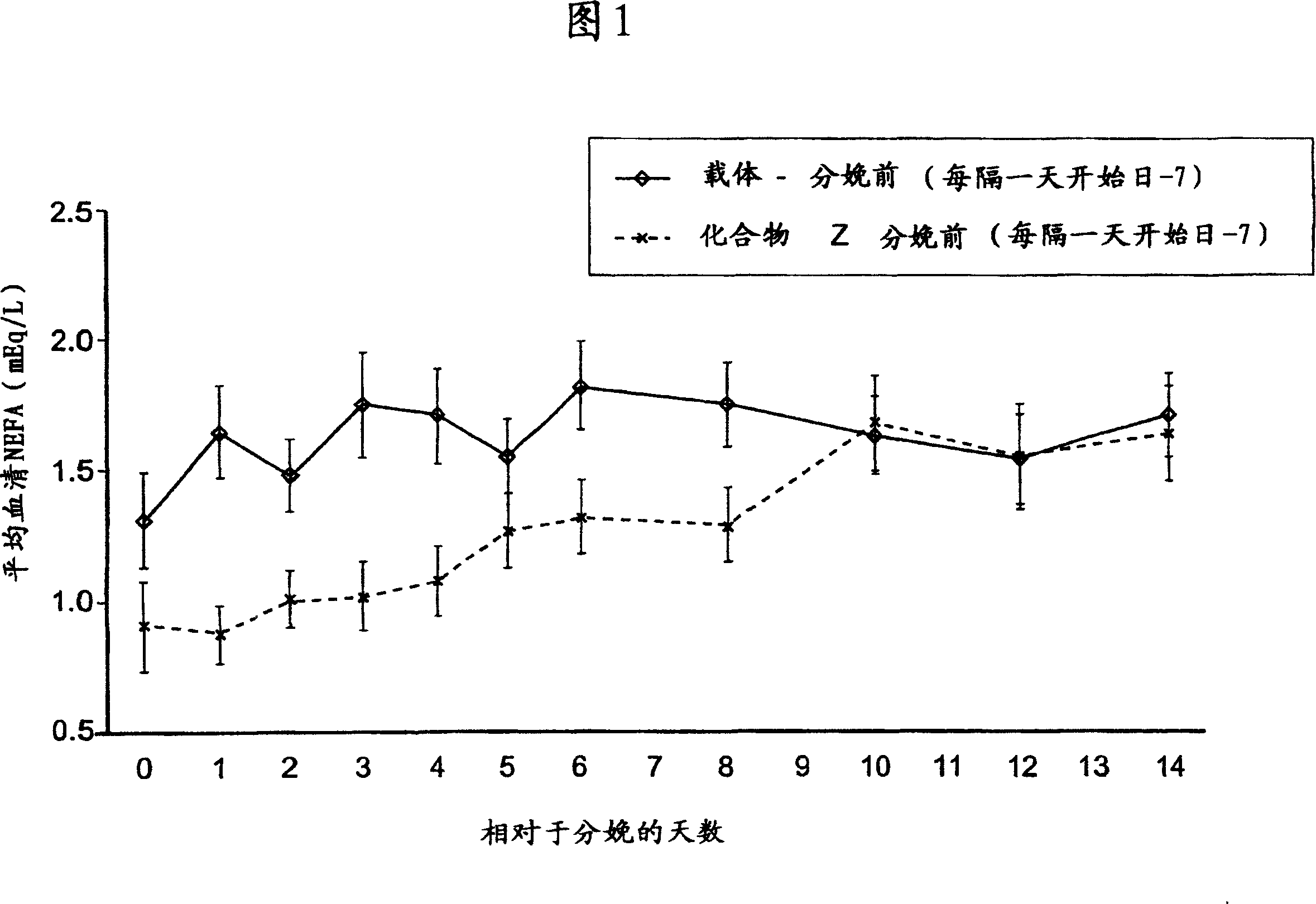
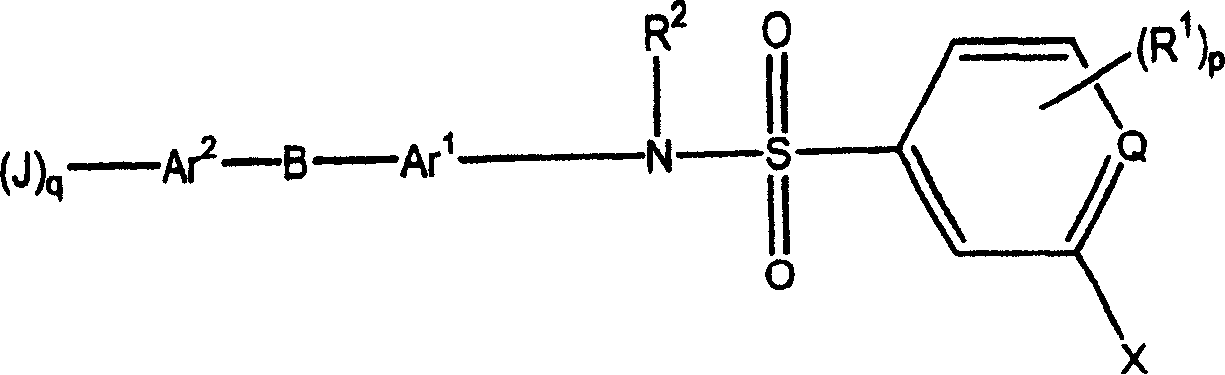
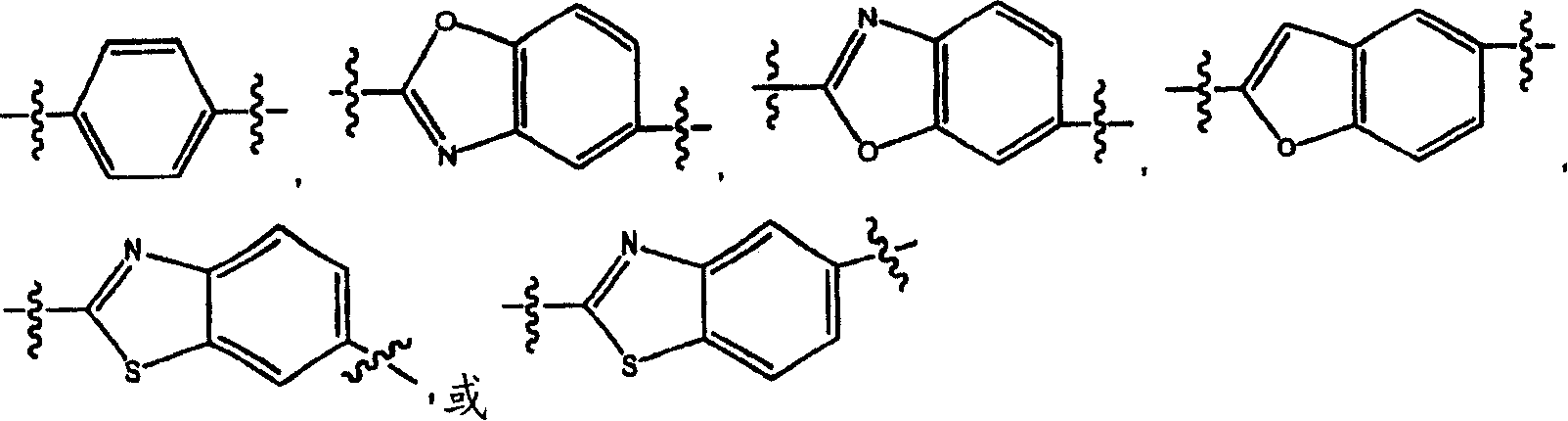
Substituted heteroaryl- and phenylsulfamoyl compounds
A technology of phenylsulfamoyl and compound, applied in the field of peroxisome proliferator-activated receptor agonists, can solve problems such as low ability and increased esterification
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0516] Example 1: 5-(4-Benzyloxy-phenylsulfamoyl)-2-methyl-benzoic acid
[0517]
[0518] Sodium bicarbonate (215 mg, 0.56mmol) in 2ml of aqueous solution. The resulting mixture was stirred overnight at room temperature. Acetone was then removed under reduced pressure and the residual mixture was partitioned between 25 ml of 1N aqueous hydrochloric acid and 25 ml of ethyl acetate. The aqueous phase was separated and extracted with 2 x 25 ml ethyl acetate. The ethyl acetate extracts were combined, dried (anhydrous sodium sulfate), and concentrated under reduced pressure. The residue was purified by flash column chromatography (silica gel, 15 gm) eluting with 9:1 chloroform / methanol to give a white solid (154 mg). The solid was triturated with dichloromethane to afford the title compound (93 mg, 28% yield) as a white solid.
[0519] MS: 395.6(M-1); 1 H NMR (400MHz, CD 3 OD): δ2.57(s, 3H), 4.96(s, 2H),...
Embodiment 2
[0521] Example 2: 2-methyl-5-[4-(6-methyl-benzothiazol-2-yl)-phenylsulfamoyl]benzoic acid
[0522]
[0523] 8% yield. MS: 439.4 (M+1); 1 H NMR (400MHz, CD 3 OD): δ2.44(s, 3H), 2.53(s, 3H), 7.23(m, 2H), 7.29(m, 1H), 7.34(m, 1H), 7.72(m, 1H), 7.75(m , 1H), 7.80(m, 1H), 7.88(m, 2H), 8.19(m, 1H).
Embodiment 3
[0524] Example 3: 2-methyl-5-[4-(5-methyl-benzoxazol-2-yl)-phenylsulfamoyl]benzoic acid
[0525]
[0526] 4% yield. MS: 423.4 (M+1): 1 H NMR (400MHz, CD 3 OD): δ2.42(s, 3H), 2.51(s, 3H), 7.17(m, 1H), 7.26(m, 2H), 7.35(m, 1H), 7.44(m, 2H), 7.56(m , 1H), 8.0(m, 2H), 8.15(m, 1H).
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap