Diamine derivatives
a technology of diamine derivatives and diamine, which is applied in the direction of drug compositions, peptides, extracellular fluid disorders, etc., can solve the problems of high-molecular weight peptides, inability to exhibit oral administration effectiveness, and inability to produce thrombin, so as to achieve potent anticoagulant effect and anticoagulant effect, potent anticoagulant effect and antithrombotic
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation process 1
[Preparation Process 1]
[0163]A compound represented by the general formula (1), a salt thereof, a solvate thereof, or an N-oxide thereof can be prepared in accordance with, for example, the following process:
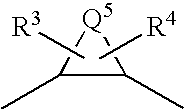
wherein Q1, Q2, Q3, Q4, R1 and R2 have the same meanings as defined above, and T1 represents a carbonyl group.
[0164]A mixed acid anhydride, acid halide, activated ester or the like, which is derived from carboxylic acid (3), may react with diamine (2), giving compound (4). The resultant compound (4) may react with carboxylic acid (5) under the same conditions, giving compound (1) according to the present invention. In the above reaction steps, reagents and conditions, which are generally used in peptide synthesis, may be applied. The mixed acid anhydride can be prepared by, for example, reaction of a chloroformate such as ethyl chloroformate or isobutyl chloroformate with carboxylic acid (3) in the presence of a base. The acid halide can be prepared by treating carboxylic acid (...
preparation process 2
[Preparation Process 2]
[0167]Compound (1) according to the present invention can also be prepared in accordance with the following process:
wherein Q1, Q2, Q3, Q4, R1 and R2 have the same meanings as defined above, T1 represents a carbonyl group, Boc represents a tert-butoxycarbonyl group, and Boc-ON represents a 2-(tert-butoxycarbonyloxyimino)-2-phenylacetonitrile.
[0168]As described above, diamine (2) is treated with Boc-ON (6) to prepare compound (7) in which one of 2 amino groups has been protected with tert-butoxycarbonyl group. The resultant compound (7) reacts with carboxylic acid (5) and affords compound (8). Compound (8) is successively treated with an acid to give compound (9). Compound (9) then reacts with the carboxylic acid (3), giving compound (1) according to the present invention. Compound (7) can be prepared by a reaction at −10° C. to 40° C. in the presence of triethylamine in a solvent such as dichloromethane. Reaction of compound (7) with the mixed acid anhydride, ...
preparation process 3
[Preparation Process 3]
[0170]Compound (1) according to the present invention can be prepared by reacting diamine (2) with sulfonyl halide (10) and then condensing the reaction product with carboxylic acid (5).
wherein Q1, Q2, Q3, Q4, R1 and R2 have the same meanings as defined above, T1 represents a sulfonyl group, and X represents a halogen atom.
[0171]Diamine (2) reacts with sulfonyl halide (10) at −10° C. to 30° C. in the presence of a base such as triethylamine in an inert solvent, giving compound (4). The inert solvent and base may be suitably chosen for use from those described in Preparation Process 1. The resultant compound (4) is condensed with carboxylic acid (5) using the reagents and conditions described in Preparation Process 1, whereby compound (1) according to the present invention can be prepared. Sulfonyl halide (10) may be synthesized in a proper base in accordance with the publicly known process (WO96 / 10022, WOO / 09480) or a process according to it.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More