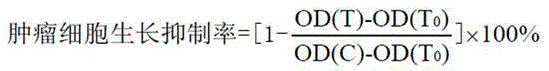Novel camptothecin derivative and antitumor application thereof
A technology of derivatives and camptothecin, which is applied in the field of novel camptothecin derivatives and their anti-tumor applications, can solve the problems of decreased drug activity and toxic reactions, and achieve the effects of low cost, readily available raw materials, and simple synthetic routes
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0030] Embodiment 1. Side chain 5,6-dihydronorcantharidin monomethyl ester 2 preparation of
[0031] 1), 5,6-Didehydronorcantharidin 1 Preparation of:
[0032] Take out a certain amount of maleic anhydride from the reagent bottle, place it in a dry grinding body and grind it finely, then weigh 12.021g of the finely ground maleic anhydride with an electronic balance, put it in a dry three-necked flask, and plug it Stopper, add diethyl ether and stir, when the volume of diethyl ether is 90 mL, the maleic anhydride is completely dissolved. After the maleic anhydride was completely dissolved, 13 mL of furan was slowly added with a dropping funnel for 13 minutes (minutes, also abbreviated as min). The temperature was controlled to start the reaction at 38°C. After reacting for 1 hour (hour, also abbreviated as h), white solids appeared in the solution, and the longer the time, the more white solids there were. Suction filtration after reacting to 24h, obtains the compou...
Embodiment 2
[0036] Example 2. Camptothecin derivatives 3 preparation of
[0037] .
[0038] 1.2 g (3.48 mmol) camptothecin was dissolved in 20 ml chloroform, followed by adding 5,6-dihydronorcantharidin monomethyl ester 2 (1.38 g, 7 mmol), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (abbreviated as EDCI; 3.2 g, 16.8 mmol) and 4-dimethylaminopyridine ( Abbreviated as DMAP; 240 mg, 2.44 mmol). React at room temperature for 10 hours. Add 30 ml of chloroform, extract and wash with water, saturated sodium carbonate solution, and saturated brine, respectively, and dry the organic layer with anhydrous magnesium sulfate. The solvent is spin-dried and separated by column chromatography to obtain a yellow solid (1.62 g, 89%). 1 HNMR (CDCl 3 ) : δ 8.40(s, 1H), 8.20(d, 1H), 7.94(d, 1H), 7.83(t, 1H), 7.67(t, 1H), 7.19(s, 1H), 6.95(q, 2H ), 5.72(d, 1H), 5.42(d, 1H), 5.29(s, 2H), 3.83(s, 3H), 2.95(s, 2H), 2.88(s, 2H), 2.17-2.38(m, 2H), 1.01 (t, 3H). 13 CNMR (CDCl 3 ) :...
Embodiment 3
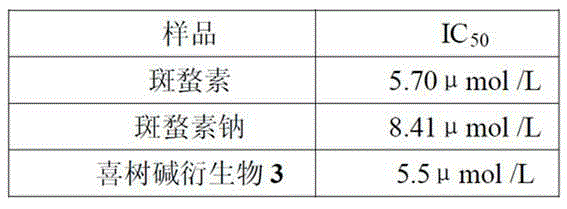
[0039] Embodiment 3, activity test of novel camptothecin derivative
[0040] Using sulforhodamine (sulforhodamine B, SRB) staining.
[0041] When inoculating cells, each cell was inoculated in two 96-well plates in parallel, one was the control plate (T0) and the other was the experimental plate. After culturing in a CO2 incubator for 20 h, the control plate (T0) was taken out, fixed with 50% trichloroacetic acid (TCA), and tested. The test compound was added to the test plate (final concentrations were 5, 2.5, 1.25, 0.625, 0.313 μg·mL-1), and a negative control group (C), an experimental group (T), and a solvent control group were set up. Set up 5 duplicate wells in each group, take out the culture plate after continuing to culture for 48 h, fix it with pre-cooled 50% TCA (final concentration is 10%), put it in a refrigerator at 4 °C for 1 h, rinse it with deionized water, and dry it naturally. Stain with 100 μL of 0.4% SRB, rinse with 0.1% acetic acid after 10 min, dry i...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Ld50 | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap