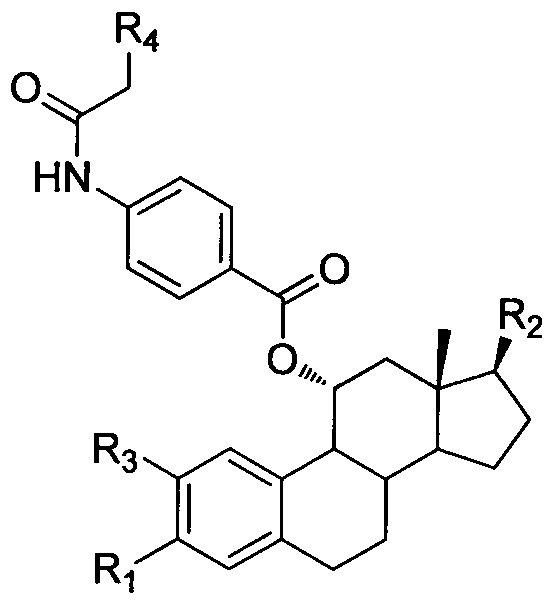
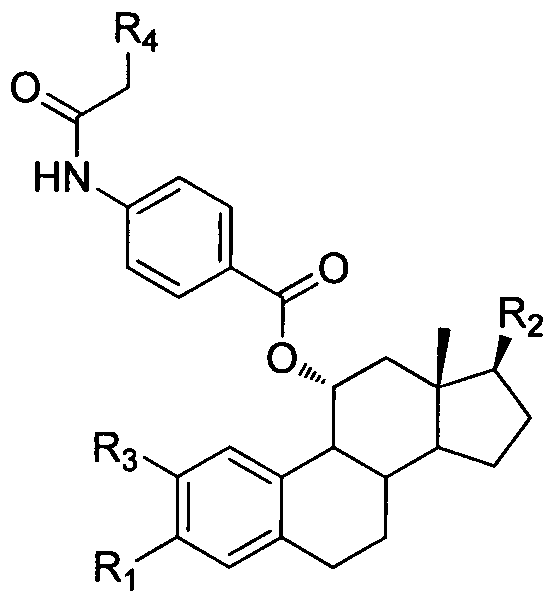
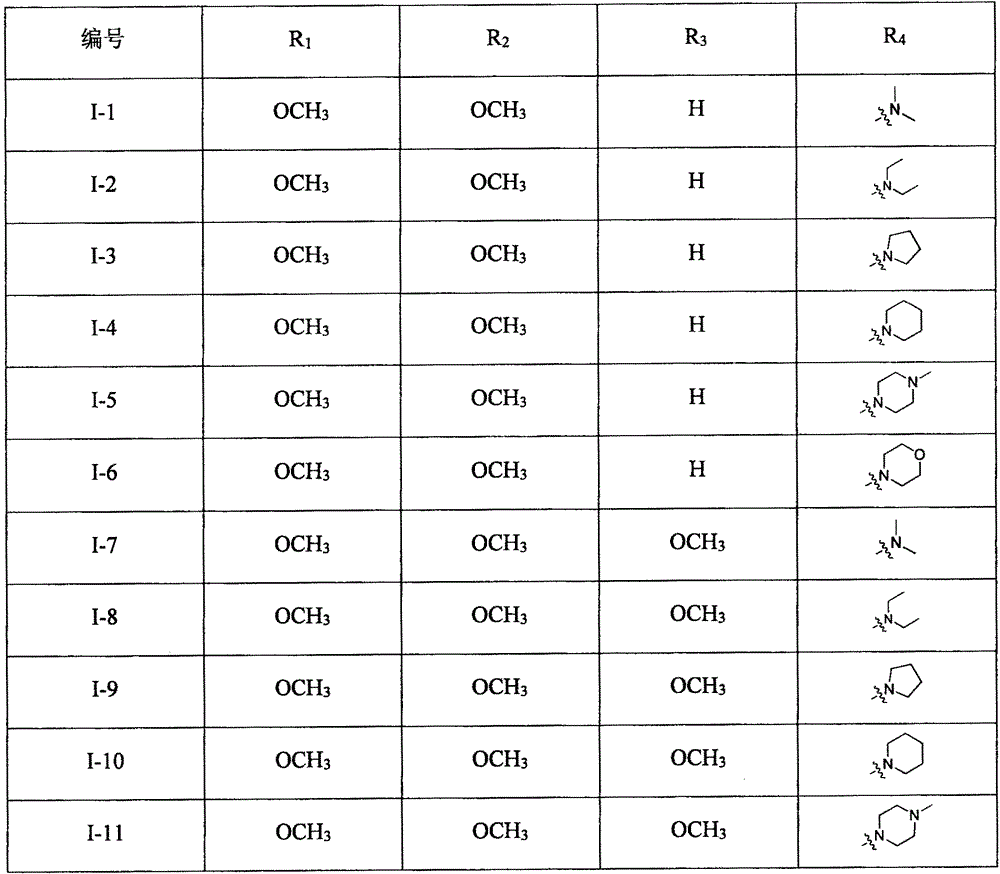
Novel steroid selective estrogen receptor modulator as well as preparation method and pharmaceutical application thereof
A pharmacy and carrier technology, applied in pharmaceutical formulations, sexually transmitted diseases, anti-tumor drugs, etc., can solve problems such as clinical treatment effects that are not as good as Tamoxifen, venous thrombosis, vasodilation, endometrial cancer and drug resistance.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
specific Embodiment approach
[0032] The specific embodiment (described embodiment is only used to illustrate the present invention, is not used to limit the present invention)
[0033] The preparation of some compounds is carried out as follows:
[0034] 1 H-NMR nuclear magnetic resonance was measured by BrukerAV300 (300MHz) nuclear magnetic resonance instrument (TMS was the internal standard), and mass spectrometry was determined by Shimadzu GC / MS-QP2010 mass spectrometer (EI-MS), Agilent1100LC-MSD-Trap / SL Mass spectrometer (ESI-MS) determination.
[0035]The silica gel used for column chromatography is 100-200 mesh, 200-300 mesh or 300-400 mesh silica gel (Qingdao Ocean Chemical Factory), and the eluent is petroleum ether-ethyl acetate system or chloroform-methanol system. Thin-layer chromatography (TLC) uses a GF254 thin-layer chromatography plate (Yantai Jiangyou Silica Gel Development Co., Ltd.); the TLC development system is a petroleum ether-ethyl acetate system or a chloroform-methanol system, a...
Embodiment 1
[0037] Synthesis of 1,3,5(10),9(11)-Estratratene-3,17β-diol (IIIa)
[0038] Dissolve 20.0g (73.53mmol) of estradiol (IIa) in 300ml of dry methanol, add 25g (110.13mmol) of 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ), and heat to reflux under nitrogen protection 3-5 hours, the end point is determined by TLC, the reaction solution is cooled, concentrated under reduced pressure to remove the solvent, the concentrate is dissolved in 300ml of ethyl acetate, and 300ml of saturated NaHCO is added 3 solution, stirred until no bubbles are generated, separated, saturated NaHCO 3 The solution was washed twice (300ml×2), the organic layer was dried over anhydrous sodium sulfate for 3 hours, concentrated under reduced pressure to remove the solvent, and 18.4g of a brownish yellow solid was obtained. Column chromatography (PE:EA=5:1) yielded 7.8 g of light yellow solid, yield 39.29%. MS (ESI, m / z): 269 [M-H] + .
Embodiment 2
[0040] Synthesis of 1,3,5(10),9(11)-estradiene-2,3,17β-triol-2-methyl ether (IIIb)
[0041] Dissolve 10g (33.1mmol) 2-methoxyestradiol (IIb) with 150ml of dry methanol, add 5.65g (49.7mmol) 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ), Under the protection of nitrogen, heat and reflux for 3-5 hours, and determine the end point by TLC. After the reaction liquid is cooled, it is concentrated under reduced pressure to remove the solvent. The concentrate is dissolved in 150ml of ethyl acetate, and 150ml of saturated NaHCO is added. 3 solution, stirred until no bubbles are generated, separated, saturated NaHCO 3 The solution was washed twice (150ml×2), the organic layer was dried over anhydrous sodium sulfate for 3 hours, concentrated under reduced pressure to remove the solvent, and 18.3g of a brownish yellow solid was obtained. Column chromatography (PE:EA=4:1) gave 4.3 g of light yellow solid, yield 43.29%. MS (ESI, m / z): 299 [M-H] + .
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap