Streptococcus pneumoniae capsular polysaccharides and immunogenic conjugate thereof
A Streptococcus pneumoniae, immunogenic technology, applied in the direction of bacterial antigen components, antibacterial drugs, drug combinations, etc., can solve the problem of lack of effective prevention or treatment of serotype infection
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
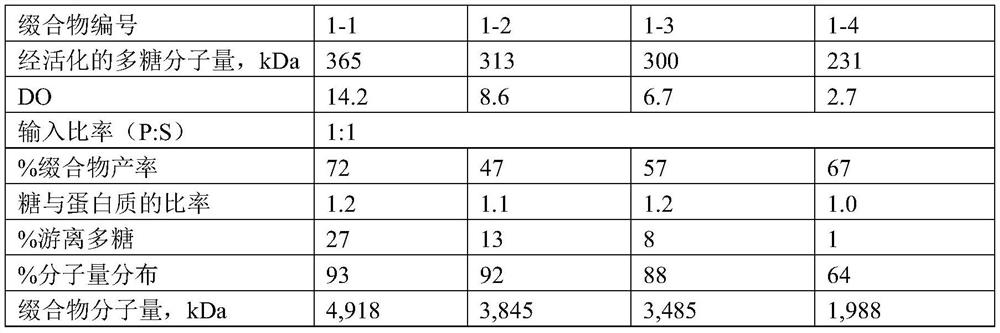
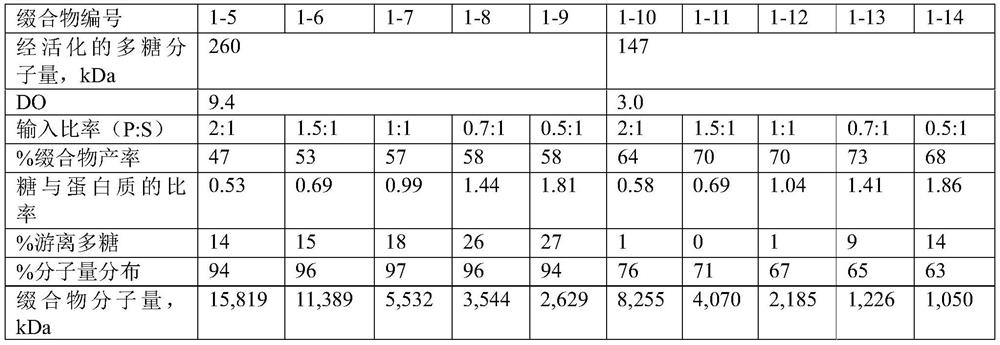
[0309] Example 1. Preparation of polysaccharide-protein conjugate vaccines derived from serotypes 2, 9N, 17F or 20
[0310] [1. Polysaccharide-protein conjugate derived from Streptococcus pneumoniae serotype 2]
[0311] Preparation of polysaccharide-protein conjugates from Streptococcus pneumoniae serotype 2
[0312] Making master cell banks and preparative cell banks
[0313] S. pneumoniae serotype 2 was obtained from the American Type Culture Collection (ATCC) (strain ATCC 6302). The seed stock was grown for several generations in order to strengthen the strain and remove components of animal origin. Seed vials were frozen (<-70°C) using synthetic glycerol as a cryopreservative. To make cell banks, all cultures were propagated in soy-based media. Prior to freezing, cells are concentrated by centrifugation and spent medium is removed, and the cell pellet is then resuspended in new medium containing a cryopreservative (eg, synthetic glycerol).
[0314] fermentation
[03...
Embodiment 2
[0439] Example 2. Preparation of polyvalent Streptococcus pneumoniae polysaccharide-protein conjugates
[0440] [5. Streptococcus pneumoniae 15-valent polysaccharide-protein conjugate]
[0441] Preparation of 15-valent polysaccharide-protein conjugate of Streptococcus pneumoniae
[0442] A formulation comprising polysaccharide-protein conjugates derived from S. pneumoniae serotypes 1, 2, 3, 4, 5, 6A, 6B, 7F, 9N, 9V, 14, 18C, 19A, 19F, and 23F each conjugated to CRM197 alone 15-valent conjugate composition (15vPnC).
[0443] For serotypes 2 and 9N, conjugates were prepared by the method described above, and for other serotypes, conjugates were prepared according to the method disclosed in Korean Patent Application No. 2012-0065893.
[0444] Calculate the desired volume of final bulk concentrate based on the batch volume and bulk sugar concentration. The required amounts of 0.85% Sodium Chloride, Polysorbate 80, and Succinate Buffer were added to pre-labeled formulation conta...
PUM
| Property | Measurement | Unit |
|---|---|---|
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
| molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap