Novel recombinant coronavirus protein vaccine as well as preparation method and application thereof
A new type of coronavirus technology, which is applied in the field of recombinant new coronavirus protein vaccines, can solve problems such as concerns about the effectiveness of new crown vaccines, and achieve the effects of vaccine production capacity advantages and economic cost advantages
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
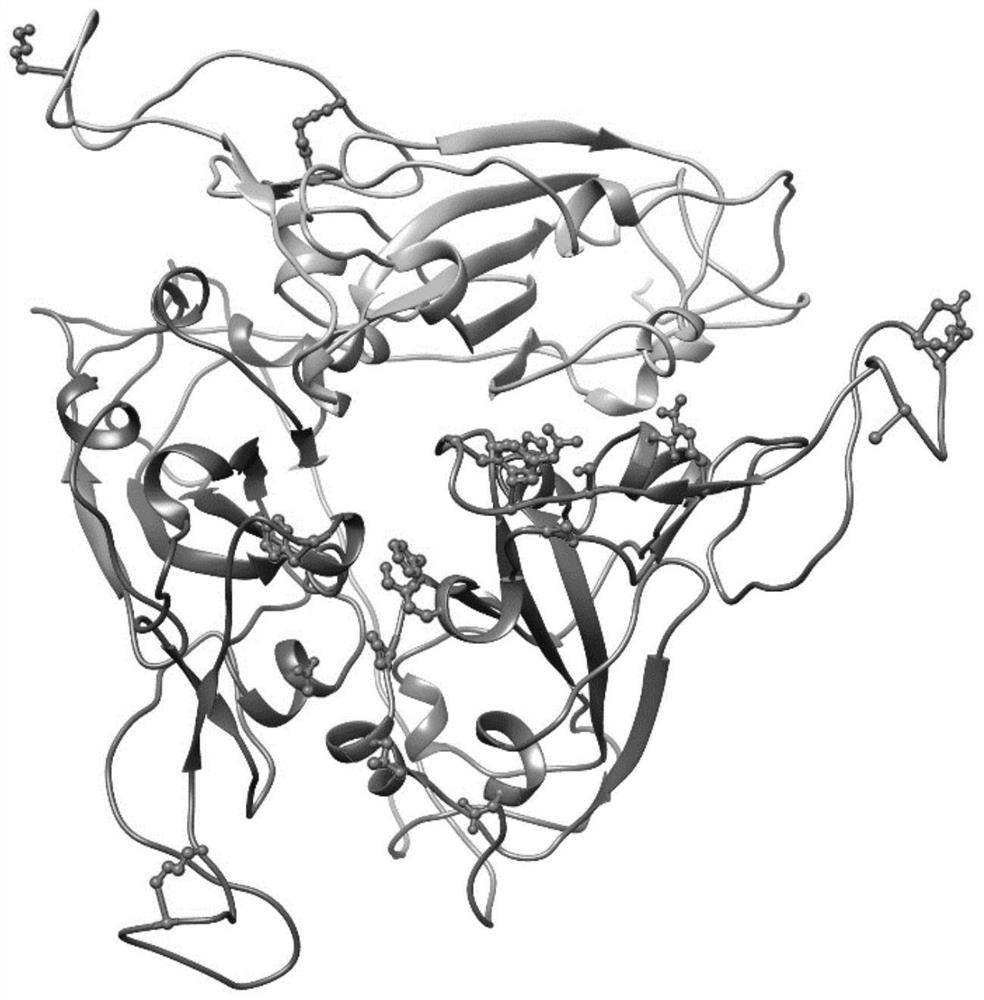
[0099] Example 1: Recombinant novel coronavirus RBD trimeric protein based on protein structure and computational biology design
[0100]The new coronavirus continues to mutate, and the emergence of multiple mutant strains has led to multiple rounds of outbreaks. According to the different transmissibility, pathogenicity or immune escape ability of the mutant strains, the World Health Organization classifies five mutant strains as variants of concern (VOCs), including Alpha (B.1.1.7) , Beta (B.1.351), Gamma (P.1), Delta (B.1.617.2), and Omicron (B.1.1.529). Among the five VOCs, a large number of studies have confirmed that the Beta variant has a strong immune escape ability, and the Delta variant has a strong transmission ability. Preliminary evidence shows that the Omicron mutant strain has both strong transmission ability and strong immune evasion ability, and its immune evasion ability is even much higher than that of the Beta strain. The emergence of these strains with s...
Embodiment 2
[0105] Example 2: Recombinant protein expression, purification and identification
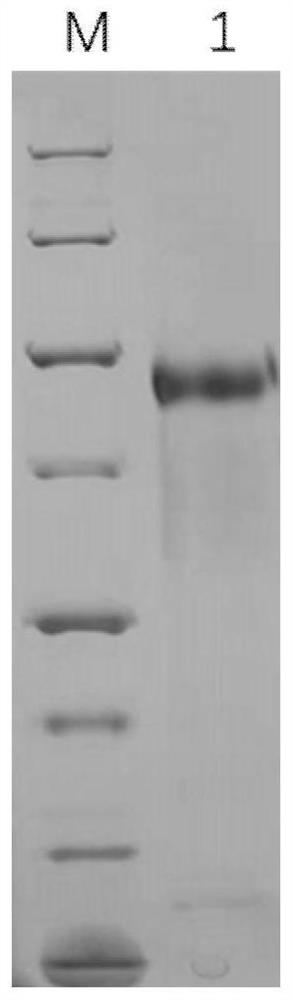
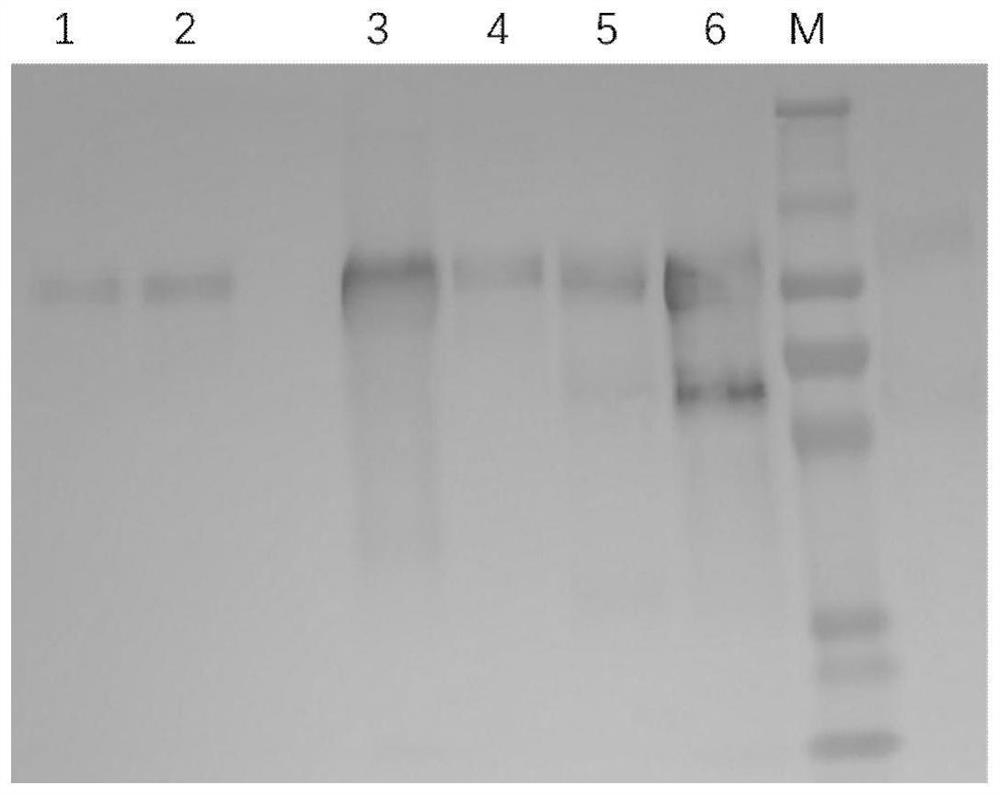
[0106] According to the codon bias of the CHO cell expression system, the nucleotide sequence encoding the recombinant protein C05G11 (amino acid sequence shown in SEQ ID NO.1) was codon optimized, and the optimized nucleotide sequence was shown in SEQ ID NO. 2. The CHO cell expression vector was constructed and then transfected into 293FT cells or CHO cells to construct a recombinant cell line. The cell line capable of stably secreting and expressing the recombinant protein C05G11 was screened by the limiting dilution method. After cell culture, the supernatant was harvested and purified by serial chromatography Finally, the recombinant protein C05G11 with a purity ≥ 95% was obtained. SDS-PAGE detection results such as figure 2 As shown, the molecular weight of the protein is 70-100kD, and some product-related substances can be seen at the same time, such as dimer protein and monomer protei...
Embodiment 3
[0108] Example 3: Detection of Physicochemical Properties and Biological Activity of C05G11 Protein
[0109] Purified C05G11 protein, C05 protein (amino acid sequence shown in SEQ ID No.23, obtained through recombinant expression and chromatography purification of 293FT cells or CHO cells) and C05C protein (amino acid sequence shown in SEQ ID No.24, Recombinantly expressed in 293FT cells or CHO cells and purified by chromatography), the RBD protein of the prototype strain (manufacturer: Beijing Yiqiao Shenzhou Technology Co., Ltd.; article number: 40592-V08B), the RBD protein with the same mutation site as the Beta strain virus (K417N , E484K, N501Y; manufacturer: Beijing Yiqiao Shenzhou Technology Co., Ltd.; article number: 40592-V08H85), RBD protein (L452R, T478K; manufacturer: Beijing Yiqiao Shenzhou Technology Co., Ltd.; article number: 40592-V02H3), the RBD protein (G339D, S371L, S373P, S375F, K417N, N440K, G446S, S477N, T478K, E484A, Q493R, G496S, Q498R, N501Y, Y505H) co...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap