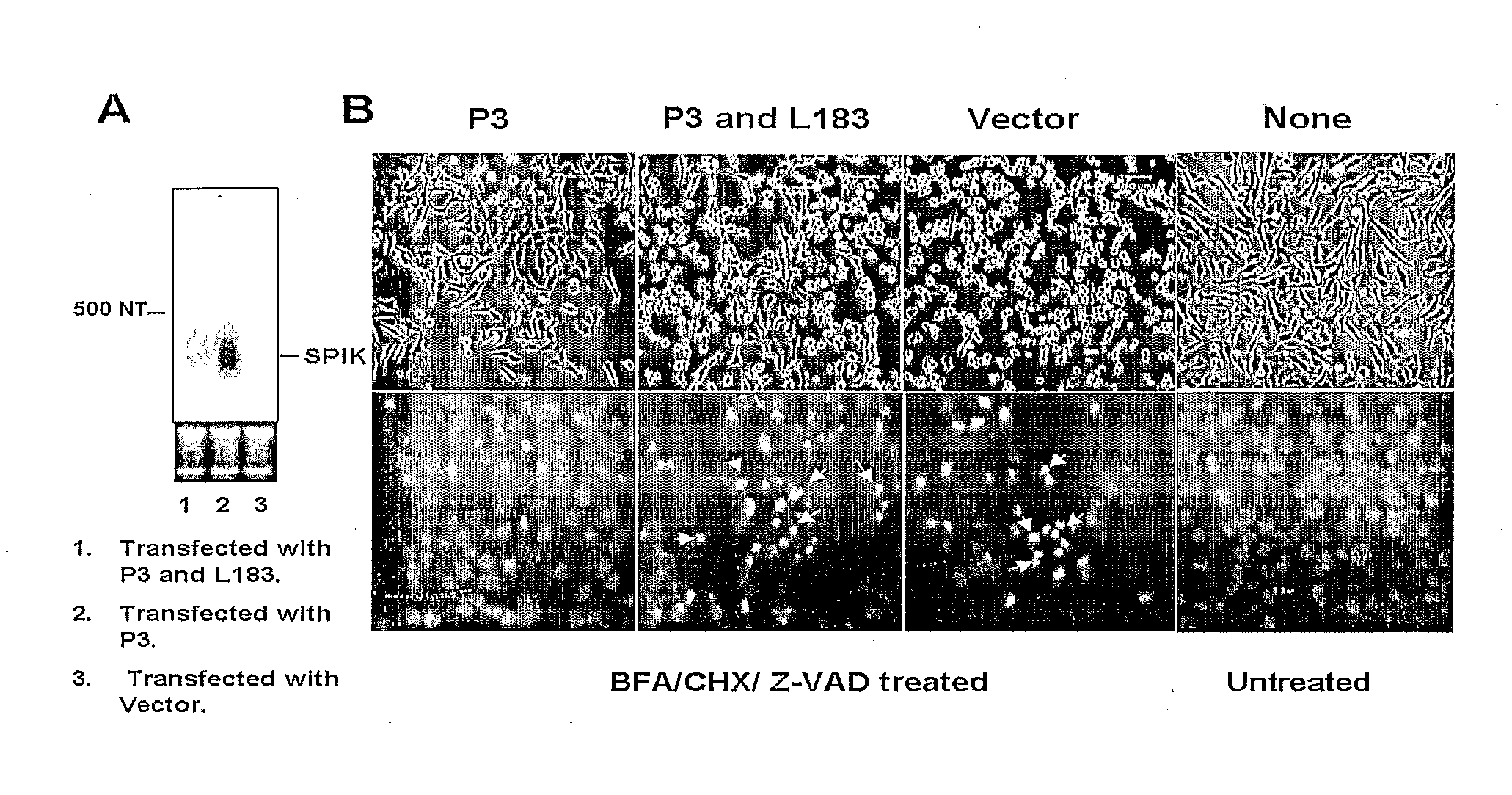
Identification of modulators of serine protease inhibitor kazal and their use as Anti-cancer and Anti-viral agents
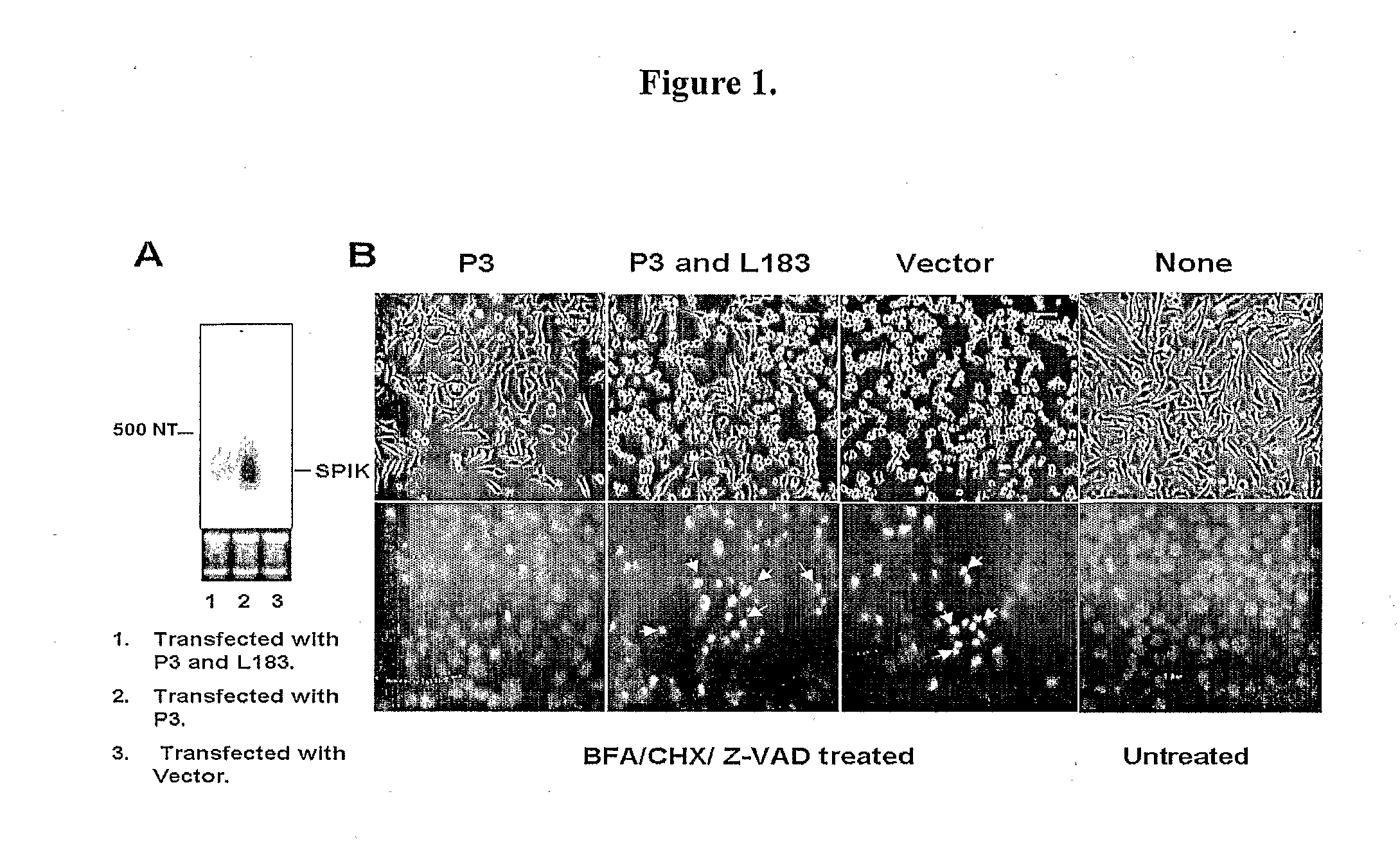
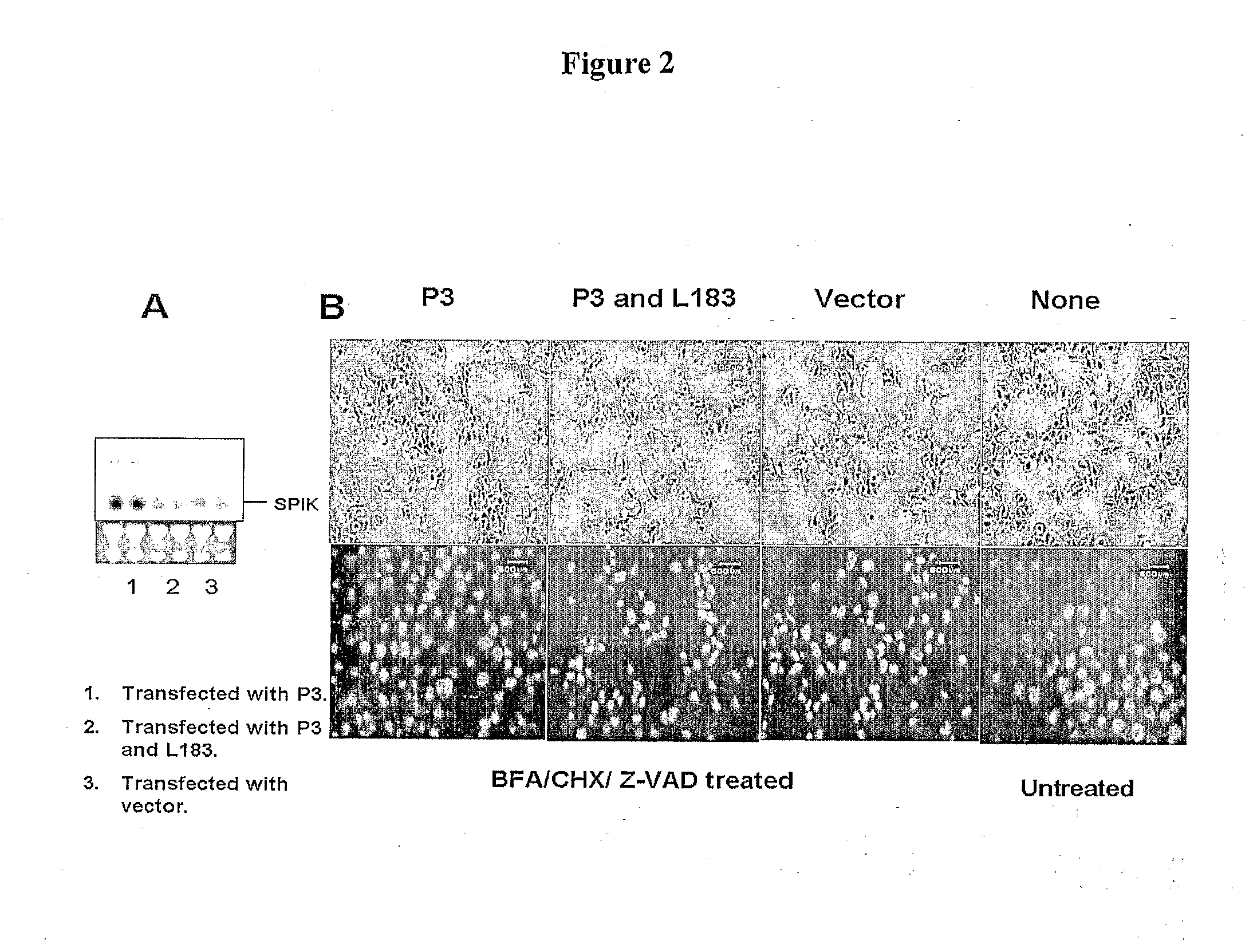
InactiveUS20090017457A1Suppressed SPIK expressionRestore sensitivityCompound screeningApoptosis detectionEtiologyAnticarcinogen
This invention describes a relevant etiology of cancer and a novel anti-cancer therapeutic strategy, based on the discovery that a protein named serine protease inhibitor (SPIK / SPINK / PSTI) was up-regulated by hepatitis B and C virus infections consequently suppressing the cell apoptosis. Accordingly, this invention provides an inhibitor of SPIK and / or a technology of suppression of over-expression of SPIK in cells. The inhibitors include: 1) chemical compounds, which can inhibit SPIK transcripts, protein activity, and gene expression, 2) SPIK siRNA (RNAi gene silence or dsRNA of SPIK, 3) DNA anti-sense and anti-SPIK antibody. Further, this invention provides a method of using the inhibitor as an anti-cancer agent to re-instate cancer cell apoptosis (e.g., serine protease dependent cell apoptosis).
Owner:PHILADELPHIA HEALTH & EDUCATION CORP
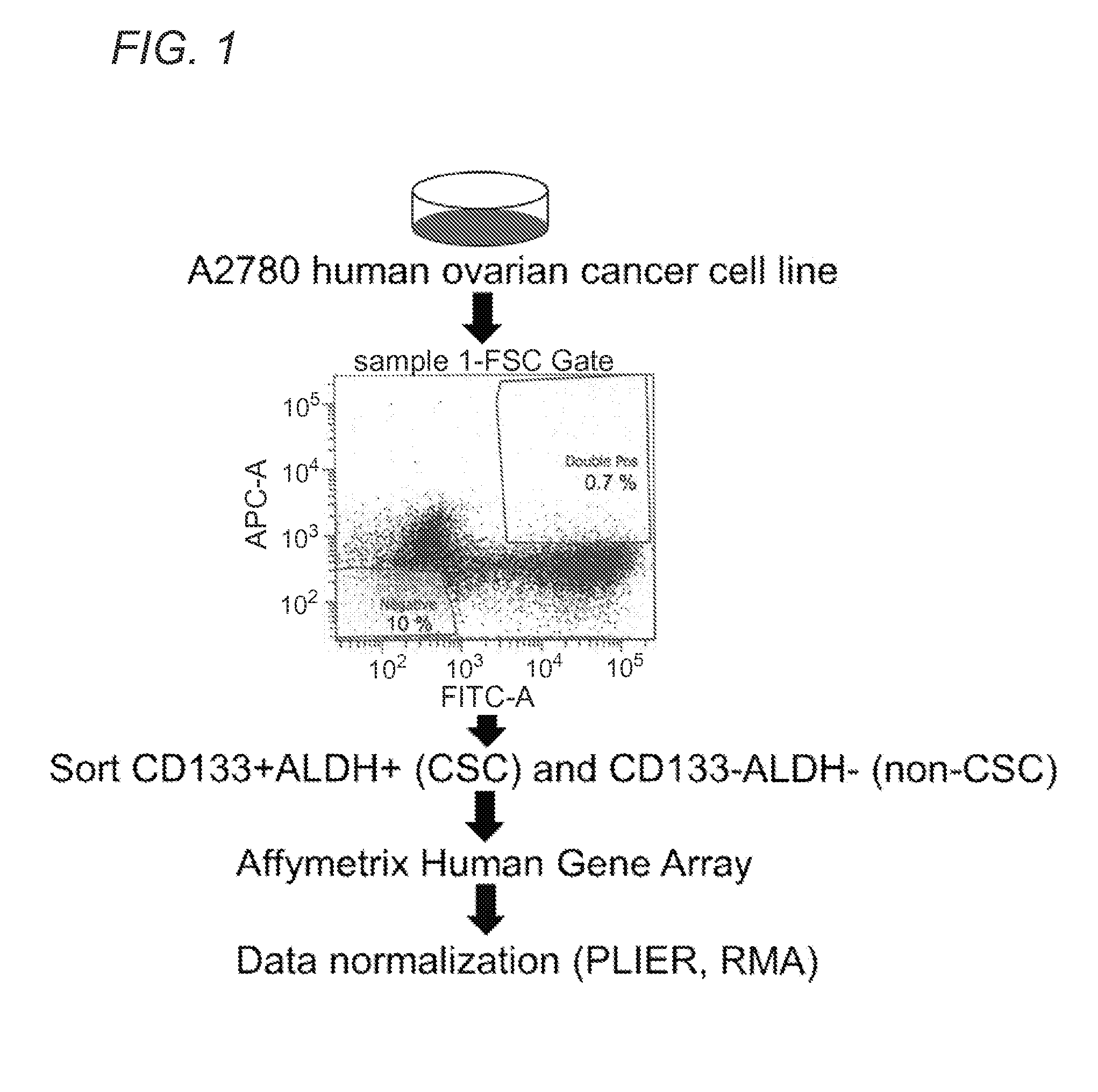
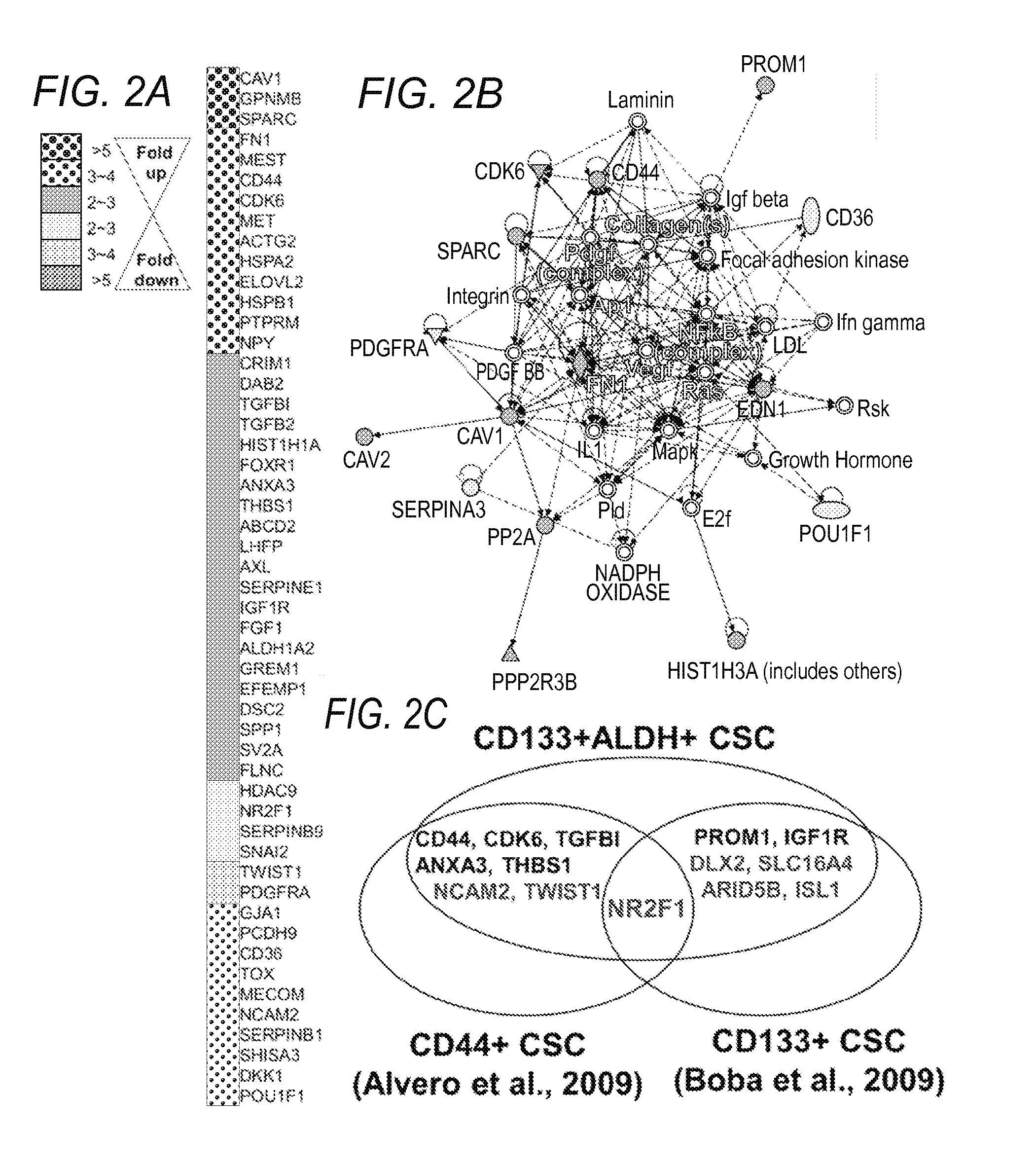
Molecular signatures of ovarian cancer
ActiveUS20150322530A1Optimal cytoreductionHeavy metal active ingredientsOrganic active ingredientsTriageOncology
Described herein are gene signatures providing prognostic, diagnostic, treatment and molecular subtype classifications of ovarian cancers through generation of ovarian cancer disease signatures (OCDSs) that account for molecular heterogeneity present in gynecological cancers. An ovarian cancer fixed signature (OCFS) is described which relates to the core programming of disease development, in addition to an ovarian cancer stem cell (OCSC) signature. Development various disease signature, suggests personalized treatment strategies focused on molecular subtypes of gynecological cancers, such as triage tests for patients.
Owner:CEDARS SINAI MEDICAL CENT
Compositions and Methods for Treating Proliferative Disorders Such as Nk-Type Ldgl
InactiveUS20070231322A1Limit pathological effectUseful in treatmentImmunoglobulins against cell receptors/antigens/surface-determinantsAntibody ingredientsDiseasePresent method
Owner:INNATE PHARMA SA +1
DNA encoding the prostate-specific membrane antigen-like gene and uses thereof
InactiveUS20050064504A1Avoid cell deathSugar derivativesPeptide/protein ingredientsProstate specific membraneDna encoding
Owner:SLOAN KETTERING INST FOR CANCER RES
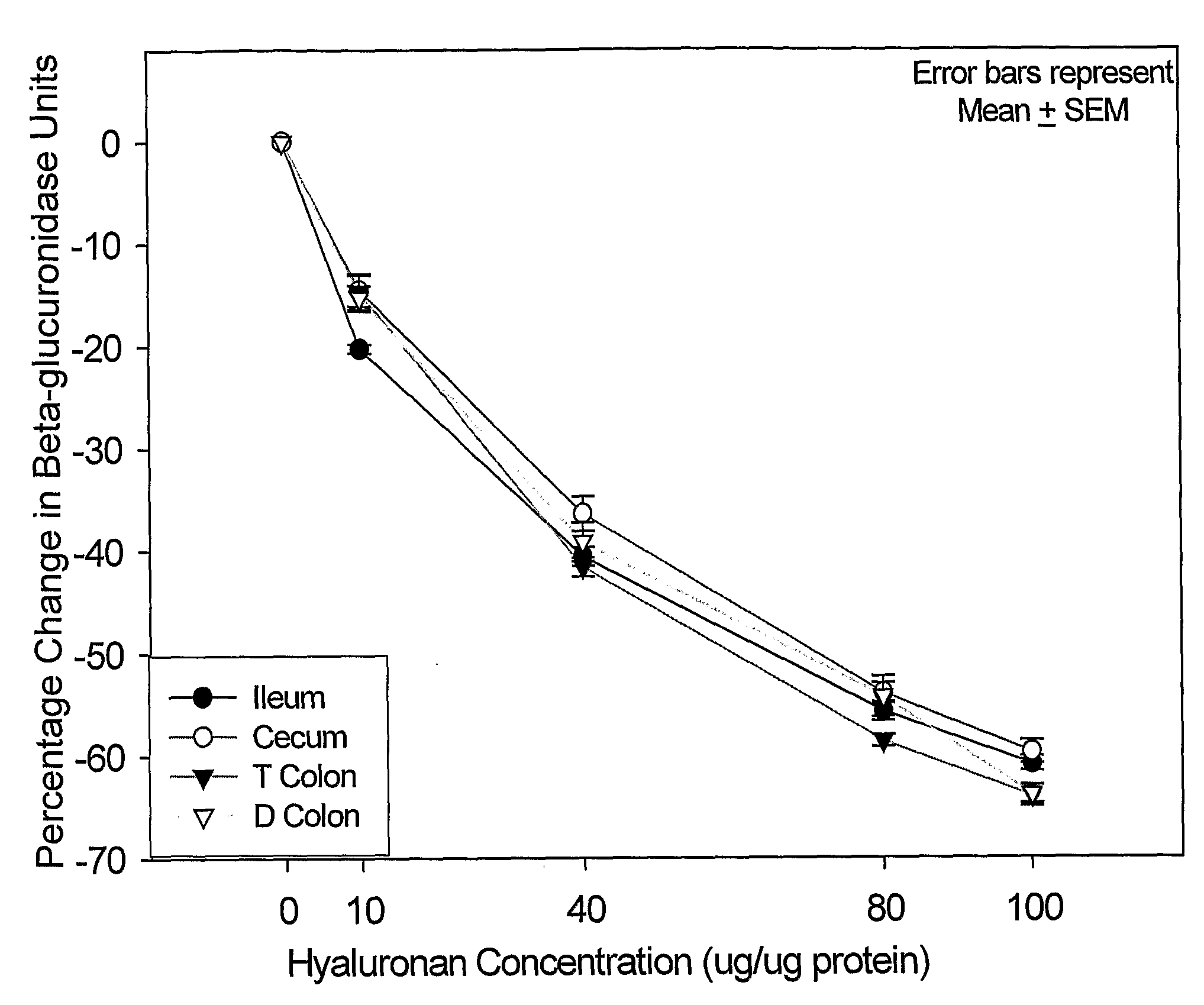
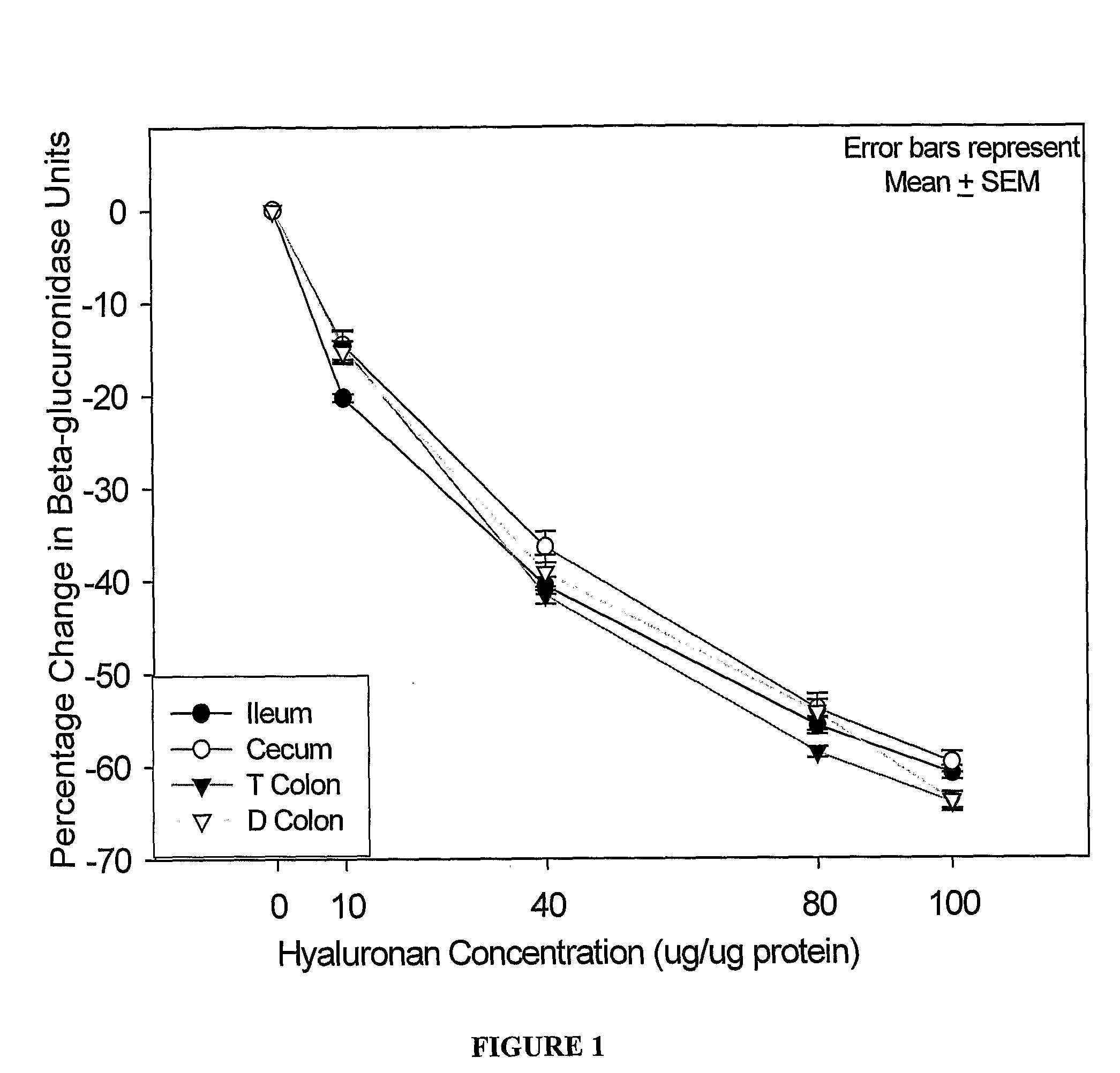
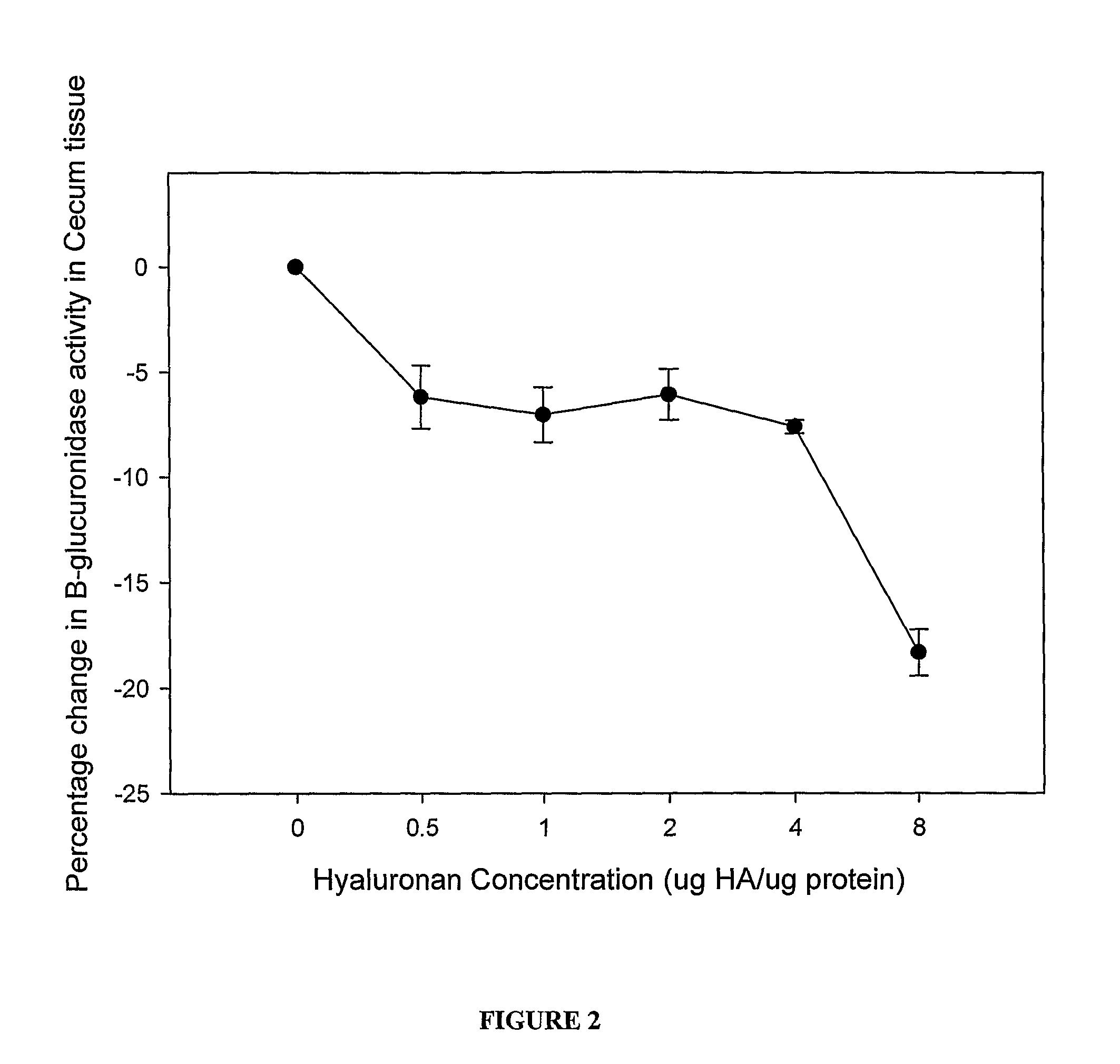
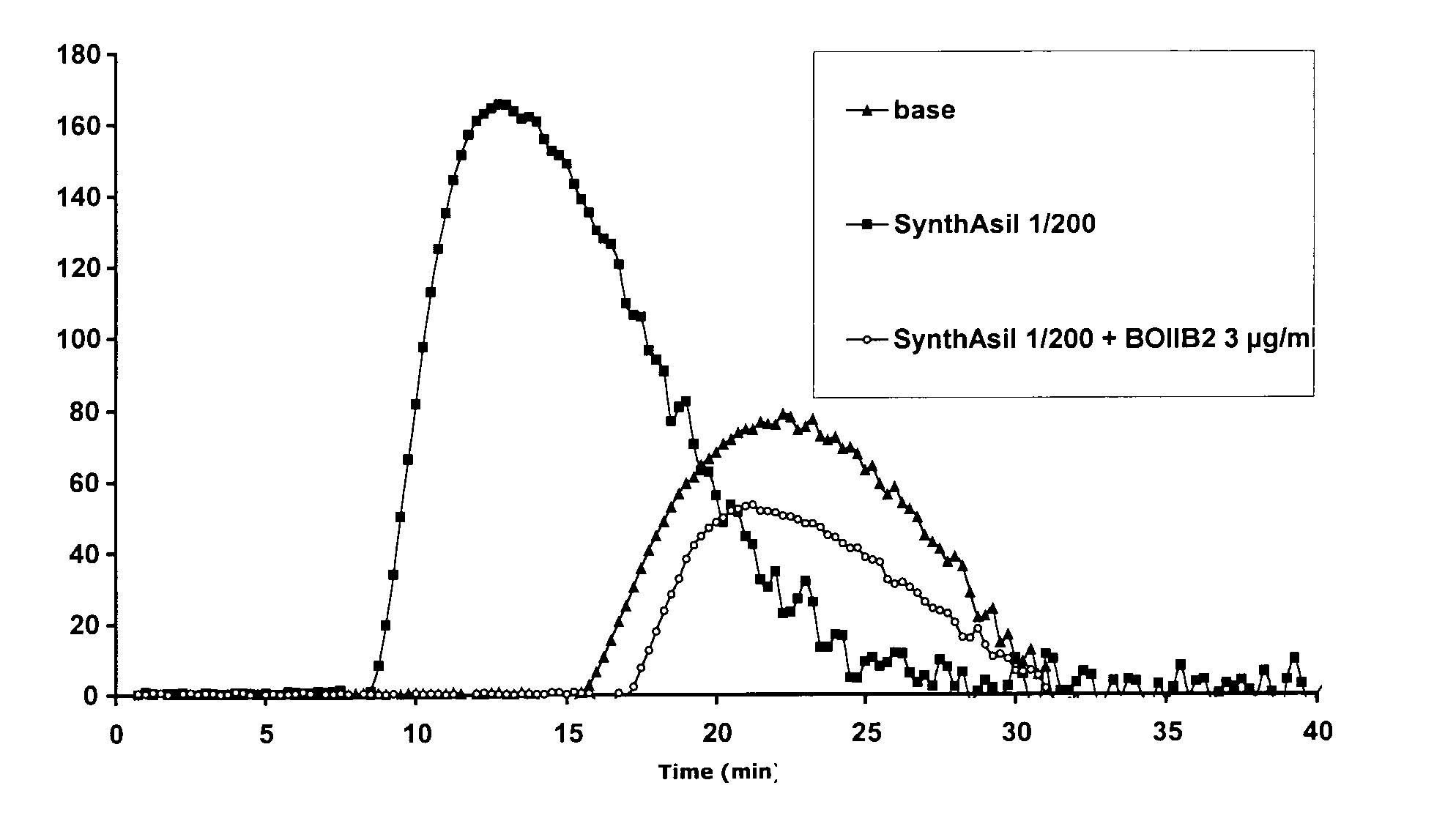
Therapeutic protocols using hyaluronan
InactiveUS20090054537A1Many symptomGood treatment effectBiocideDigestive systemMedicineHyaluronic acid
The present invention relates generally to the field of therapeutics and in particular, chemotherapy. Even more particularly, the present invention provides therapeutic strategies which reduce the toxicity or enhance the efficacy of chemotherapeutic agents. Compositions and methods of treatment and prophylaxis are also contemplated by the present invention.
Owner:ALCHEMIA ONCOLOGY PTY LTD
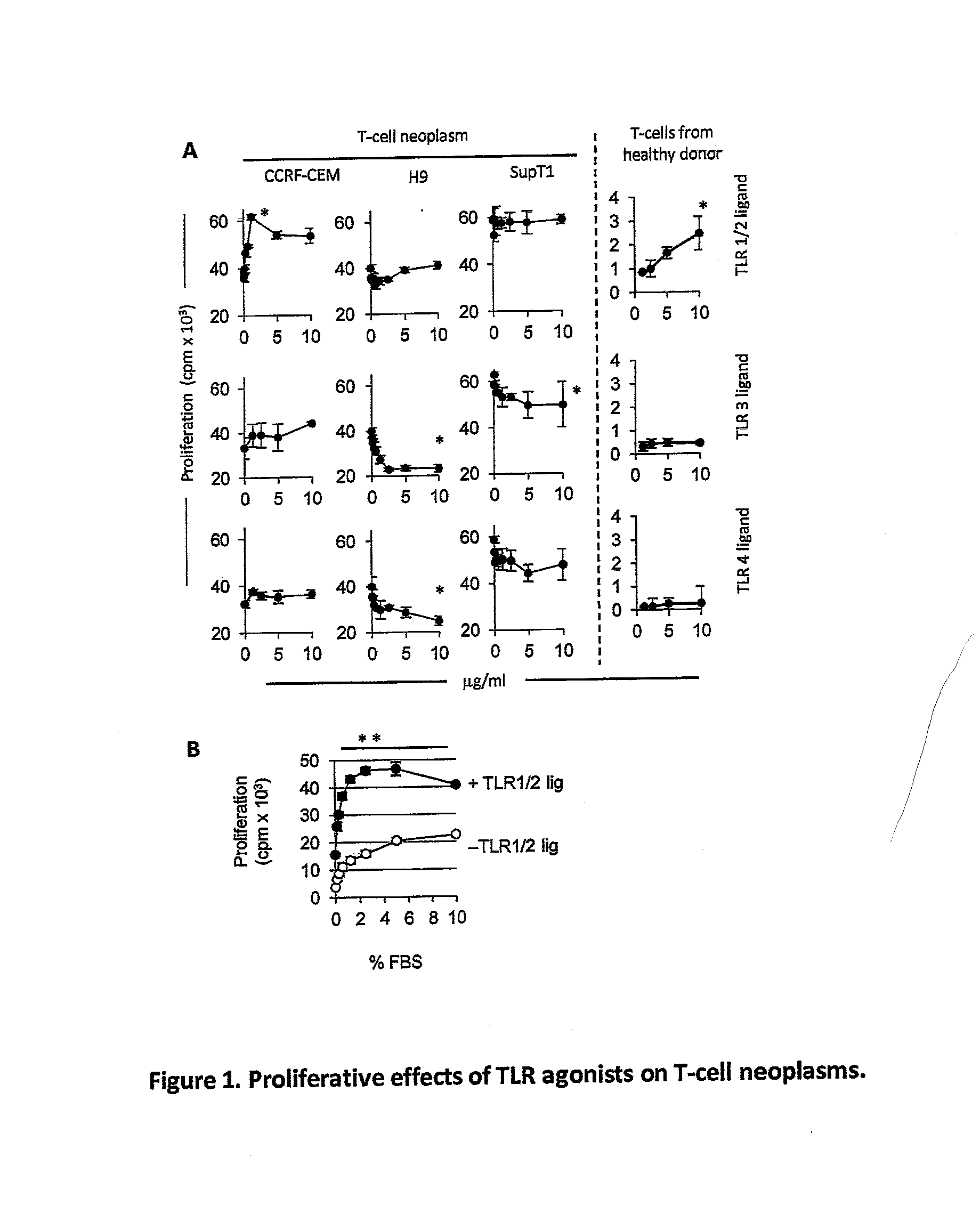
Total and Phosphorylated IL-1 Receptor-Associated Kinase-1 and IL-1 Receptor-Associated Kinase-4 as a Biomarker for Cancer Progression and Chemotherapy Resistance
ActiveUS20130280264A1Reduced T-ALLReduce the overall heightMicrobiological testing/measurementLibrary screeningMelanomaPhosphorylation
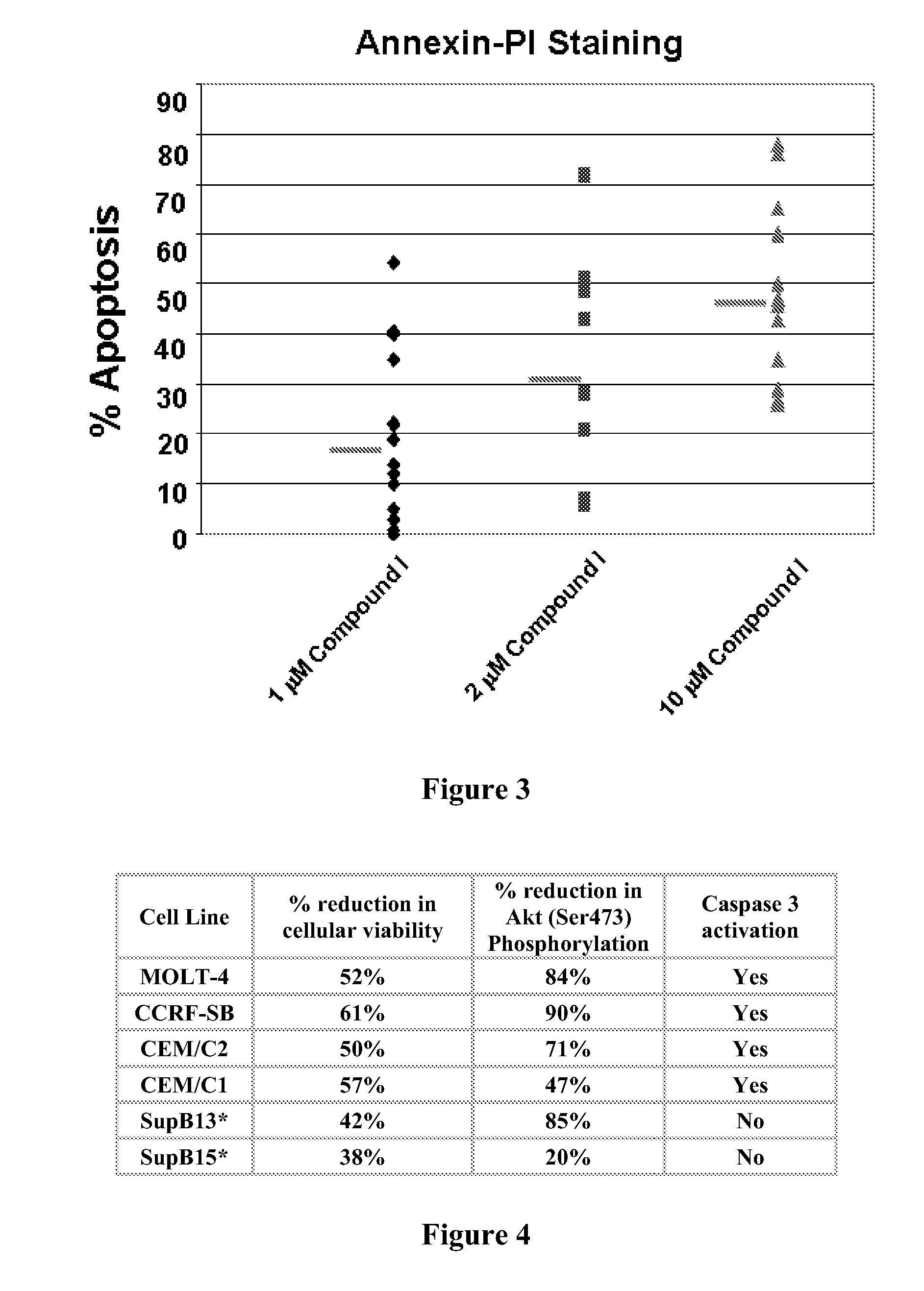
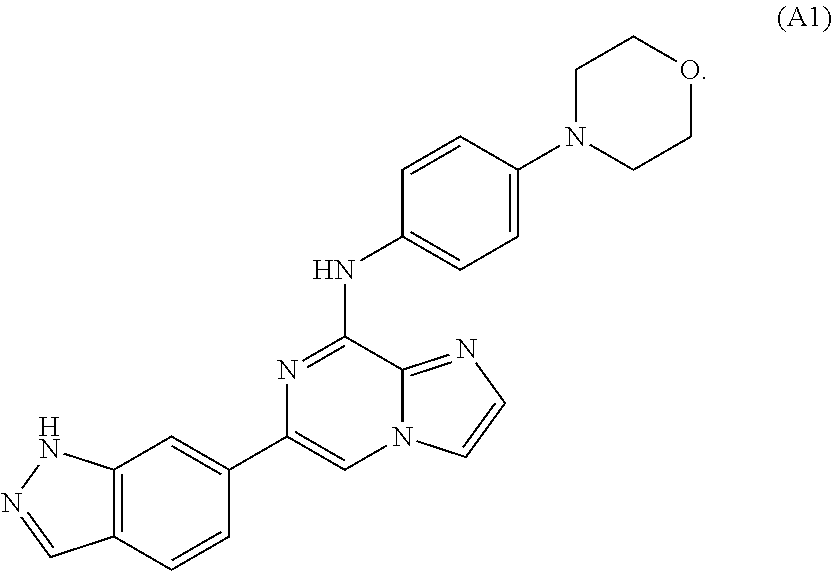
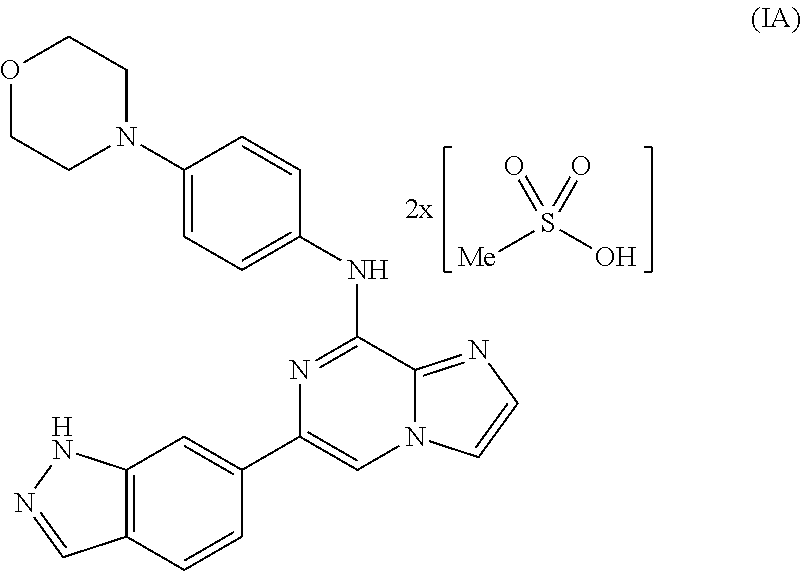
Toll-like receptors (TLR) are expressed by a variety of cancers, including melanoma and T-ALL. TLR signaling plays an important role in T cell malignancies and melanoma. The effects of stimulating or inhibiting the TLR / IL-1 