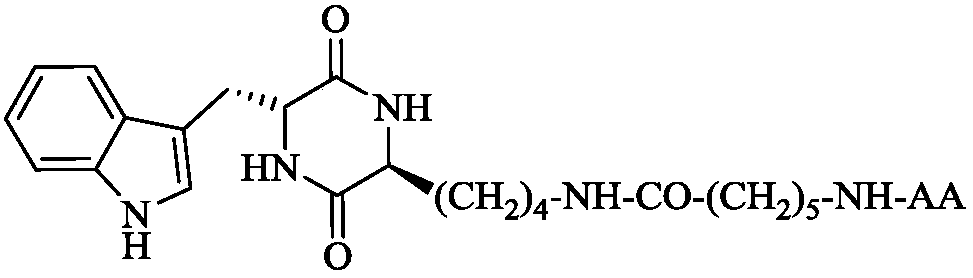
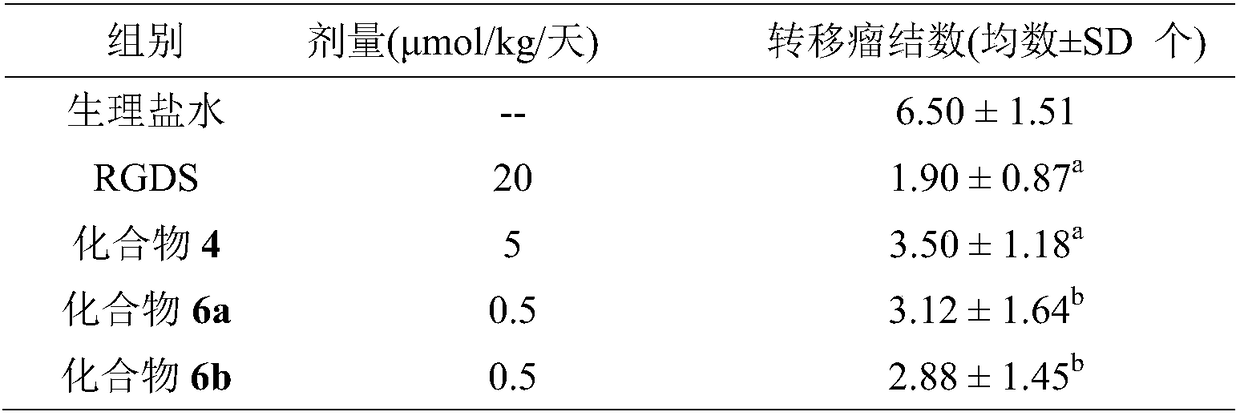
3S-indole methyl-6R-aromatic amino acid modified piperazine-2,5-dione, synthesis, activities and applications thereof
A technology of n-hexanoylamino, -d-trp-obzl, applied in the field of biomedicine, can solve the problems of no anti-tumor metastasis effect and unsatisfactory clinical curative effect of tumor chemotherapy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
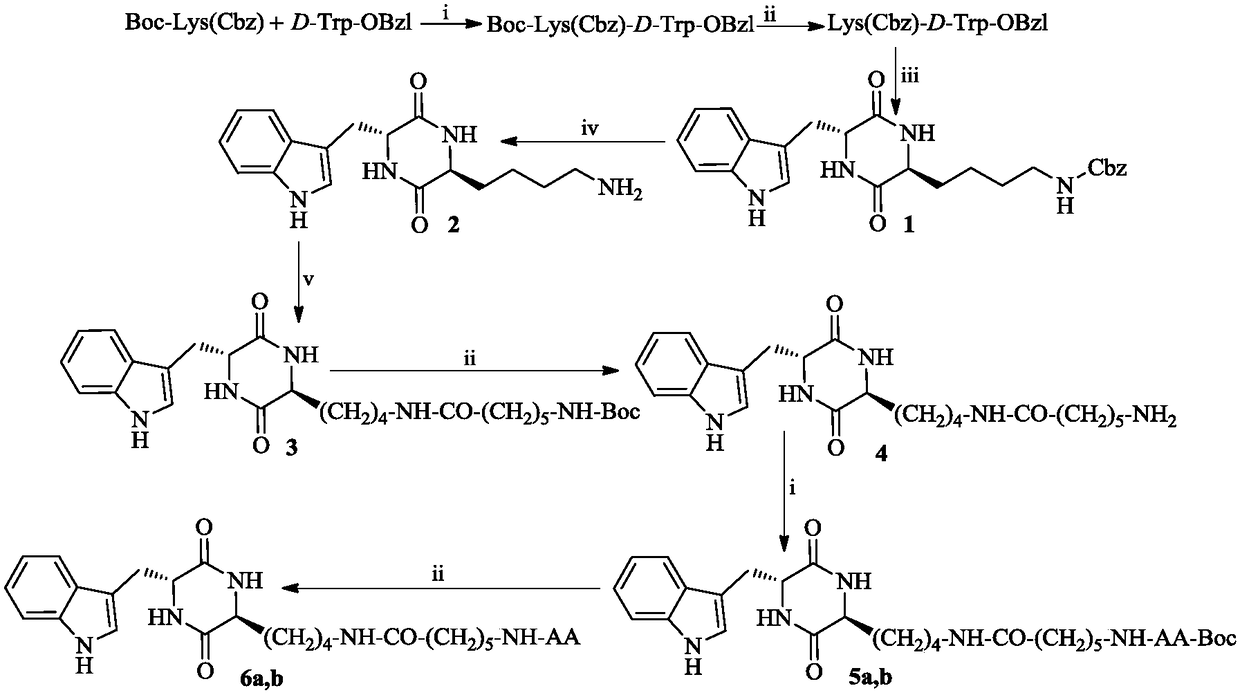
[0019] Example 1 Preparation of Boc-Lys(Cbz)-D-Trp-OBzl
[0020] Suspend 4.56g (10mmol) L-Boc-Lys (Cbz) in 100mL of anhydrous tetrahydrofuran (THF), and add 1.35g (10mmol) 1-hydroxybenzotriazole ( HOBt) and 2.47g (12mmol) of dicyclohexylcarbodiimide (DCC), then stirred for 30min. Afterwards, 3.31 g (10 mmol) of D-Trp-OBzl were added. The reaction compound was added dropwise to N-methylmorpholine (NMM) to adjust the pH to 9. The reaction mixture was first stirred under ice bath for 1 h, and then stirred at room temperature for 12 h. The reaction mixture was filtered, the filtrate was concentrated under reduced pressure, and the residue was dissolved in 150 mL of ethyl acetate solution. The obtained ethyl acetate solution was sequentially washed with 5% KHSO 4 Wash 3 times with aqueous solution and 3 times with saturated NaCl aqueous solution. Ethyl acetate layer with anhydrous Na 2 SO 4 Dry for 12h, filter, and concentrate the filtrate to dryness under reduced pressure. ...
Embodiment 2
[0021] Example 2 Preparation of Lys(Cbz)-D-Trp-OBzl
[0022] 3.28 g (5 mmol) Boc-Lys(Cbz)-D-Trp-OBzl was slowly mixed with 52 mL of hydrogen chloride in ethyl acetate under stirring in an ice bath. The resulting solution was stirred for 5 h in an ice bath. After that, the reaction mixture was concentrated under reduced pressure. The residue was dissolved in 50 mL of anhydrous ethyl acetate, and the resulting solution was concentrated under reduced pressure. This operation was repeated three times. The residue was washed well with anhydrous ether to obtain 2.36 g (85%) of the title compound as a colorless powder. ESI-MS(m / e):557[M+H] + .
Embodiment 3
[0023] Example 3 Preparation of (3S,6R)-3-(benzyloxycarbonylamino-n-butyl)-6-(indole-3-methyl)-piperazine-2,5-dione (1)
[0024] Dissolve 2.22 g (4 mmol) of L-Lys(Cbz)-D-Trp-OBzl in 100 ml of ethyl acetate, adjust the pH to 9 with triethylamine, and stir at 80° C. for 100 hours. A colorless solid was filtered off. The filtrate was concentrated under reduced pressure, and the residue was purified by silica gel column chromatography (CH 2 Cl 2 / CH 3 OH, 100 / 1). A total of 1.32 g (65%) of the title compound was obtained. ESI-MS(m / e):449[M+H] + .
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap