Patents
Literature
49 results about "Streptococcus pneumoniae capsular polysaccharide" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Vaccine against streptococcus pneumoniae capsular polysaccharides
InactiveUS20030147922A1Antibacterial agentsSenses disorderAntigenStreptococcus pneumoniae capsular polysaccharide
The present invention relates to the field of bacterial polysaccharide antigen vaccines. In particular, the present invention relates to specific advantageous pnumococcal polysaccharide conjugates adjuvanted with 3D-MPL and substantially devoid aluminium-based adjuvant.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
Multivalence pneumococcus capsular polysaccharide-protein conjugate composition and preparation method thereof
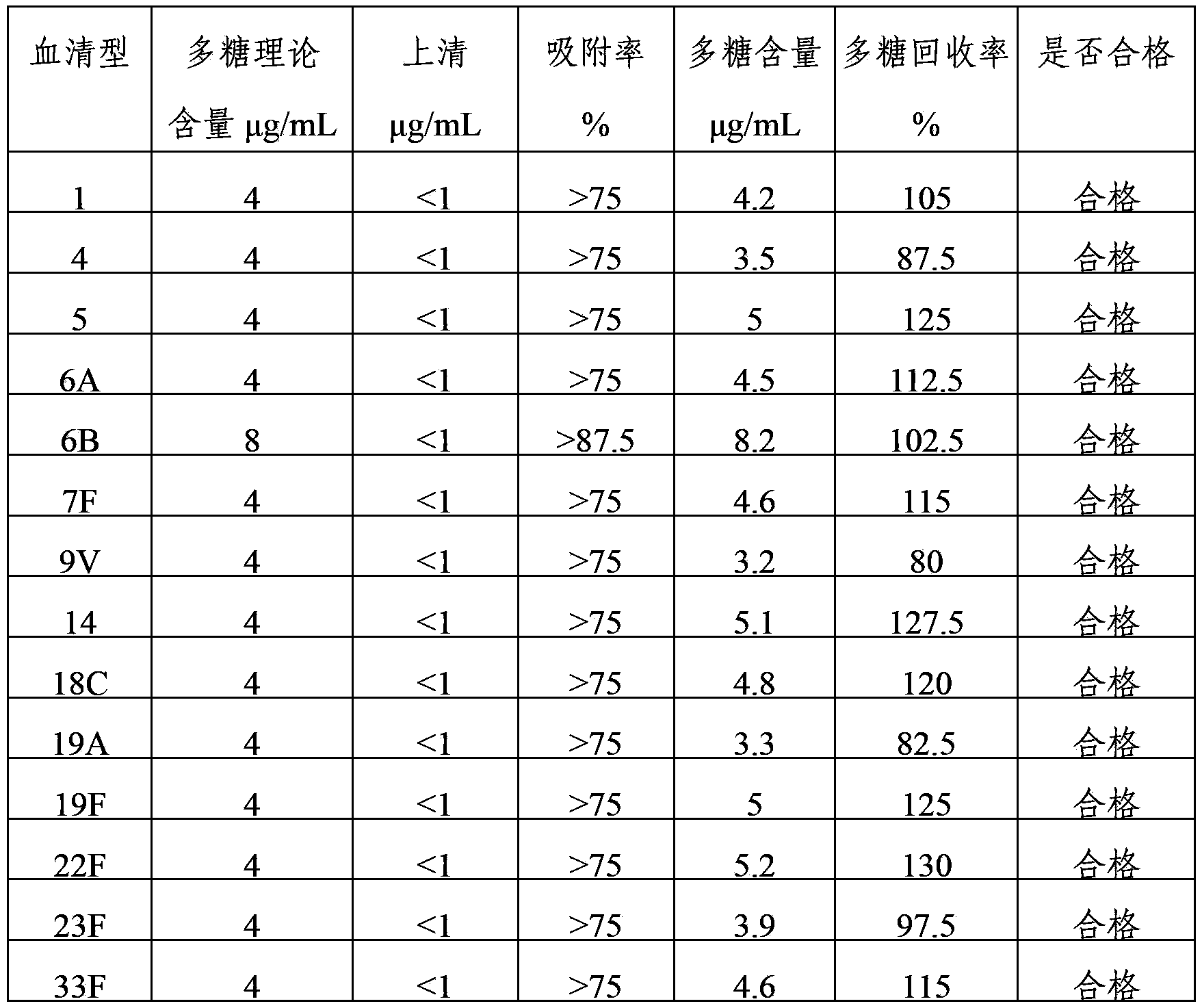
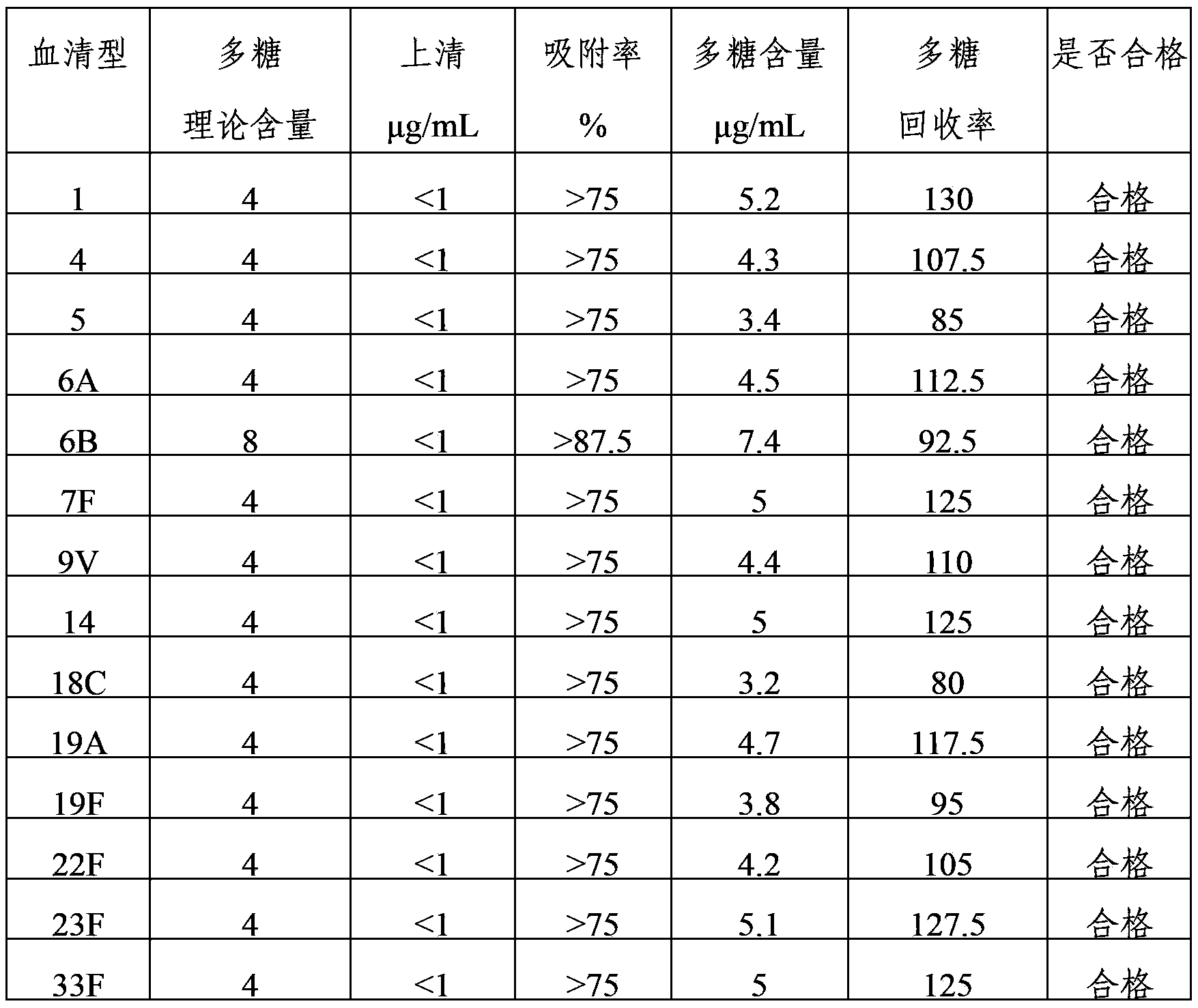
ActiveCN103656631AImprove adsorption capacityImprove stabilityAntibacterial agentsBacterial antigen ingredientsConjugate vaccineDisease
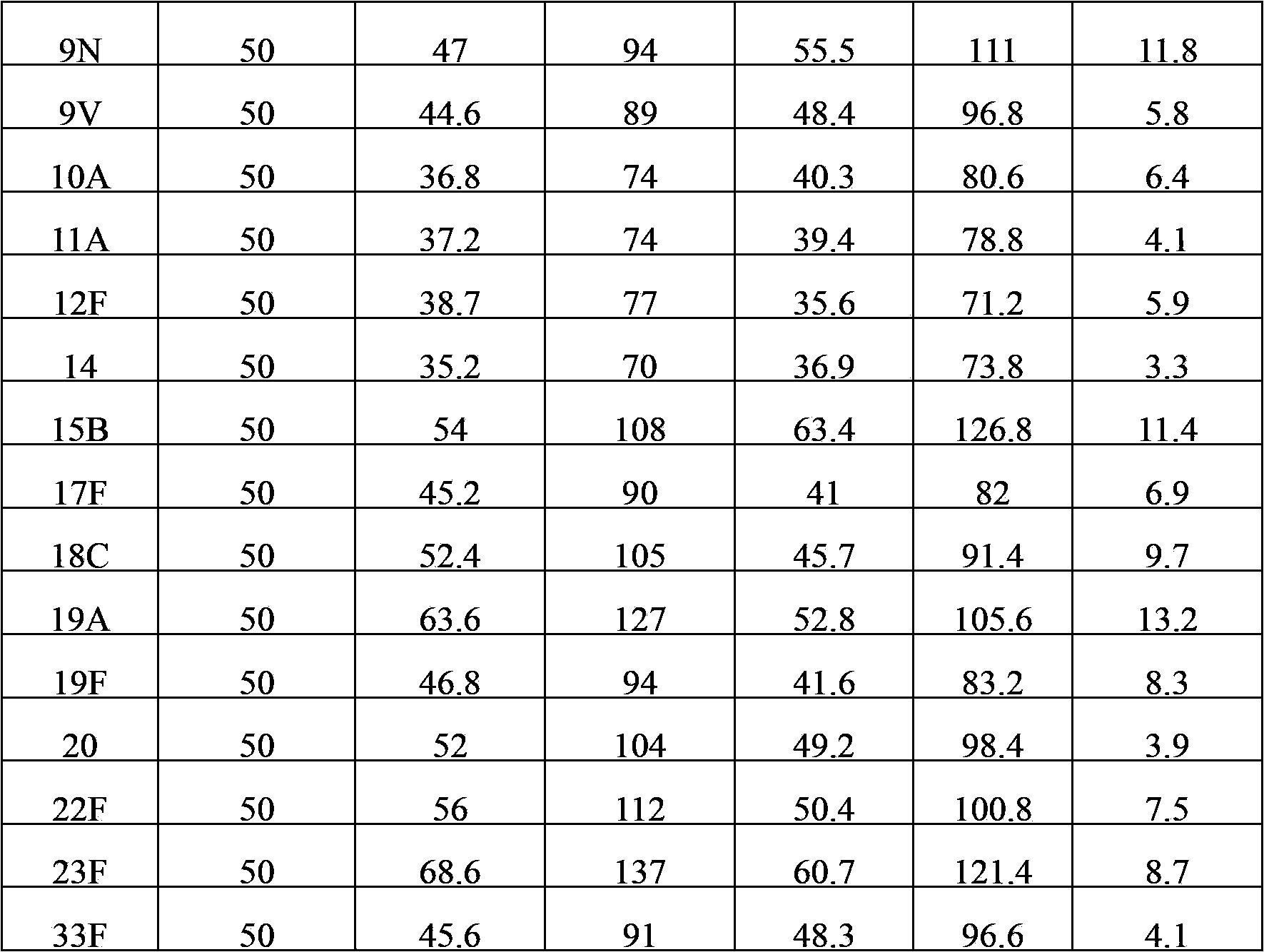
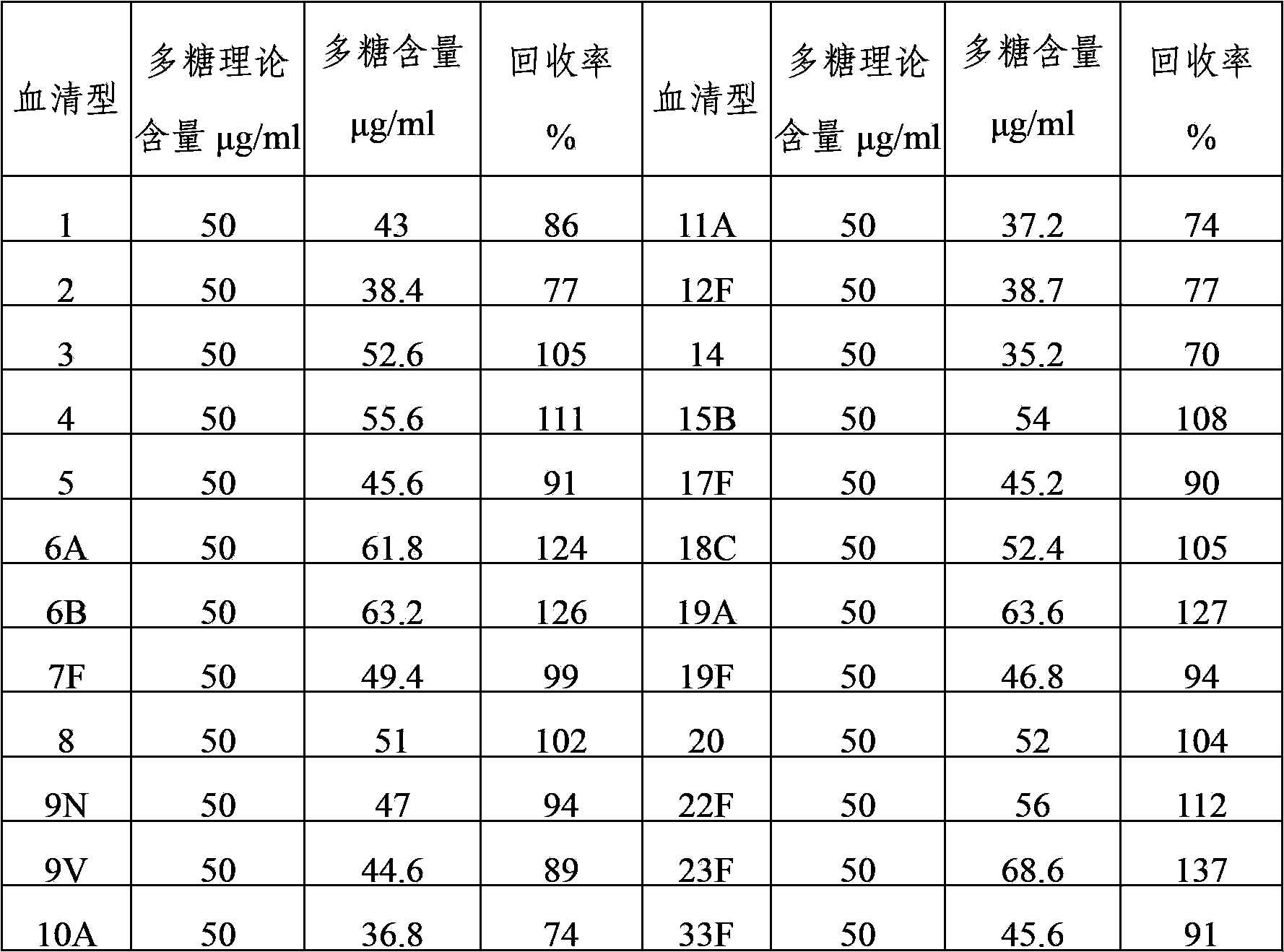
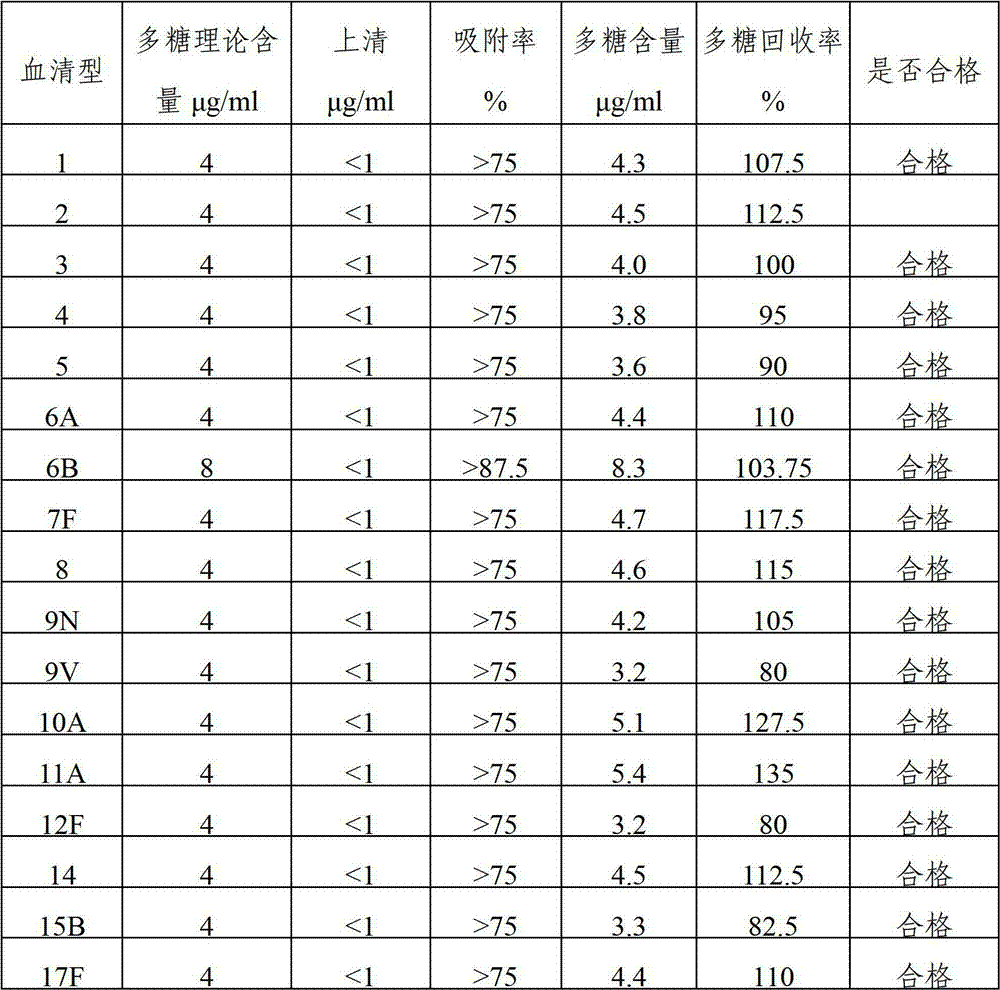
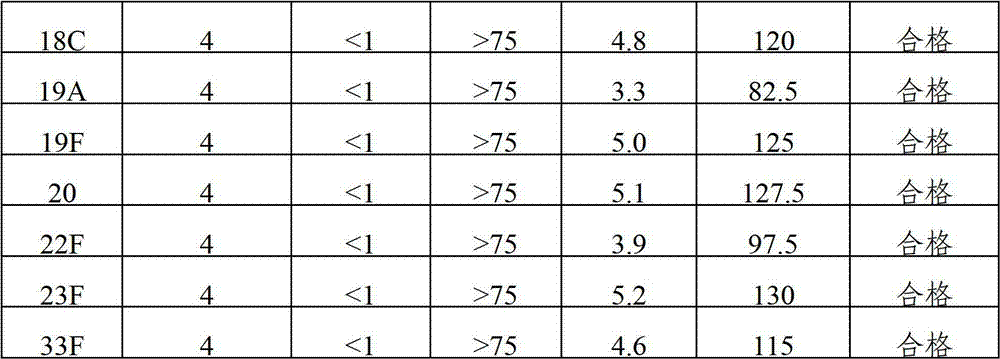
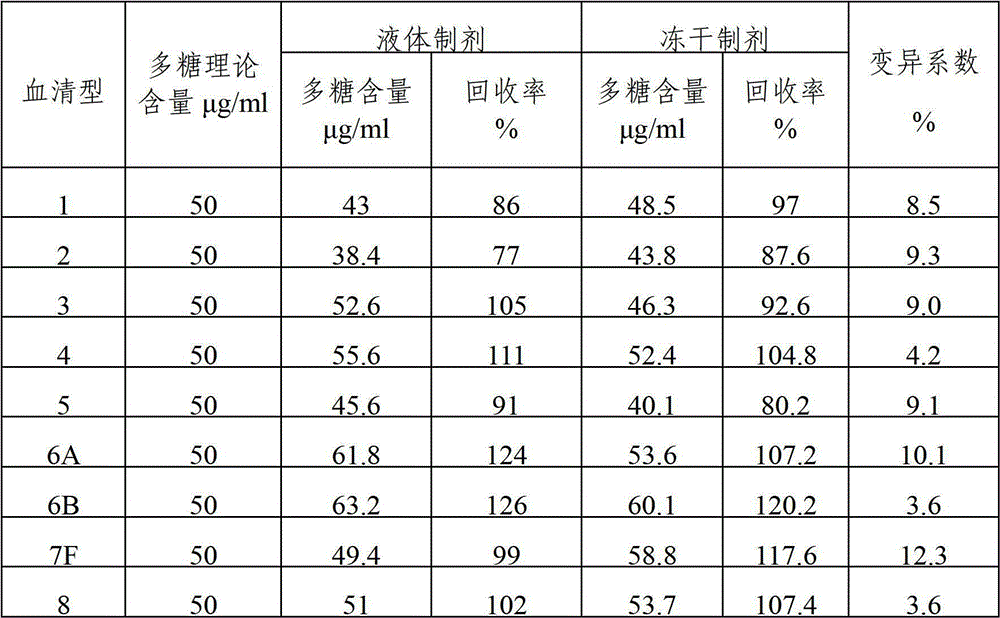
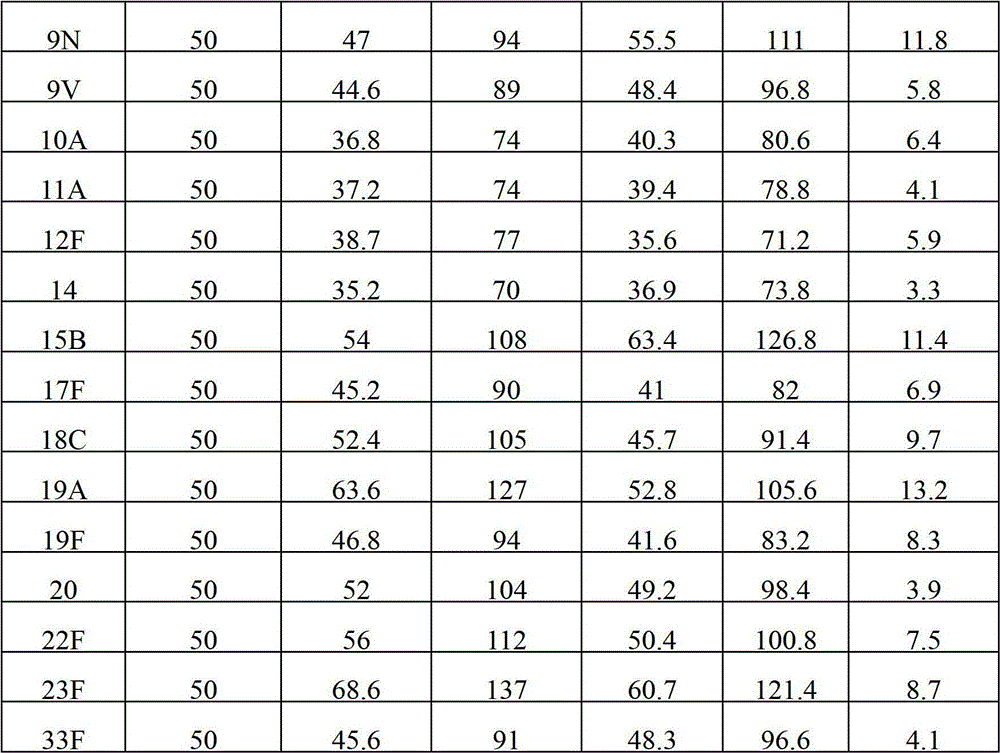
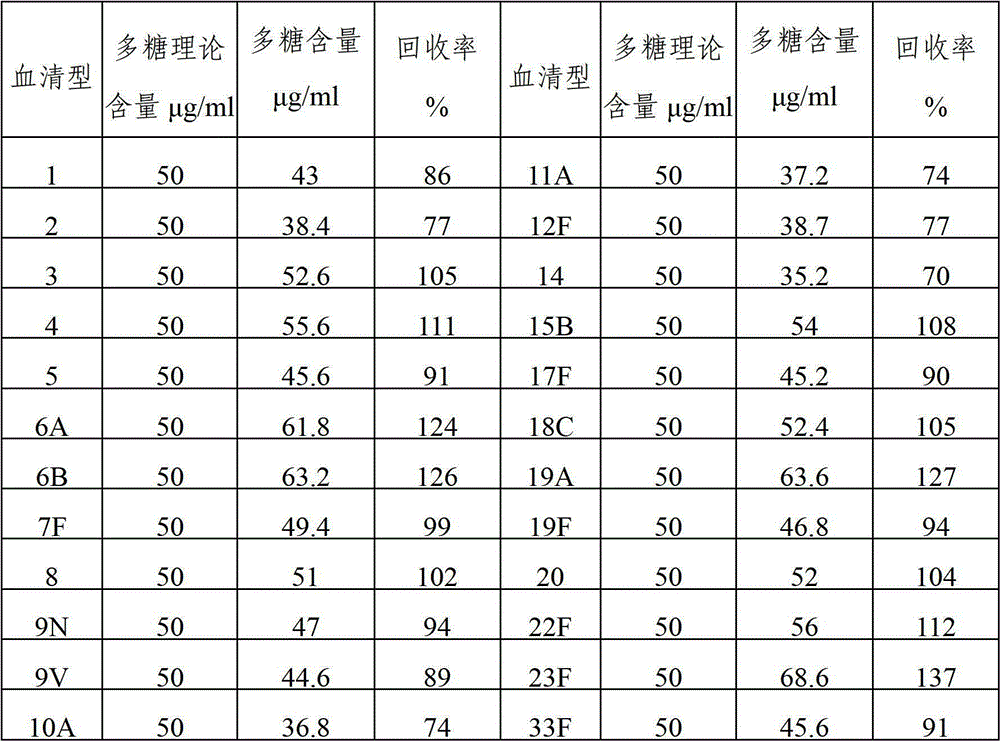
The invention provides a multivalence pneumococcus capsular polysaccharide-protein conjugate composition and a preparation method thereof. The conjugate composition is prepared from capsular polysaccharides of pneumococcus of 24 different serotypes and a carrier protein in a covalence connection manner, wherein the 24 different serotypes are 1, 2, 3, 4, 5, 6A, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F and 33F. The conjugate composition is good in adsorption effect and stability, has multiple immunogenicity and protection properties aiming at invasion of pneumococcus of the 24 serotypes, and is superior to the low-valence pneumonia composition in the market, and the immune response is higher than that of uncombined compositions. By inoculating a multivalence pneumococcus capsular polysaccharide conjugate vaccine prepared from the conjugate composition, the inoculation injection times can be reduced, the immunization procedure can be simplified, and meanwhile human beings and animals can be effectively prevented from diseases resulted from the pneumococcus of the 24 serotypes, and the conjugate composition is wide in coverage range and good in immune effect.
Owner:SINOVAC RES & DEV
Vaccine comprising streptococcus pneumoniae capsular polysaccharide conjugates
InactiveUS20100209450A1Antibacterial agentsSenses disorderConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The present invention is in the field of pneumococcal capsular saccharide conjugate vaccines. Specifically, a multivalent Streptococcus pneumoniae immunogenic composition is provided with various conjugated capsular saccharides from different S. pneumoniae serotypes conjugated to 2 or more different carrier proteins, where the composition comprises serotype 19F capsular saccharide conjugated to diphtheria toxoid. Methods of making and uses thereof are also described.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
Vaccine Comprising Streptococcus Pneumoniae Capsular Polysaccharide Conjugates
InactiveUS20090017059A1Antibacterial agentsSenses disorderStreptococcus pneumoniae capsular polysaccharideStreptococcus mitis
The present invention discloses an immunogenic composition comprising S. pneumoniae capsular saccharide conjugates from serotypes 19A and 19F wherein 19A is conjugated to a first bacterial toxoid and 19F is conjugated to a second bacterial toxoid. Vaccines, methods of making vaccines and uses of the vaccines are also described.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
Vaccine comprising streptococcus pneumoniae capsular polysaccharide conjugates
InactiveUS20100183662A1Antibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The present invention is in the field of pneumococcal capsular saccharide conjugate vaccines. Specifically, an immunogenic composition is provided comprising a multivalent Streptococcus pneumoniae vaccine comprising 13 Streptococcus pneumoniae capsular saccharide conjugates. Methods of making and uses thereof are also described.
Owner:GLAXOSMITHKLINE BIOLOGICALS SA
Multivalent pneumococcal capsular polysaccharide composition as well as preparation method and application thereof
ActiveCN103656632AStable physical and chemical propertiesPrevent diseaseAntibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The invention provides a multivalent pneumococcal capsular polysaccharide composition as well as a preparation method and application thereof. The multivalent pneumococcal capsular polysaccharide composition contains a serotype 6A and at least one extra serotype selected from the group consisting of 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F and 33F. The multivalent pneumococcal capsular polysaccharide composition provided by the invention can be used for inducing an organism to generate humoral immunity, can generate a relatively good protecting effect for infectious diseases caused by the 24 common serotype pneumococcuses and is wide in immunity coverage rate and better in effect as comparison with various existing pneumococcal polysaccharide vaccines and conjugate vaccines sold on the market.
Owner:SINOVAC RES & DEV
Pneumococcal polysaccharide and protein conjugated vaccine and preparation method thereof
ActiveCN103893751AEnhance immune responseImproving immunogenicityAntibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The invention provides a pneumococcal polysaccharide and protein conjugated vaccine and a preparation method thereof. The pneumococcus polysaccharide protein conjugated vaccine comprises one or more streptococcus pneumoniae capsular polysaccharide and protein coupled immune conjugates, and at least one of the immune conjugates is formed by coupling a single streptococcus pneumoniae capsular polysaccharide with two or more proteins. Compared with the existing pneumococcal conjugated vaccine, the pneumococcal polysaccharide and protein conjugated vaccine has stronger immunogenicity, and can cause immune response in a wider crowd; meanwhile, the two carrier proteins are covalently coupled through the polysaccharide, the protective protein antigen epitope induces higher immune response compared with the mixed injection of the two proteins, and through mutual synergy, the immunogenicity of the carrier protein is further improved and the immune response of the body to the polysaccharide is improved; the preparation method is simple, and meets the requirement of large-scale industrial production.
Owner:CANSINO BIOLOGICS INC
Pneumo-streptococcal-polysaccharide adventitia jointed vaccine and preparing method
InactiveCN101024079AEasy to manufactureImprove immunityAntibacterial agentsBacterial antigen ingredientsAntigenDisease
The present invention relates to a streptococcus pneumoriae polysaccharide-outer membrane protein combined vaccine and its preparation method, belonging to the field of streptococcus pneumoriae vaccine. The main antigen component of said vaccine is a streptococcus pneumoriae capsular polysaccharide-outer membrane protein combined product obtained by covalently connecting the capsular polysaccharide produced by streptococcus pneumoriae with its outer membrane protein. Said capsular polysaccharide is the capsular polysaccharide of one or several kinds of streptococcus pneumoriae, its molecular weight is about 200-500 KDa, every polysaccharide molecule has about 300-700 repeating units. The outer membrane protein is the outer membrane protein of one or several kinds of streptococcus pneumoriae, its molecular weight is about 30-100 Kda. Said vaccine can be used for preventing or curing the diseases induced by streptococcus pneumoriae.
Owner:CHANGHUI BIOLOGICAL ENG FUZHOU
Multivalent pneumococcus capsular polysaccharide-protein conjugated composition and preparation method thereof
InactiveCN103623401AImprove adsorption capacityImprove stabilityAntibacterial agentsBacterial antigen ingredientsConjugate vaccineDisease
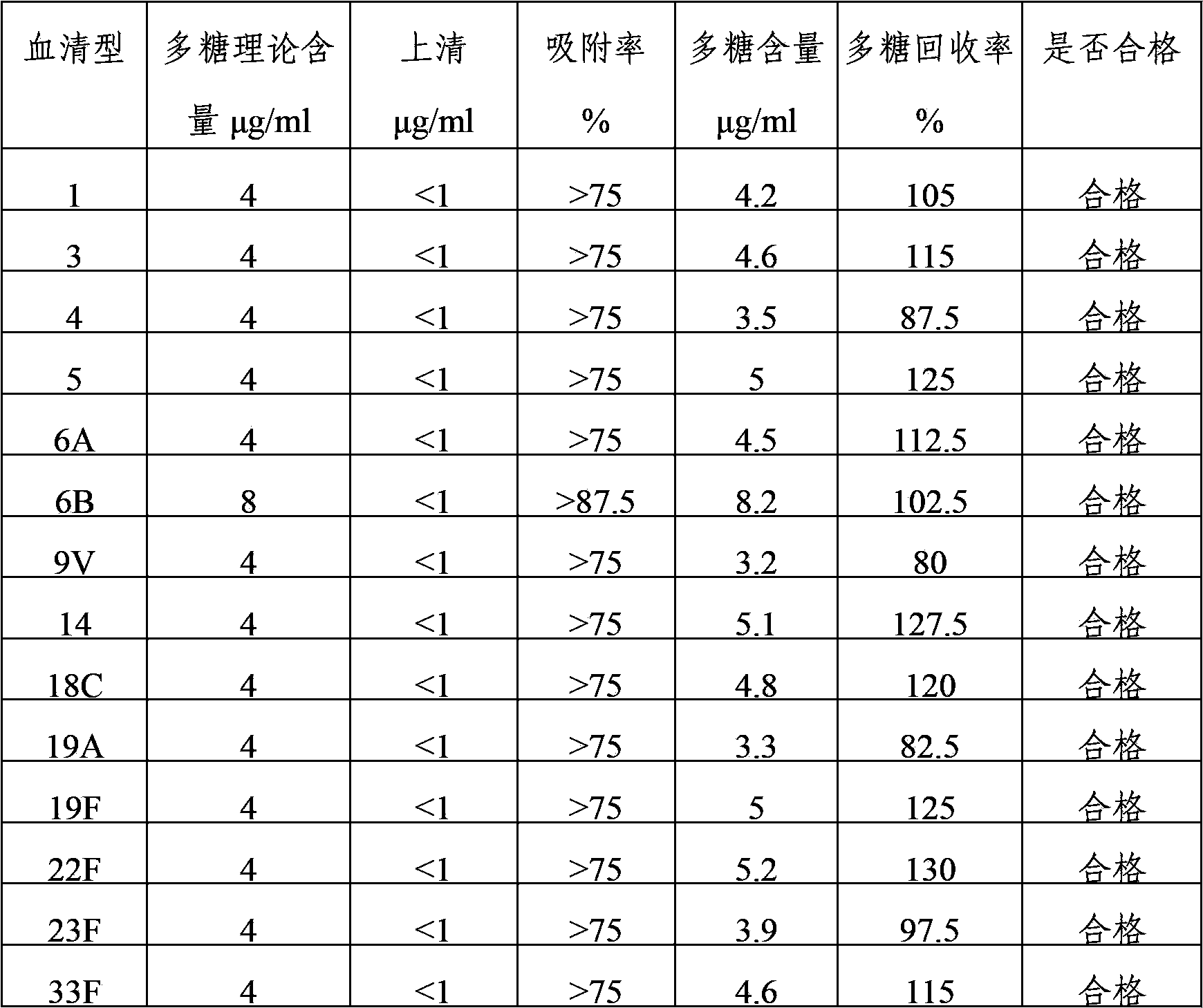
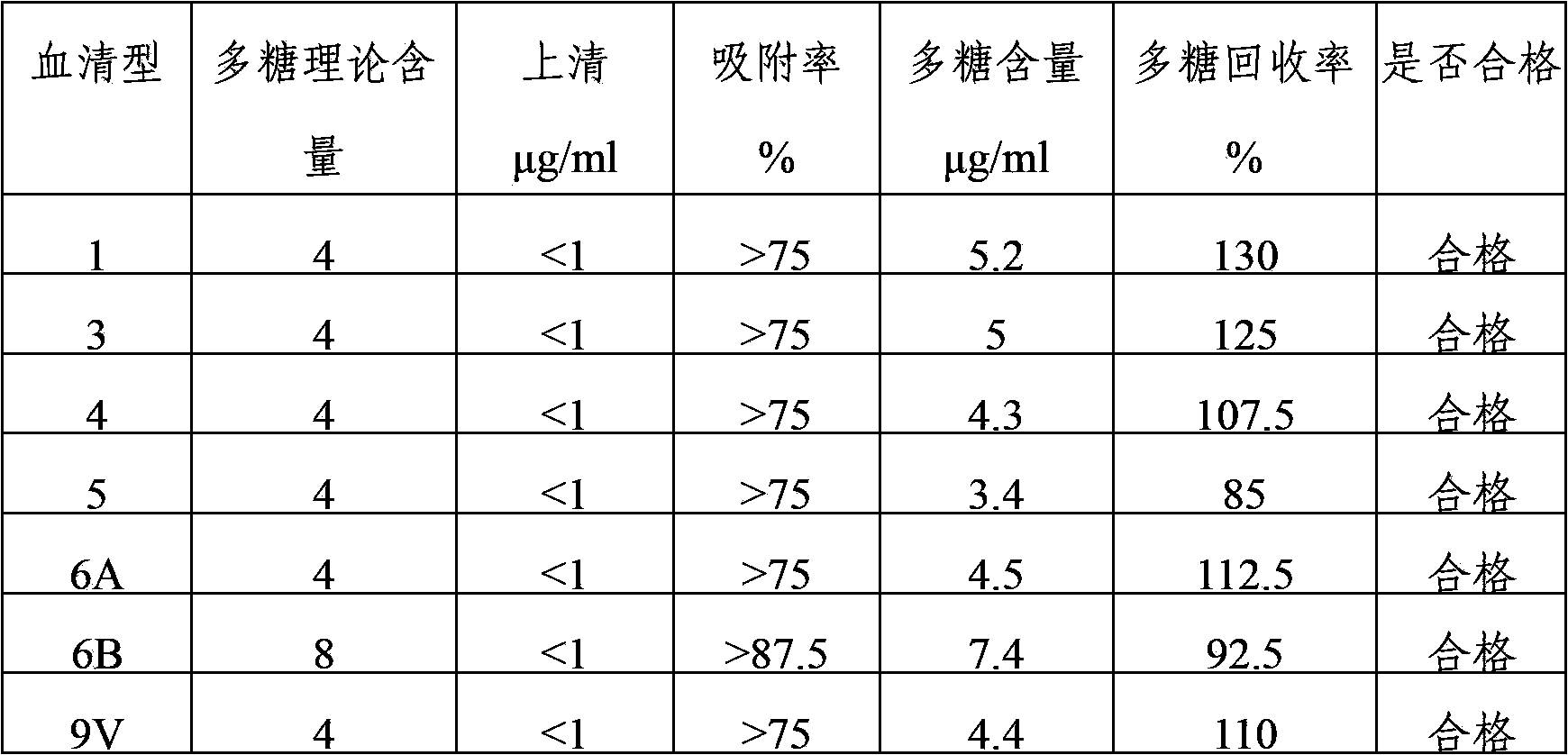
The invention provides a multivalent pneumococcus capsular polysaccharide-protein conjugated composition and a preparation method thereof. The conjugated composition is formed through covalent linkages between pneumococcus capsular polysaccharides of 14 different serotypes and a carrier protein, wherein the 14 different serotypes are: 1, 3, 4, 5, 6A, 6B, 9V, 14, 18C, 19A, 19F, 22F, 23F, and 33F. The conjugated composition has a good absorption effect and stability, and has multiple immunogenicity and protective property aiming at the invasion of pneumococcus of the 14 serotypes; the effect of the conjugated composition is better than that of the low price and low quality pneumonia-treating compositions on the market, and moreover the immune response of the conjugated composition is higher than that of the composition that has not been combined. By using the multivalent pneumococcus capsular polysaccharide conjugate vaccine containing the conjugated composition, the inoculation times can be reduced, the immune procedure is simplified, diseases of humans and animals caused by the pneumococcus of the 14 serotypes can be effectively prevented, moreover the disease coverage is wider, and the immune effect is better.
Owner:SINOVAC RES & DEV
Streptococcus pneumoniae capsular polysaccharides and conjugates thereof
InactiveUS20160324949A1Antibacterial agentsOrganic active ingredientsStreptococcus pneumoniae capsular polysaccharideCarrier protein
The invention relates to isolated Streptococcus pneumoniae serotype 15B capsular polysaccharide and processes for their preparation. The invention also relates to immunogenic conjugates comprising Streptococcus pneumoniae serotype 15B capsular polysaccharide covalently linked to a carrier protein, processes for their preparation and immunogenic compositions comprising them.
Owner:PFIZER INC
Multivalent pneumococcus capsular polysaccharide-protein conjugated composition and preparation method thereof
InactiveCN104069488AImprove adsorption capacityImprove stabilityAntibacterial agentsBacterial antigen ingredientsDiseaseConjugate vaccine
The invention provides a multivalent pneumococcus capsular polysaccharide-protein conjugated composition and a preparation method thereof. The conjugated composition is formed by covalent linkage of multivalent pneumococcus capsular polysaccharides of 14 different serotypes and carrier protein, wherein the 14 serotypes include 1, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 22F, 23F and 33F. The conjugated composition has good adsorption effect and good stability, has multiple immunogenicity and protective performance against invasion of the pneumococcus of 14 serotypes, is superior to on-sale low-valent pneumonia compositions, and the immune response of the conjugated composition disclosed by the invention is higher than that of an uncombined composition. The inoculating injection frequency can be reduced by using the multivalent pneumococcus capsular polysaccharide conjugate vaccine containing the conjugated composition, the immune process can be simplified, and diseases of human and animals caused by the 14 serotypes of pneumococcal bacteria can be effectively prevented. The conjugated composition has wider coverage and better immune effect.
Owner:SINOVAC RES & DEV
Preparation method of streptococcus pneumoniae capsular polysaccharide protein conjugate vaccine
InactiveCN103830723AIncrease coverageImprove cross protectionAntibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The invention discloses a preparation method of a streptococcus pneumoniae capsular polysaccharide protein conjugate vaccine, which comprises the following steps: degrading extracted streptococcus pneumoniae capsular polysaccharide; and conjugating the degraded streptococcus pneumoniae capsular polysaccharide with a carrier protein, and preparing the vaccine preparation.
Owner:TIANJIN TASLY PHARMA CO LTD
Vaccine compositions
InactiveUS8226959B2Organic active ingredientsBiocideAntigenStreptococcus pneumoniae capsular polysaccharide
Owner:NEWCASTE INNOVATION LTD
Vaccine compositions
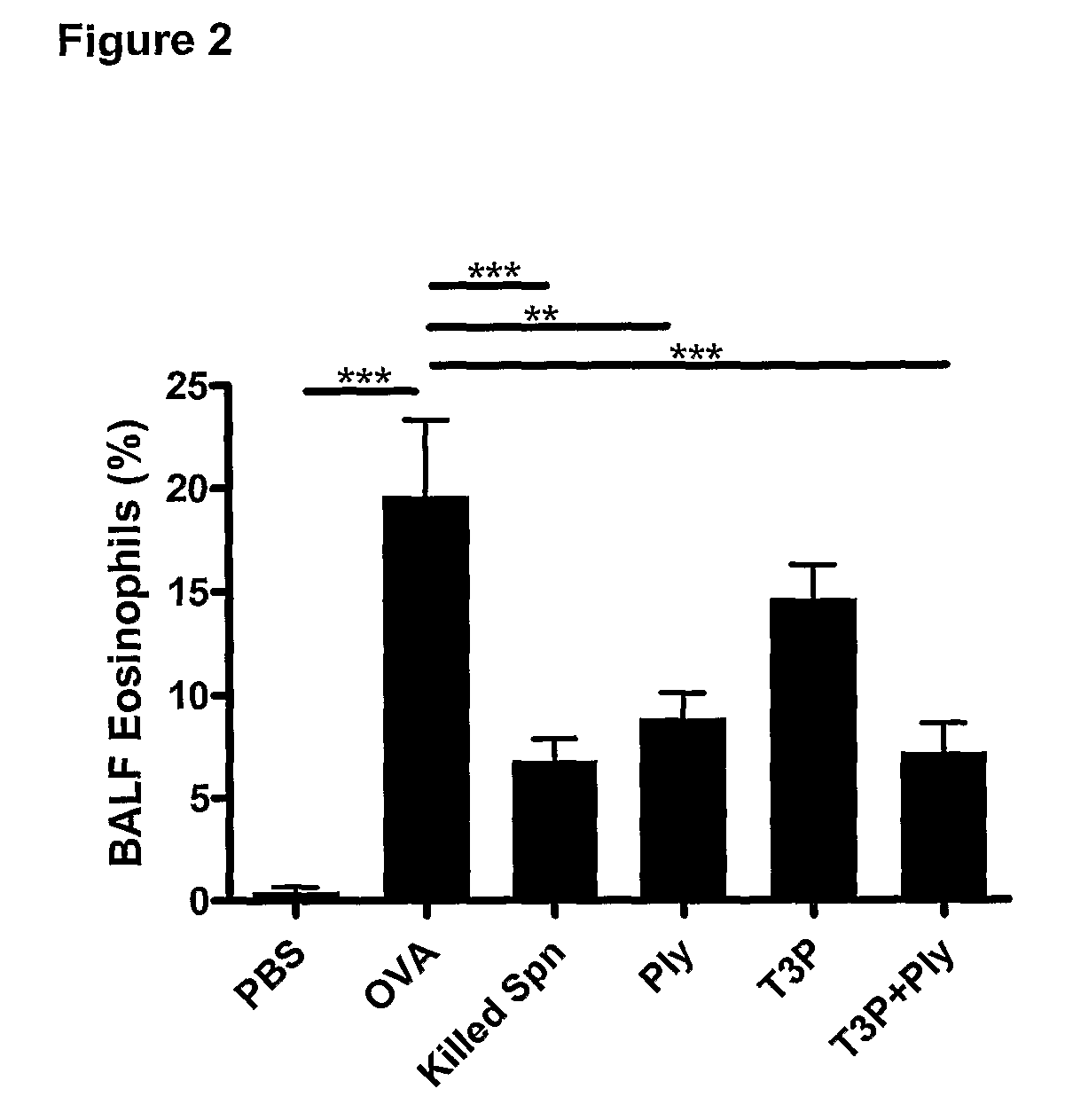
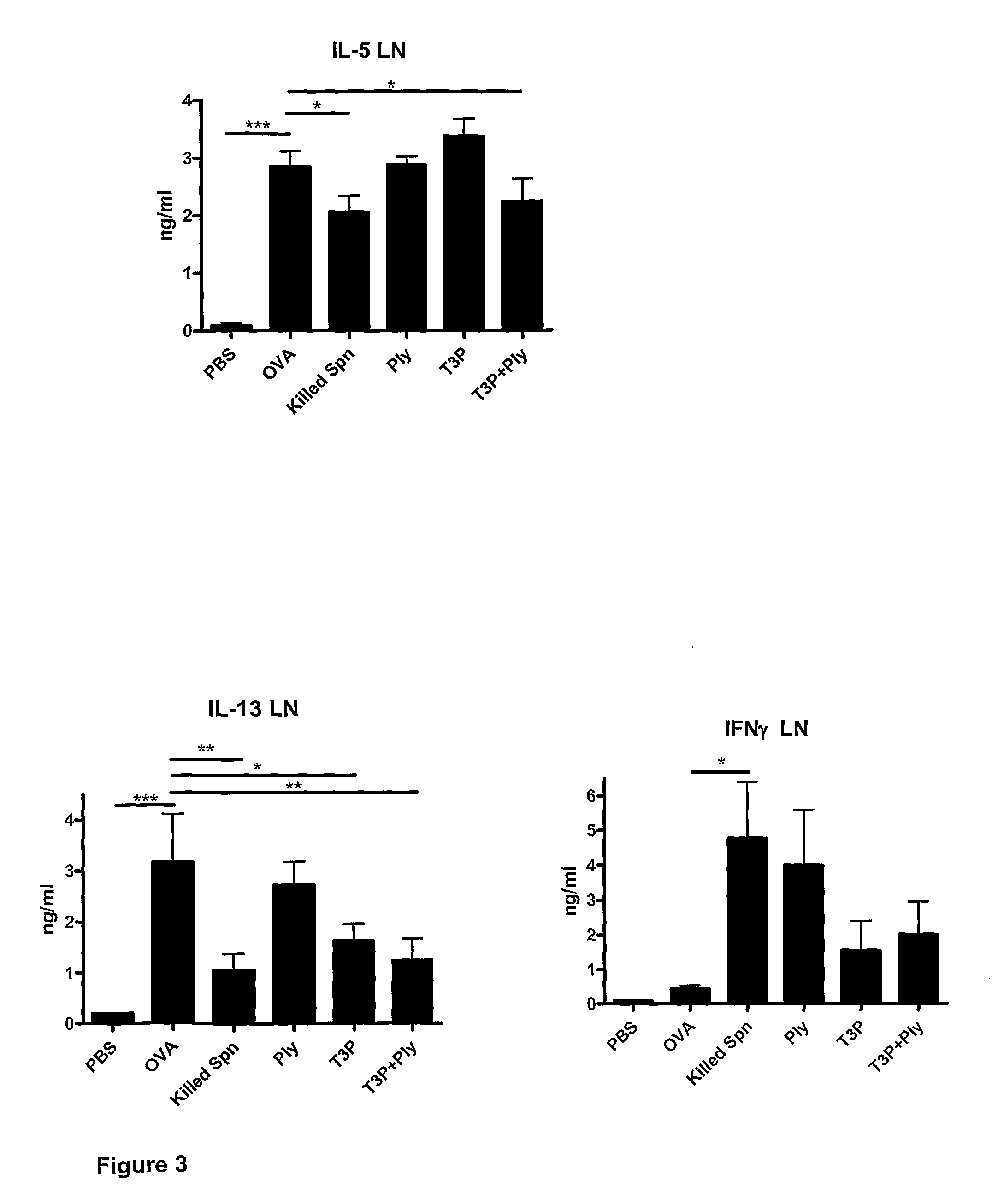
The present invention provides methods for the treatment or prevention of allergic airways diseases, the suppression of allergic immune responses, and the induction protective immunity against allergic airways diseases wherein the methods comprise administering to subjects in need thereof an effective amount of a Streptococcus pneumoniae capsular polysaccharide and a Streptococcus pneumoniae exotoxin or exotoxoid, optionally together with one or more additional antigenic or immunomodulatory constituents, components or fractions of Streptococcus pneumoniae and / or immunopotentiators. Administration of individual components is also contemplated. Also provided are vaccine compositions suitable for use in accordance with methods disclosed herein.
Owner:NEWCASTE INNOVATION LTD
Streptococcus pneumoniae capsular polysaccharides and conjugates thereof
InactiveUS20190070283A1Antibacterial agentsOrganic active ingredientsStreptococcus pneumoniae capsular polysaccharideStreptococcus mitis
The invention relates to isolated Streptococcus pneumoniae serotype 15B capsular polysaccharide and processes for their preparation. The invention also relates to immunogenic conjugates comprising Streptococcus pneumoniae serotype 15B capsular polysaccharide covalently linked to a carrier protein, processes for their preparation and immunogenic compositions comprising them.
Owner:PFIZER INC
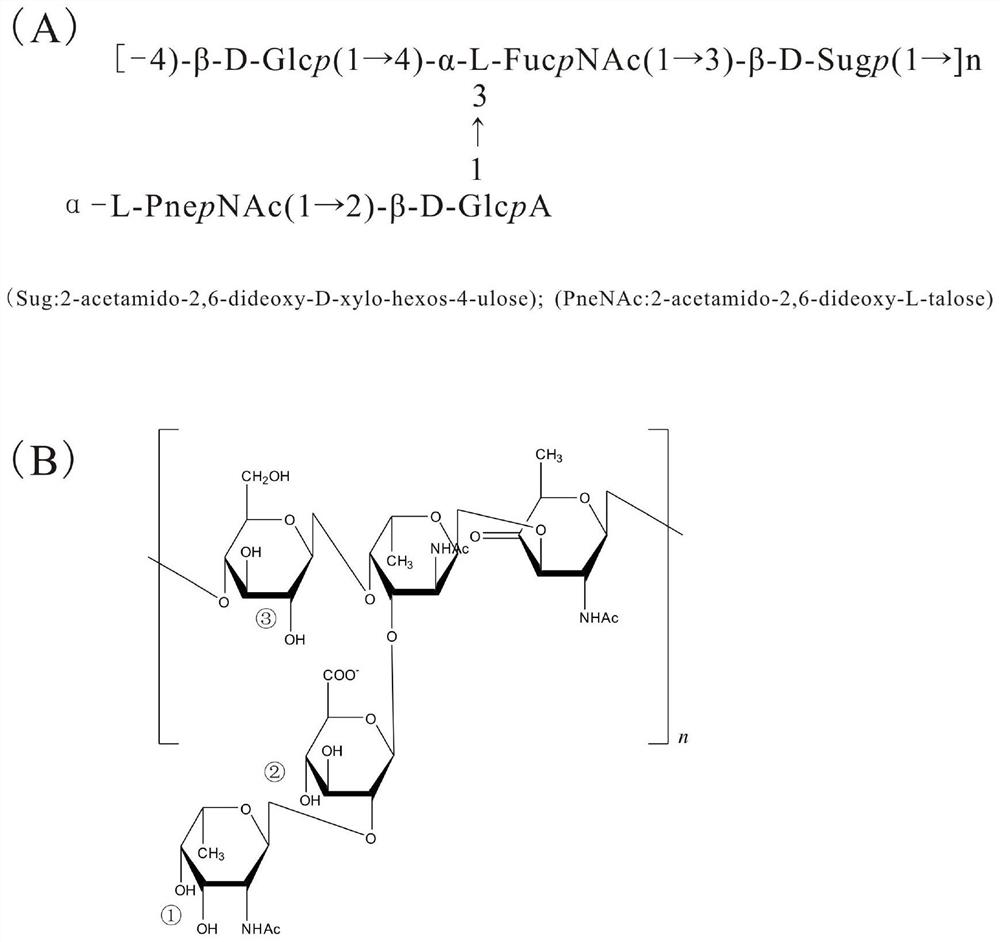
Oligosaccharide conjugate based on Streptococcus pneumoniae type 3 capsular polysaccharide, preparation method and applications thereof
ActiveCN107224577APromote maturityAntibacterial agentsBacterial antigen ingredientsStreptococcus pneumoniae capsular polysaccharideStreptococcus mitis
The present invention relates to an oligosaccharide conjugate based on Streptococcus pneumoniae type 3 capsular polysaccharide, a preparation method and applications thereof, wherein the structure general formula of the oligosaccharide conjugate is defined in the specification. According to the present invention, the system of the Streptococcus pneumoniae type 3 capsular polysaccharide related oligosaccharide derivative is firstly discloses, and can be used for preparing a novel oligosaccharide-protein binding vaccine against Streptococcus pneumoniae type 3.
Owner:SHANDONG UNIV
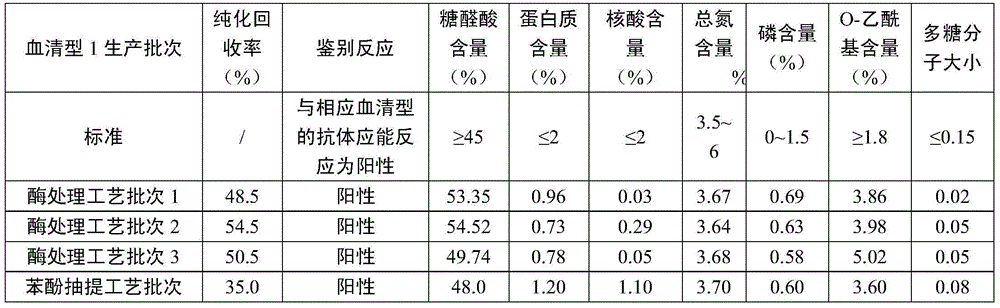
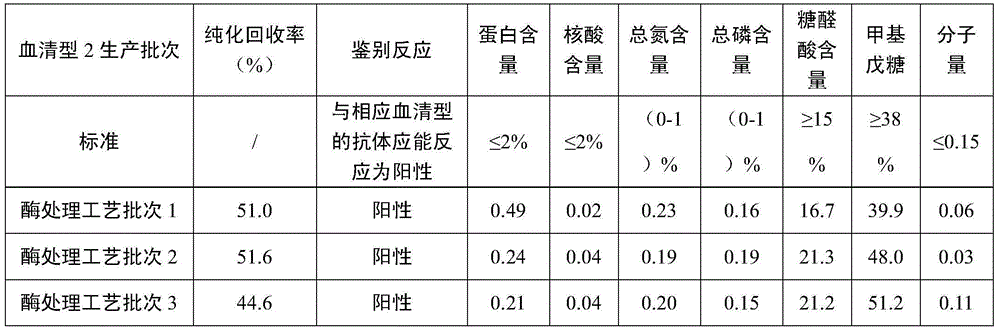
Method for purifying pneumococcal capsular polysaccharide
InactiveCN104530250AEasy to degradeReduce the impactSynechococcusStreptococcus pneumoniae capsular polysaccharide
The invention relates to a method for purifying pneumococcal capsular polysaccharide and belongs to the field of biological products. The method for purifying pneumococcal capsular polysaccharide comprises the following steps: cracking bacteria of a streptococcus pneumoniae inactivated and fermented liquid, carrying out clarification and ultra-filtration to obtain ultra-filtration concentration liquid, precipitating by using low alcohols and carrying out ultra-filtration, degrading protein and nucleic acid into micromolecule fragments through enzymic catalytic reaction under the specific reaction condition by using protease and nuclease, further finely purifying after purifications processes of ultra-filtration and chromatography in subsequent processes to obtain purified pneumococcal capsular polysaccharide. The method is mild in reaction condition, simple and convenient to operate; the process is liable to control; the defects of the prior art can be overcome; poisonous and harmful chemical reagents such as phenols, chloroform and acetone are not used; the method is environmentally friendly and friendly to operator; the prepared capsular polysaccharide is high in recovery rate while the content of protein and nucleic acid is reduced.
Owner:SINOVAC BIOTECH
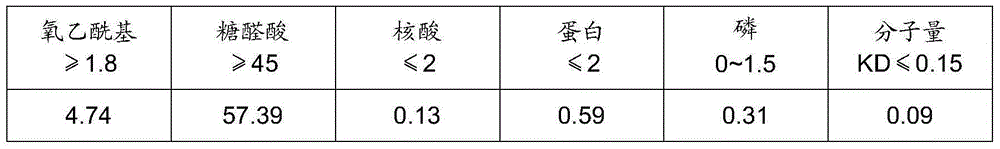
Purification method of streptococcus pneumoniae capsular polysaccharide
ActiveCN105131139AReduce usageEliminate potential safety hazardsPurification methodsStreptococcus pneumoniae capsular polysaccharide
The invention relates to a purification method of streptococcus pneumoniae capsular polysaccharide. The purification method comprises the following steps: subjecting streptococcus pneumoniae lysate to ultrafiltration and condensation, adding sodium deoxycholate, adjusting the pH to 4-5, carrying out a centrifugal treatment, collecting the supernate, adding nuclease into the supernate, carrying out reactions for 3-8 hours to obtain acidified lysate, adding sodium deoxycholate into the acidified lysate, subjecting acidified lysate to ultrafiltration, performing acidification and centrifugation, collecting the supernate, and finally subjecting the supernate to ultrafiltration and condensation to obtain a purified polysaccharide solution. In the provided purification method, phenol is not used; after ethanol drying, the obtained purified streptococcus pneumoniae capsular polysaccharide is the cell lysate after one nuclease treatment, the product is more environment-friendly and safer, the process time is short, the production cost is low, and massive production can be achieved.
Owner:LANZHOU INST OF BIOLOGICAL PROD
Polyvalent pneumococcal capsular polysaccharide-protein conjugate composition and preparation method thereof
ActiveCN103656631BImprove adsorption capacityImprove stabilityAntibacterial agentsBacterial antigen ingredientsDiseaseImmune effects
The invention provides a multivalence pneumococcus capsular polysaccharide-protein conjugate composition and a preparation method thereof. The conjugate composition is prepared from capsular polysaccharides of pneumococcus of 24 different serotypes and a carrier protein in a covalence connection manner, wherein the 24 different serotypes are 1, 2, 3, 4, 5, 6A, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F and 33F. The conjugate composition is good in adsorption effect and stability, has multiple immunogenicity and protection properties aiming at invasion of pneumococcus of the 24 serotypes, and is superior to the low-valence pneumonia composition in the market, and the immune response is higher than that of uncombined compositions. By inoculating a multivalence pneumococcus capsular polysaccharide conjugate vaccine prepared from the conjugate composition, the inoculation injection times can be reduced, the immunization procedure can be simplified, and meanwhile human beings and animals can be effectively prevented from diseases resulted from the pneumococcus of the 24 serotypes, and the conjugate composition is wide in coverage range and good in immune effect.
Owner:SINOVAC RES & DEV
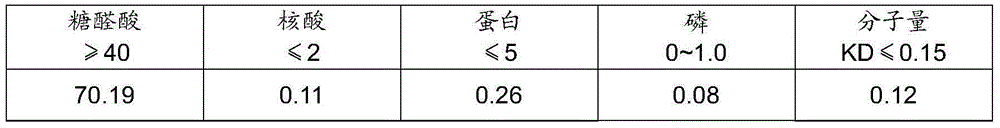
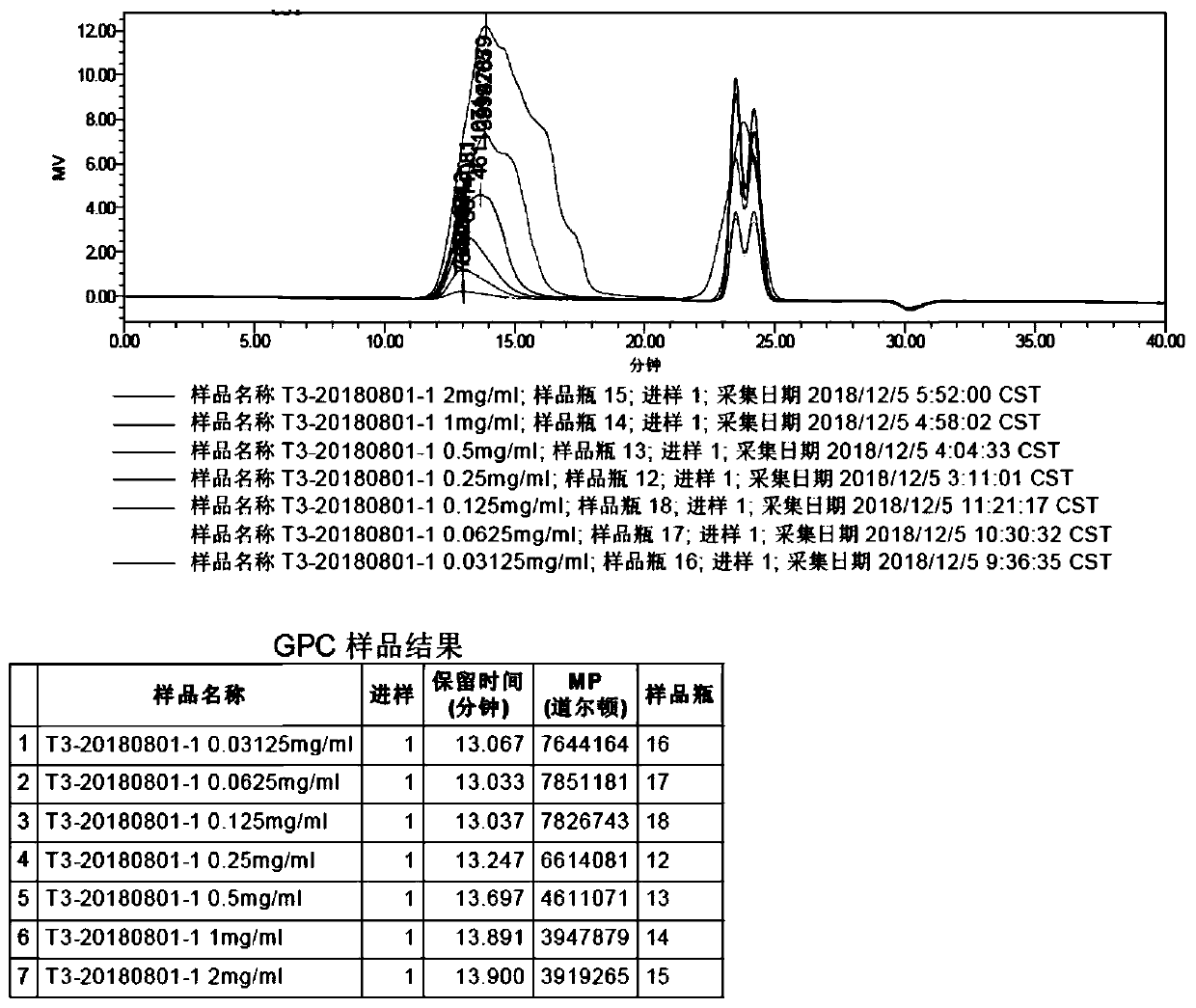
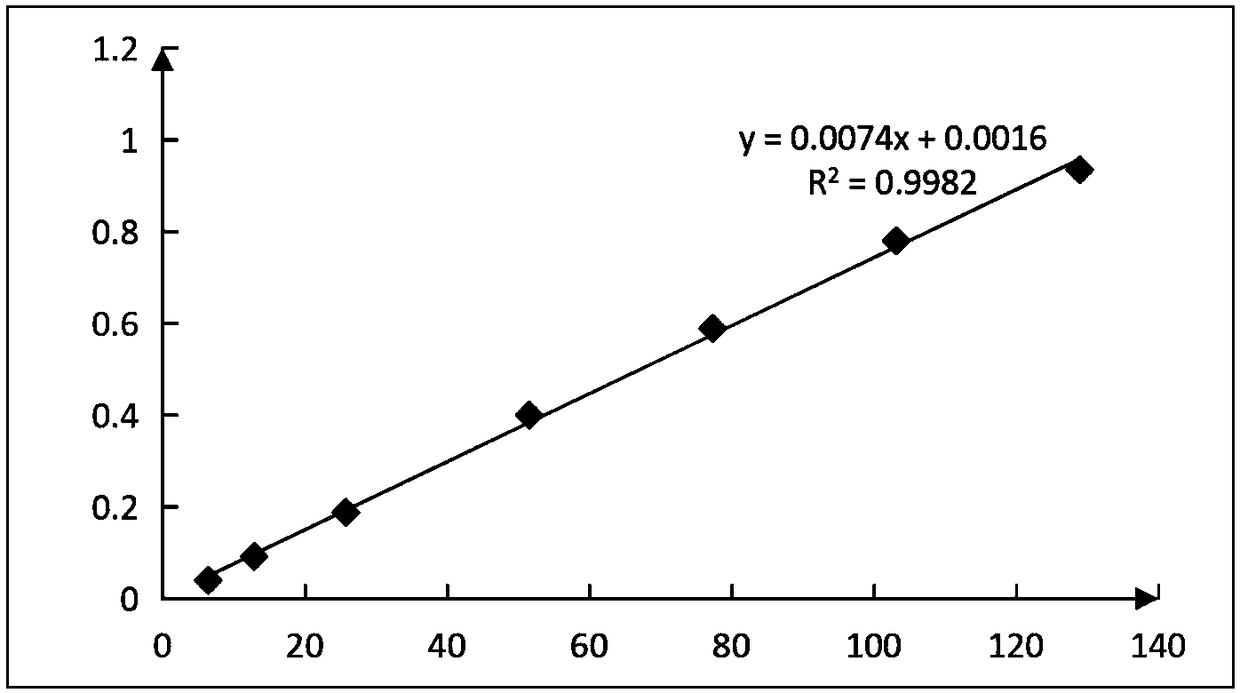
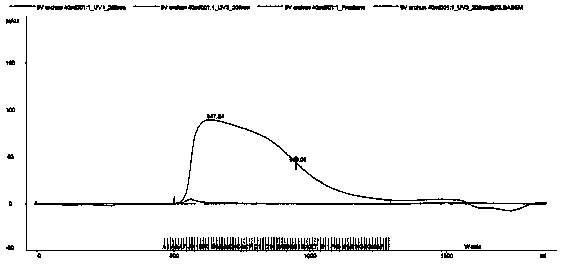
Method of detecting molecular weight of pneumococcal capsular poly-saccharides
ActiveCN110736796AImprove accuracyIncreased Viscosity ControlComponent separationStreptococcus pneumoniae capsular polysaccharideFluid phase
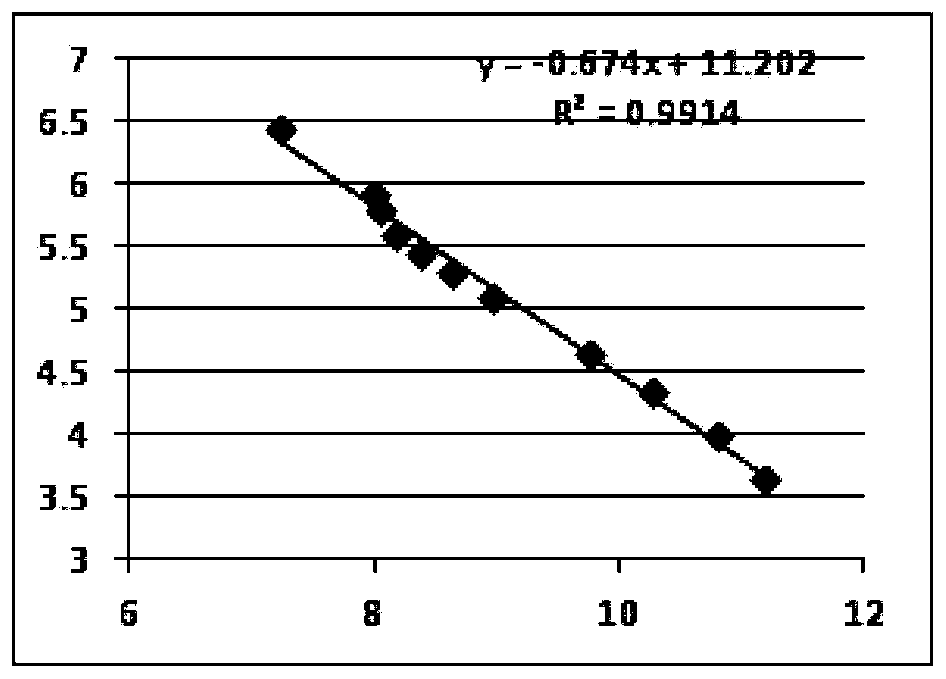
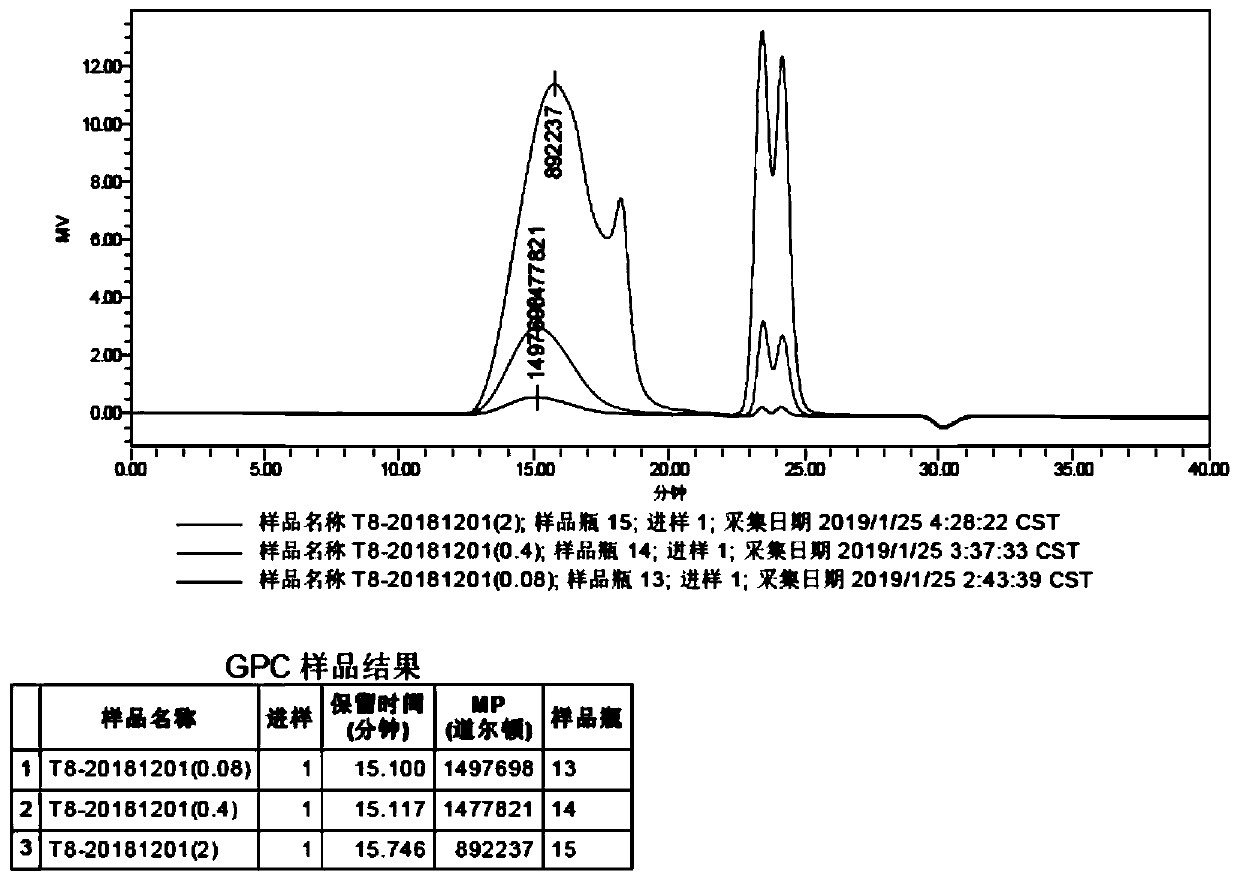
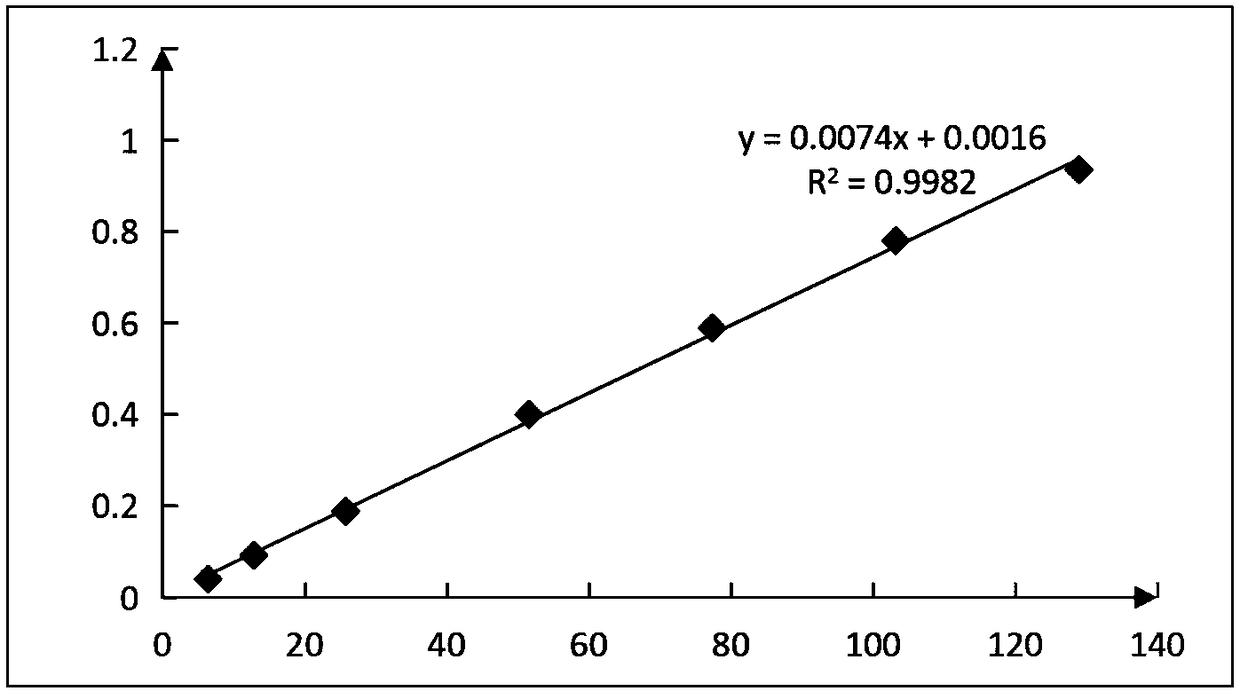
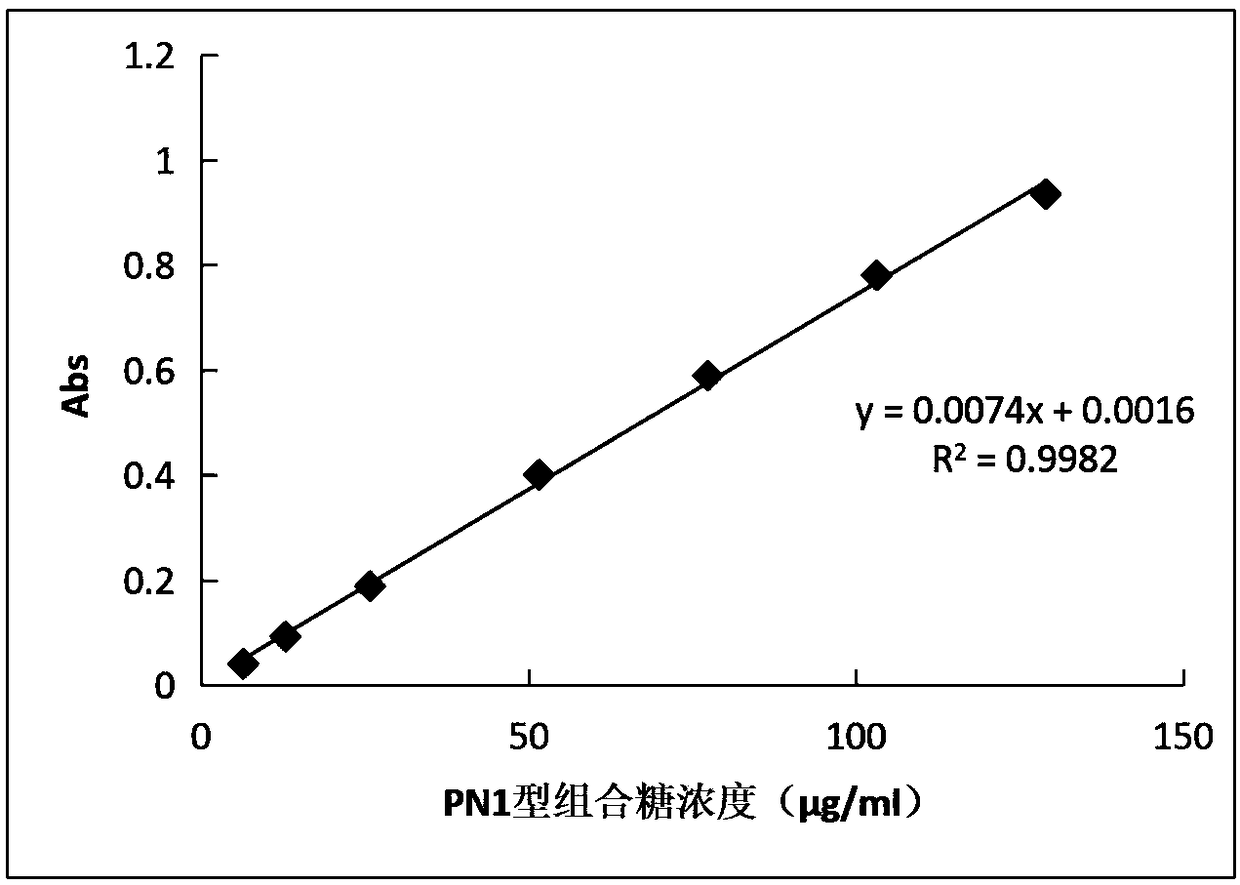
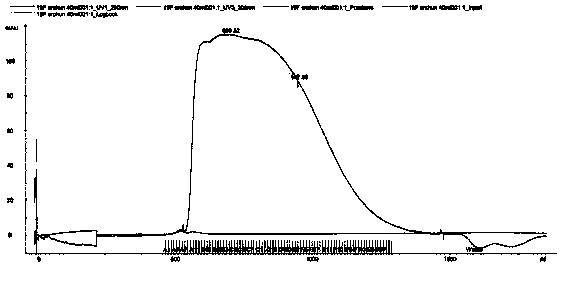
The invention discloses a method of detecting molecular weight of pneumococcal capsular poly-saccharides (PnPs) by an HPSEC-RI method. The method comprises the steps of preparing a mobile phase, precisely weighing the PnPs, adding the mobile phase, performing dissolution, performing dilution by the mobile phase, detecting the viscosity of the PnPs to allow a viscosity value of a diluted sample tobe within a certain range, performing filtering as a test sample solution, selecting a plurality of glucose standard substances with different molecular weight, respectively performing dilution by themobile phase, performing sample injection, performing analysis by liquid chromatography, establishing a molecular weight standard curve and a regression equation, detecting and recording a test sample under the same test condition, and calculating the molecular weight of the sample by applying the established standard curve and the established regression equation. A buffer system and a sample processing method are high in precision and good in repeatability.
Owner:BEIJING ZHIFEI LVZHU BIOPHARM +2
Vaccine containing capsular polysaccharide of type 5 streptococcus pneumoniae and preparation method thereof
PendingCN112741901AAvoid damageReduce lossAntibacterial agentsBacterial antigen ingredientsStreptococcus pneumoniae capsular polysaccharideCarrier protein
The invention provides a vaccine containing capsular polysaccharide of type 5 streptococcus pneumoniae and a preparation method of the vaccine, and belongs to the technical field of biological product preparation. The preparation method provided by the invention comprises the following steps: respectively degrading each serotype capsular polysaccharide at least containing type 5 streptococcus pneumoniae; performing activation; carrying out freeze-drying or precipitation; redissolving or dissolving the activated capsular polysaccharide of streptococcus pneumoniae, and conjugating the activated capsular polysaccharide with carrier protein; and purifying the conjugated vaccine stock solution. The antigen-specific epitope in the vaccine stock solution prepared by the method is not lost, the integrity of the antigen is maintained, the immunogenicity in the preparation process of the polysaccharide-protein conjugate is ensured not to be reduced to the greatest extent, and meanwhile, the generation of protein autopolymer is avoided. The streptococcus pneumoniae capsular polysaccharide-protein conjugate vaccine stock solution prepared by the method can be used for preparing streptococcus pneumoniae capsular polysaccharide monovalent and multivalent conjugate vaccines, and can also be used for preparing combined vaccines containing streptococcus pneumoniae capsular polysaccharide.
Owner:SINOVAC RES & DEV
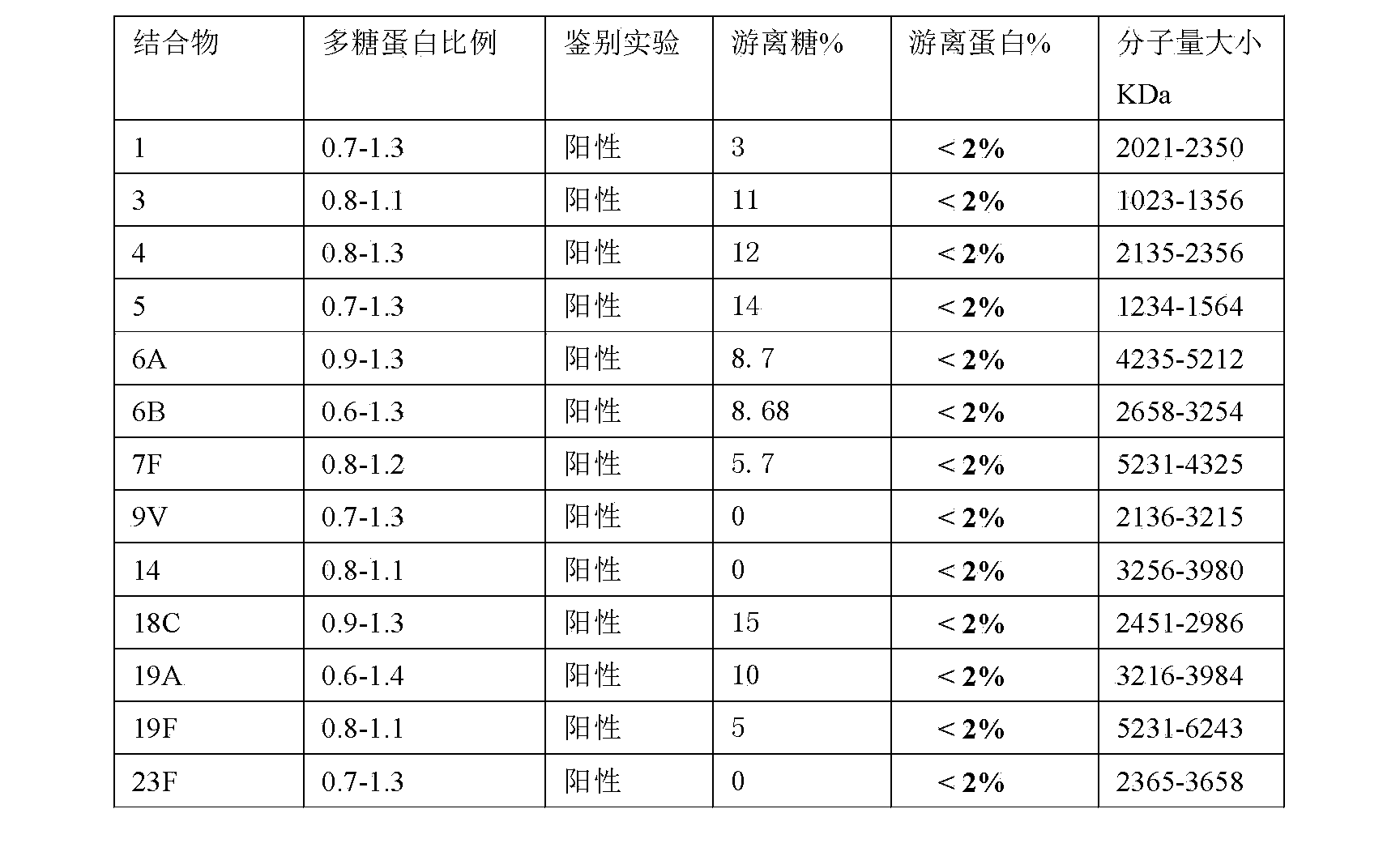
Method for determining free polysaccharide in streptococcus pneumoniae capsular polysaccharide conjugate
InactiveCN108387543AReduce analysis costsEasy to operateColor/spectral properties measurementsStreptococcus pneumoniae capsular polysaccharideQuantitative determination
The invention provides a method for determining the content of free polysaccharide in a streptococcus pneumoniae capsular polysaccharide conjugate. The scheme comprises: adding a sodium deoxycholate solution to streptococcus pneumoniae capsular polysaccharide and a conjugate corresponding to the streptococcus pneumoniae capsular polysaccharide, adjusting the pH to 2-6, and performing centrifugation to collect the supernatant; adding a color reagent to the streptococcus pneumoniae capsular polysaccharide supernatant to carry out a color development reaction; adding a color reagent to the supernatant of the conjugate corresponding to the streptococcus pneumoniae capsular polysaccharide to carry out a color development reaction; and determining the absorbance and calculating the content of free polysaccharide. The method for quantitative determination of free polysaccharide in a streptococcus pneumoniae capsular polysaccharide conjugate is low in analytical cost and simple to operate.
Owner:华兰生物疫苗股份有限公司 +2
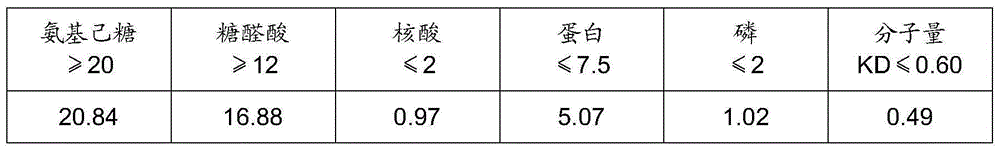
Method for preparing streptococcus pneumoniae capsular polysaccharide
ActiveCN103833865AEfficient separationAvoid harmAntibacterial agentsBacterial antigen ingredientsStreptococcus pneumoniae capsular polysaccharideStreptococcus mitis
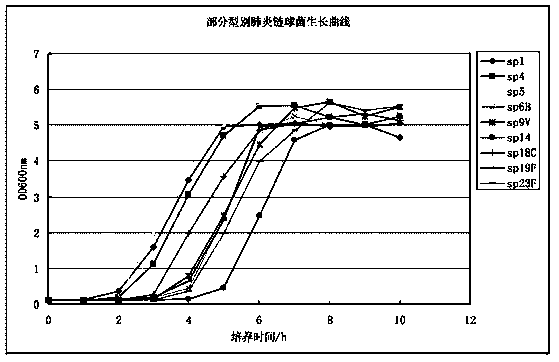
The invention relates to a method for preparing streptococcus pneumoniae capsular polysaccharide. The method comprises the following steps: 1, culturing streptococcus pneumoniae; 2, inactivating; 3, extracting streptococcus pneumoniae capsular polysaccharide; 4, refining streptococcus pneumoniae capsular polysaccharide.
Owner:TASLY BIOPHARMACEUTICALS CO LTD
16-valent streptococcus pneumoniae conjugate vaccine composition
PendingCN109091668ABroad-spectrum high-efficiency protectionAntibacterial agentsBacterial antigen ingredientsStreptococcus pneumoniae capsular polysaccharideCarrier protein
The invention discloses a 16-valent streptococcus pneumoniae conjugate vaccine composition. The vaccine composition is obtained in a manner that streptococcus pneumoniae capsular polysaccharide afterseparation and purification and carrier protein are subjected to coupling combination to be mixed, wherein the capsular polysaccharide includes 16 streptococcus pneumoniae serotypes of 1, 2, 3, 4, 5,6A, 6B, 7F, 9V, 11A, 14, 15B, 18C, 19A, 19F and 23F. The 16-valent streptococcus pneumoniae conjugate vaccine composition can cover most of common pathogenic bacterial types or drug-resistance bacterial types of our country, and has the broad-spectrum and efficient protection effect on high-risk susceptible populations such as new-born infants, old people and children under two years old.
Owner:SHANGHAI RUIZHOU BIOTECH CO LTD
A kind of pneumococcal polysaccharide protein conjugated vaccine and preparation method thereof
ActiveCN103893751BEnhance immune responseImproving immunogenicityAntibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The present invention provides a streptococcus pneumonia polysaccharide protein conjugated vaccine and a preparation method therefor. The streptococcus pneumonia polysaccharide protein conjugated vaccine comprises one or multiple immune conjugates coupling streptococcus pneumonia capsular polysaccharide with protein. At least one immune conjugate in the immune conjugates is formed by coupling a single streptococcus pneumonia capsular polysaccharide with two or more protein.
Owner:CANSINO BIOLOGICS INC
Method for determining dissociation amylase content of pneumonia streptococcus capsular polysaccharide combo
ActiveCN1963500AAccurate measurementAccurate contentMicrobiological testing/measurementPreparing sample for investigationStreptococcus pneumoniae capsular polysaccharideTest sample
This invention relates to one test method for free polysaccharide content of pneumococcal peritonitis compound in biological technique field, which comprises the following steps: a, fetching same content of the compound and adding ethanol without water and common salt for 70 to 90 hours under -20 degrees then removing upper liquid by anticentripetal and using proper agent to clear deposition to collect upper liquid through filter salt and ethanol; b, using anthranone to test sample one and two to compute free polysaccharide content.
Owner:云南沃森生物技术股份有限公司
Method for determining dissociation amylase content of pneumonia streptococcus capsular polysaccharide combo
ActiveCN1963500BMicrobiological testing/measurementPreparing sample for investigationStreptococcus pneumoniae capsular polysaccharidePolysaccharide binding
This invention relates to one test method for free polysaccharide content of pneumococcal peritonitis compound in biological technique field, which comprises the following steps: a, fetching same content of the compound and adding ethanol without water and common salt for 70 to 90 hours under -20 degrees then removing upper liquid by anticentripetal and using proper agent to clear deposition to collect upper liquid through filter salt and ethanol; b, using anthranone to test sample one and two to compute free polysaccharide content.
Owner:云南沃森生物技术股份有限公司
Preparation method of streptococcus pneumoniae capsular polysaccharide protein conjugate
ActiveCN106039300AReduce investmentImprove production efficiencyAntibacterial agentsBacterial antigen ingredientsStreptococcus pneumoniae capsular polysaccharideMicrobiology
The invention relates to a preparation method of a streptococcus pneumoniae capsular polysaccharide protein conjugate. The preparation method comprises the following steps: dissolving streptococcus pneumoniae capsular polysaccharide in a borate buffer solution, so that a capsular polysaccharide solution is obtained; making the capsular polysaccharide solution contact with CDAP and activating the capsular polysaccharide solution, so that an activated solution is obtained; and making the activated solution contact with carrier protein and promoting bonding, so that the streptococcus pneumoniae capsular polysaccharide protein conjugate is obtained. According to the method provided by the invention, a CDAP method is improved; the streptococcus pneumoniae capsular polysaccharide is dissolved by virtue of the borate buffer solution; the preparation method can avoid an operation of regulating pH value in a reaction and can reduce the use of chemical reagents; and the preparation method is simple in preparation process and high in production efficiency.
Owner:BEIJING MINHAI BIOTECH
Polyvalent pneumococcal capsular polysaccharide composition, its preparation method and application
ActiveCN103656632BStable physical and chemical propertiesPrevent diseaseAntibacterial agentsBacterial antigen ingredientsConjugate vaccineStreptococcus pneumoniae capsular polysaccharide
The invention provides a multivalent pneumococcal capsular polysaccharide composition as well as a preparation method and application thereof. The multivalent pneumococcal capsular polysaccharide composition contains a serotype 6A and at least one extra serotype selected from the group consisting of 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F and 33F. The multivalent pneumococcal capsular polysaccharide composition provided by the invention can be used for inducing an organism to generate humoral immunity, can generate a relatively good protecting effect for infectious diseases caused by the 24 common serotype pneumococcuses and is wide in immunity coverage rate and better in effect as comparison with various existing pneumococcal polysaccharide vaccines and conjugate vaccines sold on the market.
Owner:SINOVAC RES & DEV
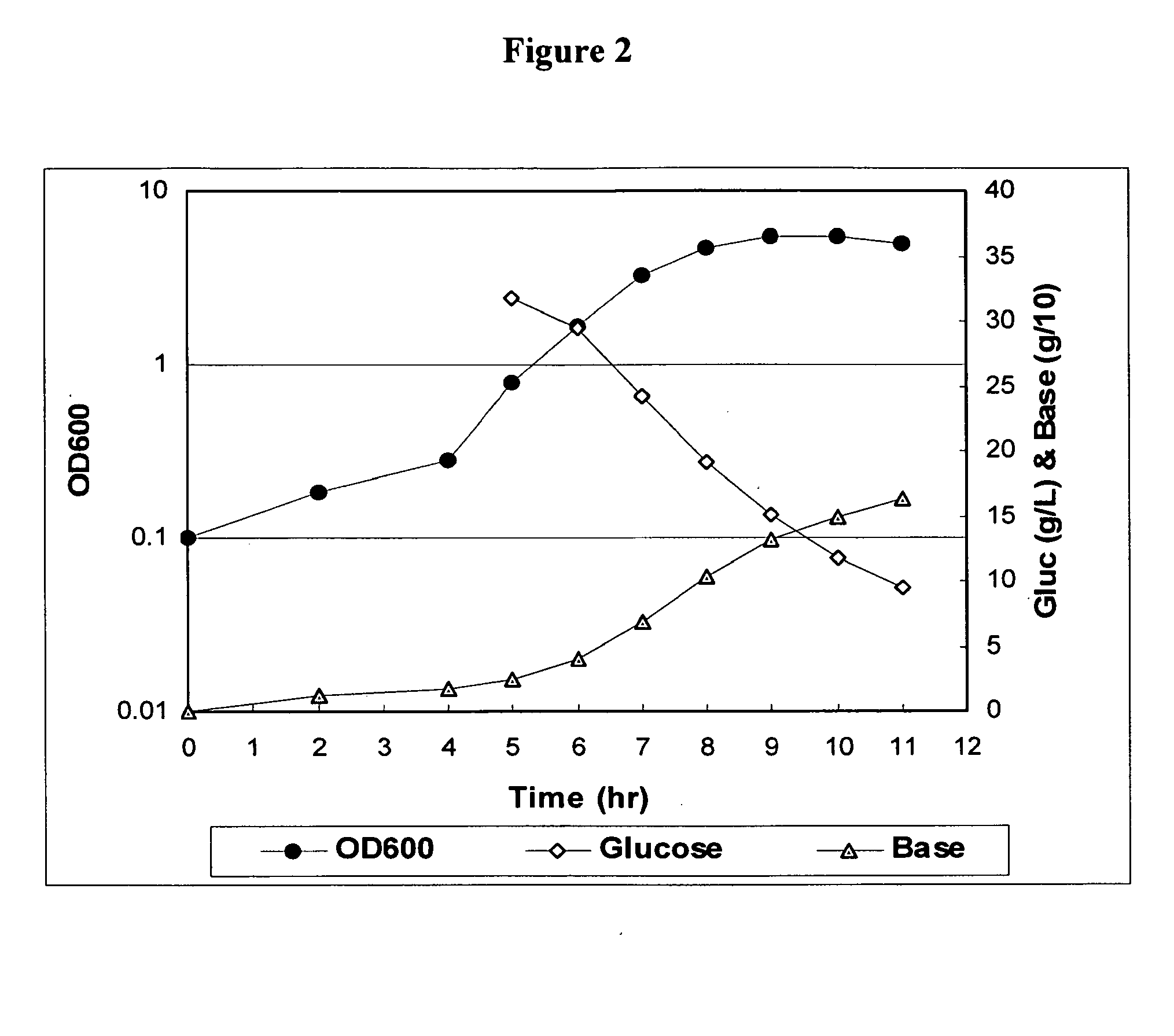
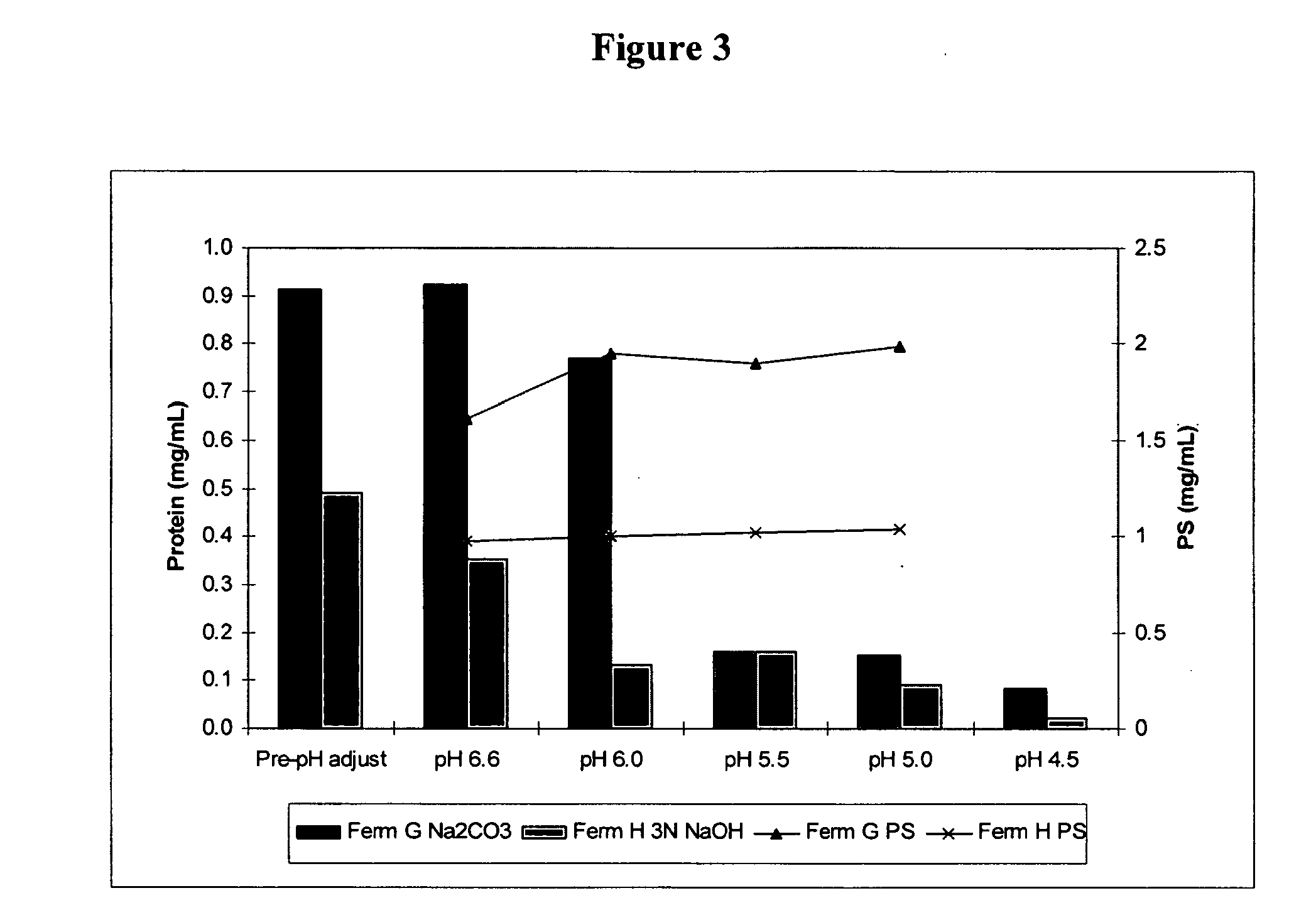
Method for controlling streptococcus pneumoniae polysaccharide molecular weight using carbon dioxide
InactiveUS20100160622A1Esterified saccharide compoundsBacteriaBacteroidesStreptococcus pneumoniae capsular polysaccharide
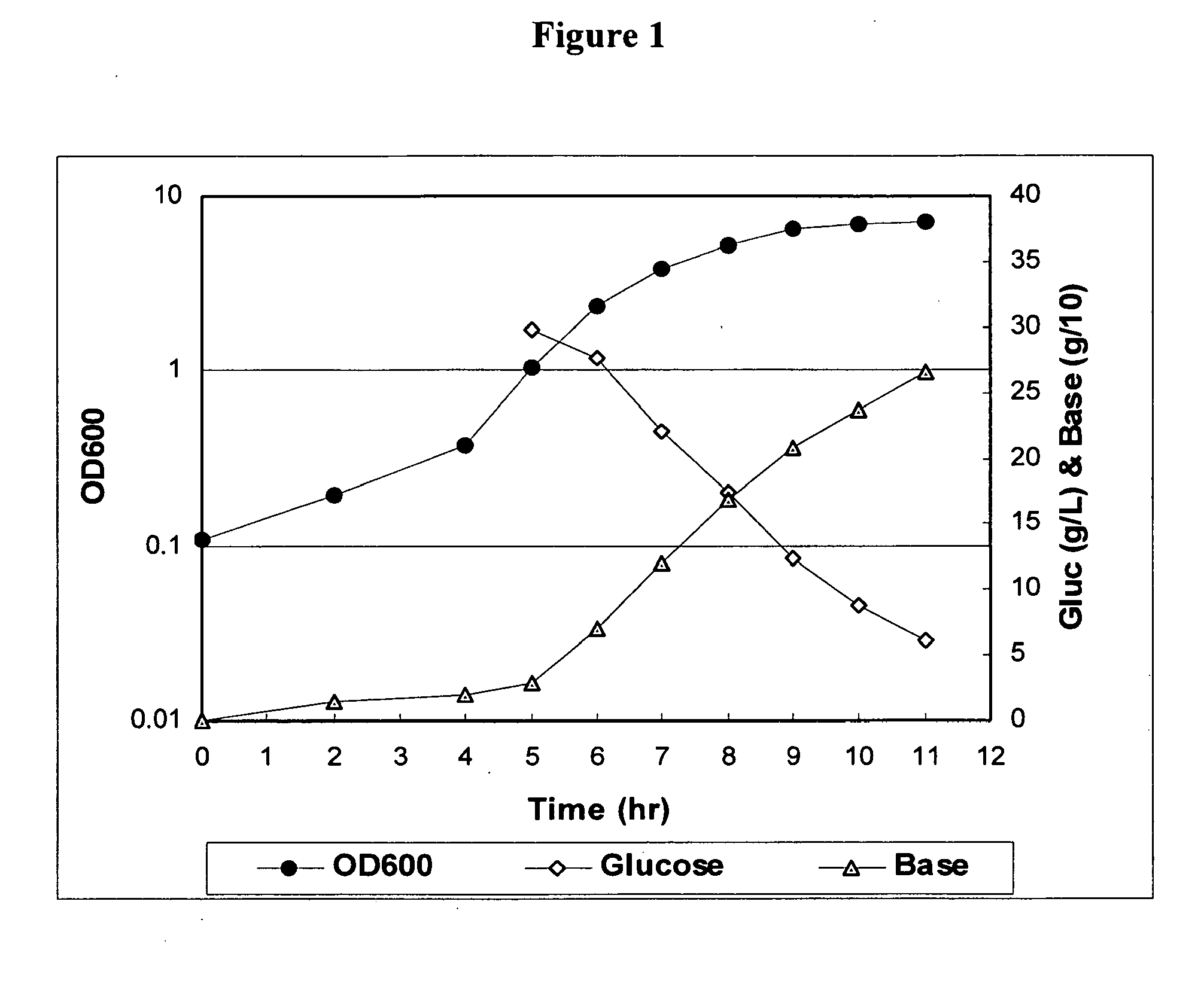
The present invention provides improved methods for producing a solution containing high molecular weight isolated Streptococcus pneumoniae capsular polysaccharides having phosphodiester linkages between saccharide repeat units. In certain methods, CO2 is supplied to a fermentation culture of Streptococcus pneumoniae bacterial cells that produce capsular polysaccharide serotypes containing phosphodiester linkages between saccharide repeat units. Exemplary Streptococcus pneumoniae serotypes containing a phosphodiester linkage between saccharide repeat units include serotypes 6A, 6B, 19A, and 19F. Supplying CO2 to the fermentation culture includes adding bicarbonate ions to the fermentation culture, adding carbonate ions to the fermentation culture, adding mixtures of bicarbonate and carbonate ions to the fermentation culture, and overlaying the fermentation culture with CO2.
Owner:WYETH LLC