Patents
Literature
14491 results about "Vitamin C" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
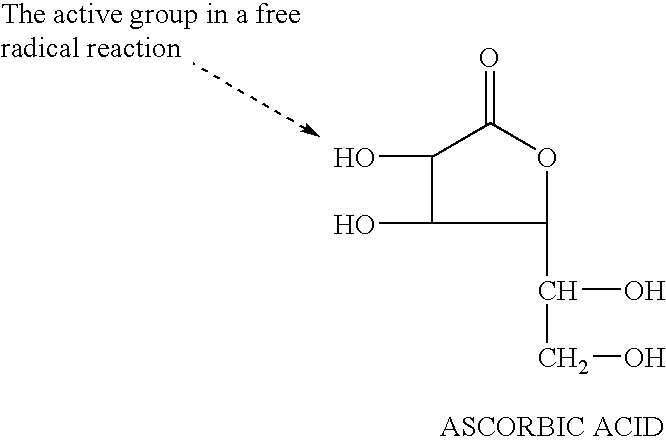
Vitamin C, also known as ascorbic acid and l-ascorbic acid, is a vitamin found in various foods and sold as a dietary supplement. It is used to prevent and treat scurvy. Vitamin C is an essential nutrient involved in the repair of tissue and the enzymatic production of certain neurotransmitters. It is required for the functioning of several enzymes and is important for immune system function. It also functions as an antioxidant.
Herbal enhanced analgesic formulations
InactiveUS20110028412A1Good analgesic effectIncrease productionBiocideNervous disorderVitamin CWhite willow bark
The analgesic properties of L-tryptophan and 5-HTP can be safely enhanced with the coadministration of salacin. Salacin can be effectively provided in the form of white willow bark along with other ingredients to further enhance the formulation's analgesic effect. As salacin can cause the loss of vitamin C in humans, the formulation advantageously includes a supplemental amount of vitamin C.
Owner:CAPPELLO INC
Composition for promoting healthy bone structure
InactiveUS6447809B1Increase bone densityPrevents radial bone lossBiocideHeavy metal active ingredientsVitamin CRegimen
A dietary supplement for benefitting human bone health includes a calcium source, a source of vitamin D activity, and an osteoblast stimulant. A preferred calcium source is microcrystalline hydroxyapatite, which also contains protein (mostly collagen), phosphorus, fat, and other minerals. A preferred source of vitamin D activity is cholecalciferol, and a preferred osteoblast stimulant is ipriflavone. In addition to these basic ingredients, the composition can further include various other minerals known to occur in bone, vitamin C, and glucosamine sulfate, all of which exert beneficial effects on growth and maintenance of healthy bone. A method for benefitting human bone health involves administering a daily regimen of the dietary supplement.
Owner:PHOENIX DICHTUNGSTECHN +1
Novel multifunctional jam
The invention provides novel multifunctional jam, which comprises fruit flesh particles, wherein the jam comprises the following ingredients through being calculated according to the weight: 70 to 80 parts of fruit flesh, 0.1 to 0.3 parts of vitamin C, 0.3 to 0.8 parts of citric acid, 80 to 120 parts of sweeteners and 0.5 to 1.2 parts of thickeners. The novel multifunctional jam has the beneficial effects that the pigment-free, essence-free and preservative-free green healthy jam integrating nutrition, delicacy and multifunctional eating is provided, the use additives of the jam are few, meanwhile, the jam can be made into fruit tea, fruity milk or fruity yoghourt by warm water, milk, yoghourt and the like, and the requirements of different user groups can be met.
Owner:TIANJIN PENGFANCHENG FOOD
Nutraceutical composition and method of use for treatment / prevention of cancer
InactiveUS20070248693A1Function increaseAbility to createBiocideAlgae medical ingredients1,4-BenzoquinonePantothenic acid
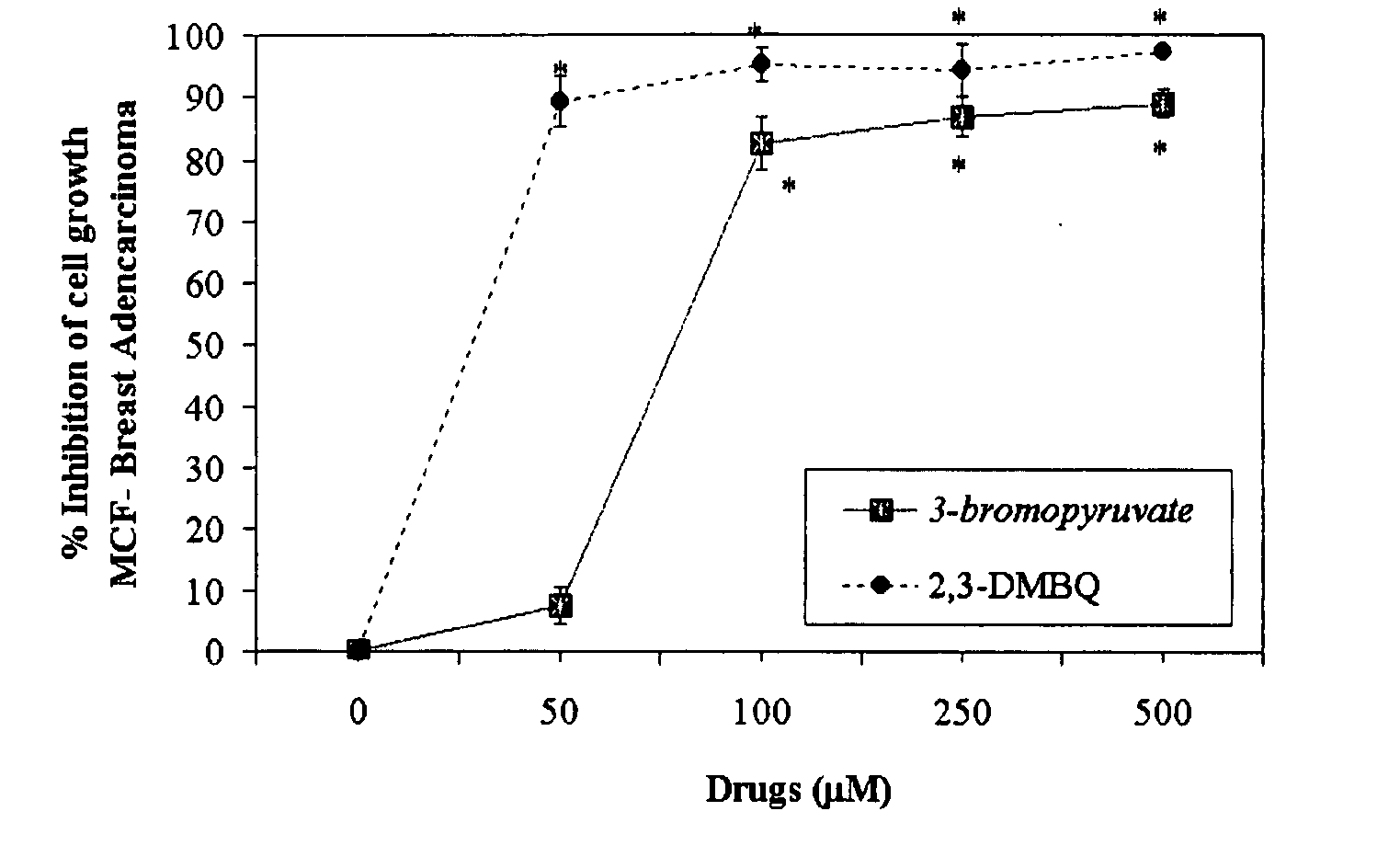
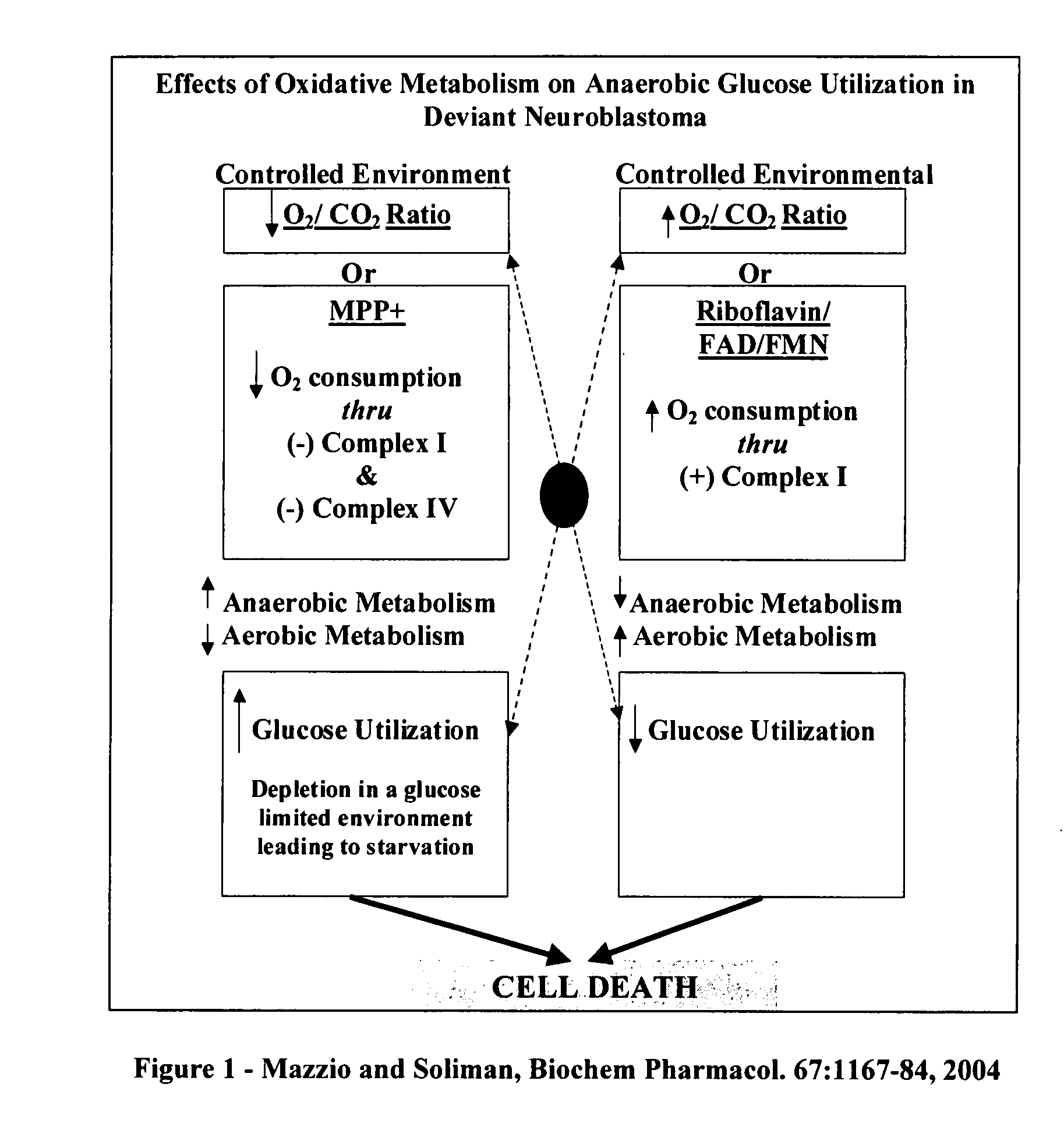
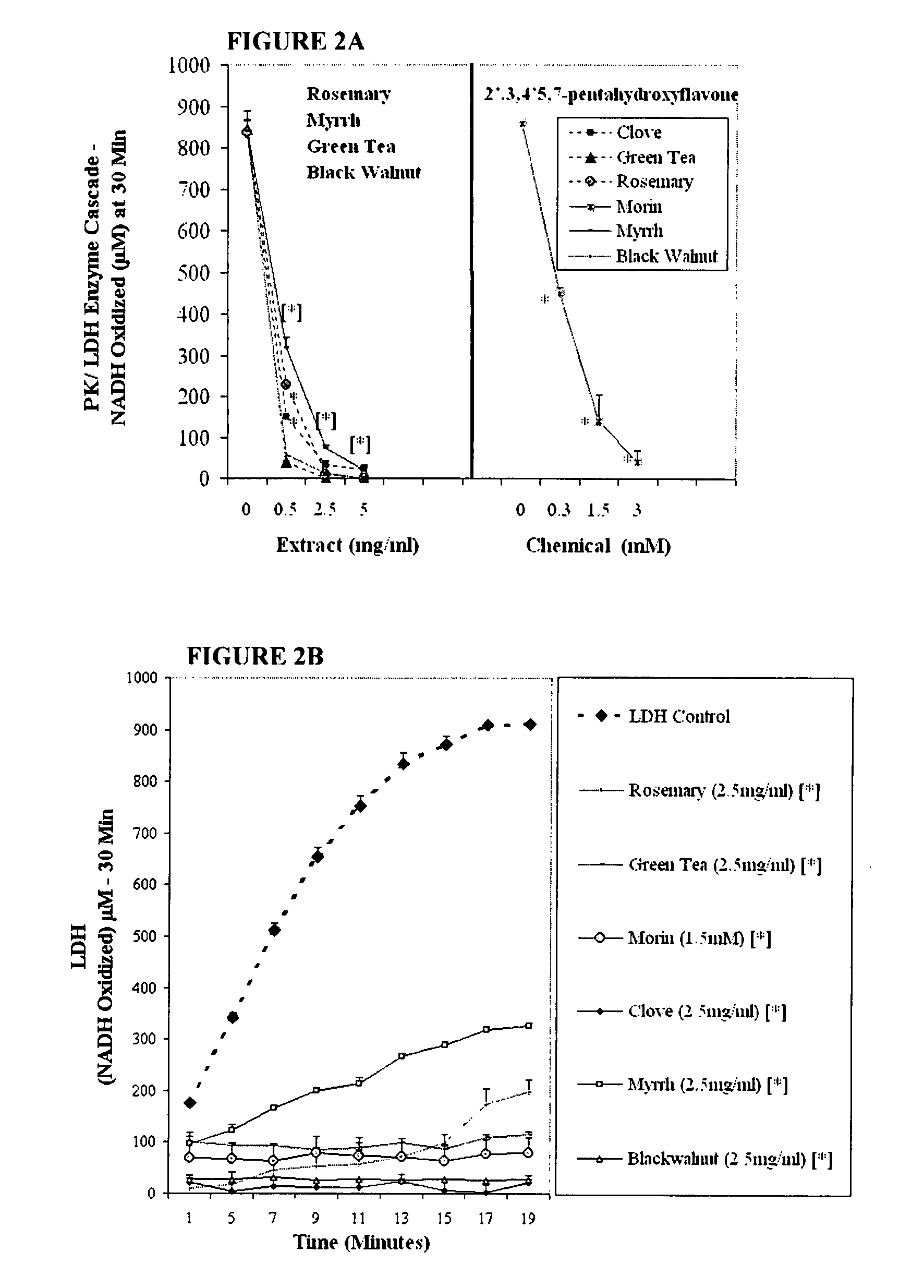
The invention describes a pharmaceutical composition and method for treating cancer comprised of A) 2,3-dimethoxy-5-methyl-1,4-benzoquinone and / or B) at least one of wild yam root, teasel root, balm of gilead bud, bakuchi seed, dichroa root, kochia seed, kanta kari, bushy knotweed rhizome, arjun, babul chall bark, opopanax and bhumy amalaki; optionally one or more of frankincense, garcinia fruit, vitex, dragons blood, mace, sage and red sandalwood with at least c) one compound capable of maximizing oxidative mitochondrial function preferably riboflavin or vitamin B2 derivatives, FAD, FMN, 5-amino-6-(5′-phosphoribitylamino)uracil, 6,7-Dimethyl-8-(1-D-ribityl)lumazine, ribitol, 5,6-dimethylbenzimidazole, tetrahydrobiopterin, vitamin B1, lipoic acid, biotin, vitamin B6, vitamin B12, folate, niacin, vitamin C and pantothenate and / or d) at least one lactic acid dehydrogenase inhibitor (preferably 2′,3,4′5,7-pentahydroxyflavone) and optionally f) an alkalizing agent (aloe vera, chlorella, wheat grass, sodium or potassium bicarbonate, potassium) g) an antiproliferative herb (speranskia or goldenseal) and h) a pharmaceutically acceptable carrier.
Owner:MAZZIO ELIZABETH +1
Pharmaceutical composition and method for the transdermal delivery of calcium
InactiveUS20070292493A1Reduce disadvantagesReduce and prevent likelihoodHalogenated hydrocarbon active ingredientsBiocideArginineTryptophan
The present invention relates to a method and transdermal pharmaceutical composition for preventing or reducing the likelihood of calcium deficiency or imbalances caused by calcium deficiency. The transdermal pharmaceutical composition includes a therapeutically effective amount of a pharmaceutically acceptable salt of calcium and a pharmaceutically acceptable carrier constituting a pluronic lecithin organogel. In addition to calcium, the transdermal pharmaceutical composition may also contain a therapeutically effective amount of: (1) a pharmaceutically acceptably salt of other minerals such as magnesium, zinc, selenium, manganese, or chromium; (2) a vitamin such as vitamin A, vitamin D, vitamin C, vitamin E or B-complex vitamins, choline, lecithin, inositol, PABA, biotin, or bioflavomoids; (3) a carotenoid such as lycopene or lutein; (4) a hormone such as dehydroepiandrosterone, progesterone, pregnenolone, or melatonin; (5) an amino acid such as arginine, glutamine, lysine, phenylalanine, tyrosine, GABA, tryptophan, carnitine, or acetyl-l-carnitine; (6) a fatty acid such as a fish oil or flax seed oil; (7) a vita-nutrient such as coenzyme Q10; (8) a cartilage building nutrient such as glucosamine, chondroitin, or MSM, (9) a herb such as ginkgo biloba, echinacea, 5-HTP, St. John's wort, or saw palmetto; or (9) any combination thereof. The transdermal pharmaceutical composition may be topically administered to a human to prevent or reduce the likelihood of calcium deficiency or imbalances caused by calcium deficiency such as hypertension, high cholesterol, colon and rectal cancer, osteomalacia, rickets, osteoporosis, cardiovascular disease, preeclampsia, tooth decay, and premenstrual syndrome.
Owner:BRIERRE BARBARA T
Nutritional composition for treating an immune condition
InactiveUS6929793B2Treat conditionPrevention or inhibition of diarrheaOrganic active ingredientsBacteriaVitamin CPhysiology
A nutritional composition is described for prevention or treatment of an immune condition. The composition includes at least vitamin E, vitamin C, vitamin B6, folic acid, vitamin B12, copper, zinc, selenium, fructo-oligosaccharides and / or gum acacia, a probiotic lactic acid bacterium. For example, in an embodiment it comprises per 300 g: 150 IU Vitamin E, 120 mg Vitamin C, 2 mg Vitamin B6, 400 ug Folic acid, 3.8 ug Vitamin B12, 1.5 mg Copper, 15 mg Zinc, 100 ug Selenium, 3 g Fructo-oligosaccharides and / or gum acacia, 10E10 cfu ST11 lactobacillus. Also disclosed are a method for making the nutritional composition, a method for manufacturing a functional food or medicament; and a method of prevention or treatment of an immune condition by administering an effective amount of the composition, functional food or medicament.
Owner:SOC DES PROD NESTLE SA
Prostate formula
InactiveUS6197309B1Good effectAlleviates and eliminates symptomHeavy metal active ingredientsOrganic active ingredientsDiseaseVitamin C
A composition providing an all-natural, non-surgical preventative of or improvement to disorders of the prostate gland is described. The invention relates to a composition for the prevention of or improvement of prostatitis, and for relieving symptoms and improving objective signs of prostatitis. The formula of the composition preferably includes the following ingredients each in a therapeutically effective amount: Vitamin C, Vitamin B6, Vitamin E, zinc, glycine, L-alanine, Glutamic acid, Saw palmetto, Pygeum extract, Pumpkin seed, Stinging nettle, Echinacea, garlic, Ginkgo leaves, and selenium.
Owner:WHEELER RONALD E
Nutritional system for nervous system disorders
A novel composition for treating nervous system disorders. The composition is formed by preparing a mixture comprising an effective amount of vitamin B-6, folic acid, vitamin C, magnesium, vitamin B-3, copper, probiotics, fructo-oligosaccharide (FOS), betaine, pancreatin, papain, pepsin, vitamin B-1, vitamin B-2, vitamin B-12, biotin, pantothenic acid, chromium polynicotinate and a digestive support ingredient selected from the group consisting of dandelion root, juniper, aloe vera, burdock, ginger root, artichoke, and kelp. Other ingredients may include: beta carotene, vitamin E, selenium, zinc, sea vegetation, alfalfa, trace minerals and molybdenum.
Owner:C&D FOREMAN
Anti-aging skin care composition and uses thereof
The present invention is directed toward anti-aging skin care compositions comprising Vitamin B1, Vitamin B5, Vitamin C, N-acetyl-cysteine and, optionally, lipoic acid. The present invention is further directed toward methods for therapeutically or prophylactically treating the consequences of aging on the condition or appearance of the skin. The present invention further provides one or more kits that are useful for delaying, treating or preventing the consequences of aging on the condition or appearance of the skin.
Owner:KLYSZ BEATRICE M
Method and synergistic composition for treating attention deficit/hyperactivity disorder
InactiveUS6541043B2Minimize side effectsBiocideHydroxy compound active ingredientsBeta-CaroteneBetaine
A composition and method for treating Attention Deficit / Hyperactivity Disorder (ADHD) is provided which can be used both with and without ethical drugs now used to treat ADHD. The composition contains dimethylaminoethanol (DMAE), omega 3-fatty acids, betaine, oligomeric proanthocyanidins (OPC), folic acid, vitamins C, E, B12, B6, B5 and beta-carotene and minerals (calcium, magnesium, zinc and selenium). Ethical drugs such as amphetamines, methylphenidate HCl and pemoline are known to control ADHD, but each has significant side effects when used in their therapeutic dose. When combining the composition with such ethical drugs, the amount of the ethical drug can be lowered below a level which causes undesirable side effects which is an important feature. Preferred compositions contain one or more of lecithin, choline, 5-hydroxytryptophan, tyrosine, Reishi Extract, Kava Extract, Gingko, Ginseng and St. John's Wort.
Owner:PHILIP C LANG
Blackthorn pear fruit-juice solid product and its production
The invention is concerned with the solid product of roxburgh rose juice and the preparation method, it is: prepares the roxburgh rose juice or combined roxburgh rose juice to be granule, tablet, capsule or effervesce agent in order to improve the stability of vitamin C, vitamin p and superoxide dismutase in it, and reduce the nutritive value loss of roxburgh rose juice. The preparation method is: 1) prepares clear roxburgh rose juice; 2) prepares roxburgh rose condensing juice; 3) adds 1% -50% accessories into the clear roxburgh rose juice or roxburgh rose condensing juice; 4) gets grains by wet-process, or makes instant powder of roxburgh rose then gets grains by wet-process; 5) the above grains dry at the temperature of 30 centigrade - 70 centigrade in order to get the solid products.
Owner:SHANGHAI NORMAL UNIVERSITY
Food compositions, compositions for oral cavity and medicinal compositions for preventing or treating periodontosis and method for preventing or treating periodontosis
InactiveUS6814958B1Preventing and treating periodontosisEffectiveCosmetic preparationsBiocideBeta-CaroteneVitamin C
Food compositions, oral compositions and pharmaceutical compositions for preventing or treating periodontosis, the compositions comprising (A) one or more extracts containing natural polyphenol; and (B) one or more members selected from the group consisting of vitamin C, vitamin E, vitamin A and beta-carotene; and methods for preventing or treating periodontosis using the above compositions.
Owner:SUNSTAR INC
Blackthorn pear products and its production
InactiveCN101073370AImprove stabilityReduce the loss of nutritional valueConfectionerySweetmeatsPEARVitamin C
The invention is concerned with a kind of Rosa roxburghii Tratt candy produce. It produces the syrup of pure Rosa roxburghii Tratt or compound Rosa roxburghii Tratt into horniness candy, horniness candy with filling, soft candy, milk lipid candy, polishing candy, glue-based candy, charging candy, chocolate candy or jelly, and so on. It increases the stability of vitamin C, vitamin P and superoxide dismutase and affords a kind of high quality candy food with double functions of nutritional health care and relaxing. The produce method relates to prepare clarified Rosa roxburghii Tratt concentrated syrup with good stability, add assistant stuff as the amount of 1 to 50 percent of whole amount into the said syrup, prepare Rosa roxburghii Tratt grain or Rosa roxburghii Tratt fast-dissolving powder and follow the routine technology to produce horniness candy, horniness candy with filling, gel candy, glue-based candy, charging candy, chocolate candy or jelly, and so on.
Owner:SHANGHAI NORMAL UNIVERSITY
Free radical quenching composition and a method to increase intracellular and/or extracellular antioxidants
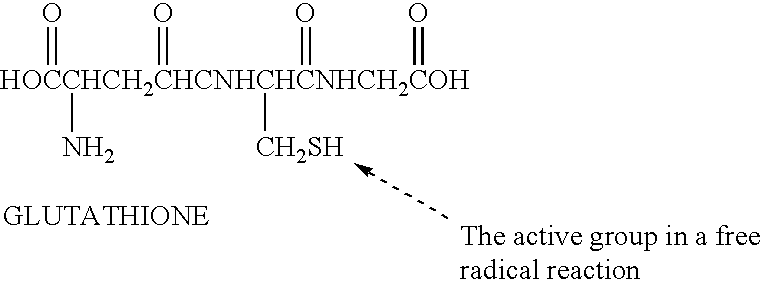
A free radical quenching composition is disclosed comprising a liposome containing at least two antioxidants selected from the following group: beta-carotene, vitamin E, vitamin C, glutathione, niacin, and optionally at least one trace metal (Zn, Se, Cr, Cu, Mn). Also disclosed is a method for reducing the undesirable side effects of free radicals in a mammal by administering to a mammal in need of such antioxidants an effective amount of liposomes containing at least two antioxidants.
Owner:AMAOX
Composition and method for treatment of inflammation and pain in mammals
A composition for treating inflammation and pain in mammals, particularly humans. Effective amounts of a boswellic acid, a curcuminoid, a gingerol, a capsaicinoid, a bioflavonoid, and a vitamin C source, in various combinations, all preferably from a botanical source, are blended to form a dose for oral administration. Administration of the dose provides relief from pain and inflammation of connective tissue. The dose may be administered as a tablet, a liquid, or a powder.
Owner:METAGENICS INC
Nutritional supplement for treatment of ocular diseases
A nutritional supplement composition that promotes visual health and reduces or reverses visual acuity loss by a reduced Vitamin E content from standard supplements with the addition of taurine, omega-3 fatty acids, and non proform Vitamin A carotenoids including lutein and zeaxanthin. The essential ingredients of the nutritional or dietary supplements are Vitamin C, no more than 300 IUs of Vitamin E, Vitamin A at least a portion of which is provided in the form of a proform Vitamin A carotenoid, omega-3 fatty acids, and non proform Vitamin A carotenoids including lutein and / or zeaxanthin. The essential ingredients are provided in a form suitable of oral ingestion or other forms of administration in one or more doses per day.
Owner:PAUL JR EDWARD L
Animal chew toy containing solid food
InactiveUS20080314333A1Easy to processImprove and protect healthPeptide/protein ingredientsBoron compound active ingredientsVitamin CManganese
An animal toy containing food having a first portion with food delivery means integrated therein adapted to securely house solid food treats, including a nutritional pet supplement, to be removed by the animal during play or through chewing action, and a second side with a plurality of gum stimulation teeth integrated within and projecting therefrom that act to massage the gums of the animal. Food delivery means may be provided as a plurality of cavities integrated within the first portion (and optionally, the second portion), preferably having a grooved or threaded interior for securely holding food pieces securely therein. Alternatively, the food delivery means is provided as a food portion composing the first portion. A pet supplement may be utilized, including joint preserving and joint rebuilding compositions comprising chicken collagen type II, glucosamine hydrochloride and chondroitin sulfate, a vitamin composition comprising vitamins C, D and K, a mineral composition comprising calcium, magnesium, zinc, copper, manganese and boron, a herbal anti-oxidant cofactor blend comprising citrus bioflavonoids, red grapes anthocyanins, turmeric rhizome, boswellia resin and fennel seed.
Owner:I DID IT
Treatment and prevention of abnormal scar formation in keloids and other cutaneous or internal wounds or lesions
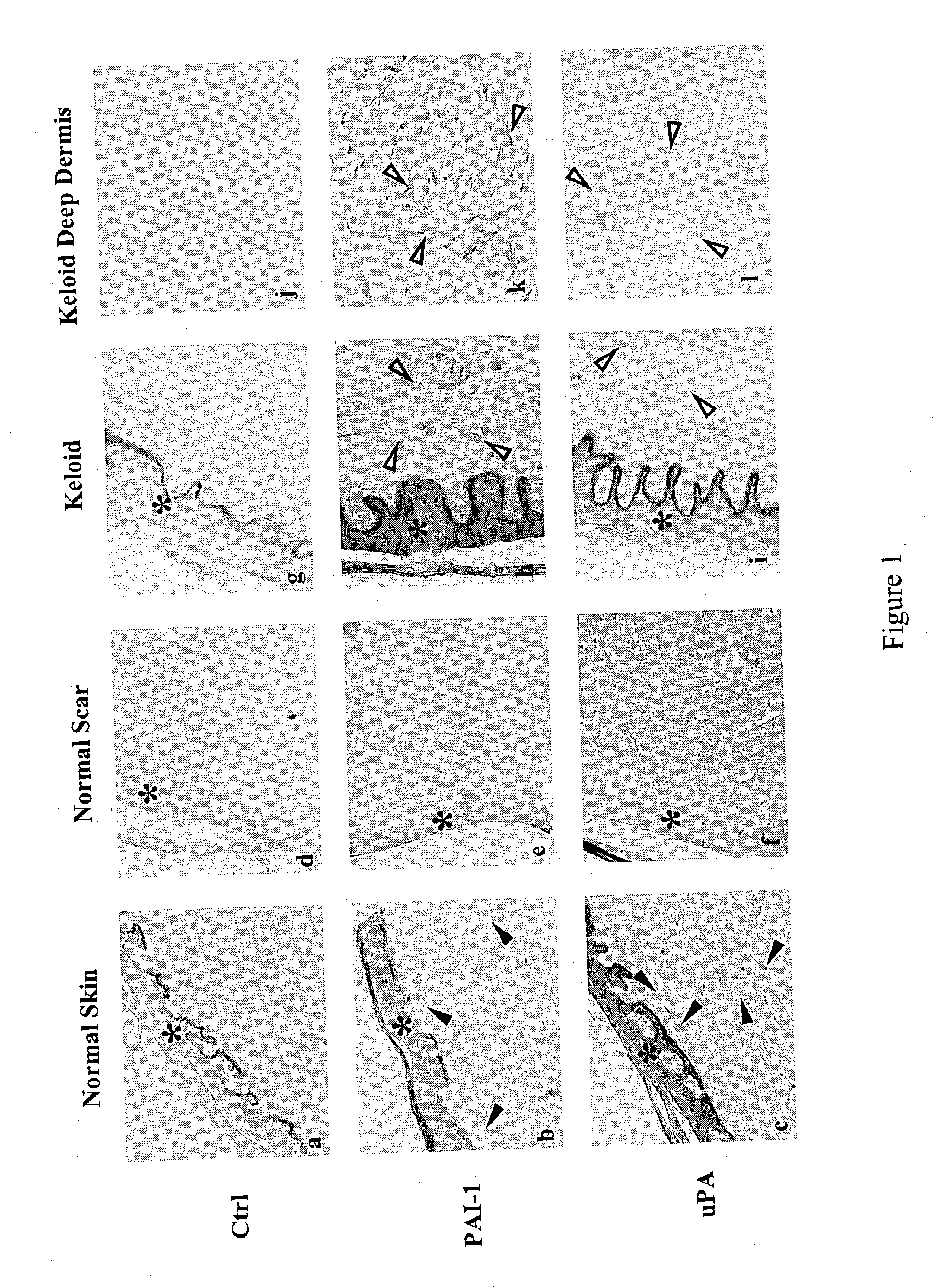
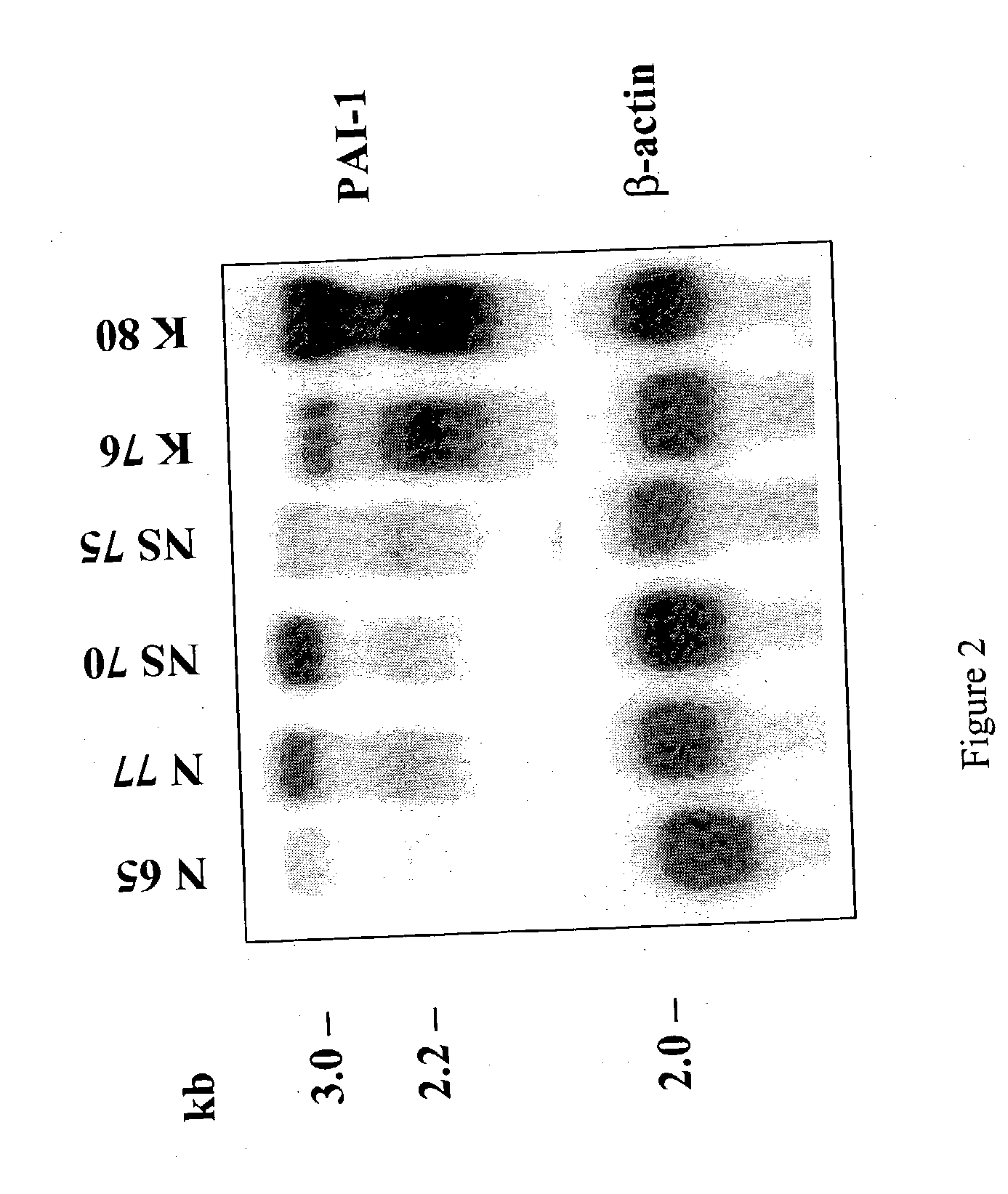
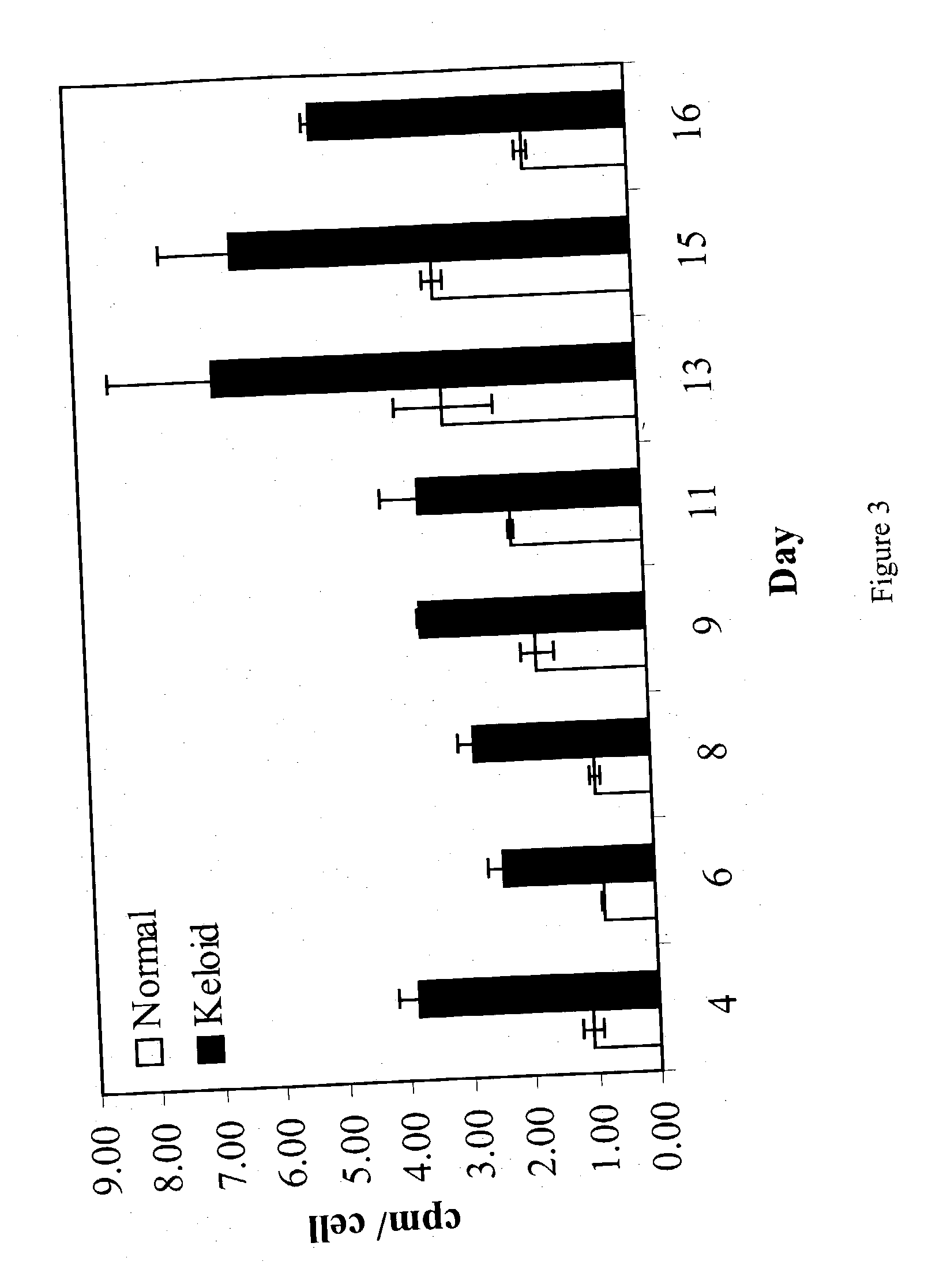
The present invention relates to findings that reducing the activity of Plasminogen Activator Inhibitor-1 (PAI-1) suppresses an excessive deposition of collagen which is known as a cause for the formation of abnormal scars. These abnormal scars include but are not limited to keloids, adhesions, hypertrophic scars, skin disfiguring conditions, fibrosis, fibrocystic conditions, contractures, and scleroderma, all of which are associated with or caused by an excessive deposit of collagen in a wound healing process. Accordingly, aspects of the present invention are directed to the reduction of PAI-1 activity to decrease an excessive accumulation of collagen, prevent the formation of an abnormal scar, and / or treat abnormal scars that result from an excessive accumulation of collagen. The PAI-1 activity can be reduced by PAI-1 inhibitors which include but are not limited to PAI-1 neutralizing antibodies, diketopiperazine based compounds, tetramic acid based compounds, hydroxyquinolinone based compounds, Enalapril, Eprosartan, Troglitazone, Vitamin C, Vitamin E, Mifepristone (RU486), and Spironolactone to name a few. Another aspect of the present invention is directed to methods of measuring PAI-1 activity in a wound healing process and determining the propensity of the formation of an abnormal scar.
Owner:CHILDRENS HOSPITAL OF LOS ANGELES +1
Hen feedstuff for improving egg-laying performance
ActiveCN101181016AIncrease egg productionImprove immunityAnimal feeding stuffAccessory food factorsEggshellVitamin C
The invention relates to a layer foodstuff used for enhancing egg laying performance, which has the components in weight portion as follow: 100 to 160 portions of calcium hydrogen phosphate, 250 to 350 portions of mountain flour, 40 to 70 portions of common salt, 20 to 40 portions of saleratus, 12 to 20 portions of choline, 30 to 40 portions of complex trace elements, 8 to 10 portions of complex vitamins, 1 to 3 portions of vitamin C, 25 to 30 portions of methionine, 50 to 100 portions of fish meal, and 250 to 400 portions of zeolite powder. Such additives as nutritive additive are added into the foodstuff in the invention, which has the advantages of apparently strengthening immunity of the layer, maintaining a long egg laying peak period, good quality of an egg shell, high egg laying rate, low layer culling rate, and obviously improving egg laying performance.
Owner:KUNMING HEMEIHUA FEED
Skin lightening compositions
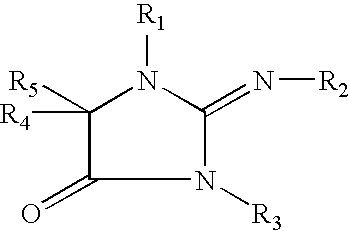
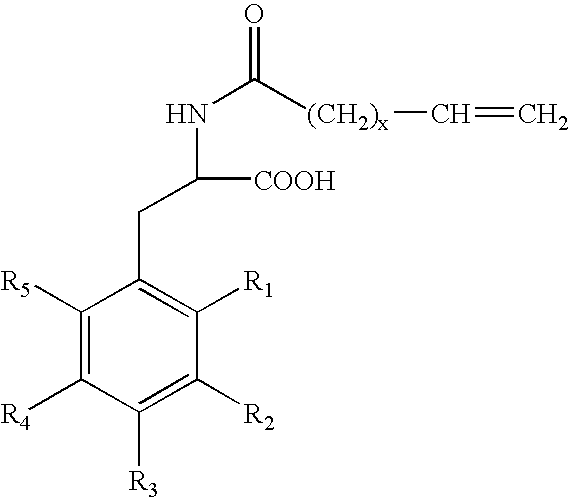
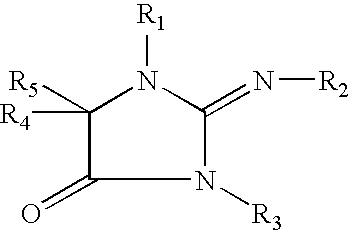
The present invention concerns methods and compositions that can be used, for example, in skin whitening or hyperpigmentation applications. The composition, in non-limiting aspects, can include a vitamin C derivative, niacinamide, an extract formulation comprising cucumber and lemon extract or compounds that include the following formulas: where R1, R2, R3, R4, and R5 are independently an H, an alkyl group, a hydroxyalkyl group, or a carboxyalkyl group where R1, R2, R3, R4, and R5 are independently an H, an alkyl group, a hydroxyalkyl group, or a carboxyalkyl group and where x is an integer from 1 to 30.
Owner:MARY KAY INC
Serum-free culture medium for mesenchymal stem cells
ActiveCN102827807AAvoid instabilityClear chemical compositionSkeletal/connective tissue cellsCell phenotypeSodium bicarbonate
The invention relates to the field of biology, and discloses a serum-free culture medium which essentially comprises an IMDM (Iscove Modified Dulbecco Medium), L-glutamine, sodium bicarbonate, Hepes, recombinant human insulin, recombinant human transferrin, recombinant human albumin, 2-mercaptoethanol, protocatechuic acid, lipid, amino acid, vitamins, trace elements, Pluronic F-68, hydrocortisone, vitamin C, bonding amine or recombinant human fibronectin, progesterone, putrescine, heparin, serotonin, epidermal growth factors (EGFs), b-fibroblast growth factors (FGF), platelet derive growth factor (PDGF)-BB and insulin-like growth factor (IGF)-I. The serum-free culture medium is clear in chemical components, free from animal sources and serum and safe and ideal in cell cultivation and avoids the doped animal components and unstable batches, and the results of the cultured mesenchymal stem cells show that the total cellular score, the cell phenotype and the secretory cell factors are normal, so that the serum-free culture medium has good industrial application prospect.
Owner:内蒙古干细胞医学工程技术研究中心
Antioxidant composition for topical/transdermal prevention and treatment of wrinkles
InactiveUS6180133B1Significant free radical protectionHead bandagesCosmetic preparationsWrinkle skinVitamin C
An anti-wrinkle skin treating composition comprises a pressure sensitive matrix patch having dissolved in the adhesive a mixture of antioxidants in the form of a Vitamins C ester and Vitamin E. Also preferably dissolved in the adhesive are glycerine and a polydiorganosiloxane adhesion-adjusting agent. Optionally dissolved in the adhesive is also one or more members selected from the group consisting of moisturizing agents, skin collagen synthesis promoting agents and exfoliating agents. When applied to a wrinkled skin area the composition acts to diminish fine wrinkles and improves the overall thickness, elasticity, firmness and smoothness of the skin. The modified adhesive properties of the patch are sufficient to maintain the patch in place on the skin for the recommended treatment period while allowing the patch to be readily removed without causing skin irritation or leaving adhesive residue on the skin.
Owner:ACTAVIS HOLDCO US INC
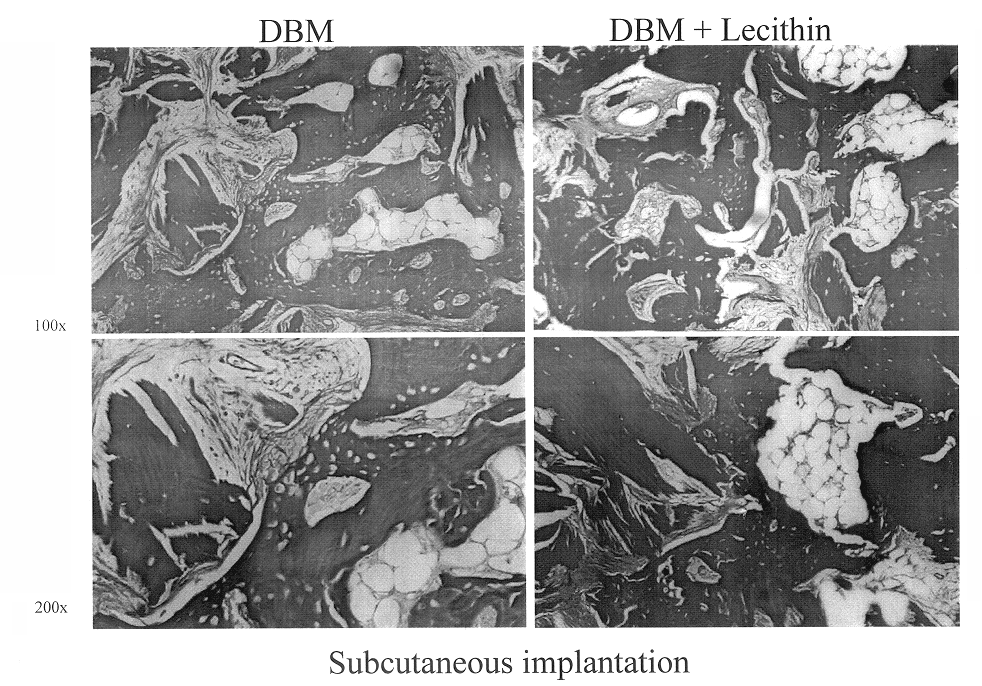
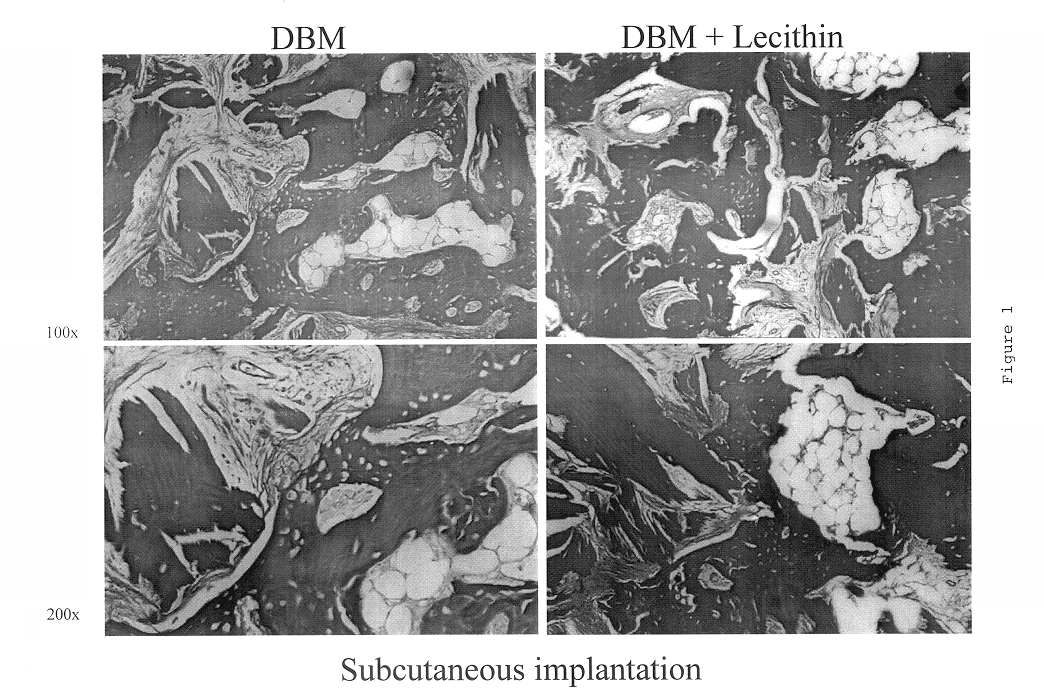
Bone graft material incorporating demineralized bone matrix and lipids
InactiveUS6565884B2Good osteoinductivityEasy to operateBiocidePowder deliveryHydrophilic polymersVitamin C
A demineralized bone putty composition comprises: (1) demineralized bone matrix (DBM); and (2) a lipid fraction selected from the group consisting of lecithin and a mixture of lecithin and triglycerides containing unsaturated fatty acids. The putty composition is moldable, biocompatible, slowly resorbable, and soluble in tissue fluids, and non-extrudable. The composition delivers a biologically active product to animals and humans that will enhance bone formation at sites where bone is lost, deficient, or present in suboptimal amounts. The composition can further comprise calcium, an antioxidant such as Vitamin E or Vitamin C, or a hydrophilic polymer such as methylcellulose or hydroxypropyl methylcellulose.
Owner:BIOMET MFG CORP
Freezing flour-dough improver and uses thereof
ActiveCN101411344AOvercome stabilityOvercome volumeDough treatmentPre-baking dough treatmentYeastVitamin C
The invention discloses a frozen dough modifying agent, which is prepared by evenly mixing an enzyme preparation (including one or a plurality of alpha-amylase, cellulose, hemicellulase, pentosanase, lipase, glucose oxidase, glutamine transaminage and so on), vitamin C, an emulsifying agent, a thickening agent, wheat gluten powder, lecithin and stuffing according to certain proportion. The frozen dough modifying agent has the advantages that the frozen dough modifying agent can effectively improve the stability of dough during fermentation and roasting processes, improve the freezing resistance property of yeasts, increase the volume of finished products, improve the tissue of the finished products, reduce the loss of the vitality of the yeasts during the freezing process, and effectively delay the aging of the finished products.
Owner:ANGELYEAST CO LTD
Cosmetic composition with anti-glycosylation and anti-aging effects and application thereof
PendingCN111329808AEfficient removalAvoid damageCosmetic preparationsToilet preparationsBiotechnologyVitamin C
The invention provides a cosmetic composition with the anti-glycosylation and anti-aging effects and an application thereof, and belongs to the technical field of functional compositions. The cosmeticcomposition comprises 0.5-4 parts of vitamin C, 0.5-4 parts of SOD-containing yeast powder, 0.2-5 parts of haematococcus pluvialis, 0.5-10 parts of rooibos tea, 0.5-8 parts of licorice extract, 0.1-6parts of waxberry extract, 0.5-8 parts of longan fruit shell extract, 0.1-8 parts of burdock polyphenol, 0.5-8 parts of cinnamon extract, 0.1-5 parts of kudzu root extract, 1-10 parts of purslane extract, 2-40 parts of carbohydrates, 2-30 parts of proteins and 0.5-5 parts of lipids. The cosmetic composition can achieve the melanogenesis-blocking and full-whitening effects, and the comprehensive anti-aging and whitening effects are achieved by a basic prescription.
Owner:西安力邦医美科技有限公司
Cell preservation solution and preparation method and applications thereof
ActiveCN104542578AExcellent maintenance of biological characteristicsMaintain biological characteristicsDead animal preservationVitamin CAmino Acid Injection
The invention relates to the technical field of clinical medicines, and particularly relates to a cell preservation solution and a preparation method and applications thereof. The cell preservation solution comprises albumin, glucose, vitamin C and basic preservation liquid, wherein the basic preservation liquid is a mixture of compound electrolyte injection and compound amino acid injection. The cell preservation solution disclosed by the invention achieves a good maintaining effect on the viability and morphology of more than two kinds of seed cells, and can preserve the seed cells for a long time, and maintain the biological characteristics of the seed cells, thereby significantly improving the therapeutic effect of the seed cells.
Owner:GUANGZHOU SALIAI STEMCELL SCI & TECH CO LTD
Fatty acid compositions and methods of use
InactiveUS20090011012A1Easy to carryConvenient travelBiocideCapsule deliveryCyclosporine toxicityAntioxidant
The invention relates to highly concentrated DHA and EPA formulations in a soft gel capsule. A capsule may contain at least 80% omega-3 fatty acids, salts or derivatives thereof, where EPA and DHA are present in relative amounts of greater than or equal to 3:1 or less than or equal to 1:3, and constitute at least 75% to greater than 95% of the total fatty acids present in the capsule. Capsules of the invention may be provided in a blister package so as to provide clean and protected oils that are easy to travel with. Compliance is improved with one-pill-a-day dosing and the days of the week imprinted on the foil packing. Anitoxidant protection may be provided by rosemary and vitamin C. The invention also provides a methods of treatment, modulation or prophalaxis of coronary disease, altering serum LDL-cholesterol and / or HDL-cholesterol, lowering serum triglycerides, lowering blood pressure, pulse rate, altering the activity of the blood coagulation factor VII complex, mild hypertension, protection from cyclosporine toxicity in kidney transplant, rheumatoid arthritis, development and progression of retinopathy, hypertriglyceridemia, and neurological disorders in a subject.
Owner:BAUM SETH J
Color changing skin sealant
Iodine is used in about 80 percent of surgeries in the US to remove some level of microbial load on the skin prior to making an incision. Skin sealants are applied over skin preps to seal the skin and hold any remaining bacteria in place. Iodine produces a characteristic orange-brown color on skin. A skin sealant is provided that has a decolorant that reacts with the iodine found in most skin preps, rendering the skin prep colorless. A skin sealant containing ascorbic acid (vitamin C), Indigo Carmine or Indigo will react with the iodine, thus visually indicating where the skin prep and sealant have been applied and allowing an unobstructed view of the incision.
Owner:KIMBERLY-CLARK WORLDWIDE INC
Antioxidant Compositions and Methods of Using the Same
ActiveUS20140315995A1Prevent degradationFacilitate preventionCosmetic preparationsBiocideVitamin CNitrogen
The invention provides non-irritating, stable topical compositions including at least Vitamin C, Vitamin E and a polyphenol antioxidant. Such compositions can be used to facilitate the prevention or treatment of free oxygen, nitrogen, and / or other free radical related skin damage. Also provided are methods for modifying free radical damage to skin by administering such compositions in an amount sufficient to treat and / or prevent free radical damage to the skin.
Owner:ANTEIS SA
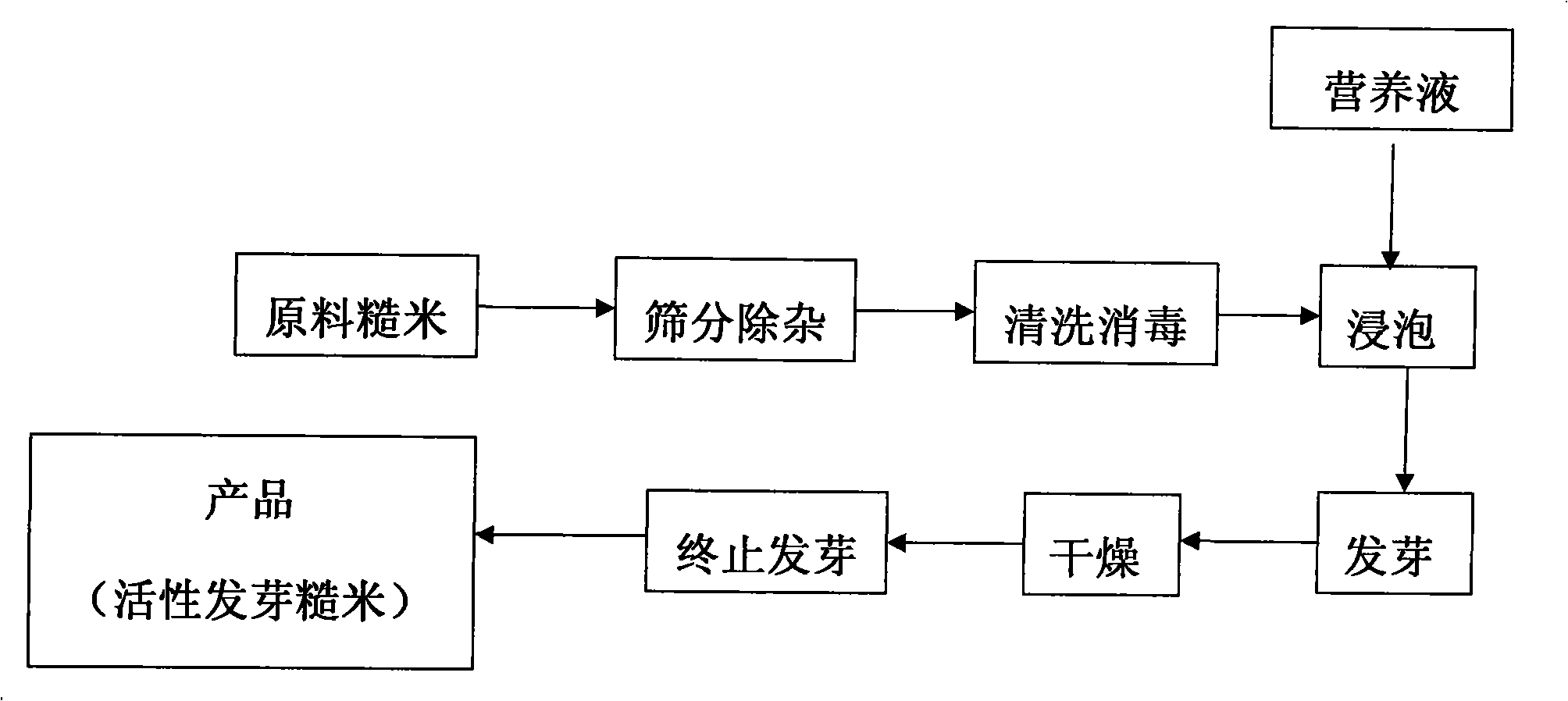
Production method of active germinated unpolished rice and its products
InactiveCN101283754APromote digestion and absorptionRich varietyFood preparationFertilizer mixturesNutritive valuesVitamin C
The invention belongs to the technology field of grain processing, specifically a method for producing active germinated brown rice and products thereof. The method comprises the following steps: screening brown rice raw material to remove impurity, washing, sterilizing, soaking in a nutrient fluid supplemented with calcium lactate, gibberellin, phytase, cellulase and vitamin C for 10-36 hours at 10-40 DEG C, draining, incubating for 10-36 hours at 10-40 DEG C under relative humidity of 50-90% to allow germination, drying with hot air of 40-55 DEG C for 0.5-3 hours until the sprout grows about 0.5-1.2 mm in height, drying with 2-10 w / g microwave for 2-10 minutes until the water content of germinated brown rice is below 15%. Compared with the prior art, the germinated brown rice is nutritious, retains almost all nutrients of brown rice and has high nutritional value.
Owner:HUAZHONG AGRI UNIV