A pro-antibody conjugated drug targeting tumor cells expressing EGFR and its application
An antibody-conjugated drug, tumor cell technology, used in anti-tumor drugs, drug combinations, anti-receptor/cell surface antigen/cell surface determinant immunoglobulin and other directions, can solve serious side effects and other problems, achieve low toxicity Side effects, high potency effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0044] Embodiment 1, preparation of proantibody conjugated drug
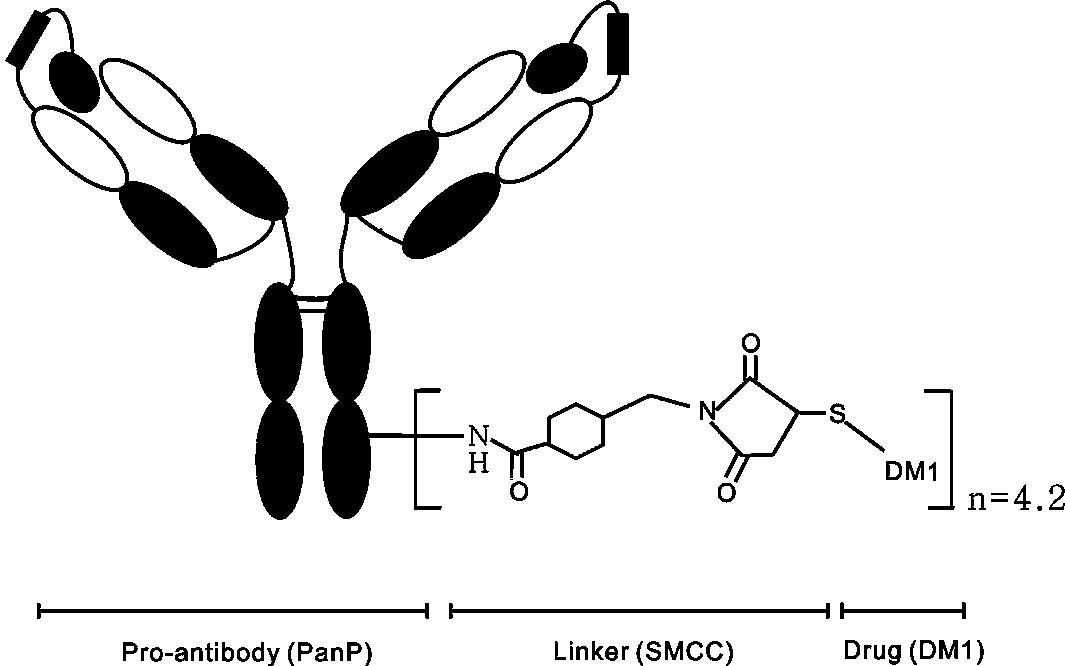
[0045]According to the patent US 2011 / 0003969, PanP-DM1 was prepared by coupling DM1-SMCC with the lysine residue of antibody PanP. The main process is as follows: 5mg / mL antibody was mixed with 6.4mg / mL DM1-SMCC, the reaction buffer was phosphate buffer (50mM, PH=7.5) containing 2mM EDTA and 50mM Nacl, and the reaction time was 2 hours. According to the above method, Pan was also coupled with DM1-SMCC to form Pan-DM1.
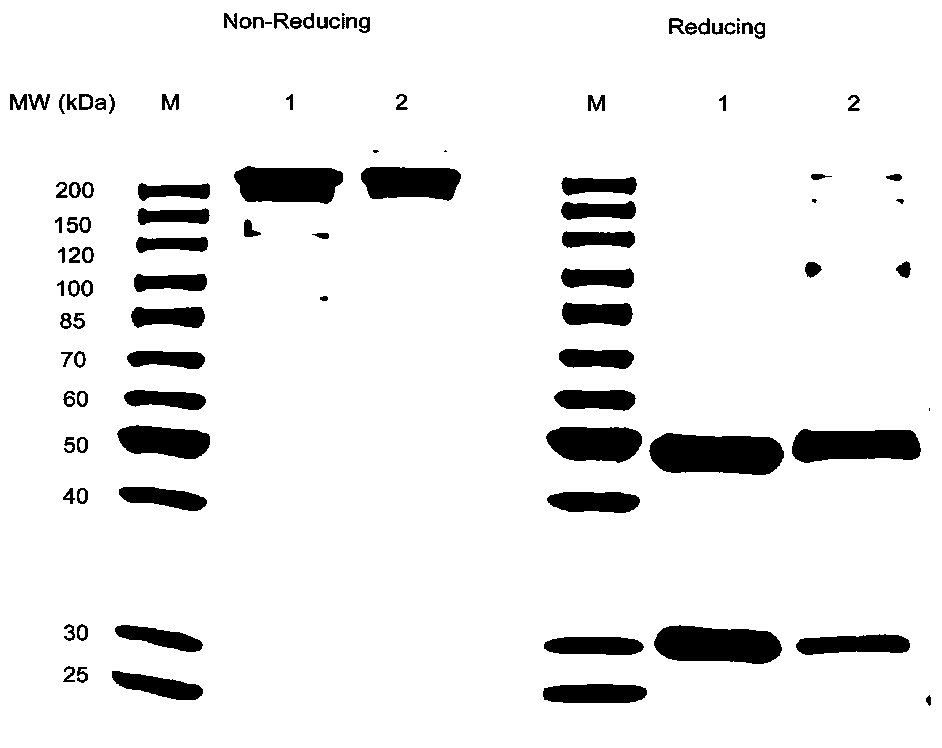
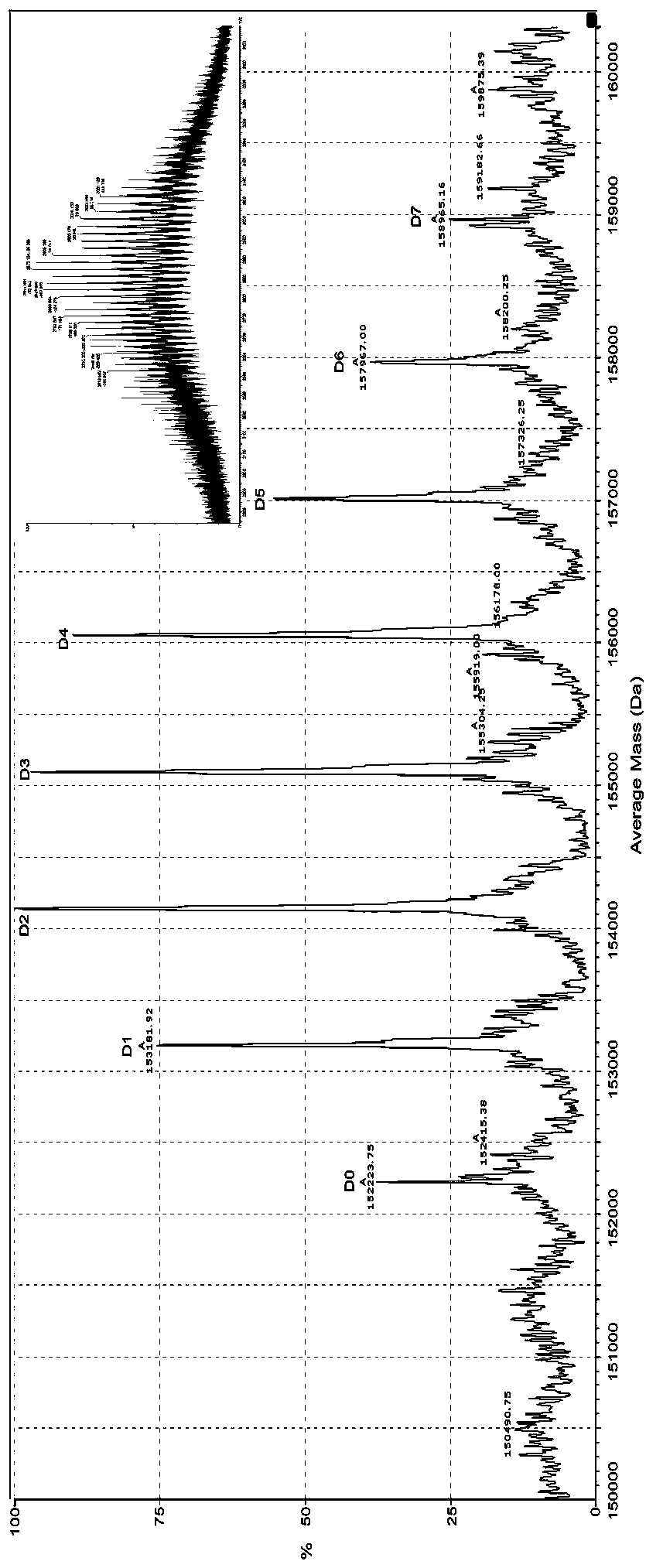
[0046] In addition, the anti-TNFα antibody was coupled with DM1 to prepare a control antibody-conjugated drug. According to the patent US2010 / 0092495, after measuring the absorbance of the antibody-conjugated drug at 252nm and 280nm by ultraviolet spectrophotometry, the coupling ratio of the drug-antibody is calculated. The distribution of drugs was determined by reversed-phase ultra-high performance liquid chromatography-tandem mass spectrometry (RP-UPLC / ESI-MS). Under reducing and non-reduc...
experiment example 1
[0053] Experimental example 1, PDC activity detection
[0054] EGFR binding determined by ELISA and flow cytometry
[0055] EGFR-Fc was coated onto a 96-well plate and blocked with PBS containing 10% skimmed milk powder. After washing, add the indicated concentration of antibody and incubate at room temperature for 1 hour. After washing, add HRP-labeled anti-human F(ab')2, incubate at 37°C for 30min, and finally add TMB substrate solution. Absorbance was read at 450 nm and fitted using a 4 parameter curve.
[0056] When performing flow cytometry analysis, A431 cells overexpressing EGFR were mixed with the antibody to be tested in FACS buffer (PBS buffer containing 1% fetal bovine serum) and incubated for 1 hour. After washing, FITC-labeled anti-human secondary antibody was added and incubated for 30 minutes. Next, wash again with FACS buffer and analyze on BD's FACSCalibur system.
[0057] PanP-DM1 showed similar enzyme activation properties to PanP.
[0058] To further ...
experiment example 2
[0060] Experimental example 2, PDC cytotoxicity test in vitro
[0061] The effect of PanP and PanP-DM1 conjugates on tumor cell viability was evaluated using CCK-8 kit. Spread the cells cultured with 1% calf serum into a 96-well cell culture plate (5000 per well for BT-474, MCF-7 and DiFi cells; 3000 per well for H292 cells; 2000 per well for A431 cells ), cultured overnight to allow it to adhere to the wall. The next day, corresponding concentrations of activated PanP, activated PanP-DM1 or anti-TNFα-DM1 antibody as a control group were added to each well and incubated for different times (48 hours for A431 cells, 72 hours for other cells). Cell viability was determined using CCK-8 kit.
[0062] Calculate the survival cell ratio according to the following formula: [(A450 value of experimental group-A450 value of background group) / (A450 value of untreated group-A450 value of background group)]*100
[0063] Comparing the inhibitory activity of PanP-DM1, activated PanP-DM1 an...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap