Functionalized nano-graphene oxide-loaded insulin derivative material and its preparation and application in the preparation of drugs for the treatment of diabetes
A technology of insulin derivatives and nano-graphite oxide, which can be used in drug combination, metabolic diseases, pharmaceutical formulations, etc., can solve problems such as undiscovered, and achieve the effects of simple steps, alleviating physical and mental pain, and direct preservation and use.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0037] (1) Preparation of nano-graphene oxide
[0038]Adopt conventional Hummers method to prepare graphene oxide, weigh 3g natural graphite powder and 18g potassium permanganate and mix evenly, add concentrated sulfuric acid 360mL and concentrated phosphoric acid 40mL mixed acid (volume ratio 9:1), stir and heat to keep the heating temperature of reactant Reaction between 35 ~ 40 ℃ for 1h. Then the reactant was rapidly heated to 50°C and the reaction was continued for 12h. After the reaction was completed, it was naturally cooled to room temperature. Quickly add 100mL of ice water into the above system, stir for 5min and then dropwise add 4mL of 30% H 2 o 2 The solution turned from black to khaki. Let it stand for 2 hours, pass through a metal standard test screen (W.S.Tyler, 300 μm), centrifuge the filtrate at 15,000 rpm for 30 minutes, discard the supernatant, and wash the lower layer of solid matter twice with 200 mL of water, 200 mL of 30% HCl and 200 mL of ethanol ,...
Embodiment 2
[0044] (1) the preparation of nanometer graphene oxide is the same as embodiment 1;
[0045] (2) the preparation of aminated polyethylene glycol functionalized nano-graphene oxide is the same as in Example 1;
[0046] (3) Preparation of Aminated Polyethylene Glycol Functionalized Nano Graphene Oxide Loaded Insulin Derivative Materials
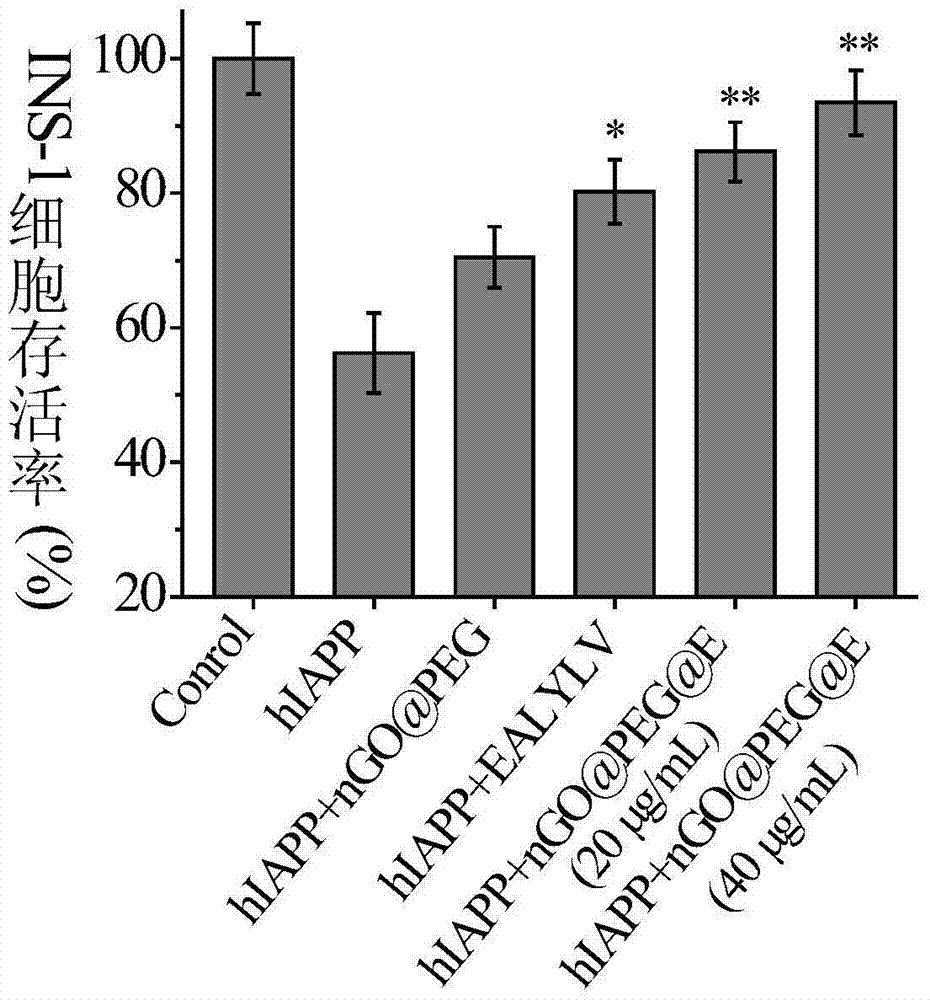
[0047] Weigh 10 mg of nGO@PEG prepared in step (2) and dissolve in 10 mL of PBS to prepare a 1 mg / mL solution. Dissolve 2 mg of insulin-derived short peptide EALYLV (relative molecular weight: 707) with 1 mL of PBS, and divide 1 mL of the solution into 5 parts, each with a concentration of 2.83×10 -3 mol / L as a stock solution. Ultrasonic disperse 10mL of nGO@PEG solution at 570W for 10min, then add 20mg EDC·HCl and 40mg NHS to maintain the pH value of the solution at about 7, then add 400μM EALYLV stock solution with a rubber dropper, and stir at 25°C Reaction 2h. After the reaction was completed, centrifuge at 6000 rpm for 30 min, discard ...
Embodiment 3
[0049] (1) the preparation of nanometer graphene oxide is the same as embodiment 1;
[0050] (2) the preparation of aminated polyethylene glycol functionalized nano-graphene oxide is the same as in Example 1;
[0051] (3) Preparation of Aminated Polyethylene Glycol Functionalized Nano Graphene Oxide Loaded Insulin Derivative Materials
[0052] Weigh 20 mg of nGO@PEG prepared in step (2) and dissolve in 10 mL of PBS to prepare a 1 mg / mL solution. Dissolve 2 mg of insulin-derived short peptide EALYLV (relative molecular weight: 707) with 1 mL of PBS, and divide 1 mL of the solution into 5 parts, each with a concentration of 1.83×10 -3 mol / L as a stock solution. Ultrasonic disperse 10mL of nGO@PEG solution at 300W for 10min, then add 40mg EDC·HCl and 80mg NHS to maintain the pH value of the solution at about 7, then add 400μM EALYLV stock solution with a rubber dropper, and stir at 25°C Reaction 2h. After the reaction was completed, centrifuge at 6000 rpm for 30 min, discard ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| particle diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap