Post-synthetic chemical modification of RNA at the 2'-position of the ribose ring via "click" chemistry
a ribose ring and chemical modification technology, applied in the field of post-synthetic can solve the problems of low-throughput chemical modification of rna prior to synthesis, bear sensitive functional groups, and limited scop
Inactive Publication Date: 2013-05-09
SIRNA THERAPEUTICS INC
View PDF4 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
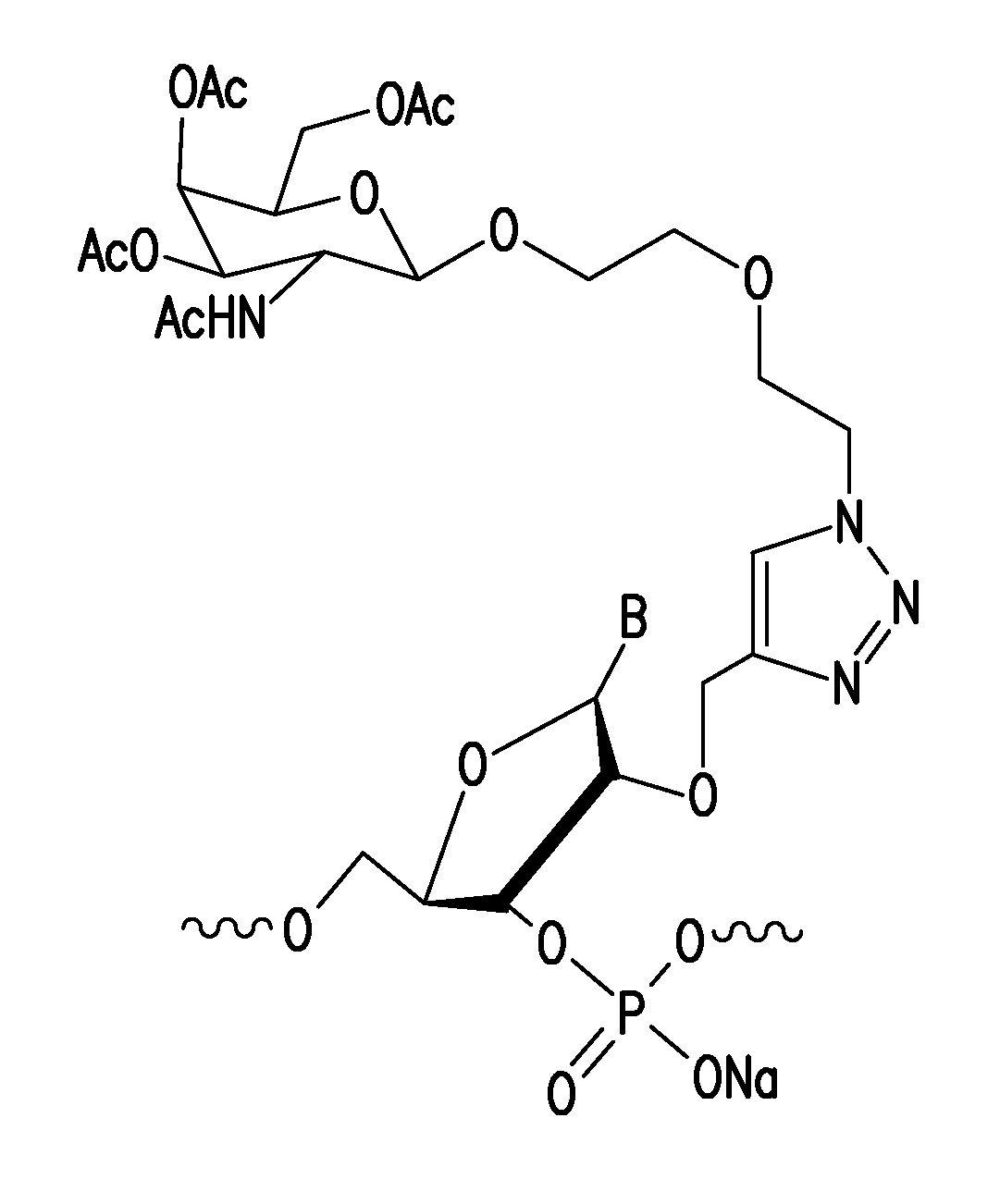
[0015]This invention relates to the post-synthetic chemical modification of RNA at the 2′-position on the ribose ring via a copper catalyzed Huisgen cycloaddition (“click” chemistry: Kolb, Sharpless Drug Discovery Today 2003, 8, 1128). The invention 1) avoids complex, tedious multi-step syntheses of each desired modified ribonucleoside; 2) allows diverse chemical modifications using high-fidelity chemistry that is completely orthogonal to commonly used alkylamino, carboxylate and disulfide linker reactivities; 3) allows introduction of functional groups that are incompatible with modern automated solid-phase synthesis of RNA and subsequent cleavage-deprotection steps; 4) allows introduction of functional groups useful as targeting ligands; and 5) enables high-throughput structure-activity relationship studies on chemically modified RNA in 96-well format.
Problems solved by technology
Chemical modifications of RNA have relied heavily on work-intensive, cumbersome, multi-step syntheses of structurally novel nucleoside analogues and their corresponding phosphoramidites prior to RNA assembly.
Furthermore, some chemical modifications bear sensitive functional groups that may be incompatible with state-of-the-art automated synthesis of RNA as well as subsequent downstream cleavage-deprotection steps.
These attributes have made chemical modification of RNA prior to synthesis rather low-throughput and limited in scope.
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
example 1
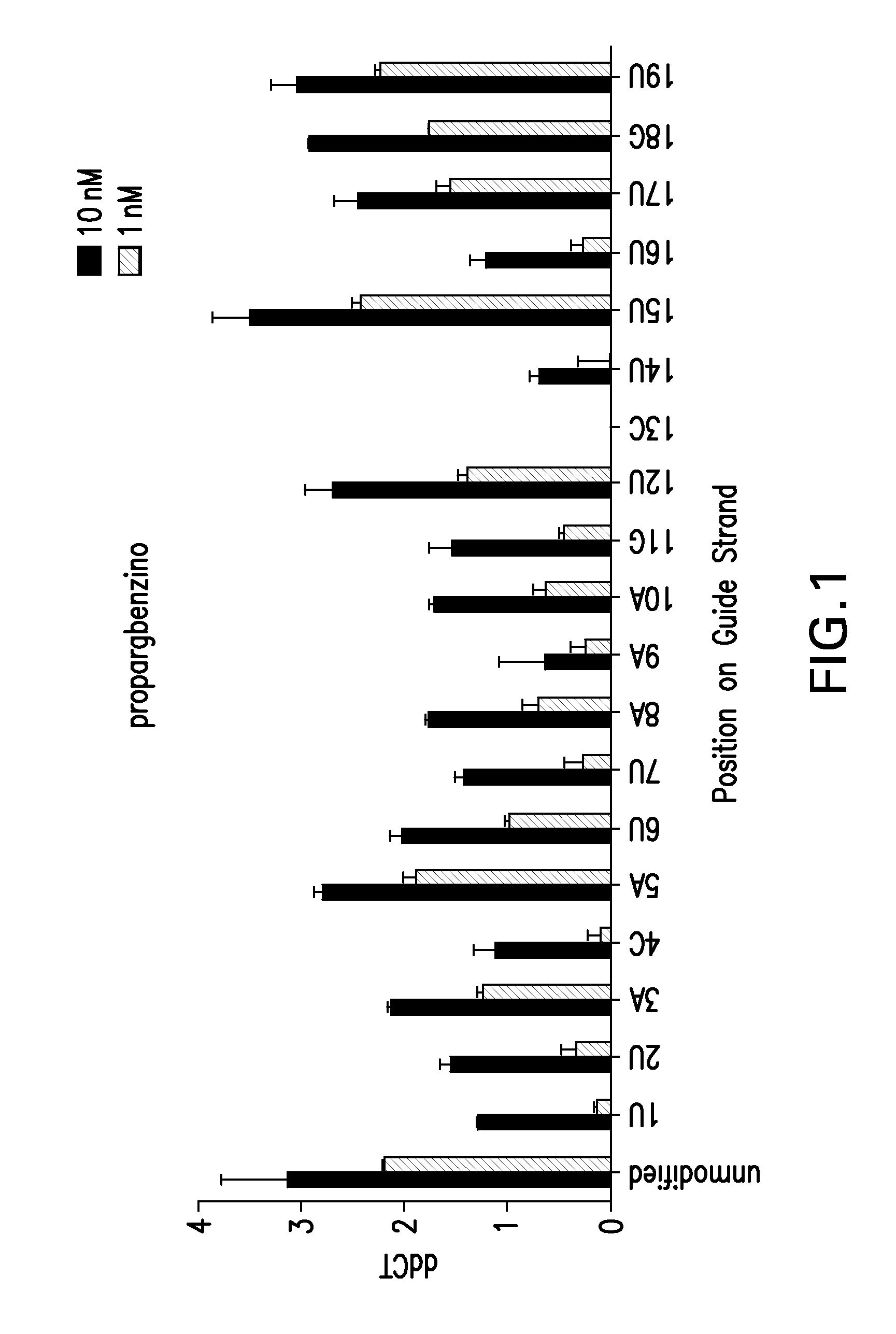
[0231]In FIG. 1, the impact on knockdown of the 2′-O-benzyl-triazole inosine chemical modification was systematically evaluated along positions 1 through 19 of the guide strand of an siRNA targeting mRNA SSB(291).
example 2
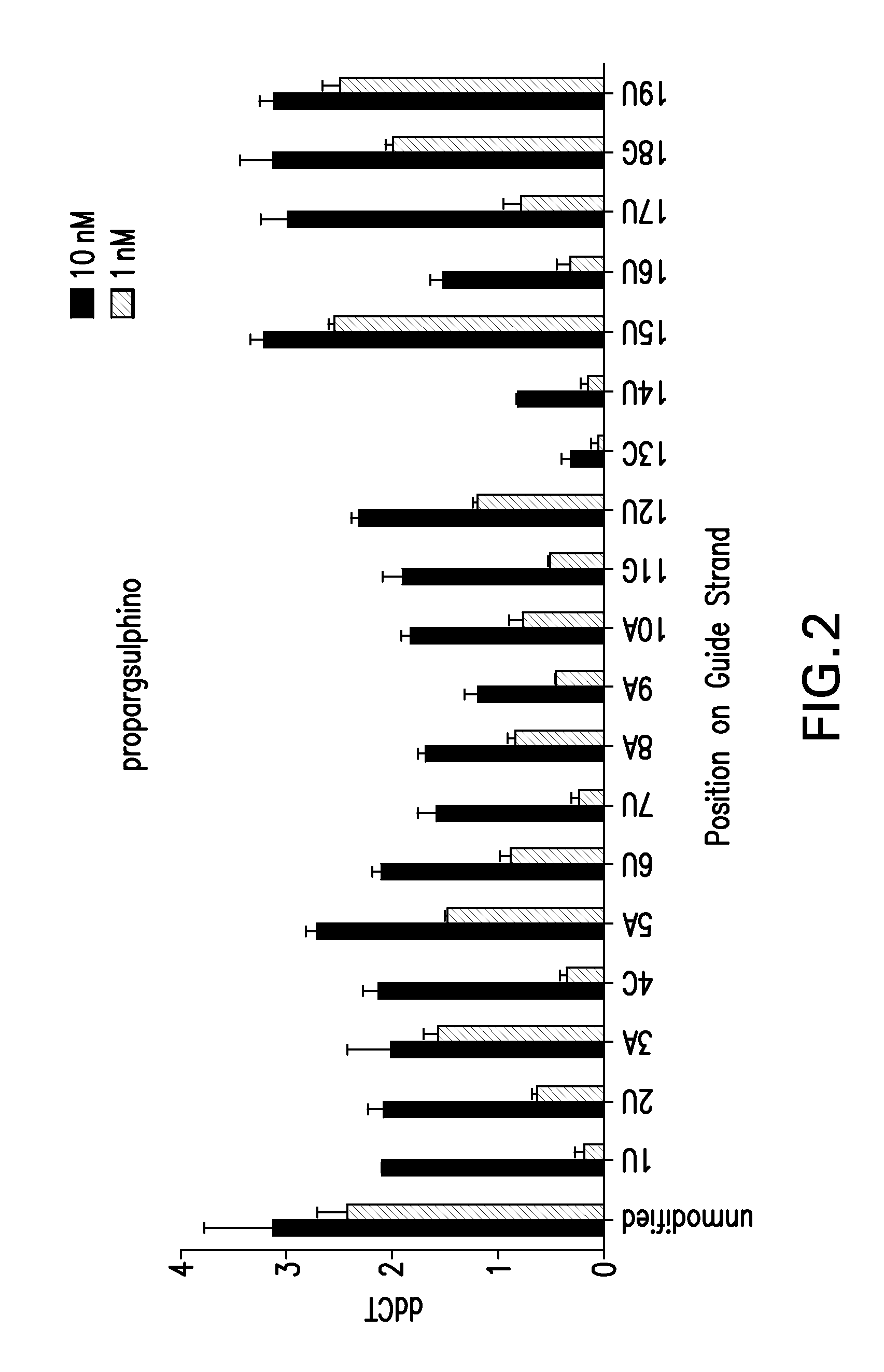
[0232]In FIG. 2, the impact of the 2′-O-phenylthiomethyl-triazole inosine chemical modification was systematically evaluated along positions 1 through 19 of the guide strand of an siRNA targeting mRNA SSB(291).
example 3
[0233]In FIG. 3, the impact on knockdown of the 2′-O-benzyl-triazole inosine chemical modification was systematically evaluated along positions 1 through 19 of the guide strand of an siRNA targeting mRNA Luc(80).
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperatures | aaaaa | aaaaa |
Login to View More
Abstract
This invention relates to the post-synthetic chemical modification of RNA at the 2′-position on the ribose ring via a copper catalyzed Huisgen cycloaddition (“click” chemistry: Kolb, Sharpless Drug Discovery Today 2003, 8, 1128). The invention 1) avoids complex, tedious multi-step syntheses of each desired modified ribonucleoside; 2) allows diverse chemical modifications using high-fidelity chemistry that is completely orthogonal to commonly used alkylamino, carboxylate and disulfide linker reactivities; 3) allows introduction of functional groups that are incompatible with modern automated solid-phase synthesis of RNA and subsequent cleavage-deprotection steps; 4) allows introduction of functional groups useful as targeting ligands; and 5) enables high-throughput structure-activity relationship studies on chemically modified RNA in 96-well format.
Description
BACKGROUND OF THE INVENTION[0001]RNA interference (RNAi) is an evolutionarily conserved cellular mechanism of post-transcriptional gene silencing found in fungi, plants and animals that uses small RNA molecules to inhibit gene expression in a sequence-specific manner. The RNAi machinery can be harnessed to destruct any mRNA of a known sequence. This allows for suppression (knock-down) of any gene from which it was generated and consequently preventing the synthesis of the target protein. Smaller siRNA duplexes introduced exogenously were found to be equally effective triggers of RNAi (Zamore, P. D., Tuschl, T., Sharp, P. A., Bartel, D. P. Cell 2000, 101, 25-33). Synthetic RNA duplexes can be used to modulate therapeutically relevant biochemical pathways, including ones which are not accessible through traditional small molecule control.[0002]Chemical modification of RNA leads to improved physical and biological properties such as nuclease stability (Damha et al Drug Discovery Today ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(United States)
IPC IPC(8): C07H21/02
CPCC07C217/40C07C217/52C07C2101/02C07D207/08C07D207/12C07H21/02C07D211/22C07D211/44C07D211/46C07D295/088C07D209/48C07C2601/02C12N15/113C12N2310/14C12N2310/141C12N2310/321C12N15/1137C12N2330/30
Inventor ZEWGE, DANIELGOSSELIN, FRANCIS
Owner SIRNA THERAPEUTICS INC