New use of Panax Notoginseng and aspirin combination
A technology of aspirin and combined application, applied in the new use field of combination or combination of Panax notoginseng and aspirin, to achieve good effects and reduce gastric mucosal damage
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
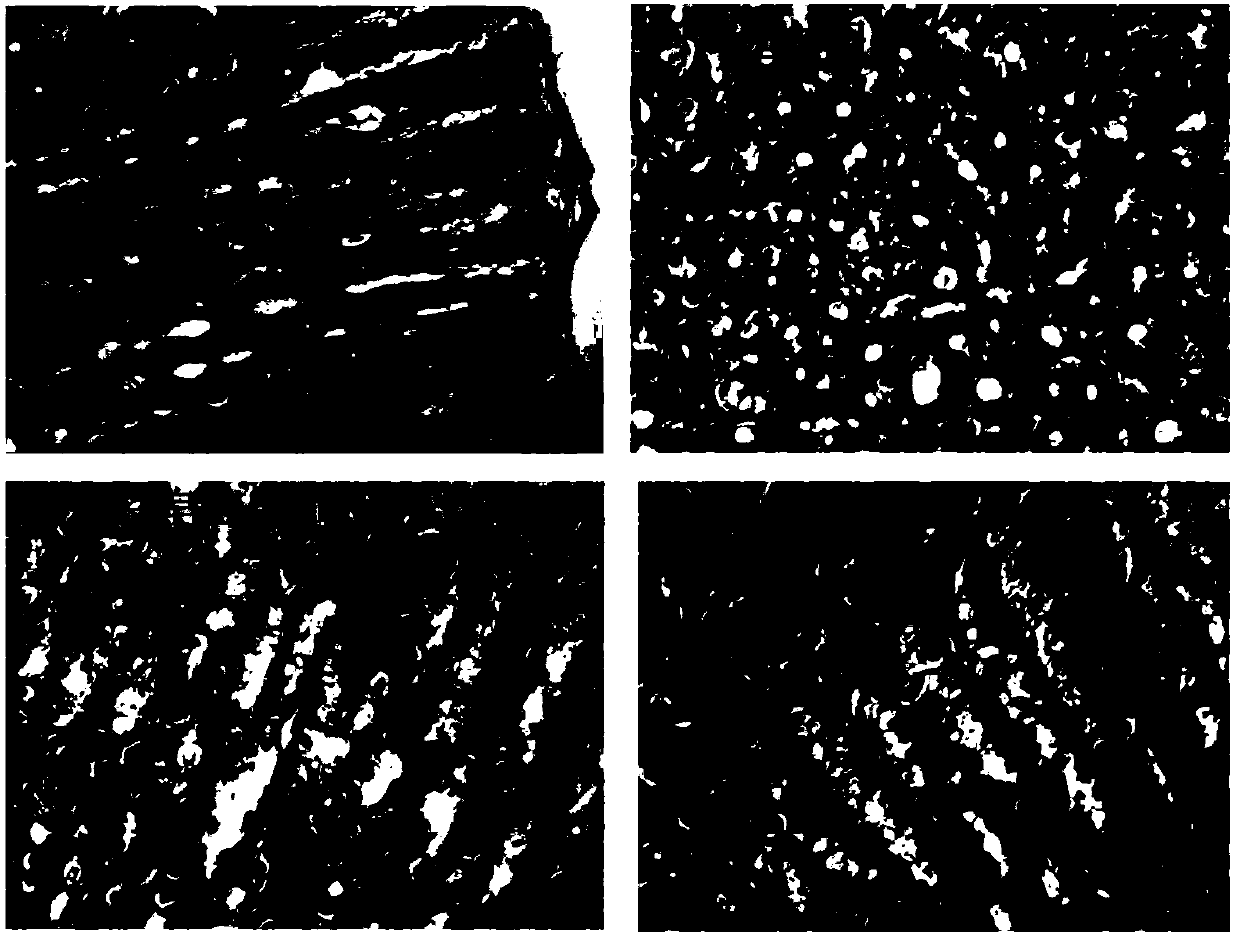
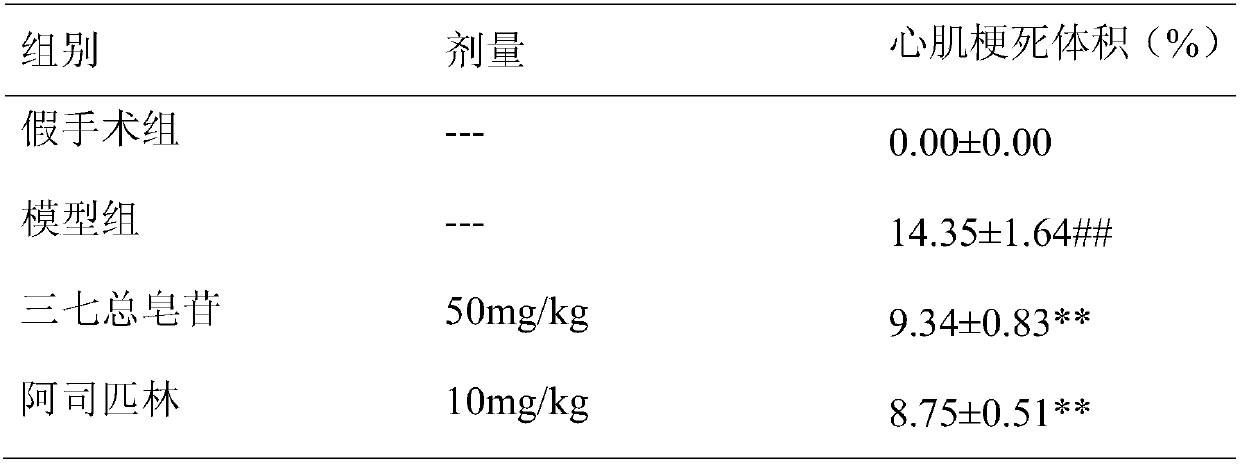
[0022] Example 1: The synergistic effect of combined use of Panax notoginseng saponins and aspirin on myocardial ischemia-reperfusion injury in rats.
[0023] 1. Test animals
[0024] Healthy male SD rats (body weight 280-300 g) were provided by Beijing Weitong Lihua Experimental Animal Company, and were raised in the Animal Center of the Institute of Medicinal Botany, Chinese Academy of Medical Sciences. Animals were fed adaptively for 1 week before testing. Before the experiment was carried out, it was reviewed and approved by the Experimental Animal Management and Animal Welfare Committee of the Institute of Medicinal Plants, Chinese Academy of Medical Sciences / Peking Union Medical College.
[0025] 2. Test method
[0026] 2.1 Animal grouping and drug treatment
[0027] 240 male SD rats were randomly divided into 8 groups, 30 in each group, respectively: sham operation group, myocardial ischemia-reperfusion injury model group, Panax notoginseng saponins (50mg / kg) group, ...
Embodiment 2
[0044] Example 2: The synergistic effect of combined use of Panax notoginseng saponins and aspirin on cerebral ischemia-reperfusion injury in rats.
[0045] 1. Test animals
[0046] Healthy male SD rats were provided by Beijing Weitong Lihua Experimental Animal Company and raised in the Animal Center of Institute of Medicinal Botany, Chinese Academy of Medical Sciences. Animals were fed adaptively for 1 week before testing. Before the experiment was carried out, it was reviewed and approved by the Experimental Animal Management and Animal Welfare Committee of the Institute of Medicinal Plants, Chinese Academy of Medical Sciences / Peking Union Medical College.
[0047] 2. Test method
[0048] 2.1 Animal grouping and drug treatment
[0049] 240 male SD rats were randomly divided into 8 groups, 30 in each group, respectively: sham operation group, middle cerebral artery embolism (MCAO / R) model group, Panax notoginseng saponins (50mg / kg) group, aspirin (10mg / kg) group, aspirin...
Embodiment 3
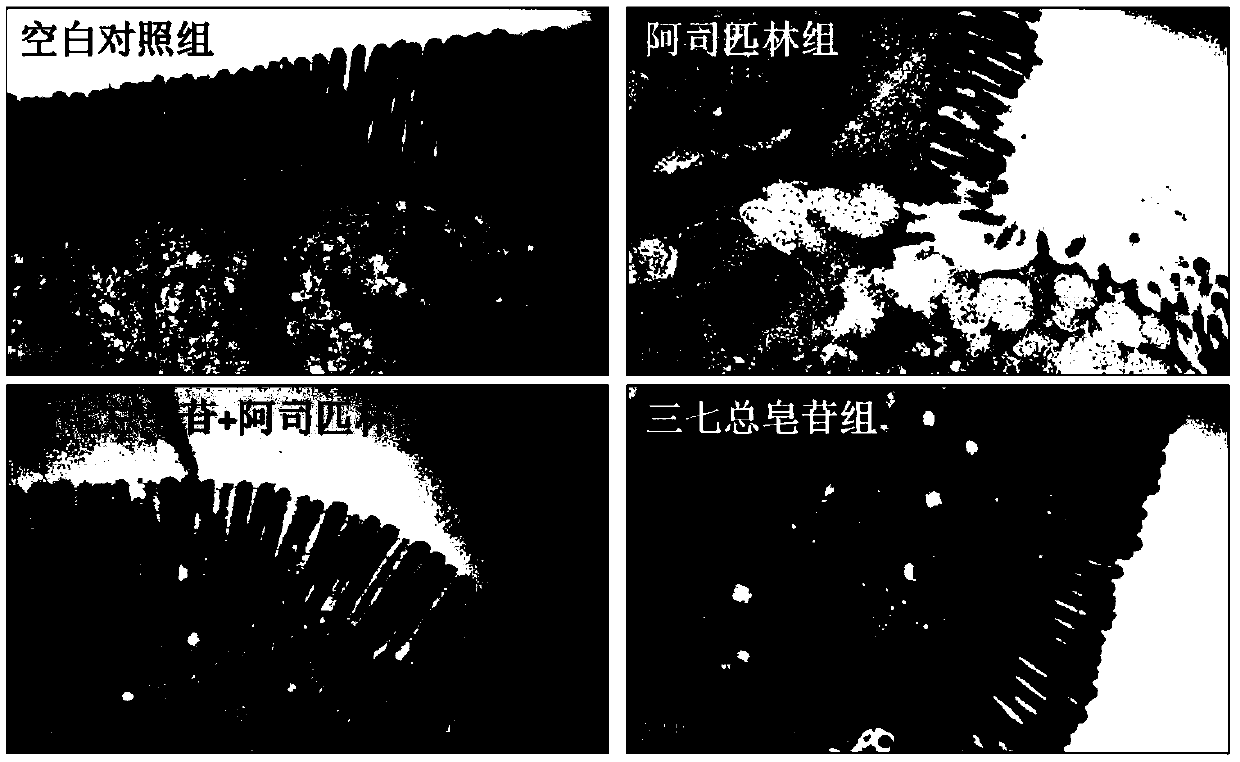
[0087]Example 3: Panax notoginseng saponins can inhibit aspirin-induced gastrointestinal mucosal damage
[0088] 1. Test animals
[0089] Healthy male SD rats (body weight 280-300 g) were provided by Beijing Weitong Lihua Experimental Animal Company, and were raised in the Animal Center of the Institute of Medicinal Botany, Chinese Academy of Medical Sciences. Animals were fed adaptively for 1 week before testing. Before the experiment was carried out, it was reviewed and approved by the Experimental Animal Management and Animal Welfare Committee of the Institute of Medicinal Plants, Chinese Academy of Medical Sciences / Peking Union Medical College.
[0090] 2. Test method
[0091] 2.1 Animal grouping and drug treatment
[0092] 40 male SD rats were randomly divided into 4 groups, 10 in each group, respectively: blank control group, aspirin (10mg / kg) group, Panax notoginseng saponins (100mg / kg)+aspirin (10mg / kg) group, Panax notoginseng total saponins (100mg / kg) group. The...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap