Conjugated polymer based on naphthothio dibenzofuran unit as well as preparation method and application thereof
A technology of naphthothiofluorene and conjugated polymer is applied in the field of conjugated polymer and its preparation, and achieves the effects of high fluorescence quantum yield, good solubility and improved device efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0055] Preparation of methyl 1-bromo-naphthoate
[0056] Under an argon atmosphere, 1-bromo-2-naphthoic acid (10g, 39.83mmol) was added to a two-neck flask, then 100mL of methanol was added, and then concentrated sulfuric acid (39.06mg, 398.29umol) was added dropwise, heated to 110°C, The reaction was carried out for 18 hours; the reaction mixture was poured into water, extracted with ethyl acetate, and the organic layer was washed completely with brine, and dried by adding anhydrous magnesium sulfate. After the solution was concentrated, a crude white solid was obtained, which was purified by silica gel column chromatography (petroleum ether / dichloromethane=3 / 1, v / v as the eluent). The product was placed in a refrigerator to obtain a white solid with a yield of 85%. 1 H NMR, 13 CNMR, MS and elemental analysis results show that the obtained compound is the target product, and the chemical reaction equation of the preparation process is as follows:
[0057]
Embodiment 2
[0059] Preparation of 2,8-Dibromothiofluorene
[0060] Under an argon atmosphere, add thiofluorene (20 g, 108.54 mmol) into a 250 ml two-necked bottle, then add 100 ml of chloroform to completely dissolve it, add 0.5 g of iodine, and add liquid bromine ( 38.16g, 238.80mmol), the reaction solution was stirred in an ice bath at 0°C for 2 hours, and then stirred at room temperature for 2 hours, adding saturated sodium bisulfite to quench liquid bromine, pouring the reaction mixture into water, and washing with ethyl acetate After extraction, the organic layer was washed completely with brine, and dried over anhydrous magnesium sulfate. After the solution was concentrated, a crude white solid was obtained, which was then recrystallized from chloroform with a yield of 85%. 1 H NMR, 13 CNMR, MS and elemental analysis results show that the obtained compound is the target product, and the chemical reaction equation of the preparation process is as follows:
[0061]
Embodiment 3
[0063] Preparation of 2,8-diboronate thiofluorene
[0064] Under an argon atmosphere, 2,8-dibromothiofluorene (10 g, 29.24 mmol) was dissolved in 180 mL of refined tetrahydrofuran (THF), and 1.6 mol L-1 of n-butyllithium was gradually added dropwise at -78 °C 28mL, react for 2 hours, then quickly add 25mL of 2-isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborane, and continue to react for 1 hour at -78°C , slowly warming up to room temperature and reacting for 24 hours. The reaction mixture was poured into water, extracted with ethyl acetate, and the organic layer was washed with brine and dried over anhydrous magnesium sulfate. After the solution was concentrated, a light yellow viscous crude product was obtained, which was purified by silica gel column chromatography (petroleum ether / ethyl acetate=20 / 1, v / v was selected as the eluent), and the product was placed in a box to obtain a white solid. The yield was 70%. 1 H NMR and GC-MASS tests show that it is the target product, ...
PUM
| Property | Measurement | Unit |
|---|---|---|
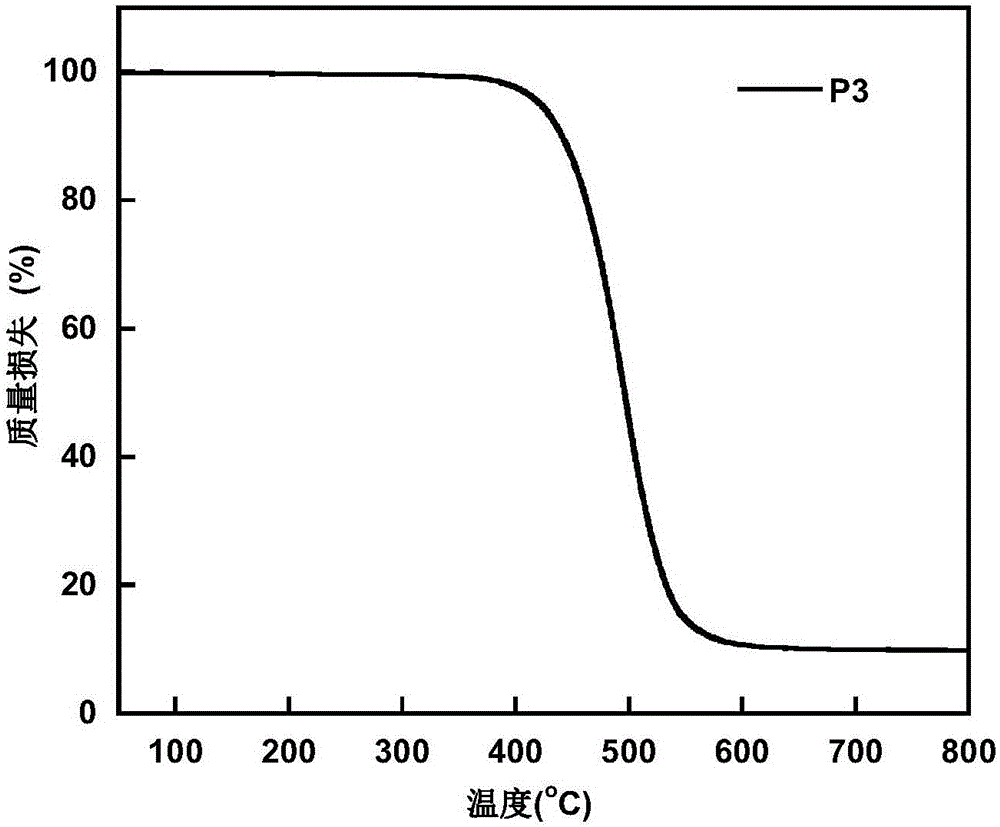
| thermal decomposition temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap