Compositions for oral drug administration
a technology of compositions and oral drugs, applied in the direction of carbohydrate active ingredients, animal husbandry, biocide, etc., can solve the problems of not being able to demonstrate the use of alkylsaccharides as absorption enhancers in solid dosage forms such as tablets, and achieve the effects of increasing the absorption and bioavailability of drugs, avoiding various adverse drug effects, and high bioavailability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Alkyl Glycoside and / or Sucrose Ester Formulations do not Cause Mucosa Irritation or Disruption
[0196]The nasal mucosa is highly vascularized and hence optimal for high drug permeation. Moreover, absorption of drug(s) through the nasal mucosa is available to the central nervous system (CNS). Although local application of drugs is desirable, a challenge for this method of administration is mucosal irritancy.
[0197]A formulation consisting of an alkyl glycoside (0.125% TDM) in a commercial over-the-counter (OTC) nasal saline was administered in vivo to human nasal epithelium over a period of over one month. The 0.125% TDM formulation is compared to the control, namely the same commercial (OTC) nasal saline, over the same period of time. Results show that during and after 33 days of daily TDM administration (i.e., the duration of the study), there is no observable irritation of the nasal mucosa (data not shown). Thus, compositions of the invention are non-toxic and non-irritable providing...
example 2
Alkyl Glycoside and / or Sucrose Ester Compositions Stabilize Drugs by Increasing Drug Bioavailability and Reducing Drug Bioavailability Variance
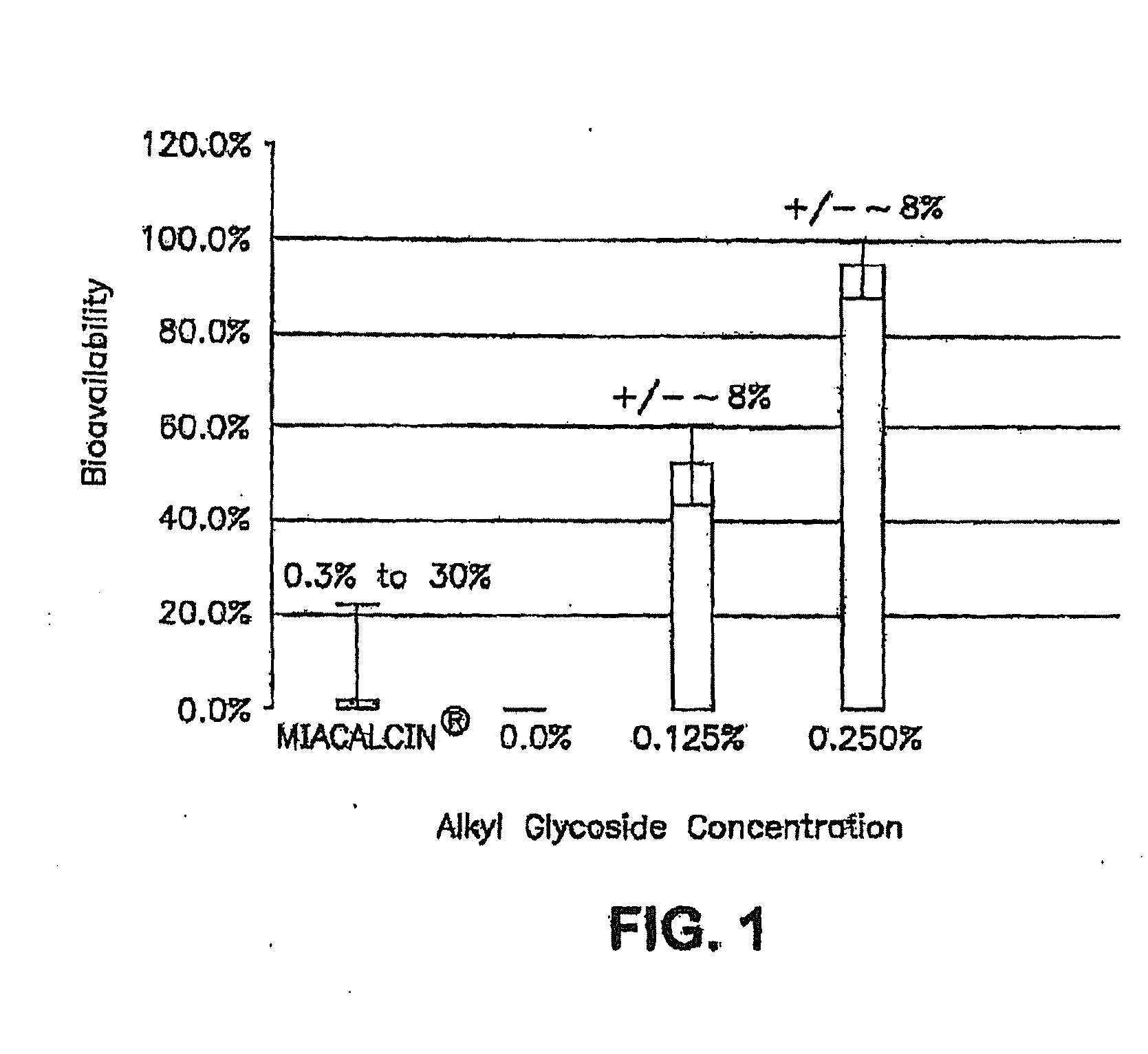
[0199]Stability of the alkyl glycoside depends, in part, on the number of carbon atoms or length of the alkyl chain and other long alkyl chains, with tetradecylmaltoside (TDM) having the greatest effect; but other highly branched alkyl chains including DDM also have stabilizing effects. In contrast to Hovgaard-1, which described the preference for a high alkyl glycoside to drug ratio, the instant invention shows that this ratio is much lower. For example, alkyl glycosides in the range of about 0.01% to about 6% by weight result in good stabilization of the drug; whereas Hovgaard-1 shows stabilization is only achieved at much higher ratios of alkyl glycosides to drug (10:1 and 16:1). Even more interesting, alkyl glycosides of the invention in the range of about 0.01% to about 6% have increased bioavailability (see FIG. 1). This is in sharp con...
example 3
Ocular Administration of Alkyl Saccharides Plus Insulin Produces Hypoglycemic Effects In Vivo
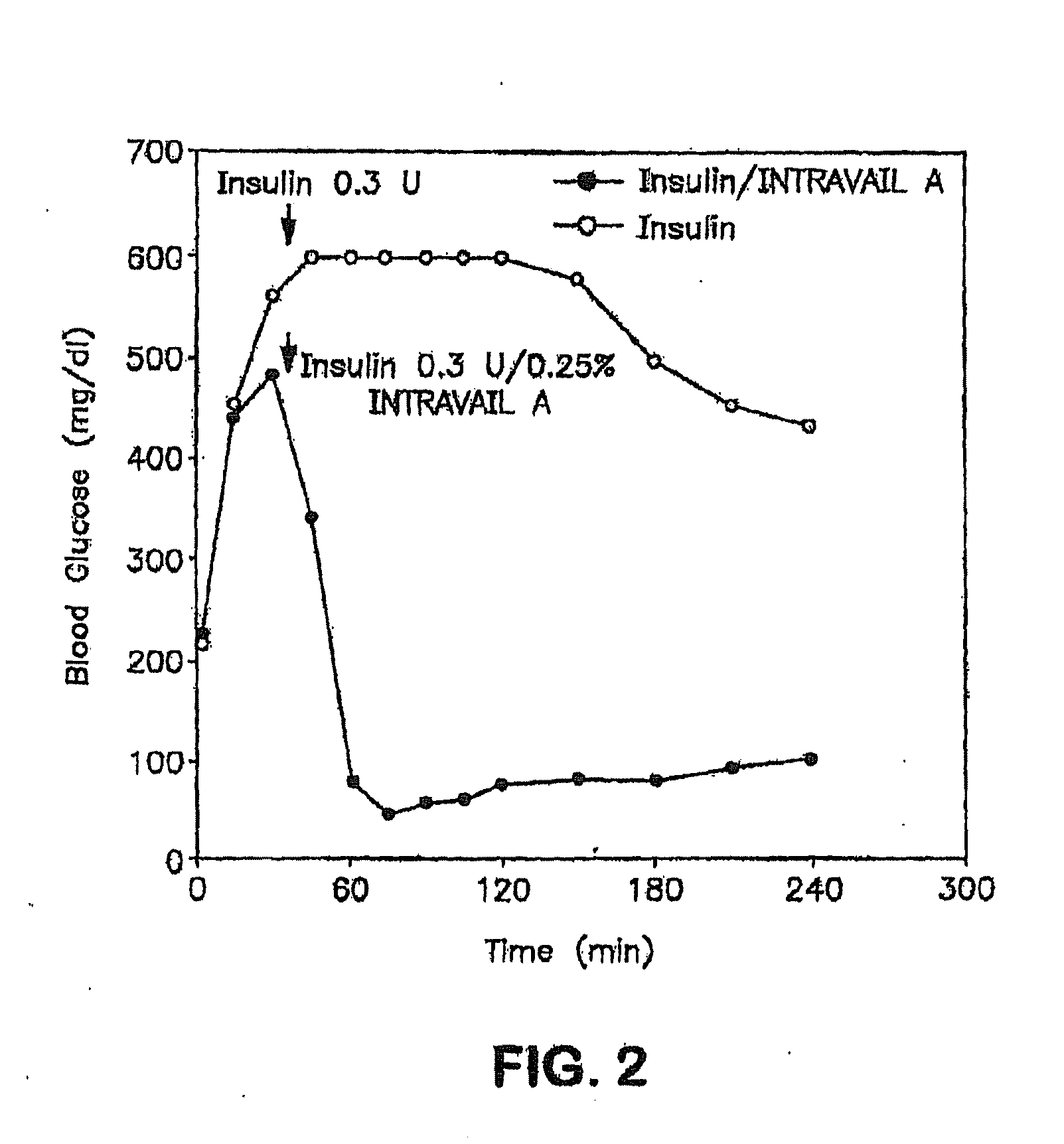
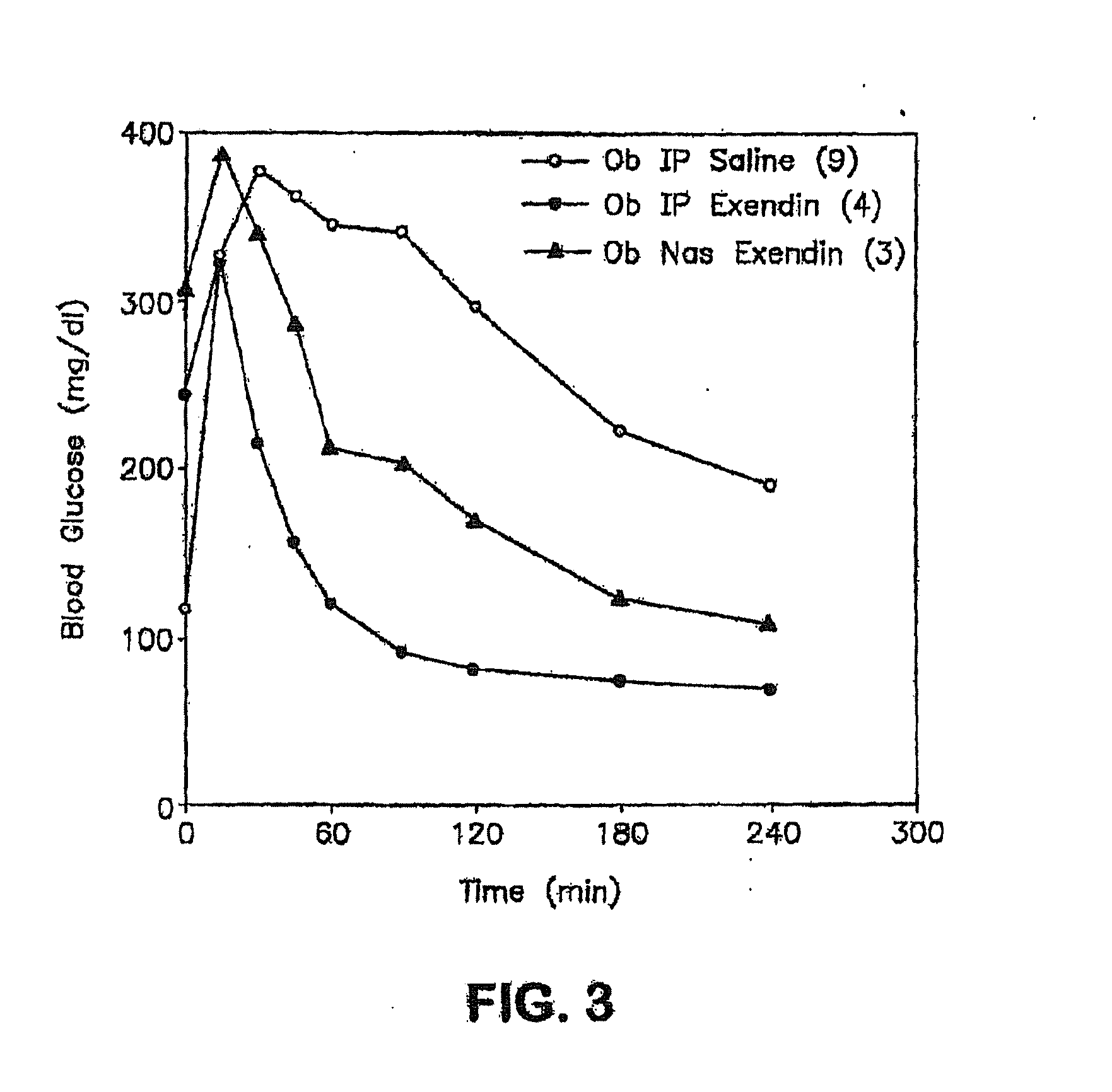
[0203]Normal rats were anesthetized with a mixture of xylazine / ketamine to elevate their blood glucose levels. The elevated levels of D-glucose that occur in response to anesthesia provide an optimal system to measure the systemic hypoglycemic action of drug administration, e.g. insulin-containing eye drops. This animal model mimics the hyperglycemic state seen in diabetic animals and humans. In the experimental animal group, anesthetized rats are given eye drops containing insulin. Blood glucose levels from the experimental group are compared to anesthetized animals which received eye drops without insulin. The change in blood glucose levels and the differential systemic responses reflects the effect of insulin absorbed via the route of administration, e.g. ocular route.
[0204]Adult male Sprague-Dawley rats (250-350 g) were fed ad libitum, and experiments were conducted between 10:00 a.m. an...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More