Stable aqueous compositions of prostglandin agonist prodrugs and methods for use thereof
A prostaglandin and composition technology, which is applied in the directions of drug combination, drug delivery, pharmaceutical formulation, etc., can solve the problems of inactive esters and inactive therapeutically active compounds, etc.
Inactive Publication Date: 2012-10-31
ALLERGAN INC
View PDF7 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Usually, but not necessarily, the ester is inactive or less active than the therapeutically active compound to which it is converted
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment
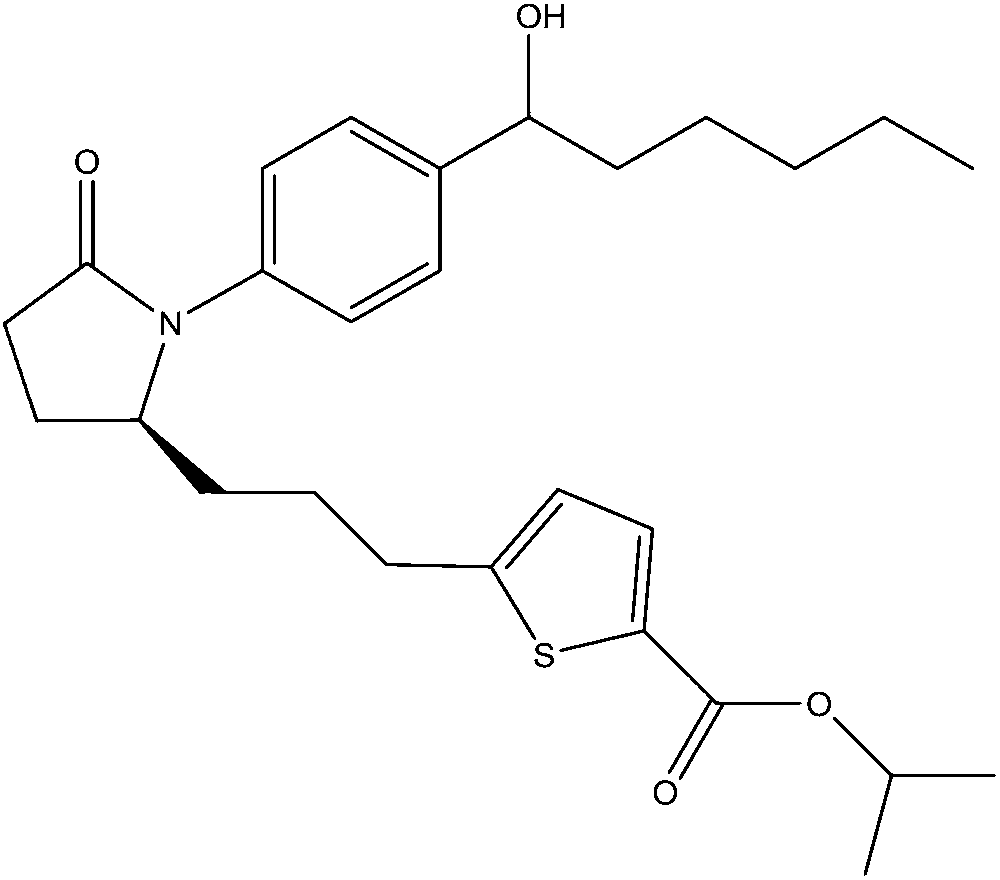
[0070] Compounds 2 and 3 were used to evaluate the aqueous stability of the compositions of the invention. Four formulations were prepared for each compound as shown in the table below.
[0071] Table 1
[0072]
[0073] Table 2
[0074]
[0075] The formulations were analyzed by HPLC using the following measurement parameters.
[0076] Column: BioWidePore C18 (SUPELCO), 4.6mmx25cm, 5μm
[0077] Mobile phase A: 0.1% (V / V) trifluoroacetic acid (TFA) in deionized water, 0.8 micron filtered
[0078] Mobile phase B: 100% acetonitrile, 0.8 micron filtered
[0079] Column temperature: ambient temperature
[0080] Injection volume: 30μL
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The present invention is based on the discovery that a marked increase in aqueous stability (and thereby shelf life) of prostanoid agonist prodrug compositions is achieved by incorporating into the compositions certain well-defined carboxylic acids, and thereafter adjusting the pH of the compositions from about 4.0 to about 8.0. As a result, the compositions and methods of the invention provide the aqueous stability required for marketable topical drug treatments of a wide variety of ocular disorders.
Description
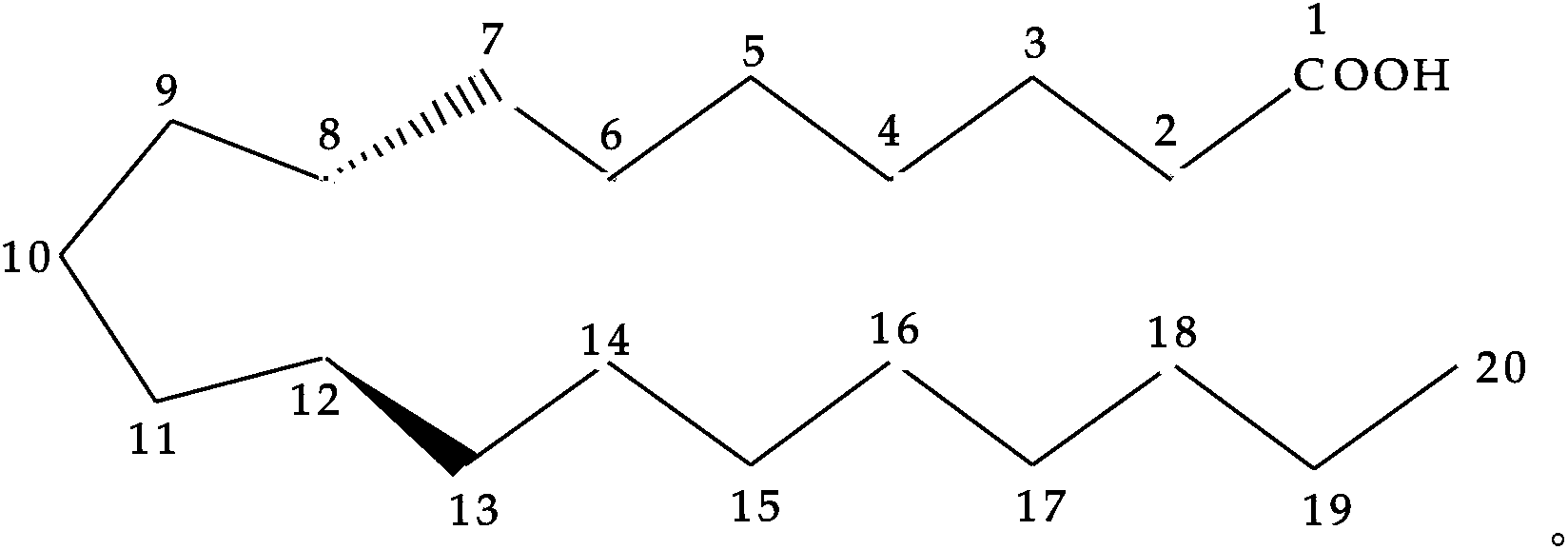
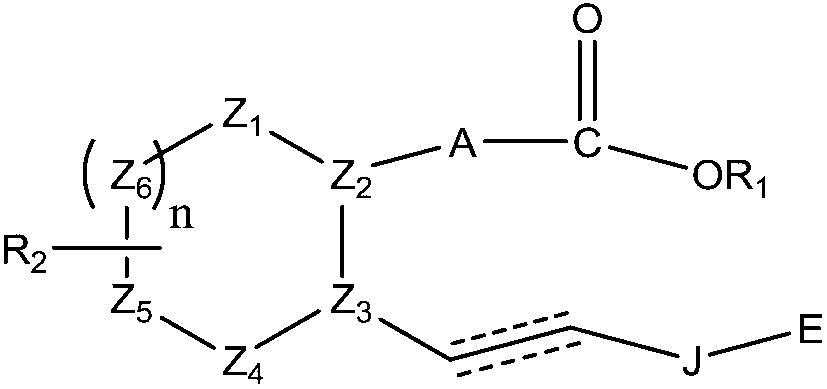
[0001] related application [0002] This application claims the benefit of US Provisional Application Serial No. 61 / 267,897, filed December 9, 2009, the disclosure of which is incorporated herein by reference in its entirety. technical field [0003] The present invention relates generally to prodrug compositions of prostanoid antagonists, and more particularly, to stable aqueous compositions of prodrugs of prostanoid antagonists and methods of use thereof. Background technique [0004] Ocular hypotensive agents are used in the treatment of a large variety of ocular hypertensive conditions, such as post-surgery and post-laser trabeculectomy ocular hypertensive episodes, glaucoma, and as a preoperative adjunct. [0005] Glaucoma is an eye disease characterized by elevated intraocular pressure. Based on its etiology, glaucoma can be classified as primary or secondary glaucoma. For example, primary glaucoma (congenital glaucoma) in adults can be open angle or acute, or chroni...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A61K9/08A61K31/4025A61K31/4436A61K47/12A61P27/06
CPCA61K31/4436A61K47/12A61K31/4025A61K9/0048A61P27/00A61P27/02A61P27/06A61P27/10A61K31/559A61K47/02A61K47/10A61K47/26
Inventor R·M·伯克W·B·艾姆
Owner ALLERGAN INC