receptor-associated kinases IRAK-1 and IRAK-4 in melanoma and T-ALL cells were evaluated. Pharmacological treatment with an IRAK-1,-4 inhibitor delays tumor growth and prolongs survival in vitro and in vivo, indicating that TLR signaling contributes to T-ALL and melanoma progression and interfering with this signaling is a novel therapeutic strategy to control T-ALL and melanoma proliferation.
Owner:UNIV OF MARYLAND BALTIMORE
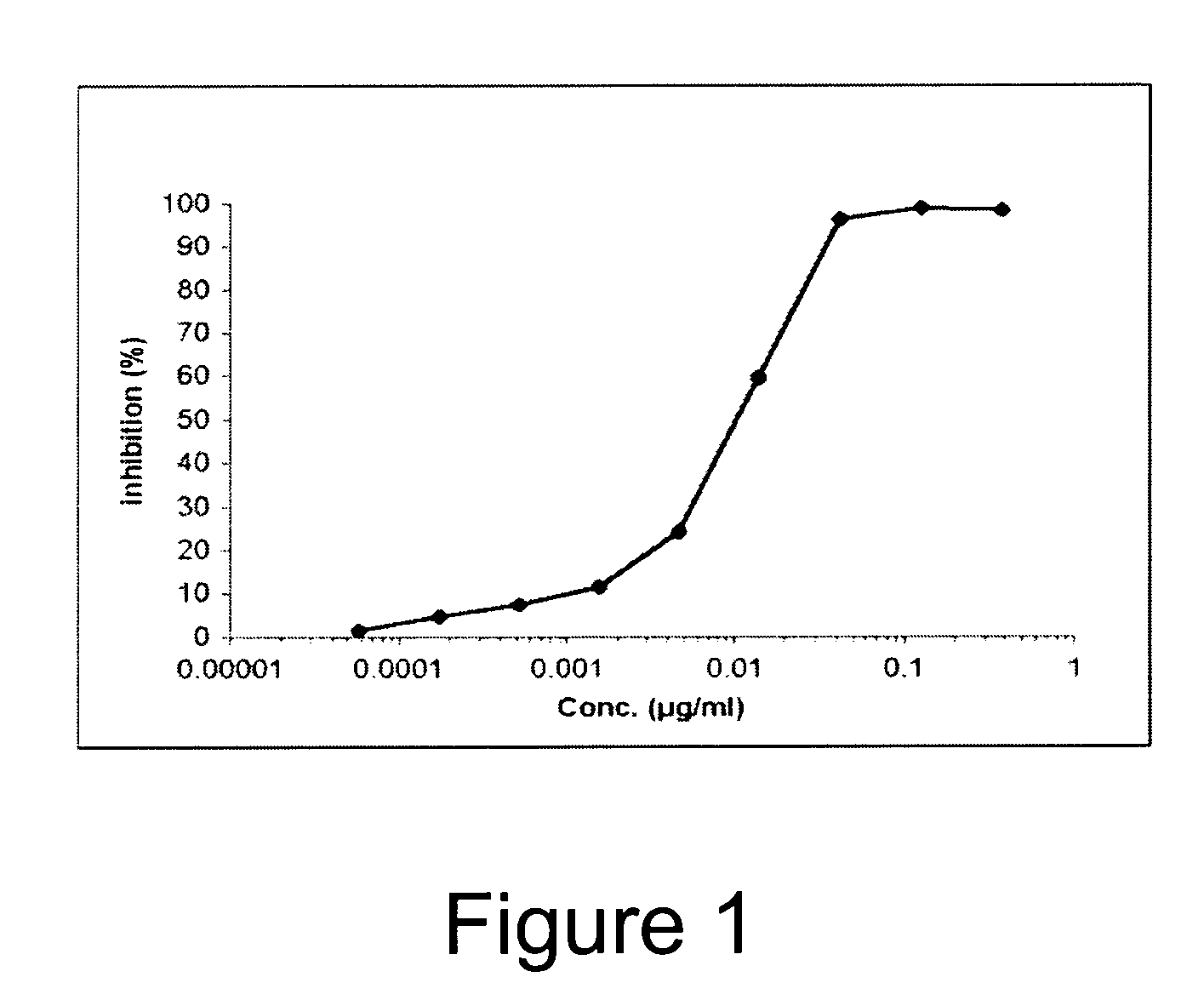
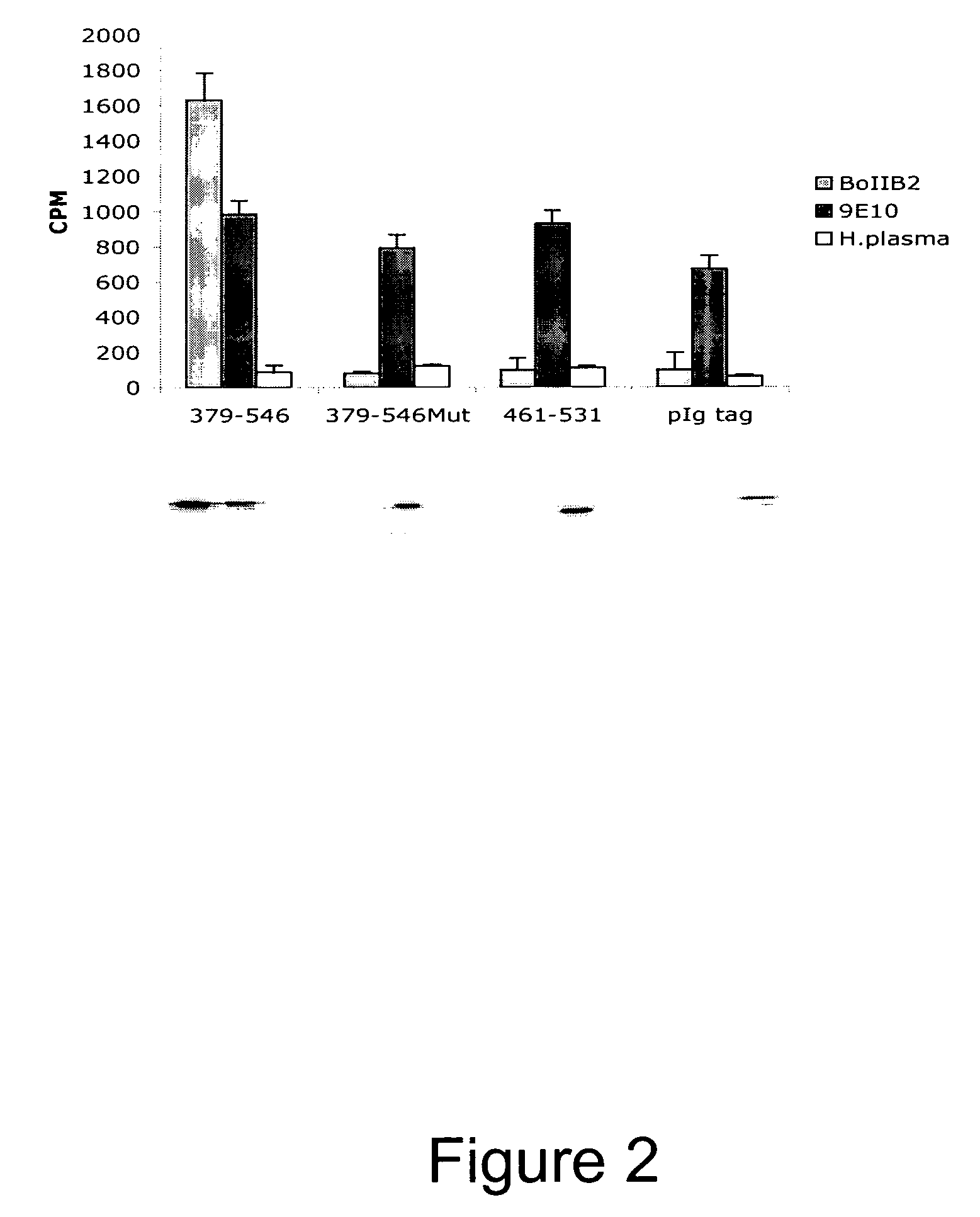
Novel Anti-Factor VIII Antibodies
InactiveUS20080206254A1Immunoglobulins against blood coagulation factorsFactor VIIStructure and functionFactor ii
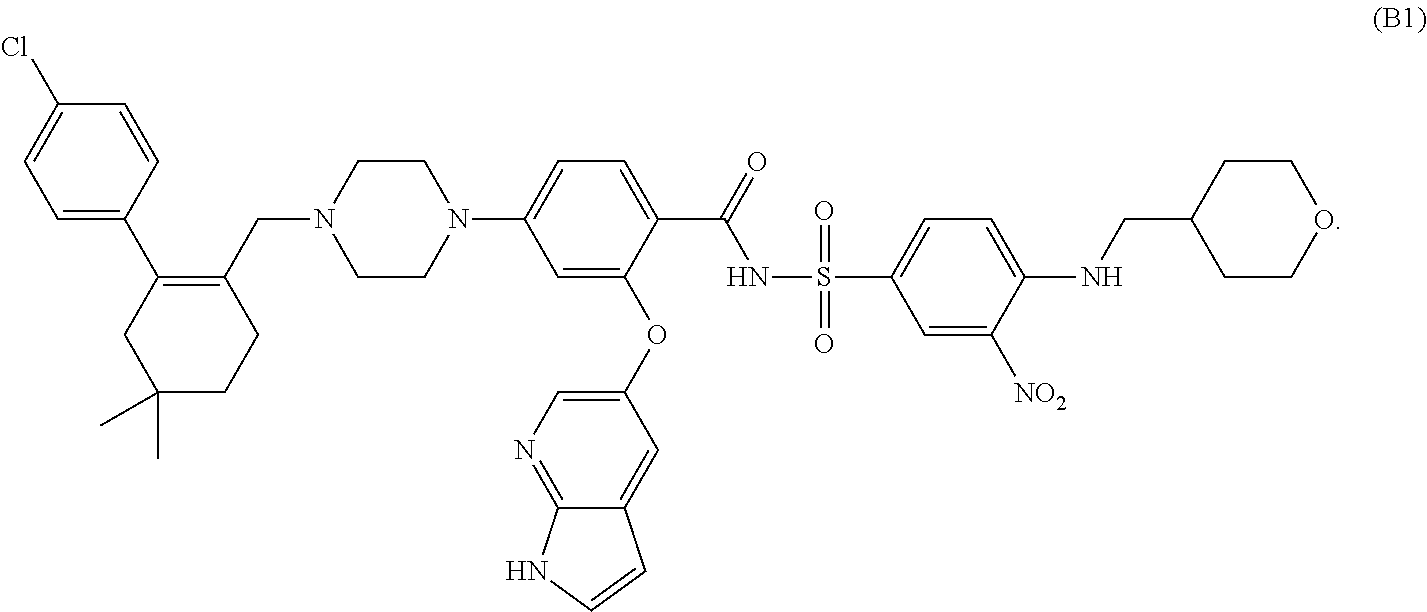
The present invention relates to new antibodies and fragments and derivatives thereof, which are particularly suited for the characterization of the structure and function of Factor VIII (FVIII) of the coagulation pathway, for the design of therapeutic strategies for eradication of FVIII inhibitors and for the use as a medicament The invention also relates to cell lines producing the specific antibodies. The present invention furthermore relates to pharmaceutical compositions comprising the antibodies, fragments and / or derivatives of the Invention and to methods of preventing and treating cardiovascular disorders by using the antibodies or fragments and derivatives thereof or pharmaceutical compositions thereof.
Owner:LIFE SCI RES PARTNERS VZW