Benzodiazepine derivatives tosylate salts, their polymorphic forms, preparation methods and uses thereof
A technology of tosidic acid salt and citric acid, which is applied in the preparation of sulfonate salts, medical preparations containing active ingredients, drug combinations, etc., and can solve problems such as unsatisfactory chemical and optical purity and affecting the stability of compounds
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0046] Example 1: Synthesis of salt:
[0047] Take 1g of compound base of formula (I) (prepared according to patent US200,700,934,75A) and dissolve 1g in 6ml ethyl acetate, then dissolve 0.39g tosidic acid (equal molar ratio) in 1ml methanol, and add dropwise to formula ( I) In the ethyl acetate solution of the compound base, stirred for crystallization, suction filtration, and dried under reduced pressure to obtain the tosidate of the compound of formula (I), 0.94 g of white solid, and the yield was 75.0%. HPLC: 99.18%, optical purity: 99.87%.
Embodiment 2
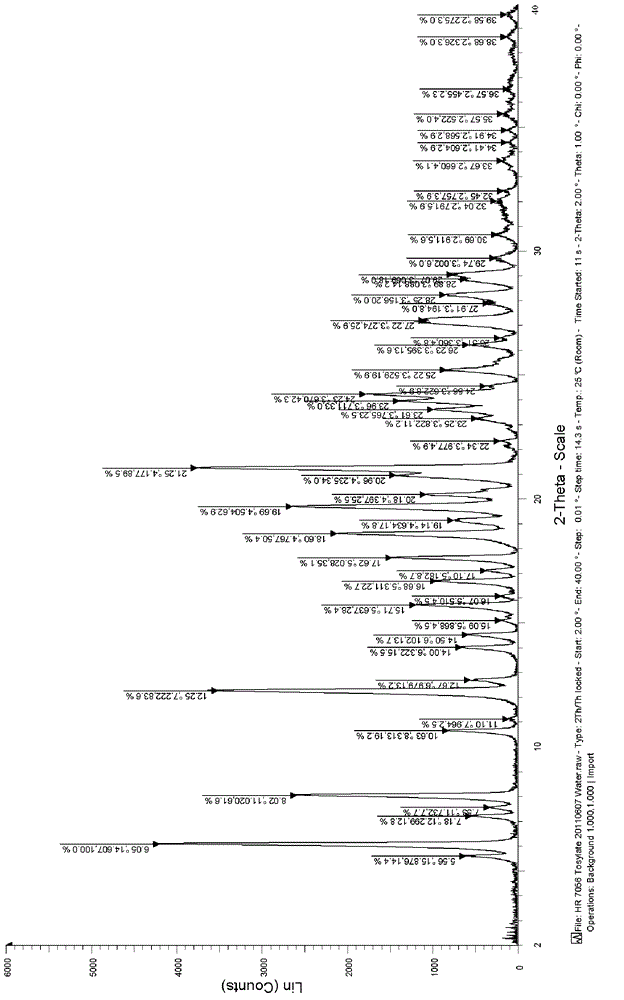
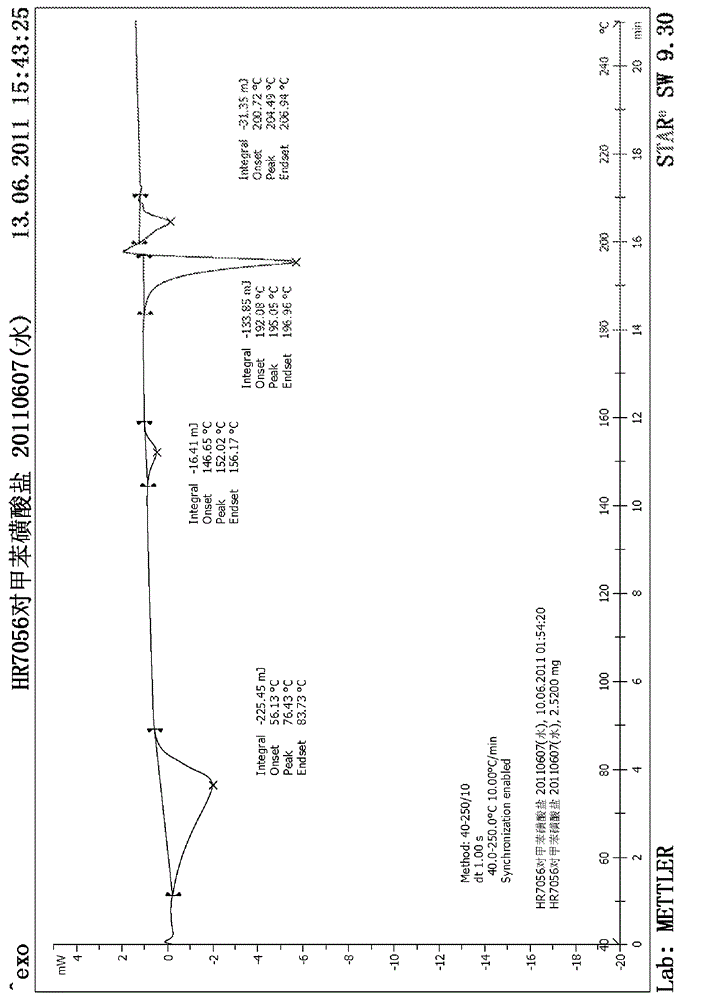
[0049] Add 1.0 g of tosidate of the compound of formula (I) obtained in Example 1 into a 50 ml round bottom flask, add 20 ml of water, and heat to reflux for 10 minutes to completely dissolve the solid, stop heating, filter while hot, and cool to crystallize The resultant product was dried under reduced pressure at 45° C. overnight to obtain 0.64 g of a white solid with a yield of 64%. The X-ray diffraction spectrum of the crystalline sample is shown in figure 1 . The crystals are about 6.05 (14.61), 7.18 (12.30), 8.02 (11.02), 12.25 (7.22), 14.00 (6.32), 14.50 (6.10), 15.71 (5.64), 16.68 (5.31), 17.62 (5.03), 18.60 (4.77), 19.69 (4.50), 21.25 (4.18), 24.23 (3.67), 25.22 (3.53) and 27.22 (3.27) have characteristic peaks, see the DSC spectrum figure 2 , There are characteristic absorption peaks at about 152.02°C, 195.05°C and 204.49°C, defining this crystal form as I crystal form.
Embodiment 3
[0051] Add 1.0g of the tosidate of the compound of formula (I) obtained in Example 1 into a 10ml round bottom flask, add 50% isopropanol aqueous solution to 5.0ml, heat to reflux to completely dissolve the solid, stop heating, and cool After crystallization, the obtained product was dried under reduced pressure at 45° C. overnight to obtain 0.72 g of white solid with a yield of 72%. The X-ray diffraction and DSC spectra of the crystalline sample were studied and compared, and it was determined that the product was crystal form I.
PUM
| Property | Measurement | Unit |
|---|---|---|
| optical purity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap