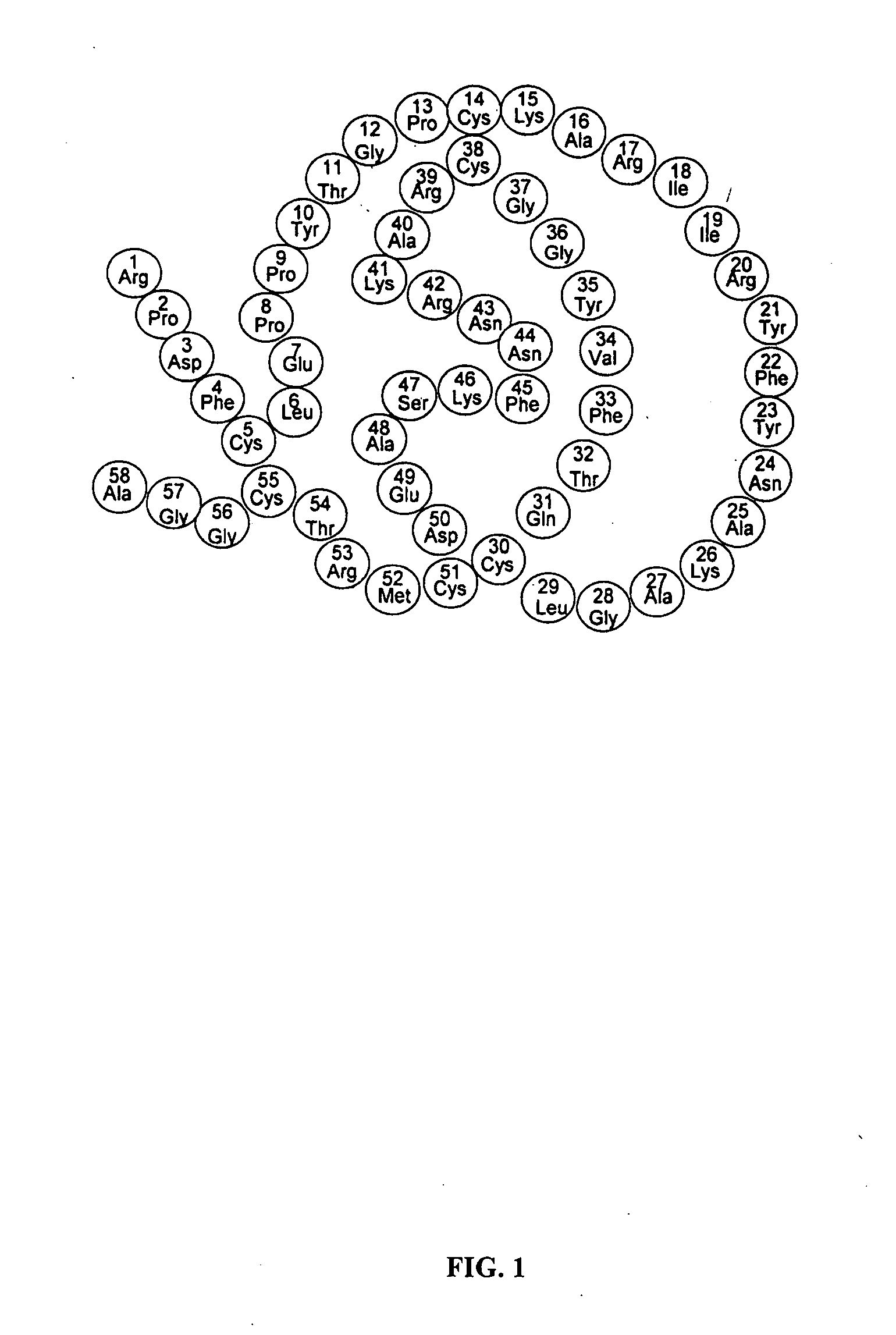Broad specturm anti-viral therapeutics and prophylaxis
a broad-specturm, antiviral technology, applied in the field of therapeutic compositions, can solve the problems of drug-resistant viruses, inability to effectively treat bronchopneumonia, and inability to meet the needs of patients,
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
Synthesizing Aprotinin Genes, Purifying and Testing Aprotinin Fusion Proteins
Introduction
[0100] Influenza viral protein hemagglutinin (HA) is the major influenza envelope protein. It plays an essential role in viral infection. The importance of HA is evidenced by the fact that it is the major target for protective neutralizing antibodies produced by the host immune response (Hayden, F G. (1996) In Antiviral drug resistance (ed. D. D. Richman), pp. 59-77. Chichester, UK: John Wiley & Sons Ltd.). It is now clear that HA has two different functions in viral infection. First, HA is responsible for the attachment of the virus to sialic acid cell receptors. Second, HA mediates viral entry into target cells by triggering fusion of the viral envelope with cellular membranes.
[0101] HA is synthesized as a precursor protein, HA0, which is transferred through the Golgi apparatus to the cell surface as a trimeric molecular complex. HA0 is further cleaved to generate the C terminus HA1 (resid...
example 2
Establishing Improved Tissue Culture Models for Studies on Influenza Virus Infection
Stocks of Influenza Viruses
[0121] Influenza viral strains are obtained from ATCC and the repository at St. Jude Children's Research Hospital. All experiments involving influenza viruses are conducted at Bio-safety level II.
[0122] Viruses are propagated by injection into the allantoic cavity of nine-day-old chicken embryos as described (Zhirnov O P, Ovcharenko A V and Bukrinskaya A G. (1985) J Gen Virol 66:1633-1638). Alternatively, viral stocks are grown on Madin-Darby canine kidney (MDCK) cells in minimal essential medium (MEM) supplemented with 0.3% bovine serum albumin and 0.5 micrograms of trypsin per ml. After incubating for 48 to 72 hours, the culture medium is clarified by low speed centrifugation. Viral particles are pelleted by ultracentrifugation through a 25% sucrose cushion. Purified viruses are suspended in 50% glycerol-0.1M Tris buffer (pH 7.3) and stored at −20° C.
Plaque Assays
[...
example 3
Comparing Functions of the Aprotinin Fusion Proteins In Vitro
Anti-Viral Effects of Aprotinin Fusion Proteins
[0131] 1. Pre-infection treatment. Aprotinin fusion proteins are added to primary human cell cultures at various concentrations and allowed to incubate with the cells for 1 hour. The cells are washed with fresh medium and immediately inoculated with influenza viruses at MOI 0.01 to 1. Cells are washed again after 1 hour and cultured for 3 to 5 days. Titer and infectivity of viruses in the supernatant are measured at various time points by two plaque assays. The cytopathic effect caused by viral infection is evaluated by staining viable cells with crystal violet and quantifying by measuring absorption at 570 nm at the end of the experiment. The percentage of cell protection by aprotinin fusion proteins is calculated by 100.times.{(aprotinin treated sample-untreated infected sample) / (uninfected control-untreated infected sample)}. The drug efficacy for cell protection is desc...
PUM
| Property | Measurement | Unit |
|---|---|---|
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| dissociation constant | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More