Difluoromethyl aldehyde hydrazone compound and preparation method thereof
A technology of difluoromethylated aldehyde hydrazone and difluoromethyl, which is applied in the field of difluoromethylated aldehyde hydrazone compounds and their preparation, can solve the problems of high cost and complex process, and achieve environmental friendliness and step-by-step operation Simple, high-yield results
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0062]
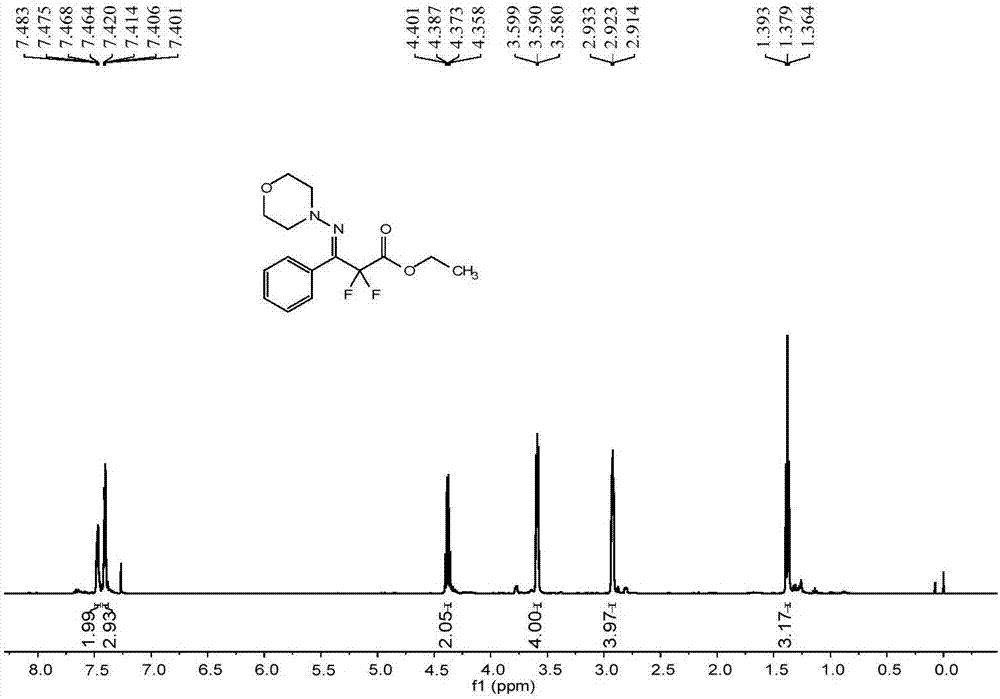
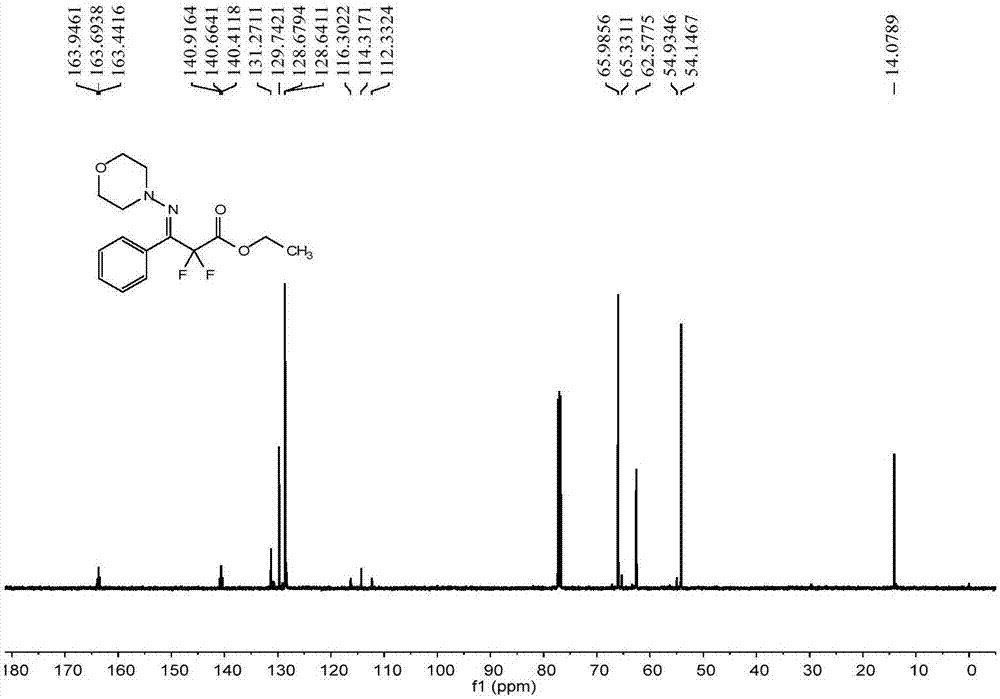
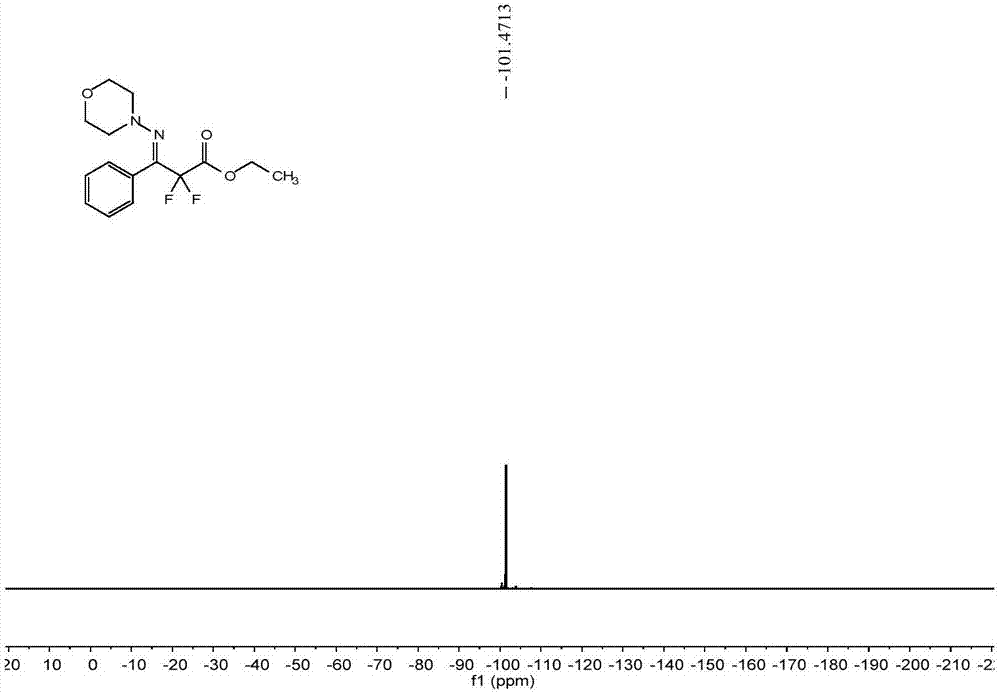
[0063] The reactor was evacuated and replaced with argon three times, and 0.2mmol (21.2mg) benzaldehyde (1a), 0.24mmol (24.5mg) N-aminomorphine (2a), 0.40mmol (80.8mg) difluorobromide were added successively Ethyl acetate (3a), 0.006 mmol (4.73 mg) 4CzIPN, 75 mg anhydrous MgSO 4 , 1mL DMF, 8W blue LED, stirred at room temperature for 12 hours. After the reaction was finished, the solvent was removed with a rotary evaporator, and the crude product was subjected to column chromatography, and the eluent was sherwood oil: ethyl acetate=10:1 mixed solvent to obtain 44.32 mg of difluoromethyl-substituted phenylaldehyde hydrazone Compound 4a. The isolated yield was 71%.
[0064] The characterization data of compound 4a are as follows:
[0065] E:Z=19:1. 1 H NMR (500MHz, CDCl 3 ):δ7.48-7.46(m,2H),7.42-7.40(m,2H),4.38(q,J=7.0Hz,2H),3.59(t,J=4.5Hz,4H),2.92(t, J=5.0Hz, 4H), 1.38(t, J=7.0Hz, 3H). 13 C NMR (125MHz, CDCl 3 ): δ163.7(t, 31.5Hz), 140.7(t, 31.5Hz), 131.3, 1...
Embodiment 2
[0067]
[0068] The reactor was evacuated and replaced with argon three times, and 0.2mmol (21.2mg) p-chlorobenzaldehyde (1b), 0.24mmol (24.5mg) N-aminomorphine (2a), 0.40mmol (80.8mg) di Ethyl fluorobromoacetate (3a), 0.006mmol (4.73mg) 4CzIPN, 75mg anhydrous MgSO 4 , 1mL DMF, 8W blue LED, stirred at room temperature for 12 hours. After the reaction was finished, the solvent was removed with a rotary evaporator, and the crude product was subjected to column chromatography, and the eluent was sherwood oil: ethyl acetate=10:1 mixed solvent to obtain 64.37 mg of difluoromethyl-substituted p-chlorophenylaldehyde Hydrazone compound 4b. The isolated yield was 93%.
[0069] The characterization data of compound 4b are as follows
[0070] E:Z=19:1. 1 H NMR (500MHz, CDCl 3 ):δ7.45-7.40(m,4H),4.38(q,J=7.0Hz,2H),3.61(t,J=4.5Hz,4H),2.94(t,J=5.0Hz,4H),1.38 (t,J=7.0Hz,3H); 13 C NMR (125MHz, CDCl 3 ): δ=163.4(t, J=31.3Hz), 139.5(t, J=31.8Hz), 135.9, 130.1, 129.6, 129.1, 114.2(t, ...
Embodiment 3
[0072]
[0073] The reactor was evacuated and replaced with argon three times, and 0.2mmol (21.2mg) p-tolualdehyde (1c), 0.24mmol (24.5mg) N-aminomorphine (2a), 0.40mmol (80.8mg) were added successively Ethyl difluorobromoacetate (3a), 0.006 mmol (4.73 mg) 4CzIPN, 75 mg anhydrous MgSO 4 , 1mL DMF, 8W blue LED, stirred at room temperature for 12 hours. After the reaction was finished, the solvent was removed with a rotary evaporator, and the crude product was subjected to column chromatography, and the eluent was sherwood oil: ethyl acetate=10:1 mixed solvent to obtain 54.80 mg of difluoromethyl-substituted p-methylphenyl Aldehydrazone compound 4c. The isolated yield was 84%.
[0074] The characterization data of compound 4c are as follows
[0075] E:Z=18:1. 1 H NMR (500MHz, CDCl 3 ): δ7.39(d, J=8.0Hz, 2H), 7.23(d, J=8.0Hz, 2H), 4.40(q, J=7.0Hz, 2H), 3.62(t, J=4.5Hz, 4H ), 2.95(t, J=4.5Hz, 4H), 2.40(s, 3H), 1.40(t, J=7.0Hz, 3H); 13 C NMR (125MHz, CDCl 3 ): δ163.8(t, ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap