A kind of highly functional yellow fever virus human monoclonal antibody and its application
A yellow fever virus and antibody technology, applied in the field of medicine, can solve problems such as the danger of non-immune individuals, frequent outbreaks of diseases, and vaccine shortages
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
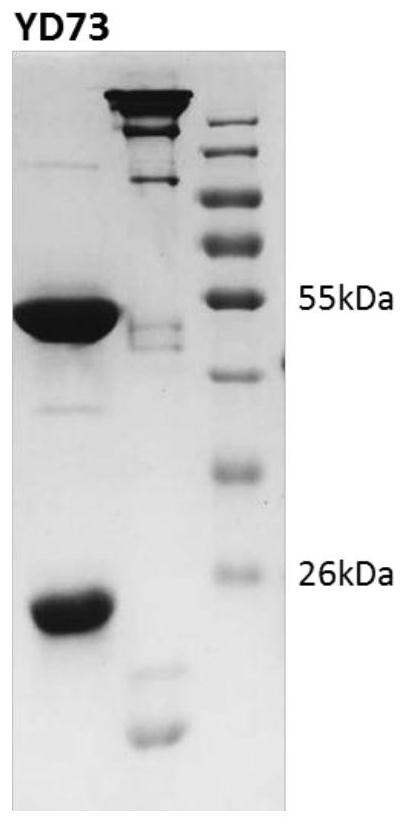
[0031] Embodiment 1: Expression and purification of yellow fever virus E protein
[0032] YFV CNYF01 / 2016 (YFV-China) strain membrane protein E protein (amino acid sequence shown in SEQ ID NO: 11, nucleotide sequence shown in SEQ ID NO: 12) extracellular region DNA fragment by NdeI and XhoI enzymes After cutting, it was connected to the pET21a vector. The 3' end of the YFV E protein coding region is connected with the coding sequence of 6 histidine tags (hexa-His-tag) and the translation stop codon. Then the ligation product was transformed into BL21 Escherichia coli competent cells. Single clones were inoculated into 40mL LB medium and cultured for 6-8 hours. Inoculate into 4L of LB medium and culture to OD at 37°C 600 =0.4-0.6, add IPTG to a final concentration of 1 mM, and continue culturing at 37°C for 4-6 hours. Inclusion bodies were harvested and refolded by dilution. The refolding solution was concentrated and replaced with 20mM Tris, 150mM NaCl, pH8.0 buffer solut...
Embodiment 2
[0033] Example 2: Isolation of specific memory B cells combined with YFV China-E protein
[0034] With the patient's informed consent, 20 mL of blood was collected and PBMCs were isolated. Separated PBMCs in 10 7 / mL density and final concentration of 400nM YFV-E protein was incubated on ice for half an hour, then washed twice with PBS, and then incubated with the following antibodies: anti-human CD3 / PE-Cy5, anti-human CD16 / PE- Cy5, anti-human CD235a / PE-Cy5, anti-human CD19 / APC-Cy7, anti-human CD27 / Pacific Blue, anti-human CD38 / APC, anti-human IgG / FITC, and anti-His / PE. After the antibody was incubated on ice for half an hour, it was washed twice with PBS. Collect PE-Cy5 by FACSAria III sorting - APCs - APC-Cy7 + Pacific Blue + FITC + PE + The cells were directly collected into a 96-well plate, 1 cell / well.
Embodiment 3
[0035] Example 3: Single B cell PCR, sequence analysis and human antibody design
[0036] The B cells obtained in Example 2 were reverse-transcribed with Superscript III reverse transcriptase (Invitrogen), and the reverse transcription primers are shown in Table 1 (sequences are shown in SED ID NO.13 to SED ID NO.20), and reacted at 55° C. for 60 min.
[0037] Table 1. Primers for reverse transcription reactions
[0038]
[0039] Using this reverse transcription product as a template, PCR was performed with HotStar Tap Plus enzyme (QIAgen) to amplify the antibody variable region sequence (PCRa). The corresponding primers were designed, and the reaction conditions were as follows: 95°C, 5min; 95°C for 30s, 55°C (heavy chain / κ chain) / 50°C (λ chain) for 30s, 72°C for 90s, 35 cycles; 72°C, 7min. Use this as a template for the second round of PCR (PCRb), the conditions are as follows: 95°C, 5min; 95°C for 30s, 58°C (heavy chain) / 60°C (κ chain) / 64°C (λ chain) for 30s, 72°C 90s, 3...
PUM
| Property | Measurement | Unit |
|---|---|---|
| affinity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com