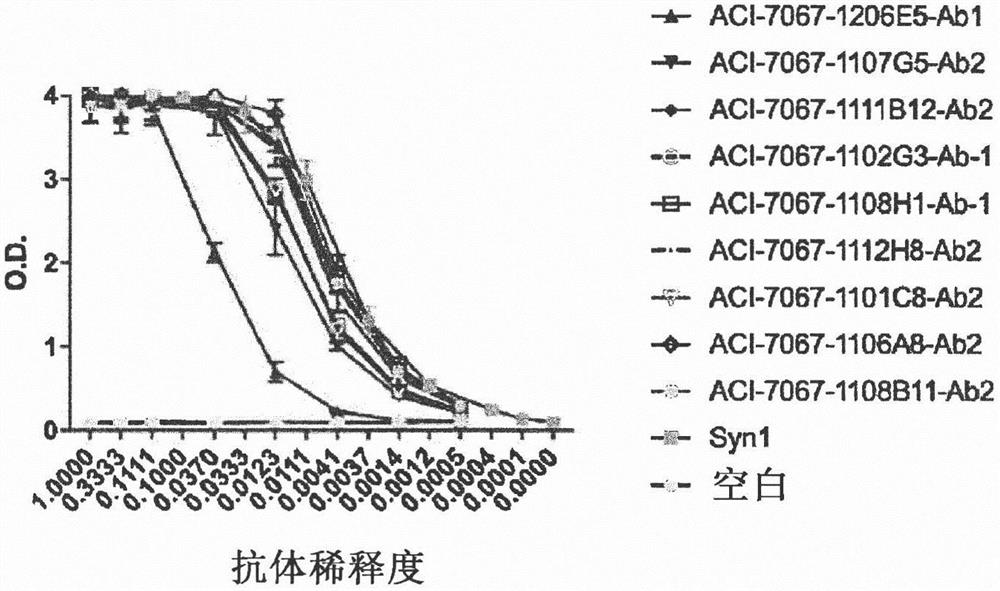
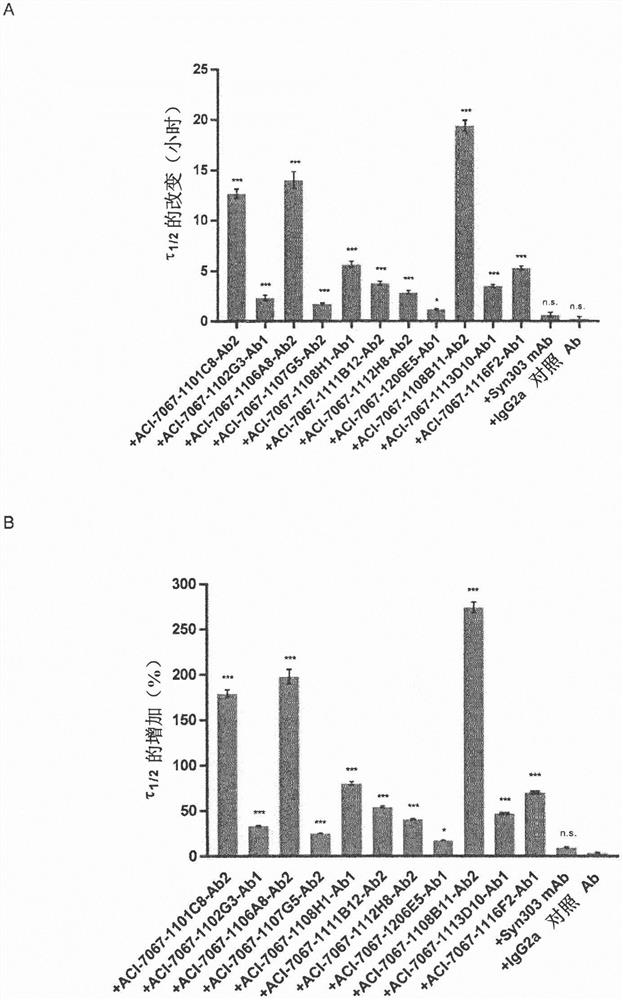
Novel molecules for therapy and diagnosis
A technology for binding molecules, synuclein, applied in the field of α-synuclein antibody or its antigen-binding fragment or its derivatives
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0930] Preparation of α-synuclein liposome vaccine composition
[0931] Liposome-based antigenic constructs were prepared according to the protocol disclosed in WO2012 / 055933. Liposomal vaccine with human full-length α-synuclein as antigen for antibody production (Table 2, SEQ ID NO: 1) or liposomal vaccine with α-synuclein peptide as antigen for antibody production .
[0932] Table 2: Antigen description
[0933]
[0934] mouse immunization
[0935] Female C57BL / 6JOlaHsd and BALB / cOlaHsd mice (Envigo, USA) were vaccinated at 10 weeks of age. The C57BL / 6JOlaHsd substrain is known to have a spontaneous deletion of the α-synuclein gene. Mice were vaccinated with a vaccine in which synthetic monophosphorylhexayl lipid A 3-deacylated (3D-(6-acyl) ) (Avanti Polar Lipids, USA) containing α-synuclein peptide or human full-length α-synuclein present on the surface of liposomes.
[0936] Mice were vaccinated by subcutaneous injection (s.c.) on days 0, 5, 8, 21, 35, 84 and in ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com