Alpha-naphthalenesulfonamide base quintuple heterocyclic compound and anti-tumor activity thereof
A technology of naphthalene sulfonamide group and five-membered heterocyclic ring, which is applied in the fields of active ingredients of heterocyclic compounds, organic active ingredients, organic chemistry, etc.
Inactive Publication Date: 2010-08-25
MEDICINE & BIOENG INST OF CHINESE ACAD OF MEDICAL SCI
View PDF3 Cites 11 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The α-naphthalenesulfonamido five-membered heterocyclic compound of the present invention and its new application in anti-tumor aspect have not yet seen relevant reports at home and abroad
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
Embodiment 2
Embodiment 3
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
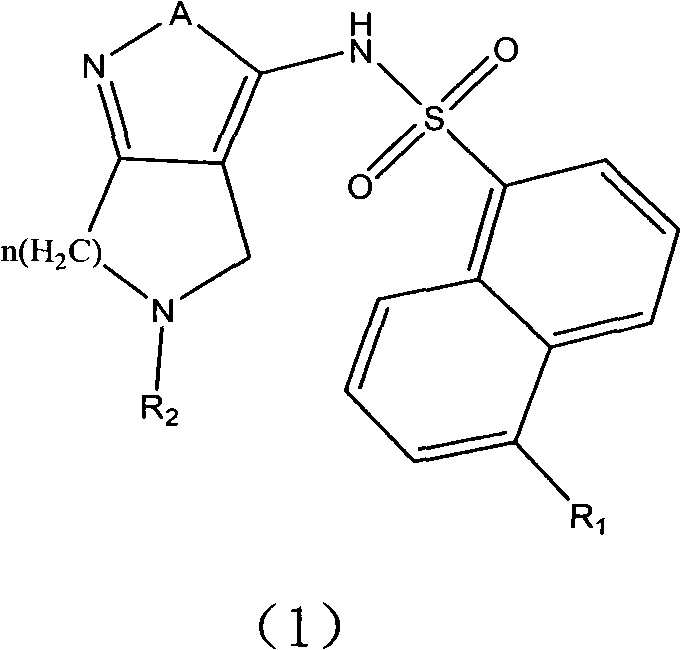
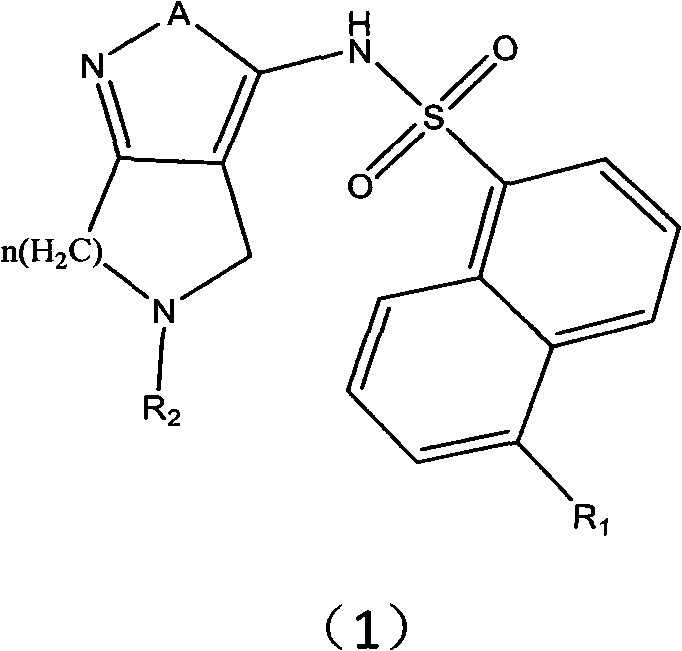
The invention relates to an alpha-naphthalenesulfonamide base quintuple heterocyclic compound and application thereof in anti-tumor treatment. Shown as research results, the alpha-naphthalenesulfonamide base quintuple heterocyclic compound, an isomer thereof, a stereomer or a mixture of the stereomer and a pharmaceutically acceptable salt or a precursor all have suppression effects on tumor cells, wherein the suppression ratios of about half compounds on the cell level are superior to that of ML-7 positive control medicine. The invention is expected to lay the foundation for developing anti-tumor medicaments.
Description
Technical field: The invention relates to α-naphthalene sulfonamide five-membered heterocyclic compounds and their application in antitumor therapy. Background technique: In 2003, U.S. Patent (US20030171357A1) disclosed a series of 3-aminopyrazole compounds that can be used as protein kinase inhibitors, especially related documents [P.Pevarello, et al.J.Med.Chem.2004, 47 (13) , 3367-3380]; P.Pevarello, et al.Bioorg.Med.Chem.Lett.2006, 16, 1084-1090; Daniele Fancelli, et al.J.Med.Chem.2005, 48, 3080-3084] It was found that as a CDK2 inhibitor, it can effectively inhibit the malignant growth of tumor cells, and has good selectivity for CDK2. Cyclin-dependent kinase (CDK) is a serine / threonine protein kinase, which determines whether the cell cycle proceeds in an orderly manner, and it is only active when combined with the corresponding cell cycle protein. There are seven main CDKs (CDK1-7), the content of which is basically stable throughout the cell cycle. CDK2, CDK4 and C...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More IPC IPC(8): C07D487/04C07D498/04C07D471/04A61K31/4162A61K31/42A61K31/437A61P35/00
Inventor 王玉成白晓光邵荣光王菊仙郭欣李祎亮任开环
Owner MEDICINE & BIOENG INST OF CHINESE ACAD OF MEDICAL SCI
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com