Active ingredient for corylifolinin and preparation method thereof
A technology of psoralen B and psoralen, applied in the field of medicine
Inactive Publication Date: 2010-09-08
北京卓越同舟药物研究院有限公司
View PDF0 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
But so far there is no new drug prepared from the active ingredients extracted from psoraleae, especially the research on the impurities of the active ingredients
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
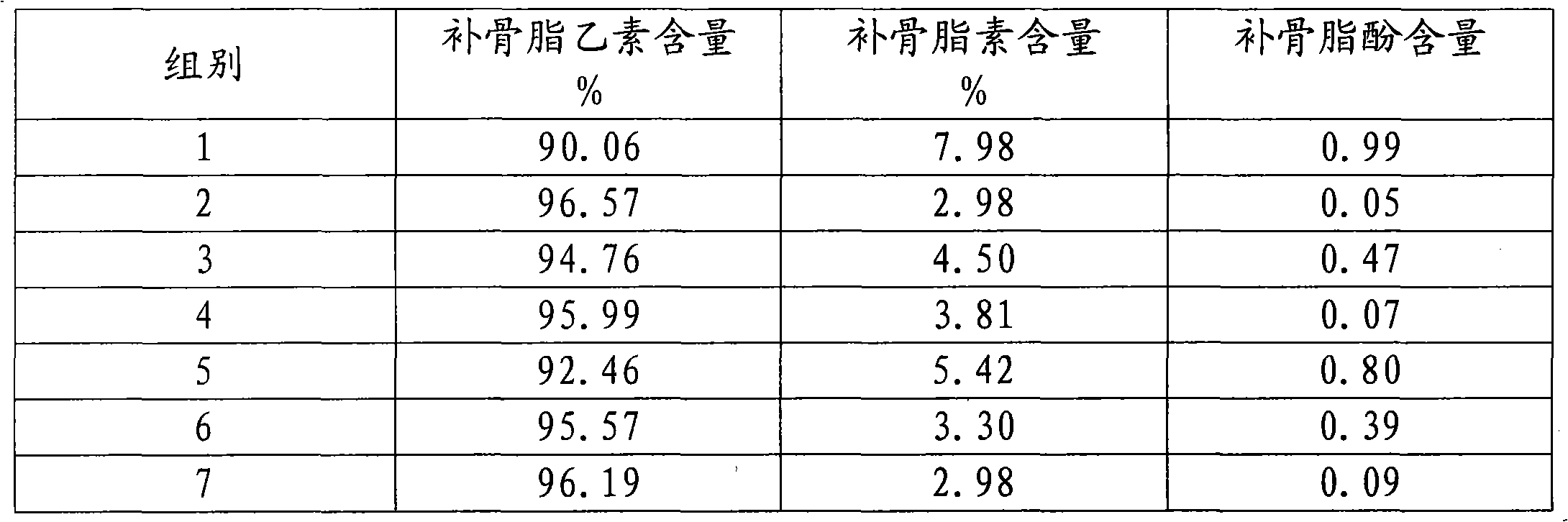
[0065] A kind of psoralen B, the content of psoralen B is 90.01%, and it contains two impurities, the content of impurity psoralen is 7.97%, and the content of impurity bakuchiol is 0.98%.
Embodiment 2
[0067] A kind of psoralen B, the content of psoralen B is 96.94%, and it contains two impurities, the content of impurity psoralen is 2.80%, and the content of impurity bakuchiol is 0.21%.
Embodiment 3
[0069] A kind of psoralen B, the content of psoralen B is 94.44%, and it contains two impurities, the content of impurity psoralen is 4.03%, and the content of impurity bakuchiol is 0.47%.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a natural medicament active ingredient and a preparation method thereof, which are characterized in that: the content of the corylifolinin is 90 to 97 percent; the corylifolinin contains two impurities; and the content of the impurity psoralen is 3 to 8 percent, and the content of the impurity bakuchiol is less than 1 percent. Preferably, the content of the psoralen is 95 to 97 percent; the content of the impurity psoralen is 3 to 4.5 percent, and the content of the impurity bakuchiol is less than 0.5 percent. More preferably, the content of the psoralen is 96 to 97 percent; the content of the impurity psoralen is 3 to 4 percent, and the content of the impurity bakuchiol is less than 0.1 percent. Pharmacological experiments prove that the natural medicament active ingredient has excellent function for treating male erectile dysfunction.
Description
technical field [0001] The invention relates to the technical field of medicine, in particular to a natural effective ingredient of psoralen B and a preparation method thereof. Background technique [0002] The "Measures for the Administration of Drug Registration" implemented on October 1, 2007 gave greater encouragement to the research and development of innovative drugs, and the number of domestic innovative drug applications also showed a clear upward trend. However, compared with the research on new drug impurities in developed countries, there is still a certain gap in the research level of our country. The main gap in impurity research is reflected in the systematicness and standardization of impurity research. This directly limits the level of drug research and the improvement of drug quality in our country. It is an urgent problem for many medical workers to clarify the research purpose, research ideas and related technologies for impurities. [0003] The traditi...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): C07D493/04A61K31/37A61P15/10A61P15/00
Inventor 王爱民安晓雪周亚君
Owner 北京卓越同舟药物研究院有限公司