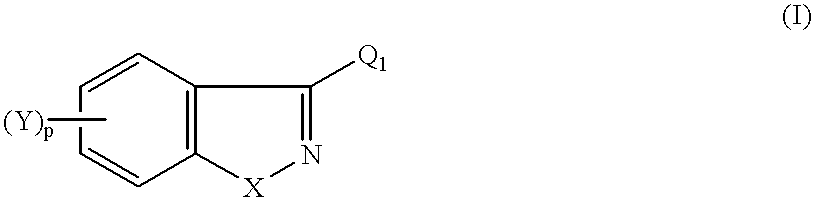
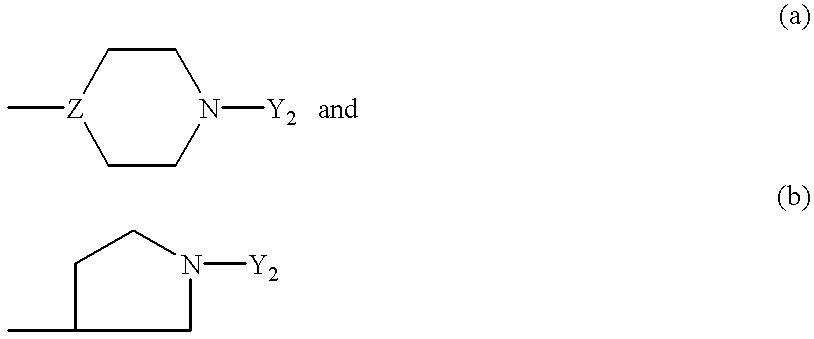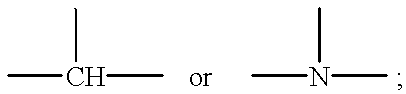1-(arylthioalkyl, arylaminoalkyl, or arylmethylenealkyl)-4-(heteroaryl)piperazines and related compounds useful as antipsychotics and analgesics
a technology of arylthioalkyl and arylmethylenealkyl, which is applied in the field of heteroarylpiperidines, pyrrolidines and piperazines, can solve the problems of known neuroleptics producing unwanted side effects, unable to cure psychotic patients, and almost certainly relapse, and achieves long-acting effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
1-[4-[3-[4-(1H-Indazol-3-yl)-1-piperazinyl]propoxy]-3-methoxyphenyl]ethanon e
(A) 2-Bromobenzoic acid 2-phenylsulfonylhydrazide
To a solution of 2-bromobenzoic acid hydrazide (132 g) in pyridine (1.2 L) cooled to about 10 C. with an ice bath, was added benzensulfonyl chloride (78.3 ml). After complete addition, the reaction was stirred at ambient temperature for four hours, and then poured into ice-hydrochloric acid to precipitate a yellow solid, 135 g. The material was recrystallized from isopropanol to yield 125 g of 2-bromobenzoic acid 2-phenylsulfonylhydrazide, m.p.=154.degree.-156.degree. C.
(B) .alpha.-Chloro-2-bromobenzaldehyde phenylsulfonylhydrazone
A mixture of 2-bromobenzoic acid phenylsulfonylhydrazide (125 g, 350 mmol) and thionyl chloride (265 ml) was stirred and refluxed for 2 hours. After about 15 minutes of reflux, the solid went into solution. The reaction was permitted to cool, and then it was poured into hexane. The resultant white solid was collected to afford 124 g...
example 2
1-[4-[3-[4-(1,2-Benzisoxazol-3-yl)-1-piperidinyl]propoxy]-1-methyoxyphenyl] -ethanone
A mixture of 3-(4-piperidinyl)-1,2-benzisoxazole hydrochloride (4.8 g, 20 mmol), K.sub.2 CO.sub.3 (5.2 g, 40 mmol), 1-[4-(3-chloropropoxy)-3-methyoxyphenyl]ethanone (5.3 g, 22 mmol), a few crystals of KI and dimethylformamide (60 ml) was stirred at 90 C. for 16 hours. The reaction was poured into water and the aqueous mixture was extracted with ethyl acetate. The extract was washed (water), dried (MgSO.sub.4) and concentrated to afford a brown oil. The oil was chromatographed on a Waters Prep 500 utilizing silica gel columns and ethyl acetate-diethylamine (2%), as eluent. Concentration of the appropriate fractions afforded 3.9 g of product as an off-white solid. Recrystallization from absolute ethyl alcohol afforded 2.6 g (33%) of 1-[4-[3-[4-(1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methoxypehenyl ]ethanone, m.p.=102.degree.-104.degree. C., as colorless needles.
ANALYSIS: Calculated for C.sub....
example 3
1-[4-[3-[4-(6-Fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-methox y-phenyl]ethanone
A stirred mixture of 6-fluoro-3-(4-piperidinyl)-1,2 benzisoxazole hydrochloride (5.1 g, 20 mmol), K.sub.2 CO.sub.3 (5.2 g, 40 mmol), 1-[4-(3-chloropropoxy)-3-methoxyphenyl]ethanone (5.3 g, 22 mmol), and dimethylformamide (60 ml) was heated at 90.degree. C. for 16 hours. The reaction was poured into water, and the aqueous mixture was extracted with ethyl acetate. The ethyl acetate was washed (water), dried (MgSO.sub.4) and concentrated to afford a moist solid. Recrystallization (twice) from ethyl alcohol afforded 5.0 g (58%) of 1-[4-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3-metho xyphenyl]-ethanone as a beige solid, m.p.=118.degree.-120.degree. C.
ANALYSIS: Calculated for C.sub.24 H.sub.27 FN.sub.2 O.sub.4 : 67.60% C 6.38% H 6.57% N Found: 67.47% C 6.40% H 6.53% N.
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More